 Open Access Article
Open Access ArticleWaste-to-worth: nano activated carbon from fruit peels for sulfur-free sugarcane juice clarification
Ramesh Duraisamya,
Wabi Regasa Bogaleab and
Gada Muleta Fanta *acd
*acd
aDepartment of Chemistry, College of Natural and Computational Sciences, Arba Minch University, P.O. Box 21, Arba Minch, Ethiopia
bDepartment of Industrial Chemistry, College of Natural and Computational Sciences, Bule Hora University, P.O. Box 144, Bule Hora, Ethiopia
cDepartment of Materials Science and Engineering, Adama Science and Technology University, P.O. Box 1888, Adama, Ethiopia. E-mail: gada.muleta@astu.edu.et
dCenter of Advanced Materials Science and Engineering, Adama Science and Technology University, P.O. Box 1888, Adama, Ethiopia
First published on 8th May 2026
Abstract
The sugar industry faces significant challenges in achieving high purity and maximizing yield during production, particularly in the crucial clarification stage. Effective clarifying agents are essential for this process. Activated carbon has emerged as a highly suitable adsorbent for removing impurities from sugarcane juice, due to its strong adsorption capacity. This study focuses on preparing nano activated carbon (NAC) derived from mixed fruit peels for clarifying sugarcane juice. Using Design-Expert software, we optimized the experimental parameters for NAC production, determining the optimal conditions: carbonization at 410 °C for 100 min with an acid impregnation ratio of 1. Under these conditions, the resulting NAC achieved a yield of 74.86%, a fixed carbon content of 89.25 ± 0.38%, and a specific surface area of 650.87 ± 0.26 m2 g−1. Characterization by X-ray diffraction and scanning electron microscopy revealed that the NAC had an average particle size of 24.46 nm, with a predominant particle size distribution between 20 and 30 nm. The optimization of NAC for cane juice clarification yielded impressive results under optimal conditions: a NAC dose of 0.01%, pH of 4, temperature of 90 °C, duration of 20 min, and agitation speed of 100 rpm, resulting in a turbidity reduction to 1760.6 NTU, a settling rate of 0.09 cm min−1, a mud volume of 6.66 mL per 100 mL, and a color removal efficiency of 97.4%. Additionally, we examined the physicochemical properties of the NAC, the juice posttreatment, and the final sugar product. The findings demonstrate that NAC synthesized from mixed fruit peels exhibits a high carbon yield and a high specific surface area, highlighting its potential as an effective biosorption material. The synthesized NAC demonstrated excellent clarification efficiency, making it an advantageous clarifying agent for sugarcane juice processing and sulfur-free sugar production. This research has significant implications for waste management, particularly in reducing landfill pollution, while also contributing to the development of value-added products that enhance bioresource management.
1 Introduction
Sugarcane (Saccharum officinarum) and sugar beets (Beta vulgaris) are the primary sources for producing table sugar (sucrose). The industrial process for extracting sucrose involves several steps of isolation and purification to separate it from non-sugar components. Traditional cane sugar production involves four main activities: purifying the raw juice extracted from the cane, evaporating the clarified juice, crystallizing the syrup, and separating the sucrose crystals from molasses (the thick liquid containing uncrystallized sucrose).1 To remove color and colloidal substances from sugarcane juice, it undergoes clarification through coagulation, flocculation, and precipitation. This process may result in an insoluble precipitate that effectively absorbs and extracts undesired components from the juice, followed by decanting and filtration.2 The liming process produces a low-turbidity liquor but may result in higher mud levels and slower-settling flocs. Additionally, the clarified juice may contain higher levels of calcium, which can cause scaling in evaporators.3Alternative methods for clarifying cane juice include the use of synthetic polymer acrylamide, which accelerates the settling of impurities. However, this method is expensive and has raised concerns regarding potential cancer and neurotoxicity risks.4 Furthermore, many countries are moving away from using SO2 in the sulfitation process for refining sugar due to the health risks associated with residual sulfur in sugar products,4 and it is an expensive treatment, requiring the use of SO2 in addition to the health issues. There is growing interest in developing cost effective, efficient alternatives to conventional commercial clarifying agents. Exploring the use of affordable, effective activated carbon (AC) not only promotes environmental sustainability but also offers benefits for future commercial applications.5
Nano activated carbon is gaining immense popularity for its ability to purify and remove pollutants from water, due to its significantly larger surface area and higher adsorption rates than traditional bulk AC. This cutting-edge technology offers a faster, more efficient alternative to conventional water purification methods, resulting in higher impurity removal in aqueous media.6,7 Among the various options, nano activated carbon (as adsorbents) stands out as a cost-effective solution for treating contaminated water due to its unique characteristics, availability, and high efficacy with smaller particles (on the nanometer scale, 10−9 meters), in contrast to regular activated carbon (AC), which has larger particles measured in micrometers.8,9
Agro by-products have emerged as promising raw materials for producing activated carbon (AC). Recent advances in using fruit peels as precursors for AC underscore growing interest in converting agricultural waste into value-added products. According to Lee et al.,10 fruit peels, often abundant and discarded, are a potential source of carbonized materials because of their high content of cellulose, hemicellulose, and lignin, key components of carbonized materials. Studies have investigated the viability of using fruit peels from citrus fruits (such as oranges, lemons, and grapefruits), bananas, mangoes, and pineapples as precursors for AC synthesis.9,11–16
The preparation of nano activated carbon from fruit peel waste represents a groundbreaking interdisciplinary research initiative that merges materials science, waste management, and nanotechnology to create sustainable, high-performance carbonaceous materials with a wide range of applications. This activated carbon can be used as an adsorbent or clarifier to effectively remove pollutants from wastewater and industrial effluents.9,13–15 Additionally, it can be expected to remove impurities (non-sucrose materials) from sugarcane juice during the sugar manufacturing process.
Research in this field has concentrated on fine-tuning activation conditions to enhance the porosity, surface area, and adsorption characteristics of the resulting activated carbon (AC) materials.17 Various factors, including temperature, duration, and impregnation ratio during carbon material activation, have been investigated to optimize the yield and quality of AC produced from fruit peels. The use of phosphoric acid (H3PO4) as an activating agent in the synthesis of activated carbon involves a chemical activation process that transforms carbonaceous precursors into porous structures, yielding greater surface areas and adsorption capacities than with other activating agents.7–9
The costs associated with chemically treating sugarcane juice and the environmental pollution resulting from sludge disposal have prompted the exploration of innovative materials such as NAC.3,9 Research has shown that certain cellulosic materials in fruit peel waste and sugarcane bagasse can be converted into nano activated carbon. Similarly, alternative clarifiers using the cane juice clarification approach substitute traditional chemical treatments with NAC-like materials derived from fruit peel waste, such as banana, avocado, and papaya peels. Utilizing mixed fruit peel waste with a higher organic carbon content to produce activated carbon represents an alternative method for recycling resources and generating value-added products that enhance its adsorption capacity, selectivity, and stability.18,19
Activated carbon made from fruit peel (a form of agricultural carbon, in nano-scale, NAC) has shown promising cost-effective cane juice clarifier, with studies on similar biomass carbons (like bagasse AC) reporting up to 98% color removal under lab conditions without chemical additives, which is comparable to the performance of commercial powdered activated carbon.20 In contrast, conventional clarification methods such as SO2 (sulfitation) and carbonation (lime + CO2) remain industry standards but can introduce residual chemicals (e.g., sulfur) and require precise pH/temperature control.
Commercial PAC or GAC combined with lime generally improves turbidity and color, but at a higher cost and sometimes greater sludge generation than biomass NAC. Polymers like those in polysaccharides enhance flocculation, but synthetic residues and dosing complexity also affect flocculation.21,22 A control sample with no clarifier typically yields poor color and high turbidity, highlighting that NAC from fruit peels not only achieves effective impurity adsorption analogous to commercial PAC/GAC under identical conditions but also offers a practical advantage of reduced chemical usage, lower cost, and minimal residual contaminants in the clarified juice.20
The novelty of this study, which focused on preparing NAC from mixed fruit peels (from avocado, banana, and papaya), lies in mixing heterogeneous agro-wastes into a single nanoscale carbon precursor, producing synergistically improved pore structures and surface functionalities compared with single-biomass sources. In contrast, the mixed-peel approach achieved compositional diversity (cellulose, lignin, pectin, polyphenols) to produce hierarchical nano-porosity and improved biomass strategy; therefore, it represents a more sustainable and possibly higher-performance pathway for nano-carbon production compared with conventional single-precursor methods. This novelty contributes to improved sustainability, waste reduction, and multifunctional performance compared with traditional single-biomass activated carbons. Continued research in this area is expected to optimize the production process further and expand the use of cellulose derived from fruit peels across various technological and environmental applications in terms of mixed fruit peel-derived NAC, which reduces organic waste accumulation and provides a low-cost, eco-friendly adsorbent for minimizing pollutants/organic materials (from aqueous media) and reliance on non-renewable materials.
The possibility of using NAC derived from mixed fruit peel (MFP) to clarify cane juice has not been examined in the existing literature, offering an opportunity to safeguard the environment from pollution while benefiting local communities and the industrial sector. Hence, the main objective of this research focuses on the preparation, optimization, and characterization of an effective NAC derived from fruit peel waste for clarifying sugarcane juice. Specifically, we developed cost-effective NACs using a mixture of banana, papaya, and avocado peels (as MFP). This study also investigated the effects of temperature, time, and acid impregnation ratio on the mixed fruit peel powder during the preparation of nano activated carbon (NAC). Additionally, it assessed the morphological, structural, and particle size characteristics of the NACs. Furthermore, the research explored a novel source of nano activated carbon for clarifying sugarcane juice, with potential applications in the sugar industry.
2. Materials and methods
2.1. Sample collection
In this study, waste from mixed fruit peels, specifically avocado, banana, and papaya, was utilized as a precursor. In February 2024, samples of each peel type were collected separately using a random sampling method in Arbaminch, southern Ethiopia, East Africa. The samples were placed in separate moisture-free polyethene bags and promptly transported to the laboratory for further processing.2.2. Selection and preparation of mixed fruit peel powder
In this study, the peels were selected based on local abundance and their complementary proximate composition, as these peels are widely generated agro-wastes and differ in carbon yield and ash content. The report indicates that banana peel contains high volatile matter, and papaya peel contributes lower ash and higher oxygenated groups. In contrast, avocado peel yields higher lignin and carbon, which are favorable for NAC preparation.23,24 Therefore, blending reflects a material-driven rationale rather than random selection.The collected fruit peels were thoroughly cleaned in the laboratory using tap water to remove any dirt or debris. After cleaning, they were sliced and dried in an oven at 110 °C for 24 h until sufficiently dry for grinding into powder. The dried peels were then ground in a laboratory grinder.25 and passed through a 500 micrometre sieve to ensure uniform particle size. The ground peel powders were blended to a total of 100 g at various proportions, as outlined in Table S1. The resulting finely powdered, homogenized fruit peel samples were stored in a moisture-proof, airtight container until they were activated and carbonized.26 Additional details on the optimization of the mixing proportions for the fruit peels are available in the SI.
2.3. Preparation and optimization of nano activated carbon
The process consisted of a single step that involved activation and carbonization to optimize the proportions of the fruit peel. This was achieved by activating the optimized proportions of dried mixed fruit peel, then carbonizing them in a fruit peel mixer. The experimental design used the RSM-CCD quadratic model approach, as detailed in Table S1. The independent variables included factor A, which represented the impregnation ratio (between sample and acid) of the fruit peel with phosphoric acid (40% H3PO4) at varying levels (1 to 2); factor B, which indicated the processing temperature range (400–700 °C); and factor C, which pertained to the processing time (40–100 min). These processing conditions were selected based on previous research.27–29Optimized dried, powdered mixed peel waste samples (100 g) were treated with 40% H3PO4 at varying ratios, as detailed in Table S1. The treated mixture was then placed in an oven at 100 °C for 12 h to eliminate moisture. Following this, the dried samples were activated at different temperatures and activation durations, as specified in Table 1, ranging from 40 to 100 min.27,29 After activation, the activated carbon (AC) was cooled to room temperature and rinsed with distilled water until the wash water was neutral. Finally, the AC was dried in an oven at 105 °C overnight.
| Run | Independent variables: amount of fruit peels (g) | Dependent variable: % carbon yield | |||
|---|---|---|---|---|---|
| A: Avocado peel (g) | B: Banana peel (g) | C: Papaya peel (g) | Experiment results | Predicted results | |
| a Data expressed as means ± standard deviation of triplicate experiments (n = 3); mean values (in the column) share different superscripts, which are called significantly different (p < 0.05). | |||||
| 1 | 27.03 | 27.03 | 45.94 | 70.36 ± 0.32 cd | 70.42 |
| 2 | 14.3 | 42.85 | 42.85 | 69.11 ± 0.24d | 69.05 |
| 3 | 42.85 | 42.85 | 14.3 | 73.84 ± 0.21a | 73.80 |
| 4 | 33.33 | 33.33 | 33.34 | 71.77 ± 0.67b | 71.72 |
| 5 | 42.85 | 14.3 | 42.85 | 71.23 ± 0.30bc | 71.18 |
| 6 | 43.5 | 43.5 | 13.0 | 71.31 ± 0.32bc | 71.35 |
| 7 | 27.03 | 45.94 | 27.03 | 69.80 ± 0.37d | 69.85 |
| 8 | 45.94 | 27.03 | 27.03 | 73.32 ± 0.49a | 73.37 |
| 9 | 43.5 | 13.0 | 43.5 | 74.92 ± 0.34a | 73.97 |
| 10 | 33.33 | 33.33 | 33.34 | 67.52 ± 0.61e | 67.47 |
| 11 | 33.33 | 33.33 | 33.34 | 71.77 ± 0.67b | 71.72 |
| 12 | 13.0 | 43.5 | 43.5 | 66.05 ± 0.62f | 66.10 |
| ANOVA model summary: Reg. coefficient, R2 | 0.9996 | ||||
| Adjusted R2 | 0.9976 | ||||
| Predicted R2 | 0.9469 | ||||
| Adeq precision | 72.6031 | ||||
| p-value | 0.0019 | ||||
| CV | 0.1676 | ||||
After drying, the synthesized activated carbon was ground, sieved, and stored in a glass bottle until ready for analysis.29 All prepared activated carbons were assessed and optimized for their high fixed carbon content and surface area using Design-Expert software (v.13.0.5.0, USA) to evaluate the adsorbent material.
2.4. Characterization of MFP and NAC
This study aims to enhance the quality of raw materials and prepared products by examining the physicochemical properties of mixed fruit peels (MFP) and the activated carbon produced from them. Key characteristics assessed include moisture, ash content, volatile matter, fixed carbon, carbon yield, and active surface area.26,30 Additionally, Fourier transform infrared spectroscopy (FTIR) was used to analyze changes in functional groups in the optimized NAC. The surface morphology of the NAC was investigated using scanning electron microscopy (SEM), and X-ray diffraction (XRD) was used to evaluate the crystalline structure and particle size of the synthesized and optimized NAC.2.4.1.1. Moisture content. To estimate the moisture content of MFP and NAC, 10 g of each sample was weighed and transferred into a pre-weighed Petri dish. The dish containing the sample was then placed in a hot air oven at 105 ± 0.5 °C for 3 hours to ensure complete drying. The percentage moisture was calculated using the following eqn (1):
| The moisture content of MFPW (%) = [(C − D)/(C − B)] × 100 | (1) |
2.4.1.2. Volatile matter. The sample (10 g) was weighed and transferred to a pre-weighed crucible, which was then covered with its lid. The crucible was heated in a muffle furnace at 950 °C for 7 min. After cooling, the crucible was reweighed to assess the residual mass. The weight loss of the sample was calculated as follows:
| Weight loss (%) = [(C − D)/(C − B)] × 100 | (2) |
| Volatile matter content, VM (%) = Weight loss (%) − Moisture content (%) | (3) |
2.4.1.3. Ash content. To assess the ash content, we first weighed 10 g of the dried sample and placed it into a pre-weighed crucible. The sample was then incinerated in a muffle furnace at 650 °C for 3 h. After allowing the crucible to cool, we reweighed it to determine the mass of the residual ash. The ash content of the sample was calculated using eqn (4):
| Ash content (%) = [(C − D)/(C − B)] × 100 | (4) |
2.4.1.4. Fixed carbon content. Fixed carbon refers to the solid combustible residue remaining after the removal of volatile matter, moisture, and ash from a sample. It is calculated by subtracting the total of volatile matter, moisture, and ash content from 100.
| Percent fixed carbon = 100 − (volatile matter + ash + moisture) | (5) |
2.4.1.5. Percentage of carbon yield of NAC. The weight of the mixed fruit peel and the NAC produced was measured using an electronic balance. The carbon yield for each sample was calculated using the following equation:33
| Carbon yield (%) = (Weight of NAC produced/Weight of mixed fruit peel) × 100 | (6) |
2.4.1.6. Measurement of the active surface area using the iodine adsorption method. Activated carbon is well known for its extensive surface area, attributed to its porous structure that creates numerous adsorption sites. As the surface area expands, its ability to adsorb molecules, such as iodine, also increases. This correlation arises from the greater availability of adsorption sites on the carbon surface. Consequently, higher iodine values are associated with activated carbons that possess larger surface areas. This relationship is frequently leveraged across several industries that use activated carbon for purification, filtration, and adsorption.34
As outlined in the literature,26,34 the adsorption of I2 from solution is a common method for evaluating the surface area of activated carbon. The surface area of the prepared NAC was determined using the iodine adsorption method described by Al Jebur & Alwan,26 with some minor modifications. This technique quantifies the surface area of NAC by measuring the amount of iodine ions adsorbed per gram of NAC. To assess the surface area, 1 g of the prepared NAC was combined with 2 mL of 5% HCl and heated at room temperature for 30 min. Following this, the mixture was added to 100 mL of a 0.1 N iodine solution (N1) and shaken for an additional 30 min. The resulting solution was then filtered, and 10 mL of the filtrate was transferred to a conical flask. The iodine content in 10 mL of the filtrate was titrated with 0.1 N anhydrous sodium thiosulfate (N2) in the presence of a starch indicator. The volume of anhydrous sodium thiosulfate used during the titration was recorded to calculate the iodine number of the NAC, which reflects the amount of iodine adsorbed. The same procedure was used for the blank (without the NAC). Then, the iodine of NAC adsorption was utilized to calculate the surface area of the NAC using the following equation:26
| Adsorbed weight of iodine (WI, in mg I2) = a − [2.2 × b × (v)]. |
| Iodine number (mg I2/g of NAC) = (1.6 WI/Weight of NAC × Correction coefficient) | (7) |
2.4.1.7. Point of zero charge. The point of zero charge (PZC) of a material is defined as the pH at which the positive and negative charges on its surface balance each other, resulting in a net charge of zero. To determine the PZC of NAC, the pH drift method was employed. In this procedure, 0.03 g of NAC is added to 50 mL of 0.1 M KNO3 solution in an Erlenmeyer flask. The pH of the solution is then adjusted to various values ranging from 1 to 12 using HCl or NaOH. After sealing the flask, it is allowed to sit at room temperature for 48 h. The pH of the solution is subsequently measured again. The PZC is identified as the pH at which the initial and final pH values are identical.35
2.5. Extraction of cane juice
Cane juice was extracted from the selected sugarcane using the method outlined.37 The cane was thoroughly washed with tap water and cleaned before milling. A pre-measured amount of cane was sliced and processed in a laboratory mill to extract the juice. The juice was collected in a clean, moisture-free plastic container. A portion of the extracted juice was stored in the refrigerator for subsequent analysis and processing. The juice yield was calculated using eqn (8):
 | (8) |
2.6. Experimental design for optimization of cane juice clarification using NAC
The ideal processing conditions for clarifying extracted cane juice (maintaining a constant juice volume of 100 mL) were determined using a central composite design (CCD) within a response surface methodology framework, facilitated by Design-Expert software (v. 13.0.5.0, USA). Details of the experimental design and the associated procedures for optimizing the clarification of cane juice with NAC are provided in Table S2.2.7. Production of raw sugar
Clarified juice was evaporated to remove water while maintaining a temperature of 102 °C in a temperature-controlled water bath. This process removed most of the water, forming syrup (concentrated juice). The syrup was then allowed to crystallize at room temperature. To initiate crystallization, a small amount of seed sucrose crystals was added, which initiated the formation of sugar (sucrose). To minimize residual water, a freeze dryer was used. Subsequently, the sugar crystals were separated from the molasses by centrifugation. The sugar was then dried in a conventional oven at 105 °C for 15 minutes. Once dried and cooled, the sugar was stored in a moisture-free container until further analysis. The percentage yield of the produced sugar was calculated by comparing the mass of the sugar obtained to the mass of the clarified juice.2.8. Physicochemical characterization of raw cane juice, clarified cane juice, and produced raw sugar
Physicochemical analyses were conducted on raw cane juice, clarified cane juice, and raw sugar. The findings for raw cane juice were compared with those for clarified cane juice, and the results for raw sugar were evaluated against international sugar quality standards.
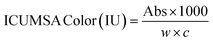 | (9) |
| Percent pol juice = Measured Pol × Pol factor of Brix | (10) |
| Juice or sugar purity (%) = (Pol/Brix) × 100 | (11) |
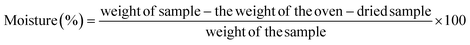 | (12) |
 | (13) |
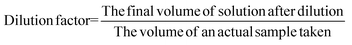 | (14) |
| Boiling house recovery = (Sucrose in sugar/Sucrose in raw juice) × 100 | (15) |
2.9. Statistical analysis
Design-Expert (v.13.0.5.0, USA) was used to analyze the data. The ANOVA output for each response facilitated the identification of significant terms in the models. Significant factors were ascertained based on p-values, and the Tukey method was applied for mean comparisons. At a 95% confidence level, significant differences in mean values were identified (p ≤ 0.05).3. Results and discussion
3.1. Optimization of mixed fruit peels processing conditions
Optimizing the proportion of nano activated carbon derived from mixed fruit peels is essential to enhance its adsorptive properties for various applications, including water and wastewater treatment, dye effluent processing, and juice clarification in the food and sugar industries. This optimization process typically involves identifying the ideal activation conditions and the best combination of fruit peel types to achieve the desired surface area and adsorption capacity. For this study, maximizing the yield of fruit peel waste is critical for producing nano activated carbon (NAC), making it a key factor in selecting the raw material. The study indicated that chemical activation yields higher carbon output than physical activation.29 Consequently, this study employed chemical activation with 40% H3PO4, which is expected to yield greater carbon and thus enhance NAC production.This section focused on determining the optimal proportions of powdered fruit peel waste, with carbon yield as the dependent variable. The study investigates the impact of varying mixing ratios of peel powders on carbon yield. The independent variables, proportions of mixed peels, and the corresponding predicted and experimental yields are summarized in Table 1.
The experimental results closely align with the predicted values, indicating strong compatibility. As shown in Table 1, the carbonized matter yield from mixed peels (carbonized at 550 °C for 30 min) ranged from 66.05 ± 0.62% to 74.92 ± 0.34%. The carbon yield percentages from the experimental runs are statistically significant (p-value = 0.0019). Among the experiments, run 12, with a fruit peel proportion (g) of 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 43.5
43.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 43.5 (avocado, banana, and papaya, respectively), yielded the lowest carbon percentage at 66.05 ± 0.62%. In contrast, run 9, with a proportion (g) of 43.5
43.5 (avocado, banana, and papaya, respectively), yielded the lowest carbon percentage at 66.05 ± 0.62%. In contrast, run 9, with a proportion (g) of 43.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13
13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 43.5, produced the highest carbon yield of 74.92 ± 0.34%.
43.5, produced the highest carbon yield of 74.92 ± 0.34%.
This variation is likely attributed to the higher carbon content in avocado peels, which is 46.16 ± 0.32%,42 surpassing that of banana and papaya peels at 39.08% and 50.8%, respectively, as reported in previous studies.43,44 The findings suggested that the mixed peel waste used in this study yields higher carbon outputs than the individual peel wastes examined earlier, potentially due to the synergistic effects of the carbonized matter and the specific processing methods and conditions employed. Additionally, different fruit peels contain various forms of organic carbon, enhancing their biosorptive properties and improving the surface chemistry (e.g., electrostatic attraction) of NAC, thereby contributing to significant waste reduction in actual waste treatment scenarios.45
The results were analyzed through analysis of variance (ANOVA), as detailed in Table S3. The model yielded an F-value of 512.50 and a corresponding p-value of 0.0019, which is lower than the 0.05 threshold, indicating that the regression model is statistically significant. This implies there is only a 0.019% probability that such a high F-value could arise from random noise. The linear variables (A, B, C) within the group showed significant differences, as evidenced by their low p-values (p < 0.05). Additionally, the interaction terms (AC and BC) also demonstrated significant differences (p < 0.05). The lack-of-fit F-value was 4.11 and non-significant, while the low pure error reflects the high precision of the experimental yield, as shown in Table S3. The non-significance of the lack-of-fit test suggests that the independent variables adequately account for the variability in the response variable.46
Moreover, this study assessed the model's goodness-of-fit using several metrics: the coefficient of determination (R2), the adjusted R2, the predicted R2, the coefficient of variation (CV), and the signal-to-noise ratio (S/N). The high R2 value of 0.9996 and adjusted R2 of 0.9976, presented in Table 1, indicate a strong representation of the relationship between the response and independent variables, reflecting the model's high precision and reliability. The low CV value of 0.1676 further supports the consistency and reliability of the experimental results. The predicted R2 of 0.9469 suggests only a 0.27% chance that the lack-of-fit F-value could result from noise. Furthermore, the predicted R2 aligns well with the adjusted R2, as the difference between them is less than 0.2. Adequate precision, which measures the signal-to-noise ratio, was 72.6031, indicating excellent model fit.46
| S. no | Avocado peel (g) | Banana peel (g) | Papaya peel (g) | Yield (%) | Desirability | |
|---|---|---|---|---|---|---|
| 1 | 51.92 | 21.64 | 26.44 | 75.87 | 1.0000 | Selected |
| 2 | 50.80 | 20.20 | 29.00 | 75.64 | 1.0000 | |
| 3 | 49.80 | 29.40 | 20.80 | 75.56 | 1.0000 | |
| 4 | 50.00 | 19.40 | 30.60 | 75.40 | 1.0000 | |
| 5 | 49.30 | 24.70 | 26.00 | 75.37 | 1.0000 | |
Among these five optimized conditions, the most favorable (sample number 1) included mixture of avocado, banana, and papaya peels of 51.92 g, 21.64 g, and 26.44 g, respectively, with a carbon yield of 75.87% and a desirability value of 1.0000. This desirability score indicates that the model developed for predicting carbon yield is highly accurate and desirable.
3.2. Optimization of processing conditions for the synthesis of NAC using mixed fruit peels by the quadratic regression model
Generally, the combined nature of avocado, papaya, and banana peels enriches NAC with diverse lignocellulosic and mineral compositions, generating hierarchical porosity and varied functional groups. This synergy enhances surface reactivity, adsorption capacity, and structural stability, yielding a more efficient, selective, and sustainable nano activated carbon compared to single-biomass precursors.The optimization of various parameters, treated as independent variables, included the impregnation ratio (with 1–2 levels of the MFP activating agent and H3PO4), carbonization temperature (ranging from 400 to 700 °C), and heating time (40–100 min). This optimization process, detailed in Table 3, aimed to achieve optimal conditions for NAC synthesis, resulting in high fixed carbon content and surface area.
| Run | Independent variables | Dependent variables | |||
|---|---|---|---|---|---|
| Impregnation ratio (%) | Temperature (°C) | Time (min) | Fixed carbon contents (%) | Surface area (m2 g−1) | |
| a Triplicate experiments were performed (n = 3). All values are presented as mean ± standard deviation; mean values (in the column) with different superscripts are significantly different (p < 0.05). | |||||
| 1 | 33.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 66.7 66.7 |
700 | 100 | 76.61 ± 0.53c | 390.02 ± 0.40e |
| 2 | 33.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 66.7 66.7 |
400 | 100 | 81.44 ± 0.43a | 565.44 ± 0.50a |
| 3 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50 |
700 | 40 | 60.50 ± 0.60f | 224.66 ± 0.40i |
| 4 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50 |
400 | 70 | 80.20 ± 0.50a | 320.03 ± 0.79f |
| 5 | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 75 75 |
550 | 70 | 78.54 ± 0.52b | 409.45 ± 0.39c |
| 6 | 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60 60 |
550 | 70 | 68.30 ± 0.20d | 143.08 ± 0.04j |
| 7 | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 75 75 |
338 | 70 | 65.36 ± 0.60e | 290.07 ± 0.51g |
| 8 | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 75 75 |
762 | 28 | 68.09 ± 0.70d | 240.44 ± 0.43h |
| 9 | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 75 75 |
550 | 112 | 68.51 ± 0.40d | 393.48 ± 0.51d |
| 10 | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 75 75 |
550 | 70 | 78.16 ± 0.60b | 408.54 ± 0.41c |
| 11 | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 75 75 |
550 | 70 | 78.26 ± 0.60b | 408.84 ± 0.41c |
| 12 | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 75 75 |
550 | 100 | 77.85 ± 0.40bc | 443.02 ± 0.41b |
| ANOVA summary: regression coefficient, R2 | 0.9956 | 0.9958 | |||
| Adj. R2 | 0.9941 | 0.9943 | |||
| Pre. R2 | 0.9914 | 0.9933 | |||
| CV% | 0.702 | 2.68 | |||
| Adeq. precision | 78.3959 | 87.1136 | |||
The results presented in Table 3 were analyzed using the Response Surface Methodology with a Central Composite Design (RSM-CCD), as implemented in Design-Expert software (v.13.0.5.0, USA). The analysis showed significant variations in the responses (p < 0.05), as indicated in Table 3.
In experimental run two, the highest fixed carbon content (81.44 ± 0.43%) and surface area (565.44 ± 0.50 m2 g−1) were observed, achieved with a 33.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 66.7 impregnation ratio (%), a carbonization temperature of 400 °C, and a carbonization time of 100 minutes. Conversely, the lowest fixed carbon content and surface area, 60.50 ± 0.6% and 143.08 ± 0.04 m2 g−1, were recorded in runs 3 and 6 under different experimental conditions, as detailed in Table 3.
66.7 impregnation ratio (%), a carbonization temperature of 400 °C, and a carbonization time of 100 minutes. Conversely, the lowest fixed carbon content and surface area, 60.50 ± 0.6% and 143.08 ± 0.04 m2 g−1, were recorded in runs 3 and 6 under different experimental conditions, as detailed in Table 3.
The results process parameters, including interaction effects, were analyzed using variance analysis (ANOVA), with findings presented in Table S4. The model p-values for the responses of fixed carbon content (FCC) and surface area (SA) were 0.0008 and 0.0103, respectively, indicating statistical significance and revealing insights into interaction patterns.48,49 Notably, interaction terms (AB, AC, and BC) demonstrated significance with p-values below 0.05 for both FCC and SA, as shown in Table S4.
The model F-values for FCC (1227.41) and surface area, SA (96.15) suggested that the quadratic model employed is significant. A high F-statistic coupled with a low p-value indicates that the regression model is likely statistically significant, suggesting a good fit for the data and a significant relationship between the independent and dependent variables. Table S4 illustrated that the F-values for most linear interactions and quadratic terms were notably high, with p-values below 0.05, suggesting the model equation effectively explains the responses.48,49
However, the term A (impregnation ratio) was found to be non-significant (p-value = 0.3237) for FCC, whereas in the case of SA, terms A (p-value = 0.2754) and B2 (p-value = 0.2261) were also non-significant. The model's pure error was minimal (zero), reflecting the high precision of the experimental results for both fixed carbon content and surface area. This is further supported by the significant F-value for carbonization time (1268.59 for FCC), which was substantially greater than that for SA (64.64), as shown in Table S4, with temperature and impregnation ratio following in significance.
The results in the graphs shown in Fig. 1a and b are plotted between this study's predicted and actual values. Indicates that the actual values are distributed near the straight line, confirming the model's better fit to the experimental data. Furthermore, the results were found to be adjacent for FCC and SA. Thus, the study reveals the lowest deviation and competent agreement between the tangible and predicted data.
Fig. 1c and d describe the plots of the internal Studentized residuals versus the experimental runs. These plots are used to check the fit of the developed model to the experimental data. As shown in Fig. 1c and d, all data points were within the allowable permissible limits (−2 to +2). However, the higher limit suggested for the model (−3.59538 to +3.59538, as shown in Fig. 1c and d). The results show that the developed model fits the experimental data well.50
The perturbation plot was used to identify the most influential independent variables in the responses. The comparative influence of the studied factors on %FCC and SA at a particular design point is shown in the perturbation plot in Fig. 1e and f. In the current study, the carbonization time (factor C) primarily influences the percentage of fixed carbon and the surface area, more than the carbonization temperature and the impregnation ratios. It is revealed that the slope of the carbonization time curve is the steepest, as shown in Fig. 1e and f. In contrast, the slopes of the other independent variables in the perturbation plot are flatter, confirming that factors A and B are less significant.
In a comparative analysis, the interaction term BC outperformed the other interaction terms, AB and AC. It showed a higher NAC surface area (600 m2 g−1) and a higher fixed carbon content (>80%) at lower temperatures. The F-values for the interaction terms also corroborate this, with BC yielding higher F-values (FCC: 4589.93 and SA: 131.63) across both responses compared to AB and AC, as demonstrated in Table S4. Consequently, the optimized processing parameters and the evaluated responses for NAC synthesis align closely, indicating that the interaction term BC is particularly effective.
3.3. Validation of optimal conditions for MFP-NAC synthesis
Using Design-Expert software (v.13.0.5.0, USA), numerical optimization for maximum desirability was conducted to optimize the independent variables (impregnation ratio, carbonization temperature, and carbonization time) for the responses of FCC and SA. Based on the desirability values and the elevated levels of FCC and SA, four optimal experimental conditions were selected, as shown in Table 4. Among the four experimental runs listed in Table 4, the optimal conditions (optimized sample 1) identified were an impregnation ratio (%) between MFP and acid ratio 38.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 61.5, a carbonization temperature of 409.9 °C, and a carbonization time of 100 minutes, yielding a fixed carbon content of 89.63% and a surface area of 620.71 m2 g−1, achieving a unit desirability value.
61.5, a carbonization temperature of 409.9 °C, and a carbonization time of 100 minutes, yielding a fixed carbon content of 89.63% and a surface area of 620.71 m2 g−1, achieving a unit desirability value.
| S. no. | Impregnation ratio (%) | Temperature (°C) | Time (min) | Fixed carbon contents (%) | Surface area (m2 g−1) | Desirability | |
|---|---|---|---|---|---|---|---|
| 1 | 38.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 61.5 61.5 |
409.9 | 100 | 89.63 | 620.71 | 1.0000 | Selected |
| 2 | 38.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 61.2 61.2 |
401.4 | 98 | 89.54 | 613.57 | 1.0000 | |
| 3 | 38.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 61.7 61.7 |
403.9 | 97 | 89.04 | 611.42 | 1.0000 | |
| 4 | 37.6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 62.4 62.4 |
409.5 | 98 | 87.24 | 606.59 | 1.0000 |
The optimal process variables identified in Table 4 are clearly demonstrated and validated by the desirability contour plots in Fig. 2. The analysis indicated that the predicted FCC yield is 89.63% and the surface area is 620.71 m2 g−1, which align with the best-optimized conditions for producing NAC. To verify the appropriateness of the selected optimized conditions (impregnation ratio: 38.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 61.5; carbonization temperature: 409.9 °C; carbonization time: 100 min), experiments were conducted in triplicate. The results showed a fixed carbon content of 89.25 ± 0.38% and a surface area of 650.87 ± 0.26 m2 g−1, which closely matched the software's predictions.
61.5; carbonization temperature: 409.9 °C; carbonization time: 100 min), experiments were conducted in triplicate. The results showed a fixed carbon content of 89.25 ± 0.38% and a surface area of 650.87 ± 0.26 m2 g−1, which closely matched the software's predictions.
 | ||
| Fig. 2 The contour plots for the best-optimized variables of the interaction terms AB (i), AC (ii), and BC (iii) of the surface area and fixed carbon contents were obtained to prepare NAC. | ||
The reproducibility of the mixture of peels described as the use of mixed fruit peels (avocado, banana, and papaya) introduces compositional variability due to differences in lignin, cellulose, and ash content. However, reproducibility is ensured through: (i) controlled mixing ratios optimized via RSM, (ii) standardized preprocessing (drying, grinding, sieving), and (iii) fixed activation conditions. Although minor variability between fruit batches may occur, the strong statistical agreement between predicted and experimental values (R2 = 0.9996) confirms high reproducibility. Additionally, synergistic interactions among the different peel types enhance consistency in adsorption performance compared to single-source biomass. This also demonstrates the robustness of the method against batch-to-batch variability due to process optimization.
3.4. Physicochemical properties of MFP and NAC
Analyses of raw materials (MFP) and synthesized NAC were conducted to evaluate several physicochemical characteristics, including moisture, volatile matter, ash, and fixed carbon content of the optimized NAC, as shown in Table 5. Additionally, the percentage yield of synthesized NAC, the point of zero charge, and the surface area were assessed.| Parameters (%) | Obtained results | |
|---|---|---|
| Raw material, MFP | Prepared NAC | |
| a Data expressed as means ± standard deviation of triplicate experiments (n = 3). Mean values within the same row that have different superscripts indicate statistical significance (p < 0.05). NAC refers to NAC. | ||
| Moisture | 4.62 ± 0.03a | 3.43 ± 0.32b |
| Ash | 11.23 ± 0.26a | 4.38 ± 0.48b |
| Volatile content | 73.89 ± 0.06a | 4.94 ± 0.05b |
| Fixed carbon content | 10.28 ± 0.11b | 89.25 ± 0.38a |
| The yield of NAC (%) | — | 74.86 ± 0.01 |
| Surface area (m2 g−1) | — | 650.87 ± 0.26 |
At pH values below the pHpzc (pH < 2.01), the NAC surface carries a positive charge due to the high concentration of H+. This condition enhances the adsorption of anionic impurities, organic molecules, and colorants from aqueous solutions, such as cane juice, beverages, and wastewater, due to increased electrostatic attraction. Conversely, at pH values above the pHpzc (pH > 2.01), the NAC surface becomes negatively charged, leading to competition with positively charged cations present in the solution. This competition can fill the adsorbent's available adsorption sites, reducing the uptake of contaminants from polluted aqueous media.
3.5. Characterization of MFP and NAC
 | ||
| Fig. 4 FTIR spectrum of mixed avocado, banana, and papaya peel powder (a), and NAC prepared from the mixed fruit peel powder under optimized process conditions (b). | ||
3.5.1.1. FTIR analysis of MFPs. Fig. 4a illustrates the FT-IR spectrum of MFP, which displays notable bands at 3285 cm−1, 2921 cm−1, 2853 cm−1, 1738 cm−1, 1605 cm−1, 1373 cm−1, 1234 cm−1, 1022 cm−1, 774 cm−1, and 524 cm−1. The broad band around 3285 cm−1 corresponds to O–H stretching, indicating the presence of alcoholic and phenolic –OH functional groups in MFP.61
The bands at 2921 cm−1 and 2853 cm−1 represent a doublet of C–H stretching (–CH2) groups, reflecting symmetric and asymmetric stretching modes typically associated with carbohydrates (polysaccharides) and lipids.62 The bands observed at 1738 cm−1, 1605 cm−1, and 1373 cm−1 are attributed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching of aldehydes or carboxylic acids (–COOH), asymmetric stretching of carboxylic C
O stretching of aldehydes or carboxylic acids (–COOH), asymmetric stretching of carboxylic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, and O–H bending, respectively.63,64 Additionally, the band at 1234 cm−1 is indicative of aromatic C
O, and O–H bending, respectively.63,64 Additionally, the band at 1234 cm−1 is indicative of aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching.65 The peaks at 524 cm−1 are linked to Si–O stretching and Si–O bending, signifying the presence of silica.66 The band at 1022 cm−1 is associated with C–N and C–O stretching from alcohol and lignin. In the fingerprint region (1200–800 cm−1), characteristic symmetric stretching bands of typical carbohydrates, including C–C, C–OH, and C–O–C groups, are revealed.67
C stretching.65 The peaks at 524 cm−1 are linked to Si–O stretching and Si–O bending, signifying the presence of silica.66 The band at 1022 cm−1 is associated with C–N and C–O stretching from alcohol and lignin. In the fingerprint region (1200–800 cm−1), characteristic symmetric stretching bands of typical carbohydrates, including C–C, C–OH, and C–O–C groups, are revealed.67
3.5.1.2. FTIR analysis for NACs. The FTIR spectra of MFP and NAC, as presented in Fig. 4a and b, reveal notable differences. It appears that certain functional groups are diminished in NAC, which is derived from MFP via carbonization and phosphoric acid activation at elevated temperatures. Additionally, the carbonization and activation process leads to the release of less stable volatile compounds, primarily oxygen-containing derivatives, thereby increasing the spacing between carbon atoms.68 In Fig. 4b, the most prominent and broad peak at 3343 cm−1 corresponds to the stretching vibrations of hydroxyl (–OH) groups associated with alcohols, adsorbed water, and phenols. The lignin component, which possesses an aromatic structure, undergoes degradation and is reconfigured into various compounds featuring C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds in NAC.25 As depicted in Fig. 4b, the strong and pronounced peak at 1575 cm−1 signifies the presence of C
C bonds in NAC.25 As depicted in Fig. 4b, the strong and pronounced peak at 1575 cm−1 signifies the presence of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds, while the broad peak at 893 cm−1 is attributed to nitrogen-containing bioligands, as corroborated by existing literature.64
C bonds, while the broad peak at 893 cm−1 is attributed to nitrogen-containing bioligands, as corroborated by existing literature.64Overall, the FTIR spectra indicate that the acid oxidation process enhanced the NAC surface by increasing the number of oxygen-containing functional groups, such as carboxyl and hydroxyl groups. These functional groups can bind charged ions via biosorption. The FTIR analysis further indicates that NAC derived from MFP is rich in hydroxyl and carboxylic groups, likely originating from hemicellulose, cellulose, lignin, and amines.69 These functional groups (–COOH, –NH2, –OH, etc.) on the adsorbent's surface can form hydrogen bonds with other molecules, facilitated by the attractive electrostatic interactions between the hydrogen atoms in these functional groups. This characteristic allows the NAC to attract and bind negatively charged impurities and activated carbon.
In this study, Fig. 5a and b illustrate the XRD patterns for MFP and NAC, respectively. The XRD pattern of the precursor MFP in Fig. 5a showed peaks that suggest an amorphous character of the raw materials. The cellulose XRD pattern, shown in Fig. 5a, exhibits well known diffraction peaks at approximately 16.5° and 22.5°, corresponding to the (110) and (200) planes, respectively. These findings aligned with the characteristic diffraction peaks expected for cellulose, attributable to the heterogeneous composition of MFP, which includes complex matrix components like proteins, lipids, and carbohydrates.
The analysis revealed that both strong and weak diffraction peaks at 2θ = 24.28° and 2θ = 44.3°, corresponding to interplanar spacings D 002 and D 100, respectively, indicate the presence of graphite crystallites within the activated carbon derived from peel.70 Additionally, a prominent peak at 2θ = 24.28° is noted in Fig. 5b, suggesting the partially crystalline nature of the synthesized NAC.11 This transition is likely due to the carbonization process, during which the sample is subjected to high temperatures, leading to the loss of its amorphous characteristics as most organic components are eliminated.69,71 The crystalline size of the synthesized NAC was determined using the standard material shape tool in ImageJ, yielding an average crystalline size of 24.46 nm.
Further, the effectiveness of the activated carbons obtained from mixed fruit peels, supported by literature,23,72 accordingly, prepared NAC typically exhibits turbostratic carbon, characterized by randomly oriented, poorly stacked graphene layers that generate broad (002) and (100) XRD reflections and enlarged interlayer spacing compared with crystalline graphite. This disorder arises from biomass heterogeneity and chemical activation, which disrupt aromatic-layer alignment and promote defect-rich, porous structures favorable for adsorption. However, crystalline sizes estimated using the Scherrer equation are only semi-quantitative for such materials, as peak broadening also reflects lattice strain, rotational disorder, and amorphous content rather than true domain size alone.23,73
 | ||
| Fig. 6 SEM image of raw MFP (a) and synthesized NAC before clarification (b), and after clarification (c) at different magnifications. | ||
The SEM image of NAC in Fig. 6b shows numerous irregularly shaped pores, which is advantageous because they provide a larger surface area for adsorption. The activated carbon produced in this study exhibits a morphology comprised of nano-sized spherical particles distributed across its surface, with an average particle diameter of 24.46 nm, as indicated in Fig. 6b. This finding aligns with the observations made in earlier,74 who noted that chemical and thermal modifications impact the surface morphology of activated carbon. Additionally, this research builds upon previous findings reported,75 which demonstrated that cellulose-based materials can yield activated carbon with irregular (amorphous) and non-uniform particle structures, including nano-sized particles. Thus, the activated carbon examined here is confirmed to be nanoscale.
Additionally, the particle size distributions of MFP and NAC are illustrated in Fig. 6. The particle size distribution of activated carbon was estimated using ImageJ software, assuming irregular particle shapes. This distribution was calculated from particle areas, following the methodology outlined.76 The particle size distribution for MFP ranged from 0 to 300 µm, while that for NAC ranged from 10 to 60 nm, as depicted in Fig. 7a and b, respectively. The peak particle sizes for MFP and NAC were observed at 50–100 µm and 20–30 nm, respectively. These findings were further validated by the SEM images presented in Fig. 6. Based on SEM analysis, the researchers inferred the particle size distributions of the samples. They plotted the corresponding curves in Fig. 7. The average MFP diameter was determined to be 64.66 µm. At the same time, MFP-NAC in Fig. 7b exhibited a range of surface areas and an average particle size of 24.46 nm. This difference is attributed to the treatment of MFP with phosphoric acid, pyrolysis, and grinding.
 | ||
| Fig. 7 Size distribution of MFP (a) and NAC (b), with an average particle size of 64.66 µm and 24.46 nm, respectively, using Image J software. | ||
The results of this study on nano activated carbon can be compared with those of similar research, as summarized in Table 6. According to the current study, the nano activated carbon produced exhibits impressive characteristics, with a specific surface area of 650.71 m2 g−1 and a nanoparticle size of 24.46 nm. These measurements are favorable compared with those reported for activated carbon derived from fruit peel waste in related studies that employed low-temperature physical and chemical activation methods. Moreover, the approach taken in this study is both cost effective and environmentally friendly, as it avoids the use of chemical reagents and utilizes mixed fruit peel waste, a type of market waste, as the precursor. One significant and expanding application of activated carbon, particularly in its nano-sized form, is its effectiveness as an adsorbent. As such, the nano activated carbon produced in this study has the potential to efficiently remove heavy metals, dyes, and various organic pollutants or impurities from aqueous solutions.9,25,28,29,56,64,77
| Raw material | Activating agents | Surface area (m2 g−1) | Applications | References |
|---|---|---|---|---|
| Annatto peel | H3PO4 | Not reported | — | 9 |
| Banana peel | ZnCl2 | Not reported | Reducing the air pollutants (mainly CO) from the motorcycle muffler | 25 |
| Lemon/citrus peel | ZnCl2/KOH | 850–1100 | Water purification | 56 |
| Avocado peel | — | 87.52 | Removal of naphtha blue black, reactive black 5 dye, and basic blue 41 dye | 28 |
| Orange peel | ZnCl2 | 1228.2 | Removal of 99.12% Cr(VI) from aqueous medium | 29 |
| Papaya peel | 1 M H3PO4 | — | Removal of Pb2+ from aqueous medium | 64 |
| Pineapple peel | H3PO4/KOH | 850–1200 | Aqueous dye adsorption | 77 |
| Mixed peels: banana + papaya + avocado peels | H3PO4 | 650.87 ± 0.26 | Prepared nano activated carbon can be used as an adsorbent to remove dyes, heavy metals, and other organic impurities from aqueous media, reduce CO gas emissionsetc. | Present study |
In the preparation of nano activated carbon (NAC) from mixed fruit peel wastes using phosphoric acid activation, wastewater is mainly generated during the post-activation washing step. This effluent contains residual H3PO4, dissolved organic matter, and phosphate ions, with volumes typically proportional to the pH-neutralization requirements of the wash. Proper treatment or reuse is necessary to minimize environmental impact.
The NAC surface coverage likely results from the adsorption of non-sugar molecules from cane juice, potentially forming a uniform monolayer. This is evidenced by the appearance of a white layer (referred to as a molecular cloud) of consistent thickness and spread, as observed in the SEM image of NAC used for cane juice clarification in Fig. 6c. This observation aligns well with previous literature.77 Fig. 6c highlights the significant changes on the NAC surface, indicating that impurities have been adsorbed onto functional groups within the pores, a finding further supported by FTIR analysis following cane juice clarification, as shown in Fig. 6b using NAC as the adsorbent. Additionally, the pores on the NAC surface appear to be blocked, with the adsorbent particles aggregating. After the clarification process, the average particle diameter of NAC increased to 45.12 nm, as shown in Fig. 6c. This increase can be attributed to NAC's tendency to agglomerate, consistent with previous studies.30
3.6. Yield of sugarcane juice
The quality of the sugarcane and the efficiency of the juice extraction process significantly influence the juice yield. High-quality cane typically has higher juice content and higher sugar levels. In contrast, even if lower-quality cane has a similar juice content, it tends to have reduced sugar levels, especially if harvested prematurely.79 The current study demonstrated that sugarcane can yield a substantial amount of juice, with an extraction of 74.6 ± 0.8%.3.7. Optimization of sugarcane juice clarification using NAC
Optimizing pretreatment conditions is a critical step in developing an effective, cost-efficient pretreatment method. A key objective of this optimization was to identify the precise process parameters that maximize processing benefits, such as clarification efficiency, while also assessing their relative importance. Traditional methods that analyze one factor at a time are time-consuming and overlook potential interactions between multiple factors. In contrast, response surface methodology-central composite design (RSM-CCD) provides a more effective optimization strategy by evaluating several factors and their interactions simultaneously.In this study, 21 experimental settings, including 2 central replicates and 23 total runs in Table 7, were used to optimize cane juice clarification. The process variables optimized were temperature, pH, clarification dosage, agitation speed, and time, and the responses were analyzed using Design-Expert software (v.13.0.5.0, USA). The data were fit to multiple regression quadratic polynomial models, setting the temperature, pH, time, agitation speed, and clarification amount to “in range”. For turbidity and MV, the system responses were set to “minimized”, while the conditions for color removal efficiency and settling rate were set to “maximized”.
| Run | Independent variables | Dependent variables (responses) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| A | B | C | D | E | Turbidity (NTU) | Settling rate (cm min−1) | Mud volume (mL) | Color removal efficiency (%) | |
| a Triplicate experiments were performed for each run (n = 3); A: nano activated carbon; B: pH; C: time (minutes); D: temperature (°C); E: agitation speed (rpm); all values are means ± standard deviation; mean values (along the column) share different superscripts that are significantly different (p < 0.05). | |||||||||
| 1 | 0.020 | 8 | 20 | 90 | 100 | 1833.15 ± 0.11k | 0.14 ± 0.01ghi | 6.98 ± 0.02cde | 73.35 ± 0.31g |
| 2 | 0.027 | 6 | 60 | 65 | 200 | 1399.86 ± 0.12q | 0.36 ± 0.03c | 7.56 ± 0.06bc | 90.40 ± 0.24d |
| 3 | 0.011 | 9.6 | 60 | 65 | 200 | 2816.36 ± 0.60f | 0.04 ± 002lm | 5.51 ± 0.05fgh | 67.16 ± 0.21 kl |
| 4 | 0.011 | 6 | 60 | 19 | 200 | 2733.06 ± 0.99h | 0.07 ± 01jkl | 5.81 ± 0.21 fg | 70.72 ± 0.40h |
| 5 | 0.020 | 8 | 20 | 40 | 250 | 3033.03 ± 0.20d | 0.06 ± 0.01jkl | 5.83 ± 0.11 fg | 76.54 ± 0.41f |
| 6 | 0.011 | 6 | 60 | 65 | 200 | 2133.12 ± 0.11i | 0.20 ± 0.02ef | 6.27 ± 0.21ef | 73.55 ± 0.22g |
| 7 | 0.011 | 6 | 60 | 65 | 200 | 2133.12 ± 0.11i | 0.21 ± 0.02ef | 6.27 ± 0.21ef | 73.55 ± 0.22g |
| 8 | 0.011 | 6 | 60 | 111 | 200 | 1316.54 ± 0.32r | 0.46 ± 0.01a | 7.57 ± 0.11bc | 92.22 ± 0.23b |
| 9 | 0.011 | 6 | 12.8 | 65 | 200 | 2932.39 ± 0.59e | 0.44 ± 0.01a | 5.21 ± 0.41gh | 69.25 ± 0.30i |
| 10 | 0.002 | 8 | 100 | 40 | 250 | 3166.35 ± 0.89a | 0.06 ± 0.02 kl | 4.49 ± 0.49h | 63.88 ± 0.51m |
| 11 | 0.002 | 4 | 20 | 40 | 100 | 3133.05 ± 0.62b | 0.08 ± 0.03jk | 4.86 ± 0.81gh | 66.55 ± 0.52L |
| 12 | 0.005 | 6 | 60 | 65 | 200 | 2766.39 ± 0.87g | 0.01 ± 0.02m | 4.82 ± 0.62gh | 68.35 ± 0.40ij |
| 13 | 0.020 | 4 | 100 | 90 | 100 | 1299.87 ± 0.22 s | 0.40 ± 0.02b | 7.83 ± 0.41bc | 93.27 ± 0.31a |
| 14 | 0.020 | 8 | 100 | 40 | 100 | 1833.15 ± 0.11k | 0.12 ± 0.01hi | 6.29 ± 0.31def | 77.65 ± 0.33e |
| 15 | 0.002 | 8 | 100 | 90 | 100 | 1766.49 ± 0.82l | 0.16 ± 0.02gh | 6.96 ± 0.22cde | 73.46 ± 0.41g |
| 16 | 0.002 | 4 | 100 | 90 | 175 | 1633.17 ± 0.13n | 0.17 ± 0.04 fg | 7.31 ± 0.31bcd | 77.11 ± 0.11ef |
| 17 | 0.011 | 6 | 132.8 | 65 | 200 | 1566.44 ± 0.10o | 0.11 ± 0.03ij | 7.91 ± 0.11bc | 74.05 ± 0.39g |
| 18 | 0.011 | 6 | 60 | 65 | 250 | 1683.16 ± 0.7m | 0.14 ± 0.03ghi | 7.36 ± 0.17bc | 73.26 ± 0.22g |
| 19 | 0.011 | 6 | 60 | 65 | 175 | 1849.95 ± 0.43j | 0.07 ± 0.03jkl | 6.94 ± 0.32cde | 69.33 ± 0.37i |
| 20 | 0.020 | 4 | 20 | 90 | 250 | 1499.85 ± 0.89p | 0.32 ± 0.01d | 7.97 ± 0.27bc | 91.12 ± 0.17 cd |
| 21 | 0.002 | 8 | 20 | 90 | 250 | 3099.84 ± 0.23c | 0.07 ± 0.07kl | 4.93 ± 0.32gh | 67.81 ± 0.21jk |
| 22 | 0.02 | 4 | 100 | 40 | 175 | 1233.18 ± 0.36 t | 0.22 ± 0.021e | 8.13 ± 0.29b | 91.54 ± 0.32bc |
| 23 | 0.011 | 2.4 | 60 | 65 | 100 | 932.91 ± 0.65u | 0.40 ± 0.03b | 9.31 ± 0.31a | 93.44 ± 0.34a |
Several parameters were optimized to determine the optimal conditions for maximizing cane juice clarification efficiency. The effects of NAC dose (0.002–0.02%), pH (4–8), contact time (20–100 minutes), heating temperature (40 to 90 °C), and agitation speed (100 to 250 rpm) on clarification responses were studied. The experimental data were analyzed using RSM-CCD, facilitated by Design-Expert software (v.13.0.5.0, USA).
The experimental design and associated responses are summarized in Table 7, which demonstrates significant variability in the outcomes. Regression analysis of the independent and dependent variables was conducted using the methodology described in an earlier study.46 Results indicated significant relationships (p ≤ 0.05), as shown in Tables S5 and S6. Among the linear terms, pH emerged as the most significant factor, positively influencing the response with a coefficient of 523.10 and the highest F-value of 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 019.27. Lower pH levels within the studied range were more effective at reducing turbidity.
019.27. Lower pH levels within the studied range were more effective at reducing turbidity.
The NAC dose also showed a significant negative effect (−774.61), indicating that lower NAC doses led to increased turbidity, suggesting the need for higher NAC doses for effective clarification. A moderate NAC dose is essential for optimal results. Additionally, interactions among pH and temperature (BD), temperature alone (D), and time (C) demonstrated significant negative impacts on turbidity removal, with coefficients of −507.4, −384.92, and −528.66, respectively, highlighting their critical roles in the clarification process of sugarcane juice using NAC.
Among all the process parameters, factor A (NAC dose) emerged as the most influential across all response cases, as indicated in Tables S5 and S6. This was followed in significance by factors B (pH), C (time), and D (temperature). Notably, factor E (agitation speed) did not rank among the top five influential factors, suggesting it has the least impact on the outcomes. The F-value further supports this for factor E, which is lower than those of the other factors in Tables S5 and S6.
The analysis of variance (ANOVA) conducted on the juice clarification process parameters, detailed in Tables S5 and S6, revealed that the p-values for all regressions were less than 0.0001. This indicates that the model is statistically significant at a 99% confidence level and provides insights into the interaction patterns.80,81 The respective higher F-values for the model were 7660.89, 37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 614.13, 5062.25, and 10
614.13, 5062.25, and 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 842.68 for turbidity, settling rate, mud volume, and color removal efficiency, respectively, confirming the model's significance. Generally, a low “p” value for Fisher's statistic indicates a high level of significance for the regression model.
842.68 for turbidity, settling rate, mud volume, and color removal efficiency, respectively, confirming the model's significance. Generally, a low “p” value for Fisher's statistic indicates a high level of significance for the regression model.
The lack-of-fit F-values, presented in Tables S5 and S6, were 1.29, 0.2232, 0.0457, and 1.59, with corresponding lack-of-fit p-values of 0.3122, 0.6388, 0.8317, and 0.2141 for turbidity, settling rate, mud volume, and color removal efficiencies, respectively. These non-significant results indicate that the model fits well. Additionally, the model exhibited low pure error, reflecting the high precision of the experimental results, as evidenced by the residual errors that accounted for the variation in responses.
The significance of the determination coefficient for the process parameters was further validated using the Student's t-test. A higher t-value and a smaller p-value suggest greater significance in the coefficient terms. The results from the Student's t-test and p-values demonstrate that most linear, quadratic, and interaction terms were highly or moderately significant, as shown in Tables S5 and S6, with p-values below 0.05. However, the interaction terms AB and C2 in mud volume displayed p-values of 0.1299 and 0.3590, respectively, indicating non-significance. In terms of colour removal efficiency, the terms CE (p-value: 0.3506) and AC (p-value: 0.9532) were also non-significant.
In the present study, the sharp turbidity reduction observed in optimized runs highlights the effectiveness of fruit-peel-derived NAC in destabilizing colloids and adsorbing suspended impurities via surface charge interactions and porous adsorption. Compared with high-turbidity control-like runs (minimal clarification effect), optimized NAC conditions achieved >65–70% turbidity reduction in Fig. 8, indicating a clear practical advantage. The low SEM values (0.5 NTU in most cases) indicate statistically reliable performance, supporting NAC as a robust clarifier for cane juice applications.
Similarly, removal efficiency upon NAC-assisted cane juice clarification ranged from 63.88 ± 0.51% to 93.44 ± 0.34% in Fig. 8, indicating strong sensitivity to operating conditions. The highest decolorization was achieved in run 23 (93.44%), closely followed by runs 13 (93.27%), 8 (92.22%), 22 (91.54%), and 20 (91.12%), demonstrating the excellent performance of NAC under optimized variable conditions. Studied results found to be narrow SD and SEM bars across all experiments confirm high analytical precision and reproducibility.
The high color removal efficiencies observed in optimized runs highlight the strong adsorptive affinity of fruit-peel-derived NAC for melanoids, phenolics, and colored colloids in cane juice. Enhanced decolorization correlated with improved settling rates and higher mud volumes, indicating effective floc formation and impurity capture. Experimental runs with lower color removal exhibit control-like behavior, underscoring the importance of appropriate NAC dosage and process conditions. The consistently low SEM values (0.35%) demonstrate statistically reliable performance, confirming that NAC provides a robust, reproducible, and efficient alternative to conventional clarifiers for cane juice decolorization.
3.7.2.1. Effect of NAC dosage and pH on juice clarification. The impact of NAC dosage and pH on the clarification of sugarcane juice was examined by varying NAC concentration (0.002% to 0.02%) and pH (4 to 8), while keeping other experimental variables constant. Fig. 9a and b present the 3D response surface and contour plot, illustrating the interaction between NAC dosage and pH in the clarification process. The results indicate that increasing the NAC dosage from 0.002% to 0.02% increases the clarification efficiency. This improvement is likely due to the increased availability of adsorption sites and surface area provided by the higher NAC dosage, which facilitates greater adsorption of impurities (non-sugars) during cane juice clarification.
 | ||
| Fig. 9 The 3D surface and contour plot for the interaction effect of NAC dose and pH on color removal efficiency (a) and turbidity (b). | ||
In some cases, the highest NAC surface area did not guarantee the maximum colour removal or turbidity reduction because cane juice clarification is controlled not only by surface area but also by pore accessibility, surface chemistry, and flocculation behaviour. A large fraction of the surface area in micropores is more effective for decolorization. In addition, surface function groups and charge interactions at pH 4 govern impurity adsorption and floc formation. Excessively high surface area can hinder particle bridging, producing poorly settleable flocs. Consequently, colour removal and turbidity reduction show a nonlinear relationship with BET surface area, with optimum performance achieved at balanced textural and surface properties rather than at maximum surface area.21,23,56
In the juice, both the surface charge of the NAC and the degree of ionization of certain impurities are influenced by pH. The pH level of the juice affects the adsorption of various ions, and the experiments were conducted within a pH range of 4 to 8. As depicted in Fig. 9a, the pH also plays a significant role in the clarification of cane juice.
The effectiveness of clarification, as shown in Fig. 9a, decreased as the solution pH increased from 4 to 8. This decline in efficiency may be attributed to increased surface attraction of the adsorbent or to an excess of H+ ions, which promote charge interactions at the adsorption sites, thereby improving clarification in an acidic medium. Additionally, at lower pH levels, the reduction in negatively charged sites may strengthen electrostatic attraction, further enhancing clarification efficiency. Conversely, Fig. 9b indicates that turbidity was higher at lower NAC dosages and elevated pH, likely due to insufficient surface area at lower NAC concentrations and the greater influence of −OH ions.
Also, the study highlights that, unlike conventional clarifiers such as lime or phosphoric acid, which primarily remove impurities via coagulation and precipitation by forming insoluble salts, NAC derived from fruit peels primarily functions through adsorption onto its high surface area (600 m2 g−1) and mesoporous structure. This allows selective capture of melanoidins, phenolics, and colloidal particles via hydrogen bonding, π–π interactions, and electrostatic attraction, rather than bulk chemical reactions. While minor flocculation may assist settling, adsorption dominates, enabling lower sludge volumes and residues, demonstrating a novel, sustainable alternative for cane juice treatment.23,56
3.7.2.2. Effect of NAC dosage and contact time on cane juice clarification. Fig. 10a shows that clarification efficiency increases to over 90% with higher NAC dosage and extended time, up to 60 min. This improvement suggests that a greater NAC dosage provides a larger surface area, enhancing the availability of adsorption sites for impurities. A similar pattern has been reported in the literature regarding the removal efficiency as a function of adsorbent dosage when using activated carbon derived from tea waste.81,82
 | ||
| Fig. 10 The 3D surface and contour plot for the interaction effect of NAC dose and time on color removal efficiency (a) and turbidity (b). | ||
In general, the duration of juice retention in the clarifiers significantly impacts both the juice and its components. Juice with a high concentration of suspended matter requires a longer retention time in the clarifier. This time can vary based on the clarification method used, as different methods may result in slower settling or longer reaction times for the defecant. For the Rapi-Dorr 444 clarifier, the recommended minimum retention time is 2 h, with a maximum of 3 h83
In the current study, clarification efficiency exceeds 90% with increasing contact time, as illustrated in Fig. 10a, and then reaches a saturation point. However, beyond this optimal threshold, a slight decrease in clarification efficiency was observed. This decline may be attributed to the saturation of available sites and the reduced impurities present in the cane juice. Additionally, the experimental results demonstrate that the clarification efficiency of NAC rises significantly during the initial phase of clarification, followed by a gradual increase with extended contact time. Eventually, this increase slows down as equilibrium is approached. This behavior suggests that a substantial number of vacant surface areas are initially available for adsorption. As the adsorption contact time increases, these surfaces become occupied, leaving fewer available sites. Consequently, the remaining surface area becomes less effective at retaining impurities, slowing the process due to repulsive forces between the solute molecules and the adsorbent.
3.7.2.3. Effect of NAC dosage and heating temperature on cane juice clarification. The clarification of sugarcane juice is influenced by both NAC and temperature. In a study using 100 mL of sugarcane juice, the effects of temperature ranging from 40 to 100 °C and NAC concentrations from 0.002% to 0.2% on juice clarification were examined in Fig. 11. As depicted in Fig. 11, the efficiency of clarification with NAC improves as the temperature increases. This enhancement may be attributed to the reduced viscosity of the cane juice at higher temperatures.
 | ||
| Fig. 11 The 3D surface and contour plot for the interaction effect of NAC dose and temperature on turbidity (a) and color removal efficiency (b). | ||
Elevated temperatures facilitate a greater diffusion rate of adsorbate molecules through the external boundary layer and into the internal pores of the adsorbent particles. As the temperature rises, the kinetic energy of the sugarcane juice increases, thereby enhancing NAC's effectiveness in the clarification process. The temperature rise not only increases the number of active sites available on the NAC but also alters the clarifying agent's ability to reach equilibrium with impurities present. The endothermic nature of this reaction is evident as well. Furthermore, the interaction between the clarifying agent and contaminants may create new active sites. At the same time, the accelerated intraparticle diffusion of clarifying molecules into the NAC pores contributes to the overall improvement in clarification capacity.
3.7.2.4. Effect of NAC dosage and agitation speed on clarification of cane juice. Using an initial juice volume of 100 mL, the effects of NAC dosage and agitation speed were examined by varying NAC concentration from 0.002% to 0.02% and agitation speed from 100 to 250 rpm. The interaction between NAC dosage and agitation speed on the clarification of sugarcane juice is illustrated in a 3D contour plot, as presented in Fig. 12.
 | ||
| Fig. 12 The 3D surface and contour plot for the interaction effect of NAC dose and Agitation speed on turbidity (a) and color removal efficiency (b). | ||
This plot indicates that higher agitation speeds facilitate the time required for the adsorbate to achieve the desired equilibrium. However, prolonged agitation reduces clarity by occupying active sites on the adsorbent. Notably, an agitation speed of around 100 rpm was found to positively influence clarity. Conversely, as agitation speed increased further, the clarification process's effectiveness diminished, likely due to increased interactions between the contaminants and the adsorbent surface.
 | ||
| Fig. 13 Ramp desirability plots for the optimization of factors and responses for cane juice clarification using NAC under RSM. | ||
In the current study, the operating pH of 4 was found to clarify cane juice using NAC, whereas pHpzc was 2.0 in Fig. 3, indicating that the adsorbent surface becomes negatively charged at pH 4, favoring electrostatic attraction with cationic pollutants while repelling anions. Optimal clarification at pH 4 is described by a synergistic mechanism: π–π (n–π) interactions between aromatic domains and adsorbates, and hydrogen between surface –OH/–COOH groups and polar molecules. Thus, adsorption efficiency arises from a combination of electrostatic attraction and non-electrostatic forces, ensuring effective contaminant removal under mildly acidic conditions.85,86 The suitable adsorption mechanism, as shown in this study in Fig. 14.
3.8. Physicochemical characteristics of raw cane juice, clarified juice, and produced raw sugar
Essential quality assessments are crucial, as they can reveal distinct patterns and characteristics of sugar, including purity (measured by polarization), color, humidity, and the presence of insoluble contaminants (impurities and insoluble solids). The key parameters evaluated included moisture content, ash content, Brix, Pol%, pH, reducing sugars, color, turbidity, settling rate, and mud volume for raw sugarcane juice, treated/clarified juice, and the final produced sugar. The results of these assessments are detailed in Table 8.| Studied parameters | Raw juice (untreated) | Treated (clear) juice | Produced sugar |
|---|---|---|---|
| a Data expressed as means ± standard deviation of triplicate experiments (n = 3). | |||
| Yield (%) | 74.6 ± 0.87 | 94.64 ± 0.05 | 13.15 ± 0.48 |
| BHR (%) | — | — | 85.88 ± 1.05% |
| pH | 5.4 ± 0.02 | 4.00 ± 0.01 | 6.99 ± 0.07 |
| Pol | 15.8 ± 0.05 | 15.4 ± 0.02 | 13.57 ± 0.09 |
| Brix | 19.03 ± 0.05 | 17.02 ± 0.17 | 14.12 ± 0.12 |
| Purity (%) | 83.1 ± 0.49 | 90.4 ± 0.39 | 96.18 ± 0.24 |
| Reducing sugar (%) | 0.46 ± 0.01 | 0.48 ± 0.03 | 0.38 ± 0.02 |
| Turbidity (NTU) | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 260.5 ± 0.5 260.5 ± 0.5 |
1285.78 ± 0.25 | — |
| Color (IU) | 9623.54 ± 0.24 | 470.16 ± 0.05 | 422.85 ± 0.15 |
| Settling rate (cm min−1) | 0.59 ± 0.01 | — | — |
| Mud volume (mL/100 mL) | 5.5 ± 0.03 | — | — |
| Moisture contents (%) | — | — | 0.89 ± 0.03 |
| Ash contents (%) | — | — | 0.62 ± 0.01 |
In this study, the turbidity levels of the unclarified and treated juice were measured at 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 260.5 ± 0.5 NTU and 1285.78 ± 0.25 NTU, respectively. They reported that turbidity in treated juice ranged from 4669 to 11
260.5 ± 0.5 NTU and 1285.78 ± 0.25 NTU, respectively. They reported that turbidity in treated juice ranged from 4669 to 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 534 NTU, depending on the amount of clarificant used.4 The results of this investigation indicated that the clarificant for sugarcane juice treatments exceeded expectations. The initial turbidity of the raw juice was approximately 16
534 NTU, depending on the amount of clarificant used.4 The results of this investigation indicated that the clarificant for sugarcane juice treatments exceeded expectations. The initial turbidity of the raw juice was approximately 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 260.5 ± 0.5 NTU, but after clarification, it decreased to 1285.78 ± 0.25 NTU.
260.5 ± 0.5 NTU, but after clarification, it decreased to 1285.78 ± 0.25 NTU.
The raw juice in this study, containing 0.46 ± 0.01% RS (which includes glucose, fructose, etc.), complies with ICUMSA standards. Additionally, the reducing sugar content of the clarified juice (processed with NAC) was determined to be 0.48 ± 0.03%. There was no significant difference between the raw and clarified juices, as both met the national standard for reducing sugar content, which limits it to 0.5%. The sugar produced in this study exhibited a reducing sugar content of 0.38 ± 0.02%.
A higher RS level suggests that the selected raw material may not have been fully matured. Furthermore, inadequate control of processing temperatures can degrade monosaccharides, leading to color that may worsen during storage.95 This indicates that, in this study, the sugarcane was fully mature and that processing temperatures were maintained at optimal levels, minimizing saccharide breakdown and resulting in high-quality sugar production. The ideal RS concentration for white sugar should be between 0.20% and 0.65%.93 The RS values of the sugar samples analyzed in this study fall within the recommended range.,93 suggesting that the NAC clarification method could be a promising clarifying agent for sugar production.
Pol represents the apparent sucrose content of a substance, expressed as a percentage by mass and determined using the single or direct polarization method. One advantage of employing NAC to clarify sugarcane juice is its dual benefit of enhancing juice clarity (purity) and Brix, as the pol% reflects both the quantity and purity of sucrose in the juice. The pol and brix values of the sugar currently produced were measured at 13.57 ± 0.09 and 14.12 ± 0.12, respectively. Several variables significantly influence the importance of sugar polarization.98 Variability in processing conditions and the presence of coloring agents may contribute to the lower pol values observed, suggesting that impurities could impact this measurement. Higher pol values might indicate a greater quantity of sugar present.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 957–12712 IU, which necessitates processes such as sulphation, defecation, and carbonation for clarification.101 The color of the juice can vary depending on the chosen clarifier, as noted.4 Consequently, a method is required to render the sugar produced in this study more palatable and suitable for human consumption.
957–12712 IU, which necessitates processes such as sulphation, defecation, and carbonation for clarification.101 The color of the juice can vary depending on the chosen clarifier, as noted.4 Consequently, a method is required to render the sugar produced in this study more palatable and suitable for human consumption.The current color value is considered excessively high and must be reduced to ensure the production of high-quality sugar. However, the clarified juice color was significantly lowered to 470.78 ± 0.05 IU, demonstrating that NAC (Nano activated carbon) achieves optimal results in cane juice clarification. Given NAC's remarkable efficiency in removing color-causing substances from raw juice, it has the potential to serve as a viable alternative to chemical clarifiers commonly used in many sugar factories today. This shift to NAC could enhance the effectiveness of the clarification process, reduce reliance on imports, and promote the health of consumers of the produced sugar. The color values of the sugars synthesized in this study were measured at 422.85 ± 0.15 IU.
The purity of sugar is influenced by processing methods, the number of chemical additives used in cane juice, and the variety of sugarcane employed.104 For instance, Demerara sugar in Brazil has purity values ranging from 93.09% to 98.88%, crystal sugar purity ranges from 98.87% to 100.00%, and refined sugar values span from 93.39% to 99.70%.103 The current study determined the sugar's purity to be 96.18 ± 0.24%, categorizing it as high quality according to the ICUMSA standard.95 This high purity indicates that sulphited sugar is considerably better refined. However, the application of technology can yield sugar products with varying levels of color and turbidity.79 Therefore, the sugar produced in this study still requires additional processing steps to enhance its palatability and make it suitable for human consumption.
The sugar currently produced has an ash content of 0.62 ± 0.01% (see Table 8), which falls well within the Codex maximum limit of 3.5%.2 These findings align with the values reported.106 Additionally, the ash consumption limit set by standards in India, East Africa, and Ethiopia is 0.76%, indicating that our results are nearly within this recommended range. Consequently, the produced sugar is largely compliant with the standards of various countries and is likely to be considered acceptable. Moreover, the use of natural NAC clarificant enhances the quality of the sugar, keeping it within the recommended ash limits.
3.9. Regenerative, cost, and scalability of NAC from fruit peels
Nano activated carbon derived from fruit peel waste offers a highly attractive economic profile for industrial cane juice clarification, thanks to its low-cost, abundant feedstock and minimal reliance on harmful chemicals during preparation. Agro-waste fruit peels, such as banana, papaya, and avocado, are locally available, often considered disposal liabilities, and can be used to produce NAC, making it both cost effective and environmentally beneficial.23 Preliminary cost analyses indicate that NAC can be produced at a fraction of the cost of commercial powdered activated carbon (PAC) or granular activated carbon (GAC), particularly when utilizing simple chemical activation (e.g., phosphoric acid) and low-energy carbonization.56From a regenerative perspective, NAC demonstrates promising stability, with multiple adsorption–desorption cycles showing minimal loss in color removal efficiency or turbidity reduction, suggesting that the material can be recycled several times, reducing operational costs and waste generation. The process is scalable, as carbonization and activation units can be integrated into agro-industrial setups with continuous feedstock supply, supporting large-scale applications. Moreover, NAC's robustness under variable pH, temperature, and turbidity conditions further enhances its industrial applicability. Collectively, the combination of low feedstock cost, ease of preparation, regenerability, and scalability positions NAC as a practical, sustainable alternative to conventional clarifiers, potentially reducing both economic and environmental burdens of cane juice clarification.23,107
4. Conclusions
The preparation of nano activated carbon (NAC) involved activating H3PO4, followed by carbonization and subsequent reduction to the nanoscale. Using Response Surface Methodology – Central Composite Design (RSM–CCD), optimal synthesis conditions were determined: an impregnation ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.6 (sample-to-acid), a carbonization temperature of 410 °C, and a carbonization duration of 100 min. Characterization of the samples was conducted using FTIR, XRD, and SEM. Results from XRD and SEM indicated that the NAC particles exhibited an average size of 24.46 nm and a surface area of 650.87 ± 0.26 m2 g−1.
1.6 (sample-to-acid), a carbonization temperature of 410 °C, and a carbonization duration of 100 min. Characterization of the samples was conducted using FTIR, XRD, and SEM. Results from XRD and SEM indicated that the NAC particles exhibited an average size of 24.46 nm and a surface area of 650.87 ± 0.26 m2 g−1.
In sugar production, clarifying sugarcane juice is a crucial step. Using a clarifying agent made from MFP presents a practical, cost effective solution for this process. This study investigated the use of NAC synthesized from MFP under optimal conditions as a clarifying agent for sugarcane juice. The established optimal conditions for clarification included a NAC concentration of 0.01%, a pH of 4, a clarification time of 20 min, a temperature of 90 °C, and an agitation speed of 100 rpm. The application of MFP-NAC resulted in a turbidity of 1760.6 NTU, a settling rate of 0.9 mL min−1, a mud volume of 6.6 mL/100 mL, and a color removal efficiency of 97.4%.
Additionally, the clarified juice was processed into raw sugar, and its physicochemical properties were analyzed and found to comply with ICUMSA and Ethiopian sugar standards, demonstrating favorable outcomes. Thus, this study concludes that the low-temperature phosphoric acid-treated nanoactivated carbon is a promising adsorbent for the removal of pigments, dyes, inorganic substances, and organic impurities from aqueous solutions, as well as for reducing CO gas emissions. Furthermore, the results support the potential of MFP-NAC as an effective clarifying agent for sugarcane juice in sugar production.
Author contributions
Conceptualization, data curation, methodology, software, validation, and writing – original draft, W. R. B; supervision, conceptualization, data curation, validation, software, and writing – review and editing, G. M. F., R. D. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declare that there is no conflicts of interest regarding the publication of this paper.Data availability
The supporting data has been provided as part of the supplementary information (SI). Supplementary information: Table S1: experimental design for optimizing the fruit peel powder mixing proportion and the nano activated carbon preparation; Table S2: experimental design for the clarification of cane juice for raw sugar production; Table S3: ANOVA results for the quadratic model of percent carbon yield obtained from the mixed fruit peels (MFP) under carbonization; Table S4: analysis of variance (ANOVA) results for the fixed carbon contents and surface area on synthesized nano activated carbon (NAC) from mixed fruit peel wastes; Table S5: results of analysis of variance for the turbidity and settling rate in clarification of cane juice using NAC; Table S6: results of analysis of variance for the mud volume and color removal efficiencies in clarification of cane juice using NAC. See DOI: https://doi.org/10.1039/d6ra01408a.Acknowledgements
The authors thank the College of Natural and Computational Sciences, Arba Minch University, Department of Chemistry, for access to the laboratory facilities. Also, thank you to the Department of Materials Science and Engineering at Adama Science and Technology University for the sample characterization techniques using FTIR, XRD, and SEM.References
- G. Eggleston, Hot and cold lime clarification in raw sugar manufacture II. Lime addition and settling behavior, Int. J. Sugar Technol., 2000, 102(1221), 453–457 CAS.
- P. Patricia and R. H. Moretti, Study of clarification process of sugar cane juice for consumption. Ciênc. eTecnolgia de Alimentos, Campinas, Food Sci. Technol., 2010, 30, 776–783 CrossRef.
- W. O. S. Doherty, Improved sugar cane juice clarification by understanding calcium oxide-phosphate-sucrose systems, J. Agric. Food Chem, 2011, 59, 1829–1836 CrossRef CAS PubMed.
- T. Birhanu, M. M. Makur and R. Duraisamy, Evaluation of Okra (Abelmoschus esculentus) Powder and Nano CaO as Alternative Clarifiers for Lime in the Processing of Sugarcane Juice, Sugar Tech, 2024, 26, 446–468 CrossRef.
- A. Mani, S. Palanivel, S. Palanivel and Th. Palvannan, Optimization of orange-G dye adsorption by activated carbon of Thespesia populnea pods using response surface methodology, J. Haz. Mate, 2011, 186, 827–834 CrossRef PubMed.
- M. T. Amin, A. A. Umair and U. Manzoor, A review of removal of pollutants from water/wastewater using different types of nanomaterials, Adv. Mate. Sci. Engg, 2014, 1, 1–24 Search PubMed.
- K. Vinod, I. T. Gupta, H. Sadeg, S. Behnam and M. Behnam, Nanoparticles as an adsorbent; A positive approach for removal of noxious metal ions: A review, Sci. Technol. Develop, 2015, 34, 195–214 CrossRef.
- Z. Jianming, Z. Shang-Ru, Li. Shi and Ge. Tian, Pb (II) removal of Fe3O4-SiO2-NH2 core-shell nanomaterials prepared via a controlled sol-gel process, Chemi. Engg. J, 2013, 215, 461–471 Search PubMed.
- R. Cucun, S. Muhamad and M. A. Yohanes, Synthesis and characterization of nano activated carbon from annatto peels (Bixaorellana L.) viewed from temperature activation and impregnation ratio of H3PO4’ EKSAKTA, J. Sci. Data Anal, 2020, 20, 44–50 Search PubMed.
- J. L. Park, C. Ryu, K. S. Gang, W. Yang, Y. K. Park, J. Jung and S. Hyun, Comparison of biochar properties from biomass residues produced by slow pyrolysis at 500°C, Bioresource, Technol, 2013, 148, 196–201 Search PubMed.
- Li. Koon-Yang, Q. Hui, H. T. Feng and B. Alexander, Bacterial cellulose is a source of activated nanosized carbon for electric double-layer capacitors, J. Mate. Sci, 2013, 48, 367–376 CrossRef.
- M. E. Fernandez, G. V. Nunell, P. R. Bonelli and A. L. Cukierman, Activated carbon developed from orange peels: batch and dynamic competitive adsorption of basic dyes, Ind. Crops and Prod., 2014, 62, 437–445 CrossRef CAS.
- P. Nowicki, J. Kazmierczak-Razna and R. Pietrzak, Physicochemical and adsorption properties of carbonaceous sorbents prepared by activation of tropical fruit skins with potassium carbonate, Mate. & Design, 2016, 90, 579–585 Search PubMed.
- N. S. Razali, A. S. Abdulhammeed, A. H. Jawad, A. L. Othman, Z. A. Yosuf, T. A. Al-Duaij and O. K. N. S. Alsaiari, High-surface-area-activated carbon derived from mango peels and seeds wastes via microwave-induced ZnCl2 activation for adsorption of methylene blue dye molecules: statistical optimization and mechanism, Molecules, 2022, 27, 6947 CrossRef CAS PubMed.
- J. Darayen, W. Theerawong, N. Sriprach and C. Vukthong, Comparative study on acetic acid adsorption using activated carbon derived from banana and pineapple peels, J. Info. Syst. Engg. Mangt, 2022, 10, 472–478 Search PubMed.
- R. K. Widi, P. Setyopratomo, P. E. Danatha, V. D. Nanlohy and E. Savitri, Preparation and characterization of activated carbon from Cavendish banana (Musa acuminata) peels for ferric ions adsorption, Inter. J. Tech, 2022, 16, 1362–1374 CrossRef.
- G. Yirdaw, A. Dessie and T. A. Birhan, Optimization of process variables to prepare activated carbon from Noug (Guizotia abyssinica cass.) stalk using response surface methodology, Heliyon, 2023, 9, e17254 CrossRef PubMed.
- N. Rambabu, B. V. Rao, V. R. Surisetty, U. Das and A. K. Dalai, Production, characterization, and evaluation of activated carbons from de-oiled canola meal for environmental applications, Industrial Crops and Products, 2015, 65, 572–578 CrossRef CAS.
- A. K. Tolkou, K. N. Maroulas, D. Theologis, I. A. Katsoyiannis and G. Z. Kyzas, Comparison of Modified Peels: Natural Peels or Peels-Based Activated Carbons for the Removal of Several Pollutants Found in Wastewaters, C., 2024, 10, 22 CAS.
- J. A. Solis-Fuentes, F. Galan-Mendez, M. D. R. Hernandez-Medel, R. S. Garcia-Gomez, M. Bernal-Gonzalez, S. Mendoza-Perez and M. D. C. Duran-Dominguez-de-Bazua, Effectiveness of bagasse activated carbon in raw cane juice clarification, Food Bioscience, 2019, 32, 100437 CrossRef CAS.
- W. O. S. Doherty, C. M. Fellows, S. Gorjian, E. Senogles and W. H. Cheung, Flocculation and sedimentation of cane sugar juice particles with cationic homo- and copolymers, J. Appl. Poly. Sci, 2003, 90, 316–325 CrossRef CAS.
- K. Salelign and R. Duraisamy, Sugar and ethanol production potential of sweet potato (Ipomoea batatas) as an alternative energy feedstock: processing and physicochemical characterizations, Heliyon, 2021, 7, e08402 CrossRef CAS PubMed.
- H. Marsh and F. Rodriguez-Reinoso, Activated Carbon, Elsevier: Oxford, 2006 Search PubMed.
- A. A. Tadesse, Production and optimization of bioethanol from mixed banana and papaya peels using Saccharomyces cerevisiae, Am. J. Chem. Biochem. Engg, 2024, 8, 45–57 CrossRef.
- A. Chafidz, W. Astuti, D. Hartanto, A. S. Mutia and P. R. Sari, Preparation of activated carbon from banana peel waste for reducing air pollutant from motorcycle muffler, EDP Sci, 2018, 154, 01021 Search PubMed.
- L. A. Al Jebur and L. H. Alwan, Development of nano-activated carbon and apply it for dyes removal from water, Water Practice and Technology, 2022, 17, 297–310 CrossRef.
- C. Palma, L. Lloret, A. Puen, M. Tobar and E. Contreras, Production of carbonaceous material from avocado peel for its application as alternative adsorbent for dyes removal, Chinese J, Chemical Eng, 2016, 24, 521–528 CAS.
- A. El. Nemr, R. M. Aboughaly, S. El Sikaily, M. S. Ragab, M. S. Masoud and M. S. Ramadan, Microporous nano-activated carbon type I derived from orange peel and its application for Cr (VI) removal from aquatic environment, Biomass Conversion and Biorefinery, 2020, 2, 1–9 Search PubMed.
- J. B. Njewa, E. Vunain and T. Biswick, Synthesis and Characterization of Activated Carbons Prepared from Agro-Wastes by Chemical Activation, J. Chem, 2022, 2022, 9975444 Search PubMed.
- M. F. Elkady, M. M. Hussein and M. M. Salama, Synthesis and characterization of nano-activated carbon from elmaghara coal, Sinai, Egypt to be utilized for wastewater purification, Am. J. Applied Chem, 2015, 3, 1–7 CrossRef CAS.
- ASTM International, D.3172, D.3173, D.3174, D.3175, West Conshohocken, PA, 2004.
- O. O. Onawumi, A. A. Sangoremi and O. S. Bello, Preparation and characterization of activated carbon from groundnut and egg shells as viable precursors for adsorption, J Applied Sci. and Envir. Management, 2021, 25, 1707–1713 CAS.
- I. A. Tan, A. L. Ahmad and D. B. Hameed, Preparation of activated carbon from coconut husk: Optimization study on removal of 2,4,6-trichlorophenol using response surface methodology, J. hazardous materials, 2008, 153, 709–717 CrossRef CAS PubMed.
- R. Ganjoo, S. Sharma, A. Kumar and M. M. Daouda, Activated carbon: Fundamentals, classification, and properties, Book Chapter I, The Royal Soc. Chem, 2023, 1–12 Search PubMed.
- G. Vijayakumar, R. Tamilarasan and M. Dharmendirakumar, Adsorption, Kinetic, Equilibrium, and Thermodynamic studies on the removal of basic dye Rhodamine-B from aqueous solution by the use of natural adsorbent perlite, J. Mater. Environ. Sci, 2012, 3, 157–170 CAS.
- O. Oginni, K. Singh, G. Oporto, B. Dawson-Andoh, L. McDonald and E. Sabolsky, Effect of one-step and two-step H3PO4 activation on activated carbon characteristics, Bioresource Techn, Reports, 2019, 8, 100307 Search PubMed.
- L. M. Humphrey, G. John, M. Duncan, Local Production of high-quality cane juice and improved Jaggery products in Rang we district, Kenya, Project Report, 2011.
- C. C. Thai, Studies on the Clarification of Juice from Whole Sugar Cane Crop, Doctoral Dissertation, Queensland University of Technology, 2013 Search PubMed.
- Z. Din and G. Rasool, Physico-chemical analysis and polarization value estimation of raw sugar from refining point of view, American J. Plant Sci, 2015, 6, 1–5 CrossRef.
- ICUMSA Method Book, International Commission for Uniform Methods of Sugar Analysis. Berlin, Germany: Bartens, 2011, 128 Search PubMed.
- P. W. Sangura and C. A. Serrem, Determination of Sucrose Recovery Maximization in Nzoia Sugar Factory, Kenya, Afri. J. Edu. Sci. Technol, 2014, 2, 128–135 Search PubMed.
- L. T. Tran, M. Q. Nguyen, H. T. Hoang, H. T. Nguyen and T. H. Vu, Catalytic hydrothermal carbonization of avocado peel, J. Chem, 2022, 1–10 CAS.
- H. Zheng and L. Wang, Banana peel carbon that containing functional groups applied to the selective adsorption of Au (III) from waste printed circuit boards, Soft Nanosci. Lett, 2013, 3, 29–36 CrossRef CAS.
- N. A. Hamid and N. Z. Zulkifli, Papaya peels as source of hydro char via hydrothermal carbonization, IOP Conference Series: Earth and Envir. Sci, 2021, 765, 01200 Search PubMed.
- A. Vimalkumar, J. Thilagan, K. Rajasekaran, C. Raja and M. N. Flora, Preparation of activated carbon from mixed peels of fruits with chemical activation (K2CO3)-application in adsorptive removal of methylene blue from aqueous solution, Inter. J. Envir. and Waste Management, 2018, 22, 260–271 CrossRef CAS.
- T. Mada, R. Duraisamy and F. Guesh, Optimization and characterization of pectin extracted from banana and papaya mixed peels using response surface methodology, Food Science & Nutrition, 2022, 10, 1222–1238 Search PubMed.
- C. Guo, L. Ding, X. Jin, H. Zhang and D. Zhang, Application of response surface methodology to optimize chromium (VI) removal from aqueous solution by cassava sludge-based activated carbon, J. Environ. Chemi. Engg, 2021, 9, 104785 CrossRef CAS.
- J. Zheng, K. Wang, E. Luo, D. Wu, F. Zhu, R. Jiang, C. Su, C. Wei and G. Ouyang, Monodisperse microporous carbon nanospheres: an efficient and stable solid phase microextraction coating material, Anal. Chim. Acta, 2015, 884, 44–45 CrossRef CAS PubMed.
- M. Blachnio, A. Derylo-Marczewska, B. Charmas, M. Zienkiewicz-Strzalka, V. Bogatyrov and M. Galaburda, Activated carbon from agricultural wastes for adsorption of organic pollutants, Molecules, 2020, 25, 5105 CrossRef CAS PubMed.
- B. H. Hameed, A. T. M. Din and A. L. Ahmad, Adsorption of methylene blue onto bamboo- based activated carbon: Kinetics and equilibrium studies, J. Hazard. Mate, 2007, 141, 819–825 CrossRef CAS PubMed.
- A. A. Ayalew and T. A. Aragaw, Utilization of treated coffee husk as low-cost bio-sorbent for adsorption of methylene blue, Adsorption Sci. and Technology, 2020, 38, 205–222 CrossRef CAS.
- K. Qureshi, I. Bhatti, R. Kazi and A. K. Ansari, Physical and chemical analysis of activated carbon prepared from sugarcane bagasse and use for sugar decolorization, Inter. J. Chemi. Biomole. Engg, 2008, 1, 145–149 CAS.
- N. Nachat, P. Tobarameekul and P. Worathanakul, Activated carbon from bagasse for syrup decolorization as an alternative for waste management and the assessment of carbon footprint, Envir, Nat. Resour. J, 2014, 12, 66–73 Search PubMed.
- M. Danish, R. Hashim, M. M. Ibrahim and O. Sulaiman, Optimized preparation for large surface area activated carbon from date (Phoenix dactylifera L.) stone biomass, Biomass and bioenergy, 2014, 61, 167–178 CrossRef CAS.
- O. Ioannidou and A. Zabaniotou, Agricultural residues as precursors for activated carbon production – A review, Renewable and Sustainable Energy Reviews, 2007, 11, 1966–2005 CrossRef CAS.
- K. Y. Foo and B. H. Hameed, Preparation and characterization of activated carbon from melon (Citrullus vulgaris) seed hull by microwave induced NaOH activation, Desalination Water Treat, 2012, 47, 130–138 CrossRef CAS.
- M. Ahmedna, W. E. Marshall and R. M. Rao, Surface properties of granular activated carbons from agricultural by-products and their effects on raw sugar decolorization, Bioresource technology, 2000, 71, 103–112 CrossRef CAS.
- H. Aljohani, Y. Ahmed, O. El-Shafey, S. El-Shafey, R. Fouad and K. Shoueir, Decolorization of turbid sugar juice from sugar factory using waste powdered carbon, Appl. Water Sci., 2018, 8, 48 CrossRef.
- X. Y. Cui, F. Jia, Y. X. Chen and J. Gan, Influence of single-walled carbon nanotubes on microbial availability of phenanthrene in sediment, Ecotoxicology, 2011, 20, 1277–1285 CrossRef CAS PubMed.
- S. D. Gisi, G. Lofrano, M. Grassi and M. Notarnicola, Characteristic and adsorption capacities of low-cost sorbents for wastewater treatment: a review, Sust. Mate. Technols, 2016, 9, 10–40 Search PubMed.
- V. Viena, M. Nizar and S. Wardani, Preparation of activated carbons from banana (Musa acuminate L.) peels for carbon monoxide adsorption, Proceedings of MICoMS, 2017, 1, 381–386 Search PubMed.
- A. Ş. Yargıç, R. Y. Şahin, N. Özbay and E. Önal, Assessment of toxic copper (II) biosorption from aqueous solution by chemically-treated tomato waste, J. Cleaner Prod, 2015, 88, 152–159 CrossRef.
- S. Abbaszadeh, S. R. Alwi, C. Webb, N. Ghasemi and I. I. Muhamad, Treatment of lead-contaminated water using activated carbon adsorbent from locally available papaya peel biowaste, J, Cleaner Production, 2016, 118, 210–222 CrossRef CAS.
- J. K. Fatombi, S. A. Osseni, E. A. Idohou, I. Agani, D. Neumeyer, M. Verelst, R. Mauricot and T. Aminou, Characterization and application of alkali-soluble polysaccharide of Carica papaya seeds for removal of indigo carmine and Congo red dyes from single and binary solutions, J. Envir. Chemical Eng, 2019, 7, 103343 CrossRef CAS.
- S. Kamsonlian, C. Balomajumder and S. A. Chand, A potential of biosorbent derived from banana peel for removal of As (III) from contaminated water, Inter. J. Chemical Sci. and Applications, 2012, 3, 269–275 CAS.
- W. Jaihan, V. Mohdee, S. Sanongraj, U. Pancharoen and K. Nootong, Biosorption of lead (II) from aqueous solution using Cellulose-based Bio-adsorbents prepared from unripe papaya (Carica papaya) peel waste: Removal Efficiency, Thermodynamics, kinetics and isotherm analysis, Arabian J. Chem, 2022, 15, 103883 CrossRef CAS.
- J. Maharjan and V. K. Jha, Activated Carbon Obtained from Banana Peels for the Removal of as (III) from Water, Scientific World, 15, 2022, 145–157 Search PubMed.
- V. Tucureanu, A. Matei and A. M. Avram, FTIR spectroscopy for carbon family study, Critical reviews in analytical chemistry, 2016, 46, 502–520 CrossRef CAS PubMed.
- Z. Xie, X. Z. Gang, G. W. G. Wei, Ji. Fang, J. F. Ying, S. Z. Rong, S. Z, Z. Yan and Z. Y. Ling, Production of biologically activated carbon from orange peel and landfill leachate, and subsequent treatment technology, J. Chem., 2014, 4, 1–9 Search PubMed.
- C. Bouchelta, M. S. Medjram, O. Bertrand and J. P. Bellat, Preparation and characterization of activated carbon from date stones by physical activation with steam, J. Analy. and Applied Pyrolysis, 2008, 82, 70–77 CrossRef CAS.
- W. W. Ngah and M. M. Hanafiah, Removal of heavy metal ions from wastewater by chemically modified plant wastes as adsorbents: a review, Bioresour. Technol, 2008, 99, 3935–3948 CrossRef PubMed.
- B. D. Cullity, and S. R. Stock, Elements of X-Ray Diffraction, Prentice Hall, 2001 Search PubMed.
- S. Rigollet, T. Beguerie, E. Weiss-Hortala, G. Flamant and A. Nzihou, Synthesis of graphitic biocarbons from lignin fostered by concentrated solar energy, Scientific Reports, 2025, 15, 6418 CrossRef CAS PubMed.
- R. Ubago-Pérez, F. Carrasco-Marín, D. Fairén-Jiménez and C. Moreno-Castilla, Granular and monolithic activated carbons from KOH-activation of olive stones, Micropor. Mesopor. Mate, 2006, 92, 64–70 CrossRef.
- L. K. Shrestha, L. Adhikari, R. G. Shrestha, M. P. Adhikari, R. Adhikari, J. P. Hill, R. R. Pradhananga and K. Ariga, Nanoporous carbon materials with enhanced supercapacitance performance and non-aromatic chemical sensing with C1/C2 alcohol discrimination, Sci. and Techno. Adva. MaTerialS, 2016, 17, 483–492 CrossRef CAS PubMed.
- M. Abbas, Experimental investigation of activated carbon prepared from apricot stone material (ASM) adsorbent for the removal of malachite green (MG) from aqueous solution, Ads. Sci. & Techn., 2020, 1, 38 Search PubMed.
- C. Namasivayam and D. Kavitha, Removal of Congo Red from water by adsorption onto activated carbon prepared from coir pith, an agricultural solid waste, Dyes and Pigments, 2002, 54, 47–58 CrossRef CAS.
- C. Agarwal and A. K. Pandey, Remediation and recycling of inorganic acids and their green alternatives for sustainable industrial chemical processes, Envrion, Sci. Adv, 2023, 2, 1306–1339 RSC.
- P. I. Bekker and H. N. Stolz, Enhancement of sugar production part 1: Production of white sugar in the raw house, In Procs Afar Sug Technol Ass, 2001, 75, 341–345 Search PubMed.
- K. Ravikumar, K. Pakshirajan, T. Swaminathan and K. J. Balu, Optimization of batch process parameters using response surface methodology for dye removal by a novel adsorbent, Chemical Engineering J, 2005, 105, 131–138 CrossRef CAS.
- Z. Y. Zhong, Q. Yang, M. X. Li, K. Luo, Y. Liu and G. M. Zeng, Preparation of peanut hull-based activated carbon by microwave-induced phosphoric acid activation and its application in Remazol Brilliant Blue R adsorption, J. Indu. Crops Prod, 2012, 37, 178–185 CrossRef CAS.
- M. T. Uddin, M. A. Islam, S. Mahmud and M. Rukanuzzaman, Adsorptive removal of methylene blue by tea waste, J. Haz. Mate, 2009, 164, 53–60 CrossRef CAS PubMed.
- G. Asfaw, Optimization of Pre-liming and Sulphation pH of Cane Juice on Clarification, Doctoral dissertation, Addis Ababa University, 2015 Search PubMed.
- M. F. Majid, H. F. Zaid, C. F. Kait, K. Jumbri, L. C. Yuan and S. Rajasuriyan, Futuristic advance and perspective of deep eutectic solvents for extractive desulfurization of fuel oil: A review, J. Molecular Liquids, 2020, 306, 112870 CrossRef CAS.
- M. Kosmulski, The pH dependent surface charging and points of zero charge X. Update, Adv. in Coll. Interface Sci, 2023, 319, 102973 CrossRef CAS PubMed.
- El. H. Benaddi and M. R. Laamari, Facile synthesis of low-cost porous activated carbon and the adsorption properties: Characterization, optimization, kinetic models, and thermodynamic study, The J. Chemi. Thermo., 2026, 214, 107625 CrossRef CAS.
- K. Panpae, W. Jaturonrusmee, W. Mingvanish, C. Nuntiwattanawong, S. Chunwiset, K. Santudrob and S. Triphanpitak, Minimization of sucrose losses in sugar industry by pH and temperature optimization, Malaysian J. Anal. Sci, 2008, 12, 513–519 Search PubMed.
- H. Kassa, Handbook of Laboratory Methods and Chemical Control for Ethiopian Sugar Factories, Ethiopian Sugar Development Agency Research Directorate, Wonji, Ethiopia, 2010 Search PubMed.
- H. C. De. Whalley, ICUMSA Methods of Sugar Analysis: Official and Tentative Methods Recommended by the International Commission for Uniform Methods of Sugar Analysis (ICUMSA), Elsevier, 2013 Search PubMed.
- G. A. Abdel-Razig, I. A. Ahmed, E. E. Babiker and A. E. Yagoub, Effect of the addition of separan at different concentrations as a flocculant on the quality of sugar cane juice, Inter. J. Nutrition and Food Eng, 2010, 4, 138–141 Search PubMed.
- H. C. C. Bourzutschky, Color formation and removal-options for the sugar and sugar refining industries: a review (part 1), Sugar Industry, 2005, 130, 470–475 CAS.
- W. Doherty and D. Rackemann, Some aspects of calcium phosphate chemistry in sugarcane clarification, Inter. Sugar J, 2009, 111, 448–454 CAS.
- O. P. Chauhan, D. Singh, S. M. Tyagi and D. K. Balyan, Studies on preservation of sugarcane juice, Inter. J. food prop., 2002, 5, 217–229 CrossRef.
- K. Begum, S. Arefin, S. Islam and J. Islam, Preservation of sugarcane juice using herbal clarificant, Int. J. Nutr. Food Sci, 2015, 4, 530–534 Search PubMed.
- ICUMSA Method Book, International Commission for Uniform Methods of Sugar Analysis. Berlin, Germany: Bartens,128, 2011 Search PubMed.
- D. Oliveira, M. M. M. Esquiaveto and J. F. S. Júnior, Sugar specification parameters and their impact on the food industry, Food Sci. Technol, 2007, 27, 99–102 CrossRef.
- C. C. D. Thai, H. Bakir and W. O. Doherty, Insights to the clarification of sugar cane juice expressed from sugar cane stalk and trash, J. of Agric. Food Chem, 2012, 60, 2916–2923 CrossRef CAS PubMed.
- N. Ramadan, S. I. Samy El-Saylad, S. M. I. Darwish, E. A. Ramadan and M. A. Ahmed, Physicochemical properties and polarization value in raw and refined sugar, Egypt, Sugar J., 2022, 19, 1982–1990 Search PubMed.
- L. Rozsa, J. Rozsa and S. Kilpinen, Crystal growth and crystallization control tactics in industrial sugar crystallizers Part 1, Crystal growth, Inter. Sugar J, 2016, 118, 746–755 Search PubMed.
- G. H. Costa, I. D. Masson, L. A. Freita, J. P. Roviero and M. J. Mutton, Use of Moringa oleífera Lamarck leaf extract as sugarcane juice clarifier: effects on clarified juice and sugar, Food Science and Technology, 2014, 34, 204–209 CrossRef.
- M. Saska, B. S. Zossi and H. L. Liu, Removal of color in sugar cane juice clarification by defecation, sulfitation and carbonation, Inter. Sugar J, 2010, 112, 258–260 CAS.
- B. Andrzejewski, G. Eggleston, S. Lingle and R. Powell, Development of a sweet sorghum juice clarification method in the manufacture of industrial feedstocks for value-added fermentation products, Indus. Crops Prod, 2013, 44, 77–87 CrossRef CAS.
- A. F. Zailer, R. N. T. Vania, B. O. Eduardo, D. S. Jane and J. Selia, Telis-Romero, Rheology and fluid dynamics properties of sugarcane juice, Biochem. Engg. J, 2011, 53, 260–265 CrossRef.
- Brazil National Sanitary Surveillance: Resolution Board of National Sanitary Surveillance, 2003, 175 Search PubMed.
- P. W. Rein, L. S. M. Bento and R. Cortes, The direct production of white sugar in a cane sugar mill. Inte, Sugar J, 2007, 109, 286–299 CAS.
- P. Umesh Kumar, B. Birendra Kumar and N. Padmalochan, Green synthesis and characterization of gold nanoparticles using onion (Allium cepa) extract, World J. Nano Sci. Engg, 2011, 1, 93–98 CrossRef.
- R. C. Bansal and M. A. Goyal, Activated Carbon Adsorption, CRC Press, 2005 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |





