Open Access Article
Open Access ArticleSynthesis, characterization, in vitro and in silico studies of novel lophine clubbed acylthioureas
Sayyed Aqib Ullaha,
Hafsa Jabeenb,
Areeba Javeda,
Abdul Manana,
Nadeem Irshad b,
Kashif Maqbool Khanc,
Aneela Khushald,
Sara Khande,
Umar Farooqd,
Kashif Bashir
b,
Kashif Maqbool Khanc,
Aneela Khushald,
Sara Khande,
Umar Farooqd,
Kashif Bashir *b and
Aamer Saeed
*b and
Aamer Saeed *a
*a
aDepartment of Chemistry, Quaid-i-Azam University, Islamabad 45320, Pakistan. E-mail: asaeed@qau.edu.pk
bDepartment of Pharmacy, Quaid-i-Azam University, Islamabad 45320, Pakistan. E-mail: kbashir@qau.edu.pk
cInstitute of Pharmaceutical Sciences, University of Veterinary and Animal Sciences, Lahore 54000, Pakistan
dDepartment of Chemistry, COMSATS University Islamabad, Abbottabad Campus, Abbottabad, 22060, Pakistan
eDepartment of Chemistry, Pennsylvania State University, University Park, PA, USA
First published on 23rd April 2026
Abstract
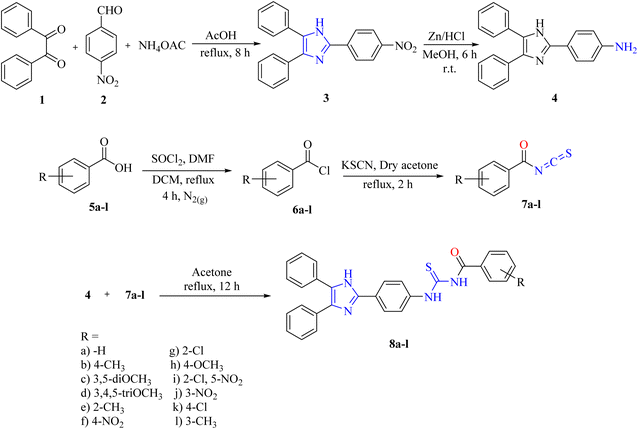
In the present study, a chemical library of novel lophine clubbed acylthioureas (8a–l) was designed and synthesized through a multistep sequence. 2-(4-Nitrophenyl)-4,5-diphenyl-1H-imidazole (3) was synthesized from benzil, 4-nitrobenzaldehyde, and ammonium acetate via multicomponent reaction. Then the nitro group of the synthesized compound was reduced using zinc and hydrochloric acid to furnish 4-(4,5-diphenyl-1H-imidazol-2-yl) aniline (4). On the other hand, the acid chloride (6a–l) of substituted benzoic acids was prepared by reacting acids (5a–l) with thionyl chloride, followed by their reaction with potassium thiocyanate to afford corresponding acyl isothiocyanates (7a–l), which were further reacted with 4-(4,5-diphenyl-1H-imidazol-2-yl)aniline (4) to produce acylthioureas (8a–l) in good yield. The synthesized series (8a–l) was characterized by using a combination of spectroscopic techniques, including FT-IR, 1H NMR, and 13C NMR. Moreover, the synthesized acylthiourea derivatives were assayed for their in vitro and in silico xanthine oxidase and thymidine phosphorylase inhibitory activities. In the tested series, compounds 8j, 8d, and 8i showed robust dual inhibition profiles in low micromolar ranges. Among them, compound 8j revealed the most potent activity, with IC50 values of 1.87 ± 0.26 µM against xanthine oxidase and 1.62 ± 0.27 µM against thymidine phosphorylase. Compound 8d sits in the next position, with an IC50 value of 3.34 ± 0.37 µM (xanthine oxidase) and 3.31 ± 0.06 µM (thymidine phosphorylase), while compound 8i demonstrated considerable xanthine oxidase inhibition with an IC50 of 2.06 ± 0.36 µM. These values have outperformed the standard drug allopurinol (IC50 = 7.4 ± 0.07 µM), which was found to be less potent against xanthine oxidase. Considering the established involvement of these enzymes in oxidative stress, hyperuricemia, and inflammation-based disorders, the results endorse the idea of these derivatives being promising multifunctional therapeutic agents in the treatment of metabolic, inflammatory, and neoplastic diseases.
1 Introduction
In medicinal chemistry, molecular hybridization is an intriguing technique for creating pharmaceuticals with two or more moieties of distinct pharmacological properties.1 It is feasible to develop hybrid compounds that are more effective by considering the biological behaviors of the moieties. These molecules would have improved binding interactions and inhibitor pocket packing with the receptors. Imidazole is a five-membered planar aromatic ring that contains two nitrogen atoms at positions 1 and 3. C3H4N2 is the molecular formula of the simplest imidazole.2 Imidazole was discovered in the early 1840s, and the name imidazole was coined by Arthur Rudolf Hantzsch in 1887.3 The name glyoxaline was used before 1887 for the compounds because it was synthesized for the first time by Henrich Debus in 1858 from glyoxal and ammonia.4Research on the synthesis of imidazole-based organic compounds is being carried out due to their diverse applications as agrochemicals, artificial acceptors, medicinal drugs, biomimetic catalysts, man-made materials, supramolecular ligands, and so on.5–9 Imidazole contains a peptide backbone unit and shows similarity with carboxamide in physicochemical and biological properties; thus, these are considered as bioisosteres of one another and provide metabolic stability against amidases. Imidazole consists of two nitrogen atoms, one acts as an H-bond acceptor, and the other one as a donor, and the same is the case for carboxamide; the only difference is the presence of one oxygen instead of a nitrogen atom.10,11 Similarly, imidazole is found as an isostere replacement of various 5-membered heterocycles such as thiazole,12 oxazole,13 tetrazole,14 pyrazole,15 and triazole16 because all these compounds show similarity in biological and physicochemical properties due to the same valence electronic distribution, and all compounds have the same hydrogen bonding and π–π stacking interactions with biological systems.17
2,4,5-Triphenyl-1H-imidazole, also called lophine, is an attractive chemiluminescent and fluorescence compound. Radziszewski reported the chemiluminescence properties of lophine for the first time in 1877. He observed that when lophine reacts with oxygen and a strong base, it emits yellow light.18 Thioureas are organosulfur compounds with the general formula RNHC(S)NHR′ and are similar in structure to urea except for the replacement of the oxygen atom of urea by a sulfur atom; consequently, the chemical characteristics of both are considerably different.19 Thiourea was synthesized in 1873 by Marceli Nencki as a white solid crystal with a melting point of 176–178 °C for the first time as a urea analog.20 The reason for extensive research and interest in thioureas is due to their diverse applications, such as plastics,21 formation of photographic film,22 and dyes.23 The derivatives of thiourea have also been reported to have biological applications, such as preservatives,24 insecticides,25 and as precursors of pharmaceutical drugs.26
Acylthiourea with the general formula (RCONHC(S)NHR′) is obtained by replacing one H atom of NH2 in thiourea with an acyl group (RC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), where R may be an aryl, alkyl, aralkyl, or heterocyclic group. Acylthioureas have two coordinating sulfur and nitrogen atoms, thus acting as bidentate ligands and leading to the formation of metal complexes.27 Extensive research on acyl thiourea was due to its effective, diverse nature of applications in various fields, such as organo-catalysts,28,29 molecular recognition, pharmaceuticals, agriculture, material science, and biological activities.30 Some important biological activities include antimicrobial,31 antidiabetic,32 herbicidal,33 fungicidal,34 antiparasitic,35 pesticidal,36 urease inhibition,37 and anticancer agents.38 Some biologically active compounds containing imidazole, thiourea, and acylthiourea moieties are shown in Fig. 1.
O), where R may be an aryl, alkyl, aralkyl, or heterocyclic group. Acylthioureas have two coordinating sulfur and nitrogen atoms, thus acting as bidentate ligands and leading to the formation of metal complexes.27 Extensive research on acyl thiourea was due to its effective, diverse nature of applications in various fields, such as organo-catalysts,28,29 molecular recognition, pharmaceuticals, agriculture, material science, and biological activities.30 Some important biological activities include antimicrobial,31 antidiabetic,32 herbicidal,33 fungicidal,34 antiparasitic,35 pesticidal,36 urease inhibition,37 and anticancer agents.38 Some biologically active compounds containing imidazole, thiourea, and acylthiourea moieties are shown in Fig. 1.
Keeping in mind the above-mentioned molecular hybridization concept and the wide spectrum of imidazole and acylthioureas activities, herein we report the synthesis of imidazole-linked acylthioureas to enhance effectiveness and broaden the pharmaceutical scope of acylthioureas.
Much attention has been directed in recent years to the therapeutic targeting of enzymes involved in oxidative stress and nucleotide metabolism with respect to inflammation-related metabolic disorders. Of these, xanthine oxidase and thymidine phosphorylase have emerged as clinically relevant enzymes because of their respective roles in purine catabolism and the generation of pro-oxidant metabolites. For example, xanthine oxidase represents a key source of ROS and uric acid, contributing to oxidative damage and gout disease, while thymidine phosphorylase is implicated in the production of 2-deoxy-D-ribose, an agent that evokes inflammation and endothelial dysfunction. Thus, compounds that can inhibit both xanthine oxidase and thymidine phosphorylase may afford therapeutic benefits in the management of diseases such as gout, atherosclerosis, and related disorders driven by oxidative and inflammatory mechanisms.39,40
A scaffold of acylthiourea has been considered a privileged pharmacophore in medicinal chemistry, as it can interact with an enzyme active site through hydrogen bonding, metal coordination, and favorable electrostatic interactions. Therefore, this would turn out to be particularly suitable for interacting with the catalytic or cofactor-binding domains of oxidoreductases and phosphorylases.41,42 Structurally based on this, a series of lophine-conjugated acylthiourea derivatives has been designed to explore their potential for dual inhibition of xanthine oxidase and thymidine phosphorylase enzymes. The biological implication of those substances was further studied by molecular docking and in vitro enzyme inhibition studies to probe their binding affinity and functional activity.39,41
2 Results and discussions
2.1 Chemistry
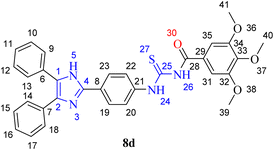
A novel class of lophine-linked acyl thiourea derivatives was synthesized via a multistep synthesis. These steps include heterocyclization, reduction, nucleophilic substitution, and addition reactions. 2-(4-Nitrophenyl)-4,5-diphenyl-1H-imidazole was synthesized from benzil, 4-nitrobenzaldehyde, and ammonium acetate via a multicomponent heterocyclization reaction. Then the nitro group of the synthesized compound was reduced using zinc and hydrochloric acid to furnish 4-(4,5-diphenyl-1H-imidazol-2-yl) aniline. On the other hand, the acid chloride of substituted benzoic acids was prepared through nucleophilic substitution reaction by reacting acids with thionyl chloride followed by their reaction with potassium thiocyanate to afford corresponding acyl isothiocyanates, which were further reacted with 4-(4,5-diphenyl-1H-imidazol-2-yl)aniline to produce acyl thioureas via nucleophilic addition reaction (8a–l) in good yield as can be seen in Scheme 1.The structures of all novel synthesized lophine clubbed acylthiourea pharmacophores were confirmed via spectroscopic analysis. A representative example (8d) is discussed here.
Synthesized compound 8d was characterized and confirmed by FT-IR spectroscopy. The stretching bands for Csp2–H and Csp3–H bonds were observed at 3031 and 2888 cm−1, respectively. A strong stretch at 1680 cm−1 was attributed to the stretching of carbonyl C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O functionality. A stretching band appeared at 1580 cm−1 corresponding to the C
O functionality. A stretching band appeared at 1580 cm−1 corresponding to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching of the aromatic part. Another characteristic stretching band manifested at 1235 cm−1 for the C
C stretching of the aromatic part. Another characteristic stretching band manifested at 1235 cm−1 for the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S bond, further confirming the synthesis of the respective compound. Additionally, there was another strong stretch at 1118 cm−1, indicating the presence of a C–N bond. The presence of sulphur was confirmed by Lassaigne's test.
S bond, further confirming the synthesis of the respective compound. Additionally, there was another strong stretch at 1118 cm−1, indicating the presence of a C–N bond. The presence of sulphur was confirmed by Lassaigne's test.
The characteristic and most deshielded signals at 12.971 and 11.748 ppm appeared as singlets, corresponding to the two NH protons at positions 26 and 24 of the thioamide moiety, which confirmed the synthesis of the respective compound. The proton present at position 26 appeared downfield because of the deshielding effect caused by the carbonyl and thiocarbonyl group attached directly to the nitrogen atom containing the proton. The carbonyl and thiocarbonyl groups here withdraw electrons both inductively and by resonance effect, which makes the proton more deshielded. The signal for the two protons at positions 19 and 23 appeared as doublet at 8.373 ppm with 3J = 8.7 Hz by coupling with protons at positions 20 and 22. Signal at 8.057 ppm corresponds to the protons at positions 22 and 24, which appeared as doublet with 3J = 8.7 Hz by virtue of coupling with protons at positions 19 and 23. The signal at 8.373 ppm was assigned to the protons at positions 19 and 23 because these protons are ortho to the electron-withdrawing imidazole ring, which deshields them through a resonance effect. Signal for four protons at positions 9, 13, 14, and 18 manifested as a multiplet at 7.599–7.569 ppm; these protons appeared slightly deshielded because they are present ortho to the electron-withdrawing imidazole ring. Signals for six protons at positions 10, 11, 12, 15, 16, and 17 appeared as a multiplet at 7.521–7.477 ppm. Signals for protons at positions 31 and 35 appeared as a singlet at 7.413 ppm; these protons are slightly shielded compared to the other aromatic protons because of electron-donating methoxy groups, which increase electron density at the ortho position through the resonance effect. The most shielded two signals at 3.889 and 3.761 ppm appeared as singlets corresponding to the protons of methoxy groups present at meta and para positions 39, 41, and 40 of the acylated aromatic part, respectively. pKa value of the proton at position 5 in the imidazole ring is 14.5, rendering it more acidic than alcohols. Due to this acidity, the signal for the proton does not appear as it can exchange with the solvent.
The most deshielded signals appeared at 179.49 and 167.69 ppm, corresponding to the carbon atoms (28) and (25) of the thiocarbonyl and carbonyl functional groups, respectively, confirming the synthesis of the desired compound. The thiocarbonyl carbon was more deshielded because it relates to three electron-withdrawing electronegative heteroatoms directly. Signals at 153.04 ppm correspond to the carbon of the imidazole ring present between two nitrogen atoms. Carbons (32) and (34) of the aromatic ring, to which the methoxy groups were directly attached, manifested at 143.89 ppm. The signal at 142.13 ppm corresponds to carbon (33) of the aromatic ring to which the methoxy group was directly attached. Signal at 141.28 was assigned to carbon (21) of the aromatic ring. The most shielded signal at 106.79 in the aromatic region was assigned to the carbons (31) and (35) of the aromatic ring present ortho to the electron-donating methoxy group, which increases electron density at these positions by the resonance effect. Remaining sp2 hybridized carbon atoms (1, 2, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 22, 23, 29) appeared in the range of 130.04–121.51 ppm. Signals at 60.64 and 56.63 ppm were assigned to the carbon (40) of the methoxy group at the para position and carbons (39) and (41) of the methoxy groups at the meta position of the aromatic ring, respectively.
2.2 Enzyme inhibition in silico studies
Molecular docking was conducted to investigate the interactions of synthesized lophine-based acylthiourea derivatives with xanthine oxidase and thymidine phosphorylase at the molecular level. The co-crystallized ligand was extracted from the protein PDB file and re-docked into the protein using the same parameters to check docking consistency. RMSD values were calculated using the DSV tool, comparing docked vs. co-crystallized ligand poses. RMSD values were 1.21 and 1.01 Å, showing significant overlap as shown in Fig. 2. This validates the docking experiment's accuracy. | ||
| Fig. 2 Re-docking of co-crystallized ligands in the active pockets of selected proteins. (a) Thymidine phosphorylase, and (b) xanthine oxidase. | ||
From molecular docking studies, the binding affinity values revealed that several compounds formed stable complexes with both enzymes. Although the docking scores do not exactly match the IC50 values, the docking results still provide useful hints on how the synthesized compounds interact with the enzyme's active sites, including their binding modes and interaction with the enzyme. Among them, compounds 8j and 8d exhibited strong binding energies against both targets, while compound 8j had the most favorable interaction pattern, indicating a good fit within the active sites, which correlated well with its strong enzymatic inhibition observed in the biological assays. Their docking scores were more favorable than those of the reference drugs allopurinol and 7-deazaxanthine, highlighting the potential of such molecules to act as effective dual inhibitors. Such computational results are in good agreement with the experimental enzyme inhibition data, further supporting the therapeutic relevance of the synthesized series. The detailed binding energies of all the compounds are summarized in Table 1.
| Sr. # | Code | Binding energy (kcal mol−1) | |
|---|---|---|---|
| Xanthine oxidase | Thymidine phosphorylase | ||
| 1 | 8a | −6.32 | −8.31 |
| 2 | 8b | −8.23 | −6.82 |
| 3 | 8c | −6.18 | −7.34 |
| 4 | 8d | −6.47 | −8.50 |
| 5 | 8e | −5.84 | −7.51 |
| 6 | 8f | −7.76 | −6.38 |
| 7 | 8g | −6.62 | −8.22 |
| 8 | 8h | −7.47 | −7.86 |
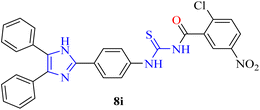
| 9 | 8i | −5.88 | −7.63 |
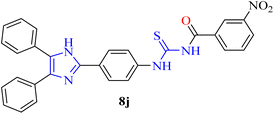
| 10 | 8j | −6.99 | −8.43 |
| 11 | 8k | −6.11 | −6.76 |
| 12 | 8l | −8.02 | −6.5 |
| 13 | 2b (IMA) | −8.46 | −6.90 |
| 14 | 7-Deazaxanthine | −5.62 | |
| 15 | Allopurinol | −5.93 | |
The docking orientations observed in this study provide insight into the mechanism through which the newly synthesized derivatives might act as inhibitors of the enzymes. From the structural conformation of the enzyme, most inhibitors have been observed to bind within the tight catalytic channel that leads to the molybdenum site, effectively blocking the substrate and preventing the conversion of hypoxanthine to uric acid.43,44 In this regard, aromatic thiourea derivatives have been observed as competitive or mixed inhibitors that form hydrogen bonds around the catalytic site and hydrophobic stacking interaction within the substrate channel.44 Previous studies on the molecular docking of small molecule inhibitors have revealed that a number of them bind to residues near the catalytic site and prevent substrate binding at the catalytic site.45,46 In the current study, the binding of compounds 8j, 8d, and 8b to the catalytic cleft and their interaction through hydrogen bonds to residues such as Ser144, Glu33, and Asp309 suggest a similar mechanism of catalytic site blocking. Such interactions are likely to hold the inhibitor within the gorge of the enzyme and prevent the binding of physiological substrates to the catalytic site containing molybdenum.
The observed binding affinities seem to arise from a combination of π–π stacking, dipolar stabilization, and directional hydrogen bonding with both the thiocarbonyl and imidazole nitrogen atoms. This type of interaction is in line with previous studies on acylthiourea-derived heterocycles, in which these key pharmacophoric features are important for effective inhibition of xanthine oxidase and thymidine phosphorylase enzymes.47,48
In addition, the extended aromatic system of the lophine core is likely to contribute to the stability of the ligand by forming aromatic–aromatic interactions with the hydrophobic residues in the binding pocket of the enzyme.49 The compounds 8j and 8d can make more effective interactions, and this is probably because substituents, whether electron-withdrawing or electron-donating, alter the electronic properties of the benzamide ring and guide the ligand into a more favorable orientation in the catalytic pocket. Orientation effects are in good agreement with SAR trends obtained in computational analyses of nitro- and methoxy-substituted enzyme inhibitors.50
Several important residues in the binding pocket that enable the inhibitor to remain within the active site are revealed by the crystal structure of xanthine oxidase. These consist of two loops with color-coded green and blue residues ranging from 25–52 and 142–153, respectively. Three helices, spanning residues 263–272, 285–308, and 342–345, are also involved in the color-coded yellow, pink, and purple region of the active site. The active site residue of the protein, color-coded brown, black, and orange, include three loops (309–341, 356–361, and 420–436) that are essential for inhibitor binding and retention. Another loop containing residues 1220–1230 also forms part of the cyan-colored region of the active site (Fig. 3).
Docking overlays reveal that the compounds 8d and 8j, containing multiple methoxy groups or a meta-nitro substituent, orient their substituents toward polar microenvironments within the binding pocket. This action may enhance the ligand–enzyme complex through additional donor–acceptor interactions. The findings were corroborated by Feng et al. (2022) in docking studies of methoxy-rich and nitro-substituted phenylthioureas, where substituent-directed orientation influences docking stability.47 The small discrepancies between the docking results and the experimental IC50 values arise from the limitations of the docking scoring function. While the scoring function provides a good estimate of the binding affinity between the ligand and the enzyme, it does not take into consideration the dynamic nature of the enzyme's binding sites.
Hydrophobic residues such as Ile182, Leu316, Val40, Ala317, and Phe269 form a nonpolar niche that facilitates van der Waals and π–π-alkyl interactions with the ligand's aromatic rings. Collectively, these interactions work together to hold the inhibitor in place in the catalytic cleft of xanthine oxidase. This results in reduced oxidative stress and excessive uric acid production that is mechanistically similar to clinically established xanthine oxidase inhibitors like allopurinol and febuxostat. Indeed, similar binding motifs have been described for other aromatic thiourea inhibitors, which display a remarkable increase in affinity upon hydrophobic enclosure of the aromatic system, combined with anchoring via the thiocarbonyl unit in a polar fashion.48 This mechanistic pattern forms the basis for the combined influence of electronic density and substituent polarity in controlling strong xanthine oxidase binding.
Thymidine phosphorylase forms an S-shaped dimer made up of two subunits when it is co-crystallized with 3-azido-2-fluoro-dideoxyuridine (ONP), according to a thorough structural analysis. The active site is located at the interface between the larger α/β domain and the smaller α-helical domain that make up each subunit. Arg-171, Ser-186, and Lys-190 are important residues that are probably involved in ligand binding. Furthermore, residues 115–123 and 206–212 compose the hydrophobic area of the active site, as shown in Fig. 5.
Docking analyses further indicate that the lophine core complex significantly stabilizes thymidine phosphorylase via π–π stacking interactions with aromatic residues, including Tyr168 and His85. These residues have been identified as key contributors to the binding of both nucleoside analogues and non-nucleoside inhibitors.47 The acylthiourea moiety appears to stabilize this orientation by acting as a polar hinge, enabling the benzamide substituent to align suitably with the catalytic region. This dual anchoring has been reported in structure–activity relationship studies of thiourea- and urea-based thymidine phosphorylase inhibitors, wherein the concurrent presence of an aromatic head group and polar linker markedly enhances binding.51
The 3D interaction plot of compound 8a, shown in Fig. 6, illustrates its positioning within the catalytic pocket of thymidine phosphorylase and highlights the key amino acids involved in ligand recognition. The enzyme catalyzes the reversible phosphorolysis of thymidine to thymine and 2-deoxy-D-ribose-1-phosphate and plays a role in tumor angiogenesis. Multiple hydrogen bonds (green dashed lines) are established with residues like Lys84, Ser95, Thr128, and Gly121, which assist in the orientation of ligand binding. Ionic interactions (orange dashed lines) with residues like Asp92 and Lys130 also reinforce the binding by complementing the charged groups of the ligand. Moreover, hydrophobic and aromatic interactions (pink and purple dashed lines) with residues such as His85, Met111, Leu117, and Tyr168 further underscore the positioning of the ligand 8a in the binding site by π–π stacking and van der Waals contacts. These interactions facilitate the binding of compound 8a to the thymidine phosphorylase catalytic pocket.
The pronounced accommodation of compound 8a within thymidine phosphorylase, despite the absence of electron-withdrawing or electron-donating substituents on the terminal phenyl ring, aligns with recent thymidine phosphorylase inhibitor studies. These studies indicate that planar aromatic systems can interact effectively through π–π stacking and hydrophobic complementarity within the enzyme active site.52 The current docking results indicate that the lophine–acylthiourea scaffold alone provides sufficient electronic and steric complementarity to offset the lack of benzamide substituents, enabling compound 8a to capitalize on the same stacking and hydrophobic interactions documented for other aromatic thymidine phosphorylase inhibitor chemotypes.53
The 3D interaction plot of compound 8j is shown in Fig. 7. The compound 8j is involved in a complicated hydrogen bonding, electrostatic, and hydrophobic network of interactions with some catalytically and structurally significant residues of thymidine phosphorylase. Interestingly, hydrogen bonds (green dashed lines) are noted between compound 8j and residues Gln156, Lys190, and Ile183, contributing to directional polar interactions with the catalytic residues. Electrostatic interactions (orange and red dashed lines) with Asp164 reflect strong ionic contact and charge complementarity between the negatively charged carboxylate group of Asp164 and the positively polarized areas of the ligand. This contact probably plays a central role in stabilizing the orientation of compound 8j in the catalytic pocket. In addition, π–π stacking and hydrophobic interactions (pink dashed lines) with His85, Tyr168, and Met111 aid nonpolar stabilization and aromatic ring stacking, which are vital for compound 8j accommodation in the enzyme's hydrophobic channel. Further interactions with amino acids such as Ser113 and Gly114 facilitate the proper positioning of the ligand.
The binding profile of compound 8j is in good agreement with previously reported docking results for nitro-substituted phenyl inhibitors of thymidine phosphorylase, where the nitro group supports high-affinity binding by orienting its oxygen atoms toward basic residues and thus forming a stable dipolar anchor within the active site.47 Specifically, the meta placement of the nitro group has been shown to preserve aromatic planarity while allowing for favorable hydrogen-bond geometry, thus providing a rationale for the observed marked stability of compound 8j in the current docking simulations.
2.3 Enzyme inhibition in vitro studies
The xanthine oxidase and thymidine phosphorylase inhibition activities of the synthesized compounds are summarized in Table 2. The enzyme concentration used for the inhibition activity was 2 mg ml−1. GraphPad Prism software was used to determine the IC50 values at various concentrations.| Sr. # | Compound | % inhibition ± STD (xanthine oxidase) | IC50 (µM) ± SEM | % inhibition ± STD (thymidine phosphorylase) | IC50 (µM) ± SEM |
|---|---|---|---|---|---|
| a The IC50 values were obtained only for those compounds that achieved at least 50% inhibition at a particular concentration. | |||||
| 1 | 8a | 67.5 ± 1.16 | 6.18 ± 1.04 | 83.5 ± 1.16 | 4.17 ± 0.03 |
| 2 | 8b | 53.8 ± 1.09 | 410.00 ± 1.18 | 49.8 ± 1.09 | _ |
| 3 | 8c | 84.6 ± 1.01 | 2.64 ± 0.19 | 44.6 ± 1.01 | _ |
| 4 | 8d | 81.3 ± 0.91 | 3.34 ± 0.37 | 89.3 ± 0.91 | 3.31 ± 0.06 |
| 5 | 8e | 70.0 ± 1.23 | 4.40 ± 0.30 | 37.0 ± 1.23 | _ |
| 6 | 8f | 41.1 ± 2.14 | _ | 65.1 ± 2.14 | 4.39 ± 0.21 |
| 7 | 8g | 46.3 ± 1.12 | _ | 39.3 ± 1.12 | _ |
| 8 | 8h | 51.3 ± 0.89 | 482.00 ± 1.01 | 52.3 ± 0.89 | 452.00 ± 0.94 |
| 9 | 8i | 83.8 ± 1.23 | 2.06 ± 0.36 | 33.8 ± 1.23 | _ |
| 10 | 8j | 87.5 ± 1.67 | 1.87 ± 0.26 | 88.5 ± 1.67 | 1.62 ± 0.27 |
| 11 | 8k | 50.0 ± 1.86 | 500.00 ± 0.84 | 55.5 ± 1.92 | 393.00 ± 1.16 |
| 12 | 8l | 65.5 ± 1.92 | 4.73 ± 0.29 | 81.0 ± 1.86 | 2.96 ± 0.14 |
| 13 | 2b (IMA) | 60.8 ± 1.15 | 6.23 ± 0.17 | 54.0 ± 1.15 | 372.00 ± 0.92 |
| 14 | 7-deazaxanthine | 89.0 ± 1.63 | 41.00 ± 0.24 | ||
| 15 | Allopurinol | 86.5 ± 1.37 | 7.40 ± 0.07 | ||
Nine compounds showed the strongest inhibition against xanthine oxidase and thymidine phosphorylase. The rest of the compounds are also moderate to good inhibitors of both enzymes. Among the synthesized derivatives, compound 8j was the most potent dual inhibitor, with IC50 values of 1.87 µM for xanthine oxidase and 1.62 µM for thymidine phosphorylase.
It was also found that compound 8d exhibited high potency against both enzymes, whereas compound 8i demonstrated selective and strong inhibition of xanthine oxidase. This was further supported by the dose–response inhibition curves, in which compounds such as 8j and 8i exhibited a sharp, concentration-dependent decrease in absorbance, confirming their potent inhibition of xanthine oxidase. These sigmoidal inhibition trends, plotted through nonlinear regression as shown in Fig. 8, illustrate the effectiveness of these molecules in suppressing enzymatic activity at micromolar levels.
Notably, compound 8j inhibited thymidine phosphorylase at 88.5%, while 8d achieved a similar magnitude of activity at 89.3%, demonstrating its strong dual-target activity. In corroboration, inhibition graphs for thymidine phosphorylase in Fig. 9 indicated steep inhibition curves for compounds 8j, 8d, and 8i, which are consistent with their higher percentage inhibition values.
The concentration-response behaviour reflected in these plots provides further experimental evidence of their efficiency as dual enzyme inhibitors. These data indicate that the above conjugates represent promising scaffolds for the design of novel multifunctional agents with potential applications in the therapy of oxidative and neoplastic disorders.
The structure–activity relationships observed in this study can be rationalized in the context of the lophine–acylthiourea hybrid scaffold, in which the acylthiourea fragment functions as a privileged pharmacophore. Acylthiourea derivatives are well documented to exhibit strong enzyme-binding ability due to their dual hydrogen-bond donor–acceptor character and the presence of a thiocarbonyl sulfur capable of engaging in metal coordination and polar interactions. Comprehensive SAR analyses have demonstrated that such features significantly enhance binding affinity toward a wide range of enzyme targets, particularly metalloenzymes and phosphoryl-processing enzymes.30
The dose–response profiles shown in Fig. 8 and 9 reinforce the SAR that underpins the inhibitory behavior of the synthesized lophine-clubbed acylthiourea derivatives for xanthine oxidase, while compounds 8j, 8i, 8c, and 8d display steeper slopes with earlier inflection points, indicative of high-affinity binding and rapid achievement of maximal inhibition. The curve morphologies are in good agreement with the kinetic characteristics reported in the literature for several potent non-purine xanthine oxidase inhibitors, in which electron-withdrawing substituents and polymethoxy motifs are strongly associated with enhanced suppression of xanthine oxidase turnover.54 For thymidine phosphorylase, the curves representing 8j, 8d, and 8a reveal early saturation combined with a large magnitude of slopes, which points toward favorable aromatic alignment and high-affinity recognition in the narrower thymidine phosphorylase catalytic groove.55
The selectivity of enzyme inhibition by thiourea scaffolds has been a recurring theme in the field of medicinal chemistry. Thiourea and acylthiourea derivatives contribute unique structural features to a molecule, such as the presence of a thiocarbonyl functional group and NH groups that facilitate their use as both hydrogen bond donors and acceptors. This increases the degree of selectivity in the binding of enzymes to their ligands.56 The sulfur atom in the thiocarbonyl group is also responsible for the formation of polar and electrostatic interactions that facilitate the binding of ligands in the active sites of enzymes. Due to these structural characteristics of the derivatives, thiourea derivatives have been widely evaluated as inhibitors of some important enzymes involved in biological processes and have been found to exhibit good binding selectivity in both biological assays and computational docking studies.57,58 The presence of the acylthiourea linker in the synthesized lophine derivatives introduces a pharmacophore that is capable of making stable interactions with the residues of the catalytic pockets of both xanthine oxidase and thymidine phosphorylase enzymes, as evidenced by the inhibition trends observed in the biological assays.
In vitro results on xanthine oxidase inhibition showed that electron-withdrawing substituents and polymethoxy substitution patterns substantially enhance activity. Electron-withdrawing groups have enhanced the inhibition of xanthine oxidase in this series of compounds. In compound 8j, for example, the presence of the meta-nitro group makes this compound the most potent in the series, with an IC50 value of 1.87 µM. The electron-withdrawing ability of the nitro group increases the hydrogen-bond acceptor ability of the acylthiourea linker.54
The presence of electron-donating groups enhances activity in various derivatives. The presence of multiple methoxy groups in various derivatives, such as 8c (3,5-di-OCH3) and 8d (3,4,5-tri-OCH3), demonstrated robust xanthine oxidase inhibitory activity. The presence of methoxy groups enhances the aromatic polarizability and favors π–π stacking.59
Steric effects also seem to play a role in inhibitory activity. Inhibitors with ortho-substituted groups, such as 8e and 8g, demonstrated lower inhibitory activity. This lower inhibitory activity may be due to steric effects that interfere with optimal ligand orientation and π-stacking.54
Compound 8i (2-Cl,5-NO2) is an example of the effect of halogens on inhibitory activity. The addition of chlorines generally enhances hydrophobic interactions within the binding pocket and extends the residence time of the compound. The nitro group is responsible for strong polar interactions with amino acids in the active site, which explains the potent xanthine oxidase inhibitory activity of this compound.60 On the other hand, compounds with weakly polar or electronically neutral functional groups, such as 8a, 8b, and 8l, displayed moderate xanthine oxidase inhibitory activity.
A comparison of their behavior from a side-by-side perspective illustrates a clear structure–activity relationship. The series demonstrates that electron-withdrawing groups, such as the meta-nitro group in 8j, are the most potent inhibitors of xanthine oxidase activity. Compounds containing multiple methoxy groups (8c and 8d) also outperform expectations, possibly because additional aromatic interactions within the binding site contribute to binding affinity. Conversely, compounds containing ortho groups (8e and 8g) are less potent due to unfavorable steric interactions that prevent optimal binding orientation. In conclusion, the findings demonstrate the impact of both electronic and steric factors on the inhibitory activity of the lophine acylthiourea series.
Compound 8d (3,4,5-tri-OCH3) is a very potent thymidine phosphorylase inhibitor (IC50 = 3.31 µM). The high density of methoxy substituents increases aromatic stacking with thymidine phosphorylase-binding residues and supports additional hydrogen bonding by oxygen lone pairs. Similar SAR trends have been observed for inhibitors containing a thiocarbonyl group, where aromatic rings containing multiple methoxy functional groups enhance the binding affinity within the catalytic pocket of the enzyme.62
Interestingly, compounds 8a (H) and 8l (3-CH3) retain high thymidine phosphorylase activity without strong polar substituents. This is due to the persistence of molecular planarity and the fulfillment of the ideal hydrophobic fit of the lophine aromatic surface within the narrow thymidine phosphorylase channel. Similar results have been found in SAR studies of thymidine phosphorylase inhibitors, where flat aromatic rings are maintained if the central scaffold has enough hydrogen-bonding potential.61
In contrast, ortho-substituted compounds such as 8e (2-CH3), 8g (2-Cl), and 8i (2-Cl,5-NO2) show reduced thymidine phosphorylase inhibition. This is because steric crowding at the ortho position invokes out-of-plane distortion of the benzamide phenyl ring, disrupting π-stacking and misaligning pharmacophoric atoms. Similar steric penalties associated with ortho-substitution have been consistently observed during SAR studies of thymidine phosphorylase inhibitors.62
Inspection of how these substituents influence the activity pattern of the synthesized compounds reveals a good relationship between structure and activity for inhibiting thymidine phosphorylase. The compounds bearing electron-withdrawing groups, such as the meta-nitro group of 8j, demonstrated the most potent inhibitory activity on thymidine phosphorylase, which suggests favourable electrostatic interactions with the enzyme's active site. The compounds bearing multiple methoxy groups (8d) also demonstrated good inhibitory potential, likely due to increased opportunities for aromatic stacking interactions. The compounds bearing ortho-substituted groups, however, were less potent (8e, 8g, 8i), likely due to steric hindrance preventing optimal binding within the narrow thymidine phosphorylase channel.
A comparison of docking scores with experimental IC50 values reveals a clear trend: molecules with higher predicted binding energies, such as 8j and 8d, are generally more potent inhibitors in vitro. This supports the docking analysis as a valid explanation for the observed structure–activity relationship. Table 3 summarizes the key substituent effects observed in this set of compounds, highlighting the role of electron-withdrawing, electron-donating, and steric substituents on xanthine oxidase and thymidine phosphorylase inhibition.
| Substituent type | Compound | Xanthine oxidase activity | Thymidine phosphorylase activity | SAR trend |
|---|---|---|---|---|
| Electron-withdrawing (NO2) | 8j | Very strong (IC50 = 1.87 µM) | Very strong (IC50 = 1.62 µM) | Enhances electrostatic interactions and hydrogen bonding |
| Electron-donating (multi-OCH3) | 8c, 8d | Strong | Strong | Improves π-stacking and aromatic interactions |
| Halogen + nitro | 8i | Strong | Moderate | Halogens enhance hydrophobic interactions |
| Neutral/weak | 8a, 8b, 8l | Moderate | Moderate-good | Activity maintained by scaffold planarity |
| Ortho-substitution | 8e, 8g | Weak | Weak | Steric hindrance disrupts ligand orientation |
Based on the SAR trends that we have observed, some design concepts can be proposed to further optimize the lophine–acylthiourea core structure. First, incorporating electron-withdrawing functional groups such as nitro substituents can improve binding affinity by enhancing electrostatic interactions. Second, the presence of multiple methoxy functional groups can facilitate in the facilitation of aromatic stacking interactions within the catalytic pocket of the enzyme. Third, it is best to avoid bulky substituents at the ortho-position due to steric clashes that can disrupt optimal ligand orientation.
From a pharmacology perspective, targeting xanthine oxidase and thymidine phosphorylase simultaneously may have potential advantages. Xanthine oxidase inhibitors have been employed in the treatment of hyperuricemia and gout because the inhibition of the enzyme reduces the production of uric acid and oxidative stress associated with various metabolic syndromes.63 In addition, thymidine phosphorylase has been identified as a significant enzyme in cancer because it is associated with the metabolism of nucleotides, which promotes cancer progression.64,65 Therefore, compounds that can modulate both targets and are sufficiently small to access both binding sites have the potential of providing a dual therapeutic strategy that combines antioxidant and anti-angiogenic activities.
Although further pharmacokinetic and in vivo studies are required to fully explore this possibility, the potent enzyme inhibition and docking of compounds 8j and 8d suggest that the lophine–acylthiourea template has the potential for the development of multifunctional agents. Collectively, the results have consistently highlighted compound 8j as the most promising dual-target inhibitor among the synthesized compounds.
3 Materials and methods
3.1 Procedure and material
All reagents used were devoid of any purification and bought from Sigma-Aldrich. Synthetic reactions were carried out in a cleaned and oven-dried apparatus. Solvents were dried using appropriate methods before their use in reactions. The melting point of all the synthesized solid compounds was determined in open-end capillaries using the Stuart SMP3 melting point apparatus. All the synthesized compounds were characterized by IR spectroscopy using a Thermo Fischer FT-IR instrument and 1H and 13C-NMR spectroscopy using Bruker Advance 300 and 75 MHz, respectively. NMR spectra were recorded in deuterated solvents such as DMSO-d6 and acetone-d6 using tetramethylsilane (TMS) as reference. Chemical shift values were reported in delta (δ) ppm, and coupling constant (J) values were calculated in hertz (Hz). The progress of each reaction was monitored by TLC using pre-coated silica-gel plates (Merck F254, 0.2 mm). Spots on TLC plates were visualized using UV light of 254 and 365 nm wavelength. Ninhydrin stain was used for confirmation of the amine functional group. Lassaigne's test was used for the detection of sulfur atoms in synthesized organic compounds.3.2 General synthetic pathway for synthesis of lophine clubbed acylthiourea derivatives (8a–l)
Steps involved in the synthesis of lophine clubbed acylthiourea derivatives are given below.3.3 In silico studies
Xanthine oxidase and thymidine phosphorylase coordinates were acquired from the Protein Data Bank using PDB codes 3NRZ and 4EAD.67,68 To determine whether the produced compounds might occupy the protein's active pocket in a fashion that would explain the enzyme's activity, a molecular docking investigation was carried out. 3D-structure of protein was downloaded from PDB and co-crystallized ligands and heteroatoms were removed. Then, molecular docking was performed by using the molecular operating environment (MOE) software.69 The structure of synthesized compounds was carried out by ChemOffice 3D (2015), and optimization of energy was performed by Gaussian 09 by using the density functional theory and 6-311G basis set.70 First, optimization of protein structure was performed by using the MOE software. Then, molecular docking was performed. Discovery visualizer (DSV) is used for the identification of 2D and 3D models for the docked compounds. The electrostatic and Lennard-Jones van der Waals interactions were found to be responsible for the retention in the active pocket. Each compound was given various poses because of the docking of compounds into the active pocket.3.4 Experimental data
White solid, m.p.: 228–229 °C, yield: 84%, Rf: 0.44 (n-hexane/EtOAc 7
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), FT-IR: ν (cm−1) 3215 (N–H), 3021 (Csp2–H stretch), 1668 (C
3), FT-IR: ν (cm−1) 3215 (N–H), 3021 (Csp2–H stretch), 1668 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, carbonyl), 1594 (C
O, carbonyl), 1594 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, aromatic), 1259 (C
C, aromatic), 1259 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 1148 (C–N). 1H-NMR: (300 MHz, DMSO-d6): δ (ppm) 12.780 (s, 1H, NH), 11.660 (s, 1H, NH), 8.137 (d, 3J = 8.4 Hz, 2H, Ar), 8.007 (d, 3J = 7.5 Hz, 2H, Ar), 7.891 (d, 3J = 8.4 Hz, 2H, Ar), 7.582–7.297 (m, 13H, Ar). 13C-NMR: (75 MHz, DMSO-d6): δ (ppm) 179.18 (C
S), 1148 (C–N). 1H-NMR: (300 MHz, DMSO-d6): δ (ppm) 12.780 (s, 1H, NH), 11.660 (s, 1H, NH), 8.137 (d, 3J = 8.4 Hz, 2H, Ar), 8.007 (d, 3J = 7.5 Hz, 2H, Ar), 7.891 (d, 3J = 8.4 Hz, 2H, Ar), 7.582–7.297 (m, 13H, Ar). 13C-NMR: (75 MHz, DMSO-d6): δ (ppm) 179.18 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 168.78 (C
S), 168.78 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 145.40 (C-imidazole), 138.48, 133.66, 133.17, 132.58, 129.19, 128.95, 128.28, 127.80, 126.04, 124.64 (C-aromatic). Anal. calcd. For C29H22N4OS (474.15): C, 73.39; N, 11.81; S, 6.76; H, 4.69% found: C, 73.37; N, 11.80; S7, 6.75; H, 4.66%.
O), 145.40 (C-imidazole), 138.48, 133.66, 133.17, 132.58, 129.19, 128.95, 128.28, 127.80, 126.04, 124.64 (C-aromatic). Anal. calcd. For C29H22N4OS (474.15): C, 73.39; N, 11.81; S, 6.76; H, 4.69% found: C, 73.37; N, 11.80; S7, 6.75; H, 4.66%.Yellow solid, m.p.: 215–217 °C, yield: 78%, Rf: 0.45 (n-hexane/EtOAc 7
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), FT-IR: ν (cm−1) 3310 (N–H), 2971 (Csp3–H), 1669 (C
3), FT-IR: ν (cm−1) 3310 (N–H), 2971 (Csp3–H), 1669 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, carbonyl), 1590 (C
O, carbonyl), 1590 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, aromatic), 1260 (C
C, aromatic), 1260 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 1146 (C–N). 1H-NMR: (300 MHz, DMSO-d6): δ (ppm) 12.862 (s, 1H, NH), 11.598 (s, 1H, NH), 8.147 (d, 3J = 8.7 Hz, 2H, Ar), 7.968–7.915 (m, 4H, Ar), 7.562–7.535 (m, 4H, Ar), 7.465–7.347 (m, 8H, Ar), 2.501 (s, 3H, aliphatic). 13C-NMR: (75 MHz, DMSO-d6): δ (ppm) 179.37 (C
S), 1146 (C–N). 1H-NMR: (300 MHz, DMSO-d6): δ (ppm) 12.862 (s, 1H, NH), 11.598 (s, 1H, NH), 8.147 (d, 3J = 8.7 Hz, 2H, Ar), 7.968–7.915 (m, 4H, Ar), 7.562–7.535 (m, 4H, Ar), 7.465–7.347 (m, 8H, Ar), 2.501 (s, 3H, aliphatic). 13C-NMR: (75 MHz, DMSO-d6): δ (ppm) 179.37 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 168.56 (C
S), 168.56 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 144.82 (C-imidazole), 144.23, 139.59, 131.90, 131.26, 129.54, 129.30, 129.15, 128.57, 126.80, 125.59, 124.69 (C-aromatic), 21.63 (C-methyl). Anal. calcd. For C30H24N4OS (488.17): C, 73.75; N, 11.47; S, 6.56; H, 4.95% found: C, 73.74; N, 11.46; S, 6.54; H, 4.94%.
O), 144.82 (C-imidazole), 144.23, 139.59, 131.90, 131.26, 129.54, 129.30, 129.15, 128.57, 126.80, 125.59, 124.69 (C-aromatic), 21.63 (C-methyl). Anal. calcd. For C30H24N4OS (488.17): C, 73.75; N, 11.47; S, 6.56; H, 4.95% found: C, 73.74; N, 11.46; S, 6.54; H, 4.94%.Yellow solid, m.p.: 232–233 °C, yield: 74%, Rf: 0.35 (n-hexane/EtOAc 7
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), FT-IR: ν (cm−1) 3315 (N–H), 3001 (Csp2–H), 1672 (C
3), FT-IR: ν (cm−1) 3315 (N–H), 3001 (Csp2–H), 1672 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, carbonyl), 1589 (C
O, carbonyl), 1589 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, aromatic), 1207 (C
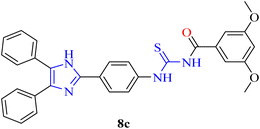
C, aromatic), 1207 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 1146 (C–N). 1H-NMR: (300 MHz, DMSO-d6): δ (ppm) 12.806 (s, 1H, NH), 11.679 (s, 1H, NH), 8.244 (d, 3J = 8.4 Hz, 2H, aromatic), 7.974 (d, 3J = 8.7 Hz, 2H, aromatic), 7.580–7.549 (m, 4H, aromatic), 7.478–7.401 (m, 6H, aromatic), 7.189 (d, 3J = 2.1 Hz, 2H, aromatic), 6.784 (t, 3J = 2.1 Hz, 1H, aromatic), 3.838 (s, 6H, methyl). 13C-NMR: (75 MHz, DMSO-d6): δ (ppm) 179.25 (C
S), 1146 (C–N). 1H-NMR: (300 MHz, DMSO-d6): δ (ppm) 12.806 (s, 1H, NH), 11.679 (s, 1H, NH), 8.244 (d, 3J = 8.4 Hz, 2H, aromatic), 7.974 (d, 3J = 8.7 Hz, 2H, aromatic), 7.580–7.549 (m, 4H, aromatic), 7.478–7.401 (m, 6H, aromatic), 7.189 (d, 3J = 2.1 Hz, 2H, aromatic), 6.784 (t, 3J = 2.1 Hz, 1H, aromatic), 3.838 (s, 6H, methyl). 13C-NMR: (75 MHz, DMSO-d6): δ (ppm) 179.25 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 168.03 (C
S), 168.03 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 160.73 (C-aromatic), 144.58 (C-imidazole), 140.01, 134.38, 131.40, 130.46, 129.18, 128.89, 128.77, 127.28, 124.60, 106.82, 105.93 (C-aromatic), 56.10 (C-methyl). Anal. calcd. For C31H26N4O3S (534.17): C, 69.64; N, 10.48; S, 6.00; H, 4.90% found: C, 69.62; N, 10.47; S, 5.98; H, 4.89%.
O), 160.73 (C-aromatic), 144.58 (C-imidazole), 140.01, 134.38, 131.40, 130.46, 129.18, 128.89, 128.77, 127.28, 124.60, 106.82, 105.93 (C-aromatic), 56.10 (C-methyl). Anal. calcd. For C31H26N4O3S (534.17): C, 69.64; N, 10.48; S, 6.00; H, 4.90% found: C, 69.62; N, 10.47; S, 5.98; H, 4.89%.Pale yellow solid, m.p.: 223–224 °C, yield: 78%, Rf: 0.25 (n-hexane/EtOAc 7
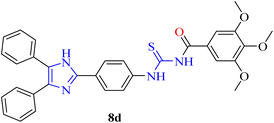
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), FT-IR: ν (cm−1) 3140 (N–H), 3031 (Csp2–H), 2888 (Csp3–H), 1680 (C
3), FT-IR: ν (cm−1) 3140 (N–H), 3031 (Csp2–H), 2888 (Csp3–H), 1680 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, carbonyl), 1580 (C
O, carbonyl), 1580 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, aromatic), 1235 (C
C, aromatic), 1235 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 1118 (C–N). 1H-NMR: (300 MHz, DMSO-d6): δ (ppm) 12.971 (s, 1H, NH), 11.748 (s, 1H, NH), 8.373 (d, 3J = 8.7 Hz, 2H, Ar), 8.057 (d, 3J = 8.7 Hz, 2H, Ar), 7.599–7.569 (m, 6H, Ar), 7.492–7.471 (m, 4H, Ar), 7.413 (s, 2H, Ar), 3.889 (s, 6H, methyl), 3.761 (s, 3H, methyl). 13C-NMR: (75 MHz, DMSO-d6): δ (ppm) 179.49 (C
S), 1118 (C–N). 1H-NMR: (300 MHz, DMSO-d6): δ (ppm) 12.971 (s, 1H, NH), 11.748 (s, 1H, NH), 8.373 (d, 3J = 8.7 Hz, 2H, Ar), 8.057 (d, 3J = 8.7 Hz, 2H, Ar), 7.599–7.569 (m, 6H, Ar), 7.492–7.471 (m, 4H, Ar), 7.413 (s, 2H, Ar), 3.889 (s, 6H, methyl), 3.761 (s, 3H, methyl). 13C-NMR: (75 MHz, DMSO-d6): δ (ppm) 179.49 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 167.69 (C
S), 167.69 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 143.89 (C-imidazole), 153.04, 142.13, 141.28, 130.05, 129.79, 129.35, 129.22, 128.38, 128.14, 127.04, 124.52, 121.52, 106.79 (C-aromatic), 60.64 (C-methyl), 56.63 (C-methyl). Anal. calcd. For C32H28N4O4S (564.18): C, 68.07; N, 9.92; S, 5.68; H, 4.90% found: C, 68.06; N, 9.90; S, 5.67; H, 4.88%.
O), 143.89 (C-imidazole), 153.04, 142.13, 141.28, 130.05, 129.79, 129.35, 129.22, 128.38, 128.14, 127.04, 124.52, 121.52, 106.79 (C-aromatic), 60.64 (C-methyl), 56.63 (C-methyl). Anal. calcd. For C32H28N4O4S (564.18): C, 68.07; N, 9.92; S, 5.68; H, 4.90% found: C, 68.06; N, 9.90; S, 5.67; H, 4.88%.Light orange solid, m.p.: 212–214 °C, yield: 83%, Rf: 0.61 (n-hexane/EtOAc 7
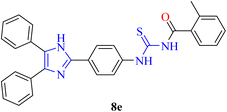
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), FT-IR: ν (cm−1) 3110 (N–H), 3010 (Csp2–H), 2924 (Csp3–H), 1674 (C
3), FT-IR: ν (cm−1) 3110 (N–H), 3010 (Csp2–H), 2924 (Csp3–H), 1674 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, carbonyl), 1591 (C
O, carbonyl), 1591 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, aromatic), 1255 (C
C, aromatic), 1255 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 1148 (C–N). 1H-NMR: (300 MHz, acetone-d6): δ (ppm) 13.098 (s, 1H, NH), 10.647 (s, 1H, NH), 8.400 (d, 3J = 8.7 Hz, 2H, Ar), 8.177 (d, 3J = 8.7 Hz, 2H, Ar), 7.666–7.617 (m, 5H, Ar), 7.513–7.452 (m, 7H, Ar), 7.374–7.348 (m, 2H, Ar), 2.537 (s, 3H, methyl). 13C-NMR: (75 MHz, acetone-d6): δ (ppm) 178.78 (C
S), 1148 (C–N). 1H-NMR: (300 MHz, acetone-d6): δ (ppm) 13.098 (s, 1H, NH), 10.647 (s, 1H, NH), 8.400 (d, 3J = 8.7 Hz, 2H, Ar), 8.177 (d, 3J = 8.7 Hz, 2H, Ar), 7.666–7.617 (m, 5H, Ar), 7.513–7.452 (m, 7H, Ar), 7.374–7.348 (m, 2H, Ar), 2.537 (s, 3H, methyl). 13C-NMR: (75 MHz, acetone-d6): δ (ppm) 178.78 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 170.71 (C
S), 170.71 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 144.15 (C-imidazole), 141.18, 136.93, 134.23, 133.66, 131.45, 131.07, 130.06, 129.28, 128.87, 128.79, 128.04, 127.94, 127.85, 125.77, 123.52, 123.37, 121.12 (C-aromatic), 19.15 (C-methyl). Anal. calcd. For C30H24N4OS (488.17): C, 73.75; N, 11.47; S, 6.56; H, 4.95% found: C, 73.74; N, 11.46; S, 6.54; H, 4.93%.
O), 144.15 (C-imidazole), 141.18, 136.93, 134.23, 133.66, 131.45, 131.07, 130.06, 129.28, 128.87, 128.79, 128.04, 127.94, 127.85, 125.77, 123.52, 123.37, 121.12 (C-aromatic), 19.15 (C-methyl). Anal. calcd. For C30H24N4OS (488.17): C, 73.75; N, 11.47; S, 6.56; H, 4.95% found: C, 73.74; N, 11.46; S, 6.54; H, 4.93%.Dark orange solid, m.p.: 204–206 °C, yield: 75%, Rf: 0.55 (n-hexane/EtOAc 7
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), FT-IR: ν (cm−1) 3110 (N–H), 3037 (Csp2–H), 1675 (C
3), FT-IR: ν (cm−1) 3110 (N–H), 3037 (Csp2–H), 1675 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, carbonyl), 1587 (C
O, carbonyl), 1587 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, aromatic), 1261 (C
C, aromatic), 1261 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 1314 and 1529 (NO2), 1130 (C–N). Anal. calcd. For C29H21N5O3S (519.14): C, 67.04; N, 13.48; S, 6.17; H, 4.07% found: C, 67.02; N, 13.49; S, 6.16; H, 4.06%.
S), 1314 and 1529 (NO2), 1130 (C–N). Anal. calcd. For C29H21N5O3S (519.14): C, 67.04; N, 13.48; S, 6.17; H, 4.07% found: C, 67.02; N, 13.49; S, 6.16; H, 4.06%.Dark orange solid, m.p.: 185–187 °C, yield: 80%, Rf: 0.41 (n-hexane/EtOAc 7
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), FT-IR: ν (cm−1) 3125 (N–H), 3017 (Csp2–H), 1677 (C
3), FT-IR: ν (cm−1) 3125 (N–H), 3017 (Csp2–H), 1677 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, carbonyl), 1585 (C
O, carbonyl), 1585 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, aromatic), 1259 (C
C, aromatic), 1259 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 1151 (C–N). Anal. calcd. For C29H21ClN4OS (508.11): C, 68.43; N, 11.01; S, 6.30; H, 4.16% found: C, 68.42; N, 11.00; S, 6.32; H, 4.14%.
S), 1151 (C–N). Anal. calcd. For C29H21ClN4OS (508.11): C, 68.43; N, 11.01; S, 6.30; H, 4.16% found: C, 68.42; N, 11.00; S, 6.32; H, 4.14%.Dark orange solid, m.p.: 230–231 °C, yield: 85%, Rf: 0.35 (n-hexane/EtOAc 7
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), FT-IR: ν (cm−1) 3305 (N–H), 3026 (Csp2–H), 1648 (C
3), FT-IR: ν (cm−1) 3305 (N–H), 3026 (Csp2–H), 1648 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, carbonyl), 1591 (C
O, carbonyl), 1591 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, aromatic), 1256 (C
C, aromatic), 1256 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 1147 (C–N). Anal. calcd. For C30H24N4O2S (504.16): C, 71.41; N, 11.10; S, 6.35; H, 4.79% found: C, 71.39; N, 11.11; S, 6.35; H, 4.78%.
S), 1147 (C–N). Anal. calcd. For C30H24N4O2S (504.16): C, 71.41; N, 11.10; S, 6.35; H, 4.79% found: C, 71.39; N, 11.11; S, 6.35; H, 4.78%.Pale yellow solid, m.p.: 275–277 °C, yield: 70%, Rf: 0.55 (n-hexane/EtOAc 7
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), FT-IR: ν (cm−1) 3128 (N–H), 3033 (Csp2–H), 2893 (Csp3–H), 1681 (C
3), FT-IR: ν (cm−1) 3128 (N–H), 3033 (Csp2–H), 2893 (Csp3–H), 1681 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, carbonyl), 1580 (C
O, carbonyl), 1580 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, aromatic), 1235 (C
C, aromatic), 1235 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 1108 (C–N). Anal. calcd. For C29H20ClN5O3S (553.10): C, 62.87; N, 12.64; S, 5.79; H, 3.64% found: C, 62.85; N, 12.63; S, 5.80; H, 3.63%.
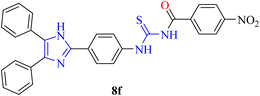
S), 1108 (C–N). Anal. calcd. For C29H20ClN5O3S (553.10): C, 62.87; N, 12.64; S, 5.79; H, 3.64% found: C, 62.85; N, 12.63; S, 5.80; H, 3.63%.White solid, m.p.: 215–217 °C, yield: 72%, Rf: 0.21 (n-hexane/EtOAc 7
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), FT-IR: ν (cm−1) 3301 (N–H), 2995 (Csp2–H), 2863 (Csp3–H), 1644 (C
3), FT-IR: ν (cm−1) 3301 (N–H), 2995 (Csp2–H), 2863 (Csp3–H), 1644 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, carbonyl), 1600 (C
O, carbonyl), 1600 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, aromatic), 1253 (C
C, aromatic), 1253 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 1128 (C–N). Anal. calcd. For C29H21N5O3S (519.14): C, 67.04; N, 13.48; S, 6.17; H, 4.07% found: C, 67.03; N, 13.47; S, 6.15; H, 4.08%.
S), 1128 (C–N). Anal. calcd. For C29H21N5O3S (519.14): C, 67.04; N, 13.48; S, 6.17; H, 4.07% found: C, 67.03; N, 13.47; S, 6.15; H, 4.08%.Beige color solid, m.p.: 237–238 °C, yield: 70%, Rf: 0.55 (n-hexane/EtOAc 7
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), FT-IR: ν (cm−1) 3118 (N–H), 3052 (Csp2–H), 1646 (C
3), FT-IR: ν (cm−1) 3118 (N–H), 3052 (Csp2–H), 1646 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, carbonyl), 1588 (C
O, carbonyl), 1588 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, aromatic), 12
C, aromatic), 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 338 (C
338 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 1089 (C–N). Anal. calcd. For C29H21ClN4OS (508.11): C, 68.43; N, 11.01; S, 6.30; H, 4.16% found: C, 68.41; N, 11.00; S, 6.31; H, 4.15%.
S), 1089 (C–N). Anal. calcd. For C29H21ClN4OS (508.11): C, 68.43; N, 11.01; S, 6.30; H, 4.16% found: C, 68.41; N, 11.00; S, 6.31; H, 4.15%.Orange solid, m.p.: 230–232 °C, yield: 74%, Rf: 0.37 (n-hexane/EtOAc 7
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), FT-IR: ν (cm−1) 3106 (N–H), 3049 (Csp2–H), 2833 (Csp3–H), 1669 (C
3), FT-IR: ν (cm−1) 3106 (N–H), 3049 (Csp2–H), 2833 (Csp3–H), 1669 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, carbonyl), 1598 (C
O, carbonyl), 1598 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, aromatic), 1254 (C
C, aromatic), 1254 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 1188 (C–N). Anal. calcd. For C30H24N4OS (488.17): C, 73.75; N, 11.47; S, 6.56; H, 4.95% found: C, 73.74; N, 11.45; S, 6.57; H, 4.94%.
S), 1188 (C–N). Anal. calcd. For C30H24N4OS (488.17): C, 73.75; N, 11.47; S, 6.56; H, 4.95% found: C, 73.74; N, 11.45; S, 6.57; H, 4.94%.4 Conclusion
A facile multistep synthesis of lophine clubbed acylthiourea derivatives is reported in this study involving heterocyclization, reduction, nucleophilic substitution, and addition reactions. In view of their relevance to advanced medicinal chemistry, the compounds were synthesized using straightforward procedures that avoided harsh conditions and minimized purification requirements. Beyond synthetic convenience, the lophine–acylthiourea derivatives were found to exert significant in vitro dual inhibition of xanthine oxidase and thymidine phosphorylase. Indeed, within this series, the analogs 8j, 8d, and 8i displayed very potent activity, with IC50 values in the low micromolar range. Molecular docking studies further supported these findings, as they indicated the binding interactions of the active compounds with the catalytic sites of both enzymes were favorable, and their accommodation was stable. The close agreement between in vitro results and computational predictions underlined the potential of this compound series as a versatile scaffold for modulating both oxidative stress and purine metabolism. Overall, the results provide a solid basis for future studies focusing on the rational design and development of dual-target enzyme inhibitors with therapeutic relevance.Conflicts of interest
There are no conflicts to declare.Data availability
The manuscript includes all relevant data which was generated or analyzed during this study. Additional information is included in the supplementary information (SI). Supplementary information: spectroscopic characterization data. See DOI: https://doi.org/10.1039/d6ra01337f.References
- C. Viegas-Junior, A. Danuello, V. da Silva Bolzani, E. J. Barreiro and C. A. M. Fraga, Molecular hybridization: a useful tool in the design of new drug prototypes, Curr. Med. Chem., 2007, 14, 1829–1852 CrossRef CAS PubMed
.
- A. Bhatnagar, P. Sharma and N. Kumar, A review on “Imidazoles”: Their chemistry and pharmacological potentials, Int. J. PharmTech Res., 2011, 3, 268–282 CAS
.
- A. Gueiffier, S. Mavel, M. Lhassani, A. Elhakmaoui, R. Snoeck, G. Andrei, O. Chavignon, J.-C. Teulade, M. Witvrouw and J. Balzarini, Synthesis of imidazo [1, 2-a] pyridines as antiviral agents, J. Med. Chem., 1998, 41, 5108–5112 CrossRef CAS PubMed
.
- S. Y. Atanasova-Stamova, S. F. Georgieva and M. B. Georgieva, Reaction strategies for synthesis of imidazole derivatives: a review, Scr. Sci. Pharm., 2018, 5, 7–13 Search PubMed
.
- R. A. Hill, Marine natural products, Annu. Rep. Prog. Chem., Sect. B:Org. Chem., 2009, 105, 150–166 RSC
.
- B. Forte, B. Malgesini, C. Piutti, F. Quartieri, A. Scolaro and G. Papeo, A submarine journey: The pyrrole-imidazole alkaloids, Mar. Drugs, 2009, 7, 705–753 CrossRef CAS PubMed
.
- Z. Jin, Muscarine, imidaozle, oxazole and thiazole alkaloids, Nat. Prod. Rep., 2013, 30, 869–915 RSC
.
- G. Gao, R. Xiao, Y. Yuan, C.-H. Zhou, J. You and R.-G. Xie, Efficient imidazolium catalysts for the benzoin condensation, J. Chem. Res., 2002, 2002, 262–263 CrossRef
.
- H.-Y. Jiang, C.-H. Zhou, K. Luo, H. Chen, J.-B. Lan and R.-G. Xie, Chiral imidazole metalloenzyme models: Synthesis and enantioselective hydrolysis for α-amino acid esters, J. Mol. Catal. A: Chem., 2006, 260, 288–294 CrossRef CAS
.
- H. V. Tolomeu and C. A. M. Fraga, Imidazole: synthesis, functionalization and physicochemical properties of a privileged structure in medicinal chemistry, Molecules, 2023, 28, 838 CrossRef CAS PubMed
.
- V. R. Pattabiraman and J. W. Bode, Rethinking amide bond synthesis, Nature, 2011, 480, 471–479 CrossRef CAS PubMed
.
- K. Liaras, A. Geronikaki, J. Glamočlija, A. Ćirić and M. Soković, Thiazole-based chalcones as potent antimicrobial agents. Synthesis and biological evaluation, Bioorg. Med. Chem., 2011, 19, 3135–3140 CrossRef CAS PubMed
.
- X. Lu, X. Liu, B. Wan, S. G. Franzblau, L. Chen, C. Zhou and Q. You, Synthesis and evaluation of anti-tubercular and antibacterial activities of new 4-(2, 6-dichlorobenzyloxy) phenyl thiazole, oxazole and imidazole derivatives. Part 2, Eur. J. Med. Chem., 2012, 49, 164–171 CrossRef CAS PubMed
.
- D. Varadaraji, S. S. Suban, V. R. Ramasamy, K. Kubendiran, J. S. K. Raguraman, S. K. Nalilu and H. N. Pati, Synthesis and evaluation of a series of 1-substituted tetrazole derivatives as antimicrobial agents, Org. Commun., 2010, 3, 45–56 CAS
.
- F. K. Keter and J. Darkwa, Perspective: the potential of pyrazole-based compounds in medicine, BioMetals, 2012, 25, 9–21 CrossRef CAS PubMed
.
- C.-H. Zhou and Y. Wang, Recent researches in triazole compounds as medicinal drugs, Curr. Med. Chem., 2012, 19, 239–280 CrossRef CAS PubMed
.
- L. Zhang, X. M. Peng, G. L. Damu, R. X. Geng and C. H. Zhou, Comprehensive review in current developments of imidazole-based medicinal chemistry, Med. Res. Rev., 2014, 34, 340–437 CrossRef CAS PubMed
.
- B. Radziszewski, Untersuchungen über hydrobenzamid, amarin und lophin, Ber. Dtsch. Chem. Ges., 1877, 10, 70–75 CrossRef
.
- R. Gandhaveeti, R. Konakanchi, P. Jyothi, N. S. Bhuvanesh and S. Anandaram, Unusual
coordination mode of aroyl/acyl thiourea ligands and their π-arene ruthenium (II) piano-stool complexes: Synthesis, molecular geometry, theoretical studies and biological applications, Appl. Organomet. Chem., 2019, 33, 4899 CrossRef
.
- F. Salomon, Ueber die Schwefelkohlensäureäther, J. Prakt. Chem., 1873,(6), 433–453 CrossRef
.
- D. C. Schroeder, Thioureas, Chem. Rev., 1955, 55, 181–228 CrossRef CAS
.
- Z.-n. Wang, H. Zhong, S. Wang, G.-y. Liu and Q. Zhang, Synthesis of 1, 4-benzenedicarbonyl thiourea resins and their adsorption properties for Ag (I), J. Cent. South Univ., 2011, 18, 361–366 CrossRef CAS
.
- Y. Nural, E. Karasu, E. Keleş, B. Aydıner, N. Seferoğlu, Ç. Efeoğlu, E. Şahin and Z. Seferoğlu, Synthesis of novel acylthioureas bearing naphthoquinone moiety as dual sensor for high-performance naked-eye colorimetric and fluorescence detection of CN− and F− ions and its application in water and food samples, Dyes Pigm., 2022, 198, 110006 CrossRef CAS
.
- E. M. Hoshall, Report on Preservatives and Artificial Sweeteners, J. Assoc. Off. Agric. Chem., 1964, 47, 68 CrossRef
.
- W. T. Lambert, M. E. Goldsmith and T. C. Sparks, Insecticidal activity of novel thioureas and isothioureas, Pest Manage. Sci., 2017, 73, 743–751 CrossRef CAS PubMed
.
- I. L. Goncalves, G. O. de Azambuja, D. F. Kawano and V. L. Eifler-Lima, Thioureas as building blocks for the generation of heterocycles and compounds with pharmacological activity: An overview, Mini-Rev. Org. Chem., 2018, 15, 28–35 CAS
.
- M. K. Rauf, A. Badshah, M. Gielen, M. Ebihara, D. de Vos and S. Ahmed, Synthesis, structural characterization and in vitro cytotoxicity and anti-bacterial activity of some copper (I) complexes with N, N′-disubstituted thioureas, J. Inorg. Biochem., 2009, 103, 1135–1144 CrossRef CAS PubMed
.
- J. M. Andrés, A. Maestro, M. Valle and R. Pedrosa, Chiral Bifunctional Thioureas and Squaramides and Their Copolymers as Recoverable Organocatalysts. Stereoselective Synthesis of 2-Substituted 4-Amino-3-nitrobenzopyrans and 3-Functionalized 3, 4-Diamino-4 H-Chromenes, J. Org. Chem., 2018, 83, 5546–5557 CrossRef PubMed
.
- T. Parvin, R. Yadav and L. H. Choudhury, Recent applications of thiourea-based organocatalysts in asymmetric multicomponent reactions (AMCRs), Org. Biomol. Chem., 2020, 18, 5513–5532 RSC
.
- S. A. Ullah, A. Saeed, M. Azeem, M. B. Haider and M. F. Erben, Exploring the latest trends in chemistry, structure, coordination, and diverse applications of 1-acyl-3-substituted thioureas: a comprehensive review, RSC Adv., 2024, 14, 18011–18063 RSC
.
- A. Maalik, H. Rahim, M. Saleem, N. Fatima, A. Rauf, A. Wadood, M. I. Malik, A. Ahmed, H. Rafique and M. N. Zafar, Synthesis, antimicrobial, antioxidant, cytotoxic, antiurease and molecular docking studies of N-(3-trifluoromethyl) benzoyl-N′-aryl thiourea derivatives, Bioorg. Chem., 2019, 88, 102946 CrossRef CAS PubMed
.
- M. Khan, J. Patujo, I. Mushtaq, A. Ishtiaq, M. N. Tahir, S. Bibi, M. S. Khan, G. Mustafa, B. Mirza and A. Badshah, Anti-diabetic potential, crystal structure, molecular docking, DFT, and optical-electrochemical studies of new dimethyl and diethyl carbamoyl-N, N′-disubstituted based thioureas, J. Mol. Struct., 2022, 1253, 132207 CrossRef CAS
.
- J.-h. Li, Y. Wang, Y.-p. Wu, R.-h. Li, S. Liang, J. Zhang, Y.-g. Zhu and B.-j. Xie, Synthesis, herbicidal activity study and molecular docking of novel pyrimidine thiourea, Pestic. Biochem. Physiol., 2021, 172, 104766 CrossRef CAS PubMed
.
- H. Wang, Z.-W. Zhai, Y.-X. Shi, C.-X. Tan, J.-Q. Weng, L. Han, B.-J. Li and X.-H. Liu, Novel trifluoromethylpyrazole acyl thiourea derivatives: synthesis, antifungal activity and docking study, Lett. Drug Des. Discovery, 2019, 16, 785–791 CrossRef CAS
.
- G. M. Viana, D. C. Soares, M. V. Santana, L. H. do Amaral, P. W. Meireles, R. P. Nunes, L. C. R. P. da Silva, L. C. de Sequeira Aguiar, C. R. Rodrigues and V. P. de Sousa, Antileishmanial thioureas: synthesis, biological activity and in silico evaluations of new promising derivatives, Chem. Pharm. Bull., 2017, 65, 911–919 CrossRef CAS PubMed
.
- M. Irani, K. R. Ranjbar and H. Izadi, The Synthesis of Some New Benzoyl Thioureas and Investigation of Pesticide Activity, 2014 Search PubMed
.
- T. A. Fattah, A. Saeed, Z. Ashraf, Q. Abbas, P. A. Channar, F. A. Larik and M. Hassan, 4-Aminocoumarin based aroylthioureas as potential jack bean urease inhibitors; synthesis, enzyme inhibitory kinetics and docking studies, Med. Chem., 2020, 16, 229–243 CrossRef CAS PubMed
.
- İ. Koca, A. Özgür, K. A. Coşkun and Y. Tutar, Synthesis and anticancer activity of acyl thioureas bearing pyrazole moiety, Bioorg. Med. Chem., 2013, 21, 3859–3865 CrossRef PubMed
.
- P. Reigan, A. Gbaj, I. J. Stratford, R. A. Bryce and S. Freeman, Xanthine oxidase-activated prodrugs of thymidine phosphorylase inhibitors, Eur. J. Med. Chem., 2008, 43, 1248–1260 CrossRef CAS PubMed
.
- S. Kalra, G. Jena, K. Tikoo and A. K. Mukhopadhyay, Preferential inhibition of xanthine oxidase by 2-amino-6-hydroxy-8-mercaptopurine and 2-amino-6-purine thiol, BMC Biochem., 2007, 8, 8 CrossRef PubMed
.
- S. Prachayasittikul, R. Pingaew, A. Worachartcheewan, N. Sinthupoom, V. Prachayasittikul, S. Ruchirawat and V. Prachayasittikul, Roles of pyridine and pyrimidine derivatives as privileged scaffolds in anticancer agents, Mini-Rev. Med. Chem., 2017, 17, 869–901 CAS
.
- L.-X. Zhao, K. Luo, X.-D. Guo, Y.-L. Zou, S. Gao, Y. Fu and F. Ye, Design, Synthesis, and Biological Activity Evaluation of Novel Phenoxypyridine Derivatives Containing Acylthiourea Fragments as Protoporphyrinogen Oxidase Inhibitor Herbicides, J. Agric. Food Chem., 2025, 73, 5020–5032 CrossRef CAS PubMed
.
- K. Okamoto, B. T. Eger, T. Nishino, E. F. Pai and T. Nishino, Mechanism of inhibition of xanthine oxidoreductase by allopurinol: crystal structure of reduced bovine milk xanthine oxidoreductase bound with oxipurinol, Nucleosides, Nucleotides Nucleic Acids, 2008, 27, 888–893 CrossRef CAS PubMed
.
- H. Xue, M. Xu, D. Gong and G. Zhang, Mechanism of flavonoids inhibiting xanthine oxidase and alleviating hyperuricemia from structure–activity relationship and animal experiments: A review, Food Front., 2023, 4, 1643–1665 CrossRef CAS
.
- A. Mehmood, J. Li, A. U. Rehman, R. Kobun, I. U. Llah, I. Khan, F. Althobaiti, S. Albogami, M. Usman, F. Alharthi, M. M. Soliman, S. Yaqoob, K. A. Awan, L. Zhao and L. Zhao, Xanthine oxidase inhibitory study of eight structurally diverse phenolic compounds, Front. Nutr., 2022, 9, 966557 CrossRef PubMed
.
- J. Chen, Z. He, S. Yu, X. Cai, D. Zhu and Y. Lin, Xanthine oxidase inhibitory kinetics and mechanism of ellagic acid: In vitro, in silico and in vivo studies, IET Nanobiotechnol., 2023, 17, 368–375 CrossRef CAS PubMed
.
- Q. Feng, W. Yang, Z. Peng and G. Wang, Recent advances in the synthetic thymidine phosphorylase inhibitors for cancer therapy, Eur. J. Pharmacol., 2022, 934, 175319 CrossRef CAS PubMed
.
- M. F. S. de Abreu, C. A. Wegermann, M. S. Ceroullo, I. G. M. Sant'Anna and R. C. S. Lessa, Ten Years Milestones in Xanthine Oxidase Inhibitors Discovery: Febuxostat-Based Inhibitors Trends, Bifunctional Derivatives, and Automatized Screening Assays, Organics, 2022, 3, 380–414 CrossRef CAS
.
- A. Gul, S. M. Saad, H. Zafar, W. Atia Tul, K. M. Khan and M. I. Choudhary, In vitro and In silico Xanthine Oxidase Inhibitory Activities of
3-Aryl-2- thioxo-2,3-dihydroquinazolin-4(1H)-one Derivatives, Med. Chem., 2023, 19, 384–392 CrossRef CAS PubMed
.
- O. L. Kobzar, I. M. Mischenko, A. V. Tatarchuk, V. S. Vdovin, S. S. Lukashov, S. M. Yarmoluk and A. I. Vovk, Nitro-substituted aurones as xanthine oxidase inhibitors, Ukr. Bioorg. Acta, 2021, 16, 12–17 Search PubMed
.
- M. A. Sajid, Z. A. Khan, S. A. Shahzad, S. A. R. Naqvi, M. Usman and A. Iqbal, Recent advances in thymidine phosphorylase inhibitors: Syntheses and prospective medicinal applications, Turk. J. Chem., 2017, 41, 1–28 CrossRef CAS
.
- K. Zaman, F. Rahim, M. Taha, A. Wadood, S. A. A. Shah, Q. U. Ahmed and Z. A. Zakaria, Synthesis of new isoquinoline-base-oxadiazole derivatives as potent inhibitors of thymidine phosphorylase and molecular docking study, Sci. Rep., 2019, 9, 16015 CrossRef PubMed
.
- R. Hussain, W. Rehman, F. Rahim, S. Khan, A. S. Alanazi, M. M. Alanazi, L. Rasheed, Y. Khan, S. A. A. Shah and M. Taha, Synthesis, in vitro thymidine phosphorylase inhibitory activity and molecular docking study of novel pyridine-derived bis-oxadiazole bearing bis-schiff base derivatives, Arabian J. Chem., 2023, 16, 104773 CrossRef CAS
.
- N. Zhai, C. Wang, F. Wu, L. Xiong, X. Luo, X. Ju and G. Liu, Exploration of Novel Xanthine Oxidase Inhibitors Based on 1,6-Dihydropyrimidine-5-Carboxylic Acids by an Integrated in Silico Study, Int. J. Mol. Sci., 2021, 22, 8122 CrossRef CAS PubMed
.
- T. M. Cui, M. Altaf, A. Aldarhami, A. S. Bazaid, N. H. Saeedi, A. A. Alkayyal, F. M. Alshabrmi, F. Ali, M. Aladhadh, M. Y. Khan, A. A. Alsaiari and Y. R. Ma, Dihydropyrimidone Derivatives as Thymidine Phosphorylase Inhibitors: Inhibition Kinetics, Cytotoxicity, and Molecular Docking, Molecules, 2023, 28, 3634 CrossRef CAS PubMed
.
- H. Zaman, A. Saeed, H. Ismail and M. Rashid, Unveiling the cyclopropyl appended acyl thiourea derivatives as antimicrobial, α-amylase and proteinase K inhibitors: Design, synthesis, biological evaluation, molecular docking, DFT and ADMET studies, Arch. Biochem. Biophys., 2025, 765, 110304 CrossRef CAS PubMed
.
- F. U. Rahman, M. Bibi, E. Khan, A. B. Shah, M. Muhammad, M. N. Tahir, A. Shahzad, F. Ullah, M. Zahoor, S. Alamery and G. E. Batiha, Thiourea Derivatives, Simple in Structure but Efficient Enzyme Inhibitors and Mercury Sensors, Molecules, 2021, 26, 4506 CrossRef CAS PubMed
.
- R. Ronchetti, G. Moroni, A. Carotti, A. Gioiello and E. Camaioni, Recent advances in urea- and thiourea-containing compounds: focus on innovative approaches in medicinal chemistry and organic synthesis, RSC Med. Chem., 2021, 12, 1046–1064 RSC
.
- E. Noriega-Iribe, L. Díaz-Rubio, A. Estolano-Cobián, V. W. Barajas-Carrillo, J. M. Padrón, R. Salazar-Aranda, R. Díaz-Molina, V. García-González, R. A. Chávez-Santoscoy, D. Chávez and I. Córdova-Guerrero, In Vitro and In Silico Screening of 2,4,5-Trisubstituted Imidazole Derivatives as Potential Xanthine Oxidase and Acetylcholinesterase Inhibitors, Antioxidant, and Antiproliferative Agents, Appl. Sci., 2020, 10, 2889 CrossRef CAS
.
- N. Arshad, U. Parveen, P. A. Channar, A. Saeed, W. S. Saeed, F. Perveen, A. Javed, H. Ismail, M. I. Mir, A. Ahmed, B. Azad and I. Khan, Investigation of Newly Synthesized Bis-Acyl-Thiourea Derivatives of 4-Nitrobenzene-1,2-Diamine for Their DNA Binding, Urease Inhibition, and Anti-Brain-Tumor Activities, Molecules, 2023, 28, 2707 CrossRef CAS PubMed
.
- T. Glomb, K. Szymankiewicz and P. Świątek, Anti-Cancer Activity of Derivatives of 1,3,4-Oxadiazole, Molecules, 2018, 23, 3361 CrossRef PubMed
.
- A. Murmu, P. Banjare, B. W. Matore, P. P. Roy and J. Singh, 1, 3, 4-Oxadiazole: an emerging scaffold to inhibit the thymidine phosphorylase as an anticancer agent, Curr. Med. Chem., 2024, 31, 6227–6250 CrossRef CAS PubMed
.
- T. Nishikawa, N. Nagata, T. Shimakami, T. Shirakura, C. Matsui, Y. Ni, F. Zhuge, L. Xu, G. Chen, M. Nagashimada, T. Yamashita, Y. Sakai, T. Yamashita, E. Mizukoshi, M. Honda, S. Kaneko and T. Ota, Xanthine oxidase inhibition attenuates insulin resistance and diet-induced steatohepatitis in mice, Sci. Rep., 2020, 10, 815 CrossRef CAS PubMed
.
- B. Huang, Q. Yuan, J. Sun, C. Wang and D. Yang, Thymidine phosphorylase in nucleotide metabolism: physiological functions and its implications in tumorigenesis and anti-cancer therapy, Front. Immunol., 2025, 16, 1561560 CrossRef CAS PubMed
.
- T. Furukawa, S. Tabata, M. Yamamoto, K. Kawahara, Y. Shinsato, K. Minami, M. Shimokawa and S.-i. Akiyama, Thymidine phosphorylase in cancer aggressiveness and chemoresistance, Pharmacol. Res., 2018, 132, 15–20 CrossRef CAS PubMed
.
- A. Puratchikody, M. Nallu and S. Gopalakrishnan, Synthesis and Pharmacological Evaluation of Some Potent 2-(4-Substitutedphenyl)-4,5-Diphenyl-1H-Imidazoles, Indian J. Pharm. Sci., 2005, 67, 725–731 CAS
.
- S. A. Shahzad, M. Yar, Z. A. Khan, L. Shahzadi, S. A. R. Naqvi, A. Mahmood, S. Ullah, A. J. Shaikh, T. A. Sherazi and A. T. Bale, Identification of 1, 2, 4-triazoles as new thymidine phosphorylase inhibitors: Future anti-tumor drugs, Bioorg. Chem., 2019, 85, 209–220 CrossRef CAS PubMed
.
- S. Chakravarty and K. Kannan, Drug-protein interactions: refined structures of three sulfonamide drug complexes of human carbonic anhydrase I enzyme, J. Mol. Biol., 1994, 243, 298–309 CrossRef CAS PubMed
.
- S. Vilar, G. Cozza and S. Moro, Medicinal chemistry and the molecular operating environment (MOE): application of QSAR and molecular docking to drug discovery, Curr. Top. Med. Chem., 2008, 8, 1555–1572 CrossRef CAS PubMed
.
- A. Frisch, Gaussian 09W Reference, Wallingford, USA, 2009, vol. 470, p. , p. 25 Search PubMed
.
- S. A. Shahzad, M. Yar, M. Bajda, L. Shahzadi, Z. A. Khan, S. A. R. Naqvi, S. Mutahir, N. Mahmood and K. M. Khan, Synthesis, thymidine phosphorylase inhibition and molecular modeling studies of 1, 3, 4-oxadiazole-2-thione derivatives, Bioorg. Chem., 2015, 60, 37–41 CrossRef CAS PubMed
.
- D. A. Kostić, D. S. Dimitrijević, G. S. Stojanović, I. R. Palić, A. S. Đorđević and J. D. Ickovski, Xanthine oxidase: isolation, assays of activity, and inhibition, J. Chem., 2015, 294858 CrossRef
.
- J. Zhao, L. Huang, C. Sun, D. Zhao and H. Tang, Studies on the structure-activity relationship and interaction mechanism of flavonoids and xanthine oxidase through enzyme kinetics, spectroscopy methods and molecular simulations, Food Chem., 2020, 323, 126807 CrossRef CAS PubMed
.
| This journal is © The Royal Society of Chemistry 2026 |