Open Access Article
Open Access ArticleSynthesis of 2-(α-trifluoromethylamino)indoles via stepwise cascade transformation of imidoyl sulfoxonium ylides with ortho-chloromethyl anilines
Yang Pana,
Yin Wangc,
Xiwei Zhouc,
Tengxiaoxiao Liuc,
Xiaoke Zhang *c and
Tao Jiang*b
*c and
Tao Jiang*b
aCollege of Pharmacy, Guiyang Kangyang University, Guiyang, 550081, Guizhou, P.R. China
bDepartment of Radiology, Fuling Hospital Affiliated to Chongqing University, Fuling, 408099, Chongqing, P.R. China. E-mail: laijiangtaotao@163.com
cThe Key Laboratory of Pharmaceutical Research for Tumor Prevention and Treatment of the Education Department of Guizhou Province, Zunyi, 563000, Guizhou, P.R. China. E-mail: xiaokezhang53@163.com
First published on 8th May 2026
Abstract
We have developed an efficient one-pot method for the synthesis of 2-(α-trifluoromethylamino)indole scaffolds from CF3-imidoyl sulfoxonium ylides and ortho-chloromethyl anilines. This transformation exhibits broad functional group tolerance under ambient conditions, giving yields ranging from 49–83% across 23 examples. Mechanistic investigations suggest that the reaction pathway involves an initial cyclization, which is followed by a base-mediated isomerization and rearomatization sequence. This approach offers a novel strategy for introducing α-CF3 amino groups into heterocycles and presents a valuable alternative for indole synthesis.
Introduction
The α-trifluoromethylamino group is a key structural moiety in medicinal chemistry which is widely found in a diverse range of biologically active molecules, including an agonist for LXRβ, the cathepsin K inhibitor Odanacatib, a selective inhibitor of PI3Kδ, anticancer agents, and an HCV NS4B inhibitor (Fig. 1).1 In addition, the α-trifluoromethyl amine motif could serve as a viable amide bioisostere.2 In contrast to amides, this motif features CF3CH2NH moieties, which could eliminate susceptibility to hydrolysis by amidases, esterases, and proteases.1d Moreover, the sp3-hybridized nature of α-carbon confers resistance to proteolytic cleavage, enhancing metabolic stability and reducing toxicity arising from the hydrolyzed amide.1e,f Therefore, the development of methods for the introduction of α-trifluoromethylamine is an important synthetic goal. Current strategies for accessing α-trifluoromethylamine primarily rely on the reductive or alkylative amination of trifluoromethyl ketones,3,4 reduction of trifluoromethylated enamines,5 nucleophilic addition to trifluoromethyl iminium species,6,7 or trifluoromethyl anion addition to functionalized imines.8 Furthermore, the direct α-trifluoromethylamination of unactivated alkenes and the reductive coupling of α-CF3 amino acetates represent valuable complements to current methodologies.9,10 However, these existing methods typically require multistep sequences and the use of metals. Consequently, the development of efficient and sustainable methodologies for the introduction of α-trifluoromethylamine remains highly desirable.Recently, the synthesis of indole skeletons bearing an α-trifluoroethylamine moiety has garnered significant attention in medicinal chemistry, driven by the privileged status of indoles in pharmaceutical agents and the unique properties of trifluoroethylamino groups.2,11 Although a diverse range of methodologies have been established for the synthesis of 3-substituted indoles,12 the construction of 2-substituted indoles remains significantly less explored due to the inherently lower reactivity of the C2 position (Scheme 1a). Furman and co-workers developed a method to access 2-(α-trifluoromethylamino)indoles involving the reduction of fluoroacetamides with Schwartz's reagent, followed by a one-pot addition to in situ generated imines (Scheme 1b).12b Additionally, 2-substituted indoles incorporating a trifluoromethyl moiety were obtained via a chiral phosphoric acid-catalyzed Friedel–Crafts alkylation of 4,7-dihydroindoles with trifluoromethyl ketimines, followed by oxidation with DDQ (Scheme 1c).13 Furthermore, Zard's group established an intermolecular radical addition reaction between 3-substituted indoles and xanthates initiated by dilauroyl peroxide (DLP). This methodology provides an effective approach for the direct incorporation of the trifluoroethylamino moiety into such heterocyclic frameworks (Scheme 1d).14 Despite these advances, there is still a strong demand for an efficient and eco-friendly methods to construct 2-(α-trifluoromethylamino)indole.
CF3-Imidoyl sulfoxonium ylides (TFISYs) are versatile nitrogen-containing trifluoromethylated dipoles that have proven to be valuable building blocks in cascade annulation chemistry.15 They react efficiently with various synthons to afford diverse trifluoromethyl-functionalized heterocycles, ranging from simple monocyclic to complex polycyclic systems.16 In particular, cascade annulations involving TFISYs serve as a powerful strategy for the synthesis of trifluoromethylated indole derivatives. Liu's group reported a visible-light-promoted cascade annulation of TFISYs with azides for the construction of 2-trifluoromethyl indoles through thermal Curtius rearrangement of azides, intermolecular nucleophilic addition, and photoinduced intramolecular cyclization followed by a 1,3-hydrogen shift.17 In addition, Chen and co-workers established an Rh(III)-catalyzed chemodivergent approach to provide CF3-substituted indoles via the annulation of 2-arylpyridines with TFISYs, involving triple C–H activation.18 Despite these progress, annulation strategies employing TFISYs for the construction of 2-(α-trifluoromethylamino)indoles are still underexplored.
The chemistry of aza-ortho-quinone methides (aza-o-QMs), which are generated from N-(ortho-chloromethyl)aryl amides under basic conditions, has garnered significant attention owing to their remarkable reactivity.19 In recent years, substantial progress has been made in the development of cycloaddition reactions for the synthesis of diverse heterocyclic skeletons.20 Notably, the cascade annulation of aza-o-QMs has emerged as a powerful strategy to access indole derivatives. For instance, Xiao and co-workers reported an efficient cascade reaction between sulfur ylides and N-(ortho-chloromethyl)aryl amides that furnished 2-acyl indoles via an E1cb elimination pathway (Scheme 2a).21 In 2014, 2-aryl indoles were synthesized through the trapping of a transient aza-ortho-quinone methide intermediate by an NHC-generated acyl anion equivalent (Scheme 2b).22 More recently, a tandem phospha-Michael addition/N-acylation/intramolecular Wittig reaction of aza-o-QMs was developed as an effective route for the construction of indole frameworks (Scheme 2c).23 Despite these advances, the application of aza-o-QM cycloaddition to the synthesis of 2-(α-trifluoromethylamino)indoles remains unexplored. Furthermore, current methodologies for aza-o-QM-based indole synthesis are predominantly limited to pathways involving cycloaddition followed by E1cb elimination or dehydrative isomerization. Herein, we described a stepwise cascade transformation of imidoyl sulfoxonium ylides with ortho-chloromethyl anilines, providing an efficient method to 2-(α-trifluoromethylamino)indoles. Notably, the reaction proceeds without the formation of E1cb elimination byproducts.
Results and discussion
To initiate the study, (E)-3-(dimethyl(oxo)-l6-sulfaneylidene)-1,1,1-trifluoro-N-phenylpropan-2-imine 1a and N-(2-(chloromethyl)phenyl)methanesulfonamide 2a were selected as model substrates and treated with a base in dichloromethane at 40 °C under air. To our delight, our hypothesis was validated, giving 2-(α-trifluoromethylamino)indole 3a in 57% yield (Table 1, entry 1). The structure was established by NMR analysis and confirmed by X-ray crystallographic analysis of 3f. Replacing the base from 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) with 1,5,7-triazabicyclo[4.4.0]dec-5-ene (TBD), 1,5-diazabicyclo[4.3.0]non-5-ene (DBN), 1,1,3,3-Tetramethylguanidine (TMG), or 1,4-diazabicyclo[2.2.2]octane (DABCO) resulted in decreased yields of 3a (Table 1, entries 2–5). Notably, inorganic bases proved ineffective for this transformation (Table 1, entries 6–8). Additionally, the reaction failed to afford product 3a in the absence of DBU (entry 9). A screening of various solvents (Table 1, entries 10–16) identified toluene as the most effective solvent for the generation of 3a. Variation of the reaction temperature (Table 1, entries 17–18) did not lead to any improvement in the yield of 3a. Further investigation disclosed neither prolonging nor shortening the reaction time increased the yield of desired product (Table 1, entries 19–20). In addition, further optimization of the base equivalents did not result in a higher yield (Table 1, entries 21–22).| Entry | Base | Solvent | Yield (%)b |
|---|---|---|---|
| a Reaction conditions: 1a (0.2 mmol), 2a (0.1 mmol), base (0.15 mol), solvent (0.05 M), 40 °C, 8 h.b Yield of isolated product.c Reaction conducted at 25 °C.d Reaction conducted at 100 °C.e Reaction time was 4 h.f Reaction time was prolonged to 10 h.g Using Na2CO3 (0.1 mmol) as a base.h Using Na2CO3 (0.3 mmol) as a base. | |||
| 1 | DBU | CH2Cl2 | 57 |
| 2 | TBD | CH2Cl2 | 31 |
| 3 | DBN | CH2Cl2 | 36 |
| 4 | TMG | CH2Cl2 | 14 |
| 5 | DABCO | CH2Cl2 | Trace |
| 6 | Na2CO3 | CH2Cl2 | Trace |
| 7 | Cs2CO3 | CH2Cl2 | Trace |
| 8 | KOH | CH2Cl2 | Trace |
| 9 | — | CH2Cl2 | Trace |
| 10 | DBU | DCE | 22 |
| 11 | DBU | THF | 42 |
| 12 | DBU | CH3CN | 30 |
| 13 | DBU | Toluene | 83 |
| 14 | DBU | CH3CO2C2H5 | 71 |
| 15 | DBU | Acetone | 38 |
| 16 | DBU | CHCl3 | 49 |
| 17c | DBU | Toluene | 36 |
| 18d | DBU | Toluene | 63 |
| 19e | DBU | Toluene | 60 |
| 20f | DBU | Toluene | 77 |
| 21g | DBU | Toluene | 52 |
| 22h | DBU | Toluene | 79 |
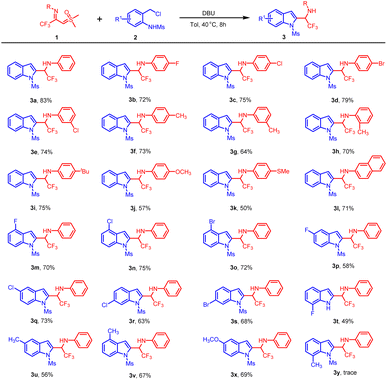
With the optimal conditions established (Table 1, entry 13), we investigated the substrate scope of CF3-imidoyl sulfoxonium ylides 1. Our investigation commenced with an evaluation of substituents on the phenyl moiety. As outlined in Table 2, substrates bearing electron-withdrawing groups (e.g., F, Cl, Br) at the para-position of the benzene ring were well tolerated, delivering the expected products 3b–3d in good yields (72–79%). Similarly, the meta-chlorophenyl substituted ylide proved to be a suitable substrate, providing 3e in a moderate yield. Furthermore, the reaction scope was successfully extended to substrates with electron-donating groups at the para-, meta-, and ortho-positions, furnishing the corresponding products 3f–3h in yields ranging from 64–73%. Intriguingly, the sterically hindered 4-tert-butyl-substituted ylide was also amenable to this transformation under standard conditions, delivering 3i in 75%. Additionally, TFISYs featuring ether and thioether substituents at the 4-position of the arene moiety were well tolerated, providing the expected products 3j and 3k in acceptable yields. Notably, the sulfoxonium ylide bearing a naphthyl group was smoothly converted into the corresponding product 3l by reacting with N-(ortho-chloromethyl)aryl amide 2. Encouraged by these results, we proceeded to investigate the substrate scope of the reaction using N-(ortho-chloromethyl)aryl amides 2 bearing various R1 groups. A variety of substrates were efficiently transformed into 2-(α-trifluoromethylamino)indoles 3 in good to excellent yields. The protocol was successfully applied to N-(ortho-chloromethyl)aryl amides 2 substituted with electron-withdrawing groups at the 6-position, affording products 3m–3o in moderate yields. N-ortho-chloromethyl anilines 2 containing halogen substituents (e.g., F, Cl) at the 5-position underwent the cyclization/isomerization/aromatization cascade to afford 3p and 3q. Furthermore, 6-substituted 2-(α-trifluoromethylamino)indoles 3r and 3s were successfully obtained utilizing the corresponding starting materials. Notably, the employment of N-(2-(chloromethyl)-6-fluorophenyl)meth-anesulfonamide 2 led to the formation of the deprotected product 3t in 49% yield. Comparative analysis of 3t with other derivatives suggested that the position of the substituent on the N-ortho-chloromethyl amides 2 significantly influences the efficiency of this cascade process. Furthermore, aza-ortho-quinone methide precursors 2 bearing electron-donating groups were well tolerated under the optimized conditions, delivering the corresponding products 3u–3x in moderate yields. However, N-(ortho-chloromethyl)aryl amides 2 with a 3-methyl substituent failed to provide the desired product 3y under the standard conditions.
To further investigate the effect of the N-substituent, the methanesulfonyl (Ms) group was replaced with benzoyl (Bz), benzyloxycarbonyl (Cbz), p-toluenesulfonyl (Ts), tert-butoxycarbonyl (Boc), H, methyl (Me), or acetyl groups (Ac). Unfortunately, the expected 2-(α-trifluoromethylamino)indoles 4 were not detected under the optimized conditions (Scheme 3a). The scalability of this transformation was further verified on a gram scale. Under standard conditions, the reaction of 1a and 2a in the presence of DBU proceeded smoothly to afford 3a (1.16 g) in 79% yield (Scheme 3b). Furthermore, the control experiments were performed. Trifluoroacetimidoyl-substituted indoline 5 was obtained utilizing 4-fluorophenyl substituted 1b and 2a as substrate in the absence of DBU (Scheme 3c). Subsequently, treatment of 5 with DBU promoted the isomerization and aromatization cascade, affording 2-(α-trifluoromethylamino)indole 3b (Scheme 3d). To further elucidate the reaction mechanism, the reaction of 1b and 2a was conducted under standard conditions in the absence of DBU, and no target product 3b was observed. However, upon the addition of DBU, 3b was obtained in 76% isolated yield after 8 hours.
Based on the results of control experiments and previous work,16b,24 a plausible mechanism for the synthesis of 2-(α-trifluoromethylamino)indole was proposed. As depicted in Scheme 4, the zwitterionic intermediate Int I, derived from imidoyl sulfoxonium ylide 1, reacts with the ambiphilic reagent 2a to furnish the intermediate Int II. Subsequently, trifluoroacetimidoyl-substituted indoline Int III is formed via an SN2 ring closure of Int II. This is followed by a DBU-mediated imine-enamine tautomerization of Int III to give Int IV, which finally undergoes base-promoted aromatization to afford the expected product 3.
Conclusions
In summary, we have discovered a DBU-promoted one-pot protocol to offer the 2-(α-trifluoromethylamino)indole core using CF3-imidoyl sulfoxonium ylides and N-(ortho-chloromethyl)aryl amides as substances. This approach offers a novel strategy for installing α-CF3 amino moieties into heterocycles under mild conditions, as well as a new route for indole construction. Mechanistic studies suggests that this reaction might proceed via a stepwise cascade to afford the corresponding indole analogs.Author contributions
Conceptualization, Tao Jiang and Xiaoke Zhang; methodology, Yang Pan; investigation, Yang Pan, Yin Wang, Xiwei Zhou, Tengxiaoxiao Liu, and Xiaoke Zhang; writing—original draft preparation, Yang Pan and Xiaoke Zhang; writing—review and editing, Tao Jiang; supervision, Tao Jiang, Xiaoke Zhang; project administration, Tao Jiang.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2504930 contains the supplementary crystallographic data for this paper.25The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental procedures, characterization data, and copies of the 1H NMR, 13C NMR and HRMS spectra of all compounds are included. See DOI: https://doi.org/10.1039/d6ra01734g.
Acknowledgements
We are grateful for financial support from The Scientific Research Foundation of Guiyang Kangyang University, grant number (GKUK2025-003), Guizhou Province Science and Technology plan program of China, grant number (QN[2025] 446), the Guizhou Province Science and Technology plan program of China, grant number (QKHJC-ZK[2023]-495), the joint Fund of Zunyi Municipal Bureau of Industry and Techonology with Zunyi Medical University, grant number (ZSKH-HZ-2024-323, QKPTRC-[2021]1350-058), Dr Jiang Tao's Research Startup Fund, Chongqing Mid-career and Young Medical High end Talent Project, grant number (YXGD202401), and Chongqing Municipal Clinical Key Specialty Construction Project, and Project of the Science and Technology Bureau of Fuling District, Chongqing, grant number (FLKJ, 2024AAN3060), Scientific and Technological Research Program of Chongqing Municipal Education Commission, grant number (KJQN202500136). Provincial-level Undergraduate Innovation and Entrepreneurship Training Program Project, grant number (ZYDC202402210).Notes and references
- For selected examples: (a) C. S. Li, D. Deschenes, S. Desmarais, J. P. Falgueyret, J. Y. Gauthier, D. B. Kimmel, S. Léger, F. Massé, M. E. McGrath, D. J. McKay, M. D. Percival, D. Riendeau, S. B. Rodan, M. Thérien, V. L. Truong, G. Wesolowski, R. Zamboni and W. C. Black, Bioorg. Med. Chem. Lett., 2006, 16, 1985–1989 CrossRef CAS PubMed; (b) N. Zhang, S. Ayral-Kaloustian, T. Nguyen, J. Afragola, R. Hernandez, J. Lucas, J. Gibbons and C. Beyer, J. Med. Chem., 2007, 50, 319–327 CrossRef CAS PubMed; (c) J. Y. Gauthier, N. Chauret, W. Cromlish, S. Desmarais, L. T. Duong, J. P. Falgueyret, D. B. Kimmel, S. Lamontagne, S. Léger, T. LeRiche, C. S. Li, F. Massé, D. J. McKay, D. A. Nicoll-Griffith, R. M. Oballa, J. T. Palmer, M. D. Percival, D. Riendeau, J. Robichaud, G. A. Rodan, S. B. Rodan, C. Seto, M. Thérien, V. L. Truong, M. C. Venuti, G. Wesolowski, R. N. Young, R. Zamboni and W. C. Black, Bioorg. Med. Chem., 2008, 18, 923–928 CrossRef CAS PubMed; (d) N. A. Meanwell, J. Med. Chem., 2018, 61, 5822–5880 CrossRef CAS PubMed; (e) M. Zanda, New J. Chem., 2004, 28, 1401–1411 RSC; (f) M. Sani, A. Volonterio and M. Zanda, ChemMedChem, 2007, 2, 1693–1700 CrossRef CAS PubMed.
- C. I. Onyeagusi and S. J. Malcolmson, ACS Catal., 2020, 10, 12507–12536 CrossRef CAS PubMed.
- M. W. Chen, Q. Yang, Z. Deng, Y. Zhou, Q. Ding and Y. Peng, J. Org. Chem., 2018, 83, 8688–8694 CrossRef CAS PubMed.
- F. Gosselin, P. D. O'Shea, S. Roy, R. A. Reamer, C. Y. Chen and R. P., Org. Lett., 2005, 7, 355–358 CrossRef CAS PubMed.
- M. W. Chen, Y. Duan, Q. A. Chen, D. S. Wang, C. B. Yu and Y. G. Zhou, Org. Lett., 2010, 12, 5075–5077 CrossRef CAS PubMed.
- H. Wang, T. Jiang and M. H. Xu, J. Am. Chem. Soc., 2013, 135, 971–974 CrossRef CAS PubMed.
- J. Zhu, L. Huang, W. Dong, N. Li, X. Yu, W. P. Deng and W. Tang, Angew. Chem., Int. Ed., 2019, 58, 16119–16123 CrossRef CAS PubMed.
- H. Kawai, A. Kusuda, S. Nakamura, M. Shiro and N. Shibata, Angew. Chem., Int. Ed., 2009, 48, 6324–6327 CrossRef CAS PubMed.
- S. L. Li, T. H. Chen, Y. H. Xi, X. J. Li and W. C. Huang, ACS Catal., 2025, 15, 11944–11952 CrossRef CAS.
- X. Chen, Y. Yang, S. L. Li, T. H. Chen, H. G. Gong, X. T. Wang and W. W. Huang, ACS Catal., 2025, 15, 17558–17567 CrossRef CAS.
- For selected examples: (a) N. Cao and C. H. Wang, Chin J Nat Med., 2021, 19, 591–607 CAS; (b) A. Dhiman, R. Sharma and R. K. Singh, Acta Pharm. Sin. B, 2022, 12, 3006–3027 CrossRef CAS PubMed.
- For selected examples: (a) L. M. Wu, C. Xie, J. Zhou, H. B. Mei, V. A. Soloshonok, J. L. Han and Y. Pan, J. Fluorine Chem., 2015, 170, 57–65 CrossRef CAS; (b) P. J. Czerwiński and B. Furman, Chem. Commun., 2019, 55, 9436–9439 RSC; (c) G. K. Zhao, S. S. Samanta, J. Michieletto and S. P. Roche, Org. Lett., 2020, 22, 5822–5827 CrossRef CAS PubMed; (d) P. J. Czerwiński, J. J. Najczuk and B. Furman, ACS Sustainable Chem. Eng., 2022, 10, 10486–10492 CrossRef; (e) R. Yonesaki, Y. Kondo, W. Akkad, M. Sawa, K. Morisaki, H. Morimoto and T. Ohshima, Chem.–Eur. J., 2018, 24, 15211–15214 CrossRef CAS PubMed.
- T. Uchikura, R. Suzuki, Y. Suda and T. Akiyama, ChemCatChem, 2020, 12, 4784–4787 CrossRef CAS.
- M. Braun, G. Castanedo, L. Qin, P. Salvo and S. Z. Zard, Org. Lett., 2017, 19, 4090–4093 CrossRef CAS PubMed.
- X. P. Chen, Q. L. Wang, X. H. Hou, C. C. Wang, Z. W. Ma and Y. J. Zhang, Chin. J. Org. Chem., 2025, 45, 2796–2814 CrossRef CAS.
- For selected examples: (a) S. Wen, Q. Tian, Y. Chen, Y. Zhang and G. Cheng, Org. Lett., 2021, 23, 7407–7411 CrossRef CAS PubMed; (b) D. Yang, Y. Wang, T. Wang, Q. Dou, K. Zhou, H. Zhai and B. Cheng, Adv. Synth. Catal., 2023, 365, 88–95 CrossRef CAS; (c) Y. Sun, S. Ling, Y. Duan, J. Li, Z. Chen and X. F. Wu, Adv. Synth. Catal., 2023, 365, 1521–1525 CrossRef CAS; (d) Z. Yang, J. Tang, Z. Chen and X. F. Wu, Org. Lett., 2022, 24, 7288–7293 CrossRef CAS PubMed; (e) J. Liao, R. Zhai, X. Gao, H. Wu, D. Kong, S. Wang and X. Chen, Adv. Synth. Catal., 2025, 367, e202401220 CrossRef CAS; (f) Q. Zhou, S. Yan, C. Ma, Y. J. Gong, X. Zhang and X. Fan, Adv. Synth. Catal., 2024, 366, 5190–5196 CrossRef CAS.
- J. H. Cui, Q. Y. Chen, J. Zhang, Y. He, X. Li and P. N. Liu, Chem. Commun., 2025, 61, 488–491 RSC.
- X. Gao, R. Zhai, J. Liao, G. Yao, H. Meng, Y. Luo, D. Kong, S. Wang and X. Chen, Org. Lett., 2025, 27, 657–662 CrossRef CAS PubMed.
- X. Y. He, Y. H. Ma, Q. Q. Yang and W. J. Xiao, Synthesis, 2022, 54, 953–964 CrossRef.
- For selected examples: (a) X. K. Zhang, Y. Pan, P. Liang, L. Pang, X. F. Ma, W. Jiao and H. W. Shao, Adv. Synth. Catal., 2019, 361, 5552–5557 CrossRef CAS; (b) X. K. Zhang, Y. Pan, P. Liang, L. Pang, X. F. Ma, W. Jiao and H. W. Shao, Adv. Synth. Catal., 2018, 360, 3015–3019 CrossRef CAS; (c) X. K. Zhang, H. B. Wang, Z. W. Li, Y. Shu, S. Gan, X. F. Zhang, H. W. Shao and C. Y. Wang, ACS Omega, 2022, 7, 40963–40972 CrossRef CAS PubMed; (d) X. K. Zhang, L. He, X. Zhang, M. Lin, X. J. Huang, L. J. Meng, H. W. Shao, W. Jiao and Q. L. Xing, Tetrahedron, 2025, 180, 134665 CrossRef CAS; (e) L. Chen, G. M. Yang, J. Wang, Q. F. Jia, J. Wei and Z. Y. Du, RSC Adv., 2015, 5, 76696 RSC; (f) L. Lei, Y. Y. Yao, L. J. Jiang, X. Q. Lu, C. Liang and D. L. Mo, J. Org. Chem., 2020, 85, 3059–3070 CrossRef CAS PubMed; (g) L. Pang, S. J. Fang, P. S. Zou, W. Wang, J. C. Su, X. Q. Liu, C. X. Pan, D. L. Mo and G. F. Su, Org. Chem. Front., 2023, 10, 1780–1787 RSC.
- Q. Q. Yang, C. Xiao, L. Q. Lu, J. An, F. Tan, B. J. Li and W. J. Xiao, Angew. Chem., Int. Ed., 2012, 51, 9137–9140 CrossRef CAS PubMed.
- M. T. Hovey, C. T. Check, A. F. Sipher and K. A. Scheidt, Angew. Chem., Int. Ed., 2014, 53, 9603–9607 CrossRef CAS PubMed.
- T. B. Hua, F. Chao, L. Wang, C. Y. Yan, C. Xiao, Q. Q. Yang and W. J. Xiao, Adv. Synth. Catal., 2020, 362, 2615–2679 CrossRef CAS.
- (a) Y. Sun, Z. G. Yang, S. N. Lu, Z. K. Chen and X. F. Wu, Org. Lett., 2022, 24(37), 6822–6827 CrossRef CAS PubMed; (b) H. Huang, Y. Yang, X. Y. Zhang, Z. L. Zeng and Y. Liang, Tetrahedron Lett., 2013, 54, 6049–6052 CrossRef CAS.
- CCDC 2504930: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q2l6m.
| This journal is © The Royal Society of Chemistry 2026 |