 Open Access Article
Open Access ArticleCharge-transfer interactions govern the trans influence and electron-induced bond activation in linear Au(I) complexes
Akef T. Afaneh *a,
Mansour H. Almatarneh
*a,
Mansour H. Almatarneh bc,
Ibrahim A. Daboubasha,
Ali Marashdeh
bc,
Ibrahim A. Daboubasha,
Ali Marashdeh *ad and
Oqab M. Abu Rummanb
*ad and
Oqab M. Abu Rummanb
aDepartment of Chemistry, Faculty of Science, Al-Balqa Applied University, 19117 Al-Salt, Jordan. E-mail: umafaneh@bau.edu.jo; a.marashdeh@bau.edu.jo
bDepartment of Chemistry, University of Jordan, Amman 11942, Jordan
cDepartment of Chemistry, College of Science, Imam Mohammad Ibn Saud Islamic University (IMSIU), Riyadh 11623, Saudi Arabia
dLeiden Institute of Chemistry, Gorlaeus Laboratories, Leiden University, Leiden, The Netherlands
First published on 17th March 2026
Abstract
Linear Au(I) complexes with the general formula of X–Au–L (X = Cl, Br, or I; L = OMe2, SMe2, SeMe2, or TeMe2) are model systems that exhibit trans influence and are directly relevant to focused electron-beam-induced deposition (FEBID), where low-energy electrons trigger precursor fragmentation. Using DFT/NBO together with ETS-NOCV/EDA and TD-DFT hole–electron analyses, we show that trends in Au–L bond strengthening and Au–X bond weakening are governed primarily by orbital-interaction/charge-transfer channels, including σ(Au–X)→σ*(Au–L) trans delocalization and ligand lone-pair donation into Au acceptor orbitals, which together modulate ΔEorb and the Au–L Wiberg bond order. Electron-attachment thermodynamics identify the most favorable dissociation channels and define an electron-induced lability metric Glab ≡ −ΔGfrag, min, where ΔGfrag, min is the most favorable (most negative) fragmentation free energy. The overlap metric Sr provides a trigger proxy for localized low-threshold excitations. To integrate these observables without double-counting, we construct a parsimonious precursor viability index (PVI3), combining Glab and interaction stabilization Sint = −ΔEint as benefits and Sr as a penalty; ΔC and WBI are retained as mechanistic correlates rather than as independent score components. The resulting ranking highlights ClAu(S/Se)Me2 as the most balanced candidate within this series, and this work provides a reproducible framework for FEBID-style screening.
Introduction
Predicting and rationalizing molecular stability, reactivity, and spectroscopic responses underpin advances from catalysis to nanofabrication. Frontier molecular orbitals (FMOs) offer a comprehensive understanding of these properties; however, in larger or highly polarizable systems, orbital delocalization can create difficulties in understanding which fragments control bond activation, charge transfer, or excited-state localization.1–5 Fragment-aware descriptors and energy-decomposition analyses therefore provide a quantitative, fragment-resolved complement to FMO interpretations, particularly when both donor–acceptor interactions and Pauli repulsion contribute appreciably to bonding and response.Gold is often described as a soft Lewis acid, and the linear, two-coordinate Au(I) motif is especially attractive due to its broad ligand tolerance and well-defined trans influence. In Au(I) halide adducts, X–Au–L (X = Cl, Br, or I; L = neutral donor), both experimental and computational results show that d(Au–X) increases, Au–X weakens and polarizability increases from Cl to I.6–21 These complexes are also central to focused electron-beam-induced deposition (FEBID), in which adsorbed precursors are dissociated by low-energy electrons to directly fabricate metal nanostructures.22–27 Gold FEBID remains compelling for plasmonic and electronic applications, but its progress hinges on the choice of the precursor: molecules must be stable enough for delivery while remaining sufficiently labile to fragment under the beam.6–9,23,28–30
In the present series of X–Au–L, electronegativity and polarizability covary for both X (Cl → Br → I) and L (O → S → Se → Te). Accordingly, we do not interpret any single HSAB “softness” metric as a standalone causal predictor. Instead, we interpret trends using decomposed computed observables, including CDA (charge-decomposition analysis) and NPA (natural population analysis) charge redistributions, as well as ETS–EDA (extended transition state energy-decomposition analysis) partitioning into ΔEelstat (electrostatic interaction), ΔEPauli (Pauli/exchange repulsion), and ΔEorb (the orbital-interaction/charge-transfer term), together with the dominant ETS-NOCV (ETS with natural orbitals for chemical valence) channels, thereby explicitly separating polarization, Pauli repulsion, and covalent/orbital interactions.
Here, we investigate how the anionic ligand X and neutral donor L modulate the ground- and excited-state electronic structure of linear X–Au–L complexes, and how these changes translate into FEBID-relevant trade-offs between precursor stability and electron-induced activation. We use DFT/NBO/EDA for ground states and TD-DFT for excited states to quantify fragment coupling, charge transfer (CT), and the balance among electrostatics, Pauli repulsion, and orbital interactions in the interaction energy (ΔEint). We introduce a compact frontier descriptor, ΔC ≡ C%(HOMO)L − C%(LUMO)L, and show its inverse relationship with the Au–L Wiberg bond order across the series (quantified in Results; see SI, TSI-7I). We also correlate the Au NBO (NPA) charge with the HOMO–LUMO gap, using Au charge as a simple electronic indicator of gap tuning, and we combine the hole–electron overlap metric Sr with the excitation energy Eex to identify low-threshold, localized excitations relevant to electron-induced bond activation.
To translate these observables into FEBID precursor selection, we introduce a normalization-based precursor viability index (PVI). Because several candidate inputs—WBI(Au–L) (taken here as the NBO Wiberg bond index of the Au–L bond), the frontier-balance descriptor ΔC ≡ CL(HOMO) − CL(LUMO) (where CL(ψ) is the fraction or percentage of an orbital's AO weight residing on ligand AOs), and the electron-induced lability metric Glab ≡ −ΔGfrag,min—are strongly collinear within the present 12-complex dataset, the main-text score is formulated in a parsimonious, collinearity-aware form (PVI3) based on Glab and Sint ≡ −ΔEint as benefits and Sr as a penalty, while WBI(Au–L) and ΔC are retained as mechanistic correlates (see the PVI section and SI for diagnostics; SI. 8–10). Across the trans axis, σ(Au–X) → σ*(Au–L) delocalization provides a trans-coupling channel that attenuates the Au–X bond and tunes the Au–L interaction; this is quantified using NBO/Wiberg indices, ETS-NOCV/EDA, and CDA.
Prior studies of Au(I) bonding and trans influence have established qualitative trends with donor strength and halide type. Here we add three compact, operational elements aimed at FEBID-style screening: (i) a mechanistic ΔC–WBI relationship, linking ligand-referenced frontier balance to WBI(Au–L) across X–Au–L (used as an interpretation tool rather than an independent screening axis); (ii) a charge-transfer/orbital-interaction map, which shows that interaction trends are dominated by the orbital term ΔEorb and its ETS-NOCV channels, rationalizing access to the σ(Au–X) → σ*(Au–L) trans-coupling pathway; and (iii) a collinearity-aware precursor-viability index used for ranking, reported as PVI3 in the main text and accompanied by a PC1-compressed sensitivity variant (PVIPC) in the SI (see SI, TSI-7A–TSI-7H).
Computational methods
All ground-state structures were optimized with Kohn–Sham DFT at the B3LYP level31–33 using aug-cc-pVTZ for main-group atoms and aug-cc-pVTZ-PP (effective-core potentials) for Au, Te, and I.34 Harmonic vibrational analyses at the same level confirmed each reported structure at its energy minimum (no imaginary modes). Unless otherwise stated, calculations were performed in the gas-phase; thermochemical corrections to Gibbs free energies employed the rigid-rotor harmonic-oscillator (RRHO) approximation at 298.15 K at the optimization level. All Gaussian runs used Gaussian 09.35Vertical singlet excitations were computed by linear-response TD-DFT/TD-SCF36,37 using CAM-B3LYP13 with the same basis/ECP partitioning (aug-cc-pVTZ used on main-group atoms; aug-cc-pVTZ-PP used on Au, Te, and I) in the gas phase. For each complex, the lowest 20 singlet states were evaluated to characterize the near-UV/visible spectral manifold. Spin–orbit effects were assessed qualitatively on a representative subset using SOC-TDDFT (RI-SOMF) in ORCA 6.0 (ref. 38) at the same functional/basis level.12,37,39 Scalar-relativistic effects for Au and I were treated using the effective-core potentials in aug-cc-pVTZ-PP (ref. 34); no additional DKH or ZORA corrections were applied in the Gaussian calculations.
Bonding and population analyses used NBO 3.1 (ref. 40) (as interfaced in Gaussian) to obtain Wiberg bond indices (WBI) and second-order donor → acceptor stabilization energies; atomic charges were obtained from the natural population analysis (NPA) section of the NBO output. Multiwfn 3.8 (ref. 41) was used for real-space hole–electron analysis to compute the overlap metric Sr and fragment population decompositions. For the frontier-balance descriptor, the ligand contribution to a molecular orbital ψ is defined as  , where cµ are MO coefficients in the AO basis, and the numerator sums AOs on the ligand fragment L. We define ΔC ≡ CL(HOMO) − CL(LUMO) and report it in Table 1 as the difference in ligand contribution between the HOMO and LUMO. In the present 12-complex series, ΔC was retained primarily for mechanistic interpretation (and shown alongside WBI and fragmentation thermodynamics), but it was not treated as an independent ingredient in the main-text PVI3 ranking due to descriptor collinearity (SI. 9). Bond order between the EDA fragments F1 = XAu and F2 = L was reported as an interfragment Wiberg bond index, computed by summing NBO Wiberg indices over all atom pairs across the two fragments:
, where cµ are MO coefficients in the AO basis, and the numerator sums AOs on the ligand fragment L. We define ΔC ≡ CL(HOMO) − CL(LUMO) and report it in Table 1 as the difference in ligand contribution between the HOMO and LUMO. In the present 12-complex series, ΔC was retained primarily for mechanistic interpretation (and shown alongside WBI and fragmentation thermodynamics), but it was not treated as an independent ingredient in the main-text PVI3 ranking due to descriptor collinearity (SI. 9). Bond order between the EDA fragments F1 = XAu and F2 = L was reported as an interfragment Wiberg bond index, computed by summing NBO Wiberg indices over all atom pairs across the two fragments: 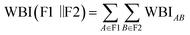 . For the present linear monodentate X–Au–L series, this interfragment index was numerically dominated by (and effectively equivalent to) Au–Y WBI (Y = donor atom); hence, we also refer to it as WBI(Au–L) for brevity.
. For the present linear monodentate X–Au–L series, this interfragment index was numerically dominated by (and effectively equivalent to) Au–Y WBI (Y = donor atom); hence, we also refer to it as WBI(Au–L) for brevity.
| FO contribution (C%) | ΔC | WBI(F1‖F2) | ||
|---|---|---|---|---|
| HOMO | LUMO | |||
| a For each ligand L, values are reported as ClAuL (BrAuL) [IAuL]. | ||||
| OMe2 | 96.3 (96.5) [96.8] | 3.7 (3.5) [3.2] | 92.60 (93.00) [93.60] | 0.328 (0.294) [0.209] |
| SMe2 | 81.3 (84.7) [84.9] | 18.8 (15.4) [17.1] | 62.50 (69.30) [67.80] | 0.459 (0.422) [0.336] |
| SeMe2 | 78.2 (78.8) [79.9] | 21.9 (21.1) [20.2] | 56.30 (57.70) [59.70] | 0.468 (0.426) [0.366] |
| TeMe2 | 73.6 (74.3) [75.7] | 26.3 (25.8) [24.1] | 47.30 (48.50) [51.60] | 0.590 (0.553) [0.491] |
Energy-decomposition analyses followed the Morokuma–Ziegler partitioning (ΔEelstat, ΔEPauli, ΔEorb, ΔEdisp) and were carried out as ETS-NOCV in ORCA 6.0 (ref. 38) using neutral singlet fragments F1 = XAu (S = 0) and F2 = L (S = 0) unless stated otherwise. Charge-decomposition/ETS-NOCV charge-flow analyses were evaluated in both directions (X → Au–L and L → Au–X).42–44 For comparison with conceptual-DFT reactivity indices, we evaluated the ligand-condensed dual descriptor Δf = f+ − f−, where f+ and f− are the condensed Fukui functions for nucleophilic and electrophilic attack, respectively, summed over the ligand atoms, following Morell, Grand, and Toro-Labbé.45 To compare Au–L and Au–X bond lengths across donors/halides of different sizes, we calculated size-normalized distances  (Y = L or X) using covalent radii from Cordero et al.46 to define the bond-distance cutoff (i.e., atoms were considered bonded when dAB ≤ rcov(A) + rcov(B) [±tolerance, if you used one]); the radii and resulting dnorm values are tabulated in the SI (TSI-8).
(Y = L or X) using covalent radii from Cordero et al.46 to define the bond-distance cutoff (i.e., atoms were considered bonded when dAB ≤ rcov(A) + rcov(B) [±tolerance, if you used one]); the radii and resulting dnorm values are tabulated in the SI (TSI-8).
Neutral bond-dissociation Gibbs free energies, ΔBDG, were evaluated as adiabatic RRHO Gibbs free-energy changes at 298.15 K for X–Au–L → X + AuL (and related neutral channels), with all species optimized at the B3LYP/aug-cc-pVTZ(-PP) level (with aug-cc-pVTZ-PP ECPs for Au and I); electron-induced fragmentation free energies, ΔGfrag, were analogously defined for X–Au–L + e− → [X–Au–L]−, followed by dissociation to the indicated fragments. Unless otherwise noted, ΔGfrag values reported in Table 3 correspond to the adiabatic thermochemical cycle (optimized [X–Au–L]− and optimized fragments prior to RRHO free-energy evaluation) and do not include a separate vertical (fixed-geometry) attachment step. Charge/multiplicity conventions, definitions of vertical versus adiabatic steps, and the treatment of spin–orbit coupling (SOC) for Au/I (where applicable) are documented in the SI (SI. 4; Tables TSI-14 and TSI-15), together with the Cartesian coordinates and representative input files (SI. 6). Transition-state (TS) searches were attempted for relevant substitution/dissociation steps using the QST2/QST3 and TS optimizations seeded from relaxed coordinate scans at the same level of theory used for the corresponding energy minima; however, no converged first-order saddle point (single imaginary frequency) was obtained for any complex, with candidate optimizations relaxing to nearby energy minima or failing to converge on a shallow potential-energy surface. Accordingly, we did not report TS-derived activation parameters; the mechanistic discussion was restricted to scan-guided qualitative trends and the relative stability of the identified energy minima, and |ΔGfrag|min was used as a thermodynamic proxy for scission propensity in the PVI analysis (SI. 4).
All metrics were mapped onto a common [0,1] scale using min–max normalization over the full set of complexes:  ;
;  . For penalty metrics (Sr), we used the transformed contribution (1 − z(j)m) so that the larger values always indicated a more promising precursor. The resulting three-term index was
. For penalty metrics (Sr), we used the transformed contribution (1 − z(j)m) so that the larger values always indicated a more promising precursor. The resulting three-term index was  , which lies between 0 and 1 because each z-score is min–max normalized to [0,1] and the expression is the average of three terms in [0,1].
, which lies between 0 and 1 because each z-score is min–max normalized to [0,1] and the expression is the average of three terms in [0,1].
For transparency, we also calculated the original five-term index (PVI5), which combined the delivery- and lability-relevant descriptors using fixed weights. Benefit terms were entered as z(j)m, whereas penalty terms were entered as (1−z(j)m). Using WBI(Au–L), Glab, and Sint as benefits and ΔC and Sr as penalties, we defined PVI5 = 0.25 zBOj + 0.20 (1 − zΔCj) + 0.20 z|ΔG|j + 0.20 (1 − zSrj) + 0.15 zSintj. As ΔC, WBI(Au–L), and Glab were strongly collinear within this series, PVI5 was provided mainly for transparency and sensitivity checks (SI), together with a PC1-compressed variant (PVIPC); these checks do not change the qualitative conclusions for the present dataset.
Results and discussion
Across the donor series L = OMe2 → SMe2 → SeMe2 → TeMe2, the Au–L interaction strengthens electronically even though the direct Au–L distance increases with the donor size. Because direct distances conflate size with bonding, we consider the size-normalized contact metric dnorm(Au–Y) = d(Au–Y)/[Rcov(Au) + Rcov(Y)] (covalent radii from Cordero et al.; SI: TSI-8)46 only as a geometric comparator and do not use it to infer covalency. Instead, electronic strengthening is assessed using Wiberg bond indices (WBI from NBO) and ETS-EDA/ETS-NOCV descriptors, in particular the orbital-interaction/charge-transfer term ΔEorb and dominant NOCV channels (SI: TSI-16A, B).42–44 Using these observables, both WBI(Au–L) and ΔEorb increase from OMe2 to heavier chalcogen donors, indicating enhanced electronic coupling, while ΔEint reflects the balance among electrostatics, Pauli repulsion, and orbital contributions. In parallel, σ(Au–X) → σ*(Au–L) delocalization provides a trans-coupling pathway that decreases Au–X bonding and adjusts Au–L bonding, consistent with the classical trans influence in linear d10 Au(I) complexes.21,39,47 Detailed structural benchmarks and full geometric trends are provided in the SI (SI. 1).Charge-decomposition analysis (CDA) and ETS-EDA map fragment coupling by relating CT(L → XAu) to the interaction energy ΔEint(XAu‖L)42–44
Across OMe2 → SMe2 → SeMe2 → TeMe2 at fixed X, CT increases, and the interaction becomes more stabilizing; across Cl → Br → I at fixed L, stabilization is generally attenuated. Within this dataset, the orbital-interaction term (ΔEorb), which captures donor–acceptor mixing/CT, tracks the overall interaction energy most closely (Pearson r(ΔEint, ΔEorb) = 0.980, R2 = 0.960, N = 12; SI Table TSI-16A; see also TSI-7I), while ΔEelstat and ΔEPauli,total are strongly anti-correlated (r = −0.984, R2 = 0.969, N = 12; SI Table TSI-16A; see also SI Table TSI-7I) and therefore partially cancel one another, so their combined contribution varies more weakly across the series than either term individually. Accordingly, we treat CT/ΔEorb as a decisive contribution when rationalizing trends and avoid over-interpreting individual electrostatic or Pauli components in isolation.13,15,16,41,48 The observed increase in WBI(Au–L) cannot be attributed to increased occupation of σ*(Au–L), because donation into σ*(Au–L)-for example via a σ(Au–X) → σ*(Au–L) charge-transfer pathway-would weaken the Au–L bond rather than strengthen it. The net increase arises from enhanced n(Ch) → Au covalency (more negative ΔEorb), which dominates over the trans-weakening channel (Fig. 1).A compact, EDA-independent descriptor, ΔC, is defined from ligand frontier-orbital contributions as ΔC ≡ CL(HOMO) − CL(LUMO), where CL(ψ) is the fraction (or percentage) of the AO-weight of orbital ψ residing on ligand AOs (see the Computational methods section); CL(HOMO) and CL(LUMO) are reported in Table 1. Across the X–Au–L series, a larger ΔC coincides with a smaller WBI(F1∥F2), whereas a smaller ΔC accompanies larger WBI: OMe2 exhibits the largest ΔC and smallest WBI, while TeMe2 shows the smallest ΔC and largest WBI (Pearson r = −0.881, R2 = 0.776, N = 12; see SI, TSI-7I). Thus, smaller ΔC signals more balanced ligand participation at the frontier level and tighter electronic coupling, consistent with the strengthening observed along O → S → Se → Te. Importantly, within this chemically narrow 12-complex set, ΔC is strongly collinear with other lability proxies (e.g., Glab and WBI); therefore, ΔC is used here primarily as a mechanistic frontier-localization indicator rather than as an independent component in the main-text PVI3 ranking (see SI).
Time-dependent DFT provides orthogonal cues relevant to FEBID
Within a given chemical subset, lower excitation energies often coincide with larger hole–electron overlap (Sr), reflecting more localized X–Au–L excitations (Pearson r = −0.646, R2 = 0.417, N = 12; see SI, TSI-7I).13,36,41 Because Eex and Sr are spectroscopic descriptors rather than thermodynamic metrics, we do not equate them directly with stability. OMe2 complexes deliberately deviate by displaying more metal- or halide-centered excitations, explaining why Au–X versus Au–L scission propensity can switch across X. As a trigger heuristic, larger Sr at lower Eex suggests lower activation thresholds and a greater likelihood of populating dissociative channels under low-energy electron impact (Table 2).| Complexes | Eex | Sr | Complexes | Eex | Sr |
|---|---|---|---|---|---|
| ClAuOMe2 | 6.547 | 0.512 | ClAuSeMe2 | 6.627 | 0.502 |
| BrAuOMe2 | 6.470 | 0.637 | BrAuSeMe2 | 6.428 | 0.507 |
| IAuOMe2 | 6.265 | 0.504 | IAuSeMe2 | 6.238 | 0.539 |
| ClAuSMe2 | 7.178 | 0.468 | ClAuTeMe2 | 6.466 | 0.613 |
| BrAuSMe2 | 6.831 | 0.475 | BrAuTeMe2 | 6.275 | 0.614 |
| IAuSMe2 | 6.425 | 0.567 | IAuTeMe2 | 5.911 | 0.631 |
To quantify how the lowest excitation redistributes density between the Au–X and Au–L sides of the complex, we analyzed the corresponding NTO hole/electron densities using two complementary fragment partitions: (XAu)|(L) and (X)|(AuL) (SI, TSI-3). In the (X)|(AuL) partition, the particle NTO density on X is generally minor-to-moderate (typically <20%, maximum 25.5%), indicating that the lowest excitation is not dominated by the σ*(Au–X) character but by a measurable σ*(Au–X)-like (X-involving) acceptor admixture. Notably, BrAuOMe2 shows the strongest halide participation (25.5% on Br), consistent with our observation that OMe2 can display more metal-/halide-involved low-energy excitations along with the reported switching of Au–X versus Au–L scission propensity across X. This excited-state partitioning is independent of the ground-state bond-length trends discussed above, which arise from the trans-axis CT/orbital-interaction effects in S0.
A gap–charge coupling (Fig. 2) emerges when the HOMO–LUMO gap, Egap, is plotted against the Au(I) NPA charge qAu. For a fixed ligand L, the halide series Cl → Br → I shows a monotonic decrease in both qAu and Egap, consistent with the increased Au–X covalency and charge redistribution. Across the donor series, at fixed X, OMe2 lies at the high-qAu/high-Egap end, while S/Se/Te progressively reduce qAu and compress Egap as donor polarizability increases (and electronegativity decreases) and Au–L covalency increases. Thus, OMe2 is not anomalous in trend direction; it is offset relative to the chalcogen donors, and the smaller Egap is associated with a less cationic Au center and a higher reactivity potential.
Fragmentation windows for neutral channels (ΔBDG, 298 K) indicate that at a fixed halide, heavier chalcogens generally yield less favorable neutral dissociation, while at a fixed ligand, moving from Cl to I tends to make electron-induced fragmentation more exergonic. For electron-impact conditions relevant to FEBID, we report electron-induced fragmentation free energies, ΔGfrag (298 K), for X–Au–L + e− → [X–Au–L]− → fragments (Table 3). Consistent with prior dissociative electron-attachment studies that have reported preferential Au–Cl scission upon electron attachment,15 the ClAuL members of our set display strongly exergonic Au–Cl cleavage channels in the anionic fragmentation thermodynamics (Table 3; SI TSI-9). Because first-order saddle points could not be located for the corresponding substitution/dissociation pathways (SI. 4; TSI-15), we do not report TS-derived activation parameters; instead, the magnitude of the most exergonic channel, Glab, is used as the thermodynamic proxy for scission propensity.
| Reaction scheme: X–Au–L + e− → [X–Au–L]− → fragments (channels listed below)a | ||||
|---|---|---|---|---|
| Fragmentation channel (products) | OM2 | SMe2 | SeMe2 | TeMe2 |
| a All-channels conserve total charge (−1) after electron attachment; e.g., “ClAu− + L” denotes dissociation into an anionic Au–X fragment and neutral ligand, while “X− + AuL” denotes the halide loss with a neutral AuL fragment. Channel definitions follow SI. 4 (Table TSI-9). | ||||
| ClAu− + L | −1.678 | −0.970 | −0.896 | −0.767 |
| BrAu− + L | −1.751 | −1.073 | −0.995 | −0.862 |
| IAu− + L | −1.831 | −1.200 | −1.118 | −0.981 |
| Cl− + AuL | −0.459 | −0.096 | −0.122 | −0.159 |
| Br− + AuL | −0.581 | −0.248 | −0.270 | −0.302 |
| I− + AuL | −0.675 | −0.389 | −0.408 | −0.436 |
| Cl− + Au + L | −0.658 | 0.050 | 0.124 | 0.252 |
| Br− + Au + L | −0.780 | −0.102 | −0.024 | 0.109 |
| I− + Au + L | −0.874 | −0.243 | −0.162 | −0.024 |
To summarize the screening outcomes, we report a precursor viability index, PVI, constructed from min–max normalized descriptors. In the main text, we use the parsimonious, collinearity-aware PVI3 (Glab and Sint as benefits, Sr as a penalty), while WBI(Au–L) and ΔC are reported alongside the score as mechanistic correlates (Table 4). Full component tables and sensitivity checks (including the original five-term PVI5) are provided in the SI.
| Rank (PVI3) | Complexes | X | L | ΔC | WBI(Au–L) = WBI(F1‖F2) | Glab (eV) | Sr | Sint (eV) | PVI3 |
|---|---|---|---|---|---|---|---|---|---|
| 1 | ClAuSMe2 | Cl | SMe2 | 62.56 | 0.459 | 1.349 | 0.468 | 7.362 | 0.829 |
| 2 | ClAuSeMe2 | Cl | SeMe2 | 56.28 | 0.468 | 1.424 | 0.502 | 7.195 | 0.747 |
| 3 | BrAuSMe2 | Br | SMe2 | 69.28 | 0.422 | 1.228 | 0.475 | 7.023 | 0.705 |
| 4 | ClAuOMe2 | Cl | OMe2 | 92.57 | 0.328 | 0.643 | 0.512 | 7.872 | 0.646 |
| 5 | BrAuSeMe2 | Br | SeMe2 | 57.69 | 0.426 | 1.306 | 0.507 | 6.861 | 0.630 |
| 6 | ClAuTeMe2 | Cl | TeMe2 | 47.33 | 0.590 | 1.553 | 0.613 | 6.924 | 0.507 |
| 7 | IAuOMe2 | I | OMe2 | 93.67 | 0.209 | 0.419 | 0.504 | 7.366 | 0.485 |
| 8 | IAuSeMe2 | I | SeMe2 | 59.76 | 0.366 | 1.131 | 0.539 | 6.609 | 0.460 |
| 9 | IAuSMe2 | I | SMe2 | 65.76 | 0.336 | 1.050 | 0.567 | 6.768 | 0.416 |
| 10 | BrAuTeMe2 | Br | TeMe2 | 48.51 | 0.553 | 1.439 | 0.614 | 6.595 | 0.400 |
| 11 | BrAuOMe2 | Br | OMe2 | 93.00 | 0.294 | 0.549 | 0.637 | 7.563 | 0.304 |
| 12 | IAuTeMe2 | I | TeMe2 | 51.62 | 0.491 | 1.269 | 0.631 | 6.344 | 0.262 |
Within each halide or ligand subgroup, the descriptor values are internally consistent and highlight the same qualitative trade-offs (increasing WBI(Au–L) along O → S → Se → Te, while the net interaction stabilization Sint can be moderated by countervailing the terms in EDA).
These outcomes translate into three practical implications for FEBID precursor selection. First, coupling balance is captured by the joint behavior of ΔC and WBI: WBI(F1‖F2) is larger and XAu‖L coupling is stronger when the ligand contributes more evenly to HOMO and LUMO (smaller ΔC); thus, a full EDA decomposition is not required to obtain a first-pass trans influence diagnostic.49–51 Second, the Egap–qAu relationship (HOMO–LUMO gap versus Au NPA charge) is used only as a qualitative frontier/hardness proxy and is interpreted alongside fragment-resolved observables (CDA/NBO/ETS-NOCV) that explicitly separate polarization and covalency. Finally, electron triggering is assessed using the thermodynamic lability metric Glab together with Sr and Eex: larger Sr at lower Eex indicates localized, low-threshold excitations that are more likely to yield bond-selective scission upon electron impact, while Glab screens out overly exergonic fragmentation that could compromise delivery stability. In the collinearity-aware PVI3 ranking (Table 4), Glab and Sint provide the primary balance between lability and robustness, with Sr acting as a conservative trigger penalty.
Conclusion
The bonding- and electron-induced reactivities in linear Au(I) complexes, X–Au–L, are significantly affected by the identities of the ligands and halides. The electron density at Au increases as the donor transitions from OMe2 to TeMe2. Delocalization along the trans axis (e.g., σ(Au–X) → σ*(Au–L)) contributes to Au–X weakening and represents a trans influence (antibonding) channel that would, in isolation, oppose Au–L strengthening. The observed net strengthening of Au–L along the donor series is instead reflected by increased WBI/WBI (Au–L) and more stabilizing ΔEorb from ETS-EDA/ETS-NOCV. The CT–ΔEint quadrant map facilitates the categorization of fragment-pairing regimes, while energy-decomposition analysis elucidates the contributions of orbital interactions, electrostatics, and Pauli repulsion in defining ΔEint. Ligand involvement in HOMO and LUMO is linked to bond order and coupling strength, ΔC, which serves as a small frontier descriptor that characterizes trans influence patterns. In FEBID, excited-state descriptors provide distinct insights: an increased Sr at reduced Eex suggests a more direct pathway to dissociative channels, influenced by the thermodynamic window indicated by Glab. We introduce a simple index that ranks precursors using only the reported tabulated descriptors, after scaling each descriptor by min–max normalization to enable consistent combination. This index outlines the process for actualizing these concepts and highlights the primary design requirement of FEBID: molecules must possess sufficient fragility for disintegration while maintaining adequate stability for delivery. Furthermore, it provides a systematic list of options and experimental variables that can influence the deposition outcomes, including dose, duration, temperature, and co-reactants.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this study are available within the article and its supporting information (SI). Optimized Cartesian coordinates, computed descriptors (NBO charges, Wiberg bond indices, ΔC, and HOMO–LUMO gaps), and ETS-EDA/ETS-NOCV energy-decomposition data (including dominant NOCV channel populations and energies) are provided in the SI Tables (e.g., TSI-16A and TSI-16B) and associated SI sections. Additional raw input/output files (Gaussian/ORCA job files) are available from the corresponding author upon reasonable request. Supplementary information is available. See DOI: https://doi.org/10.1039/d6ra01199c.References
- R. B. Woodward, and R. Hoffmann, The Conservation of Orbital Symmetry, Elsevier, 2013 Search PubMed.
- I. Fleming, Molecular Orbitals and Organic Chemical Reactions, John Wiley & Sons, 2011 Search PubMed.
- R. G. Parr and W. Yang, Density functional approach to the frontier-electron theory of chemical reactivity, J. Am. Chem. Soc., 1984, 106, 4049–4050 CrossRef CAS.
- I. Fleming, Frontier Orbitals and Organic Chemical Reactions, Wiley, 1977 Search PubMed.
- K. Fukui, T. Yonezawa and H. Shingu, A molecular orbital theory of reactivity in aromatic hydrocarbons, J. Chem. Phys., 1952, 20, 722–725 CrossRef CAS.
- G. Pakeltis, Z. Hu, A. G. Nixon, E. Mutunga, C. P. Anyanwu, C. A. West and P. D. Rack, Focused electron beam induced deposition synthesis of 3d photonic and magnetic nanoresonators, ACS Appl. Nano Mater., 2019, 12, 8075–8082 CrossRef.
- W. G. Carden, R. M. Thorman, I. Unlu, K. A. Abboud, D. H. Fairbrother and L. McElwee-White, Design, Synthesis, and Evaluation of CF3AuCNR Precursors for Focused Electron Beam-Induced Deposition of Gold, ACS Appl. Mater. Interfaces, 2019, 11, 11976–11987 CrossRef CAS PubMed.
- A. T. M. Addaraidi, An Investigation of Structural Motifs in Gold Complexes, The University of Manchester, United Kingdom, 2018 Search PubMed.
- I. Utke, P. Hoffmann, B. Dwir, K. Leifer, E. Kapon and P. Doppelt, Focused electron beam induced deposition of gold, J. Vac. Sci. Technol., B: Microelectron. Nanometer Struct.–Process., Meas., Phenom., 2000, 18, 3168 CrossRef CAS.
- P. Nava, D. Hagebaum-Reignier and S. Humbel, Bonding of gold with unsaturated species, ChemPhysChem, 2012, 13, 2090–2096 CrossRef CAS PubMed.
- P. Pyykkö and P. Zaleski-Ejgierd, Basis-set limit of the aurophilic attraction using the MP2 method: The examples of [ClAuPH3]2 dimer and [P(AuPH3)4]+ ion, J. Chem. Phys., 2008, 128, 124309 CrossRef.
- S. G. Wang and W. E. Schwarz, Quasi-relativistic density functional study of aurophilic interactions, J. Am. Chem. Soc., 2004, 126, 1266–1276 CrossRef CAS PubMed.
- T. Yanai, D. P. Tew and N. Handy, A new hybrid exchange-correlation functional using the Coulomb-attenuating method (CAM-B3LYP), Chem. Phys. Lett., 2004, 393, 51–57 CrossRef CAS.
- F. Rabilloud, Structure and Stability of Coinage Metal Fluoride and Chloride Clusters (MnFn and MnCln, M 5Cu, Ag, Or Au; n 51–6), J. Comput. Chem., 2012, 33, 2083–2091 CrossRef CAS.
- J. Kopyra, F. Rabilloud, P. Wierzbicka and H. Abdoul-Carime, Energy-Selective Decomposition of Organometallic Compounds by Slow Electrons: The Case of Chloro(dimethyl sulfide)gold(I), J. Phys. Chem. A, 2021, 125, 966–972 CrossRef CAS.
- L. Antes, S. Dapprich, G. Frenking and P. Schwerdtfeger, The Stability of Group 11 ClMCO complexes (M=Cu, Ag, Au), Inorg. Chem., 1996, 35, 2089–2096 CrossRef.
- D. Sorbelli, L. Belpassi, F. Tarantelli and P. Belanzoni, Ligand Effect on Bonding in Gold(III) Carbonyl Complexes, Inorg. Chem., 2018, 57, 6161–6175 CrossRef CAS.
- D. Sorbelli, L. Belpassi and P. Belanzoni, Mechanistic Study of Alkyne Insertion into Cu–Al and Au–Al Bonds: A Paradigm Shift for Coinage Metal Chemistry, Inorg. Chem., 2022, 61, 21095–21106 CrossRef CAS PubMed.
- L. M. Reynard, C. J. Evans and M. C. L. Gerry, The Pure Rotational Spectrum of AuI, J. Mol. Spectrosc., 2001, 205, 344–346, DOI:10.1006/jmsp.2000.8274.
- C. J. Evans and M. C. L. Gerry, The Pure Rotational Spectra of AuCl and AuBr, J. Mol. Spectrosc., 2000, 203, 105–117, DOI:10.1006/jmsp.2000.8150.
- X. Li, Metalophilic Interaction in Gold Halide: Quantum Chemical Study of AuX (X = F-At), J. Comput. Chem., 2014, 35, 923–931 CrossRef CAS PubMed.
- M. Huth, F. Porrati, C. Schwalb, M. Winhold, R. Sachser, M. Dukic, J. Adams and G. Fantner, Focused electron beam induced deposition: A perspective, Beilstein J. Nanotechnol., 2012, 3, 597–619, DOI:10.3762/bjnano.3.70.
- W. F. van Dorp and C. W. Hagen, A critical literature review of focused electron beam induced deposition, J. Appl. Phys., 2008, 104, 081301, DOI:10.1063/1.2977587.
- I. Utke, P. Hoffmann and J. Melngailis, Gas-assisted focused electron beam and ion beam processing and fabrication, J. Vac. Sci. Technol., B: Microelectron. Nanometer Struct.–Process., Meas., Phenom., 2008, 26, 1197–1276, DOI:10.1116/1.2955728.
- N. Silvis-Cividjian and C. W. Hagen, Electron-Beam–Induced Nanometer-Scale Deposition, Adv. Imaging Electron Phys., 2006, 143, 1–235, DOI:10.1016/s1076-5670(06)43001-9.
- K. Höflich, R. B. Yang, A. Berger, G. Leuchs and S. Christiansen, The Direct Writing of Plasmonic Gold Nanostructures by Electron-Beam-Induced Deposition, Adv. Mater., 2011, 23, 2657–2661, DOI:10.1002/adma.201004114.
- S. Graells, S. Aćimović, G. Volpe and R. Quidant, Direct Growth of Optical Antennas Using E-Beam-Induced Gold Deposition, Plasmonics, 2010, 5, 135–139, DOI:10.1007/s11468-010-9128-9.
- M. Mostafa, P. T. Shawrav, H. D. Wanzenboeck, M. Schinnerl, M. Stöger-Pollach, S. Schwarz and A. Steiger-Thirsfeld, Emmerich Bertagnolli Highly conductive and pure gold nanostructures grown by electron beam induced deposition, Sci. Rep., 2016, 6, 34003 CrossRef.
- I. Utke, P. Swiderek, K. Höflich, K. Madajska, J. Jurczyk, P. Martinović and I. B. Szymańska, Coordination and organometallic precursors of group 10 and 11: Focused electron beam induced deposition of metals and insight gained from chemical vapour deposition, atomic layer deposition, and fundamental surface and gas phase studies, Coord. Chem. Rev., 2022, 458, 213851–213915 CrossRef CAS.
- W. F. v. Dorp, X. Wu, J. J. L. Mulders, S. Harder, P. Rudolf and J. T. M. D. Hosson, Gold Complexes for Focused-Electron-Beam-Induced Deposition, Langmuir, 2014, 30, 12097–12105 CrossRef PubMed.
- W. Kohn, A. D. Becke and R. G. Parr, Density Functional Theory of Electronic Structure, J. Phys. Chem., 1996, 100, 12974–12980, DOI:10.1021/jp960669l.
- A. D. Becke, Modern Electronic Structure Theory, Part II Yarkony, ed. D. R. Yarkony, World Scientific, Singapore, 1995, p. 1022 Search PubMed.
- T. Ziegler, Approximate density functional theory as a practical tool in molecular energetics and dynamics, Chem. Rev., 1991, 91, 651–667, DOI:10.1021/cr00005a001.
- B. P. Pritchard, D. Altarawy, B. Didier, T. D. Gibson and T. L. A. Windus, New Basis Set Exchange: An Open, Up-to-date Resource for the Molecular Sciences Community, J. Chem. Inf. Model., 2019, 59, 4814–4820 CrossRef CAS PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, and et al., Gaussian 09, Revision B.01, Gaussian, Inc., 2010 Search PubMed.
- M. E. Casida, C. Jamorski, K. C. Casida and D. R. Salahub, Molecular excitation energies to high-lying bound states from time-dependent density-functional response theory: Characterization and correction of the time-dependent local density approximation ionization threshold, J. Chem. Phys., 1998, 108, 4439–4449 CrossRef CAS.
- C. Adamo and D. Jacquemin, The calculations of excited-state properties with Time-Dependent Density Functional Theory, Chem. Soc. Rev., 2013, 42, 845 RSC.
- F. Neese, Software Update: The ORCA Program System—Version 6.0, Wiley Interdiscip. Rev.:Comput. Mol. Sci., 2025, 15(2), e70019 Search PubMed.
- P. Schwerdtfeger, Relativistic Effects in Properties of Gold, Angew. Chem., Int. Ed., 2003, 42, 892–1895 CrossRef PubMed.
- F. Weinhold, and J. E. Carpenter in The Structure of Small Molecules and Ions, 1988, pp. 227–236 Search PubMed.
- T. Lu and F. C. Multiwfn, A multifunctional wavefunction analyzer, J. Comput. Chem., 2012, 33, 580–592 CrossRef CAS.
- K. Kitaura and K. Morokuma, A new energy decomposition scheme for molecular interactions within the Hartree-Fock approximation, Int. J. Quantum Chem., 1976, 10, 325–340, DOI:10.1002/qua.560100211.
- T. Ziegler and A. Rauk, On the calculation of bonding energies by the Hartree Fock Slater method I. The transition state method, Theor. Chim. Acta, 1977, 46, 1–10, DOI:10.1007/BF02401406.
- S. Dapprich and G. Frenking, Investigation of Donor-Acceptor Interactions: A Charge Decomposition Analysis Using Fragment Molecular Orbitals, J. Phys. Chem., 1995, 99, 9352–9362 Search PubMed.
- C. Morell, A. Grand and A. Toro-Labbe, New Dual Descriptor for Chemical Reactivity, J. Phys. Chem. A, 2005, 109, 205–212 CrossRef CAS PubMed.
- B. Cordero, V. Gomez, A. E. Pletro-Prats, M. Reves, J. Echeverrıa, E. Cremades, F. Barragan and S. Alvarez, Covalent radii revisited, Dalton Trans., 2008, 2832–2838 Search PubMed.
- S. Komeda, R. Takahashi and Y. Yamamoto, Trans Influence in Gold(I) Complexes, Inorg. Chem., 2023, 62, 14032–14040 Search PubMed.
- F. Rabilloud, Structure and Bonding in Coinage Metal Halide Clusters MnXn, M = Cu, Ag, Au; X = Br, I; n = 1−6, J. Phys. Chem. A, 2012, 116, 3474–3480 Search PubMed.
- R. G. Parr and R. G. Pearson, Absolute Hardness: Companion Parameter to Absolute Electronegativity, J. Am. Chem. Soc., 1983, 105, 7512–7516 Search PubMed.
- R. G. Pearson, The Principle of Maximum Hardness, Acc. Chem. Res., 1993, 26, 250–255 CrossRef CAS.
- R. G. Pearson, Absolute Electronegativity and Hardness: Application to Inorganic Chemistry, Inorg. Chem., 1988, 27, 734–740 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |


