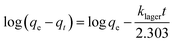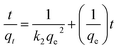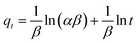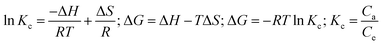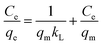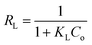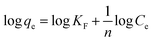Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Development of ionic polymers: novel [SPVP]TfO-graphene nanocomposites for sustainable water treatment and enhancing dye removal efficiency
Mahmoud A. Hussein *ab,
Layla R. Alfahmya,
Dina A. Bakhotmaha,
Mohamed A. Abdel-Fadeela,
Tamer S. Saleh
*ab,
Layla R. Alfahmya,
Dina A. Bakhotmaha,
Mohamed A. Abdel-Fadeela,
Tamer S. Saleh c and
Salsabeel Al-Sodies
c and
Salsabeel Al-Sodies d
d
aChemistry Department, Faculty of Science, King Abdulaziz University, P.O. Box 80203, Jeddah, 21589, Saudi Arabia. E-mail: mahussein74@yahoo.com; maabdo@kau.edu.sa
bChemistry Department, Faculty of Science, Assiut University, Assiut, 71516, Egypt
cDepartment of Chemistry, College of Science, University of Jeddah, Jeddah, 80327, Saudi Arabia
dDepartment of Chemistry, Faculty of Science, Taibah University, Al-Madinah Al-Munawarah 30002, Saudi Arabia
First published on 8th April 2026
Abstract
The synthesis and application of a novel nanocomposite material for dye removal from aqueous solutions is described in this work. The nanocomposite consisted of sulfonated poly(vinylpyrrolidonium) triflate [SPVP]TfO reinforced with graphene nanosheets (G). Various concentrations of graphene (0.2–10 wt%) were incorporated into the [SPVP]TfO matrix via in situ polymerization. The resulting nanocomposites were extensively characterized using multiple analytical techniques, including FT-IR, Raman spectroscopy, XRD, SEM, TEM, and thermal analysis, confirming the successful integration of graphene and the formation of well-defined nanocomposite structures. The adsorption performance of the [SPVP]TfO-G nanocomposites for Acid Red 1 (AR) dye removal was thoroughly investigated under various experimental conditions. Optimal adsorption was achieved at pH 2 with an adsorbent dosage of 20 mg and contact time of 90 min. Kinetic studies revealed that the adsorption process followed pseudo-second-order kinetics, while thermodynamic analysis indicated the endothermic and spontaneous nature of adsorption. The Langmuir isotherm model best described the adsorption equilibrium, with a maximum adsorption capacity of 21.96 mg g−1 for AR dye. The nanocomposites demonstrated excellent performance in removing AR dye from real water samples, including seawater, wastewater, and tap water, with removal efficiencies above 93%. In addition, the nanocomposites exhibited good reusability over four adsorption cycles, highlighting their potential as efficient and sustainable adsorbents for the removal of dye pollutants from aqueous environments.
1. Introduction
The development of efficient and sustainable methods to remove dyes from wastewater remains an important environmental challenge. Without proper treatment, dyes from various industries can contaminate water sources and pose a risk to aquatic ecosystems and human health. Recent studies have focused on the synthesis and utilization of innovative nanocomposite materials that combine the characteristics of polymers and nanomaterials. These materials are favored owing to their simplicity, cost-effectiveness, and high efficiency, which improve their adsorption capacity and selectivity.1–8In recent years, ionic polymers and their nanocomposites have garnered considerable attention from researchers due to their distinctive properties and potential uses across various domains, including water treatment and dye removal.9–14 Sulfonated ionic polymers, in particular, have emerged as promising adsorbents for eliminating organic dyes from aqueous solutions.15–18 Numerous studies have investigated the synthesis and characterization of poly(vinylpyrrolidone) (PVP) and its derivatives for various applications.19–22
Furthermore, PVP has demonstrated significant potential in water treatment and dye removal applications,23 particularly when combined with other materials to form nanocomposites or membranes.24–26 For instance, PVP has been successfully used in combination with titanium dioxide nanotubes27 and tungsten oxide nanostructures28 to develop porous nanocomposite beads for the removal of methylene blue from aqueous solutions. PVP has also been used in combination with other polymers to produce nanofibrous membranes for water filtration. For instance, polyvinylidene fluoride PVDF/PVP-Cu2O composite membranes demonstrate high photocatalytic degradation ratios for multiple dyes, including methyl orange, methylene blue, and Congo red.29
Incorporating graphene nanosheets into polymer matrices has been shown to imparts unique characteristics to the resulting nanocomposites.30–32 The mechanical strength and thermal stability of PVP have been reported to improve in graphene-reinforced PVP nanocomposites33–35 making these materials appealing for environmental remediation purposes. When combined with graphene-based materials, sulfonated PVP (SPVP) has been shown to enhance the adsorption capacity and efficiency of composite materials for various dyes, facilitating dye removal from wastewater through adsorption that follows pseudo-second-order kinetics and the Langmuir isotherm model.36–38
The selection of counter anions in the synthesis of ionic SPVP significantly influences the adsorption performance of the material in dye removal applications. Triflate anions, known as trifluoromethanesulfonate (TfO), have shown promise for dye removal, especially when integrated into ionic frameworks. Research has explored the integration of TfO anions into ionic liquids, poly(ionic liquids), and polyamide structures with various organic cations to achieve efficient and selective adsorption and elimination of harmful dyes from wastewater.39–41
The triflate (TfO−) counter-anion significantly influences the surface chemistry of sulfonated poly(vinylpyrrolidonium) (SPVP). Compared with common anions such as Cl−, BF4−, and PF6−, TfO− possesses a larger ionic radius and a delocalized charge distribution, which modifies the hydration shell and dye-binding sites of SPVP. This alteration promotes enhanced hydrophobic interactions and facilitates stronger π–π stacking and hydrogen bonding between the polymer matrix and aromatic azo and anthraquinone dyes. While electrostatic interactions remain dominant due to the sulfonate groups, the triflate anion synergistically tunes the polymer's surface environment, thereby improving dye adsorption efficiency. Recent studies have underscored the critical role of ionic polymers and nanocomposites in dye removal from contaminated water, emphasizing the importance of counter-anion selection for optimizing adsorbent performance.42–48
Although significant progress has been made, additional research is necessary to fine-tune the composition and structure of these composites for specific dye-removal scenarios. This study explored the synthesis and characterization of a new class of nanocomposite materials based on sulfonated poly(vinylpyrrolidonium) triflate [SPVP]TfO, reinforced with varying amounts of graphene nanosheets (G). This study marks the first time that the triflate anion has been examined as a counteranion within the SPVP framework.
The incorporation of both TfO anions and graphene nanostructures into the ionic polymer matrix enhances the adsorption capacity of the material. This study comprehensively characterizes materials using a range of analytical techniques, such as FT-IR, Raman spectroscopy, XRD, SEM, TEM, and thermal analysis. Furthermore, the adsorption performance of the nanocomposites for Acid Red 1 dye (AR dye) removal was assessed by examining factors such as pH, adsorbent dosage, contact time, temperature, and ionic strength. Kinetic and thermodynamic studies were conducted to elucidate the adsorption mechanism and the process efficiency. Finally, the practical applicability of the developed nanocomposites was assessed using real water samples including seawater, wastewater, and tap water.
The results of this research are anticipated to aid to the development of new and effective adsorbents for water treatment and to offer insights into the fundamental properties and behavior of ionic polymer nanocomposites enhanced with graphene nanosheets.
2. Results and discussions
2.1. Chemistry
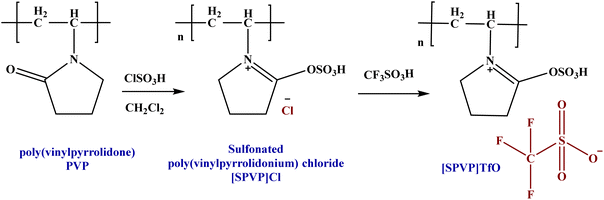
This paper details the synthesis of sulfonated poly(vinylpyrrolidonium) triflate and its nanocomposites reinforced with graphene nanosheets. The process involves two main stages: (1) the formation of the ionic polymer through the sulfonation of polyvinylpyrrolidone and anion exchange with triflate and (2) the incorporation of graphene nanosheets via an in situ polymerization method.49 Initially, [SPVP]Cl was formed using dichloromethane and chlorosulfonic acid, followed by counter-anion substitution with triflate acid to produce [SPVP]TfO (Scheme 1). This novel ionic polymer served as the base material for the subsequent nanocomposite development.The second stage focused on the integration of graphene nanosheets into the [SPVP]TfO polymer matrix. Using an ultrasonic technique, varying percentages of graphene nanosheets (0.2, 0.5, 1, 2, 5, and 10%) were incorporated into the polymer (Table 1).50,51 This resulted in a series of nanocomposites with different graphene contents. An in situ polymerization method, similar to that used for the ionic [SPVP]TfO, was employed for nanocomposite synthesis (Scheme 2). The use of ultrasound during this process ensures the uniform dispersion of the nanoparticles throughout the polymer matrix, which is crucial for achieving the desired properties in the final nanocomposite materials.
| Composite | Loading percentage of graphene nanosheets |
|---|---|
| [SPVP]TfO-G0.2% | 0.2% |
| [SPVP]TfO-G0.5% | 0.5% |
| [SPVP]TfO-G1% | 1% |
| [SPVP]TfO-G2% | 2% |
| [SPVP]TfO-G5% | 5% |
| [SPVP]TfO-G10% | 10% |
 | ||
| Scheme 2 The formulation of [SPVP] triflate loaded with different percentage of graphene nanosheets. | ||
2.2. IR and Raman analysis
FT-IR and Raman spectroscopic analyses of the newly fashioned nanocomposites revealed the successful counter anion replacement and the structural and compositional information of the G reinforcement (Fig. 1 and 2). The IR spectra of the triflate ionic polymer [SPVP]TfO-G0.2-10% exhibited characteristic vibrational bands associated with the CF3SO3− group, providing evidence of their presence in the polymer matrix. The observed stretching vibration at 1160 cm−1 for CF3 and the bending mode near 764 cm−1 confirm the incorporation of the triflate group. Additionally, the asymmetric and symmetric stretching vibrations of the SO3 group at 1226 and 1030 cm−1, respectively, further supported the successful integration of the triflate ionic polymer complex (Fig. 1).As the graphene content in the nanocomposites increased from 0.2 to 10%, notable broadening and deformation of the peaks in the ionic polymer complex was observed in the spectra. This peak broadening suggests a significant interaction between the [SPVP] network and graphene nanosheets, indicating the formation of a well-integrated nanocomposite structure.50,51 The presence of all the remaining peaks for [SPVP] and [SPVP]Cl, as confirmed in a previous study, further validates the successful synthesis of the nanocomposites.49 These spectroscopic findings provide valuable insights into the molecular structure and interactions within the newly created nanocomposites, demonstrating the effectiveness of the synthesis process for combining ionic polymer complexes with graphene nanosheets.
The incorporation of graphene nanosheets into the polymer matrix was further analyzed using Raman spectroscopy, focusing on the [SPVP]TfO-G2% nanocomposite as a representative sample (Fig. 2). The Raman spectra revealed three primary bands of interest: the G-band, D-band, and 2D-band. The G-band observed at 1560 cm−1 indicates the presence of in-plane stretching vibrations of sp2 bonded carbon atoms. This slight shift from the typical monolayer peak position of 1585 cm−1 suggested an increase in the number of graphene layers, as the peak tends to shift to lower wavenumbers with increasing layer count. The intensity of the G-band also increases with the number of layers, providing a useful metric for assessing the layer thickness.52
The 2D-band, observed at 2698 cm−1, resulted from a double resonance-enhanced two-phonon lateral vibrational process and was also sensitive to graphene folding. An I2D/IG ratio of less than 1 further confirmed the presence of multiple graphene layers in the nanocomposite. The D-band, a narrow and less intense band around 1348 cm−1, indicated a lower number of defects and reduced disorder within the carbon lattice of the polymer matrix. These Raman spectroscopy results provide valuable insights into the structural characteristics and quality of graphene nanosheets incorporated into the designed polymer nanocomposite, demonstrating the successful integration of graphene layers with minimal defects.52
2.3. Morphology study
SEM and TEM analyses of the [SPVP]TfO-G2% and [SPVP]TfO-G10% nanocomposites provided crucial insights into their structural characteristics (Fig. 3 and 4). For [SPVP]TfO-G2%, the SEM images revealed a surface composed of small asymmetrical spheres with varying porosities. These pores became more pronounced at higher magnifications, contributing to the increased surface area and enhanced absorption properties of the polymer.49 This structure suggests the potential for improved interactions with the dye molecules, facilitating their removal from the solution. However, the morphology of the [SPVP]TfO-G10% nanocomposite with higher graphene loading showed smooth black graphene sheets layered on the surface of the spheres, indicating the successful deposition and coating of graphene nanosheets. This network/layer structure, characterized by a rough and well-defined topography, is particularly advantageous for nanocomposite functionality.34,50,51 The combination of the increased surface area and the presence of graphene sheets likely enhance the capacity of the material to adsorb and remove dye species from solution, potentially improving the overall efficiency of dye removal applications.The transmission electron microscopy (TEM) analysis of nanocomposites containing 2% and 10% graphene nanosheets within the ionic polymer matrix revealed distinct morphological characteristics (Fig. 4). The images of both [SPVP]TfO-G2% and [SPVP]TfO-G10% nanocomposites exhibit an archetypal sheet-graphene morphology, characterized by a folded, film-like structure dispersed across the ionic polymer surface. This observation confirms the successful incorporation of graphene nanosheets into the network and provides insights into their distribution and interaction with the ionic matrix. A notable difference is observed between the 2% and 10% graphene-loaded nanocomposites. The higher concentration of graphene nanosheets in the 10% nanocomposite resulted in a more noticeable and prevalent sheet-like morphology compared to the 2% nanocomposite.34,50,51 This increased visibility of the graphene structures in the 10% nanocomposite suggests a greater degree of graphene dispersion and potentially enhanced interaction with the polymer matrix.
The X-ray diffraction analysis was conducted to provide insights into the integration of nanoparticles within the polymer matrix and the crystalline characteristics of the resulting nanocomposites (Fig. 5). All synthesized ionic polymers exhibited an amorphous microstructure, as evidenced by their crystallographic patterns. A broad diffraction peak was observed in the XRD spectra between 16° and 26°, corresponding to the (002) plane associated with the d-spacing between graphitic layers.49,53 The intensity of this peak increased progressively as the graphene content increased from 0.2% to 10%, indicating a correlation between the graphene concentration and structural properties of the nanocomposites.54 Interestingly, nanocomposites containing 10% nanostructure displayed a crystalline peak at 21°, suggesting that higher graphene loading influences the semicrystalline structure of ionic polymer nanocomposites. This observation implies that the incorporation of graphene at higher concentrations may induce some degree of crystallinity in the otherwise amorphous polymer matrix.
2.4. Thermal study
Thermogravimetric analysis (TGA) of [SPVP]triflate nanostructures revealed a complex thermal degradation process that occurred in multiple stages. The initial weight loss observed below 120 °C can be attributed to the evaporation of the residual moisture within the samples. Following this, the primary degradation of the polymer matrix (PDTmain) occurred rapidly between 185 and 510 °C, resulting in a significant 70% reduction in the polymer weight.49–51 The main degradation phase is further divided into two distinct stages. The first stage, occurring between 185 and 250 °C, is associated with the thermal decomposition of the pendant sulfonic groups and triflate counter anions, while the subsequent stage involves the breakdown of the poly(vinylpyrrolidinium) [PVP] matrix.The incorporation of graphene nanosheets in the nanocomposites, ranging from 0.2% to 10%, led to subtle variations in the thermal behavior of the materials. These differences may be attributed to the influence of graphene on the polymer matrix, which potentially alters the thermal stability and decomposition kinetics of nanocomposites. The observed changes in the thermal characteristics with increasing graphene content suggest that the nanosheets play a role in modifying the overall thermal properties of the [SPVP]triflate nanostructures (Fig. 6a).
 | ||
| Fig. 6 (a) TGA curves of [SPVP]TfO-G0.5% to [SPVP]TfO-G10% nanocomposites. (b) DTG curves of [SPVP]TfO-G0.5% to [SPVP]TfO-G10% nanocomposites. | ||
The derivative thermogravimetric (DTG) analysis confirms a two-stage degradation process: initial decomposition of pendant sulfonic and triflate groups between 185–250 °C with the maximum degradation temperature of the polymer (PDTmax) followed by polymer backbone degradation at higher temperatures, supporting the thermal degradation discussion (Fig. 6b).
2.5. Removal and adsorption study
The adsorption performance of the nanocomposites was evaluated using Acid Red 1 as the model dye pollutant. Batch adsorption experiments were conducted to examine the effects of various parameters, including pH, adsorbent dosage, contact time, temperature, and ionic strength, on dye removal efficiency.The absorption spectrum of Acid Red 1 in water exhibited a prominent peak at 530 nm, which is characteristic of the molecular structure of this dye and its interaction with light. This peak indicates the dye concentration in the aqueous solution, and serves as a baseline for assessing the effectiveness of the extraction methods (Fig. 7A). When the solution was agitated with [SPVP]TfO-G10%, a notable reduction in the intensity of this peak was observed, signifying a substantial decrease in the concentration of AR dye in water (Fig. 7B).
 | ||
| Fig. 7 The electronic spectra of 20 ppm concentration of AR dye in aqueous phase (A), and after shaking with 20 mg of formed solid phase (B). | ||
This significant decrease in the absorption peak demonstrates the high efficacy of the ionic polymer triflate nanocomposites in extracting the AR dye from an aqueous solution. The [SPVP]TfO-G10% nanocomposite likely interacts with dye molecules through various mechanisms, such as electrostatic interactions, π–π stacking, and/or hydrogen bonding. This interaction results in the removal of the dye from the aqueous phase, thereby effectively decolorizing the solution. The ability of these nanocomposites to efficiently extract dyes suggests their potential application in water treatment processes, particularly in industries where dye-contaminated wastewater is a concern.
The combination of TfO− counterions with graphene nanosheets in [SPVP]TfO-G nanocomposites creates synergistic selectivity toward dye classes. TfO− anions enhance electrostatic attraction toward cationic dyes, while graphene nanosheets provide π–π stacking and hydrophobic interactions for neutral and aromatic dyes, leading to improved adsorption efficiency. The G10% nanocomposite was selected for detailed experiments based on SEM and TEM analyses showing optimal graphene dispersion, while adsorption tests confirmed that 10 wt% graphene provided the best balance between adsorption capacity and mechanical stability.
The impact of adsorbent mass on the adsorption process was also investigated (Fig. 8b). As the [SPVP]TfO-G10% dosage increased from 5 mg to 60 mg, the percentage of adsorbed AR dye increased significantly from 35.1% to 98.1%. This trend was attributed to the increased surface area of the ionic polymer nanocomposite, which provided more active adsorption sites. To evaluate other factors affecting adsorption, the researchers chose to use 20 mg of [SPVP]TfO-G10%, which corresponded to a 60% removal rate. These findings underscore the critical role of both the pH and adsorbent dosage in optimizing the adsorption process for dye removal, thereby providing valuable insights for the development of efficient water treatment technologies.
Temperature also plays a crucial role in the adsorption process. Experiments conducted at 293, 303, 313, and 323 K demonstrated that increasing the solution temperature led to a notable enhancement in the percentage of the AR dye removed by [SPVP]TfO-G10%. This positive correlation between the temperature and adsorption efficiency suggests that the process is endothermic in nature. This endothermic characteristic implies that higher temperatures facilitate the adsorption of AR dye onto the nanocomposite surface, potentially due to the increased molecular mobility and enhanced interaction between the adsorbate and adsorbent at elevated temperatures (Fig. 8d).
The impact of ionic strength on the adsorption of AR dye is a complex phenomenon that involves the interplay of electrostatic interactions between ionic polymer triflate graphene nanosheet surfaces and dye molecules. As the concentration of potassium nitrate (KNO3) increased, the ionic strength of the solution changed, leading to different adsorption scenarios. This study revealed that higher KNO3 concentrations resulted in a slight decrease in the percentage of the AR dye removed from the solution. This observation can be attributed to the presence of K+ cations, which interfere with the adsorption process by competing with the dye molecules for binding sites on the [SPVP]TfO-G10% nanocomposite surface (Fig. 8e).
The relationship between ionic strength and adsorption efficiency is further complicated by the charge characteristics of the adsorbent and adsorbate. The [SPVP]TfO-G10% nanocomposite surface possessed a specific charge distribution that influenced its interaction with AR dye molecules. As the ionic strength increases, the electrostatic interactions between the adsorbent and adsorbate may be either enhanced or diminished depending on the nature of the charges involved. In this case, the presence of K+ cations appears to create a screening effect, reducing the attractive forces between the nanocomposite surface and the dye molecules. This screening effect ultimately leads to a decrease in adsorption efficiency, highlighting the importance of considering ionic strength when optimizing adsorption processes for dye removal applications.56
The Weber–Morris model, as applied to the experimental results, offers a quantitative approach for analyzing the kinetics of dye removal:57,58
| qt = Rd(t)1/2 + C |
The model parameters, including the rate constant for intraparticle transport (Rd) and the constant percentage of the border-thickness layer (C), provided valuable information regarding the adsorption process. The graph of qt versus time (Fig. 9a) and the derived Rd values from the two distinct slopes of the Weber–Morris plots (1.599 and 0.605 mg g−1) with high correlation coefficients (R2 = 0.997 and R2 = 0.979) indicate a good fit of the experimental data to the model. This analysis suggested the adsorption kinetics and potentially optimized the [SPVP]TfO-G10% nanocomposite sorbent for more effective pollutant removal in water treatment applications.
The kinetic model of the fractional power function is represented by the following equation and provides insights into the adsorption process of the AR dye onto [SPVP]TfO-G10%.59
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qt = ln qt = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a + b a + b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) t t |
The model parameters a and b are mathematical coefficients, with b less than one. When applied to the experimental adsorption data (Fig. 9b), the model showed alignment with the R2 values for the AR dye. The numerical values of a and b are presented in Table 2. This alignment suggests that the fractional power function model effectively describes the adsorption behavior of AR dye on the [SPVP]TfO-G10% surface.
| Fractional power function kinetic model | |||
|---|---|---|---|
| a | b | ab | R2 |
| 6.945 | 0.2377 | 1.651 | 0.994 |
| The pseudo-first-order kinetic (Lagergren) model | |||
|---|---|---|---|
| qe,exp (mg g−1) | qe,calc (mg g−1) | k1 | R2 |
| 19.91 | 17.37 | 0.049 | 0.968 |
| The pseudo-second-order kinetic model | |||
|---|---|---|---|
| qe,exp (mg g−1) | qe,calc(mg g−1) | k2 | R2 |
| 19.91 | 22.22 | 4.1 × 10−3 | 0.996 |
| Elovich kinetic model | ||
|---|---|---|
| α, (g mg−1 min−1) | β, (mg g−1 min−1) | R2 |
| 0.882 | 3.521 | 0.982 |
To further investigate adsorption kinetics, the Lagergren equation, commonly used for liquid-phase systems, was applied.60 This first-order kinetic model is expressed as
The pseudo-second-order kinetic model is a widely used approach for describing adsorption processes. This model is based on two key assumptions: the total number of binding sites is determined by the amount of adsorbate at equilibrium and the adsorbate concentration remains constant over time.62,63
The model is represented by an equation that relates the amount of dye adsorbed per unit weight of adsorbent at equilibrium (qe) and at any given time (qt), along with the pseudo-second-order coefficient (k2). When applied to the adsorption of AR dye using triflate nanocomposites, the model demonstrated a good fit, as evidenced by the linear plot of t/qt versus t. The intercept and slope of these plots were used to calculate k2 and qe for the dye species, and the results confirmed the suitability of this model for describing the AR dye-removal process (Fig. 9d and Table 2).
In addition to the pseudo-second-order model, the Elovich model was applied to analyze adsorption kinetics.64 This model is particularly useful for systems with non-uniform adsorption surfaces and is often associated with chemisorption processes.
The Elovich equation incorporates the parameters α and β, which represent the initial adsorption rate and desorption coefficient, respectively. A linear plot of qt versus ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) t was obtained, allowing determination of the α and β coefficients (Fig. 9e and Table 2). A comparison of the correlation coefficient values and experimental data from various kinetic models, including the Lagergren pseudo-first-order, pseudo-second-order, and Elovich models, revealed that the pseudo-second-order kinetic model provided the best fit for characterizing the adsorption of AR dye onto [SPVP]TfO-G10%. This comprehensive analysis of the adsorption kinetics provides valuable insights into the mechanism and efficiency of the dye removal process using the triflate nanocomposites.
t was obtained, allowing determination of the α and β coefficients (Fig. 9e and Table 2). A comparison of the correlation coefficient values and experimental data from various kinetic models, including the Lagergren pseudo-first-order, pseudo-second-order, and Elovich models, revealed that the pseudo-second-order kinetic model provided the best fit for characterizing the adsorption of AR dye onto [SPVP]TfO-G10%. This comprehensive analysis of the adsorption kinetics provides valuable insights into the mechanism and efficiency of the dye removal process using the triflate nanocomposites.
Analysis of the data revealed a linear relationship between ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kc and 1000/T for AR dye retention on [SPVP]TfO-G10% within the temperature range – 285–318 K. The increasing equilibrium constant with increasing temperature indicates the endothermic nature of AR dye retention on the sorbents. Calculated thermodynamic values at 293 K showed a positive enthalpy (55.94 ± 0.1 kJ mol−1) and entropy (192.5 ± 0.15 J mol−1 K−1), while Gibbs free energy was negative (−0.46 ± 0.02 kJ mol−1). These results provide insights into the thermodynamic behavior of the adsorption process, suggesting a spontaneous and endothermic reaction with increased disorder at the solid–liquid interface, likely due to desolvation effects during dye binding as illustrated in the Van 't Hoff plots (Fig. 10a).
Kc and 1000/T for AR dye retention on [SPVP]TfO-G10% within the temperature range – 285–318 K. The increasing equilibrium constant with increasing temperature indicates the endothermic nature of AR dye retention on the sorbents. Calculated thermodynamic values at 293 K showed a positive enthalpy (55.94 ± 0.1 kJ mol−1) and entropy (192.5 ± 0.15 J mol−1 K−1), while Gibbs free energy was negative (−0.46 ± 0.02 kJ mol−1). These results provide insights into the thermodynamic behavior of the adsorption process, suggesting a spontaneous and endothermic reaction with increased disorder at the solid–liquid interface, likely due to desolvation effects during dye binding as illustrated in the Van 't Hoff plots (Fig. 10a).
The positive enthalpy (ΔH) value of 55.94 ± 0.1 kJ mol−1 indicates an endothermic uptake process, suggesting that energy is required to break existing bonds and form new bonds between the dye and the adsorbent. The positive entropy (ΔS) value of 192.5 ± 0.15 J mol−1 K−1 implies increased randomness at the solid–liquid interface, likely due to the release of water molecules from the hydration sphere during adsorption. The negative Gibbs free energy (ΔG) value of −0.46 ± 0.02 kJ mol−1 at 293 K confirms the spontaneous nature of the adsorption process.
In addition, the adsorption isotherms provided valuable insights into the interaction mechanism between the AR dye and [SPVP]TfO-G10% sorbent surface. Equilibrium studies are crucial for determining the maximum adsorption capacity and identifying the key surface properties of sorbents. The retention profiles were analyzed across a range of equilibrium concentrations (5–40 mg L−1) under optimal conditions. The relationship between the amount of AR dye retained on [SPVP]TfO-G10% and the equilibrium concentration in the solution exhibits a linear trend at moderate analyte concentrations. The adsorption capacity of AR dye onto [SPVP]TfO-G10% was found to be 21.96 mg g−1 (Fig. 10b).
The Langmuir isotherm model was used to analyze the retention of the AR dye on the [SPVP]TfO-G10% sorbent.66
The plot of Ce/qe against Ce exhibited a linear relationship with an R2 value of 0.999, indicating uniform adsorption of the AR dye on the [SPVP]TfO-G10% surface and good alignment with the Langmuir model. The Langmuir parameters, qm and kL, were derived from the slope and intercept of the linear graph, respectively (Fig. 10c and Table 3). The dimensionless separation factor RL, a crucial feature of this model, was calculated as 0.079 (0 < RL < 1), suggesting favorable adsorption conditions for the AR dye onto the [SPVP]TfO-G10% sorbent.67
| Sorption isotherms models | Values | |
|---|---|---|
| Langmuir | qm, mg g−1 | 22.73 |
| KL, L g−1 | 0.584 | |
| RL | 0.079 | |
| R2 | 0.999 | |
| Freundlich | KF, mg g−1 | 1.083 |
| 1/n | 0.226 | |
| R2 | 0.873 | |
The Freundlich model is a widely used adsorption isotherm that describes the relationship between the amount of adsorbate (in this case, AR dye) retained on the adsorbent surface and its concentration in the solution at equilibrium.68
This model assumes a heterogeneous surface with multiple adsorption sites of varying energy. In this study, the Freundlich parameters KF and 1/n were determined from the intercept and slope of the linear plot (Fig. 10d and Table 3). The KF value represents the adsorption capacity and 1/n is the adsorption intensity or surface heterogeneity.
The obtained 1/n value of 0.226, which is less than 1, suggests that the [SPVP]TfO-G10% sorbent surface is heterogeneous, which is favorable for the adsorption of AR dye. This heterogeneity implies that the adsorption sites have different affinities for the dye molecules, potentially leading to a more efficient removal process. However, the correlation coefficient (R2) for the Freundlich model was 0.873, which, while indicating a reasonable fit, was lower than that of the Langmuir model. This suggests that the Langmuir model, which assumes monolayer adsorption on a homogeneous surface, provides a better description of the AR dye adsorption process on [SPVP]TfO-G10%. The superior fit of the Langmuir model may indicate that despite the surface heterogeneity, the adsorption process predominantly occurs through the formation of a monolayer on the adsorbent surface.
Real-water adsorption tests were conducted at pH 2 to maximize dye removal efficiency. However, acidification to pH 2 may not be feasible for large-scale field applications. Additional experiments at native pH values of seawater, wastewater, and tap water (∼7) revealed a moderate decrease in adsorption efficiency by approximately 10–15% (Table 4). Despite this reduction, the nanocomposite maintains significant dye uptake, demonstrating practical applicability without stringent pH adjustment.
| Water sample | Native pH | Removal efficiency (%) at pH 2 | Removal efficiency (%) at native pH |
|---|---|---|---|
| Seawater | ∼8 | 93.05 | ∼80 |
| Wastewater | ∼7 | 94.76 | ∼82 |
| Tap water | ∼7 | 96.57 | ∼85 |
Subsequently, to assess the reusability of [SPVP]TfO-G10%, the adsorbent was retrieved, washed with acetone to remove the dye, dried, and reused over multiple adsorption cycles. The results demonstrated that [SPVP]TfO-G10% maintained a nearly consistent adsorption efficiency (%E) over four cycles (Fig. 11b). This finding indicates that the ionic polymer designed with triflate counter anions and graphene nanosheets can be effectively recycled and reused multiple times without significant loss in its adsorption capacity. The ability to maintain high adsorption efficiency over multiple cycles highlights the potential of [SPVP]TfO-G10% as a sustainable and cost-effective solution for dye removal from various water sources (Table 5).
| Adsorbent material | Adsorption capacity (mg g−1) | Contact time (min) | Optimal pH | Regeneration cycles | Remarks | Ref. |
|---|---|---|---|---|---|---|
| [SPVP]TfO-G10% nanocomposite | 21.96 | 90 | 2 | 4 | High removal efficiency; good reusability | This study |
| Activated carbon | 15–30 | 90–120 | 4–6 | 3–5 | Widely used; moderate regeneration cost | 70 and 71 |
| Commercial resin (e.g., Amberlite) | 20–25 | 90–120 | 3–5 | 5 | Effective for cationic dyes | 72 and 73 |
| Polymer–graphene composite (recent study) | 18–23 | 60–120 | 2–4 | 3–4 | Enhanced π–π interactions; selective adsorption | 74–77 |
3. Experimental
3.1. Materials
Organic solvents were sourced from commercial suppliers and utilized in their original form unless otherwise specified. Other chemicals were obtained from Merck, Aldrich, or Acros, and were used without additional purification. In this study, powdered polyvinylpyrrolidone (PVP) was employed to modify the nanocomposites as a matrix, while graphene nanosheets (G) served as a reinforcement. Dichloromethane (CH2Cl2), ethyl acetate (C4H8O2), and ethanol (C2H5OH) were used in the preparation of PVP, and chlorosulfonic acid (HSO3Cl) along with triflate acid (CF3SO3H) were utilized for the chemical modification of PVP.A stock solution of Acid Red 1 at a concentration of 500 µg mL−1 was prepared using chemicals from Aldrich Chemical Co. Ltd (Milwaukee, WC, USA). This stock solution was then diluted with deionized water to create standard solutions ranging from 10 to 100 µg mL−1. For the sorption process of Acid Red using modified composites, a series of Britton–Robinson (BR) buffers with pH values from 2 to 11 and 0.5 mol per L HCl were utilized as the extraction medium.
3.2. Apparatus
Various instruments were employed to characterize and identify the chemically modified PVP and nanocomposite materials. The FT-IR spectra of all the nanocomposites were recorded using a PerkinElmer Spectrum100 FT-IR spectrometer, covering a range of 4000–450 cm−1. X-ray diffraction patterns for all resulting nanocomposites were obtained using a D8 ADVANCE X-ray diffractometer, with 2θ ranging from 5° to 80°. The thermal properties were assessed by thermogravimetric analysis (TGA) and derivative thermal gravimetric analysis (DTG) using a Shimadzu TG-50H thermal analyzer. Scanning electron microscopy (SEM) was conducted using a JSM-7610FPlus Schottky (JEOL-Japan) to examine the surface morphology supported by EDX. Transmission electron microscopy (TEM) was performed using a JEOL JEM-F200 multipurpose instrument operating at 200 kV. Raman spectroscopy, including 3D and 2D confocal Raman images and Raman shift curve measurements, was carried out using a Raman spectrometer (Lab. RAM-HR Evolution HORIBA France SAS Co.). The sorption progress of Acid Red was measured using a PerkinElmer UV-visible spectrophotometer (model Lambda 25, USA) equipped with a quartz cell with a 10 mm path width, covering a wavelength range of 190–1100 nm. An Orion pH meter (Model EA 940) was used for pH measurements and testing solutions, and a digital micropipette (Volac) was employed to prepare the standard Acid Red solutions. A digital sensitive balance (Citizen Scales Inc., USA) with a precision of up to four decimal places was used. Deionized water sourced from a Milli-Q Plus system (Millipore, Bedford, MA, USA) was used to prepare the solutions.3.3. Standard protocols for preparing modified sulfonated poly(vinylpyrrolidonium) (SPVP)
4. Conclusion
In this study, a novel nanocomposite material based on sulfonated poly(vinylpyrrolidonium) triflate [SPVP]TfO reinforced with graphene nanosheets was developed for the efficient removal of Acid Red 1 dye from aqueous solutions. FT-IR, Raman spectroscopy, XRD, SEM, TEM, and thermal analysis confirmed the successful integration of graphene and the formation of well-defined nanocomposite structures. The optimal adsorption conditions were determined to be a pH of 2, adsorbent dosage of 20 mg, and contact time of 90 min. The adsorption process followed pseudo-second-order kinetics and was best described by the Langmuir isotherm model, with a maximum adsorption capacity of 21.96 mg g−1 for AR dye. Thermodynamic analysis indicated that the adsorption process was endothermic and spontaneous in nature. The nanocomposites demonstrated excellent performance in removing AR dye from real water samples (seawater, wastewater, and tap water) with removal efficiencies above 93%. Good reusability was observed over the four adsorption cycles, highlighting its potential for sustainable applications. The [SPVP]TfO-graphene nanocomposites developed in this study show great promise as efficient and reusable adsorbents for the removal of anionic dyes from aqueous environments. The combination of the ionic polymer matrix and graphene nanosheets resulted in enhanced adsorption properties compared with those of conventional materials. Further optimization and scale-up studies are recommended to fully realize the potential of these nanocomposites for large-scale water-treatment applications.Conflicts of interest
There are no conflicts to declare.Data availability
The authors confirm that all data are included in the manuscript.Acknowledgements
This project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah, under grant no. (G: 276-247-1442). The authors, therefore, acknowledge with thanks DSR for technical and financial support.References
- M. I. Khan, M. K. Almesfer, A. Elkhaleefa, I. Shigidi, M. Z. Shamim, I. H. Ali and M. Rehan, Polymers, 2021, 13(21), 3810 CrossRef CAS PubMed.
- A. I. Rehan, A. I. Rasee, M. E. Awual, R. M. Waliullah, M. S. Hossain, K. T. Kubra and M. R. Awual, Colloids Surf., A, 2023, 673, 131859 CrossRef CAS.
- H. Roy, T. U. Rahman, M. A. J. R. Khan, M. R. Al-Mamun, S. Z. Islam, M. A. Khaleque and M. R. Awual, J. Water Process Eng., 2023, 54, 104069 CrossRef.
- S. S. Sana, R. Haldhar, J. Parameswaranpillai, M. Chavali and S. C. Kim, Cleaner Mater., 2022, 6, 100161 CrossRef.
- M. Liu, W. Yin, T. L. Zhao, Q. Z. Yao, S. Q. Fu and G. T. Zhou, Sep. Purif. Technol., 2021, 272, 118901 CrossRef CAS.
- P. Koohi, A. Rahbar-Kelishami and H. Shayesteh, Environ. Technol. Innovation, 2021, 23, 101559 CrossRef CAS.
- G. Hitkari, P. Chowdhary, V. Kumar, S. Singh and A. Motghare, Clean. Chem. Eng., 2022, 1, 100003 CrossRef.
- U. Kamran, H. N. Bhatti, S. Noreen, M. A. Tahir and S. J. Park, Chemosphere, 2022, 291, 132796 CrossRef CAS PubMed.
- A. V. B. Reddy, R. Rafiq, A. Ahmad, A. S. Maulud and M. Moniruzzaman, Molecules, 2022, 27(22), 7775 CrossRef CAS PubMed.
- H. Gao, T. Kan, S. Zhao, Y. Qian, X. Wu, W. Cheng and L. Zheng, J. Hazard. Mater., 2013, 261, 83–90 CrossRef CAS PubMed.
- E. N. Zare, A. Motahari and M. Sillanpää, Environ. Res., 2018, 162, 173–195 CrossRef CAS PubMed.
- W. Song, Y. Liu, L. Qian, L. Niu, L. Xiao, Y. Hou and X. Fan, Chem. Eng. J., 2016, 287, 482–491 CrossRef CAS.
- M. Makrygianni, Z. G. Lada, A. Manousou, C. A. Aggelopoulos and V. Deimede, J. Environ. Chem. Eng., 2019, 7(3), 103163 CrossRef CAS.
- B. M. Thamer, A. Aldalbahi, A. M. Moydeen, M. Rahaman and M. H. El-Newehy, Polymers, 2020, 13(1), 20 CrossRef PubMed.
- W. Zhao, Y. Jiao, R. Gao, L. Wu, S. Cheng, Q. Zhuang and W. Dong, Chem. Eng. J., 2020, 391, 123591 CrossRef CAS.
- J. Hızal, N. Kanmaz and M. Yılmazoğlu, J. Mol. Liq., 2021, 322, 114761 CrossRef.
- L. Tian, S. Zhou, J. Zhao, Q. Xu, N. Li, D. Chen and J. Lu, J. Hazard. Mater., 2023, 441, 129873 CrossRef CAS PubMed.
- N. S. Salman and H. A. Alshamsi, J. Polym. Environ., 2022, 30(12), 5100–5118 CrossRef CAS.
- B. Wang, D. Wang, S. Zhao, X. Huang, J. Zhang, Y. Lv and X. Ma, Eur. J. Pharm. Sci., 2017, 96, 45–52 CrossRef CAS PubMed.
- R. Kumar, J. Macromol. Sci., 2024, 63(9), 772–789 CAS.
- J. Barzin, S. S. Madaeni, H. Mirzadeh and M. Mehrabzadeh, J. Appl. Polym. Sci., 2004, 92(6), 3804–3813 CrossRef CAS.
- M. G. Tardajos, L. Garcia-Fernandez, H. Reinecke, M. R. Aguilar, A. Gallardo and J. S. Roman, J. Bioact. Compat Polym., 2012, 27(5), 453–466 CrossRef.
- Z. Oksida-Polivinilpirolidon, Malays. J. Anal. Sci., 2018, 22(4), 693–701 Search PubMed.
- P. Zhao, R. Li, W. Wu, J. Wang, J. Liu and Y. Zhang, Composites, Part B, 2019, 176, 107208 CrossRef CAS.
- G. Pandey, S. Singh and G. Hitkari, Int. Nano Lett., 2018, 8, 111–121 CrossRef CAS.
- A. G. Khorshidi and A. D. Khalaji, J. Polym. Environ., 2024, 32(9), 4577–4588 CrossRef CAS.
- N. A. Elessawy, A. Hashim, H. F. Zahran, M. M. Abd El-Latif, M. H. Gouda, M. S. Elnouby and D. M. F. Santos, Appl. Sci., 2021, 11(19), 9186 CrossRef CAS.
- E. M. Elsayed, M. H. Gouda, M. S. Elnouby, D. M. F. Santos and N. A. Elessawy, Materials, 2020, 13(8), 1905 CrossRef CAS PubMed.
- Q. Qing, L. Li, S.-Z. Hu, S.-Y. Chen, Y. Wang, N. Zhang and T. Huang, Langmuir, 2024, 40(2), 1447–1460 CrossRef CAS PubMed.
- P. Govindaraj, A. Sokolova, N. Salim, S. Juodkazis, F. K. Fuss, B. Fox and N. Hameed, Composites, Part B, 2021, 226, 109353 CrossRef CAS.
- L. Zeng, Z. Zhu and D. W. Sun, Crit. Rev. Food Sci. Nutr., 2022, 62(14), 3705–3722 CrossRef CAS PubMed.
- X. Sun, C. Huang, L. Wang, L. Liang, Y. Cheng, W. Fei and Y. Li, Adv. Mat., 2021, 33(6), 2001105 CrossRef CAS PubMed.
- Y. Tang, X. Wang, J. Chen, X. Wang, D. Wang and Z. Mao, Carbon, 2021, 174, 98–109 CrossRef CAS.
- F. Q. Li, B. H. Sun, Y. L. Qin, X. Y. Liu, Z. Y. Liu, D. R. Ni and Z. Y. Ma, Vacuum, 2021, 184, 109857 CrossRef CAS.
- J. S. Jayan, K. Pal, A. Saritha, B. D. S. Deeraj and K. Joseph, J. Mol. Struct., 2021, 1230, 129873 CrossRef CAS.
- O. Mouhtady, E. Obeid, M. Abu-Samha, K. Younes and N. Murshid, Gels, 2022, 8(7), 447 CrossRef CAS PubMed.
- A. Sayed, G. A. Mahmoud, H. Said and A. A. Diab, Mater. Chem. Phys., 2022, 280, 125731 CrossRef CAS.
- W. K. Abdulsahib, L. S. Jasim, M. A. Mahdi and S. H. Ganduh, Int. J. Appl. Pharm., 2020, 12, 100–106 CrossRef CAS.
- X. Tian, F. Luo, X. Dong, L. Liang, Y. Duan, J. Zhuang and Z. Chen, Colloids Surf., A, 2024, 682, 132883 CrossRef CAS.
- H. Ma, Y. Liu and Y. Wang, Ionic Liquid-Based Adsorbents for the Removal of Toxic Dyes from Wastewater, in Encyclopedia of Ionic Liquids, Springer Nature Singapore, Singapore, 2023, pp. 602–610 Search PubMed.
- A. Shen, X. Liao and Y. Li, Colloids Surf., A, 2021, 623, 126666 CrossRef CAS.
- Y. Zheng, Y. Liu and A. Wang, Ind. Eng. Chem. Res., 2012, 51(30), 10079–10087 CrossRef CAS.
- M. Zare, E. R. Ghomi, P. D. Venkatraman and S. Ramakrishna, J. Appl. Polym. Sci., 2021, 138(38), 50969 CrossRef CAS.
- Z. Iqbal, M. Alam and M. S. Tanweer, ACS Omega, 2023, 8(7), 6376–6390 CrossRef CAS PubMed.
- H. A. Alsabbagh, K. M. Aldamestani, W. A. Amer, J. Iqbal, O. M. Lemine, N. Madkhali and K. El-Boubbou, Phys. Chem. Chem. Phys., 2025, 27(33), 17341–17359 RSC.
- K. Ansari, M. S. Tanweer, D. Kumar and S. R. Samadder, Sep. Purif. Technol., 2025, 378, 134661 CrossRef CAS.
- M. S. Tanweer, K. Ansari, M. Alam and Z. Iqbal, Environ. Processes, 2024, 11(4), 63 Search PubMed.
- M. S. Tanweer, M. Alam and Z. Iqbal, Langmuir, 2022, 38(29), 8837–8853 CrossRef CAS PubMed.
- F. Shirini, P. N. Moghadam, S. Moayedi and M. Seddighi, RSC Adv., 2014, 4(73), 38581–38588 RSC.
- S. Al-Sodies, A. M. Asiri, M. M. Alam, K. A. Alamry, M. M. Rahman and M. A. Hussein, Anal. Methods, 2024, 16(26), 4333–4346 RSC.
- S. Al-Sodies, A. M. Asiri, S. H. Ismail, K. A. Alamry and M. A. Hussein, Mater. Res. Express, 2024, 11(4), 045701 CrossRef CAS.
- J. Patel and A. Kiani, Comparative study of tribological behaviours of different base greases enhanced by graphene nano platelets, in Proceedings of the 3rd International Conference on Theoretical and Applied Nanoscience and Nanotechnology (TANN’19), Ottawa, Canada, 2019, pp. 12–14 Search PubMed.
- G. Surekha, K. V. Krishnaiah, N. Ravi and R. P. Suvarna, FTIR, Raman and XRD analysis of graphene oxide films prepared by modified Hummers method, J. Phys.:Conf. Ser., 2020, 1495(1), 012012 CrossRef CAS.
- Y. Zhang, P. Wang, Z. Hussain, H. Zhang, H. Wang, N. Chang and F. Li, J. Cleaner Prod., 2022, 342, 130789 CrossRef CAS.
- S. Zhang, S. Liao, F. Qi, R. Liu, T. Xiao, J. Hu and Y. Min, J. Hazard. Mater., 2020, 384, 121367 CrossRef CAS PubMed.
- S. A. Dastgheib and D. A. Rockstraw, Carbon, 2002, 40(11), 1853–1861 CrossRef CAS.
- A. N. Chaleshtori, F. M. Meghadddam, M. Sadeghi, R. Rahimi, S. Hemati and A. Ahmadi, J. Environ. Sci. Manag., 2017, 20(2), 9–16 Search PubMed.
- W. J. Weber Jr and J. C. Morris, J. Sanit. Eng. Div., 1963, 89(2), 31–59 CrossRef.
- M. A. Gabal, N. G. Al-Zahrani, Y. M. Al Angari, A. A. Al-Juaid, M. A. Abdel-Fadeel, S. R. Alharbi and R. M. El-Shishtawy, Mater. Res. Express, 2019, 6(10), 105059 CrossRef CAS.
- H. M. Al-Saidi, M. A. Abdel-Fadeel and S. S. Alharthi, J. Saudi Chem. Soc., 2021, 25(8), 101301 CrossRef CAS.
- S. A. Hameed, M. A. Abdel-Fadeel, H. M. Al-Saidi and M. A. Salam, Desalin. Water Treat., 2020, 188, 266–276 CrossRef CAS.
- M. A. Salam, L. Alkhateeb and M. A. Abdel-Fadeel, Desalin. Water Treat., 2018, 129, 168–176 CrossRef.
- T. Braun, J. D. Navratil and A. B. Farag, Polyurethane Foam Sorbents in Separation Science, CRC press, 2018, pp. 1–228 Search PubMed.
- A. S. Al-Soihi, W. Bajafar, M. A. Abdel-Fadeel, Q. A. Alsulami, T. S. Saleh and M. M. M. Mostafa, J. Water Process Eng., 2024, 61, 105361 CrossRef.
- R. H. Althomali, K. A. Alamry, M. A. Hussein, A. Khan, S. S. Al-Juaid and A. M. Asiri, Int. J. Environ. Anal. Chem., 2022, 102(16), 3673–3693 CrossRef CAS.
- M. Dubinin and L. Raduskhevich, Proceedings of the academy of sciences of the USSR, Phys. Chem., 1947, 55, 327–329 CAS.
- G. A. El Naeem, A. I. Abd-Elhamid, O. O. Farahat, A. A. El-Bardan, H. M. Soliman and A. A. Nayl, J. Mater. Res. Technol., 2022, 19, 3241–3254 CrossRef.
- A. S. Sidjou, A. N. Tchakounte, V. Shikuku, I. Lenou, R. Djimtibaye and M. M. Dika, Hybrid Adv., 2023, 4, 100113 CrossRef.
- Z. Marczenko, Separation and Spectrophotometric Determination of Elements, 1986 Search PubMed.
- D. Dimbo, M. Abewaa, E. Adino, A. Mengistu, T. Takele, A. Oro and M. Rangaraju, Res. Eng., 2024, 21, 101910 CAS.
- A. Ahmadfazeli, H. Akbari, P. Pourali, A. Adibzadeh, Y. Rashtbari and Y. Poureshgh, J. Water, Sanit. Hyg. Dev., 2021, 11(6), 1083–1096 CrossRef.
- F. Taleb, M. Ben Mosbah, E. Elaloui and Y. Moussaoui, Korean J. Chem. Eng., 2017, 34(4), 1141–1148 CrossRef CAS.
- G. Kyriakopoulos, D. Doulia and A. Hourdakis, Int. J. Environ. Anal. Chem., 2006, 86(3–4), 207–214 CrossRef CAS.
- P. L. Yap, D. Losic, S. Kabiri and D. N. H. Tran, ACS Appl. Mater. Interfaces, 2018, 11(6), 6350–6362 CrossRef PubMed.
- M. A. Gado, Sep. Sci. Technol., 2018, 53(13), 2016–2033 CrossRef CAS.
- C. Jiao, Z. Du, Y. Chen, X. Han, T. Li, X. Zhang, J. Wang, H. Wang and Y. Shang, J. Polym. Environ., 2020, 28(5), 1492–1502 CrossRef CAS.
- F. Sharif and E. P. L. Roberts, Catalysts, 2020, 10(2), 263 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |