Open Access Article
Open Access ArticleHigh-sensitivity optical thermometry in Tb3+/Eu3+ co-doped Li2Y4(MoO4)7 phosphors synthesized via solid-state reaction
Fadwa Ayachia,
Kamel Saidi ab,
Mohamed Dammak
ab,
Mohamed Dammak *a,
I. Mediavilla-Martínezc and
J. Jimenezc
*a,
I. Mediavilla-Martínezc and
J. Jimenezc
aLaboratoire de Physique Appliquée, Faculté des Sciences de Sfax, Département de Physique, Université de Sfax, BP 1171, Sfax, Tunisia. E-mail: mohamed.dammak@fss.usf.tn; madidammak@yahoo.fr
bDepartment of Physics, Sfax Preparatory Engineering Institute, University of Sfax, 1172-3000, Sfax, Tunisia
cGdS Optronlab, Department of Condensed Matter Physics, LUCIA Building University of Valladolid, Paseo de Belen 19, 47011, Valladolid, Spain
First published on 27th March 2026
Abstract
Developing ratiometric optical thermometers with high sensitivity and signal stability remains a critical challenge in non-contact temperature sensing. Herein, a series of 5% Eu3+, 40% Tb3+ doped, and 40% Tb3+/5% Eu3+ co-doped Li2Y4(MoO4)7 (LiYMoO:Tb/Eu) phosphors were synthesized utilizing a conventional solid-state reaction route. Structural refinement and morphological inspections confirmed the phase purity and crystallinity of the resulting molybdate host. Under 325 nm ultraviolet excitation, the photoluminescence spectra revealed a multi-center emission profile, featuring characteristic transitions of Tb3+ (green) and Eu3+ (red) host framework, indicating an efficient energy transfer network among the dopants and the host. To evaluate the material's potential for thermometry, the fluorescence intensity ratio (FIR) technique was applied using the non-thermally coupled transitions of Tb3+ (5D4 → 7F5 at 542 nm) and Eu3+ (5D0 → 7F4 at 701 nm). The thermometric performance was investigated over the physiological and industrial temperature range of 300–403 K. Notably, the phosphor exhibited a superior maximum relative sensitivity (Sr) of 6.5% K−1 at 300 K, significantly outperforming many existing molybdate-based sensors. These findings suggest that the 40% Tb3+/5% Eu3+ co-doped Li2Y4(MoO4)7 system is a promising candidate for precise optical temperature sensing applications.
1. Introduction
In recent years, lanthanide-doped luminescent materials have attracted significant scientific interest due to their multifunctional capabilities in next-generation solid-state lighting (SSL), optical display technologies, and non-contact temperature sensing.1–6 The development of high-performance phosphors requires a careful selection of the host matrix, as its crystal field environment and phonon energy critically influence the radiative and non-radiative relaxation pathways of the dopant ions. Among the various oxide hosts, molybdate compounds have emerged as exceptional candidates owing to their high chemical stability, excellent thermal durability, and moderate phonon energy, which minimizes quenching losses.7,8A strategic approach to optimizing luminescent performance involves the co-doping of rare-earth (RE3+) ions to exploit energy transfer (ET) mechanisms. Europium (Eu3+) is widely recognized as a superior activator for red emission, characterized by its intense 5D0 to 7F2 transition, which is essential for improving the color rendering index (CRI) and warm-light components in white light-emitting diodes (W-LEDs).9,10 Conversely, terbium (Tb3+) provides a distinct green emission via the 5D4 to 7F5 transition at approximately 542 nm. While singly doped materials are limited to monochromatic outputs, the Tb3+/Eu3+ co-doped system has garnered considerable attention because it enables color tunability through efficient Tb3+ to Eu3+ energy transfer. This mechanism allows for the precise adjustment of emission chromaticity from green to red by modulating dopant concentrations, offering a versatile platform for tunable LED components.11,12 Beyond illumination, these RE3+-doped phosphors are increasingly utilized for optical thermometry, a technique that overcomes the limitations of conventional contact sensors in harsh or electromagnetic environments. The Fluorescence Intensity Ratio (FIR) method, which relies on the intensity ratio of two distinct emission bands, is particularly favored for its immunity to fluctuations in excitation power and spectrum losses.13 Building upon our previous investigations into rare-earth-doped phosphors, our group has extensively explored the thermometric potential of various host matrices. We have previously demonstrated high-sensitivity sensing in systems such as Er3+/Yb3+ co-doped molybdates and vanadates, where energy transfer plays a pivotal role in signal stability.14,15 For instance, our recent work on Na3Y(VO4)2:Eu3+ highlighted the efficacy of Eu3+ transitions for temperature monitoring,16 while our studies on phosphate matrices elucidated the mechanisms of color tuning via Tb3+/Eu3+ interaction.17 However, the exploration of the Li2Y4(MoO4)7 host remains limited.
The present work does not aim to provide a comprehensive study of energy transfer dynamics between Tb3+ and Eu3+ ions, but rather to exploit the thermally distinct emission responses of their characteristic bands as the basis for high-sensitivity non-contact LIR thermometry in the novel Li2Y4(MoO4)7 host.
In this study, we report the synthesis of a novel series of Tb3+, Eu3+, and Tb3+/Eu3+ co-doped Li2Y4(MoO4)7 (LiYMoO) phosphors via a conventional solid-state reaction. We present a systematic analysis of their structural phase purity and morphological characteristics. Furthermore, we investigate the photoluminescence properties and the Tb3+ to Eu3+ energy transfer mechanism in detail. A key focus of this work is to evaluate the optical thermometry performance of the co-doped system using the FIR technique under 325 nm excitation. The results are compared with our previous findings to demonstrate the superior relative sensitivity and signal reproducibility of the Li2Y4(MoO4)7 matrix, establishing its suitability for sensing applications.
2. Experimental section
2.1. Sample synthesis
A series of LiYMoO phosphors, including singly doped (Eu3+, Tb3+) and co-doped (Tb3+/Eu3+) samples, were synthesized via a conventional high-temperature solid-state reaction method. The starting precursors employed were analytical grade lithium carbonate (Li2CO3, 99.99%), molybdenum trioxide (MoO3, 99.99%), yttrium oxide (Y2O3, 99.99%), terbium oxide (Tb4O7, 99.99%), and europium oxide (Eu2O3, 99.99%). Stoichiometric amounts of the raw materials were accurately weighed according to the nominal composition of Li2Y{4−x−y}TbxEuy(MoO4)7. The powders were thoroughly ground in an agate mortar with a small quantity of ethanol to facilitate homogenization. After drying, the mixtures were transferred into alumina crucibles and sintered in an electric furnace at 1273 K for 8 h in an air atmosphere. Finally, the obtained products were naturally cooled to room temperature and ground into fine powders for further characterization.2.2. Characterization techniques
The phase purity and crystal structure of the as-prepared phosphors were identified by X-ray diffraction (XRD) using a Bruker AXS D8 diffractometer equipped with Cu K alpha radiation (lambda = 1.5406 Å). The surface morphology and particle size distribution were examined using a field-emission scanning electron microscope (FE-SEM, Hitachi SU-4800). Photoluminescence (PL) measurements were conducted at room temperature using a high-resolution Horiba-Jobin Yvon LabRAM HR 800 UV Raman spectrometer coupled with an Olympus BX41 confocal microscope. The excitation source was the 325 nm line of a He–Cd continuous-wave laser. The emitted signal was detected by a charge-coupled device (CCD) detector. Spectral resolution was controlled using holographic gratings with groove densities of 2400 lines per mm (for high-resolution scans) and 150 lines per mm (for broad surveys) and the spectral resolution was approximately ∼1 nm. To prevent thermal heating effects during measurement, the laser power incident on the sample surface was maintained below 1 mW, corresponding to an irradiance of less than 100 kW cm−2, for which no line shifts were observed.3. Results and discussion
3.1. Structural analysis and phase purity
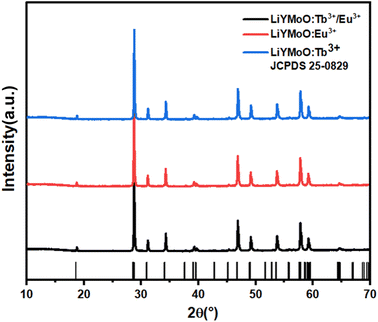
The phase composition and crystal structure of the synthesized LiYMoO phosphors singly doped with 5% Eu3+ and 40% Tb3+, as well as co-doped with ion patterns are presented in Fig. 1. The detected diffraction peaks are in accordance well with the standard Na0.5Gd0.5MoO4 (JCPDS 25-0829) which is of a single phase with tetragonal scheelite structure and a space group I41/a. The diffraction data are consistent with the general family of defect scheelite-type double molybdates with composition A2Ln4(MoO4)7 (where A = alkali metal, Ln = rare-earth element). No secondary phases or impurity peaks related to unreacted precursors were detected, confirming that the synthesis conditions (1273 K for 8 h) were sufficient to obtain a pure, single-phase solid solution. The sharp and intense diffraction peaks attest to the high crystallinity of the samples. Furthermore, the incorporation of Eu3+ and Tb3+ activators into the host lattice did not induce any significant shift in peak positions or observable changes in the diffraction profiles. This structural stability is attributed to the successful isovalent substitution of Y3+ ions by Eu3+ and Tb3+ ions at the same crystallographic site, which is facilitated by the close similarity of their ionic radii. According to Shannon's crystal ionic radii tables,18 all three ions share coordination number CN = 8 with the following values: Y3+ (1.019 Å), Tb3+ (1.040 Å), and Eu3+ (1.066 Å), corresponding to lattice mismatch values of only +2.1% and +4.6% for Tb3+ and Eu3+ with respect to Y3+, respectively. These marginal size differences are well within the tolerance range for solid-solution substitution without structural distortion. Consequently, the structural integrity of the Li2Y4(MoO4)7 host matrix is fully preserved across all doping compositions, as evidenced by the absence of systematic lattice parameter shifts and the conservation of the tetragonal scheelite-type structure throughout the entire series.To further confirm the phase purity and crystal structure of the synthesized LiYMoO:Tb3+, LiYMoO:Eu3+ and LiYMoO:5% Eu3+/40% Tb3+ phosphors, Rietveld refinement of the powder XRD data were performed using the tetragonal scheelite-type structural model with space group I41/a. The refined patterns are presented in Fig. 2. Rietveld refinement of the XRD patterns confirmed that both Tb3+, Eu3+ and Eu3+/Tb3+ doped LiYMoO phosphors crystallize in the tetragonal scheelite-type structure (space group I41/a) without secondary phases. The good agreement between experimental and calculated patterns indicates high crystallinity and phase purity. Eu3+, Tb3+ and Eu3+/Tb3+ ions enter into Y3+ sites without inducing structural distortion, thereby preserving the structural stability of the host lattice. Table 1 lists the crystals cell and structural properties.
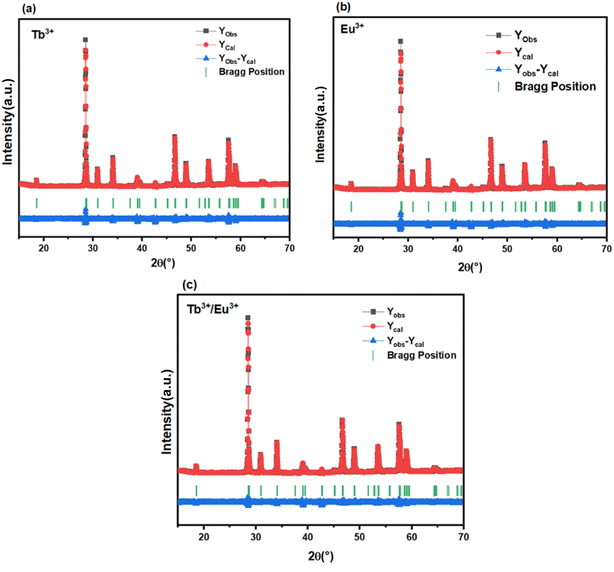 | ||
| Fig. 2 Refined powder X-ray diffraction pattern for Li2Y4(MoO4)7 samples (a) 40% Tb3+, (b) 5% Eu3+ and (c) 40% Tb3+/5% Eu3+. | ||
| Sample | α = β = γ | a (Å) | b (Å) | c (Å) | V (Å3) | Rwp (%) | χ2 |
|---|---|---|---|---|---|---|---|
| Li2Y4(MoO4)7:40% Tb3+ | 90° | 5.26361 | 5.26361 | 11.56449 | 320.400 | 6.60 | 2.2 |
| Li2Y4(MoO4)7:5% Eu3+ | 90° | 5.26353 | 5.26353 | 11.56436 | 320.387 | 5.00 | 2.4 |
| Li2Y4(MoO4)7:40% Tb3+/5% Eu3+ | 90° | 5.26361 | 5.26361 | 11.56458 | 320.404 | 9.4 | 2.1 |
3.2. Morphological characterization
The surface morphology and particle microstructure were examined by Scanning Electron Microscopy (SEM). Fig. 3 displays representative SEM micrographs of the undoped, singly doped, and co-doped LiYMoO samples.The micrographs reveal that the samples consist of agglomerated particles with irregular shapes and distinct boundaries, a characteristic feature of ceramic phosphors prepared via the high-temperature solid-state reaction route. The particle sizes generally range from 3 to 10 µm. Comparative analysis of the images indicates that the introduction of Eu3+ and Tb3+ dopants had no significant impact on the overall morphology or grain size distribution. The microstructure remains uniform across all compositions, suggesting that the doping concentration does not alter the grain growth kinetics during the sintering process. This micron-scale particle size is favorable for phosphor applications, as it typically enables easier coating and better light extraction efficiency in packaged devices.
4. Luminescence properties
4.1. Photoluminescence properties and spectral analysis
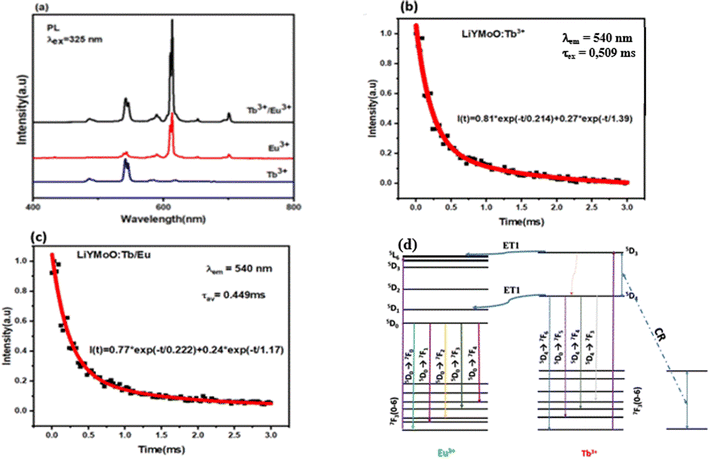
The room-temperature photoluminescence (PL) spectra of the LiYMoO phosphors doped with Tb3, Eu3+, and codoped with Tb3+/Eu3+ under near-UV excitation (325 nm) are plotted in Fig. 4(a). The excitation at 325 nm is well-suited for this system for two complementary reasons: (i) it falls within the charge-transfer absorption band of the [MoO4]2− polyanionic groups, which act as sensitizers that transfer energy to both Tb3+ and Eu3+ ions; and (ii) it overlaps with 4f–4f absorption transitions of both lanthanide ions, enabling their direct excitation.23The spectrum of the Tb3+ doped sample exhibits a series of sharp emission bands characteristic of the 5D4 to 7FJ (J=6,5,4,3) transitions. The peaks centered at 488, 542, 585, and 621 nm are ascribed to the 5D4 → 7F6, 5D4 → 7F5, 5D4 → 7F4, and 5D4 → 7F3 transitions, respectively. Among these, the green emission at 542 nm (5D4 → 7F5) is the most dominant. The Eu3+ doped sample displays a distinct spectral profile featuring multiple narrow peaks arising from the intra-configurational 4f–4f transitions of Eu3+. These are identified as the 5D0 → 7F0 (578 nm), 5D0 → 7F1 (592 nm), 5D0 → 7F2 (612 nm), 5D0 → 7F3 (653 nm), and 5D0 → 7F4 (701 nm) transitions. The hypersensitive electric dipole transition (5D0 → 7F2) at 612 nm exhibits the highest intensity, resulting in a strong red emission for the Tb3+/Eu3+ co-doped sample, the PL spectrum reveals a dual-emission character, containing the characteristic peaks of both Tb3+ (green) and Eu3+ (red) ions, notably, the simultaneous presence of these bands under an excitation wavelength that primarily targets the host and Tb3+ levels suggest the existence of an efficient energy transfer pathway within the matrix.
4.2. Energy transfer mechanism
Under typical conditions, Tb3+ ions can act as sensitizers for Eu3+ ions, enhancing the Tb3+ emission. In this study, the doping concentrations of Tb3+ and Eu3+ ions were fixed at 40% and 5%, respectively.Therefore, to carefully estimate the ET efficiency (ηET) from Tb3+ to Eu3+ ions we measured the lifetimes of Tb3+ ion and Tb3+/Eu3+ as illustrated in Fig. 4(b) and (c), respectively and this can be expressed by the equation of Paulose:19,20
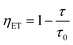 | (1) |
To elucidate the interaction between the dopants, the energy transfer (ET) mechanism in the Tb3+/Eu3+ co-doped system is schematically illustrated in Fig. 4(d). The mechanism relies on Tb3+ acting as an effective sensitizer for Eu3+. Upon excitation at 325 nm, Tb3+ ions are promoted to high-energy excited states and subsequently relax non-radiatively to the metastable 5D4 level. While a portion of this energy is released radiatively (green emission), a significant fraction is transferred non-radiatively to the neighboring Eu3+ ions via phonon-assisted energy transfer. This process is facilitated by the spectral overlap between the emission energy of the Tb3+ donor (5D4 → 7F{6,5}) and the absorption energy levels of the Eu3+ acceptor. Consequently, the Eu3+ ions are excited to the 5D0 state and decay radiatively to the ground state manifold (7FJ), intensifying the red emission (5D0 → 7F2). This synergistic interaction not only enhances the overall luminescence efficiency, but also enables the tuning of the emission color by modulating the Tb/Eu ratio.
4.3. Chromaticity coordinates (CIE)
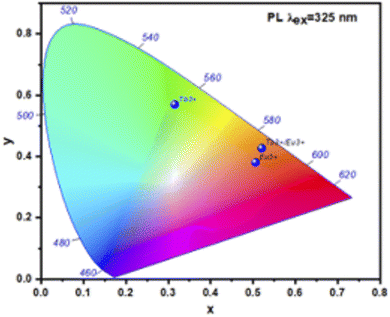
The influence of co-doping on the perceived emission color was quantified using Commission Internationale de l'Éclairage (CIE 1931) chromaticity coordinates, as depicted in Fig. 5.The chromaticity analysis reveals a clear evolution of the emission color depending on the dopant composition. In the Tb3+-doped sample, the coordinates are located in the deep green region of the CIE diagram, which is consistent with the dominant 5D4 → 7F5 transition centered at 542 nm. In contrast, the Eu3+-doped sample exhibits chromaticity coordinates situated in the reddish-orange region, mainly driven by the intense 5D0 → 7F2 electric-dipole transition at 612 nm. For the Tb3+/Eu3+ codoped sample, the chromaticity coordinates shift toward the orange to warm-white region. This shift represents the macroscopic manifestation of the microscopic energy transfer mechanism between Tb3+ and Eu3+ ions, where the effective mixing of the green emission from Tb3+ and the red emission from Eu3+ results in a tunable orange luminescence. By adjusting the [Tb3+]/[Eu3+] ratio, the emission color can be precisely controlled.
5. Temperature-dependent luminescence and optical thermometry
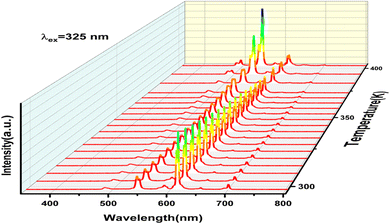
To assess the potential of the synthesized material for contactless temperature sensing, the photoluminescence properties of the Tb3+/Eu3+ co-doped LiYMoO phosphor were systematically investigated under 325 nm excitation over the temperature range of 300–403 K. The temperature-dependent PL spectra are illustrated in Fig. 6.The Eu3+ emission (5D0 → 7F2, 615 nm) undergoes progressive thermal quenching with increasing temperature, while the Tb3+ emission (5D4 → 7F5, 545 nm) exhibits a thermally activated enhancement in the range 300–403 K. This asymmetric and opposite thermal behavior of the two emission centers constitutes the physical basis enabling high-sensitivity LIR thermometry in this system, as it maximizes the rate of change of the intensity ratio with temperature.
The temperature-dependent luminescence behavior of the Tb3+/Eu3+ co-doped Li2Y4(MoO4)7 phosphor reveals an asymmetric thermal response between the two emitting centers. While the Eu3+ emission (5D0 → 7F2, 612 nm) undergoes progressive thermal quenching with increasing temperature consistent with thermally activated non-radiative relaxation via the host lattice phonons: the Tb3+ emission (5D4 → 7F5, 542 nm) exhibits a thermally activated enhancement in the range 300–403 K. This behavior arises from the interplay of three physical mechanisms: (i) thermally enhanced multi-phonon relaxation from the higher-lying 5D3 level of Tb3+ to the emitting 5D4 level, which increases 5D4 population at elevated temperatures; (ii) a temperature-dependent reduction in the Tb3+ → Eu3+ non-radiative energy transfer efficiency, which decreases with rising temperature due to the increased phonon-assisted back-transfer probability, thereby retaining more excitation energy in the Tb3+ 5D4 level; and (iii) the lower activation energy for non-radiative decay of Eu3+ relative to Tb3+ in the molybdate host, causing preferential quenching of Eu3+ emission. Taken together, these effects produce the observed asymmetric thermal behavior, where the Eu3+ emission weakens while the Tb3+ emission is temporarily enhanced, maximizing the rate of change of the LIR with temperature and thereby providing the physical basis for the high thermometric sensitivity of this system.
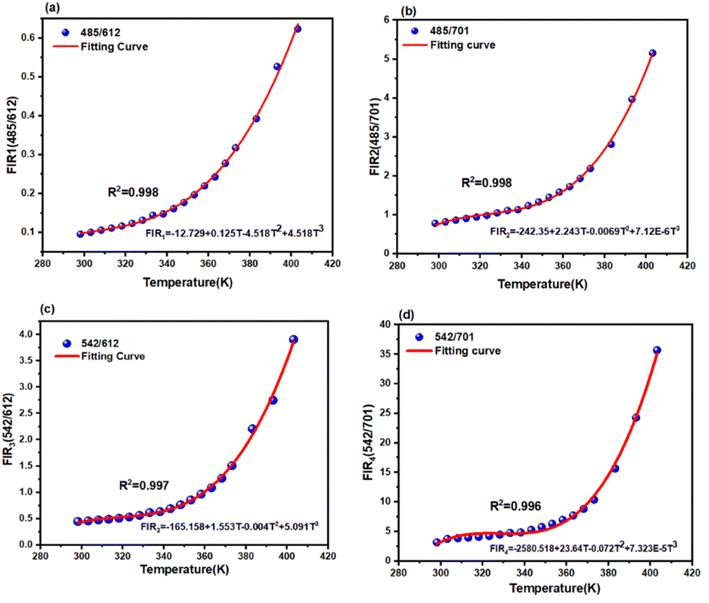
The diverse thermal response of the two emission centers (Tb3+ and Eu3+) allows for the application of the Fluorescence Intensity Ratio (FIR). This method mitigates measurement errors arising from fluctuations in excitation power or sample geometry. To identify the optimal thermometric signal, four distinct FIR modes were defined based on the ratio of integrated intensities of the non-thermally coupled levels of Tb3+ and Eu3+: FIR1 = I485/I612, FIR2 = I485/I701, FIR3 = I542/I612, FIR4 = I542/I701.
As shown in Fig. 7, all calculated FIR values exhibit a monotonic and non-linear evolution with temperature from 300 to 403 K.
To quantify this behavior, the experimental data for all FIR modes were fitted using a cubic polynomial function, given by:21,22
| FIR = A + B × T + C × T2 + D × T3 | (2) |
| Transitions | λ (nm) | Equation | Sr (max) (% K−1) |
|---|---|---|---|
| Tb3+:5D4 → 7F6, Eu3+:5D0 → 7F2 | 485/612 | −12.729 + 0.125T − 4.518T2 + 4.518T3 | 2.5 (400 K) |
| Tb3+:5D4 → 7F6, Eu3+:5D0 → 7F4 | 485/701 | −242.35 + 2.243T − 0.0069T2 + 7.12 × 10−6T3 | 2.7 (300 K), 3 (400 K) |
| Tb3+:5D4 → 7F5, Eu3+:5D0 → 7F2 | 542/612 | −165.158 + 1.553T − 0.004T2 + 5.091T3 | 3.4 (400 K) |
| Tb3+:5D4 → 7F5, Eu3+:5D0 → 7F4 | 542/701 | −2580.518 + 23.64T − 0.072T2 + 7.323 × 10−5T3 | 6.62 (300 K) |
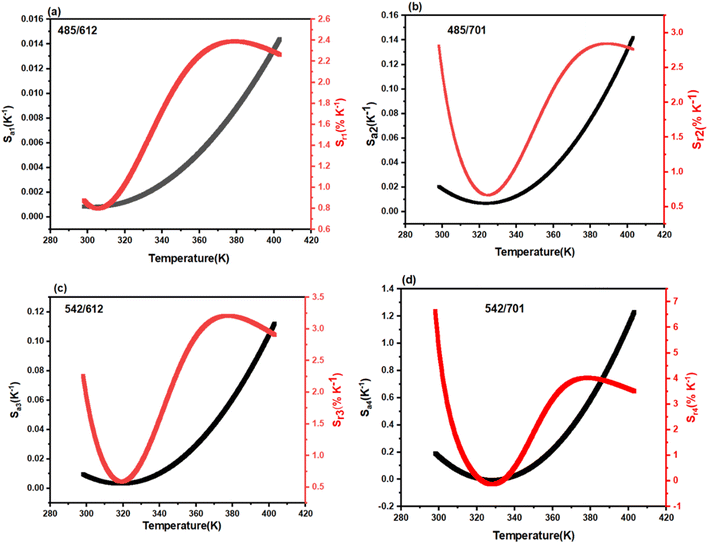
The performance of an optical thermometer is critically evaluated using two figures of merit: absolute sensitivity (Sa) and relative sensitivity (Sr). These parameters are defined as:23–25
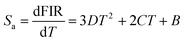 | (3) |
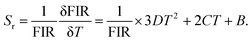 | (4) |
Fig. 8 depicts the temperature dependence of Sa and Sr for the investigated FIR modes. Among the four studied ratios, the FIR4(I542/I701) mode demonstrates the superior performance. It achieves a maximum absolute sensitivity (Sa) of 1.22 K−1 at 403 K and a remarkable relative sensitivity (Sr) of 6.5% K−1 at 300 K. This high relative sensitivity value significantly surpasses that of many previously reported rare-earth doped molybdate phosphors,26 positioning the Tb3+/Eu3+ co-doped LiYMoO system as a highly competitive candidate for high-precision optical thermometry in the physiological and industrial temperature ranges.
To properly evaluate the thermometric performance of the synthesized phosphors, a comparative analysis was conducted against other recently reported Tb3+/Eu3+ co-doped hosts. Table 3 summarizes the host matrix, temperature range, FIR mode, and maximum relative sensitivity (Sr) for these materials. As evidenced by the data, the LiYMoO:Tb3+/Eu3+ phosphor investigated in this work exhibits a remarkably high maximum relative sensitivity of 6.5% K−1 (300 K). This value significantly exceeds that of many established oxide, fluoride, and phosphate hosts, such as CaWO4 (4.01% K−1) and LiLaP4O12 (1.00% K−1). The superior sensitivity in our system can likely be attributed to the efficient phonon-assisted energy transfer between the Tb3+ and Eu3+ ions within the molybdate host. This confirms that the prepared material is a top-tier candidate for high-sensitivity optical thermometry applications. The temperature range of 300–403 K was chosen to ensure reliable and reproducible measurements while avoiding potential thermal degradation of the phosphors. Within this range, the FIR exhibits a clear and monotonic increase, allowing accurate evaluation of the temperature sensitivity. At higher temperatures, although the FIR trend suggests that sensitivity may continue to increase, thermal quenching and possible lattice distortions could reduce emission intensity and measurement stability. Therefore, the selected range provides a practical balance between sensitivity and the structural and optical stability of the phosphor material, ensuring accurate temperature sensing while maintaining reproducible luminescence performance.
| Host matrix | Temp. range (K) | FIR transition pair | Max Sr (% K−1) | Ref. |
|---|---|---|---|---|
| Li2Y4(MoO4)7 (LiYMoO) | 300–403 | Tb(5D4)/Eu(5D0) | 6.5 | This work |
| CaWO4 | 303–573 | Tb(5D4)/Eu(5D0) | 4.01 | 26 |
| LiLaP4O12 | 303–753 | Tb(5D4)/Eu(5D0) | 1.00 | 27 |
| NaCaPO4 (glass ceramic) | 298–573 | Tb(5D4)/Eu(5D0) | 0.66 | 28 |
| Sr8MgLa(PO4)7 | 298–523 | Tb(5D4)/Eu(5D0) | 0.16 | 29 |
| NaGd(MoO4)2 | 300–500 | Tb(5D4)/Eu(5D0) | 1.25 | 30 |
| Gd2O3 | 303–873 | Tb(5D4)/Eu(5D0) | 0.82 | 31 |
To evaluate the reliability of the temperature sensing performance, the thermometric parameters (FIR values) were systematically measured over repeated heating–cooling cycles between 300 K and 403 K, as illustrated in Fig. 9. The repeatability (R) of the sensor was calculated using the following expression:27
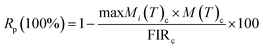 | (5) |
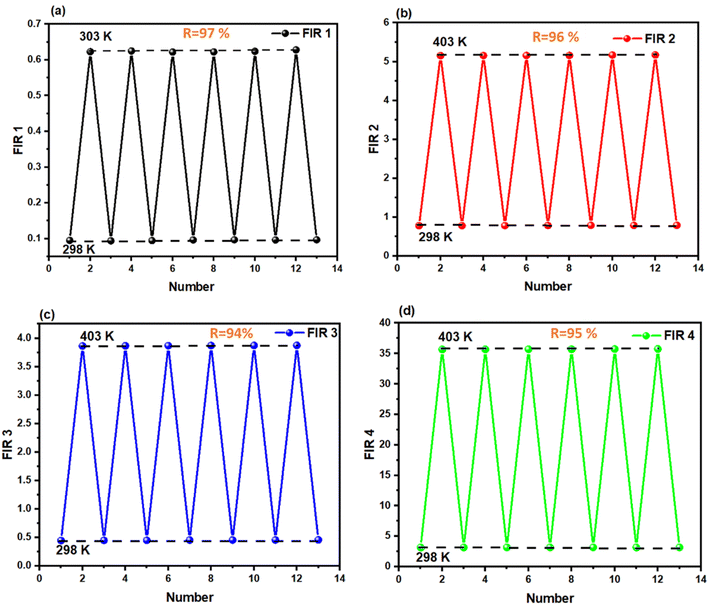 | ||
| Fig. 9 Repeatability assessment (R) (a) FIR1(I585/I612) (b) FIR2(I485/I701), (c) FIR3(I542/I612) and (d) FIR4(I542/I701) for LiYMoO phosphors. | ||
6. Conclusions
In summary, single-phase Li2Y4(MoO4)7 (LiYMoO) phosphors doped with 40% Tb3+, 5% Eu3+, and co-doped with 40% Tb3+/5% Eu3+ were successfully synthesized via a high-temperature solid-state reaction method. Structural and morphological characterizations confirmed that the incorporation of activator ions preserved the tetragonal crystal lattice and resulted in uniform microparticles suitable for luminescent applications. Under near-UV excitation (325 nm), the co-doped samples exhibited a tunable dual-emission profile, featuring the characteristic transitions of both Tb3+ (green) and Eu3+ (red) ions. The spectral evolution provided clear evidence of an efficient Tb3+ to Eu3+ non-radiative energy transfer mechanism. Crucially, the material demonstrated exceptional performance as a radiometric optical thermometer. By employing the Fluorescence Intensity Ratio (FIR) technique based on the non-thermally coupled transitions of Tb3+ (5D4 → 7F5 at 542 nm) and Eu3+ (5D0 → 7F4 at 701 nm), a reliable thermal response was established over the working range of 300–403 K. The sensor achieved a superior maximum relative sensitivity (Sr) of 6.5% K−1 at 300 K, significantly outperforming many comparable molybdate and phosphate hosts. These findings position the Tb3+/Eu3+ codoped Li2Y4(MoO4)7 system as a highly promising candidate for high-sensitivity contactless temperature sensing devices.Conflicts of interest
There are no conflicts to declare.Data availability
All data underlying the results are available as part of the article and no additional source data are required.References
- F. Ayachi, K. Saidi, K. Soler-Carracedo, M. Dammak and I. R. Martín, J. Alloys Compd., 2023, 961, 171146 CrossRef CAS.
- F. Ayachi, K. Saidi, W. Chaabani and M. Dammak, J. Lumin., 2021, 240, 118451 CrossRef CAS.
- B. Koudsi, H. Refai and J. J. Sluss, J. Disp. Technol., 2015, 11, 266–272 Search PubMed.
- X. Zhang, J. Wang, L. Huang, F. Pan, Y. Chen, B. Lei, M. Peng and M. Wu, ACS Appl. Mater. Interfaces, 2015, 7, 10044–10054 CrossRef CAS PubMed.
- H. M. Yang, J. X. Shi and M. L. Gong, J. Solid State Chem., 2005, 178, 917–920 CrossRef CAS.
- E. Castellano-Hernández, X. Han, M. Rico, L. Roso, C. Cascales and C. Zaldo, Opt. Express, 2015, 23, 11135–11140 CrossRef PubMed.
- B. Kukliński, D. Wileńska, S. Mahlik, K. Szczodrowski, M. Grinberg and A. M. Kłonkowski, Opt. Mater., 2014, 36, 633–638 CrossRef.
- F. Ayachi, K. Saidi, M. Dammak, I. Mediavilla and J. Jiménez, RSC Adv., 2025, 15, 655–664 RSC.
- Z. E. A. A. Taleb, I. Kachou, K. Saidi, M. Dammak, I. Mediavilla and J. Jiménez, Mater. Adv., 2025, 6, 2385–2396 RSC.
- X. Zhang, L. Zhou, J. Shi and M. Gong, Mater. Lett., 2014, 137, 32–35 CrossRef CAS.
- Z. Yahiaoui, M. A. Hassairi, M. Dammak, E. Cavalli and F. Mezzadri, J. Lumin., 2018, 194, 96–101 CrossRef CAS.
- F. Ayachi, K. Saidi, M. Dammak, J. J. Carvajal and M. C. Pujol, RSC Adv., 2024, 14, 13494–13504 RSC.
- K. Saidi, M. Dammak, K. Soler-Carracedo and I. R. Martín, Dalton Trans., 2022, 51, 5108–5117 RSC.
- K. Saidi and M. Dammak, J. Solid State Chem., 2021, 300, 122214 CrossRef CAS.
- N. Ben Amar, K. Saidi, C. Hernández-Álvarez, M. Dammak and I. R. Martín, ACS Appl. Nano Mater., 2025, 8, 17133–17143 CrossRef CAS PubMed.
- I. Kachou, K. Saidi, R. Salhi and M. Dammak, RSC Adv., 2022, 12, 7529–7539 RSC.
- K. Saidi and M. Dammak, RSC Adv., 2020, 10, 21867–21875 RSC.
- C. Harriswangler, J. C. Frías, M. T. Albelda, L. Valencia, E. García-España, D. Esteban-Gómez and C. Platas-Iglesias, Inorg. Chem., 2023, 62, 17030–17040 CrossRef CAS PubMed.
- M. Fan, L. Zhao, X. Jin, W. Sun, W. Qi and Y. Li, Anal. Chim. Acta, 2022, 1221, 340026 CrossRef CAS PubMed.
- B. Wang, Q. Ren, O. Hai and X. Wu, RSC Adv., 2017, 7, 15222–15227 RSC.
- Q. Han, H. Hao, J. Yang, Z. Sun, J. Sun, Y. Song, Y. Wang and X. Zhang, J. Alloys Compd., 2019, 786, 770–778 CrossRef CAS.
- S. Bhushan and M. V. Chukichev, J. Mater. Sci. Lett., 1988, 7, 319–321 CrossRef CAS.
- Y. Bahrouni, I. Kachou, K. Saidi, C. Hernández-Álvarez, M. Dammak and I. R. Martín, RSC Adv., 2026, 16, 4452–4460 RSC.
- M. Fhoula, M. Khitouni and M. Dammak, RSC Adv., 2024, 14, 39373–39380 RSC.
- Y. Jiang, Y. Tong, S. Chen, W. Zhang, F. Hu, R. Wei and H. Guo, Chem. Eng. J., 2021, 413, 127470 CrossRef CAS.
- M. D. Dramićanin, J. Appl. Phys., 2020, 128, 040902 CrossRef.
- Y. Bahrouni, I. Kachou, K. Saidi, C. Hernández-Álvarez, M. Dammak and I. R. Martín, J. Alloys Compd., 2026, 1056, 186657 CrossRef.
- Y. Zhang, W. Gong, J. Yu, Z. Cheng and G. Ning, RSC Adv., 2016, 6, 30886–30894 RSC.
- L. Marciniak and A. Bednarkiewicz, Phys. Chem. Chem. Phys., 2016, 18, 15584–15592 RSC.
- L.-Q. Yao, G.-H. Chen, T. Yang, S.-C. Cui, Z.-C. Li and Y. Yang, Ceram. Int., 2016, 42, 13086–13090 CrossRef CAS.
- W. Xia, L. Li, P. Yang, F. Ling, Y. Wang, Z. Cao, S. Jiang, G. Xiang, X. Zhou and Y. Hua, J. Lumin., 2021, 239, 118383 CrossRef CAS.
- L. Peng, Q. Meng and W. Sun, Ceram. Int., 2019, 45, 20656–20663 CrossRef CAS.
- I. E. Kolesnikov, D. V. Mamonova, M. A. Kurochkin, V. A. Medvedev, E. V. Borisov and E. Y. Kolesnikov, Ceram. Int., 2023, 49, 6899–6905 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |