Open Access Article
Open Access ArticlePhotoredox-enabled late-stage functionalization of pyrrolidines in batch and flow: rapid access to benzothiazole hybrids as a new class of cholinesterase inhibitors
A. Filipovića,
M. Vučkovskib,
D. Mićovićb,
A. M. Bondžić b and
B. P. Bondžić
b and
B. P. Bondžić *a
*a
aUniversity of Belgrade-Institute of Chemistry, Technology and Metallurgy, National Institute of the Republic of Serbia, Njegoševa 12, 11000 Belgrade, Serbia. E-mail: bojan.bondzic@ihtm.bg.ac.rs
bVinča Institute of Nuclear Sciences, National Institute of the Republic of Serbia, University of Belgrade, P.O. Box 522, 11000 Belgrade, Serbia
First published on 7th April 2026
Abstract
A visible-light-driven photoredox protocol for the α-amino C(sp3)–H heteroarylation of N-aryl pyrrolidines and piperidines with 2-chlorobenzothiazole derivatives in batch and in flow conditions is developed. The transformation proceeds under mild conditions using an iridium photocatalyst in the presence of water, providing efficient access to benzothiazole-amine hybrid structures. After initial optimization in batch, the reaction was successfully translated to microfluidic flow, where enhanced light utilization and improved mass and heat transfer enabled higher productivity and superior energy efficiency compared to batch procedures. The scope of the method encompasses a broad range of substituted N-aryl pyrrolidines and benzothiazoles, affording the desired products in generally good to excellent yields. In total, a diverse library of benzothiazole–pyrrolidine and –piperidine derivatives was synthesized. The obtained compounds were evaluated for their physicochemical properties and biological activity against acetyl- and butyrylcholinesterase enzymes. Several derivatives exhibited inhibition of these enzymes in the micromolar range, with few molecules showing selectivity towards butyrylcholinesterase. In silico ADME analysis indicated favorable lipophilicity and predicted blood–brain barrier permeability. The synergistic combination of photoredox catalysis and microflow technology allowed an efficient and sustainable approach to the late-stage functionalization of biologically relevant amines and the rapid generation of a neuroactive small-molecule library.
1. Introduction
Over the past decade photoredox catalysis has become one of the fastest growing areas in organic chemistry. Using light as a readily available reagent, coupled with transition metal or organic photocatalysts led to the development of a plethora of light driven processes.1–5 A significant area of research in the photoredox catalysis field is late stage functionalizations,6–9 particularly C–H functionalizations,10,11 a promising approach in the synthesis of new pharmaceuticals. In that regard α-amino radical formation via visible light promoted single electron oxidation of amines and further functionalization of this open-shell species is an important tool for the synthesis of α-functionalized amines, promising structures with a potential wide field of biological activities. In the first example of a photoredox catalyzed α-amino C–H arylation reaction of aliphatic amines with electron deficient heteroarenes, only two examples of reaction with benzothiazole and benzoxazole were presented.12 This study was extended showcasing the direct α-heteroarylation of tertiary amines with a variety of five- and six-membered chloro-heteroarenes.13 In addition, C–H functionalization of tertiary aliphatic amines with a number of 2-chloroazole derivatives catalyzed by tris-fac-Ir(ppy)3 under the blue light irradiation was reported.14 Transition-metal-free reaction of 2-chlorobenzazoles with aliphatic carbamates, alcohols, and ethers was achieved using benzophenone as a photocatalyst under the UV-A irradiation at ambient temperature.15 Dual-catalytic platform for the cross dehydrogenative-coupling between (benzo-)thiazoles and amines combined low loadings of an iridium photoredox catalyst and a cobaloxime catalyst under blue light irradiation.16Functionalized pyrrolidines and piperidines are present in the nature in the form of bioactive alkaloids and are extensively used by pharma industry and academic groups in the pursuit for biologically active molecules. Piperidine and pyrrolidine in particular are very often found in approved as well as in the investigational and experimental drugs.17 Pyrrolidine is the most commonly found non-aromatic nitrogen heterocycle in the marketed drugs.18 They are present as a core structure as well as a pharmacophore used to impose certain desired properties upon larger molecule. Among the well-known drugs with a pyrrolidine or piperidine ring, some of the very recently approved molecules (2022) containing pyrrolidine ring include daridorexant (insomnia), pacritinib (JAK-2 inhibitor), and futibatinib 14 (FGFR-4 inhibitor) (Fig. 1).19 Additionally, pyrrolidine and derivatives are very often used as ligands in transition metal catalysis and as catalysts in organo-catalysis.20–22
On the other hand, benzothiazoles represent prominent structural motif in a wide range of biologically active molecules and are attractive target structures in medicinal chemistry.23 Their biological activity is reflected in ability to inhibit various therapeutic targets such as the cyclooxygenase-2 (COX-2), the enzyme linked with inflammation, pain, and fever,24 the vascular endothelial growth factor receptor-2 (VEGFR-2), the receptor crucial for tumor growth and angiogenesis,25 the tyrosinase, the enzyme involved in melanin production, with potential for skin-whitening agents and antioxidants.26 Recently published study also clearly demonstrated the inhibitory potential of benzothiazole derivatives against enzymes involved in the pathogenesis of neurodegenerative diseases, such as the monoamine oxidase B (MAO-B),27 acetyl- (AChE) and butyrylcholinesterase (BuChE),28,29 highlighting this scaffold as a potential chromophore in the development of new anti-neurodegenerative drugs. Additionally, there are several notable drugs that incorporate this moiety (Fig. 2).
Functionalization of (benz)azoles was previously achieved with a variety of transition-metal-catalyzed C–C and C–N coupling reactions using the copper, nickel, or palladium catalysis.30 In our study we wanted to perform thorough examination of photoredox catalyzed α-amino C(sp3)–H heteroarylation of N-aryl pyrrolidines and piperidines with 2-chlorobenzazole derivatives to obtain number of potentially active molecules against cholinergic enzymes. Various pyrrolidine derivatives have already shown cholinergic activity, such as dispiro–indeno pyrrolidine/pyrrolothiazole–thiochroman hybrids,31 spirooxindole-pyrrolidine derivatives,32 or N-benzylpyrrolidine derivatives.33
We expected that merger of these biological active scaffolds, pyrrolidine's ring with benzothiazoles, should provide good starting point for enhancement of biological activity of hybrid compounds as for instance shown in a series of benzimidazole pyrrolidine hybrid molecules that were synthesized by Min et al. as inhibitors of poly(ADP-ribose) for treatment of cancer.34 This endeavor is in line with our recent work in the field of cholinergic enzymes inhibitors as potential Alzheimer's disease therapies.35–37 In addition, to make the functionalization practical and more efficient we also envisaged use of the microfluidic chemistry methodology as a green and efficient synthetic method.
2. Experimental
2.1. General remarks
All reactions were monitored by thin-layer chromatography using Merck 60 F254 precoated silica gel plates (0.25 mm thickness). Preparative thin layer chromatography was performed using Merck 60 F254 silica gel purchased from Merck KGA. Column chromatography was carried out on silica gel (12–26, ICN Biomedicals) using petrol ether/ethyl acetate as eluents. 1H-NMR and 13C-NMR spectra were measured on a Bruker Ultrashield Advance III spectrometer (1H at 500 MHz, 13C at 125 MHz) and Varian 400 sprectrometer (1H at 400 MHz, 13C at 100 MHz) using CDCl3 as solvent with TMS as internal standard. Chemical shifts (δ) are given in parts per million (ppm) and coupling constants are given in Hertz (Hz).The proton spectra are reported as follows δ per ppm (multiplicity, coupling constant J Hz−1, number of protons). High-resolution mass spectral analyses (HRMS) were carried out using Bruker ESI-TOF MS. IR spectra were measured on a PerkineElmer FT-IR 1725X spectrophotometer using ATR technique. The peak intensities are defined as very strong (vs.), strong (s), middle (m) or weak (w).2.2. Materials and methods
3. Results and discussion
3.1. Synthesis of benzothiazole–pyrrolidine derivatives in batch
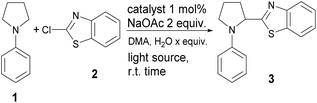
We started our study by firstly optimizing reaction conditions in batch using N-phenyl pyrrolidine and 2-chlorobenzthiazole as model reactants, using different photocatalysts in the presence of DMA as a solvent and various amounts of water as a co-solvent. Small amounts of water have already been shown to be beneficial for this reaction and NaOAc has been established as the base of choice for this type of reaction.12,13 Solubility of an inorganic base is a very important factor when establishing synthetic protocol that would also be viable under microfluidic conditions. Solids could precipitate leading to unrepeatable results or reaction interruption upon clogging of channels. In that regard we examined the effect of amount water on the reaction outcome. In the presence of 1 mol% [Ir(dtbbpy)(ppy)2]PF6 as a catalyst and 2 equivalents of NaOAc as a base while increasing amounts of water from 0 to 65 equivalents (Table 1, entries 1–4) we observed an increase in the reaction yield. Best results were obtained with 65 equivalents of water in which case reaction mixture was homogeneous. Further increase up to 185 equivalents of water led to deteriorating yield and only 15% of desired compound was isolated (Table 1, entry 5). Other Ir catalysts and organic catalysts such as TPP and acridinium type catalyst did not give better yields compared to initial Ir catalyst (Table 1 entries 6–10). After reaction conditions were optimized series of N-aryl substituted pyrrolidines were reacted with various 2-chloro benzthiazoles in order to establish the scope of the reaction.| Entrya | Photocatalyst (x mol%) | H2O (equiv.) | Light | Yield (%)b |
|---|---|---|---|---|
| a Reaction conditions: phenylpyrrolidine (0.5 mmol, 2 equiv.), Ir cat. (0.0025 mmol, 1 mol%), NaOAc (0.5 mmol, 2 equiv.) and benzothiazole (0.25 mmol, 1 equiv.) were added to DMA (1 mL) and H2O (see table) mixture, and sparged with argon for 15 minutes to remove oxygen. Solution was irradiated with 24 W blue LED and stirred for designated period of time.b Isolated yield after column chromatography. | ||||
| 1 | [Ir(dtbbpy)(ppy)2]PF6 (1 mol%) | 0 | Blue LED | 51 |
| 2 | [Ir(dtbbpy)(ppy)2]PF6 (1 mol%) | 10 | Blue LED | 72 |
| 3 | [Ir(dtbbpy)(ppy)2]PF6 (1 mol%) | 25 | Blue LED | 76 |
| 4 | [Ir(dtbbpy)(ppy)2]PF6 (1 mol%) | 65 | Blue LED | 87 |
| 5 | [Ir(dtbbpy)(ppy)2]PF6 (1 mol%) | 185 | Blue LED | 35 |
| 6 | Ir[dF(CF3)ppy]2(dtbpy)PF6 (1 mol%) | 65 | Blue LED | 77 |
| 7 | Ir(ppy)3 (1mol%) | 65 | Blue LED | 60 |
| 8 | TPP (5 mol%) | 65 | Blue LED | 10 |
| 9 | 9-Mesityl-3,6-di-tert-butyl-10-phenylacridinium tetrafluoroborate (5 mol%) | 25 | Blue LED | n.r. |
| 10 | 9-Mesityl-10-methylacridinium perchlorate (5 mol%) | 25 | Blue LED | n.r. |
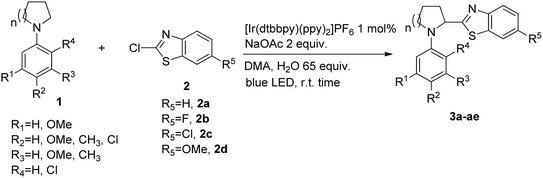
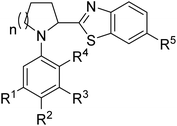
In all cases better yields were obtained with unsubstituted 2a or Cl-substituted 2c. Methoxy substituted benzothiazole 2d in general gave bit lower yields of desired product in the most of the cases while fluorine substituted 2b gave very good yields in general a bit lower than Cl substituted 2c probably due to the pronounced mesomeric effect of fluorine in 2b. This behavior can be explained by the fact that according to previously established mechanism reaction most likely proceeds through addition of α-amino radical to heteroarene, in which case electron density at heteroarene's C-2 plays a very significant role. Various substituents at the aryl part of pyrrolidine are tolerated in this reaction, unsubstituted (Table 2, entries 1–4) or Me substituted substrates at R2 (Table 2, entries 5–8), R2 and R3 (Table 2, entries 13–16) and at R3 (Table 2, entries 17–20), react efficiently giving products in good to excellent yields. Electron donating OMe at position R2 (Table 2, entries 21–24) and at R1–R3 (Table 2, entries 29–31) also gave very good yields of desired products, reaction is efficient and bit faster judging by the TLC analysis which might be in line with increased electron density of nucleophilic α-amino radical in this case. Electron withdrawing Cl substituent at R4 (Table 2, entries 9–12) or at R2 (Table 2, entries 25–28) also gave very good yields of desired products. In addition, N-aryl substituted piperidines also react with 2-chloro benzthiazoles in the similar manner to pyrrolidines under optimized reaction conditions using 1 mol% [Ir(dtbbpy)(ppy)2]PF6 as a catalyst and DMA/water as a solvent under 24W LED irradiation. In general, lower yields compared to reactions with pyrrolidines are obtained. Reaction proceeds with benzthiazoles 2a–d under optimized conditions to give desired products in 56–76% yield (Table 2, entries 32–35). In total, 35 compounds with different substitution pattern were synthesized. Wide array of available substituents at the aryl part of the molecule is accompanied by the various substituents at the benzothiazole part of the hybrid molecule.
| Entrya | Compound | Structure | Yieldb (%) | |||||
|---|---|---|---|---|---|---|---|---|
| n | R1 | R2 | R3 | R4 | R5 | |||
| a Reaction conditions: phenylpyrrolidine (0.5 mmol, 2 equiv.), Ir cat. (0.0025 mmol, 1 mol%) sodium acetate (0.5 mmol, 2 equiv.) and benzothiazole (0.25 mmol, 1 equiv.) were added to solvent DMA (1 mL) and water (16.25 mmol, 65 equiv., 0.3 mL) mixture and sparged with argon for 15 minutes to remove oxygen. Solution was irradiated with 24 W blue LED and stirred for designated period of time.b Isolated yield after column chromatography. | ||||||||
| 1 | 3a | 1 | H | H | H | H | H | 87 |
| 2 | 3b | 1 | H | H | H | H | F | 61 |
| 3 | 3c | 1 | H | H | H | H | Cl | 69 |
| 4 | 3d | 1 | H | H | H | H | OCH3 | 75 |
| 5 | 3e | 1 | H | CH3 | H | H | H | 75 |
| 6 | 3f | 1 | H | CH3 | H | H | F | 66 |
| 7 | 3g | 1 | H | CH3 | H | H | Cl | 70 |
| 8 | 3h | 1 | H | CH3 | H | H | OCH3 | 82 |
| 9 | 3i | 1 | H | H | CH3 | H | H | 77 |
| 10 | 3j | 1 | H | H | CH3 | H | F | 78 |
| 11 | 3k | 1 | H | H | CH3 | H | Cl | 81 |
| 12 | 3l | 1 | H | H | CH3 | H | OCH3 | 82 |
| 13 | 3m | 1 | H | CH3 | CH3 | H | H | 67 |
| 14 | 3n | 1 | H | CH3 | CH3 | H | F | 73 |
| 15 | 3o | 1 | H | CH3 | CH3 | H | Cl | 89 |
| 16 | 3p | 1 | H | CH3 | CH3 | H | OCH3 | 79 |
| 17 | 3q | 1 | H | OCH3 | H | H | H | 88 |
| 18 | 3r | 1 | H | OCH3 | H | H | F | 77 |
| 19 | 3s | 1 | H | OCH3 | H | H | Cl | 76 |
| 20 | 3t | 1 | H | OCH3 | H | H | OCH3 | 81 |
| 21 | 3u | 1 | OCH3 | OCH3 | OCH3 | H | H | 84 |
| 22 | 3v | 1 | OCH3 | OCH3 | OCH3 | H | F | 74 |
| 23 | 3w | 1 | OCH3 | OCH3 | OCH3 | H | Cl | 75 |
| 24 | 3x | 1 | H | Cl | H | H | H | 79 |
| 25 | 3y | 1 | H | Cl | H | H | F | 63 |
| 26 | 3z | 1 | H | Cl | H | H | Cl | 69 |
| 27 | 3aa | 1 | H | Cl | H | H | OCH3 | 72 |
| 28 | 3ab | 1 | H | H | H | Cl | H | 75 |
| 29 | 3ac | 1 | H | H | H | Cl | F | 80 |
| 30 | 3ad | 1 | H | H | H | Cl | Cl | 79 |
| 31 | 3ae | 1 | H | H | H | Cl | OCH3 | 70 |
| 32 | 4a | 2 | H | H | H | H | H | 62 |
| 33 | 4b | 2 | H | H | H | H | F | 64 |
| 34 | 4c | 2 | H | H | H | H | Cl | 56 |
| 35 | 4d | 2 | H | H | H | H | OMe | 76 |
Plausible reaction mechanism of α-amino C(sp3)–H heteroarylation of N-aryl pyrrolidines has been previously proposed and involves single-electron oxidation of amine I followed by deprotonation to give α-amino radical III (Scheme 1).13 Radical III reacts with neutral chloro-heteroarene IV via a homolytic aromatic substitution pathway giving intermediate V. Reduction of this intermediate with IrII followed by release of chlorine anion provides final product VI.
3.2. Synthesis of benzothiazole–pyrrolidine derivatives in microfluidic reactor
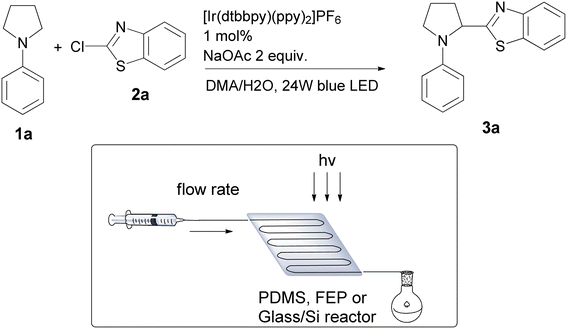
Next, we turned our attention to transferring our optimized reactions to microfluidic reactors. In general, it is very often possible to apply optimized batch conditions to microflow directly, with required optimization of the residence time and light intensity. Choice of microreactor type is important in regard to applied reaction conditions such as oxygen sensitivity, reactor transparency for various wavelengths, heat and mass transfer capability.Application of flow chemistry in the synthesis of valuable organic molecules is a well-established method.39–42 Lately, visible light promoted photoredox catalysis is one of the areas in which microfluidic chemistry has shown great potential for application and improvement of batch processes.43–47 High surface-area-to-volume ratios characteristic of these systems allow improved light utilization and better heat and mass transfer effects in photochemical transformations. Our group recently used readily available Ru catalyst and polydimethyl siloxane (PDMS) matrices in the microfluidic functionalization of tetrahydroisoquinolines (THIQs) using Manich, Strecker or alkynilation protocols.48 In addition, we showcased one of the first examples of formation and functionalization of THIQ derived α-aminoradicals under microflow conditions.49
We started by testing PDMS, Glass/Silicon and fluorinated ethylene propylene (FEP) polymer tube reactors (Table 3). Among tested reactors FEP tube reactor gave the best yields of 64% of the desired product after 4 h or residence time (Table 3, entry 2). PDMS and Glass/Si reactor gave lower yields of desired product most probably due to oxygen porosity in case of PDMS and much thinner channels and occasional clogging in Glass/Si reactor leading to uneven reaction flow and pressure drops. After 4 h of residence time there is still unreacted starting material in FEP reactor, increase of the residence time to 6 h led to full conversion of starting material and 92% yield of the desired product (Table 3, entry 5).
| Entrya | Reactor type | Reactor volume (cm3) | Residence time (h) | Yieldb (%) | % W−1 h−1 | STYc (mmol l−1 h−1) | Productivityc (mg h−1) |
|---|---|---|---|---|---|---|---|
| a Reaction conditions: N-phenyl pyrrolidine (0.5 mmol, 2 equiv.), Ir cat. (0.0025 mmol, 1 mol%) sodium acetate (0.5 mmol, 2 equiv.) and benzothiazole (0.25 mmol, 1 equiv.) were added to DMA (1 mL) and water (16.25 mmol, 65 equiv., 0.3 mL) mixture and sparged with argon for 15 minutes to remove oxygen. Solution was pumped through the microreactor using syringe pump and irradiated with 24 W blue LED for designated period of time.b Isolated yield after column chromatography.c Space-time yield (STY) and productivity are calculated according to formulae shown in the SI. | |||||||
| 1 | Batch | 1.5 | 24 | 87 | 0.15 | 7.2 | 2.54 |
| 2 | FEP | 0.5 | 4 | 64 | 0.66 | 30.8 | 2.80 |
| 3 | PDMS | 0.5 | 4 | 43 | 0.45 | 20.4 | 1.88 |
| 4 | Glass/Si | 0.025 | 4 | 21 | 0.22 | 10.8 | 0.048 |
| 5 | FEP | 0.5 | 6 | 92 | 0.64 | 29.4 | 2.69 |
Surface to volume ratio for PDMS, Glass/Si and FEP microreactor are similar, around 5000 m2 m−3 which is in the range for a microreactor device according to De Santis.50 Energy efficiency of the microreactors was also calculated and data are given in the Table 3. The energy efficiency of different light sources and reactors can be assessed through parameter defined as the ratio of percent product yield per W h−1 (Table 3).51 The best values of 0.64 of energy efficiency of light sources and the energy efficiency of the reactor are obtained for the FEP microreactor with a residence time of 6 h and LED irradiation of 24 W (Table 3, entry 5). It can be concluded that FAP microreactor has the best utilization of light and is the most energy efficient compared to two other reactors.
Besides product yield and energy efficiency, essential metrics for evaluating reaction performance in batch and microflow conditions are space-time yield (STY) and productivity. These metrics define the efficiency of the reactor and enable a comparison of the different reactors.52 STY is defined as a measure of reaction effectiveness quantified through the product mass obtained for a certain time in a certain reactor volume. The results for these parameters regarding applied microreactors in our process are summarized in Table 3. As can be seen, STY is 2–3 times higher in PDMS and FEP microreactor compared to batch system (Table 3, entries 2, 3 and 5). The glass/silicon microreactor (Table 3, entry 4) has STY value just slightly higher than the batch most probably due to reaction clogging and uneven flow in the very narrow channels. These results are in accordance with previous reports where STY is 3–4 times higher in microfluidic devices vs. batch.53,54 The best productivity was achieved in FEP microreactor, with a residence time of 4 h (Table 3, entry 2).
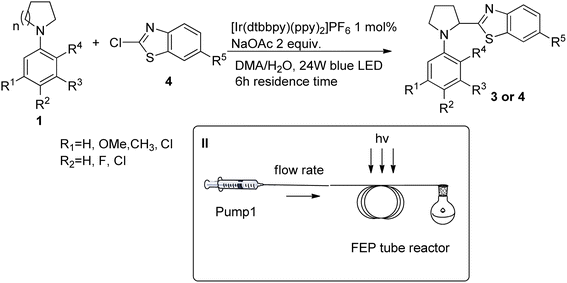
Upon reaction conditions optimization under microfluidic conditions generality of the setup was confirmed with several examples (Table 4). In al tested cases better yields were obtained compared to batch conditions. Reactions were run with 6 h residence time irradiated with 24 W blue LEDs.
| Entrya | Compound | n | R1 | R2 | R3 | R4 | R5 | Yieldb (%) | STYc (mmol l−1 h−1) | Productivityc (mg h−1) |
|---|---|---|---|---|---|---|---|---|---|---|
| a Reaction conditions: N-phenyl pyrrolidine or N-phenyl piperidine (0.5 mmol, 2 equiv.), Ir cat. (0.0025 mmol, 1 mol%) sodium acetate (0.5 mmol, 2 equiv.) and benzothiazole (0.25 mmol, 1 equiv.) were added to DMA (1 mL) and water (16.25 mmol, 65 equiv., 0.3 mL) mixture and sparged with argon for 15 minutes to remove oxygen. Solution was pumped through the microreactor using syringe pump and irradiated with 24 W blue LED for designated period of time.b Isolated yield after column chromatography.c Space-time yield (STY) and productivity are calculated according to formulae shown in the SI. | ||||||||||
| 1 | 3a | 1 | H | H | H | H | H | 92 | 29.4 | 2.69 |
| 2 | 3b | 1 | H | H | H | H | F | 81 | 25.8 | 2.52 |
| 3 | 3c | 1 | H | H | H | H | Cl | 87 | 27.6 | 2.85 |
| 4 | 3e | 1 | Me | H | H | H | H | 85 | 27.1 | 2.60 |
| 5 | 3g | 1 | H | Me | H | H | Cl | 89 | 28.2 | 3.05 |
| 6 | 3o | 1 | H | Me | Me | H | Cl | 93 | 30.1 | 3.32 |
| 7 | 3u | 1 | OMe | OMe | OMe | H | H | 91 | 29.4 | 3.51 |
| 8 | 3y | 1 | Cl | H | H | H | F | 85 | 27.1 | 2.95 |
| 9 | 3z | 1 | Cl | H | H | H | Cl | 88 | 28.2 | 3.20 |
| 10 | 3aa | 1 | Cl | H | H | H | OMe | 92 | 29.4 | 3.31 |
| 11 | 3ab | 1 | H | H | H | Cl | H | 84 | 27.0 | 2.75 |
| 12 | 4a | 2 | H | H | H | H | H | 79 | 25.2 | 2.42 |
3.3. Assessment of log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) P, blood–brain permeability and the biological activity of the synthesized benzothiazole derivatives
P, blood–brain permeability and the biological activity of the synthesized benzothiazole derivatives
In the early stages of drug discovery and development, particularly for compounds intended to treat neurological disorders, adequate lipophilicity and blood–brain barrier (BBB) permeability represent essential prerequisites. Accordingly, log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P, as an indicator of compound lipophilicity, together with the BBB permeability of the synthesized compounds, was theoretically assessed using SwissADME software, and the results are summarized in Table 5. All compounds displayed log
P, as an indicator of compound lipophilicity, together with the BBB permeability of the synthesized compounds, was theoretically assessed using SwissADME software, and the results are summarized in Table 5. All compounds displayed log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P values within the optimal range and were predicted to cross the BBB,52,55 suggesting their potential to inhibit cholinesterase enzymes, which are closely associated with Alzheimer's disease. On this basis, the synthesized benzothiazole derivatives were further evaluated for their inhibitory activity against acetyl- and butyrylcholinesterase, and the corresponding results are presented in Table 5. Overall, the compounds predominantly exhibited selectivity toward BuChE, with IC50 values in the micromolar range, whereas lower or no inhibitory activity against AChE was generally observed. An exception was found among the chloro-substituted derivatives, which displayed inhibitory activity toward both enzymes and yielded the most potent AChE inhibitor, compound 3o.
P values within the optimal range and were predicted to cross the BBB,52,55 suggesting their potential to inhibit cholinesterase enzymes, which are closely associated with Alzheimer's disease. On this basis, the synthesized benzothiazole derivatives were further evaluated for their inhibitory activity against acetyl- and butyrylcholinesterase, and the corresponding results are presented in Table 5. Overall, the compounds predominantly exhibited selectivity toward BuChE, with IC50 values in the micromolar range, whereas lower or no inhibitory activity against AChE was generally observed. An exception was found among the chloro-substituted derivatives, which displayed inhibitory activity toward both enzymes and yielded the most potent AChE inhibitor, compound 3o.
| Compound | Structure | Swiss ADME | IC50,a (µM) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| n | R1 | R2 | R3 | R4 | R5 | log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P P |
BBB | AChE | BuChE | |
a n.a. – no activity; data given represent mean ± standard error (SE) values of the results from three independent experiments performed in triplicates. Predicted log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P and BBB permeability for synthesized benzothiazole derivatives obtained using Swiss ADME. P and BBB permeability for synthesized benzothiazole derivatives obtained using Swiss ADME. |
||||||||||
| 3a | 1 | H | H | H | H | H | 3.42 | Yes | >10 | 6.17 ± 0.98 |
| 3b | 1 | H | H | H | H | F | 3.8 | Yes | >10 | >10 |
| 3c | 1 | H | H | H | H | Cl | 3.92 | Yes | >10 | 2.74 ± 1.09 |
| 3d | 1 | H | H | H | H | OCH3 | 3.04 | Yes | n.a. | 3.83 ± 0.47 |
| 3e | 1 | H | CH3 | H | H | H | 3.65 | Yes | >10 | >10 |
| 3f | 1 | H | CH3 | H | H | F | 4.04 | Yes | >10 | >10 |
| 3g | 1 | H | CH3 | H | H | Cl | 4.15 | Yes | 0.95 ± 0.08 | 2.96 ± 0.40 |
| 3h | 1 | H | CH3 | H | H | OCH3 | 3.27 | Yes | >10 | >10 |
| 3i | 1 | H | H | CH3 | H | H | 3.65 | Yes | >10 | >10 |
| 3j | 1 | H | H | CH3 | H | F | 4.04 | Yes | >10 | >10 |
| 3k | 1 | H | H | CH3 | H | Cl | 4.15 | Yes | 5.65 ± 0.05 | 3.96 ± 0.06 |
| 3l | 1 | H | H | CH3 | H | OCH3 | 3.27 | Yes | >10 | >10 |
| 3m | 1 | H | CH3 | CH3 | H | H | 3.89 | Yes | >10 | >10 |
| 3n | 1 | H | CH3 | CH3 | H | F | 4.27 | Yes | >10 | >10 |
| 3o | 1 | H | CH3 | CH3 | H | Cl | 4.38 | Yes | 0.78 ± 0.08 | 2.72 ± 0.02 |
| 3p | 1 | H | CH3 | CH3 | H | OCH3 | 3.50 | Yes | >10 | >10 |
| 3q | 1 | H | OCH3 | H | H | H | 3.04 | Yes | n.a. | >10 |
| 3r | 1 | H | OCH3 | H | H | F | 3.43 | Yes | >10 | >10 |
| 3s | 1 | H | OCH3 | H | H | Cl | 3.54 | Yes | >10 | >10 |
| 3t | 1 | H | OCH3 | H | H | OCH3 | 2.62 | Yes | n.a. | >10 |
| 3u | 1 | OCH3 | OCH3 | OCH3 | H | H | 2.36 | Yes | >10 | >10 |
| 3v | 1 | OCH3 | OCH3 | OCH3 | H | F | 2.74 | Yes | >10 | >10 |
| 3w | 1 | OCH3 | OCH3 | OCH3 | H | Cl | 2.85 | Yes | >10 | >10 |
| 3x | 1 | H | Cl | H | H | H | 3.92 | Yes | >10 | >10 |
| 3y | 1 | H | Cl | H | H | F | 4.31 | Yes | >10 | 10 |
| 3z | 1 | H | Cl | H | H | Cl | 4.42 | Yes | >10 | >10 |
| 3aa | 1 | H | Cl | H | H | OCH3 | 3.54 | Yes | >10 | 10 |
| 3ab | 1 | H | H | H | Cl | H | 3.92 | Yes | 2.81 ± 0.05 | >10 |
| 3ac | 1 | H | H | H | Cl | F | 4.31 | Yes | 10 | >10 |
| 3ad | 1 | H | H | H | Cl | Cl | 4.42 | Yes | >10 | >10 |
| 3ae | 1 | H | H | H | Cl | OCH3 | 3.54 | Yes | 10 | 10 |
| 4a | 2 | H | H | H | H | H | 3.65 | Yes | >10 | >10 |
| 4b | 2 | H | H | H | H | F | 4.04 | Yes | >10 | >10 |
| 4c | 2 | H | H | H | H | Cl | 4.15 | Yes | >10 | 8.93 ± 0.05 |
| 4d | 2 | H | H | H | H | OCH3 | 3.27 | Yes | >10 | >10 |
Structure-activity relationship analysis revealed that the presence of chlorine in the benzothiazole moiety significantly influenced biological activity. Specifically, chlorination led to a twofold increase in BuChE inhibitory potency, as observed for compound 3c compared with its non-chlorinated analogue, compound 3a. Furthermore, variations in substituents (CH3, Cl, and OCH3) on the aryl moiety of the pyrrolidine part of the Cl-substituted derivatives resulted in distinct biological activity profiles. Among these, CH3 substitution markedly enhanced inhibitory potency and additionally enabled AChE inhibition, yielding compounds with dual inhibitory properties (3g, 3k, and 3o). Notably, the synergistic effect of two CH3 groups proved particularly important, resulting in potent inhibition of both enzymes, with IC50 values for AChE below 1 µM, as exemplified by compound 3o. In contrast, the introduction of Cl at the ortho position of the aryl moiety led to selective AChE inhibition without affecting BuChE activity (compound 3ab).
In contrast to the chlorinated analogues, fluorine substitution in the benzothiazole ring led to a decrease in BuChE inhibitory activity compared with compound 3a, irrespective of the substituents on the aryl moiety of the pyrrolidine unit. Moreover, no inhibitory activity toward AChE was detected for the fluorinated derivatives in any case. On the other hand, introduction of a methoxy group into the non-substituted compound 3a resulted in an approximately 1.5-fold increase in BuChE inhibitory potency (IC50 = 6.17 µM for 3a vs. 3.83 µM for compound 3d). However, further structural modification of compound 3d on the aryl moiety of the pyrrolidine part did not yield biologically active derivatives.
Evaluation of the biological activity of piperidine-based benzothiazole derivatives against cholinesterase enzymes revealed generally low inhibitory activity, with IC50 values higher than 10 µM, except for compound 4c, which contains a chlorine substituent on the benzothiazole moiety. Comparison of the IC50 value of compound 4c with that of its structural analogue, compound 3c, suggests that the size of the heterocyclic ring (piperidine vs. pyrrolidine) influences inhibitory activity. The pyrrolidine ring, likely due to its conformational properties or smaller dimensions, appears to confer enhanced inhibitory potency.
Taken together, these results indicate that the most biologically active synthesized benzothiazole aryl pyrrolidines were those containing a 6-Cl-substituted benzothiazole core combined with CH3 groups on the aryl moiety. This highlights the important role of chloride involvement in enzyme-ligand interactions, as well as the significant contribution of methyl substituents to enhanced binding and inhibitory activity.
4. Conclusion
In conclusion, we have developed robust synthetic protocol for the visible light promoted photoredox functionalization of N-aryl pyrrolidines and piperidines with benzothiazoles in the presence of water. 35 compounds were synthesized under batch conditions in good yields showing wide applicability of this method. These hybrid molecules were tested in inhibition assays with cholinergic enzymes important in the development of neurological disorders such as Alzheimer's disease and showed low micromolar activities and good potential for further improvement of the inhibition potentials. Several derivatives showed selectivity towards BuChE enzyme. These hybrid molecules represent a new class of neurologicaly active molecules. In silico ADME study supports this conclusion since most active molecules are predicted to be able to pass blood–brain barrier which is a prerequisite for molecules intended for use in the treatment of neurological disorders. Furthermore, optimized conditions were transferred to microfluidic conditions allowing access to practical and greener synthetic pathways for the synthesis of our compounds. Good to excellent yields were obtained using FAP tube custom made reactors.Author contributions
Ana Filipović: investigation, data curation, formal analysis, visualization, Marija Vučkovski: investigation, data curation, formal analysis, visualization, Dunja Mićović: investigation, data curation, formal analysis, visualization, Aleksandra Bondžić: resource, writing – review and editing, methodology, conceptualization, funding acquisition, supervision, resources, Bojan Bondžić: conceptualization, methodology, writing – original draft, writing – review & editing, supervision, project administration, funding acquisition, resources.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
All relevant data supporting this article are included within the manuscript and supplementary information (SI). Supplementary information: detailed experimental procedures and characterization data for the synthesized compounds, including 1H, 13C NMR and FT-IR spectra. See DOI: https://doi.org/10.1039/d6ra01041e.Acknowledgements
This research was supported by the Science Fund of the Republic of Serbia, Grant No. 7451, Plasmonic-based light harvesting for photocatalytic microfluidic devices – PlasmaHarvest. In addition, this research has been financially supported by the Ministry of Science, Technological Development, and Innovation of the Republic of Serbia (contract numbers: 451-03-33/2026-03/200017, 451-03-33/2026-03/200026 and 451-03-33/2026-03/200168).References
- M. A. Ischay, M. E. Anzovino, J. Du and T. P. Yoon, J. Am. Chem. Soc., 2008, 130, 12886–12887 CrossRef CAS PubMed.
- D. A. Nicewicz and D. W. C. MacMillan, Science, 2008, 322, 77–80 CrossRef CAS PubMed.
- N. J. W. Straathof, B. J. P. Tegelbeckers, V. Hessel, X. Wang and T. Noël, Chem. Sci., 2014, 5, 4768–4773 RSC.
- A. Talla, B. Driessen, N. J. W. Straathof, L.-G. Milroy, L. Brunsveld, V. Hessel and T. Noël, Adv. Synth. Catal., 2015, 357, 2180–2186 CrossRef CAS.
- M. März, J. Chudoba, M. Kohout and R. Cibulka, Org. Biomol. Chem., 2017, 15, 1970–1975 RSC.
- G. Laudadio, Y. Deng, K. van der Wal, D. Ravelli, M. Nuño, M. Fagnoni, D. Guthrie, Y. Sun and T. Noël, Science, 2020, 369, 92–96 CrossRef CAS PubMed.
- S. Kamijo, T. Hoshikawa and M. Inoue, Org. Lett., 2011, 13, 5928–5931 CrossRef CAS PubMed.
- T. Hoshikawa and M. Inoue, Chem. Sci., 2013, 4, 3118–3123 RSC.
- J. N. Capilato, C. R. Pitts, R. Rowshanpour, T. Dudding and T. Lectka, J. Org. Chem., 2020, 85, 2855–2864 CrossRef CAS PubMed.
- J. Xie, M. Rudolph, F. Rominger and A. S. K. Hashmi, Angew. Chem. Int. Ed., 2017, 56, 7266–7270 CrossRef CAS PubMed.
- D. Mazzarella, G. E. M. Crisenza and P. Melchiorre, J. Am. Chem. Soc., 2018, 140, 8439–8443 CrossRef CAS PubMed.
- A. McNally, C. K. Prier and D. W. C. MacMillan, Science, 2011, 334, 1114 CrossRef CAS PubMed.
- C. K. Prier and D. W. C. MacMillan, Chem. Sci., 2014, 5, 4173–4178 RSC.
- A. Singh, A. Arora and J. D. Weaver, Org. Lett., 2013, 15, 5390–5393 CrossRef CAS PubMed.
- A. Lipp, G. Lahm and T. Opatz, J. Org. Chem., 2016, 81, 4890–4897 CrossRef CAS PubMed.
- E. Bergamaschi, C. Weike, V. J. Mayerhofer, I. Funes-Ardoiz and C. J. Teskey, Org. Lett., 2021, 23, 5378–5382 CrossRef CAS PubMed.
- C. Bhat and S. G. Tilve, RSC Adv., 2014, 4, 5405–5452 RSC.
- E. Vitaku, D. T. Smith and J. T. Njardarson, J. Med. Chem., 2014, 57, 10257–10274 CrossRef CAS PubMed.
- D. B. Tiz, L. Bagnoli, O. Rosati, F. Marini, C. Santi and L. Sancineto, Pharmaceutics, 2022, 14, 2538 CrossRef PubMed.
- P. I. Dalko and L. Moisan, Angew. Chem., Int. Ed., 2004, 43, 5138–5175 CrossRef CAS PubMed.
- K. Higashiyama, H. Inoue and H. Takahashi, Tetrahedron, 1994, 50, 1083–1092 CrossRef CAS.
- S. Llopis, T. García, Á. Cantín, A. Velty, U. Díaz and A. Corma, Catal. Sci. Technol., 2018, 8, 5835–5847 RSC.
- C. S. Demmer and L. Bunch, Eur. J. Med. Chem., 2015, 97, 778–785 CrossRef CAS PubMed.
- M. Kokane, D. Bhosale, D. Raut, P. Hadimani, A. Narale, S. Chaudhari, P. Choudhari and A. Lawand, Biochem. Biophys. Res. Commun., 2025, 793, 152998 CrossRef CAS PubMed.
- F. A. A. A. Mohamed, D. Kłopotowska, M. K. Abd El-Gaber, A. S. Aboraia, J. Wietrzyk and A. F. Youssef, Bioorg. Chem., 2025, 166, 109066 CrossRef CAS PubMed.
- M. Usman, A. Alam, Zainab, M. Khan, A. A. Elhenawy, M. Ayaz, M. M. Alanazi, A. Latif, S. A. A. Shah, M. Ali and M. Ahmad, J. Mol. Struct., 2025, 1319, 139504 CrossRef CAS.
- M.-H. Nam, M. Park, H. Park, Y. Kim, S. Yoon, V. S. Sawant, J. W. Choi, J.-H. Park, K. D. Park, S.-J. Min, C. J. Lee and H. Choo, ACS Chem. Neurosci., 2017, 8, 1519–1529 CrossRef CAS PubMed.
- D. H. Dawood and M. M. Anwar, Eur. J. Med. Chem., 2025, 287, 117331 CrossRef CAS PubMed.
- R. A. Mohamed-Ezzat and G. H. Elgemeie, RSC Adv., 2025, 15, 41724–41832 RSC.
- L. Ackermann, S. Barfusser and J. Pospech, Org. Lett., 2010, 12, 724–727 CrossRef CAS PubMed.
- C. Bharkavi, S. V. Kumar, M. A. Ali, H. Osman, S. Muthusubramanian and S. Perumal, Bioorg. Med. Chem. Lett., 2016, 24, 5873–5883 CrossRef CAS PubMed.
- Y. Huang, W. Min, Q. W. Wu, J. Sun, D. H. Shi and C. G. Yan, New J. Chem., 2018, 42, 16211–16216 RSC.
- P. K. Choubey, A. Tripathi, P. Sharma and S. K. Shrivastava, Bioorg. Med. Chem., 2020, 28, 115721 CrossRef CAS PubMed.
- R. Min, W. Wu, M. Wang, Y. Li, Z. Zhang and L. Chen, Molecules, 2019, 24, 1901 CrossRef CAS PubMed.
- D. Jovanović, A. Filipović, R. Suručić, L. Korićanac, J. Žakula, B. P. Bondžić and A. M. Bondžić, Int. J. Biol. Macromol., 2025, 319, 145524 CrossRef PubMed.
- M. Vučkovski, A. Filipović, M. Jadranin, L. Korićanac, J. Žakula, B. P. Bondžić and A. M. Bondžić, Int. J. Mol. Sci., 2024, 25, 13076 CrossRef PubMed.
- D. Jovanović, A. Filipović, G. Janjić, T. Lazarević-Pašti, Z. Džambaski, B. P. Bondžić and A. M. Bondžić, Int. J. Mol. Sci., 2024, 25, 1033 CrossRef PubMed.
- A. Daina, O. Michielin and V. Zoete, Sci. Rep., 2017, 7, 42717 CrossRef PubMed.
- K. Geyer, J. D. C. Codée and P. H. Seeberger, Chem. Eur. J., 2006, 12, 8434–8442 CrossRef CAS PubMed.
- P. Watts and C. Wiles, Chem. Commun., 2007, 443–467 RSC.
- D. Webb and T. F. Jamison, Chem. Sci., 2010, 1, 675–680 RSC.
- C. Stephenson, T. Yoon and D. W. C. MacMillan, Visible Light Photocatalysis in Organic Chemistry, Wiley-VCH, Weinheim, 2018 Search PubMed.
- J. W. Beatty and C. R. J. Stephenson, J. Am. Chem. Soc., 2014, 136, 10270–10273 CrossRef CAS PubMed.
- Y. Su, N. J. W. Straathof, V. Hessel and T. Noël, Chem. Eur. J., 2014, 20, 10562–10589 CrossRef CAS PubMed.
- E. M. Schuster and P. Wipf, Isr. J. Chem., 2014, 54, 361–370 CrossRef CAS.
- F. Lévesque and P. H. Seeberger, Org. Lett., 2011, 13, 5008–5011 CrossRef PubMed.
- A. C. Gutierrez and T. F. Jamison, Org. Lett., 2011, 13, 6414–6417 CrossRef CAS PubMed.
- A. Filipović, Z. Džambaski, D. Vasiljević-Radović and B. P. Bondžić, Org. Biomol. Chem., 2021, 19, 2668–2675 RSC.
- A. Filipović, Z. Džambaski, A. M. Bondžić and B. P. Bondžić, Photochem. Photobiol. Sci., 2023, 22, 2259–2270 CrossRef PubMed.
- P. De Santis, L.-E. Meyer and S. Kara, React. Chem. Eng., 2020, 5, 2155–2184 RSC.
- A. Sugimoto, T. Fukuyama, Y. Sumino, M. Takagi and I. Ryu, Tetrahedron, 2009, 65, 1593–1598 CrossRef CAS.
- C. A. Hone and C. O. Kappe, Chem.–Methods, 2021, 1, 454–467 CrossRef CAS.
- O. Shvydkiv, A. Yavorskyy, S. B. Tan, K. Nolan, N. Hoffmann, A. Youssef and M. Oelgemöller, Photochem. Photobiol. Sci., 2011, 10, 1399–1404 CrossRef CAS PubMed.
- A. Yavorskyy, O. Shvydkiv, K. Nolan, N. Hoffmann and M. Oelgemöller, Tetrahedron Lett., 2011, 52, 278–280 CrossRef CAS.
- M. Markowicz-Piasecka, A. Markiewicz, P. Darłak, J. Sikora, S. K. Adla, S. Bagina and K. M. Huttunen, Neurotherapeutics, 2022, 19, 942–976 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |