Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Synthesis, characterization, in vitro and computational assessment of new xanthene hydrazone derivatives as promising vasorelaxant agents
Mohammed El Mesky *a,
Ismail bouadidb,
Fatimazahra Guerguerc,
Hicham Zguenia,
Yassine Rhazi
*a,
Ismail bouadidb,
Fatimazahra Guerguerc,
Hicham Zguenia,
Yassine Rhazi d,
Mohammed Chalkha
d,
Mohammed Chalkha *ad,
Farid Khalloukib,
Samir Chtita
*ad,
Farid Khalloukib,
Samir Chtita c,
Sahar Abdulaziz AlSedairye,
Mourad A. M. Aboul-Soud*f,
John P. Giesyghi,
Driss Chebabe
c,
Sahar Abdulaziz AlSedairye,
Mourad A. M. Aboul-Soud*f,
John P. Giesyghi,
Driss Chebabe a,
El Houssine Mabroukad and
Mohamed Eddouksb
a,
El Houssine Mabroukad and
Mohamed Eddouksb
aLaboratory of Materials Engineering for the Environment and Natural Resources, Faculty of Sciences and Techniques, Moulay Ismail University of Meknès, B.P. 509, Boutalamine, 52000, Errachidia, Morocco. E-mail: m.elmesky@edu.umi.ac.ma; mohammed.chalkha1@usmba.ac.ma
bTeam of Ethnopharmacology and Pharmacognosy, Faculty of Sciences and Techniques Errachidia, Moulay Ismail University of Meknes, Errachidia, Morocco
cLaboratory of Analytical and Molecular Chemistry, Faculty of Sciences Ben M'Sik, Hassan II University of Casablanca, Casablanca, Morocco
dLaboratory of Engineering of Organometallic, Molecular Materials, Environment, and Innovative Pedagogy (LIMOMEPI), Faculty of Sciences Dhar EL Mahraz, Sidi Mohamed Ben Abdellah University, P.O. Box 1796 (Atlas), 30000 Fez, Morocco
eDepartment of Food Sciences and Nutrition, College of Food and Agricultural Sciences, King Saud University, P.O. Box 2460, Riyadh 11451, Saudi Arabia
fCenter of Excellence in Biotechnology Research (CEBR), College of Applied Medical Sciences, King Saud University, P.O. Box 17 10219, Riyadh 11433, Saudi Arabia. E-mail: maboulsoud@ksu.edu.sa
gDepartment of Veterinary Biomedical Sciences and Toxicology Centre, Western College of Veterinary Medicine, University of Saskatchewan, Saskatoon, SK S7N 5B4, Canada. E-mail: Jgiesy@aol.com
hDepartment of Integrative Biology and Center for Integrative Toxicology, Michigan State University, East Lansing, MI 48824, USA
iDepartment of Environmental Sciences, Baylor University, Waco, 76706, USA
First published on 17th April 2026
Abstract
Functionalized xanthene hydrazone derivatives were synthesized using two distinct pathways. Two routes of synthesis, involving condensation and O-alkylation reactions, were adopted to synthesize the target compounds. The structures of the synthetised intermediates and xanthene hydrazone derivatives were confirmed through high-resolution mass spectrometry (HRMS), 1H-nuclear magnetic resonance (NMR), and 13C-NMR. Vasorelaxation assays indicated that derivatives modified solely by alkylation or condensation remained inactive. It was observed that compounds became active only when both functional groups were present simultaneously, highlighting the importance of the complementarity of the ester and hydrazone in the mechanism of action. Among the derivatives examined, compounds F3 and F4 exhibited the greatest potency as vasorelaxants, with EC50 values of 38.204 and 41.300 µg mL−1 and Emax values of 80.61% and 83.13%, respectively. While F3 and F4 displayed somewhat lower potency than verapamil (EC50 = 18.000 ± 1.00 µg mL−1; Emax = 61.67 ± 4.10%), they demonstrated significantly greater maximal effectiveness. These findings highlighted the great potential of compounds F3 and F4 as new vasorelaxant agents. Results of the in vitro tests on rat aortic rings that were pre-contracted using epinephrine revealed a clear correlation between both the chemical structure and biological activity. The results of molecular docking and molecular dynamics simulations show that F3 and F4 formed stable and robust binding modes with the human L-type calcium channel (CaV1.2). Furthermore, the in silico findings suggest that F3 and F4 possess acceptable ADMET properties, supporting their potential as promising drug candidates. Overall, the results indicate that these functionalized xanthene-hydrazone derivatives represent a promising foundation for the development of novel vasorelaxant agents.
1. Introduction
Blood pressure is essential for adequate perfusion of vital organs. It depends on two main mechanisms: the resistance of blood vessels,1 which depends on the degree of vascular contraction, and cardiac output, which is linked to the frequency and strength of the heartbeat. When blood pressure falls to abnormally low levels, tissues lack oxygen, which can impair organ function. Conversely, chronic hypertension eventually damages the walls of blood vessels and gradually affects the heart, brain, kidneys and even the eyes.Hypertension is defined as persistently elevated blood pressure, according to the WHO. It is a major health problem that affects more than one billion individuals worldwide and leads to nearly 10 million deaths each year.2 It is frequently referred to as the ‘silent killer’ since it usually does not show any warning signs for years. If left undetected and untreated, it can result in severe complications such as heart problems, strokes, kidney disease and retinal damage.3
Antihypertensive drugs are the standard treatment for high blood pressure. They are effective in controlling blood pressure and reducing cardiovascular risks. However, long-term use is often associated with side effects such as palpitations, persistent dry cough, constant fatigue, and even kidney complications. For many patients, these side effects make long-term adherence to treatment difficult.
One of the key mechanisms for combating hypertension is the relaxation of the smooth muscles of the blood vessels. This is why research is focusing on the development of more potent and better-tolerated vasorelaxant agents.4–6 The design of hybrid molecules, which combine several pharmacophores in a single structure, appears particularly promising. The concept is to combine complementary mechanisms capable of acting simultaneously on several biological pathways. By carefully assembling these elements, it is possible to improve the potency, selectivity and tolerance of the treatment, paving the way for a new generation of antihypertensive drugs that are both more effective and safer.7,8
In medicinal chemistry, the xanthene scaffold is considered highly versatile. For years, researchers have been exploring its derivatives, xanthones, fluorescein, rhodamine, with continuous discovery of new properties. These molecules exhibit a wide range of biological activities: antiviral,9 antihyperlipidemic effects,10 antibacterial action,11 anti-inflammatory,12 antimalarial activity,13 antimicrobial,14 anticancer,15 antileukemic,16 free radical scavenging,17 antitumor,18 and even capable of fighting certain cancers or promoting apoptosis. Some, such as certain xanthones and norathyriol, also show interesting potential for regulating blood pressure (Fig. 1).19
Recently, hydrazones and their derivatives have attracted considerable interest. They are relatively simple to synthesize and have a wide range of pharmacological effects.20 They possess a flexible and stable structure capable of forming hydrogen bonds. This explains their broad pharmacological activities: antihypertensive,21 antibacterial,22 antifungal,23 antiviral and even anticancer.24,25 By combining them with other functional groups, hybrid molecules with unique physicochemical properties can be obtained (Fig. 2). Their versatility often allows them to target several mechanisms at once, improving both their efficacy and selectivity. For these reasons, they are considered promising candidates for the design of new multifunctional drugs.26,27
Xanthene derivatives have interesting properties, particularly due to their ester and hydrazone groups. Therefore, we designed and synthesized new functionalised xanthene-based molecules and evaluated their vasorelaxant activity. Two synthesis routes were successfully used, and all compounds, both intermediates and final products, were characterised by NMR spectroscopy and high-resolution mass spectrometry. To investigate their vasorelaxant effects, we tested the compounds step by step, first the intermediates, then the final hybrids. This stepwise approach allowed us to better understand how the order of functionalisation influences the pharmacological behaviour of xanthene-hydrazone compounds. The results obtained provide valuable insights for improving and selecting the most promising candidates as potential vasodilators. In addition to the experiments, we conducted computational studies, including molecular docking and molecular dynamics simulations, to better understand the interactions with biological targets. An ADMET analysis was also performed to assess the therapeutic potential of these molecules.
2. Materials and methods
2.1. Chemistry
Melting points of compounds were determined by use of a Kofler bank. The evolution of the synthesis was determined by thin-layer chromatography (TLC) on silica 60 F254 (E. Merck). NMR spectra were measured on a Bruker Advanced 300 WB at 300 MHz and 600 MHz for solutions in DMSO-d6, with chemical shifts reported in δ ppm relative to tetramethyl silane 7 (TMS) as an internal control. Mass spectra were obtained by using an UltiMate 3000 (Thermo Scientific).2.2. Synthesis of FH and F1–7
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ethyl acetate/hexane combination. Product FH was obtained as a fragile, white solid.
1 ethyl acetate/hexane combination. Product FH was obtained as a fragile, white solid.The yield was 76% of a light off-white solid; 1H NMR (500 MHz, DMSO-d6): δ (ppm) = 4.40 (s, 2H, NH2), 9.02 (s, 2H, 2(–OH)), 7.91–7.85 (m, 1H, ArH), 7.58 (dtd, J = 21.4, 7.4, 1.2 Hz, 2H, ArH), 7.38 (dd, J = 6.8, 3.0 Hz, 2H, ArH), 7.31 (dd, J = 5.1, 1.9 Hz, 3H, ArH), 7.10 (dd, J = 7.4, 1.2 Hz, 1H, ArH), 6.62 (d, J = 2.3 Hz, 2H, ArH), 6.46 (d, J = 8.6 Hz, 2H, ArH), 6.42 (dd, J = 8.6, 2.4 Hz, 2H, ArH); 13C NMR (126 MHz, DMSO-d6): δ (ppm) = 164.17 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 159.13, 152.81, 150.91, 149.80, 135.04, 134.52, 130.89, 129.66, 129.65, 129.36, 128.54, 127.29, 124.36, 123.73, 112.85, 110.75, 103.01 (aromatic carbons), 65.93 (spiro carbon); HRMS (m/z): calculated for C20H14N2O4 345.09356, found [M–H]−: 345.08569.
O), 159.13, 152.81, 150.91, 149.80, 135.04, 134.52, 130.89, 129.66, 129.65, 129.36, 128.54, 127.29, 124.36, 123.73, 112.85, 110.75, 103.01 (aromatic carbons), 65.93 (spiro carbon); HRMS (m/z): calculated for C20H14N2O4 345.09356, found [M–H]−: 345.08569.
The yield was 87%, a yellow solid, m. p.: 168–170 °C, f. r. = 0.52; 1H NMR (500 MHz, DMSO-d6): δ 7.77 (dd, J = 5.7, 3.1 Hz, 1H), 7.47 (dd, J = 5.7, 3.0 Hz, 2H), 7.00–6.94 (m, 1H), 6.78 (d, J = 2.6 Hz, 2H), 6.61 (dd, J = 8.5, 2.6 Hz, 2H), 6.51 (d, J = 8.8 Hz, 2H), 4.78 (s, 4H), 4.43 (s, 2H), 4.14 (q, J = 7.1 Hz, 4H), 1.21–1.15 (m, 6H); 13C NMR (126 MHz, DMSO-d6): δ 169.03, 166.17, 158.87, 152.79, 151.67, 133.32, 129.78, 129.24, 128.65, 123.97, 123.07, 112.86, 112.18, 102.32, 65.43, 64.95, 61.26, 14.57.
2-(Benzylideneamino)-3′,6′-dihydroxyspiro[isoindolin-3-one-1,9′-xanthene] F5 had a yield of 82.9%; m. p.: 188–190 °C, brown. F. r. (Ac![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Hex) (3
Hex) (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) = 0.50; 1H NMR (500 MHz, DMSO-d6) δ 9.84 (s, 2H), 9.02 (s, 1H), 7.91–7.85 (m, 1H), 7.58 (dtd, J = 21.4, 7.4, 1.2 Hz, 2H), 7.38 (dd, J = 6.8, 3.0 Hz, 2H), 7.31 (dd, J = 5.1, 1.9 Hz, 3H), 7.10 (dd, J = 7.4, 1.2 Hz, 1H), 6.62 (d, J = 2.3 Hz, 2H), 6.46 (d, J = 8.6 Hz, 2H), 6.42 (dd, J = 8.6, 2.4 Hz, 2H); 13C NMR (500 MHz, DMSO-d6) δ 164.17, 159.13, 152.81, 150.91, 149.80, 135.04, 134.52, 130.89, 129.66, 129.65, 129.36, 128.54, 127.29, 124.36, 123.73, 112.85, 110.75, 103.01, 65.93; HRMS (m/z): calculated for C27H18N2O4 435.12666, found 435.12768.
2) = 0.50; 1H NMR (500 MHz, DMSO-d6) δ 9.84 (s, 2H), 9.02 (s, 1H), 7.91–7.85 (m, 1H), 7.58 (dtd, J = 21.4, 7.4, 1.2 Hz, 2H), 7.38 (dd, J = 6.8, 3.0 Hz, 2H), 7.31 (dd, J = 5.1, 1.9 Hz, 3H), 7.10 (dd, J = 7.4, 1.2 Hz, 1H), 6.62 (d, J = 2.3 Hz, 2H), 6.46 (d, J = 8.6 Hz, 2H), 6.42 (dd, J = 8.6, 2.4 Hz, 2H); 13C NMR (500 MHz, DMSO-d6) δ 164.17, 159.13, 152.81, 150.91, 149.80, 135.04, 134.52, 130.89, 129.66, 129.65, 129.36, 128.54, 127.29, 124.36, 123.73, 112.85, 110.75, 103.01, 65.93; HRMS (m/z): calculated for C27H18N2O4 435.12666, found 435.12768.
2-((4-Methoxybenzylidene)amino)-3′,6′-dihydroxyspiro[isoindolin-3-one-1,9′-xanthene] F6 had a yield of 85%; m. p: 192–194 °C, light yellow, f. r. (Ac![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Hex) (3
Hex) (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) = 0.60; 1H NMR (500 MHz, DMSO-d6) δ 9.92–9.75 (m, 2H), 8.99 (s, 1H), 7.89–7.84 (m, 1H), 7.57 (dtd, J = 19.0, 7.3, 1.2 Hz, 2H), 7.35–7.32 (m, 2H), 7.08 (d, J = 7.1 Hz, 1H), 6.88 (d, J = 1.9 Hz, 1H), 6.62 (d, J = 2.2 Hz, 2H), 6.47–6.40 (m, 4H), 5.71 (s, 1H), 3.70 (s, 3H); 13C NMR (500 MHz, DMSO-d6) δ 161.64, 159.07, 150.33, 134.29, 129.59, 128.97, 128.56, 124.30, 123.60, 114.87, 112.79, 102.97, 55.82; HRMS (m/z): calculated for C28H20N2O5 465.14722, found 465.14145.
2) = 0.60; 1H NMR (500 MHz, DMSO-d6) δ 9.92–9.75 (m, 2H), 8.99 (s, 1H), 7.89–7.84 (m, 1H), 7.57 (dtd, J = 19.0, 7.3, 1.2 Hz, 2H), 7.35–7.32 (m, 2H), 7.08 (d, J = 7.1 Hz, 1H), 6.88 (d, J = 1.9 Hz, 1H), 6.62 (d, J = 2.2 Hz, 2H), 6.47–6.40 (m, 4H), 5.71 (s, 1H), 3.70 (s, 3H); 13C NMR (500 MHz, DMSO-d6) δ 161.64, 159.07, 150.33, 134.29, 129.59, 128.97, 128.56, 124.30, 123.60, 114.87, 112.79, 102.97, 55.82; HRMS (m/z): calculated for C28H20N2O5 465.14722, found 465.14145.
2-((4-Nitrobenzylidene)amino)-3′,6′-dihydroxyspiro[isoindolin-3-one-1,9′-xanthene] F7 had a yield of 90.5%; m. p: 180–182 °C, yellow. F. r. (Ac![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Hex) (3
Hex) (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) = 0.66; 1H NMR (500 MHz, DMSO-d6) δ 10.02 (d, J = 112.9 Hz, 2H), 9.11 (s, 1H), 8.16 (d, J = 8.6 Hz, 2H), 7.91 (d, J = 7.5 Hz, 1H), 7.64 (dd, J = 7.9, 5.2 Hz, 3H), 7.58 (t, J = 7.4 Hz, 1H), 7.12 (d, J = 7.6 Hz, 1H), 6.64 (d, J = 2.3 Hz, 2H), 6.47 (d, J = 8.6 Hz, 2H), 6.42 (dd, J = 8.7, 2.4 Hz, 2H); 13C NMR (500 MHz, DMSO-d6) δ 164.56, 159.27, 152.77, 150.98, 148.54, 146.54, 141.20, 134.99, 129.82, 129.09, 128.50, 128.14, 124.65, 124.47, 123.97, 112.99, 110.44, 103.11, 66.14.; HRMS (m/z): calculated for C27H17N3O6 480.11174, found 480.11212.
2) = 0.66; 1H NMR (500 MHz, DMSO-d6) δ 10.02 (d, J = 112.9 Hz, 2H), 9.11 (s, 1H), 8.16 (d, J = 8.6 Hz, 2H), 7.91 (d, J = 7.5 Hz, 1H), 7.64 (dd, J = 7.9, 5.2 Hz, 3H), 7.58 (t, J = 7.4 Hz, 1H), 7.12 (d, J = 7.6 Hz, 1H), 6.64 (d, J = 2.3 Hz, 2H), 6.47 (d, J = 8.6 Hz, 2H), 6.42 (dd, J = 8.7, 2.4 Hz, 2H); 13C NMR (500 MHz, DMSO-d6) δ 164.56, 159.27, 152.77, 150.98, 148.54, 146.54, 141.20, 134.99, 129.82, 129.09, 128.50, 128.14, 124.65, 124.47, 123.97, 112.99, 110.44, 103.11, 66.14.; HRMS (m/z): calculated for C27H17N3O6 480.11174, found 480.11212.
For the second method, a flask containing one equivalent of the F5 and F7 derivatives was completely dissolved in DMF. Then 2.2 equivalents of K2CO3 were added, and a catalytic amount of BTBA (phase transfer catalyst) was added. At room temperature, the reaction mixture was stirred for thirty minutes. 2.2 equivalents of ethyl bromoacetate were then added to the mixture after it had been stirred until the desired xanthene hydrazone derivatives F2, F3 and F4 were generated. The mixture was added to cold water after the reaction, and the precipitate that formed was filtered off. The crude material was further purified by column chromatography using an appropriate solvent mixture as eluent to afford the pure products F2, F3 and F4.
The yield of diethyl 2,2′-((2-((4-nitrobenzylidene)amino)spiro[isoindolin-3-one-1,9′-xanthene]-3′,6′-diyl)bis(oxy))(E)-diacetate F2 was 93%, light yellow, m. p.: 151–153 °C, f. r. = 0.40; 1H NMR (500 MHz, DMSO-d6): δ 9.33 (s, 1H), 8.23–8.17 (m, 1H), 8.13 (ddd, J = 8.2, 2.4, 1.1 Hz, 1H), 7.98–7.87 (m, 1H), 7.79 (dt, J = 7.9, 1.3 Hz, 1H), 7.67–7.57 (m, 2H), 7.17 (dt, J = 7.8, 0.9 Hz, 1H), 6.88 (t, J = 1.5 Hz, 2H), 6.63 (d, J = 1.5 Hz, 5H), 4.79 (s, 4H), 4.11 (q, J = 7.1 Hz, 4H), 1.13 (t, J = 7.1 Hz, 6H). 13C NMR (126 MHz, DMSO-d6): δ 168.92, 164.39, 159.24, 152.81, 150.18, 148.74, 148.12, 136.83, 134.98, 133.30, 130.95, 130.09, 128.65, 125.11, 124.51, 124.12, 121.43, 112.95, 112.46, 102.52, 65.96, 65.45, 61.23, 40.33, 14.49. HRMS (m/z): calculated for C35H29N3O10 652.18529, found 652.18378 [M + H]+.
The yield of diethyl 2,2′-((2-(benzylideneamino)spiro[isoindoline-3-one-1,9′-xanthene]-3′,6′-diyl)bis(oxy))(E)-diacetate F3 was 78%, light yellow, m. p: 123–125 °C, f. r. = 0.37; 1H NMR (500 MHz, DMSO-d6): δ 9.21 (s, 1H), 7.93–7.88 (m, 1H), 7.65–7.56 (m, 2H), 7.40–7.33 (m, 2H), 7.31 (d, J = 7.3 Hz, 3H), 7.13 (d, J = 7.1 Hz, 1H), 6.86 (d, J = 1.9 Hz, 2H), 6.61 (d, J = 2.0 Hz, 4H), 4.79 (s, 4H), 4.12 (q, J = 7.1 Hz, 4H), 1.14 (t, J = 7.1 Hz, 6H); 13C NMR (126 MHz, DMSO-d6): δ 168.95, 164.14, 159.14, 152.72, 151.11, 150.29, 134.93, 134.66, 131.03, 129.97, 129.82, 129.33, 128.74, 127.35, 124.42, 123.92, 113.08, 112.40, 102.43, 65.43, 61.23, 14.53; HRMS (m/z): calculated for C35H30N2O8 607.20022, found 607.20333.
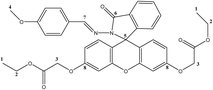
The yield of diethyl 2,2′-((2-((4-methoxybenzylidene)amino)spiro[isoindolin-3-one-1,9′-xanthene]-3′,6′-diyl)bis(oxy))(E)-diacetate F4 was 83.5%, white, m. p.: 136–138 °C, f. r. = 0.37; 1H NMR (500 MHz, DMSO-d6): δ 9.16 (s, 1H), 7.93–7.85 (m, 1H), 7.64–7.54 (m, 2H), 7.36–7.29 (m, 2H), 7.14–7.09 (m, 1H), 6.90–6.83 (m, 4H), 6.64–6.55 (m, 4H), 4.79 (s, 4H), 4.12 (q, J = 7.1 Hz, 4H), 3.70 (s, 3H), 1.14 (t, J = 7.1 Hz, 6H); 13C NMR (126 MHz, DMSO-d6): δ 168.97, 163.90, 161.73, 159.07, 152.71, 151.61, 150.26, 130.03, 129.00, 128.75, 127.46, 113.16, 112.33, 65.71, 65.39, 61.24, 14.54; HRMS (m/z): calculated for C36H32N2O9 637, 21078, found 636, 21326.
2.3. Biological activity
To measure the relaxation capacity of a compound, we tested different concentrations. After an equilibration phase, the aortic rings were contracted with epinephrine (10 µM). Once the contraction had stabilized, we gradually added increasing concentrations of the compound (12.5, 25, 37.5 and 50 µg mL−1), waiting 10 minutes between each addition for the response to stabilize. The compounds were solubilized in 1% DMSO. The DMSO concentrations used as controls were 0.0025%, 0.005%, 0.0075% and 0.01%. Control tests confirmed that the solvent had no significant effect on the vascular tone of the rings. Relaxation was expressed as a percentage of epinephrine-induced contraction, following a protocol already described in the literature.28
2.4. In silico studies
2.5. Statistical analysis
Data are presented as mean ± standard deviation. The Shapiro–Wilks test was used to guarantee normality, and Levene's test was used to assess the assumption of homogeneity of variance. Two-way ANOVA and Bonferroni's post hoc test for multiple comparisons were used for statistical analysis (Prism 8, GraphPad). A P-value of less than 0.05 was considered statistically significant.3. Results and discussion
3.1. Chemistry
The novel functionalized xanthene-hydrazone derivatives were synthesized using two distinct methodological approaches (Scheme 1). The first protocol is based on sequential functionalization, beginning with O-alkylation carried out by phase transfer catalysis, followed by functionalization at the nitrogen (N) position to form the hydrazone function. This last step was carried out under reflux in an acidic medium using a conventional method. In the second protocol, these steps are applied in reverse order, i.e., N-functionalization was carried out first, followed by O-alkylation.The structures of intermediates FH, F1, and F5 through 7, as well as that of the newly synthesized target molecules F2, F3 and F4 were checked using various spectroscopic techniques and mass spectrometry. The NMR results for compound F4 are presented as a representative example of this series. In the 1H NMR spectrum, a singlet is observed, corresponding to the six protons of the terminal methyl groups (–CH3) of the ethoxy chains. Another singlet at 4.79 ppm is assigned to the four protons of the two O–CH2 groups bonded to the aromatic ring, while a signal at 4.12 ppm corresponds to the O–CH2 protons associated with the ester function. A singlet at 3.70 ppm is assigned to the three protons of the methoxy group carried by the benzene ring. In addition, a singlet signal at 9.16 ppm is characteristic of the proton of the imine function (N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH). Finally, the signals between 6.5 and 8 ppm are assigned to the aromatic protons. The 13C NMR spectrum of compound F4 shows four main signals at 65.39 ppm, 61.24 ppm, 55.78 ppm, and 14.54 ppm, corresponding respectively to the carbons of the O–CH2 methylene groups bonded to the aromatic ring, the ester methylene (CO–O–CH2), the C–CH3 carbon of the ethoxy group, and the methoxy group of the benzene ring. An additional signal at 65.71 ppm is attributed to the spiro carbon characteristic of the spiro-lactam form of fluorescein. The most relevant NMR signals for compound F4 are shown in Table 1.
CH). Finally, the signals between 6.5 and 8 ppm are assigned to the aromatic protons. The 13C NMR spectrum of compound F4 shows four main signals at 65.39 ppm, 61.24 ppm, 55.78 ppm, and 14.54 ppm, corresponding respectively to the carbons of the O–CH2 methylene groups bonded to the aromatic ring, the ester methylene (CO–O–CH2), the C–CH3 carbon of the ethoxy group, and the methoxy group of the benzene ring. An additional signal at 65.71 ppm is attributed to the spiro carbon characteristic of the spiro-lactam form of fluorescein. The most relevant NMR signals for compound F4 are shown in Table 1.
High-resolution mass spectrometry of F4 revealed the pseudomolecular ion [M + H]+ at m/z 637.21328, consistent with the expected molecular mass. All NMR and HRMS spectra of the other synthesized compounds are given in the SI section.
3.2. Vasorelaxant activity
We conducted the evaluation in several stages. First, we tested the vasorelaxant effect of fluorescein hydrazine (FH) itself. Next, we examined the impact of alkylation on the hydroxyl group (OH), particularly for compound F1. Then, we studied separately the effect of hydrazone formation on the amine function (NH2), which corresponds to compounds F5 to F7. Finally, we analysed the combined influence of O-alkylation and hydrazone formation on compounds F2, F3 and F4. This allowed us to evaluate the synergistic role of these chemical modifications on vasorelaxant activity. The results are given in Fig. 3 and Table 2.| Compounds | EC50 (µg mL−1) | Emax (%) |
|---|---|---|
| FH | 0.000 ± 0.000 | 00.00 ± 0.00 |
| F1 | 0.000 ± 0.000 | 00.00 ± 0.00 |
| F2 | 113.000 ± 8.00 | 19,75 ± 4.10 |
| F3 | 38.204 ± 5.00 | 80.61 ± 6.00 |
| F4 | 41.300 ± 5.000 | 83.13 ± 6.20 |
| F5 | 0.000 ± 0.000 | 00.00 ± 0.00 |
| F6 | 0.000 ± 0.000 | 00.00 ± 0.00 |
| F7 | 0.000 ± 0.000 | 00.00 ± 0.00 |
| Verapamil | 18.000 ± 1.000 | 61.67 ± 4.10 |
The results show that fluorescein hydrazine (FH) does not exhibit significant vasorelaxant activity. This lack of efficacy suggests that its chemical structure, as designed, does not interact effectively with the membrane receptors or signalling pathways involved in vascular relaxation. Furthermore, derivatives modified at the O position, such as product F1, were also found to be inactive. This indicates that simple substitution on the hydroxyl group is not sufficient to confer pharmacological activity. Evaluation of N-functionalization, corresponding to the formation of the hydrazone function, showed that N-functionalized compounds (F5–F7) exert no significant vasorelaxant activity, with EC50 and Emax parameters remaining zero under our experimental conditions.
Double functionalization, combining modifications at the O and N positions, aimed to simultaneously modulate the polarity, lipophilicity and electron density of the fluorescein nucleus to optimize molecular recognition at the level of vascular receptors. This strategy yielded very different results: marked vasorelaxant activity, unlike the simple derivatives, which remained inactive. The EC50 and Emax values for compounds F2 to F4 reveal a clear hierarchy, which is dependent on the position and nature of the aromatic substituents introduced. This confirms the extent to which the chemistry of these groups influences pharmacological activity.
Compounds F3, bearing an unsubstituted aromatic ring, and F4, substituted by a methoxy group in the para position, exhibit moderate to good vasorelaxant activity. They differ markedly from fluorescein hydrazone (FH) and its simple derivatives F1 and F5 to F7, which are inactive. Their EC50 values are 38.204 and 41.3 µg mL−1 respectively, while their Emax values (80.61% and 83.13%) indicate significant activity. This improvement suggests that a moderate increase in the electron density of the aromatic ring promotes the recognition of compounds by receptors or channels involved in vasodilation. In contrast, the F2 derivative, substituted by a nitro group (NO2), shows a notable reduction in activity (EC50 = 113 µg mL−1; Emax = 19.75%). This substitution disrupts conjugation and impairs interaction with the biological target.
These collective findings establish a definitive structure–activity relationship that constitutes the central discovery of this study: dual functionalization at both O- and N-positions is strictly required for vasorelaxant activity. The complete inactivity of singly modified derivatives (F1, F5–F7) versus the marked potency of doubly functionalized congeners (F3, F4) demonstrates that neither the ethyl ester groups alone, nor the hydrazone moiety alone can activate the underlying biological mechanism. Instead, the simultaneous presence of both pharmacophores appears necessary to achieve the optimal balance of polarity, lipophilicity, and electron density required for effective interaction with vascular receptors or ion channels.
3.3. Molecular docking studies
To understand the mechanism of action and binding affinities of the synthesized compounds toward the human L-type calcium channel (CaV1.2), molecular docking simulations were performed. CaV1.2 was chosen as the molecular target owing to its essential role in regulating smooth muscle contraction and promoting vascular relaxation.38 The channel therefore represents a favourable target in the treatment of cardiovascular disorders, particularly those characterized by an excessive rate of smooth muscle contraction, since it controls the vascular tone by modulating the calcium ion influx into the cells.39,40 The crystal structure with PDB ID: 8HMB, co-crystallized with verapamil, was used to compare the binding affinities and interaction profiles of the synthesized derivatives with the reference drug. Before the docking simulations, re-docking of the native ligand within the protein's active site was carried out. The RMSD value of 1.27 Å resulting from this practice, which is significantly below the accepted threshold of 2.0 Å, validates the accuracy and reliability of the docking methodology used (Fig. 4).Results of docking simulations indicated that the two active compounds (F3 and F4) were all favorably situated within the CaV1.2 binding pocket, with binding energies of approximately −7.0 to −7.1 kcal mol−1, which were comparable to that of verapamil (−7.0 kcal mol−1) (Table 3). Supporting the findings of the biological tests, the compounds F3 (EC50 = 38.204 µg mL−1) and F4 (EC50 = 41.300 µg mL−1) were found to be the strongest CaV1.2 inhibitors, thus indicating their high binding affinity. 2D and 3D visual representations of the compounds F3 and F4, compared with verapamil (Fig. 5), were generated for analysis of the key interactions responsible for their enhanced affinity toward the CaV1.2 target. Verapamil exhibits a stabilizing network comprising hydrogen bonds and multiple hydrophobic contacts. Specifically, it forms a conventional hydrogen bond with Thr1056 (2.53 Å) and several carbon-hydrogen bonds with Ser1532 (3.45 Å), Met1509 (3.16 Å), and Ala1512 (3.55 Å), ensuring an optimal orientation of the ligand within the binding pocket. Additional hydrophobic interactions, including π-alkyl contacts with Val1053 (5.81 Å), Met1177 (5.20 Å), Met1178 (5.18 Å), and Ala1512 (4.59 Å), together with π-sigma interactions with Phe1181 (3.90 Å), further stabilize the complex. Compounds F3 and F4 exhibit a highly comparable interaction profile, forming conventional hydrogen bonds with Thr1057 (2.41 Å) and Ser1532 (2.75 Å), along with carbon–hydrogen bonds with Thr1056 (2.85 Å) and Ser1132 (3.30 Å). Stabilization is also reinforced by π-sigma interactions with Met1509 (3.70 Å) and π-alkyl contacts with Val1053 (5.45 Å), Ala1512 (5.37 Å), and Met1509 (4.54 Å and 4.59 Å). Notably, F3 forms an additional π-alkyl interaction that is absent in F4, probably because the methoxy substituent in F4 alters the aromatic orientation of the ligand within the binding pocket. Overall, both derivatives recapitulate almost all the important interactions observed for verapamil, taking the same polar and hydrophobic residues in the active site. This similarity in the interaction pattern explains why docking gave such close binding energies of (−7.0 to −7.1 kcal mol−1) and aligns with the experimental activity values. These results highlight the capability of F3 and F4 as high-affinity CaV1.2 ligands and provide a strong rationale for additional structural optimization and the development of congeners with increased activity.
| Compounds | F3 | F4 | Verapamil |
| Binding affinity kcal mol−1 | −7.1 | −7.1 | −7.0 |
 | ||
| Fig. 5 2D and 3D binding interactions of compounds F3, F4, and verapamil within the active site of CaV1.2 (PDB ID: 8HMB). | ||
3.4. ADME-Tox predictions
The ADMET properties of the active compounds were evaluated using the pkCSM web server to assess their pharmacokinetic potential as promising drug candidates (Table 4). Both molecules showed excellent human intestinal absorption (HIA = 100%), which is a sign of good oral bioavailability. Their Caco-2 permeability values (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Papp = 0.407 and 0.346, respectively) imply that their absorption is moderate, thus confirming their passive diffusion through the cell membranes. The volume of distribution (log
Papp = 0.407 and 0.346, respectively) imply that their absorption is moderate, thus confirming their passive diffusion through the cell membranes. The volume of distribution (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) VDss) values was negative (−1.385 and −1.496 L kg−1, respectively), indicating limited tissue distribution, while the predicted log
VDss) values was negative (−1.385 and −1.496 L kg−1, respectively), indicating limited tissue distribution, while the predicted log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) BB values, all less than −1.0, confirm poor penetration across the blood–brain barrier (BBB), which could potentially reduce central side effects. Regarding metabolism, both compounds are predicted to act as substrates of CYP3A4, and neither compound inhibits CYP1A2, CYP2D6, or CYP3A4, except for F4, which shows slight inhibition of CYP3A4. However, both compounds display moderate inhibitory potential toward CYP2C19 and CYP2C9, suggesting an acceptable level of metabolic interaction and maintaining overall metabolic stability. The total clearance values (0.838 and 0.824 mL min−1 kg−1, respectively) further indicate adequate excretory efficiency. Concerning toxicity predictions (Table 5), both F3 and F4 were determined to be non-sensitizing to the skin, non-hepatotoxic, and non-mutagenic (negative Ames test), reflecting an overall favourable safety profile. However, both compounds were identified as potential hERG II inhibitors, which may raise concerns regarding cardiotoxicity. As hERG channel inhibition is associated with QT interval prolongation and arrhythmias, this aspect is particularly relevant for cardiovascular agents. Nevertheless, this prediction is based solely on in silico models, while the present study focused on vasorelaxant activity in rat aortic rings and interaction with the CaV1.2 channel. Therefore, further experimental studies are required to confirm this potential liability and ensure cardiac safety. Overall, these in silico findings suggest that F3 and F4 possess acceptable ADME-Tox properties, supporting their potential as promising drug candidates.
BB values, all less than −1.0, confirm poor penetration across the blood–brain barrier (BBB), which could potentially reduce central side effects. Regarding metabolism, both compounds are predicted to act as substrates of CYP3A4, and neither compound inhibits CYP1A2, CYP2D6, or CYP3A4, except for F4, which shows slight inhibition of CYP3A4. However, both compounds display moderate inhibitory potential toward CYP2C19 and CYP2C9, suggesting an acceptable level of metabolic interaction and maintaining overall metabolic stability. The total clearance values (0.838 and 0.824 mL min−1 kg−1, respectively) further indicate adequate excretory efficiency. Concerning toxicity predictions (Table 5), both F3 and F4 were determined to be non-sensitizing to the skin, non-hepatotoxic, and non-mutagenic (negative Ames test), reflecting an overall favourable safety profile. However, both compounds were identified as potential hERG II inhibitors, which may raise concerns regarding cardiotoxicity. As hERG channel inhibition is associated with QT interval prolongation and arrhythmias, this aspect is particularly relevant for cardiovascular agents. Nevertheless, this prediction is based solely on in silico models, while the present study focused on vasorelaxant activity in rat aortic rings and interaction with the CaV1.2 channel. Therefore, further experimental studies are required to confirm this potential liability and ensure cardiac safety. Overall, these in silico findings suggest that F3 and F4 possess acceptable ADME-Tox properties, supporting their potential as promising drug candidates.
| Compounds | ADME properties | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Absorption | Distribution | Metabolism | Excretion | |||||||||
Caco2 perm. log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Papp in 10−6 cm s−1 Papp in 10−6 cm s−1 |
HIA% | VDss (log L kg−1) | BBB perm. (BB log) | CYP450 | Total clearance log ml min−1 kg−1 | |||||||
| Substrate | Inhibitor | |||||||||||
| 2D6 | 3A4 | 1A2 | 2C19 | 2C9 | 2D6 | 3A4 | ||||||
| Acceptable range | Log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Papp > 0.90: high Papp > 0.90: high |
<30%: poorly absorbed | log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) VDss >0.45: high VDss >0.45: high |
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) BB < – 1: poorly distributed BB < – 1: poorly distributed |
Categorical (yes/no) | — | ||||||
| F3 | 0.407 | 100 | −1.385 | −1.565 | No | Yes | No | Yes | Yes | No | No | 0.838 |
| F4 | 0.346 | 100 | −1.496 | −1.802 | No | Yes | No | Yes | Yes | No | Yes | 0.824 |
| Compounds | Toxicity | ||||
|---|---|---|---|---|---|
| AMES | Categorical (yes/no) | ||||
| hERG I | hERG II | Hepatotoxicity | Skin Sensitization | ||
| F3 | No | No | Yes | No | No |
| F4 | No | No | Yes | No | No |
3.5. MD simulations
MD simulations of compounds F3 and F4, as well as verapamil, in complex with the CaV1.2 protein (PDB ID: 8HMB) were performed over a 100 ns timescale to validate the docking results and evaluate the conformational stability of the complexes. The Cα RMSD values of all systems were explored to evaluate the structural stability during the simulation. Both complexes, 8HMB-F3 and 8HMB-F4, exhibited relatively low backbone deviations ranging between 5.0 and 6.0 Å and quickly reached equilibrium after approximately 10 ns, maintaining stable trajectories for the rest of the simulation (Fig. 6A). The RMSD profile of the 8HMB-verapamil complex also showed relatively low backbone deviations (5.0–6.0 Å) during the first 30 ns, followed by pronounced fluctuations, with RMSD values increasing up to 13 Å. After about 70 ns, the system stabilized at around 11 Å, indicating significant conformational rearrangements within the complex. The average ligand RMSD values for all complexes remained consistent overall during the simulation (Fig. 6B). Specifically, compound F3 exhibited RMSD fluctuations between 2.0 and 3.5 Å, suggesting a stable orientation and minimal movement within the active site. Compound F4 displayed moderate variations ranging from 3.0 to 5.0 Å, while verapamil showed the highest ligand RMSD values (5.0–8.0 Å), indicating greater flexibility. This suggests that the F3 ligand maintained a more stable conformation and experienced fewer structural changes compared to F4 and verapamil throughout the simulation. RMSF values are also computed to establish the dynamic behavior of protein residues (Fig. 6C). The structural stability of the protein was confirmed as most residues in all systems showed a fluctuation below 5 Å. Maximum fluctuations of 15.0 Å and 10.0 Å are detected in residues Arg114, Arg590, Asn898 and Leu1563. Visual examination of the MD simulation trajectories revealed that all compounds exhibited substantial binding interactions with the hotspot residues, specifically, Gln1060, Met1178, Phe1181, Ala1512, Met1509 and Tyr1508 of the CaV1.2 protein. It was found that all these interacting residues had low RMSF values, indicating the stability of F3 and F4 complexes compared to the verapamil complex during simulation. Greater structural stability is reflected by smaller fluctuations. The radius of gyration (RGyr) was used to assess the structural compactness of complexes. The radius of gyration represents the root-mean-square distance of a molecule's atoms from its centre of mass, serving as a measure of the overall compactness and three-dimensional organisation of a macromolecule under different conditions.41,42 The RGyr values (Fig. 6D) for all three complexes remained nearly constant, ranging from 5.2 to 5.8 Å, throughout the 100 ns trajectory. These relatively low and favourable values suggest that the CaV1.2 protein maintained its overall structural stability throughout the simulation.Interaction analysis identified the key residues contributing to the stabilization of the complexes. The 8HMB-F3 complex (Fig. 7A) showed that the residues Tyr1508, Phe1181, and Met1509 with the highest interaction fractions of about 1.1, 0.9, and 0.7, respectively. These values reflect predominant hydrophobic and π–π interactions that persisted throughout the simulation, playing a crucial role in maintaining complex stability. The 8HMB-F4 complex (Fig. 7B) displayed a more evenly distributed interaction pattern, with interaction fractions exceeding 0.7 for Phe118, around 0.6 for Tyr1108, Met1129, and Ser1506, and between 0.4 and 0.5 for several additional residues. These interactions, having hydrophobic contacts, hydrogen bonds, and water bridges, also contributed to the overall stability of the complex. In contrast, verapamil (Fig. 7C) had an interaction with Gln1060 as the strongest one with a fraction around 1.0, along with secondary contacts with Phe1129 and Phe1181 (0.6, 0.7, respectively), reflecting the stabilization within the CaV1.2 active site. Overall, MD studies indicate that both F3 and F4 formed stable complexes with CaV1.2, with a slight preference for F3, as demonstrated by its decreased residue fluctuations and lower protein and ligand RMSD values at the active site, and maintained structural compactness. These results confirm the strong and stable binding modes of F3 and F4, supporting their potential as promising vasorelaxant agents.
4. Conclusions
The present study enabled the successful synthesis and characterization of a novel series of functionalized molecules (F2–F4) incorporating xanthene and hydrazone motifs using two distinct methods, combining O-alkylation and condensation at the NH2 group. In vitro evaluation of vasorelaxant activity on isolated rat aortic rings showed that dual functionalization, simultaneously modulating the polarity, lipophilicity and electron density of the fluorescein nucleus, optimizes molecular recognition at the level of vascular receptors. This strategy led to significant vasorelaxant activity of the hybrid molecules in the series. Molecular docking and MD analyses showed a high binding affinity and improved stability in the active site compared to verapamil, thus suggesting that these compounds are valuable candidates for vasodilator therapeutic applications. Furthermore, results of in silico ADMET studies revealed a good pharmacokinetic profile for compounds F3 and F4, with favourable safety profiles as evidenced by negative Ame's test results (non-mutagenic). The predicted hERG II inhibition remains the primary safety consideration requiring further experimental evaluation. Overall, these experimental and theoretical findings underscore the potential of F3 and F4 as promising vasorelaxant agents and provide a strong rationale for further structural optimization and development of congeners with enhanced activity. Further experimental studies are needed to substantiate the predicted mechanism of action.Conflicts of interest
Authors declare no conflict of interest.Data availability
All additional data analysed during this study can be found in the supplementary information (SI) section of this article. Supplementary information: 1H NMR, 13C NMR, and HRMS spectra of the synthesized compounds. See DOI: https://doi.org/10.1039/d6ra01067a.Acknowledgements
The authors gratefully acknowledge the staff of the National Centre for Scientific and Technical Research (CNRST) in Rabat for their support in spectroscopic measurements. The Authors extend their appreciation to the Ongoing Research Funding Program (ORF-2026-861), King Saud University, for funding this project.References
- Y. Rhazi, I. Bouadid, A. Nakkabi, U. Raza, L. T. Nagarajappa, N. Deak, A. Soran, M. El Yazidi, M. Alaqarbeh, A. Tounsi, M. A. Harrad and M. Eddouks, Eur. J. Med. Chem., 2025, 291, 117515 CrossRef CAS PubMed.
- K. Ghosh Dastidar, S. Maitra, L. Goswami, D. Roy, K. P. Das and A. L. Majumder, Plant Physiol., 2006, 140, 1279–1296 CrossRef PubMed.
- N. L. Tilney, W. D. Whitley, J. R. Diamond, J. W. Kupiec-Weglinski and D. H. Adams, Transplantation, 1991, 52, 389 CrossRef CAS PubMed.
- A. H. Gradman, J. N. Basile, B. L. Carter, G. L. Bakris and American Society of Hypertension Writing Group, J. Am. Soc. Hypertens., 2010, 4, 42–50 CrossRef CAS PubMed.
- G. R. C. Millogo, R. F. E. Zongo, A. Benao, E. N. H. Youl, B. A. B. Bassoleth, M. Ouédraogo, P. Zabsonré and I. P. Guissou, Pan Afr. Med. J., 2018, 29, 29–84 Search PubMed.
- M. Radosavljevic, D. Vučević, J. Samardžić, M. Radenkovic and T. Radosavljević, Curr. Med. Chem., 2024, 31, 4602–4620 CrossRef PubMed.
- H. Singh and D. K. Agrawal, Bioorg. Med. Chem., 2022, 62, 116706 CrossRef CAS PubMed.
- Y. Rhazi, I. Bouadid, F. Z. Guerguer, M. El mesky, A. Arzine, A. Nakkabi, M. Bakhouch, N. Deak, S. Chtita, E. H. Mabrouk, G. Nemes, M. E. Yazidi and M. Eddouks, Chem. Biodiversity, 2025, 22, e01062 CrossRef CAS PubMed.
- B. Dean, G. Cooper, M. Shivkumar and T. J. Snape, Bioorg. Med. Chem. Lett., 2023, 84, 129211 CrossRef CAS PubMed.
- M. El Mesky, H. Zgueni, Y. Rhazi, O. El-Guourrami, O. Abchir, M. Jabha, A. Nakkabi, S. Chtita, S. Achamlale, M. Chalkha, D. Chebabe and E. H. Mabrouk, J. Mol. Struct., 2024, 1313, 138705 CrossRef CAS.
- X. Liu, J. Shen and K. Zhu, RSC Med. Chem., 2022, 13, 107–116 RSC.
- N. Singh, A. K. Shreshtha, M. S. Thakur and S. Patra, Heliyon, 2018, 4, e00829 CrossRef PubMed.
- J. X. Kelly, R. Winter, D. H. Peyton, D. J. Hinrichs and M. Riscoe, Antimicrob. Agents Chemother., 2002, 46, 144–150 CrossRef CAS PubMed.
- H. Li, W.-T. Kang, Y. Zheng, Y. He, R. Zhong, S. Fang, W. Wen, S. Liu and S. Lin, Sci. Adv., 2025, 11, eadt4723 CrossRef CAS PubMed.
- M. Abualhasan, M. Hawash, S. Aqel, M. Al-Masri, A. Mousa and L. Issa, ACS Omega, 2023, 8, 38597–38606 CrossRef CAS PubMed.
- M. L. C. Caxito, R. R. Correia, A. C. C. Gomes, G. Justo, M. G. P. Coelho, C. M. Sakuragui, R. M. Kuster and K. C. C. Sabino, J. Evidence-Based Complementary Altern. Med., 2015, 2015, 384267 Search PubMed.
- A. M. L. Seca, S. B. Leal, D. C. G. A. Pinto, M. C. Barreto and A. M. S. Silva, Molecules, 2014, 19, 8317–8333 CrossRef PubMed.
- X. Zhao, L. Ren, X. Wang, G. Han, S. Wang, Q. Yao and Y. Qi, Exp. Ther. Med., 2022, 23, 1–9 Search PubMed.
- L.-W. Wang, J.-J. Kang, I.-J. Chen, C.-M. Teng and C.-N. Lin, Bioorg. Med. Chem., 2002, 10, 567–572 CrossRef CAS PubMed.
- S. Rollas and S. G. Küçükgüzel, Molecules, 2007, 12, 1910–1939 CrossRef CAS PubMed.
- C. L. M. Silva, F. Noël and E. J. Barreiro, Br. J. Pharmacol., 2002, 135, 293–298 CrossRef CAS PubMed.
- S. Verma, S. Lal, R. Narang and K. Sudhakar, ChemMedChem, 2023, 18, e202200571 CrossRef CAS PubMed.
- P. C. Sharma, D. Sharma, A. Sharma, N. Saini, R. Goyal, M. Ola, R. Chawla and V. K. Thakur, Mater. Today Chem., 2020, 18, 100349 CrossRef CAS.
- Y. Biliz, B. Hasdemir, H. Başpınar Küçük, M. Zaim, A. M. Şentürk, A. Müdüroğlu Kırmızıbekmez and İ. Kara, ACS Omega, 2023, 8, 20073–20084 CrossRef CAS PubMed.
- Ł. Popiołek, Biomed. Pharmacother., 2023, 163, 114853 CrossRef PubMed.
- H. Zara Tariq, A. Saeed, S. Ullah, N. Fatima, S. Ahsan Halim, A. Khan, H. R. El-Seedi, M. Zaman Ashraf, M. Latif and A. Al-Harrasi, RSC Adv., 2023, 13, 26229–26238 RSC.
- S. Charan, N. Maksud, A. Gupta, S. Devi, M. M. Alam, M. Akhter, S. Tasneem, D. Parveen, Ahasan and M. Shaquiquzzaman, Eur. J. Med. Chem., 2026, 302, 118305 CrossRef CAS PubMed.
- I. Bouadid, A. Qabouche and M. Eddouks, Cardiovasc. Hematol. Agents Med. Chem., 2024, 22, 432–440 CrossRef CAS PubMed.
- Y. Wei, Z. Yu, L. Wang, X. Li, N. Li, Q. Bai, Y. Wang, R. Li, Y. Meng, H. Xu, X. Wang, Y. Dong, Z. Huang, X. C. Zhang and Y. Zhao, Nat. Commun., 2024, 15, 2772 CrossRef CAS PubMed.
- N. Guex and M. C. Peitsch, Electrophoresis, 1997, 18, 2714–2723 CrossRef CAS PubMed.
- M. D. Hanwell, D. E. Curtis, D. C. Lonie, T. Vandermeersch, E. Zurek and G. R. Hutchison, J. Cheminf., 2012, 4, 17 CAS.
- J. Eberhardt, D. Santos-Martins, A. F. Tillack and S. Forli, J. Chem. Inf. Model., 2021, 61, 3891–3898 CrossRef CAS PubMed.
- K. J. Bowers, E. Chow, H. Xu, R. O. Dror, M. P. Eastwood, B. A. Gregersen, J. L. Klepeis, I. Kolossvary, M. A. Moraes, F. D. Sacerdoti, J. K. Salmon, Y. Shan and D. E. Shaw, in Proceedings of the 2006 ACM/IEEE Conference on Supercomputing, Association for Computing Machinery, New York, NY, USA, 2006, p. 84 Search PubMed.
- F. Z. Guerguer, B. Rossafi, O. Abchir, Y. S. Raouf, D. B. Albalushi, A. Samadi and S. Chtita, PLoS One, 2025, 20, e0317261 CrossRef CAS PubMed.
- B. Rossafi, O. Abchir, F. Z. Guerguer, M. Khedraoui, M. E. Kouali and S. Chtita, ChemistrySelect, 2025, 10, e02869 CrossRef CAS.
- K. Roos, C. Wu, W. Damm, M. Reboul, J. M. Stevenson, C. Lu, M. K. Dahlgren, S. Mondal, W. Chen, L. Wang, R. Abel, R. A. Friesner and E. D. Harder, J. Chem. Theory Comput., 2019, 15, 1863–1874 CrossRef CAS PubMed.
- O. Abchir, I. Yamari, H. Nour, O. Daoui, S. Elkhattabi, A. Errougui and S. Chtita, ChemistrySelect, 2023, 8, e202301092 CrossRef CAS.
- D. E. V. Pires, T. L. Blundell and D. B. Ascher, J. Med. Chem., 2015, 58, 4066–4072 CrossRef CAS PubMed.
- J. P. Bannister, M. D. Leo, D. Narayanan, W. Jangsangthong, A. Nair, K. W. Evanson, J. Pachuau, K. S. Gabrick, F. A. Boop and J. H. Jaggar, J. Physiol., 2013, 591, 2987–2998 CrossRef CAS PubMed.
- S. Mowlazadeh Haghighi, M. Fathalipour, M. Abbasi, A. Purkhosrow, S. Oftadehgan, A. Khalafi-Nezhad and E. Sattarinezhad, Trends Pharm. Sci. Technol., 2023, 9, 105–112 Search PubMed.
- J. El-Akhal, A. P. Oliveira, R. Bencheikh, P. Valentão, P. B. Andrade and M. Morato, Molecules, 2022, 27, 8752 CrossRef CAS PubMed.
- K. Tabti, I. Ahmad, I. Zafar, A. Sbai, H. Maghat, M. Bouachrine and T. Lakhlifi, Comput. Biol. Chem., 2023, 104, 107855 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |