Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A2Se2C2 (A = Na–Cs): crystalline compounds featuring −Se–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/h2_char_e002.gif) C–Se− dianions with short-range structural disorder
C–Se− dianions with short-range structural disorder
Tim Mattick ,
Marc Hetzert and
Uwe Ruschewitz
,
Marc Hetzert and
Uwe Ruschewitz *
*
Department of Chemistry, Greinstraße 6, 50939 Köln, Germany. E-mail: uwe.ruschewitz@uni-koeln.de; Fax: +49 221 470 3933; Tel: +49 221 470 3285
First published on 5th May 2026
Abstract
Crystalline powders of A2Se2C2 (A = Na–Cs) were prepared either by reacting A2C2 with two equivalents of elemental (grey) selenium in liquid ammonia or by thermally decomposing ASeC2H at 200 °C under dynamic vacuum. IR and Raman spectra unambiguously confirm the presence of −Se–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Se− anions. X-ray powder diffraction patterns exhibit only a few very broad reflections, consistent with a primitive cubic arrangement of the alkali metal cations. The anions occupy the interstitial voids between these cations and display local orientational disorder. Only through a combined approach employing Rietveld and pair distribution function (PDF) analyses of synchrotron powder diffraction data, supplemented by density functional theory (DFT) calculations, it was possible to establish a consistent structural model for the anion disorder.
C–Se− anions. X-ray powder diffraction patterns exhibit only a few very broad reflections, consistent with a primitive cubic arrangement of the alkali metal cations. The anions occupy the interstitial voids between these cations and display local orientational disorder. Only through a combined approach employing Rietveld and pair distribution function (PDF) analyses of synchrotron powder diffraction data, supplemented by density functional theory (DFT) calculations, it was possible to establish a consistent structural model for the anion disorder.
Introduction
In his pioneering work more than a century ago, Moissan reported on the synthesis of alkali metal acetylides with the composition A2C2 (A = Li–Cs).1,2 Several decades later, based on X-ray powder diffraction data, the crystal structures of these acetylides were investigated, confirming the existence of C22− anions, as postulated by Moissan.3–5 Notably, in all these crystal structures, the C22− dumbbells are ordered at ambient conditions. Only at higher temperatures, a rotational disorder of the acetylide anions was observed for Li2C2 (>770 K),6 Na2C2 (>580 K),7 and K2C2 (>420 K).7In 1963, the first report of a ternary acetylide, namely KAgC2, was published.8 Its crystal structure revealed the existence of linear 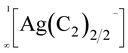 chains, which are separated by K+ cations.9 Similar
chains, which are separated by K+ cations.9 Similar 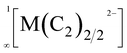 chains were found in compounds with the composition A2MC2 (A = Na–Cs; M = Pd, Pt).10–12 All these chain-type structures are not unexpected, as Ag(I) as well as Pd(0) and Pt(0) feature a d10 electron configuration, for which a linear coordination, as found in these chains, is very common.13,14
chains were found in compounds with the composition A2MC2 (A = Na–Cs; M = Pd, Pt).10–12 All these chain-type structures are not unexpected, as Ag(I) as well as Pd(0) and Pt(0) feature a d10 electron configuration, for which a linear coordination, as found in these chains, is very common.13,14
Therefore, it was very surprising, when back in 2015 Németh et al. proposed similar linear chain-type structures 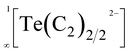 within the acetylides Li2TeC2 and Na2TeC2.15 Te(0), which should exist in these compounds, is not known to prefer a linear coordination. Quite the contrary, in elemental tellurium a tilted arrangement with Te–Te–Te angles of approx. 102.3° was reported.16 Nonetheless, the ternary tellurium acetylides Li2TeC2 and Na2TeC2 seem to be intriguing materials with several potential applications as photocathodes or novel electrode materials for batteries, as predicted on the basis of density functional theory (DFT) calculations.17 They inspired us to have a closer look at acetylides with the main group elements sulphur, selenium, and tellurium. In these works, we were able to synthesise and characterise the elusive and mainly unprecedented anions −S–C
within the acetylides Li2TeC2 and Na2TeC2.15 Te(0), which should exist in these compounds, is not known to prefer a linear coordination. Quite the contrary, in elemental tellurium a tilted arrangement with Te–Te–Te angles of approx. 102.3° was reported.16 Nonetheless, the ternary tellurium acetylides Li2TeC2 and Na2TeC2 seem to be intriguing materials with several potential applications as photocathodes or novel electrode materials for batteries, as predicted on the basis of density functional theory (DFT) calculations.17 They inspired us to have a closer look at acetylides with the main group elements sulphur, selenium, and tellurium. In these works, we were able to synthesise and characterise the elusive and mainly unprecedented anions −S–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–H,18 −Se–C
C–H,18 −Se–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–H,19,20 and −Se–C
C–H,19,20 and −Se–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Se−.21 However, we have never been able to observe a chain-type structure as published by Németh et al.15 With regard to the present work, it is important to note that the above-mentioned anions typically occur in an ordered arrangement in their crystal structures. Only in NaSC2H18 and NaSeC2H,20 the anions show an orientational disorder over two positions. Also at low temperatures down to 100 K, no ordering was observed.
C–Se−.21 However, we have never been able to observe a chain-type structure as published by Németh et al.15 With regard to the present work, it is important to note that the above-mentioned anions typically occur in an ordered arrangement in their crystal structures. Only in NaSC2H18 and NaSeC2H,20 the anions show an orientational disorder over two positions. Also at low temperatures down to 100 K, no ordering was observed.
One obvious drawback of the work by Németh et al. is the very low crystallinity of Li2TeC2 and Na2TeC2, resulting in X-ray powder diffraction patterns with only very few and very broad reflections, from which their crystal structures had to be solved and refined (Li2TeC2: P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1; Na2TeC2: I4/mmm).15 In our work on selenium acetylides, we observed very similar diffraction patterns after heating ASeC2H (A = Na–Cs) in a dynamic vacuum. In contrast to the work of Nemeth et al.,15 we were able to index these patterns in small cubic unit cells.19 As the release of acetylene (C2H2) was proven during heating, we postulated a reaction according to20
m1; Na2TeC2: I4/mmm).15 In our work on selenium acetylides, we observed very similar diffraction patterns after heating ASeC2H (A = Na–Cs) in a dynamic vacuum. In contrast to the work of Nemeth et al.,15 we were able to index these patterns in small cubic unit cells.19 As the release of acetylene (C2H2) was proven during heating, we postulated a reaction according to20
This assumption would challenge the findings of Németh et al. At this point, it is notable that their DFT calculations on Li2TeC2 predicted a much smaller unit cell with an inverted c/a ratio, already pointing out possible inaccuracies in their structural model.15 In the following, we will give a clear proof that the cubic selenium compounds we found in our investigations19 have the composition A2Se2C2 and contain −Se–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Se− dianions, which have been observed before in Li2Se2C2·2NH3.21 Due to orientational disorder of these anions on a local scale, the reflections are broadened and PDF (pair distribution function) analysis accompanied by DFT calculations is needed to develop a reasonable structural model for this disorder. In our view, this approach provides an engaging example of how reliable structural information can be obtained even from a poorly crystalline sample by combining PDF analyses and DFT calculations with conventional spectroscopic studies and Rietveld refinements.
C–Se− dianions, which have been observed before in Li2Se2C2·2NH3.21 Due to orientational disorder of these anions on a local scale, the reflections are broadened and PDF (pair distribution function) analysis accompanied by DFT calculations is needed to develop a reasonable structural model for this disorder. In our view, this approach provides an engaging example of how reliable structural information can be obtained even from a poorly crystalline sample by combining PDF analyses and DFT calculations with conventional spectroscopic studies and Rietveld refinements.
Experimental
Synthesis
ASeC2H (A = Na–Cs) was synthesised according to a previously published method.19,20 The resulting compound was transferred to a Schlenk tube and heated at 200 °C for 1 h under a dynamic vacuum.Alternatively, a mixture of A2C2 (A = Na–Cs) and two equivalents of grey selenium was suspended in liquid ammonia. The suspension was stirred for 1 h at −78 °C, after which the ammonia was evaporated at room temperature.
In both cases, the product was obtained in the form of a brown powder. The SI provides more details regarding the syntheses.
Vibrational spectroscopy
IR spectra were measured using a Bruker ALPHA FT-IR spectrometer. Measurements were carried out inside a glovebox under inert conditions (argon atmosphere).Raman analysis was performed using a Renishaw inVia Qontor® Raman Microscope. Samples were transferred into glass capillaries sealed under argon before the measurements. Spectra were measured using a laser wavelength of 457 nm and a Centrus 05 TJ CCD detector.
All vibrational spectra were measured on samples synthesised in liquid ammonia.
SEM/EDX
The morphology and elemental distribution were analyzed using a Zeiss Sigma 300 VP Rise field emission SEM with an integrated Oxford Instruments Xplore 30 EDX. SEM data was measured at 2 kV, EDX data was measured at 15 kV. Samples were transferred onto a carbon grid under inert conditions (argon atmosphere) prior to the measurement.Powder diffraction
High-resolution synchrotron powder patterns were recorded at the DELTA facility (Dortmund, Germany), beamline BL9.22 Samples were measured at ambient conditions (25 keV, Dectris PILATUS 100K detector, glass capillaries ⌀ = 0.7 mm sealed under argon). All samples were synthesised in liquid ammonia.Total scattering data was collected at the German Electron Synchrotron (DESY, Hamburg (Germany), beamline P21.1).23 Samples were measured under ambient conditions (101.35(10) keV, PerkinElmer XRD1621 detector, glass capillaries ⌀ = 1.0 mm sealed under argon). The 2D data was integrated using pyFAI,24 pair distribution functions were calculated using PDFgetX3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 on a Nyquist-Shannon grid26 with Qmax = 22.9 Å−1. Na2Se2C2 and K2Se2C2 were synthesised in liquid ammonia, while Rb2Se2C2 and Cs2Se2C2 were synthesised by heating RbSeC2H and CsSeC2H, respectively. This ensured phase purity for all samples.
25 on a Nyquist-Shannon grid26 with Qmax = 22.9 Å−1. Na2Se2C2 and K2Se2C2 were synthesised in liquid ammonia, while Rb2Se2C2 and Cs2Se2C2 were synthesised by heating RbSeC2H and CsSeC2H, respectively. This ensured phase purity for all samples.
Simultaneous Rietveld and pair distribution function refinements were performed using TOPAS-Academic Version 6.27–29 To obtain a smooth convergence, the C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C distance was soft restrained to 1.20(2) Å and the Se–C distance to 1.85(2) Å, as observed in KSeCN.30 Alkali metal cations were fixed to a primitive cubic lattice and Se and C atoms were restrained so that they could only move along the Se⋯Se axis in an anion.
C distance was soft restrained to 1.20(2) Å and the Se–C distance to 1.85(2) Å, as observed in KSeCN.30 Alkali metal cations were fixed to a primitive cubic lattice and Se and C atoms were restrained so that they could only move along the Se⋯Se axis in an anion.
Quantum chemical calculations
Solid state DFT calculations were performed using Quantum ESPRESSO.31,32 For total energy calculations and geometry optimizations, the PBEsol functional,33 pseudopotentials from the SSSP PBEsol Precision v1.3.0 library34–38 and D3 corrections39 were employed. For calculations of spectroscopic data, the LDA functional40 was used in combination with ONCVPSP v0.4.1 pseudopotentials41 from PseudoDojo.37Gas phase DFT calculations were performed using ORCA,42–47 the B3LYP hybrid functional,48–51 ma-def2-QZVPP52,53 basis sets and D4 corrections.54–56 Bonds within the calculated molecules were analysed using JANPA.57,58 Further details regarding these calculations are available in the SI.
Results and discussion
Structural characterisation
Although the PXRD patterns of A2Se2C2 (A = Na–Cs) can be indexed in small, primitive cubic unit cells,19,20 a complete ordering of the linear −Se–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Se− anions within these small unit cells is not possible. Similar behaviour has already been observed for CsSeH, which appears to crystallise in the CsCl structure type with obviously rotationally disordered SeH− anions, according to the analysis of Bragg diffraction data.59 For this reason, a structural model of orientationally disordered anions in the a, b, and c directions was envisaged for the compounds A2Se2C2. For the Rietveld and PDF fits, a 4 × 4 × 4 supercell of the primitive cubic unit cell containing statistically disordered anions (Fig. 1) was constructed for each compound. Statistical, orientational disorder was simulated using the Metropolis–Hastings algorithm60 with local edge flips in a Markov chain Monte Carlo simulation61 comprising 10
C–Se− anions within these small unit cells is not possible. Similar behaviour has already been observed for CsSeH, which appears to crystallise in the CsCl structure type with obviously rotationally disordered SeH− anions, according to the analysis of Bragg diffraction data.59 For this reason, a structural model of orientationally disordered anions in the a, b, and c directions was envisaged for the compounds A2Se2C2. For the Rietveld and PDF fits, a 4 × 4 × 4 supercell of the primitive cubic unit cell containing statistically disordered anions (Fig. 1) was constructed for each compound. Statistical, orientational disorder was simulated using the Metropolis–Hastings algorithm60 with local edge flips in a Markov chain Monte Carlo simulation61 comprising 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 steps.
000 steps.
 | ||
Fig. 1 4 × 4 × 4 supercell of K2Se2C2 with disordered −Se–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Se− anions. Colour code: K: light grey, C: dark grey, Se: red. C–Se− anions. Colour code: K: light grey, C: dark grey, Se: red. | ||
The Rietveld fits of all A2Se2C2 compounds are in good agreement with the PXRD measurements (Fig. 2; S1, S3, S5, SI). The Rietveld fit of Rb2Se2C2 shows small amounts of RbSeC2H19 impurities. This suggests an analogous equilibrium for the anion couple SeC2H−/Se2C22− as found for C2H−/C22− in liquid ammonia.62
 | ||
| Fig. 2 Rietveld fit of K2Se2C2 in a 4 × 4 × 4 supercell (Fig. 1) with disordered anions (λ = 0.496 Å). | ||
The PDF fits of the A2Se2C2 compounds also show a good agreement with the experimental PDFs (Fig. 3; Fig. S2, S4, S6, SI). The only major discrepancies appear in the very low r-region, where the experimental PDFs are significantly higher than the fits. This is most likely a result of termination ripples63 caused by the cutoff at Qmax = 22.9 Å−1 as well as some noise in F(Q) at high Q, and is therefore not indicative of any structural features.
 | ||
| Fig. 3 PDF fit of K2Se2C2 in a 4 × 4 × 4 supercell (Fig. 1) with disordered anions (Qmax = 22.9 Å−1). | ||
Each Se2C22− anion bridges two cubes spanned by the alkali metal cations (Fig. 4 right). The acetylidic C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C dumbbell is orientated perpendicular to the square face that connects the two cubes and is coordinated by four cations in a side-on fashion (Fig. 4 left).
C dumbbell is orientated perpendicular to the square face that connects the two cubes and is coordinated by four cations in a side-on fashion (Fig. 4 left).
 | ||
Fig. 4 Coordination sphere of the −Se–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Se− anion in K2Se2C2. Left: side-on coordination of the C C–Se− anion in K2Se2C2. Left: side-on coordination of the C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C unit. Right: Se sitting in the cubic K8 void. Colour code: K: light grey, C: dark grey, Se: red. C unit. Right: Se sitting in the cubic K8 void. Colour code: K: light grey, C: dark grey, Se: red. | ||
The Se atoms sit within the cubic voids spanned by the alkali metal cations. They are slightly shifted out of the cube's centre along the Se–C axis (Fig. 4 right). The distortion from the coordination within a platonic cube is small and decreases with a larger cation radius and, consequently, larger unit cell parameters (Table S2, SI), as would be expected. The intraanionic Se⋯Se distance is 4.90(3) Å and the alkali metal distance (i.e. the lattice parameter of the primitive unit cell) approaches this value for the larger alkali metals. The alkali metal-selenium distances are consistently longer than those found in reference compounds (Table 1), indicating weaker interactions.
To assess the hypothesis of disordered −Se–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Se− anions, a structural model with ordered anions was also fitted to the data. This model belongs to the tetragonal space group type P4/mmm (no. 123) and contains two crystallographically independent alkali metal cations, with the anion aligned along the c axis (Fig. S7, SI). However, with the exception of the Rb compound, this model consistently provides a poorer fit to the data (Table 2). The difference in Rwp values for Rb2Se2C2 is negligible and does not prefer any of the possible models significantly. However, a model of locally disordered anions also in Rb2Se2C2 is supported by DFT calculations and Raman spectroscopy, as will be demonstrated in the following.
C–Se− anions, a structural model with ordered anions was also fitted to the data. This model belongs to the tetragonal space group type P4/mmm (no. 123) and contains two crystallographically independent alkali metal cations, with the anion aligned along the c axis (Fig. S7, SI). However, with the exception of the Rb compound, this model consistently provides a poorer fit to the data (Table 2). The difference in Rwp values for Rb2Se2C2 is negligible and does not prefer any of the possible models significantly. However, a model of locally disordered anions also in Rb2Se2C2 is supported by DFT calculations and Raman spectroscopy, as will be demonstrated in the following.
| Rwp 4 × 4 × 4 (%) | Rwp P4/mmm (%) | |
|---|---|---|
| Na2Se2C2 | 12.35 | 16.06 |
| K2Se2C2 | 8.46 | 12.13 |
| Rb2Se2C2 | 6.50 | 5.84 |
| Cs2Se2C2 | 6.94 | 8.65 |
The tetragonal structure model with ordered anions shows clear deviations in reflection intensities as well as additional reflections in the Rietveld fit. For example, there are additional reflections for K2Se2C2 between 10° and 13° (Fig. S9, SI). These errors are significantly smaller in the Rietveld fit of the 4 × 4 × 4 supercell containing disordered anions (Fig. S8, SI). The remaining discrepancies are probably due to the finite size of the supercell. However, refinement of even larger supercells was not computationally feasible.
This evident orientational disorder of the anions may be rationalised by the resulting increase of the intermolecular anion–anion distances. If the anions were ordered, the partially negatively charged Se atoms would approach each other along the c axis (Fig. 5 left). For K2Se2C2 e.g., this would result in a Se⋯Se distance of 3.6272(9) Å. Notably, this is less than twice the van der Waals radius of selenium, which is 1.9 Å.66 If the anions are orientationally disordered, only some anions directly point at each other like in the model with P4/mmm symmetry (Fig. 5 left), while most of them evade each other with respect to short Se⋯Se distances (Fig. 5 right). For the K2Se2C2 supercell (Fig. 1), this results in an average Se⋯Se distance of 3.810(4) Å with a mode (i.e. the most common value) of 3.9410(9) Å. This now is more than twice the van der Waals radius of selenium.
 | ||
| Fig. 5 Interanionic distance between Se atoms in K2Se2C2. Left: ordered anions (P4/mmm). Right: disordered anions (4 × 4 × 4 supercell). Colour code: K: light grey, C: dark grey, Se: red. | ||
SEM/EDX
The EDX (energy-dispersive X-ray spectroscopy) spectrum of K2Se2C2 demonstrates a K![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Se ratio of 1
Se ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 within experimental errors (Fig. 6), which further indicates that its composition is A2Se2C2. The C signals are stronger than expected as the measurement was carried out on a carbon grid. The presence of small amounts of oxygen in the sample is probably due to a slight oxidation during transfer into the device. SEM (scanning electron microscopy) images of the sample demonstrate the cubic morphology of K2Se2C2 (Fig. S10, SI).
1 within experimental errors (Fig. 6), which further indicates that its composition is A2Se2C2. The C signals are stronger than expected as the measurement was carried out on a carbon grid. The presence of small amounts of oxygen in the sample is probably due to a slight oxidation during transfer into the device. SEM (scanning electron microscopy) images of the sample demonstrate the cubic morphology of K2Se2C2 (Fig. S10, SI).
Solid state DFT calculations
To further analyse the structural motif of the A2Se2C2 compounds, the disordered model in a 4 × 4 × 4 supercell and the ordered model with P4/mmm symmetry were optimised using DFT. The resulting formation enthalpies show that all structures are thermodynamically stable compared to the elements and that the disorder of the anions leads to a favourable energy gain (Fig. 7). This effect decreases from Na to Cs, which further hints at the increase of Se⋯Se distances as the driving force for the orientational disorder. As the alkali metal radii and subsequently the lattice parameters increase, the Se⋯Se distances increase as well. As a result, even if the anions point at each other in the ordered P4/mmm model, the interanionic Se⋯Se distances are larger than twice the van der Waals radius of selenium for the larger alkali metal cations. | ||
| Fig. 7 Formation enthalpies per atom for A2Se2C2 compounds after optimisation in a 4 × 4 × 4 supercell and in P4/mmm symmetry. | ||
Furthermore, the lattice parameters of the orientationally disordered model of A2Se2C2 barely decreased during optimisation (Table S3–S6, SI), which is common for these calculations.67 The unit cell angles all stayed very close to 90°. In contrast, the c parameter of the P4/mmm model changed significantly during optimisation, leading to large distortions of the pseudo-cubic cell (Table S3–S6, SI).
Vibrational spectroscopy
The IR spectra clearly show modes from 740 cm−1 to 768 cm−1 for all A2Se2C2 (A = Na–Cs) compounds (Fig. S11, SI). These can be attributed to the Se–C stretching vibration, which can be reproduced using solid-state density functional perturbation theory (DFPT)68 (Table 3). The absence of modes characteristic of ammonia clearly shows that, in contrast to lithium,21 no ammoniate is formed for A = Na–Cs. As already observed in the PXRD data, weak modes that can be attributed to minor impurities of RbSeC2H and CsSeC2H, respectively, are evident in the IR spectra of Rb2Se2C2 and Cs2Se2C2.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 21 are given for reference
21 are given for reference
νSe–C,exp![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
νSe–C,calc | νC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C,exp C,exp![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
νC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C,calc C,calc |
|
|---|---|---|---|---|
| Na2Se2C2 | 754 cm−1 | 754.24 cm−1 | 1977 cm−1 | 1970.88 cm−1 |
| K2Se2C2 | 768 cm−1 | 765.32 cm−1 | 2004 cm−1 | 1998.04 cm−1 |
| Rb2Se2C2 | 767 cm−1 | 761.17 cm−1 | 2009 cm−1 | 2004.32 cm−1 |
| Cs2Se2C2 | 740 cm−1 | 748.95 cm−1 | 2014 cm−1 | 2007.10 cm−1 |
| Li2Se2C2·2NH3 | 756 cm−1 | 760 cm−1 | 2059 cm−1 | 2057 cm−1 |
The Raman spectra show modes that can be assigned to the C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C stretching vibration. The modes for K2Se2C2, Rb2Se2C2 and Cs2Se2C2 are all very similar, with wavenumbers ranging from 2004 cm−1 to 2014 cm−1, whereas the mode for Na2Se2C2 is shifted to 1977 cm−1 (Fig. 8). These results could be reproduced in solid-state DFPT68 calculations (Table 3). All Raman spectra show a shoulder (A = Na) or a second signal at lower wavenumbers (A = K, Rb, Cs). These are caused by the two different possible interanionic arrangements (Fig. 5). As the disordered structure model contains both motifs (vide supra), the C
C stretching vibration. The modes for K2Se2C2, Rb2Se2C2 and Cs2Se2C2 are all very similar, with wavenumbers ranging from 2004 cm−1 to 2014 cm−1, whereas the mode for Na2Se2C2 is shifted to 1977 cm−1 (Fig. 8). These results could be reproduced in solid-state DFPT68 calculations (Table 3). All Raman spectra show a shoulder (A = Na) or a second signal at lower wavenumbers (A = K, Rb, Cs). These are caused by the two different possible interanionic arrangements (Fig. 5). As the disordered structure model contains both motifs (vide supra), the C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C stretching vibration leads either to the anions swinging directly into each other (Fig. 5 left) or swinging side by side (Fig. 5 right), resulting in slightly different Raman modes. Both the IR and the Raman modes of the −Se–C
C stretching vibration leads either to the anions swinging directly into each other (Fig. 5 left) or swinging side by side (Fig. 5 right), resulting in slightly different Raman modes. Both the IR and the Raman modes of the −Se–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Se− anion are consistent with the modes found for Li2Se2C2·2NH3 (Table 3).21
C–Se− anion are consistent with the modes found for Li2Se2C2·2NH3 (Table 3).21
 | ||
Fig. 8 Raman spectra of the A2Se2C2 (A = Na–Cs) compounds. Wavenumbers of the C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C stretching vibration are given. C stretching vibration are given. | ||
Gas phase DFT calculations reveal a Raman mode of 2080.97 cm−1 for the formal −Se–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Se− dianion and a mode of 2013.93 cm−1 for the formal .Se–C
C–Se− dianion and a mode of 2013.93 cm−1 for the formal .Se–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Se− radical monoanion. This difference might explain the shifted Raman mode for Na2Se2C2. As Na (5.139 eV) has a significantly higher first ionization energy than K (4.341 eV), Rb (4.177 eV) and Cs (3.894 eV),69 the electronic structure of the anion in Na2Se2C2 will be closer to a formal monoanion, whereas the similar first ionization energies of K, Rb and Cs lead to comparable Raman shifts for these compounds. This is most likely caused by a weaker C
C–Se− radical monoanion. This difference might explain the shifted Raman mode for Na2Se2C2. As Na (5.139 eV) has a significantly higher first ionization energy than K (4.341 eV), Rb (4.177 eV) and Cs (3.894 eV),69 the electronic structure of the anion in Na2Se2C2 will be closer to a formal monoanion, whereas the similar first ionization energies of K, Rb and Cs lead to comparable Raman shifts for these compounds. This is most likely caused by a weaker C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C bond in the slightly electron deficient monoanion compared to the dianion. This weaker bond in Se2C2− compared to Se2C22− is corroborated by CLPO58 bond analysis. This analysis indicates a bond order of 2.78 for the dianion, but only 2.58 for the monoanion, as the additional electron of the dianion mainly occupies the bonding π-orbital of the C2 unit.
C bond in the slightly electron deficient monoanion compared to the dianion. This weaker bond in Se2C2− compared to Se2C22− is corroborated by CLPO58 bond analysis. This analysis indicates a bond order of 2.78 for the dianion, but only 2.58 for the monoanion, as the additional electron of the dianion mainly occupies the bonding π-orbital of the C2 unit.
Conclusions
We were able to synthesise polycrystalline A2Se2C2 (A = Na–Cs) by either reacting A2C2 with grey selenium in liquid ammonia, or by heating ASeC2H. Their crystal structures were modelled and solved from synchrotron powder data and pair distribution functions. The resulting structure features a primitive cubic lattice of alkali metal cations with Se2C22− anions positioned between them. The presence of the anions is corroborated by IR and Raman spectra. The anions are orientationally disordered along the crystallographic a, b, and c axes. Models with ordered Se2C22− anions resulted in poorer fits of PXRD and PDF data, and reduced stability according to DFT calculations.As a continuation of this work, we plan to investigate the corresponding tellurium-containing compounds. As previously demonstrated,19 we were able to synthesise these compounds from A2C2 (A = Na, K, Rb) and elemental tellurium, which show similar PXRD patterns as those presented in this work. The resulting patterns could be indexed in small primitive cubic unit cells, e.g. a = 4.144 Å for the sodium compound.19 It is in our opinion very likely, that these Te compounds exhibit similar structures as presented for the Se compounds in this work, which would challenge the results of Németh et al.15 Unfortunately, the crystal structures of these compounds could not be fully elucidated until now, partly due to their increased chemical and mechanical sensitivity.
Author contributions
T. M.: data curation, investigation, methodology, software, visualisation, writing – original draft. M. H.: conceptualisation, data curation, investigation, methodology, writing – review & editing. U. R.: funding acquisition, resources, supervision, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
Data supporting this article, has been included as part of the supplementary information (SI). Supplementary information: Tables S1–S7, further Rietveld and PDF fits, IR spectra, further experimental and computational details. IR and Raman data for this article are available at Chemotion Repository at https://dx.doi.org/10.14272/collection/tmattic2_2026-02-04. The code for the supercell generation is available at https://doi.org/10.5281/zenodo.18611408. See DOI: https://doi.org/10.1039/d6ra01467d.CCDC 2529945–2529948 contain the supplementary crystallographic data for this paper.70a–d
Acknowledgements
The authors thank Dr Lisa Körtgen (sample preparation), Dr Christoph Lenting (Raman), Tom Schneider (SEM/EDX), and Dr Christian Sternemann (synchrotron powder data) for their help and support with data acquisition and DELTA for providing synchrotron radiation. We acknowledge DESY (Hamburg, Germany), a member of the Helmholtz Association HGF, for the provision of experimental facilities. Parts of this research were carried out at PETRA III. Data was collected using beamline P21.1 operated by DESY Photon Science. We would like to thank Jiatu Liu and Philipp Glaevecke for assistance during the experiments. Beamtime was allocated for proposal I-20240621 EC. The authors thank the groups of Sabrina Disch and Germán Salazar Alvarez for their help with data acquisition. The authors furthermore thank the ITCC (IT Center University of Cologne) for providing compute resources on the DFG-funded HPC (High Performance Computing) system RAMSES (Research Accelerator for Modeling and Simulation with Enhanced Security) as well as support (DFG funding number: INST 216/512-1 FUGG). Finally, we would like to acknowledge the generous financial support provided for this research project by the DFG (RU 546/13-2) and TM acknowledges the Fonds der Chemischen Industrie for a Kekulé fellowship.References
- M. H. Moissan, C. R. Acad. Sci., 1898, 126, 302–308 Search PubMed.
- M. H. Moissan, C. R. Acad. Sci., 1898, 127, 911–918 Search PubMed.
- R. Juza, V. Wehle and H. Schuster, Z. Anorg. Allg. Chem., 1967, 352, 252–257 CrossRef CAS.
- H. Föppl, Angew. Chem., 1958, 70, 401 Search PubMed.
- U. Ruschewitz, P. Müller and W. Kockelmann, Z. Anorg. Allg. Chem., 2001, 627, 513–522 CrossRef CAS.
- U. Ruschewitz and R. Pöttgen, Z. Anorg. Allg. Chem., 1999, 625, 1599–1603 CrossRef CAS.
- S. Hemmersbach, B. Zibrowius and U. Ruschewitz, Z. Anorg. Allg. Chem., 1999, 625, 1440–1446 CrossRef CAS.
- R. Nast and H. Schindel, Z. Anorg. Allg. Chem., 1963, 326, 201–208 CrossRef CAS.
- W. Kockelmann and U. Ruschewitz, Angew. Chem., Int. Ed., 1999, 38, 3492–3495 CrossRef CAS PubMed.
- M. Weiß and U. Ruschewitz, Z. Anorg. Allg. Chem., 1997, 623, 1208–1210 CrossRef.
- S. Hemmersbach, B. Zibrowius, W. Kockelmann and U. Ruschewitz, Chem. - Eur. J., 2001, 7, 1952–1958 CrossRef CAS PubMed.
- U. Ruschewitz, Z. Anorg. Allg. Chem., 2001, 627, 1231–1235 CrossRef CAS.
- M. A. Carvajal, J. J. Novoa and S. Alvarez, J. Am. Chem. Soc., 2004, 126, 1465–1477 CrossRef CAS PubMed.
- L. P. Wolters and F. M. Bickelhaupt, ChemistryOpen, 2013, 2, 106–114 CrossRef CAS PubMed.
- K. Németh, A. K. Unni, C. Kalnmals, C. U. Segre, J. Kaduk, I. D. Bloom and V. A. Maroni, RSC Adv., 2015, 5, 55986–55993 RSC.
- A. J. Bradley, London Edinburgh Philos. Mag. J. Sci., 1924, 48, 477–496 CrossRef CAS.
- J. Z. Terdik, K. Németh, K. C. Harkay, J. H. Terry, L. Spentzouris, D. Velázquez, R. Rosenberg and G. Srajer, Phys. Rev. B, 2012, 86, 035142 CrossRef.
- M. Hetzert, L. Körtgen and U. Ruschewitz, Z. Anorg. Allg. Chem., 2025, 651, e202500075 CrossRef CAS.
- M. Hetzert, M. Werker and U. Ruschewitz, Angew. Chem., Int. Ed., 2018, 57, 16475–16479 CrossRef CAS PubMed.
- M. Hetzert, C. Tobeck and U. Ruschewitz, Z. Anorg. Allg. Chem., 2020, 646, 1583–1587 CrossRef CAS.
- M. Hetzert, Q. Yuan, W. Cao, X.-B. Wang, J. Hempelmann, R. Dronskowski and U. Ruschewitz, Inorg. Chem., 2022, 61, 18769–18778 CrossRef CAS PubMed.
- C. Krywka, C. Sternemann, M. Paulus, N. Javid, R. Winter, A. Al-Sawalmih, S. Yi, D. Raabe and M. Tolan, J. Synchrotron Radiat., 2007, 14, 244–251 CrossRef CAS PubMed.
- M. v. Zimmermann, O. Ivashko, F. Igoa Saldaña, J. Liu, P. Glaevecke, O. Gutowski, R. Nowak, K. Köhler, B. Winkler, A. Schöps, H. Schulte-Schrepping and A.-C. Dippel, J. Synchrotron Radiat., 2025, 32, 802–814 CrossRef CAS PubMed.
- J. Kieffer, J. Orlans, N. Coquelle, S. Debionne, S. Basu, A. Homs, G. Santoni and D. De Sanctis, J. Appl. Crystallogr., 2025, 58, 138–153 CrossRef CAS PubMed.
- P. Juhás, T. Davis, C. L. Farrow and S. J. L. Billinge, J. Appl. Crystallogr., 2013, 46, 560–566 CrossRef.
- C. L. Farrow, M. Shaw, H. Kim, P. Juhás and S. J. L. Billinge, Phys. Rev. B, 2011, 84, 134105 CrossRef.
- A. A. Coelho, J. Appl. Crystallogr., 2018, 51, 210–218 CrossRef CAS.
- A. A. Coelho, P. A. Chater and A. Kern, J. Appl. Crystallogr., 2015, 48, 869–875 CrossRef CAS.
- P. A. Chater, Topas, Online, 2023, https://github.com/pachater/, last visited 24.11.2025 Search PubMed.
- D. D. Swank and R. D. Willett, Inorg. Chem., 1965, 4, 499–501 CrossRef CAS.
- P. Giannozzi, O. Andreussi, T. Brumme, O. Bunau, M. B. Nardelli, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, M. Cococcioni, N. Colonna, I. Carnimeo, A. D. Corso, S. de Gironcoli, P. Delugas, R. A. D. Jr, A. Ferretti, A. Floris, G. Fratesi, G. Fugallo, R. Gebauer, U. Gerstmann, F. Giustino, T. Gorni, J. Jia, M. Kawamura, H.-Y. Ko, A. Kokalj, E. Küçükbenli, M. Lazzeri, M. Marsili, N. Marzari, F. Mauri, N. L. Nguyen, H.-V. Nguyen, A. O. de-la Roza, L. Paulatto, S. Poncé, D. Rocca, R. Sabatini, B. Santra, M. Schlipf, A. P. Seitsonen, A. Smogunov, I. Timrov, T. Thonhauser, P. Umari, N. Vast, X. Wu and S. Baroni, J. Phys.:Condens. Matter, 2017, 29, 465901 CrossRef CAS PubMed.
- P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. L. Chiarotti, M. Cococcioni, I. Dabo, A. Dal Corso, S. de Gironcoli, S. Fabris, G. Fratesi, R. Gebauer, U. Gerstmann, C. Gougoussis, A. Kokalj, M. Lazzeri, L. Martin-Samos, N. Marzari, F. Mauri, R. Mazzarello, S. Paolini, A. Pasquarello, L. Paulatto, C. Sbraccia, S. Scandolo, G. Sclauzero, A. P. Seitsonen, A. Smogunov, P. Umari and R. M. Wentzcovitch, J. Phys.:Condens. Matter, 2009, 21, 395502 CrossRef PubMed.
- J. P. Perdew, A. Ruzsinszky, G. I. Csonka, O. A. Vydrov, G. E. Scuseria, L. A. Constantin, X. Zhou and K. Burke, Phys. Rev. Lett., 2008, 100, 136406 CrossRef.
- G. Prandini, A. Marrazzo, I. E. Castelli, N. Mounet and N. Marzari, npj Comput. Mater., 2018, 4, 72 CrossRef.
- A. Dal Corso, Comput. Mater. Sci., 2014, 95, 337–350 CrossRef CAS.
- M. Schlipf and F. Gygi, Comput. Phys. Commun., 2015, 196, 36–44 CrossRef CAS.
- M. van Setten, M. Giantomassi, E. Bousquet, M. Verstraete, D. Hamann, X. Gonze and G.-M. Rignanese, Comput. Phys. Commun., 2018, 226, 39–54 CrossRef CAS.
- K. F. Garrity, J. W. Bennett, K. M. Rabe and D. Vanderbilt, Comput. Mater. Sci., 2014, 81, 446–452 CrossRef CAS.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, J. Chem. Phys., 2010, 132, 154104 CrossRef PubMed.
- P. Hohenberg and W. Kohn, Phys. Rev., 1964, 136, B864–B871 CrossRef.
- D. R. Hamann, Phys. Rev. B, 2013, 88, 085117 CrossRef.
- F. Neese, WIREs Comput. Mol. Sci., 2025, 15, e70019 CrossRef.
- F. Neese, J. Comput. Chem., 2003, 24, 1740–1747 Search PubMed.
- F. Neese, F. Wennmohs, A. Hansen and U. Becker, Chem. Phys., 2009, 356, 98–109 CrossRef CAS.
- B. Helmich-Paris, B. de Souza, F. Neese and R. Izsák, J. Chem. Phys., 2021, 155, 104109 CrossRef CAS PubMed.
- F. Neese, J. Comput. Chem., 2022, 44, 381–396 CrossRef.
- L. Wittmann, I. Gordiy, M. Friede, B. Helmich-Paris, S. Grimme, A. Hansen and M. Bursch, Phys. Chem. Chem. Phys., 2024, 26, 21379–21394 RSC.
- A. D. Becke, J. Chem. Phys., 1993, 98, 5648–5652 CrossRef CAS.
- C. Lee, W. Yang and R. G. Parr, Phys. Rev. B, 1988, 37, 785–789 CrossRef CAS PubMed.
- S. H. Vosko, L. Wilk and M. Nusair, Can. J. Phys., 1980, 58, 1200–1211 CrossRef CAS.
- P. J. Stephens, F. J. Devlin, C. F. Chabalowski and M. J. Frisch, J. Phys. Chem., 1994, 98, 11623–11627 CrossRef CAS.
- F. Weigend and R. Ahlrichs, Phys. Chem. Chem. Phys., 2005, 7, 3297–3305 RSC.
- J. Zheng, X. Xu and D. G. Truhlar, Theor. Chem. Acc., 2010, 128, 295–305 Search PubMed.
- E. Caldeweyher, C. Bannwarth and S. Grimme, J. Chem. Phys., 2017, 147, 034112 CrossRef PubMed.
- E. Caldeweyher, S. Ehlert, A. Hansen, H. Neugebauer, S. Spicher, C. Bannwarth and S. Grimme, J. Chem. Phys., 2019, 150, 154122 CrossRef PubMed.
- E. Caldeweyher, J.-M. Mewes, S. Ehlert and S. Grimme, Phys. Chem. Chem. Phys., 2020, 22, 8499–8512 RSC.
- T. Y. Nikolaienko, L. A. Bulavin and D. M. Hovorun, Comput. Theor. Chem., 2014, 1050, 15–22 CrossRef CAS.
- T. Y. Nikolaienko and L. A. Bulavin, Int. J. Quantum Chem., 2018, 119, e25798 CrossRef.
- W. Teichert and W. Klemm, Z. Anorg. Allg. Chem., 1939, 243, 86–98 CrossRef CAS.
- N. Metropolis, A. W. Rosenbluth, M. N. Rosenbluth, A. H. Teller and E. Teller, J. Chem. Phys., 1953, 21, 1087–1092 CrossRef CAS.
- C. Robert and G. Casella, Stat. Sci., 2011, 26, 102–115 Search PubMed.
- M. Hamberger, S. Liebig, U. Friedrich, N. Korber and U. Ruschewitz, Angew. Chem., Int. Ed., 2012, 51, 13006–13010 CrossRef CAS PubMed.
- X. Qiu, E. S. Božin, P. Juhas, T. Proffen and S. J. L. Billinge, J. Appl. Crystallogr., 2004, 37, 110–116 CrossRef CAS.
- E. Zintl, A. Harder and B. Dauth, Z. Elektrochem. Angew. Phys. Chem., 1934, 40, 588–593 CrossRef CAS.
- H. Sommer and R. Hoppe, Z. Anorg. Allg. Chem., 1977, 429, 118–130 CrossRef CAS.
- A. Bondi, J. Phys. Chem., 1964, 68, 441–451 Search PubMed.
- S. F. Yuck, I. Sargin, N. Meyer, J. T. Krogel, S. P. Beckmann and V. R. Cooper, Sci. Rep., 2024, 14, 20219 CrossRef PubMed.
- S. Baroni, S. de Gironcoli, A. Dal Corso and P. Giannozzi, Rev. Mod. Phys., 2001, 73, 515–562 CrossRef CAS.
- A. Kramida, Y. Ralchenko, J. Reader and NIST ASD Team, NIST Atomic Spectra Database (Ver. 5.12), Online, 2014, https://physics.nist.gov/asd, last visited 17.02.2026 Search PubMed.
- (a) CCDC 2529945: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxm4f; (b) CCDC 2529946: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxm5g; (c) CCDC 2529947: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxm6h; (d) CCDC 2529948: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxm7j.
| This journal is © The Royal Society of Chemistry 2026 |