DOI:
10.1039/D6RA01666A
(Paper)
RSC Adv., 2026,
16, 17676-17689
Tuning the electrical and optical performance of PVA/PANI films via Fe2O3 nanorods for advanced energy storage and optoelectronic devices
Received
25th February 2026
, Accepted 27th March 2026
First published on 1st April 2026
Abstract
PVA/PANI-Fe2O3 nanocomposite films were successfully fabricated via a solution casting method with Fe2O3 nanorod loadings ranging from 1.0 to 4.5 wt% to tailor their structural, optical, and electrical properties for optoelectronic applications. XRD analysis revealed a progressive reduction in crystallite size (from ∼4.2 to ∼3.0 nm) and enhanced amorphous character, attributed to the disruption of the native hydrogen-bonding network in the polymer matrix by Fe2O3 nanorods. FTIR results confirmed strong interfacial interactions through hydrogen bonding between Fe2O3 and PVA/PANI chains. Optical measurements demonstrated a red shift in absorption edges and a significant narrowing of both direct and indirect band gaps, accompanied by an increase in Urbach energy, indicating the formation of localized states and increased structural disorder. These changes are associated with the modified electronic structure induced by Fe2O3 incorporation. Furthermore, dielectric analysis revealed multiple conduction regimes, with electrical conductivity improving by nearly two orders of magnitude due to the formation of interconnected charge transport pathways facilitated by the nanorods. Overall, the incorporation of Fe2O3 nanorods effectively tunes the physicochemical properties of PVA/PANI films, making them promising candidates for flexible optoelectronic and energy-related applications.
1 ntroduction
Polymer nanocomposites have become essential in modern technological development and play a significant role in various aspects of daily life.1–3 Recent research primarily focuses on designing innovative polymer nanocomposites with improved physical and functional properties.4,5 These polymers are highly attractive for both scientific and industrial applications due to their affordability, biodegradability, ease of fabrication, and favorable optoelectronic characteristics.6–9 Poly(vinyl alcohol) is a bio-compatible and thermally stable material,10 that has been widely employed as a base material for diverse nanofiller additives due to its advantageous properties, including water solubility, non-corrosive behavior, excellent film-forming ability, and high optical transparency. In our previous studies, a range of nanoparticles, including CoFe2O4.11 Bi2O3/MWCNT hybrid nanofillers,12 Al2O3/V2O5 nanoparticles,13 and niobium oxide and praseodymium oxide nanoparticles14 have been incorporated into PVA matrices to tailor and enhance their optical properties for potential industrial applications. In addition, polyaniline (PANI), a conducting polymer with semiconducting or metallic behavior,15 has attracted attention due to its good electrical conductivity, electrochemical performance, stability, accessibility, and low-cost synthesis. Laourari et al. synthesized NiCu-PANI/PVA quaternary nanocomposite films and demonstrated their strong antibacterial performance and Cu-enhanced antifungal activity against selected pathogens.16 Li et al.17 synthesized a flexible APH-PANi hydrogel sample with vertically aligned channels that serves as an integrated, self-healable supercapacitor exhibiting high capacitance, excellent mechanical flexibility, and stable electrochemical performance. Abdelhamied et al.18 prepared PVA/PANI/Ag nanocomposite films and showed that oxygen-ion irradiation significantly modifies their structural, linear, and nonlinear optical properties, with the highest fluence producing films most suitable for optoelectronic applications. Arenas et al.19 synthesized PVA-PANI in situ nanocomposite films using surfactant and acid dopants, demonstrating low percolation thresholds and enhanced electrical conductivity suitable for antistatic electronic applications. Alsulami and Rajeh20 fabricated PANI/PMMA-TiO2 nanocomposite films and demonstrated that increasing TiO2 doping enhances thermal stability and significantly modifies the optical properties, making the films promising for optoelectronic applications.
Magnetic nanoparticles have attracted considerable attention as nanofillers due to their distinctive properties, including enhanced performance relative to bulk materials and the ability to be precisely controlled by external magnetic field.21 Among magnetic iron nanoparticles, Fe2O3 (hematite) is particularly appealing because it combines exceptional corrosion resistance, cost-effective synthesis, bio-compatibility, and eco-friendly due to its non-toxic nature.22 Furthermore, hematite nanoparticles exhibit a high surface-to-volume ratio, which results in elevated surface energy and enhanced reactivity.23 By incorporating these nanoparticles into polymer matrices, their unique characteristics can be exploited to improve the versatility and physicochemical properties of the resulting nanocomposite materials.24–26 Badawi et al.27 prepared PVA/graphene nanocomposite films loaded with Fe2O3 nanoparticles and demonstrated that varying the iron oxide content significantly tailors their structural and optical parameters, making them suitable for diverse optical and storage applications. Ragab et al.28 fabricated PEO@NaAlg sample reinforced with Fe2O3 nanorods and showed that increasing nanorod content significantly modifies their optical, magnetic, and electrical properties, enhancing their suitability for magneto-optical and energy-storage applications. El Sayed and Morsi29 fabricated α-Fe2O3-reinforced (PVA + PEG) nanocomposite films and demonstrated that increasing hematite content significantly tunes their optical and dielectric properties through enhanced refractive index, reduced band gap, and improved AC conductivity.
To address this gap, the present study focuses on the incorporation of α-Fe2O3 nanorods into PVA/PANI hybrid nanocomposites and investigates how their unique rod-like morphology influences the structural, optical, and electrical properties. Despite extensive studies on PVA/PANI composites with metal oxides, limited attention has been given to the role of nanorod morphology in simultaneously tuning the optical band gap, Urbach energy, and AC/DC conductivity. In this work, α-Fe2O3 nanorods with high aspect ratio were incorporated over a controlled loading range (1–4.5 wt%), allowing a systematic correlation between nanorod dispersion, structure, and multifunctional properties. The main findings demonstrate that Fe2O3 nanorods enhance the amorphous nature of the polymer matrix, reduce the optical band gap, increase Urbach energy, and significantly improve electrical conductivity through efficient charge transport pathways. These results highlight the novelty of this work and underscore the strong potential of PVA/PANI-Fe2O3 nanocomposites as tunable multifunctional materials for advanced optoelectronic and energy-related applications.
2 Methodology
2.1 Chemicals
Polyvinyl alcohol (PVA) with an average molecular weight of about 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1 was purchased from Merck (Germany). Polyaniline (PANI), having a molecular weight exceeding 19
000 g mol−1 was purchased from Merck (Germany). Polyaniline (PANI), having a molecular weight exceeding 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1, was sourced from Sigma-Aldrich. Ferric chloride (FeCl3, ≥98% purity, Sigma-Aldrich), deionized water, and aqueous ammonia solution (NH4OH, 25–28%, Sigma-Aldrich) were used in the preparation of the Fe2O3 nanorods.
000 g mol−1, was sourced from Sigma-Aldrich. Ferric chloride (FeCl3, ≥98% purity, Sigma-Aldrich), deionized water, and aqueous ammonia solution (NH4OH, 25–28%, Sigma-Aldrich) were used in the preparation of the Fe2O3 nanorods.
2.2 Synthesis of Fe2O3 nanorods
Fe2O3 nanorods were synthesized using a co-precipitation approach. A 2 M FeCl3 solution was prepared in 150 mL of deionized water and heated to 90 °C. Ammonia was gradually introduced under continuous stirring for 3 h to induce precipitation. The resulting product was centrifuged at 8000 rpm, and the obtained precipitate was repeatedly washed with deionized water. The Fe2O3 nanorods were then dried at 90 °C for 48 h and subsequently calcined at 500 °C for 3 h.
2.3 Preparation of PVA/PANI-Fe2O3 nanocomposites
PVA/PANI blend films with an 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 ratio was fabricated using the solution casting technique. First, 1.6 g of PVA was dissolved in 100 mL of deionized water at 80 °C under continuous stirring until a clear homogeneous solution was formed. Separately, 0.4 g of PANI was dispersed in 40 mL of deionized water at room temperature. The two solutions were then combined, as the 80
20 ratio was fabricated using the solution casting technique. First, 1.6 g of PVA was dissolved in 100 mL of deionized water at 80 °C under continuous stirring until a clear homogeneous solution was formed. Separately, 0.4 g of PANI was dispersed in 40 mL of deionized water at room temperature. The two solutions were then combined, as the 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 PVA/PANI proportion was found to yield highly uniform and flexible films. The blended mixture was stirred for an additional hour to ensure complete homogenization. Fe2O3 nanorods were incorporated into the polymer blend at various loadings (1.0, 2.5, 3.5, and 4.5 wt%). Each amount of nanofiller was incorporated gradually to the solution under constant stirring for 1 hour, followed by ultrasonic treatment for 20 min at room temperature to minimize particle agglomeration. The required nanorod content M (1.0–4.5 wt%) was computed by eqn (1):
20 PVA/PANI proportion was found to yield highly uniform and flexible films. The blended mixture was stirred for an additional hour to ensure complete homogenization. Fe2O3 nanorods were incorporated into the polymer blend at various loadings (1.0, 2.5, 3.5, and 4.5 wt%). Each amount of nanofiller was incorporated gradually to the solution under constant stirring for 1 hour, followed by ultrasonic treatment for 20 min at room temperature to minimize particle agglomeration. The required nanorod content M (1.0–4.5 wt%) was computed by eqn (1):| |
 | (1) |
Finally, the prepared mixtures were cast into Petri dishes and allowed to dry at 55 °C for four days. Uniform films with an approximate thickness of 0.08 mm were obtained by gently peeling them from the dishes. A schematic illustration of the synthesis procedure is presented in Scheme 1, which outlines the formation process of the PVA/PANI-Fe2O3 nanocomposites films.
 |
| | Scheme 1 A schematic illustration of the synthesis procedure of the PVA/PANI-Fe2O3 nanocomposites films. | |
2.4 Device characterizations
The structural, optical, and electrical properties of the PVA/PANI-Fe2O3 nanocomposites were characterized using a range of analytical techniques. X-ray diffraction (XRD) patterns were recorded using a PANalytical X'Pert Pro diffractometer with Cu Kα radiation (λ = 0.15406 nm), operating at 40 kV and 30 mA, over a 2θ range of 3–70°. FTIR spectra were recorded using a Bruker Vertex 80 spectrometer in the range of 400–4000 cm−1. Optical absorption spectra were obtained using a Shimadzu UV-3600 spectrophotometer operating between 200 and 1000 nm under ambient conditions. Dielectric properties were measured by broadband dielectric spectroscopy using a Novocontrol Alpha-A Analyzer within the frequency range of 0.1–10 MHz.
3 Results and discussion
3.1 XRD analysis
Fig. 1 presents the XRD patterns of the pure PVA/PANI blend alongside those of the nanocomposite films containing various loadings of Fe2O3 nanorods (NRs). The broad diffraction halo centered at 19.73°, corresponding to the (101) plane, is typical of semi-crystalline polymer blend. Upon incorporating Fe2O3 NRs into the PVA/PANI polymer blend, this halo becomes progressively wider and less intense, indicating a reduction in structural order as the nanorod content increases. Additionally, diffraction peaks appearing at 33.76° and 36.10°, assigned to rhombohedral Fe2O3 (JCPDS 01-076-8394), are detected and become increasingly distinct at higher nanorod concentrations.30 This behavior signifies that introducing Fe2O3 NRs enhances the amorphous character of the PVA/PANI blend.31 The more noticeable Fe2O3 reflections at elevated filler content, along with their absence at lower levels, suggest an increase in particle size due to agglomeration as the Fe2O3 loading rises. The interactions between Fe2O3 nanorods and PVA/PANI chains are further supported by the formation of hydrogen bonds between the nanorods and the hydroxyl groups of PVA/PANI. A similar trend was observed by Abdelhamied et al.,18 who observed a decrease in semicrystallinity when Ag nanoparticles were incorporated into PVA/PANI polymer blend. To quantify structural parameters, the main (101) peak of the PVA/PANI-based samples was fitted using a Gaussian function (Fig. 2). From this analysis, the values of (D nm), (ε), and (δ) were determined using the standard relations provided in ref. 32:| |
 | (2) |
| |
 | (3) |
| |
 | (4) |
 |
| | Fig. 1 XRD patterns for pure PVA/PANI polymer blend and PVA/PANI-Fe2O3 nanocomposites samples. | |
 |
| | Fig. 2 Gaussian fitting of (101) peak for pure PVA/PANI polymer blend and PVA/PANI-Fe2O3 nanocomposites samples. | |
The calculated structural parameters are summarized in Table 1 and illustrated in Fig. 2. It is evident that the crystallite size (D) decreases progressively as the Fe2O3 nanorod content in the PVA/PANI matrix increases. This reduction is attributed to the disruption of the original hydrogen-bonding network within the PVA/PANI blend when Fe2O3 nanorods are introduced, as new hydrogen bonds form between the nanorods and the polymer chains instead.33 Conversely, both the internal strain (ε) and dislocation density (δ) exhibit an upward trend with increasing Fe2O3 nanorod concentrations. The elevated values of ε and δ further support the conclusion that the crystallinity of the PVA/PANI blend diminishes upon incorporation of Fe2O3 nanorods.34
Table 1 Geometrical parameters of PVA/PANI-Fe2O3 nanocomposites samples
| Composites |
β (radians) |
2θ |
D (nm) |
ε |
δ (nm−2) |
| PVA/PANI (blend) |
2.65 |
19.82 |
4.20 |
0.0564 |
0.124 |
| Blend-1.0% Fe2O3 |
2.87 |
20.02 |
3.3 |
0.0624 |
0.146 |
| Blend-2.5% Fe2O3 |
3.14 |
20.33 |
3.2 |
0.0781 |
0.152 |
| Blend-3.5% Fe2O3 |
3.49 |
19.97 |
3.1 |
0.0762 |
0.178 |
| Blend-4.5% Fe2O3 |
3.38 |
20.38 |
3.0 |
0.0812 |
0.169 |
3.2 FTIR study
FTIR spectroscopy was employed to examine how the functional groups within the PVA/PANI polymer matrix interact with the incorporated Fe2O3 nanorods. Fig. 3 shows the FT-IR spectra of pristine PVA/PANI and composites containing different concentrations of Fe2O3 nanorods. In the pristine PVA/PANI sample, a broad and intense absorption band appears at 3286 cm−1, which is ascribed to OH stretching vibration, confirming the presence of hydroxyl group.35 As the loading of Fe2O3 nanorods increases, this band progressively diminishes and eventually disappears, indicating the creation of intermolecular hydrogen bond between the OH group of poly(vinyl alcohol) and the Fe2O3 surface.36 This modification implies that the CH2 units in the side chain of PVA/PANI may coordinate with Fe2+ ions. The absorption at 2933 cm−1 corresponds to the asymmetric stretching of CH3 groups.37 Additionally, the pure PVA/PANI exhibits a peak at 1609 cm−1, associated with the stretching of the –COO functional group. The band at 1481 cm−1 is linked to CH2 scissoring vibrations, while the signal at 1318 cm−1 arises from ether group stretching. The characteristic C–O stretching vibration appears near 1134 cm−1,38 and the peak detected at 1084 is due to asymmetric C–C stretching modes.37 Another notable peak at 858 corresponds to CH2 stretching vibrations.36 The incorporation of Fe2O3 nanorods into the polymer blend caused noticeable shifts in key absorption peaks, particularly those at 3286, 1609, and 1134 cm−1, toward lower wavenumbers, confirming strong interactions between the nanofiller and the host matrix. Minor spectral changes also emerged with increasing Fe2O3 content, consistent with modifications in the polymer environment upon doping. The overall reduction in characteristic vibrational frequencies provides clear evidence of bonding between PVA/PANI and Fe2O3 nanorods, as illustrated in Scheme 2. These interactions contribute to a decrease in the crystallin degree within the amorphous region, in agreement with the XRD results.
 |
| | Fig. 3 FT-IR spectra of the PVA/PANI blend and the nanocomposite films containing various loadings of Fe2O3 nanorods (NRs). | |
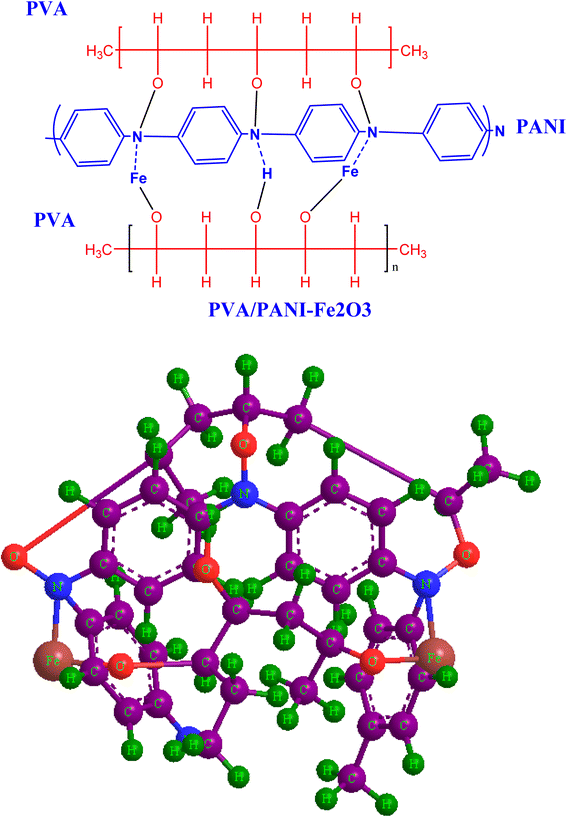 |
| | Scheme 2 Schematic representation of the proposed interaction mechanism between PVA/PANI and Fe2O3 nanorods, illustrated in both 2D and 3D formats. | |
3.3 TEM image
The TEM image corresponding to the highest Fe2O3 nanorod loading (4.5 wt%) reveals a dense and well-dispersed distribution of nanorods within the PVA/PANI polymer matrix (Fig. 4). The nanorods exhibit a distinct elongated morphology with a high aspect ratio, confirming the successful synthesis of rod-like Fe2O3 structures via the co-precipitation method.39 At this concentration, the nanorods are closely packed, forming a semi-interconnected network throughout the polymer matrix, which is expected to facilitate charge transport pathways. Despite the increased filler content, the nanorods remain relatively uniformly distributed due to the combined effect of prolonged stirring and ultrasonication, although slight agglomeration can be observed in some localized regions. This homogeneous dispersion with limited aggregation indicates strong interfacial interactions between Fe2O3 nanorods and the PVA/PANI chains, which plays a crucial role in enhancing the electrical conductivity and optical performance of the nanocomposite films.
 |
| | Fig. 4 TEM image of PVA/PANI-Fe2O3 (4.5 wt%) showing well-dispersed nanorods with slight aggregation; inset shows higher magnification. | |
3.4 UV-visible spectroscopy
The optical behavior of the prepared samples was examined using UV-visible spectroscopy. The corresponding spectra for the pristine PVA/PANI blend and the Fe2O3-incorporated PVA/PANI nanocomposites are presented in Fig. 5. In the UV region of the PVA/PANI spectrum, medium-intensity absorption bands appear at approximately 233, 292, and 398 nm, which are commonly associated with the carbonyl functionalities of PVA.40 Also, these bands originate from the π → π* electronic transitions of the benzenoid and quinoid segments of PANI. Upon introducing Fe2O3 nanorods into the polymer matrix, the ternary nanocomposites exhibit a noticeable red shift in the characteristic absorption peaks of the PVA/PANI blend. Furthermore, samples loaded with 2.5%, 3.5%, and 4.5% Fe2O3 show a marked increase in absorbance within the UV region.41 This shift is indicative of strong interactions between the Fe2O3 nanorods and the PVA/PANI host polymer. An additional absorption feature observed around 531 nm is attributed to the presence of iron oxide nanorods.42
 |
| | Fig. 5 The plots of absorption versus wavelength for PVA/PANI matrix and the nanocomposite films containing various loadings of Fe2O3 nanorods. | |
3.5 Optical parameters
3.5.1 Energy gap. The energy gap (Eg) of the PVA/PANI-Fe2O3 nanocomposites was calculate by the Davis–Mott equation:43Here α(cm−1) is the absorption coefficient, hν (eV) is the photon energy, and r takes the values 2 and 1/2 for direct (Egd) (eV) and indirect (Egi) (eV) allowed transitions, respectively. Here, Eg denotes the optical band gap, while C is a constant related to the absorption edge. The extracted Egd and Egi values, calculated from the plots in Fig. 6(a and b), are summarized in Table 2. A progressive reduction in both the direct and indirect band gap energies is observed with increasing Fe2O3 loading in the PVA/PANI matrix. This trend, evident in Fig. 6 and Tables 2, is ascribed to the creation of charge transfer complexes between the Fe2O3 nanorods and the functional group of the PVA/PANI blend, which modifies the electronic structure and narrows the band gap. Additionally, the incorporation of nanofillers introduces structural defects that generate localized states within the band gap, further contributing to the observed decrease in Eg.44 Furthermore, the observed band gap narrowing can be attributed to the combined effect of interfacial interactions, defect-induced localized states, and charge transfer mechanisms. The strong interfacial interaction between α-Fe2O3 nanorods and the PVA/PANI chains leads to electronic coupling, which modifies the density of states near the band edges. In addition, the incorporation of Fe2O3 introduces structural disorder and defect levels within the forbidden band gap, acting as intermediate energy states that facilitate electronic transitions at lower photon energies.
 |
| | Fig. 6 The relationship between (a) (αhυ)2 and (b) (αhυ)1/2 and hυ for PVA/PANI-Fe2O3 nanocomposites samples. | |
Table 2 The optical and electrical parameters for PVA/PANI-Fe2O3 nanocomposite films
| Composites |
Egd (eV) |
Egi (eV) |
Eu (eV) |
σdc (S.cm−1) |
σac (S.cm−1) |
| PVA/PANI (blend) |
3.91 |
2.45 |
0.1736 ± 0.0014 |
3.58 × 10−13 |
5.39 × 10−8 |
| Blend-1.0% Fe2O3 |
3.53 |
2.04 |
0.1922 ± 0.0037 |
4.09 × 10−12 |
1.14 × 10−7 |
| Blend-2.5% Fe2O3 |
3.01 |
1.91 |
0.2185 ± 0.0027 |
1.48 × 10−11 |
5.89 × 10−7 |
| Blend-3.5% Fe2O3 |
2.90 |
1.63 |
0.2318 ± 0.0052 |
6.41 × 10−11 |
9.92 × 10−7 |
| Blend-4.5% Fe2O3 |
2.59 |
1.09 |
0.2537 ± 0.0034 |
2.64 × 10−10 |
3.32 × 10−6 |
3.5.2 Urbach energy. The incorporation of Fe2O3 nanorods increases the structural disorder within the polymeric matrix. This enhancement in disorder can be evaluated by estimating the Urbach energy (EU), which is derived from the empirical relationship between the absorption coefficient and photon energy:45| |
 | (6) |
where αo (cm−1) is a material-dependent constant. Fig. 7 presents the plot of ln(α) Vs. hυ in the region near the absorption edge, yielding a linear dependence consistent with eqn (6). The Urbach energy values, determined from the inverse slope of these linear fits, are listed in Table 2. An increase in EU is observed with higher Fe2O3 content in the PVA/PANI matrix, indicating that the introduction of nanofiller enhances structural disorder and results in the creation of additional localized states inside the band gap. This broadening of the tail states increases the probability of electronic transitions such as tail-to-tail and band-to-tail, further reflecting the higher degree of disorder in the prepared nanocomposite films.
 |
| | Fig. 7 The plots of ln(α) vs. photon energy for pure PVA/PANI polymer blend and PVA/PANI-Fe2O3 nanocomposites samples. | |
3.6 Electrical properties
3.6.1 AC conductivity. The electrical conductivity of materials, especially their alternating current conductivity (σac), plays a vital role in explaining charge transport behavior in polymer nanocomposites.46 The electrical conductivity of the PVA/PANI system containing different loadings of Fe2O3 nanorods was evaluated at ambient temperature over a wide frequency window, as displayed in Fig. 8. The conductivity values were calculated using eqn (7):47| |
 | (7) |
Here Gp (S (Siemens)) is the parallel conductance of the film, A (cm2) its surface area, and T(cm) is the film thickness. The conductivity curves reveal three characteristic frequency-dependent regions. Low-frequency region (dispersion): σac shows a reduction due to electrode polarization. In this regime, charge carriers gather at the electrode/electrolyte interface, forming a barrier and restricting charge movement. The conductivity remains almost constant here since charge transport is mainly controlled by freely moving ions within the polymer matrix. Intermediate frequency region (σdc plateau): conductivity approaches the DC limit (σdc), attributed to ions migrating through the bulk of the material.48 This plateau reflects the intrinsic ion-transport capability of the polymeric matrix under an applied electric field. High-frequency region (rise in σac): conductivity increases sharply, signifying improved carrier mobility. This behavior is associated with the release or activation of trapped ions, which can readily follow the rapidly alternating field at higher frequencies.49 Introducing Fe2O3 into the PVA/PANI blend substantially improves σac. For instance, the pristine polymer blend shows an AC conductivity (σac) of 5.39 × 10−8 S cm−1 at high frequency, which rises to 3.32 × 10−6 S cm−1 at 4.5 wt% Fe2O3. This remarkable enhancement is attributed to the formation of additional charge transport pathways and a higher density of mobile carriers generated by the dispersed nanofiller. The Fe2O3 nanorods promote efficient charge movement by increasing the number of conduction channels and enabling long-range ion hopping. The XRD results further suggest that increased amorphous content provides localized states that facilitate ionic migration, while the recorded reduction in energy gap (UV/Vis.) implies a higher density of energy states, contributing to easier charge transfer.50 Overall, the results confirm that Fe2O3 incorporation effectively tunes the AC conductivity of PVA/PANI nanocomposites. A similar trend has been widely reported in various nanofiller-reinforced polymer systems, where a higher nanofiller loading results in enhanced electrical conductivity.51 The reported conductivity values correspond to the AC conductivity (σac) evaluated at a high-frequency region (e.g., 106 Hz), where the conductivity reaches its maximum and becomes less affected by electrode polarization effects. In addition, the DC conductivity (σdc) was extracted from the frequency-independent plateau observed in the intermediate frequency region, as shown in Fig. 8 and summarized in Table 2.
 |
| | Fig. 8 The plots of σac versus log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (F) for pure PVA/PANI polymer blend and PVA/PANI-Fe2O3 nanocomposites samples. (F) for pure PVA/PANI polymer blend and PVA/PANI-Fe2O3 nanocomposites samples. | |
3.6.2 Dielectric parameters. Dielectric behavior plays a crucial role in determining the suitability of polymer-based materials for electrical insulation, charge storage, and next-generation flexible electronics. The complex dielectric permittivity of any dielectric medium is expressed as:| |
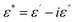 | (8) |
Here ε′ (dimensionless) corresponds to the energy stored inside the materials, and ε″(dimensionless) denotes the energy dissipated during each AC field cycle. These parameters can be determined through the following equations:52| |
 | (9) |
| |
 | (10) |
Fig. 9(a and b) depicts the variation of the (ε′) and (ε″) permittivity parts with log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (f) for PVA/PANI-Fe2O3 nanocomposites containing different filler contents. The values of (ε′) is strongly influenced by frequency due to Maxwell/Wagner interfacial polarizations. At lower frequencies, all samples exhibit high ε′ values as charges accumulate at the electrode–polymer interface, enabling dipoles to align effectively with the applied fields. When the frequency increases, dipole relaxation becomes delayed, reducing their ability to orient with the oscillating field, resulting in a gradual decline in ε′.53,54 At sufficiently high frequencies, only electronic and atomic polarization mechanisms remain active, while interfacial and orientational contributions gradually diminish. The observed enhancement in ε′ upon Fe2O3 incorporation is attributed to increased interfacial polarization sites and improved charge storage capability as a result of the creation of extra interfaces between the polymer and nanofiller.
(f) for PVA/PANI-Fe2O3 nanocomposites containing different filler contents. The values of (ε′) is strongly influenced by frequency due to Maxwell/Wagner interfacial polarizations. At lower frequencies, all samples exhibit high ε′ values as charges accumulate at the electrode–polymer interface, enabling dipoles to align effectively with the applied fields. When the frequency increases, dipole relaxation becomes delayed, reducing their ability to orient with the oscillating field, resulting in a gradual decline in ε′.53,54 At sufficiently high frequencies, only electronic and atomic polarization mechanisms remain active, while interfacial and orientational contributions gradually diminish. The observed enhancement in ε′ upon Fe2O3 incorporation is attributed to increased interfacial polarization sites and improved charge storage capability as a result of the creation of extra interfaces between the polymer and nanofiller.
 |
| | Fig. 9 The plot of (a) ε′ and (b) ε″ with log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (f) for the PVA/PANI-Fe2O3 nanocomposite samples. (f) for the PVA/PANI-Fe2O3 nanocomposite samples. | |
The dielectric loss (ε″), shown in Fig. 9(b), also exhibits a strong frequency dependence. At low frequencies, ε″ values are high owing to the large segmental mobility of polymer chains and dominant dipolar polarization. As frequency rises, polymer segments can no longer follow the rapidly oscillating field, leading to a decrease in dielectric loss.55 An increase in ε″ with higher Fe2O3 concentration suggests stronger interfacial polarization and enhanced dipolar relaxation within the nanocomposite matrix. This effect is advantageous for energy-storage applications, such as capacitors, where high permittivity and charge retention are required.56 Conversely, the decreasing dielectric parameters at high frequencies indicate reduced energy dissipation, rendering these materials suitable for use as dielectric layers in micro- and nano-electronic systems.57
3.6.3 Complex electric modulus. The complex electric modulus (M*) is closely related to the dielectric permittivity (ε*) and is expressed as follows:58,59| |
 | (11) |
where M′ and M″ represent the real and imaginary components of the modulus, respectively. The modulus formalism is particularly valuable for analyzing relaxation dynamics and understanding ion-transport mechanisms in polymer nanocomposites. Fig. 10(a) and (b) display the variation of (M′) and (M″) with frequency for samples containing different Fe2O3 loadings. At low frequencies, both M′ and M″ approach zero, indicating negligible electrode polarization and limited charge-carrier behaviour within this frequency region. This behaviour suggests that charge transport is highly restricted and that the restoring force acting on the charge carriers is insufficient to generate polarization, implying that electrode effects do not dominate the relaxation process. As frequency increases, distinct and broadened peaks appear in the M′ and M″ spectra, demonstrating the occurrence of relaxation phenomena inside the nanocomposite system. These peaks correspond to the relaxation frequency (fmax), which is inversely related to the relaxation time τ of mobile ions via the relation τ = 1/(2πfmax). The existence of well-defined peaks confirms that ionic hopping is the primary conduction mechanism in these materials.60,61 The shift of the M″ peaks to higher frequencies with increasing Fe2O3 content indicates shorter relaxation times and enhanced ion mobility within the nanocomposite. The noticeably small M″ values at low frequencies additionally suggest minimal ion displacement and reduced polarization at the electrodes, consistent with restricted charge dynamics.62 Notably, the M″ peak intensity becomes higher upon incorporating Fe2O3 nanorods compared to the pristine PVA/PANI polymer blend, reflecting an altered relaxation time and enhanced ion mobility induced by the nanofiller. The broadening of the peaks suggests a distribution of relaxation times, likely due to heterogeneity introduced by the nanorods and local structural disorder. Overall, the electric modulus analysis confirms that α-Fe2O3 nanorods promote faster ion hopping and facilitate charge transport, consistent with the observed enhancement in AC conductivity.
 |
| | Fig. 10 The plot of (a) M′ and (b) M″ with log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (f) for the PVA/PANI-Fe2O3 nanocomposites samples. (f) for the PVA/PANI-Fe2O3 nanocomposites samples. | |
3.6.5 Nyquist analysis. Nyquist plots of the PVA/PANI-Fe2O3 nanocomposites exhibit a characteristic semicircular arc in the higher frequencies range and subsequently showing a linear tail at low frequency, reflecting two distinct electrochemical processes, as shown in Fig. 11. The impedance spectra were interpreted using an equivalent electrical circuit model consisting of the bulk resistance (Rb) in parallel with two constant phase elements (CPE1 and CPE2). The value of Rb, obtained from the intercept of the semicircle with the Z′ axis, represents the intrinsic resistance of the material.65,66 The high-frequency semicircle corresponds to the bulk response of the system, where Rb and CPE1 dominate the relaxation process. CPE1 accounts for interfacial effects at the electrode–electrolyte boundary and participates directly in charge-transfer mechanisms. In contrast, the low-frequency inclined line is associated with diffusion-controlled transport or a more complex relaxation that cannot be described by Rb and CPE alone. A progressive decrease in the diameter of the semicircle with increasing Fe2O3 content confirms a reduction in bulk resistance and enhanced electrical conductivity.67 This improvement arises from the formation of new ion-transport pathways facilitated by the nanofiller, which reduces the activation energy required for ionic motion.68 The finding aligns well with FTIR and XRD analyses, which verify strong interactions between the filler and polymer chains. The addition of Fe2O3 significantly boosts ionic conductivity by promoting more continuous conduction channels along the polymer network, enabling easier ion migration.69 The parameters (Q1), (n1), (Q2), and (n2) corresponding to (CPE1) and (CPE2) were extracted through circuit fitting (Table 3) to provide quantitative insight into relaxation dynamics within the system. A marked decrease in Rb [from 1.00 × 109 Ω to 1.51 × 108 Ω] was observed at high filler loading, indicating the presence of more efficient conduction pathways and improved ion mobility under alternating electric fields, a behavior consistent with semiconductor conduction mechanisms.70 Meanwhile, the shrinking semicircle at high frequency reflects reduced impedances at interfaces and higher carrier mobility Fig. 12. Collectively, these features demonstrate that Fe2O3-enriched PVA/PANI nanocomposites possess favorable conductivity, strong capacitive characteristics, and high charge-storage efficiency.71
Table 3 Optimized circuit-model fitting parameters for the PVA/PANI-Fe2O nanocomposites
| Composites |
Fitting parameters |
| Rb (Ω) |
Q1 (F) |
n1 |
Q2 (F) |
n2 |
| PVA/PANI (blend) |
1.00 × 109 |
1.61 × 10−10 |
0.93 |
4.72 × 10−9 |
0.37 |
| Blend-1.0% Fe2O3 |
4.99 × 108 |
1.69 × 10−10 |
0.83 |
1.08 × 10−8 |
0.46 |
| Blend-2.5% Fe2O3 |
4.72 × 108 |
1.72 × 10−10 |
0.78 |
8.14 × 10−9 |
0.45 |
| Blend-3.5% Fe2O3 |
2.51 × 108 |
2.44 × 10−10 |
0.74 |
1.21 × 10−8 |
0.35 |
| Blend-4.5% Fe2O3 |
1.51 × 108 |
2.01 × 10−10 |
0.79 |
2.21 × 10−8 |
0.28 |
 |
| | Fig. 12 The relation between (Z′) and (Z″) for the PVA/PANI-Fe2O3 nanocomposites samples. | |
4 Conclusion
This study presents a comprehensive evaluation of PVA/PANI-Fe2O3 nanocomposites, demonstrating the strong influence of Fe2O3 nanorod loading on their structural, optical, and electrical properties. XRD and FTIR confirmed the successful incorporation of Fe2O3 into the PVA/PANI matrix, leading to reduced crystallinity, increased disorder, and strong filler–polymer interactions. Optical assessment revealed a red shift in absorption spectra along with significant band gap narrowing (Egd: 3.91 → 2.59 eV; Egi: 2.45 → 1.09 eV), while Urbach energy increased, confirming enhanced defect-state density and improved electronic transition capability. The nanocomposites also exhibited substantial enhancement in AC electrical conductivity from 5.39 × 10−8 to 3.32 × 10−6 S cm−1, attributed to improved charge carrier mobility and interfacial polarization. Overall, increasing Fe2O3 content effectively tunes the optical band structure, increases electrical conductivity, and improves dielectric response, positioning these nanocomposites as strong candidates for next-generation optoelectronic, photonic, and energy-related devices. This work demonstrates that the incorporation of α-Fe2O3 nanorods, rather than conventional nanoparticles, plays a key role in enhancing charge transport and tailoring the optical band gap. The combined improvement in optical and electrical properties highlights the potential of these nanocomposites for optoelectronic applications.
Author contributions
H. M. Ragab: methodology, formal analysis, investigation, writing – review and editing. N.S. Diab: investigation, writing – review & editing Rosilah Ab Aziz: methodology, formal analysis, investigation. Shimaa Mohammed Aboelnaga: investigation, writing – review & editing. Tahani M. Alresheedi: investigation, writing – review & editing. M.O. Farea: conceptualization, methodology, writing – review & editing. A Al Ojeery: investigation, writing – review & editing.
Conflicts of interest
The authors declare no conflicts of interest.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgements
This research has been funded by Scientific Research Deanship at University of Ha'il – Saudi Arabia through project number <<RG-25 066>>
References
- S. Kuvshinova, et al., PVC carbon nanostructure composite materials: Approaches to their fabrication and properties, Nanotechnol. Russ., 2015, 10(1), 1–12 CrossRef CAS.
- E. Abdelrazek, I. Elashmawi and H. Ragab, Manifestation of MnCl2 fillers incorporated into the polymer matrices in their dielectric properties, Phys. B, 2008, 403(18), 3097–3104 CrossRef CAS.
- J. Y. Lu, et al., Facile microwave-assisted synthesis of Sb2O3-CuO nanocomposites for catalytic degradation of p-nitrophenol, J. Mol. Liq., 2024, 409, 125503 CrossRef CAS.
- S. Wang, et al., Multifunctional tribovoltaic coating for self-powered in situ sensing with exceptional tribological robustness and charge transport, Adv. Funct. Mater., 2026, 36(8), e14190 CrossRef CAS.
- Š. Krobot, et al., Optimizing printability and mechanical properties of poly (3-hydroxybutyrate) biocomposite blends and their biological response to Saos-2 cells, Int. J. Bioprint., 2024, 11(1), 400 Search PubMed.
- M. S. Ismail, et al., Improving the refractive index by engineering PbS/PVA nano polymer composite for optoelectronic applications, Opt. Mater., 2022, 131, 112639 CrossRef CAS.
- A. I. Al-Sulami, et al., Enhanced Structural, Optical, and Electrical Properties of Polymer Composites Induced by CoFe2O4/MWCNTs Nanohybrids, Polym. Eng. Sci., 2026, 66(2), 896–904 CrossRef CAS.
- H. Ragab, et al., Enhanced detection of ammonia (NH3) using a TiO2/PANI/GO composite for real-time environmental monitoring, Chem. Phys. Lett., 2025, 869, 142044 CrossRef CAS.
- T. Zhang, et al., KNN-based frequency-adjustable ferroelectric heterojunction and biomedical applications, Nat. Commun., 2025, 16(1), 7120 CrossRef CAS PubMed.
- H. E. Ali, et al., Microstructure analysis and nonlinear/linear optical parameters of polymeric composite films based PVAL for wide optical applications, Phys. Scr., 2021, 96(11), 115804 CrossRef CAS.
- A. B. Alruqi, et al., PVA/NaAlg–CoFe2O4 nanocomposites: Structural, electrical, magnetic, optical, and dielectric characterization for energy storage and magneto-electronic devices, Ceram. Int., 2025, 54831–54839 CrossRef CAS.
- A. I. Al-Sulami, et al., Structural, optical, and electrical properties of Bi2O3/MWCNT-doped PVA/NaAlg nanocomposite films for flexible electronic applications, J. Sci.:Adv. Mater. Devices, 2025, 100979 CAS.
- A. Alshehri, et al., Dielectric properties and optical characteristics of PVA-chitosan blend incorporated by Al2O3 and V2O5 nanoparticles prepared via lased ablation and their potential for electrical applications, J. Mater. Sci.: Mater. Electron., 2025, 36(11), 654 CrossRef CAS.
- K. H. Alharbi, et al., Investigating electrical conductivity and antimicrobial performance of niobium oxide and praseodymium oxide nanoparticles filled Chitosan-PVA polymer blends, Inorg. Chem. Commun., 2025, 174, 113994 CrossRef CAS.
- O. Norfazlinayati, et al., Optical characterization of PANI/functionalized-MWCNTs/PVA nanocomposites induced by gamma irradiation, Synth. Met., 2021, 276, 116755 CrossRef CAS.
- I. Laourari, et al., Antimicrobial and antifungal properties of NiCu-PANI/PVA quaternary nanocomposite synthesized by chemical oxidative polymerization of polyaniline, Chemosphere, 2022, 291, 132696 CrossRef CAS PubMed.
- W. Li, et al., Flexible poly (vinyl alcohol)–polyaniline hydrogel film with vertically aligned channels for an integrated and self-healable supercapacitor, ACS Appl. Energy Mater., 2020, 3(9), 9408–9416 CrossRef CAS.
- M. Abdelhamied, et al., Oxygen ion induced variations in the structural and Linear/Nonlinear optical properties of the PVA/PANI/Ag nanocomposite film, Inorg. Chem. Commun., 2021, 133, 108926 CrossRef CAS.
- M. Arenas, et al., Electrical and morphological properties of polyaniline–polyvinyl alcohol in situ nanocomposites, Composites, Part B, 2014, 56, 857–861 CrossRef CAS.
- Q. A. Alsulami and A. Rajeh, Structural, thermal, optical characterizations of polyaniline/polymethyl methacrylate composite doped by titanium dioxide nanoparticles as an application in optoelectronic devices, Opt. Mater., 2022, 123, 111820 CrossRef CAS.
- N. Yang, et al., Achieving broadband electromagnetic absorption in laminated composites through progressive Bayesian optimization, Composites, Part B, 2025, 112882 CrossRef.
- M. Tadic, et al., Magnetic properties of hematite (α-Fe2O3) nanoparticles prepared by hydrothermal synthesis method, Appl. Surf. Sci., 2014, 320, 183–187 CrossRef CAS.
- D. E. Fouad, et al., Improved size, morphology and crystallinity of hematite (α-Fe2O3) nanoparticles synthesized via the precipitation route using ferric sulfate precursor, Results Phys., 2019, 12, 1253–1261 CrossRef.
- N. Wadatkar and S. Waghuley, Complex optical studies on conducting polyindole as-synthesized through chemical route, Egypt. j. basic appl. sci., 2015, 2(1), 19–24 Search PubMed.
- A. Al Ojeery and M. Farea, Optimizing and boosting the physicochemical properties of some polymer nanocomposites for high-performance and flexible energy storage systems, J. Energy Storage, 2024, 80, 110309 CrossRef CAS.
- N. Wadatkar and S. Waghuley, Studies on properties of as-synthesized conducting polythiophene through aqueous chemical route, J. Mater. Sci.: Mater. Electron., 2016, 27, 10573–10581 CrossRef CAS.
- A. Badawi, et al., The effect of iron oxide content on the structural and optical parameters of polyvinyl alcohol/graphene nanocomposite films, J. Vinyl Addit. Technol., 2022, 28(1), 235–246 CrossRef CAS.
- H. Ragab, Influence of α–Hematite Nanorods (αFe2O3 NRs) on the Optical, Magnetic, and Electrical Properties of PEO/NaAlg Blend for Magneto-Optical Applications, J. Inorg. Organomet. Polym. Mater., 2023, 33(2), 484–494 CrossRef CAS.
- A. El Sayed and W. J. J. O. M. S. Morsi, α-Fe2O3/(PVA+ PEG) nanocomposite films; synthesis, optical, and dielectric characterizations, J. Mater. Sci., 2014, 49(15), 5378–5387 CrossRef CAS.
- R. Ahmed, et al., Influence of Fe2O3@ reduced graphene oxide nanocomposite on the structural, morphological, and optical features of the polyvinyl alcohol films for optoelectronic applications, Phys. Scr., 2023, 98(5), 055928 CrossRef CAS.
- S. Chapi, Optical, electrical and electrochemical properties of PCL5/ITO transparent conductive films deposited by spin-coating–Materials for single-layer devices, J. Sci.:Adv. Mater. Devices, 2020, 5(3), 322–329 Search PubMed.
- H. Ragab, et al., Tailoring optical and electrical properties of hybrid polymer nanodielectrics: Synthesis and characterization of CuO/TiO2 nanoparticle-embedded HPMC/NaAlg blend, Ceram. Int., 2025, 17302–17310 CrossRef CAS.
- C. Bao, et al., Poly (vinyl alcohol) nanocomposites based on graphene and graphite oxide: a comparative investigation of property and mechanism, J. Mater. Chem., 2011, 21(36), 13942–13950 RSC.
- S. Chapi and H. J. J. O. M. S. M. I. E. Devendrappa, Optical, electrical, thermal and electrochemical studies of spin-coated polyblend-ZnO nanocomposites, J. Mater. Sci.: Mater. Electron., 2016, 27(11), 11974–11985 CrossRef CAS.
- H. El-Mallah, et al., Structural characterization and optical parameter of silicon phthalocyanine dichloride thin films dependence with gamma ray radiation, Radiat. Phys. Chem., 2020, 176, 109012 CrossRef CAS.
- T. Rajavardhana Rao, et al., Electron paramagnetic resonance and optical absorption studies of manganese ions doped in polyvinyl (alcohol) complexed with polyethylene glycol polymer films, Ionics, 2012, 18(7), 695–701 CrossRef.
- F. K. M. Genova, et al., Study on blend polymer (PVA-PAN) doped with lithium bromide, Polym. Sci., 2015, 57(6), 851–862 CAS.
- M. T. Taghizadeh and N. Sabouri, Study of enzymatic degradation and water absorption of nanocomposites polyvinyl alcohol/starch/carboxymethyl cellulose blends containing sodium montmorillonite clay nanoparticle by cellulase and α-amylase, J. Taiwan Inst. Chem. Eng., 2013, 44(6), 995–1001 CrossRef CAS.
- Z. Li, et al., Direct hydrothermal synthesis of single-crystalline hematite nanorods assisted by 1, 2-propanediamine, Nanotechnology, 2009, 20(24), 245603 CrossRef PubMed.
- T. Siddaiah, et al., Structural, optical and thermal characterizations of PVA/MAA: EA polyblend films, Mater. Res., 2018, 21(5), e20170987 CAS.
- A. A. Ahmad, et al., Characterization of As-prepared PVA-PEO/ZnO-Al2O3-NPs hybrid nanocomposite thin films, Polym. Bull., 2022, 79(11), 9881–9905 CrossRef CAS.
- A. Mohd Meftah, et al., Structural, optical and electrical properties of PVA/PANI/Nickel nanocomposites synthesized by gamma radiolytic method, Polymers, 2014, 6(9), 2435–2450 CrossRef.
- E. Elesh, D. El Damhogi and Z. Mohamed, Gamma radiation induced variation in structure formation and optical characteristics of evaporated tris [2-phenylpyridinato-C2, N] iridium (III) films prepared by electron beam evaporator in photovoltaic applications, Phys. Scr., 2023, 98(3), 035817 CrossRef CAS.
- H. El-Mallah, et al., Structural characterization and optical parameter of silicon phthalocyanine dichloride thin films dependence with gamma ray radiation, Radiat. Phys. Chem., 2020, 176, 109012 CrossRef CAS.
- E. Elesh, et al., Thermal annealing enhanced morphological, nonlinear characteristic, and optical features of Victoria blue nanofilms for photonic application, Optik, 2023, 295, 171486 CrossRef CAS.
- Q. Zhou, et al., Co3S4-pyrolysis lotus fiber flexible textile as a hybrid electrocatalyst for overall water splitting, J. Energy Chem., 2024, 89, 336–344 CrossRef CAS.
- F. Hanash, et al., Tunable structural, optical, and electrical performance of PEMA/PMMA–CoCl2 composites for advanced optoelectronics and energy storage applications, Sci. Rep., 2025, 15(1), 42157 CrossRef CAS PubMed.
- F. H. Alkallas, et al., Influence of Al2O3 nanoparticles on the structural, optical, and electrical properties of PVC/PS nanocomposite for use in optoelectronic devices, Surf. Interfaces, 2024, 51, 104651 CrossRef CAS.
- A. A. Al-Ghamdi, et al., Enhanced structural, optical, dielectric, and electrical properties in polymer nanocomposite films via loading AgNO3 NPs for emerging optical and electrical applications, J. Mater. Sci.: Mater. Electron., 2024, 35(17), 1134 CrossRef CAS.
- M. Maniruzzaman, et al., ITO free MoO3/Au/MoO3 structures using Al2O3 as protective barrier between MoO3 and PEDOT: PSS in organic solar cells, Renewable Energy, 2014, 71, 193–199 CrossRef CAS.
- A. M. Alghamdi, Fabrication and comprehensive characterization of HPMC/PVA/CMC-MoO3 bio-nanocomposites: Enhanced mechanical, electrical, and antibacterial properties for food packaging applications, Int. J. Biol. Macromol., 2025, 287, 138612 CrossRef CAS PubMed.
- J. C. Pandey and M. Singh, Dielectric polymer nanocomposites: past advances and future prospects in electrical insulation perspective, SPE Polym., 2021, 2(4), 236–256 CrossRef CAS.
- A. Menazea, et al., Enhanced dielectric properties of α-MoO3 nanobelts dispersed polyethylene oxide-carboxymethyl cellulose matrix-based nanocomposites, Mater. Chem. Phys., 2024, 312, 128585 CrossRef CAS.
- Z. Chen, et al., A highly efficient and recyclable living biocatalyst using Shewanella@ polydopamine@ NH2-doped carbon dot biohybrids and polypyrrole immobilized melamine foam for microbial-photoreduction of Cr (VI), J. Clean. Prod., 2024, 435, 140497 CrossRef CAS.
- N. A. Kattan, et al., Optimizing PEO/HPMC polymer blends with Al2O3/MoO3 as hybrid nanofiller for enhanced dielectric performance and energy storage systems, Reactive Funct. Polym., 2025, 106432 CrossRef CAS.
- E. Aldosari, et al., Novel PEVA/PMMA-based nanocomposites containing ZnO–Co nanoparticles: investigation of optical, dielectric and electrical properties for energy storage and organic optoelectronic devices, Opt. Quant. Electron., 2025, 57(4), 1–21 CrossRef.
- J. Yu, et al., Influence of interface structure on dielectric properties of epoxy/alumina nanocomposites, Macromol. Res., 2012, 20(8), 816–826 CrossRef CAS.
- S. Karadaş, et al., Complex dielectric, complex electric modulus, and electrical conductivity in Al/(Graphene-PVA)/p-Si (metal-polymer-semiconductor) structures, J. Phys. Chem. Solid., 2021, 148, 109740 CrossRef.
- A. S. Almuslem, et al., Enhanced electrical conductivity, dielectric properties, and thermal stability of polyvinyl alcohol filled by bismuth oxide and iron oxide for advanced dielectric applications, J. Mater. Sci.: Mater. Electron., 2025, 36(7), 436 CrossRef CAS.
- R. Quader, L. K. Narayanan and E. B. Caldona, Dielectric characterization of fiber-and nanofiller-reinforced polymeric materials, J. Appl. Polym. Sci., 2024, 141(19), e55362 CrossRef CAS.
- Z. Wang, et al., Tribological Behavior of Zwitterionic-Polyester Composite Films with Phase-Separated Structures, Langmuir, 2025, 41(45), 30167–30173 CrossRef CAS PubMed.
- G. R. Gajula, et al., Study on electric modulus, complex modulus and conductivity properties of Nb/Sm, Gd doped barium titanate-lithium ferrite ceramic composites, Results Phys., 2020, 17, 103076 CrossRef.
- M. Morsi, et al., Reinforced PEO/Cs polymers blend with Al2O3/TiO2 hybrid nanofillers: nanocomposites for optoelectronics and energy storage, J. Energy Storage, 2024, 88, 111554 CrossRef CAS.
- A. I. Al-Sulami, et al., Tunable band gap and ionic conductivity in PMMA and PANI blend with WO3 nanocomposites for optoelectronics and energy storage devices, Sci. Rep., 2025, 15(1), 34543 CrossRef CAS PubMed.
- A. A. Khan, et al., Temperature dependent charge conduction and relaxation mechanism study of nano-structure WO3 by impedance spectroscopy, Mater. Res. Express, 2019, 6(12), 1250a5 CrossRef CAS.
- E. Wu, et al., W-shaped antiambipolar transistors based on h-BN/MoTe2/BP heterostructures, ACS Nano, 2025, 19(40), 35701–35711 CrossRef CAS PubMed.
- L. Maaza, et al., Evaluation of the influence of Al2O3 nanoparticles on the thermal stability and optical and electrochemical properties of PANI-derived matrix reinforced conducting polymer composites, J. Phys. Chem. Solid., 2021, 152, 109970 CrossRef CAS.
- H. Ragab, et al., Exploring the optical properties of CuCo2O4-doped polyethylene oxide and carboxymethylcellulose for optoelectronic application, Opt. Quant. Electron., 2024, 56(3), 323 CrossRef CAS.
- S. N. A. M. Johari, et al., A review: ionic conductivity of solid polymer electrolyte based polyethylene oxide, Int. J. Electrochem. Sci., 2021, 16(10), 211049 CrossRef CAS.
- X. Zhao, et al., Frequency-dependence of electric double layer capacitance of TiO2-based composite nanotube arrays, J. Electroanal. Chem., 2016, 779, 199–206 CrossRef CAS.
- M. Pandey, et al., Electrical properties and thermal degradation of poly (vinyl chloride)/polyvinylidene fluoride/ZnO polymer nanocomposites, Polym. Int., 2016, 65(9), 1098–1106 CrossRef CAS.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article *d
*d
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1 was purchased from Merck (Germany). Polyaniline (PANI), having a molecular weight exceeding 19
000 g mol−1 was purchased from Merck (Germany). Polyaniline (PANI), having a molecular weight exceeding 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1, was sourced from Sigma-Aldrich. Ferric chloride (FeCl3, ≥98% purity, Sigma-Aldrich), deionized water, and aqueous ammonia solution (NH4OH, 25–28%, Sigma-Aldrich) were used in the preparation of the Fe2O3 nanorods.
000 g mol−1, was sourced from Sigma-Aldrich. Ferric chloride (FeCl3, ≥98% purity, Sigma-Aldrich), deionized water, and aqueous ammonia solution (NH4OH, 25–28%, Sigma-Aldrich) were used in the preparation of the Fe2O3 nanorods.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 ratio was fabricated using the solution casting technique. First, 1.6 g of PVA was dissolved in 100 mL of deionized water at 80 °C under continuous stirring until a clear homogeneous solution was formed. Separately, 0.4 g of PANI was dispersed in 40 mL of deionized water at room temperature. The two solutions were then combined, as the 80
20 ratio was fabricated using the solution casting technique. First, 1.6 g of PVA was dissolved in 100 mL of deionized water at 80 °C under continuous stirring until a clear homogeneous solution was formed. Separately, 0.4 g of PANI was dispersed in 40 mL of deionized water at room temperature. The two solutions were then combined, as the 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 PVA/PANI proportion was found to yield highly uniform and flexible films. The blended mixture was stirred for an additional hour to ensure complete homogenization. Fe2O3 nanorods were incorporated into the polymer blend at various loadings (1.0, 2.5, 3.5, and 4.5 wt%). Each amount of nanofiller was incorporated gradually to the solution under constant stirring for 1 hour, followed by ultrasonic treatment for 20 min at room temperature to minimize particle agglomeration. The required nanorod content M (1.0–4.5 wt%) was computed by eqn (1):
20 PVA/PANI proportion was found to yield highly uniform and flexible films. The blended mixture was stirred for an additional hour to ensure complete homogenization. Fe2O3 nanorods were incorporated into the polymer blend at various loadings (1.0, 2.5, 3.5, and 4.5 wt%). Each amount of nanofiller was incorporated gradually to the solution under constant stirring for 1 hour, followed by ultrasonic treatment for 20 min at room temperature to minimize particle agglomeration. The required nanorod content M (1.0–4.5 wt%) was computed by eqn (1):










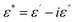


![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (f) for PVA/PANI-Fe2O3 nanocomposites containing different filler contents. The values of (ε′) is strongly influenced by frequency due to Maxwell/Wagner interfacial polarizations. At lower frequencies, all samples exhibit high ε′ values as charges accumulate at the electrode–polymer interface, enabling dipoles to align effectively with the applied fields. When the frequency increases, dipole relaxation becomes delayed, reducing their ability to orient with the oscillating field, resulting in a gradual decline in ε′.53,54 At sufficiently high frequencies, only electronic and atomic polarization mechanisms remain active, while interfacial and orientational contributions gradually diminish. The observed enhancement in ε′ upon Fe2O3 incorporation is attributed to increased interfacial polarization sites and improved charge storage capability as a result of the creation of extra interfaces between the polymer and nanofiller.
(f) for PVA/PANI-Fe2O3 nanocomposites containing different filler contents. The values of (ε′) is strongly influenced by frequency due to Maxwell/Wagner interfacial polarizations. At lower frequencies, all samples exhibit high ε′ values as charges accumulate at the electrode–polymer interface, enabling dipoles to align effectively with the applied fields. When the frequency increases, dipole relaxation becomes delayed, reducing their ability to orient with the oscillating field, resulting in a gradual decline in ε′.53,54 At sufficiently high frequencies, only electronic and atomic polarization mechanisms remain active, while interfacial and orientational contributions gradually diminish. The observed enhancement in ε′ upon Fe2O3 incorporation is attributed to increased interfacial polarization sites and improved charge storage capability as a result of the creation of extra interfaces between the polymer and nanofiller.









