DOI:
10.1039/D6RA00997B
(Paper)
RSC Adv., 2026,
16, 12282-12294
Build-up shielding-factors, and physical & mechanical properties of borosilicate glasses with varied CeO2 contents
Received
4th February 2026
, Accepted 17th February 2026
First published on 4th March 2026
Abstract
Heavy-metal borosilicate glasses of composition 10BaO–10CdO–12PbO–3Al2O3–5SiO2–(Z)CeO2–(60 − Z)B2O3 (where Z = 0.5–4.0 mol%) were synthesized via a melt-quenching technique to systematically evaluate the influence of CeO2 incorporation on their γ-ray shielding and mechanical characteristics. XRD and XPS confirmed amorphous glass formation with mixed Ce3+/Ce4+ oxidation states (42%/58%), while SEM-EDX evidenced compositional homogeneity and densification with increasing Ce content. Density rose by 8.55%, molar volume decreased by 4.21%, and oxygen packing density increased by 3.48%, signifying structural compaction. Mechanical assessments, based on the Makishima–McKenzie approach, showed that Young's modulus and bulk modulus increased from 74.53 to 77.43 GPa and 51.99 to 56.26 GPa, respectively, up to 2 mol% CeO2, reflecting enhanced rigidity. Gamma-ray attenuation parameters determined using the Py-MLBUF platform revealed a substantial improvement in linear attenuation coefficient (LAC) by 21.75%, and a reduction in half-value layer (HVL) by 13.64% at 0.015 MeV, confirming superior shielding efficiency. The double-layer exposure buildup factors (DLEBF) reached a maximum of 6.79 at 0.05 MeV for GC2–GC0, demonstrating effective secondary photon suppression. These results highlight that moderate CeO2 substitution enhances glass compactness, mechanical resilience, and γ-ray attenuation, establishing Ce-doped borosilicate glasses as robust candidates for advanced radiation shielding applications.
1 Introduction
In recent times, rapid and successive development in many aspects of industrial, agricultural and medical life has become an inevitable necessity. In addition, there is an urgent need for energy, which is the backbone of technological development, and providing new sources for it has become extremely important. Today, the use of various types of radioactive materials in many aspects of life, including medicine, military science, space science, and power generation, in addition to many different technological applications, has become inevitable. Therefore, the preparation and development of new types of materials with unique properties for radiation protection has now become an important and vital branch of materials science to avoid the health risks associated with ionizing radiation.1–3 The efficiency and effectiveness of radiation protection materials can be estimated according to their ability to scatter or absorb ionizing radiation to reduce its danger.4,5 In the field of radiation shielding materials, many different materials have been used, the most common being concrete and lead. However, in reality, these materials have not met the necessary requirements, resulting in numerous health and environmental effects, in addition to their limited protective efficiency and their degradation over time.1,6,7 It also requires a significant curing time and specific conditions for optimal performance.1,6,8
In the field of materials science for protection from harmful radiation, glass has been greatly appreciated for its promising physical, chemical and optical properties, in addition to its unlimited compositions that meet the changing conditions and requirements of protection.9–11 Glass systems based on silicates (SiO2), phosphates (P2O5), tellurium oxide (TeO2), borate (B2O3), and a mixture of two or more of these types are the basic nucleus for forming a glass network that is resistant to harmful rays. Of these several glass systems, borate-based glass is the most important and widely used because of its high neutron thermal cross-section and excellent shielding capabilities for both neutron shielding and gamma ray attenuation. The borate glass network can encapsulate various radioactive elements and stabilize them in a stable, non-reactive form.12–14 Moreover glass optical qualities and radiation shielding capabilities are improved by the adding of subordinate components like heavy metal oxides like ZnO, CdO, BaO and PbO. Recent studies have shown that adding CdO, BaO and lead in varying quantities to borate-based glass modifies the atoms in the internal structure of the glass network and improving its optical properties.15–17 Furthermore, these elements have strong absorption of ionizing radiation due to their high density. Lanthanide-containing glasses are highly researched and praised for having a low vibration frequency.18 Cerium (Ce) ion is part of the lanthanide rare earth (RE) ion group and displays promising properties linked to its surface electronic configuration.19,20 Cerium oxide (CeO2) is frequently utilized in glass structures because of its beneficial optical, physical, and radiation-shielding qualities.21,22 To improve shielding glass's ability to absorb radiation, especially gamma and X-ray radiation, cerium oxide (CeO2) is utilized. It is added to glass compositions to enhance their half-value layer, radiation protection efficiency, and mass attenuation coefficient, making them more potent shields. Additionally, CeO2 can enhance the glass's optical qualities. The ratios of cerium ions in the host glass composition affect the concentration of the two oxidation states (Ce4+/Ce3+) in which cerium can be integrated into the glassy matrix.19,23 Although several studies have reported the structural and compositional properties of modified glass systems, a systematic investigation combining structural (XRD), surface chemical (XPS), and microstructural/compositional (SEM-EDS) analyses for the present glass composition and modification range remains limited. The novelty and involvement of the present work lie in providing a comprehensive correlation between compositional modifications and the resulting structural, surface, and elemental characteristics of the glass samples (GC0–GC4), supported by experimental evidence. In addition, the objectives of the present study have been clearly stated in a point-wise manner at the end of the Introduction section to improve clarity. In the current studies, the assortment of 10BaO–10CdO–12PbO–5SiO2–3Al2O3–60B2O3 base glass for radiation shielding applications is predicated on their remarkable optical and shielding qualities as well as their exceptional resistance to thermal shocks. Therefore, this work is aimed at doping this unique glass system with varying proportions of cerium oxide (0.5, 1.0, 2.0, and 4.0 mol%) instead of B2O3 to investigate how it affects the optical and radiation shielding properties of the prepared glasses. Therefore, the objectives of the present work are as follows: (i) to synthesize and modify glass samples with controlled compositions (GC0–GC4), (ii) to investigate the structural properties using XRD, (iii) to examine surface chemical states and bonding environments using (XPS), (iv) to analyze the microstructure and semi-quantitative elemental composition using SEM-EDS, and (v) to correlate compositional modifications with the observed structural and compositional changes.
2 Materials and methods
2.1 Glass synthesis
Barium cadmium lead aluminum borosilicate glasses doped with CeO2 as a rare earth elements with the composition 10BaO–10CdO–12PbO–3Al2O3–5SiO2–(Z)CeO2–(60 − Z)B2O3 glass system (where Z = 0, 0.5, 1, 2, and 4 mol%) were effectively synthesized using the melt quenching technique, as shown in Table 1. Various highly pure chemical reagents of barium carbonate (BaCO3) (99.50%, Loba-Chemie), cadmium carbonate (CdCO3), aluminium oxide (Al2O3) (99.90%, Alfa Aesar), Boric acid (H3BO3) (ACS reagent, ≥98% Sigma-Aldrich), lead sesquioxide (Pb2O3), cerium oxide (CeO2) oxide with purities of 99.98, and quartz (SiO2) (99.99%, Alfa Aesar) were weighed to prepare the glass batches. The oxides were thoroughly mixed in an agate mortar to form homogeneous mixtures before melting, which took 90 min in platinum crucibles at temperatures of about 1250 °C in an electrical furnace. The molten glass was stirred several times to remove air bubbles and increase the consistency of the melt. The molten glass was then poured into preheated steel molds and placed in an annealing furnace to relieve stress. The annealing process took place at temperatures around 400 °C for two hours in a muffle furnace, followed by cooling to room temperature at a rate of 1 °C per minute.
Table 1 The elemental fractional abundance and density of the prepared samples
| Sample ID |
Glass oxide constitutes (mol%) |
| BaO |
CdO |
PbO |
SiO2 |
Al2O3 |
CeO2 |
B2O3 |
| GC0 |
10 |
10 |
12 |
5 |
3 |
0 |
60 |
| GC0.5 |
10 |
10 |
12 |
5 |
3 |
0.5 |
59.5 |
| GC1 |
10 |
10 |
12 |
5 |
3 |
1 |
59 |
| GC2 |
10 |
10 |
12 |
5 |
3 |
2 |
58 |
| GC4 |
10 |
10 |
12 |
5 |
3 |
4 |
56 |
2.2 Experimental techniques
XPS measurements were performed using a Thermo Scientific K-Alpha spectrometer (Thermo Fisher Scientific, USA) equipped with a monochromatic Al Kα X-ray source (hν = 1486.6 eV). The analysis was conducted under ultra-high vacuum conditions (10−9 mbar) with a spot size of 400 µm. Survey spectra were recorded with a pass energy of 200 eV, while high-resolution spectra were acquired using a pass energy of 50 eV. Binding energies were calibrated using the C 1s peak at 284.8 eV. Peak deconvolution was carried out after background subtraction using a Shirley background, and curve fitting was performed with appropriate Gaussian–Lorentzian functions. X-ray diffraction (XRD) analysis was carried out using a Bruker D8 Discover diffractometer (Bruker AXS GmbH, Germany) equipped with a Cu target providing CuKα radiation (λ = 1.5406 Å). The instrument was operated at an accelerating voltage of 40 kV and a current of 40 mA. Data were collected over the selected 2θ range using a step size of 0.02°. SEM observations were carried out using a Prisma E-SEM instrument (Thermo Fisher Scientific Inc., Waltham, MA, USA) equipped with an energy-dispersive X-ray spectroscopy (EDX, Ultradry EDX detector) detector. The analyses were performed at an accelerating voltage of 20–30 kV, with a counting time of 60–120 s and an electron beam diameter of approximately 1–2 µm.
2.3 Density calculations
The density was determined using Archimedes' Principle, as expressed in eqn (1).| |
 | (1) |
The symbol ρ represents the density of the glass sample, whereas (Wair) denotes the weight of the glass sample in air. The variable (Wxylene) represents the weight of the glass sample while immersed in xylene, and ρxylene is the density of see xylene (??).
2.4 Mechanical study
The primary challenge in the Makishima–McKenzie principle is utilizing the following relations to determine the total ionic packing density (Vt) and the total dissociation energy per unit volume (Gt) for the glass system oxides:| |
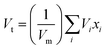 | (2) |
| |
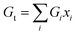 | (3) |
The packing density factor (Vi) and the dissociation energy per unit volume of the supplied glass systems oxides (Gi) are represented by these variables.24–26 The synthesized specimens' elastic modulus was designed to theoretically extend the Makishima–McKenzie model.24–26
2.5 % of change
To investigate the change % on changing from A value to B value, the eqn (4) was used| |
 | (4) |
2.6 Gamma shielding
Py-MLBUF27,28 were used to calculate linear attenuation coefficient (LAC), mass attenuation coefficient (MAC), half-value layer (HVL), tenth value layer (TVL) and effective atomic number for attenuation (Zeff). The atomic interaction cross section for attenuation (σ-atomic) and electron interaction cross section for attenuation (σ-electron) against energy were also investigated. The buildup factors are correction variables that take into consideration the impact of secondary particles in the materials and scattered radiation. The exposure buildup factor (EBF) and energy absorption buildup factor (EABF) were investigated for single layer and the double layer buildup factors (DLEABF and DLEBF) were examined for double layer shield. The combination of double layers was set as AB and BA with all glass samples as 1st and then as 2nd layer.
Py-MLBUF platform was validated in detailed for 32 materials including standard, plastics and polymers, pure-compounds, fatty-acids, building-materials.27,29 Fundamental parameters including atomic weights and gamma-photons’ cross-sections for the first 92 elements of the periodic table were obtained from NIST: XCOM and XAAMDI30,31 and the standard values of GPF-parameters for 23 elements were obtained from the ANS-standard.32 The effects of photons scattering while passing through a Gamma-ray Shielding Parameters (GSP) are described by BUF. The online platforms supports up to 15 layers, before applying the overestimation-correction in the Py-MLBUF this led to some errors in the calculation. In current study only single and double layers were investigated for more accurate results.33
The total MAC (µ·ρ−1)Total for the material is the sum of the coefficients of photoelectric absorption (µ·ρ−1)pe, Compton scattering (µ·ρ−1)cs and pair production (µ·ρ−1)pp (eqn (5)).34
| | |
(µ·ρ−1)Total = (µ·ρ−1)pe + (µ·ρ−1)cs + (µ·ρ−1)pp
| (5) |
where
µ is LAC and
ρ is the density of the selected sample. The true mass absorption coefficient is given by
eqn (6)| |
 | (6) |
where
E is the incident photon energy. Using
eqn (7) Zeq can be interpolation whose ratio (
µ·
ρ−1)
cs/(
µ·
ρ−1)
Total lies in between two successive ratios of elements.
| |
 | (7) |
where
Z1 and
Z2 are the elemental atomic numbers,
R1 and
R2 corresponding to the ratios, respectively, and
R is the ratio for the selected material at the specified energy;
R1 <
R <
R2.
The equivalent atomic numbers are averaged over the 25 incident photon-E and the atomic number so obtained is treated as the effective atomic number Zeff of that sample for given energy range (eqn (8))
| |
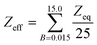 | (8) |
The buildup factors were calculated according to eqn (9) and eqn (9) using GP within the energy spectrum of 0.015–15 MeV and up to a penetration depth of 40 mean free paths (mfp)
| |
 | (9) |
| | |
B(E, x) = 1 + (b − 1)x for K = 1
| (10) |
The expression K(E, x) denotes the photon dose multiplication factor, which is determined by eqn (11) for x ≤ 40 mfp.
| |
 | (11) |
where
b is the accumulation factor at 1 mfp,
E denotes the energy of the incident photon, and
x refers to the distance from the source to the detector within the medium, measured in units of mfp.
DLEABF and DLEBF are required for calculating the buildup factors for double-layered shields. Eqn (12) and (13) for estimating DLEBF and DLEABF.
| |
 | (12) |
| |
 | (13) |
Eqn (12) and (13) B(X1, X2) refers to DLEBF (or DLEABF) for the double-layered shields with X1 and X2 as mfp of first and second layer's, respectively.
3 Results and discussion
3.1 XPS
The XPS survey spectra of GCe0 and GCe4 are shown in Fig. 1a and b. The spectra of all samples showed the presence of Al-2p (72.08–72.25 eV), Si-2p (94.08–102.98), Pb-4f (139.07–143.13 eV), B-1s (192.01–195.00 eV), C-1s (285.23–287.94 eV), Cd-3d (412.41–416.08), O-1s (531.97–534.18 eV), and Ba-3d (780.38–782.19 eV). The extra peak at 901.94 eV in the spectrum of GCe4 was attributed to Ce-3d. These results support the proposed structures. The fitting of XPS high-resolution spectra (HRSf) was good for most elements except aluminum, which showed a noisy unclear spectrum due to its low content. HRSf of silicon are shown in Fig. 1c and d. The absence of Si0 is supported by the presence of all peaks above 100 eV.35 The spectrum of GCe0 show two doublets at (100.55, 101.13 eV) and (101.84, 102.24 eV) which were attributed to Si1+ (O–Si–Si3, 2p3/2 and 2p1/2) and Si3+ (O3–Si–Si, 2p3/2 and 2p1/2) with a shift from Si0 (2p3/2) of 1.25 and 2.54 eV, respectively. The ratio of Si3+ may be calculated from eqn (14).36–38 The average oxidation sate may be calculated as “1 × 0.55 + 3 × 0.45 = 1.91” where 0.55 and 0.45 are the ratio of Si1+ and Si3+, respectively. The spectrum of GCe0 show on doublet at 100.99 and 101.39 which were attributed to Si2+ (SiO) 2p3/2 and 2p1/2, respectively a Shift from Si0 (2p3/2) of 1.69 eV.36–38| |
 | (14) |
 |
| | Fig. 1 (a and b) survey spectra, (c and d) HRSf Si, (e and f) HRSf Pb, (g and h) HRSf B and (i and j) HRSf C of GCe0 and GCe4. | |
HRSf of lead showed a doublet @ 142.95–144.29 and 139.64–138.05 eV which may assigned to Pb-4f5/2 and Pb-4f7/2, respectively for PbO.39–41 The extra peak appearing around 414 eV in Fig. 2a and b was assigned to Pb-4d5/2which further support forming Pb–O bonds.39,42 HRSf of B are shown in Fig. 1g and h. The absence of any peak around 187 eV indicate the absence of elemental Boron.43 Peaks in 188.95–183.54 eV region were attributed to B–B44 while those in 191.11–191.47 and 192.27–192.99 eV corresponding to B2O3 and substitutional B that occupy O sites, respectively.40 Peaks in 193.48–194.84 eV region were attributed B![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in B2O3.45 HRSf of C are shown in Fig. 1i and j. The peaks around 284 and 286 eV were assigned to sp2 hybridized carbon (C
O in B2O3.45 HRSf of C are shown in Fig. 1i and j. The peaks around 284 and 286 eV were assigned to sp2 hybridized carbon (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) and C–OH, respectively.46,47 The peaks @ 285.21, 288.45, and 292.76 eV were attributed to sp3 C–C bonding, (C
C) and C–OH, respectively.46,47 The peaks @ 285.21, 288.45, and 292.76 eV were attributed to sp3 C–C bonding, (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)–O, and C–O/π–π * shake-up satellite.29,48,49 HRSf of Cd (Fig. 2a and b) show doublet in 411.54–411.81 and 404.65–404.98 eV regions which were attributed to Cd-3d3/2 and Cd-3d5/2 with separation around 6.8 for a single CdO-related phase.50 HRSf of O are shown in Fig. 1c and d. The peaks in 529.73–531.24 and 532.03–532.28 eV are attributed to MO (Pb–O–Si/PbO2/CdO) and chemisorbed oxygen/Si–O–Si in bridging oxygen (BO)/Pb–O–Pb.49,51–53 The peak at 533.14 eV was assigned to Si–OH, which further support the presence of Si1+ while that around 537 eV was due to Pb–Cd–O.53–57 HRSf of Ba (Fig. 2e and f) show doublet in 794.77–795.19 and 779.57–779.99 eV regions which were attributed to Ba-3d3/2 and Ba 3d5/2 peaks of BaO2, respectively.58 The absence of any peaks around 775 eV indicate the absence of BaO.58 HRSf of Ce are shown in Fig. 2g.
O)–O, and C–O/π–π * shake-up satellite.29,48,49 HRSf of Cd (Fig. 2a and b) show doublet in 411.54–411.81 and 404.65–404.98 eV regions which were attributed to Cd-3d3/2 and Cd-3d5/2 with separation around 6.8 for a single CdO-related phase.50 HRSf of O are shown in Fig. 1c and d. The peaks in 529.73–531.24 and 532.03–532.28 eV are attributed to MO (Pb–O–Si/PbO2/CdO) and chemisorbed oxygen/Si–O–Si in bridging oxygen (BO)/Pb–O–Pb.49,51–53 The peak at 533.14 eV was assigned to Si–OH, which further support the presence of Si1+ while that around 537 eV was due to Pb–Cd–O.53–57 HRSf of Ba (Fig. 2e and f) show doublet in 794.77–795.19 and 779.57–779.99 eV regions which were attributed to Ba-3d3/2 and Ba 3d5/2 peaks of BaO2, respectively.58 The absence of any peaks around 775 eV indicate the absence of BaO.58 HRSf of Ce are shown in Fig. 2g.
 |
| | Fig. 2 (a and b) HRSf Cd, (c and d) HRSf, (e and f) HRSf Ba of GCe0 and GCe4 and (g) HRSf Ce of GCe4. | |
The spectra are complicated because they contain contributions of many components. The interpretation of these spectra indicates the coexistence of both Ce3+ (v0 + v′ + u0 + u′) and Ce4+ (v + v′ + v′′′ + u + u′ + u′u′′′ + u′′′) oxidation states.48,59 The peak u′′′ is indicated as a fingerprint for +4 state. In these spectra, the u′′′ was found at 919.70 eV. For +3 state v0, v′, u0 and u′ were found in 880.65, 887.57, 899.55 and 905.17 eV region, respectively. The +4 state v, v′′, v′′′, u, u′′, u′u′′′ and u′′′ were found at 884.33, 891.10, 896.29, 901.93, 908.70, 914.24 and 919.70 eV region, respectively.40,48,59,60 The binding energies u/v, u0/v0 and u′/v′/u′′/v′′ are the result of O 2p and Ce 4f screening level hydration corresponds to Ce3d94f4O2p4, Ce3d94f2O2p4, Ce3d94f1O2p5 and while u′′′/v′′′ results from 3d94f0O2p6 final state.40,48,59,60 The presence of Ce3+ can be calculated using the eqn (15). The % Ce3+ is 42.00%.
| |
 | (15) |
3.2 XRD
To further confirm the glassy nature of the prepared samples, XRD were performed on three selected samples GCe0, GCe2, and GCe4 as shown in Fig. 3. The XRD analysis of the three specimens showed two distinct broad halos and no sharp diffraction peaks, confirming the amorphous nature of the prepared samples. The presence of two distinct broad halos, with maxima around 28° and 43°, indicates phase separation within the glass [1]. In synthesized glass, separation can be the result from two interpenetrating lead-silicate and borate networks.
 |
| | Fig. 3 XRD fitting of (a) GCe0, (b) GCe2 and (c) GCe4. | |
3.3 SEM-EDX
The SEM micrographs (Fig. 4a) reveal a clear morphological evolution with Ce addition. The undoped glass (Ce0), numerous bright inclusions, and surface defects indicate segregation of Pb-, Ba-, and Cd-rich domains within the matrix. These inclusions are often associated with polishing pits and microcracks, reflecting weak mechanical integrity. With progressive Ce addition (Ce0.5–Ce1), the microstructure becomes increasingly uniform; bright heavy-metal-rich features diminish in both number and size, and the background appears smoother and darker (i.e., pull-outs or pits are reduced), indicating reduced modifier segregation and improved glass homogeneity. At higher Ce levels (Ce2–Ce4), the surfaces display a nearly featureless, homogeneous matrix with only sparse, submicron inclusions, confirming the depletion of heavy-metal modifiers and enhanced polymerization of the boro-alumino-silicate network. The reduction of pull-outs and cracks further suggests improved stability and mechanical strength with Ce doping. Notably, the bright regions are likely artifacts caused by sample charging under the electron beam.61
 |
| | Fig. 4 SEM micrograph and corresponding EDX spectra for samples Ce0 to Ce4 (a–e). | |
The EDX data reveal a complex narrative far beyond a simple doping process for this Ce-doped heavy-metal oxide glass. The presence of B, Si, and Al confirms a boro-alumino-silicate network, providing chemical durability and thermal stability, while the heavy metals (Cd, Ba, Pb) act as modifiers that disrupt the network to impart high density and specific optical properties. The EDX spectra (Fig. 4) confirm the presence of all expected elements in the glass matrix. Cerium is identified by its characteristic Lα (4.840 keV), Lβ1 (5.262 keV), and Lγ (6.052 keV) peaks. Strong Kα signals corresponding to B (0.183 keV), O (0.523 keV), Al (1.487 keV), and Si (1.740 keV) further verify the glass-forming network.61 Pb is distinguished by its Lα2 (10.731 keV) and Lβ2 (12.980 keV) lines, while Cd exhibits Lα1 (3.133 keV), Lβ1 (3.317 keV), and Lβ2 (3.528 keV) peaks. Ba is detected via its Lα2 (4.634 keV), Lα1 (4.651 keV), Lβ1 (5.042 keV), and Lβ2 (5.384 keV) signals. EDX analysis was conducted on the undoped control sample (Ce0) and the Ce-doped samples (Ce0.5 to Ce4) to assess material purity and trace changes in elemental composition.62 This technique confirms the material purity and provides semi-quantitative information on the elemental composition of the samples. EDX analysis was employed to compare and monitor changes in the elemental composition of the glass samples before modification (GC0; as a control) and after modification for samples GC0.5–GC4. The semi-quantitative results are provided in Table 2. Although the analysis successfully confirms the increasing incorporation of cerium from 0 to 4.9% weight percent (wt%), the most striking observation is the profound compositional shift within the glass matrix itself. The introduction of CeO2 appears to trigger a substantial restructuring, evidenced by a dramatic decrease in the concentration of the heavy metal constituents and a corresponding increase in the glass formers (B, O, Si, Al). The key trends and observations are as follows:
Table 2 EDX analysis for the Ce0 to Ce4 glass samples
| Sample element |
Ce0 |
Ce0.5 |
Ce1 |
Ce2 |
Ce4 |
| Atomic % |
Weight % |
Atomic % |
Weight % |
Atomic % |
Weight % |
Atomic % |
Weight % |
Atomic % |
Weight % |
| B |
4.4 ± 0.3 |
0.7 ± 0.0 |
8.9 ± 0.3 |
3.2 ± 0.1 |
6.8 ± 0.4 |
2.4 ± 0.1 |
8.3 ± 0.3 |
2.8 ± 0.1 |
9.3 ± 0.3 |
3.4 ± 0.1 |
| O |
51.2 ± 1.2 |
12.1 ± 0.3 |
71.1 ± 0.6 |
37.3 ± 0.3 |
68.0 ± 0.7 |
35.2 ± 0.3 |
69.8 ± 0.5 |
34.9 ± 0.3 |
72.2 ± 0.5 |
39.1 ± 0.3 |
| Al |
1.8 ± 0.2 |
0.7 ± 0.1 |
3.5 ± 0.1 |
3.1 ± 0.1 |
5.9 ± 0.1 |
5.1 ± 0.1 |
3.9 ± 0.1 |
3.3 ± 0.1 |
2.9 ± 0.2 |
2.6 ± 0.1 |
| Si |
5.1 ± 0.2 |
2.1 ± 0.1 |
5.6 ± 0.2 |
5.2 ± 0.1 |
9.2 ± 0.1 |
8.4 ± 0.1 |
6.8 ± 0.2 |
6.0 ± 0.2 |
5.3 ± 0.1 |
5.1 ± 0.1 |
| Cd |
12.4 ± 0.3 |
20.6 ± 0.5 |
4.2 ± 0.1 |
15.7 ± 0.3 |
3.4 ± 0.2 |
12.4 ± 0.5 |
3.3 ± 0.1 |
11.5 ± 0.4 |
3.4 ± 0.1 |
13.1 ± 0.4 |
| Ba |
12.7 ± 0.1 |
25.7 ± 0.3 |
4.2 ± 0.1 |
18.7 ± 0.4 |
3.2 ± 0.1 |
14.3 ± 0.5 |
3.8 ± 0.1 |
16.3 ± 0.4 |
3.8 ± 0.1 |
17.5 ± 0.2 |
| Ce |
0.0 ± 0.0 |
0.0 ± 0.0 |
0.2 ± 0.1 |
0.9 ± 0.3 |
0.2 ± 0.1 |
1.0 ± 0.2 |
0.6 ± 0.1 |
2.8 ± 0.2 |
1.0 ± 0.1 |
4.9 ± 0.1 |
| Pb |
12.5 ± 0.1 |
38.1 ± 0.4 |
2.4 ± 0.0 |
16.0 ± 0.2 |
3.2 ± 0.1 |
21.2 ± 0.3 |
3.5 ± 0.1 |
22.5 ± 0.3 |
2.0 ± 0.0 |
14.3 ± 0.2 |
| Total |
100 |
100 |
100 |
100 |
100 |
100 |
100 |
100 |
100 |
100 |
• Cerium content successfully increases from 0.0 to 1.0 in at% (∼0.0 to 4.9 in wt%), confirming effective doping. The progression is not perfectly linear (e.g., Ce0.5 and Ce1 are similar), which is common in synthesis due to factors like volatilization or solubility limits. The significant jump between Ce1 and Ce2/Ce4 is notable.
• The atomic concentrations of the heavy metal modifiers (Cd, Ba, and Pb), plummet from a combined ∼37 at% in the Ce0 blank to roughly ∼13% in atomic percent (at%) for all doped samples. Their concentrations plummet at Ce0 then remain low: Pb: (drops from 12.5 at% to 3.6 at%), Cd (drops from 12.4 at% to 5.0 at%), Ba (drops from 12.7 at% to ∼4.6 at%).
• Glass formers and intermediates (O, B, Si, Al): their concentrations in atomic percent (at%) increase significantly as follows: O increases from 51.2% to over 72.2%, B increases from 4.4% to 9.3%. While Si and Al display fluctuating, non-linear concentration trends across the series. Despite this variability, their atomic percentages are consistently higher in the Ce-doped samples relative to the blank (Ce0) glass.
• The significant increase in oxygen atomic percent (from ∼51% to ∼72%) cannot be explained by the cerium oxide (CeO2 or Ce2O3) addition alone. This serves as a crucial clue that the doped samples (Ce0.5–Ce4) were synthesized from a base composition that was fundamentally different from the Ce0 blank from the outset.
These observed restructuring suggests two interpretations: the doped series may have utilized an intentionally altered base composition to aid cerium incorporation, or, if a single batch was targeted, the addition of oxidizing CeO2 potentially initiated redox reactions that volatilized heavy metals or enhanced crucible corrosion. Consequently, the Ce4 glass constitutes a distinct material—enriched in oxides and depleted in heavy metals—rather than a simple cerium-admixed variant of Ce0. This is a pivotal finding; clarifying its origin by consulting batch sheets and employing XPS and XRD to determine cerium valence and crystallinity is essential before linking these structural changes to the material's functional properties.
3.4 Physical properties
Density is one of the most important physical properties of glass.41,49 Table 3 indicate an improvement in density with CeO2/B2O3 replacement by 2.11%, 3.68%, 6.47% and 8.55% for  =
=  (GC0.5),
(GC0.5),  (GC1 G),
(GC1 G),  (GC2) and
(GC2) and  (GC4), respectively. This results may be due to the density difference between CeO2 (7.22 g cm−3) and B2O3 (2.46 g cm−3).
(GC4), respectively. This results may be due to the density difference between CeO2 (7.22 g cm−3) and B2O3 (2.46 g cm−3).
Table 3 Physical properties of prepared glass depending on variation in CeO2 and B2O3 concentration
| |
GC0 |
GC0.5 |
GC1 |
GC2 |
GC4 |
| Density × 10−1 |
38.81 |
39.63 |
40.24 |
41.32 |
42.13 |
| Vm value |
26.49 |
26.07 |
25.80 |
25.37 |
25.37 |
| OPD |
87.22 |
88.43 |
89.15 |
90.25 |
89.47 |
| VO value |
11.47 |
11.31 |
11.22 |
11.08 |
11.18 |
| VmB |
33.11 |
32.18 |
31.46 |
30.21 |
28.83 |
| dB–B × 10−8 |
3.80 |
3.77 |
3.74 |
3.69 |
3.63 |
| nb × 1022 m−3 |
8.78 |
8.98 |
9.13 |
9.40 |
9.64 |
| N × 1020 CeO2 |
0 |
1.16 |
2.33 |
4.75 |
9.50 |
| Rp (angstrom) CeO2 |
— |
8.28 |
6.55 |
5.17 |
4.10 |
| Ri (angstrom) CeO2 |
— |
20.53 |
16.24 |
12.82 |
10.17 |
The samples molar volume (Vm) were calculated using eqn (16):
| |
 | (16) |
where,
xi = molar fraction and
Mwt−i = molar mass fraction.
Table 3 and Fig. 5 show a decrease in Vm value by 1.58%, 2.59%, 4.20% and 4.21% as the CeO2 content increase. These values indicate a compression in the glass which were attributed to the increase in cation coordination number (CeO2: 8, B2O3: 3) and the density difference between CeO2 (7.22 g cm−3) and B2O3 (2.46 g cm−3). The difference between GC2 and GC4 is very low which may be due the small difference in density and the with double the CeO2 content and also the number of oxygen atoms per molecule (CeO2 (2 atoms) and B2O3 (3 atoms)). Oxygen packing density (OPD) was calculated using eqn (17):
| |
 | (17) |
where,
c = the number of oxygen's in the glasses,
ρ = the density and
Mwt = sum of molar mass fraction.
 |
| | Fig. 5 Effect of CeO2 replacement of B2O3 on (a) density, and Vm, (b) OPD, and Vo, (c) VBm, and dB–B, (d) Rp CeO2 and Ri CeO2. | |
The OPD is the next most important physical property of glass after density. Table 3 and Fig. 5b show the OPD values, which showed a general increase from by 1.39%, 2.22%, 3.48% and 2.58% with increasing CeO2 content. The increase in OPD values is strongly related to the increase in density and also the higher Cno of CeO2 (8) compared to that of B2O3 (3). Other physical parameters were calculated using:
| |
 | (18) |
| |
 | (19) |
| |
 | (20) |
| |
 | (21) |
| |
 | (22) |
| |
 | (23) |
| |
 | (24) |
where
Vo: the oxygen molar volume,
VBm: boron atom molar volume,
dB–B: average boron–boron separation,
xB: molar fraction of B
2O
3, Na: Avogadro's number (6.0228 × 1023 g mol
−1), N: ionic concentration,
Rp: polaron radius,
Ri: inter-ionic distance, and
nb: bonds per unit volume.
Vo values (Table 3 and Fig. 5b) show the exact same behavior as Vm, decreasing from 11.47 (GC0) to 11.08 at GC2 then increases to 11.18 at GC2. These values indicate a compression in Vo which as stated before is due to the higher density and cation coordination number of CeO2. After CeO2 the most effective factor is the number of oxygen atoms in molecule. The decrease in both Vo and Vm indicate a compression in glass structural network which in turns explain the decrease in d<B–B> and VBm values (Table 3 and Fig. 5c). The value of N, Rp and Ri were calculated depending on the variation of CeO2 concentrations as can be seen in Table 3 and Fig. 5d. The values of N, Rp and Ri agree with the proposed composition, starting at 0.00 (and no value for Rp and Ri) with no CeO2 content then increases as the its molar ratio increases.
3.5 Elastic properties RD
The elastic modulus is a fundamental mechanical parameter that measures the stiffness of radiation shielding materials. It plays a crucial role in achieving long-term stability of mechanical strength under various and harsh environmental conditions. The high modulus of elasticity of various materials provides a great ability to resist stresses, enhancing their resistance to structural deformation during use. The calculated basic elastic moduli of the prepared glass materials and the effect of CeO2/B2O3 replacements on the change in these values are shown in Fig. 6 and Table 4. An established approach for figuring out the mechanical characteristics of glass systems is the Makishima and Mackenzie model.63,64 The elastic modules parameters for the synthesized non-crystalline materials were as shadows: Young's modulus (E) range from 74.527 GPa to 77.425 GPa; Bulk modulus (B) range from 51.994 GPa to 56.260 GPa; Shear modulus (S) range from 29.548 GPa to 30.467 GPa; and Longitudinal modulus (L) range from 74.155 GPa to 79.110 GPa. The mechanical characteristics indicate an increasing trend with CeO2 content up to 2 mol%, as illustrated in Fig. 6 and Table 4. The reason for the increase in mechanical resistance of the sample prepared by adding the rapid up to 2 mol percent can be attributed to the increase in the number of bridging oxygen compared to the non-bridging oxygen. There is a direct relationship between the amount of bound oxygen and its effect on increasing the strength of the internal structure of the glass and its reflection on the increase in the calculated mechanical resistance.65 It was found that adding CeO2 to alkali borosilicate glasses works primarily to strengthen the glass network and is reflected in increasing the mechanical resistance of the prepared glass.66 On the other hand, the slightly decrease in elastic modulus with increase in cerium dioxide up to 4 mol% can be attributed to role of cerium in increasing the BO3/BO4 ratio and decrease the strength of the glass network.66 It is clear from Table 4 that the relationship between atomic packing density (Vt) and Poisson ratio (σ) is a direct relationship, and the values of both are increased with the increase cerium oxide up to 2 mol%. Poisson's ratio (σ) is frequently used to explain the compactness of glass and the effect of changes in composition on modifications in the cross link density of glass network.67 Increasing Poisson's ratio values indicate that the internal structure of the glass has become stronger and more toughness.67 However, the weakened glass network is responsible for the drop in the (Vt) and σ values at 4 mol% (Fig. 7).
 |
| | Fig. 6 The elastic modulus of the synthesized glasses Young's modulus, E; bulk modulus, B; shear modulus, S; and longitudinal modulus L. | |
Table 4 The elemental fractional abundance and density of the prepared samplesa
| Glass ID |
GC0 |
GC0.5 |
GC1 |
GC2 |
GC4 |
| All values × 10−1. |
| Young's modulus EM-M (GPa) |
745.27 |
756.13 |
762.85 |
774.25 |
769.02 |
| Shear modulus SM-M (GPa) |
295.48 |
298.94 |
301.07 |
304.67 |
302.84 |
| Longitudinal modulus LM-M (GPa) |
741.55 |
759.74 |
771.26 |
791.10 |
783.59 |
| Bulk modulus BM-M (GPa) |
519.94 |
535.54 |
545.46 |
562.60 |
556.45 |
| Vt |
5.81 |
5.90 |
0.5.96 |
6.06 |
0.6.03 |
| Poisson ratio (σ) |
2.61 |
2.65 |
2.67 |
2.71 |
2.70 |
 |
| | Fig. 7 Atomic packing density (Vt) and Poisson ratio (σ) of the investigated YSBP. | |
3.6 Shielding properties
Single layer shielding properties of current glass samples were investigated using Py-MLBUF by calculating LAC, MAC, HVL, TVL and Zeff in the energy ranges from 0.015 to 15 MeV (Table 5 and Fig. 8). In all samples, LAC values show maximum value at 0.05 eV followed by a sharp decrease then gradual decrease with increasing γE. This general behavior for most materials.29,46 These values depend mainly on the dominant interaction mechanism. At low γE (0.015 eV) the photoelectric absorption is the dominant but with increasing energy the Compton scattering became more dominant. LAC is a very important factor in considering the efficiency of current glass shielding. The maximum difference between glass samples were noted at lowest γE. LAC of basic glass was improved by 3.71%, 6.93%, 13.06% and 21.75% with  =
=  (GC0.5),
(GC0.5),  (GC1),
(GC1),  (GC2) and
(GC2) and  (GC4), respectively. This order agrees with the increase in densities (Table 3). MAC show similar behavior to LAC (Table 5 and Fig. 8b), showing maximum value at 0.05 eV then the values decrease sharply with increasing γE followed by a gradual decrease. The decrease of MAC values as the γE increases may be due to the decrease in the effectiveness of Compton scattering and photoelectric absorption. MAC of basic glass was improved by 1.57%, 3.13%, 6.20% and 5.57% with
(GC4), respectively. This order agrees with the increase in densities (Table 3). MAC show similar behavior to LAC (Table 5 and Fig. 8b), showing maximum value at 0.05 eV then the values decrease sharply with increasing γE followed by a gradual decrease. The decrease of MAC values as the γE increases may be due to the decrease in the effectiveness of Compton scattering and photoelectric absorption. MAC of basic glass was improved by 1.57%, 3.13%, 6.20% and 5.57% with  =
=  (GC0.5),
(GC0.5),  (GC1 G),
(GC1 G),  (GC2) and
(GC2) and  (GC4), respectively, indicating the superior effect of higher density and molecular weight (CeO2: 7.22 g cm−3, 174 g mol−1, and B2O3: 2.46 g cm−3, 69.62 g mol−1).37,41,68 The lower Zeff values indicate a higher shielding power at lower γE. The increase of Zeff values (at γE = 1.00 eV: 0.83% (GC0.5), 1.66% (GC1), and 3.32% (GC2) and 6.65% (GC4)) with
(GC4), respectively, indicating the superior effect of higher density and molecular weight (CeO2: 7.22 g cm−3, 174 g mol−1, and B2O3: 2.46 g cm−3, 69.62 g mol−1).37,41,68 The lower Zeff values indicate a higher shielding power at lower γE. The increase of Zeff values (at γE = 1.00 eV: 0.83% (GC0.5), 1.66% (GC1), and 3.32% (GC2) and 6.65% (GC4)) with  replacement is due to adding Ce (58) with higher atomic number than B (5).48,69 σ-atomic, and σ-electron (Table 5 and Fig. 8e, f) show the same behavior, their values decrease as the γE increases, since the probability of the photoelectric effect decreases as photon energy increases.48,70 HVL and TVL show similar behavior (Table 5 and Fig. 8c, d) showing minimum values at 0.015 eV and increase as the γE increases which indicate the need to increase the thickness with increasing photon energy.47,71,72 At γE = 0.0150 eV:
replacement is due to adding Ce (58) with higher atomic number than B (5).48,69 σ-atomic, and σ-electron (Table 5 and Fig. 8e, f) show the same behavior, their values decrease as the γE increases, since the probability of the photoelectric effect decreases as photon energy increases.48,70 HVL and TVL show similar behavior (Table 5 and Fig. 8c, d) showing minimum values at 0.015 eV and increase as the γE increases which indicate the need to increase the thickness with increasing photon energy.47,71,72 At γE = 0.0150 eV:
Table 5 MAC, LAC and Zeff for prepared glass calculated using Py-MLBUF
| Energy MeV |
Total MAC (cm2 g−1) |
LAC (cm−1) |
Zeff |
| GC0 |
GC0.5 |
GC1 |
GC2 |
GC4 |
GC0 |
GC0.5 |
GC1 |
GC2 |
GC4 |
GC0 |
GC0.5 |
GC1 |
GC2 |
GC4 |
| 0.0150 |
41.03 |
41.308 |
41.585 |
42.128 |
43.185 |
159.24 |
163.7 |
167.34 |
174.07 |
181.94 |
59.4448 |
59.4482 |
59.4522 |
59.4587 |
59.4715 |
| 0.0200 |
27.381 |
27.469 |
27.556 |
27.727 |
28.059 |
106.27 |
108.86 |
110.88 |
114.57 |
118.21 |
64.6337 |
64.593 |
64.5536 |
64.4758 |
64.3283 |
| 0.0263 |
13.379 |
13.419 |
13.459 |
13.538 |
13.69 |
51.925 |
53.181 |
54.16 |
55.937 |
57.675 |
63.6235 |
63.5993 |
63.5759 |
63.5296 |
63.4415 |
| 0.0300 |
12.974 |
12.985 |
12.996 |
13.017 |
13.059 |
50.352 |
51.459 |
52.295 |
53.787 |
55.016 |
57.9692 |
57.9878 |
58.0062 |
58.0418 |
58.1113 |
| 0.0400 |
8.8346 |
8.8256 |
8.817 |
8.7995 |
8.7659 |
34.287 |
34.976 |
35.48 |
36.36 |
36.931 |
55.5623 |
55.5907 |
55.6196 |
55.6756 |
55.7864 |
| 0.0500 |
4.9664 |
5.0444 |
5.1219 |
5.2742 |
5.5701 |
19.275 |
19.991 |
20.611 |
21.793 |
23.467 |
53.0818 |
53.201 |
53.317 |
53.5352 |
53.9302 |
| 0.0595 |
3.1645 |
3.2138 |
3.2628 |
3.359 |
3.546 |
12.282 |
12.736 |
13.129 |
13.879 |
14.939 |
50.2044 |
50.3775 |
50.546 |
50.8648 |
51.4456 |
| 0.0600 |
3.1023 |
3.1506 |
3.1986 |
3.2929 |
3.4762 |
12.04 |
12.486 |
12.871 |
13.606 |
14.645 |
50.0786 |
50.254 |
50.4246 |
50.7476 |
51.3361 |
| 0.0800 |
1.5006 |
1.5227 |
1.5447 |
1.588 |
1.672 |
5.8238 |
6.0346 |
6.216 |
6.5615 |
7.044 |
43.3936 |
43.6594 |
43.9192 |
44.417 |
45.34 |
| 0.1000 |
1.8807 |
1.8876 |
1.8946 |
1.9082 |
1.9346 |
7.2989 |
7.4807 |
7.6238 |
7.8846 |
8.1507 |
52.3212 |
52.4228 |
52.5234 |
52.72 |
53.0985 |
| 0.1500 |
0.72192 |
0.72399 |
0.72605 |
0.7301 |
0.73796 |
2.8018 |
2.8692 |
2.9216 |
3.0168 |
3.109 |
38.7078 |
38.8818 |
39.055 |
39.3972 |
40.0684 |
| 0.2000 |
0.39131 |
0.39217 |
0.39302 |
0.39471 |
0.39798 |
1.5187 |
1.5542 |
1.5815 |
1.6309 |
1.6767 |
29.4158 |
29.5912 |
29.7662 |
30.1148 |
30.807 |
| 0.3000 |
0.19401 |
0.19424 |
0.19448 |
0.19493 |
0.19582 |
0.75296 |
0.76979 |
0.78258 |
0.80547 |
0.82499 |
20.3608 |
20.5024 |
20.6444 |
20.9286 |
21.5001 |
| 0.4000 |
0.13376 |
0.13384 |
0.13392 |
0.13407 |
0.13437 |
0.51911 |
0.53039 |
0.53888 |
0.55398 |
0.56612 |
16.7619 |
16.8811 |
17.0008 |
17.2411 |
17.7263 |
| 0.5000 |
0.10648 |
0.10651 |
0.10653 |
0.10658 |
0.10668 |
0.41326 |
0.42209 |
0.42869 |
0.4404 |
0.44944 |
15.0602 |
15.1672 |
15.2748 |
15.491 |
15.9284 |
| 0.6000 |
0.091007 |
0.091008 |
0.091011 |
0.091014 |
0.09102 |
0.3532 |
0.36067 |
0.36623 |
0.37607 |
0.38347 |
14.1402 |
14.2403 |
14.3408 |
14.543 |
14.9527 |
| 0.6620 |
0.084257 |
0.084251 |
0.084245 |
0.084233 |
0.08421 |
0.327 |
0.33388 |
0.339 |
0.34805 |
0.35478 |
13.7592 |
13.8564 |
13.954 |
14.1504 |
14.5485 |
| 0.8000 |
0.073551 |
0.073534 |
0.073519 |
0.073487 |
0.073425 |
0.28545 |
0.29142 |
0.29584 |
0.30365 |
0.30934 |
13.2174 |
13.3102 |
13.4036 |
13.5914 |
13.972 |
| 1.0000 |
0.063485 |
0.063463 |
0.063442 |
0.063399 |
0.063317 |
0.24639 |
0.2515 |
0.25529 |
0.26196 |
0.26675 |
12.7789 |
12.8682 |
12.958 |
13.1386 |
13.5054 |
| 1.1730 |
0.057473 |
0.05745 |
0.057428 |
0.057383 |
0.057297 |
0.22305 |
0.22767 |
0.23109 |
0.23711 |
0.24139 |
12.575 |
12.6627 |
12.7509 |
12.9284 |
13.2886 |
| 1.3330 |
0.053352 |
0.05333 |
0.053309 |
0.053266 |
0.053184 |
0.20706 |
0.21135 |
0.21451 |
0.2201 |
0.22407 |
12.4747 |
12.5618 |
12.6494 |
12.8256 |
13.1834 |
| 1.5000 |
0.050063 |
0.050043 |
0.050024 |
0.049985 |
0.049909 |
0.1943 |
0.19832 |
0.2013 |
0.20654 |
0.21027 |
12.45 |
12.537 |
12.6245 |
12.8006 |
13.1581 |
| 2.0000 |
0.043632 |
0.043621 |
0.043611 |
0.043589 |
0.043549 |
0.16934 |
0.17287 |
0.17549 |
0.18011 |
0.18347 |
12.5922 |
12.6814 |
12.771 |
12.9515 |
13.3177 |
| 2.5060 |
0.039788 |
0.039787 |
0.039787 |
0.039787 |
0.039786 |
0.15442 |
0.15768 |
0.1601 |
0.1644 |
0.16762 |
12.8974 |
12.9906 |
13.0843 |
13.2726 |
13.6548 |
| 3.0000 |
0.037356 |
0.037365 |
0.037375 |
0.037393 |
0.03743 |
0.14498 |
0.14808 |
0.1504 |
0.15451 |
0.15769 |
13.2394 |
13.337 |
13.435 |
13.6321 |
14.0316 |
| 4.0000 |
0.034462 |
0.034489 |
0.034516 |
0.034569 |
0.034671 |
0.13375 |
0.13668 |
0.13889 |
0.14284 |
0.14607 |
14.012 |
14.119 |
14.2264 |
14.4423 |
14.8791 |
| 5.0000 |
0.032989 |
0.03303 |
0.033072 |
0.033153 |
0.033311 |
0.12803 |
0.1309 |
0.13308 |
0.13699 |
0.14034 |
14.7974 |
14.9135 |
15.0302 |
15.2642 |
15.7372 |
| 6.0000 |
0.032231 |
0.032285 |
0.032339 |
0.032444 |
0.032648 |
0.12509 |
0.12795 |
0.13013 |
0.13406 |
0.13755 |
15.5558 |
15.6803 |
15.8054 |
16.056 |
16.5617 |
| 8.0000 |
0.031807 |
0.031881 |
0.031955 |
0.0321 |
0.032382 |
0.12344 |
0.12634 |
0.12859 |
0.13264 |
0.13642 |
16.9514 |
17.0904 |
17.2298 |
17.509 |
18.0704 |
| 10.0000 |
0.032068 |
0.032158 |
0.032248 |
0.032425 |
0.032769 |
0.12445 |
0.12744 |
0.12977 |
0.13398 |
0.13806 |
18.1704 |
18.3208 |
18.4716 |
18.7732 |
19.3783 |
| 15.0000 |
0.033636 |
0.033757 |
0.033878 |
0.034115 |
0.034577 |
0.13054 |
0.13378 |
0.13633 |
0.14096 |
0.14567 |
20.5458 |
20.7158 |
20.8858 |
21.2251 |
21.9026 |
 |
| | Fig. 8 (a) LAC, (b) total MAC, (c) HVL, (d) TVL, (e) σ-atomic, (f) σ-electron, and (g) Zef versus log10 of photon energy of single layer samples. | |
• HVL: the decrease in valued with increasing  content (−4.55% (GC0.5), −6.82% (GC1), −9.09% (GC2), −13.64% (GC4)) indicate a higher shielding power (less thickness of the glass).
content (−4.55% (GC0.5), −6.82% (GC1), −9.09% (GC2), −13.64% (GC4)) indicate a higher shielding power (less thickness of the glass).
• TVL: Shaw the same trends (−2.76% (GC0.5), −4.83% (GC1), −8.97% (GC2), −12.41% (GC4))
EABF and EBF (single layer) were calculated at 1 mfp as shown in Fig. 9. These are vital in the correction of the attenuation calculation by taking in account the secondary gamma ray emission. EBFmax (2.84–3.47) and EABFmax (1.63–1.66) are shown in all glass samples in 0.04–0.06 eV region, which result from multiple scattering caused by Compton interaction. To investigate the use of double layer, DLEBF and DLEABF for AB (GC0 as 1st layer and other samples as 2nd layer), and BA (GC0 as 2nd layer and other samples as 1st layer) were calculated at X1 = 1 mfp and X2 = 1 mfp penetration depths (Fig. 9). DLEBFmax (0.05–0.06) order of current samples was: GC2–GC0 (6.792) > GC0–GC4 (6.465) > GC1–GC0 (4.514) > GC0–GC0.5 (4.505) > GC4–GC0 (4.317) > GC0–GC2 (4.054) > GC0–GC1 (4.017) while that of DLEABFmax (0.6–1.0) was GC0–GC4 (3.362) > GC0–GC2 (3.359) > GC0–GC1 (3.357) > GC4–GC0 (3.335) > GC2–GC0 (3.307) > GC0.5–GC0 (3.227) > GC1–GC0 = GC0–GC0.5 (3.166) (Table 6).
 |
| | Fig. 9 (a) EBF, (b) EABF and (c & d) DEBF, (e & f) DEABF versus log10 of photon energy of single layer samples (selected penetration depth: X1 = 1 mfp) and of double layers (penetration depth: X1 = X2 = 1 mfp). | |
Table 6 HVL, and TVL for prepared glass calculated using Py-MLBUF
| Energy MeV |
HVL (cm) |
TVL (cm) |
| GC0 |
GC0.5 |
GC1 |
GC2 |
GC4 |
GC0 |
GC0.5 |
GC1 |
GC2 |
GC4 |
| 0.0150 |
0.0044 |
0.0042 |
0.0041 |
0.004 |
0.0038 |
0.0145 |
0.0141 |
0.0138 |
0.0132 |
0.0127 |
| 0.0200 |
0.0065 |
0.0064 |
0.0063 |
0.0061 |
0.0059 |
0.0217 |
0.0212 |
0.0208 |
0.0201 |
0.0195 |
| 0.0263 |
0.0133 |
0.013 |
0.0128 |
0.0124 |
0.012 |
0.0443 |
0.0433 |
0.0425 |
0.0412 |
0.0399 |
| 0.0300 |
0.0138 |
0.0135 |
0.0133 |
0.0129 |
0.0126 |
0.0457 |
0.0447 |
0.044 |
0.0428 |
0.0419 |
| 0.0400 |
0.0202 |
0.0198 |
0.0195 |
0.0191 |
0.0188 |
0.0672 |
0.0658 |
0.0649 |
0.0633 |
0.0623 |
| 0.0500 |
0.036 |
0.0347 |
0.0336 |
0.0318 |
0.0295 |
0.1195 |
0.1152 |
0.1117 |
0.1057 |
0.0981 |
| 0.0595 |
0.0564 |
0.0544 |
0.0528 |
0.0499 |
0.0464 |
0.1875 |
0.1808 |
0.1754 |
0.1659 |
0.1541 |
| 0.0600 |
0.0576 |
0.0555 |
0.0539 |
0.0509 |
0.0473 |
0.1912 |
0.1844 |
0.1789 |
0.1692 |
0.1572 |
| 0.0800 |
0.119 |
0.1149 |
0.1115 |
0.1056 |
0.0984 |
0.3954 |
0.3816 |
0.3704 |
0.3509 |
0.3269 |
| 0.1000 |
0.095 |
0.0927 |
0.0909 |
0.0879 |
0.085 |
0.3155 |
0.3078 |
0.302 |
0.292 |
0.2825 |
| 0.1500 |
0.2474 |
0.2416 |
0.2372 |
0.2298 |
0.2229 |
0.8218 |
0.8025 |
0.7881 |
0.7633 |
0.7406 |
| 0.2000 |
0.4564 |
0.446 |
0.4383 |
0.425 |
0.4134 |
1.5162 |
1.4816 |
1.4559 |
1.4118 |
1.3733 |
| 0.3000 |
0.9206 |
0.9004 |
0.8857 |
0.8606 |
0.8402 |
3.058 |
2.9912 |
2.9423 |
2.8587 |
2.791 |
| 0.4000 |
1.3353 |
1.3069 |
1.2863 |
1.2512 |
1.2244 |
4.4356 |
4.3413 |
4.2729 |
4.1564 |
4.0673 |
| 0.5000 |
1.6773 |
1.6422 |
1.6169 |
1.5739 |
1.5422 |
5.5718 |
5.4552 |
5.3712 |
5.2284 |
5.1232 |
| 0.6000 |
1.9625 |
1.9219 |
1.8927 |
1.8431 |
1.8076 |
6.5192 |
6.3843 |
6.2873 |
6.1228 |
6.0046 |
| 0.6620 |
2.1197 |
2.076 |
2.0447 |
1.9915 |
1.9537 |
7.0415 |
6.8963 |
6.7922 |
6.6157 |
6.4902 |
| 0.8000 |
2.4283 |
2.3786 |
2.343 |
2.2827 |
2.2407 |
8.0665 |
7.9014 |
7.7832 |
7.5831 |
7.4435 |
| 1.0000 |
2.8133 |
2.756 |
2.7151 |
2.646 |
2.5985 |
9.3455 |
9.1553 |
9.0195 |
8.7897 |
8.6319 |
| 1.1730 |
3.1075 |
3.0445 |
2.9995 |
2.9234 |
2.8715 |
10.323 |
10.1135 |
9.964 |
9.7112 |
9.5388 |
| 1.3330 |
3.3476 |
3.2797 |
3.2312 |
3.1493 |
3.0935 |
11.1204 |
10.8949 |
10.7339 |
10.4617 |
10.2764 |
| 1.5000 |
3.5675 |
3.4951 |
3.4434 |
3.356 |
3.2965 |
11.8509 |
11.6104 |
11.4388 |
11.1486 |
10.9507 |
| 2.0000 |
4.0933 |
4.0097 |
3.9498 |
3.8485 |
3.778 |
13.5978 |
13.3198 |
13.121 |
12.7843 |
12.5502 |
| 2.5060 |
4.4888 |
4.396 |
4.3293 |
4.2163 |
4.1352 |
14.9116 |
14.6033 |
14.3818 |
14.0061 |
13.737 |
| 3.0000 |
4.7811 |
4.681 |
4.6088 |
4.4861 |
4.3956 |
15.8823 |
15.5499 |
15.3101 |
14.9026 |
14.6018 |
| 4.0000 |
5.1825 |
5.0713 |
4.9905 |
4.8527 |
4.7454 |
17.2157 |
16.8465 |
16.5781 |
16.1203 |
15.7637 |
| 5.0000 |
5.414 |
5.2953 |
5.2085 |
5.06 |
4.9391 |
17.9848 |
17.5907 |
17.3022 |
16.8088 |
16.4075 |
| 6.0000 |
5.5412 |
5.4175 |
5.3265 |
5.1705 |
5.0393 |
18.4075 |
17.9966 |
17.6943 |
17.1761 |
16.7403 |
| 8.0000 |
5.6151 |
5.4862 |
5.3905 |
5.2259 |
5.0808 |
18.653 |
18.2246 |
17.9068 |
17.3601 |
16.8782 |
| 10.0000 |
5.5695 |
5.4389 |
5.3415 |
5.1735 |
5.0207 |
18.5014 |
18.0676 |
17.7439 |
17.1859 |
16.6786 |
| 15.0000 |
5.3098 |
5.1812 |
5.0845 |
4.9172 |
4.7583 |
17.6387 |
17.2116 |
16.8902 |
16.3345 |
15.8068 |
4 Conclusion
The present study demonstrates that CeO2-modified heavy-metal borosilicate glasses exhibit enhanced structural and functional properties, making them promising γ-ray shielding materials. XRD confirmed the fully amorphous nature of all compositions, indicating stable glass networks. SEM-EDS analysis revealed homogeneous microstructures with effective cerium incorporation up to 4.9 wt%. XPS results confirmed mixed Ce3+ (42%) and Ce40 (58%) states, contributing to improved network cross-linking and structural stability. The addition of CeO2 significantly influenced the physical and mechanical properties, with glass density increasing by 8.55% from 3.881 g cm−3 (GC0) to 4.213 g cm−3 (GC4), accompanied by a 4.21% reduction in molar volume, reflecting structural compaction. Mechanical modeling revealed enhanced elastic performance, with Young's modulus, bulk modulus, and shear modulus reaching maximum values of 77.43 GPa, 56.26 GPa, and 30.47 GPa, respectively, at approximately 2 mol% CeO2. γ-ray shielding performance improved markedly with CeO2 incorporation, as evidenced by a 21.75% increase in the LAC and a 6.20% rise in the MAC, alongside reductions of 13.64% and 12.41% in the HVL and TVL, respectively, confirming improved photon attenuation efficiency. The Zeff increased by 6.65%, and the maximum double-layer exposure buildup factor (DLEBFmax = 6.79 at 0.05 MeV) confirmed efficient suppression of secondary radiation. Thus, moderate CeO2 doping (2 mol%) provides an optimal balance between structural integrity, mechanical strength, and γ-ray attenuation efficiency, positioning these glasses as promising, eco-friendly alternatives to conventional lead-based shielding materials.
Ethical approval
This article doesn't contain any studies involving animals performed by any authors. Also, this article does not contain any studies involving human participants performed by any of the authors.
Consent for publication
All authors have read and agreed to the published version of the manuscript.
Consent to participate
All authors agree to participate in the published version of the manuscript.
Author contributions
H. A. Abo-Mosallam & Mortaga M. Abou-Krisha: synthesis of glass & mechanical study; Ahmed A. Galhoum: SEM-EDX; A. M. A. El-Seidy: shielding/build-up shielding-factors & physical properties; H. A. Abo-Mosallam, Mortaga M. Abou-Krisha, Ahmed A. Galhoum & A. M. A. El-Seidy: writing – review & editing, writing – original draft, validation, methodology, investigation, formal analysis, data curation, conceptualization.
Conflicts of interest
The authors declare that there is no conflict of interest.
Data availability
All data generated or analyzed during this study is included in this published article.
Acknowledgements
This work was supported and funded by the Deanship of Scientific Research at Imam Mohammad Ibn Saud Islamic University (IMSIU) (grant number IMSIU-DDRSP2602).
Notes and references
- C. G. Hernández-Murillo, L. A. Escalera-Velasco, J. R. M. Contreras, H. R. Vega-Carrillo and H. A. de Leon-Martíne, in Advanced Radiation Shielding Materials, Elsevier, 2024, DOI:10.1016/C2021-0-02858-3.
- A. Poddar, M. Roy, A. Chamuah and S. Bhattacharya, Phys. B, 2026, 723, 418143 CrossRef CAS.
- A. Chamuah, K. Bhattacharya, S. Ojha, C. K. Ghosh, J. Ghosh, T. Chakraborty, P. Ranjan and S. Bhattacharya, Adv. Theory Simul., 2025, e01031 Search PubMed.
- M. R. Ahmed, N. A. Alsaif, N. Siddiqui, A. S. Prasad, M. Srinivas, S. K. Ahmmad, K. M. Kaky, A. J. Kadhim and Y. Rammah, Opt. Mater., 2025, 158, 116461 CrossRef CAS.
- N. A. Alsaif, N. Alfryyan, H. Al-Ghamdi, Y. Rammah, E. A. Mahdy, H. Abo-Mosallam and F. El-Agawany, Opt. Mater., 2024, 157, 116081 CrossRef CAS.
- S. Barbhuiya, B. B. Das, P. Norman and T. Qureshi, Struct. Concr., 2024, 26, 1809–1855 CrossRef.
- J. Ghosh, M. S. Ali, R. Sarkar, R. K. Shukla, J. Chowdhury, P. Bhowmick and S. Bhattacharya, J. Electron. Mater., 2025, 54, 10710–10732 CrossRef CAS.
- P. Halder, N. D. Sherpa, B. Pahari, N. Roy and S. Bhattacharya, J. Alloys Compd., 2025, 1019, 179197 CrossRef CAS.
- G. Annadurai, W. Chaiphaksa, C. Mutuwong, S. Yonphan, Y. Ruangtaweep, W. Cheewasukhanont, N. Intachai, M. Tungjai, S. Kothan, H. Kim and J. Kaewkhao, Radiat. Phys. Chem., 2026, 238, 113137 CrossRef CAS.
- D. Keerthana, K. Naseer, S. Arul Raja Sekaran, M. Sayyed, M. S. Alqahtani and K. Marimuthu, Radiat. Phys. Chem., 2026, 238, 113153 CrossRef CAS.
- P. Halder, M. S. Ali and S. Bhattacharya, J. Non-Cryst. Solids, 2023, 616, 122459 CrossRef CAS.
- O. Sallam, M. S. Mansy, Y. Mohamed and N. Elalaily, Radiat. Phys. Chem., 2025, 237, 113056 CrossRef CAS.
- E. Ilik, S. H. Guler, O. Guler, H. Durmus, E. Kavaz, S. Birdogan, G. ALMisned, G. Kilic and H. Tekin, J. Alloys Compd., 2025, 1036, 181904 CrossRef CAS.
- S. Ojha, A. Chamuah, M. Roy and S. Bhattacharya, Front. Phys., 2023, 11, year Search PubMed.
- J. Alyami, Y. Al-Hadeethi, O. A. Fallatah, S. Biradar, M. Sayyed and F. Almutairi, Ann. Nucl. Energy, 2025, 212, 111069 CrossRef CAS.
- Y. Maghrbi, H. Heryanto, S. Biradar, M. Y. Hanfi and M. Sayyed, J. Sci.:Adv. Mater. Devices, 2025, 10, 100949 CAS.
- K. Bhattacharya and D. Das, Bull. Mater. Sci., 2008, 31, 467–471 CrossRef CAS.
- H. H. Negm, A. A. Sdeek and A. A. Ebrahim, J. Electron. Mater., 2024, 53, 3965–3979 CrossRef CAS.
- E. Abou Hussein, S. Shaban and S. A. Makhlouf, Radiat. Phys. Chem., 2025, 235, 112762 CrossRef CAS.
- D. Das and K. Bhattacharya, Jpn. J. Appl. Phys., 2007, 46, L1006 CrossRef CAS.
- A. Acikgoz, G. Demircan, D. Yılmaz, B. Aktas, S. Yalcin and N. Yorulmaz, Mater. Sci. Eng. B, 2022, 276, 115519 CrossRef CAS.
- K. S. Shaaban, D. A. Aloraini, K. Alsafi, H. M. Almutairi, W. M. Al-Saleh and A. S. Alzahrani, Mater. Today Commun., 2024, 38, 108309 CrossRef CAS.
- E. A. Hussein, A. Madbouly, F. E. Eldin and N. ElAlaily, Mater. Chem. Phys., 2021, 261, 124212 CrossRef CAS.
- A. Makishima and J. Mackenzie, J. Non-Cryst. Solids, 1973, 12, 35–45 CrossRef CAS.
- A. Abd El-Moneim, M. A. Azooz, H. A. Hashem, A. M. Fayad and R. L. Elwan, Sci. Rep., 2023, 13, 1–15 Search PubMed.
- A. Amat, H. M. Kamari, I. Mansor, N. Osman, N. N. S. Nidzam and N. I. M. Kamal, Appl. Phys. A, 2021, 127, year CrossRef.
- K. S. Mann and S. S. Mann, Ann. Nucl. Energy, 2021, 150, 107845 CrossRef CAS.
- O. F. Ozpolat, B. Alım, E. Sakar, M. Büyükyıldız and M. Kurudirek, Radiat. Environ. Biophys., 2020, 59, 321–329 CrossRef CAS PubMed.
- A. A. Al-Ghamdi, E. E. Bayoumi, S. Al-Farraj, M. Sillanpää and A. M. El-Seidy, Surf. Interfaces, 2025, 67, 106567 CrossRef CAS.
- S. Seltzer, XCOM-Photon Cross Sections Database, NIST Standard Reference Database 8, 1987, DOI:10.18434/T48G6X.
- S. Seltzer, Tables of X-Ray Mass Attenuation Coefficients and Mass Energy-Absorption Coefficients, NIST Standard Reference Database, 1995, p. 126, DOI:10.18434/T4D01F.
- P. Basu, R. Sarangapani and B. Venkatraman, Appl. Radiat. Isot., 2019, 154, 108864 CrossRef CAS PubMed.
- K. S. Mann, Nucl. Eng. Technol., 2017, 49, 792–800 CrossRef CAS.
- K. S. Mann, M. Kurudirek and G. Sidhu, Appl. Radiat. Isot., 2012, 70, 681–691 CrossRef CAS PubMed.
- A. A. Sokolov, E. O. Filatova, V. V. Afanasev, E. Y. Taracheva, M. M. Brzhezinskaya and A. A. Ovchinnikov, J. Phys. D: Appl. Phys., 2008, 42, 035308 CrossRef.
- Defects in SiO2 and Related Dielectrics: Science and Technology, ed. G. Pacchioni, L. Skuja and D. L. Griscom, Springer Netherlands, Dordrecht, 2000, DOI:10.1007/978-94-010-0944-7.
- M. S. El-Okaily, A. M. A. El-Seidy, E. H. Ismail, R. M. Allam, A. A. Saeed, A. Bhaumik and A. A. Mostafa, J. Mater. Res., 2024, 39, 1741–1757 CrossRef CAS.
- A. M. A. El-Seidy, M. S. El-Okaily, I. M. Nabil and A. A. Mostafa, Sci. Rep., 2025, 15, year Search PubMed.
- H.-H. T. Nguyen, Y.-H. Choi, E.-B. Kim, Y.-H. Jeong, J.-W. Lee, K.-H. Park, Y.-J. Woo, S. Ameen and D.-H. Kwak, Carbon Trends, 2025, 19, 100465 CrossRef CAS.
- O. I. Sallam, Y. S. Rammah, I. M. Nabil and A. M. A. El-Seidy, Sci. Rep., 2024, 14, 24478 CrossRef CAS PubMed.
- N. A. M. Alsaif, N. Alfryyan, H. Al-Ghamdi, A. M. A. El-Seidy, A. M. Abdelghany, Y. S. Rammah and A. S. Abouhaswa, J. Inorg. Organomet. Polym. Mater., 2024, 34, 3623–3631 CrossRef CAS.
- B. Joanne James, R. Cameron and C. Baskcomb, Res. Lett. Mater. Sci., 2008, 2008, 1–4 CrossRef.
- T. Susi, T. Pichler and P. Ayala, Beilstein J. Nanotechnol., 2015, 6, 177–192 CrossRef PubMed.
- S. Zhao, Y. Wu, B. Zhou and X. Liu, Sci. Rep., 2019, 9, 17337 CrossRef PubMed.
- J. Qu, Q. Li, C. Luo, J. Cheng and X. Hou, Coatings, 2018, 8, 214 CrossRef.
- A. M. A. El-Seidy, I. E. El-Sayed, M. Linnolahti, E. E. Bayoumi, H. I. Mira and A. A. Galhoum, RSC Adv., 2025, 15, 28269–28279 RSC.
- A. Elseidy, S. Bashandy, F. Ibrahim, S. Abd El-Rahman, O. Farid, S. Moussa and M. El-Baset, Egypt. J. Chem., 2022, 497 Search PubMed.
- A. M. El-Seidy, M. A. Elbaset, F. A. Ibrahim, S. A. Abdelmottaleb Moussa and S. A. Bashandy, J. Trace Elem. Med. Biol., 2023, 80, 127312 CrossRef CAS PubMed.
- A. M. A. El-Seidy, O. I. Sallam, I. M. Nabil, Y. S. Rammah, M. S. El-Okaily and H. Alshater, Sci. Rep., 2024, 14, 25354 CrossRef CAS PubMed.
- S. Rajappan Achary, S. Agouram, J. F. Sánchez-Royo, M. C. Martínez-Tomás and V. Muñoz-Sanjosé, RSC Adv., 2014, 4, 23137 RSC.
- R. M. Allam, N. M. A. El-Nasr, M. A. Elbaset, D. O. Saleh and A. M. A. El-Seidy, Sci. Rep., 2025, 15, 15477 CrossRef CAS PubMed.
- R. Svoboda, L. P. Maqueda, V. Podzemná, A. Perejon and O. Svoboda, J. Non-Cryst. Solids, 2020, 528, 119738 CrossRef CAS.
- J. Du, Z. Zhu, J. Yang, J. Wang and X. Jiang, Heritage Sci., 2021, 9, 45 CrossRef CAS.
- I. V. Gordovskaya, T. Hashimoto, J. Walton, M. Curioni, G. E. Thompson and P. Skeldon, J. Electrochem. Soc., 2014, 161, C601–C606 CrossRef CAS.
- J. F. Watts and J. Wolstenholme, Comparison of XPS and AES with Other Analytical Techniques, in An Introduction to Surface Analysis by XPS and AES, ed. J. F. Watts and J. Wolstenholme, 2019, DOI:10.1002/9781119417651.ch8.
- J. Am. Ceram. Soc., 2016, 99, 363 Search PubMed.
- D. Du, Y. Zeng, C. Wang, M. Li, F. Wang, Z. Xu and H. Wang, Opt. Mater., 2021, 114, 110957 CrossRef CAS.
- S. Chakrabarti, S. Ginnaram, S. Jana, Z.-Y. Wu, K. Singh, A. Roy, P. Kumar, S. Maikap, J.-T. Qiu, H.-M. Cheng, L.-N. Tsai, Y.-L. Chang, R. Mahapatra and J.-R. Yang, Sci. Rep., 2017, 7, 4735 CrossRef PubMed.
- S. A. E. Bashandy, M. A. Elbaset, F. A. A. Ibrahim, S. S. Abdelrahman, S. A. A. Moussa and A. M. A. El-Seidy, Sci. Rep., 2025, 15, 5709 CrossRef PubMed.
- S. A. Bashandy, M. A. Elbaset, F. A. Ibrahim, S. A. A. Moussa, N. A. A. El-Latif, S. M. Afifi, T. Esatbeyoglu and A. M. El-Seidy, J. Trace Elem. Miner., 2025, 14, 100260 CrossRef.
- A. Mohamed, A. A. Galhoum, A. Saad and S. Wageh, Int. J. Biol. Macromol., 2025, 321, 146329 CrossRef CAS PubMed.
- N. Hamad, A. A. Galhoum, A. Saad and S. Wageh, J. Mol. Liq., 2024, 409, 125349 CrossRef CAS.
- M. Sayyed, M. Mhareb, M. Hamad, A. J. Kadhim, K. M. Kaky and Y. Maghrbi, Results Phys., 2025, 75, 108356 CrossRef.
- J. V. Gunha, A. Somer, S. d. R. F. Sabino, R. El-Mallawany, A. Novatski and R. F. Muniz, Ceram. Int., 2025, 51, 42377–42388 CrossRef CAS.
- G. Almisned, Z. Khattari, D. Sen Baykal, G. Susoy, G. Kilic, A. Ene and H. Tekin, J. Mater. Res. Technol., 2023, 27, 7582–7592 CrossRef CAS.
- M. M. Ismail, A. Darwish and H. Abo-Mosallam, Ceram. Int., 2025, 51, 42496–42508 CrossRef CAS.
- I. Mahmoud, M. Gaafar, S. Marzouk, A. Okasha and H. Elsaghier, Phys. B, 2025, 714, 417526 CrossRef CAS.
- I. M. Nabil, A. M. A. El-Seidy, A. T. Mosleh, H. Y. Zahran, S. H. Zyoud and I. S. Yahia, J. Mater. Sci.: Mater. Electron., 2024, 35, year Search PubMed.
- S. A. E. Bashandy, A. M. A. El-Seidy, F. A. A. Ibrahim, S. S. Abdelrahman, S. A. Abdelmottaleb Moussa and M. A. ElBaset, Sci. Rep., 2023, 13, 16010 CrossRef CAS PubMed.
- G. A. Alharshan, A. M. A. El-Seidy, M. I. Elamy, I. M. Nabil, A. M. El-Refaey, R. A. Elsad, M. S. Shams, A. M. Abdelghany and Y. S. Rammah, J. Mater. Sci.: Mater. Electron., 2024, 35, 862 CrossRef CAS.
- M. Abd Elwanees, Mat. J. Vet. Med., 2023, 3, 50–57 Search PubMed.
- M. A. E. M. Sayed, N. A. A. E. Latif, F. A. H. A. S. Ibrahim, A. M. A. El-Seidy, S. A. A. Moussa and S. A. E. M. Bashandy, Braz. Arch. Biol. Technol., 2024, 67, 1 Search PubMed.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article c and
Ahmed M. A. El-Seidy
c and
Ahmed M. A. El-Seidy *d
*d

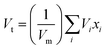
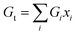



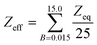






![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in B2O3.45 HRSf of C are shown in Fig. 1i and j. The peaks around 284 and 286 eV were assigned to sp2 hybridized carbon (C
O in B2O3.45 HRSf of C are shown in Fig. 1i and j. The peaks around 284 and 286 eV were assigned to sp2 hybridized carbon (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) and C–OH, respectively.46,47 The peaks @ 285.21, 288.45, and 292.76 eV were attributed to sp3 C–C bonding, (C
C) and C–OH, respectively.46,47 The peaks @ 285.21, 288.45, and 292.76 eV were attributed to sp3 C–C bonding, (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)–O, and C–O/π–π * shake-up satellite.29,48,49 HRSf of Cd (Fig. 2a and b) show doublet in 411.54–411.81 and 404.65–404.98 eV regions which were attributed to Cd-3d3/2 and Cd-3d5/2 with separation around 6.8 for a single CdO-related phase.50 HRSf of O are shown in Fig. 1c and d. The peaks in 529.73–531.24 and 532.03–532.28 eV are attributed to MO (Pb–O–Si/PbO2/CdO) and chemisorbed oxygen/Si–O–Si in bridging oxygen (BO)/Pb–O–Pb.49,51–53 The peak at 533.14 eV was assigned to Si–OH, which further support the presence of Si1+ while that around 537 eV was due to Pb–Cd–O.53–57 HRSf of Ba (Fig. 2e and f) show doublet in 794.77–795.19 and 779.57–779.99 eV regions which were attributed to Ba-3d3/2 and Ba 3d5/2 peaks of BaO2, respectively.58 The absence of any peaks around 775 eV indicate the absence of BaO.58 HRSf of Ce are shown in Fig. 2g.
O)–O, and C–O/π–π * shake-up satellite.29,48,49 HRSf of Cd (Fig. 2a and b) show doublet in 411.54–411.81 and 404.65–404.98 eV regions which were attributed to Cd-3d3/2 and Cd-3d5/2 with separation around 6.8 for a single CdO-related phase.50 HRSf of O are shown in Fig. 1c and d. The peaks in 529.73–531.24 and 532.03–532.28 eV are attributed to MO (Pb–O–Si/PbO2/CdO) and chemisorbed oxygen/Si–O–Si in bridging oxygen (BO)/Pb–O–Pb.49,51–53 The peak at 533.14 eV was assigned to Si–OH, which further support the presence of Si1+ while that around 537 eV was due to Pb–Cd–O.53–57 HRSf of Ba (Fig. 2e and f) show doublet in 794.77–795.19 and 779.57–779.99 eV regions which were attributed to Ba-3d3/2 and Ba 3d5/2 peaks of BaO2, respectively.58 The absence of any peaks around 775 eV indicate the absence of BaO.58 HRSf of Ce are shown in Fig. 2g.

 =
=  (GC0.5),
(GC0.5),  (GC1 G),
(GC1 G),  (GC2) and
(GC2) and  (GC4), respectively. This results may be due to the density difference between CeO2 (7.22 g cm−3) and B2O3 (2.46 g cm−3).
(GC4), respectively. This results may be due to the density difference between CeO2 (7.22 g cm−3) and B2O3 (2.46 g cm−3).











 =
=  (GC0.5),
(GC0.5),  (GC1),
(GC1),  (GC2) and
(GC2) and  (GC4), respectively. This order agrees with the increase in densities (Table 3). MAC show similar behavior to LAC (Table 5 and Fig. 8b), showing maximum value at 0.05 eV then the values decrease sharply with increasing γE followed by a gradual decrease. The decrease of MAC values as the γE increases may be due to the decrease in the effectiveness of Compton scattering and photoelectric absorption. MAC of basic glass was improved by 1.57%, 3.13%, 6.20% and 5.57% with
(GC4), respectively. This order agrees with the increase in densities (Table 3). MAC show similar behavior to LAC (Table 5 and Fig. 8b), showing maximum value at 0.05 eV then the values decrease sharply with increasing γE followed by a gradual decrease. The decrease of MAC values as the γE increases may be due to the decrease in the effectiveness of Compton scattering and photoelectric absorption. MAC of basic glass was improved by 1.57%, 3.13%, 6.20% and 5.57% with  =
=  (GC0.5),
(GC0.5),  (GC1 G),
(GC1 G),  (GC2) and
(GC2) and  (GC4), respectively, indicating the superior effect of higher density and molecular weight (CeO2: 7.22 g cm−3, 174 g mol−1, and B2O3: 2.46 g cm−3, 69.62 g mol−1).37,41,68 The lower Zeff values indicate a higher shielding power at lower γE. The increase of Zeff values (at γE = 1.00 eV: 0.83% (GC0.5), 1.66% (GC1), and 3.32% (GC2) and 6.65% (GC4)) with
(GC4), respectively, indicating the superior effect of higher density and molecular weight (CeO2: 7.22 g cm−3, 174 g mol−1, and B2O3: 2.46 g cm−3, 69.62 g mol−1).37,41,68 The lower Zeff values indicate a higher shielding power at lower γE. The increase of Zeff values (at γE = 1.00 eV: 0.83% (GC0.5), 1.66% (GC1), and 3.32% (GC2) and 6.65% (GC4)) with  replacement is due to adding Ce (58) with higher atomic number than B (5).48,69 σ-atomic, and σ-electron (Table 5 and Fig. 8e, f) show the same behavior, their values decrease as the γE increases, since the probability of the photoelectric effect decreases as photon energy increases.48,70 HVL and TVL show similar behavior (Table 5 and Fig. 8c, d) showing minimum values at 0.015 eV and increase as the γE increases which indicate the need to increase the thickness with increasing photon energy.47,71,72 At γE = 0.0150 eV:
replacement is due to adding Ce (58) with higher atomic number than B (5).48,69 σ-atomic, and σ-electron (Table 5 and Fig. 8e, f) show the same behavior, their values decrease as the γE increases, since the probability of the photoelectric effect decreases as photon energy increases.48,70 HVL and TVL show similar behavior (Table 5 and Fig. 8c, d) showing minimum values at 0.015 eV and increase as the γE increases which indicate the need to increase the thickness with increasing photon energy.47,71,72 At γE = 0.0150 eV:

 content (−4.55% (GC0.5), −6.82% (GC1), −9.09% (GC2), −13.64% (GC4)) indicate a higher shielding power (less thickness of the glass).
content (−4.55% (GC0.5), −6.82% (GC1), −9.09% (GC2), −13.64% (GC4)) indicate a higher shielding power (less thickness of the glass).



