 Open Access Article
Open Access ArticleRecyclable chitosan–biochar biocomposite films for sustainable removal of congo red dye pollutants from water
René Blaise Ngouateu Lekene *ab,
Mária Kováčová
*ab,
Mária Kováčová b,
Christian Brice Dantio Nguela
b,
Christian Brice Dantio Nguela a,
Matej Mičušík
a,
Matej Mičušík b,
Alena Opálková Šišková
b,
Alena Opálková Šišková b,
Anna Vykydalová
b,
Anna Vykydalová b,
Hamed Peidayesh
b,
Hamed Peidayesh b,
Daouda Kouotou
b,
Daouda Kouotou a,
Peter Machata
a,
Peter Machata b,
Ndi Juluis Nsami
b,
Ndi Juluis Nsami a,
Angela Kleinováb,
Julien Parmentier
a,
Angela Kleinováb,
Julien Parmentier c and
Zdenko Špitalský
c and
Zdenko Špitalský b
b
aApplied Physical and Analytical Chemistry Laboratory, Department of Inorganic Chemistry, Faculty of Science, University of Yaoundé I, P. O. Box 812, Yaoundé, Cameroon. E-mail: blaise.lekene@univ-yaounde1.cm
bPolymer Institute, Slovak Academy of Sciences, Dúbravská cesta 9, 845 41, Bratislava 45, Slovakia
cInstitut de Science des Matériaux de Mulhouse – IS2M – UMR 7361, Université de Haute-Alsace, 15 rue Jean Starcky, 68057 Mulhouse, France
First published on 21st April 2026
Abstract
Synthetic dyes, such as congo red (CR), are among the most hazardous pollutants released into aquatic environments due to their toxicity, persistence, and potential health risks. This study prepared and extensively characterized novel chitosan–biochar biocomposite films using SEM-EDS, N2 adsorption–desorption, FTIR, XPS, TGA, tensile testing, water contact angle measurement, and water absorption and solubility testing to efficiently remove CR dye from aqueous solutions. These characterization techniques revealed that the films exhibited excellent mechanical strength, surfaces enriched with –OH, –NH2, and –COOH functional groups and low porosity. Furthermore, the prepared films could be easily separated from the solution after adsorption. The CR removal percentages of the composites were ∼74%, 73%, and 85% for pristine biochar (CH2BC), acid-modified (CH2ABC), and base-modified (CH2BBC), respectively, after 420 minutes with a solution pH of 6.5 at 25 °C. The adsorption data suggested that the kinetic models were those of pseudo-first-order and Elovich models, while the Freundlich model best described the adsorption isotherm. Moreover, CH2BBC retained up to 79% of its adsorption capacity after four adsorption–desorption cycles, confirming its reusability. The CR adsorption mechanism involves electrostatic attraction, π–π stacking, H-bonding, n–π interaction, imine and sulfone bridging. The environmentally friendly preparation, low cost, and easy regeneration of the prepared chitosan–biochar biocomposite films offer a promising sustainable solution for remediating dye-contaminated water.
1. Introduction
Congo red (CR), a synthetic dye, is classified as an anionic diazo dye due to the presence of two –N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– bonds in its structure. CR has been widely used in various industries for dyeing products such as paper, rubber, textile, and leather,1 as well as in the formulation of histological stains.2,3 As the use of CR is constantly increasing, huge amounts of wastewater containing CR are streamed into surface waters, causing its depletion and negatively affecting the aquatic flora and fauna by inhibiting light penetration.4–6 In terms of its impact on human health effects, CR has been identified as carcinogenic and causes allergic dermatitis and skin irritation.4 As a result, various techniques have been reported for the treatment of dye-contaminated water, including ozonation, coagulation, chemical oxidation, nanofiltration, electrochemical oxidation, adsorption, and photodegradation.7–10 Among these techniques, adsorption has been identified as the most convenient method for removing dyes from water due to its low cost, high efficiency, ease of implementation, and the absence of secondary pollution.9,11 The type of interaction between the adsorbed species and the adsorbent determines which type of adsorption occurs. Chemisorption is a selective process characterized by strong interactions (covalent or ionic) between the adsorbent and the adsorbate, resulting in an irreversible process. In contrast, physisorption involves weak interactions, such as hydrogen bonding, dipole–dipole, van der Waals, and polar interactions. Physisorption is preferred for water treatment due to its nonselective and reversible process.10,12
N– bonds in its structure. CR has been widely used in various industries for dyeing products such as paper, rubber, textile, and leather,1 as well as in the formulation of histological stains.2,3 As the use of CR is constantly increasing, huge amounts of wastewater containing CR are streamed into surface waters, causing its depletion and negatively affecting the aquatic flora and fauna by inhibiting light penetration.4–6 In terms of its impact on human health effects, CR has been identified as carcinogenic and causes allergic dermatitis and skin irritation.4 As a result, various techniques have been reported for the treatment of dye-contaminated water, including ozonation, coagulation, chemical oxidation, nanofiltration, electrochemical oxidation, adsorption, and photodegradation.7–10 Among these techniques, adsorption has been identified as the most convenient method for removing dyes from water due to its low cost, high efficiency, ease of implementation, and the absence of secondary pollution.9,11 The type of interaction between the adsorbed species and the adsorbent determines which type of adsorption occurs. Chemisorption is a selective process characterized by strong interactions (covalent or ionic) between the adsorbent and the adsorbate, resulting in an irreversible process. In contrast, physisorption involves weak interactions, such as hydrogen bonding, dipole–dipole, van der Waals, and polar interactions. Physisorption is preferred for water treatment due to its nonselective and reversible process.10,12
Several adsorbent materials have been used for the removal of dye from water, including metal oxide,13,14 silica gel, resin, and zeolite,9 clay,15 silica, carbon-based materials (activated carbon, carbon quantum dots, carbon nanotubes, graphitic carbon).12,16 However, they are difficult to use for large-scale applications due to their drawbacks, including poor reusability and high cost. Biochar, an emerging carbonaceous material with outstanding physicochemical properties, produced by the pyrolysis of biomass in a limited or oxygen-free atmosphere, has demonstrated its effectiveness in water treatment.17,18 To further enhance its adsorptive capacity for targeted pollutants, biochar has been modified using several advanced techniques that improve its physicochemical properties.19,20 Even with its great capacity for removing pollutants from water, recovering and/or regenerating biochar, especially in powder form, can be challenging in practical applications. Once dispersed in water, the powder can become a source of secondary waste. Thus, by extending its lifetime, the disposal of the biochar as a secondary hazardous material is considerably reduced. One solution is to shape the biochar by incorporating it into an appropriate polymer matrix. This could yield a material that not only increases its adsorption capacity but also facilitates its handling, recovery, and reuse after treatment.21
Chitosan, a biodegradable and environmentally friendly polymer, has received considerable attention for water treatment due to its nontoxicity and high content of hydroxyl (–OH) and amine (–NH2) groups, which allow strong interaction with various classes of pollutants such as dyes, heavy metals, and other organic/inorganic contaminants has gained significant attention for water treatment.22,23 However, the use of chitosan is limited by its low mechanical strength and instability in acidic environments, which can be overcome by using cross-linking agents and/or combining it with other materials, such as carbonaceous material, to form composites. In this regard, various chitosan–biochar composites have been used in several studies for the removal of dyes,24,25 heavy metals,26,27 nitrate and phosphate ions28,29 and pharmaceuticals30,31 from water.
Although the reported studies have demonstrated the efficiency of chitosan–biochar composites for water treatment, most of these studies have focused on using them in powder form. This poses the problem of separation (filtration and/or centrifugation), secondary pollution generation, and reusability after treatment, which significantly increases processing duration and cost. In this study, in addition to its interesting surface functional groups (–OH and –NH2), chitosan was used as a binder polymer for the preparation of biochar film to prevent its dispersion and ensure its environmental application. To the best of our knowledge, there are no studies on using chitosan–biochar composites in the form of films for congo red dye removal from wastewater. Another important aspect of this study is the conversion of agricultural waste into a resource for biochar production. West African countries, including Nigeria, Ghana, the Ivory Coast, and Cameroon, are the main producers of kola nuts. In 2020, more than 300![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons were produced.32,33 Unfortunately, the resulting waste, kola nut shells, is often disposed of untreated in the landfills, causing serious soil and groundwater depletion. Using kola nut shells as the starting material for biochar production could be a good alternative for reducing the environmental footprint. The prepared chitosan–biochar films were easily separated from the solution after the sorption process. They were easily regenerated and could be used for more than five adsorption–desorption cycles. We also investigated the effects of acid/base modification of the biochar surface, as well as the effects of the chitosan/biochar ratio, on the physicochemical, mechanical, and textural properties and adsorption capacities of the resulting biocomposite films towards CR. Finally, we proposed an adsorption mechanism of CR on the prepared biocomposite films.
000 tons were produced.32,33 Unfortunately, the resulting waste, kola nut shells, is often disposed of untreated in the landfills, causing serious soil and groundwater depletion. Using kola nut shells as the starting material for biochar production could be a good alternative for reducing the environmental footprint. The prepared chitosan–biochar films were easily separated from the solution after the sorption process. They were easily regenerated and could be used for more than five adsorption–desorption cycles. We also investigated the effects of acid/base modification of the biochar surface, as well as the effects of the chitosan/biochar ratio, on the physicochemical, mechanical, and textural properties and adsorption capacities of the resulting biocomposite films towards CR. Finally, we proposed an adsorption mechanism of CR on the prepared biocomposite films.
2. Material and methods
2.1. Reagents
Kola nut shells were obtained from a local farm in Donga Mantum division, Cameroon. Chitosan (degree of deacetylation ≥ 80%, average M.W.: 190–310 kDa) from TCI Europe, glutaraldehyde (25%, w/v) from Sigma-Aldrich, congo red from Roth, acetic acid (CH3COOH), sodium hydroxide (NaOH), nitric acid 65% (HNO3) from Lachner, hydrochloride acid (HCl), sodium chloride (NaCl), sodium iodide (NaI), sodium sulfate (Na2SO4), sodium carbonate (Na2CO3), disodium phosphate (Na2HPO4), potassium chloride (KCl), calcium chloride (CaCl2), iron(III) chloride (FeCl3), magnesium chloride (MgCl2), barium chloride (BaCl2) were purchased from Centralchem (Bratislava, Slovakia). All the chemicals were used as received. Deionized water (18.2 MΩ cm−1) received from the Millipore Direct-Q3 device was used to prepare solutions.2.2. Sample preparation
Cola nut shells were washed and sun-dried, followed by pulverization, and the particles with sizes less than 0.8 mm were retained. The obtained particles were pyrolyzed at 500 °C in a muffle furnace (Carbolite Chamber Furnace, Germany) with a temperature rising of 10 °C min−1 for 1 hour. After cooling the furnace to room temperature, the biochar was ground, passed through an 80 µm sieve, and stored for further use.18 To modify the surface properties of biochar, 20 g of biochar was reacted with 200 mL of one molar HNO3 or NaOH solution under magnetic stirring at room temperature and stored overnight to ensure adequate reaction. Acid and alkaline-modified biochars (ABC and BBC) were washed with deionized water until the pH of the supernatant was stable, followed by oven drying at 80 °C for 12 hours.25The chitosan–biochars films were prepared according to the procedure described by Rizzi et al. (2019)34 with some slight modifications. Briefly, 1% (w/v) of chitosan solution was obtained by dissolving a known amount of chitosan powder in aqueous acetic acid solution (0.8%, v/v) under vigorous stirring for 3 hours at room temperature. Then, 750 µL of glutaraldehyde (cross-linking agent) was added to each 100 mL of chitosan solution, under constant stirring. After 24 hours of stirring the hydrogel solution was filtered with the coarse sintered glass followed by ultrasound sonication for 15 min to remove bubbles. The chitosan–biochars films were obtained by adding 50 or 100 mg of biochars (BC, ABC, or BBC) to 100 mL of chitosan hydrogel solution, followed by ultrasound sonication for 15 min and stirring at 450 rpm for 6 hours. The resulting blackish solution (10 mL) was then cast onto a glass Petri dish (10 cm diameter), oven-dried for 16 hours at 60 °C, and cut into small pieces with an approximate size of 1 cm × 1 cm. Based on the amount and type of biochar added to the chitosan hydrogel solution, the biocomposite films were labeled CHxBC, CHxABC, and CHxBBC. Where x = 1 or 2 represents the mass ratio of chitosan to biochar. Fig. 1 shows the simplified schematic diagram of the preparation of chitosan–biochar films and their application.
 | ||
| Fig. 1 Diagram of the preparation biochars and biocomposite chitosan–biochar films, adsorption, and simple solid–liquid phase separation. | ||
2.3. Characterization
A series of characterization techniques, including SEM, N2 adsorption–desorption, FTIR, XPS, pHpzc, tensile properties, contact angle, water absorption, solubility in water, and TGA, as described in the SI (S1), were used to evaluate the physicochemical and mechanical properties of prepared materials.2.4. Adsorption experiments
All the adsorption experiments were performed in closed reactor by mixing the adsorbent materials and CR solution at a mass to volume ratio of 1 g L−1. A 15 mg L−1 CR solution was used for the kinetic studies and was sampled at regular time intervals (10–420 min). The isotherm adsorption experiments were carried out for 24 hours at room temperature in the concentration range 15–340 mg L−1. Kinetic and isotherm studies were conducted at room temperature (RT) and at the natural pH of the CR's solution (6.5). The environmental parameters affecting the adsorption process such as agitation speed (0–320 rpm), pH (3–11), temperature (30–45 °C) leading to thermodynamic studies and interfering anions (Cl−, I−, SO42−, HPO42−, CO32−) and cations (Na+, K+, Ca2+, Fe2+, Mg2+, Ba2+) were also studied. The pH of the solution was adjusted with a molar solution of NaOH and HCl, while the interfering ions concentration was aligned with that of CR and set at 15 mg L−1.Only samples containing powder adsorbent (BC, ABC, or BBC) were filtered with a 0.45 µm filter syringe prior to analysis. The CR concentrations in the solution before and after adsorption experiments were evaluated by measuring the absorbance of the solution at a sensitive maximum wavelength of 498 nm (Fig. S1a) using a UV-vis spectrophotometer (Shimadzu UV-1650PC, Japan) based on a pre-established calibration curve (Fig. S1b). The uptake capacity (qe, mg g−1) and the removal efficiency (R, %) were calculated using the respective following equations:
 | (1) |
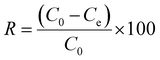 | (2) |
2.5. Modeling for congo red adsorption
Adsorption kinetic data were analyzed using pseudo-first and pseudo-second order, Elovich, and intra-particle diffusion models, while the adsorption equilibrium data were fitted by Langmuir, Freundlich, and Temkin isotherm models. Thermodynamic data were analyzed using the Gibbs–Helmholtz equation to evaluate the thermodynamic properties of CR adsorption onto biocomposite films. All models and parameters information are provided in the SI (S2).2.6. Regeneration and reusability experiments
The reusability of biocomposite films was investigated by four adsorption–desorption cycles using 0.5 mol L−1 sodium hydroxide solution as eluent.1 Initially, biofilm (CH2BC, CH1BC, CH2ABC, CH1ABC, CH2BBC, or CH1BBC) was contacted with a 15 mg L−1 CR solution at a ratio of 1 g L−1 and agitated at 320 rpm for 24 hours. After adsorption, regeneration was performed by mixing the CR-loaded films with 0.5 mol L−1 NaOH solution and agitating for 30 min, followed by extensive washing with deionized water, drying at 60 °C, and using for another adsorption–desorption cycle.All adsorption and desorption experiments were performed at least in triplicates, and the average values and standard deviations were reported.
3. Results and discussion
3.1. Characterization of materials
Based on the elemental analysis obtained from EDS (Fig. S2, Tables 1 and S1), nitrogen was not detected in any of the analyzed materials. The main contents were C and O, while some alkaline mineral elements (K, Ca, and Mg) were detected. Acid treatment considerably reduces the mineral content in biochar from approximately 3.68% in BC to ∼0.96% in ABC while it increases to ∼4.30% in BBC. This finding was supported by the work of Collivignarelli et al.37 On the other hand, the O/C ratios of BC, ABC, and BBC were found to be 0.21, 0.33, and 0.23 respectively, indicating that acid treatment provided more oxygenated groups (–COOH, –OH).38,39 The finding in EDS elemental composition was not easily comparable to that of XPS since, in the latter, only the uppermost layers (∼10 nm) were analyzed and with high sensitivity indicating that the nitrogen is only found on the surface and not within the bulk of the material. Nevertheless, XPS was complementary to EDS. All the biocomposite films exhibited low C and higher O content compared to their respective based biochar indicating successful obtention of biocomposite.
| Samples | C (EDS/XPS) (%) | O (EDS/XPS) (%) | N (EDS/XPS) (%) | Minerals (EDS/XPS) (%) | Specific surface area (m2 g−1) | Pore volume (cm3 g−1) | Pore width (nm) | pHpzc |
|---|---|---|---|---|---|---|---|---|
| a n.d. not detected by EDS. | ||||||||
| BC | 79.83/75.96 | 16.49/16.40 | n.d./2.32 | 3.68/5.3 | 7.542 | 0.0313 | 35.450 | — |
| CH2BC | 59.16/62.48 | 39.04/28.45 | n.d./4.54 | 1.79/4.5 | 0.731 | 0.0012 | 7.203 | 6.62 |
| CH1BC | — | — | — | — | 1.034 | 0.0038 | 7.877 | 6.63 |
| ABC | 74.21/78.03 | 24.78/17.41 | n.d./3.70 | 0.96/0.9 | 2.899 | 0.0077 | 15.333 | — |
| CH2ABC | 69.32/64.51 | 30.10/28.99 | n.d./5.74 | 0.57/0.8 | 0.027 | n.a. | n.a. | 6.64 |
| CH1ABC | — | — | — | — | 7.075 | 0.0073 | 5.653 | 7.09 |
| BBC | 78.00/68.71 | 17.70/21.70 | n.d./2.40 | 4.30/7.2 | 2.552 | 0.0078 | 21.565 | — |
| CH2BBC | 61.25/62.77 | 37.00/29.39 | n.d./6.07 | 1.75/1.7 | 1.255 | 0.0015 | 7.722 | 6.52 |
| CH1BBC | — | — | — | — | 1.949 | 0.0047 | 11.973 | 6.77 |
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O/C
O/C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, and O–C
C, and O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds,27,41 respectively. These results are in agreement with elemental analysis, suggesting that the acid/alkaline treatment successfully increased the oxygen-containing groups, which are important in the mechanism of obtaining chitosan–biochar composite.39,42 Compared to biochar, chitosan displayed many more peaks (Fig. 3b). The peak at 3374 cm−1 could be assigned to the vibration of overlapping O–H and N–H bonds, while the peaks at 2941 and 2887 cm−1 could be the stretching vibration of C–H bonds methyl and methylene groups respectively.29 The stretching vibration of the C
O bonds,27,41 respectively. These results are in agreement with elemental analysis, suggesting that the acid/alkaline treatment successfully increased the oxygen-containing groups, which are important in the mechanism of obtaining chitosan–biochar composite.39,42 Compared to biochar, chitosan displayed many more peaks (Fig. 3b). The peak at 3374 cm−1 could be assigned to the vibration of overlapping O–H and N–H bonds, while the peaks at 2941 and 2887 cm−1 could be the stretching vibration of C–H bonds methyl and methylene groups respectively.29 The stretching vibration of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond of the primary amide appeared at 1644 cm−1, while the band at 1382 cm−1 was assigned to the overlapping of –CH3 symmetric angular deformation and C–N stretching. The peak at 1071 cm−1 corresponds to C–O stretching in the β(1→4) glycosidic bond.43,44 In addition to the functions present in chitosan, the composites also exhibit the characteristics of biochars indicating the successful fabrication of biocomposite. On the other hand, the drastic reduction of the intensities of the peaks related to C
O bond of the primary amide appeared at 1644 cm−1, while the band at 1382 cm−1 was assigned to the overlapping of –CH3 symmetric angular deformation and C–N stretching. The peak at 1071 cm−1 corresponds to C–O stretching in the β(1→4) glycosidic bond.43,44 In addition to the functions present in chitosan, the composites also exhibit the characteristics of biochars indicating the successful fabrication of biocomposite. On the other hand, the drastic reduction of the intensities of the peaks related to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and O–C
O and O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in the composite (Fig. 3b) indicates the coverage of biochar by chitosan and the possible reaction between the carboxylic group on the surface of biochar and the amine function on chitosan as the XPS will confirm.
O in the composite (Fig. 3b) indicates the coverage of biochar by chitosan and the possible reaction between the carboxylic group on the surface of biochar and the amine function on chitosan as the XPS will confirm.
Full scan XPS spectra indicated that C, O, and N were the primary constituents of BC, ABC, BBC, CH2BC, CH2ABC, and CH2BBC (Fig. S4). The higher content of oxygenated carbons (C–O, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, and O–C
O, and O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) of ABC and BBC compared to BC (Tables 1 and S2) indicated successful acidic/alkaline biochar oxidation consistent with EDS and FTIR analysis. In all the biocomposites, there is an increase in the proportion of N 1s and O 1s and a decrease in C 1s which may be due to the surface exposure of many nitrogen/oxygen-containing groups (Table S2),45 further confirming the biocomposite formation. The peak of Na 1s detected on the spectra of BBC and CH2BBC (with low intensity) (Fig. S4) may be due to the introduction of Na+ during the NaOH treatment which reacted with –COOH to form –COONa.25 The deconvoluted C 1s spectrum of BBC showed that it has the characteristics of graphitized/graphenized materials containing nitrogen with six peaks (Fig. 4a) centered at 284.75 (C–C/C–H), 284.25 (C
O) of ABC and BBC compared to BC (Tables 1 and S2) indicated successful acidic/alkaline biochar oxidation consistent with EDS and FTIR analysis. In all the biocomposites, there is an increase in the proportion of N 1s and O 1s and a decrease in C 1s which may be due to the surface exposure of many nitrogen/oxygen-containing groups (Table S2),45 further confirming the biocomposite formation. The peak of Na 1s detected on the spectra of BBC and CH2BBC (with low intensity) (Fig. S4) may be due to the introduction of Na+ during the NaOH treatment which reacted with –COOH to form –COONa.25 The deconvoluted C 1s spectrum of BBC showed that it has the characteristics of graphitized/graphenized materials containing nitrogen with six peaks (Fig. 4a) centered at 284.75 (C–C/C–H), 284.25 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 286.07 (C–O/C–N), 287.71 (C
C), 286.07 (C–O/C–N), 287.71 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 298.11 (O–C
O), 298.11 (O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), and 291.40 (π–π* satellite) eV.26,46,47 In the spectra of the biocomposite (Fig. 4b), there is the disappearance of a π–π* peak and the formation of the N–C
O), and 291.40 (π–π* satellite) eV.26,46,47 In the spectra of the biocomposite (Fig. 4b), there is the disappearance of a π–π* peak and the formation of the N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond (288.08 eV). In addition, there is an increase in the percentages of C–O/C–N (from 8.32 to 51.44%) and C–C/C–H (from 15.31 to 22.16%) accompanied by peak shifts, indicating that interactions occurred between BBC and chitosan.27 In the O 1s spectra of BBC, three peaks were present at 531.30 eV (C
O bond (288.08 eV). In addition, there is an increase in the percentages of C–O/C–N (from 8.32 to 51.44%) and C–C/C–H (from 15.31 to 22.16%) accompanied by peak shifts, indicating that interactions occurred between BBC and chitosan.27 In the O 1s spectra of BBC, three peaks were present at 531.30 eV (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, 63.04%), 532.75 eV (aliphatic –OH, 31.94%), and 535.00 eV (aromatic –OH, 5.02%) (Fig. 4c), whereas in CH2BBC (Fig. 4d), we noticed the disappearance of the latter peak and significant increases in the proportion of aliphatic –OH (86.59%), respectively, attributed to the surface coverage of BBC by chitosan and the abundant presence of hydroxyl on this latter.25,48 Deconvolution of the N 1s spectrum of BBC (Fig. 4e) revealed four peaks at 398.3 eV (29.17%), 400.00 eV (56.25%), 402.00 eV (6.67%), and 405.42 eV (7.92%) assigned to C
O, 63.04%), 532.75 eV (aliphatic –OH, 31.94%), and 535.00 eV (aromatic –OH, 5.02%) (Fig. 4c), whereas in CH2BBC (Fig. 4d), we noticed the disappearance of the latter peak and significant increases in the proportion of aliphatic –OH (86.59%), respectively, attributed to the surface coverage of BBC by chitosan and the abundant presence of hydroxyl on this latter.25,48 Deconvolution of the N 1s spectrum of BBC (Fig. 4e) revealed four peaks at 398.3 eV (29.17%), 400.00 eV (56.25%), 402.00 eV (6.67%), and 405.42 eV (7.92%) assigned to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–C, –NH2, –NH2+–/–NH3+ and nitro (–NO2) respectively, while, these peaks shifted slightly and changed considerably in intensity on CH2BBC (Fig. 4f).26,49 The results of XPS were in agreement with those of FTIR.
N–C, –NH2, –NH2+–/–NH3+ and nitro (–NO2) respectively, while, these peaks shifted slightly and changed considerably in intensity on CH2BBC (Fig. 4f).26,49 The results of XPS were in agreement with those of FTIR.
For BC- and BBC-based biocomposites, the solubility (dissolution) slightly increased with the amount of biochar, while the opposite was observed for ABC-based films (Fig. 5c). As shown by EDS results, ABC possessed a more oxygenated group which can strongly react with amine groups of chitosan, and contributed more to the reduction of the solubility of CH1ABC than CH2ABC. Nevertheless, the solubility of the films did not exceed 16%, indicating that they can be reused during many adsorption–desorption cycles.
3.2. Adsorption of congo red onto biocomposite films
 | ||
| Fig. 6 Adsorption kinetic ([CR]0 = 15 mg L−1, RT, pH 6.5, 240 rpm); pseudo-first-order, pseudo-second-order, and Elovich kinetic model (a–c), intraparticle diffusion fitting model (d–f). | ||
Pseudo-first-order, pseudo-second-order, and Elovich models were used to fit the adsorption kinetics data of CR (Fig. 6a–c), and the corresponding fitted parameters are given in Table S3. The kinetics data of ABC and BBC were well fitted by the pseudo-second-order and Elovich models, while the pseudo-first-order and Elovich models were suitable for BC according to their high values of R2 and low values of RMSE and χ2, indicating that the adsorption process is governed by the chemisorption on heterogeneous surfaces. Indeed, the calculated values of qe from the pseudo-second-order model were closer to those observed experimentally. Moreover, α, the initial rate constant of Elovich is larger than the desorption constant β, indicating a strong affinity between CR and the biochar surfaces.11,18 However, for the biocomposite films, based on their error functions (R2, RMSE and χ2) we rather observed that the data are well fitted by the pseudo-second-order which often characterizes the chemisorption mechanism56 and the Elovich model. Nevertheless, the similarity between the experimental qe and those derived from the pseudo-first-order model also makes it suitable to describe the adsorption kinetics of CR on films, suggesting the involvement of the physisorption in the adsorption process.57 This latter assertion is supported by the Elovich model, in which the desorption constant β is higher than the initial adsorption α, indicating that CR interacts weakly with the surface films.
To determine the rate-controlling step of the adsorption of the dye onto the prepared adsorbent materials, the kinetic data were analyzed with the Weber–Morris intra-particle diffusion model.58 Since it was not possible to distinguish the two or three subdivisions corresponding to the different stages of the diffusion process, the fitted curves of the Weber–Morris intra-particle diffusion model showed a straight line (Fig. 6d–f) for all the films. This observation indicates that the multiple diffusion steps of the adsorption process are smoothly overlapped. In addition, the linear regression of the plots qt vs. t1/2 did not pass through the origin, resulting in the values of the boundary layer thickness constant C different from zero in all cases (Table S3), suggesting that the intra-particle diffusion was not the only controlling step for CR adsorption onto biocomposite films, as boundary layer diffusion also played a significant role in the process.11 Due to their low R2 (Table S3), the intra-particle diffusion model was not suitable to fit the kinetic data of the adsorption of CR onto the different biochars (BC, ABC and BBC). Due to their low adsorption capacities and the intervention of the filtration step in the experimental procedure, which significantly affected the time and the cost of treatment, the biochars were ignored and the experiments continued with biocomposite films only.
The CR removal performance of the prepared biocomposite films was evaluated by comparing their Langmuir monolayer adsorption capacity with those of other reported materials listed in Table 2. In general, the biocomposite films exhibit high adsorption capacity. Their low-cost, environmentally friendly synthesis process, easy recovery, and facile regeneration allowing multiple reuse cycles as shown below, highlight their potential as promising adsorbent materials for CR remediation in aqueous solutions.
| Adsorbents | pH | Temperature (°C) | Adsorbent dose (g L−1) | Concentration (mg L−1) | Type | Qmax | Ref. |
|---|---|---|---|---|---|---|---|
| a Room temperature. | |||||||
| Sugarcane bagasse | 5 | 30 | 10 | 100–500 | Powder | 38.20 | 63 |
| Polypyrrole modified nanocellulose | 2 | 50 | — | 10–50 | Powder | 46.78 | 64 |
| Activated carbon based-cocoa pod husks | 2 | 50 | 1 | 20–100 | Powder | 43.67 | 65 |
| Fe3O4@carbon@ZIF-8 | 4 | RTa | 3 | 30–1000 | Powder | 566.53 | 66 |
| Eucalyptus wood sawdust | 7 | 30 | 4 | 5–30 | Powder | 31.25 | 67 |
| KOH-activated biochar | 4 | 25 | 2 | 25–100 | Powder | 6.238 | 68 |
| Fly ash | Natural | RTa | 1 | 10–50 | Powder | 22.12 | 14 |
| PANI@ZnO | 5 | 25 | 0.4 | 50–200 | Powder | 69.82 | 69 |
| Pine bark | Natural | 25 | 10 | 5–100 | Powder | 3.92 | 5 |
| Cellulose/chitosan hydrogel | — | 25 | 2 | 10–70 | Bead | 40.00 | 70 |
| Banana peel | 7.9 | 30 | 1 | 10–120 | Powder | 18.20 | 71 |
| Orange peel | 7.9 | 30 | 1 | 10–120 | Powder | 14.00 | 71 |
| Wastepaper nanocomposite | 7 | 30 | 2 | 20–100 | Membrane | 71.00 | 9 |
| Kaolin | 3 | 30 | 1 | 20–60 | Powder | 5.41 | 15 |
| Aspergillus niger | 6 | — | 2.67 | — | Powder | 14.72 | 72 |
| CH1BC | 6.5 | RTa | 1 | 15–340 | Film | 48.95 | This work |
| CH2BC | 55.26 | ||||||
| CH1ABC | 28.94 | ||||||
| CH2ABC | 44.72 | ||||||
| CH1BBC | 32.98 | ||||||
| CH2BBC | 62.23 | ||||||
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–) present in the CR molecule is the most affected by the pH variation, leaving the sulfonate group (–SO3−) unprotonated,2,3 which would thus be the most reactive site for the adsorption process.13 Regarding the adsorbents, added to the surface functional groups discussed by XPS and FTIR, the pHpzc evaluated by the drift method75 were found to be approximately in the range of 6.27–7.08 (Table 1 and Fig. S7). Therefore, at pH < pHpzc, the surface charges of the films are dominated by positive charges and vice versa. Under strongly acidic conditions, most of the surface functional groups (–NH2, –COOH, and –OH) are protonated due to the availability of more protons in the solution, resulting in a high electrostatic attraction of the sulfonate group (–SO3−), leading to higher adsorption capacities at pH 3. With increasing pH, the surface becomes less and less protonated until the pH > pHpzc, where the surface becomes negatively charged, reducing the adsorption capacity due to less electrostatic attraction. In addition, the presence of hydroxyl ions in the solution at higher pH may compete with CR for adsorption sites. At a high pH range, the adsorption of CR onto films was still effective, indicating that other interactions (e.g., H-bonding, n–π and π–π stacking), besides electrostatic attraction, were involved in the adsorption mechanisms of CR.76
N–) present in the CR molecule is the most affected by the pH variation, leaving the sulfonate group (–SO3−) unprotonated,2,3 which would thus be the most reactive site for the adsorption process.13 Regarding the adsorbents, added to the surface functional groups discussed by XPS and FTIR, the pHpzc evaluated by the drift method75 were found to be approximately in the range of 6.27–7.08 (Table 1 and Fig. S7). Therefore, at pH < pHpzc, the surface charges of the films are dominated by positive charges and vice versa. Under strongly acidic conditions, most of the surface functional groups (–NH2, –COOH, and –OH) are protonated due to the availability of more protons in the solution, resulting in a high electrostatic attraction of the sulfonate group (–SO3−), leading to higher adsorption capacities at pH 3. With increasing pH, the surface becomes less and less protonated until the pH > pHpzc, where the surface becomes negatively charged, reducing the adsorption capacity due to less electrostatic attraction. In addition, the presence of hydroxyl ions in the solution at higher pH may compete with CR for adsorption sites. At a high pH range, the adsorption of CR onto films was still effective, indicating that other interactions (e.g., H-bonding, n–π and π–π stacking), besides electrostatic attraction, were involved in the adsorption mechanisms of CR.76
The appearance of the peaks at about 1225, 1174, and 1063 cm−1 respectively assigned to C–N, S–O, and S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O vibrations in the CR molecule4 on the biocomposite after adsorption (Fig. 9a) indicated that the dye was adsorbed on the surface of biocomposite. The narrowing and intensification of the –NH/–OH joint band centered at 3362 cm−1 on the biocomposite after CR adsorption indicated the formation of H-bonding between the –NH/–OH of CH2BBC and –NH2 of CR.19 The peak at 1563 cm−1 on CH2BBC which was assigned to the C
O vibrations in the CR molecule4 on the biocomposite after adsorption (Fig. 9a) indicated that the dye was adsorbed on the surface of biocomposite. The narrowing and intensification of the –NH/–OH joint band centered at 3362 cm−1 on the biocomposite after CR adsorption indicated the formation of H-bonding between the –NH/–OH of CH2BBC and –NH2 of CR.19 The peak at 1563 cm−1 on CH2BBC which was assigned to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C (aromatic) vibration shifted to 1584 cm−1 after CR adsorption indicating the π–π interaction between the aromatic ring of the dye and that of the film derived from BC.75
C (aromatic) vibration shifted to 1584 cm−1 after CR adsorption indicating the π–π interaction between the aromatic ring of the dye and that of the film derived from BC.75
 | ||
| Fig. 9 FTIR spectra (a), XPS full scan (b), and high-resolution spectra of C 1s (c), O 1s (d), N 1s (e), and S 2p (f). | ||
Two main features were observed on the XPS spectrum full scan of CH2BBC-CR (Fig. 9b): (1) the rising of a peak attributed to S 2p attributed to sulfur present in CR molecule and clearly indicated that it was immobilized on the surface of CH2BBC, and (2) the disappearance of the Na 1s peak indication the occurrence of ion exchange between –COONa and CR.25 Moreover, in the high-resolution spectrum, we noticed a variation in peak intensities and movement of the binding energy of C 1s, O 1s, and N 1s indicating/confirming the change of the surface chemistry of CH2BBC after CR adsorption. The deconvoluted peak of C 1s (Fig. 9c) revealed that the content of C–C/C–H and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C increased indicating that a large amount of CR was adsorbed onto the surface of CH2BBC.83 The reductions of C–O/C–N content from 51.44 to 30.69% and that of N–C
C increased indicating that a large amount of CR was adsorbed onto the surface of CH2BBC.83 The reductions of C–O/C–N content from 51.44 to 30.69% and that of N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O from 12.79 to 11.23% indicated their contribution to CR removal which may be through H-bonding.83,84 The increase (from 3.14 to 3.79%) of O–C
O from 12.79 to 11.23% indicated their contribution to CR removal which may be through H-bonding.83,84 The increase (from 3.14 to 3.79%) of O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O content after adsorption may be due to the reaction between N–C
O content after adsorption may be due to the reaction between N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (–COOH) of CH2BBC and the sulfonate group of CR.85 On O 1s spectra (Fig. 9d), the increase in the percentage of –OH from 86.59 to 93.56% may correlated to the interaction between –SO3− and the protonated functions of CH2BBC. The C
O (–COOH) of CH2BBC and the sulfonate group of CR.85 On O 1s spectra (Fig. 9d), the increase in the percentage of –OH from 86.59 to 93.56% may correlated to the interaction between –SO3− and the protonated functions of CH2BBC. The C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O content significantly decreases from 13.41 to 6.44% due to the sharing of free electron pair bond of O with CR through n–π interactions.66 This reduction may also be due to the reaction between C
O content significantly decreases from 13.41 to 6.44% due to the sharing of free electron pair bond of O with CR through n–π interactions.66 This reduction may also be due to the reaction between C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (carbonyl) of CH2BBC and –NH2 of CR to form an imine function as confirmed by the increase of the C
O (carbonyl) of CH2BBC and –NH2 of CR to form an imine function as confirmed by the increase of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–C fraction from 8.57 to 28.43% in N 1s spectra (Fig. 9e), which among other things, contributes to confirm the chemisorption hypothesis presented above. The –NH2 present on the film may be involved in some interactions with CR including H-bonding, n–π (π electron of the CR benzene ring), confirmed by the slight increase of –NH2+–/–NH3+. The S 2p peak after deconvolution was split into two (Fig. 9f) centered at 168.68 and 167.36 eV attributed to the sulfone bridge (–C–S(O)2–C–) between the CR and CH2BBC.86 Although the study of the effect of pH revealed the importance of the electrostatic interactions, additional investigations using zeta potential–pH would be valuable to strengthen this assertion. The overall mechanism of CR adsorption onto biocomposite is illustrated in Fig. 10.
N–C fraction from 8.57 to 28.43% in N 1s spectra (Fig. 9e), which among other things, contributes to confirm the chemisorption hypothesis presented above. The –NH2 present on the film may be involved in some interactions with CR including H-bonding, n–π (π electron of the CR benzene ring), confirmed by the slight increase of –NH2+–/–NH3+. The S 2p peak after deconvolution was split into two (Fig. 9f) centered at 168.68 and 167.36 eV attributed to the sulfone bridge (–C–S(O)2–C–) between the CR and CH2BBC.86 Although the study of the effect of pH revealed the importance of the electrostatic interactions, additional investigations using zeta potential–pH would be valuable to strengthen this assertion. The overall mechanism of CR adsorption onto biocomposite is illustrated in Fig. 10.
 | ||
| Fig. 11 Reusability of biofilms (a) and reduction efficiency after each adsorption–desorption cycle (b). | ||
It is important to note that the main objective of the desorption process is to regenerate the adsorbent and transfer the pollutant from the solid phase to a smaller volume of the liquid phase. This concentrates the pollutant. To address the eventual question of the fate of the concentrated desorbed solution containing CR and NaOH, further treatment is required. This treatment could be an advanced oxidation process or electrochemical degradation. This would ensure complete mineralization of the CR before discharged into the environment.89
4. Conclusion
In the present study, the authors discussed the preparation and extensive characterization of novel biocomposite films based on pristine, HNO3-, and KOH-modified biochar and glutaraldehyde-crosslinked chitosan, which were applied for the removal of congo red from aqueous solution. Despite their low porosity, the biocomposite films exhibited a rich surface chemistry with oxygenated and aminated groups favorable for CR uptake. Their excellent mechanical strength enabled easy separation and reuse without filtration, offering a significant advantage over the powder adsorbents. Among the prepared adsorbent materials, the biocomposite film derived from KOH-treated biochar with a chitosan–BBC ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (CH2BBC) achieved the highest adsorption capacity (62.23 mg g−1). A comprehensive study of the effect of pH, kinetics, isotherms, and thermodynamics supported by the results of SEM-EDS, FTIR, and XPS of CH2BBC after CR adsorption revealed that multiple interactions, including electrostatic attraction, π–π stacking, H-bonding, n–π interaction, and sulfone bridging, governed the adsorption mechanism. CH2BBC retained up to 79% of its adsorption after four consecutive adsorption–desorption cycles, highlighting its strong reusability potential for CR removal from aqueous solution. In addition, these biocomposite films need to be tested in real wastewater effluents to evaluate their applicability for industrial-scale applications.
1 (CH2BBC) achieved the highest adsorption capacity (62.23 mg g−1). A comprehensive study of the effect of pH, kinetics, isotherms, and thermodynamics supported by the results of SEM-EDS, FTIR, and XPS of CH2BBC after CR adsorption revealed that multiple interactions, including electrostatic attraction, π–π stacking, H-bonding, n–π interaction, and sulfone bridging, governed the adsorption mechanism. CH2BBC retained up to 79% of its adsorption after four consecutive adsorption–desorption cycles, highlighting its strong reusability potential for CR removal from aqueous solution. In addition, these biocomposite films need to be tested in real wastewater effluents to evaluate their applicability for industrial-scale applications.
Author contributions
René Blaise Ngouateu Lekene: conceptualization, methodology, investigation, writing – original draft, funding acquisition; Mária Kováčová: investigation, resources; Christian Brice Dantio Nguela: writing – review and editing; Matej Mičušík: resources, investigation, data curation; Alena Opálková Šišková: investigation, resources; Anna Vykydalová: resources, investigation; Hamed Peidayesh: resources, investigation, data curation; Daouda Kouotou: resources, writing – review and editing, Peter Machata: resources, investigation, data curation; Ndi Juluis Nsami: resources, writing – review and editing; Angela Kleinová: investigation, resources; Julien Parmentier: writing – review and editing; Zdenko Špitalský: supervision, writing – review and editing, funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6ra01186a.Acknowledgements
René Blaise Ngouateu Lekene thanks the National Scholarship Programme (NSP) of the Slovak Republic for the financial support of his stay at the Polymer Institute, Slovak Academy of Sciences, and Dr Cyrille Vaulot (IS2M-CNRS) for N2 adsorption–desorption.References
- N. El Messaoudi, M. El Khomri, N. Chlif, Z. G. Chegini, A. Dbik, S. Bentahar and A. Lacherai, Groundw. Sustain. Dev., 2021, 12, 100552 CrossRef.
- E. Pigorsch, A. Elhaddaoui and S. Turrell, Spectrochim. Acta, Part A, 1994, 50, 2145–2152 CrossRef.
- K. Csillag, T. Emri, D. E. N. Rangel and I. Pócsi, Fungal Biol., 2023, 127, 1180–1186 CrossRef CAS PubMed.
- R. Kaur and H. Kaur, Port. Electrochim. Acta, 2016, 34, 185–196 CrossRef CAS.
- K. Litefti, M. S. Freire, M. Stitou and J. González-Álvarez, Sci. Rep., 2019, 9, 16530 CrossRef PubMed.
- N. F. Zainudin, S. T. Sam, Y. S. Wong, H. Ismail, S. Walli, K. Inoue, G. Kawamura and W. K. Tan, Polymers, 2023, 15, 237 CrossRef CAS PubMed.
- P. O. Oladoye, M. O. Bamigboye, O. D. Ogunbiyi and M. T. Akano, Groundw. Sustain. Dev., 2022, 19, 100844 CrossRef.
- R. R. M. Khan, H. Qamar, A. Hameed, A. ur Rehman, M. Pervaiz, Z. Saeed, A. Adnan and A. R. Ch, Water, Air, Soil Pollut., 2022, 233, 468 CrossRef CAS.
- R. Goswami, A. Mishra, A. Mirza, W. Ahmad and R. Rana, J. Appl. Polym. Sci., 2024, 141, e56275 CrossRef CAS.
- F. Mashkoor and A. Nasar, J. Magn. Magn. Mater., 2020, 500, 166408 CrossRef CAS.
- G. Agbor Tabi, L. Ngouateu Rene Blaise, K. Daouda, A. Naphtali Odogu, A. Aime Victoire, N. Nsami Julius and K. Joseph Mbadcam, Arabian J. Chem., 2022, 15, 103515 CrossRef CAS.
- H. Li, V. L. Budarin, J. H. Clark, M. North and X. Wu, J. Hazard. Mater., 2022, 436, 129174 CrossRef CAS PubMed.
- F. B. D. Saïah and N. Bettahar, Desalin. Water Treat., 2022, 254, 260–273 CrossRef.
- M. Harja, G. Buema and D. Bucur, Sci. Rep., 2022, 12, 6087 CrossRef CAS PubMed.
- T. A. Aragaw and A. N. Alene, Emerging Contam., 2022, 8, 59–74 CrossRef CAS.
- T. Panczyk, P. Wolski, A. Jagusiak and M. Drach, RSC Adv., 2014, 4, 47304–47312 RSC.
- J. Lu, C. Zhang, J. Wu and Y. Luo, Water, Air, Soil Pollut., 2017, 228, 327 CrossRef.
- R. B. N. Lekene, T. M. M. Ntep, M. N. A. Fetzer, T. Strothmann, J. N. Nsami and C. Janiak, Environ. Sci. Pollut. Res., 2023, 30, 100095–100113 CrossRef CAS PubMed.
- X. Huang, F. Yu, Q. Peng and Y. Huang, RSC Adv., 2018, 8, 29781–29788 RSC.
- J. Wang and M. Zhang, Int. J. Environ. Res. Public Health, 2020, 17, 1075 CrossRef CAS PubMed.
- N. Somsesta, V. Sricharoenchaikul and D. Aht-Ong, Mater. Chem. Phys., 2020, 240, 122221 CrossRef CAS.
- J. Desbrieres and E. Guibal, Polym. Int., 2018, 67, 7–14 CrossRef CAS.
- J. O. Gonçalves, M. M. Strieder, L. F. O. Silva, G. S. Dos Reis and G. L. Dotto, Int. J. Biol. Macromol., 2024, 270, 132307 CrossRef PubMed.
- A. H. Jawad, A. S. Abdulhameed, L. D. Wilson, M. A. K. M. Hanafiah, W. I. Nawawi, Z. A. ALOthman and M. Rizwan Khan, J. Polym. Environ., 2021, 29, 2855–2868 CrossRef CAS.
- X. Su, X. Wang, Z. Ge, Z. Bao, L. Lin, Y. Chen, W. Dai, Y. Sun, H. Yuan, W. Yang, J. Meng, H. Wang and S. C. Pillai, Chem. Eng. J., 2024, 486, 150387 CrossRef CAS.
- J. Song, S. A. Messele, L. Meng, Z. Huang and M. Gamal El-Din, Water Res., 2021, 194, 116930 CrossRef CAS PubMed.
- H. Chen, Y. Gao, A. El-Naggar, N. K. Niazi, C. Sun, S. M. Shaheen, D. Hou, X. Yang, Z. Tang, Z. Liu, H. Hou, W. Chen, J. Rinklebe, M. Pohořelý and H. Wang, J. Hazard. Mater., 2022, 425, 127971 CrossRef CAS PubMed.
- Y. Huang, X. Lee, M. Grattieri, M. Yuan, R. Cai, F. C. Macazo and S. D. Minteer, Chem. Eng. J., 2020, 380, 122375 CrossRef CAS.
- R. B. N. Lekene, N. O. Ankoro, D. Kouotou, G. B. N. Yemeli, S. A. Benedoue, J. N. Nsami and J. K. Mbadkam, Biomass Convers. Biorefin., 2024, 14, 20855–20872, DOI:10.1007/s13399-023-04239-0.
- J. Liu, B. Zhou, H. Zhang, J. Ma, B. Mu and W. Zhang, Bioresour. Technol., 2019, 294, 122152 CrossRef CAS PubMed.
- V. Son Tran, H. Hao Ngo, W. Guo, T. Ha Nguyen, T. Mai Ly Luong, X. Huan Nguyen, T. Lan Anh Phan, V. Trong Le, M. Phuong Nguyen and M. Khai Nguyen, Bioresour. Technol., 2023, 385, 129384 CrossRef CAS PubMed.
- A. J. Asuquo, X. Zhang, L. Lin and J. Li, Int. J. Green Energy, 2024, 21, 2218–2227 CrossRef CAS.
- J. Ndi Nsami and J. Ketcha Mbadcam, J. Chem., 2013, 2013, 469170 CrossRef.
- V. Rizzi, D. Lacalamita, J. Gubitosa, P. Fini, A. Petrella, R. Romita, A. Agostiano, J. A. Gabaldón, M. I. Fortea Gorbe, T. Gómez-Morte and P. Cosma, Sci. Total Environ., 2019, 693, 133620 CrossRef CAS PubMed.
- Z. Wang, J. Li, G. Zhang, Y. Zhi, D. Yang, X. Lai and T. Ren, Materials, 2020, 13, 2270 CrossRef CAS PubMed.
- B. Hayati and N. M. Mahmoodi, Desalin. Water Treat., 2012, 47, 322–333 CrossRef CAS.
- M. C. Collivignarelli, W. A. M. A. N. Illankoon, C. Milanese, S. Calatroni, F. M. Caccamo, M. Medina-Llamas, A. Girella and S. Sorlini, Water, 2024, 16, 698 CrossRef CAS.
- R. B. N. Lekene, D. Kouotou, N. O. Ankoro, A. P.-M. S. Kouoh, J. N. Ndi and J. M. Ketcha, J. Saudi Chem. Soc., 2021, 25, 101316 CrossRef CAS.
- Y. Xue, W. Cheng, M. Cao, J. Gao, J. Chen, Y. Gui, W. Zhu and F. Ma, Environ. Sci. Pollut. Res., 2022, 29, 77536–77552 CrossRef CAS PubMed.
- M. Thommes, K. Kaneko, A. V. Neimark, J. P. Olivier, F. Rodriguez-Reinoso, J. Rouquerol and K. S. W. Sing, Pure Appl. Chem., 2015, 87, 1051–1069 CrossRef CAS.
- R. B. N. Lékéné, J. N. Nsami, A. Rauf, D. Kouotou, P. D. B. Belibi, M. I. Bhanger and J. K. Mbadcam, Am. J. Anal. Chem., 2018, 09, 439 CrossRef.
- İ. Teğin, S. Öc and C. Saka, Biomass Convers. Biorefin., 2025, 15, 6805–6816, DOI:10.1007/s13399-024-05477-6.
- M. Monier, D. M. Ayad, Y. Wei and A. A. Sarhan, React. Funct. Polym., 2010, 70, 257–266 CrossRef CAS.
- J. K. Fatombi, E. A. Idohou, S. A. Osseni, I. Agani, D. Neumeyer, M. Verelst, R. Mauricot and T. Aminou, Fibers Polym., 2019, 20, 1820–1832 CrossRef CAS.
- Y. Zhou, B. Gao, A. R. Zimmerman, J. Fang, Y. Sun and X. Cao, Chem. Eng. J., 2013, 231, 512–518 CrossRef CAS.
- M. C. Biesinger, Appl. Surf. Sci., 2022, 597, 153681 CrossRef CAS.
- S. Ge, S. Wang, W. Mai, K. Zhang, M. Tanveer, L. Wang and C. Tian, Environ. Sci. Pollut. Res., 2023, 30, 66113–66124 CrossRef CAS PubMed.
- L. Zhang, S. C. Jiang and Y. Guan, Environ. Res., 2021, 194, 110667 CrossRef CAS PubMed.
- Y. Yu, W. Liu, Y. Zhang, B. Zhang, Y. Jin, S. Chen, S. Tang, Y. Su, X. Yu and G. Chen, Arabian J. Chem., 2024, 17, 105746 CrossRef CAS.
- H. Kandil, A. E. Abdelhamid, R. M. Moghazy and A. Amin, Polym. Eng. Sci., 2022, 62, 145–159 CrossRef CAS.
- H. Zeng, H. Hao, X. Wang and Z. Shao, Int. J. Biol. Macromol., 2022, 213, 369–380 CrossRef CAS PubMed.
- M.-X. Huo, Y.-L. Jin, Z.-F. Sun, F. Ren, L. Pei and P.-G. Ren, Carbohydr. Polym., 2021, 254, 117473 CrossRef CAS PubMed.
- V. Rizzi, J. Gubitosa, P. Fini, R. Romita, S. Nuzzo, J. A. Gabaldón, M. I. F. Gorbe, T. Gómez-Morte and P. Cosma, J. Environ. Sci. Health, Part A: Toxic/Hazard. Subst. Environ. Eng., 2020, 1–12 Search PubMed.
- A. Kwaśniewska, M. Świetlicki, A. Prószyński and G. Gładyszewski, Polymers, 2021, 13, 4406 CrossRef PubMed.
- J. Wang, X. Sun, H. Zhang, M. Dong, L. Li, H. Zhangsun and L. Wang, Food Hydrocolloids, 2022, 124, 107258 CrossRef CAS.
- Y. S. Ho and G. McKay, Process Biochem., 1999, 34, 451–465 CrossRef CAS.
- E. D. Revellame, D. L. Fortela, W. Sharp, R. Hernandez and M. E. Zappi, Clean Eng. Technol., 2020, 1, 100032 CrossRef.
- W. J. Weber and J. C. Morris, J. Sanit. Eng. Div., Am. Soc. Civ. Eng., 1963, 89, 31–59 CrossRef.
- G. Y. Boris, A. N. Odogu, L. N. R. Blaise, K. Daouda, N. J. Nsami, N. H. Manga and K. J. Mbadcam, J. Mater. Sci. Chem. Eng., 2023, 11, 75–105 CAS.
- J. Gubitosa, V. Rizzi, P. Fini, S. Nuzzo and P. Cosma, Int. J. Mol. Sci., 2023, 24, 8552 CrossRef CAS PubMed.
- Y. Tan, Y. Kang, W. Wang, X. Lv, B. Wang, Q. Zhang, C. Cui, S. Cui, S. Jiao, G. Pang and S. Feng, Appl. Surf. Sci., 2021, 569, 150970 CrossRef CAS.
- A. Naphtali Odogu, K. Daouda, L. Paul Keilah, G. Agbor Tabi, L. Ngouateu Rene, N. Julius Nsami and K. Josoph Mbadcam, Arabian J. Chem., 2020, 13, 5241–5253 CrossRef.
- Z. Zhang, L. Moghaddam, I. M. O'Hara and W. O. S. Doherty, Chem. Eng. J., 2011, 178, 122–128 CrossRef CAS.
- T. Shahnaz, S. Mohamed Madhar Fazil, V. C. Padmanaban and S. Narayanasamy, Int. J. Biol. Macromol., 2020, 151, 322–332 CrossRef CAS PubMed.
- M. O. Olakunle, A. A. Inyinbor, A. O. Dada and O. S. Bello, Int. J. Sustain. Eng., 2018, 11, 4–15 CrossRef.
- Z. Xiong, H. Zheng, Y. Hu, X. Hu, W. Ding, J. Ma and Y. Li, Sep. Purif. Technol., 2021, 277, 119053 CrossRef CAS.
- V. S. Mane and P. V. Vijay Babu, J. Taiwan Inst. Chem. Eng., 2013, 44, 81–88 CrossRef CAS.
- N. Kaya, Z. Yıldız Uzun, C. Altuncan and H. Uzun, Biomass Convers. Biorefin., 2022, 12, 5293–5315, DOI:10.1007/s13399-021-01856-5.
- I. Toumi, H. Djelad, F. Chouli and A. Benyoucef, J. Inorg. Organomet. Polym., 2022, 32, 112–121 CrossRef CAS.
- M. Li, Z. Wang and B. Li, Desalin. Water Treat., 2016, 57, 16970–16980 CrossRef CAS.
- G. Annadurai, R.-S. Juang and D.-J. Lee, J. Hazard. Mater., 2002, 92, 263–274 CrossRef CAS PubMed.
- Y. Fu and T. Viraraghavan, Adv. Environ. Res., 2002, 7, 239–247 CrossRef CAS.
- G. L. Dotto and L. A. A. Pinto, J. Hazard. Mater., 2011, 187, 164–170 CrossRef CAS PubMed.
- M. R. Islam and M. G. Mostafa, Sustain. Water Resour. Manag., 2022, 8, 52 CrossRef.
- Y. M. Correa-Navarro, J. C. Moreno-Piraján and L. Giraldo, Braz. J. Chem. Eng., 2022, 39, 933–948 CrossRef CAS.
- Y. Dago-Serry, K. N. Maroulas, A. K. Tolkou, N. AbdelAll, A. N. Alodhayb, G. A. Khouqeer and G. Z. Kyzas, J. Mol. Struct., 2024, 1298, 137044 CrossRef CAS.
- Z. Zeng, S. Ye, H. Wu, R. Xiao, G. Zeng, J. Liang, C. Zhang, J. Yu, Y. Fang and B. Song, Sci. Total Environ., 2019, 648, 206–217 CrossRef CAS PubMed.
- S. Dong, Y. Wang, Y. Zhao, X. Zhou and H. Zheng, Water Res., 2017, 126, 433–441 CrossRef CAS PubMed.
- Y. Wang, C. Wang, X. Huang, Q. Zhang, T. Wang and X. Guo, Chemosphere, 2024, 349, 140736 CrossRef CAS PubMed.
- D. Akin Sahbaz, Polymers, 2023, 15, 3246 CrossRef CAS PubMed.
- N. J. Hahn, Can. J. Microbiol., 1966, 12, 725–733 CrossRef CAS PubMed.
- G. Han, Y. Du, Y. Huang, W. Wang, S. Su and B. Liu, Chemosphere, 2022, 289, 133109 CrossRef CAS.
- C. Zhang, L. Meng, Z. Fang, Y. Xu, Y. Zhou, H. Guo, J. Wang, X. Zhao, S. Zang and H. Shen, Molecules, 2024, 29, 4517 CrossRef CAS PubMed.
- Y. Liang, X. Xu, F. Yuan, Y. Lin, Y. Xu, Y. Zhang, D. Chen, W. Wang, H. Hu and J. Z. Ou, Carbon, 2023, 205, 40–53 CrossRef CAS.
- X. Zhang, F. Li, X. Zhao, J. Cao, S. Liu, Y. Zhang, Z. Yuan, X. Huang, C. F. De Hoop, X. Peng and X. Huang, Gels, 2023, 9, 40 CrossRef CAS PubMed.
- L. Qie, W. Chen, X. Xiong, C. Hu, F. Zou, P. Hu and Y. Huang, Adv. Sci., 2015, 2, 1500195, DOI:10.1002/advs.201500195.
- D. Li, H. Zhang, K. Ren, D. Zhu, G. Xiao, M. Vakili, W. Wang and J. Ye, Sep. Purif. Technol., 2025, 361, 131305 CrossRef CAS.
- M. Turk Sekulic, N. Boskovic, A. Slavkovic, J. Garunovic, S. Kolakovic and S. Pap, Process Saf. Environ. Prot., 2019, 125, 50–63 CrossRef CAS.
- S. Mehmood, W. Ahmed, M. Rizwan, J. Bundschuh, A. S. M. Elnahal and W. Li, Sep. Purif. Technol., 2025, 361, 131305 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |








