Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Metal-free Minisci C–H alkylation of hydrazones using aldehydes: an unexpected route to hydrazone-containing pyrimidine derivatives
Atefeh Tirehdasta,
Volodymyr Semeniuchenko b and
Ali Shiri
b and
Ali Shiri *a
*a
aDepartment of Chemistry, Faculty of Science, Ferdowsi University of Mashhad, Mashhad, Iran. E-mail: alishiri@um.ac.ir
bDepartment of Chemistry and Biomolecular Sciences, Faculty of Science, University of Ottawa, Ottawa, Canada
First published on 29th April 2026
Abstract
In this research, a novel method for synthesizing various heterocyclic derivatives via the Minisci radical substitution reaction has been presented. The Minisci reaction offers a very efficient and valuable approach for directly functionalizing heteroarenes with an electron-deficient group, allowing the formation of carbon–carbon bonds straightforwardly and efficiently. We have developed a metal-free, light-independent Minisci-type strategy for the direct C–H functionalization of pyrimidines, thereby facilitating the design of heteroaromatic derivatives containing hydrazone groups. The target derivatives were successfully synthesized in good yields using aromatic aldehydes as alkyl radical precursors. The reaction proceeds through a sequence of hydrogen atom abstraction (HAA) from the aldehyde, followed by decarbonylation under oxidative conditions using K2S2O8 and trifluoroacetic acid (TFA). This method provides a practical and scalable approach to accessing structurally heterocyclic compounds of various classes.
Introduction
The Minisci reaction, first developed in the late 1960s by Francesco Minisci, is a significant strategy for the direct radical functionalization of electron-deficient nitrogen-containing heteroarenes.1–5 One of the key advantages of this process is its ability to directly attach alkyl and aryl groups onto heteroaromatic frameworks without needing prior substrate activation.6–11 This notable property has made the Minisci reaction essential for the synthesis of complex bioactive compounds and producing pharmaceuticals and important synthetic intermediates.12–14Even if there are such merits, classical Minisci protocols generally experience inherent deficiencies. The traditional protocols generally rely on metal catalysis, photochemical initiation, or stoichiometric amounts of strong oxidants.15 Such needs not only become prohibitively expensive and environmentally unfavorable but also impose severe limitations on scalability and functional group toleration.16–19 Consequently, efforts in the recent past were aimed at the synthesis of more practical alternatives. Metal-free and photocatalyst-free forms became the future Minisci-type protocols through the accomplishment of the conversion under milder, more sustainable conditions, in alignment with the principles of the green chemistry movement.20–24 Of the many radical precursors studied for Minisci reactions, the aldehyde case is particularly unique.25 It simplifies the process by inclusion of aldehydes, and offers access to a vast new product range.26–28
Recent studies have demonstrated that aldehydes can serve as effective radical precursors in Minisci-type C–H functionalization of heteroarenes. In several reported systems, these transformations proceed under visible-light irradiation through photochemically initiated radical pathways.29 In addition, related approaches have utilized metal catalysts in combination with oxidants to enable oxidative C–H functionalization using aldehydes as coupling partners.30 While these studies highlight the versatility of aldehydes in radical C–H functionalization chemistry, many of the existing methods rely on either external light sources or metal catalysts. In contrast, the present study describes a metal-free and light-independent Minisci-type protocol for the C–H functionalization of pyrimidines using readily available aldehydes, providing straightforward access to hydrazone-containing heterocyclic derivatives.
On the other hand, pyrimidines, among the nitrogen heterocycles, have long been a focus of medicinal chemists due to their vast array of biological activities, including anticancer,31 antiviral,32,33 antibacterial,34,35 and enzyme inhibitory properties.36,37 Their unique properties and strong ability to engage biological targets render them popular scaffolds in modern drug design. Moreover, hydrazone-containing frameworks have attracted significant attention owing to their diverse biological properties, including anticancer, anti-inflammatory, antifungal, and antiviral activities.38,39 The incorporation of the hydrazone substructures on heteroaromatic rings such as pyrimidines has the potential to endow heterocycles with promising pharmacological properties. One among the subsets of the hydrazone compounds occupies central roles in various drugs and natural products, for instance, nifuroxazide (A) functions as an intestinal antiseptic; compounds (B), exhibit anti-tubercular activity, compounds (C) show anti-thyroid cancer properties, while compound (D) displays analgesic and antibacterial activities (Fig. 1).40–43
Motivated by such remarks and in continuation of our previous studies,44–47 the present work looks at the potential use of aldehydes as radical precursor sources in the synthesis of hydrazone-containing heterocyclic derivatives via Minisci-type reactions. This work attempts to offer a convenient, metal-free, and light-independent synthetic process that not only adheres to the principles of green chemistry but also offers a viable process for accessing novel heteroaromatic hydrazones of remarkable pharmaceutical promise.
Results and discussion
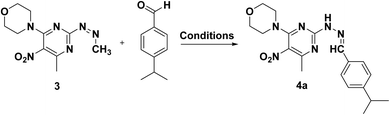
For the synthesis of heterocycles 4a–f, initially, 2,4-dichloro-6-methyl-5-nitropyrimidine (1, synthesized by known method)48 was stirred at room temperature with morpholine in chloroform for 2 h to obtain precursor 2.49 In the subsequent investigation, we hypothesized that the 2, when treated with hydrazine hydrate in a mixture of triethylamine (Et3N) and dimethylformamide (DMF), would undergo direct hydrazination. However, analysis revealed the formation of compound 3 (Scheme 1).Several intermediates were isolated at different time points and their structures confirmed through spectral analysis. In the proposed mechanism, hydrazine hydrate and DMF initially react, and the resulting product acts as a nucleophile attacking the chlorine atom in compound 2 at position 4, forming structure I.50–52 Then the reaction leads to the formation of a pyrazole ring, as in compound II. The presence of hydrazine hydrate subsequently causes the CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N double bond to be reduced.53–55 Through sigmatropic shift of H[1,5] under thermal conditions a more stable product forms by rearranging π bonds, ultimately leading to the final compound 3 (Scheme 2).
N double bond to be reduced.53–55 Through sigmatropic shift of H[1,5] under thermal conditions a more stable product forms by rearranging π bonds, ultimately leading to the final compound 3 (Scheme 2).
In order to assay the Minisci reaction, the optimization studies were carried out using compound 3 and 4-isopropylbenzaldehyde as model substrates (Table 1). We investigated different oxidants and solvents to find suitable reaction conditions. Experiments conducted in acetonitrile, whether using trifluoroacetic acid (TFA) or sulfuric acid (H2SO4) (entries 1 and 2), using ammonium persulfate as the oxidant and dichloroethane (DCE) as the solvent, resulted in negligible product formation (entry 3), while replacing TFA with H2SO4 under the same conditions (entry 4) completely inhibited the reaction. In contrast, the combination of DCE, TFA, and potassium persulfate produced the desired product (yield = 85%) (entry 5), indicating that these conditions were optimal. The superior performance of K2S2O8 compared to (NH4)2S2O8 may be attributed to its higher stability under the reaction conditions, which likely enables a more efficient and controlled generation of sulfate radical species. Alternative oxidants, such as benzoyl peroxide (entry 6), and solvents, such as DMF (entry 7), failed to improve the reaction efficiency. Furthermore, reactions conducted in DCE without the addition of acid (entry 8) or in the absence of solvent (entry 9) did not yield the desired product. The reaction was also performed at room temperature (entry 10); however, no product was observed, likely due to insufficient thermal activation of K2S2O8 required to generate sulfate radical species. Overall, these studies identify dichloroethane as the appropriate solvent, trifluoroacetic acid as the acid, and potassium persulfate as the optimal oxidant in reflux conditions.
| Entry | Solvent | Acid | Oxidant | Time (h) | Temperature (°C) | Yield (%) |
|---|---|---|---|---|---|---|
| 1 | CH3CN | TFA | K2S2O8 | 24 | Reflux | — |
| 2 | CH3CN | H2SO4 | K2S2O8 | 24 | Reflux | — |
| 3 | DCE | TFA | (NH4)2S2O8 | 20 | Reflux | Trace |
| 4 | DCE | H2SO4 | (NH4)2S2O8 | 24 | Reflux | — |
| 5 | DCE | TFA | K2S2O8 | 16 | Reflux | 85 |
| 6 | DCE | TFA | Benzoyl peroxide | 24 | Reflux | — |
| 7 | DMF | TFA | K2S2O8 | 24 | 100 | — |
| 8 | DCE | — | K2S2O8 | 24 | Reflux | — |
| 9 | — | TFA | K2S2O8 | 24 | 100 | — |
| 10 | DCE | TFA | K2S2O8 | 24 | 25 | — |
In the final step, products 4a–f were synthesized by reacting compound 3 with various aldehyde derivatives in K2S2O8 as a radical source, TFA as an acid catalyst, and dichloroethane as solvent under reflux conditions (Scheme 3).
Based on the literature reports56,57 and our studies, a plausible mechanism is proposed in Fig. 2. The Minisci reaction involves radical C–H functionalization of electron-deficient heteroarenes, in which aldehydes act as precursors to alkyl radicals via a decarbonylation pathway. Protonation of compound 3 with trifluoroacetic acid (TFA) increases its electrophilicity and directs radical addition to the most electron-deficient sites. Initially, potassium persulfate decomposes under the reaction conditions to generate sulfate radicals, which initiate the transformation of aldehydes into alkyl radicals through a sequence of radical steps. Mechanistically, the sulfate radical first abstracts the formyl hydrogen of the aldehyde, yielding a transient acyl radical intermediate. This intermediate rapidly undergoes decarbonylation, extruding CO to furnish the corresponding alkyl radical. The alkyl radical then adds to the protonated heteroarene to form a radical cation, which ultimately provides the products 4a–f.
All the structures were characterized, and the spectral data of one of the synthesized derivatives, 4-(2-(2-(4-isopropylbenzylidene)hydrazineyl)-6-methyl-5-nitropyrimidin-4-yl)morpholine 4a, were reported. In the IR spectrum of this derivative, the stretching vibration bands appear at νmax 3277 cm−1 (NH), whereas the IR spectrum of compound 3 reveals the absence of an NH moiety. The C–H stretching vibrations of both aromatic and aliphatic groups appear in the range of ν = 2868–3133 cm−1, and symmetric and asymmetric stretching vibrations of the nitro group in the range of ν = 1573, 1327 cm−1. In the 1H NMR spectrum, a doublet signal at δ 1.23 ppm corresponds to six hydrogens of the isopropyl moiety (–CH(C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif) 3)2). There is a single signal at δ 2.44 ppm corresponding to three hydrogens of the methyl of the pyrimidine ring, and a peak as a multiplet integrating for one hydrogen is associated, corresponding to (–C
3)2). There is a single signal at δ 2.44 ppm corresponding to three hydrogens of the methyl of the pyrimidine ring, and a peak as a multiplet integrating for one hydrogen is associated, corresponding to (–C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif) (CH3)2) and two peaks appear as triplets in δ 3.48 ppm and δ 3.69 ppm, each integrating for four hydrogens, confirm the presence of the morpholine ring. Two peaks as a doublet signal at δ 7.32 ppm and δ 7.61 ppm, each integrating for two hydrogens, corresponded to the phenyl protons, and a signal at δ 8.16 ppm, corresponding to one hydrogen, is associated with (–CH
(CH3)2) and two peaks appear as triplets in δ 3.48 ppm and δ 3.69 ppm, each integrating for four hydrogens, confirm the presence of the morpholine ring. Two peaks as a doublet signal at δ 7.32 ppm and δ 7.61 ppm, each integrating for two hydrogens, corresponded to the phenyl protons, and a signal at δ 8.16 ppm, corresponding to one hydrogen, is associated with (–CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N). Also, the spectrum shows a singlet D2O-exchangeable signal at δ 11.53 ppm, indicating the NH moiety. In the 13C NMR spectrum, two groups of peaks are observed: five peaks in the aliphatic region corresponding to
N). Also, the spectrum shows a singlet D2O-exchangeable signal at δ 11.53 ppm, indicating the NH moiety. In the 13C NMR spectrum, two groups of peaks are observed: five peaks in the aliphatic region corresponding to ![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H3-pyrimidine, and carbons of isopropyl and morpholine moieties. Nine signals appear in the aromatic region, eight of which correspond to the carbons of the pyrimidine and phenyl rings. In contrast, one signal at δ 144.4 ppm corresponds to the carbon of CH
H3-pyrimidine, and carbons of isopropyl and morpholine moieties. Nine signals appear in the aromatic region, eight of which correspond to the carbons of the pyrimidine and phenyl rings. In contrast, one signal at δ 144.4 ppm corresponds to the carbon of CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N, confirming the true structure of this compound. Furthermore, Nuclear Overhauser Effect Spectroscopy (NOESY) was conducted on compound 4e. The two-dimensional NOESY spectrum, resembling the 1H NMR spectrum, confirmed the spatial relationship between the C
N, confirming the true structure of this compound. Furthermore, Nuclear Overhauser Effect Spectroscopy (NOESY) was conducted on compound 4e. The two-dimensional NOESY spectrum, resembling the 1H NMR spectrum, confirmed the spatial relationship between the C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif)
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N and NH protons, as evidenced by the presence of cross-peaks. This indicates that these two hydrogen atoms are in close proximity within the molecular structure. Additionally, the spatial relationship between the C
N and NH protons, as evidenced by the presence of cross-peaks. This indicates that these two hydrogen atoms are in close proximity within the molecular structure. Additionally, the spatial relationship between the C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif)
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N and the morpholine protons is confirmed by the presence of cross-peaks (Fig. 3).
N and the morpholine protons is confirmed by the presence of cross-peaks (Fig. 3).
Eventually, the mass spectrum of this sample shows the molecular ion peak at m/z = 384 and a peak at m/z = 219, which corresponds to the cleavage of the nitro and isopropylbenzene groups from the main structure, confirming the synthesis of the desired compound 4a. Attempted crystallography for compound 4a confirmed the connectivity of the atoms, but significant twinning did not allow the collection of publishable data for the crystal structure of this compound.
Conclusion
In summary, a protocol for the preparation of new derivatives of pyrimidine containing hydrazone substituents 4a–f has been reported and obtained by the metal-free and light-independent Minisci reaction. The synthetic method used radical precursors in the form of aldehydes and an oxidative medium mediated by K2S2O8–TFA, providing an efficient method for the direct functionalization of the pyrimidine derivatives containing the hydrazone 4a–f via C–C cross-coupling. The derivatives obtained were carefully characterized by using spectroscopic and microanalytical methods to confirm the structural framework. Pyrimidine–hydrazone moieties are of great interest because they have a wide biological spectrum.Experimental
Melting points were measured by an Electrothermal Type 9200 melting point apparatus. The 1H NMR (300 MHz) and the 13C NMR (75 MHz) spectra were obtained on a Bruker Avance DRX-300 Fourier transfor spectrometer. An Avatar 370 FT-IR Thermo Nicolet spectrometer was employed to record the IR spectra, and a Varian Mat CH-7 instrument for collecting mass spectra with 70 eV. Microanalytical data were obtained on a Thermo Finnigan Flash EA 1112 microanalyzer.2-Chloro-6-methyl-4-morpholino-5-nitropyrimidine (2)
Yellow needle crystals; yield = 90%; m. p. 115–116 °C; IR (KBr disc, cm−1): ν 3427, 2986, 2925, 2864, 1580, 1495, 1423, 1344, 1290, 1228, 1141. 1H NMR (300 MHz, CDCl3): δH 2.50 (s, 3H, CH3), 3.60 (t, J = 4.8 Hz, 4H, morpholine), 3.78–3.82 (m, 4H, morpholine). 13C NMR (75 MHz, CDCl3): δC 21.3, 46.6, 66.3, 131.0, 154.8, 158.8, 163.5. MS (m/z) = 258 (M+), 198 [M+ − (Me, NO2)], 128 [M+ − (morpholine, NO2)]. Anal. calcd for C9H11ClN4O3 (%): C, 41.79; H, 4.29; N, 21.66. Found: C, 41.77; H, 4.27; N, 21.64.General procedure for the synthesis of compound (3)
To prepare 6-methyl-2-(methyldiazenyl)-4-morpholino-5-nitropyrimidine 3, a solution of compound 2 (1 mmol, 0.258 g) in DMF (1 mL) was added to a mixture of hydrazine hydrate (0.24 mL, 5 mmol), triethylamine (0.3 mL, 2 mmol), and DMF (2 mL), which had been preheated to 50 °C. The resulting mixture was then stirred at 50 °C for 10 h. The progress of the reaction was monitored by TLC using chloroform![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) methanol (20
methanol (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent. After the completion of the reaction, the solvent was removed under reduced pressure, and the resulting solid was washed with water (2 × 20 mL) and dried.
1) as eluent. After the completion of the reaction, the solvent was removed under reduced pressure, and the resulting solid was washed with water (2 × 20 mL) and dried.
N,N-Dimethyl-N′-(4-methyl-6-morpholino-5-nitropyrimidin-2-yl)formohydrazonamide (I)
Brown powder; yield = 54%; m. p. 130–131 °C; IR (KBr disc, cm−1): ν 3338, 3260, 3203, 3039, 2960, 2900, 1634, 1567, 1511, 1470, 1442, 1330, 1263. 1H NMR (300 MHz, DMSO-d6): δH 2.26 (s, 3H, CH3-pyrimidine), 2.32–2.38 (m, 6H, N(CH3)2), 3.62–3.65 (m, 8H, morpholine), 5.80 (s, 1H, CH), 8.20 (s, 1H, NH). 13C NMR (75 MHz, DMSO-d6): δC 24.9, 37.2, 47.2, 66.2, 123.9, 143.3, 158.6, 158.7, 163.8. MS (m/z) = 309 (M+), 177 [M+ − (morpholine, NO2)]. Anal. calcd for C12H19N7O3 (%): C, 46.59; H, 6.19; N, 31.70. Found: C, 46.57; H, 6.17; N, 31.68.4-(5-Methyl-6-nitro-[1,2,4]triazolo[4,3-a]pyrimidin-7-yl)morpholine (II)
Yellow powder; yield = 44%; m. p. 123–124 °C; IR (KBr disc, cm−1): ν 3208, 3139, 3090, 2988, 2898, 2864, 1585, 1524, 1480, 1420, 1379, 1358, 1307, 1280, 1187. MS (m/z) = 264 (M+), 218 [M+ − NO2], 178 [M+ − (morpholine)]. Anal. calcd for C10H12N6O3 (%): C, 45.45; H, 4.58; N, 31.80. Found: C, 45.43; H, 4.56; N, 31.78.6-Methyl-2-(methyldiazenyl)-4-morpholino-5-nitropyrimidine (3)
Yellow powder; yield = 65%; m. p. 102–103 °C; IR (KBr disc, cm−1): ν 3035, 2970, 2925, 2858, 1558, 1486, 1445, 1402, 1370, 1323, 1267, 1232, 1141. 1H NMR (300 MHz, CDCl3): δH 2.51 (s, 3H, CH3-pyrimidine), 3.22 (s, 3H, CH3), 3.47–3.50 (m, 4H, morpholine), 3.76–3.79 (m, 4H, morpholine). 13C NMR (75 MHz, CDCl3): δC 23.6, 37.2, 47.2, 66.8, 124.3, 157.1, 158.7, 164.1. MS (m/z) = 266 (M+), 220 [M+ − NO2], 135 [M+ − (morpholine, NO2)]. Anal. calcd for C10H14N6O3 (%): C, 45.11; H, 5.30; N, 31.56. Found: C, 45.09; H, 5.28; N, 31.54.General procedure for the synthesis of compound (4a–f)
A mixture of 6-methyl-2-(methyldiazenyl)-4-morpholino-5-nitropyrimidine 3 (1 mmol, 0.266 g), various aldehyde derivatives (3 mmol), K2S2O8 (2.3 mmol, 0.621 g), and TFA (1 mmol, 0.07 mL) in DCE (3 mL) was heated under reflux for 16 h. After completion of the reaction, which was monitored by TLC using CHCl3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH (30
MeOH (30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), the solvent was evaporated under reduced pressure. Water (5 mL) was added, and the mixture was neutralized with an aqueous 5% NaHCO3 solution. The mixture was extracted with EtOAc (3 × 10 mL), and the organic phase was dried over anhydrous Na2SO4 and evaporated under reduced pressure. The crude product was purified by column chromatography on silica gel using CHCl3
1), the solvent was evaporated under reduced pressure. Water (5 mL) was added, and the mixture was neutralized with an aqueous 5% NaHCO3 solution. The mixture was extracted with EtOAc (3 × 10 mL), and the organic phase was dried over anhydrous Na2SO4 and evaporated under reduced pressure. The crude product was purified by column chromatography on silica gel using CHCl3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH (30
MeOH (30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent to yield the pure product.
1) as eluent to yield the pure product.
Single crystals of compound 4a were grown by slow evaporation of acetonitrile solution. For all observed crystals non-merohedral twinning with the presence of multiple domains was observed, and a non-twinned crystal could not be found. Application of other solvents did not allow single crystal growth. X-ray diffraction data collection (Mo Kα radiation, 0.71073 Å wavelength) at 100 K temperature allowed to determine unit cell and space group (a = 11.9529 Å, b = 13.3518 Å, c = 15.7124 Å, α = 93.8315°, β = 111.2006°, γ = 113.9578°, space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ). The asymmetric unit contained two molecules of 4a and one MeCN molecule. Although structure solution and refinement confirmed the atoms connectivity for compound 4a, final R-factors (R1 = 0.2704, wR2 = 0.4368) made the structure not publishable. High R-factors are observed due to crystal twinning and inability to detwin the data using TWINABS software (in Bruker APEX II suite). To our surprise, the structure has not shown any disorder, and all hydrogen atoms were resolved in the difference Fourier map after location and anisotropic refinement of heavy atoms. Located H atoms as well confirmed the structure 4a.
). The asymmetric unit contained two molecules of 4a and one MeCN molecule. Although structure solution and refinement confirmed the atoms connectivity for compound 4a, final R-factors (R1 = 0.2704, wR2 = 0.4368) made the structure not publishable. High R-factors are observed due to crystal twinning and inability to detwin the data using TWINABS software (in Bruker APEX II suite). To our surprise, the structure has not shown any disorder, and all hydrogen atoms were resolved in the difference Fourier map after location and anisotropic refinement of heavy atoms. Located H atoms as well confirmed the structure 4a.
Conflicts of interest
The authors declare that they have no conflicts of interest.Data availability
The datasets used and analyzed in the current study are available in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6ra01465h.Acknowledgements
The authors gratefully acknowledge the Research Council of Ferdowsi University of Mashhad (3/62964).References
- S. Jin, X. Geng, Y. Li and K. Zheng, Eur. J. Org Chem., 2021, 2021, 969–972 CrossRef CAS.
- D. Xie, Y. Liu, X. Liu, Q. Yang and Y. Peng, Eur. J. Org Chem., 2024, 27, e202300993 CrossRef CAS.
- K. A. Rinderspacher, Prog. Heterocycl. Chem., 2015, 27, 393–450 CAS.
- R. S. J. Proctor and R. J. Phipps, Angew. Chem., Int. Ed., 2019, 58, 13666–13699 CrossRef CAS PubMed.
- F. Minisci, R. Bernardi, F. Bertini, R. Galli and M. Perchinummo, Tetrahedron, 1971, 27, 3575–3579 CrossRef CAS.
- O. Patel, N. Nandwana, L. J. Legoabe and A. Kumar, Adv. Synth. Catal., 2020, 362, 4226–4255 CrossRef CAS.
- J. Li, S. Siang Tan, S. H. Kyne and P. Wai Hong Chan, Adv. Synth. Catal., 2022, 364, 802–810 CrossRef CAS.
- M. Kemmochi, Y. Miyamoto, Y. Sumida and H. Ohmiya, Asian J. Org. Chem., 2022, 11, e202100640 CrossRef CAS.
- J. Jin and D. W. C. MacMillan, Angew. Chem., 2015, 127, 1585–1589 CrossRef.
- F. J. A. Troyano, K. Anwar, F. Mohr, G. Robert and A. Gómez-Suárez, Eur. J. Org Chem., 2023, 26, e202201176 CrossRef CAS.
- P. Chandra Tiwari, A. Pulcinella, E. Hodzic and T. Noel, ACS Cent. Sci., 2025, 11, 910–917 CrossRef PubMed.
- M. A. J. Duncton, Med. Chem. Commun., 2011, 2, 1135–1161 RSC.
- X.-Z. Wang and C.-C. Zeng, Tetrahedron, 2019, 75, 1425–1430 CrossRef CAS.
- A.-L. Lee, D. T. Mooney and H. McKee, Org. Biomol. Chem., 2024, 22, 9145–9164 RSC.
- E. Le Saux, E. Georgiou, I. A. Dmitriev, W. C. Hartley and P. Melchiorre, J. Am. Chem. Soc., 2023, 145, 47–52 CrossRef CAS PubMed.
- M. A. Graham, G. Noonan, J. H. Cherryman, J. J. Douglas, M. Gonzalez, L. V. Jackson, K. Leslie, Z. Liu, D. McKinney, R. H. Munday, C. D. Parsons, D. T. E. Whittaker, E. Zhang and J. Zhang, Org. Process Res. Dev., 2021, 25, 57–67 CrossRef CAS.
- J. Dong, F. Yue, J. Liu, H. Song, Y. Liu and Q. Wang, Green Chem., 2021, 23, 7963–7968 RSC.
- E. R. Lopat’eva, I. B. Krylov, O. O. Segida, V. M. Merkulova, A. I. Ilovaisky and A. O. Terent’ev, Molecules, 2023, 28, 934 CrossRef PubMed.
- R. S. J. Proctor, H. J. Davis and R. J. Phipps, Science, 2018, 360, 419–422 CrossRef CAS PubMed.
- A. M. Truscello and C. Gambarotti, Org. Process Res. Dev., 2019, 23, 1450–1457 CrossRef CAS.
- W. Ali, A. Behera, S. Guin and B. K. Patel, J. Org. Chem., 2015, 80, 5625–5632 CrossRef CAS PubMed.
- M. T. Westwood, C. J. C. Lamb, D. R. Sutherland and A.-L. Lee, Org. Lett., 2019, 21, 7119–7123 CrossRef CAS PubMed.
- S. Mandal, T. Bera, G. Dubey, J. Saha and J. K. Laha, ACS Catal., 2018, 8, 5085–5144 CrossRef CAS.
- F. Doraghi, S. Kermaninia, E. S. Ghalehsefid, B. Larijani and M. Mahdavi, RSC Adv., 2025, 15, 14691–14716 RSC.
- F. S. Meger and J. A. Murphy, Molecules, 2023, 28, 6127 CrossRef CAS PubMed.
- K. Matcha and A. P. Antonchick, Angew. Chem., 2013, 125, 2136–2140 CrossRef.
- Y. Siddaraju, M. Lamani and K. R. Prabhu, J. Org. Chem., 2014, 79, 3856–3865 CrossRef CAS PubMed.
- M. Qi and A.-W. Xu, Org. Biomol. Chem., 2024, 22, 2654–2661 RSC.
- Z. Wang, X. Ji, J. Zhao and H. Huang, Green Chem., 2019, 21, 5512–5516 RSC.
- O. Basl, J. Bidange, Q. Shuai and C.-J. Li, Adv. Synth. Catal., 2010, 352, 1145–1149 CrossRef.
- A. Mahapatra, T. Prasad and T. Sharma, Future J. Pharm. Sci., 2021, 7, 123 CrossRef.
- S. Eg and B. Lakshmanan, Asian J. Pharm. Clin. Res., 2020, 13, 30–34 CrossRef.
- F. N. Gallo, A. B. Marquez, D. M. Fidalgo, A. Dana, M. Dellarole, C. C. García and M. Bollini, Eur. J. Med. Chem., 2024, 272, 116465 CrossRef CAS PubMed.
- Y.-M. Tan, D. Li, F.-F. Li, M. Fawad Ansari, B. Fang and C.-H. Zhou, Bioorg. Med. Chem. Lett., 2022, 73, 128885 CrossRef CAS PubMed.
- M. S. Raghu, C. B. Pradeep Kumar, K. Yogesh Kumar, M. K. Prashanth, F. Alharethy and B.-H. Jeon, Bioorg. Med. Chem. Lett., 2024, 103, 129707 CrossRef CAS PubMed.
- H. Kuday, F. Sonmez, C. Bilen, E. Yavuz, N. Gençer and M. Kucukislamoglu, BioMed Res. Int., 2014, 2014, 594879 Search PubMed.
- M. Afzal, R. Mehmood, E. U. Mughal, N. Naeem, Z. Ashraf, Y. Nazir, F. M. Shalaby, A. El-Sayed Abd El Hady and A. Sadiq, RSC Adv., 2024, 14, 22769–22780 RSC.
- S. Rollas and S. G. Küçükgüzel, Molecules, 2007, 12, 1910–1939 CrossRef CAS PubMed.
- D. A. Tafere, M. Gebrezgiabher and F. Elemo, RSC Adv., 2025, 15, 6191–6207 RSC.
- S. N. Mali, B. R. Thorat, D. R. Gupta and A. Pandey, Eng. Proc., 2021, 11, 21 Search PubMed.
- L. Yurttaş, Z. A. Kaplancıklı, Z. Cantürk and K. Gencer, Phosphorus, Sulfur Silicon Relat. Elem., 2015, 190, 1183–1191 CrossRef.
- H. Li, M.-Q. Jia, Z.-L. Qin, C. Lu, W. Chu, Z. Zhang, J. Niu, J. Song, S.-Y. Zhang and L. Fu, J. Enzyme Inhib. Med. Chem., 2024, 39, 2423875 CrossRef PubMed.
- B. Padmashali, B. N. Chidananda, B. Govindappa, S. M. Basavaraj, S. Chandrashekharappa and K. N. Venugopala, J. Appl. Pharm. Sci., 2019, 9, 117–124 CAS.
- A. Mirfarah, S. Hatami and A. Shiri, RSC Adv., 2025, 15, 42121–42175 RSC.
- E. Vatankhah, H. Eshghi and A. Shiri, J. Heterocycl. Chem., 2025, 62, 1111–1118 CrossRef CAS.
- A. Tirehdast, S. Sheikhi-Mohammareh, H. Sabet-Sarvestani, M. G. Organ, V. Semeniuchenko and A. Shiri, RSC Adv., 2024, 14, 29122–29133 RSC.
- P. Moghimi, H. Sabet-Sarvestani, O. Kohandel and A. Shiri, J. Org. Chem., 2022, 87, 3922–3933 CrossRef CAS PubMed.
- K. J. Hodgetts, T. Yoon, J. Huang, M. Gulianello, A. Kieltyka, R. Primus, R. Brodbeck, S. De Lombaert and D. Doller, Bioorg. Med. Chem. Lett., 2003, 13, 2497–2500 CrossRef CAS PubMed.
- S. M. Banihashemi, H. Hassani and J. Lari, Russ. J. Org. Chem., 2020, 56, 322–326 CrossRef CAS.
- Z.-B. Wu, X. Zhou, Y.-Q. Ye, P.-Y. Wang and S. Yang, Chin. Chem. Lett., 2017, 28, 121–125 CrossRef CAS.
- A. Katrusiak, A. Katrusiak and S. Bałoniak, Tetrahedron, 1994, 50, 12933–12940 CrossRef CAS.
- S. H. Kwak and Y.-D. Gong, Tetrahedron, 2013, 69, 7107–7111 CrossRef CAS.
- N. Menges and M. Balci, Synlett, 2014, 25, 671–676 CrossRef CAS.
- B. J. Marsh and D. R. Carbery, J. Org. Chem., 2009, 74, 3186–3188 CrossRef CAS PubMed.
- C. Smit, M. W. Fraaije and A. J. Minnaard, J. Org. Chem., 2008, 73, 9482–9485 CrossRef CAS PubMed.
- X. Wang, X. Shao, Z. Cao, X. Wu and C. Zhu, Adv. Synth. Catal., 2022, 364, 1200–1204 CrossRef CAS.
- R.-J. Tang, L. Kang and L. Yang, Adv. Synth. Catal., 2015, 357, 2055–2060 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |