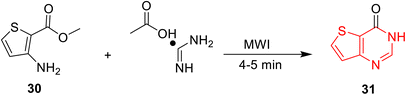Open Access Article
Open Access ArticleRecent green approaches for the synthesis of thienopyrimidine scaffolds: an overview (2007–2025)
Rofaida M. Rabeh
 a,
Asmaa E. Kassab
a,
Asmaa E. Kassab
 b,
Rasha A. Hassan
b,
Rasha A. Hassan
 *b and
Inas G. Shahin
a
*b and
Inas G. Shahin
a
aOrganic Chemistry Department, Faculty of Pharmacy, October University for Modern Sciences and Arts (MSA), Giza 12451, Egypt
bPharmaceutical Organic Chemistry Department, Faculty of Pharmacy, Cairo University, Kasr El-Aini Street, Cairo 11562, Egypt. E-mail: rasha.hasan@pharma.cu.edu.eg
First published on 27th March 2026
Abstract
Thienopyrimidines are considered purine bioisosteres as they consist of a thiophene ring fused with pyrimidine. Thienopyrimidines are present in three isomeric forms: thieno[2,3-d]pyrimidines, thieno[3,2-d]pyrimidines, and thieno[3,4-d]pyrimidines. They are of interest in drug discovery owing to their extensive pharmacological and biological activities. Several drugs incorporating the thienopyrimidine scaffold are marketed commercially, while others are presently in the experimental and development stages. Green chemistry, also known as sustainable chemistry, aims to minimize the environmental impact of chemical products and processes. Thus, applying green techniques has become a necessity in organic chemistry. The current review discusses the different synthetic approaches to obtain thienopyrimidine scaffolds using various green techniques, including microwave-assisted synthesis, green solvents, one-pot reactions, solvent-less reactions, and catalysis, published between 2007 and 2025. Additionally, a brief overview of the conventional synthetic routes is provided. This review emphasizes how green chemistry is becoming increasingly important in promoting the synthesis of bioactive heterocycles such as thienopyrimidines.
1. Introduction
1.1. Green chemistry
Green chemistry is defined by the U.S. Environmental Protection Agency (EPA) as chemistry that prevents pollution of the environment through the design of chemical products and processes that are harmless to the environment.1,2 Also, green chemistry is known as sustainable chemistry that aims to minimize the environmental impact of chemical products and processes. The 12 principles of green chemistry involve the use of renewable resources, the reduction of energy waste, the reduction of hazardous chemicals, and the use of safer solvents. One of the most prominent challenges in the synthesis of organic chemicals is avoiding costly and harmful reagents as well as reducing energy consumption.3,4 As a result, one of the most prominent goals of green chemistry is the creation of environmentally safe chemical processes,1 which produce chemical products that eventually break down after their application into components that are harmless and do not remain in the environment.2 Recently, the industrial and chemical research fields have focused on developing eco-friendly green reactions.5,6 In addition, both local and large companies are focused on enhancing synthetic techniques to produce a variety of heteroatom moieties with maximum yields.7 While most other conventional methods are highly profitable, they are not eco-friendly because they produce a considerable amount of waste and by-products.8 Therefore, green chemistry provides a promising approach for meeting the needs of the medical sector.9 Various green techniques can be utilized in chemical processes, including microwave-assisted techniques, synthesis using green solvents, catalysis, solvent-less synthesis, and one-pot reactions.To quantitatively measure how much these green techniques make a synthesis method more efficient and eco-friendly, some indicators can be used for evaluating its greenness. These indicators are known as “green chemistry metrics”. Green chemistry metrics include atom economy (AE) or atom efficiency, process mass intensity (PMI), real atom economy (RAE), environmental factor (E-factor), and reaction mass efficiency (RME). We will focus on AE, which is the ratio between the product formula weight to the formula weight of all reactants used. The higher the AE%, the greener the process (optimal value is 100%). Also, we focus on PMI, which is the ratio between the total mass of all utilized reactants to the mass of the product. The lower the PMI, the less waste produced (optimal value is 1). Additionally, we focus on the E-factor, which is the ratio between the mass of waste to the mass of the target product. The lower the E-factor, the less waste produced (optimal value is 0).21
Heterocyclic compounds, especially nitrogen-containing heterocycles, play an important role in organic research and therapeutic chemistry due to their diverse pharmacological and biological actions.22–26 Thienopyrimidine is one of these nitrogen-containing heterocycles that has gained momentum. Thienopyrimidine scaffolds are of interest in drug discovery owing to their extensive pharmacological and biological activities.27–32 Thienopyrimidine derivatives have been discovered to exhibit anti-cancer,33–43 anti-infective,44 antioxidant,45 antimicrobial,46–48 and anti-tuberculosis49 activities. Furthermore, currently, different thienopyrimidine derivatives are undergoing clinical trials and are utilized in medical applications. For instance, relugolix (TAK-385) is in phase III and has been approved for the treatment of advanced hormone-sensitive prostate cancer in the European Union and for advanced prostate cancer in the USA.50 Another example of a thienopyrimidine derivative undergoing phase a II trial is PRX-08066, which acts as a potent serotonin 5-HT subtype 2B receptor antagonist and is utilized for pulmonary diseases, particularly pulmonary arterial hypertension. Furthermore, it was revealed that it can prevent the activation of fibroblasts, making it a good candidate as an antitumor agent.51,52 In addition, apitolisib (GDC-0980) showed a significant anti-tumor effect in PI3K hyperactivation or PTEN loss tumor models.53,54 Similarly, pictilisib (GDC-0941) and fimepinostat (CUDC-907) are in phase II. Pictilisib is used for the treatment of non-Hodgkin's lymphoma, metastatic breast cancer, and advanced solid tumors.53,55,56 Fimepinostat is used against breast cancer, multiple myeloma, and lymphoma.57 Finally, olmutinib is a thienopyrimidine marketed as an EGFR tyrosine kinase inhibitor; it demonstrated high efficacy and showed controlled safety in patients with T790M-positive non-small cell lung cancer58 (Fig. 1). Although several reviews discussed the synthesis and biological importance of thienopyrimidine derivatives,36,44,59–65 this review focuses specifically on the green synthetic strategies reported over the last two decades.
2. Synthesis of thienopyrimidine derivatives
2.1. Conventional synthesis
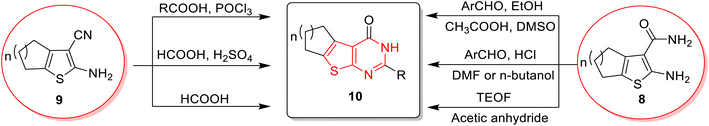
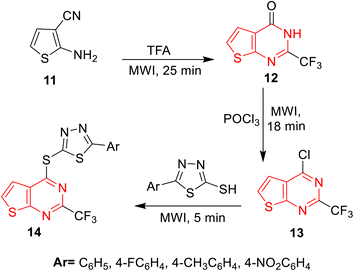
Thienopyrimidine can be synthesized through different synthetic pathways. The most prominent pathway is the Gewald reaction, which provides the key precursors for thienopyrimidine synthesis. The reaction usually starts from 2-aminothiophene-3-carboxylate 1, 2-aminothiophene-3-carboxamide 8, or 2-aminothiophene-3-carbonitrile 9. Elmongy and colleagues45 synthesized 3-amino-2-methyl thienopyrimidine derivatives 2 through the reaction of 2-aminothiophene-3-carboxylate 1 with acetic anhydride, followed by cyclization using hydrazine hydrate in ethanol. Kim and co-workers66 reported the synthesis of thienopyrimidine derivatives 3 using 2-aminothiophene-3-carboxylate derivative 1 as a precursor, which reacted with 1,1′-thiocarbonylbis(pyridin-2(1H)-one) to convert the amino group into an isothiocyanate group. The isothiocyanate product reacted with substituted anilines to produce thiourea derivatives, which were then cyclized with the help of alcoholic sodium hydroxide. 2-Mercapto-thienopyrimidin-4-one 5 was afforded by reacting 2-aminothiophene-3-carboxylate with different isothiocyanates, giving rise to thiourea derivatives 4, followed by cyclization with potassium hydroxide in ethanol;67,68 likewise it was cyclized using hydrazine hydrate69 or potassium carbonate in acetonitrile.70 Another pathway for the synthesis of thienopyrimidinone from thiophene aminoester is through reaction with formamidine acetate in dimethyl formamide (DMF)71–73 or with formamide.74–76 Sheta et al.77 prepared imidazothienopyrimidine 7 by reacting the starting material, substituted 2-aminothiophene-3-carboxylate 1, with alkylated thioimidazolone in acetic acid and ethanol (Scheme 1).A different approach for thienopyrimidine synthesis was the reaction of 2-aminothiophene-3-carboxamide 8 with substituted benzaldehyde in glacial acetic acid and DMSO78 or substituted benzaldehyde and HCl in DMF79,80 or in n-butanol,69 yielding thienopyrimidinone derivatives 10.81 Moreover, they can be obtained by reacting 2-aminothiophene-3-carboxamide 8 with triethyl orthoformate (TEOF) and acetic anhydride.79 Furthermore, 2-aminothiophene-3-carbonitrile 9 provided thienopyrimidinone derivatives through a reaction with aliphatic acids and phosphorus oxychloride (POCl3),82 with formic acid and sulphuric acid,83 or formic acid only84–86 (Scheme 2). However, these conventional methods have a negative impact on the environment due to the use of harmful solvents, high energy consumption, and waste production. Thus, it has become necessary to replace these harmful conventional methods with more green ones that avoid the use of solvents or use greener solvents and the use of high temperatures, and involve multiple-step reactions. Therefore, applying green techniques is more eco-friendly, time-saving, and cost-saving.87–89
2.2. Green synthesis
In the same year, Poojari and coworkers,91 described an efficient microwave-assisted approach for synthesizing 4-hydroxythieno[2,3-d]pyrimidine compounds. This method utilized 2-aminothiophene-3-carboxamide derivatives 15 as precursors, which underwent a one-pot reaction with various acids 16 at 120 °C for 20 min in the presence of propylphosphonic anhydride (T3P) and triethylamine (TEA), furnishing amide intermediate 17, which was then cyclized to produce the target product 18 in a yield of up to 99% and an AE of 88.1% (Scheme 4). This method is highly efficient, rapid, and gentle, as the use of T3P avoids the use of a strong acidic medium.
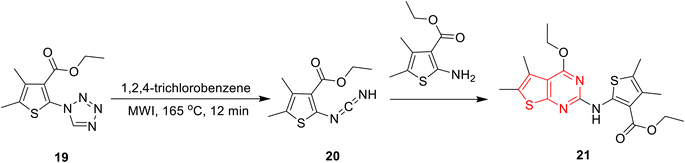
According to Soares et al.,92 4-ethoxythieno[2,3-d]pyrimidine 21 can be obtained through microwave-induced thermolysis of 4,5-dimethyl-2-(1H-tetrazol-1-yl)thiophene-3-carboxylate (19) (Scheme 5). This was explained by initial nitrogen elimination, followed by rearrangement into the respective carbodiimide and subsequent cyclization owing to the nucleophilic attack of the in situ-generated aminothiophene. Although the yield of this method is slightly low at 46%, the AE is very high (93.5%), with a PMI of 1.04 and E-factor of 0.04.
Di Fruscia and co-authors93 reported the synthesis of thieno[2,3-d]pyrimidin-4-one scaffold through the Gewald condensation reaction, where ketone 22 reacted with methyl cyanoacetate and elemental sulfur in the presence of diethylamine (DEA) as a catalyst to produce compound 23. Microwave-assisted cyclization is further performed in the presence of formamide and ammonium formate to afford the desired thienopyrimidone derivative 24, which shows a neuroprotective effect in Parkinson's disease (Scheme 6).
Gill et al.94 outlined the synthesis of 4-chloro-5,6-disubstituted-thieno[2,3-d]pyrimidine via the Gewald reaction under MWI for 2–9 min to yield 2-amino-3-carboethoxy thiophenes 26. Subsequently, cyclization of newly formed thiophene derivatives 26 was conducted with formamide under neat conditions and MWI for 20–25 min to yield compounds 27, followed by chlorination with POCl3 under MWI for 12–15 min to produce 4-chloro-5,6-disubstituted thieno[2,3-d]pyrimidine 28. Then, nucleophilic displacement of the chloride atom with substituted aniline afforded the desired product 29, which has antibacterial activity (Scheme 7). Compared to traditional procedures, MWI enables the synthesis of target products with enhanced purity, yields, and reaction time.
Meanwhile, Wang et al.95 introduced microwave-assisted cyclization of methyl-3-aminothiophene-2-carboxylate (30) with formamidine acetate to give thieno[3,2-d]pyrimidine-4(3H)-one (31) (Scheme 8). This reaction provides a good yield of 76% and a shorter reaction time, but its PMI and E-factor are high at 2.4 and 1.4, respectively, indicating high loss of the reactants.
Accordingly, Sureja and collaborators47 reported the reaction of 2-amino-4,5-substituted thiophene-3-carbonitrile 32 with different acids. The reaction was carried out in the presence of aluminium oxide and POCl3 under MWI for 2–4 min, affording the antimicrobial 2,5,6-substituted thieno[2,3-d]pyrimidine-4(3H)-one derivatives 33 in yields ranging from 78% to 90%. This method is superior to the conventional method, which requires reflux for 1.5–2.5 h and gives yields of 70–83% (Scheme 9).
In 2018, a series of N-substituted thieno[2,3-d]pyrimidin-4-amines was synthesized starting with the typical Gewald reaction, but the reaction was upgraded with 4-methylpiperidine, which is an efficient and inexpensive catalyst; this led to a shorter reaction time and provided a higher yield of 2-aminothiophene-3-carbonitrile 34. Eventually, polymerization, cyclization, and Dimroth rearrangement occurred under MWI, affording the desired product 36 in 71–95% yield (Scheme 10). This method is efficient as it is simple, shortens the reaction time, and produces higher yields compared to the conventional method, which requires a longer time of 0.5–5 h and gives yields of 64–90%.96
Two years later, Sun et al.97 applied the same method to synthesize thiophene[2,3-d]pyrimidine derivatives 39 (Scheme 11). MWI was used in all steps of the synthesis, thereby minimizing the reaction time and providing a higher yield.
Li and coworkers developed a new method for the synthesis of a thieno[2,3-d]pyrimidine-2,4(1H,3H)-dione derivative (42) using an ionic liquid, [HDBN+][TFE−], which is a simple and easily prepared solvent. The reaction occurs in the presence of CO2, which is inexpensive, abundant, and renewable, and a green source of carbon atoms. This reaction is simple, effective, and produces a high yield of 94%, with the optimal AE (100%), PMI (1), and E-factor (0)99 (Scheme 13).
In 2017, Abaee et al.101 developed a new approach for the synthesis of 2-phenylthieno[2,3-d]pyrimidine through a one-pot Gewald reaction. The reaction was performed by concurrently mixing the four reaction components: cyclohexanone, malononitrile, elemental sulfur, and benzonitrile simultaneously in the presence of NaOH as a base and tert-butanol as a solvent for 3 h at 50 °C. The targeted product 2-phenylthieno[2,3-d]pyrimidin-4-amine 47 was afforded in a very high yield of 95%, with an AE of 75.35%, a low PMI of 1.3, and an E-factor of 0.3 (Scheme 15).
Meanwhile, Wilding and colleagues65 reported a one-pot method for the synthesis of 2-aminothiophene[2,3-d]pyrimidin-4-one (50). Initially, reacting 4-chloropyrimidine with sodium hydrosulfide provided mercaptopyrimidine derivative 49, which was not separated from the reaction mixture. Hence, α-chloroacetaldehyde and K2CO3 as a catalyst were added to obtain the desired product 50. Dimerization of the sulphydryl intermediate was prevented by carrying out the reaction under inert conditions (Scheme 16).
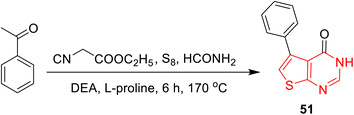
Shi and associates102 reported a new strategy for the synthesis of 5-phenylthieno[2,3-d]pyrimidine-4(3H)-one (51) through a one-pot reaction without intermediate isolation, instead of the conventional method that involved three steps. The reaction comprises coupling all the reaction components, acetophenone, ethyl cyanoacetate, sulfur, and formamide, in one vessel at 170 °C for 6 h. In addition, the reaction was performed in the presence of DEA and L-proline as catalysts, affording AE of 73.5%, PMI of 2.5, and E-factor of 1.5 (Scheme 17).
 | ||
| Scheme 17 Multicomponent approach to the synthesis of 5-phenylthieno[2,3-d]pyrimidin-4(3H)-one (51). | ||
Shi et al.103 synthesized 5,6-substituted thieno[2,3-d]pyrimidin-4-amine derivatives 52 through a multicomponent reaction; hence, the reaction was rapid, efficient, provided a high yield reaching up to 96%, and shortened the reaction time immensely. The procedure involved incorporating ketone, malononitrile, sulfur, and formamide under catalysis with Na2HPO4 and triphenylphosphine (PPh3), which inhibits the dimerization of product 52 (Scheme 18). The product shows multi-targeted kinase inhibitory effect. The high AE (80.8%), low PMI (1.3), and low E-factor (0.3) indicate the effective incorporation of all the reactants in the reaction with minimal waste production.
 | ||
| Scheme 18 Synthesis of 5,6-substituted thieno[2,3-d]pyrimidin-4-amine 52 via a one-pot Na2HPO4-catalyzed reaction. | ||
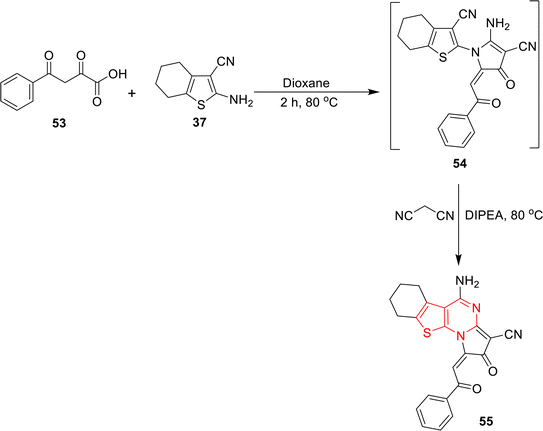
A series of substituted pyrrolo[1,2-α]thieno[3,2-e]pyrimidine 55 was synthesized via a one-pot reaction. In this reaction, the three components, 2,4-dioxo-4-phenylbutanoic acid (53), malononitrile, and 2-amino-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carbonitrile (37), were combined with dioxane as the solvent and N,N-diisopropylethylamine (DIPEA) as a catalyst. The reaction was refluxed at 80 °C for 2 h to obtain product 55 with a high AE (78.28%).104 The obtained product shows antitumor activity (Scheme 19).
In their study, Bakavoli and collaborators107 investigated an innovative and efficient method for the synthesis of 2-aryl-5,6-dimethylthieno[2,3-d]pyrimidine 60, which has antibacterial activity. According to the Gewald reaction, butan-2-one, malononitrile, S8, and DEA reacted to provide 2-amino-4,5-dimethylthiophene-3-formonitrile (58). Later, derivative 58 underwent acidic hydrolysis to produce 59, which undergoes nucleophilic addition with a series of aromatic aldehydes in acetonitrile under the catalysis of iodine at room temperature for only 15–30 min and finally cyclization to obtain the target product 60 (Scheme 21). The reaction proceeded with a low PMI (1.1), good E-factor (0.1), and high AE (89.6%).
An organo-catalyzed method was investigated for the synthesis of 2-phenylbenzo[4,5]thieno[2,3-d]pyrimidin-4(3H)-one (62). The reaction was conducted by aerobic cross-coupling of 2-amino-3-carboxamide derivative 61 with benzylamine using ortho-naphthoquinone (o-NQ1) catalyst and TFA as a co-catalyst.108 The use of these catalysts helps to overcome the defects of the method reported by the Nguyen group involving closed systems, high temperatures, and metal catalysts, resulting in lower yields109 (Scheme 22).
In a recent study, 2-phenylthieno[2,3-d]pyrimidin-4-one 65 could be prepared under catalysis by either the same method (Scheme 22) of using an ortho-naphthoquinone (o-NQ1) catalyst and TFA as a co-catalyst108 or by cyclization with benzaldehyde, which was performed in acetonitrile and iodine was used as a catalyst. Consequently, as with all the catalyzed reactions, the conditions were substantially improved, as seen by the shorter reaction time and stirring at room temperature. In addition, this reaction showed greenness with a high AE (82.2%), low PMI (1.2), and good E-factor (0.2)110 (Scheme 23).
In 2016, Narender et al.112 investigated a new cyclization method for 2-substituted aminothieno[2,3-c]pyridine-3-carboxylate derivatives 66. Cyclization occurs through stirring overnight with hydrazine hydrate and ethanol at room temperature. The reaction afforded the desired thieno[2,3-d]pyrimidine derivatives 67 in a good yield of about 65–71%. Later, 67 reacted with substituted aldehydes to give derivatives 68, which have antimycobacterial activity (Scheme 24).
In 2019, a cyclization reaction between aminothiophene derivatives and formamide at room temperature was researched to provide the 7-substituted thieno[3,2-d]pyrimidin-4(3H)-one with a good yield of 60–65% and good AE of 75.2%. Additionally, it could be synthesized via acetylation of aminothiophene with acetic anhydride or by reacting aminothiophene with α-bromoketone in the presence of TEA and dichloromethane (DCM) under mild conditions. Product 70 was reacted with 30% NH4OH to afford product 71 in high yields of 75–85% (ref. 113) (Scheme 25).
Davoodnia and co-authors114 investigated a one-pot, base-catalyzed cyclocondensation of 2-aminothiophene-3-carboxylate with aryl isocyanates to obtain a series of 3-arylthieno[2,3-d]pyrimidin-2,3(1H,3H)-diones 74 (Scheme 26).
A new approach was established for the synthesis of thieno[2,3-d]pyrimidine derivatives 77 using various 2-aminothiophene-3-carboxylates with different arylamines 76 and TEOF. The reaction was performed under MWI and zinc acetate Zn(OAc)2 catalysis. This approach reduced the reaction time to only 5 min and increased the yield to 72–95%115 (Scheme 27).
Through the same previous method, 2,5-diaminothiophene-3,4-dicarboxylate 78 reacted with aniline to afford compound 79 in only 5 min, 88% yield, and 68.2% AE115 (Scheme 28).
In 2017, 3-hydroxythieno[2,3-d]pyrimidine derivatives 81 were prepared through the Gewald reaction, in which 4-chloroacetophenone reacted with ethyl cyanoacetate in the presence of sulfur and morpholine as a base, but the reaction was enhanced by MWI. After that, the produced ortho amino ester intermediate 80 was cyclized under the catalysis of p-toluenesulfonic acid (p-TSA hydrate) in methanol at room temperature for 2–3 h (ref. 116) (Scheme 29). This reaction provides a very high AE (93.9%) and low PMI (1.06) and E-factor (0.06), which align with the greenness. Also, the prepared 3-hydroxythieno[2,3-d]pyrimidine derivatives 81 have considerable antiviral activity.
Ghayour and associates117 developed a metal oxide nanocomposite-catalyzed synthetic route for 2-arylthieno[2,3-d]pyrimidine 83. In this method, 2-aminothiophene-3-carboxamide condensed with a panel of aromatic aldehydes under zinc oxide–cerium oxide (ZnO–CeO2) catalysis and neat conditions for 1.5 h. After that, cyclization and oxidation occur to produce the desired thieno[2,3-d]pyrimidine derivatives in a good yield of 95% (Scheme 30). Metal oxide nanocomposites are of great interest in catalysis because they are easily available, affordable, have a large surface area, and are reusable, meeting green chemistry requirements. The reaction conditions allow achieving a high AE (93.4%) and very low E-factor (0.07) and PMI (1.07), minimizing waste production.
Shyyka and collaborators118 reported a thermally activated trans-annulation for 2-aminothieno[2,3-d]pyrimidine 87. In this method, substituted-2-tetrazolylthiophene-3-carboxylate 84 was reacted with different cage-like amines in one-pot under solvent-free conditions at 150 °C-160 °C for 35–40 min, yielding the transannular thieno[2,3-d]pyrimidine with anticancer activity in a yield of 84–89%. This reaction involves three steps: cleavage of the tetrazole ring, nucleophilic addition, and finally cyclization (Scheme 31).
3. Conclusion and future insights
Thienopyrimidine scaffolds represent distinctive lead compounds in drug discovery due to their various biological activities. Therefore, synthesizing new compounds containing thienopyrimidine is worthwhile. Developing eco-friendly synthetic pathways as alternatives to traditional ones helps to minimize waste production and reduce pollution. In this review, we have outlined different green principles and their applications in thienopyrimidine synthesis, for instance, microwave irradiation, green solvents, catalysis, and solvent-free and one-pot techniques. Despite the prominent progress achieved in designing eco-friendly approaches for the synthesis of thienopyrimidines, further progress is required to improve selectivity, efficiency, and scalability. Future research should focus on using renewable feedstock and biocatalysts that offer higher efficiency and selectivity. In addition, a combination of more than one green technique simultaneously can help in reducing the energy consumption, reaction time, and cost. We hope that this review helps medicinal chemists design novel thienopyrimidines for the treatment of many diseases through eco-friendly methods and avoiding the traditional polluting methods.Conflicts of interest
The authors declare no competing financial or personal interests that could have influenced this work.Data availability
No new data were generated or analyzed in support of this review article. All data discussed are from previously published studies and are properly cited within the manuscript.References
- A. Kumar, Y. Kuang, Z. Liang and X. Sun, Mater. Today Nano, 2020, 11, 100076 CrossRef
.
- A. Ivanković, A. Dronjić, A. M. Bevanda and S. Talić, Int. J. Sustainable Energy, 2017, 6, 39–48 CrossRef
.
- B. Borah, M. Patat, S. Swain and L. R. Chowhan, ChemistrySelect, 2022, 7, e202202484 CrossRef CAS
.
- J. Płotka-Wasylka, A. Kurowska-Susdorf, M. Sajid, M. de la Guardia, J. Namieśnik and M. Tobiszewski, ChemSusChem, 2018, 11, 2845–2858 CrossRef PubMed
.
- K. Kajal, R. Shakya, M. Rashid, V. Nigam, B. D. Kurmi, G. D. Gupta and P. Patel, Sustainable Chem. Pharm., 2024, 37, 101374 CrossRef CAS
.
- V. Bressi, A. Ferlazzo, D. Iannazzo and C. Espro, Nanomaterials, 2021, 11, 1120 CrossRef CAS PubMed
.
- M. Rafiq, M. Shafique, A. Azam, M. Ateeq, I. A. Khan and A. Hussain, Molecules, 2020, 25, 3901 CrossRef CAS PubMed
.
- M. Fraga-Corral, P. Ronza, P. Garcia-Oliveira, A. Pereira, A. Losada, M. Prieto, M. Quiroga and J. Simal-Gandara, Trends Food Sci. Technol., 2022, 119, 23–35 CrossRef CAS
.
- F. G. Calvo-Flores and C. Mingorance-Sánchez, ChemistryOpen, 2021, 10, 815–829 CrossRef CAS PubMed
.
- M. S. Phoujdar, M. K. Kathiravan, J. B. Bariwal, A. K. Shah and K. S. Jain, Tetrahedron Lett., 2008, 49, 1269–1273 CrossRef CAS
.
- A. Imtiyaz and A. Singh, J. Inorg. Organomet. Polym. Mater., 2023, 33, 3027–3048 CrossRef CAS
.
- Z. Kou, Z. Zhao, H. Li and X. Gao, Chem. Eng. Process., 2023, 184, 109300 CrossRef CAS
.
- S. Kumar and S. Kumar, Tetrahedron, 2024, 134437 Search PubMed
.
- X. Pi, J. Lu, S. Li, J. Zhang, Y. Wang and H. He, Sep. Purif. Technol., 2025, 361, 131585 CrossRef CAS
.
- B. Martinengo, E. Diamanti, E. Uliassi and M. L. Bolognesi, ACS Infect. Dis., 2024, 10, 1856–1870 CrossRef CAS PubMed
.
- G. Rothenberg, Catalysis: Concepts and Green Applications, John Wiley & Sons, 2017 Search PubMed
.
- B. M. Shaoo and B. K. Banik, in Green Approaches in Medicinal Chemistry for Sustainable Drug Design, Elsevier, 2024, pp. 387–408 Search PubMed
.
- L. Xian, C.-T. Ma, Y.-G. Ouyang, J.-Q. Di and Z.-H. Zhang, Appl. Organomet. Chem., 2020, 34, e5921 CrossRef CAS
.
- R. Kamani, D. Raval, K. Patel, V. Prajapati, R. Prajapati, U. Shah, N. Patel, A. Patel, M. Patel and S. N. Mali, J. Mol. Struct., 2025, 1322, 140295 CrossRef CAS
.
- J. Guo, Z. Yu, M. Das and L. Huang, ACS Nano, 2020, 14, 5075–5089 CrossRef CAS PubMed
.
- N. Fantozzi, J.-N. Volle, A. Porcheddu, D. Virieux, F. García and E. Colacino, Chem. Soc. Rev., 2023, 52, 6680–6714 RSC
.
- S. E. Seif, Z. Mahmoud, W. W. Wardakhan, A. M. Abdou and R. A. Hassan, Drug Dev. Res., 2023, 84, 839–860 CrossRef CAS PubMed
.
- S. E. Seif, W. W. Wardakhan, R. A. Hassan, A. M. Abdou and Z. Mahmoud, Drug Dev. Res., 2024, 85, e70007 CrossRef CAS PubMed
.
- J. Dhuguru, E. Zviagin and R. Skouta, J. Pharm., 2022, 15, 66 CAS
.
- W. Hou and H. Xu, J. Med. Chem., 2022, 65, 4436–4456 CrossRef CAS PubMed
.
- Zabiulla, F. H. Al-Ostoot, M. Al-Ghorbani and S. A. Khanum, J. Iran. Chem. Soc., 2022, 19, 23–54 CrossRef CAS
.
- M. T. M. Sayed, P. A. Halim, A. K. El-Ansary and R. A. Hassan, Drug Dev. Res., 2023, 84, 1299–1319 CrossRef CAS PubMed
.
- A. E. Mghwary, R. A. Hassan, P. A. Halim and M. K. Abdelhameid, Bioorg. Chem., 2025, 107985 CrossRef CAS PubMed
.
- A. E. Rashad, A. H. Shamroukh, R. E. Abdel-Megeid and W. A. El-Sayed, Synth. Commun., 2010, 40, 1149–1160 CrossRef CAS
.
- S. Abuelhassan, E. A. G. Bakhite, A. E. Abdel-Rahman and A. F. El-Mahdy, J. Heterocycl. Chem., 2021, 58, 1784–1801 CrossRef CAS
.
- D. R. Gorja, K. S. Kumar, K. Mukkanti and M. Pal, Beilstein J. Org. Chem., 2011, 7, 338–345 CrossRef CAS PubMed
.
- A. E. Rashad, A. H. Shamroukh, H. H. Sayed, S. M. Awad and N. A. Abdelwahed, Synth. Commun., 2011, 41, 652–661 CrossRef CAS
.
- R. A. Hassan, A. Sonousi, A. M. Abdou and Y. El-Dash, Bioorg. Chem., 2025, 108839 CrossRef CAS PubMed
.
- M. T. M. Sayed, Z. Mahmoud, M. M. Said, A. M. Abdou and R. A. Hassan, Bioorg. Chem., 2025, 109003 CrossRef CAS PubMed
.
- H. K. Abd El-Mawgoud, A. M. AboulMagd, M. T. Nemr, M. M. Hemdan, A. I. Hassaballah and P. S. Farag, Bioorg. Chem., 2024, 150, 107622 CrossRef CAS PubMed
.
- M. T. M. Sayed, R. A. Hassan, P. A. Halim and A. K. El-Ansary, Med. Chem. Res., 2023, 32, 659–681 CrossRef CAS
.
- A. Rogova, I. A. Gorbunova, T. E. Karpov, R. Y. Sidorov, A. E. Rubtsov, D. A. Shipilovskikh, A. R. Muslimov, M. V. Zyuzin, A. S. Timin and S. A. Shipilovskikh, Eur. J. Med. Chem., 2023, 254, 115325 CrossRef CAS PubMed
.
- T. Wang, F. Wu, L. Luo, Y. Zhang, J. Ma and Y. Hu, J. Mol. Struct., 2022, 1256, 132497 CrossRef CAS
.
- S. N. Milik, A. K. Abdel-Aziz, M. M. El-Hendawy, R. I. El-Gogary, M. K. Saadeldin, S. Minucci, C. D. Klein and K. A. Abouzid, J. Mol. Struct., 2022, 1259, 132724 CrossRef CAS
.
- M. Sharaky, M. Kamel, M. A. Aziz, M. Omran, M. M. Rageh, K. A. Abouzid and S. A. Shouman, J. Enzyme Inhib. Med. Chem., 2020, 35, 1641–1656 CrossRef CAS PubMed
.
- S. Zhang, F. Liu, X. Hou, J. Cao, X. Dai, J. Yu and G. Huang, Biomolecules, 2019, 9, 631 CrossRef CAS PubMed
.
- A. T. Mavrova, S. Dimov, D. Yancheva, M. Rangelov, D. Wesselinova and J. A. Tsenov, Eur. J. Med. Chem., 2016, 123, 69–79 CrossRef CAS PubMed
.
- P. Bánhegyi, G. Kéri, L. Örfi, Z. Szekélyhidi, F. Wáczek, US Pat., 8802849, 2014 Search PubMed
.
- P. Lagardère, C. Fersing, N. Masurier and V. Lisowski, J. Pharm., 2022, 15, 35 Search PubMed
.
- E. Elmongy, M. Kedr, N. Abotaleb and S. Abbas, Egypt. J. Chem., 2021, 64, 6857–6867 Search PubMed
.
- R. A. Hassan, M. I. Hamed, A. M. Abdou, R. H. Hedia, E. S. Ibrahim, E. A. Fouad, S. S. Alam and Y. El-Dash, Bioorg. Chem., 2025, 108549 CrossRef CAS PubMed
.
- D. K. Sureja and K. R. Vadalia, J. Saudi Chem. Soc., 2018, 22, 248–253 CrossRef CAS
.
- M. El-Sherbeny, M. El-Ashmawy, H. El-Subbagh, A. El-Emam and F. Badria, Eur. J. Med. Chem., 1995, 30, 445–449 CrossRef CAS
.
- V. Finger, M. Kufa, O. Soukup, D. Castagnolo, J. Roh and J. Korabecny, Eur. J. Med. Chem., 2023, 246, 114946 CrossRef CAS PubMed
.
- D. E. Spratt, D. J. George, N. D. Shore, M. S. Cookson, D. R. Saltzstein, R. Tutrone, A. Bossi, B. A. Brown, S. Lu and M. Fallick, JAMA Oncol., 2024, 10, 594–602 CrossRef PubMed
.
- S. L. Porvasnik, S. Germain, J. Embury, K. S. Gannon, V. Jacques, J. Murray, B. J. Byrne, S. Shacham and F. Al-Mousily, J. Pharmacol. Exp. Ther., 2010, 334, 364–372 CrossRef CAS PubMed
.
- B. Svejda, M. Kidd, F. Giovinazzo, K. Eltawil, B. I. Gustafsson, R. Pfragner and I. M. Modlin, Cancer, 2010, 116, 2902–2912 CrossRef CAS PubMed
.
- D. P. Sutherlin, L. Bao, M. Berry, G. Castanedo, I. Chuckowree, J. Dotson, A. Folks, L. Friedman, R. Goldsmith and J. Gunzner, J. Med. Chem., 2011, 54, 7579–7587 CrossRef CAS PubMed
.
- M. McKenna, S. McGarrigle and G. P. Pidgeon, Biochim. Biophys. Acta, 2018, 1870, 185–197 Search PubMed
.
- X. Zhang, O. Vadas, O. Perisic, K. E. Anderson, J. Clark, P. T. Hawkins, L. R. Stephens and R. L. Williams, Mol. Cell, 2011, 41, 567–578 CrossRef CAS PubMed
.
- A. Berndt, S. Miller, O. Williams, D. D. Le, B. T. Houseman, J. I. Pacold, F. Gorrec, W.-C. Hon, P. Ren and Y. Liu, Nat. Chem. Biol., 2010, 6, 117–124 CrossRef CAS PubMed
.
- C. Qian, C.-J. Lai, R. Bao, D.-G. Wang, J. Wang, G.-X. Xu, R. Atoyan, H. Qu, L. Yin and M. Samson, Clin. Cancer Res., 2012, 18, 4104–4113 CrossRef CAS PubMed
.
- K. Park, P. A. Jänne, D. W. Kim, J. Y. Han, M. F. Wu, J. S. Lee, J. H. Kang, D. H. Lee, B. C. Cho and C. J. Yu, Cancer, 2021, 127, 1407–1416 CrossRef CAS PubMed
.
- M. Abdel-Megid, K. M Elmahdy, A. M Elkazak, M. H Seada, O. F Mohamed and J. Pharm, Appl. Chem., 2016, 2, 78–102 Search PubMed
.
- S. Nadar, M. Borkar and T. Khan, Chem. Biol. Drug Des., 2025, 105, e70146 CrossRef CAS PubMed
.
- S. Thakur, S. Arora, M. K. Katiyar, G. Joshi and R. Kumar, ChemistrySelect, 2024, 9, e202400514 CrossRef CAS
.
- F. Islam and T. M. Quadery, Curr. Drug Targets, 2021, 22, 1944–1963 CrossRef CAS PubMed
.
- E. I. Elmongy, J. Heterocycl. Chem., 2020, 57, 2067–2078 CrossRef CAS
.
- E. M. Ali, M. S. Abdel-Maksoud and C.-H. Oh, Bioorg. Med. Chem., 2019, 27, 1159–1194 CrossRef CAS PubMed
.
- B. Wilding and N. Klempier, Org. Prep. Proced. Int., 2017, 49, 183–215 CrossRef CAS
.
- T. Kim, M. N. Morshed, A. M. Londhe, J. W. Lim, H. E. Lee, S. Cho, S. J. Cho, H. Hwang, S. M. Lim and J. Y. Lee, J. Enzyme Inhib. Med. Chem., 2021, 36, 831–846 CrossRef CAS PubMed
.
- H. A. Elsebaie, T. F. El-Moselhy, E. A. El-Bastawissy, K. M. Elberembally, R. M. Badi, E. B. Elkaeed, M. A. Shaldam, W. M. Eldehna and H. O. Tawfik, Bioorg. Chem., 2024, 143, 107101 CrossRef CAS PubMed
.
- Y. El-Dash, E. Elzayat, A. M. Abdou and R. A. Hassan, Bioorg. Chem., 2021, 114, 105137 CrossRef CAS PubMed
.
- E. I. Elmongy and H. A. Henidi, Molbank, 2022, 2022, M1352 CrossRef
.
- K. Suryanarayana, S. Maddila, K. Nagaraju and S. B. Jonnalagadda, J. Mol. Struct., 2022, 1250, 131713 CrossRef CAS
.
- H. Khalifa, A. K. ElHady, T. Liu, W. A. Elgaher, O. Filhol-Cochet, C. Cochet, A. H. Abadi, M. M. Hamed, M. Abdel-Halim and M. Engel, Eur. J. Med. Chem., 2025, 282, 117048 CrossRef CAS PubMed
.
- S. Malasala, A. Polomoni, M. N. Ahmad, M. Shukla, G. Kaul, A. Dasgupta, S. Chopra and S. Nanduri, J. Mol. Struct., 2021, 1234, 130168 CrossRef CAS
.
- A. Faraji, T. O. Bakhshaiesh, Z. Hasanvand, R. Motahari, E. Nazeri, M. A. Boshagh, L. Firoozpour, H. Mehrabi, A. Khalaj and R. Esmaeili, Eur. J. Med. Chem., 2021, 209, 112942 CrossRef CAS PubMed
.
- S. Arora, S. Upadhayay, P. Kumar, P. Kumar and R. Kumar, Bioorg. Chem., 2025, 154, 108043 CrossRef CAS PubMed
.
- M. S. Biradar, S. L. Nargund and S. Thapa, Results Chem., 2023, 5, 100926 CrossRef CAS
.
- K. I. Eissa, M. M. Kamel, L. W. Mohamed, M. A. Galal and A. E. Kassab, Drug Dev. Res., 2022, 83, 1394–1407 CrossRef CAS PubMed
.
- Y. S. Sheta, M. T. Sarg, F. G. Abdulrahman, E. S. Nossier and E. M. Husseiny, Bioorg. Chem., 2024, 149, 107471 CrossRef CAS PubMed
.
- A. S. Chiriapkin, I. P. Kodonidi, D. I. Pozdnyakov and A. A. Glushko, Chim. Techno Acta, 2022, 9, 20229204 CrossRef CAS
.
- M. A. Farag, M. M. Kandeel, A. E. Kassab and S. I. Faggal, J. Mol. Struct., 2025, 1334, 141884 CrossRef CAS
.
- F. M. Elmenier, D. S. Lasheen and K. A. Abouzid, J. Enzyme Inhib. Med. Chem., 2022, 37, 315–332 CrossRef CAS PubMed
.
- S. Elsayed, A. S. Abdelkhalek, S. Rezq, M. E. A. Kull, D. G. Romero and H. Kothayer, Eur. J. Med. Chem., 2023, 260, 115724 CrossRef CAS PubMed
.
- D. P. Kawade, D. R. Chaple and P. B. Khedekar, Int. J. Pharm. Chem. Anal., 2020, 7, 32–38 CrossRef CAS
.
- M. S. Salem, Y. M. A. Aziz, M. S. Elgawish, M. M. Said and K. A. Abouzid, Bioorg. Chem., 2020, 94, 103472 CrossRef CAS PubMed
.
- R. G. Yousef, S. A. El-Metwally, M. M. S. Al Ward, A. A. Alsfouk, D. Z. Husein, O. A. Soliman, E. B. Elkaeed, H. Elkady, A. M. Metwaly and I. H. Eissa, J. Mol. Struct., 2025, 142586 CrossRef CAS
.
- B. Nagaraju, M. Rajeswari, T. Mounika, G. Rajitha, G. S. Kumar, C. V. Rao and S. Maddila, J. Mol. Struct., 2024, 1295, 136485 CrossRef CAS
.
- S. A. El-Metwally, M. M. Abou-El-Regal, I. H. Eissa, A. B. Mehany, H. A. Mahdy, H. Elkady, A. Elwan and E. B. Elkaeed, Bioorg. Chem., 2021, 112, 104947 CrossRef CAS PubMed
.
- J. Sperry and J. García-Álvarez, Molecules, 2016, 21, 1527 CrossRef PubMed
.
- H. R. El-Shanawany, N. M. Abdelazeem, M. El-Hussieny, E. F. Ewies and N. F. El-Sayed, Egypt. J. Chem., 2023, 66, 523–540 Search PubMed
.
- S. Kushwaha, J. Baranwal, S. Singh and A. Jyoti, Curr. Green Chem., 2022, 9, 174–195 CrossRef CAS
.
- X. J. Song, Z. C. Duan, Y. Shao and X. G. Dong, Chin. Chem. Lett., 2012, 23, 549–552 CrossRef CAS
.
- S. Poojari, P. P. Naik and G. Krishnamurthy, Tetrahedron Lett., 2012, 53, 4639–4643 CrossRef CAS
.
- M. I. Soares, A. C. de Lyra, M. S. Henriques, J. A. Paixao and T. M. P. e Melo, Tetrahedron, 2015, 71, 3343–3350 CrossRef CAS
.
- P. Di Fruscia, E. Zacharioudakis, C. Liu, S. Moniot, S. Laohasinnarong, M. Khongkow, I. F. Harrison, K. Koltsida, C. R. Reynolds and K. Schmidtkunz, ChemMedChem, 2015, 10, 69–82 CrossRef CAS PubMed
.
- R. K. Gill, H. Singh, T. Raj, A. Sharma, G. Singh and J. Bariwal, Chem. Biol. Drug Des., 2017, 90, 1115–1121 CrossRef CAS PubMed
.
- J. Wang, M. Su, T. Li, A. Gao, W. Yang, L. Sheng, Y. Zang, J. Li and H. Liu, Eur. J. Med. Chem., 2017, 128, 293–299 CrossRef CAS PubMed
.
- C. Zong, L. Zhang, M. Gu and Y. Sun, Chin. J. Org. Chem., 2018, 38, 1422 CrossRef CAS
.
- X. Sun, C. Sun, L. Feng and C. Kang, Chin. J. Org. Chem., 2020, 40, 2626 CrossRef CAS
.
- F. Han, S. Lin, P. Liu, X. Liu, J. Tao, X. Deng, C. Yi and H. Xu, ACS Med. Chem. Lett., 2015, 6, 434–438 CrossRef CAS PubMed
.
- C. Li, X. Lu, Y. Yang, S. Yang and L. Zhang, Tetrahedron Lett., 2018, 59, 2463–2466 CrossRef CAS
.
- A. Cohen, P. Suzanne, J.-C. Lancelot, P. Verhaeghe, A. Lesnard, L. Basmaciyan, S. Hutter, M. Laget, A. Dumètre and L. Paloque, Eur. J. Med. Chem., 2015, 95, 16–28 CrossRef CAS PubMed
.
- M. S. Abaee, A. Hadizadeh, M. M. Mojtahedi and M. R. Halvagar, Tetrahedron Lett., 2017, 58, 1408–1412 CrossRef CAS
.
- T. Shi, L. Kaneko, M. Sandino, R. Busse, M. Zhang, D. Mason, J. Machulis, A. J. Ambrose, D. D. Zhang and E. Chapman, ACS Sustainable Chem. Eng., 2018, 7, 1524–1528 CrossRef PubMed
.
- T. Shi, C. Zerio, J. Sivinski, A. J. Ambrose, K. T. Moore, T. Buckley, L. Kaneko, M. Zhang, D. D. Zhang and E. Chapman, Eur. J. Org Chem., 2019, 2019, 3269–3272 CrossRef CAS PubMed
.
- I. A. Gorbunova, A. Rogova, D. R. Akhmetova, R. Y. Sidorov, E. E. Priakhin, R. R. Makhmudov, D. A. Shipilovskikh, O. S. Epifanovskaya, A. S. Timin and S. A. Shipilovskikh, Bioorg. Chem., 2024, 148, 107468 CrossRef CAS PubMed
.
- A. Davoodnia, M. Bakavoli, G. Barakouhi and N. Tavakoli-Hoseini, Chin. Chem. Lett., 2007, 18, 1483–1486 CrossRef CAS
.
- A. A. Ibrahim, R. S. Salama, S. A. El-Hakam, A. S. Khder and A. I. Ahmed, Colloids Surf., A, 2021, 631, 127753 CrossRef CAS
.
- M. Bakavoli, G. Bagherzadeh, M. Vaseghifar, A. Shiri and P. Pordeli, J. Chem. Res., 2009, 2009, 653–655 CrossRef
.
- K. Kim, H. Y. Kim and K. Oh, RSC Adv., 2020, 10, 31101–31105 RSC
.
- T. B. Nguyen, J. Le Bescont, L. Ermolenko and A. Al-Mourabit, Org. Lett., 2013, 15, 6218–6221 CrossRef CAS PubMed
.
- A. S. Abdelkhalek, H. Kothayer, S. Rezq, K. Y. Orabi, D. G. Romero and O. I. El-Sabbagh, Eur. J. Med. Chem., 2023, 256, 115443 CrossRef CAS PubMed
.
- D. Wu, N. Gu, J. Yao, Y. Cao, L. Wang, I. Shakir, Y. Sun and Y. Xu, Chem. Sci., 2025, 16, 5447–5463 RSC
.
- M. Narender, K. Umasankar, J. Malathi, A. R. Reddy, K. Umadevi, A. Dusthackeer and K. V. Rao, Bioorg. Med. Chem. Lett., 2016, 26, 836–840 CrossRef CAS PubMed
.
- X. Shao, A. AbdelKhalek, N. S. Abutaleb, U. K. Velagapudi, S. Yoganathan, M. N. Seleem and T. T. Talele, J. Med. Chem., 2019, 62, 9772–9791 CrossRef CAS PubMed
.
- A. Davoodnia, H. Behmadi, A. Z. Bidaki, M. Bakavoli and N. T. Hoseini, Chin. Chem. Lett., 2007, 18, 1163–1165 CrossRef CAS
.
- X. Jing, Z. Li, L. Wu and C. Yan, J. Iran. Chem. Soc., 2011, 8, 107–114 CrossRef CAS
.
- J. Kankanala, K. A. Kirby, A. D. Huber, M. C. Casey, D. J. Wilson, S. G. Sarafianos and Z. Wang, Eur. J. Med. Chem., 2017, 141, 149–161 CrossRef CAS PubMed
.
- F. Ghayour, M. R. Mohammad Shafiee and M. Ghashang, Main Group Met. Chem., 2018, 41, 21–26 CrossRef CAS
.
- O. Y. Shyyka, N. T. Pokhodylo, V. A. Palchykov, N. S. Finiuk, R. S. Stoika and M. D. Obushak, Chem. Heterocycl. Compd., 2020, 56, 793–799 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2026 |