 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Eggshell waste derived nano-hydroxyapatite/metakaolin composites for bone scaffold applications
Zaid Kareem *ab and
Ersan Eyiler
*ab and
Ersan Eyiler bcd
bcd
aProsthetics and Orthotics Engineering Department, University of Kerbala, 1152, Karbala, Iraq. E-mail: zaltaey88@gmail.com; Tel: +964-772-377-9470
bAdvanced Materials and Nanotechnology Department, Cukurova University, 01250, Adana, Turkey
cDepartment of Chemical Engineering, Cukurova University, 01250, Adana, Turkey
dTissue Engineering Department, Cukurova University, 01250, Adana, Turkey
First published on 17th April 2026
Abstract
This study presents a novel, zero-waste wet chemical precipitation method for synthesizing eggshell-derived nano-hydroxyapatite (HA) reinforced with metakaolin (MK) for potential biomedical applications. Calcium oxide obtained from calcined eggshells served as the calcium source, and MK was incorporated at varying weight ratios (25–100% of HA mass) prior to cold pressing. The composites were characterized using XRD, FTIR, SEM-EDX, and BET analyses to assess phase composition, functional groups, microstructure, and surface area. Results confirmed successful HA formation with rod-like nanocrystals, while increasing MK content reduced crystallinity and crystallite size (37.3–21.1 nm) due to aluminosilicate incorporation. Cold pressing decreased porosity (63–55%), increased bulk density (1.57–1.66 g cm−3), and improved mechanical performance, with compressive strength and diametral tensile strength reaching 16.4 MPa and 11.8 MPa, respectively, within the range of cancellous bone. In vitro bioactivity tests revealed progressive apatite layer formation over 21 days, while antimicrobial assays showed broad-spectrum inhibition against Gram-positive, Gram-negative, and fungal strains. MTT cytotoxicity assays using MC3T3-E1 pre-osteoblasts demonstrated high cell viability (>95%) at ≤12.5 µg mL−1, indicating good biocompatibility. The developed HA/MK composites exhibited promising mechanical, bioactive, and antimicrobial properties, supporting their potential use in bone tissue engineering scaffolds.
1. Introduction
Eggshells are biowaste materials, constituted, structurally speaking, by a three-layered structure, namely the cuticle, the spongeous layer and the lamellar layer.1 The cuticle layer represents the outermost surface, and it consists of several proteins, spongeous and lamellar layers form a matrix constituted by protein fibers bonded to calcite (calcium carbonate) crystals in the proportion of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50.2 The eggshell represents 11% of the total weight of the egg and is composed by calcium carbonate 94%, calcium phosphate 1%, organic matter 4% and magnesium carbonate 1%.3
50.2 The eggshell represents 11% of the total weight of the egg and is composed by calcium carbonate 94%, calcium phosphate 1%, organic matter 4% and magnesium carbonate 1%.3
According to FAO, in 2023, world egg production reached 97 million tons in an increasingly trend from the previous year.4 Consequently, the management of large amounts of eggshell waste annually produced in the world is problematic as generally this material is only disposed at landfills with odor production and microbial growth.5 The utilization of bio-calcium found in eggshells in various applications contributes to reducing the negative impacts of untreated eggshell waste, as well as saving disposal costs in landfills.
Commercially available HAs are expensive due to the use of high purity reagents. On the other hand, HAs derived from natural materials such as bovine bone, fish bone or coral have the advantage that they inherit some properties of the raw materials such as the pore structure, carbonated HA etc.3 In biomaterials fields, eggshells are exploited in HA (Ca10(PO4)6(OH)2) synthesis via wet chemical precipitation. Many researchers have extensively studied the synthesis of HA based on eggshells via wet chemical precipitation in last decade,6–8 and they focused on advancing and optimizing the process of producing HA from eggshells.
Wet chemical precipitation method is common method for nanoscale hydroxyapatite synthesis, and many researchers followed this method in producing HA based eggshells. The wet chemical precipitation method permits chemists to control morphologies, particle size, and incorporating ions into the chemical structure of HA, however all these options aimed to enhance the characteristics of HA. Despite the many positive features of this method, it is still having a limitation presenting in the low yielding (limited for laboratory-scale). In conventional wet chemical precipitation methods for hydroxyapatite synthesis, the overall material yield is often reduced due to multiple washing and filtration steps required to remove residual ions from the reaction medium. Hydroxyapatite particles are typically nanosized and highly dispersed, a portion of the precipitated material may remain suspended in the supernatant and be lost during washing or filtration processes. In addition, the incomplete precipitation and the formation of transient calcium phosphate phases may further reduce the effective recovery of the final HA product. In the present approach, the elimination of conventional washing and filtration steps minimizes the loss of fine particles and enables nearly complete recovery of the precipitated material, thereby improving overall process efficiency.
Literately, several researchers have explored methods to scale up the production of HA from eggshell biowaste, aiming to improve yield and process efficiency. For instance, Muthu et al. optimized HA synthesis using a microwave-assisted wet chemical precipitation method at both laboratory and pilot scales, achieving a 45% conversion of eggshell biowaste into HA with enhanced process speed and energy efficiency.9 Similarly, Agbeboh et al. compared two wet precipitation approaches, revealing that the orthophosphoric acid route produced HA with higher purity, whereas the nitric acid-based method offered finer particle size, improved safety, faster reaction time, and greater environmental sustainability.10 These studies highlight innovative approaches to increasing yield, improving particle characteristics, and ensuring eco-friendly production, which are critical steps toward large-scale application of eggshell-derived HA. Another major challenge in utilizing hydroxyapatite for scaffold fabrication lies in its inherent mechanical limitations, despite its excellent bioactivity. Pure HA is brittle and exhibits low fracture toughness, which restricts its application in load-bearing biomedical implants. To address this issue, recent research has focused on reinforcing HA with complementary materials to enhance its physical and mechanical properties without compromising its biocompatibility. In literature, hydroxyapatite has been widely reinforced with various organic and inorganic materials to enhance its mechanical, thermal, and biological properties. For instance, natural polymers such as chitosan have been incorporated into HA matrices to improve biocompatibility, antibacterial properties, and biodegradability.11,12 Similarly, synthetic polymers like polycaprolactone (PCL) have been used to enhance flexibility and processability, particularly in scaffold fabrication for tissue engineering applications.13 Polylactic acid (PLA) has also been employed as a reinforcing agent due to its excellent biodegradability and suitability for 3D printing and biomedical implants.14,15 In addition to polymeric reinforcements, inorganic materials such as silica have been introduced to HA to improve its mechanical strength, chemical stability, and osteoconductive behavior.11 Furthermore, kaolin, a naturally occurring aluminosilicate clay, has been reported as a cost-effective reinforcement for HA-based composites, offering improved compressive strength and structural stability.16–18 These reinforcements collectively aim to overcome the inherent brittleness and low fracture toughness of pure HA, thereby expanding its potential applications in orthopedic, dental, and tissue engineering fields.
This study aimed to synthesize hydroxyapatite from eggshells using a modified wet chemical precipitation method that ensured zero material loss, as it eliminates conventional waste-generating steps such as washing and filtration for the precipitated nano-HA. In addition, the developed process was utilized to produce HA reinforced with calcined kaolin (the common term is metakaolin), creating a composite material suitable for cold pressing and molding. This approach not only maximizes material yield and sustainability but also enhances the mechanical properties of HA, making the composite promising for potential biomedical applications.
2. Materials and method
2.1 Materials
The eggshells were collected from domestic waste. Ortho-phosphoric acid solution (85.5% purity) and sodium hydroxide pellets (EMSURE®, assay ≥99%) were supplied from Sigma Aldrich. Calcined kaolin or metakaolin (MK) (KAOCAL ® 80 Calcined kaolin) was delivered from AVS mineral company, Türkiye.2.2 Preparation of calcined kaolin reinforced hydroxyapatite composites from eggshells
Eggshells were collected, cleaned with tap water and brush, followed by a water-boiling process for 1 h. Then they were dried in oven for 2 h at 150 °C. Grinding process was applied to the dried eggshells for 5 min using ordinary grinding machine (200 g capacity and 1000 W power) to obtain eggshell powder. Next, it was transferred to muffle furnace for decarbonization by heat treatment at 900 °C for 3 h. The resultant ash was soaked in distilled water and stirred (magnetically) for the hydrating process, then the water was spelt out, and the rest powder was dried at 100 °C overnight. The calcined eggshell powder (CES) was grinded again for 5 min and sieved using 75 µm sieve.Nanoscale hydroxyapatite was synthesized using eggshell-derived calcium as the primary calcium source. 0.4 L of H3PO4 solution of 0.67 M was added (10 drops per minute) under magnetic stirring (fixed at 700 rpm) to solution of 1 M of Ca(OH)2, prepared by dissolving 29.6 g in 0.4 L distilled water, due to the limited solubility of Ca(OH)2, the system represents a suspension rather than a completely dissolved solution. 0.05 L of NaOH solution of 1 M was used as a buffer solution to adjust pH of the solution. After the dropwise addition of the H3PO4 solution was completed (about 6 h), the mixture was stirred continuously for an additional 6 h, and then the suspension was left to precipitate overnight. From the experiment, it was concluded that the selected conditions and concentrations yielded 45 g of HA. Subsequently, MK was added to the HA suspension in four proportions (25%, 50%, 75%, and 100% of the synthesized HA mass (i.e., 45 g)) and stirred for 6 h. Then, the suspension was poured into shallow dish to increase surface area and dried under vacuum at 60 °C and 10 mbar. Then, the powder was dried again in oven overnight at 100 °C. The oven dried powder (OD) was grinded again for 1 min and mixed with water at 10% by mass of OD. The wet mixture was put into stainless steel mold for CP under 10 MPa, 150 °C and 30 min (holding time) (Fig. 1). A mold with a diameter of 1.2 cm was used, and the height of each sample was controlled by adjusting the weight of each batch prepared for cold pressing process. Cylindrical samples of 1.2 cm in diameter and 2.4 cm in height were produced for compressive strength test, while cylindrical discs of 1.2 cm in diameter and 0.6 cm in height were used for other mechanical, physical and biological tests.
2.3 Characterization
The Vickers hardness of HA/MK composites was measured in accordance with ASTM E384 using a Vickers microhardness tester (HMV-2, Shimadzu, Japan) with a specified load and dwell time. Hardness values were calculated using:
| VHN = 1.854(P/d2) | (1) |
Diametral tensile strength (DTS) was determined using a universal testing machine (Instron 3369, Instron Corp., USA) at a crosshead speed of 1 mm min−1. DTS values were calculated from:
| DTS = 2P/πDt | (2) |
Porosity percentage was calculated from the eqn (3):
 | (3) |
Theoretical density of the composite was calculated using the rule of mixtures, which considers the contribution of each constituent based on its weight fraction. The eqn (4) was applied:
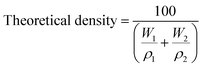 | (4) |
The densities used in the calculation were 2.50 g cm−3 for MK, 3.16 g cm−3 for HA. Bulk density was determined using Archimedes' principle by measuring the dry and submerged weights of the samples with a density tester. The values were then used in the eqn (5):
 | (5) |
Biodegradability studies were conducted to assess the gradual weight loss of the samples in a phosphate buffer solution (PBS, pH 7.4) for 7, 14 and 21 days. Each pellet was immersed in 10 mL of PBS solution and kept at RT in closed container. Readings were taken for the pH variation and weight loss of each respective pellet before and after the incubation using a pH meter and an analytical balance (accuracy: 0.001 g). The samples were then dried in oven at 100 °C for 3 h before weight recording. The percentage of weight decrease (Wd) was calculated using the eqn (6):
 | (6) |
Cytotoxicity was assessed via MTT assay on MC3T3-E1 pre-osteoblastic mouse cells; MC3T3-E1 cells were obtained from the Iranian Biological Resource Center (IBRC). Cells were cultured in α-MEM with 10% FBS and 1% penicillin/streptomycin at 37 °C, 5% CO2. They were seeded (5000–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cells per well) in 96-well plates and incubated for 24 h. Serial dilutions of the material were applied for 48 h, followed by MTT addition (5 mg mL−1). Formazan was dissolved in DMSO, and absorbance was read at 570 nm to determine cell viability (%). The antimicrobial activity of the samples was evaluated using the agar well diffusion method against Staphylococcus aureus, Staphylococcus epidermidis (Gram-positive), Escherichia coli, Pseudomonas aeruginosa (Gram-negative), and Candida albicans (fungal model). Microbial suspensions were prepared from fresh cultures, standardized to a 0.5 McFarland turbidity standard (1.5 × 108 CFU mL−1) for reproducibility, and inoculated (0.1 mL) onto Mueller–Hinton agar plates to achieve uniform lawn growth. After 10–15 min equilibration, four 5 mm wells were aseptically made per plate, and 50 µL of the sterile-prepared samples at concentrations of 1000, 500, 250, and 125 µg mL−1 were loaded into the wells. Plates were incubated at 37 °C for 18–24 h for bacterial strains and up to 48 h for C. albicans. Inhibition zones were measured in millimeters using a digital caliper to quantify antimicrobial activity.
000 cells per well) in 96-well plates and incubated for 24 h. Serial dilutions of the material were applied for 48 h, followed by MTT addition (5 mg mL−1). Formazan was dissolved in DMSO, and absorbance was read at 570 nm to determine cell viability (%). The antimicrobial activity of the samples was evaluated using the agar well diffusion method against Staphylococcus aureus, Staphylococcus epidermidis (Gram-positive), Escherichia coli, Pseudomonas aeruginosa (Gram-negative), and Candida albicans (fungal model). Microbial suspensions were prepared from fresh cultures, standardized to a 0.5 McFarland turbidity standard (1.5 × 108 CFU mL−1) for reproducibility, and inoculated (0.1 mL) onto Mueller–Hinton agar plates to achieve uniform lawn growth. After 10–15 min equilibration, four 5 mm wells were aseptically made per plate, and 50 µL of the sterile-prepared samples at concentrations of 1000, 500, 250, and 125 µg mL−1 were loaded into the wells. Plates were incubated at 37 °C for 18–24 h for bacterial strains and up to 48 h for C. albicans. Inhibition zones were measured in millimeters using a digital caliper to quantify antimicrobial activity.
2.4 ANOVA statistical analysis
The compressive strength, DTS, hardness and Young's modulus were performed in triplicate. Data are presented as mean ± standard deviation. Statistical significance between groups was assessed using one-way ANOVA using Minitab software (Minitab® 22.1). A p-value ≤0.05 was considered statistically significant.3. Results and discussion
3.1 X-ray diffraction
X-ray diffraction patterns for both oven dried (OD) and (CP) HA/MK composites with varying MK contents (25%, 50%, 75%, and 100%) were analyzed in comparison with reference reacted precursor; calcined eggshell powder (CES), MK, and HA (Fig. 2). The XRD pattern of CES exhibited characteristic calcite peaks primarily at 2θ = 29.4°, 39.5°, and 47°, confirming the crystalline nature of the CES.20–22 The HA showed sharp, well-defined characteristic peaks at approximately 2θ = 25.9°, 31.8°, 32.9°, 34.0°, and 49.5°, corresponding to planes (002), (211), (300), (202), and (213), respectively, matching the standard hexagonal hydroxyapatite (JCPDS 09-0432). The XRD pattern of pure MK demonstrated a broad amorphous halo between 15° and 35°, consistent with its disordered or amorphous aluminosilicate structure following calcination.23–25 Other estimated phases are amorphous zeolitic phases, which appears as a broad, diffuse peak or halo instead of the sharp, distinct peaks characteristic of a crystalline material.26,27 All composite samples showed characteristics, indicating the integrity of HA and confirming the successful HA formation regardless of MK incorporation levels. However, an increase in MK content led to decreased peak intensity and increased peak broadening in both before and after cold pressing, indicating reduced crystallinity. However, this reduction in crystal growth may result from structural disorder or inhibited crystal growth due to the presence of amorphous MK phases or the incorporation of MK oxides into the chemical structure of HA. Many papers have reported that doping process led to a reduction in HA crystallinity.28,29 Studies on Si- and Al-doped HA have indicated that Al tends to substitute for Ca in the HA lattice, whereas Si partially replaces phosphate (PO4)3− groups upon incorporation.30–32 However, Si doping improved the biodegradability, resorbability and mineralization ability of HA, which can enhance bone scaffold performance by facilitating faster apatite formation and promoting improved bone tissue integration.31,33 Meanwhile, Al enhanced its thermal stability, contributing to improved structural stability of the bone scaffold.34The crystallite size of the HA phase was estimated using the Scherrer equation, based on the prominent diffraction peak at 2θ ≈ 31.8°, attributed to the (211) plane:
D = Kλ/β![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ θ
| (7) |
The calculated crystallite sizes are presented in Table 1.
| Sample composition | Crystallite size (nm) of OD | Crystallite size (nm) of CP |
|---|---|---|
| 25% MK | 36.7 | 37.3 |
| 50% MK | 31.1 | 30.8 |
| 75% MK | 25.3 | 26.0 |
| 100% MK | 21.3 | 21.1 |
The crystallite size decreased significantly from approximately 36.7 nm and 37.3 nm for the 25% MK composite of OD and CP, respectively, to about 21.3 nm and 21.1 nm for the 100% MK composite of OD and CP, respectively, and this decrease in crystallite size confirms that increased MK content significantly impacts HA crystal growth, resulting in reduced crystallinity and enhanced structural disorder. The presence of the amorphous aluminosilicate phase (MK) appears to disrupt crystallization processes, causing nucleation and growth constraints. In the comparison between OD and CP samples, cold pressing had no significant effect on the crystallite size of HA. This outcome may be attributed to the low processing temperature, which was insufficient to induce any noticeable changes in the HA structure.
3.2 Fourier transform infrared spectroscopy
FTIR spectroscopy was employed to HA/MK composites of both OD and CP, HA, CES, and MK (Fig. 3). The FTIR spectrum of HA exhibits characteristic phosphate (PO43−) vibrational bands prominently at approximately 560–610 cm−1 (bending mode, ν4),35,36 960 cm−1 (symmetrical stretching mode, ν1),36 and between 1000–1100 cm−1 (asymmetrical stretching mode, ν3).35,36 Additionally, a clear hydroxyl (OH−) absorption peak near 3570 cm−1 confirms the presence of structural hydroxyl groups characteristic of stoichiometric HA.10,22 The calcined eggshell spectrum shows distinctive carbonate bands, primarily observed at approximately 1420–1480 cm−1, corresponding to the carbonate group (CO32−),22,35 and confirming residual calcium carbonate phases. The absorption peak appears at 3450 to 3600 cm−1, indicating to O–H stretching vibrations that confirm the formation of Ca(OH)2.36,37 The MK spectrum shows characteristic Si–O–Si and Al–O–Si vibrational bands in the range of 1000–1200 cm−1,38,39 and O–H stretching vibrations appear as broad absorption peaks around 3450–3600 cm−1,23,40 indicating hydroxyl groups bonded to the aluminosilicate structure. For the composite samples (25%, 50%, 75%, and 100% MK), the FTIR spectra reveals a combination of HA and MK characteristics. At lower MK contents (25–50%), prominent HA bands at around 560–610 cm−1, 960 cm−1, and 1000–1100 cm−1 remain well-defined, indicating the preservation of crystalline HA structure. The carbonate band at 1420–1480 cm−1 is still present, but significantly reduced, suggesting minor carbonate substitution within the HA lattice or incomplete carbonate removal. As MK content increases (75–100%), phosphate bands become noticeably broader and reduced in intensity, indicative of reduced crystallinity and increased amorphous character, which aligns well with XRD results. Furthermore, the intensity of the broad Si–O–Si/Al–O–Si vibration between 1000–1200 cm−1 becomes more prominent, indicating increased MK presence and contribution. The broadening in the O–H absorption between 3400–3600 cm−1 suggests increased structural disorder and hydration interactions, likely related to aluminosilicate interactions or adsorbed moisture. Overall, FTIR results corroborate the XRD analysis, confirming that HA crystallinity decreases with higher MK incorporation, leading to increased structural disorder and a more amorphous composite structure. The presence of aluminosilicate-related vibrations demonstrates chemical integration between Ca with Si or Al. Previous researchers confirmed the Ca replacement with Al or Si when HA was doped with silica and alumina oxides.30,34,41 Al or Si doped HA was primarily characterized by enhanced physicochemical, structural, and biological properties, making it superior to pure HA for biomedical applications.31,323.3 SEM-EDX
The microstructures of HA, MK and CES are shown in Fig. 4. The image revealed the formation of nanoscale HA particles with rod-like morphology. It was reported that rods like HA was synthesized successfully from eggshell.42,43 The approximated dimensions of nano-rod hydroxyapatite ranged between 150-200 nm in length and 30–50 nm in width. The pores area clearly appeared in 200 nm magnifications indicative of a large surface area.44The microstructures of MK/HA composites (OD and CP) for the four percentages (25%, 50%, 75%, and 100%) are present in Fig. 5 and 6. The images confirmed the stability of rod like HA in the four percentages even after CP. Metakaolin appeared as a microscale plate particles covered by the nanoparticles of HA. The pores between the metakaolin particles looked big in size and devour HA nanoparticles, meanwhile the pore got narrower for counterpart sample after CP, see Fig. 6. In comparison with MK microstructure shown in Fig. 4, there was disintegration in MK particle size into smaller size, and it was confirmed that the MK was disintegrated under alkaline conditions.45 The CES particles, which appeared as polygonal shapes, were clearly observed in the HA and HA/MK microstructure images, confirming that all CES was consumed during the HA synthesis process. Signs of zeolite grain initiation appeared as bright clusters of particles.
The EDX results of HA and HA/MK composites are shown in Fig. 4–6. Regarded to HA Ca/P atomic ratio was 1.86 indicating to Ca rich HA.46 The sodium of buffered solution appeared in HA EDX result. Upon the incorporation of metakaolin, new peaks corresponding to (Al) and (Si) appeared. As the MK content increased from 25% to 100%, the intensity of the Al and Si peaks progressively increased, while the Ca and P peaks decreased, reflecting the gradual replacement of the HA phase by MK or MK oxides incorporation in crystal structure of HA. It has been reported that Si was replaced by phosphate,47 while Al was replaced by Ca.48,49 At higher MK concentrations, especially at 75% and 100%, the spectra were dominated by aluminosilicate components, these observations demonstrated the effective integration of MK into the HA structure and the compositional shift in the hybrid material system. The Ca/P atomic ratios of 25%MK, 50%MK, 75%MK and 100%MK of OD and CP samples are shown in Table 2.
| Samples | Ca/P for samples of OD | Ca/P for samples of CP |
|---|---|---|
| 25%MK | 1.71 | 1.69 |
| 50%MK | 1.65 | 1.61 |
| 75%MK | 1.52 | 1.47 |
| 100%NK | 1.41 | 1.37 |
3.4 BET
The crystallite size of (HA/MK) composites was estimated via the Scherrer equation based on the (211) diffraction peak centered at approximately 2θ ≈ 31.8°, which is characteristic of the HA phase. As the MK content increased from 25% to 100%, the calculated crystallite size showed a progressive reduction, decreasing from approximately 37.3 nm in the 25%MK sample to around 21.1 nm in the 100%MK composite. This trend suggested that the incorporation of MK, an amorphous aluminosilicate phase, inhibited the long-range ordering of the HA crystal lattice and disrupted crystal growth, resulting in smaller nanocrystalline domains.30–34 The disruption was likely due to the physical presence of MK particles interfering with HA nucleation and the chemical complexity introduced by silicate and aluminate species during synthesis.This decrease in crystallite size was accompanied by a concurrent reduction in BET surface area, total pore volume, and mean pore diameter, particularly in the (CP) samples (Fig. S1–S3 and Table 3). While smaller crystallites typically led to higher surface areas, the inverse trend observed in this study may be explained by two main factors. First, the nanoscale HA particles may partially occupy the pores of the metakaolin matrix, thereby reducing the accessible pore space available for nitrogen adsorption. Second, and more importantly, the cold pressing process significantly modifies the pore structure of the composites.
| Samples | Surface area (m2 g−1) | Total pore volume (cm3 g−1) | Mean pore diameter (nm) |
|---|---|---|---|
| MK | 7.94 | 0.053 | 26.88 |
| HA | 75.32 | 0.602 | 31.96 |
| 25%MK-OD | 67.54 | 0.492 | 30.93 |
| 25%MK-CP | 62.37 | 0.281 | 24.88 |
| 50%MK-OD | 64.32 | 0.432 | 29.08 |
| 50%MK-CP | 57.42 | 0.253 | 24.19 |
| 75%MK-OD | 56.13 | 0.335 | 27.57 |
| 75%MK-CP | 44.23 | 0.224 | 23.62 |
| 100%MK-OD | 53.11 | 0.281 | 24.54 |
| 100%MK-CP | 41.35 | 0.219 | 22.04 |
During cold pressing, the applied pressure brings particles into closer contact, increasing particle packing and deforming contact points between grains. This process promotes densification and can partially collapse or occlude interconnected pores that would otherwise be accessible during nitrogen adsorption measurements. As a result, a portion of the internal porosity becomes inaccessible to nitrogen molecules during BET analysis, leading to a reduction in the measured surface area despite the smaller crystallite size.27,50 The BET surface area decreased from approximately 61 m2 g−1 in 25%MK-CP to 42 m2 g−1 in the 100%MK-CP samples. Similarly, total pore volume declined from 0.28 cm3 g−1 to 0.22 cm3 g−1, and mean pore diameter decreased from 25 nm to about 22 nm over the same composition range. These results indicated that the densification, achieved through cold pressing-along with the inherent packing behavior of fine MK particles, effectively reduced the number and size of accessible pores, thereby limiting the available surface area for nitrogen adsorption during BET analysis.51 Therefore, although the crystallite size decreased with increasing MK content, the overall accessible surface area was reduced due to pore occlusion and structural densification. This densification likely contributed to the enhanced mechanical performance observed in parallel to results.
3.5 Mechanical and physical properties of the composites
The mechanical and physical characterizations were conducted only on the CP samples, as the OD samples were prepared solely to compare the effect of the cold-pressing conditions on HA integrity.| Tests | 25%MK | 50%MK | 75%MK | 100%MK | p-Value |
|---|---|---|---|---|---|
| Compressive strength (MPa) | 10.4 ± 2.7 | 13.7 ± 1.6 | 14.9 ± 1.6 | 16.4 ± 1.3 | 0.016 |
| DTS (MPa) | 6.1 ± 1.2 | 7.5 ± 1.7 | 10.3 ± 1.7 | 11.8 ± 1.8 | 0.034 |
| Hardness (Hv) | 235 ± 25 | 235 ± 22 | 214 ± 22 | 204 ± 36 | 0.609 |
| Young's modulus (GPa) | 5.0 ± 0.29 | 5.0 ± 0.16 | 5.2 ± 0.14 | 5.6 ± 0.20 | 0.050 |
Similarly, DTS values demonstrated a positive correlation with MK content, increasing from 6.1 MPa to 11.8 MPa. The rise in tensile strength suggested that the MK matrix formed at higher ratios exhibited superior internal bonding and resistance to tensile stress, reinforcing the mechanical integrity of the composites. This improvement in tensile properties is beneficial for bone scaffold applications, as it helps the scaffold withstand physiological stresses and maintain structural stability during bone regeneration. This result is consistent with that of Ibrahim et al., who studied the effect of metakaolin alkaline activation on HA scaffolds.53
The Vickers hardness values remained relatively stable across all formulations, ranging between 204 and 235 Hv. The minimal variation implied that while the bulk matrix densification increased with MK content, surface hardness was less sensitive to MK ratio under Vickers indentation. This may have resulted from the localized nature of the indentation test and possible surface heterogeneities or residual porosity. The hardness value observed in this study was higher than those reported previously.54 Hardness values referred to surface resistance to deformation, which is beneficial for maintaining the structural integrity of bone scaffolds during implantation and service in physiological environments.
Young's modulus, representing the stiffness of the composite, also followed a similar upward trajectory. The Young's modulus values were 5.0, 5.0, 5.2, and 5.6 GPa for the samples containing 25%, 50%, 75%, and 100% MK, respectively, indicating a progressive improvement in elastic behavior with increasing MK content. The increase in modulus reflected improved load transfer across the composite and enhanced rigidity, likely resulting from the integration of amorphous metakaolin, which acted as a stiffening agent. The Young's modulus of cancellous bone ranges from 1.0 to 22.3 GPa.55
Lastly, ANOVA results indicated that the compressive strength, DTS and Young's modulus are significantly increased with increasing metakaolin content (p ≤ 0.05), between 25%MK and 100%MK. Meanwhile, hardness was not statistically significant (p ≥ 0.05).
As the MK content increased to 50% and 75%, porosity declined gradually to approximately 61% and 58%, respectively, while the bulk density increased to 1.59 and 1.61 g cm−3. This improvement can be attributed to the filler effect of metakaolin, whose finer particles can occupy interstitial spaces within the HA matrix, thus enhancing packing efficiency and reducing voids. At 100% MK, the composite reached the lowest porosity (55%) and highest bulk density (1.66 g cm−3), demonstrating the dominant densifying effect of MK when used as the primary solid phase in the composite.
The increasing bulk density with concurrent reduction in porosity indicates more effective compaction and microstructural consolidation with higher MK content under cold pressing. These structural changes were highly favorable for improving the composite's mechanical properties, as lower porosity typically correlated with greater compressive strength and stiffness. It has been reported that the desired porosity range for cortical bone is 50–80%.56
3.6 Biocompatibility and biofunctionality
Based on the results obtained from characterization analyses, mechanical testing, and physical evaluations, the 25% MK sample was selected for further biological and bioactivity assessments to ensure the structural and compositional integrity of HA. This decision was made because samples with 50% MK or higher showed a reduction in porosity and a compromised HA integrity, likely due to ion incorporation or changes in crystallinity. | ||
| Fig. 10 Antimicrobial inhibition zones of the 25%MK scaffold against Candida albicans, Staphylococcus epidermidis, Staphylococcus aureus, Escherichia coli, and Pseudomonas aeruginosa. | ||
| Isolates | 1000 µg mL−1 | 500 µg mL−1 | 250 µg mL−1 | 125 µg mL−1 |
|---|---|---|---|---|
| Candida albicans | 27 | 26 | 24 | 23 |
| Staphylococcus epidermidis | 29 | 25 | 23 | 22 |
| Staphylococcus aureus | 27 | 23 | 23 | 22 |
| Escherichia coli | 29 | 26 | 24 | 23 |
| Pseudomonas aeruginosa | 26 | 25 | 23 | 23 |
 | ||
| Fig. 11 MTT assay results of 25%MK at different concentrations after 48 h incubation with MC3T3-E1 cells. | ||
The overall performance of the HA/MK composites was governed by the interaction between HA nanocrystals and the amorphous aluminosilicate phase of metakaolin. Structural analyses (XRD and FTIR) revealed that increasing MK content reduced HA crystallinity and crystallite size, while SEM observations confirmed the formation of a hybrid microstructure where rod-like HA nanoparticles were distributed over plate-like MK particles. Cold pressing enhanced particle packing and reduced pore size, which led to decreased porosity and increased bulk density. These microstructural changes improved load transfer within the composite, resulting in higher compressive strength, tensile strength, and Young's modulus. Such mechanical properties fall within the range of cancellous bone, indicating that the scaffold can provide sufficient structural support during bone regeneration. At the same time, the presence of nanoscale HA and interconnected porosity supports biological performance by promoting apatite formation in simulated body fluid, enabling cell attachment and proliferation, and facilitating nutrient transport. The observed antimicrobial activity further enhances the scaffold's functionality by reducing the risk of post-implantation infection. Therefore, the balance between MK-induced structural densification and HA bioactivity provides a combination of mechanical stability, controlled biodegradation, and biological functionality, which are essential characteristics for bone tissue engineering scaffolds.
4. Conclusions
This study successfully developed a sustainable, zero-waste wet chemical precipitation method to synthesize nano-hydroxyapatite from eggshell biowaste and reinforced it with calcined metakaolin for potential biomedical applications. The elimination of conventional washing and filtration steps significantly enhanced material yield and process efficiency, as these steps can lead to partial loss of nanoparticles due to their nanoscale dimensions and high colloidal stability. Structural analyses (XRD, FTIR, SEM-EDX) confirmed the formation of HA with rod-like nanocrystals and effective incorporation of MK, which resulted in reduced crystallinity and crystallite size due to aluminosilicate interactions. Cold pressing improved bulk density, reduced porosity, and enhanced mechanical properties, with compressive strength (16.4 MPa) and diametral tensile strength (11.8 MPa) values within the cancellous bone range. Bioactivity tests demonstrated progressive apatite layer formation on the composite surface in SBF, while antimicrobial evaluations revealed broad-spectrum activity against Gram-positive, Gram-negative, and fungal strains. Cytotoxicity assays indicated excellent biocompatibility, with cell viability exceeding 95% at concentrations up to 12.5 µg mL−1. Overall, the developed HA/MK composite exhibits a unique combination of mechanical strength, bioactivity, antimicrobial functionality, and cytocompatibility, supporting its potential as a promising candidate for bone tissue engineering scaffolds and other biomedical applications. Future studies should focus on in vivo performance and optimization of degradation rates to further validate its clinical applicability.Conflicts of interest
There are no conflict to declare.Data availability
The data supporting the findings of this study are included within the manuscript and the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6ra01169a.Acknowledgements
This work was supported by the Scientific Research Projects (BAP) Coordination Unit at Cukurova University [grant numbers FDK-2023-16321].References
- M. T. Hincke, Y. Nys, J. Gautron, K. Mann, A. B. Rodriguez-Navarro and M. D. McKee, Front. Biosci., 2012, 17, 1266–1280 CrossRef CAS PubMed.
- G. Daculsi, N. Passuti, S. Martin, C. Deudon, R. Legeros and S. Raher, J. Biomed. Mater. Res., 1990, 24, 379–396 CrossRef CAS PubMed.
- E. M. Rivera, M. Araiza, W. Brostow, V. M. Castano, J. Dıaz-Estrada, R. Hernández and J. R. Rodrıguez, Mater. Lett., 1999, 41, 128–134 CrossRef CAS.
- FAO, World Food and Agriculture-Statistical Yearbook 2023, 2023 Search PubMed.
- S. Mignardi, L. Archilletti, L. Medeghini and C. De Vito, Sci. Rep., 2020, 10, 2436 CrossRef CAS PubMed.
- O. M. Gomez-Vazquez, B. A. Correa-Piña, L. F. Zubieta-Otero, A. M. Castillo-Paz, S. M. Londoño-Restrepo and M. E. Rodriguez-García, Ceram. Int., 2021, 47, 32775–32785 CrossRef CAS.
- P. Kamalanathan, S. Ramesh, L. T. Bang, A. Niakan, C. Y. Tan, J. Purbolaksono, H. Chandran and W. D. Teng, Ceram. Int., 2014, 40, 16349–16359 CrossRef CAS.
- S. Ummartyotin and B. Tangnorawich, Colloid Polym. Sci., 2015, 293, 2477–2483 CrossRef CAS.
- D. Muthu, G. S. Kumar, V. Kattimani, V. Viswabaskaran and E. Girija, Ceram. Int., 2020, 46, 25024–25034 CrossRef CAS.
- N. Agbeboh, I. Oladele, O. Daramola, A. Akinwekomi, M. Tanimola and O. Olasukanmi, Futa J. Eng. Eng. Technol, 2022, 16, 95–104 CrossRef.
- R. Adamski and D. Siuta, Molecules, 2021, 26, 1976 CrossRef CAS PubMed.
- A. S. Pádua, L. Figueiredo, J. C. Silva and J. P. Borges, Prog. Biomater., 2023, 12, 137–153 CrossRef PubMed.
- N. Rezania, M. Asadi-Eydivand, N. Abolfathi, S. Bonakdar, M. Mehrjoo and M. Solati-Hashjin, J. Mater. Sci.: Mater. Med., 2022, 33, 31 CrossRef CAS PubMed.
- E. Pérez, J. Mater. Sci., 2021, 56, 19915–19935 CrossRef.
- H.-S. Ko, S. Lee, D. Lee and J. Y. Jho, Nanomaterials, 2021, 11, 249 CrossRef CAS PubMed.
- P. Satish, K. Hadagalli, L. L. Praveen, M. S. Nowl, A. H. Seikh, I. A. Alnaser, H. S. Abdo and S. Mandal, Inorganics, 2023, 11, 427 CrossRef CAS.
- D. Obada, E. T. Dauda, J. Abifarin, N. D. Bansod and D. Dodoo Arhin, Mater. Today: Proc., 2020, 38, 2295–2300 Search PubMed.
- D. O. Obada, S. A. Osseni, H. Sina, K. A. Salami, A. N. Oyedeji, D. Dodoo-Arhin, N. D. Bansod, S. Csaki, A. Y. Atta, O. O. Fasanya and A. R. Sowunmi, Appl. Clay Sci., 2021, 215, 106298 CrossRef CAS.
- T. Kokubo and H. Takadama, Biomaterials, 2006, 27, 2907–2915 CrossRef CAS PubMed.
- H. Khandelwal and S. Prakash, J. Miner. Mater. Char. Eng., 2016, 4, 119–126 CAS.
- O. Agbabiaka, I. Oladele, A. Akinwekomi, A. Adediran, A. O. Balogun, O. Olasunkanm and T. Olayanju, Sci. Afr., 2020, 8, e00452 CAS.
- N. M. Pu'ad, J. Alipal, H. Abdullah, M. Idris and T. Lee, Mater. Today: Proc., 2021, 42, 172–177 Search PubMed.
- H. K. Tchakouté, S. J. Melele, A. T. Djamen, C. R. Kaze, E. Kamseu, C. N. Nanseu, C. Leonelli and C. H. Rüscher, Appl. Clay Sci., 2020, 186, 105448 CrossRef.
- Z. Khaled, A. Mohsen, A. Soltan and M. Kohail, Ain Shams Eng. J., 2023, 14, 102142 CrossRef.
- J. Davidovits and J. Ceram, Sci. Technol., 2017, 8, 335–350 Search PubMed.
- A. J. Sudagar, S. Andrejkovičová, F. Rocha, C. Patinha, M. R. Soares, A. L. Velosa and E. F. d. Silva, Minerals, 2021, 11, 486 CrossRef CAS.
- L. Li, F. Yang, C. Liu, Z. Chen, J. Ge and Z. Hou, Ceram. Int., 2025, 51, 43311–43318 CrossRef CAS.
- N. C. Reger, A. K. Bhargava, I. Ratha, B. Kundu and V. K. Balla, Ceram. Int., 2019, 45, 252–263 CrossRef CAS.
- T. Baskaran, N. F. Mohammad, S. S. Md Saleh, N. F. Mohd Nasir and F. D. Mohd Daud, Synthesis methods of doped hydroxyapatite: a brief review, J. Phys.: Conf. Ser., 2021, 012008 CrossRef.
- J. Xu and K. A. Khor, J. Inorg. Biochem., 2007, 101, 187–195 CrossRef CAS PubMed.
- M. Tavafoghi, J. M. Kinsella, C. G. Gamys, M. Gosselin and Y. F. Zhao, Ceram. Int., 2018, 44, 17612–17622 CrossRef CAS.
- M. Wang, L. Wang, C. Shi, T. Sun, Y. Zeng and Y. Zhu, Phys. Chem. Chem. Phys., 2016, 18, 21789–21796 RSC.
- N.-I. Farkas, G. L. Turdean, L. Bizo, L. Marincaş, O. Cadar, L. Barbu-Tudoran and B. Réka, Ceram. Int., 2023, 49, 25156–25169 CrossRef CAS.
- M. Goldberg, P. Protsenko, V. Smirnov, O. Antonova, S. Smirnov, A. Konovalov, K. Vorckachev, E. Kudryavtsev, S. Barinov and V. Komlev, J. Mater. Res. Technol., 2020, 9, 76–88 CrossRef CAS.
- M. Khalid, S. S. B. Jikan, S. Adzila, Z. Murni, N. Badarulzaman, R. Rosley and M. U. Hameed, Biointerface Res. Appl. Chem., 2022, 12, 5663–5671 CAS.
- S. D. Lala, E. Barua, P. Deb and A. B. Deoghare, Mater. Today Commun., 2021, 27, 102443 CrossRef.
- K. W. Goh, Y. H. Wong, S. Ramesh, H. Chandran, S. Krishnasamy, A. Sidhu and W. Teng, Ceram. Int., 2021, 47, 8879–8887 CrossRef CAS.
- S. Onutai, T. Osugi and T. Sone, Materials, 2023, 16, 985 CrossRef CAS PubMed.
- S. Onutai, J. Sato and T. Osugi, J. Solid State Chem., 2023, 319, 123808 CrossRef CAS.
- R. A. Caciatori, A. G. Dal-Bó and A. M. Bernardin, Miner. Eng., 2024, 219, 109068 CrossRef CAS.
- K. V. Tian, M. Z. Mahmoud, P. Cozza, S. Licoccia, D.-C. Fang, D. Di Tommaso, G. A. Chass and G. N. Greaves, J. Non-Cryst. Solids, 2016, 451, 138–145 CrossRef CAS.
- M. K. d. S. Horta, F. J. Moura, M. S. Aguilar, C. B. Westin, J. B. d. Campos, S. B. Peripolli, V. S. Ramos, M. I. Navarro and B. S. Archanjo, Mater. Res., 2020, 23, e20200266 CrossRef.
- A.-R. Ibrahim, Y. Zhou, X. Li, L. Chen, Y. Hong, Y. Su, H. Wang and J. Li, Mater. Res. Bull., 2015, 62, 132–141 CrossRef CAS.
- M. Trzaskowska, V. Vivcharenko, A. Benko, W. Franus, T. Goryczka, A. Barylski, K. Palka and A. Przekora, Sci. Rep., 2024, 14, 28137 CrossRef CAS PubMed.
- N. E. N'Guessan, E. Joussein, A. Courtin-Nomade, E. Paineau, M. Soubrand, O. Grauby, V. Robin, C. D. Cristina, D. Vantelon and P. Launois, Appl. Clay Sci., 2021, 205, 106037 CrossRef.
- C. Y. Tan, R. Singh, Y. C. Teh, Y. M. Tan and B. K. Yap, Int. J. Appl. Ceram. Technol., 2015, 12, 223–227 CrossRef CAS.
- A. Aminian, M. Solati-Hashjin, A. Samadikuchaksaraei, F. Bakhshi, F. Gorjipour, A. Farzadi, F. Moztarzadeh and M. Schmücker, Ceram. Int., 2011, 37, 1219–1229 CrossRef CAS.
- M. Wakamura, K. Kandori and T. Ishikawa, Colloids Surf., A, 2000, 164, 297–305 CrossRef CAS.
- Y. Nie, C. Hu and C. Kong, J. Hazard. Mater., 2012, 233, 194–199 CrossRef PubMed.
- S. Grasso, M. Biesuz, L. Zoli, G. Taveri, A. I. Duff, D. Ke, A. Jiang and M. J. Reece, Adv. Appl. Ceram., 2020, 119, 115–143 CrossRef CAS.
- P. Sinha, A. Datar, C. Jeong, X. Deng, Y. G. Chung and L.-C. Lin, J. Phys. Chem. C, 2019, 123, 20195–20209 CrossRef CAS.
- P.-Y. Chen and J. McKittrick, J. Mech. Behav. Biomed. Mater., 2011, 4, 961–973 CrossRef CAS PubMed.
- W. M. A. W. Ibrahim, M. M. A. B. Abdullah, N. H. Jamil, H. Mohamad, M. A. A. M. Salleh, A. V. Sandu, P. Vizureanu, M. S. Baltatu and P. Sukmak, Appl. Sci., 2023, 13, 2643 CrossRef CAS.
- D. Obada, E. Dauda, J. Abifarin, N. Bansod and D. Dodoo-Arhin, Mater. Today: Proc., 2021, 38, 2295–2300 CAS.
- D. Wu, P. Isaksson, S. J. Ferguson and C. Persson, Acta Biomater., 2018, 78, 1–12 CrossRef PubMed.
- V. Balasubramani, R. Jeganathan and S. D. Kumar, Mater. Today: Proc., 2023 Search PubMed , in press.
- N. Twinprai, R. Sutthi, P. Ngaonee, P. Chaikool, T. Sookto, P. Twinprai, Y. Mutoh, P. Chindaprasirt and T. Laonapakul, Arab. J. Chem., 2024, 17, 105878 CrossRef CAS.
- N. Eliaz and N. Metoki, Materials, 2017, 10, 334 CrossRef PubMed.
- J. Cabezas-Pizarro, M. Redondo-Solano, C. Umaña-Gamboa and M. L. Arias-Echandi, Rev. Argent. Microbiol., 2018, 50, 56–61 Search PubMed.
- S. M. Dizaj, F. Lotfipour, M. Barzegar-Jalali, M. H. Zarrintan and K. Adibkia, Mater. Sci. Eng., C, 2014, 44, 278–284 CrossRef CAS PubMed.
- E. Bivehed, B. Hellman, Y. Fan, J. Haglöf and S. Buratovic, Mutat. Res., Genet. Toxicol. Environ. Mutagen., 2023, 891, 503680 CrossRef CAS PubMed.
- M. G. Nurdiana Dewi, D. Gustiono, D. Kurnia and A. Cahyanto, Eur. J. Dent., 2025, 19, 563–579 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |









