Open Access Article
Open Access ArticleStrategic modular assembly of trifluoromethylated energetic materials via triazine-based scaffolds
Jiyuan Yu ,
Xiangyan Miao
,
Xiangyan Miao and
Yuchuan Li
and
Yuchuan Li *
*
School of Materials Science & Engineering, Beijing Institute of Technology, Beijing 100081, China
First published on 13th May 2026
Abstract
Nitrogen-rich frameworks with good thermal stability and low sensitivity have attracted considerable interest as potential energetic materials. Herein, a modular assembly strategy based on nucleophilic aromatic substitution (SNAr) on chloro-1,3,5-triazines was employed to construct triazine-bridged, CF3-functionalized nitrogen-rich architectures, affording compounds 3 and 6. Subsequent salt formation gave 4 and 5, and the structures of 4 and 5 were confirmed by single-crystal X-ray diffraction. Among the compounds examined, 4 exhibits a relatively high density (ρ = 1.80 g cm−3), improved detonation performance (D = 7664 m s−1, P = 25.2 GPa), very low sensitivity (IS > 40 J, FS > 360 N), and good thermal stability (Td,onset = 241.4 °C). These results show that triazine-based scaffold assembly combined with perchlorate modulation is a useful approach to tuning the balance among thermal stability, energetic performance, and low sensitivity in CF3-containing nitrogen-rich systems.
Introduction
Energetic materials (EMs) are indispensable in explosives and propellants, where their practical use requires a delicate balance among detonation performance, thermal stability, and operational safety.1 These materials are critically employed in defense, space exploration, and various civilian industries.2,3 However, optimizing all key properties simultaneously is challenging, as improvements in one aspect often compromise another.4 Representative examples of classical heat-resistant energetic compounds include 2,2′,4,4′,6,6′-hexanitrostilbene (HNS, Td = 318 °C, D = 7612 m s−1, P = 24.3 GPa) and 2,6-bis(picrylamino)-3,5-dinitropyridine (PYX, Td = 360 °C, D = 7757 m s−1, P = 25.1 GPa). However, their relatively high mechanical sensitivities (HNS: IS = 5 J, FS = 240 N; PYX: IS = 10 J, FS = 240 N) (Fig. 1) also illustrate the persistent trade-off between thermal robustness and safety.5–7 Consequently, there remains an urgent need to develop EMs that are both thermally robust and significantly less sensitive.8 | ||
| Fig. 1 (a) N-heterocycle bridging for enhanced thermal stability. (b) CF3 functionalization for increased density. (c) This work. | ||
From a structural perspective, nitrogen-rich fused heterocycles paired with robust hydrogen-bond networks in the crystal lattice are highly favorable motifs for reducing mechanical sensitivity. These frameworks benefit from dense heteroatom arrays that support directional intermolecular contacts, thereby facilitating stress delocalization.9,10 For example, TATOT (a 5/5-fused triazolo[4,3-b][1,2,4]triazole) exemplifies such a nitrogen-rich platform: it combines high intrinsic energy (ΔfH = 446.7 kJ mol−1; D = 9385 m s−1; P = 29.7 GPa) with remarkably low sensitivity (IS = 40 J, FS = 360 N). This outstanding insensitivity is attributed to an extensive amino-driven hydrogen-bonding network in the crystal (Fig. 1).11,12 However, all-amino fused systems are typically oxygen-deficient, indicating that additional design strategies are needed to maintain safety while further enhancing energetic output.13
In addition to fused-ring construction, incorporation of a CF3 group is an effective means of improving energetic materials, particularly by increasing density and lowering mechanical sensitivity.14,15 Previous studies have shown that CF3-containing nitrogen-rich fused frameworks can combine low sensitivity with relatively high density and useful detonation performance. For example, precursor 2, 6-(trifluoromethyl)-7H-[1,2,4]tri-azolo[4,3-b][1,2,4]triazole-3,7-diamine, exhibits a density of 1.82 g cm−3, a detonation velocity of 7706 m s−1, and a detonation pressure of 21.7 GPa, while maintaining low sensitivity (IS = 40 J, FS = 360 N).16 Fluorinated frameworks are also of interest for aluminium-containing energetic systems because fluorine-containing species can promote surface alumina conversion and facilitate aluminium ignition and combustion.17 Nevertheless, the intrinsic oxygen deficiency of CF3-containing scaffolds limits further improvement in energetic output and therefore calls for complementary structural regulation.
Among the available molecular design strategies for next-generation EMs, the electron-deficient 1,3,5-triazine ring is an attractive motif because it enables modular assembly via SNAr reactions and can reorganise intermolecular interactions in the solid state.18,19 Previous studies have shown that fusion of a triazine unit into a nitrogen-rich framework, followed by perchlorate formation, can improve thermal stability and sensitivity. For example, 2,4,7-triamino-8-nitropyrazolo[1,5-a][1,3,5]triazin-1-ium perchlorate exhibits a higher onset decomposition temperature than its precursor 3,5-diamino-4-nitro-1H-pyrazol-2-ium perchlorate (279 vs. 194 °C), while the impact and friction sensitivities are reduced from 5 J and 160 N to 40 J and 240 N, respectively.20 These results indicate that the combination of triazine incorporation and oxygen-rich counteranion modulation is a viable strategy for optimising the overall behaviour of nitrogen-rich energetic frameworks.21
On this basis, we sought to integrate a CF3-functionalised fused precursor with a chlorotriazine platform through a triazine-based SNAr strategy. In this work, neutral triazine-bridged frameworks 3 and 6 were constructed from precursor 2, and 3 was further converted into salts 4 and 5 through ionic modulation with perchlorate and nitrate. This design was intended to compensate for the oxygen deficiency of the CF3-containing scaffold and to tune density, decomposition behaviour, detonation performance, and mechanical sensitivity within a single modular platform.22,23 In particular, perchlorate salt 4 retains low sensitivity while increasing the density from 1.71 to 1.80 g cm−3 and improving detonation performance from 6994 m s−1/19.3 GPa to 7664 m s−1/25.2 GPa, indicating that this strategy is useful for improving the overall property balance of this CF3-containing energetic system.
Results and discussion
Synthesis
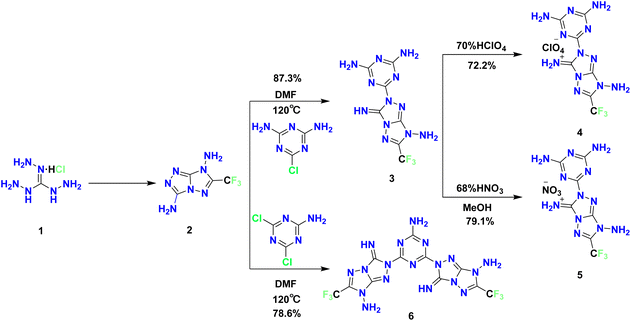
As shown in Scheme 1, precursor 2, prepared according to the reported procedure,24 reacted with 2-chloro-4,6-diamino-1,3,5-triazine to afford 3 in 87.3% isolated yield, whereas coupling with 4,6-dichloro-1,3,5-triazine-2-amine gave 6 in 78.6% yield. Subsequent salt formation of 3 with perchloric acid and nitric acid furnished 4 and 5 in 72.2% and 79.1% yields, respectively. These results indicate that the present triazine-based SNAr strategy proceeds efficiently under straightforward experimental conditions. It should be noted that the single crystals of 4·H2O and 5·5H2O were obtained from aqueous recrystallization for X-ray diffraction analysis only. In contrast, the bulk samples used for DSC measurements, density determination, sensitivity testing, and energetic calculations were isolated as solid powders from synthesis, washed with acetonitrile, and dried prior to use. Therefore, the hydrated crystal compositions observed in the single-crystal structures do not necessarily represent the state of the bulk samples employed in the physicochemical measurements discussed below.X-ray crystallographic analysis
Compound 4·H2O (CCDC: 2525729) crystallizes in the triclinic crystal system with the P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group (Fig. 2). The unit–cell parameters are a = 13.619(2) Å, b = 15.295(3) Å, c = 17.404(2) Å, α = 69.224(6)°, β = 69.711(6)°, and γ = 83.327(7)°, giving a cell volume of 3179.2(9) Å3 with Z = 2. The calculated crystal density is 1.757 g cm−3 at 170 K. The asymmetric unit comprises a protonated cation, perchlorate counterions, and lattice solvent/water species modelled over disordered sites. In the solid state, extensive intermolecular N–H⋯O hydrogen bonds between amino/imino donors and perchlorate oxygen atoms (H⋯O = 1.94–2.53 Å; N⋯O = 2.80–3.28 Å), together with N–H⋯N contacts among neighbouring nitrogen-rich heterocycles (H⋯N = 2.09–2.43 Å), connect the ionic components into a robust three-dimensional hydrogen-bonded network.
space group (Fig. 2). The unit–cell parameters are a = 13.619(2) Å, b = 15.295(3) Å, c = 17.404(2) Å, α = 69.224(6)°, β = 69.711(6)°, and γ = 83.327(7)°, giving a cell volume of 3179.2(9) Å3 with Z = 2. The calculated crystal density is 1.757 g cm−3 at 170 K. The asymmetric unit comprises a protonated cation, perchlorate counterions, and lattice solvent/water species modelled over disordered sites. In the solid state, extensive intermolecular N–H⋯O hydrogen bonds between amino/imino donors and perchlorate oxygen atoms (H⋯O = 1.94–2.53 Å; N⋯O = 2.80–3.28 Å), together with N–H⋯N contacts among neighbouring nitrogen-rich heterocycles (H⋯N = 2.09–2.43 Å), connect the ionic components into a robust three-dimensional hydrogen-bonded network.
Compound 5·5H2O (CCDC: 2530986) crystallizes in the triclinic crystal system with the P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group (Fig. 3). The calculated crystal density is 1.632 g cm−3 at 100 K. The asymmetric unit contains a protonated cation, one nitrate anion, and five lattice water molecules. The cation adopts an almost planar conformation, which is further stabilized by an intramolecular N–H⋯N contact (N18–H18B⋯N15, N⋯N = 2.770 Å). In the crystal, the amino group donates a bifurcated hydrogen bond to the nitrate oxygen atoms (N16–H16A⋯O25/O26, D⋯A = 3.128/2.991 Å), thereby anchoring the anion adjacent to the cation. Meanwhile, the lattice water molecules are linked through short O–H⋯O hydrogen bonds (O⋯O = 2.723–2.803 Å) to generate an extended hydration network that bridges the ionic components. As a result, the packing is governed predominantly by hydrogen-bonding interactions, while neighbouring π-surfaces are separated by the hydrated nitrate/water units rather than by pronounced face-to-face π–π stacking.
space group (Fig. 3). The calculated crystal density is 1.632 g cm−3 at 100 K. The asymmetric unit contains a protonated cation, one nitrate anion, and five lattice water molecules. The cation adopts an almost planar conformation, which is further stabilized by an intramolecular N–H⋯N contact (N18–H18B⋯N15, N⋯N = 2.770 Å). In the crystal, the amino group donates a bifurcated hydrogen bond to the nitrate oxygen atoms (N16–H16A⋯O25/O26, D⋯A = 3.128/2.991 Å), thereby anchoring the anion adjacent to the cation. Meanwhile, the lattice water molecules are linked through short O–H⋯O hydrogen bonds (O⋯O = 2.723–2.803 Å) to generate an extended hydration network that bridges the ionic components. As a result, the packing is governed predominantly by hydrogen-bonding interactions, while neighbouring π-surfaces are separated by the hydrated nitrate/water units rather than by pronounced face-to-face π–π stacking.
Thermal properties
The thermal behavior of compounds 3–6 was investigated by DSC under N2 at heating rates of 5, 10, 15, and 20 °C min−1 (Fig. 4). All four compounds showed a dominant exothermic decomposition event, with Tp shifting to higher temperature as the heating rate increased. No distinct low-temperature endothermic signal attributable to dehydration was observed for the measured bulk samples, suggesting that these powders did not retain the same hydrated state as the recrystallized single-crystal hydrates. At 10 °C min−1, compound 3 exhibited the highest Tonset (254 °C; Tp = 257 °C), whereas salts 4 and 5 showed earlier onsets (Tonset = 241 °C for both) but similar Tp values (258 and 257 °C, respectively). Compound 6 displayed the lowest thermal stability, with Tonset = 234 °C and Tp = 237 °C. In addition, ΔT values for 4 and 5 were much larger than those for 3 and 6, indicating a more gradual initial decomposition process in the salt forms. Compared with precursor 2 (Tonset = 215 °C), compounds 3–6 show improved decomposition thresholds, indicating that incorporation of the triazine unit enhances thermal resistance within this CF3-containing fused framework, whereas salt formation mainly modifies the onset behavior rather than further increasing Tonset. | ||
| Fig. 4 DSC curves (different heating rates) and Kissinger curves of (a) compound 3, (b) compound 4, (c) compound 5 and (d) compound 6. | ||
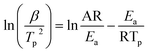 | (1) |
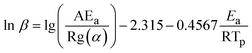 | (2) |
Ozawa plots of log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) β versus 1000/Tp yielded excellent linearity (R2 ≥ 0.98), affording Ea values of 222.3 kJ mol−1 for 3, 141.9 kJ mol−1 for 4, 147.22 kJ mol−1 for 5, and 186.3 kJ mol−1 for 6 (Fig. S19–S22). These values are in close agreement with those obtained from Kissinger's method (224.99, 140.36, 146.01, and 187.43 kJ mol−1 for 3, 4, 5 and 6, respectively), confirming the reliability of the kinetic analysis. The trend in decomposition barriers (3 > 6 > 5 > 4) parallels the order of their onset temperatures, indicating that incorporation of oxygen-containing counter-anions via salt formation markedly lowers the activation energy (by ∼75–80 kJ mol−1 relative to 3) while maintaining a similar Tp at 10 °C min−1 for the salts (Tp = 257.46 °C for 4 and 256.50 °C for 5, versus 256.78 °C for 3). Overall, compound 3 exhibits the highest thermal decomposition barrier, followed by the dimer 6, whereas the ionic salts 4 and 5 show substantially reduced Ea values, consistent with their facilitated initial decomposition. This hierarchy reflects how salt formation modulates the decomposition kinetics: the ionic salt 4/5 initiates decomposition more readily than neutral 3, yet still achieves a peak decomposition temperature on par with 3. These results indicate that targeted structural modifications can preserve thermal stability while adjusting decomposition kinetics, which is critical for designing next-generation insensitive energetic materials.
β versus 1000/Tp yielded excellent linearity (R2 ≥ 0.98), affording Ea values of 222.3 kJ mol−1 for 3, 141.9 kJ mol−1 for 4, 147.22 kJ mol−1 for 5, and 186.3 kJ mol−1 for 6 (Fig. S19–S22). These values are in close agreement with those obtained from Kissinger's method (224.99, 140.36, 146.01, and 187.43 kJ mol−1 for 3, 4, 5 and 6, respectively), confirming the reliability of the kinetic analysis. The trend in decomposition barriers (3 > 6 > 5 > 4) parallels the order of their onset temperatures, indicating that incorporation of oxygen-containing counter-anions via salt formation markedly lowers the activation energy (by ∼75–80 kJ mol−1 relative to 3) while maintaining a similar Tp at 10 °C min−1 for the salts (Tp = 257.46 °C for 4 and 256.50 °C for 5, versus 256.78 °C for 3). Overall, compound 3 exhibits the highest thermal decomposition barrier, followed by the dimer 6, whereas the ionic salts 4 and 5 show substantially reduced Ea values, consistent with their facilitated initial decomposition. This hierarchy reflects how salt formation modulates the decomposition kinetics: the ionic salt 4/5 initiates decomposition more readily than neutral 3, yet still achieves a peak decomposition temperature on par with 3. These results indicate that targeted structural modifications can preserve thermal stability while adjusting decomposition kinetics, which is critical for designing next-generation insensitive energetic materials.
Energy and sensitivity properties
The energetic performance of compounds 3–6 was evaluated using Explo5 (v6.01)27 based on their heats of formation (ΔfH), obtained from quantum-chemical calculations (Gaussian 09, Tables S8 and S9), and experimentally measured densities (ρ).28 The density values used for these evaluations were 1.71, 1.80, 1.78, and 1.66 g cm−3 for compounds 3, 4, 5, and 6, respectively. For compounds 4 and 5, these physicochemical data correspond to the bulk samples used for the measurements rather than to the hydrated single-crystal forms obtained from aqueousrecrystallization. The calculated ΔfH values are −5.73 kJ mol−1 for 3, +13.67 kJ mol−1 for 4, −619.5 kJ mol−1 for 5, and +20.8 kJ mol−1 for 6. For comparison of thermal stability, Td is defined here as the onset decomposition temperature (Tonset) measured by DSC at 10 °C min−1 under N2, giving values of 253 °C (3), DSC at 10 °C min−1 under N2, giving values of 253 °C (3), 241 °C (4), 241 °C (5), and 234 °C (6). As shown in Table 1, the neutral compounds 3 and 6 exhibit moderate detonation performance, with D/P values of 6994 m s−1/19.3 GPa and 6812 m s−1/19.2 GPa, respectively, which are close to those of TNT (7303 m s−1 and 21.3 GPa). Although compound 6 has a higher positive ΔfH than 3, its lower density results in slightly reduced detonation output. In contrast, conversion of 3 into the perchlorate salt 4 leads to a marked increase in density (1.71 → 1.80 g cm−3) and a corresponding enhancement in detonation performance (D = 7664 m s−1, P = 25.2 GPa). These values approach those of HNS (7612 m s−1, 24.3 GPa) and PYX (7757 m s−1, 25.1 GPa), although compound 4 still decomposes at a lower onset temperature than these classical thermostable explosives. The nitrate salt 5 also shows a density increase relative to 3 (1.78 g cm−3) and exhibits intermediate performance (D = 7228 m s−1, P = 20.8 GPa), lying between neutral 3 and perchlorate salt 4.| Compd | Tda (°C) | Dvb (m s−1) | Pc (GPa) | ΔfHd (kJ mol−1) | ISe (J) | FSf (N) | ρg (g cm−3) |
|---|---|---|---|---|---|---|---|
| a Densities, sensitivities and energetic calculations for 3–6 correspond to the dried bulk samples. Thermal decomposition temperature (onset) under nitrogen gas (DSC, 10 °C min−1).b Calculated detonation velocity (calculated with Explo5 6.01).c Calculated detonation pressure (calculated with Explo5 6.01).d Calculated heat of formation.e Impact sensitivity.f Friction sensitivity.g Measured densities, gas pycnometer at room temperature.h Ref. 29. | |||||||
| 2 | 215 | 7706 | 21.7 | −193.5 | 40 | 360 | 1.82 |
| 3 | 253 | 6994 | 19.3 | −5.7 | >40 | >360 | 1.71 |
| 4 | 241 | 7664 | 25.2 | 13.7 | >40 | >360 | 1.80 |
| 5 | 241 | 7228 | 20.8 | −619.5 | >40 | >360 | 1.78 |
| 6 | 234 | 6812 | 19.2 | 20.8 | >40 | >360 | 1.66 |
| TATOT | 245 | 9385 | 29.7 | 446.7 | 40 | 360 | 1.72 |
| HNS | 318 | 7612 | 24.3 | 78.2 | 5 | 240 | 1.74 |
| PYX | 360 | 7757 | 25.1 | −11.6 | 10 | 240 | 1.76 |
| TNTh | 295 | 7303 | 21.3 | −59.3 | 15 | 353 | 1.65 |
Mechanical sensitivity measurements further show that all four compounds are consistently insensitive toward external stimuli, with impact sensitivities above 40 J and friction sensitivities above 360 N. These values are significantly lower than those of HNS, PYX, and TNT, indicating that low mechanical sensitivity is a common feature across this series. Taken together, these results suggest that ionic modulation through salt formation can effectively tune density and detonation output within this CF3-containing, nitrogen-rich framework while preserving very low mechanical sensitivity. Among the compounds examined here, perchlorate salt 4 provides the most balanced combination of detonation performance and insensitivity, whereas compound 3 retains the highest resistance to initial thermal decomposition.
Structure–property relationship
To rationalize the low mechanical sensitivities (IS > 40 J, FS > 360 N) and the distinct thermal-decomposition behaviors of 3, [3H]+ and 6, electrostatic potential (ESP)30,31 analyses were performed for 3 and [3H]+, and a Hirshfeld surface (HS)32 analysis was applied to [3H]+ based on its single-crystal structure. Since suitable single crystals of 3 were not obtained, HS descriptors could not be calculated for the neutral precursor. For the ionic salt 4, the HS analysis was carried out on the basis of its crystallographically characterized protonated cation-perchlorate assembly. Nevertheless, the combined HS and ESP results provide a consistent picture of how salt formation reshapes the intermolecular contact network and the surface charge landscape.For 4, the HS mapped with d_norm (Fig. 5) displays pronounced close-contact features around the protonated N–H donors and the oxygen-rich ClO4− acceptors, indicating strong and highly directional N–H⋯O interactions. Quantitatively, the 2D fingerprint plots reveal that hydrogen-bond-related contacts dominate lattice cohesion: N⋯H/H⋯N contacts account for 15.6% and O⋯H/H⋯O contacts for 13.8%, together contributing 29.4% of the total surface contacts. In addition, H⋯H contacts (22.7%) indicate efficient close packing, while H⋯F/F⋯H contacts (12.7%) and other heteroatom-involving contacts such as C⋯N/N⋯C (7.9%) and N⋯N (6.3%) further enrich the secondary interaction network. The presence of multiple, spatially distributed short contacts (including hydrogen bonds and ancillary heteroatom contacts) is expected to facilitate stress delocalization under mechanical stimuli, consistent with the experimentally observed insensitivity of 4. For 5, the Hirshfeld surface mapped over d_norm (Fig. 5) shows multiple intense red spots concentrated around the protonated N–H donors and the oxygen atoms of the NO3− anion, evidencing strong and highly directional N–H⋯O hydrogen-bonding interactions; additional short-contact features are also distributed near ring nitrogens, consistent with auxiliary N–H⋯N contacts. Quantitatively, the 2D fingerprint plots confirm that hydrogen-bond-related interactions dominate lattice cohesion: N⋯H/H⋯N contacts contribute 26.5% and O⋯H/H⋯O contacts contribute 13.2%, giving a combined 39.7% of the total surface contacts. Beyond these primary hydrogen bonds, H⋯F/F⋯H contacts (22.4%) are prominent, reflecting close approaches between the CF3 groups and surrounding hydrogen donors, while H⋯H contacts (13.4%) indicate efficient close packing. The remaining 24.5% (“Others”) corresponds to a range of weaker secondary contacts that further consolidate the solid-state network. Overall, the dense, spatially distributed short contacts in 5—dominated by N–H⋯O/N–H⋯N interactions and supplemented by H⋯F and packing contacts—are expected to promote stress delocalization under mechanical stimuli, consistent with the experimentally observed low sensitivity of 5.
Complementary ESP analyses further support this interpretation. For neutral 3, the surface potential spans from Vs, min = −49.31 to Vs, max = +45.03 kcal mol−1, a moderate range of ∼94.34 kcal mol−1 with a balanced distribution. Upon perchlorate salt formation, [3H]+ exhibits an expanded high-potential window (22.67 to 121.46 kcal mol−1 on the mapped surface), reflecting the ionic character introduced by protonation and ClO4− pairing. Importantly, although [3H]+ shows higher extreme surface potentials, its overall potential dispersion is similar to that of 3, suggesting a broadly distributed electrostatic surface rather than a highly localized one. Combined with the extensive N–H⋯O interactions evidenced by HS analysis, this feature provides a plausible microscopic basis for the preserved low sensitivity of [3H]+. Meanwhile, salt formation can introduce additional initiation pathways because oxygen-rich anions and hydrogen-bonded ion pairs are more susceptible to early-stage reactions than the neutral framework. Overall, ionization strengthens lattice interactions and improves energetic performance, while altering thermal-decomposition kinetics via anion-assisted activation (Fig. 6).
 | ||
| Fig. 6 (a) Electrostatic potential (ESP) maps of 3, the protonated cation [3H]+ derived from 4, and 6; (b) corresponding ESP distributions shown as bar graphs. | ||
Conclusions
In summary, a series of CF3-functionalized, triazine-linked nitrogen-rich energetic compounds (3–6) were constructed through an SNAr-based modular strategy from a chlorotriazine core and a fused 5/5 triazolo[4,3-b][1,2,4]triazole precursor. Relative to precursor 2 (Td = 215 °C), incorporation of the triazine unit increases the decomposition threshold of this framework, with compound 3 showing the highest onset decomposition temperature in the present series (Td = 253 °C). Subsequent ionic modulation further tunes density and energetic output: perchlorate salt 4 reaches a density of 1.80 g cm−3 and delivers D = 7664 m s−1 and P = 25.2 GPa, whereas nitrate salt 5 shows intermediate performance (D = 7228 m s−1, P = 20.8 GPa). By contrast, the triazine-bridged dimer 6 retains very low mechanical sensitivity but exhibits comparatively lower density, detonation output, and thermal stability within this series (Td = 234 °C). Notably, all four compounds remain highly insensitive toward impact and friction (IS > 40 J, FS > 360 N). Overall, these results indicate that triazine bridging combined with ionic modulation provides a useful strategy for tuning the balance among thermal behaviour, detonation performance, and low mechanical sensitivity in CF3-containing nitrogen-rich frameworks, with perchlorate salt 4 showing the most balanced overall combination of energetic performance and insensitivity.Author contributions
Jiyuan Yu: investigation, data curation, writing – original draft. Xiangyan Miao: methodology, writing – review & editing. Yuchuan Li: conceptualization, supervision, funding acquisition, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2525729 and 2530986 contain the supplementary crystallographic data for this paper.33a,bThe data underlying this study are available in the published article and its supporting information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6ra01290f.
Acknowledgements
This work was supported by the National Natural Science Foundation of China [No. 22135003].Notes and references
- X. Miao, J. Yu, Y. Li and S. Pang, Constructing nitrogen-rich fused [5,6,5]-tricyclic frameworks through rearrangement: heat-resistant zwitterionic salt energetic materials, Org. Lett., 2024, 26, 10085–10089 CrossRef CAS PubMed.
- J. Cai, C. Xie, J. Xiong, J. Zhang, P. Yin and S. Pang, High performance and heat-resistant pyrazole-1,2,4-triazole energetic materials: tuning the thermal stability by asymmetric framework and azo-bistriazole bridge, Chem. Eng. J., 2022, 433, 134480 CrossRef CAS.
- Q. Sun, Z. Jiang, N. Ding, C. Zhao, B. Tian, S. Li and S. Pang, Assembly of three oxadiazole isomers toward versatile energetics, J. Mater. Chem. A, 2023, 11, 23228–23232 RSC.
- H. Zhang, J. Cai, Z. Li, Q. Lai, P. Yin and S. Pang, Exploring a Fused Triazole–Tetrazine Binary CN Material for a Promising Initiating Substance, ACS Appl. Mater. Interfaces, 2024, 16, 4628–4636 CrossRef CAS PubMed.
- A. K. Sikder and N. Sikder, A review of advanced high performance, insensitive and thermally stable energetic materials emerging for military and space applications, J. Hazard. Mater., 2004, 112, 1–15 CrossRef CAS PubMed.
- R. P. Singh, R. D. Verma, D. T. Meshri and J. M. Shreeve, Energetic nitrogen-rich salts and ionic liquids, Angew, Chem., Int. Ed., 2006, 45, 3584–3601 CrossRef CAS PubMed.
- T. M. Klapötke, J. Stierstorfer, M. Weyrauther and T. G. Witkowski, Synthesis and investigation of 2,6-bis(picrylamino)-3,5-dinitropyridine (PYX) and its salts, Chem. Eur. J., 2016, 22, 8619–8626 CrossRef PubMed.
- X. Miao, J. Yu, S. Jiang, Y. Li and S. Pang, Reactivity investigation of bis-tetrazole isomers prepared through Dimroth rearrangement, Org. Lett., 2026, 28, 929–934 CrossRef CAS PubMed.
- H. Gao, Q. Zhang and J. M. Shreeve, Fused heterocycle-based energetic materials (2012–2019), J. Mater. Chem. A, 2020, 8, 4193–4216 RSC.
- S. Chen, W. Zhang, Y. Wang and Q. Zhang, [1,2,4]Triazolo[4,3-b]pyridazine as a building block towards low-sensitivity high-energy materials, Chem. Eng. J., 2021, 421, 129635 CrossRef CAS.
- P. Yin, C. He and J. M. Shreeve, Fused heterocycle-based energetic salts: alliance of pyrazole and 1,2,3-triazole, J. Mater. Chem. A, 2016, 4, 1514–1519 RSC.
- J. Zhang, Y. Feng, R. J. Staples, J. Zhang and J. M. Shreeve, Taming nitroformate through encapsulation with nitrogen-rich hydrogen-bonded organic frameworks, Nat. Commun., 2021, 12, 2146 CrossRef CAS PubMed.
- M. S. Gruhne, M. Lommel, M. H. H. Wurzenberger, T. M. Klapötke and J. Stierstorfer, Investigation of ethylenedinitramine as a versatile building block in energetic salts, cocrystals, and coordination compounds, Inorg. Chem., 2021, 60, 4816–4828 CrossRef CAS PubMed.
- Z. Yan, T. Lu, Y. Liu, W. Liu, B. Zhao, Y. Wang and Z. Ge, High thermal stability and insensitive fused triazole–triazine trifluoromethyl-containing explosives (TFX), ACS Omega, 2021, 6, 18591–18597 CrossRef CAS PubMed.
- Y. Kang, Y. Dong, Y. Liu, H. Gao, Y. Wang and J. M. Shreeve, Halogen bonding (C–F···X) and its effect on creating ideal insensitive energetic materials, Chem. Eng. J., 2022, 440, 135969 CrossRef CAS.
- S. Nehe, A. K. Yadav, V. D. Ghule and S. Dharavath, Employing the Trifluoromethyl Group on a 5/5 Fused Triazolo[4,3-b][1,2,4]triazole Backbone: A Viable Strategy for Attaining Balanced Energetics, Org. Lett., 2024, 26, 10611–10615 CrossRef PubMed.
- P. Xie, X. Liao and J. Liu, Ignition and combustion characteristics of aluminum-based fluorine-containing composite powder, Thermochim. Acta, 2024, 736, 179757 CrossRef CAS.
- Q. Sun, W. Chen, N. Ding, C. Zhao, Z. Jiang, S. Li and S. Pang, Unraveling the direct effect of hydrogen bonding on density and thermostability of energetic materials through isomerism, Chem. Eng. J., 2022, 444, 136539 CrossRef CAS.
- X. Jiang, D. Yin, S. Song, Y. Wang, M. Fan, R. Wang and Q. Zhang, Achieving ultra-high heat resistance of novel energetic materials through a hydrogen bonding and extended π-conjugation strategy, J. Mater. Chem. A, 2024, 12, 13231–13239 RSC.
- J. Ma, Y. Tang, G. Cheng, G. H. Imler, D. A. Parrish and J. M. Shreeve, Energetic derivatives of 8-nitropyrazolo[1,5-a][1,3,5]triazine-2,4,7-triamine: achieving balanced explosives by fusing pyrazole with triazine, Org. Lett., 2020, 22, 1321–1325 CrossRef CAS PubMed.
- X. Jiang, R. Wang, M. Fan, S. Song, Y. Wang and Q. Zhang, Combination of 5-amino-4-nitro-1,2-dihydro-3H-pyrazol-3-one and azine frameworks for insensitive, heat-resistant energetic materials, Dalton Trans., 2025, 54, 7056–7061 RSC.
- W. Ma, J. Li, C. He and S. Pang, Synthesis of two series of isomeric triazolo-triazine fused energetic salts: high thermal stability, insensitivity, and high performances, Cryst. Growth Des., 2023, 23, 3463–3470 CrossRef CAS.
- J. Ma, G. Cheng, X. Ju, Z. Yi, S. Zhu, Z. Zhang and H. Yang, Amino-nitramino functionalized triazolotriazines: a good balance between high energy and low sensitivity, Dalton Trans., 2018, 47, 14483–14490 RSC.
- S. Kronister, D. Svatunek, C. Denk and H. Mikula, Acylation-mediated ‘kinetic turn-on’ of 3-amino-1,2,4,5-tetrazines, Synlett, 2018, 29, 1297–1302 CrossRef CAS.
- W. N. Carson, J. W. Vanderwater and H. S. Gile, Coulometric determination of plutonium, Anal. Chem., 1957, 29, 1417–1422 CrossRef CAS.
- T. Ozawa, Kinetics in differential thermal analysis, Bull. Chem. Soc. Jpn., 1965, 38, 1881–1886 CrossRef CAS.
- M. Sućeska, Explo5 (Version 6.05), OZM Research s.r.o., Pardubice, 2018 Search PubMed.
- P. Richardson, A. A. Kitos, M. Triglav, J. S. Ovens, I. Laroche, S. Delisle, B. Jolicoeur, J. L. Brusso and M. Murugesu, Synthesis and detonation performance of novel tetrazolyl–triazine nitrogen-rich energetic materials, Mater. Adv., 2023, 4, 5775–5784 RSC.
- H. Gao, Q. Zhang and J. M. Shreeve, Fused heterocyclebased energetic materials (2012–2019), J. Mater. Chem. A, 2020, 8, 4193–4216 RSC.
- T. Lu and F. Chen, Multiwfn: a multifunctional wavefunction analyzer, J. Comput. Chem., 2012, 33, 580–592 CrossRef CAS PubMed.
- W. Humphrey, A. Dalke and K. Schulten, VMD: visual molecular dynamics, J. Mol. Graphics, 1996, 14, 33–38 CrossRef CAS PubMed.
- P. R. Spackman, M. J. Turner, J. J. McKinnon, S. K. Wolff, D. J. Grimwood, D. Jayatilaka and M. A. Spackman, CrystalExplorer: a program for Hirshfeld surface analysis, visualization and quantitative analysis of molecular crystals, J. Appl. Crystallogr., 2021, 54, 1006–1011 CrossRef CAS PubMed.
- (a) CCDC 2525729: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qs74x; (b) CCDC 2530986: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qypq3.
| This journal is © The Royal Society of Chemistry 2026 |