Open Access Article
Open Access ArticleExploring the synthesis of conduritol derivatives: strategies, challenges, and advances
Soumyadip Malik
a and
Amrit Krishna Mitra
 *b
*b
aDepartment of Chemistry, Rani Rashmoni Green University Tarakeswar, Hooghly, West Bengal Pin: 712410, India
bDepartment of Chemistry, Government General Degree College, Singur, Singur, Hooghly, West Bengal Pin: 712409, India. E-mail: amritsepistles@gmail.com; Tel: +91-33-2630-0126 Tel: +91 9432164011
First published on 8th April 2026
Abstract
Conduritol derivatives represent an important class of cyclitol-based molecules characterized by structural diversity, synthetic flexibility, and notable biological activity. Built on a cyclohexene core bearing multiple hydroxyl groups, conduritols have attracted sustained interest due to their potent glycosidase inhibitory properties and their occurrence in biologically active natural products such as pancratistatin and lycoricidine. The parent compound exists as six diastereomeric forms, and extensive methodological advances over recent decades have enabled access to all stereoisomers as well as a wide range of functionalized derivatives, including epoxidized, aminated, halogenated, and selectively protected analogues. Many of these compounds have shown promise as enzyme inhibitors and as leads for anticancer, antileukemic, and antimicrobial applications. Building on earlier work focused on the synthesis of conduritol stereoisomers, this review concentrates on strategies for the preparation of conduritol derivatives, emphasizing asymmetric synthesis, stereoselective functionalization, and recent developments in catalytic and green chemistry approaches. Key challenges related to regio- and stereocontrol are critically discussed, along with comparative assessments of established and emerging synthetic routes. The therapeutic relevance of conduritol derivatives, particularly in glycosidase-targeted interventions for metabolic and proliferative diseases, is also highlighted, providing an integrated perspective on current advances and future opportunities in this field. By consolidating and critically evaluating the diverse synthetic methodologies developed to date, this review aims to serve as a comprehensive resource for the research community, highlighting existing methodological gaps and guiding the design of more efficient, selective, and sustainable strategies for the synthesis and application of conduritol-based frameworks.
1. Introduction
Cyclitols, a structurally diverse class of cyclic polyols derived from a cyclohexane framework, are broadly categorized into three principal families: inositols, quercitols, and conduritols.1 Among these, conduritols have emerged as a distinctive and highly impactful subgroup owing to their intricate stereochemical features and significant biological relevance.2,3 Since Kübler's seminal 1908 report of a novel polyhydroxylated cyclohexene from Marsdenia condurango, later identified as conduritol A, these molecules have continued to inspire extensive research in organic synthesis, medicinal chemistry, and chemical biology.4The six theoretically possible diastereomers of conduritol, designated A through F, encompass two meso forms (A and D) and four chiral enantiomeric pairs (B, C, E, and F). While conduritol F is relatively abundant in nature, conduritol A is isolated only in trace amounts. Despite their limited natural availability, the conduritols have proven indispensable in synthetic organic chemistry, functioning as versatile intermediates and privileged scaffolds for the construction of structurally and biologically complex targets.5,6
Beyond their structural diversity, conduritols possess a remarkable breadth of biological activity. Numerous derivatives, including epoxides, aminoconduritols, cyclophellitols, and other functionalized analogues, display strong glycosidase inhibitory activity and have attracted considerable interest as potential therapeutic agents for metabolic and proliferative diseases, including diabetes, cancer, HIV/AIDS, and lysosomal storage disorders such as Gaucher's disease. Recent studies have further highlighted the importance of targeting glycosidase-related pathways and cyclitol-based inhibitors in therapeutic research.7 Additional reported activities, including antifeedant, antibiotic, antileukemic, and plant growth-regulating effects, further underscore their pharmacological importance. The presence of conduritol A in traditional medicinal plants such as Gymnema sylvestre illustrates the longstanding connection between these molecules and natural therapeutic practices.3,8,9
From a synthetic perspective, conduritols also serve as strategic building blocks in the total synthesis of natural products, particularly members of the Amaryllidaceae alkaloid family. Notable examples include pancratistatin and lycoricidine, whose syntheses rely on conduramine or aminoconduritol cores. These natural products demonstrate a range of biological properties, including protein synthesis inhibition and cytotoxicity toward lymphocytic leukemia and ovarian sarcoma cell lines, positioning them as valuable leads in anticancer drug discovery and motivating continued investigation into cyclitol-based bioactive scaffolds.10–14
Progress in synthetic methodology over the past several decades has greatly expanded access to conduritol derivatives.1,2,6,15,16 Classical transformations such as aromatic dihydroxylation have been complemented by advances in chemoenzymatic, enantioselective, and microbial oxidation strategies. Particularly noteworthy is the use of engineered microorganisms such as Pseudomonas putida for highly selective cis-dihydroxylation of aromatic substrates, enabling efficient construction of key intermediates for conduritol frameworks. These developments have facilitated the synthesis not only of the parent compounds but also of complex derivatives including glycomimetics, oligosaccharide analogues, and various unnatural sugars with therapeutic potential.
Our research group recently provided a comprehensive review of the synthetic approaches to all six stereoisomeric conduritols (A–F).6 Building upon that foundation, the present review focuses specifically on conduritol derivatives, which constitute an equally important domain owing to their biological functions and synthetic utility. The literature discussed in this review was selected through a comprehensive survey of the major scientific databases and journals to provide a representative overview of synthetic strategies reported for conduritol derivatives. In contrast to our previous review, which primarily addressed the preparation of the parent conduritol stereoisomers, the present article emphasizes the rapidly expanding chemistry of functionalized conduritol derivatives, including aminated, epoxidized, halogenated, phosphorylated, and structurally diversified analogues. By systematically compiling and critically comparing these synthetic approaches, this review aims to provide the broader research community with a consolidated platform for understanding the evolution of conduritol chemistry, identifying existing methodological limitations, and guiding the development of more efficient and sustainable strategies for the synthesis and application of these biologically significant cyclitol frameworks.
2. Synthetic landscape of conduritol derivatives
In recent years, continued advances in catalytic transformations, metathesis chemistry, and chemoenzymatic methodologies have further expanded the toolbox available for constructing highly functionalized conduritol derivatives.The synthesis of conduritol derivatives has evolved through a wide variety of strategic approaches, ranging from chemoenzymatic arene dihydroxylation and carbohydrate-derived routes to modern catalytic and metathesis-based methodologies. These strategies differ not only in the origin of stereochemical information but also in the way the polyhydroxylated cyclohexene framework is constructed and functionalized. In the following sections, representative synthetic routes are discussed chronologically, highlighting key transformations, stereochemical considerations, and methodological innovations that have shaped the development of conduritol chemistry.
Over more than a century of investigation, conduritols and their nitrogen-containing congeners have evolved from rare natural curiosities into versatile molecular platforms central to modern synthetic, medicinal, and biological chemistry. Their densely functionalized cyclohexene core, rich stereochemical diversity, and close resemblance to carbohydrates have inspired an exceptional range of synthetic strategies. Across the literature, researchers have developed approaches that span classical oxidation–reduction sequences, stereocontrolled epoxidation and dihydroxylation protocols, ring-closing metathesis, Ramberg–Bäcklund transformations, and carbohydrate-derived routes. Equally transformative has been the emergence of chemoenzymatic methods, particularly selective microbial cis-dihydroxylation, which provides rapid access to key building blocks with high regio- and stereocontrol. Collectively, these methods illustrate the ingenuity and breadth of synthetic design applied to the construction of conduritols, conduramines, and inositol derivatives. The following section systematically reviews these advances, highlighting strategic innovations, stereochemical logic, and the evolving toolkit that continues to shape the synthesis of this important family of cyclitols.
2.1. Synthesis and advances in conduritol B epoxide
Conduritol B epoxide (CBE) is a well-established irreversible inhibitor of several D-glucosidases, displaying particularly high potency toward the mammalian β-glucosidase responsible for the hydrolysis of glucosylceramide. Impaired function of this enzyme underlies Gaucher disease, a major lysosomal storage disorder. Owing to its ease of synthetic access and reliable inhibitory profile, CBE has become an indispensable biochemical probe for elucidating the molecular basis of this genetic condition using diverse animal models.The first description of CBE was reported by Legler and co-workers in 1966, who demonstrated its capacity to inactivate both plant-derived glucosidases and mammalian β-glucosidases.17 A decade later, in 1977, the same group proposed a synthetic route to CBE (8) from myo-inositol (1); however, this method suffered from modest yields and required large amounts of starting material, limiting its practical utility.18
To overcome these limitations, Lee et al. (1985) developed a significantly improved and operationally convenient protocol.19 Their synthesis began with the reflux of myo-inositol (1) in cyclohexane–dimethyl sulfoxide (DMSO), catalyzed by p-toluenesulfonic acid monohydrate at 110 °C for 6 h, yielding 1,2-O-cyclohexylidene-myo-inositol (2) in ∼85% yield. Subsequent acetylation with acetic anhydride and pyridine afforded 3,4,5,6-tetra-O-acetyl-1,2-O-cyclohexylidene-myo-inositol (3) in approximately 90% yield.
Removal of the cyclohexylidene protecting group was achieved by heating compound (3) in a 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of acetic acid and water in the presence of 0.1 M HCl at 100 °C, furnishing 1,4,5,6-tetra-O-acetyl-myo-inositol (4) in ∼97% yield. Conversion of compound (4) to its corresponding thiocarbonate (5) was carried out using N,N′-thiocarbonyldiimidazole under nitrogen, also providing a ∼97% yield.
1 mixture of acetic acid and water in the presence of 0.1 M HCl at 100 °C, furnishing 1,4,5,6-tetra-O-acetyl-myo-inositol (4) in ∼97% yield. Conversion of compound (4) to its corresponding thiocarbonate (5) was carried out using N,N′-thiocarbonyldiimidazole under nitrogen, also providing a ∼97% yield.
Refluxing intermediate (5) with trimethyl phosphite for 5 h under nitrogen produced conduritol B tetraacetate (6) in ∼75% yield. Mechanistically, this transformation proceeds through a phosphite-mediated elimination of the thiocarbonate functionality, enabling efficient generation of the cyclohexene framework that ultimately serves as the precursor for epoxide formation. Subsequent deacetylation of compound (6) with triethylamine in a 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 methanol–water mixture afforded conduritol B (7) in nearly quantitative yield. Final epoxidation of compound (7) with meta-chloroperbenzoic acid (m-CPBA) in methanol for 12 h, followed by crystallization from absolute ethanol, yielded conduritol B epoxide (CBE) (8) in ∼90% overall yield (Scheme 1). The use of m-CPBA as an oxidizing agent ensures efficient epoxide formation with high selectivity; however, the reliance on peracid oxidants and multiple protection–deprotection steps may limit the overall step economy and raise practical considerations related to reagent cost and waste generation when larger-scale synthesis is contemplated.
3 methanol–water mixture afforded conduritol B (7) in nearly quantitative yield. Final epoxidation of compound (7) with meta-chloroperbenzoic acid (m-CPBA) in methanol for 12 h, followed by crystallization from absolute ethanol, yielded conduritol B epoxide (CBE) (8) in ∼90% overall yield (Scheme 1). The use of m-CPBA as an oxidizing agent ensures efficient epoxide formation with high selectivity; however, the reliance on peracid oxidants and multiple protection–deprotection steps may limit the overall step economy and raise practical considerations related to reagent cost and waste generation when larger-scale synthesis is contemplated.
Conduritol B epoxide (CBE, 8) is not only a potent irreversible inhibitor of β-glucosidase but also an indispensable molecular probe for elucidating the biochemical basis of Gaucher's disease. To study its biological activity, metabolic fate, and mechanism of enzyme inactivation, researchers frequently rely on radiolabelled analogues of CBE. In 1977, Legler et al. reported the first synthetic protocol for obtaining radiolabelled CBE using either [14C]- or [3H]-myo-inositol as the precursor.18 However, the method suffered from low overall yields and the formation of isomeric mixtures, complicating purification and stereochemical characterization.
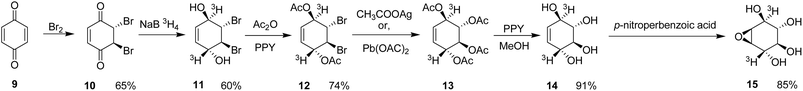
To address these shortcomings, Gal et al. (1987) developed a significantly improved route for the preparation of [3H]-labelled CBE, providing enhanced yields and superior stereochemical fidelity.20 Their synthesis commenced with the bromination of p-benzoquinone (9), producing trans-5,6-dibromo-2-cyclohexene-1,4-dione (10) in ∼65% yield. Reduction of compound (10) with sodium boro[3H]hydride furnished the tritiated diol (1α,2β,3α,4β)-(±)-2,3-dibromo-[1,4-3H]-dihydroxy-5-cyclohexene (11) in approximately 60% yield.
Acetylation of intermediate (11) with acetic anhydride in the presence of 4-pyrrolidinopyridine afforded the corresponding diacetate (12) in ∼74% yield. Oxidative debromination of compound (12) using either silver acetate or lead(II) acetate produced [3H]-labelled conduritol B tetraacetate (13) in yields of ∼68% and ∼54%, respectively. Subsequent deacetylation of compound (13) gave [3H]-conduritol B (14) in ∼91% yield.
Final epoxidation of compound (14) with p-nitroperbenzoic acid afforded [3H]-conduritol B epoxide (15) in an overall yield of ∼85% (Scheme 2). This improved synthetic protocol enabled the efficient and stereoselective preparation of radiolabelled CBE, greatly facilitating detailed mechanistic investigations into β-glucosidase inhibition and the pathophysiology of Gaucher's disease.
While conduritol B epoxide represents one of the earliest and most extensively studied functional derivatives of the conduritol framework, its chemistry also highlighted the broader synthetic potential of cyclitol scaffolds. In particular, the structural modification of conduritols through the introduction of nitrogen functionality led to the development of conduramine derivatives, which significantly expanded the biological and synthetic scope of this class of compounds.
2.2. Construction of conduramine frameworks: structural diversity and foundational synthetic methods
Aminocyclitols represent an important class of structurally diverse compounds that appear in numerous biologically active molecules. They form the core of aminoglycoside–aminocyclitol (AGAC) antibiotics, constitute key structural elements of several Amaryllidaceae alkaloids, and also function as ligands in cytostatic platinum-based complexes. Within this broader family, conduramines, which are aminated cyclohexenetriols, hold particular significance. Structurally, they are derived from the conduritol framework by replacing one hydroxyl group with an amino substituent.Conduramines exhibit potent glycosidase inhibitory activity and have therefore attracted substantial interest as both pharmacologically active agents and versatile synthetic intermediates. Owing to their well-defined stereochemistry, these aminocyclitols serve as valuable chiral building blocks in the synthesis of several biologically important natural products, including (+)-narciclasine (16), (+)-valienamine (17), and (+)-lycoricidine (18) (Fig. 1). Their synthetic utility extends further to the preparation of amino cyclitols that constitute the aglycone portion of numerous clinically relevant aminoglycoside antibiotics.
Beyond these applications, conduramines are integral to the construction of a wide range of therapeutically relevant molecular architectures such as aminosugars, sphingosines, azasugars, and various Narcissus alkaloids. Depending on the position of the amino functionality on the cyclohexene ring, conduramines may be categorized into several structural subclasses, each offering distinct reactivity patterns and synthetic potential.
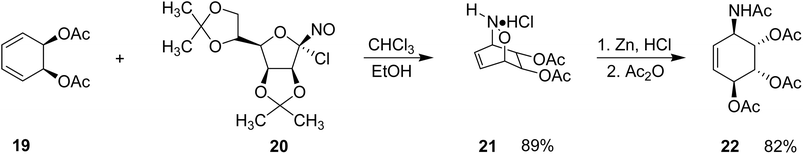
In 1990, Werbitzky et al. reported a concise and efficient strategy for the synthesis of conduramine A tetraacetate based on a hetero-Diels–Alder reaction.21 The Diels–Alder reaction and its hetero variants represent powerful cycloaddition methods for constructing six-membered frameworks and have also found applications in modern materials science through reversible Diels–Alder linkages.22 The sequence began with the cycloaddition between diacetoxydiene (19) and protected 1-chloro-1-nitrosomannose (20) in a chloroform–ethanol solvent mixture. This [4 + 2] hetero-cycloaddition proceeded smoothly to afford the corresponding dihydrooxazine adduct (21) in ∼89% yield. Subsequent reductive cleavage of the N–O bond in compound (21) using zinc in hydrochloric acid, followed by acetylation with acetic anhydride, provided conduramine A tetraacetate (22) in 82% yield (Scheme 3). This approach illustrates the effectiveness of hetero-Diels–Alder chemistry for constructing aminocyclitol frameworks with excellent regio- and stereocontrol.
Mechanistically, the hetero-Diels–Alder cycloaddition enables simultaneous formation of the cyclohexene framework and installation of the nitrogen functionality, thereby streamlining the construction of the conduramine scaffold. Nevertheless, the requirement for specifically functionalized nitroso precursors and subsequent reductive N–O bond cleavage steps may limit the broader practicality of this strategy in comparison with more modular nitrogen-installation methods developed in later studies.
The development of efficient synthetic routes to conduramine derivatives also stimulated broader interest in structurally related cyclitol frameworks that display important biological and pharmacological properties. Among these, naturally occurring inositol derivatives such as pinitol have attracted considerable attention, prompting the exploration of enantioselective synthetic strategies capable of accessing these molecules with high stereochemical fidelity.
2.3. Enantiodivergent synthetic approaches to pinitol
D-Pinitol (3-O-methyl-D-chiro-inositol) (23) is a naturally occurring cyclitol widely distributed in plant species belonging to the Leguminosae and Pinaceae families. Functionally, it serves as an essential cellular osmoprotectant and defensive metabolite, enabling plants to tolerate abiotic stresses such as drought, salinity, and oxidative damage. Through its involvement in osmotic adjustment and stress-responsive metabolic pathways, D-pinitol plays a critical role in maintaining cellular homeostasis under adverse environmental conditions.The name “D-pinitol” originates from its initial isolation from the heartwood of Pinus monticola. Later investigations established its presence in multiple plant tissues, including resin, wood, bark, and cambial sap, across many gymnosperms, particularly species of Pinus and Abies. Its distribution is not confined to conifers; the compound has also been identified in several angiosperm families, including Fabaceae, Nyctaginaceae, Asteraceae, Zygophyllaceae, Caryophyllaceae, Aristolochiaceae, Sapindaceae, Santalaceae, and Aizoaceae.
Traditionally, plants enriched in D-pinitol have been employed in the treatment of diabetes, inflammation, cancer, and microbial infections, consistent with the broad biological activities attributed to this cyclitol. In recent years, interest in D-pinitol has grown markedly due to its abundance in nutritionally and pharmacologically important plant sources such as soybean (Glycine max), carob pod (Ceratonia siliqua), ice plant (Mesembryanthemum crystallinum), fenugreek seed (Trigonella foenum-graecum), and several Retama species.
Collectively, these findings underscore the significance of D-pinitol as a bioactive phytochemical with promising applications in nutraceuticals, phytopharmaceuticals, and agro-biotechnology (Fig. 2).
In 1990, Hudlicky et al. reported a seminal enantiodivergent synthetic strategy that enabled the preparation of both enantiomers of pinitol, (+)-pinitol (+)-23 and (−)-pinitol (−)-23 from a common chiral intermediate.23 The sequence began with bromo-acetonide (24), which was subjected to osmium tetroxide/N-methylmorpholine N-oxide (OsO4/NMO)-mediated dihydroxylation to furnish the conduritol E derivative (25) in approximately 85% yield.
Reduction of compound (25) with lithium aluminium hydride (LiAlH4) afforded the corresponding olefin (ca. 85% yield), which underwent epoxidation with m-chloroperbenzoic acid (m-CPBA) to deliver epoxide (26) in 86% yield. Subsequent methanolysis followed by acetonide deprotection furnished (+)-pinitol (+)-23 in an overall yield of ∼90%. The regio- and stereochemical outcome of these transformations is governed by the conformational preference of the conduritol intermediate, which directs nucleophilic attack and oxidative addition from the less sterically hindered face of the cyclohexene ring.
In the complementary sequence leading to the opposite enantiomer, bromo-acetonide (24) was first epoxidized directly with m-CPBA in dichloromethane to produce epoxide (27) in 80% yield. Methanolysis of epoxide (27) (89%) followed by dehalogenation (85%) yielded the conduritol F derivative (28). Dihydroxylation of compound (28) using OsO4/NMO (63%) and subsequent deprotection provided (−)-pinitol (−)-23. Notably, substituting the bromo group in the acetonide precursor with chlorine markedly slowed the dehalogenation step, underscoring the importance of the bromine substituent in controlling reaction efficiency.
The OsO4/NMO system plays a crucial role in establishing the syn-dihydroxylated stereochemical motif characteristic of many cyclitol frameworks. However, the use of osmium-based oxidants presents practical limitations due to their toxicity, high cost, and environmental concerns, which has motivated ongoing efforts to develop milder and more sustainable dihydroxylation methodologies.
This six-step route, originating from a diene diol generated via microbial oxidation of bromobenzene, enabled the enantiospecific synthesis of both pinitol enantiomers with excellent stereocontrol and regiodivergence. A key feature of this strategy was the use of a highly versatile chiral synthon that allowed complete diastereofacial control (α- versus β-attack) and regioselective oxidative transformations in subsequent steps (Scheme 4).
 | ||
| Scheme 4 Enantiospecific synthesis of both pinitol enantiomers (23) using the common bromo acetonide intermediate (24). | ||
The successful application of such stereocontrolled transformations in the synthesis of cyclitol derivatives also highlighted the broader synthetic versatility of conduritol-type intermediates. In particular, similar strategies involving selective protection, oxidation, and dihydroxylation reactions have been employed in the construction of highly functionalized inositol derivatives, including biologically significant myo-inositol phosphates.
2.4. Foundational synthetic routes to myo-inositol polyphosphates
Inositol phosphates constitute a structurally diverse family of naturally occurring organophosphorus compounds. Despite their widespread presence in terrestrial and aquatic environments, they remain among the least understood components of the global phosphorus cycle. These molecules, containing up to six phosphate groups esterified to an inositol scaffold, occur in several stereoisomeric forms, including myo, D-chiro, scyllo, and neo with myo-inositol hexakisphosphate (phytate) being the most abundant, particularly in plant-derived materials.Inositol phosphates are primarily of plant origin and accumulate in soils as the dominant class of organic phosphorus species. They are also detected in notable concentrations in aquatic systems, where their partial bioavailability may influence eutrophication processes. Their environmental behavior, such as cycling, mobility, enzymatic turnover, and biological accessibility, is still poorly understood because isolating, separating, and detecting highly phosphorylated species from complex matrices remains analytically challenging.
Beyond their environmental significance, myo-inositol phosphates play pivotal roles in biological systems. Several members of this family act as indispensable secondary messengers, particularly in calcium signaling pathways, where they regulate intracellular calcium release and modulate key physiological processes.
A landmark synthetic contribution to this field was reported by Carless et al. in 1990, who developed a versatile route to biologically relevant myo-inositol phosphates.24 The synthesis originated from MEM-protected trans-benzene diol (29), derived from benzene over seven steps in an overall yield of ∼35%. Compound (29) underwent a [4 + 2] cycloaddition with singlet oxygen, followed by thiourea-mediated reduction to afford diol (30). Mechanistically, this pericyclic oxygenation step provides a rapid entry into oxygen-rich cyclohexane frameworks, demonstrating how pericyclic reactions can efficiently generate the stereochemically dense intermediates required for complex cyclitol synthesis. Oxidation of the latter using 1.5 equivalents of PCC generated a mixture of hydroxyenones (31) and (32), isolated in 50% and 20% yield, respectively.
Luche reduction of compound (31) provided the conduritol B derivative (33) in 90% yield. Alternatively, compound (33) could be accessed in ∼70% yield by oxidation of diol (30) with 3 equivalents of PCC, followed by reduction of the resulting enedione. Dibenzylation of compound (33) with NaH and benzyl bromide furnished the corresponding protected intermediate in 95% yield. Subsequent dihydroxylation using osmium tetroxide/N-methylmorpholine N-oxide (OsO4/NMO) afforded cyclitol (34) in 96% yield, serving as a key branch point for the synthesis of multiple myo-inositol phosphate isomers. The OsO4/NMO-mediated dihydroxylation plays a decisive role in establishing the syn-vicinal diol configuration required for inositol phosphate synthesis; however, the reliance on osmium-based oxidants raises practical concerns related to reagent toxicity, cost, and environmental impact, factors that have motivated the search for greener oxidative methodologies in later studies.
From cyclitol (34), regioselective introduction of a third MEM protecting group yielded compounds (35) and (36) in 60% and 10% yield, respectively. Compound (35) underwent benzylation (86% yield), MEM deprotection with 6 N HCl in THF at 20 °C (59% yield), and phosphorylation using tetrabenzyl pyrophosphate (TBPP) and NaH (47% yield). Global hydrogenolysis furnished myo-inositol 1,4,5-triphosphate (37).
Similarly, myo-inositol 2,4,5-triphosphate (38) was obtained from compound (36) through benzylation (78%), MEM deprotection (82%), phosphorylation (53%), and final hydrogenolysis (85%).
Further elaboration of compound (34) via phosphorylation produced tetrol (39), which upon hydrogenolysis furnished racemic myo-inositol 1,2,4,5-tetrakisphosphate (40). In another sequence, benzylation of compound (34) followed by MEM deprotection generated compound (41), which upon phosphorylation with TBPP/BuLi in THF and subsequent debenzylation afforded racemic myo-inositol 4,5-bisphosphate (42) (Scheme 5).
The development of such versatile cyclitol intermediates not only facilitated the synthesis of complex inositol phosphates but also underscored the broader synthetic potential of carbohydrate-derived precursors for constructing conduritol frameworks. Building on these concepts, subsequent studies explored stereocontrolled strategies that utilized naturally occurring sugars as chiral starting materials to access structurally diverse conduritol derivatives.
2.5. Stereodivergent synthesis of tetra-O-methylconduritol derivatives of conduritol A and C from L-arabinose
In 1991, McIntosh et al. reported a stereospecific and stereodivergent synthetic route to the tetra-O-methyl derivatives of conduritol A and conduritol C, starting from the naturally occurring pentose L-arabinose. The synthesis began with the conversion of L-arabinose (43) into the corresponding dithioacetal trimethyl ether (44) through a three-step sequence.25Selective protection of the primary hydroxyl group in compound (44) was achieved using TBDMSCl, imidazole, and DAMP in DMF, affording the silyl ether (45) in 86% yield. Subsequent deprotection of the thioacetal moiety with HgO/HgCl2 in an acetone–water mixture furnished aldehyde (46) in 98% yield.
Aldehyde (46) underwent Wittig olefination to yield dibromide (47) in 81% yield, which was then transformed into silylacetylene (48) in 73% yield following the Corey–Fuchs protocol. Hydrogenation of compound (48) using Pd/BaSO4 under an atmosphere of H2 in pyridine provided vinylsilane (49) in 91% yield. Hydrolytic removal of the silyl group with aqueous acetic acid afforded alcohol (50) in 80% yield, and oxidation of this primary alcohol produced vinylsilane aldehyde (51) with an excellent yield of 98%.
A key feature of this strategy was the stereochemical control achieved during intramolecular cyclization of aldehyde (51), governed by the choice of Lewis acid. In this context, the contrasting use of BF3·OEt2 and SnCl4 highlights the decisive role of Lewis acid coordination in controlling the transition-state geometry of the cyclization process, thereby directing the formation of either the anti or syn cyclohexenol framework. However, the requirement for strong Lewis acids and carefully controlled reaction temperatures may impose practical limitations when considering the scalability and operational simplicity of such transformations. Treatment with BF3·OEt2 in dichloromethane at ambient temperature led to the formation of 1,2-anti-cyclohexenol (52) in 86% yield and with >30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 isomeric purity. Conversely, treatment with SnCl4 at −78 °C followed by gradual warming to room temperature afforded the 1,2-syn-cyclohexenol (54), also in 86% yield and >30
1 isomeric purity. Conversely, treatment with SnCl4 at −78 °C followed by gradual warming to room temperature afforded the 1,2-syn-cyclohexenol (54), also in 86% yield and >30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 isomeric ratio.
1 isomeric ratio.
Final methylation of the hydroxyl groups using Ag2O and methyl iodide completed the divergent sequence: the 1,2-anti intermediate (52) furnished tetra-O-methylconduritol A (53) in 71% yield, while the 1,2-syn intermediate (54) delivered tetra-O-methylconduritol C (55) in 91% yield (Scheme 6). This methodology elegantly demonstrates how Lewis acid-controlled cyclizations can direct stereochemical outcomes, enabling efficient access to distinct conduritol isomers from a common chiral precursor.
 | ||
| Scheme 6 Synthesis of tetra-O-methylconduritol A (53) and tetra-O-methylconduritol C (55) from L-arabinose (43). | ||
The ability to control stereochemical outcomes through strategic choice of reagents and reaction conditions also proved valuable in the synthesis of nitrogen-containing cyclitol derivatives. Building on the principles of stereoselective cyclization and functional group manipulation illustrated above, subsequent studies explored routes to aminocyclitol frameworks, which are of considerable importance in antibiotic development and medicinal chemistry.
2.6. Stereospecific synthesis of cis-1,3-diaminocyclitols
The selective synthesis of aminocyclitol isomers with retention of amino group configuration is of considerable interest, particularly due to their relevance in the mutasynthesis of new aminoglycoside antibiotics and their utility as ligands in cytostatic platinum(II) complexes. In 1991, Schürrle et al. developed an elegant and stereospecific synthetic approach to cis-1,3-diamino-1,3-dideoxycyclitols, employing a conduramine A-1 derivative as the central intermediate.26The key transformation in their strategy was a hetero-Diels–Alder reaction between compound (56), prepared from cis-cyclohexa-3,5-diene-1,2-diol, and (−)-2,3:5,6-di-O-isopropylidene-1-nitroso-α-D-manno-furanosyl chloride (−)-57. This cycloaddition furnished the oxazine (+)-58 in 82% yield. Reductive cleavage of (+)-58 with aluminum amalgam in aqueous THF then produced the conduramine A-1 derivative (−)-59 in 94% yield.
Reaction of compound (−)-59 with N-ethoxycarbonylphthalimide (60) and sodium carbonate afforded the protected amino derivative (−)-61 in 90% yield. Hydrolysis of (−)-61 subsequently delivered the triol (−)-62 in 92% yield. Protection of the three hydroxyl groups as isopropyldimethylsilyl (IPDMS) ethers using chloro(isopropyl)dimethylsilane and imidazole afforded compound (−)-63 in 98% yield.
Epoxidation of (−)-63 with p-nitroperbenzoic acid, followed by desilylation with acetic acid, generated epoxy triol (−)-64 in 87% yield. Subsequent treatment with trichloroacetonitrile and DBU furnished the trichloroacetimidate (−)-65 in 94% yield. A crucial stereoselective intramolecular epoxide opening mediated by triethylaluminum led to exclusive formation of the 2-trichloromethyl-4,5-dihydro-1,3-oxazole (−)-66 in 80% yield. The use of triethylaluminum as a Lewis acidic promoter is central to this transformation, facilitating controlled intramolecular epoxide opening through coordination to the oxygen atom and directing nucleophilic attack along a well-defined transition-state pathway. Nevertheless, the requirement for highly moisture-sensitive organoaluminum reagents and multistep protection–deprotection sequences may pose practical challenges when considering the operational robustness and scalability of this methodology.
Cleavage of the oxazole ring in (−)-66 using HCl followed by treatment with anhydrous hydrazine afforded (−)-1L-2,4-diamino-2,4-dideoxy-chiro-inositol (−)-67 in 79% yield. The opposite enantiomer, (+)-67, was obtained analogously starting from (+)-57.
In a complementary sequence, compound (−)-61 was converted into its trichloroacetamidate derivative (−)-68 in 91% yield using trichloroacetonitrile in dichloromethane. Subsequent iodination with N-iodosuccinimide (NIS) afforded a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of (−)-69 (66%, major) and (−)-70 (21%). Further iodination of (−)-69 with NIS produced the diiodide (−)-71 in 95% yield.
1 mixture of (−)-69 (66%, major) and (−)-70 (21%). Further iodination of (−)-69 with NIS produced the diiodide (−)-71 in 95% yield.
Radical dehalogenation of compound (−)-71 provided (+)-72 in 92% yield. Final global deprotection of (+)-72 with hydrazine in a chloroform–ethanol mixture delivered the target aminocyclitol, (−)-1L-2,4-diamino-2,3,4-trideoxy-allo-inositol (−)-73, in 73% yield. The enantiomeric form, (+)-73, was obtained similarly using the enantiomer (+)-57 (Scheme 7).
 | ||
| Scheme 7 Synthesis of 2,4-diamino-2,4-dideoxy-chiro-inositol (67) and 2,4-diamino-2,3,4-trideoxy-allo-inositol (73). | ||
While the strategy described above highlights the power of hetero-Diels–Alder reactions and intramolecular epoxide openings for constructing aminocyclitol frameworks with precise stereochemical control, alternative approaches have also been explored to access conduritol and conduramine derivatives more directly from simple aromatic precursors. In particular, microbial arene oxidation has emerged as a highly effective strategy for generating cis-dihydroxylated intermediates that can serve as versatile building blocks for diverse cyclitol syntheses.
2.7. Synthesis of deoxyconduritol E and conduramine A-1 derivatives
In 1991, Hudlicky et al. reported a versatile and stereoselective synthetic strategy enabling access to a broad array of natural and synthetic conduritols.27 The central feature of this unified approach was the microbial oxidation of chloro- or bromobenzene to generate the corresponding cis-cyclohexadienediols. These bio-oxidized intermediates, with their well-defined symmetry and inherent stereochemical bias, provided an ideal platform for introducing functional groups with excellent stereocontrol. The microbial arene dihydroxylation step is particularly valuable because it introduces vicinal cis-diol functionality with high enantioselectivity under mild conditions; however, practical implementation may depend on access to specialized microbial strains and biocatalytic facilities, which can limit the widespread application of this approach in laboratories lacking biotransformation infrastructure.For the synthesis of deoxyconduritol E, bromocyclohexadienediol (74), obtained from bromobenzene oxidation, was first transformed into its acetonide-protected derivative. Epoxidation of this intermediate with m-CPBA in dichloromethane furnished epoxide (27). Similarly, chloro-cyclohexadienediol (77) was converted into the corresponding epoxide (78) following acetonide protection and m-CPBA epoxidation.
Reduction of epoxides (27) and (78) with LiAlH4 in ether afforded alcohols (75) and (79) in ∼99% and 80% yields, respectively. Radical dehalogenation of these alcohols produced a common intermediate (76) in 80% yield. Subsequent acidic deprotection of compound (76) delivered Deoxyconduritol E (80) in quantitative yield.
In a complementary sequence, Hudlicky and co-workers also developed an efficient route to conduramine A-1 tetraacetate, dihydroconduramine A-1, and its tetraacetate derivative.1,27 The route began with a hetero-Diels–Alder reaction between bromocyclohexadienediol acetonide (24) and an in situ-generated nitrosyl dienophile derived from acetohydroxamic acid. This cycloaddition produced the oxazine (81) as a single enantiomer, installing four contiguous stereocenters in a single step.
Reductive cleavage of the N–O bond in oxazine (81) using aluminum amalgam furnished the amino alcohol (82) in 77% yield. Acidic deprotection of (82) then yielded triol (83) in ∼99% yield. Acetylation of triol (83) afforded conduramine A-1 tetraacetate (22) in 63% yield. Moreover, palladium-catalyzed hydrogenation of triol (83) provided dihydroconduramine A-1 (84) in 99% yield, which upon acetylation in a THF–water mixture furnished dihydroconduramine A-1 tetraacetate (85) in 97% yield (Scheme 8).
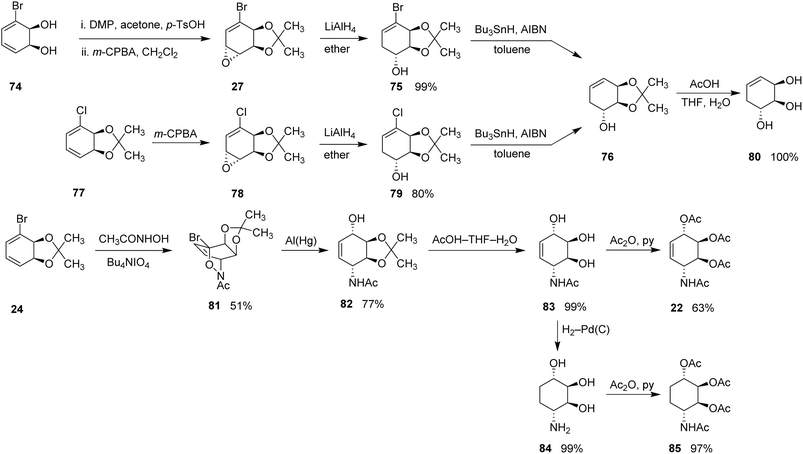 | ||
| Scheme 8 Synthesis of deoxyconduritol E (80), conduramine A-1 tetraacetate (22) tetraacetate along with dihydroconduramine A-1 (85). | ||
The successful utilization of microbial arene dihydroxylation in these transformations highlighted the broader potential of biocatalytic oxidation for constructing highly functionalized cyclitol frameworks from simple aromatic precursors. Building upon this concept, subsequent studies expanded the scope of such chemoenzymatic strategies to access structurally modified inositol derivatives, including C-methyl-substituted cyclitols of biological significance.
2.8. Chemoenzymatic synthesis of C-methyl inositols: enantioselective access to (−)-laminitol and C-methyl muco-inositol
The identification of D-myo-inositol-1,4,5-trisphosphate (IP3) as a pivotal second messenger that mediates intracellular calcium ion release highlighted the need for efficient synthetic routes to inositol phosphates and related analogues. These compounds became invaluable tools in probing signal transduction pathways. While several synthetic methods for myo-inositol phosphates had been developed, they often relied on naturally abundant myo-inositol as the starting material. Alternative strategies frequently employed benzene or cyclohexa-1,2-diene-1,2-diols (in cis or trans forms) to access the desired inositol phosphate analogues.Although the biological functions of other inositol stereoisomers remained comparatively less explored, two naturally occurring C-methyl inositols, (−)-laminitol and mytilitol (a symmetrical derivative of scyllo-inositol) were isolated from marine algae. Of these, (−)-laminitol, featuring myo-inositol stereochemistry and a methyl group at C-4, demonstrated biological activity by inhibiting the growth of Neurospora crassa. While a racemic synthesis of laminitol from myo-inositol had been reported earlier, Carless et al. (1991) provided the first enantioselective total synthesis of (−)-laminitol (−)-93 and C-methyl muco-inositol (94), uniquely starting from toluene (86).28
Their synthetic route began with microbial oxidation of toluene using Pseudomonas putida, affording cis-cyclohexadienediol (87). Protection of the diol with 2,2-dimethoxypropane yielded the corresponding isopropylidene derivative (88) in ∼65% yield. Epoxidation of compound (88) afforded epoxide (89), which was converted into the corresponding trans-diol (90) via acid-mediated ring opening. Subsequent deprotection of the isopropylidene group led to C-methyl conduritol F (91) in ∼78% yield.
Hydroxyl-directed epoxidation of the remaining double bond in compound (91) yielded epoxide (92) in ∼73% yield. The final step, an acid-catalyzed epoxide ring opening produced a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of (−)-laminitol (−)-93 and C-methyl muco-inositol (94), with an excellent combined yield of ∼95%.
1 mixture of (−)-laminitol (−)-93 and C-methyl muco-inositol (94), with an excellent combined yield of ∼95%.
Additionally, Carless et al. described an alternative synthetic route to C-methyl muco-inositol (94). This involved the reaction of cis-cyclohexadienediol (87) with singlet oxygen to form endoperoxide (95). Reductive cleavage of the peroxide bond using thiourea yielded C-methyl conduritol A (96). Subsequent dihydroxylation of compound (96) with osmium tetroxide afforded C-methyl muco-inositol (94) in ∼55% isolated yield (Scheme 9). The use of osmium tetroxide enables highly stereoselective syn-dihydroxylation of the cyclohexene framework; however, similar to other osmium-mediated oxidations discussed earlier, the toxicity and high cost of this reagent remain important considerations when evaluating the scalability and environmental sustainability of such methodologies.
 | ||
| Scheme 9 Chemoenzymatic synthesis of (−)-laminitol (−)-93 and C-methyl muco-inositol (94) from toluene (86). | ||
The versatility of microbial arene dihydroxylation in generating highly functionalized cyclitol intermediates also inspired its application in the synthesis of structurally complex natural products. In particular, the stereochemically rich cyclohexadienediol intermediates obtained from aromatic biooxidation proved especially valuable for constructing the core frameworks of several Amaryllidaceae alkaloids, including lycoricidine and related compounds.
2.9. Chemoenzymatic synthetis of (+)-lycoricidine from chlorobenzene
The Narcissus alkaloids pancratistatin, narciclasine, and lycoricidine belong to a prominent class of bioactive natural products isolated from members of the Amaryllidaceae family. These compounds exhibit remarkable cytotoxic and antitumor activities, with pancratistatin emerging as the most promising therapeutic candidate since the initial structural elucidations reported in the late 1960s. However, the extremely low natural abundance of these alkaloids, coupled with the complexity of their extraction and purification from plant sources, severely limits their availability. These challenges highlight the need for synthetic approaches that are synthetically efficient, cost-effective, environmentally sustainable, and stereochemically well-controlled, ideally providing scalable access to all members of this alkaloid family.In 1992, Hudlicky et al. disclosed an enantioselective total synthesis of (+)-lycoricidine that leveraged a biooxidative strategy to establish the core stereochemical framework.29 The synthesis began with microbial dihydroxylation of chlorobenzene (97) to afford cis-cyclohexadienediol (98), which was subsequently protected as its acetonide derivative (77). A hetero-Diels–Alder cycloaddition between acetonide (77) and dienophiles (99) or (100), promoted by Bu4NiO4 in dichloromethane, furnished the corresponding oxazines (101a) and (101b) in 74% and 80% yield, respectively.
Reductive cleavage of the N–O bond in these oxazines using aluminum amalgam generated the protected conduramine D derivatives (102a) and (102b). Compound (102a) underwent silylation of the secondary hydroxyl group in 98% yield, followed by lithiation with n-BuLi in THF and subsequent acylation with 2-bromopiperonyloyl chloride (103) to deliver amide (104) in 77% yield. Alternatively, amide (104) could be obtained from compound (102b) via silylation followed by CBZ protection and analogous acylation.
The pivotal macrocyclization of amide (104) was accomplished through a modified Heck reaction employing Pd(OAc)2, Tl(OAc), and 1,2-bis(diphenylphosphino)ethane (DPPE) in anisole, furnishing the cyclized product (105) in 27% yield. Removal of the CBZ protecting group delivered amide (106) in nearly quantitative yield. Final acid-mediated removal of the acetonide and silyl protecting groups using trifluoroacetic acid (TFA) completed the synthesis, yielding (+)-lycoricidine (+)-18 in 85% yield (Scheme 10).
Chemoenzymatic arene cis-dihydroxylation strategies provide a powerful entry into densely functionalized cyclitol frameworks because they introduce multiple stereocentres in a single step with excellent stereochemical control. Nevertheless, their broader application can sometimes be limited by substrate scope, the need for specialized microbial systems, and challenges associated with scaling up biotransformations for large-scale synthetic applications.
Beyond their application in the total synthesis of complex natural products such as lycoricidine, these chemoenzymatically generated cyclitol intermediates also provide versatile platforms for the preparation of structurally diverse conduramine derivatives. Consequently, several studies have explored strategies that combine microbial oxidation with enzymatic resolution and stereospecific functionalization to access enantiomerically pure aminocyclitols with defined stereochemical configurations.
2.10. Stereoselective construction of conduramine C-1 and related conduramine A derivatives
Similar to the parent conduritols, conduramines display potent glycosidase inhibitory activity and therefore serve as valuable scaffolds for the design of enzyme inhibitors. In 1992, Johnson et al. reported an enantioselective synthesis of both enantiomers of conduramine C-1 (114), employing a strategy that combined microbial oxidation, enzymatic resolution, and Mitsunobu-type reactions.30The synthesis commenced with meso-cyclohexa-3,5-dien-1,2-diol (108), generated from benzene (107) via microbial dihydroxylation. Protection of this diol afforded the corresponding conduritol A derivative (109). Selective enzymatic acetylation using Pseudomonas cepacia lipase and isopropenyl acetate provided monoacetate (110). After hydroxyl protection and subsequent deacetylation, compound (111) was isolated in ∼98% yield.
Exposure of compound (111) to Mitsunobu conditions in the presence of phthalimide furnished protected conduramine C-1 (112) in 67% yield. The Mitsunobu reaction plays a crucial role in this transformation by enabling stereospecific inversion of configuration during nucleophilic substitution, thereby allowing efficient introduction of the nitrogen functionality into the cyclitol framework. However, the use of stoichiometric quantities of azodicarboxylate reagents and triphenylphosphine generates significant by-product waste, which may limit the environmental sustainability of this approach when considered on larger scales. Global deprotection of (112) with p-toluenesulfonic acid (p-TSA) yielded intermediate (113), which upon treatment with 40% aqueous methylamine delivered (−)-conduramine C-1 (−)-114 in quantitative yield.
In an alternative sequence, monoacetate (110) was subjected directly to a Mitsunobu reaction with phthalimide, affording protected intermediate (115) in 61% yield. Treatment of (115) with 40% aqueous methylamine produced compound (116) in ∼81% yield, and a subsequent methylamine-induced deprotection step yielded (+)-conduramine C-1 (+)-114 in ∼99% yield.
Johnson et al. also described the preparation of partially protected conduramine A derivatives. The sequence began with a hetero-Diels–Alder reaction between diene (56) and an in situ-generated nitroso dienophile (formed by tetraethylammonium periodate oxidation of benzohydroxamic acid), producing racemic oxazine (117). Reductive cleavage of the N–O bond using aluminum amalgam in THF furnished racemic alcohol (118).
Resolution of racemic (118) was achieved using amano P-30 lipase in isopropenyl acetate, providing a mixture of acetate (119) and unreacted alcohol (−)-118. Deacetylation of (119) produced alcohol (120), which upon acidic deprotection with p-TSA yielded partially protected conduramine A (+)-121. The opposite enantiomer (−)-121 was obtained by direct p-TSA-mediated deprotection of (−)-118 (Scheme 11).
 | ||
| Scheme 11 Synthesis of both enantiomers of conduramine C-1 (114) and partially protected conduramine A (121). | ||
These studies demonstrate how combinations of microbial oxidation, enzymatic resolution, and stereospecific functionalization can provide efficient access to structurally diverse conduramine derivatives. In addition to such aminocyclitol frameworks, related synthetic efforts have also focused on the preparation of electrophilic conduritol epoxides, which serve as valuable mechanism-based inhibitors of glycosidases and important probes for studying carbohydrate-processing enzymes.
2.11. Unified route to conduritol E and F epoxides via carbocyclization of a mannose-derived intermediate
Epoxide derivatives of conduritols have attracted considerable attention owing to their function as mechanism-based inactivators of glycosidases. These electrophilic cyclitols act as potent biochemical probes for elucidating the mechanistic features of glycoside hydrolases. For example, epoxy-conduritol F has been shown to irreversibly inactivate a liver-specific isoform of β-mannosidase isolated from goat liver, underscoring the biological significance of such compounds.In a seminal contribution published by Chretien et al. in 1993, a concise and efficient synthetic route to both conduritol E and F epoxides was developed starting from a D-mannose derivative.31 The synthesis began with the transformation of methyl α-D-mannopyranoside (122) into the crystalline 6-iodo peracetate (123) via a modified Garegg protocol followed by acetylation, affording the product in 91% yield.
Treatment of compound (123) with silver fluoride in pyridine induced an elimination reaction to furnish alkene (124) in 91% yield. Subsequent deacetylation of compound (124), followed by benzylation, provided the dibenzylated intermediate (125), again in 91% yield. The key carbocyclization step was accomplished using catalytic HgSO4 in sulfuric acid, converting compound (125) into ketol (126) in 80% yield. This mercury-mediated carbocyclization efficiently constructs the cyclohexene framework characteristic of conduritols while preserving the stereochemical information derived from the carbohydrate precursor. However, the use of mercury salts raises environmental and safety concerns, and the development of alternative metal-catalyzed or organocatalytic cyclization strategies would be desirable for improving the sustainability of such transformations.
Elimination of the hydroxyl group in ketol (126) with tosyl chloride in pyridine generated enone (127) in 82% yield. Stereoselective Luche-type reduction of enone (127) with NaBH4 in the presence of CeCl3 produced a mixture of partially protected conduritol E (128) and conduritol F (129). Oxidation of these intermediates with m-chloroperbenzoic acid (m-CPBA) yielded the corresponding epoxides: partially protected conduritol E epoxide (130) in ∼40% yield and conduritol F epoxide (131) in ∼80% yield (Scheme 12).
 | ||
| Scheme 12 Synthesis of partially protected conduritol E epoxide (130) and conduritol F epoxide (131) from methyl α-D-mannopyranoside (122). | ||
This sequence provides an elegant carbohydrate-derived approach to conduritol epoxides, enabling the stereocontrolled preparation of two biologically important epoxide isomers from a readily available chiral pool precursor.
In addition to carbohydrate-derived strategies, complementary approaches have employed chemoenzymatic arene dihydroxylation to generate versatile cyclitol intermediates from simple aromatic substrates. Such biooxidative methods have proven particularly powerful for accessing diverse inositol stereoisomers through controlled transformations of conduritol epoxide intermediates.
2.12. Chemoenzymatic divergent synthesis of inositol stereoisomers from chlorobenzene
In 1993, Hudlicky et al. developed a versatile synthetic strategy for the preparation of various inositols starting from chlorobenzene, with a conduritol E epoxide derivative serving as a key intermediate.32 The sequence commenced with the biotransformation of chlorobenzene (97) to chlorocyclohexadienediol (98) using Pseudomonas putida strain 39D. This microbial oxidation product was subsequently converted into the chloro epoxide (132), a pivotal branching point for constructing multiple inositol stereoisomers.Hydrolysis of epoxide (132) over alumina furnished the corresponding ketone (133) in approximately 85% yield, and catalytic hydrogenation of (133) produced allo-inositol (134) in yields exceeding 90%. Under radical dehalogenation conditions, epoxide (132) was transformed into epoxide (135), which upon hydrolysis either using Amberlite IR-118 resin or under neutral aqueous conditions afforded a 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 mixture of (+)-D-chiro-inositol (+)-137 and neo-inositol (138). The major isomer, (+)-137, was isolated in 60% yield and could be readily separated from neo-inositol via crystallization.
3 mixture of (+)-D-chiro-inositol (+)-137 and neo-inositol (138). The major isomer, (+)-137, was isolated in 60% yield and could be readily separated from neo-inositol via crystallization.
Further refinement of this route was achieved by treating epoxide (135) with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of amberlyst A-21 and Amberlite IRA-904, or alternatively with sodium benzoate, to promote highly selective formation of (+)-D-chiro-inositol via the acetoneide intermediate (139). This optimized method delivered (+)-137 in 98% yield with >95% selectivity, accompanied by only trace amounts (≤5%) of neo-inositol (138).
1 mixture of amberlyst A-21 and Amberlite IRA-904, or alternatively with sodium benzoate, to promote highly selective formation of (+)-D-chiro-inositol via the acetoneide intermediate (139). This optimized method delivered (+)-137 in 98% yield with >95% selectivity, accompanied by only trace amounts (≤5%) of neo-inositol (138).
Under acidic hydrolytic conditions, epoxide (135) underwent a Payne rearrangement, progressing through intermediates (140), (141), and (142) to ultimately furnish muco-inositol (143) (Scheme 13).
The Payne rearrangement plays a crucial role in this transformation by enabling intramolecular migration of the epoxide under basic or acidic conditions, thereby allowing controlled redistribution of stereochemical information across the cyclitol framework. Such rearrangements provide an efficient means of accessing multiple inositol stereoisomers from a common intermediate, although careful control of reaction conditions is necessary to minimize competing side reactions.
The ability to generate diverse inositol stereoisomers from common cyclitol intermediates highlights the remarkable versatility of chemoenzymatic arene dihydroxylation strategies. Beyond the synthesis of inositol derivatives, similar intermediates have also been employed as chiral building blocks for constructing biologically significant aminocyclitol antibiotics and related natural products. One notable example is the stereocontrolled synthesis of the aminocyclitol antibiotic fortamine.
2.13. Efficient stereocontrolled route to (+)-fortamine dihydrochloride from cis-cyclohexadiene diol acetonide
In 1994, Vandewalle et al. reported the total synthesis of (+)-fortamine dihydrochloride through a highly efficient and stereocontrolled sequence originating from the acetonide-protected cis-cyclohexa-3,5-diene-1,2-diol derivative (56).33 Following the protocol of Balcı et al., the starting material was transformed into diol (109) in an excellent 94% yield. Subsequent esterification of (109) with butanoyl chloride in the presence of triethylamine and 4-dimethylaminopyridine (DMAP) in dichloromethane afforded ester (144) in quantitative yield. Enzymatic hydrolysis of (144) using PGL (a recombinant Fusarium solani pisi cutinase) furnished the optically active alcohol (+)-145 in approximately 83% yield. The use of an enzyme-catalysed hydrolysis step is particularly advantageous in this sequence, as it enables highly selective kinetic resolution under mild conditions while avoiding the need for harsh reagents typically required for chemical resolution methods. However, the efficiency of such biocatalytic processes can depend strongly on enzyme availability, substrate compatibility, and reaction optimization.Alcohol (+)-145 was then protected as its methoxyphenylmethyl (MPM) ether, and hydrolysis of the butanoate ester generated alcohol (146) in 98% yield. Under Mitsunobu conditions, compound (146) underwent regioselective inversion to give alcohol (147) in 97% yield. Subsequent silyl protection of the free hydroxyl group followed by acetonide deprotection furnished diol (148) in 90% yield.
Treatment of diol (148) with sulfuryl chloride and triethylamine in dichloromethane led to formation of the cyclic sulfate (149) in 89% yield. Nucleophilic displacement of the sulfate moiety with lithium azide produced a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 mixture of regioisomers (150) and (151), which were separated by column chromatography. The major regioisomer, (151), was desilylated and then subjected to olefin epoxidation with m-CPBA to yield epoxide (152) in 93% yield. Benzylation of the hydroxyl groups followed by removal of the MPM protecting group afforded intermediate (153) in 98% yield.
3 mixture of regioisomers (150) and (151), which were separated by column chromatography. The major regioisomer, (151), was desilylated and then subjected to olefin epoxidation with m-CPBA to yield epoxide (152) in 93% yield. Benzylation of the hydroxyl groups followed by removal of the MPM protecting group afforded intermediate (153) in 98% yield.
Subsequent treatment of (153) with methyl isothiocyanate and triethylamine in dichloromethane produced the epoxy-urethane (154), which upon sequential reaction with sodium hydride and methyl iodide provided compound (155) in quantitative yield. Finally, catalytic hydrogenolysis of the benzyl protecting groups in the presence of formic acid, followed by acid hydrolysis of (155) using 6 N HCl, delivered (+)-fortamine dihydrochloride (+)-156 in approximately 98% yield for the final transformation (Scheme 14).
 | ||
| Scheme 14 Synthesis of (+)-fortamine dihydrochloride (+)-156 employing an enzyme-catalysed ester hydrolysis. | ||
The efficient construction of the aminocyclitol framework in this synthesis further illustrates the versatility of cyclohexadienediol-derived intermediates for accessing structurally diverse aminocyclitol natural products. In addition to antibiotic scaffolds such as fortamine, closely related synthetic strategies have also been applied to the preparation of conduramine derivatives, which serve as valuable intermediates for the synthesis of glycosidase inhibitors and other biologically active cyclitols.
2.14. Stereoselective construction of conduramine C-1: chemoenzymatic desymmetrization and carbohydrate-derived routes
Conduritols (cyclohex-5-ene-1,2,3,4-tetrols) and their amino analogues, conduramines in which one hydroxyl group is replaced by an amino functionality, have long attracted interest owing to their value as key intermediates in the synthesis of amino- and diaminocyclitols. These structural motifs constitute the aglycone cores of several aminoglycoside antibiotics, and numerous conduramine derivatives have demonstrated potent glycosidase inhibitory activity, underscoring their pharmacological relevance.The first optically pure conduramines were reported in 1981 by Paulsen and co-workers, who prepared conduramine F from the naturally occurring cyclitol quebrachitol.34 Subsequent advances in stereoselective synthesis further expanded access to this family of compounds. Notably, the groups of Ogawa and Knapp independently employed Ferrier-type cyclizations of D-glucose to furnish a variety of conduramine analogues, including the C1, C2, and F1 series.
Over the last few decades, numerous asymmetric methodologies have been developed for the construction of conduritols and conduramines. These include Diels–Alder cycloadditions of nitroso derivatives to cyclohexa-3,5-diene-1,2-diol systems as well as microbial oxidation of halogenated benzenes (e.g., bromo- or chlorobenzene), which provides access to optically enriched cyclohexadienediol precursors.
In 1992, Johnson et al. reported the first enantioselective syntheses of both (−)-conduramine C1 and (+)-conduramine C1.34 Their strategy began with the microbial oxidation of benzene to yield cyclohexa-3,5-diene-1,2-diol, which was subsequently converted into meso-2,3-O-isopropylideneconduritol A. The key desymmetrization step involved enzymatic monoacetylation using isopropenyl acetate in the presence of Pseudomonas cepacia lipase (Amano P-30), furnishing optically enriched intermediates suitable for divergence toward either enantiomer. Such chemoenzymatic desymmetrization strategies are particularly attractive because they enable efficient access to enantiomerically enriched intermediates from meso substrates under mild conditions. However, the outcome of enzymatic transformations may depend strongly on enzyme selectivity, substrate compatibility, and reaction optimization, which can influence their broader applicability in synthetic planning.
Further refinement came in 1994, when Vogel et al. developed an alternative synthesis of (−)-conduramine C1.35 Beginning with the sugar derivative (+)-157, a six-step sequence delivered ketone (+)-158 in an overall yield of approximately 46%. Stereoselective reduction with sodium cyanoborohydride afforded the endo-alcohol (−)-159 in 97% yield, which was subsequently acetylated using acetic anhydride and pyridine to give acetate (−)-160 in 75% yield. Treatment of (−)-160 with hydrobromic acid in acetic acid induced a regio- and stereoselective oxa-ring opening to furnish (−)-161 in 56% yield. Cyclization mediated by DBU then produced the protected conduramine C1 derivative (−)-162 in 95% yield. Final acidic deprotection of (−)-162 furnished the target compound, (−)-conduramine C1 (−)-114, also in 95% yield (Scheme 15).
 | ||
| Scheme 15 Stereoselective synthesis of (−)-conduramine C-1 (−)-114 via a carbohydrate-derived approach involving regioselective oxa-ring opening and intramolecular cyclization. | ||
The synthetic routes described above highlight the effectiveness of chemoenzymatic desymmetrization and carbohydrate-derived strategies for accessing stereochemically defined conduramine frameworks. Complementary approaches have also explored oxidative transformations of cyclohexadiene derivatives, where photooxygenation and subsequent rearrangements provide alternative entry points to aminocyclitol architectures.
2.15. Stereoselective synthesis of conduramine F-4 via photooxygenation-derived endo-peroxides
Aminocyclitols have garnered considerable attention owing to their potent glycosidase inhibitory activity and their value as key intermediates in the synthesis of aminoinositols and other biologically active cyclitol derivatives. Hasegawa et al. demonstrated the successful preparation of several aminocyclitols starting from cis- and trans-benzene diol isomers, highlighting the versatility of these precursors.36In 1994, Seçen et al. reported an efficient synthetic approach to conduramine F-4 (166), employing a concise sequence involving photooxygenation, reductive rearrangement, and nucleophilic epoxide opening.36 The route began with the photooxygenation of cyclohexadiene acetonide (56) using a 150-W projection lamp and tetraphenylporphyrin as a singlet oxygen sensitizer, affording the corresponding endo-peroxide (163) in an excellent 95% yield after silica gel purification. The singlet oxygen-mediated photooxygenation step is particularly valuable because it rapidly installs the peroxide functionality with high regio- and stereoselectivity, thereby generating a versatile intermediate that can be transformed into epoxides and aminocyclitols through subsequent reductive and nucleophilic processes. However, such photochemical transformations often require specialized irradiation equipment and careful control of reaction conditions.
Reductive cleavage of the endo-peroxide (163) with triethyl phosphite in chloroform furnished a single epoxide product (164) in 55% yield. This epoxide was then subjected to nucleophilic ring opening with ammonia in methanol, delivering the conduramine F-4 precursor (165) in approximately 95% yield. Final deprotection of the acetonide functionality under acidic hydrolytic conditions afforded conduramine F-4 (166) in near-quantitative yield (Scheme 16).
 | ||
| Scheme 16 Synthesis of conduramine F-4 (166) via a photooxygenation-derived endo-peroxide intermediate. | ||
This concise and stereoselective sequence illustrates the synthetic utility of photooxygenation-derived endo-peroxides and epoxide intermediates in accessing biologically relevant aminocyclitols.
While photooxygenation-based strategies provide an efficient route to aminocyclitol frameworks, alternative synthetic approaches have also been explored to access structurally diverse conduramine derivatives. In particular, cycloaddition-based methodologies have proven valuable for constructing highly functionalized intermediates that can be transformed into multiple conduramine isomers through divergent synthetic sequences.
2.16. Divergent synthesis of conduramine A-1, F-1, and C-1 from an N-Boc pyrrole Diels–Alder adduct
In 1994, Muchowski and co-workers reported a series of comprehensive synthetic routes to various conduramine derivatives using an N-substituted pyrrole framework as the key starting scaffold.37 One of their significant achievements was the synthesis of (±)-conduramine A-1 (±)-177. The sequence commenced with a Diels–Alder cycloaddition between N-Boc-pyrrole (167) and tosylacetylene (168) in a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio at 85 °C for 24 hours, furnishing cycloadduct (169) in 80–85% yield. The use of an N-substituted pyrrole as the diene component in this cycloaddition provides a versatile platform for constructing densely functionalized intermediates that can be diversified into multiple conduramine frameworks. However, the requirement for elevated temperatures and multistep downstream functionalization highlights the synthetic complexity associated with such cycloaddition-based approaches.
1 molar ratio at 85 °C for 24 hours, furnishing cycloadduct (169) in 80–85% yield. The use of an N-substituted pyrrole as the diene component in this cycloaddition provides a versatile platform for constructing densely functionalized intermediates that can be diversified into multiple conduramine frameworks. However, the requirement for elevated temperatures and multistep downstream functionalization highlights the synthetic complexity associated with such cycloaddition-based approaches.
Reduction of (169) with sodium borohydride in methanol produced an 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of endo/exo isomers of alcohol (170) in 94% yield. Subsequent treatment of this mixture with lithium bis(trimethylsilyl)amide (LiHMDS) in THF generated the conjugated diene (171) in 70–75% yield. Protection of the amino group in (171) with (Boc)2O in the presence of catalytic 4-DMAP afforded diene (172) in 85% yield.
1 mixture of endo/exo isomers of alcohol (170) in 94% yield. Subsequent treatment of this mixture with lithium bis(trimethylsilyl)amide (LiHMDS) in THF generated the conjugated diene (171) in 70–75% yield. Protection of the amino group in (171) with (Boc)2O in the presence of catalytic 4-DMAP afforded diene (172) in 85% yield.
Cis-dihydroxylation of (172) using osmium tetroxide/NMO provided diol (173) in 91% yield. Reductive desulfonation of (173) with sodium amalgam in buffered methanol furnished compound (174) in 42–50% yield. Protection of the resulting diol as an acetonide followed by epoxidation with m-CPBA delivered epoxide (175) in 90% yield. Regioselective epoxide opening with phenylselenide, followed by oxidative elimination with hydrogen peroxide, produced the protected conduramine A-1 intermediate (176) in 76% yield. Global deprotection furnished (±)-conduramine A-1 (±)-177 in 92% final yield.
The authors also developed a route to conduramine F-1 (183). Starting from diene (171), epoxidation with m-CPBA in dichloromethane gave epoxide (178) in approximately 90% yield. Acid-catalyzed hydrolysis afforded trans-diol (179) in 73% yield. Reductive desulfonation using sodium amalgam yielded compound (180) in 76% yield. Acetylation followed by epoxidation produced epoxide (181) in 68% yield, which was subsequently transformed into the protected conduramine F-1 derivative (182) in 51% yield. Final deprotection provided conduramine F-1 (183) in 95% yield.
Muchowski et al.37 also described the synthesis of (±)-conduramine C-1 (±)-114. The sequence began with syn-dihydroxylation of cycloadduct (169), followed by acetonide protection to afford compound (184) in 55–65% overall yield. Reduction with sodium borohydride delivered intermediate (185) in 97% yield. Treatment with LiHMDS afforded diene (186) in 72–75% yield, which underwent reductive desulfonylation to produce alkene (187) in 28–52% yield. Epoxidation of (187) furnished epoxide (188) in 99% yield. Subsequent epoxide opening followed by oxidation gave the protected conduramine C-1 intermediate (189) in 83% yield. Global deprotection ultimately delivered (±)-conduramine C-1 (±)-114 in 65% yield (Scheme 17).
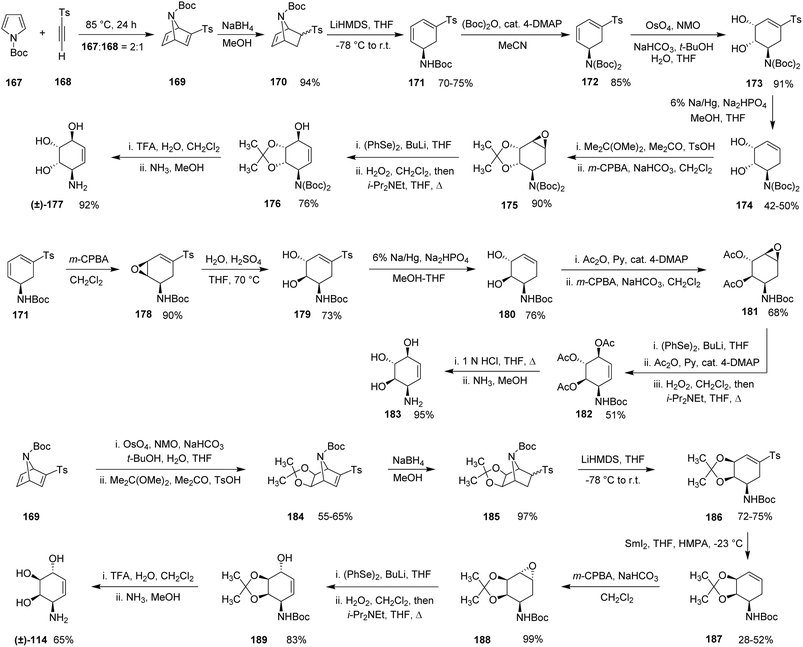 | ||
| Scheme 17 Synthesis of (±)-conduramine A-1 (±)-177, conduramine F-1 (183) and (±)-conduramine C-1 (±)-114 from a single N-Boc pyrrole (167). | ||
While the Diels–Alder-based approach provides an effective strategy for constructing conduramine frameworks from heterocyclic precursors, alternative synthetic routes have also been explored to access structurally modified cyclitol derivatives. In particular, photochemical oxygenation reactions have proven valuable for generating oxygen-rich intermediates that can be transformed into higher homologues of conduritols through controlled ring functionalization.
2.17. Synthesis of bis-homo-conduritol-D and -F via photooxygenation of cyclooctatetraene
In 1994, Balcı and co-workers reported an efficient and stereocontrolled synthetic route to bis-homo-conduritols starting from cyclooctatetraene (190).38 The sequence began with the transformation of cyclooctatetraene into dibromobicyclo[4.2.0]octa-2,4-diene (191), which was subsequently subjected to photooxygenation using tetraphenylporphyrin as a singlet oxygen sensitizer. This reaction afforded the corresponding endo-peroxide (192) in approximately 80% yield. The singlet-oxygen mediated photooxygenation of cyclooctatetraene provides an efficient entry into highly oxygenated bicyclic intermediates, enabling rapid construction of the functionalized frameworks required for bis-homo-conduritols. However, the requirement for photochemical irradiation and the inherent reactivity of peroxide intermediates may present practical challenges in terms of reaction control and scalability.Reductive cleavage of the endo-peroxide functionality in (192) with thiourea in methanol, followed by acetylation with acetic anhydride in pyridine, furnished diacetate (193) in 75% yield. Epoxidation of (193) using m-CPBA in chloroform produced epoxide (194), which upon debromination with zinc in DMSO yielded compound (195) in 84% yield. Regioselective epoxide ring opening under acidic conditions in the presence of acetic anhydride, followed by deacetylation, provided bis-homo-conduritol-F (196).
In a complementary sequence, syn-dihydroxylation of diacetate (193) with ethanolic KMnO4 generated diol (197), which was subsequently acetylated to furnish tetraacetate (198) with an overall yield of approximately 68%. Debromination of (198) using zinc in DMSO, followed by methanolysis to remove the acetyl groups, afforded bis-homo-conduritol-D (199) in quantitative yield (Scheme 18).
 | ||
| Scheme 18 Synthesis of bis-homo-conduritol-D (199) and bis-homo-conduritol-F (196) utilizing a photooxygeantion procedure. | ||
While the photooxygenation strategy described above provides efficient access to structurally expanded cyclitol frameworks such as bis-homo-conduritols, conduritol derivatives have also played a crucial role as chiral building blocks in the synthesis of complex biologically active natural products. In particular, the inherent stereochemical richness of conduritol scaffolds has enabled their application in the asymmetric synthesis of pharmacologically important alkaloids, including pancratistatin.
2.18. Stereocontrolled construction of (+)-pancratistatin from a conduritol-based chiral building block
One of the major limitations of contemporary cancer chemotherapy is the lack of selectivity, wherein cytotoxic agents frequently damage healthy cells alongside malignant ones. This persistent challenge has motivated the search for natural products exhibiting inherent selectivity toward cancer cells. Among these, pancratistatin, a natural alkaloid isolated from Pancratium littorale (spider lily), has attracted significant attention due to its remarkable and highly selective anticancer properties.In 1995, Trost et al. reported a stereoselective and efficient asymmetric total synthesis of (+)-pancratistatin based on a conduritol-derived chiral building block.39 Their synthetic sequence began with the protected conduritol A derivative (109), which was treated with two equivalents of n-butyllithium in THF at 0 °C. Quenching this dianion with methyl chloroformate afforded the decarbonate intermediate (200) in approximately 87% yield.
Azidation of (200) using trimethylsilyl azide, the chiral ligand (201), and π-allylpalladium chloride furnished azide (202) in 82% yield. The palladium-catalyzed azidation step represents a key stereochemical control element in this synthesis, enabling efficient installation of the nitrogen functionality while preserving the stereochemical integrity of the conduritol-derived framework. Nevertheless, the use of transition-metal catalysts and organometallic reagents may introduce considerations related to cost, reagent handling, and large-scale applicability. Subsequent reaction of azide (202) with Grignard reagent (203) in the presence of CuCN produced the corresponding 1,4-addition product (204). Syn-dihydroxylation of (204) with osmium tetroxide then delivered diol (205) in 62% yield. Protection of the resulting diol as its triethylsilyl (TES) ethers gave compound (206) quantitatively, which upon NBS bromination provided bromide (207) in 75% yield.
Compound (207) was further transformed through a sequence involving treatment with trimethylphosphine in THF/water, followed by reaction with phosgene in THF and triethylamine, affording intermediate (208). Exposure of (208) to tert-butyllithium induced metal–halogen exchange and intramolecular nucleophilic cyclization, yielding lactone (209) in 60–65% yield.
Removal of the TES protecting groups from (209) using TBAF in THF generated diol (210). Subsequent treatment with thionyl chloride and triethylamine, followed by RuCl3-catalyzed oxidation with NaIO4, furnished cyclic sulfate (211) in 72% yield. Regioselective ring opening of (211) with phenylsulfonyl cesium (PhSO2Cs), followed by acidic hydrolysis, afforded alcohol (212) in 85% yield.
Final global deprotection of (212) furnished (+)-pancratistatin (+)-213 in approximately 85% yield, completing an elegant and highly stereocontrolled total synthesis (Scheme 19).
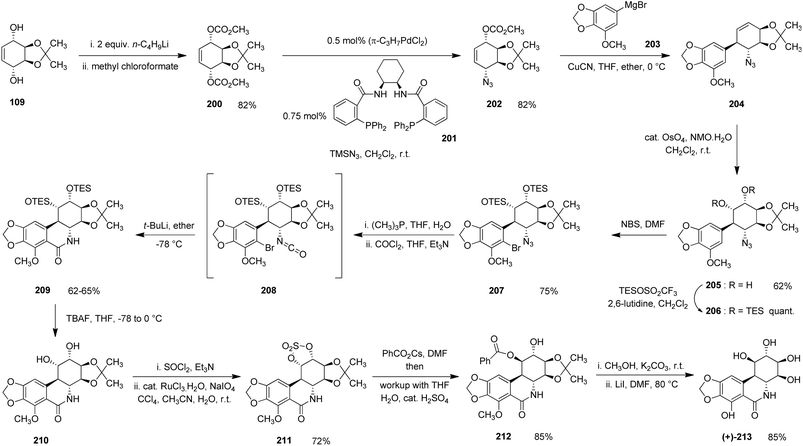 | ||
| Scheme 19 Enantioselective synthesis of (+)-pancratistatin (+)-213 from partially protected conduritol A. | ||
While conduritol derivatives have proven highly effective as chiral building blocks for the asymmetric synthesis of complex natural products such as pancratistatin, they have also been widely employed for the construction of aminocyclitol motifs present in biologically active antibiotics. In particular, conduritol-based intermediates provide convenient access to amino-deoxy-inositol frameworks, which constitute essential structural elements in several antibacterial natural products.
2.19. Construction of the amino-deoxy-neo-inositol framework of hygromycin A
Hygromycin A, an antibiotic produced by several Streptomyces species, demonstrates potent activity against both Gram-positive and Gram-negative bacteria. A defining structural feature of this molecule is the presence of a distinctive aminocyclitol moiety, an amino-deoxy-neo-inositol derivative. Recent structure–activity relationship studies on Hygromycin A analogues have emphasized the indispensable role of this aminocyclitol core in sustaining antibacterial potency.In 1995, Arjona et al. reported a synthetic route to this key aminocyclitol unit starting from a conduritol B derivative.40 Their sequence began with compound (214), which underwent sequential ring opening, Luche reduction, and acidic work-up to afford diol (215) in approximately 35% yield. Protection of the resulting diol with tert-butyldimethylsilyl triflate (TBSOTf) in the presence of triethylamine furnished the corresponding TBS ether (216) in 66% yield.
Mesylation of compound (216) using methanesulfonyl chloride in pyridine afforded the mesylate in about 90% yield. Subsequent nucleophilic substitution with tetrabutylammonium azide (n-Bu4NN3) produced azide (217) in 82% yield with clean inversion of configuration, an essential transformation for establishing the desired stereochemical framework. The azide displacement step is particularly valuable in this sequence because it enables stereospecific introduction of the nitrogen functionality through an SN2-type mechanism, thereby allowing precise control over the configuration of the aminocyclitol scaffold. However, the use of azide reagents requires careful handling due to their potential toxicity and safety considerations in large-scale operations.
The azide (217) was then subjected to syn-dihydroxylation using osmium tetroxide and trimethylamine N-oxide as the co-oxidant, delivering diol (218) in 75% yield with a diastereomeric ratio of 86![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 (neo
14 (neo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) epi). The OsO4/TMAO system provides efficient syn-dihydroxylation of the cyclohexene framework, enabling stereoselective installation of vicinal diol functionality required for the aminocyclitol architecture; nevertheless, the toxicity and cost of osmium reagents remain important considerations for large-scale applications. This diol (218) represents a versatile intermediate for assembling aminocyclitol architectures, thereby enabling the synthesis of hygromycin A analogues with potential antibacterial applications (Scheme 20).
epi). The OsO4/TMAO system provides efficient syn-dihydroxylation of the cyclohexene framework, enabling stereoselective installation of vicinal diol functionality required for the aminocyclitol architecture; nevertheless, the toxicity and cost of osmium reagents remain important considerations for large-scale applications. This diol (218) represents a versatile intermediate for assembling aminocyclitol architectures, thereby enabling the synthesis of hygromycin A analogues with potential antibacterial applications (Scheme 20).
In 1995, Trost et al. described a concise and highly efficient synthetic route to conduramine E.41 The sequence began with azide (219), which underwent a Staudinger reduction upon treatment with trimethylphosphine, affording amine (220) in an excellent 94% yield. Subsequent acidic hydrolysis of the acetonide protecting group in (220) furnished conduramine E (221) in approximately 88% yield. Final acetylation of conduramine E (221) with acetic anhydride in the presence of pyridine provided conduramine E tetraacetate (222) in about 70% yield (Scheme 21).
 | ||
| Scheme 21 Synthesis of conduramine E (221) and conduramine E tetraacetate (222) via staudinger reduction of an azide followed by deprotection and acetylation. | ||
While the preceding strategies highlight the use of conduritol derivatives as versatile intermediates for constructing aminocyclitol frameworks, alternative approaches have focused on the direct stereoselective synthesis of conduritol derivatives from readily available polyols. In this context, chiral pool precursors such as sorbitol have proven particularly valuable, providing an efficient and scalable platform for accessing stereochemically defined conduritol derivatives.
2.20. Enantioselective synthesis of (−)-conduritol F tetraacetate from D-sorbitol
In 1995, Chiara et al. reported an enantioselective synthesis of (−)-conduritol F tetraacetate (−)-230 starting from D-sorbitol (223).42 The sequence began with protection of D-sorbitol using acetone in the presence of sulfuric acid, followed by treatment with 70% aqueous acetic acid and subsequent benzoylation with benzoyl chloride in pyridine/dichloromethane, affording the protected intermediate (224). This compound then underwent disilylation with TBDPSCl and imidazole, and selective deprotection of the benzoate ether using DIBAL-H in toluene furnished alcohol (225).Oxidation of (225) under Swern conditions, followed by Pinacol coupling mediated by samarium(II) iodide, generated a diastereomeric mixture of myo-inositol (226) and L-chiro-inositol (227), strongly favoring the latter in a 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 94 ratio. The SmI2-mediated Pinacol coupling represents a powerful reductive carbon–carbon bond-forming transformation that enables rapid construction of the cyclitol framework from acyclic precursors. However, the requirement for low-valent samarium reagents and strictly controlled reaction conditions may limit the practicality of this approach for large-scale applications. The major isomer, L-chiro-inositol (227), was isolated and converted to thiocarbonate (228) via reaction with N,N′-thiocarbonyl diimidazole in toluene. Formation of the thiocarbonate intermediate is crucial for enabling subsequent elimination reactions that generate the cyclohexene framework characteristic of conduritol derivatives. Thermal treatment of (228) with trimethyl phosphite yielded the (−)-conduritol F derivative (229).
94 ratio. The SmI2-mediated Pinacol coupling represents a powerful reductive carbon–carbon bond-forming transformation that enables rapid construction of the cyclitol framework from acyclic precursors. However, the requirement for low-valent samarium reagents and strictly controlled reaction conditions may limit the practicality of this approach for large-scale applications. The major isomer, L-chiro-inositol (227), was isolated and converted to thiocarbonate (228) via reaction with N,N′-thiocarbonyl diimidazole in toluene. Formation of the thiocarbonate intermediate is crucial for enabling subsequent elimination reactions that generate the cyclohexene framework characteristic of conduritol derivatives. Thermal treatment of (228) with trimethyl phosphite yielded the (−)-conduritol F derivative (229).
Final deprotection was accomplished through a sequential protocol involving pyridinium p-toluenesulfonate in dichloromethane/methanol, followed by desilylation with TBAF in THF, and concluding with acetylation using acetic anhydride in pyridine. This sequence furnished (−)-conduritol F tetraacetate (−)-230 (Scheme 22).
While the chiral pool strategy described above enables efficient access to conduritol derivatives from readily available polyols, alternative synthetic approaches have employed functionalized cyclohexadienediols as versatile intermediates for constructing structurally diverse cyclitol and pseudo-sugar frameworks. In particular, halogenated cyclohexadienediol derivatives have proven valuable for introducing additional functional groups through metal–halogen exchange and related transformations.
2.21. Construction of pseudo-β-D-altropyranose derivatives from iodocyclohexadienediol
In 1995, Hudlicky and co-workers reported an efficient synthetic route to pseudo-sugars featuring a conduritol E intermediate as the key scaffold.43 The sequence began with the acetonide protection of the hydroxyl groups in iodocyclohexadienediol (231), affording compound (232). Subsequent dihydroxylation with osmium tetroxide and N-methylmorpholine N-oxide (NMO) provided the partially protected iodoconduritol E derivative (233). The OsO4/NMO-mediated dihydroxylation step plays a central role in establishing the vicinal diol motif required for the conduritol E scaffold, providing high stereochemical control. However, as noted for several related transformations discussed earlier, the toxicity and cost of osmium reagents represent important considerations when evaluating the practicality and environmental sustainability of this methodology. The remaining free hydroxyl groups in (233) were then masked using 2,2-dimethoxypropane (DMP) under acid catalysis to furnish the corresponding diacetonide (234) in an overall yield of approximately 75% from starting material (231).A metal–halogen exchange followed by carboxylation with CO2 and methylation with methyl iodide delivered ester (235) in ∼90% yield. This metal–halogen exchange strategy provides a convenient means of introducing carboxyl functionality at the iodinated position, thereby enabling further functionalization of the cyclitol framework toward pseudo-sugar architectures. Stereoselective hydrogenation over palladium generated ester (236) in 92% yield. Subsequent reduction of ester (236) with DIBAL-H in toluene afforded alcohol (237) in 74% yield. Finally, deprotection of the acetonide groups in (237) using amberlyst-15 resin in wet methanol, followed by acetylation with acetic anhydride in pyridine, furnished the target pentaacetate (238), a pseudo-β-D-altropyranose derivative in 89% overall yield (Scheme 23).
 | ||
| Scheme 23 Stepwise synthesis of pseudosugars via a conduritol E intermediate derived from iodocyclohexadienediol. | ||
While the above strategy illustrates how conduritol intermediates can be elaborated into structurally diverse pseudo-sugar frameworks, alternative methodologies have focused on exploiting enzymatic transformations to achieve stereoselective functionalization of conduritol derivatives. In particular, lipase-mediated kinetic resolution has emerged as an effective approach for accessing enantiomerically enriched conduritols and conduramines under mild conditions.
2.22. Synthesis of (+)-conduritol C and (+)-conduramine C-4 via enzymatic resolution
In 1996, Nicolosi et al. described a stereoselective approach for converting conduritol D tetraacetate (239) into (+)-conduritol C and (+)-conduramine C-4.44 The synthetic sequence commenced with the enzymatic resolution of compound (239) using various lipases in the presence of n-butanol, leading to the formation of alcohol (241). Among the enzymes screened, porcine pancreatic lipase (PPL) demonstrated the highest stereoselectivity, furnishing exclusively alcohol (+)-241, whereas the other lipases afforded mixtures of diol (+)-240 and alcohol (+)-241. The lipase-mediated kinetic resolution represents a particularly attractive strategy for generating enantiomerically enriched conduritol derivatives, as it proceeds under mild conditions and avoids the need for stoichiometric chiral auxiliaries or metal catalysts. Nevertheless, such enzymatic processes may be sensitive to substrate structure and reaction parameters, which can influence their broader synthetic applicability.The enantiomerically enriched alcohol (+)-241 was subsequently subjected to a Mitsunobu esterification with benzoic acid to yield (−)-benzoate (−)-242. The Mitsunobu reaction enables stereospecific inversion of configuration during nucleophilic substitution, providing an efficient means of introducing functional groups while maintaining control over the stereochemical outcome. Methanolysis of (−)-242 then provided (+)-conduritol C (+)-243. In a parallel transformation, (+)-conduramine C-4 (+)-244 was synthesized from alcohol (+)-241 via a Mitsunobu reaction employing diethyl azodicarboxylate (DEAD), triphenylphosphine, and phthalimide, followed by nucleophilic cleavage with methylamine (Scheme 24).
 | ||
| Scheme 24 Synthesis of (+)-conduritol C (+)-243 and (+)-conduramine C-4 (+)-244 using enzyme-catalysed selective ester hydrolysis. | ||
While enzymatic resolution provides an effective strategy for obtaining enantiomerically enriched conduritol and conduramine derivatives, complementary synthetic approaches have also focused on constructing aminocyclitol frameworks through direct functionalization of unsaturated precursors. In particular, aminohydroxylation reactions of dienyl systems have emerged as valuable transformations for introducing nitrogen and oxygen functionalities simultaneously, thereby enabling rapid assembly of conduramine scaffolds.
2.23. Synthesis of a partially protected conduramine E derivative
In 1997, Angelaud et al. developed a concise synthetic route to a partially protected conduramine E derivative starting from dienylsilane (245).45 The sequence began with an aminohydroxylation reaction, which afforded compound (246) in approximately 75% yield. The aminohydroxylation transformation is particularly valuable in this context because it enables simultaneous introduction of both nitrogen and oxygen functionalities across the unsaturated framework, thereby rapidly generating the key aminocyclitol architecture. However, the success of such reactions often depends strongly on substrate geometry and reagent compatibility, which can influence regio- and stereochemical outcomes. Oxidative cleavage of the C–Si bond using hydrogen peroxide, followed by acetonide protection of the resulting hydroxy-carbamate, furnished allylic alcohol (247) in about 70% yield. Subsequent epoxidation of (247) with m-CPBA generated epoxide (248) in 84% yield. Finally, treatment of epoxide (248) with 3.5 equivalents of lithium diisopropylamide (LDA) afforded the partially protected conduramine E derivative (249) in approximately 89% yield (Scheme 25). | ||
| Scheme 25 Synthesis of a partially protected conduramine E derivative via aminohydroxylation, epoxidation, and base-induced cyclization. | ||
The use of the strong, non-nucleophilic base LDA facilitates regioselective ring opening of the epoxide intermediate, enabling controlled formation of the aminocyclitol framework while preserving the established stereochemical relationships.
While the above strategy highlights the use of aminohydroxylation and epoxide ring-opening reactions for constructing conduramine frameworks, complementary approaches have explored chemoenzymatic transformations to achieve selective functionalization of conduritol derivatives. In particular, lipase-catalysed acylation reactions have proven highly effective for introducing protecting groups in a stereoselective manner, thereby enabling access to enantiomerically enriched conduritol intermediates.
2.24. Chemoenzymatic preparation of partial esters of conduritol E via lipase-catalysed selective acylation
In 1997, Sanfilippo and co-workers reported a chemoenzymatic strategy for the preparation of partial esters of conduritol E.46 The synthesis began with the dihydroxylation of syn-cyclohexadienediacetate (19) using osmium tetroxide, furnishing conduritol E diacetate (±)-250 in nearly quantitative yield. Subsequent enzymatic acylation of (±)-250 with lipozyme IM (lipase from Mucor miehei) and vinyl acetate in tert-butyl methyl ether (t-BME) afforded a diastereomeric mixture consisting of unreacted (+)-conduritol E diacetate (+)-250 and (−)-conduritol E triacetate (−)-251. Lipase-catalysed acylation represents a powerful tool for achieving selective esterification of polyhydroxylated substrates, offering high regio- and stereoselectivity under mild reaction conditions. However, the efficiency of such enzymatic transformations can depend strongly on solvent choice, enzyme activity, and substrate structure. The two components were separated by column chromatography. These partially esterified derivatives serve as useful intermediates for the enantioselective synthesis of both enantiomers of conduritol E and related analogues, including conduramines (Scheme 26). | ||
| Scheme 26 Synthesis of partial esters of conduritol E utilizing enzyme-catalysed selective ester hydrolysis. | ||
While chemoenzymatic strategies such as lipase-mediated acylation provide efficient routes to selectively functionalized conduritol derivatives, purely chemical transformations have also been exploited to construct the cyclohexene framework characteristic of conduritols. Among these, rearrangement reactions capable of converting sulfone precursors into olefinic cyclitol derivatives have emerged as particularly valuable tools for generating conduritol scaffolds with high efficiency.
2.25. Synthesis of 1,4-dimethoxy conduritol E via Ramberg–Bäcklund rearrangement of a D-mannitol-derived sulfone
In 1997, Cerè et al. developed an efficient route to 1,4-dimethoxy conduritol E starting from D-mannitol.47 The sequence began with the oxidation of the D-mannitol-derived intermediate (1S,2S,6S,7S)-(−)-2,6-dimethoxy-9,9-dimethyl-8,10-dioxa-4-thiabicyclo[5.3.0]decane (252), which furnished the corresponding sulfone (253) in 98% yield. Treatment of sulfone (253) with KOH in tert-butanol and tetrachloromethane induced a Ramberg–Bäcklund rearrangement, providing the conduritol E derivative (254) in 94% yield. The Ramberg–Bäcklund rearrangement represents a powerful method for converting sulfone precursors into olefinic systems, enabling efficient construction of the cyclohexene framework characteristic of conduritol derivatives. Nevertheless, the reaction typically requires strong base and halogenating conditions, which may impose limitations when sensitive functional groups are present. Hydrolysis of the acetonide protecting group in (254) subsequently afforded 1,4-dimethoxy conduritol E (255), also in 94% yield. Importantly, this intermediate can be converted to (−)-conduritol E in excellent yield without the need for further purification, underscoring the efficiency of the overall sequence (Scheme 27).While rearrangement reactions such as the Ramberg–Bäcklund transformation provide efficient access to olefinic conduritol frameworks, alternative strategies have focused on the construction of structurally modified conduritols through nucleophilic transformations of epoxide intermediates. In particular, epoxide ring-opening reactions offer a versatile approach for introducing nitrogen-containing bridges and generating conformationally constrained aminocyclitol architectures.
2.26. Synthesis of an amino-bridged conduritol via nucleophilic epoxide opening and acetonide deprotection
In 1997, Lallemand et al. reported a synthetic strategy for the construction of an amino-bridged conduritol.48 The sequence began with the bicyclic conduramine derivative (256), which was heated in a sealed tube with tert-butanol and epoxide (257), yielding the bridged amine (258) in approximately 50%. The nucleophilic opening of the epoxide ring plays a crucial role in this transformation by enabling formation of the nitrogen-bridged cyclitol framework while simultaneously establishing the required connectivity between the two ring systems. However, such intramolecular or intermolecular epoxide-opening reactions can sometimes suffer from moderate yields and competing side reactions, depending on the reaction conditions and nucleophile employed. Subsequent hydrolysis of the acetonide protecting group in (258) using TFA in aqueous THF afforded the desired amino-bridged conduritol (259) in about 90% yield (Scheme 28). Acid-mediated deprotection of the acetonide group provides a straightforward route to liberate the polyhydroxylated cyclitol framework while maintaining the stereochemical integrity of the bridged system. | ||
| Scheme 28 Synthesis of amino-bridged conduritol (259) via nucleophilic epoxide opening followed by acetonide deprotection. | ||
While epoxide-opening reactions provide an effective route for constructing bridged aminocyclitol architectures, alternative strategies have explored the use of rearrangement reactions to generate functionalized conduritol derivatives bearing multiple nitrogen substituents. In particular, the Ramberg–Bäcklund rearrangement has proven valuable for converting sulfone precursors into cyclohexene frameworks that can subsequently be transformed into diamino-conduritol derivatives.
2.27. Synthesis of a diamino conduritol B derivative via Ramberg–Bäcklund rearrangement
In 1998, Cerè et al. described an efficient synthetic route to a diamino conduritol B derivative.49 The sequence began with the nucleophilic substitution of (−)-(3S,4R,5R,6S)-4,5-dimesyl-3,6-dimethoxythiepane (260) with sodium azide in DMSO, affording diazide (261) in approximately 94% yield. Oxidation of (261) with m-CPBA in dichloromethane then provided the corresponding sulfone (262) in 98% yield. The sulfone underwent a Ramberg–Bäcklund transformation to give the cyclohexene derivative (263) with ∼78% efficiency. In this transformation, the Ramberg–Bäcklund rearrangement enables efficient conversion of the sulfone intermediate into the unsaturated cyclohexene framework characteristic of conduritol derivatives. However, the reaction typically requires strong base and halogenating conditions, which may restrict its compatibility with sensitive functional groups. Finally, simultaneous reduction of both the double bond and the azide groups in (263) was accomplished by refluxing with LiAlH4, delivering the target diamino conduritol B derivative (264) in 91% yield (Scheme 29). | ||
| Scheme 29 Synthesis of diamino conduritol B derivative (264) via a Ramberg–Bäcklund rearrangement and subsequent reduction. | ||
While rearrangement-based strategies such as the Ramberg–Bäcklund transformation provide efficient access to nitrogen-functionalized conduritol derivatives, conduritol frameworks have also served as versatile intermediates in the synthesis of biologically important natural products. In particular, the well-defined stereochemical arrangement of hydroxyl groups within conduritol derivatives has enabled their application in the synthesis of structurally related polyhydroxylated molecules such as L-ascorbic acid.
2.28. Conduritol F–based synthetic route to L-ascorbic acid
In 1998, Banwell and co-workers reported a synthetic strategy for the preparation of L-ascorbic acid (272) that employed a conduritol F derivative as a key intermediate.50 The sequence began with the protection of the hydroxyl groups of diol (98) using 2,2-dimethoxypropane, affording compound (77). Epoxidation of (77) with m-CPBA furnished epoxide (78), which was subsequently opened by benzyl alcohol in the presence of trifluoromethanesulfonic acid to provide the protected conduritol F derivative (265) in approximately 50% yield.Ozonolysis of compound (265) in methanol, followed by in situ reduction with sodium cyanoborohydride under acidic conditions, afforded compound (266) in 74% yield. Palladium-catalyzed hydrogenolysis of (266) in the presence of cyclohexa-1,4-diene (267) generated alcohol (268) in 53% yield. Reaction of (268) with benzaldehyde under acidic conditions then furnished acetal (269) in 47% yield.
Transformation of the primary hydroxyl group of (269) into its TBDMS ether gave compound (270) in 74% yield. Oxidation of (270) with Dess–Martin periodinane in dichloromethane afforded compound (271), which upon hydrolysis in acetic acid delivered the target molecule, L-ascorbic acid (272), in 66% yield (Scheme 30).
 | ||
| Scheme 30 Synthesis of L-ascorbic acid (272) synthesis of L-ascorbic acid from a conduritol F derivative. | ||
2.29. Synthesis of conduramine D-1 and its tetraacetate
In 1998, Leung-Toung et al. reported a multistep synthetic strategy for the preparation of conduramine D-1 starting from N-tert-butoxycarbonylpyrrole.51 A Diels–Alder reaction between N-Boc-pyrrole (167) and tosyl acetylene (168) furnished cycloadduct (169) in 84% yield. The Diels–Alder cycloaddition between the N-protected pyrrole and tosyl acetylene provides an efficient entry into a densely functionalized bicyclic framework that can be strategically manipulated toward conduramine architectures. Such cycloaddition-based approaches enable rapid construction of molecular complexity; however, subsequent functional group interconversions are often required to convert the bicyclic intermediate into the desired cyclitol skeleton. Syn-dihydroxylation of (169) with osmium tetroxide followed by treatment with PhB(OH)2 in tert-butanol produced borate (273) in 92% yield, which upon oxidation with hydrogen peroxide gave diol (274) in 88% yield. Alternatively, (274) could be obtained directly from (169) in 73% yield via syn-dihydroxylation with OsO4/NMO in tert-butanol.The osmium tetroxide-mediated syn-dihydroxylation is crucial for establishing the vicinal diol functionality with high stereochemical control, which subsequently enables efficient protection and downstream transformations leading to the conduramine framework. Nevertheless, the toxicity and cost associated with osmium reagents remain important considerations when evaluating the scalability of such methodologies.
Protection of the vicinal diol (274) as its acetonide using 2,2-dimethoxypropane and p-toluenesulfonic acid afforded compound (275) in 85% yield. Reduction of (275) with sodium borohydride generated a mixture of endo- and exo-isomers of (276), which underwent bicyclic fragmentation upon treatment with Li(NTMS)2 to furnish (277) in 79% yield. Deprotection of the acetonide group in (277) yielded diol (278) in 89% yield, and subsequent reductive desulfonylation of (278) afforded diol (279).
Stereoselective epoxidation of (279) with m-CPBA provided epoxide (280) in 79% yield. Regioselective ring opening of (280) with (PhSe)2 in the presence of n-BuLi, followed by acetylation with acetic anhydride, afforded triacetate (281) in 70% yield. Oxidative elimination of (281) then furnished the protected conduramine D-1 derivative (282) in 90% yield. Global deprotection of (282) by refluxing in 5 N HCl produced conduramine D-1 (283), which upon acetylation gave conduramine D-1 tetraacetate (284) in 95% combined yield over the final two steps (Scheme 31).
 | ||
| Scheme 31 Synthesis of conduramine D-1 (283) and conduramine D-1 tetraacetate (284) via a Diels–Alder-based strategy. | ||
Beyond the synthesis of conduramines, cyclohexadienediol-derived conduritol frameworks have also proven valuable intermediates for the preparation of structurally diverse natural products. In this context, Donohoe and co-workers reported a stereoselective route to the bioactive cyclitol natural product (+)-pericosine B using a bromocyclohexadienediol precursor.52
2.30. Synthesis of (+)-pericosine B from bromocyclohexadienediol
In 1998, Donohoe et al. reported a stereoselective synthesis of (+)-pericosine B (+)-291, beginning from bromocyclohexadienediol (74).52 Protection of one hydroxyl group with TIPSOTf in the presence of triethylamine afforded compound (285) in 95% yield. The remaining free hydroxyl group was methylated using sodium hydride and methyl iodide, and subsequent removal of the TIPS group with TBAF furnished alcohol (286) in 61% yield.Dihydroxylation of the diene (286) with osmium tetroxide yielded a mixture of anti-diol (287) and syn-diol (288), corresponding to conduritol E and D derivatives, respectively. The ratio of products was strongly influenced by the reaction medium and co-catalysts; notably, use of osmium tetroxide in dichloromethane with quinuclidine favored the syn-diol, giving an anti![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) syn ratio of 1
syn ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.2 and a combined yield of approximately 71%.
2.2 and a combined yield of approximately 71%.
The diastereoselectivity of osmium-mediated dihydroxylation is known to be sensitive to solvent effects and coordinating additives. In this case, the presence of quinuclidine likely modulates the approach of the osmium species to the diene framework, thereby influencing the facial selectivity of the oxidation and favoring formation of the syn-diol derivative.
Protection of the hydroxyl groups of the conduritol D derivative (288) as triethylsilyl ethers afforded compound (289) in 95% yield. Subsequent metal–halogen exchange using t-BuLi followed by trapping with methyl chloroformate produced ester (290) in ∼95% yield. The metal–halogen exchange step provides an efficient method for introducing a carbonyl functionality at the halogen-bearing position of the cyclohexene framework, enabling rapid functionalization of the conduritol-derived intermediate and facilitating conversion into the ester precursor required for completion of the natural product synthesis. Final desilylation with trifluoroacetic acid in dichloromethane furnished the target natural product, (+)-pericosine B (+)-291, in 90% yield (Scheme 32).
In addition to natural product synthesis, the development of new catalytic strategies has significantly expanded the synthetic toolbox for constructing conduritol frameworks. In this context, Chang and co-workers reported an elegant approach to 3,6-di-O-methyl conduritol E employing a ruthenium-catalysed ring-closing metathesis reaction.53
2.31. Synthesis of 3,6-di-O-methyl conduritol E via Ru-catalysed RCM
In 1999, Chang et al. reported an efficient synthetic route to 3,6-di-O-methyl conduritol E (255).53 The synthesis began with the partial reduction of (2R,3R)-2,3-O-isopropylidenetartrate (292), followed by nucleophilic addition of vinylmagnesium bromide, furnishing bis-allyl alcohol (293) in approximately 54% yield. Subsequent methylation of the resulting diol with sodium hydride and methyl iodide afforded the dimethoxy ether (294) in ∼94% yield.Cleavage of the acetonide protecting group with 10% HCl in methanol produced diol (295) in nearly quantitative yield (∼99%). Acetylation of (295) with acetic anhydride in the presence of triethylamine provided diacetate (296) in 92% yield. The key step involved a ring-closing metathesis (RCM) of (296) catalyzed by Grubbs' first-generation Ru–benzylidene complex [Cl2(PCy3)2Ru![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHPh], giving the conduritol E derivative (297) in ∼73% yield. Final deacetylation of (297) furnished the target 3,6-di-O-methyl conduritol E (255) in ∼99% yield (Scheme 33).
CHPh], giving the conduritol E derivative (297) in ∼73% yield. Final deacetylation of (297) furnished the target 3,6-di-O-methyl conduritol E (255) in ∼99% yield (Scheme 33).
Beyond the development of new catalytic approaches for conduritol synthesis, cyclohexadienediol-derived intermediates have also played an important role in the preparation of complex bioactive natural products. A notable example is the chemoenzymatic synthesis of the anticancer alkaloid narciclasine reported by Gonzalez and co-workers.11
2.32. Chemoenzymatic route to narciclasine involving microbial cis-dihydroxylation, Suzuki coupling, and intramolecular lactamization
Narciclasine, a biologically active alkaloid renowned for its potent anticancer and anti-inflammatory properties, was synthesized by Gonzalez et al. in 1999 via a chemoenzymatic route beginning with the biotransformation of m-dibromobenzene (298) using E. coli JM109 (pDTG601G).11 This process afforded syn-diol (299), which was converted into oxime (300) in approximately 70% yield through sequential acetonide protection of the hydroxyl groups followed by reaction with methyl carbamate (NHCO2Me) in the presence of sodium periodate (NaIO4).A Suzuki cross-coupling between oxime (300) and borate (301), catalyzed by Pd(PPh3)4, yielded compound (302) in ∼30% yield. Radical-mediated deoxygenation of (302) using TTMSS (tris(trimethylsilyl)silane) and AIBN furnished the key α,β-unsaturated ketone (303) in approximately 80% yield. Alternatively, a one-pot sequence involving Suzuki coupling of (300) with (301) followed by Mo(CO)6-mediated reduction in refluxing acetonitrile provided enone (303) in ∼45% yield.
Luche reduction of enone (303) gave allylic alcohol (304) in ∼80% yield, which underwent a Mitsunobu reaction with tributylphosphine, DEAD, and benzoic acid to furnish benzoate (305) in ∼65% yield. Acetonide deprotection of (305) using Dowex 50X8 resin in methanol, followed by acetylation of the resulting hydroxyl groups with acetic anhydride in pyridine, afforded diacetate (306) in ∼70% yield.
Subsequent treatment of (306) with trifluoromethanesulfonic anhydride and DMAP induced lactam formation, yielding compound (307) in ∼40% yield. Finally, global deprotection of the ester and methyl ester functionalities provided narciclasine (16) in ∼20% overall yield from the lactam stage (Scheme 34).
 | ||
| Scheme 34 Synthesis of narciclasine (16) utilizing enzyme-catalysed regio- and stereoselective dihydroxylation. | ||
In addition to enabling the synthesis of complex natural products, microbial cis-dihydroxylation of aromatic substrates has also been widely applied in the preparation of cyclitol frameworks. An illustrative example is the chemoenzymatic synthesis of neo-inositol reported by Hudlicky and co-workers.54
2.33. Chemoenzymatic preparation of neo-inositol
In 1999, Hudlicky et al. described a chemoenzymatic route to neo-inositol (138) beginning from bromobenzene (308).54 The synthesis commenced with a regio- and stereoselective enzymatic cis-dihydroxylation of bromobenzene using E. coli JM109 (pDTG601), furnishing the bromo-cyclohexadiene-cis-diol (74). Protection of the resulting diol as its acetonide was achieved using 2,2-dimethoxypropane and p-toluenesulfonic acid in acetone. The protected intermediate was then brominated with 1,3-dibromo-5,5-dimethylhydantoin (DBH) in aqueous acetone to yield bromohydrin (309).Treatment of bromohydrin (309) with aqueous KOH induced intramolecular cyclization to generate an unisolated epoxide (310), which rearranged upon reflux to yield the trans-diol (311). Radical debromination of (311) with tributyltin hydride (Bu3SnH) and AIBN in benzene afforded the corresponding conduritol C derivative (312).
Dihydroxylation of the alkene moiety in (312) with osmium tetroxide and NMO in a tert-butanol–acetone–water mixture produced the tetrol (313). Final acidic removal of the acetonide protecting group using methanolic HCl furnished neo-inositol (138) in approximately 77% yield from tetrol (313) (Scheme 35).
 | ||
| Scheme 35 Synthesis of neo-inositol (138) utilizing enzyme-catalysed regio- and stereoselective dihydroxylation. | ||
In addition to chemoenzymatic strategies, synthetic approaches based on classical functional group transformations and modern catalytic reactions have also been developed for constructing conduritol frameworks. In this context, Gallos and co-workers reported an efficient route to tetrabenzylated conduritol derivatives employing a ring-closing metathesis strategy.55
2.34. Synthesis of tetrabenzylated conduritols A, E, and F
In 1999, Gallos et al. reported an efficient synthetic strategy for the preparation of tetrabenzylated derivatives of conduritols A, E, and F, starting from the readily available alditols galactitol (314), D-mannitol (319), and D-glucitol (323), respectively.55 The synthetic sequence began with selective protection and benzylation steps: each alditol was sequentially treated with triphenylmethyl chloride, 4-dimethylaminopyridine (DMAP), and triethylamine in DMF, followed by benzylation with sodium hydride and benzyl chloride in DMF, and final acidic detritylation with sulfuric acid in a methanol–dichloromethane mixture. These transformations furnished the partially protected alcohol intermediates (315), (320), and (324) in 34%, 50%, and 39% yields, respectively.Swern oxidation of the primary alcohols afforded the corresponding dialdehydes, which underwent Wittig olefination with methyltriphenylphosphonium bromide in the presence of 12-crown-4 to give the dienes (316), (321), and (325) in 50%, 52%, and 53% yields, respectively. The final ring-closing metathesis (RCM) of these dienes, catalyzed by Grubbs' first-generation catalyst (317) in refluxing dichloromethane, produced the tetrabenzylated conduritol derivatives: conduritol A (318) in 99% yield, conduritol E (322) in 58% yield, and conduritol F (326) in 77% yield (Scheme 36).
 | ||
| Scheme 36 Synthesis of tetra-O-benzyl derivatives of conduritol A (318), conduritol E (322) and conduritol F (326). | ||
In addition to alditol-based approaches, carbohydrate-derived intermediates have also been widely employed for the stereoselective construction of conduramine frameworks. An illustrative example is the synthesis of a protected conduramine E derivative reported by Ovaa and co-workers.56
2.35. Carbohydrate-mediated synthesis of protected conduramine E
In 1999, Ovaa et al. reported a concise synthetic route to a protected conduramine E derivative starting from the readily available p-methoxyphenyl-β-D-galactopyranoside (327).56 The synthesis began with protection of the primary hydroxyl group as its tert-butyldiphenylsilyl (TBDPS) ether using TBDPSCl and pyridine. The syn-diol unit was then converted into an acetonide using dimethoxypropane under acidic conditions, followed by benzylation of the remaining free hydroxyl group with benzyl bromide and sodium hydride. Subsequent desilylation with TBAF furnished alcohol (328) in 88% yield.Alcohol (328) was transformed into iodide (329) in 95% yield, and chemoselective reduction of (329) with zinc in methanol afforded the open-chain aldehyde (330) in ∼99% yield. Wittig olefination of the aldehyde with methyl triphenylphosphoranylidene in acetonitrile provided the E-alkene (331) in 95% yield, which was reduced with lithium aluminium hydride in THF to furnish the allylic alcohol (332) in 72% yield.
Treatment of (332) with trichloroacetonitrile and DBU produced intermediate (333) in 95% yield. An Overman rearrangement of (333), catalyzed by PdCl2(MeCN)2 in toluene, delivered amide (334) in 73% yield. Hydrolysis of the amide followed by Boc-protection of the liberated amine using (Boc)2O and triethylamine afforded compound (335) in 75% yield. Finally, ring-closing metathesis (RCM) mediated by Grubbs' catalyst generated the target protected conduramine E (336) in 65% yield (Scheme 37).
In addition to conduramine derivatives, conduritol frameworks have also served as valuable intermediates in the synthesis of biologically active cyclitol natural products. A notable example is the stereocontrolled synthesis of (+)-cyclophellitol reported by Trost and co-workers.57
2.36. Stereocontrolled construction of (+)-cyclophellitol from conduritol B tetraacetate
In 1999, Trost et al. reported an elegant asymmetric synthesis of (+)-cyclophellitol, a potent β-glucosidase and HIV inhibitor, starting from racemic conduritol B tetraacetate (±)-6.57 The strategy commenced with a kinetic resolution mediated by [η3-C3H5PdCl]2 in combination with the chiral ligand (337), which, at 50% conversion, afforded a mixture consisting of unreacted (+)-conduritol B tetraacetate (+)-6 in near-quantitative yield, the desired monopivalate (−)-338 in ∼88% yield, and only ∼1% of dipivalate (339).The monopivalate (−)-338 was subjected to ammonium hydroxide in methanol, effecting global deacetylation and furnishing triol (340) in ∼95% yield. Benzylation of (340) with benzyl bromide in the presence of tetrabutylammonium iodide (TBAI) delivered the tribenzyl ether (341) in ∼79% yield. Reduction of the pivalate ester in (341) using DIBAL-H in dichloromethane afforded alcohol (342) in ∼90% yield.
Alkylation of (342) with (C4H9)3SnCH2I and potassium iodide provided compound (343) in ∼90% yield. A tin–lithium exchange followed by a 2,3-sigmatropic rearrangement established the requisite regio- and stereochemistry, giving compound (344) in ∼72% yield. Subsequent epoxidation of (344) with m-CPBA in dichloromethane produced epoxide (345) in ∼78% yield. A final palladium-catalyzed hydrogenolysis furnished (+)-cyclophellitol (+)-346 in an excellent ∼91% yield (Scheme 38).
In addition to serving as intermediates in the synthesis of biologically active cyclitols, conduritol frameworks have also been accessed through divergent strategies starting from carbohydrate precursors. An illustrative example is the diastereodivergent conversion of D-xylopyranose into conduritol and inositol derivatives reported by Kornienko and co-workers.58
2.37. Diastereodivergent conversion of D-xylopyranose into conduritol and inositol derivatives
In 1999, Kornienko et al. described a versatile approach for synthesizing partially protected derivatives of conduritols, L-chiro-inositol, and myo-inositol via protected conduritol intermediates.58 The sequence began with 2,3,4-tri-O-benzyl-D-xylopyranose (347), which was oxidatively transformed into aldehyde (348). Nucleophilic addition of vinylmagnesium bromide to aldehyde (348) afforded a mixture of diastereomeric dienes (349) and (350), with the ratio of these diastereomers tunable through modifications of the reaction conditions, as illustrated in Scheme 39. | ||
| Scheme 39 Synthesis of partially protected L-chiro and myo-inositols from a single sugar derivative. | ||
Both dienes (349) and (350) underwent efficient ring-closing olefin metathesis (RCM) in the presence of Grubbs' catalyst, yielding the partially protected conduritol derivatives conduritol F (351) in ∼99% yield and conduritol B (353) in ∼95% yield, respectively. Subsequent benzylation of these intermediates using benzyl bromide, sodium hydride, and DMF, followed by dihydroxylation with OsO4 and NMO in an acetone–water mixture, furnished the corresponding partially protected inositol derivatives: L-chiro-inositol (352) from compound (351) and myo-inositol (354) from compound (353) (Scheme 39).
In addition to carbohydrate-derived strategies, alternative synthetic approaches employing bicyclic precursors have also been developed for constructing conduritol and inositol frameworks. An illustrative example is the synthesis of allo-inositol and related derivatives reported by Mehta and co-workers.59
2.38. Synthesis of allo-inositol and MK 7607 from norbornyl-derived precursors
In 2000, Mehta et al. reported a synthetic route to allo-inositol (134) via a protected conduritol E intermediate.59 The sequence began with norbornyl derivative (355), which was treated with sodium methoxide in methanol to afford compound (356). Compound (356) was subsequently converted into the bis-acetonide tosylate (357). Heating compound (357) with sodium iodide in acetone for 30 hours furnished the corresponding iodide in ∼92% yield. Treatment of this iodide with potassium tert-butoxide in tert-butyl alcohol effected an elimination to give olefin (358) in 70% yield.Ozonolysis of olefin (358) provided cyclohexanone (359) in ∼90% yield. Reduction of (359) with sodium borohydride in methanol afforded the corresponding alcohol in 89% yield, which was then mesylated using methanesulfonyl chloride and triethylamine in dichloromethane to yield mesylate (360) in 90% yield. Elimination of (360) with potassium tert-butoxide in DMSO furnished the conduritol E derivative (361) in ∼75% yield. Subsequent dihydroxylation of (361) with osmium tetroxide in the presence of NMMO provided diol (362) in 70% yield. Deprotection of the acetonide groups using 5% HCl afforded allo-inositol (134).
Olefin (358) also served as a precursor for the synthesis of MK 7607 (367). Dihydroxylation of (358) with osmium tetroxide and NMMO generated a 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 mixture of diols (363) and (364) in an overall 95% yield. Acetylation of diol (363) with acetic anhydride produced the corresponding diacetate in nearly quantitative yield. Treatment of this diacetate with thionyl chloride and pyridine gave a 1
30 mixture of diols (363) and (364) in an overall 95% yield. Acetylation of diol (363) with acetic anhydride produced the corresponding diacetate in nearly quantitative yield. Treatment of this diacetate with thionyl chloride and pyridine gave a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 mixture of acetates (365) and (366) in ∼45% yield. Finally, MK 7607 (367) was obtained in >95% yield through one-pot acetonide deprotection and hydrolysis of acetate (365) using 5% HCl (Scheme 40).
2 mixture of acetates (365) and (366) in ∼45% yield. Finally, MK 7607 (367) was obtained in >95% yield through one-pot acetonide deprotection and hydrolysis of acetate (365) using 5% HCl (Scheme 40).
 | ||
| Scheme 40 Divergent synthesis of allo-inositol (134) and MK 7607 (367) from norbornyl-derived precursors via a conduritol E intermediate. | ||
Beyond the synthesis of simple cyclitol derivatives, conduritol intermediates have also been employed in the preparation of biologically significant inositol phosphates. A representative example is the synthesis of myo-inositol polyphosphates reported by Plettenburg and co-workers using a conduritol B intermediate.
2.39. Synthesis of myo-inositol polyphosphates via a conduritol B intermediate
In 2000, Plettenburg et al. reported a synthesis of myo-Ins(1,2,3,4)P4 (376) starting from p-benzoquinone (9) via an intermediate conduritol B derivative.60 The racemic diacetate (±)-368, prepared in three steps from p-benzoquinone, was subjected to enzymatic resolution using pig pancreas lipase, affording diol (369) and the unreacted diacetate (368), both in high yields and separable by recrystallization. Hydrolysis of the diacetate with KOH in anhydrous THF followed by intramolecular nucleophilic substitution furnished the diepoxide (370) in approximately 80% yield. Subsequent reaction of (370) with dibenzyl benzoate resulted in double allylic epoxide opening to generate diphosphoconduritol B (371) in ∼80% yield.Acetylation of diphosphoconduritol B (371) with acetic anhydride in pyridine provided the corresponding diacetate (372) in nearly quantitative yield. Oxidation of (372) using RuCl3 and NaIO4 furnished diol (373) in ∼86% yield. Phosphorylation of diol (373) with 3-diethylamino-2,4,3-benzodioxaphosphepane (374) in the presence of 1H-tetrazole, followed by oxidation with m-CPBA, afforded compound (375) in ∼80% yield. Finally, hydrogenolysis of (375) over Pd/C, followed by hydrolysis of the acetate groups, delivered myo-Ins(1,2,3,4)P4 (376) in nearly quantitative yield (Scheme 41).
In addition to their application in the synthesis of inositol phosphates, conduritol derivatives have also served as valuable precursors for the preparation of aminocyclitols such as conduramines. An illustrative example is the chemoenzymatic synthesis of (−)-conduramine E reported by Prinzbach and co-workers.61
2.40. Chemoenzymatic synthesis of (−)-conduramine E via enzymatic resolution and Mitsunobu functionalization
In 2000, Prinzbach et al. reported a synthesis of (−)-conduramine E (−)-221.61 The sequence began with diacetate (377), which was subjected to enzymatic resolution using SP 523 in n-hexane and a 0.2 N phosphate buffer (pH ≈ 7), affording alcohol (+)-378 in 80–90% yield with 94% enantiomeric excess. Alcohol (+)-378 was then converted into compound (379) via a Mitsunobu reaction employing NHZ2 as the nucleophile, along with triphenylphosphine and DEAD, giving the product in 80–92% yield.Deprotection of (379) with ammonia in methanol furnished primary amine (380) in nearly quantitative yield. Treatment of (380) with aqueous acetic acid generated urethane (381) in ∼92% yield. Hydrolysis of urethane (381) with barium hydroxide then delivered (−)-conduramine E (−)-221 in approximately 86% yield (Scheme 42).
While chemoenzymatic approaches provide an efficient strategy for accessing enantiomerically enriched conduramines, alternative routes to related cyclitol derivatives have been developed using naturally occurring chiral pool precursors. In this context, Falshaw and co-workers demonstrated enantiodivergent syntheses of conduritol B epoxides starting from naturally occurring inositol derivatives such as quebrachitol and pinitol.62
2.41. Enantiodivergent syntheses of 1L- and 1D-conduritol B epoxides from quebrachitol and pinitol
In 2000, Falshaw et al. reported the synthesis of both enantiomers of conduritol B epoxide.62 For the preparation of 1L-conduritol B epoxide (1L-8), the synthesis commenced from the natural product 1L-quebrachitol (382), which was demethylated with hydroiodic acid to afford 1L-chiro-inositol (1L-137). Subsequent treatment with 2,3-butanedione and trimethyl orthoformate in the presence of an acid catalyst furnished the corresponding diacetal (383). The free hydroxyl groups of (383) were then converted into allyl ethers using allyl bromide and sodium hydride, providing compound (384). Acidic hydrolysis of the acetal moiety with trifluoroacetic acid in water yielded the tetraol (385), which upon benzylation with benzyl bromide and sodium hydride gave benzyl ether (386). Cleavage of the allyl protecting groups in (386) using p-toluenesulfonic acid produced diol (387), which was subsequently transformed into epoxide (388) via a Mitsunobu reaction employing triphenylphosphine and DEAD. Final palladium-catalyzed hydrogenolysis of (388) afforded 1L-conduritol B epoxide (1L-8) in an overall yield of approximately 9%.The synthesis of 1D-conduritol B epoxide (1D-8) began from 1D-pinitol (23), which underwent demethylation with hydroiodic acid to produce 1D-chiro-inositol (1D-137). Protection of the vicinal diols with 2,2-dimethoxypropane in the presence of catalytic TsOH furnished the diacetonide (389). Benzylation of the remaining free hydroxyl groups with benzyl bromide, tetrabutylammonium iodide, and sodium hydride provided dibenzyl ether (390). Subsequent removal of the acetonide groups using trifluoroacetic acid generated tetraol (391). Regioselective stannylene-mediated benzylation of the 2,5-equatorial hydroxyl groups delivered the tetrabenzyl intermediate (392), which was converted into epoxide (393) through a Mitsunobu reaction. Final palladium-catalyzed hydrogenolysis of (393) furnished 1D-conduritol B epoxide (1D-8) in an overall yield of approximately 29% (Scheme 43).
 | ||
| Scheme 43 Synthesis of both enantiomers of conduritol B epoxide (1L-8) and (1D-8) from 1L-quebrachitol (382). | ||
While chiral pool strategies based on naturally occurring inositols provide efficient routes to stereochemically defined conduritol derivatives, alternative approaches have explored the construction of more complex architectures incorporating multiple cyclitol units. In this context, Mehta and co-workers reported the synthesis of unusual fused-ring systems in which two conduritol frameworks share a common bond.63
2.42. Construction of conduritol D–E and A–E fused ring systems
In 2000, Mehta et al. described the synthesis of a unique class of molecules incorporating two fused conduritol rings sharing a common bond.63 The sequence began with the Diels–Alder adduct (394), which was transformed into the corresponding diene (395) following a strategically modified literature protocol based on the procedures of Forman and Dailey. The diene (395) was then subjected to dihydroxylation using catalytic osmium tetroxide in the presence of N-methylmorpholine N-oxide (NMMO), delivering the tetraol (396) in approximately 66% yield.Sequential treatment of (396) with amberlyst-15 resin and 4 Å molecular sieves afforded a dehydrated intermediate in 75% yield, which underwent reductive dechlorination to give compound (397) in 49% yield. Deketalisation of (397) with amberlyst-15 in acetone produced the ketone (398) in 98% yield. Subsequent decarbonylation of (398) with nitrobenzene furnished the cyclohexadiene derivative (399) in 62% yield. Dihydroxylation of (399) using OsO4/NMMO afforded diol (400) in 85% yield. Final acetonide deprotection with trifluoroacetic acid delivered compound (401)—a fused system comprising conduritol D and conduritol E in ∼95% yield.
In a complementary pathway, the Diels–Alder adduct (394) was converted to diol (402) following a previously reported method. Deketalisation of (402) using amberlyst-15 in acetone yielded the ketone (403) in 95% yield. Decarbonylation of (403) with nitrobenzene provided the cyclohexadiene (404) in 34% yield, which upon dihydroxylation with OsO4/NMMO afforded diol (405) in 73% yield. Final acetonide deprotection using trifluoroacetic acid furnished compound (406), featuring a fused conduritol A–conduritol E framework, in 90% yield (Scheme 44).
 | ||
| Scheme 44 Synthesis of fused conduritol D–E and A–E bicyclic systems from a Diels–Alder-derived precursor. | ||
While the construction of fused conduritol frameworks illustrates the structural diversity accessible from cyclitol intermediates, alternative strategies have focused on functional group transformations of conduritol epoxides to access aminocyclitol derivatives. In this regard, Lee and co-workers reported a regioselective aminolysis of conduritol epoxides for the preparation of protected conduramine F derivatives.64
2.43. Regioselective aminolysis of conduritol epoxides for constructing conduramine F frameworks
In 2000, Lee et al. described an efficient approach for the synthesis of protected derivatives of conduramine F.64 The strategy began with the cis-diols (98) and (74), obtained through the microbial oxidation of chlorobenzene and bromobenzene, respectively, using Pseudomonas putida. These diols were converted into their corresponding acetonides (77) and (24) via treatment with 2,2-dimethoxypropane, providing the protected derivatives in 86% and 90% yields, respectively.Epoxidation of acetonides (77) and (24) using m-chloroperbenzoic acid (m-CPBA) in dichloromethane furnished the epoxides (78) and (27) in 85% and 84% yields, respectively. Subsequent reaction of epoxides (78) and (27) with aniline in the presence of neutral alumina resulted in regioselective epoxide ring opening, affording the protected conduramine F derivatives (407) and (408) in 72% and 70% yields, respectively. Likewise, treatment of epoxide (27) with benzylamine under analogous conditions provided the corresponding protected amine (409) in approximately 70% yield.
The derivatives (407), (408), and (409) serve as versatile intermediates for the synthesis of conduramine F, dihydroconduramine F, and a range of structurally related analogues (Scheme 45).
 | ||
| Scheme 45 Regioselective aminolysis of conduritol epoxides for the synthesis of protected conduramine F derivatives. | ||
While regioselective aminolysis of conduritol epoxides provides an efficient route to aminocyclitol derivatives such as conduramines, conduritol intermediates have also been exploited as versatile chiral building blocks for the synthesis of other biologically active carba-sugars. A notable example is the enantioselective synthesis of gabosine derivatives reported by Banwell and co-workers.65
2.44. Conduritol E–based chiral pool approach to (−)-gabosine A
Gabosines constitute a distinctive class of carba-sugars noted for their enzyme-inhibitory activity, plant growth-regulating properties, and DNA-binding capabilities. In 2001, Banwell et al. developed an enantioselective synthetic route to (−)-gabosine A (−)-415, employing a conduritol E–derived intermediate as a central building block.65The synthesis began with the regioselective protection of the less hindered hydroxyl group of iodocyclohexadienediol (231) using tert-butyldiphenylsilyl chloride (TBDPS–Cl), affording the silyl ether (410). Subsequent dihydroxylation of (410) with osmium tetroxide in the presence of N-methylmorpholine N-oxide (NMMO) delivered the partially protected conduritol E derivative (411).
The newly formed syn-diol in (411) was then protected as an acetonide using 2,2-dimethoxypropane and catalytic p-toluenesulfonic acid, providing compound (412) in an overall yield of ∼78% from starting material (231). Swern oxidation of (412) afforded the corresponding ketone (413) in 85% yield. Nucleophilic addition of MeMgCl to (413), promoted by FeCl3 and 2-methyl-2-nitropropane (MNP), produced compound (414) in 94% yield. Finally, acidic removal of the acetonide protecting group furnished (−)-gabosine A (−)-415 in ∼85% yield (Scheme 46).
While conduritol E-derived intermediates have been successfully utilized as chiral building blocks for the synthesis of biologically active carba-sugars such as gabosines, conduritol derivatives have also served as valuable precursors in asymmetric catalytic approaches toward aminocyclitol-based natural products. An illustrative example is the enantioselective palladium-catalyzed synthesis of cyclophellitol reported by Trost and co-workers.66
2.45. Synthesis of (−)-cyclophellitol via enantioselective Pd catalysis
In 2001, Trost et al. reported an enantioselective synthesis of (−)-cyclophellitol (−)-346.66 The route began with the coupling of the protected conduritol B derivative (+)-416 with the sodium salt of (phenylsulfonyl)nitromethane (417), catalyzed by 2.5% [Pd2(dba)3]·CHCl3 in the presence of the chiral ligand (R,R)-337. This transformation afforded compound (418) in 81% yield and 88% enantiomeric excess.Treatment of (418) with N,N,N′,N′-tetramethylguanidine (TMG) in dichloromethane, followed by oxidation with dimethyldioxirane (DMDO), furnished the carboxylic acid (419) in 78% yield. Subsequent reduction of (419) provided the alcohol (420) in 70% yield, which upon epoxidation generated a mixture of epoxides (421) and (422). Among the methods evaluated, the MTO/H2O2 system proved optimal, delivering epoxide (421) in 15% yield and epoxide (422) in 65% yield.
Finally, global deprotection of the major epoxide (422) furnished (−)-cyclophellitol (−)-346 in 95% yield (Scheme 47).
While enantioselective palladium-catalyzed methodologies have enabled efficient access to biologically important cyclitol derivatives such as cyclophellitol, similar asymmetric catalytic strategies have also been applied to the synthesis of aminocyclitol fragments present in clinically relevant antibiotics. A notable example is the synthesis of the aminocyclitol core of hygromycin A reported by Trost and co-workers.67
2.46. Synthesis of the hygromycin A aminocyclitol scaffold via asymmetric Pd catalysis
In 2001, Trost et al. reported an efficient synthetic route toward the aminocyclitol core of the antibiotic hygromycin A.67 The sequence commenced with the bromination of p-benzoquinone (9) in dichloromethane using elemental bromine, affording the corresponding anti-dibromo adduct in 76% yield. Subsequent reduction with sodium borohydride furnished diol (369) in 82% yield. Acetylation of the resulting hydroxyl groups with acetic anhydride in the presence of potassium carbonate, followed by refluxing in acetic acid, delivered conduritol B tetraacetate (6) in 71% overall yield. This intermediate was further converted into the tetra(2,2,2-trichloroethyl carbonate) derivative (416).The tetra(2,2,2-trichloroethyl carbonate) derivative was then subjected to an asymmetric palladium-catalyzed process employing [η3-C3H5PdCl]2, the chiral ligand (S,S)-337, benzoic acid, and tetrahexylammonium benzoate (THAB), affording dibenzoate (423) in an excellent 90% yield. Subsequent reduction of (423) with zinc in acetic acid provided the anti-diol (424) in 77% yield. Treatment of (424) with benzyl isocyanate in the presence of triethylamine generated the corresponding monocarbamate (425), which upon exposure to triflic anhydride furnished triflate (426).
Displacement of the triflate (426) with potassium bis(trimethylsilyl)amide afforded the oxazolidinone (427) in 70% yield over two steps. Dihydroxylation of (427) using osmium tetroxide with N-methylmorpholine N-oxide (NMO) as the stoichiometric reoxidant delivered diol (428) in 80% yield. Conversion of the two free hydroxyl groups of (428) to the corresponding methylene acetal (429) was achieved almost quantitatively using dimethoxymethane in the presence of trimethylsilyl triflate and 2,6-lutidine. Finally, reductive deprotection of the benzylic amine and benzoate ester functionalities in (429) with lithium in liquid ammonia furnished the partially protected aminocyclitol fragment of hygromycin A, compound (430), in 77% yield (Scheme 48).
While asymmetric catalytic strategies provide powerful approaches for constructing complex aminocyclitol frameworks, alternative methodologies have focused on the stereoselective functionalization of simpler cyclohexene derivatives to access conduritol analogues. In this context, Sousa and co-workers reported an efficient synthesis of conduritol F tetraacetate via α-hydroxylation of an allylic alcohol intermediate.68
2.47. Synthesis of conduritol F tetraacetate via α-hydroxylation of an allylic alcohol
In 2001, Sousa et al. reported an efficient synthesis of conduritol F tetraacetate (230).68 The sequence began with the epoxidation of cyclohexene (431) using m-CPBA in cyclohexane, affording the anti-epoxide (432) in 81% yield. Ring opening of (432) with the chiral base (1S,2R)-433 provided the corresponding allylic alcohol (434) in >90% yield. Subsequent oxidation of (434) with PCC in dichloromethane furnished enone (435) in 88% yield.Deprotonation of enone (435) with NaHMDS in THF generated the corresponding enolate, which was directly trapped with Davis' oxaziridine (436) to give α-hydroxylated product (437) in 91% yield. Sequential acetylation of (437) using acetic anhydride/pyridine, followed by Luche reduction, afforded alcohol (438) in nearly quantitative yield. Final desilylation of (438) with TBAF in THF, followed by global acetylation using acetic anhydride/pyridine, delivered conduritol F tetraacetate (230) in an overall yield of 87% over the last four steps (Scheme 49).
While stereoselective functionalization of cyclohexene derivatives provides efficient access to individual conduritol frameworks, further synthetic developments have explored the assembly of higher-order cyclitol architectures. In this regard, Paul and co-workers reported divergent synthetic strategies for constructing N-linked and O-linked inositol dimers derived from conduramine and conduritol intermediates.69
2.48. Divergent routes to N-linked and O-linked inositol dimers
In 2001, Paul et al. reported the synthesis of a series of N-linked and O-linked inositol oligomers from partially protected N-linked conduramine dimers and O-linked dimers derived from conduramine and conduritol F.69 The sequence commenced with the syn-dihydroxylation of bromobenzene (308) using E. coli JM109(pDTG601), affording diol (74). Protection of (74) as its acetonide using DMP under acidic conditions, followed by treatment with PhINTs, furnished N-tosyl vinylaziridine (439). Alternatively, epoxidation of the DMP-protected diol (74) with m-CPBA generated vinyloxirane (27).N-Tosyl vinylaziridine (439) was reacted with ammonia in the presence of Yb(OTf)3 and subsequently coupled with vinyloxirane (27) in dioxane to afford the N-linked conduramine dimer (440) in 98% yield. Under analogous conditions, reaction with (27) furnished the stereoisomeric N-linked dimer (445) in 79% yield. Acetylation of (440) with acetic anhydride/pyridine produced the corresponding acetate in 93% yield. Conversion of its amino group to the trifluoroacetamide yielded compound (441), which upon radical debromination furnished derivative (442) in 73% yield.
Likewise, radical debromination of compound (445) produced (446) in 83% yield. Acetylation of the hydroxyl groups of (446), followed by trifluoroacetamide formation at the amino group, afforded compound (447) in 88% yield. Compounds (442) and (447) were then subjected to sequential syn-dihydroxylation and acetonide protection of the resulting diols to provide tetraacetonide derivatives (443) and (448) in 61% and 53% yield, respectively. Subsequent hydrolysis of the amide and ester functionalities, reductive treatment with sodium in liquid ammonia, and removal of the acetonide groups delivered the N-linked inositol dimers (444) and (449) in 77% and 87% yield, respectively.
The synthesis of O-linked inositol oligomers began with epoxide ring opening using KOH to afford diol (450) in 63% yield. Reaction of (450) with vinylaziridine (439) in the presence of BF3·OEt2 produced a mixture of O-linked conduritol F derivatives (451) and (452) in 52% combined yield. Radical debromination of (451) and (452) afforded (453) and (454), respectively, in 68% yield. These intermediates underwent syn-dihydroxylation followed by acetonide protection to yield compound (455) in 60% overall yield (two steps). Finally, global deprotection of (455) furnished the O-linked inositol dimer (456) in 52% yield (Scheme 50).
 | ||
| Scheme 50 Synthesis of N-linked conduramine dimers, N-linked inositol dimers, O-linked conduritol F dimer and O-linked inositol dimer. | ||
While the construction of N-linked and O-linked inositol dimers highlights the potential of conduramine and conduritol derivatives for assembling higher-order cyclitol architectures, further synthetic developments have explored strategies for introducing multiple amino functionalities into the cyclitol framework. In this regard, Arcelli and co-workers reported the synthesis of diamino-substituted conduritol derivatives via sulfone–olefin rearrangement pathways.70
2.49. Synthesis of 2,3-diamino conduritol F and B analogues via sulfone–olefin rearrangement pathways
In 2001, Arcelli and co-workers reported the synthesis of both 2,3-diamino-1,4-dimethoxyconduritol F and 2,3-diamino conduritol B.70 The synthesis of 2,3-diamino-1,4-dimethoxyconduritol F began with the protection of the hydroxyl groups of diol (457) using methanesulfonyl chloride in pyridine, affording dimesylate (458). Subsequent oxidation of (458) with m-CPBA in dichloromethane furnished sulfone (459) in 93% yield.Sulfone (459) was treated with sodium azide in DMSO to give a mixture of the expected azide (460) and byproduct (461) in 62% and 24% yield, respectively. Purification of (460) by flash chromatography followed by a Ramberg–Bäcklund olefination furnished cyclohexene (462) in 90% yield. Reduction of (462) with triethylamine and 1,3-propanedithiol in methanol then afforded 2,3-diamino-1,4-dimethoxyconduritol F (463) in 70% yield.
The synthesis of 2,3-diamino conduritol B (472) commenced from D-mannitol (319), which was transformed into 3,6-dihydroxy-4,5-di-O-isopropylidenethiepane (464). Treatment of (464) with sodium hydride and benzoyl bromide in THF afforded dibenzyl ether (465) in 96% yield. Acidic cleavage of the acetonide group in (465) furnished diol (466) in 95% yield, which upon treatment with methanesulfonyl chloride in pyridine gave dimesylate (467) in 92% yield. Reaction of (467) with sodium azide in DMSO produced diazide (468) in 85% yield, and subsequent oxidation with m-CPBA provided sulfone (469) in 98% yield.
Application of Ramberg–Bäcklund conditions to (469) delivered cyclohexene (470) in 61% yield. Reduction of (470) using triethylamine and 1,3-propanedithiol in methanol afforded diamine (471) in 68% yield. Final deprotection of the benzyl ethers in (471) with boron trichloride in dichloromethane furnished 2,3-diamino conduritol B (472) in 72% yield (Scheme 51).
 | ||
| Scheme 51 Synthesis of 2,3-diamino-1,4-dimethoxy conduritol F (463) and 2,3-diamino conduritol B (472) employing a Ramberg–Bäcklund reaction. | ||
While sulfone–olefin rearrangement strategies provide efficient access to diamino-substituted conduritol derivatives, conduritol-based intermediates have also been widely utilized in the synthesis of complex natural products. In this context, Elango and co-workers reported a concise conduritol A-derived pathway toward the total synthesis of the biologically significant alkaloid narciclasine.12
2.50. Conduritol A-derived pathway to the total synthesis of narciclasine
In 2002, Elango and co-workers reported a concise synthetic route to narciclasine (16).12 The sequence began with a Diels–Alder cycloaddition between the chiral chloronitroso compound (473) and diene (56), furnishing cycloadduct (58). Reduction of (58) with aluminum amalgam provided the partially protected conduritol A derivative (59) in ∼85% yield. Subsequent treatment of (59) with p-nitrobenzenesulfonyl chloride, followed by silylation of the unprotected hydroxyl group with TBSCl, afforded derivative (474) in ∼78% yield. Bromination of (474) with NBS in acetone produced a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of bromohydrins (475) and (476) in ∼98% combined yield.
1 mixture of bromohydrins (475) and (476) in ∼98% combined yield.
Alkylation of this mixture with benzyl bromide (477) in acetonitrile in the presence of potassium carbonate furnished epoxide (478) in ∼88% yield. Exposure of (478) to SnCl4 in dry dichloromethane promoted regioselective opening of the epoxide, and acetylation of the resulting hydroxyl group furnished acetate (479) in ∼98% yield. Treatment of acetate (479) with two equivalents of mercaptoacetic acid in the presence of LiOH (5 equiv.) in DMF at room temperature delivered amine (480) in ∼78% yield. Conversion of (480) to imide (481) was achieved by sequential Boc protection and oxidative cleavage using RuCl3/NaIO4, affording (481) in ∼67% yield.
Heating imide (481) with DBU in benzene induced a syn-elimination to provide compound (482) in ∼97% yield. Finally, global deprotection of (482) furnished Narciclasine (16) in ∼65% yield (Scheme 52).
 | ||
| Scheme 52 Conduritol A-derived total synthesis of narciclasine (16) via a Diels–Alder-based strategy. | ||
While conduritol-derived intermediates have proven valuable in the synthesis of biologically significant alkaloids such as narciclasine, related cyclitol-based frameworks have also been employed for the preparation of other Amaryllidaceae alkaloid analogues. In this regard, Rinner and co-workers reported an epoxyconduramine-driven strategy for constructing epi-7-deoxypancrastatin.71
2.51. Epoxyconduramine-driven construction of epi-7-deoxypancrastatin
In 2002, Rinner and co-workers reported the synthesis of epi-7-deoxypancrastatin (493).71 The sequence began with the conversion of bromobenzene (308) into aziridine (483), which was subsequently subjected to RuCl3/NaIO4-mediated syn-dihydroxylation to afford diol (484) in ∼85% yield. Treatment of (484) with sulfuryl chloride and triethylamine generated the corresponding cyclic sulfate (485) in ∼93% yield. Sequential reaction of (485) with BzONH4 in DMF, followed by hydrolysis, delivered benzoate (486) in ∼90% yield.Protection of the free hydroxyl group in (486) with TBSCl/imidazole, followed by methanolysis of the benzoate using sodium methoxide in THF, furnished compound (487) in ∼63% combined yield (two steps). Treatment of (487) with t-BuLi in THF, followed by coupling with piperonyl bromide in the presence of tetrabutylammonium iodide, afforded the epoxyconduramine derivative (488) in ∼68% yield.
Lewis acid-mediated cyclization of (488) with Me2AlCl in dichloromethane furnished bicyclic intermediate (489) in ∼68% yield. Protection of the resulting free hydroxyl group as its MOM ether afforded compound (490) in ∼97% yield. Oxidation of (490) with RuCl3/NaIO4 delivered lactam (491) in ∼50% yield. Subsequent reduction of (491) using sodium/naphthalene furnished compound (492) in ∼75% yield. Finally, global deprotection of (492) yielded epi-7-deoxypancrastatin (493) in ∼68% yield (Scheme 53).
While epoxyconduramine intermediates have proven useful for the synthesis of Amaryllidaceae alkaloid analogues such as epi-7-deoxypancratistatin, conduritol derivatives have also found application as versatile chiral building blocks for the construction of complex marine natural products. In this context, Lambert and co-workers employed a conduritol E-derived scaffold for the synthesis of the C1–C14 fragment of the potent anticancer agent Halichondrin B.72
2.52. Construction of the C1–C14 halichondrin B subunit via a conduritol E-derived scaffold
In 2003, Lambert and co-workers reported the synthesis of the C1–C14 subunit of halichondrin B, beginning from (+)-conduritol E (+)-494.72 Protection of (+)-494 with TBDPSCl in pyridine/DMF afforded diol (495) in ∼83% yield. Ozonolysis of (495) furnished the bicyclic intermediate (496), which underwent rapid dehydration upon treatment with acetic anhydride/Et3N to afford hemiacetal (497). Conversion of (497) into a single diastereomeric lactone (498) was accomplished using TESCl/imidazole in ∼80% yield. Methylation of (498) with dimethyltitanocene furnished compound (499) in ∼93% yield.Hydroboration–oxidation of (499) with BH3 Me2S, followed by oxidative workup with H2O2/NaOH, provided alcohol (500) in ∼70% yield. Protection of (500) as the corresponding pivalate, followed by acidic methanolysis, furnished hemiacetal (501) in ∼99% yield. A one-carbon Wittig homologation converted (501) into enol ether (502) in ∼87% yield. Subsequent dihydroxylation of (502) using OsO4/NMO delivered diol (503) in ∼73% yield.
Acetylation of diol (503) with Ac2O/DMAP/pyridine generated diacetate (504) in ∼68% yield. Lewis acid–promoted coupling of (504) with allylic silane (505) in the presence of BF3·OEt2 afforded coupling product (506) in ∼71% yield. Desilylation of (506) with TBAF/AcOH, followed by amberlyst IRA-400 treatment in methanol, provided triol (507) in ∼62% yield. Refluxing (507) with DBU in toluene delivered stereodefined diol (508) in ∼78% yield.
Further treatment of (508) with amberlyst IRA-400 in methanol furnished triol (509) in ∼93% yield. Sequential silylation with TESCl/imidazole and Swern oxidation afforded aldehyde (510) in ∼74% yield. Wittig olefination of (510) with phosphonium salt (511) and n-BuLi in THF produced enol ethers 512(Z) and 512(E) in ∼37% and ∼42% yields, respectively; the isomers were separated by Florisil chromatography.
Finally, acidic hydrolysis of the enol ether mixture with p-TsOH in CH2Cl2/H2O/MeOH furnished the C1–C14 subunit of Halichondrin B (513) in ∼67% overall yield from triol (509) (Scheme 54).
While conduritol derivatives have proven valuable as chiral building blocks in the synthesis of complex natural product fragments such as halichondrin B, their structural versatility has also inspired the development of annulated conduritol systems. These conformationally constrained derivatives provide useful platforms for accessing novel inositol analogues and exploring new stereochemical architectures.
2.53. Strategies for the construction of cyclohexa- and cyclopenta-annulated conduritols
In 2003, Mehta and co-workers reported versatile synthetic strategies for the preparation of annulated conduritols and conformationally constrained inositols.73 Their studies commenced with 1,2,3,4,5,8-hexahydronaphthalene (514), which underwent regioselective epoxidation with m-CPBA in dichloromethane to furnish epoxide (515) in ∼85% yield. Acid-catalyzed ring opening of (515), followed by acetylation, afforded the trans-diacetate (516) in ∼90% overall yield. Allylic bromination of (516), followed by DBU-mediated dehydrobromination, produced diene (517) in ∼52% overall yield. Stereoselective mono-epoxidation of (517) yielded a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 mixture of diastereomeric epoxides (518) and (519) in ∼73% combined yield.
9 mixture of diastereomeric epoxides (518) and (519) in ∼73% combined yield.
Ring opening of the major epoxide (519) with 10% aqueous acetic acid in THF afforded a mixture of annulated conduritol F (520) and annulated conduritol B (521) in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (∼80% yield). Catalytic dihydroxylation of (520) with OsO4/NMMO in acetone–water provided triol (522) in ∼88% yield, which upon basic hydrolysis furnished 2,3-cyclohexa-annulated chiro-inositol (523) in ∼96% yield. Similarly, hydrolysis of (521) generated annulated conduritol B derivative (524) in ∼95% yield; subsequent dihydroxylation of (524) afforded 3,4-cyclohexa-annulated myo-inositol (525) in ∼80% yield.
1 ratio (∼80% yield). Catalytic dihydroxylation of (520) with OsO4/NMMO in acetone–water provided triol (522) in ∼88% yield, which upon basic hydrolysis furnished 2,3-cyclohexa-annulated chiro-inositol (523) in ∼96% yield. Similarly, hydrolysis of (521) generated annulated conduritol B derivative (524) in ∼95% yield; subsequent dihydroxylation of (524) afforded 3,4-cyclohexa-annulated myo-inositol (525) in ∼80% yield.
Catalytic hydroxylation of diene (517) produced a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of diols (526) and (527) in ∼70% yield, which upon basic hydrolysis delivered 3,4-cyclohexa-annulated chiro-inositol (528) in ∼90% yield.
1 mixture of diols (526) and (527) in ∼70% yield, which upon basic hydrolysis delivered 3,4-cyclohexa-annulated chiro-inositol (528) in ∼90% yield.
For cyclopenta-annulated derivatives, 2,3,4,7-tetrahydro-1H-indene (529) was treated with MMPP in THF/water to provide the trans-diol (530) in ∼85% yield, followed by ring opening in 10% aqueous acetic acid (∼90% yield). Bromination and subsequent acetylation of (530) afforded dibromide (531) in ∼66% and ∼89% yields, respectively. Double dehydrobromination of (531) using DBU/DMSO yielded diene (532) in ∼54% yield. Epoxidation of (532) with m-CPBA produced a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 mixture of epoxides (533) and (534) in ∼60% combined yield. Ring opening of (534) with 10% acetic acid/THF afforded annulated conduritol F (535) and conduritol B (536) derivatives in a 3
2 mixture of epoxides (533) and (534) in ∼60% combined yield. Ring opening of (534) with 10% acetic acid/THF afforded annulated conduritol F (535) and conduritol B (536) derivatives in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (∼60% yield).
1 ratio (∼60% yield).
Basic hydrolysis of (535) gave annulated conduritol F (537) in ∼81% yield, which upon stereospecific dihydroxylation provided 2,3-cyclopenta-annulated chiro-inositol (538) in ∼84% yield. Likewise, hydrolysis of (536) furnished cyclopenta-annulated myo-inositol (540) in ∼92% and ∼86% yields over two steps. Treatment of epoxide (533) with 10% acetic acid/THF generated allylic alcohol (541) in ∼74% yield; basic hydrolysis delivered conduritol E derivative (542) in ∼95% yield. Final dihydroxylation of (542) using OsO4/NMMO furnished annulated allo-inositol (543) in ∼83% yield (Scheme 55).
Beyond the synthesis of annulated conduritol frameworks, the development of conformationally restricted cyclitol derivatives has attracted considerable interest due to their potential applications in probing stereochemical effects in cyclitol chemistry. In this context, Mehta and co-workers further explored strategies for the construction of cyclohexa-annulated derivatives of conduritol C and neo-inositol.74
2.54. Construction of conformationally restricted cyclohexa-annulated conduritol C and neo-inositol
In 2003, Mehta and colleagues reported the synthesis of cyclohexa-annulated, conformationally constrained derivatives of conduritol C and neo-inositol.74 Their study began with the dihydroxylation of tetrahydronaphthalene (544) using OsO4/NMMO, which furnished a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 mixture of syn-diols (545) and (546). The major diol (546) was converted to the corresponding diacetate (547), which upon epoxidation with m-CPBA in dichloromethane afforded a 1
8 mixture of syn-diols (545) and (546). The major diol (546) was converted to the corresponding diacetate (547), which upon epoxidation with m-CPBA in dichloromethane afforded a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 mixture of epoxides (548) and (549) in ∼86% overall yield.
10 mixture of epoxides (548) and (549) in ∼86% overall yield.
Ring opening of the major epoxide (549) with 10% aqueous acetic acid yielded diol (550) in ∼85% yield. Bromination of (550) furnished dibromide (551) in ∼77% yield. Treatment of (551) with potassium tert-butoxide induced regio- and stereoselective transetherification coupled with dehydrobromination, affording bicyclic ether (552) in ∼45% yield. Subsequent dihydroxylation followed by acetylation provided diacetate (553) in ∼75% yield over two steps.
Refluxing diacetate (553) with tetrabutylammonium iodide generated iodide (554) in ∼73% yield. Treatment of (554) with potassium tert-butoxide afforded the partially protected, cyclohexa-annulated, conformationally locked conduritol C derivative (555) in ∼63% yield. Dihydroxylation of (555) with OsO4 furnished triol (556) in ∼60% yield, and final hydrolysis of the acetate groups delivered the cyclohexa-annulated, conformationally constrained neo-inositol derivative (557) in ∼67% yield (Scheme 56).
While the construction of annulated conduritol derivatives provides access to conformationally constrained cyclitol architectures, alternative synthetic strategies have focused on introducing halogen substituents and polyacetylated functionalities into conduritol frameworks. Such modifications expand the structural diversity of conduritols and provide valuable intermediates for further synthetic transformations.
2.55. Advances in the synthesis of halogenated and polyacetylated conduritol frameworks
In 2003, Baran and co-workers reported the synthesis of a series of haloconduritols and conduritol tetraacetates.75 The study commenced with a Diels–Alder cycloaddition between furan (558) and vinylene carbonate (559), generating a mixture of the exo-adduct (560) and endo-adduct (561) in ∼34% and ∼8% yields, respectively. Sequential hydrolysis and acetylation of these adducts afforded the corresponding diacetates (562) and (563), in ∼8% and ∼64% yields, respectively.Treatment of endo-diacetate (563) with BCl3 at −78 °C furnished chloroconduritol A diacetate (565) in ∼96% yield, while reaction with BBr3 under analogous conditions produced bromoconduritol A diacetate (566) in ∼94% yield. Subsequent acetylation of the free hydroxyl group in compounds (565) and (566) using acetyl chloride in dichloromethane provided the corresponding triacetates (567) and (568) in nearly quantitative yields. Global deprotection of these triacetates then yielded chloroconduritol A (569) and bromoconduritol A (570), each obtained in almost quantitative yield.
In addition, treatment of endo-diacetate (563) with acetic anhydride and sulfuric acid generated a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of conduritol A tetraacetate (564) and conduritol F tetraacetate (230) in nearly quantitative yield. Conduritol A tetraacetate (564) could also be obtained via halide displacement in compounds (565) and (566) using acetate, affording the tetraacetate in ∼78% and ∼73% yields, respectively (Scheme 57).
1 mixture of conduritol A tetraacetate (564) and conduritol F tetraacetate (230) in nearly quantitative yield. Conduritol A tetraacetate (564) could also be obtained via halide displacement in compounds (565) and (566) using acetate, affording the tetraacetate in ∼78% and ∼73% yields, respectively (Scheme 57).
 | ||
| Scheme 57 Synthesis of halogenated conduritols and conduritol tetraacetates via a Diels–Alder-based strategy. | ||
Beyond halogenated and polyacetylated conduritol derivatives, further synthetic developments have explored oxidative transformations of conduritol frameworks for accessing various inositol stereoisomers and their biologically significant phosphate derivatives. Such strategies highlight the versatility of conduritols as intermediates in the synthesis of polyhydroxylated cyclitols and their phosphorylated analogues.
2.56. Oxidative transformations of conduritol derivatives toward inositols and their polyphosphate analogues
In 2003, Podeschwa and co-workers reported a series of synthetic methods leading to conduritol tetraacetates, inositols, and their corresponding hexakis(phosphate) derivatives.76 The synthesis began with anti-dibromide (+)-368, which was sequentially treated with sodium acetate in acetic acid and subsequently with an anhydride to furnish conduritol E tetraacetate (+)-571 in ∼70% yield (67% after recrystallization). Oxidative ring opening of (+)-571 with trifluoroacetic anhydride, hydrogen peroxide, dichloromethane, and sodium bicarbonate generated epoxide (+)-572 (24% yield), which underwent further oxidation to give 2,3,4,5-tetra-O-acetyl-neo-inositol (+)-573 in 71% yield. Acetylation of (+)-573 in pyridine produced neo-inositol hexaacetate (574) in 83% yield, and subsequent deprotection afforded neo-inositol (138) in ∼99% yield. Phosphorylation of neo-inositol (138) with 3-diethylamino-2,4,3-benzodioxaphosphepane (374) and 1H-tetrazole, followed by palladium-catalyzed hydrogenolysis, provided neo-inositol hexakis(phosphate) (575) in ∼99% yield.Similarly, dihydroxylation of conduritol E tetraacetate (+)-571 using RuCl3/NaIO4 afforded 3,4,5,6-tetra-O-acetyl-allo-inositol (+)-576 in ∼90% yield. Deacetylation produced allo-inositol (134) in ∼83% yield, which upon phosphorylation via the same sequence gave allo-inositol hexakis(phosphate) (577) in ∼99% yield.
For the synthesis of epi-inositol, dihydroxylation of (+)-368 with RuCl3/NaIO4 furnished syn-diol (−)-578 in ∼81% yield. Acetylation afforded tetraacetate (−)-579 (∼99%), which underwent zinc-mediated elimination to provide (−)-conduritol C tetraacetate (−)-580 in ∼68% yield. Subsequent dihydroxylation and acetylation yielded hexa-O-acetyl-epi-inositol (581) in ∼74% yield, and deprotection furnished epi-inositol (582) in ∼99% yield.
Diol (−)-578 was also converted into orthoester (−)-583 (∼93%) using triethyl orthoformate and p-toluenesulfonic acid, followed by acidic workup. Zinc-mediated elimination afforded partially protected conduritol C (−)-584 (∼82%), which, after phosphorylation and oxidation, generated triphosphate (+)-586 (∼50%). Hydrogenolysis and deacetylation then furnished D-epi-inositol-1,4,5-triphosphate (+)-587 in ∼99% yield.
The dibromide (+)-368 was further transformed into partially protected conduritol B (−)-588 (∼80%), which upon treatment with trifluoroacetic anhydride and hydrogen peroxide afforded epoxide (−)-589 (∼71%). Acidic ring opening of (−)-589 with H2SO4 in dioxane–water yielded anti-diol (−)-590 (∼80%), which underwent hydrogenolysis to provide L-chiro-inositol (−)-137 in ∼99% yield. Phosphorylation of L-chiro-inositol (−)-137 furnished L-chiro-inositol hexakis(phosphate) (−)-591 in ∼60% yield.
Dihydroxylation of (−)-588 afforded 1,4-di-O-benzyl-myo-inositol (−)-592 in ∼82% yield. Protection of the free hydroxyl groups of epoxide (−)-589 using 2,2-dimethoxypropane and PPTS gave the corresponding acetonide (593) in ∼96% yield. Epoxide opening with an allylic alcohol, followed by acetonide removal, produced partially protected scyllo-inositol (594) in ∼60% yield. Complete deprotection yielded scyllo-inositol (595) in ∼60% yield, which upon phosphorylation afforded L-scyllo-inositol hexakis(phosphate) (596) in ∼60% yield (Scheme 58).
In addition to oxidative transformations that convert conduritol frameworks into inositol derivatives, further synthetic efforts have focused on the introduction of nitrogen-containing functionalities. Such modifications enable access to azido- and amino-substituted conduritol and inositol derivatives, which are valuable intermediates for the synthesis of biologically relevant aminocyclitols and phosphorylated inositol analogues.
2.57. Stereocontrolled synthesis of azido- and amino-functionalized conduritol and inositol derivatives
In 2003, Podeschwa and co-workers reported the synthesis of both enantiomers of 1-azidoconduritol B and a series of amino-inositol phosphates.77 The study began with the treatment of diacetate (+)-368 and diol (+)-369 with lithium hydroxide in diethyl ether/methanol, generating the corresponding epoxides (+)-597 and (−)-597 in ∼99% yield. Nucleophilic ring opening of these epoxides with sodium azide afforded azides (−)-598 and (+)-598 in ∼90% yield. Subsequent treatment with lithium hydroxide followed by p-toluenesulfonic acid provided the 1-azidoconduritol B derivatives (−)-600 and (+)-600 in ∼47% yield.Acetylation of the triols (−)-600 and (+)-600 with acetic anhydride/pyridine furnished the 2,3,4-tri-O-acetyl derivatives (−)-601 and (+)-601 in ∼96% yield. Dihydroxylation of (+)-601 with RuCl3/NaIO4 delivered a 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 mixture of 3,4,5-tri-O-acetyl-6-azido-myo-inositol (−)-602 and 4,5,6-tri-O-acetyl-3-azido-myo-inositol (+)-603 in ∼91% overall yield. Acetylation of (−)-602 afforded pentaacetate (+)-604 (∼98%), and deprotection furnished 1-deoxy-1-azido-myo-inositol (−)-605 in ∼99% yield.
6 mixture of 3,4,5-tri-O-acetyl-6-azido-myo-inositol (−)-602 and 4,5,6-tri-O-acetyl-3-azido-myo-inositol (+)-603 in ∼91% overall yield. Acetylation of (−)-602 afforded pentaacetate (+)-604 (∼98%), and deprotection furnished 1-deoxy-1-azido-myo-inositol (−)-605 in ∼99% yield.
Hydrogenation of (−)-605 produced 6-deoxy-6-amino-myo-inositol (+)-606 in ∼99% yield. Sequential hydrogenation (80%), acetylation (99%), acetate deprotection (80%), phosphorylation (43%), and hydrogenolysis (80%) of (+)-604 yielded 6-deoxy-6-acetamido-myo-inositol-1,2,3,4,5-pentakisphosphate (−)-607. Similarly, phosphorylation (51%) and hydrogenolysis (90%) of (−)-605 afforded 6-deoxy-6-amino-myo-inositol-1,2,3,4,5-pentakisphosphate (−)-608.
For the synthesis of 3-deoxy-3-azido-myo-inositol, acetylation of (+)-603 produced pentaacetate (−)-609 (∼98%), which upon deprotection furnished 3-deoxy-3-azido-myo-inositol (+)-610 (∼99%). Palladium-catalyzed hydrogenation afforded 3-deoxy-3-amino-myo-inositol (+)-611 [(+)-903] in ∼99% yield. Subsequent transformations delivered 3-deoxy-3-acetamido-myo-inositol-1,2,4,5,6-pentakisphosphate (+)-612 (∼80 → 99% overall) and 3-deoxy-3-amino-myo-inositol-1,2,4,5,6-pentakisphosphate (+)-613 (∼51 → 90% overall).
Podeschwa et al. also described routes to diazido conduritol B, diazido-myo-inositol, and the corresponding tetraphosphate.77 Treatment of (+)-369 with lithium hydroxide followed by epoxide formation and sodium azide-mediated ring opening yielded diazido conduritol B (+)-614 (∼51%). Acetylation produced diacetate (+)-615 (∼99%). Dihydroxylation of (+)-615 with RuCl3/NaIO4 afforded 4,5-di-O-acetyl-3,6-diazido-myo-inositol (−)-616 (∼99%), and deacetylation furnished 3,6-dideoxy-3,6-diazido-myo-inositol (−)-617 (∼99%). Phosphitylation using 3-diethylamino-2,4,3-benzodioxaphosphepane (374) and 1H-tetrazole gave (−)-618 (∼45%), which upon treatment with TMSBr in CH2Cl2 produced 3,6-dideoxy-3,6-diazido-1,2,4,5-myo-inositol tetrakisphosphate (−)-619 in ∼90% yield (Scheme 59).
Beyond azido- and amino-functionalized conduritol derivatives, further synthetic developments have explored the preparation of higher-order inositol polyphosphates from conduritol precursors. These strategies highlight the versatility of conduritol frameworks as intermediates for accessing structurally diverse phosphorylated cyclitols with important biological functions.
2.58. Diversified synthesis of higher-order scyllo-inositol polyphosphates from a conduritol B precursor
In 2003, Know and co-workers reported the synthesis of several scyllo-inositol polyphosphates using conduritol B acetonide as the key starting material.78 The sequence began with conduritol B acetonide (+)-620, which was treated with benzyl bromide and sodium hydride to convert the free hydroxyl groups into benzyl ethers, affording compound (+)-621 in ∼96% yield. Dihydroxylation of (+)-621 with osmium tetroxide and NMO furnished the partially protected myo-inositol derivative (−)-622 in ∼94.4% yield.The partially protected inositol (−)-622 was subjected to a Mitsunobu inversion using benzoic acid, triphenylphosphine, and DEAD to give benzoate (−)-623 in ∼79% yield. Deprotection of the benzoate ester with sodium methoxide in methanol provided the partially protected scyllo-inositol derivative (−)-624 in ∼96% yield. Phosphorylation of (−)-624 with a phosphoramidite reagent followed by oxidation with m-CPBA afforded phosphate ester (−)-625 in ∼75% yield. Global deprotection via palladium-catalyzed hydrogenolysis and alkaline hydrolysis delivered D-scyllo-inositol-(1,2)-diphosphate (−)-626 in ∼94% yield.
Heating benzoate (−)-623 in 80% aqueous acetic acid produced partially protected scyllo-inositol (−)-627 in nearly quantitative yield. Phosphorylation with a phosphoramidite reagent and oxidation with m-CPBA furnished compound (−)-628 in ∼96% yield. Subsequent palladium-catalyzed hydrogenolysis followed by complete deprotection afforded scyllo-inositol-(1,2,4)-triphosphate (629) in ∼91% yield.
Further derivatization of (−)-623 by benzoylation of the remaining hydroxyl group provided (−)-630 in ∼99% yield. Palladium-catalyzed hydrogenolysis of benzyl ethers followed by acidic cleavage of the acetonide group yielded tetraol (+)-631 in ∼96% yield. Phosphorylation and oxidation of (+)-631 produced phosphate ester (+)-632 in ∼77% yield. Final palladium-catalyzed hydrogenolysis and benzoate deprotection delivered scyllo-inositol-(1,2,3,4)-tetraphosphate (633) in ∼89% yield (Scheme 60).
 | ||
| Scheme 60 Divergent synthesis of scyllo-inositol polyphosphates from a conduritol B-derived precursor. | ||
While conduritol derivatives have proven valuable intermediates for accessing inositol polyphosphates, their synthetic versatility has also enabled the preparation of structurally modified conduritol analogues. In particular, aryl-substituted deoxyconduritol derivatives have attracted considerable attention due to their role as key intermediates in the synthesis of biologically active natural products such as pancratistatins.
2.59. D-xylose-derived routes to aryl-substituted deoxyconduritol F analogues
In 2004, Nadein and co-workers reported an efficient synthetic route to 1-aryl-1-deoxyconduritol F derivatives, which serve as key precursors in the synthesis of pancratistatins.79 The sequence commenced with D-xylose (634), which was converted into its tribenzyl ether (347) in ∼71% yield.Tribenzyl ether (347) was then subjected to a sequence of olefination steps, beginning with a Wittig reaction using H2C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) PPh3 in THF, followed by Swern oxidation, and a second Wittig reaction employing PPh3
PPh3 in THF, followed by Swern oxidation, and a second Wittig reaction employing PPh3![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHCO2Me. This sequence afforded diene (635) in ∼84% overall yield. The diene (635) underwent 1,4-conjugate addition with various Gilman reagents to furnish the corresponding substituted intermediates (636a–d) in 95–97% yield.
CHCO2Me. This sequence afforded diene (635) in ∼84% overall yield. The diene (635) underwent 1,4-conjugate addition with various Gilman reagents to furnish the corresponding substituted intermediates (636a–d) in 95–97% yield.
Reduction of compounds (636a–d) with lithium aluminum hydride, followed by selenoxide elimination, provided allylic alcohols (637a–d). Finally, ring-closing metathesis (RCM) using Grubbs' catalyst converted these intermediates into the desired 1-aryl-1-deoxyconduritol F derivatives (638a–d) (Scheme 61).
 | ||
| Scheme 61 D-Xylose-derived synthesis of 1-aryl-1-deoxyconduritol F derivatives via a ring-closing metathesis strategy. | ||
While carbohydrate-derived strategies provide versatile access to aryl-substituted conduritol derivatives, alternative approaches have also employed chemoenzymatic transformations for constructing cyclitol frameworks. In particular, enzymatic dihydroxylation combined with chemical functionalization offers an efficient route to various inositol stereoisomers.
2.60. Synthesis of epi-inositol through chemoenzymatic dihydroxylation and radical debromination
In 2004, Vitelio and co-workers reported an efficient method for the synthesis of epi-inositol.80 The sequence began with the dihydroxylation of bromobenzene (308) using toluene dioxygenase to afford diol (74). This diol was subjected to a second dihydroxylation with osmium tetroxide/NMO, and the resulting tetraol was protected as its diacetonide using 2,2-dimethoxypropane and p-toluenesulfonic acid, yielding compound (639) in ∼75% yield.Epoxidation of diacetonide (639) with m-CPBA furnished epoxide (640) in ∼70% yield. Radical debromination of (640) provided the dehalogenated epoxide (641) in ∼85% yield. Finally, global deprotection of the acetonide groups followed by epoxide ring opening delivered epi-inositol (582) in ∼90% yield (Scheme 62).
 | ||
| Scheme 62 Chemoenzymatic synthesis of epi-inositol (582) via dihydroxylation and radical debromination. | ||
While chemoenzymatic dihydroxylation strategies have proven effective for constructing inositol stereoisomers, further developments have explored structural modifications of the conduritol framework through aromatic ring substitution. Such approaches enable access to polyhydroxylated tetrahydronaphthalene derivatives with expanded structural diversity and potential biological relevance.
2.61. Aromatic extensions of the conduritol scaffold: synthesis of 7-substituted polyhydroxylated tetrahydronaphthalenes
In 2004, Orsini and co-workers developed a method for synthesizing aromatic ring-substituted conduritol derivatives.81 The sequence began with 7-substituted naphthalene derivatives (642a–f), which were converted into the corresponding 7-substituted (1R,2S)-1,2-dihydroxy-1,2-dihydronaphthalenes (643a–f) via biotransformation using E. coli JM109 engineered to express naphthalene dioxygenase from Pseudomonas fluorescens N3. Protection of these diols with dimethoxypropane (DMP) in the presence of p-toluenesulfonic acid furnished the isopropylidene derivatives (644a–f). Epoxidation of (644a–f) with m-CPBA afforded the corresponding epoxides (645a–f).Heating epoxides (645a–d) with sodium azide in DMF provided azides (646a–d). Catalytic hydrogenation of azides (646a) and (646d) over Pd/C gave the amines (647a) and (647d), and subsequent acidic deprotection of their isopropylidene groups with methanolic HCl furnished the 7-substituted 1,2,3-trihydroxy-4-amino-tetrahydronaphthalenes (648a) and (648d).
To access 7-substituted 1,2,3,4-tetrahydroxy-tetrahydronaphthalenes and additional 1,2,3-trihydroxy-4-amino derivatives, the isopropylidene diols (644a,b,d,e) were subjected to dihydroxylation using β-AD-mix in t-butanol, affording diols (649a,b,d,e). Reaction of diols (649a,b,e) with triphosgene in pyridine/CH2Cl2 generated chloroformate intermediates (650a,b,e), which were converted into azides and subsequently reduced over Pd/C to give amines (647a) and (647e). Deprotection of these amines with methanolic HCl provided the corresponding 7-substituted 1,2,3-trihydroxy-4-amino-tetrahydronaphthalenes (648a) and (648e). Similarly, acidic removal of the isopropylidene group from diols (649a,d,e) furnished the 7-substituted 1,2,3,4-tetrahydroxy-tetrahydronaphthalenes (651a,d,e) (Scheme 63).
 | ||
| Scheme 63 Synthesis of 7-substituted 1,2,3-trihydroxy-4-amino-tetrahydronaphthalenes and 7-substituted 1,2,3,4-tetrahydroxy-tetrahydronaphthalenes. | ||
While aromatic substitutions provide access to structurally diverse conduritol-derived scaffolds, another major direction in cyclitol chemistry involves the synthesis of highly phosphorylated inositol derivatives. Such compounds play important roles in cellular signaling pathways, motivating the development of efficient synthetic routes to various inositol polyphosphates.
2.62. Divergent construction of D- and L-myo-inositol polyphosphates via conduritol B intermediates
In 2004, Saito and co-workers reported a synthetic route for D-myo-inositol-3,4,5,6-tetrakisphosphate (D-664) and L-myo-inositol-3,4,5,6-tetrakisphosphate (L-674) starting from a D-glucose derivative.82 D-Glucose was first converted into compound (652), which was then transformed into aldehyde (653) in approximately 67% yield. A 1,2-addition of vinylcopper to aldehyde (653) afforded allyl alcohol (654) with around 75% yield. Protection of the allylic hydroxyl group as 4-methoxybenzoyl (PMB) or 2-methoxybenzoyl (OMB) ethers yielded compounds (655a) and (655b) in about 98% yield each. Hydrolysis of the isopropylidene acetal with sulfuric acid in THF produced hemiacetals (656a) (50% yield) and (656b) (78% yield). Wittig methylenation of these hemiacetals furnished 1,7-dienes (657a) (70% yield) and (657b) (75% yield), which underwent ring-closing metathesis (RCM) using Grubbs' catalyst to give partially protected conduritol B derivatives (658a) (87% yield) and (658b) (84% yield).For the D-myo-inositol pathway, the free hydroxyl groups of (658a) were converted into PMB ethers using PMBCl and sodium hydride, yielding compound (659) almost quantitatively. Dihydroxylation of (659) with osmium tetroxide, NMO, and quinuclidine afforded partially protected myo-inositol (660) in 80% yield. Benzylation of the free hydroxyls gave compound (661) (93% yield), and PMB deprotection with DDQ yielded tetraol (−)-662 in 68% yield. Sequential phosphorylation using (BnO)2PNEt2 and oxidation with m-CPBA produced (+)-663 (78% yield), which upon palladium-catalyzed hydrogenolysis gave D-myo-inositol-3,4,5,6-tetrakisphosphate (D-664) in nearly quantitative yield.
For the L-myo-inositol pathway, diol (658b) was converted to benzoate (665) (72% yield) and reacted with Cl3CC(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NH)O–PMB to yield compound (666) (57% yield). Dihydroxylation of (666) produced diol (667) in 86% yield, which was protected as a THP ether (668) (72% yield). Hydrolysis of the benzoate gave diol (669) (75% yield), followed by benzylation of the free hydroxyls to give (670) (71% yield). THP deprotection produced compound (671) quantitatively, and removal of PMB/OMB groups with DDQ yielded 1,2-O-dibenzyl-D-myo-inositol (+)-672 in 72% yield. Sequential phosphorylation and oxidation afforded (−)-673 in 92% yield, and final palladium-catalyzed hydrogenolysis yielded L-myo-inositol-3,4,5,6-tetrakisphosphate (L-674) in 94% yield (Scheme 64).
NH)O–PMB to yield compound (666) (57% yield). Dihydroxylation of (666) produced diol (667) in 86% yield, which was protected as a THP ether (668) (72% yield). Hydrolysis of the benzoate gave diol (669) (75% yield), followed by benzylation of the free hydroxyls to give (670) (71% yield). THP deprotection produced compound (671) quantitatively, and removal of PMB/OMB groups with DDQ yielded 1,2-O-dibenzyl-D-myo-inositol (+)-672 in 72% yield. Sequential phosphorylation and oxidation afforded (−)-673 in 92% yield, and final palladium-catalyzed hydrogenolysis yielded L-myo-inositol-3,4,5,6-tetrakisphosphate (L-674) in 94% yield (Scheme 64).
 | ||
| Scheme 64 Synthesis of D-myo-inositol-3,4,5,6-tetrakisphosphate (D-664) and L-myo-inositol-3,4,5,6-tetrakisphosphate (L-674). | ||
In 2004, Freeman et al. reported a synthetic route for conduritol F oligomers and muco-inositol oligomers.83 The synthesis began with diol (74), which was converted into epoxide (27) via protection with 2,2-dimethoxypropane and acetone in the presence of p-TsOH, followed by epoxidation using m-CPBA in dichloromethane.
Treatment of epoxide (27) with a Lewis acid and either cinnamyl alcohol or benzyl alcohol produced compounds (675a) and (675b) in 73% and 75% yields, respectively. Coupling these intermediates with epoxide (27) under Lewis acid catalysis afforded ethers (676a) and (676b) in about 55% yield.
The conduritol F dimer (677) was obtained through sequential dehalogenation, deprotection of the cinnamyl group, and hydrolysis of ether (676a). In a related approach, epoxide (27) was hydrolyzed to diol (678), which coupled with epoxide (27) in the presence of a Lewis acid to form ether (679). Subsequent dehalogenation, deprotection, and hydrolysis yielded a second conduritol F dimer, (680).
Ether (676b) underwent electrochemical reduction to give compound (681) in approximately 77% yield. Treatment of (681) with oxone and trifluoroacetone in the presence of sodium bicarbonate in acetonitrile afforded epoxide (682) in about 40% yield. Hydrolysis of epoxide (682) using amberlyst A-27 resin produced compound (683) in 61% yield. Finally, complete deprotection of all protecting groups furnished muco-di-inositol (684) with an excellent yield of 97% (Scheme 65).
In addition to oligomeric cyclitols, conduritol intermediates have also been employed as versatile precursors for the synthesis of diverse inositol phosphate derivatives with potential biological activity.
2.63. Synthesis of inositol phosphate derivatives from sugar-derived conduritol B
In 2004, Andresen et al. reported a synthetic approach for phosphatidylinositol derivatives with potential anti-tumor activity, starting from partially protected conduritol B.84 The synthesis began with 6-iodo-6-deoxy-α-D-glucopyranoside (685), which underwent sequential benzylation of its hydroxyl groups. Sonication with zinc followed by reaction with vinyl magnesium bromide yielded a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 mixture of dienes (349) and (350) in about 59% overall yield. Ring-closing metathesis (RCM) then afforded tribenzyl conduritol F (351) (14%) and tribenzyl conduritol B (353) (69%), with a combined yield of approximately 83%.
5 mixture of dienes (349) and (350) in about 59% overall yield. Ring-closing metathesis (RCM) then afforded tribenzyl conduritol F (351) (14%) and tribenzyl conduritol B (353) (69%), with a combined yield of approximately 83%.
Oxidation of compound (351) with Dess-Martin periodinane followed by reduction with sodium borohydride and CeCl3·7H2O produced compound (353) in about 70% yield. Palladium-catalyzed hydrogenation of (353) yielded 1D-4,5,6-tri-O-benzyl-2,3-dideoxy-myo-inositol (686) nearly quantitatively. Epoxidation of (353) using m-CPBA generated partially protected conduritol B epoxide (687) in ∼91% yield, which, upon reaction with PMBCl and sodium hydride, gave compound (688) with ∼90% yield.
Opening the epoxide ring of (688) with sodium borohydride and a Lewis acid yielded compound (689) in ∼66% yield. Subsequent benzylation of the free hydroxyl group produced compound (690) (∼95% yield), and PMB deprotection using DDQ gave 1D-2,4,5,6-tetra-O-benzyl-3-deoxy-myo-inositol (691) (∼85% yield).
Separately, compound (353) was benzoylated with benzoyl chloride and triethylamine to form (692), then dihydroxylated with osmium tetroxide and NMO to give (693) (∼87% yield). Benzylation of the free hydroxyl groups afforded (694) (∼85% yield), and subsequent benzoate deprotection produced 1D-2,3,4,5,6-penta-O-benzyl-myo-inositol (695) (∼95% yield).
Finally, compounds (686), (691), and (695) were converted to the corresponding inositol amidites (696a), (696b), and (696c) by reaction with benzyl-N,N-diisopropylchlorophosphoramidite. Coupling with compound (697) in the presence of 5-phenyl-1H-tetrazole followed by oxidation with tert-butylhydrogen peroxide yielded phosphates (698a), (698b), and (698c). Palladium-catalyzed hydrogenolysis of these phosphates furnished the final inositol phosphate derivatives (699a), (699b), and (699c), with yields of approximately 70%, 67%, and 66%, respectively (Scheme 66).
Carbohydrate-derived approaches benefit from the inherent chirality of readily available sugars, allowing reliable stereochemical control throughout the synthesis. However, such strategies often require extensive protecting-group manipulations and multiple redox adjustments, which can increase the overall step count and reduce synthetic efficiency.
2.64. Synthesis of 3,6-dimethoxy derivatives of conduritols A–E: solution- and solid-phase strategies
In 2005, Vanda Cerè et al. presented both solution-phase and solid-phase strategies for the synthesis of 3,6-dimethoxy-substituted conduritols.85 In the solution-phase route, thiepane derivatives (+)-700, (+)-705, and (+)-710 were first methylated with methyl iodide and sodium hydride in THF to furnish the corresponding methyl ethers (+)-701, (+)-706, and (+)-711 in 96–97% yield. Subsequent oxidation with m-CPBA in dichloromethane afforded the sulfones (+)-702, (+)-707, and (+)-712 in 90–92% yield.Application of the Ramberg–Bäcklund reaction to these sulfones provided the cyclohexene intermediates (+)-703, (+)-708, and (+)-713 in 60–64% yield. Final deprotection of the isopropylidene protecting groups using TFA in acetonitrile delivered the desired 3,6-dimethoxy derivatives of conduritol A (+)-704, conduritol C (+)-709, and conduritol D (+)-714 in ∼93% yield.
In parallel, a solid-phase synthesis was developed to access 3,6-dimethoxy derivatives of conduritol B and E. Thiepanes (715) and (457) were immobilized onto a preswollen polystyrene–CHO resin under p-TSA catalysis, generating resin-bound intermediates (716) and (717) in ∼95% yield. Oxidation of these resin-bound thiepanes with m-CPBA produced sulfones (718) and (719) in ∼98% yield. Ramberg–Bäcklund cyclization under Meyers' modified conditions furnished the corresponding cyclohexenes (720) and (721), which upon acid-mediated resin cleavage yielded 3,6-dimethoxy conduritol B (255) and conduritol E (722). The solid-phase route offered streamlined purification and a modest improvement in overall yield compared with the solution-phase method (Scheme 67).
 | ||
| Scheme 67 Synthesis of 3,6-dimethoxy derivatives of conduritol A, conduritol C and conduritol D and solid phase preparation of 3,6-dimethoxy conduritol derivatives. | ||
Beyond structural modification of the conduritol scaffold, considerable attention has also been devoted to the development of nitrogen-containing cyclitol derivatives, particularly aminoinositols, owing to their significant biological relevance.
2.65. Divergent syntheses of aminoinositol derivatives from conduritol B epoxide
In 2005, Serrano et al. developed a comprehensive strategy for accessing a diverse array of aminoinositol derivatives from tetra-O-benzylconduritol B epoxide (388).86 Nucleophilic opening of epoxide (388) with sodium azide in the presence of LiClO4 in acetonitrile afforded azido alcohol (723) in 91% yield. Subsequent benzylation of the free hydroxyl group furnished derivative (724) in 95% yield. Reduction of the azides in (723) and (724) with LiAlH4 provided the tetrabenzyl and pentabenzyl scyllo-aminoinositol derivatives (726) and (725) in excellent yields (92–99%).Mesylation of the free hydroxyl group in (723) with mesyl chloride and triethylamine generated mesylate (727) in 88–92% yield. Heating (727) in DMF promoted stereochemical inversion to give alcohol (728) in 82% yield, and reduction of its azide group with LiAlH4 furnished the tetrabenzyl myo-aminoinositol derivative (729) in 87% yield. An alternative azidation route involved treating epoxide (388) with sodium azide and ammonium chloride in aqueous methanol, producing azide (730) in 91% yield; its mesylation provided mesylate (731) in 88–92% yield.
Mesylates (727) and (731) underwent intramolecular cyclization upon treatment with LiAlH4 to generate tetra-O-benzylconduritol B aziridine (732) in 90% yield. Nucleophilic ring opening of aziridine (732) with a mixture of sodium azide and ammonium azide (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 MeOH/H2O) afforded a 1
1 MeOH/H2O) afforded a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of tetrabenzyl chiro-2-azido-1-aminoinositol (733) and scyllo-2-azido-1-aminoinositol (734) in 82% overall yield. Reduction of the azide group in (730) also gave amino alcohol (735) in 92–99% yield. Boc protection of (735) afforded (736) in 78% yield; mesylation produced (737) (84%), and heating in DMF led to a transient oxazolidine intermediate (738). Hydrolysis of this intermediate delivered the tetrabenzyl myo-inositol derivative (739) in 65% yield.
1 mixture of tetrabenzyl chiro-2-azido-1-aminoinositol (733) and scyllo-2-azido-1-aminoinositol (734) in 82% overall yield. Reduction of the azide group in (730) also gave amino alcohol (735) in 92–99% yield. Boc protection of (735) afforded (736) in 78% yield; mesylation produced (737) (84%), and heating in DMF led to a transient oxazolidine intermediate (738). Hydrolysis of this intermediate delivered the tetrabenzyl myo-inositol derivative (739) in 65% yield.
Overall, this unified sequence from epoxide (388) enabled efficient and stereochemically controlled access to scyllo-, myo-, and chiro-configured aminoinositol derivatives through judicious selection of nucleophiles, protecting groups, and azide/mesylate manipulations (Scheme 68a).
 | ||
| Scheme 68 (a) Synthesis of aminoinositol derivatives from conduritol B epoxide. (b) Synthesis of diaminoinositol derivatives. | ||
In continuation of their 2005 study, Serrano et al. expanded the synthetic utility of conduritol B epoxide-derived intermediates by accessing a diverse library of N-acylated and diamino inositol derivatives.86
Collectively, these transformations highlight the versatility of conduritol B-derived intermediates for constructing structurally diverse N-functionalized aminoinositols, offering efficient access to scyllo-, myo-, and chiro-configured frameworks (Scheme 68b).
Epoxide- and azide-mediated transformations remain among the most versatile approaches for introducing nitrogen functionality into conduritol frameworks. These reactions offer reliable regio- and stereocontrol; however, their reliance on azides, strong reducing agents, or Mitsunobu-type reagents may pose safety and environmental considerations for large-scale applications.
Beyond the preparation of aminoinositol derivatives, conduritol intermediates have also been employed for the synthesis of related aminocyclitol natural products such as conduramines.
2.66. Efficient access to (−)-conduramine B-1 using reductive and Mitsunobu transformations
In 2005, Łysek et al. reported the synthesis of (−)-conduramine B-1 (−)-772.87 The route began with the reduction of compound (−)-766 using sodium borohydride in the presence of CeCl3·7H2O, yielding a 2.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of alcohols (−)-767 and (−)-768 with an overall yield of approximately 98%. The use of CeCl3 in combination with NaBH4 (Luche reduction conditions) enables selective reduction of carbonyl functionalities while minimizing competing side reactions.
1 mixture of alcohols (−)-767 and (−)-768 with an overall yield of approximately 98%. The use of CeCl3 in combination with NaBH4 (Luche reduction conditions) enables selective reduction of carbonyl functionalities while minimizing competing side reactions.
This mixture was then subjected to a Mitsunobu-type reaction with phthalimide, DEAD, and triphenylphosphine, giving a 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of N-substituted phthalimides (−)-769 and (−)-770 in 87% yield. The two compounds were separated by flash chromatography on silica gel. Deprotection of the silyl ether in compound (−)-769 using 1% toluenesulfonic acid afforded compound (−)-771. Treatment of (−)-771 with 40% aqueous methylamine, followed by purification through acidic Dowex-50W-X2 resin, yielded (−)-conduramine B-1 (−)-772 in nearly 95% yield.
1 mixture of N-substituted phthalimides (−)-769 and (−)-770 in 87% yield. The two compounds were separated by flash chromatography on silica gel. Deprotection of the silyl ether in compound (−)-769 using 1% toluenesulfonic acid afforded compound (−)-771. Treatment of (−)-771 with 40% aqueous methylamine, followed by purification through acidic Dowex-50W-X2 resin, yielded (−)-conduramine B-1 (−)-772 in nearly 95% yield.
This method provides a concise and high-yielding route to (−)-conduramine B-1 (−)-772 (Scheme 69).
In addition to the synthesis of naturally occurring conduramines, related studies have also explored the construction of more complex aminocyclitol frameworks containing bicyclic architectures.
2.67. Synthesis of a conduramine D-2 based bicyclic framework
In 2006, Kelebekli et al. reported a synthetic route to a bicyclic aminocyclitol derivative with a conduramine D-2 configuration.88 The synthesis began with cyclooctatetraene (190), which was treated with Hg(OAc)2 to give diene (773) in approximately 84% yield. This diene underwent photooxygenation with singlet oxygen in the presence of tetraphenylporphyrin, producing endo-peroxide (774) in around 70% yield. Such photooxygenation reactions provide an efficient strategy for introducing oxygen functionality into cyclic polyene systems.Reductive cleavage of the endo-peroxide using thiourea in methanol furnished diol (775) with nearly quantitative yield (∼99%). The diol was then converted into bis-carbamate (776) via reaction with two equivalents of toluenesulfonyl isocyanate. Cyclization using triisopropylphosphine in the presence of (dba)Pd2CHCl3 yielded oxazolidinone (777) in about 48% yield.
Subsequent dihydroxylation of (777) with KMnO4 in acetone/THF afforded syn-diol (778) in roughly 84% yield. Acetylation of the free hydroxyls using sodium acetate and acetic anhydride produced compound (779) in approximately 30% yield. Finally, acidic hydrolysis of (779) yielded the target aminocyclitol (780) with close to 84% yield (Scheme 70).
Alongside efforts directed toward bicyclic aminocyclitol frameworks, related studies have also focused on the stereocontrolled synthesis of conduramine derivatives from bicyclic precursors.
2.68. Stereocontrolled synthesis of (±)-conduramine F-1 from a bicyclic enone precursor
In 2006, Łysek et al. reported an efficient route to (±)-conduramine F-1.89 The synthesis commenced with (±)-7-oxabicyclo[2.2.1]hept-5-en-2-one (±)-781, which was converted to enone (±)-766. Reduction of this enone with diisobutylaluminium hydride furnished a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5.1 mixture of alcohols (±)-767 and (±)-768 in an excellent combined yield of approximately 98%. DIBAL-H enables selective reduction of the conjugated carbonyl functionality while preserving the bicyclic framework.
5.1 mixture of alcohols (±)-767 and (±)-768 in an excellent combined yield of approximately 98%. DIBAL-H enables selective reduction of the conjugated carbonyl functionality while preserving the bicyclic framework.
A Mitsunobu reaction on this mixture using phthalimide, triphenylphosphine, and DEAD generated a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 mixture of N-phthalimido derivatives (±)-769 and (±)-770 in 53% overall yield, and these were separated by chromatographic purification.
2.5 mixture of N-phthalimido derivatives (±)-769 and (±)-770 in 53% overall yield, and these were separated by chromatographic purification.
Hydrazinolysis of compound (±)-770 produced amine (±)-782 in 56% yield; however, direct desilylation at this stage resulted in impure (±)-conduramine F-1. In contrast, mild acid-mediated deprotection of (±)-770 using 1% p-toluenesulfonic acid in methanol afforded triol (±)-783 in 93% yield. Subsequent aminolysis with 40% aqueous methylamine efficiently delivered (±)-conduramine F-1 (±)-183 in 91% yield, corresponding to an overall yield of 85% from (±)-770 (Scheme 71).
In addition to purely synthetic strategies from bicyclic intermediates, chemoenzymatic approaches have also been developed for the preparation of conduramine derivatives.
2.69. Chemoenzymatic route to (−)-conduramine C-4 from bromobenzene
In 2007, Bellomo et al. reported an enantioselective synthesis of (−)-conduramine C-4 (−)-244 starting from bromobenzene (308).90 Enzymatic dihydroxylation of bromobenzene using Pseudomonas putida F39/D furnished bromo-cyclohexadienediol (74), which was subsequently protected as its acetonide (24) using 2,2-dimethoxypropane and p-toluenesulfonic acid in ∼85% yield.Oxidation of acetonide 24 with N-bromosuccinimide in aqueous THF generated a mixture of bromohydrins (309) and (784). Cyclization of this mixture under basic conditions (NH4BuHSO4) provided epoxides (310) and (27) in a 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio and an overall yield of ∼50%; the two epoxides were separated chromatographically.
1 ratio and an overall yield of ∼50%; the two epoxides were separated chromatographically.
The major epoxide (310) underwent nucleophilic ring opening with sodium azide to give azido alcohol (785) in nearly quantitative yield (∼98%). Staudinger reduction of the azide furnished the corresponding amine (786) in 74% yield. Radical debromination of (786) using Bu3SnH and ABCC afforded compound (787) in ∼75% yield. An alternative approach from azido alcohol (785) via LiAlH4 reduction also yielded (787), though with significantly lower efficiency (31%).
Final deprotection of the acetonide group in (787) using amberlite resin catalysis provided (−)-conduramine C-4 (−)-244 in nearly quantitative yield (Scheme 72).
Beyond chemoenzymatic approaches, alternative synthetic routes have also been developed for accessing conduramine derivatives and their epoxide analogues through azidoconduritol intermediates.
2.70. Synthesis of conduramine B-1 and conduramine F-1 epoxides via azidoconduritol intermediates
In 2007, Łysek et al. described the synthesis of conduramine F-1 epoxides starting from compound (±)-781.91 Reduction of this starting material produced a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5.1 mixture of alcohols (±)-767 and (±)-768. Treatment of this mixture with DPPA (diphenylphosphoryl azide) and DEAD in toluene yielded a 1
5.1 mixture of alcohols (±)-767 and (±)-768. Treatment of this mixture with DPPA (diphenylphosphoryl azide) and DEAD in toluene yielded a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 mixture of azides (±)-788 and (±)-789, with an overall yield of about 67%.
2.5 mixture of azides (±)-788 and (±)-789, with an overall yield of about 67%.
Deprotection of the silyl ethers in azide (±)-789 afforded azidoconduritol F-1 (±)-790 in 84% yield. Subsequent epoxidation using m-CPBA produced a mixture of epoxides (±)-791 (35%) and (±)-792 (16%). Palladium-catalyzed hydrogenation of these epoxides generated conduramine F-1 epoxides (±)-793 and (±)-794 in approximately 50% yield.
Reduction of azidoconduritol F-1 (±)-790 with triphenylphosphine followed by workup furnished conduramine F-1 (±)-183 in 78% yield. Similarly, deprotection of silyl ethers in (±)-788 gave azidoconduritol B-1 (±)-600 in 77% yield, which was reduced to conduramine B-1 (±)-772 in 73% yield.
Protection of the hydroxyl groups in epoxides (±)-791 and (±)-792 as silyl ethers yielded (±)-795 and (±)-796, both in 98% yield. Subsequent palladium-catalyzed reduction of the azides produced amines (±)-797 and (±)-798 in 81% and 74% yield, respectively. Finally, deprotection of the silyl ethers of these amines afforded conduramine F-1 epoxides (±)-793 and (±)-794 in 95% and 85% yields, respectively (Scheme 73).
 | ||
| Scheme 73 Synthesis of (±)-conduramine B-1 (±)-772 and (±)-conduramine F-1 epoxides (±)-793 and (±)-794. | ||
Beyond the preparation of conduramine derivatives and their epoxide analogues, conduritol-derived intermediates have also been employed in the total synthesis of biologically important natural products.
2.71. Synthesis of (−)-narciclasine using conduritol-derived bromoconduramine and aryl boronate partners
In 2008, Matveenko et al. reported a convergent strategy for the total synthesis of (−)-narciclasine (−)-16, beginning from compound (799).13 Nucleophilic substitution of (799) with sodium cyanide in the presence of manganese dioxide and diethylamine furnished diethylamide (800) in 58% yield. Directed metallation of (800) with sec-BuLi/TMEDA, followed by trapping with trimethyl borate and oxidative workup (H2O2/AcOH), afforded alcohol (801) in 90% yield. Protection of the hydroxyl group with TBSCl provided silyl ether (802) in 94% yield. Subsequent lithiation–iodination converted (802) into iodide (803) in 90% yield. Deprotection of the TBS ether furnished alcohol (804) in 62% yield, which was transformed into the corresponding MOM ether (805) in 99% yield. A Miyaura borylation step then delivered boronate (806), albeit in a modest 44% yield.In a parallel sequence, cis-diol (74) was converted into acetal (807) using p-MBDMA and p-TsOH. Dihydroxylation of (807) with OsO4/NMO gave diol (808) in 65% yield, which was fully protected as the MOM ether (809) in 75% yield. Reduction of (809) with DIBAL-H furnished bromoconduritol E derivative (810) in 84% yield. Conversion of the remaining free hydroxyl into a MOM ether gave (811) in 90% yield, and DDQ oxidation removed the PMB group to afford alcohol (812) in 95% yield. Formation of acetimidate (813) using trichloroacetonitrile/DBU, followed by microwave-assisted cyclization with K2CO3, generated amide (814) in 78% yield. DIBAL-H reduction of (814) provided bromoconduramine A (815) in 89% yield.
The key Suzuki–Miyaura cross-coupling between boronate (806) and bromoconduramine A (815) under microwave irradiation afforded biaryl intermediate (816) in 63% yield. Global deprotection of (816) then delivered the target natural product (−)-narciclasine (−)-16 in 48% yield (Scheme 74).
 | ||
| Scheme 74 Convergent synthesis of (−)-narciclasine (−)-16 via Suzuki–Miyaura coupling of a conduritol-derived bromoconduramine and an aryl boronate. | ||
In addition to their application in natural product synthesis, conduritol-derived intermediates have also been employed for the stereocontrolled preparation of diverse conduramine analogues.
2.72. Regio- and stereocontrolled construction of conduramine F-1 and dihydroconduramine E-1
In 2008, Pandey et al. described an efficient synthetic route to conduramine F-1 (183) and dihydroconduramine E-1 (826).92 Reduction of ketone (817) with lithium aluminium hydride generated a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 mixture of alcohols (818) and (819), which were separated by silica gel chromatography. Nucleophilic ring opening of the major isomer (819) with excess methylmagnesium bromide furnished compound (820) in 80% yield. Subsequent reduction with 6% sodium amalgam afforded diol (821) in 86% yield, and protection of the free hydroxyl group as a TBS ether produced (822) in 86% yield.
9 mixture of alcohols (818) and (819), which were separated by silica gel chromatography. Nucleophilic ring opening of the major isomer (819) with excess methylmagnesium bromide furnished compound (820) in 80% yield. Subsequent reduction with 6% sodium amalgam afforded diol (821) in 86% yield, and protection of the free hydroxyl group as a TBS ether produced (822) in 86% yield.
Allylic oxidation of (822) using t-BuOOH and Pd/C generated enone (823) in 75% yield, and epoxidation with t-BuOOH/Triton B delivered epoxide (824) in 85% yield. Conduramine F-1 (183) was accessed from epoxide (824) through a sequence involving KHMDS/Comins reagent-mediated functionalization, Pd(PPh3)4-catalyzed transformation, Et3SiH reduction, and a final one-pot epoxide ring opening accompanied by global deprotection using 0.2 N H2SO4 and 10 N HCl in dioxane.
For the synthesis of dihydroconduramine E-1 (826), diol (821) was subjected to OsO4/NMO-mediated dihydroxylation to afford triol (825) in 75% yield. Subsequent carbamate deprotection with 12 N HCl furnished dihydroconduramine E-1 (826) in nearly 90% yield (Scheme 75).
Beyond these stereocontrolled approaches, ring-closing metathesis (RCM) has emerged as a powerful strategy for constructing conduritol frameworks that serve as versatile intermediates for a variety of conduramine derivatives.
2.73. Divergent access to conduramine A-1, conduramine E, and valienamine derivatives from an RCM-enabled conduritol scaffold
In 2009, Chang et al. reported an efficient and versatile strategy for the synthesis of conduramine derivatives, with particular emphasis on valienamine scaffolds.93 The sequence commenced with L-tartaric acid (L-827), which was protected as acetonide (828) using 2,2-dimethoxypropane and p-toluenesulfonic acid in methanol (∼83% yield). Sequential reduction with DIBAL-H followed by a diastereoselective addition of divinylzinc furnished diene (293) in 83% yield. Ring-closing metathesis (RCM) of this diene using Grubbs' second-generation catalyst enabled cyclization to the partially protected conduritol E derivative (829) in 70% yield, which was subsequently transformed into epoxide (830) via triflimide activation and NaH-mediated cyclization in a DMF/EtOH/H2O mixture.Epoxide (830) underwent regioselective opening with NaN3 to afford azido alcohol (831) at room temperature (66% yield) or the isomeric azido alcohol (832) at 90 °C (70% yield). Notably, (831) rearranged to (832) upon heating to 90 °C. Reduction of (831), followed by isopropylidene deprotection and acetylation, delivered conduramine A-1 tetraacetate (22) in 79% yield. Alternatively, treatment of (831) with trifluoroacetic acid induced isopropylidene migration, and subsequent reaction with methyl chloroformate/NaH furnished intermediate (202), a known precursor to (+)-pancratistatin.
In parallel, reduction, deprotection, and acetylation of azido alcohol (832) afforded conduramine E tetraacetate (222) in nearly 90% yield. Oxidation of (832) with Dess–Martin periodinane provided ketone (833) in 96% yield. Condensation of (833) with formaldehyde in the presence of NaHCO3 and imidazole yielded alcohol precursor (834) in 65% yield. Luche reduction of (834) afforded alcohol (835) in 80% yield. Final reduction, acetonide cleavage, and acetylation furnished the tetraacetate derivative of (+)-valienamine (836) in 50% yield (Scheme 76).
The increasing use of ring-closing metathesis in conduritol chemistry highlights its value as a modular strategy for constructing cyclohexene frameworks from acyclic precursors. Compared with classical pericyclic or oxidative approaches, RCM offers greater flexibility in substrate design and enables divergent access to multiple conduritol derivatives from common diene intermediates.
Complementary to metathesis-based approaches, cycloaddition strategies have also been explored for the construction of aminocyclitol frameworks.
2.74. Copper-catalyzed nitroso-Diels–Alder strategy for the synthesis of conduramine A-1 tetraacetate
In 2009, Jana et al. reported a synthesis of conduramine A-1 tetraacetate (22).94 The process began with a nitroso-Diels–Alder reaction between compound (56) and 2-nitrosopyridine (837) using [CuPF6(MeCN)4] and the Walphos-CF3 ligand, yielding adduct (838) in nearly 99% yield. Reductive cleavage of the N–O bond with sodium borohydride in the presence of [Mo(CO)6], followed by silyl protection of the hydroxyl group with TBSCl and imidazole, produced compound (839) in about 91% yield.Subsequent reaction of (839) with methyl chloroformate and methyl magnesium chloride afforded compound (840) in approximately 96% yield. Deprotection of the pyridyl group and TBS ether yielded compound (841) with around 82% yield. Final cleavage of all protecting groups followed by acetylation furnished conduramine A-1 tetraacetate (22) in roughly 57% yield (Scheme 77).
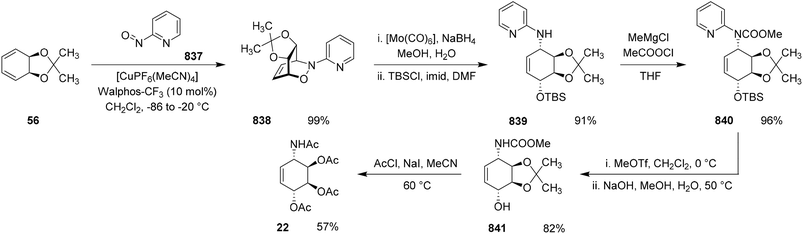 | ||
| Scheme 77 Synthesis of conduramine A-1 tetraacetate (22) using copper-catalyzed nitroso-Diels–Alder strategy. | ||
In addition to cycloaddition-based approaches, chemoenzymatic strategies combined with selenium-mediated transformations have also been explored for the preparation of functionalized conduritol derivatives.
2.75. Synthesis of deoxy-selenylconduritols and a partially protected conduritol C derivative
In 2009, Bellomo et al. developed a concise synthetic strategy for accessing deoxy-selenylconduritols and a partially protected conduritol C derivative.95 The sequence commenced with the enzymatic dihydroxylation of bromobenzene (308) using Pseudomonas putida F39/D, affording cis-diol (74). Protection of the diol as its isopropylidene acetal proceeded in nearly quantitative yield (∼98%), and subsequent epoxidation with m-CPBA furnished epoxide (27) in 85% yield.Radical debromination of epoxide (27) provided bromine-free intermediate (164) in ∼70% yield. Nucleophilic ring opening of (164) with diphenyl diselenide (DPDS) in the presence of NaBH4 afforded the selenylated intermediate (842) in ∼72% yield. Acidic resin-catalyzed hydrolysis of the acetonide protecting group in (842) gave 6-phenylselenylconduritol F derivatives (843) in 77% yield.
In an alternative transformation, compound (842) was subjected to oxidative rearrangement using RuCl3–NaIO4, generating partially protected conduritol C derivative (844) in ≈30% yield—an intermediate of utility for downstream syntheses of conduritol C. Moreover, direct treatment of epoxide (27) with DPDS/NaBH4 led to regioselective ring opening to give compound (845) in ≈89% yield, and deprotection of its acetonide group under acidic conditions delivered 4-bromo-6-phenylselenylconduritol F derivatives (846) in ≈70% yield (Scheme 78).
Halogenated conduritol derivatives have also attracted considerable interest because they serve as useful intermediates for further functionalization and biological investigations.
2.76. Divergent strategies for the synthesis of bromo-conduritol B and C
In 2009, Cantekin et al. reported efficient synthetic routes to both bromo-conduritol B and bromo-conduritol C.96 | ||
| Scheme 79 Synthesis of bromo-conduritol B and bromo-conduritol C via photooxygenation and subsequent functional group transformations. | ||
Beyond halogenated conduritol derivatives, selective dihydroxylation strategies have also been explored for constructing key intermediates in conduramine synthesis.
2.77. Construction of a conduramine E precursor via selective dihydroxylation
In 2010, Chappell et al. developed an efficient route to a key precursor of conduramine E.97 Initial bromination of compound (863) using polymer-supported bromine (Br3−) afforded intermediate (864). Sequential treatment of (864) with methanolic HCl and then triphosgene in the presence of pyridine generated urethane (865) in an overall 86% yield from (863).Elimination of urethane (865) with DBU provided diene (866) in 90% yield. Dihydroxylation of (866) furnished diols (867) and (868). Use of AD-mix-β in the presence of MeSO2NH2 enabled highly selective formation of diol (867) as the sole product (100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 ratio) in 76% overall yield, whereas dihydroxylation with K2OsO4·2H2O delivered a 4
0 ratio) in 76% overall yield, whereas dihydroxylation with K2OsO4·2H2O delivered a 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of (867) and (868) in 55% yield.
1 mixture of (867) and (868) in 55% yield.
Finally, refluxing diol (867) in trifluoroacetic acid provided compound (869) in 76% yield. This intermediate serves as a valuable building block for the synthesis of (−)-conduramine E (Scheme 80).
Beyond dihydroxylation-based approaches, chiral auxiliary-mediated strategies have also been developed to access various conduramine derivatives with high stereochemical control.
2.78. Synthesis of conduramine A-1, E-1, and F-1 via chiral auxiliary-mediated transformations
In 2010, Lu et al. reported synthetic routes for conduramine A-1, conduramine E-1, and conduramine F-1.98Compound (882) was converted to azide (883) (94% yield) using TfN3 and CuSO4. Ketal hydrolysis with TFA produced intermediate (884), which underwent a [3,3]-sigmatropic rearrangement to yield compound (885) (95% yield). Luche reduction afforded a mixture of alcohols (790) and (886). Reaction with 2,2-dimethoxypropane and PTSA gave acetonide (219) (33% yield) and unreacted (790) (53%), which were isolated.
Staudinger reaction of acetonide (219) yielded amine (220) (94% yield), and subsequent deprotection produced (+)-conduramine E-1 (+)-221 (91% yield). Similarly, treatment of (790) under Staudinger conditions furnished (+)-conduramine F-1 (+)-183 with approximately 90% yield (Scheme 81).
 | ||
| Scheme 81 Synthesis of (−)-conduramine A-1 (−)-177, (+)-conduramine E-1 (+)-221 and (+)-conduramine F-1 (+)-183. | ||
While chiral auxiliary–based approaches enable efficient access to several conduramine derivatives, carbohydrate-derived strategies have also been explored for the stereodivergent synthesis of conduramine intermediates.
2.79. Divergent syntheses of precursors to (+)- and (−)-conduramine E
In 2012, Ghosal et al. reported stereodivergent strategies for preparing key intermediates en route to both enantiomers of conduramine E.99In addition to carbohydrate-derived metathesis strategies, alternative approaches employing boronate intermediates have also been developed for the synthesis of conduramine derivatives.
2.80. Divergent access to conduramine A-1 and C-4 using boronate intermediates
In 2012, Norsikian et al. reported efficient synthetic routes to conduramine A-1 (177) and conduramine C-4 (244), employing D-ribose derivatives as versatile chiral building blocks.100Bromination of (909) using NBS/AgNO3/AcOH produced bromide (910) (69%), while an alternative halogenation using NIS or HInCl2/Et3B/I2 delivered iodide (911) in significantly higher yield (∼95%). Cross-coupling of bromide (910) with bis(pinacolato)diboron under PdCl2(PPh3)2/PPh3 catalysis afforded a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 mixture of E/Z boronic esters (912) in ∼66% combined yield. Alternatively, borylation of iodide (911) via t-BuLi, trimethyl borate, and pinacol provided (912) in ∼79% yield.
7 mixture of E/Z boronic esters (912) in ∼66% combined yield. Alternatively, borylation of iodide (911) via t-BuLi, trimethyl borate, and pinacol provided (912) in ∼79% yield.
Oxidative cleavage of (912) with sodium metaperiodate generated aldehyde (913) (73%), which upon treatment with 6 N HCl afforded a mixture of hemiacetals (914a) and (914b). Cyclization with diallylamine (All)2NH yielded aminal (915), and final deallylation using Pd(PPh3)4/NDMBA furnished conduramine A-1 (177) in ∼70% yield.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 ratio, with a combined yield of ∼73%.
11 ratio, with a combined yield of ∼73%.The major Z-isomer (Z-918) was treated sequentially with t-BuLi, trimethyl borate, pinacol, and sodium metaperiodate to produce aldehyde (919) in approximately 70% yield. Cyclization of (919) in the presence of TFA and (All)2NH furnished aminal (920), which was finally converted into conduramine C-4 (244) through Pd(PPh3)4-catalyzed deallylation using NDMBA, delivering the target structure in ∼78% yield (Scheme 83).
Beyond boronate-based approaches, alternative strategies involving diazide and diene intermediates have also been explored for the synthesis of diamino conduritol derivatives.
2.81. Construction of 2,3-diamino conduritol F via diazide and diene intermediates
In 2012, Ekmekci et al. reported a concise synthetic route to 2,3-diamino conduritol F (932), beginning from a simple anhydride precursor.101 Initial methanolysis of the anhydride in the presence of HCl generated diester (921), which upon heating with hydrazine in methanol afforded hydrazide derivative (922) in ∼59% yield. Conversion of (922) to diazide (923) was achieved via NaNO2/HCl treatment, delivering the desired intermediate in ∼77% yield. Refluxing diazide (923) in benzene promoted rearrangement to diisocyanide (924), which upon heating with methanol furnished compound (925). Subsequent bromination of (925) using Br2 in dichloromethane gave anti-dibromide (926) in ∼87% yield.Base-induced elimination of (926) using DBU provided diene (927) in ∼34% yield. Photooxygenation of (927) under tetraphenylporphyrin sensitization afforded endo-peroxide (928) in ∼82% yield. Reductive cleavage of the peroxide with thiourea generated diol (929), which was acetylated to yield diacetate (931) in ∼63% yield. Final deacetylation of (931) using NaOH in methanol furnished 2,3-diamino conduritol F (932) in ∼42% yield (Scheme 84).
Beyond diamino derivatives, conduritol frameworks have also been functionalized with heteroaromatic substituents to generate structurally diverse analogues with potential biological relevance.
2.82. Synthesis of mono- and bis-indole conduritol derivatives
In 2012, Çavdar et al. reported the synthesis of mono- and bis-indole conduritol derivatives.16 The procedure began with p-benzoquinone (9), which was brominated (∼70% yield), reduced with NaBH4 (∼84% yield), and subsequently acetylated (∼75% yield) to give diacetate (368). Treatment of diacetate (368) with KOH induced acetate hydrolysis and an intramolecular substitution, producing anti-bisepoxide (370) in ∼77% yield.Reaction of anti-bisepoxide (370) with indoline, followed by acetylation, afforded compound (934) in ∼72% yield. Oxidation of (934) with MnO2 yielded the mono-indole conduritol B diacetate (935) in ∼85% yield. Alternatively, reaction of anti-bisepoxide (370) with indoline in dichloromethane produced the bis-indolidine conduritol B (936) in ∼80% yield. Subsequent acetylation and MnO2 oxidation of (936) furnished the bis-indole conduritol B diacetate (937) with an approximate yield of 74% (Scheme 85).
In addition to heteroaromatic functionalization of conduritol scaffolds, carbohydrate-derived strategies have also been developed for constructing conduramine frameworks.
2.83. Synthesis of conduramine C and D frameworks from ribofuranose
In 2013, Rajender et al. reported the synthesis of conduramine C and D derivatives.102 The sequence began with ribofuranose (938), which was reacted with vinyl magnesium bromide to yield compound (939) in ∼75% yield. Oxidation with NaIO4 produced lactol (940) in ∼80% yield. Treatment of (940) with benzylamine, followed by vinyl magnesium bromide, gave diene (941) in ∼75% yield over the two steps. Protection of the amine in (941) using CbzCl and NaHCO3 afforded diene (942) in ∼90% yield.Diene (942) underwent ring-closing metathesis (RCM) with Grubbs' 2nd-generation catalyst to yield cyclohexene (943) in ∼80% yield. Subsequent treatment with sodium in liquid ammonia, followed by Boc protection, produced compound (944) in ∼78% yield over two steps. Compound (944) was then converted into the protected conduramine C-1 derivative (945) via a Mitsunobu reaction with tetraphenylporphyrin, DEAD, and p-nitrobenzoic acid, followed by hydrolysis, achieving ∼80% yield over two steps.
In a parallel pathway, deprotection of the acetonide and Boc groups in (944) using trifluoroacetic acid, followed by acetylation, yielded conduramine D-1 tetraacetate (284) in ∼90% yield over two steps. Both (945) and (284) serve as intermediates for the synthesis of conduramines and their derivatives (Scheme 86).
Beyond carbohydrate-derived approaches, alternative strategies have also been explored to prepare functionalized conduramine derivatives through epoxide intermediates.
2.84. Construction of N-substituted conduramine F-4 analogues
In 2014, Kuno et al. reported the synthesis of N-substituted (+)-conduramine F-4 derivatives.103 The sequence began with the protection of the hydroxyl groups of (+)-proto-quercitol (+)-946 using 2,2-dimethoxypropane and p-TsOH, yielding compound (947) in ≈90% yield. The remaining free hydroxyl group in (947) was then protected with MsCl in the presence of triethylamine, producing compound (948) in ∼97% yield.Treatment of (948) with DBU in toluene induced elimination, forming the cyclohexene derivative (949) in ∼76% yield. Subsequent methanolysis with PPTS gave the anti-diol (950) in ∼91% yield. Conversion of (950) into epoxide (164) using Martin sulfurane proceeded with ∼69% yield. Finally, the epoxide (164) underwent ring opening with a primary amine (RNH2) in acetonitrile, followed by acetonide deprotection, furnishing the N-substituted (+)-conduramine F-4 derivatives (166a–n) in yields ranging from 64% to 100% (Scheme 87).
In addition to the synthesis of diversified N-substituted conduramine derivatives, stereodefined conduramine F-4 has also been prepared through ring-closing metathesis strategies.
2.85. Synthesis of (−)-conduramine F-4 via RCM strategy
In 2016, Harit et al. reported a synthetic route to (−)-conduramine F-4 (−)-166.104 The sequence began with the oxidation of diol (951) using DMSO and oxalyl chloride to form the corresponding aldehydes, yielding intermediate (952). This intermediate was then subjected to a Wittig reaction, producing diene (953) with an overall yield of ∼60% from (951).Heating diene (953) in the presence of Grubbs' 2nd generation catalyst effected a ring-closing metathesis (RCM), furnishing cyclohexene derivative (954) in ∼70% yield. Treatment of (954) with sodium amalgam yielded compound (955) in ∼74% yield. Subsequent acetylation produced compound (956) in ∼80% yield, and final deprotection of all protecting groups afforded (−)-conduramine F-4 (−)-166 in ∼52% yield (Scheme 88).
In addition to metathesis-based approaches starting from carbohydrate derivatives, amino acid-derived building blocks have also been employed for the synthesis of conduramine frameworks.
2.86. Synthesis of (−)-conduramine A-1 from a D-serine-derived intermediate
In 2016, Myeong et al. reported an efficient synthetic route to (−)-conduramine A-1 (−)-177.105 The sequence commenced with compound (957), derived from D-serine, which underwent ozonolysis followed by nucleophilic addition of vinyl magnesium bromide to furnish a mixture of adducts (958) and (959). The ratio of these products was influenced by the choice of Lewis acid, solvent, and reaction temperature.Acetylation of the major isomer (959) provided acetate (960) in ∼96% yield. Subsequent treatment with CbzCl and sodium bicarbonate generated the carbamate (961) in ∼90% yield. Regioselective cleavage of the silyl ether in (961) using hydrofluoric acid afforded alcohol (962) in ∼78% yield. Oxidation of (962) followed by a Wittig olefination delivered diene (963) in an overall yield of ∼56%.
Exposure of diene (963) to Grubbs' 2nd generation catalyst induced a ring-closing metathesis (RCM), producing the cyclized intermediate (964) in ∼62% yield. Final global deprotection under 6 M HCl furnished (−)-conduramine A-1 (−)-177 in ∼78% yield (Scheme 89).
Beyond the synthesis of simple conduramine frameworks, related strategies have also been employed to access N-substituted conduramine derivatives.
2.87. Synthesis of N-benzyl conduramine E-1 and F-1 via RCM
In 2017, Katakam et al. reported a stereodivergent strategy for the synthesis of N-benzyl conduramine E-1 and N-benzyl conduramine F-1.106 The sequence commenced with NaIO4-mediated oxidative cleavage of compound (965), derived from D-mannitol, to afford aldehyde (966). Nucleophilic addition of vinyl magnesium bromide furnished a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.3 mixture of diastereomers (967) and (968) in an overall yield of ∼76% across two steps.
1.3 mixture of diastereomers (967) and (968) in an overall yield of ∼76% across two steps.
For the synthesis of N-benzyl conduramine F-1, selective deprotection of the primary acetonide in (967), followed by oxidative cleavage with periodic acid, generated aldehyde (969). Condensation with benzylamine gave imine (970), which upon vinylmagnesium bromide addition furnished diene (971) in ∼72% yield over three steps. Boc protection of (971) yielded diene (972) in ∼82% yield. Subsequent ring-closing metathesis (RCM) with Grubbs' 2nd generation catalyst produced cyclohexene derivative (973) in ∼79% yield. Final deprotection of the acetonide moiety using 6 N HCl afforded N-benzyl conduramine F-1 (974) in ∼74% yield.
In the parallel pathway, intermediate (975) was obtained from compound (968) in ∼70% yield across three steps. Boc protection furnished diene (976) in ∼82% yield, which underwent RCM to generate cyclohexene derivative (977) in ∼70% yield. Acetonide cleavage under acidic conditions ultimately provided N-benzyl conduramine E-1 (978) in ∼73% yield (Scheme 90).
Beyond RCM-based strategies employing carbohydrate-derived precursors, alternative approaches incorporating chiral auxiliaries and sulfinimine chemistry have also been explored for the synthesis of conduramine frameworks.
2.88. Synthesis of conduramine F-1 via sulfinimine addition and RCM
In 2018, Prasad et al. reported an efficient synthetic route to conduramine F-1 (183).107 The sequence commenced with bis-Weinreb amide (970), derived from tartaric acid, which underwent nucleophilic addition of vinylmagnesium bromide to furnish the α,β-unsaturated ketone (980) in ∼74% yield. Luche reduction of (980) provided allylic alcohol (981) in ∼88% yield, and subsequent protection of this alcohol as its TBDMS ether using TBDMSCl/imidazole afforded compound (982) in ∼89% yield.Reduction of (982) with DIBAL-H generated the corresponding aldehyde, which was transformed into sulfinimine (983) using (S)-tert-butanesulfinamide in the presence of Ti(OEt)4 (∼72% yield). Addition of vinylmagnesium bromide to sulfinimine (983) yielded sulfinamide (984) in ∼89% yield. Deprotection of the silyl ether using TBAF provided diene (985) in ∼94% yield.
Ring-closing metathesis (RCM) of (985) using Grubbs' 2nd generation catalyst delivered the cyclized intermediate (986) in ∼86% yield. Final global deprotection furnished conduramine F-1 (183) in ∼60% yield (Scheme 91).
Related strategies combining vinyl addition reactions with ring-closing metathesis have also been applied to the synthesis of other conduramine derivatives.
2.89. Synthesis of conduramine B-2 and F-2 via vinyl addition-RCM strategy
In 2019, Harit et al. described an efficient synthetic strategy for accessing conduramine B-2 (996) and conduramine F-2 (998) from a carbohydrate-derived precursor.108 The sequence began with the reduction of 2-iodo-2-deoxy-1-sulfonamido-α-D-glucose (987) using sodium borohydride to yield compound (988) in ∼95% yield. Acetylation of (988) in a ZnCl2-catalyzed mixture of acetic anhydride and acetic acid (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) afforded acetate (989) in ∼91% yield, which upon hydrolysis delivered alcohol (990) in ∼93% yield.
1) afforded acetate (989) in ∼91% yield, which upon hydrolysis delivered alcohol (990) in ∼93% yield.
Conversion of alcohol (990) to iodide (991) using iodine, triphenylphosphine, and imidazole proceeded in ∼92% yield. Sonochemical treatment of (991) with zinc induced dehydroiodination, furnishing alkene (992), which underwent nucleophilic addition of vinylmagnesium bromide to afford a mixture of dienes (993) and (994) in ∼61% and 25% yields, respectively.
Ring-closing metathesis (RCM) of dienes (993) and (994) using Grubbs' 1st generation catalyst furnished the corresponding cyclohexene derivatives (995) and (997) in ∼78% and 79% yields. Global deprotection of (995) and (997) provided conduramine B-2 (996) and conduramine F-2 (998) in ∼76% and 77% yields, respectively (Scheme 92).
Alternative strategies based on stereoselective epoxide functionalization have also been explored for the synthesis of conduramine analogues.
2.90. Stereoselective construction of (−)-conduramine A and E analogues
In 2019, Pinto et al. reported an enantioselective strategy for synthesizing (−)-conduramine A-1 (−)-177, (−)-conduramine A-2 (−)-1012, and (−)-conduramine E-2 (−)-1013.109 The sequence began with the epoxidation of cyclohexa-1,4-diene (999) using peracetic acid to afford epoxide (1000), which upon bromination in dichloromethane furnished anti-dibromide (1001) in ∼88% yield. Base-induced elimination of (1001) with DBU in diethyl ether yielded alkene (1002), which underwent regio- and stereoselective epoxide opening upon treatment with enantiopure (R)-α-methyl-p-methoxybenzylamine (1003). This produced a separable mixture of amines (1004) and (1005), isolated in ≈21% and ≈19% yield, respectively, along with an additional mixed fraction (∼21%).Further transformation of compound (1005) with 40% aqueous HBF4, followed by oxidation with m-CPBA, generated a mixture of four derivatives—(1008), (1009), (1010), and (1011)—in a 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37
37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32
32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 ratio. These were separated by preparative TLC, affording isolated yields of ∼8%, 21%, 15%, and 5%, respectively.
14 ratio. These were separated by preparative TLC, affording isolated yields of ∼8%, 21%, 15%, and 5%, respectively.
Finally, global deprotection of the α-methyl-p-methoxybenzyl (MPM) groups using Et3SiH/TFA furnished the target natural products: (−)-conduramine A-1 (−)-177, (−)-conduramine A-2 (−)-1012, and (−)-conduramine E-2 (−)-1013 in ∼82%, 79%, and 58% yields, respectively (Scheme 93).
 | ||
| Scheme 93 Synthesis of (−)-conduramine A-1 (−)-177, (−)-conduramine A-2 (−)-1012, and (−)-conduramine E-2 (−)-1013. | ||
Beyond the synthesis of conduramine analogues, conduritol-derived intermediates have also been employed for the preparation of structurally diverse inositol derivatives.
2.91. Synthesis of dimethylated inositol derivatives from conduritol B epoxide
In 2020, Aksu et al. reported an efficient synthetic route to various inositol derivatives using a conduritol B epoxide intermediate.110 The sequence commenced with bromination of p-benzoquinone (9), affording anti-dibromide (10) in nearly 95% yield. Reduction of (10) with sodium borohydride provided diol (369) in approximately 85% yield. Subsequent treatment of diol (369) with sodium methoxide in methanol furnished 3,6-dimethoxy conduritol B (255) in 87% yield. Acetylation under acidic conditions using acetic anhydride generated the corresponding diacetate (1014) in nearly 89% yield.Epoxidation of (1014) with m-CPBA afforded the protected conduritol B epoxide (1015) in about 70% yield. Ring opening of epoxide (1015) with acetic anhydride under acidic conditions produced a 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of acetoxyalcohols (1016) and (1017) in an overall yield of approximately 87%. Final deacetylation using potassium carbonate in methanol delivered two inositol derivatives: DL-2,5-di-O-methyl-chiro-inositol (1018) in 61% yield and DL-1,4-di-O-methyl-scyllo-inositol (1019) in 89% yield (Scheme 94).
1 mixture of acetoxyalcohols (1016) and (1017) in an overall yield of approximately 87%. Final deacetylation using potassium carbonate in methanol delivered two inositol derivatives: DL-2,5-di-O-methyl-chiro-inositol (1018) in 61% yield and DL-1,4-di-O-methyl-scyllo-inositol (1019) in 89% yield (Scheme 94).
3. Critical and comparative analysis of synthetic strategies toward conduritol derivatives
Beyond reporting individual synthetic routes, it is instructive to examine the broader trends that have shaped the development of conduritol chemistry. Comparative evaluation of these methodologies reveals recurring strategic themes, highlights their advantages and limitations, and provides insight into how different synthetic philosophies converge toward similar cyclitol frameworks. While advantages and limitations of individual methodologies are discussed, wherever possible, in the context of their respective synthetic approaches throughout the review, the following section provides a consolidated and comparative perspective to highlight overarching trends, strengths, and limitations across different strategies. In many cases, the original studies were developed in the context of target-oriented synthesis, and therefore the breadth of substrate scope was not always systematically explored. Nevertheless, several methodologies demonstrate significant versatility through divergent transformations and adaptation to structurally distinct conduritol, conduramine, and inositol derivatives.Taken together, the methodologies surveyed in this review highlight how the conduritol framework has evolved from a synthetic curiosity into a highly versatile chiral pool for accessing inositols, conduramines, aminocyclitols, and complex natural products. A clear trend over the last three decades is the progressive shift from linear, target-specific routes to divergent, platform-type strategies, where a single conduritol- or inositol-derived intermediate can be elaborated into families of polyols, amino derivatives, polyphosphates, and annulated analogues. Many of these transformations rely on well-established mechanistic principles, including stereospecific epoxide opening, osmium-mediated syn-dihydroxylation, and metathesis-driven ring formation, which collectively enable precise control over the stereochemical architecture of conduritol frameworks.
From the standpoint of starting material and chiral information, three broad philosophies emerge.
(i) Chemoenzymatic and microbial cis-dihydroxylation approaches (Hudlický, Banwell, Bellomo, Paul, Vitelio, Orsini, etc.) rely on arene oxidation (chlorobenzene, bromobenzene, m-dibromobenzene, naphthalenes) to introduce the initial cis-diol in a single step with excellent stereocontrol. These routes excel in step economy at the very beginning and often deliver densely functionalized cyclohexadienediols that can be channelled into conduritol A–F, inositols, and gabosine-type carbasugars. Their weaknesses are practical: dependence on whole-cell systems, oxygen transfer, fermentation-like conditions and sometimes modest substrate scope.
(ii) Carbohydrate-based strategies (Kornienko, Mehta, Pattenburg, Saito, Andresen, Nadein, Ghosal, Rajender, Harit, Katakam, etc.) transform readily available sugars (D-glucose, D-mannose, D-galactose, D-xylose, D-ribose) into conduritol and inositol derivatives via olefination-RCM manifolds or carefully choreographed protecting-group patterns. These routes are attractive for chirality “for free” and scalability, but often suffer from heavy protecting-group load and multiple redox adjustments.
(iii) Small chiral pool and auxiliary-based routes (L-tartaric acid, proto-quercitol, D-serine, camphorsultam-derived auxiliaries, sulfinimines) provide flexible access to specific stereochemical arrays, particularly for conduramine A/F series, with high enantioselectivity, though at the cost of auxiliary installation/removal steps and sometimes lower overall atom economy.
A second major axis of comparison concerns the construction of the cyclohexene/cyclohexane core. Older approaches used pericyclic reactions such as Diels–Alder and nitroso-Diels–Alder reactions (Trost's cyclophellitol and pancratistatin work, Elango's narciclasine, Jana's Cu-catalysed nitroso-DA to conduramine A-1), or photooxygenation of dienes derived from cyclooctatetraene or cyclohexadiene (Balci, Kelebekli, Cantekin, Ekmekci). These are powerful because they build complexity rapidly and allow embedded peroxide or enone functionalities that can be leveraged for downstream dihydroxylation or fragmentation. However, they can be sensitive to scale, light, and oxygen and sometimes give mixtures of endo/exo or regioisomers that must be separated or equilibrated.
By contrast, ring-closing metathesis (RCM) has emerged as perhaps the single most general tactic for assembling the conduritol core from acyclic sugar- or tartaric-derived dienes (Gallos, Chang 1999 and 2009, Mehta, Saito, Ghosal, Myeong, Harit, Prasad, Nadein, Andresen). RCM offers excellent control over ring size and olefin geometry, tolerates multiple protecting groups, and is readily adapted to divergent programmes: the same diene manifold can be pushed toward conduritol E/F/B scaffolds and then onwards to inositols, valienamine, aminocyclitols or phosphates. The trade-offs are the cost and air-sensitivity of Ru catalysts and the need to manage E/Z mixtures in some cases; nonetheless, RCM strategies are arguably the most modular and “designable” across this landscape.
The installation and manipulation of heteroatom functionality (OH, N, X, Se, P) provides another clear point of comparison. For oxygenation, almost all families rely heavily on OsO4-based syn-dihydroxylation (either classical OsO4/NMO or AD-mix-β) and, to a lesser extent, RuO4/RuCl3–NaIO4 oxidations for ring opening to inositols or oxidative rearrangements (e.g., Podeschwa's neo/allo/epi-inositols, Banwell's L-ascorbic acid, Bellomo's deoxy-selenylconduritols). These steps are highly reliable and stereocontrolled but score poorly against green-chemistry metrics due to the toxicity and cost of Os and Ru. A positive development is the regioselective dihydroxylation control achieved in some systems (e.g., Chappell's use of AD-mix-β/MeSO2NH2 to select one diastereomer exclusively), which reduces separation overhead and waste. Future work could profitably target catalytic, less toxic alternatives while preserving the exquisite stereocontrol currently provided by late transition metals.
For nitrogen introduction, several conceptually distinct manifolds can be contrasted.
(a) Epoxide and cyclic sulfate aminolysis (Lee's conduramine F, Rinner's epoxyconduramine route to epi-7-deoxypancrastatin, Pandey's epoxidation/functionalization, Kuno's Martin sulfurane epoxide, Katakam's and Prasad's RCM-based routes) provide direct access to 1-amino or 1,4-diamino motifs with good regioselectivity, especially when combined with neighbouring protecting groups to steer attack.
(b) Mitsunobu and azide/mesylate inversion strategies (Prinzbach's (−)-conduramine E, Falshaw's conduritol B epoxides, Serrano's rich aminoinositol library, Lysek's conduramine B-1/F-1, Rajender's conduramine C/D derivatives) afford powerful control over relative configuration at multiple centres and are particularly useful for accessing myo/scyllo/chiro series from a single epoxide precursor. Their downside is reliance on stoichiometric DEAD/DIAD, PPh3 and azides, which are environmentally problematic on scale.
(c) Chemoenzymatic and Pd-catalysed asymmetric approaches (Trost's allylic alkylations to conduramine C-4, Hygromycin A aminocyclitol, and pancratistatin; Lu's chiral auxiliary-mediated routes; Pinto's chiral amine-mediated openings) achieve high enantioselectivity at the cost of more elaborate catalysts or auxiliaries. These asymmetric catalysis-based routes are particularly attractive when access to a single enantiomer is crucial, as for narciclasine, pancratistatin, cyclophellitol and halichondrin fragments.
Halogenation, selenylation and phosphorylation chemistry further expand the divergent potential of the conduritol core. Baran's haloconduritols (BCl3/BBr3-mediated halodeacetalization) and Cantekin's bromo-conduritols B/C show that carefully orchestrated halogenation–elimination–acetate substitution sequences provide access to functional handles for cross-coupling or further oxidation. Bellomo's deoxy-selenylconduritols demonstrate that Se-based nucleophilic openings can be used not only to generate new C–Se and C–O patterns but also to trigger oxidative rearrangements to partially protected conduritol C. On the phosphorylation side, the work of Pattenburg, Podeschwa, Know, Saito and Andresen illustrates a robust “platform logic”: once a suitably protected inositol is in hand, phosphoramidite chemistry enables systematic construction of di-, tri-, tetra- and hexakisphosphate patterns, including azido- and amino-inositol phosphates of biological relevance. These late-stage, phosphorylation-based divergences are powerful but are inherently multistep and require extensive protecting-group choreography to control site selectivity.
Methodologically, several named reactions and motifs recur across otherwise different programmes: Ramberg–Bäcklund rearrangements from thiepane sulfones (Cerè, Arcelli, Vanda Cerè) as a general way to introduce cyclohexene unsaturation; photooxygenation of dienes to endo-peroxides followed by reductive cleavage (Balci, Kelebekli, Ekmekci) to deliver bis-homo-conduritols or diamino derivatives; Diels–Alder and nitroso-Diels–Alder cycloadditions as compact entries into highly functionalized cyclohexanes (Elango, Jana, Trost); and extensive use of RCM not just to give simple conduritol rings but also annulated systems (Mehta's cyclohexa- and cyclopenta-annulated inositols) and conformationally locked analogues. In general, Ramberg–Bäcklund and pericyclic/peroxide strategies are synthetically elegant but often less user-friendly than RCM in terms of operational simplicity and safety.
From an applications perspective, it is striking how often conduritols and conduramines are embedded in or serve as chiral building blocks for bioactive targets: (+)/(−)-cyclophellitol, pericosine B, (−)/(+)-gabosine A, (+)- and epi-7-deoxypancrastatin, pancratistatin, (−)- and (+)-narciclasine, halichondrin B fragments, Hygromycin A aminocyclitol, L-ascorbic acid, phosphatidylinositol analogues and inositol polyphosphates. Here, chemoenzymatic routes and RCM-enabled carbohydrate pathways clearly dominate because they combine reliable absolute stereocontrol with modular late-stage diversification (e.g., conversion of conduritol B epoxide into a broad library of scyllo, myo and chiro aminoinositols by Serrano, or Saito/Know/Andresen's multiple polyphosphate topologies from one conduritol B precursor). In contrast, some of the more specialized haloconduritols, selenylconduritols, annulated and indole-functionalized derivatives (Baran, Bellomo, Çavdar, Mehta) are better viewed as methodology demonstrations that enrich the structural diversity space rather than as immediately scalable routes to a single pharmacophore.
Finally, a few general advantages and limitations can be distilled.
• Strengths of the modern toolbox include: (a) extensive diastereo- and enantioselective control at each stage; (b) highly divergent use of common intermediates (epoxides, aziridines, partially benzylated or acetonide-protected conduritols); (c) compatibility with complex downstream targets and late-stage introduction of polar groups (phosphates, amines, amides); and (d) increasing use of catalytic processes (Pd, Cu, Ru-RCM, enzymatic lipases and dioxygenases).
• Weaknesses remain in the areas of step economy and sustainability: many sequences are long, rely on multiple protecting-group manipulations and redox toggling, and use toxic or precious reagents (OsO4, RuO4, Bu3SnH, DEAD, DPPA, Pb/Se chemistry, peracids). Chemoenzymatic methods, though stereochemically superb, may pose scale-up and reproducibility challenges outside of specialized laboratories.
Looking ahead, the body of work summarized here suggests that future progress will likely come from integrating the best features of these strategies: using biocatalytic or chiral-pool inputs for initial stereocontrol, employing RCM or other catalytic annulations to build the ring with minimal protecting-group overhead, and then applying site-selective C–H or C–O functionalization, milder aminating agents, and greener oxidation methods to install the desired nitrogen, phosphorus and halogen functions. Such developments would not only streamline access to classical conduritol/conduramine targets, but also enable rapid exploration of new, densely functionalized cyclitol space for medicinal chemistry and chemical biology.
From a practical standpoint, several factors influence the broader applicability of the synthetic strategies discussed in this review. While many routes provide excellent stereochemical control and access to structurally diverse conduritol derivatives, aspects such as reaction scalability, cost of reagents, and environmental considerations remain important limitations. For instance, several widely used transformations including osmium-mediated dihydroxylation, peracid epoxidation, Mitsunobu reactions, and tributyltin hydride reductions rely on reagents that may present challenges in terms of toxicity, cost, or waste generation when considered for large-scale synthesis. Moreover, detailed impurity profiles, robustness studies, and large-scale reaction data are often not reported in the primary literature, which makes systematic comparison difficult. Consequently, while many of the reported methodologies are highly effective at the laboratory scale, further optimization and development of greener, scalable alternatives will be essential for translating these strategies into practical synthetic platforms.
4. Medicinal relevance of conduritol-derived aminocyclitols
Conduritol and conduramine frameworks have also attracted significant attention in medicinal chemistry because closely related aminocyclitol structures form the core of several clinically important glycosidase inhibitors. These compounds mimic carbohydrate substrates and inhibit carbohydrate-processing enzymes, thereby modulating glucose metabolism. Several approved antidiabetic drugs, including acarbose, miglitol, and voglibose, contain highly functionalized cyclitol or aminocyclitol motifs related to conduritol-derived scaffolds. These agents act as α-glucosidase inhibitors, delaying carbohydrate digestion in the intestine and reducing post-prandial hyperglycemia. The inhibitory potency of these molecules varies depending on the enzyme source and assay conditions, but reported IC50 values typically fall within the micromolar to sub-millimolar range. The development and clinical success of these drugs highlight the broader pharmaceutical importance of densely functionalized cyclitols and underscore the value of synthetic strategies that enable efficient access to conduritol and conduramine derivatives (Table 1).111–114| Drug | Core scaffold | Therapeutic use | Target enzyme | Approximate IC50 |
|---|---|---|---|---|
| Acarbose | Valienamine-derived aminocyclitol | Type-2 diabetes | α-glucosidase/α-amylase | ∼0.78 mM |
| Miglitol | Iminosugar (aminocyclitol) | Type-2 diabetes | α-glucosidase | ∼10–20 µM |
| Voglibose | Valiolamine-derived aminocyclitol | Type-2 diabetes | α-glucosidase | ∼0.1–1 µM |
5. Future perspectives
Looking ahead, several promising directions can be envisaged for the continued development of conduritol- and conduramine-based chemistry.5.1. Greener and more step-economic oxidation and amination methods
Replacing Os-, Ru- and peracid-based protocols with milder, more sustainable catalytic systems such as organocatalytic, electrochemical, enzymatic, or earth-abundant metal-mediated oxidations will be crucial for translating many of these routes to larger scale. Similarly, safer, catalytic alternatives to azide- and Mitsunobu-based nitrogen introduction would significantly improve the environmental and safety profiles of aminocyclitol syntheses.5.2. Refined divergent platforms and late-stage functionalization
The concept of using a single conduritol or conduramine scaffold as a branching point to multiple inositol, aminoinositol and polyphosphate derivatives is already well established. Future work can focus on designing “minimal protection” or “protection-free” platforms that combine site-selective C–H or C–O functionalization, orthogonal protecting groups, and programmable phosphorylation. Such approaches would facilitate rapid assembly of focused libraries of cyclitol-based probes, inhibitors, and mimetics.5.3. Integration of biocatalysis with modern catalysis and flow chemistry
Microbial and enzymatic cis-dihydroxylation provides unparalleled stereocontrol at an early stage. Coupling these transformations with RCM, cross-coupling, and catalytic functionalizations in telescoped or continuous-flow sequences could dramatically reduce purification steps and improve overall efficiency. Engineered dioxygenases, epoxide hydrolases, and transaminases tailored to polyfunctional cyclohexene substrates may further expand the accessible structural space.Microbial and enzymatic cis-dihydroxylation provides unparalleled stereocontrol at an early stage. Coupling these transformations with RCM, cross-coupling, and catalytic functionalizations in telescoped or continuous-flow sequences could dramatically reduce purification steps and improve overall efficiency. Engineered dioxygenases, epoxide hydrolases, and transaminases tailored to polyfunctional cyclohexene substrates may further expand the accessible structural space.
It is worth noting, however, that several key transformations in conduritol chemistry—including heterogeneous catalytic reactions (e.g., Pd/C hydrogenations), enzymatic oxidations, and reactions involving insoluble reagents, pose practical challenges for direct translation into continuous-flow systems. Nevertheless, recent advances in flow chemistry, such as packed-bed reactors, immobilized catalysts and enzymes, slurry-based flow systems, and segmented (gas–liquid or liquid–liquid) flow technologies, have demonstrated that heterogeneous processes can be effectively adapted to flow conditions. These developments suggest that, with appropriate reactor design, even traditionally heterogeneous transformations relevant to conduritol and conduramine synthesis could be rendered compatible with continuous processing.
5.4. Rational design of bioactive analogues and structure–activity studies
The successful syntheses of pancratistatin, narciclasine, cyclophellitol, halichondrin fragments, gabosines, and inositol polyphosphates highlight the pharmacological relevance of conduritol and conduramine cores. Moving forward, the emphasis is likely to shift from “can we make it?” to “how do we systematically vary it?”. Efficient, divergent routes will be invaluable for generating analogues with subtle changes in stereochemistry, substitution pattern, or phosphorylation state to probe enzyme inhibition, receptor binding, signalling modulation, and transport properties.5.5. Expansion into materials, supramolecular, and glycobiology applications
The dense, polyhydroxylated nature of these frameworks makes them attractive not only as enzyme inhibitors but also as building blocks for multivalent ligands, chiral ionophores, molecular recognition motifs, and functional materials (e.g., in self-assembly or metal coordination). Polyhydroxylated cyclitol scaffolds can participate in highly organized supramolecular interactions through hydrogen bonding, metal coordination, and host–guest recognition processes, which are central to the design of molecular recognition systems and functional supramolecular assemblies. Recent studies have highlighted how such polyfunctional motifs can be incorporated into modern supramolecular and materials architectures where precise spatial arrangement of functional groups governs molecular recognition and self-assembly processes.115 Future research may exploit conduritol- and inositol-based scaffolds in polymer chemistry, nanoscience, and glycobiology, where the precise spatial arrangement of hydroxyl and amino groups is often critical. Polyhydroxylated cyclitol motifs have recently attracted attention as functional building blocks for advanced polymeric and supramolecular materials, including responsive polymer networks, dynamic covalent systems, and carbohydrate-inspired nanostructures. Such systems demonstrate how reversible covalent interactions and densely functionalized cyclitol frameworks can be integrated into modern materials design and polymer architectures.116–119In summary, the synthetic methodologies reviewed here have established conduritols, conduramines, and their inositol derivatives as exceptionally rich and flexible platforms at the interface of synthetic methodology, medicinal chemistry, and chemical biology. Continued progress in catalytic, chemoenzymatic, and diversity-oriented synthesis, guided by sustainability considerations and biological questions, can be expected to further unlock the potential of these compact yet information-dense scaffolds in both fundamental research and applied science.
6. Conclusion
Over the past few decades, conduritols and conduramines have evolved from structurally intriguing cyclitols into central scaffolds for the construction of inositols, aminocyclitols, and complex bioactive natural products. The synthetic studies summarized in this review clearly demonstrate that these frameworks are not merely end points, but highly versatile platform intermediates that can be divergently elaborated into a wide spectrum of molecular architectures.Collectively, the available methodologies span chemoenzymatic cis-dihydroxylation of simple arenes, carbohydrate-derived strategies, small chiral pool approaches, auxiliary- and catalyst-controlled asymmetric routes, and pericyclic or metathesis-based ring constructions. Among these, ring-closing metathesis (RCM) of sugar- or tartaric acid-derived dienes and microbial or enzymatic arene cis-dihydroxylation have emerged as particularly powerful tactics for assembling densely functionalized cyclohexene or cyclohexane cores with precise stereochemical control. Subsequent use of epoxidation, aziridination, Ramberg–Bäcklund rearrangements, selective dihydroxylation, and Mitsunobu or azide/mesylate inversions allows for fine-tuning of the oxygen and nitrogen substitution patterns required for conduritol, conduramine, and inositol derivatives.
A notable theme is the progressive shift from linear, target-specific syntheses toward modular, divergent strategies. Conduritol B and E derivatives, in particular, have served as nodal intermediates, enabling access to myo, scyllo, chiro, allo, neo, and epi inositols, as well as to polyphosphate, azido-, amino-, halo-, and selenyl-functionalized analogues. This platform concept has been exploited in the synthesis of structurally and biologically important compounds such as pancratistatin and its analogues, cyclophellitol, pericosine B, narciclasine, halichondrin fragments, hygromycin A aminocyclitol units, L-ascorbic acid, inositol polyphosphates, and phosphatidylinositol mimetics.
Although most reported syntheses of conduritols and conduramines have been developed within academic laboratories, these frameworks possess significant relevance in medicinal chemistry. Aminocyclitol structures derived from conduritols serve as key scaffolds in the design of glycosidase inhibitors, antiviral agents, and other bioactive molecules. Several natural products and drug candidates, including valienamine-based α-glucosidase inhibitors such as acarbose and voglibose, contain closely related aminocyclitol motifs. Consequently, synthetic methodologies developed for conduritols and conduramines provide valuable strategies for constructing highly functionalized cyclohexene and aminocyclitol architectures relevant to pharmaceutical research and lead optimization. Furthermore, scalable synthetic approaches and chemoenzymatic methods reported for conduritol derivatives offer potential pathways for future process-chemistry development.
Despite this impressive toolbox, certain limitations remain. Many routes are still relatively long and protecting-group intensive, often relying on toxic or environmentally problematic reagents such as OsO4, RuO4/RuCl3–NaIO4, tributyltin hydride, stoichiometric Mitsunobu reagents, and organoselenium or azide chemistry. Chemoenzymatic processes, while highly stereoselective, may pose challenges in terms of operational simplicity and scalability. Nevertheless, the breadth of strategies now available provides a solid foundation for more sustainable and streamlined approaches to these densely functionalized cyclitols.
From a translational and medicinal chemistry perspective, it is important to recognize that several practical challenges remain in advancing conduritol- and conduramine-based compounds toward therapeutic applications. Detailed pharmacokinetic (DMPK) and toxicity data for these scaffolds are still relatively limited. Owing to their highly polar and polyhydroxylated nature, such compounds typically exhibit high aqueous solubility but may suffer from low membrane permeability and limited oral bioavailability, a trend commonly observed for glycosidase inhibitors and related aminocyclitols. While many members of this class have demonstrated low intrinsic toxicity in biological assays, systematic investigations addressing absorption, distribution, metabolism, and long-term safety remain underexplored.
In addition, the widespread use of transition-metal catalysts (e.g., Ru in RCM, Pd in cross-coupling, Os in dihydroxylation) necessitates careful consideration of residual metal content in the final active pharmaceutical ingredient (API), in accordance with ICH Q3D guidelines. Although most methodologies discussed herein are developed at laboratory scale, established process-chemistry strategies—such as the use of metal scavengers (e.g., silica-supported thiols, amines, or phosphines), crystallization-based purification, aqueous workups, and chromatographic techniques—are effective in reducing residual metal levels to acceptable limits. Furthermore, the development of immobilized catalysts, recyclable catalytic systems, and low-metal-loading protocols provides promising avenues for minimizing metal contamination at the source.
More broadly, this review aims to provide a unified and critically connected perspective on the synthesis of conduritols, conduramines, and their related cyclitol frameworks, which have often been treated in a fragmented manner across the literature. By systematically compiling chemoenzymatic, carbohydrate-derived, chiral pool, and catalytic strategies within a single framework, this work highlights underlying design principles, recurring mechanistic motifs, and points of convergence between seemingly distinct synthetic approaches. In contrast to earlier reviews that have primarily focused on specific subclasses of cyclitols or individual synthetic methodologies, the present review emphasizes a comparative and integrative analysis, enabling clearer identification of strengths, limitations, and opportunities across the field. It is anticipated that this consolidated perspective will not only assist synthetic chemists in selecting appropriate strategies but also help bridge existing gaps between methodology development, scalability, and medicinal chemistry applications, thereby guiding future research toward more efficient, sustainable, and application-oriented cyclitol synthesis.
Taken together, these considerations highlight that, alongside synthetic innovation, future progress in this field will also depend on improving pharmacokinetic properties, ensuring regulatory compliance, and developing scalable, environmentally responsible processes. Consequently, the continued development of efficient, scalable, and sustainable synthetic strategies for conduritols and their derivatives is expected to play a pivotal role in advancing glycosidase inhibitor research, natural product synthesis, and the discovery of new cyclitol-based therapeutic agents.
Author contributions
SM initially prepared the draft following the suggestions of AKM. AKM later modified and rewrote the manuscript according to the standard of the journal.Conflicts of interest
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.Data availability
No new data were created or analyzed in this study. Data sharing is not applicable to this article.Acknowledgements
The authors would like to acknowledge Prof. Ashutosh Ghosh, Vice-Chancellor of Rani Rashmoni Green University, for his guidance and encouragement throughout the process of preparing this manuscript. SM would also like to acknowledge the invaluable support of his parents.References
- J. Duchek, D. R. Adams and T. Hudlicky, Chem. Rev., 2011, 111, 4223–4258 Search PubMed.
- M. Balci, Y. Sütbeyaz and H. Secen, Tetrahedron, 1990, 46, 3715–3742 CrossRef CAS.
- M. S. Gultekin, M. Celik and M. Balci, Curr. Org. Chem., 2004, 8, 1159–1186 CrossRef.
- K. Kubler, Arch. Pharm., 1908, 246, 620–660 CrossRef.
- G. Dangschat and H. O. L. Fischer, Naturwissenschaften, 1939, 27, 756–757 CrossRef CAS.
- S. Malik and A. K. Mitra, Tetrahedron, 2025, 178, 134605 CrossRef CAS.
- (a) N. Verma, S. Arora, A. K. Singh and A. Kumar, Targets, 2025, 3, 32 CrossRef; (b) N. Verma, S. Arora, A. K. Singh and J. Ahmed, RSC Pharm., 2025, 2, 1201 RSC.
- D. C. Billington, F. Perron-Sierra, I. Picard, S. Beaubras, J. Duhault, J. Espinal and S. Challal, Bioorg. Med. Chem. Lett., 1994, 4, 2307–2312 CrossRef CAS.
- N. Şimşek Kuş, Chem. Biodiversity, 2024, 21, e202301064 CrossRef PubMed.
- T. Hudlicky, F. Rulin, T. Tsunoda, H. Luna, C. Andersen and J. D. Price, Isr. J. Chem., 1991, 31, 229–238 CrossRef CAS.
- D. Gonzalez, T. Martinot and T. Hudlicky, Tetrahedron Lett., 1999, 40, 3077–3080 CrossRef CAS.
- S. Elango and T.-H. Yan, J. Org. Chem., 2002, 67, 6954–6959 CrossRef CAS PubMed.
- M. Matveenko, M. G. Banwell and A. C. Willis, Tetrahedron, 2008, 64, 4817–4826 CrossRef CAS.
- S. Arora and C. Mao, Nucleosides, Nucleotides Nucleic Acids, 2024, 43, 316–339 CrossRef CAS PubMed.
- Y.-U. Kwon, C. Lee and S.-K. Chung, J. Org. Chem., 2002, 67, 3327–3338 CrossRef CAS PubMed.
- H. Çavdar, O. Talaz and D. Ekinci, Bioorg. Med. Chem. Lett., 2012, 22, 7499–7503 Search PubMed.
- G. Legler, Hoppe-Seyler’s Z. Physiol. Chem., 1966, 345, 197–214 CrossRef CAS PubMed.
- G. Legler, in Methods in Enzymology, Academic Press, 1977, vol. 46, pp. 368–381 Search PubMed.
- K. J. Lee, S. A. Boyd and N. S. Radin, Carbohydr. Res., 1985, 144, 148–154 CrossRef CAS PubMed.
- A. E. Gal and J. P. Voorstad, J. Labelled Compd. Radiopharm., 1987, 24, 397–407 CrossRef CAS.
- O. Werbitzky, K. Klier and H. Felber, Liebigs Ann. Chem., 1990, 1990, 267–270 CrossRef.
- S. Arora, J. Liang, S. K. Fullerton-Shirey and J. E. Laaser, ACS Mater. Lett., 2020, 2, 331–335 CrossRef CAS.
- T. Hudlicky, J. D. Price, F. Rulin and T. Tsunoda, J. Am. Chem. Soc., 1990, 112, 9439–9440 CrossRef CAS.
- H. A. J. Carless and K. Busia, Tetrahedron Lett., 1990, 31, 3449–3452 CrossRef CAS.
- M. C. McIntosh and S. M. Weinreb, J. Org. Chem., 1991, 56, 5010–5012 CrossRef CAS.
- K. Schürrle, B. Beier and W. Piepersberg, J. Chem. Soc., Perkin Trans. 1, 1991, 2407–2412 RSC.
- T. Hudlicky, H. Luna, H. F. Olivo, C. Andersen, T. Nugent and J. D. Price, J. Chem. Soc., Perkin Trans. 1, 1991, 2907–2917 RSC.
- H. A. J. Carless and O. Z. Oak, Tetrahedron Lett., 1991, 32, 1671–1674 CrossRef CAS.
- T. Hudlicky and H. F. Olivo, J. Am. Chem. Soc., 1992, 114, 9694–9696 CrossRef CAS.
- C. R. Johnson, P. A. Plé, L. Su, M. J. Heeg and J. P. Adams, Synlett, 1992, 1992, 388–390 CrossRef.
- F. Chretien, R. Wolf and Y. Chapleur, Nat. Prod. Lett., 1993, 2, 69–75 CrossRef CAS.
- M. Mandel and T. Hudlicky, J. Chem. Soc., Perkin Trans. 1, 1993, 741–743 RSC.
- (a) L. Pingli and M. Vandewalle, Tetrahedron, 1994, 50, 7061–7074 CrossRef CAS; (b) Y. Siitbeyaz, H. Seçen and M. Balcı, J. Chem. Soc., Chem. Commun., 1988, 1330 RSC.
- (a) C. R. P. Johnson, A. Patrick, S. Lin, H. Mary Jane and P. Adams Joseph, Synlett, 1992, 1992, 388–390 CrossRef; (b) H. Paulsen, W. Roben and F. R. Heiker, Chem. Ber., 1981, 114, 3242 CrossRef CAS.
- S. Allemann and P. Vogel, Helv. Chim. Acta, 1994, 77, 1–9 CrossRef CAS.
- (a) H. Seçen, S. Gültekin, Y. Sütbeyaz and M. Balci, Synth. Commun., 1994, 24, 2103–2108 CrossRef; (b) M. Nakajima, A. Hasegawa and N. Kurihara, Chem. Ber., 1962, 95, 2709 Search PubMed.
- R. Leung-Toung, Y. Liu, J. M. Muchowski and Y.-L. Wu, Tetrahedron Lett., 1994, 35, 1639–1642 CrossRef CAS.
- Y. Kara, M. Balcı, S. A. Bourne and W. H. Watson, Tetrahedron Lett., 1994, 35, 3349–3352 CrossRef.
- B. M. Trost and S. R. Pulley, J. Am. Chem. Soc., 1995, 117, 10143–10144 CrossRef CAS.
- O. Arjona, A. de Dios, J. Plumet and B. Saez, Tetrahedron Lett., 1995, 36, 1319–1320 CrossRef CAS.
- B. M. Trost and S. R. Pulley, Tetrahedron Lett., 1995, 36, 8737–8740 CrossRef CAS.
- J. L. Chiara and N. Valle, Tetrahedron:Asymmetry, 1995, 6, 1895–1898 CrossRef CAS.
- D. A. Entwistle and T. Hudlicky, Tetrahedron Lett., 1995, 36, 2591–2594 CrossRef CAS.
- A. Patti, C. Sanfilippo, M. Piattelli and G. Nicolosi, Tetrahedron:Asymmetry, 1996, 7, 2665–2670 CrossRef CAS.
- R. Angelaud, Y. Landais and K. Schenk, Tetrahedron Lett., 1997, 38, 1407–1410 CrossRef CAS.
- C. Sanfilippo, A. Patti, M. Piattelli and G. Nicolosi, Tetrahedron:Asymmetry, 1997, 8, 2083–2084 CrossRef CAS.
- V. Cerè, F. Peri and S. Pollicino, Tetrahedron Lett., 1997, 38, 7797–7800 CrossRef.
- M.-C. Lallemand, M. Desjardins, S. Freeman and T. Hudlicky, Tetrahedron Lett., 1997, 38, 7693–7696 Search PubMed.
- V. Cerè, F. Peri, S. Pollicino and A. Ricci, Synlett, 1998, 1998, 1197–1198 CrossRef.
- M. Banwell, S. Blakey, G. Harfoot and R. Longmore, J. Chem. Soc., Perkin Trans. 1, 1998, 3141–3142 RSC.
- R. Leung-Toung, Y. Liu, J. M. Muchowski and Y.-L. Wu, J. Org. Chem., 1998, 63, 3235–3250 CrossRef CAS.
- T. J. Donohoe, K. Blades, M. Helliwell, M. J. Waring and N. J. Newcombe, Tetrahedron Lett., 1998, 39, 8755–8758 CrossRef CAS.
- W.-W. Lee and S. Chang, Tetrahedron:Asymmetry, 1999, 10, 4473–4475 CrossRef CAS.
- T. Hudlicky, N. Restrepo-Sánchez, P. D. Kary and L. M. Jaramillo-Gómez, Carbohydr. Res., 2000, 324, 200–203 CrossRef CAS PubMed.
- J. K. Gallos, T. V Koftis, V. C. Sarli and K. E. Litinas, J. Chem. Soc., Perkin Trans. 1, 1999, 3075–3077 Search PubMed.
- H. Ovaa, J. D. C. Codée, B. Lastdrager, H. S. Overkleeft, G. A. van der Marel and J. H. van Boom, Tetrahedron Lett., 1999, 40, 5063–5066 CrossRef CAS.
- B. M. Trost and E. J. Hembre, Tetrahedron Lett., 1999, 40, 219–222 Search PubMed.
- A. Kornienko and M. d'Alarcao, Tetrahedron:Asymmetry, 1999, 10, 827–829 CrossRef.
- G. Mehta and S. Lakshminath, Tetrahedron Lett., 2000, 41, 3509–3512 Search PubMed.
- O. Plettenburg, S. Adelt, G. Vogel and H.-J. Altenbach, Tetrahedron:Asymmetry, 2000, 11, 1057–1061 Search PubMed.
- D. Spielvogel, J. Kammerer, M. Keller and H. Prinzbach, Tetrahedron Lett., 2000, 41, 7863–7867 CrossRef CAS.
- A. Falshaw, J. B. Hart and P. C. Tyler, Carbohydr. Res., 2000, 329, 301–308 Search PubMed.
- G. Mehta and S. S. Ramesh, Chem. Commun., 2000, 2429–2430 RSC.
- S. H. Lee and C. S. Cheong, Bull. Korean Chem. Soc., 2000, 21, 1061–1062 CAS.
- M. G. Banwell, A. M. Bray and D. J. Wong, New J. Chem., 2001, 25, 1351–1354 RSC.
- B. M. Trost, D. E. Patterson and E. J. Hembre, Chem.–Eur. J., 2001, 7, 3768–3775 CrossRef CAS PubMed.
- B. M. Trost Jr, D. Joseph and E. J. Hembre, Chem.–Eur. J., 2001, 7, 1619–1629 CrossRef.
- S. E. de Sousa, P. O'Brien and C. D. Pilgram, Tetrahedron Lett., 2001, 42, 8081–8083 CrossRef CAS.
- B. J. M. Paul, A. Theodore, W. Jerremey and H. Tomas, Synthesis, 2001, 2001, 952–956 CrossRef.
- A. Arcelli, V. Cerè, F. Peri, S. Pollicino and A. Ricci, Tetrahedron, 2001, 57, 3439–3444 CrossRef CAS.
- U. Rinner, P. Siengalewicz and T. Hudlicky, Org. Lett., 2002, 4, 115–117 CrossRef CAS PubMed.
- W. T. Lambert and S. D. Burke, Org. Lett., 2003, 5, 515–518 Search PubMed.
- G. Mehta, R. S. Senaiar and M. K. Bera, Chem.–Eur. J., 2003, 9, 2264–2272 CrossRef CAS PubMed.
- G. Mehta and S. S. Ramesh, Tetrahedron Lett., 2003, 44, 3105–3108 CrossRef CAS.
- A. Baran, C. Kazaz, H. Seçen and Y. Sütbeyaz, Tetrahedron, 2003, 59, 3643–3648 CrossRef CAS.
- M. Podeschwa, O. Plettenburg, J. vom Brocke, O. Block, S. Adelt and H.-J. Altenbach, Eur. J. Org. Chem., 2003, 2003, 1958–1972 CrossRef.
- M. A. L. Podeschwa, O. Plettenburg and H.-J. Altenbach, Org. Biomol. Chem., 2003, 1, 1919–1929 RSC.
- Y.-U. Kwon, J. Im, G. Choi, Y.-S. Kim, K. Yong Choi and S.-K. Chung, Bioorg. Med. Chem. Lett., 2003, 13, 2981–2984 CrossRef CAS PubMed.
- O. N. Nadein and A. Kornienko, Org. Lett., 2004, 6, 831–834 CrossRef CAS PubMed.
- C. Vitelio, A. Bellomo, M. Brovetto, G. Seoane and D. Gonzalez, Carbohydr. Res., 2004, 339, 1773–1778 CrossRef CAS PubMed.
- F. Orsini, G. Sello, S. Bernasconi and G. Fallacara, Tetrahedron Lett., 2004, 45, 9253–9255 CrossRef CAS.
- S. Saito, R. Shimazawa and R. Shirai, Chem. Pharm. Bull., 2004, 52, 727–732 CrossRef CAS PubMed.
- S. Freeman and T. Hudlicky, Bioorg. Med. Chem. Lett., 2004, 14, 1209–1212 Search PubMed.
- T. L. Andresen, D. M. Skytte and R. Madsen, Org. Biomol. Chem., 2004, 2, 2951–2957 Search PubMed.
- V. Cerè, M. Minzoni, S. Pollicino, A. Ricci, F. Gasparrini, A. Ciogli and I. D'Acquarica, J. Comb. Chem., 2006, 8, 74–78 CrossRef PubMed.
- P. Serrano, A. Llebaria and A. Delgado, J. Org. Chem., 2005, 70, 7829–7840 CrossRef CAS PubMed.
- R. Łysek, C. Schütz and P. Vogel, Bioorg. Med. Chem. Lett., 2005, 15, 3071–3075 CrossRef PubMed.
- L. Kelebekli, M. Çelik, E. Şahin, Y. Kara and M. Balci, Tetrahedron Lett., 2006, 47, 7031–7035 CrossRef CAS.
- R. Łysek, C. Schütz, S. Favre, A. C. O'Sullivan, C. Pillonel, T. Krülle, P. M. J. Jung, I. Clotet-Codina, J. A. Esté and P. Vogel, Bioorg. Med. Chem., 2006, 14, 6255–6282 CrossRef PubMed.
- A. Bellomo, C. Giacomini, B. Brena, G. Seoane and D. Gonzalez, Synth. Commun., 2007, 37, 3509–3518 Search PubMed.
- R. Łysek, S. Favre and P. Vogel, Tetrahedron, 2007, 63, 6558–6572 Search PubMed.
- G. Pandey, K. N. Tiwari and V. G. Puranik, Org. Lett., 2008, 10, 3611–3614 CrossRef CAS PubMed.
- Y.-K. Chang, H.-J. Lo and T.-H. Yan, Org. Lett., 2009, 11, 4278–4281 Search PubMed.
- C. K. Jana, S. Grimme and A. Studer, Chem.–Eur. J., 2009, 15, 9078–9084 Search PubMed.
- A. Bellomo, A. Bertucci, H. Stefani, Á. Vázquez and D. Gonzalez, Tetrahedron:Asymmetry, 2009, 20, 2673–2676 Search PubMed.
- S. Cantekin, A. Baran, R. Çalışkan and M. Balci, Carbohydr. Res., 2009, 344, 426–431 Search PubMed.
- D. D. Chappell, G. B. Michael, S. Gibson, L. M. Harwood and A. T. Russell, Synlett, 2010, 2010, 517–520 CrossRef.
- P.-H. Lu, C.-S. Yang, B. Devendar and C.-C. Liao, Org. Lett., 2010, 12, 2642–2645 Search PubMed.
- P. Ghosal and A. K. Shaw, J. Org. Chem., 2012, 77, 7627–7632 Search PubMed.
- S. Norsikian, J.-F. Soulé, A. Cannillo, R. Guillot, M.-E. Tran Huu Dau and J.-M. Beau, Org. Lett., 2012, 14, 544–547 Search PubMed.
- Z. Ekmekci and M. Balci, Eur. J. Org. Chem., 2012, 2012, 4988–4995 Search PubMed.
- A. Rajender and B. V. Rao, Tetrahedron Lett., 2013, 54, 2329–2331 Search PubMed.
- S. Kuno, K. Higaki, A. Takahashi, E. Nanba and S. Ogawa, Medchemcomm, 2015, 6, 306–310 Search PubMed.
- V. K. Harit and N. G. Ramesh, J. Org. Chem., 2016, 81, 11574–11586 Search PubMed.
- I.-S. Myeong, J.-S. Kim, Y.-T. Lee, J.-C. Kang, S.-H. Park, C. Jung and W.-H. Ham, Tetrahedron:Asymmetry, 2016, 27, 823–828 Search PubMed.
- R. Katakam, R. Anugula, L. Macha and V. R. Batchu, Tetrahedron Lett., 2017, 58, 559–562 Search PubMed.
- K. R. Prasad and V. A. Rangari, Tetrahedron, 2018, 74, 6689–6693 Search PubMed.
- V. K. Harit and N. G. Ramesh, Org. Biomol. Chem., 2019, 17, 5951–5961 Search PubMed.
- S. Da Silva Pinto, S. G. Davies, A. M. Fletcher, P. M. Roberts and J. E. Thomson, Org. Lett., 2019, 21, 7933–7937 Search PubMed.
- K. Aksu, H. Akincioglu, I. Gulcin and L. Kelebekli, Arch. Pharm., 2021, 354, 2000254 Search PubMed.
- F. Laar, Vasc. Health Risk Manage., 2008, 4, 1189–1195 Search PubMed.
- N. Asano, Glycobiology, 2003, 13, 93R–104R Search PubMed.
- H. E. Lebovitz, Endocrinol. Metab. Clin. North Am., 1997, 26, 539–551 Search PubMed.
- E. Truscheit, W. Frommer, B. Junge, L. Müller, D. D. Schmidt and W. Wingender, Angew Chem. Int. Ed. Engl., 1981, 20, 744–761 Search PubMed.
- S. Arora and N. Verma, ChemistrySelect, 2026, 11, e02016 Search PubMed.
- S. Arora and N. Verma, RSC Appl. Polym., 2024, 2, 317–355 Search PubMed.
- S. Arora, J. Rozon and J. E. Laaser, Macromolecules, 2021, 54, 6466–6476 Search PubMed.
- Z. Huo, S. Arora, V. A. Kong, B. J. Myrga, A. Statt and J. E. Laaser, Macromolecules, 2023, 56, 1845–1854 Search PubMed.
- J. Liang, K. Xu, S. Arora, J. E. Laaser and S. K. Fullerton-Shirey, Materials, 2020, 13, 1089 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |