 Open Access Article
Open Access ArticleMagnetically recoverable nano-adsorbents for dual removal of organic dyes and heavy metals from aqueous media
Olufemi Ogunjimi ,
John Verdaris,
Fatema Tarannum
,
John Verdaris,
Fatema Tarannum ,
Kayla Foley
,
Kayla Foley and
Keisha B. Walters
and
Keisha B. Walters *
*
Ralph E. Martin Department of Chemical Engineering, University of Arkansas, Fayetteville, AR 72701, USA. E-mail: keisha.walters@uark.edu
First published on 11th May 2026
Abstract
Freshwater contamination by heavy metals and synthetic dyes poses serious environmental and health risks, driving the need for efficient, cost-effective, and recoverable remediation technologies. Magnetic nanoparticles (MNPs) have emerged as a promising platform due to their high surface area and easy magnetic recovery. However, tailoring their surface chemistry is essential to maximize pollutant uptake and reusability. In this work, functionalized Fe3O4 MNPs were developed via chemical co-precipitation followed by surface functionalization with 3-aminopropyl trimethoxysilane (APTES), glutaraldehyde (GA), and citric acid (CA), yielding MNP-APTES, MNP-APTES-GA, and MNP-CA, respectively, for the efficient removal of heavy metal ions and organic pollutants from aqueous solutions. Physicochemical characterization—including FTIR spectroscopy, dynamic light scattering and zeta potential, electron microscopy, and thermogravimetric analysis—confirmed successful surface modification without compromising magnetic properties of the Fe3O4 core. Adsorption performance was assessed for copper(II) ions (Cu2+) in the presence of various competing heavy metal ions as well as for methyl orange (MO) dye. MNP-APTES-GA outperformed bare MNPs, MNP-CA, and MNP-APTES, achieving 69% Cu2+ removal at pH 6 and 83% MO decolorization at pH 3. Kinetic analysis demonstrated that Cu2+ uptake followed pseudo-first-order kinetics, whereas MO dye removal followed pseudo-second-order kinetics. The maximum Cu2+ adsorption capacity from the Langmuir model was 14.39 mg g−1, and both Langmuir and Freundlich isotherms fit the experimental data well, suggesting heterogeneous, multilayer and competitive adsorption behavior. In multicomponent systems containing Co2+, Ni2+, Zn2+, and Al3+, Cu2+ removal remained 22–37%, confirming preferential Cu2+ uptake under competitive conditions. MNP regeneration experiments demonstrated robust reusability and good retention of adsorption capacity after five cycles using suitable eluents. Regeneration studies revealed strong reusability, with MO desorption remaining above 80% after five cycles, while Cu2+ desorption decreased from ∼76% to ∼30%. The enhanced dual-removal performance of MNP-APTES-GA is attributed to synergistic interactions from amine and aldehyde functionalities, enabling combined electrostatic attraction, coordination, and supramolecular interactions. These findings establish MNP-APTES-GA as a versatile, magnetically recoverable adsorbent with significant potential for scalable, multi-contaminant water remediation applications.
1. Introduction
Ensuring access to clean water is essential for human health and sustainable development. However, water pollution continues to rise due to rapid industrialization, population growth, and intensified domestic and agricultural activities, which when unabated contribute to the prevalence of life-threatening diseases.1,2 Industrial effluents originating from textile,3 chemical,4 mining,5 and metallurgical6 sectors discharge large quantities of hazardous substances such as dyes, heavy metals, and other hazardous pollutants into aquatic systems. These toxic contaminants cause severe ecological damage and present carcinogenic and mutagenic risks to human populations.7 Therefore, increasing attention has been directed toward the development of efficient and sustainable strategies for removing hazardous dyes and toxic heavy metals from wastewater discharged by manufacturing industries.8,9Various conventional remediation methods such as chemical precipitation, ion exchange, membrane filtration, coagulation, and flocculation3,10–12 have been utilized for the removal of heavy metal ions and synthetic dyes from contaminated water. Although effective under certain conditions, these methods are often constrained by drawbacks, including high operational costs, secondary sludge production, membrane fouling, and limited efficiency at low pollutant concentrations.6,13 In addition, dye removal presents further challenges due to the structural stability, high solubility, and resistance to biodegradation of many azo and aromatic compounds. By comparison, adsorption stands out as one of the most attractive alternatives due to its high removal efficiency, cost-effectiveness, ease of use, potential for reuse, and suitability for large-scale water purification.10,13–15 Among various adsorbent materials, magnetic nanoparticles (MNPs)—particularly iron oxides—have gained significant attention owing to their high specific surface area, chemical tunability, environmental compatibility, and excellent magnetic responsiveness, which enable efficient adsorption and facile separation from aqueous systems without filtration or centrifugation.16–19 Iron oxide based nano-adsorbents such as hematite (α-Fe2O3), maghemite (γ-Fe2O3), and magnetite (Fe3O4) have been widely explored for the remediation of toxic metal ions including arsenic (As3+/As5+),20,21 chromium (Cr3+/Cr4+/Cr6+),22–24 manganese (Mn2+),25 copper (Cu2+),26 lead (Pb2+),27,28 and mercury (Hg2+)29,30 from both environmental and industrial waste streams.31–35 Magnetic recoverability of Fe3O4 makes it particularly attractive for sustainable wastewater treatment applications. In addition to metal ion removal, bare iron oxide nanoparticles have also demonstrated intrinsic adsorption capability toward various dye molecules such as methylene blue (MB),36 methyl orange (MO),37 Congo red (CR),38 and rhodamine dyes.39 However, unmodified Fe3O4 often suffer from aggregation due to magnetic dipole interactions and possess limited selectivity,40 which reduces their adsorption efficiency in practical applications. To overcome these challenges, tailoring the surface chemistry of iron oxide-based MNPs has emerged as an effective strategy to improve their colloidal stability, target selectivity, and overall adsorption performance.41,42
Surface functionalization of iron oxide nanoparticles (IONPs) have been achieved using a variety of moieties such as silane coupling agents, organic vapor deposition, physiosorbed and chemisorbed polymers, and surfactants.43,44 These modifications introduce functional groups capable of forming specific interactions with pollutant species while simultaneously preventing nanoparticle aggregation.45 Among the different silane coupling agents, (3-aminopropyl)triethoxysilane (APTES) has been widely used due to its ability to form stable Fe–O–Si bonds with surface hydroxyl groups of Fe3O4 while exposing terminal amine (–NH2) groups that serve as active sites for pollutant binding.46 According to Campaña et al., APTES-modified Fe3O4 NPs exhibited effective adsorption of Cd2+ ions from aqueous solution, achieving removal efficiencies up to 94% and a retention capacity of approximately 0.49 mg g−1. Adsorption followed Langmuir/Freundlich isotherms and pseudo-first-order and pseudo-second-order kinetic models, which indicated APTES-modified Fe3O4 NPs as promising low-cost platforms for aqueous heavy metal remediation.47 Besides silane-based functionalization, organic ligands such as citric acid (CA) have also been used to stabilize Fe3O4 nanoparticles through surface carboxylate groups.48,49 CA-coated magnetite nanoparticles (Fe3O4-CA) were synthesized specifically as effective adsorbents for cadmium (Cd2+) removal from aqueous solutions.50 The adsorption performance of Cd2+ was systematically evaluated with different pH, temperature, and contact time. Equilibrium in Cd2+ adsorption onto Fe3O4-CA was achieved within 35 min at an optimized pH of 5, confirming the rapid adsorption kinetics. Similarly, Tziro and Baseri51 investigated magnetite nano-adsorbents synthesized via chemical co-precipitation and subsequently coated with different organic and inorganic materials, including CA, for the adsorption of Cu2+and Pb2+. While bare Fe3O4 exhibited removal efficiencies of approximately 75% for Pb2+ and 78% for Cu2+, CA-coated Fe3O4 NPs achieved removal efficiencies above 90%, confirming the pivotal role of surface charge and ligand coordination in metal binding.
Beyond heavy metals, functionalized iron-based nanomaterials have demonstrated high efficiency in dye removal and multifunctional applications. Yan and Wang synthesized amino-functionalized Fe3O4 nanoparticles via APTES modification and demonstrated effective adsorption of anionic dyes, achieving maximum capacities of 51.4 mg g−1 for Reactive Red 228 and 118.8 mg g−1 for CR.52 Keshta and Gemeay53 synthesized Fe3O4 nanoparticles using different methods (co-precipitation, hydrothermal, and sonochemical) and subsequently modified with APTES to introduce surface amine (–NH2) groups, resulting in enhanced adsorption of the anionic dye Eosin-Y with a maximum adsorption capacity of 38.32 mg g−1. In another study, amino-functionalized silica-coated Fe3O4 nanoparticles were synthesized and found effective for dye adsorption such as CR and MB, which indicated the versatility of amine-modified magnetic cores in treating different organic pollutants.54 Alqadami et al. prepared trisodium-citrate-functionalized Fe3O4 (Fe3O4@TSC) which showed a maximum adsorption capacity of 435 mg g−1 for malachite green, demonstrating the strong adsorption capability imparted by citrate-derived surface carboxylate groups.55 The same citrate-modified Fe3O4-TSC NPs also showed rapid and efficient adsorption of Cr3+ and Co2+, with equilibrium adsorption capacities of 50 mg g−1 for Cr3+ and 47.4 mg g−1 for Co2+.56 The importance of carboxyl groups in adsorption processes is further illustrated by Qu et al., who modified water hyacinth biomass with CA to introduce dense carboxyl functionalities. The systematic investigation of adsorption parameters (solution pH, contact time, initial adsorbate concentration, adsorbent dosage, and temperature) showed optimized conditions of pH 7.5 for Ni2+, 6.0 for Cu2+, and 5.0 for Cr2+, with contact times of 300, 180, and 120 min, respectively.57 These findings reinforce that carboxyl groups play a critical role in mediating selective ion binding, a principle directly translatable to iron oxide surfaces where carboxyl, amine, or hydroxyl groups can be tailored to maximize adsorption efficiency. Importantly, these parameter-specific optimizations provide a practical guideline for scaling up the adsorption process.57 A comprehensive comparison of these studies, including adsorbent type, target pollutants, optimal pH conditions, adsorption capacities, and kinetic models, is summarized in Table 1, which provides a broader context for evaluating the performance of surface-functionalized Fe3O4-based adsorbents reported in the literature.
| Adsorbent (with functional groups) | Pollutant | Optimal pH | Removal efficiency (%) | Adsorption capacity (mg g−1) | Kinetics/isotherm | Reference |
|---|---|---|---|---|---|---|
| a SDS: sodium dodecyl sulfate; EDTA: ethylenediaminetetraacetic acid; GA: glutaraldehyde. | ||||||
| Amine-functionalized Fe3O4 | Cr4+ | 3 | 95.25–97.94 | 232.51 | Pseudo-second-order; Langmuir | Baghani et al.58 |
| Ni2+ | 6 | 96.42–98.56 | 222.12 | |||
| Carboxyl functionalized Fe3O4 | Pb2+ | ∼5–5.5 | — | 74.63 | Pseudo-second-order; Langmuir | Shi et al.59 |
| Cd2+ | 45.66 | |||||
| Cu2+ | 44.84 | |||||
| EDTA-Fe3O4 | Cu2+ | ∼5–6 | ∼98.46 ± 0.08 | ∼46.27 | Pseudo-second-order; Langmuir | Liu et al.60 |
| Fe3O4@SiO2 | Cd2+ | 6 | 55 | 81.62 | Langmuir and Freundlich | Ghafoor et al.61 |
| Ni2+ | 44 | 63.995 | ||||
| APTES-GA-Fe3O4 | Cu2+ | 6 | 86 | 19.26 | Pseudo-first-order; Langmuir isotherm | Özmen et al.62 |
| Fe3O4-APTES | Cu2+ | 4.0–5.3 | 78.2–79.6 | 61.07 | Langmuir/Freundlich analyzed; fast equilibrium (∼15 min) | Kothavale et al.63 |
| Citric acid-coated Fe3O4 | Crystal violet | ∼6–8 | 94.5% | ∼50–120 | Pseudo-second-order; Langmuir | Jangra et al.64 |
| Fe3O4–NH2–COOH | Pb2+ | ∼5–6 | 96.8% | 125.3 | Synergistic binding (dual sites) | Yang et al.65 |
| Cd2+ | 89.4% | 98.7 | ||||
| Fe3O4-SDS | Tolonium chloride | 6 | 98 | 111.11 | Pseudo-second-order, Langmuir | Abedi et al.66 |
| Fe3O4-APTES (amine-functionalized) | Reactive red 228 | ∼3–5 | 95.26 | 51.4 | Pseudo-second-order | Yan and Wang52 |
| Congo red | 4–10 | 94.2 | 118.8 | Langmuir, Freundlich | ||
| Fe3O4-APTES (amine-functionalized) | Eosin Y (EY) | ∼4–6 (best acidic) | ∼94–100 | 38.32 | Pseudo-second-order | Keshta & Gemeay53 |
| Langmuir | ||||||
| Fe3O4-APTES-GA | Cu2+ | 3 | 69% | 21.39 | Pseudo-first-order; Langmuir and Freundlich | This work |
| Methyl orange | 6 | 83% | 17.95 | Pseudo-second-order | ||
To further improve adsorption efficiency, dual-functional surface modification strategies combining different functional groups have also been explored. In many cases, APTES is used as an intermediate functionalization layer that introduces reactive amine groups and subsequently can react with aldehydes, carboxylic acids, or other functional molecules.67 Karimi et al. synthesized Fe3O4 NPs sequentially modified with APTES and glutaraldehyde (GA), which introduced surface amine and aldehyde functional groups to provide abundant coordination sites for metal binding. The resulting Fe3O4/APTES/GA NPs exhibited superior adsorption of thorium ions (Th4+) under optimized conditions of pH 4.5, 250 mg L−1 initial thorium concentration, 1 g L−1 adsorbent dosage, and 90 min contact time. The enhanced performance was attributed to the synergistic coordination of amine and aldehyde functionalities on the particle surface.68 Similarly, Yang et al.65 synthesized dual-functional amino–carboxyl co-modified Fe3O4 nanoparticles using APTES followed by succinic anhydride, which exhibited maximum adsorption capacities of 125.3 mg g−1 for Pb2+ and 98.7 mg g−1 for Cd2+. It was attributed to the combined presence of amine and carboxyl functional groups that significantly enhances metal ion binding through cooperative coordination and electrostatic interactions. Özmen et al.62 and Kothavale et al.63 both developed APTES-glutaraldehyde (GA) functionalized Fe3O4 NPs for Cu2+ removal from aqueous solutions, where surface amine and imine groups introduced by APTES and GA provided active coordination sites. Özmen et al.69 reported rapid adsorption kinetics with equilibrium reached within ∼15 min and a high Langmuir adsorption capacity of 0.961 mmol g−1 (≈61.07 mg g−1), whereas Kothavale et al.63 observed ∼86% Cu2+ removal at pH 6 with an adsorption capacity of 19.26 mg g−1 and equilibrium reached within ∼120 min, with the adsorption behavior fitting the Langmuir isotherm and pseudo-first-order kinetics. Despite these promising results, both studies are primarily limited to a single-component removal system, and their adsorption performance is strongly influenced by solution pH and competing ions, resulting in reduced efficiency under more complex conditions. In contrast, the present work extends beyond these limitations by demonstrating the simultaneous removal of Cu2+ ions and MO dye, thereby establishing a multifunctional adsorption platform. Furthermore, the inclusion of competitive multi-ion systems provides a more realistic evaluation of adsorbent performance, while the comparative analysis of different surface functionalization strategies offers deeper insight into the relationship between surface chemistry and adsorption behavior. These features highlight the enhanced versatility and practical applicability of the present system for wastewater treatment involving mixed contaminants.
In this study, Fe3O4 MNPs were synthesized via a one-pot co-precipitation and crosslinking strategy using 3-aminopropyl triethoxysilane (APTES), glutaraldehyde (GA), and citric acid (CA) to produce three functionalized materials-MNP-APTES, MNP-APTES-GA, and MNP-CA, respectively. The main objective of this work is to comparatively evaluate amine-, aldehyde/imine-, and carboxyl-functionalized IONPs to understand how different surface functionalization influence its adsorption behavior. The combination of surface functionalities is designed to synergistically enhance adsorption affinity, accelerate binding kinetics, and improve recyclability, thereby overcoming limitations of conventional single-ligand systems. The novelty of this work lies in the integration of multiple functional groups onto Fe3O4 MNPs to prepare MNP-APTES-GA and achieve remediation of both heavy metal (Cu2+) and dye (MO) within a single platform—an advancement rarely reported in the literature.63,69 In this study, Cu2+ was selected as a representative heavy metal due to its ubiquity in industrial effluents and high toxicity even at trace levels, while MO, a persistent azo dye, was chosen as a model organic contaminant owing to its persistence and resistance to biodegradation. Together, these pollutants reflect the realistic complexity of wastewater, where metals and dyes often coexist and require concurrent removal. Comprehensive characterization by Fourier transform infrared (FTIR) spectroscopy, zeta potential, thermogravimetric analysis (TGA), scanning electron microscopy (SEM), transmission electron microscopy (TEM), dynamic light scattering (DLS), X-ray diffraction (XRD), and ultraviolet-visible spectroscopy (UV-vis) confirms robust surface grafting without compromising magnetic responsiveness. These analyses provide structural, morphological, and functional verification of successful ligand immobilization. Furthermore, systematic evaluation of adsorption parameters, including contact time, pH, adsorbent dose, initial concentration, and coexisting ions, elucidated the adsorption pathways and maximized removal efficiencies. To elucidate the adsorption mechanisms, pseudo-first-order and pseudo-second-order kinetic models were applied to evaluate adsorption rates, while Langmuir and Freundlich isotherm models were used to analyze equilibrium adsorption behavior and surface interactions. To understand the adsorption mechanisms, pseudo-first-order and pseudo-second-order kinetic models were applied to evaluate the adsorption rates of Cu2+ and MO removal, while Langmuir and Freundlich isotherm models were used to analyze equilibrium adsorption behavior and surface interactions. Overall, by comparing APTES, APTES-GA, and CA functionalized Fe3O4 nanoparticles, this study provides a systematic understanding of how surface functionalization influences adsorption performance and recyclability in magnetic nanoadsorbents. Such scalable and reusable magnetic nanoparticles offer a promising and sustainable approach for the simultaneous removal of heavy metals and dyes from complex wastewater streams.
2 Materials and methods
2.1 Materials
Iron(III) chloride hexahydrate (FeCl3·6H2O, CAS 7789-21-1, ≥99% ACS) and iron(II) chloride tetrahydrate (FeCl2·4H2O, CAS 13478-10-9, ≥98%) were procured from Beantown Chemicals. Glutaraldehyde (GA, CAS 111-30-8, 25 wt% aqueous solution) and ethanol (anhydrous, CAS 64-17-5, ≥99.5%) were obtained from VWR Chemicals. 3-Aminopropyl triethoxysilane (APTES, CAS 919-30-2, ≥98%), ammonium hydroxide (NH4OH, CAS 1336-21-6, 28–30 wt% NH3 in H2O), ethylenediaminetetraacetic acid (EDTA, CAS 6381-92-6, ≥99.5%, disodium salt), and citric acid monohydrate (CA, CAS 77-92-9, ≥99.5%) were purchased from Thermo Fisher Scientific. Copper(II) sulfate pentahydrate (CuSO4·5H2O, CAS 7758-98-7, ≥99%), aluminum sulfate (Al2(SO4)3, CAS 10043-01-3, ≥97%), nickel(II) sulfate hexahydrate (NiSO4·6H2O, CAS 10101-97-0, ≥98%), cobalt(II) sulfate heptahydrate (CoSO4·7H2O, CAS 10026-24-1, ≥98%), and zinc sulfate heptahydrate (ZnSO4·7H2O, CAS 7446-20-0, ≥98%) were obtained from Sigma-Aldrich, and methyl orange dye (MO, CAS 547-58-0, ≥85% dye content) was purchased from LabChem, Inc. Deionized (DI) water was obtained from a MilliporeSigma water system. Branson SFX 150 ultrasonifier from Marshall Scientific was used to prevent aggregation during nanoparticle synthesis. All materials were used as received unless otherwise specified.2.2 Synthesis of magnetic nanoparticles (MNP)
The reaction mechanism for the synthesis and surface functionalization of Fe3O4 magnetic nanoparticles is shown in Fig. 1. MNPs were prepared using an improved co-precipitation method with iron(III) chloride hexahydrate (6.5 g) and iron(II) chloride tetrahydrate (3.5 g) mixed in 100 mL of distilled water at 80 °C. NH4OH (40 mL) was added dropwise and vigorously stirred into the solution under an inert atmosphere, with a rapid change in the solution colour from orange to black observed, indicating the formation of MNPs. The solution mixture was continuously stirred for an additional 30 min before proceeding to the washing step. The resulting black MNP particles were separated from the solution with a magnet and washed several times with deionized water and ethanol. Finally, a portion of the MNPs was redispersed into water while another portion was dispersed into ethanol for further characterization. The remainder was dried in an oven overnight at 40 °C70 to produce Fe3O4 MNPs. | ||
| Fig. 1 Schematic illustration of the synthesis and surface functionalization of MNPs by 3-aminopropyl triethoxysilane (APTES), glutaraldehyde (GA), and citric acid (CA). | ||
2.3 Surface modification of MNPs with 3-aminopropyl triethoxysilane (MNP-APTES)
Amino-functionalized MNPs were produced according to Ozmen et al.69 Fe3O4 NPs (2 g) were dispersed in 100 mL of an ethanol/water solution (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v ratio); afterward, 7.65 g of 3-aminopropyl triethoxysilane (APTES) was added to the solution with sonication under a nitrogen atmosphere at 40 °C for 2 h. The solution was then allowed to cool to room temperature, and the resulting MNP-APTES particles were separated from the solution using a permanent neodymium magnet. Aliquots of the particles were dispersed in ethanol for further characterization, while the remainder was dried in a vacuum oven overnight at 40 °C.
1 v/v ratio); afterward, 7.65 g of 3-aminopropyl triethoxysilane (APTES) was added to the solution with sonication under a nitrogen atmosphere at 40 °C for 2 h. The solution was then allowed to cool to room temperature, and the resulting MNP-APTES particles were separated from the solution using a permanent neodymium magnet. Aliquots of the particles were dispersed in ethanol for further characterization, while the remainder was dried in a vacuum oven overnight at 40 °C.
2.4 Surface modification of MNP-APTES with glutaraldehyde (MNP-APTES-GA)
MNP-APTES particles were further functionalized with glutaraldehyde (GA) based on the method described,30,69 where APTES modified MNPs (2 g) were dispersed in a 75 mL solution containing 12.5 wt% GA and stirred for 12 h at room temperature. Subsequently, the resulting suspension was washed three times with ethanol, then the particles were separated and washed 3× with DI water and finally dried in a vacuum oven at 40 °C overnight to yield MNP-APTES-GA.2.5 Surface modification of MNPs with citric acid (MNP-CA)
Fe3O4 MNPs were separately also surface modified with citric acid (CA) following a method by Dheyab et al.,71 where 100 mL of CA was added to a mixture of 2 g of bare MNP in 25 mL of ethanol. This solution was stirred for 30 min at 65 °C. The resulting MNP-CA particles were then collected through a permanent magnet and thoroughly rinsed four times with DI water to remove unreactive or inert impurities. Finally, the MNP-CA was redispersed in the DI water for 10 min and magnetically decanted after sonication. The resultant particles were dried in the oven overnight at 40 °C.2.6 Adsorption and desorption experiment
Adsorption studies were conducted to evaluate the removal of copper(II) sulfate pentahydrate and methyl orange (MO) using surface functionalized magnetic nanoparticles as adsorbents. The residual concentration of Cu2+ ions was determined spectrophotometrically at 744 nm using ethylenediaminetetraacetic acid (EDTA) as a complexing agent, and the corresponding removal efficiency was calculated. For copper adsorption experiments, MNPs at a dosage of 5 g L−1 were placed in 500 mg L−1 (2 mM) Cu2+ ion solution and agitated in a shaker at 250 rpm and 25 °C for a specified time to attain adsorption equilibrium. For MO adsorption, an initial dye concentration of 45.8 mg L−1 (0.14 mM) was used.To investigate the effect of key experimental factors, the rate of adsorption for the Cu2+ ions using MNPs was determined by varying contact time (30–210 min), adsorbent dose (2.5–15 g L−1 of MNPs), initial concentration (100–500 mg L−1), and pH (3–7). For MO adsorption, the parameters of contact time (15–120 min), 2 g L−1 adsorbent dose, and pH (3, 5, 10) were studied. At the end of each batch experiment, MNPs were decanted using a permanent magnet. The adsorption experiments were repeated three times, and the average values were reported. Fig. 2 depicts the adsorption and desorption process using MNPs as the adsorbent along with the chemical structures of adsorbates.
 | ||
| Fig. 2 (a) Schematic illustration of the adsorption and decantation process (adapted from Panda et al.72). Chemical structures of (b) copper(II) sulfate pentahydrate and (c) MO dye. | ||
The percentage adsorption conducted for Cu2+ and MO was deduced based on the following equations:
 | (1) |
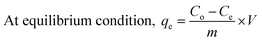 | (2) |
 | (3) |
Desorption and reusability studies were carried out after Cu2+ and MO adsorption using MNP-APTES-GA nanoparticles. For MO desorption, 2.5 g L−1 of MNP-APTES-GA was treated at pH 3, with a contact time of 60 min and an initial MO concentration of 45.8 mg L−1. In accordance with Shariati-Rad et al.,73 and Afkhami et al.,74 10 mL of 0.1 M NaOH was used as the eluent, followed by washing with distilled water before reuse. For Cu2+ desorption, MNP-APTES-GA (15 g L−1) was applied at pH 6 with a contact time of 90 min and an initial Cu2+ concentration of 100 mg L−1. Following literature reports,75–77 10 mL of 0.1 M HCl was used as the eluent, and the adsorbent was washed with distilled water before subsequent use. Dye concentration in the desorbed solution was measured spectrophotometrically.
The expression utilized for the desorption percentage was deduced from Zhai et al.78 as follows,
 | (4) |
 | (5) |
In(qe − qt) = In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe − k1t (integral expression) qe − k1t (integral expression)
| (6) |
 | (7) |
 | (8) |
2.7 Adsorption isotherms
Adsorption isotherms are essential for evaluating the adsorption capacity of materials and understanding adsorbate–adsorbent interactions at equilibrium.80–82 Among the most widely used models are the Langmuir, Freundlich, Temkin, and Dubinin–Radushkevich isotherms.79 The Langmuir model assumes that adsorption occurs as a monolayer on a homogeneous surface with a finite number of identical binding sites,83 and its linear form79 is expressed as
 | (9) |
The Freundlich isotherm, in contrast, is an empirical model that assumes a heterogeneous surface with multilayer adsorption,79 where adsorbate interactions occur and adsorption capacity increases with concentration; its linear form is given as
 | (10) |
2.8 Characterization methods
Fourier transform infrared spectroscopy (FTIR) was performed to characterize the surface functional groups of the MNPs using a ThermoFisher Scientific Nicolet iS50R FTIR spectrometer. To reduce water peak interference in the spectra, each sample was dried in a vacuum oven at 50 °C for 24 h before conducting the FTIR scans. FTIR spectra were collected over the 4000–400 cm−1 wavenumber range with an average of 64 scans at 4 cm−1 resolution and processed using OMNIC (Thermo Scientific™) software. The net surface charge of the nanoparticles was measured using an Anton Paar Litesizer 500 at room temperature, with each sample dispersed in an aqueous solution at 1 mg mL−1 concentrations.Thermogravimetric analysis (TGA) of the MNPs was conducted with a Discovery TGA 5500 instrument (TA Instruments). Approximately 15 mg of each sample was heated from 25 to 800 °C at a rate of 10 °C min−1 under a nitrogen flow rate of 20 mL min−1. The surface morphology of the samples was examined using scanning electron microscopy (SEM) on a Zeiss NEON high vacuum field-emission operated at an accelerating voltage of 15.00 kV.
Transmission electron microscopy (TEM) analysis was performed with a FEI Titan transmission electron microscope equipped with a field emission gun operating at 80 to 300 kV and an energy dispersive X-ray detector for elemental analysis. About 5 mg of each sample was ultrasonically dispersed in ethanol, and a drop of the suspension was deposited onto a carbon-coated copper grid and dried prior to imaging. A Shimadzu 1601 UV-Vis spectrophotometer was used to determine the concentration of anionic dye and Cu2+ in aqueous solution before, during, and after the adsorption process. Spectra were collected over the 1100–200 nm wavelength range at a spectral resolution of 1 nm. Calibration curves were produced by measuring the absorbance of a series of standard solutions with known concentrations of dye and Cu2+. The equipment was baseline corrected with DI water at the respective corresponding analytical wavelengths to ensure accuracy before analyzing the adsorbate solutions.
X-ray diffraction (XRD) patterns were recorded using a Philips X'Pert MPD diffractometer equipped with a 1.8 kW Cu Kα X-ray tube (λ = 0.15406 nm), a 1/2° divergence slit, a 0.2 mm receiving slit, and a flat monochromator positioned in front of the standard proportional detector. The average crystallite size (D) of the nanoparticles was estimated using the Debye–Scherrer equation.84
 | (11) |
3 Results and discussion
3.1 Characterization of MNPs
To evaluate the chemical composition of the as-synthesized (bare) MNPs and examine the success of the surface functionalization using APTES, GA, and CA, FTIR spectroscopy was used to identify the characteristic vibrational bands for the functional groups present in each sample. Fig. 3a–d displays the FTIR spectra of bare and functionalized Fe3O4 nanoparticles. The FTIR spectra for bare MNPs do not show the presence of any functional groups at absorbances above 800 cm−1, while an intense peak was observed at 537 cm−1 with this feature attributed to the Fe–O bond of bulk magnetite, as reported.30 The lack of other functional groups and the strong Fe–O band indicated the successful formation of bare magnetite nanoparticles. | ||
| Fig. 3 FTIR spectra of magnetic nanoparticle samples: (a) bare MNP; (b) MNP-APTES; (c) MNP-APTES-GA; and (d) MNP-CA. | ||
The surface modification of the MNPs with APTES was confirmed by the presence of the peaks at 1093 and 1031 cm−1 associated with Si–O groups, as well as two broad peaks at 1625 and 2927 cm−1, which were assigned to N–H stretching and the C–H stretch of the anchored propyl group, respectively. Similar peaks have been previously reported for APTES coatings by Ozmen et al.62 After the reaction of MNP-APTES with GA to obtain MNP-APTES-GA, a peak at 1709 cm−1 emerged, indicating the presence of the carbonyl group. Strong absorption bands at 937 and 1093 cm−1 were attributed to Si–O–Si groups, while peaks at 1625 and 1709 cm−1 were assigned to N–H stretching and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O carbonyl vibrations, respectively. Additional bands at 2927 and 2848 cm−1 indicated the presence of the propyl group63 for the MNP-APTES-GA, confirming that the APTES shell remained intact after the reaction with GA. In the FTIR spectra for MNP-CA, as shown in Fig. 3d, two strong bands at 1584 and 1387 cm−1 arise from vibrations of the citric-acid-derived carboxylate groups on the nanoparticle surface. Specifically, the 1584 cm−1 band corresponds to the asymmetric stretch of the –COO− moiety, while the 1387 cm−1 band is due to its symmetric stretch. All MNP samples exhibited a broad absorption band between 3200 and 3400 cm−1, characteristic of O–H stretching vibrations from surface hydroxyl groups.85 These hydroxyls are attributed to hydrogen bonding between oxygen atoms on the Fe3O4 nanoparticles and adsorbed water molecules or hydrogen species. Additionally, the intense absorption at ∼537 cm−1 is assignable to the Fe–O stretching vibration of the magnetite core. These spectral features confirm that citric acid was successfully grafted onto the MNP surface via its carboxylate functionalities, consistent with our group's previous observations and consistent with the work of Dheyab et al.71
O carbonyl vibrations, respectively. Additional bands at 2927 and 2848 cm−1 indicated the presence of the propyl group63 for the MNP-APTES-GA, confirming that the APTES shell remained intact after the reaction with GA. In the FTIR spectra for MNP-CA, as shown in Fig. 3d, two strong bands at 1584 and 1387 cm−1 arise from vibrations of the citric-acid-derived carboxylate groups on the nanoparticle surface. Specifically, the 1584 cm−1 band corresponds to the asymmetric stretch of the –COO− moiety, while the 1387 cm−1 band is due to its symmetric stretch. All MNP samples exhibited a broad absorption band between 3200 and 3400 cm−1, characteristic of O–H stretching vibrations from surface hydroxyl groups.85 These hydroxyls are attributed to hydrogen bonding between oxygen atoms on the Fe3O4 nanoparticles and adsorbed water molecules or hydrogen species. Additionally, the intense absorption at ∼537 cm−1 is assignable to the Fe–O stretching vibration of the magnetite core. These spectral features confirm that citric acid was successfully grafted onto the MNP surface via its carboxylate functionalities, consistent with our group's previous observations and consistent with the work of Dheyab et al.71
In general, colloidal solutions with |ζ| > 30 mV are considered stable, while those with |ζ| > 60 mV demonstrate exceptional stability.86 A high absolute zeta potential indicates that particles have a high net surface charge, which prevents particle aggregation due to charge repulsion. Conversely, colloidal solutions with |ζ| < 20 mV indicate poor stability, and those with a zeta potential between zero and ±5 mV tend to experience significant aggregation. Thus, the functionalized MNPs in this study exhibit superior suspension stability69 compared to bare MNPs. At near-neutral zeta potential, attraction forces overcome repulsion, leading to agglomeration; however, when two adjacent particles have sufficiently high zeta potential of the same sign, they do not agglomerate due to electrostatic repulsions. The results of this study clearly indicate that surface modification and functionalization significantly increase zeta potential, thereby enhancing colloidal stability, with the notable exception of MNP-CA, which achieves stabilization through negatively charged carboxylate groups.
To ascertain how the surface functional groups influence colloidal stability, zeta potential (ζ) measurements were measured for the various MNP samples just after synthesis and as a function of solution pH (Fig. 4). At the synthesis pH (≈6), bare MNPs exhibit a relatively low zeta potential of 10.6 ± 2.00 mV and so they are likely to have limited stability and a tendency toward aggregation, while MNP-APTES and MNP-APTES-GA have zeta potentials of 28.3 ± 1.06 mV and 39.4 ± 1.84 mV, respectively, as shown in Fig. 4a. These results suggest moderate stability for MNP-APTES and strong stability for MNP-APTES-GA, with improved dispersion relative to bare MNPs. As expected, the only MNP sample exhibiting a negative surface charge is MNP-CA, which had a measured zeta potential value of −36.0 ± 1.63 mV. This strong negative zeta potential is consistent with the presence of deprotonated carboxylate functional group,86 as confirmed by the FTIR analysis, and indicates MNP-CA should exhibit strong stability and effective dispersion in solution.
 | ||
| Fig. 4 Zeta potential of MNP samples measured (a) just after synthesis (pH ≈ 6) and (b) at different solution pH values. | ||
Understanding this pH-dependent surface charge behavior is critical for identifying conditions under which electrostatic attractions can maximize adsorption performance. The ζ vs. pH profiles (Fig. 4b) reveal how the protonation and deprotonation of fixed surface functional groups alter the nanoparticles' net charge across the pH range, shifting their isoelectric points accordingly. The isoelectric point (IEP), defined as the pH at which the particle's net surface charge is zero, corresponding to a zeta potential of 0 mV, provides a critical parameter for understanding colloidal stability. At this point, electrostatic repulsion is minimized, and particles are most prone to agglomeration due to attractive van der Waals forces. For the bare MNPs, the IEP was observed at pH 7.4, consistent with the presence of surface hydroxyl groups that undergo protonation and deprotonation around neutral conditions. Upon modification with APTES, the IEP shifted upward to pH 8.8, which reflects the contribution of protonatable amine groups on the surface; at lower pH, these groups are positively charged, enhancing surface charge density and stability. In contrast, CA functionalization shifted the IEP downward to pH 4.3 due to the abundance of deprotonated carboxylate groups, which impart a strong negative charge to the particle surface in neutral and basic media. For MNP-APTES-GA, the IEP was measured at pH 6.7, an intermediate value that reflects the combined influence of both amine (positively charged at acidic pH) and aldehyde-modified groups with partial negative character. Overall, these shifts do not imply any change in the chemical identity of the grafted moieties but rather reflect how the balance of positively and negatively charged sites evolves across the pH range, enabling precise control over colloidal stability and adsorption interactions in aqueous environments.
To corroborate the formation of coatings on the surface reflected by the deposition of organic and inorganic layers, the thermal stability of bare MNP, MNP-APTES, MNP-APTES-GA, and MNP-CA was studied using thermogravimetric analysis (TGA), as shown in Fig. 5. Upon heating from 0 to 800 °C, bare MNPs exhibited an initial drop of 1.8% between 25 °C and ∼150 °C which is attributable to the removal of physiosorbed water. Even up to 800 °C, the weight loss for the bare MNPs was minimal at 5.2%, indicating the inherent thermal stability of unmodified magnetite. In comparison, the functionalized nanoparticles displayed noticeably higher weight losses, consistent with the presence of grafted surface modifiers. A consistent weight decrease was observed between 120 and 150 °C across all samples, due to the removal of adsorbed water and trapped solvents on the surfaces of particles. For MNP-CA, the additional weight loss between 200 and 350 °C is attributed to loss of citric acid.87,88 MNP-APTES showed additional weight loss between 200–600 °C as the surface-grafted APTES layer decomposed into volatile fragments.11,30 In the case of MNP-APTES-GA, a more pronounced weight loss above 550 °C was observed, attributable to the combined decomposition of the silane and crosslinked glutaraldehyde networks.30 The relatively high weight loss recorded for MNP-APTES and MNP-APTES-GA can be attributed to solvent release from the porous, functionalized surfaces and the decomposition of the organic grafted ligands. A similar observation was reported by Ozmen et al.,69 further supporting this interpretation. The grafting ratio can be calculated through the equation:89
 | (12) |
 | ||
| Fig. 5 TGA analysis for the bare MNP and the surface functionalized MNP samples: MNP-APTES; MNP-APTES-GA; and MNP-CA. | ||
Based on the measured weight losses from TGA, the grafting ratio is calculated as 4.6 wt% for CA, 9.1 wt% for APTES, and 7.4 wt% for GA, confirming the successful immobilization of each surface modifier. In the case of the CA-functionalized nanoparticles, the grafting ratio (wt%) was determined from the difference in weight loss between MNP-bare and MNP-CA at 350 °C, the temperature of a distinct mass-loss event observed for MNP-CA, which is attributed to the thermal decomposition of surface-bound citrate moieties.87,88 The weight loss for MNP-APTES and MNP-APTES-GA was calculated from the overall weight loss difference between these samples and MNP-Bare at 800 °C. Overall, TGA analyses confirm the presence of organic and inorganic ligands on the surface of bare MNP, providing complementary evidence to FTIR and zeta potential analyses. Moreover, while surface functionalization slightly reduces thermal stability due to the introduction of organic coatings, this trade-off is offset by the enhanced surface chemistry, improved colloidal stability, and higher adsorption potential of the functionalized nanoparticles.
SEM analysis was performed to further examine the nanoparticles' morphology, as shown in Fig. 6. While both the bare MNP and MNP-APTES-GA samples showed a strong tendency to agglomerate when dried, this is expected for nanoparticles. SEM images (Fig. 6b) show that MNPs surface-functionalized with APTES and GA largely retain their spherical primary morphology, but compared to bare MNPs, the functionalized nanoparticles appear slightly larger and form more diffuse aggregates rather than packing tightly into compact clumps. The bare MNPs exhibited an average particle size of 21.25 ± 8.93 nm, while the APTES-GA functionalized MNPs showed an increased average size of 26.09 ± 5.34 nm, consistent with the presence of the surface modification layer. The surface-functionalized MNPs also appeared well-separated, indicating the formation of a consistent shell around each magnetic core. These changes highlight the effectiveness of the functionalization in stabilizing these nanoparticles, making them more suitable for use in broad applications, including wastewater treatment. Reduced agglomeration for the MNP-APTES-GA sample aligns well with the zeta potential results, which demonstrated enhanced colloidal stability due to the introduction of charged surface groups, and with the TGA data, which confirmed the presence of a significant organic surface modification layers through characteristic weight losses. These results are consistent with previous studies by Predescu et al.90 and Mohamed et al.91
The particle size distribution was examined by TEM, and Fig. 7 presents the data for bare MNPs (as-synthesized) prior to surface modification. TEM analysis reveals that the nanoparticles possess a spherical structure and exhibit a porous texture with a broad distribution. These impurity-free magnetic nanoparticles, characterized by their spherical, porous surfaces, are suitable additives for various applications and can be further functionalized to enhance superparamagnetic properties and mechanical robustness. However, it is important to note that TEM reflects the particles' dry-state morphology. In contrast, DLS had limitations in accurately measuring particles in suspension, often resulting in larger hydrodynamic sizes due to solvation layers, Brownian motion effects, and surface interactions, which can limit the accuracy of size determination in colloidal systems.92
 | ||
| Fig. 7 (a) TEM micrograph of bare magnetic nanoparticles (MNP) and (b) the corresponding particle size distribution. | ||
DLS confirmed that the optimized co-precipitation method yielded well-dispersed nanoparticle suspensions with relatively narrow size distributions, as shown in Fig. 8a. The average hydrodynamic diameters were 137.33 ± 3.96 nm, 186.23 ± 3.63 nm, 190.90 ± 2.78 nm, and 169.97 ± 2.82 nm for bare MNPs, MNP-APTES, MNP-APTES-GA, and MNP-CA, respectively. The hydrodynamic sizes of the MNPs increased upon surface functionalization due to the presence of grafted organic layers and associated solvation shells. Notably, the value for MNP-CA was 169.97 ± 2.82 nm, a slightly smaller hydrodynamic size than that of the other functionalized MNPs, likely due to the stabilizing influence of CA under optimized synthesis conditions, resulting in a narrow size distribution and reduced aggregation. Similar observations were reported by Sehlleier et al.,93 where surface modification led to a larger hydrodynamic diameter of 237 nm, attributed to the effect of the surface modification of the MNPs.
 | ||
| Fig. 8 (a) DLS particle size distribution and (b) X-ray diffractograms of bare MNPs, MNP-APTES, MNP-APTES-GA, and MNP-CA. | ||
To verify the crystalline phase and assess the structural stability of the nanoparticles after surface functionalization, X-ray diffraction (XRD) analysis was also performed. Fig. 8b presents the XRD patterns of bare MNPs, MNP-APTES, MNP-APTES-GA, and MNP-CA. All samples exhibit the characteristic diffraction peaks of magnetite (Fe3O4) at 2θ values of approximately 30.2° (220), 35.6° (311), 43.3° (400), 53.5° (422), 57.2° (511), and 62.8° (440), confirming the formation of a crystalline cubic spinel structure consistent with the JCPDS card no. 19-0629. The most intense peak was observed at 35.6° (311 plane), which is typical of magnetite and further validates the nanoparticles' crystalline nature. Importantly, no additional diffraction peaks corresponding to other iron oxide phases, such as maghemite (γ-Fe2O3) or hematite (α-Fe2O3), were detected, indicating that the crystalline structure of magnetite remained intact during surface modification. Using the Scherrer equation, the average size of MNP crystallites was estimated to be 7.67 nm. This crystal size matches other literature where the average MNP crystallite sizes obtained were 7.80 nm,84 7.66 nm,94 8.31 nm,95 and 12.0 nm.63 Moreover, the XRD analysis indicated that the functionalized samples (MNP-APTES, MNP-APTES-GA, and MNP-CA) show no discernible shift in peak positions, though a slight reduction in peak intensity and broadening is noticeable. These effects can be attributed to the presence of organic ligands on the MNP surface for the functionalized samples, which can slightly attenuate the diffraction signal and marginally reduce the apparent crystallinity signal without altering the core spinel phase.
3.2 Adsorption experiments
For Cu2+ adsorption (Fig. 9a), MNP-APTES-GA exhibited the highest removal efficiency (∼18%), followed by MNP-APTES (∼13%) and MNP-CA (∼11%), while bare MNPs achieved the lowest removal efficiency (∼8%). The removal efficiency of Cu2+ by bare MNPs, MNP-APTES, and MNP-APTES-GA increased gradually and reached equilibrium at 180 min, indicating saturation of the available active sites, On the other hand, MNP-CA showed a steady increase in Cu2+ removal until 150 min, after which equilibrium was attained. This clearly showed that the active sites were exhausted, causing no further adsorption of the Cu2+ ions.
In contrast, MO adsorption (Fig. 9b) showed a different performance order, with MNP-APTES-GA again demonstrating the highest efficiency (∼74%), followed by MNP-APTES (∼62%) and bare MNPs (∼47%), whereas MNP-CA displayed only negligible adsorption (∼3%). The adsorption of MO by bare MNPs, MNP-APTES, and MNP-APTES-GA increased steadily from 15 to 60 min and reached equilibrium at 90 min. These differences can be attributed to the distinct chemical functionalities introduced during surface modification, which alter the affinity of the nanoparticles toward different classes of pollutants. The superior uptake of Cu2+ by amine-functionalized nanoparticles is primarily due to the strong coordination ability of amine groups with metal ions, while the enhanced adsorption of MO is driven by electrostatic interactions between the negatively charged dye molecules and the functional groups present on the nanoparticle surface. Recently, Tziro and Baseri51 synthesized MNPs by co-precipitation and surface coated them with citric acid, ascorbic acid, salicylic acid, starch, and saccharose. The removal efficiency for Pb2+ increased steadily until it reached the equilibrium stage state of dynamic equilibrium,97 defined as the needed contact time required for adsorption and desorption rates to balance. Similarly, Jayalakshmi et al. reported that the adsorption of heavy metals (Pb2+ and Zn2+) and dyes (Congo red and methylene green) followed a similar pattern, characterized by rapid uptake at the beginning and a steady increase to equilibrium.98 In this study, optimization of factors such as initial concentration, pH, and adsorbent dose further confirmed that both equilibrium time and adsorption capacity depend strongly on the type of surface modification and the pollutant class being targeted.
There was a noticeable decrease in the removal efficiency of Cu2+ as the initial concentration of metal ions increased, as shown in Fig. 10b. This is because the adsorption sites are limited for a given adsorbent dose. For instance, the removal efficiency increased from 22% to 38% when the initial Cu2+ concentration decreased from 500 to 100 mg L−1. This was also confirmed in the study by Azzam et al., who reported similar behavior for Ni2+, Cu2+, Cd2+, and Pb2+ ions.101,102 The percentage of Cu2+ removal increases from 22 to 38% with a decrease in initial Cu2+ concentration from 500–100 mg L−1. The higher removal efficiencies at lower initial concentrations can be explained by the increasing driving force of the Cu2+ ions towards the adsorptive or active sites on the MNP-APTES-GA surface coupled with a higher adsorbate-to-site accessibility ratio. At elevated concentrations, the limited number of adsorption sites becomes saturated, leading to reduced percentage removal, even though the absolute amount of adsorbed metal increases. To ensure both practical relevance and analytical reliability, the range of 100–500 mg L−1 initial Cu2+ concentrations was restricted. This interval mirrors the typical pollutant load encountered in industrial effluents such as metal plating wastewater, while also aligning with the linear calibration range of the UV-vis assay. By doing so, the selected concentration window not only represents realistic environmental conditions but also guarantees sensitivity and accuracy in the measurement of Cu2+ removal.
The pH sensitivity of MNP-APTES-GA was evaluated for the adsorption of Cu2+ ion and MO. As shown in Fig. 11a, Cu2+ removal efficiency increased with rising pH, reaching a maximum of 69% at pH 6 for an initial concentration of 100 mg L−1, after which it declined. In contrast, MO adsorption exhibited its maximum removal efficiency of 83% at pH 3, as illustrated in Fig. 11b. This pH is considered as the operating pH for further experiments. Interestingly, post-adsorption measurements revealed slight shifts in solution pH toward neutrality, indicating buffering tendencies during MO adsorption. These opposite trends can be explained by changes in surface protonation of the MNP-APTES-GA. At low pH, nitrogen-containing groups introduced during APTES-GA functionalization, including imine (–CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–) linkages, are expected to undergo protonation to form –CH
N–) linkages, are expected to undergo protonation to form –CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NH+ species, thereby imparting a net positively charged surface.63 This protonation causes electrostatic repulsion toward Cu2+ ions, reducing metal adsorption, while simultaneously enhancing electrostatic attraction toward the negatively charged sulfonate group (–SO3−) of MO, resulting in higher dye uptake. Conversely, at higher pH values, decreased protonation favors the neutral imine groups for Cu2+ adsorption, enhancing metal adsorption but weakening interactions with MO. These complementary adsorption trends underscore the importance of pH in tuning the selectivity of MNP-APTES-GA for coexisting pollutants.
NH+ species, thereby imparting a net positively charged surface.63 This protonation causes electrostatic repulsion toward Cu2+ ions, reducing metal adsorption, while simultaneously enhancing electrostatic attraction toward the negatively charged sulfonate group (–SO3−) of MO, resulting in higher dye uptake. Conversely, at higher pH values, decreased protonation favors the neutral imine groups for Cu2+ adsorption, enhancing metal adsorption but weakening interactions with MO. These complementary adsorption trends underscore the importance of pH in tuning the selectivity of MNP-APTES-GA for coexisting pollutants.
Although iron oxide nanoparticles are susceptible to partial dissolution under strongly acidic conditions due to proton-assisted Fe–O bond cleavage,104 the APTES-GA surface modification provides a protective organic layer that improves resistance to acid-induced aggregation and surface degradation. Within the operational pH range (∼3–10) of this study, no observable loss of magnetic responsiveness, structural integrity, or adsorption performance was detected unlike prior studies.69 Furthermore, regeneration experiments demonstrated acceptable stability over multiple adsorption–desorption cycles. While very strong acidic conditions (pH < 3) may potentially lead to gradual iron leaching, such conditions are generally outside the typical operational range for wastewater treatment processes. Overall, the functionalized MNP-APTES-GA adsorbent demonstrates adequate chemical stability under the relevant experimental conditions.
 | ||
| Fig. 12 Effect of co-existing metal ions (Co2+, Ni2+, Zn2+, and Al3+) on the selective adsorption of Cu2+ by MNP-APTES-GA particles. | ||
The selective adsorption of Cu2+ can be explained by the coordination interactions between Cu2+ ions and the nitrogen-containing functional groups introduced during APTES-GA modification. The imine (–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–) and residual amine (–NH2) functionalities introduced through APTES-GA modification act as electron-donating ligands capable of forming surface coordination complexes with Cu2+ via lone-pair donation. Compared with competing divalent cations, Cu2+ exhibits stronger complexation affinity toward nitrogen-containing ligands due to its favorable distortion and higher ligand-field stabilization energy. These factors promote preferential surface complexation, consistent with recent reports highlighting the importance of specific coordination environments in driving selective heavy-metal adsorption in mixed-ion systems.107,108 In addition to metal-ion adsorption, the adsorbent also demonstrated effective removal of MO through a different adsorption mechanism. Selective adsorption under acidic conditions is primarily governed by electrostatic attraction and supramolecular interactions. At low pH, protonation of imine groups generates positively charged surface sites (–CH
N–) and residual amine (–NH2) functionalities introduced through APTES-GA modification act as electron-donating ligands capable of forming surface coordination complexes with Cu2+ via lone-pair donation. Compared with competing divalent cations, Cu2+ exhibits stronger complexation affinity toward nitrogen-containing ligands due to its favorable distortion and higher ligand-field stabilization energy. These factors promote preferential surface complexation, consistent with recent reports highlighting the importance of specific coordination environments in driving selective heavy-metal adsorption in mixed-ion systems.107,108 In addition to metal-ion adsorption, the adsorbent also demonstrated effective removal of MO through a different adsorption mechanism. Selective adsorption under acidic conditions is primarily governed by electrostatic attraction and supramolecular interactions. At low pH, protonation of imine groups generates positively charged surface sites (–CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NH+), enhancing electrostatic interaction with the anionic sulfonate (–SO3−) groups of MO. Furthermore, hydrogen bonding and other interactions between aromatic moieties of MO and surface functionalities contribute to chemisorption behavior, as confirmed by FTIR spectral shifts after adsorption. Unlike Cu2+, which possibly relies on coordination interactions, MO adsorption proceeds through multimodal interactions involving electrostatic attraction, hydrogen bonding, and van der Waals forces, leading to heterogeneous multilayer adsorption.109 Overall, the high selectivity observed in complex systems is not governed by a single interaction mechanism but rather by the synergistic interplay of surface complexation, electrostatic forces, and supramolecular interactions. Although, Cu2+ and MO adsorption were evaluated independently under their respective optimized conditions, the multifunctional surface chemistry of MNP-APTES-GA enables effective removal of both heavy metal ions and anionic dyes, highlighting its strong potential for application in multi-contaminant wastewater systems.
NH+), enhancing electrostatic interaction with the anionic sulfonate (–SO3−) groups of MO. Furthermore, hydrogen bonding and other interactions between aromatic moieties of MO and surface functionalities contribute to chemisorption behavior, as confirmed by FTIR spectral shifts after adsorption. Unlike Cu2+, which possibly relies on coordination interactions, MO adsorption proceeds through multimodal interactions involving electrostatic attraction, hydrogen bonding, and van der Waals forces, leading to heterogeneous multilayer adsorption.109 Overall, the high selectivity observed in complex systems is not governed by a single interaction mechanism but rather by the synergistic interplay of surface complexation, electrostatic forces, and supramolecular interactions. Although, Cu2+ and MO adsorption were evaluated independently under their respective optimized conditions, the multifunctional surface chemistry of MNP-APTES-GA enables effective removal of both heavy metal ions and anionic dyes, highlighting its strong potential for application in multi-contaminant wastewater systems.
 | ||
| Fig. 13 Contrasting desorption and reusability behavior of MNP-APTES-GA showing effective regeneration for (a) MO and limited regeneration for (b) Cu2+. | ||
In contrast, Cu2+ desorption (Fig. 13b) began at ∼76% when HCl was used as the eluent, indicating that acid effectively disrupts Cu2+–imine coordination bonds. However, unlike MO desorption, a sharper decline in efficiency was observed over successive cycles. After the second and third cycles, the desorption efficiency decreased to ∼62% and ∼52%, respectively, while a substantial reduction to ∼30% was recorded in the fifth cycle. The stronger decline can be attributed to the formation of stable coordination complexes with imine groups and potential adsorbent degradation under acidic conditions, as supported by the observed ∼15% material loss after repeated acid exposure. Although the adsorbent retained some reusability for Cu2+, these results suggest that its long-term regeneration efficiency is limited under acidic conditions and optimization of desorption conditions would be necessary for improved recovery. The findings of this reusability study highlight that while MNP-APTES-GA demonstrates excellent durability and regeneration efficiency for MO dye removal, its performance in repeated Cu2+ desorption cycles could stand to be improved through the development of more suitable eluents or milder regeneration conditions.
3.3 Adsorption kinetics
Adsorption kinetic studies were performed to examine the relationship between adsorption capacity and time, providing insights into adsorption rates and equilibrium mechanisms.79 The pseudo-first-order model generally describes physisorption, while the pseudo-second-order model often represents chemisorption processes. In this study, the kinetic modelling results revealed distinct behaviors for Cu2+ and MO adsorption on MNP-APTES-GA as shown in Fig. 14 and 15 and Table 2. For Cu2+ adsorption, the correlation coefficient (R2 = 0.8885) was higher for the pseudo-first-order model compared to the pseudo-second-order model (R2 = 0.8091), indicating that the adsorption process is better described by the pseudo-first-order kinetics. This suggests that Cu2+ adsorption is dominated by physisorption mechanisms rather than strong chemical bonding. Moreover, the theoretical adsorption capacity (qe, cal) of 18.23 mg g−1 closely matched the experimental value (qe, exp) of 21.39 mg g−1 for the pseudo-first-order model, further supporting the applicability of this kinetic model for the Cu2+ adsorption process.| Adsorbate | Pseudo-first-order model | Pseudo-second-order model | ||||
|---|---|---|---|---|---|---|
| R2 | K1 (min−1) | qe (mg g−1) | R2 | K2 (g mg−1 min−1) | qe (mg g−1) | |
| Cu2+ | 0.8885 | 0.0147 | 21.390 | 0.8091 | 1.6 × 10−4 | 34.132 |
| MO | 0.9660 | 0.0571 | 10.891 | 0.9992 | 0.0151 | 17.951 |
In contrast, MO adsorption was better fitted by the pseudo-second-order model, with a correlation coefficient (R2 = 0.9992) that was higher than that of the pseudo-first-order model (R2 = 0.9660). The good agreement between the calculated and experimental qe values in the pseudo-second-order model indicates that MO adsorption tends toward chemisorption. The correlation coefficient is closer to unity for the pseudo-second order than the first-order model, which also indicates that their adsorption tends to be chemisorption held on by chemical bonds. This process is likely governed by electrostatic attraction between the positively charged surface functional groups of MNP-APTES-GA and the negatively charged dye molecules, as also reported in a similar study by Muthukumaran et al.81 Overall, these results demonstrate that Cu2+ adsorption on MNP-APTES-GA primarily follows physisorption, whereas MO adsorption is dominated by chemisorption, highlighting the dual adsorption mechanisms enabled by the multifunctional surface chemistry of the adsorbent.
3.4 Adsorption isotherms
From the adsorption isotherm models, it is evident that there is a direct relationship between the initial Cu2+ concentration and adsorption capacity. Fig. 16 illustrates the impact of the initial concentration on Cu2+, the uptake, and the corresponding Langmuir and Freundlich isotherms, while the estimated parameters for the isotherm are summarized in Table 3.| Metal | Langmuir isotherm | Freundlich isotherm | ||||
|---|---|---|---|---|---|---|
| R2 | K1 (L mg1) | Qm (mg g−1) | R2 | Kf (L mg−1) | n | |
| Cu2+ | 0.9944 | 0.0041 | 14.390 | 0.9950 | 0.2587 | 1.6710 |
The correlation coefficients (R2) for the Langmuir and Freundlich models are 0.9944 and 0.9950, respectively, indicating that both models provide an excellent fit to the experimental data, with the Freundlich model showing slightly better linearity (Fig. 16). This suggests that Cu2+ adsorption onto MNP-APTES-GA can occur through both monolayer adsorption on a homogeneous surface (Langmuir) and multilayer adsorption on a heterogeneous surface (Freundlich). Additionally, the dimensionless separation factor RL, which characterizes the favorability of Langmuir-type adsorption, was calculated to range from 0.71 to 0.33 for initial Cu2+ concentrations between 100 and 500 mg L−1. In general, RL > 1 denotes unfavorable adsorption, RL = 1 suggests linear adsorption, 0 < RL < 1 indicates favorable adsorption, and RL = 0 signifies irreversible adsorption. Since all calculated values fall between 0 and 1, the adsorption of Cu2+ onto MNP-APTES-GA is confirmed to be favorable. The adsorption mechanism of MO was further elucidated through FTIR analysis, as shown in Fig. 17 and Table 4. After adsorption, a decrease in the O–H band suggested hydrogen bonding between surface hydroxyl groups and dye molecules. A decrease in intensity and shift in stretching vibration of –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O from 1709 to 1695 cm−1 and changes in Si–O–Si bands attest to their participation in the adsorption process. Additionally, the appearance of new sharp peaks at 1595 cm−1 (–N
O from 1709 to 1695 cm−1 and changes in Si–O–Si bands attest to their participation in the adsorption process. Additionally, the appearance of new sharp peaks at 1595 cm−1 (–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– stretching), 1275 cm−1 (C–N stretching), and 1144 cm−1 (S
N– stretching), 1275 cm−1 (C–N stretching), and 1144 cm−1 (S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching of sulfonic groups), indicates the successful anchoring of the azo dye on the sorbent surface.110–112
O stretching of sulfonic groups), indicates the successful anchoring of the azo dye on the sorbent surface.110–112
| Dye structure | Key functional groups | Wavenumbers (cm−1) | References |
|---|---|---|---|
 |
Sulfonate group (–SO3−) | 1000–1200 cm−1 | Extremera et al.,112 Imene et al.,113 Wang et al.114 |
Azo linkage (–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–) N–) |
1594, 1605 cm−1 | Bozbay et al.,115 Wang et al.114 | |
| Aromatic rings | 700–1500 cm−1 | Extremera et al.,112 Imene et al.,113 Bozbay et al.115 | |
| C–N | 1278 cm−1 | Extremera et al.112 |
After five adsorption–desorption cycles, TGA was again performed to evaluate the structural stability of the adsorbent. After adsorption of MO and Cu2+, the overall thermal decomposition profiles remained largely comparable to that of the material, indicating that the functionalized coating remained structurally intact during the adsorption process after 5 cycles, as shown in Fig. 18. The residual weights at 800 °C were 79.8% for the initial adsorbent used, 79.9% after MO adsorption, and 79.1% after Cu2+ adsorption, suggesting only minor variation in the organic content of the material. These results confirm that the grafted organic layer remains stable after adsorption and that no significant degradation of the functionalized surface occurs during the adsorption process to the adsorbent. To further examine changes in surface charge following adsorption, zeta potential measurements were performed. The pristine MNP-APTES-GA exhibited a zeta potential value of 39.4 ± 1.84 mV, indicating a highly charged surface resulting from the protonated groups. After adsorption, the zeta potential decreased to 20.1 ± 2.60 mV for Cu2+ and 1.40 ± 1.21 mV for MO adsorption respectively, reflecting the interaction of the adsorbates with the active surface sites. The decrease in surface charge suggests partial occupation or neutralization of surface functional groups during adsorption while still maintaining surface stability.116 These observations support the proposed adsorption mechanism and confirm the structural stability of the adsorbent during the adsorption process.
It is important to note that the adsorption behavior differs between Cu2+ and MO due to the distinct nature of the adsorbates. Cu2+ ions are small, hydrated cations that predominantly undergo electrostatic attraction and surface complexation with negatively charged or electron-donating groups, resulting in monolayer adsorption consistent with the Langmuir model. In contrast, MO is a larger aromatic azo dye bearing sulfonic and azo functional groups, which interact via multiple mechanisms, including electrostatic attraction, hydrogen bonding, van der Waals forces, and π–π interactions. FTIR evidence confirms the active participation of these groups, leading to a more complex and heterogeneous adsorption process. Despite these differences in adsorption mechanisms and kinetics—Cu2+ following pseudo-first-order and MO following pseudo-second-order kinetics—the multifunctional surface chemistry of MNP-APTES-GA enables effective removal of both contaminants. Furthermore, the minimal changes observed in TGA profiles and zeta potential after adsorption confirm the structural stability and sustained activity of the adsorbent. Even though the adsorption kinetics differ among pollutants, this combination of dual functionality, stability, and reusability underscores the advantages of the present system over conventional single-pollutant adsorbents and highlights its potential for practical wastewater treatment applications.
4 Conclusions
This study demonstrates that magnetic nanoparticles can be efficiently synthesized via a simple chemical co-precipitation route and subsequently functionalized with 3-aminopropyltriethoxysilane, glutaraldehyde, and citric acid. Comprehensive characterization confirmed the successful attachment of each ligand without compromising the intrinsic magnetic properties of the nanoparticle core, while also revealing that dual silane-crosslinker modification of MNP-APTES-GA significantly enhances superior colloidal stability and dispersion compared to single-ligand systems. When evaluated for pollutant uptake, MNP-APTES-GA consistently outperformed bare MNP, MNP-CA, and MNP-APTES, achieving up to ∼69% Cu2+ removal at pH 6 under optimized contact time, adsorbent dose, initial concentration, and competing ions conditions, and ∼83% methyl orange removal at pH 3. These outcomes firmly establish its dual functionality toward simultaneous remediation of heavy metals and anionic organic dyes. Kinetic analyses demonstrated that Cu2+ adsorption follows a pseudo-first-order mechanism, whereas methyl orange conforms to a pseudo-second-order model. Equilibrium data for Cu2+ adsorption fit both Langmuir and Freundlich isotherms, indicating a hybrid monolayer-heterogeneous sorption processes. Importantly, in multicomponent wastewater containing Co2+, Ni2+, Zn2+, and Al3+, MNP-APTES-GA adsorbent exhibited a strong preferential affinity toward Cu2+, suggesting a multi-surface adsorption phenomenon rather than simple competitive inhibition. Furthermore, desorption and regeneration studies confirmed that the functionalized nanoparticles retain their moderate to high removal efficiency for both Cu2+ (∼76–30%) and methyl orange (∼91–80%) over five consecutive cycles. Overall, the results position MNP-APTES-GA as a robust, scalable, dual-functional, and recyclable adsorbent with significant promise for advanced water-purification applications.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this study are available within the article. Additional data are available from the corresponding author upon reasonable request.Acknowledgements
We appreciate Dr Mourad Benamara and Serhii Kryvyi of the Institute for Nanoscience and Engineering at the University of Arkansas for providing access to TEM instrumentation and XRD data collection and interpretation. We also thank Dr Preston Larson of the Samuel Roberts Noble Microscopy Laboratory at the University of Oklahoma for support with SEM imaging.References
- P. N. Dave and L. V. Chopda, J. Nanotechnol., 2014, 2014(1), 398569 Search PubMed.
- R. Bhateria and R. Singh, J. Water Proc. Eng., 2019, 31, 100845 CrossRef.
- K. Thirumoorthy and S. Krishna, J. Environ. Chem. Eng., 2020, 8, 103582 CrossRef.
- V. K. Gupta, A. Rastogi and A. Nayak, J. Colloid Interface Sci., 2010, 342, 135–141 CrossRef CAS PubMed.
- E. Iakovleva and M. Sillanpää, Environ. Sci. Pollut. Res., 2013, 20, 7878–7899 CrossRef CAS PubMed.
- M. Chalaris, D. A. Gkika, A. K. Tolkou and G. Z. Kyzas, Environ. Sci. Pollut. Res., 2023, 30, 119627–119653 CrossRef CAS PubMed.
- S. Singh, K. Barick and D. Bahadur, Nanomater. Nanotechnol., 2013, 3, 20 CrossRef PubMed.
- F. S. A. Khan, N. M. Mubarak, M. Khalid, R. Walvekar, E. C. Abdullah, S. A. Mazari, S. Nizamuddin and R. R. Karri, Environ. Sci. Pollut. Res., 2020, 27, 24342–24356 CrossRef CAS PubMed.
- P. Saharan, G. R. Chaudhary, S. Mehta and A. Umar, J. Nanosci. Nanotechnol., 2014, 14, 627–643 CrossRef CAS PubMed.
- S. Jin, B. C. Park, W. S. Ham, L. Pan and Y. K. Kim, Colloids Surf., A, 2017, 531, 133–140 CrossRef CAS.
- A. Abdolmaleki, S. Mallakpour and S. Borandeh, RSC Adv., 2015, 5, 90602–90608 RSC.
- A. A. Joshi, K. Chaudhari and G. Ragupathy, Environ. Sci. Water Res. Technol., 2026,(12), 421–456 RSC.
- R. Kandasamy, Sep. Purif. Technol., 2017, 188, 458–467 CrossRef.
- A. Masjedi, E. Askarizadeh and S. Baniyaghoob, Mater. Chem. Phys., 2020, 249, 122917 CrossRef CAS.
- K. Al-Saad, M. Amr, D. Hadi, R. Arar, M. Al-Sulaiti, T. Abdulmalik, N. Alsahamary and J. Kwak, Arab. J. Nucl. Sci. Appl., 2012, 45, 335–346 Search PubMed.
- S. Nizamuddin, M. Siddiqui, N. Mubarak, H. A. Baloch, E. Abdullah, S. A. Mazari, G. Griffin, M. Srinivasan and A. Tanksale, Nanoscale Materials in Water Purification, 2019, pp. 447–472 Search PubMed.
- F. Almomani, R. Bhosale, M. Khraisheh and T. Almomani, Appl. Surf. Sci., 2020, 506, 144924 CrossRef CAS.
- M. Faraji, Y. Yamini and M. Rezaee, J. Iran. Chem. Soc., 2010, 7, 1–37 CrossRef CAS.
- X. Zeng, G. Zhang, J. Zhu and Z. Wu, Environ. Sci. Water Res. Technol., 2022, 8, 907–925 RSC.
- S. R. Chowdhury, E. K. Yanful and A. R. Pratt, Environ. Earth Sci., 2011, 64, 411–423 CrossRef CAS.
- A. D. Abid, M. Kanematsu, T. M. Young and I. M. Kennedy, Aerosol Sci. Technol., 2013, 47, 169–176 CrossRef CAS PubMed.
- J. Zhang, S. Lin, M. Han, Q. Su, L. Xia and Z. Hui, Water, 2020, 12, 446 CrossRef CAS.
- S. Rajput, C. U. Pittman and D. Mohan, J. Colloid Interface Sci., 2016, 468, 334–346 CrossRef CAS PubMed.
- P. Yuan, M. Fan, D. Yang, H. He, D. Liu, A. Yuan, J. Zhu and T. Chen, J. Hazard. Mater., 2009, 166, 821–829 CrossRef CAS PubMed.
- Y. Liu, J. Bai, H. Duan and X. Yin, Chin. J. Chem. Eng., 2017, 25, 32–36 CrossRef CAS.
- X. S. Wang, L. Zhu and H. J. Lu, Desalination, 2011, 276, 154–160 CrossRef CAS.
- Y. Bagbi, A. Sarswat, D. Mohan, A. Pandey and P. R. Solanki, J. Environ. Chem. Eng., 2016, 4, 4237–4247 CrossRef CAS.
- P. Chen, M. P. Prange, D. Song, S. Lee, S. S. Lee, J. Liu, Y. Zhao, X. Wang, M. H. Engelhard and Y. Zhou, Environ. Sci. Technol., 2025, 59, 11862–11874 CrossRef CAS PubMed.
- H. A. Wiatrowski, S. Das, R. Kukkadapu, E. S. Ilton, T. Barkay and N. Yee, Environ. Sci. Technol., 2009, 43, 5307–5313 CrossRef CAS PubMed.
- V. J. Inglezakis, A. Kurbanova, A. Molkenova, A. A. Zorpas and T. S. Atabaev, Sustainability, 2020, 12, 5489 CrossRef CAS.
- K. Zargoosh, H. Abedini, A. Abdolmaleki and M. R. Molavian, Ind. Eng. Chem. Res., 2013, 52, 14944–14954 CrossRef CAS.
- X. Xin, Q. Wei, J. Yang, L. Yan, R. Feng, G. Chen, B. Du and H. Li, Chem. Eng. J., 2012, 184, 132–140 CrossRef CAS.
- D. S. Chaudhari, R. P. Upadhyay, G. Y. Shinde, M. B. Gawande, J. Filip, R. S. Varma and R. Zbořil, Green Chem., 2024, 26, 7579–7655 RSC.
- Y. Iqbal, W. Ma, X. Li, S. Fu, A. Shahzad, I. Ahmad, Y. Wang, M. Adil and M. Abohashrh, Water, Air, Soil Pollut., 2025, 236, 687 CrossRef CAS.
- M. S. Alshammari, M. M. El-Tyieb, M. E. Fawzy, E. S. Al-Farraj and H. M. Ahmed, PLoS One, 2025, 20, e0329686 CrossRef CAS PubMed.
- S. T. Aly, A. Saed, A. Mahmoud, M. Badr, S. S. Garas, S. Yahya and K. H. Hamad, Sci. Rep., 2024, 14, 20100 CrossRef CAS PubMed.
- B. Rahmanivahid, F. Naderi and H. Nayebzadeh, J. Water Environ. Nanotechnol., 2020, 5, 1–16 CAS.
- A. Afkhami and R. Moosavi, J. Hazard. Mater., 2010, 174, 398–403 CrossRef CAS.
- G. Vochiţa, A. R. Fânaru-Balint, A. Agavriloaei, D. Gherghel, M. Răcuciu and D. Creangă, Molecules, 2025, 30, 4447 CrossRef.
- D. Xiao, T. Lu, R. Zeng and Y. Bi, Microchim. Acta, 2016, 183, 2655–2675 CrossRef CAS.
- Y. Wanna, A. Chindaduang, G. Tumcharern, D. Phromyothin, S. Porntheerapat, J. Nukeaw, H. Hofmann and S. Pratontep, J. Magn. Magn. Mater., 2016, 414, 32–37 CrossRef CAS.
- B. Paul, V. Parashar and A. Mishra, Environ. Sci. Water Res. Technol., 2015, 1, 77–83 RSC.
- D. Chen, T. Awut, B. Liu, Y. Ma, T. Wang and I. Nurulla, e-Polymers, 2016, 16, 313–322 CrossRef CAS.
- K. Turcheniuk, A. V. Tarasevych, V. P. Kukhar, R. Boukherroub and S. Szunerits, Nanoscale, 2013, 5, 10729–10752 RSC.
- B. Natarajan, P. Kannan, J. A. Rather and R. A. Sheikh, J. Taiwan Inst. Chem. Eng., 2023, 147, 104942 CrossRef CAS.
- Y. Liu, Y. Li, X.-M. Li and T. He, Langmuir, 2013, 29, 15275–15282 CrossRef CAS PubMed.
- A. L. Campaña, A. Guillén, R. Rivas, V. Akle, J. C. Cruz and J. F. Osma, Molecules, 2021, 26, 4150 CrossRef PubMed.
- A. A. Pawar, R. Kumar, S. Sharma and T. Satyanarayana, Part. Part. Syst. Char., 2022, 39, 2200156 CrossRef CAS.
- R. K. Sharma, K. Solanki, R. Dixit, S. Sharma and S. Dutta, Environ. Sci. Water Res. Technol., 2021, 7, 818–860 RSC.
- D. Singh, R. K. Gautam, R. Kumar, B. K. Shukla, V. Shankar and V. Krishna, J. Water Proc. Eng., 2014, 4, 233–241 CrossRef.
- H. Baseri and S. Tizro, J. Adv. Mater. Process., 2016, 4, 15–29 Search PubMed.
- T.-g. Yan and L.-j. Wang, Water Sci. Technol., 2014, 69, 612–621 CrossRef CAS PubMed.
- B. E. Keshta and A. H. Gemeay, J. Hazard. Toxic Radioact. Waste, 2022, 26, 04021042 CrossRef CAS.
- R. Wo, Q.-L. Li, C. Zhu, Y. Zhang, G.-f. Qiao, K.-y. Lei, P. Du and W. Jiang, J. Chem. Eng. Data, 2019, 64, 2455–2463 CrossRef.
- A. A. Alqadami, M. Naushad, M. A. Abdalla, M. R. Khan and Z. A. Alothman, J. Chem. Eng. Data, 2016, 61, 3806–3813 CrossRef.
- A. A. Alqadami, M. Naushad, M. A. Abdalla, T. Ahamad, Z. A. Alothman and S. M. Alshehri, RSC Adv., 2016, 6, 22679–22689 RSC.
- W. Qu, D. He, Y. Guo, Y. Tang, J. Shang, L. Zhou, R. Zhu and R.-J. Song, Ind. Eng. Chem. Res., 2019, 58, 18508–18518 CrossRef CAS.
- A. N. Baghani, A. H. Mahvi, M. Gholami, N. Rastkari and M. Delikhoon, J. Environ. Health Sci. Eng., 2016, 14, 11 CrossRef.
- J. Shi, H. Li, H. Lu and X. Zhao, J. Chem. Eng. Data, 2015, 60, 2035–2041 CrossRef CAS.
- Y. Liu, M. Chen and H. Yongmei, Chem. Eng. J., 2013, 218, 46–54 CrossRef CAS.
- S. Ghafoor and S. Ata, J. Chil. Chem. Soc., 2017, 62, 3588–3592 CrossRef CAS.
- M. Ozmen, K. Can, G. Arslan, A. Tor, Y. Cengeloglu and M. Ersoz, Desalination, 2010, 254, 162–169 CrossRef CAS.
- V. Kothavale, V. Karade, P. Waifalkar, S. C. Sahoo, P. Patil and P. Patil, AIP Conf. Proc., 2018, 1942(1), 050038 CrossRef.
- A. Jangra, J. Singh, J. Kumar, K. Rani, P. Kumar, S. Kumar, D. Singh and R. Kumar, Biointerface Res. Appl. Chem., 2023, 13, 325 CAS.
- M. Yang, S. Dang, L. Gao and J. Zhang, Sci. Rep., 2026, 14(1), 11 Search PubMed.
- M. Abedi, M. Ahmadmoazzam and N. Jaafarzadeh, Chem. Biochem. Eng. Q., 2018, 32, 205–213 CrossRef CAS.
- F. Hosseini, M. Seyedsadjadi and N. Farhadyar, Orient. J. Chem., 2014, 30(4), 1609–1618 CrossRef CAS.
- M. Karimi, S. A. Milani and H. Abolgashemi, J. Nucl. Mater., 2016, 479, 174–183 CrossRef CAS.
- M. Ozmen, K. Can, G. Arslan, A. Tor, Y. Cengeloglu and M. Ersoz, Desalination, 2010, 254, 162–169 CrossRef CAS.
- J. C. Chacón-Torres, C. Reinoso, D. G. Navas-León, S. Briceño and G. González, Sci. Rep., 2020, 10, 19004 CrossRef.
- M. A. Dheyab, A. A. Aziz, M. S. Jameel, O. A. Noqta, P. M. Khaniabadi and B. Mehrdel, Sci. Rep., 2020, 10, 10793 CrossRef CAS PubMed.
- S. K. Panda, I. Aggarwal, H. Kumar, L. Prasad, A. Kumar, A. Sharma, D.-V. N. Vo, D. Van Thuan and V. Mishra, Environ. Chem. Lett., 2021, 19, 2487–2525 CrossRef CAS.
- M. Shariati-Rad, M. Irandoust, S. Amri, M. Feyzi and F. Ja’fari, Int. Nano Lett., 2014, 4, 91–101 CrossRef CAS.
- A. Afkhami, M. Saber-Tehrani and H. Bagheri, Desalination, 2010, 263, 240–248 CrossRef CAS.
- S. Sulaiman, R. a. S. Azis, I. Ismail, H. C. Man, K. F. M. Yusof, M. U. Abba and K. K. Katibi, Nanoscale Res. Lett., 2021, 16, 1–17 CrossRef.
- J. Bayuo, M. A. Abukari and K. B. Pelig-Ba, Appl. Water Sci., 2020, 10, 171 CrossRef CAS.
- M.-W. Wan, C.-C. Kan, B. D. Rogel and M. L. P. Dalida, Carbohydr. Polym., 2010, 80, 891–899 CrossRef CAS.
- L. Zhai, Z. Bai, Y. Zhu, B. Wang and W. Luo, Chin. J. Chem. Eng., 2018, 26, 657–666 CrossRef CAS.
- X. Wang, C. Jiang, B. Hou, Y. Wang, C. Hao and J. Wu, Chemosphere, 2018, 206, 587–596 CrossRef CAS.
- S. Singh, D. Kapoor, S. Khasnabis, J. Singh and P. C. Ramamurthy, Environ. Chem. Lett., 2021, 19, 2351–2381 CrossRef CAS.
- C. Muthukumaran, V. M. Sivakumar and M. Thirumarimurugan, J. Taiwan Inst. Chem. Eng., 2016, 63, 354–362 CrossRef CAS.
- P. Xu, G. M. Zeng, D. L. Huang, C. Lai, M. H. Zhao, Z. Wei, N. J. Li, C. Huang and G. X. Xie, Chem. Eng. J., 2012, 203, 423–431 CrossRef CAS.
- J. Chen, L. An, J. W. Heo, J. H. Bae, H. Jeong and Y. S. Kim, J. Wood Chem. Technol., 2022, 42, 114–124 CrossRef CAS.
- B. Malaikozhundan, R. Krishnamoorthi, J. Vinodhini, K. S. N. Nambi and S. Palanisamy, Inorg. Chem. Commun., 2022, 144, 109843 CrossRef CAS.
- S. Liaquat, S. Farrukh, N. Ahmad, S. S. Karim, E. Pervaiz, A. Sultan and S. Ali, Environ. Sci. Water Res. Technol., 2024, 10, 1637–1652 RSC.
- O. A. Attallah, M. A. Al-Ghobashy, M. Nebsen and M. Y. Salem, RSC Adv., 2016, 6, 11461–11480 RSC.
- A. Qureashi, A. H. Pandith, A. Bashir, T. Manzoor, L. A. Malik and F. A. Sheikh, Surf. Interfaces, 2021, 23, 101004 CrossRef CAS.
- D. Portilla, A. Velázquez-López, R. Escobedo and H. Yee-Madeira, AIMS Mater. Sci., 2024, 11(4), 991–1012 CAS.
- D. Jiang, Y. Huan, C. Sun, C. Hu, J. Guo, J. Long, M. A. Khan, D. P. Young and Z. Guo, RSC Adv., 2016, 6, 91584–91593 RSC.
- A. M. Predescu, E. Matei, A. C. Berbecaru, C. Pantilimon, C. Drăgan, R. Vidu, C. Predescu and V. Kuncser, R. Soc. Open Sci., 2018, 5, 171525 CrossRef PubMed.
- A. Mohamed, R. Atta, A. A. Kotp, F. I. Abo El-Ela, H. Abd El-Raheem, A. Farghali, D. H. M. Alkhalifah, W. N. Hozzein and R. Mahmoud, Sci. Rep., 2023, 13, 7227 CrossRef CAS.
- D. Kovář, A. Malá, J. Mlčochová, M. Kalina, Z. Fohlerová, A. Hlaváček, Z. Farka, P. Skládal, Z. Starčuk and R. Jiřík, J. Nanomater., 2017, 2017(1), 7859289 Search PubMed.
- Y. Sehlleier, S. Hardt, C. Schulz and H. Wiggers, J. Environ. Chem. Eng., 2016, 4, 3779–3787 CrossRef CAS.
- R. K. Wardani, K. Dahlan, S. T. Wahyudi and S. G. Sukaryo, AIP Conf. Proc., 2019, 2194(1), 020137 CrossRef.
- N. Mufti, D. Sari, A. Muyasaroh and A. Taufiq, J. Phys.:Conf. Ser., 2020, 1595(1), 012004 CrossRef CAS.
- K. Y. Kumar, H. Muralidhara, Y. A. Nayaka, J. Balasubramanyam and H. Hanumanthappa, Powder Technol., 2013, 246, 125–136 CrossRef CAS.
- A. K. Badawi, M. Abd Elkodous and G. A. Ali, RSC Adv., 2021, 11, 36528–36553 RSC.
- R. Jayalakshmi, J. Jeyanthi and K. A. Sidhaarth, Environ. Nanotechnol. Monit. Manag., 2022, 17, 100659 CAS.
- V. Chavan, V. Kothavale, S. Sahoo, P. Kollu, T. Dongale, P. Patil and P. Patil, Phys. B, 2019, 571, 273–279 CrossRef CAS.
- P. B. Patil and V. P. Kothavale, Advances in Metal Oxides and Their Composites for Emerging Applications, Elsevier, 2022, pp. 401–424 Search PubMed.
- A. M. Azzam, S. T. El-Wakeel, B. B. Mostafa and M. El-Shahat, J. Environ. Chem. Eng., 2016, 4, 2196–2206 CrossRef CAS.
- G. Mohamed, N. Hassan, A. Shahat, A. El-Didamony and A. Ashraf, Biointerface Res. Appl. Chem., 2021, 11, 13377–13401 Search PubMed.
- M. Ghasemi, S. Mashhadi and J. Azimi-Amin, J. Water Environ. Nanotechnol., 2018, 3, 321–336 CAS.
- R. M. Cornell and U. Schwertmann, The Iron Oxides: Structure, Properties, Reactions, Occurrences and Uses, John Wiley & Sons, 2003 Search PubMed.
- L. Lv, M. P. Hor, F. Su and X. Zhao, J. Colloid Interface Sci., 2005, 287, 178–184 CrossRef CAS PubMed.
- N. N. Nassar, J. Hazard Mater., 2010, 184, 538–546 CrossRef CAS PubMed.
- Y. Xie, Q. Rong, C. Wen, X. Liu, M. Hao, Z. Chen, H. Yang, G. I. N. Waterhouse, S. Ma and X. Wang, CCS Chem., 2024, 6, 1908–1919 CrossRef CAS.
- S. Chen, R. Ding, B. Li, J. Lu and X. Zhang, Sep. Purif. Technol., 2025, 354, 129451 CrossRef CAS.
- R. Ding, Y. Zhu, L. Jing, S. Chen, J. Lu and X. Zhang, Int. J. Biol. Macromol., 2024, 282, 137037 CrossRef CAS PubMed.
- D. C. Kalyani, A. A. Telke, S. P. Govindwar and J. P. Jadhav, Water Environ. Res., 2009, 81, 298–307 CrossRef CAS PubMed.
- T. Shen, C. Jiang, C. Wang, J. Sun, X. Wang and X. Li, RSC Adv., 2015, 5, 58704–58712 RSC.
- R. Extremera, I. Pavlovic, M. R. Pérez and C. Barriga, Chem. Eng. J., 2012, 213, 392–400 CrossRef CAS.
- K. Imene, B. C. Eddine, B. A. Salah, B. Riadh, A. Gil, B.-A. Imene, G. Messaoued and B. Mokhtar, Sep. Purif. Technol., 2025, 365, 132656 CrossRef CAS.
- K. Wang, Z. Song, Z. Xu, Y. Xi, Y. Cui and H. Zhou, RSC Adv., 2025, 15, 21121–21132 RSC.
- R. Bozbay and N. Orakdogen, Int. J. Biol. Macromol., 2025, 299, 140034 CrossRef CAS.
- S. Kamble, S. Agrawal, S. Cherumukkil, V. Sharma, R. V. Jasra and P. Munshi, ChemistrySelect, 2022, 7, e202103084 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |









