 Open Access Article
Open Access ArticleSynthesis of La-Fe3O4@polydopamine core–shell magnetic composites for enhanced phosphate removal from aqueous solutions
Xinyu Cheng†
ab,
Zhihua Xu†ac,
Chenxi Guoa,
Ting Zhouac,
Alang Zhanga and
Feng Shi *a
*a
aCollege of Life Sciences, Shihezi University, China. E-mail: shifeng2314@yeah.net
bXinjiang Production and Construction Corps, Key Laboratory of Oasis Town and Mountain-basin System Ecology, China
cKey Laboratory of Xinjiang Phytomedicine Resource Utilization, Ministry of Education, China
First published on 11th May 2026
Abstract
In this work, a lanthanum-doped Fe3O4@polydopamine (PDA) core–shell composite was prepared via a hydrothermal method. XPS depth profiling revealed that La is uniformly distributed throughout the Fe3O4 particles, with a constant La/Fe ratio from the surface to 60 nm depth. The material exhibits superparamagnetic properties, enabling rapid magnetic separation within 30 s. Phosphate adsorption reaches equilibrium in 180 min, following pseudo-second-order kinetics (R2 > 0.999) and the Sips isotherm, with a maximum capacity of 123.44 mg P per g. XPS analysis revealed shifts in the La 3d spectra consistent with inner-sphere La–P complexation, providing supportive evidence for chemisorption. The adsorbent retained >85% of its capacity after five cycles using 1 M NaCl as the eluent. Negligible La leaching (<0.01 µg L−1) and low aquatic toxicity to Chlorella sp. further support its environmental safety. These results demonstrate that 2.5%La-Fe3O4@PDA provides a robust, magnetically separable, and reusable platform for phosphate removal, offering deeper mechanistic insights into La-doping and PDA synergy in established magnetic core–shell systems.
Introduction
Phosphorus (P) is an indispensable nutrient for the growth and reproduction of all organisms.1 However, the excessive discharge of phosphate into water bodies from agricultural runoff and domestic/industrial wastewater has emerged as one of the most pressing global environmental challenges of the 21st century. This leads to eutrophication, characterized by harmful algal blooms, reduced water transparency, and dissolved oxygen depletion, ultimately causing the collapse of ecosystem functions and a loss of biodiversity.2Phosphate removal employs various technologies,3 including chemical precipitation,4 biological treatment,5 membrane filtration,6 and adsorption.7,8 Among these, adsorption is widely regarded as one of the most promising methods due to its simplicity, cost-effectiveness, high efficiency, and potential for phosphate recovery.3 Nanomaterial adsorbents have demonstrated exceptional pollutant removal capabilities in the field of water treatment owing to their ultra-high specific surface area and reactivity, such as layered double hydroxides (LDHs),9 metal–organic frameworks (MOFs),10 biochar,8 and hydrogels.11 However, it is the high dispersion and low sedimentation brought about by its nanoscale size (<100 nm) that cause it to face inherent defects, such as difficulties in separation and recycling, a surge in operating costs, and potential secondary ecological risks in practical applications.
To overcome this bottleneck, superparamagnetic adsorbents have emerged as a promising solution. These materials typically use superparamagnetic nanoparticles (such as Fe3O4) as the magnetic core, allowing the adsorbents to be rapidly and efficiently separated from water using an external magnetic field after adsorption. This avoids the filtration difficulties associated with conventional powdered adsorbents and greatly facilitates the regeneration and recycling of the adsorbents.12–14 Despite the obvious advantages, pure Fe3O4 nanoparticles also have the disadvantage of being easily oxidized and aggregated in aqueous solutions, which reduces their adsorption efficiency and long-term stability.15–17 Therefore, by surface modification18–20 or composite techniques,21,22 functional groups or porous materials with specific adsorption capabilities can be combined on the Fe3O4 magnetic core, achieving both high adsorption performance and convenient magnetic-responsive separation, which is key to enhancing their practical application performance.
Among them, the rare earth element lantanth (La) has a strong affinity for Lewis bases such as phosphate radicals (PO43−) due to its unique electron layer structure and strong Lewis's acidity, which can form extremely stable lanthane phosphate (LaPO4)23,24 precipitation and is recognized as one of the most effective metals to capture phosphate. The effectiveness of this strategy has been widely confirmed by anchoring La on the magnetic carrier to construct an adsorbent with both high-efficiency adsorption and convenient separation characteristics. For instance, Wang et al.25 fabricated a magnetic La–Fe co-loaded calcined soda residue via co-precipitation, achieving a phosphate adsorption capacity of 74.87 mg P per g. Despite these advances, significant hurdles persist for La-based magnetic adsorbents, including potential La leaching under acidic conditions and the gradual deactivation of active sites over repeated adsorption–desorption cycles.26 Lu et al.27 effectively prevented the oxidation of the magnetic nucleus by introducing a dense SiO2 protective layer between its Fe3O4 magnetic nucleus and the La(OH)3 active layer, successfully constructing a core–shell structural adsorbent with a high adsorption capacity (63.72 mg P per g) and excellent magnetic separation performance. However, such inorganic layers (e.g., SiO2) often lack sufficient surface functional groups, limiting further functionalization and the multifaceted tuning of adsorption properties. Polydopamine (PDA) stands out for its exceptional biocompatibility, environmental friendliness, and strong adhesive properties.28 Its monomer dopamine can self-polymerize in a weakly alkaline aqueous solution to form a powerful coating layer on the surface of various materials.29,30 This adhesion results from the strong coordination of catechol groups and metal oxides in dopamine molecules, ensuring the mechanical stability of the shell.31 Rich functional groups provide a large number of phenol hydroxyl groups, amino groups, and other active sites, which not only serve as an ideal platform for subsequent modification but can also initially adsorb phosphate through hydrogen bonds and other interactions. In this study, we systematically investigate a La-doped Fe3O4@PDA core–shell composite to elucidate the synergistic role of bulk La incorporation and the PDA coating in enhancing phosphate adsorption, providing mechanistic validation and performance optimization of existing magnetic adsorbent strategies.
As the magnetic core, Fe3O4 magnetic nanoparticles are applied to various suitable fields depending on different preparation methods.32 Bruckmann et al.33 synthesized Fe3O4 by the co-precipitation method and coated a layer of chitosan (CS) on the basis of Fe3O4 as the core to prepare a magnetic composite (CS-Fe3O4). Magnetic nanoparticles uniformly covered with a polymer matrix can achieve efficient adsorption of tetracycline. Divakara et al.34 reviewed the green synthesis of spinel ferrite nanoparticles and their nanocomposites, and discussed the applications of these green-synthesized materials in the fields of antibacterial activity, photocatalytic degradation of organic dyes, and wastewater treatment. Aboelfetoh35 used the classical hydrothermal synthesis method to obtain Ag-doped reduced graphene oxide (rGO)-stabilized Fe3O4 composites (rGO/Fe3O4/Ag). The integration of rGO/Fe3O4/Ag enhanced the catalytic and adsorption capabilities of the nanomaterials, providing research progress for future environmental remediation applications. According to the purpose of this study, a La-doped spherical Fe3O4 magnetic core was prepared by a simple hydrothermal synthesis method. Subsequently, based on this, the polymerization characteristics of PDA were utilized to coat the surface, in order to obtain a novel integrated enhanced adsorbent.
In this work, a core–shell structured La-Fe3O4@PDA composite was synthesized via a hydrothermal method followed by PDA self-polymerization. The material was systematically characterized, and batch adsorption experiments were conducted to evaluate the effects of pH, adsorbent dosage, contact time, and initial concentration. Kinetic and isotherm models were applied to elucidate the adsorption mechanism, and the recyclability was assessed. The composite exhibits good superparamagnetism, selectivity, easy magnetic separation, and low environmental impact. This work advances the understanding of prior La-modified Fe3O4 and Fe3O4@PDA systems by providing XPS depth-profiling evidence of uniform La incorporation, spectroscopic data consistent with inner-sphere La–P interactions, and comprehensive environmental safety assessment. These features, together with the synergistic role of the PDA shell, validate 2.5%La-Fe3O4@PDA as a magnetically separable, reusable, and safe adsorbent with competitive phosphate capacity.
Materials and methods
Materials
Ferric chloride hexahydrate (FeCl3·6H2O), sodium acetate (NaOAc), ethylenediaminetetraacetic acid disodium salt (EDTA-2Na), ethylene glycol (EG), dopamine hydrochloride, potassium dihydrogen phosphate (KH2PO4, AR, ≥99%), and ascorbic acid (AR, ≥99.7%) were purchased from Aladdin Biochemical Technology Co., Ltd. Potassium antimonyl tartrate (AR, ≥99%) and ammonium molybdate tetrahydrate ((NH4)2MoO4·4H2O, AR, ≥99%) were also obtained from Aladdin. Lanthanum nitrate hexahydrate (La(NO3)3·6H2O, AR, ≥99%) and ethanol were supplied by Macklin Biochemical Technology Co., Ltd. Chlorella sp. was purchased from the Freshwater Algae Culture Collection, Institute of the Chinese Academy of Sciences. Deionized (DI) water was used throughout the experiments.Hydrothermal synthesis of Fe3O4 nanoparticles
Fe3O4 nanoparticles were synthesized via a modified solvothermal method. In a typical procedure, FeCl3·6H2O (0.68 g), NaOAc (1.2 g), and EDTA-2Na (0.034 g) were dissolved in 20 mL of ethylene glycol under magnetic stirring for 1 h. The homogeneous yellow solution was then transferred into a 50 mL Teflon-lined stainless-steel autoclave and heated at 200 °C for 10 h. After cooling naturally to room temperature, the resulting black precipitate was collected magnetically and washed several times with anhydrous ethanol and DI water, followed by drying under vacuum at 60 °C for 12 h.Hydrothermal synthesis of La-doped Fe3O4(La-Fe3O4)
The synthesis of La-Fe3O4 followed a similar procedure to that of pure Fe3O4, with the addition of a La precursor. Specifically, after the initial mixture of FeCl3·6H2O (0.68 g), NaOAc (1.2 g), and EDTA-2Na (0.034 g) in 20 mL ethylene glycol was stirred for 1 h, a predetermined volume of La(NO3)3·6H2O solution (in ethylene glycol) was added dropwise under vigorous stirring to achieve a nominal La doping concentration. The mixture was stirred for an additional 30 min before being transferred to the autoclave for hydrothermal reaction (200 °C, 10 h). The final product was washed and dried using the same protocol as for the pure Fe3O4 nanoparticles described above.Synthesis of La-Fe3O4@PDA core–shell nanoparticles
The La-Fe3O4@PDA composite was prepared via the in situ self-polymerization of dopamine on the surface of the as-synthesized La-Fe3O4 nanoparticles. First, La-Fe3O4 powder was dispersed in 50 mL of Tris–HCl buffer (10 mM, pH 8.5) by sonication for 5 min. Subsequently, 40 mg of dopamine hydrochloride was added to the suspension under mechanical stirring. The reaction was allowed to proceed at 30 °C for 24 h. The resulting black product, denoted as La-Fe3O4@PDA, was collected magnetically, thoroughly washed with ethanol and DI water, and finally freeze-dried for 12 h.Characterization techniques
The crystal structure and phase composition of the synthesized samples were determined using X-ray diffraction (XRD, Rigaku SmartLab) with Cu Kα radiation (λ = 1.5406 Å) and a scanning 2θ range from 10° to 90°. Morphology and microstructure were examined by transmission electron microscopy (TEM, JEOL JEM-2100F). Elemental composition and mapping were performed using energy-dispersive X-ray spectroscopy (EDS) attached to a scanning electron microscope (SEM, Hitachi SU8010). XPS depth profiling was carried out on a Thermo Scientific K-Alpha spectrometer using cluster ion etching (Ar1500+). Fourier transform infrared (FTIR) spectra were recorded on a Nicolet iS50 spectrometer in the range of 400–4000 cm−1 using the KBr pellet method. The magnetic properties were measured at room temperature using a vibrating sample magnetometer (VSM, Lake Shore 7404) with an applied field ranging from −20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 to 20
000 to 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 Oe.
000 Oe.
Batch adsorption experiments
Prepare a phosphate stock solution with a concentration of 100 mg P per L in a volumetric flask using KH2PO4. First, investigate the effect of pH on phosphate adsorption over a pH range of 3.0–9.0 at 2.0 pH intervals. Adjust the pH of the solution by adding 0.1 M HCl or NaOH. Add 10 mg of adsorbent to 50 mL of solution containing 100 mg P per L phosphate and then place the conical flask in a thermostatic shaker, agitating at 200 rpm for 120 min. After equilibrium is reached, separate the solid from the aqueous suspension using magnetic separation, and determine the residual phosphate concentration in the solution at a wavelength of 820 nm using a microplate reader (SpectraMax M2) according to standard procedures. The adsorption capacity is calculated using the following formula:
 | (1) |
Aquatic toxicity assessment
Chlorella sp. was selected as the model organism to evaluate the potential environmental impact of 2.5%La-Fe3O4@PDA. Algal cells in the logarithmic growth phase were inoculated into fresh culture medium to achieve an initial optical density (OD) of 0.16 at 680 nm. The 2.5%La-Fe3O4@PDA stock solution was then added to the algal suspensions to obtain final material concentrations of 0 (control), 20, 40, 80, 100, and 200 mg L−1. The exposure period lasted for 30 days, with sampling conducted every 5 days.To monitor algal growth, the optical density of the Chlorella sp. suspension was measured using a UV-Vis spectrophotometer. A full-wavelength scan from 350 to 800 nm was first performed to determine the maximum absorption wavelength, which was identified as 680 nm. Subsequently, at each sampling time point (every 5 days), the absorbance at 680 nm (OD680) was recorded. All experiments were performed in triplicate, and the results were expressed as mean ± standard deviation (SD).
Results and discussion
Characterization of the La-Fe3O4@PDA composite
As summarized in Table S1, the surface La/Fe atomic ratio is 1.92%. At depths of 30 nm (1.15%) and 60 nm (1.49%), the La/Fe atomic ratios remain essentially constant within the typical experimental uncertainty of XPS quantification, and are both significantly lower than the surface value (1.92%). This trend indicates a slight surface segregation feature, which is a common phenomenon in transition metal oxide systems. Subsequently, La exhibits a uniform distribution throughout the particles. In addition, the normalized high-resolution La 3d and Fe 2p spectra (Fig. S2) show consistent peak shapes and binding energies at different etching depths, further confirming that La and Fe maintain similar chemical coordination environments from the surface to the bulk. Overall, the constant La/Fe ratio and superimposable La 3d and Fe 2p spectra across etching depths strongly support homogeneous incorporation of La rather than discrete surface phases.
In the range of 500–600 cm−1, the peak at ∼570 cm−1 corresponds to the expansion and contraction of the tetrahedral Fe–O bond. For La-Fe3O4 samples, the Fe–O characteristic peaks are redshifted to about ∼560 cm−1, and the peak shape is slightly widened. This change can be attributed to the incorporation of larger-radius La3+ (≈1.06 Å) into the Fe3O4 particles, which leads to local structural distortion and a reduction in local symmetry, thereby decreasing the force constant of the Fe–O bond.37
For samples coated with polydopamine (PDA), both Fe3O4@PDA and La-Fe3O4@PDA exhibit a pair of clear absorption peaks at ∼1610 cm−1 and 1510 cm−1, corresponding to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C skeleton vibration and the NH surface of the aromatic ring in PDA, respectively. Bending vibration, which confirms that the PDA shell layer is successfully covered on the surface of both types of core materials. La-Fe3O4@PDA has a slightly strong absorption peak and displacement at ∼1280 cm−1 (C–O expansion vibration), and the O–H wide peak at ∼3400 cm−1 is also more significant. These changes may be attributed to the coordination of La3+ with phenol hydroxyl groups, amino groups, and other functional groups in the PDA shell, resulting in La–O–C coordinated bonds (Fig. S3). This indicates that the modification not only affects the Fe3O4 kernel but also regulates the chemical environment of the PDA shell interface.
C skeleton vibration and the NH surface of the aromatic ring in PDA, respectively. Bending vibration, which confirms that the PDA shell layer is successfully covered on the surface of both types of core materials. La-Fe3O4@PDA has a slightly strong absorption peak and displacement at ∼1280 cm−1 (C–O expansion vibration), and the O–H wide peak at ∼3400 cm−1 is also more significant. These changes may be attributed to the coordination of La3+ with phenol hydroxyl groups, amino groups, and other functional groups in the PDA shell, resulting in La–O–C coordinated bonds (Fig. S3). This indicates that the modification not only affects the Fe3O4 kernel but also regulates the chemical environment of the PDA shell interface.
Adsorption performance of La-Fe3O4@PDA
When the La doping ratio is further increased from 2.5% to 3.5%, the phosphate adsorption capacity of the material declines slightly. This may be due to excessively high La3+ content covering some active sites or even causing aggregation, resulting in a reduction of the material's effective specific surface area and the availability of active sites. Pure Fe3O4 exhibits the lowest adsorption capacity, which is attributed to the lack of effective surface functional group modifications and the tendency of magnetic particles to aggregate, significantly limiting the available adsorption sites. PDA coating treatment brings about a notable improvement. This polymer layer not only introduces abundant nitrogen- and oxygen-containing functional groups, thereby enhancing the surface complexation capability, but also improves the overall stability of the material. Moderate La doping, based on the advantages conferred by the PDA layer, further optimizes the chemical properties of the material surface and enhances its affinity for phosphate.
Therefore, La doping combined with PDA modification exhibits a clear synergistic effect in enhancing the adsorption performance of Fe3O4, jointly optimizing the surface properties and composition of adsorption sites of the material. Among them, a 2.5%La doping ratio demonstrates the best phosphate adsorption performance in this system; thus, this doping ratio was selected for further in-depth study in the subsequent experiments.
It can also be observed from the figure that as the pH value continues to increase from 3.0, the adsorption capacities of both materials show a gradual decreasing trend. Even under alkaline conditions (pH 9.0), 2.5%La-Fe3O4@PDA still maintains an adsorption capacity of 56.72 mg P per g, which is approximately 25.2% higher than that of unmodified Fe3O4, indicating that La doping effectively broadens the operational pH window of the material. Although the maximum adsorption capacity is achieved at pH 3.0 in controlled deionized water systems, the material retains substantial phosphate removal performance under near-neutral conditions commonly encountered in real wastewater. As demonstrated in spiked tap water and lake water samples (natural pH typically 6.5–8.0, initial phosphate concentration of 20 mg P per L), the removal efficiency exceeded 91% (Fig. 4f). This indicates that extensive pH adjustment may not be necessary for many practical applications, thereby enhancing the feasibility of 2.5%La-Fe3O4@PDA despite the acidic optimum observed under idealized batch conditions. Nevertheless, for applications requiring maximum capacity, mild acidification could be considered as a trade-off. Based on the above findings, subsequent adsorption experiments in simulated solutions were conducted at pH 3.0 to fully exploit the maximum adsorption potential of the material.
ln(qe − qt) = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe − k1t qe − k1t
| (2) |
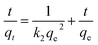 | (3) |
A linear relationship between ln(qe − qt) and t was observed at different initial phosphate concentrations (Fig. 3d). The plot was obtained by further analyzing qt versus t using the pseudo-second-order rate law (Fig. 3e). The parameters for the pseudo-first order and pseudo-second-order kinetics were determined from the slopes and intercepts of the respective curves.8 The correlation coefficient of the pseudo-second-order kinetic model (R2 = 0.99958) is higher than that of the pseudo-first-order kinetic model (R2 = 0.97167) (Table S3).
Therefore, the adsorption kinetics of 2.5%La-Fe3O4@PDA follow a pseudo-second-order model, indicating that the adsorption of phosphate by 2.5%La-Fe3O4@PDA may be consistent with chemisorption; that is, electrons are shared or exchanged between the adsorbent and the adsorbate, and phosphate ions are adsorbed through the formation of coordination bonds or ion exchange.
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe = ln qe = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qd − Kdε2 qd − Kdε2
| (4) |
 | (5) |
 | (6) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(1 + Ce−1), Ce (mg P per L) is the phosphate concentration at equilibrium); K [(mol g−1) (mol L−1)−1/n] can be regarded as the adsorption amount per unit concentration of phosphate; 1/n is an indicator of adsorption intensity. qm is the maximum adsorption capacity (mg P per g); β is an indicator of adsorption strength; b [L per mg P] is the Sips equilibrium constant.
ln(1 + Ce−1), Ce (mg P per L) is the phosphate concentration at equilibrium); K [(mol g−1) (mol L−1)−1/n] can be regarded as the adsorption amount per unit concentration of phosphate; 1/n is an indicator of adsorption intensity. qm is the maximum adsorption capacity (mg P per g); β is an indicator of adsorption strength; b [L per mg P] is the Sips equilibrium constant.In addition, equilibrium parameters RL = (1 + bC0)−1, the highest initial solute concentration in the concentration gradient C0 (mg P per L), and the mean free energy of adsorption Ed[(2Kd)−1/2, kJ mol−1] were also estimated.
The fitted correlation coefficients indicate that the Sips model has the highest correlation coefficient (R2 = 0.977), outperforming the D–R model (R2 = 0.945) and the Freundlich model (R2 = 0.912) (Table S4). The Sips model, a combination of the Langmuir and Freundlich models, suggests that the adsorption process exhibits both a tendency toward monolayer adsorption and characteristics of a heterogeneous surface. This is highly consistent with the material design: La3+ sites with high surface affinity contribute to monolayer adsorption, while polydopamine (PDA) functional groups with a broader energy distribution contribute to heterogeneous adsorption behavior, synergistically forming a composite adsorption interface.
The Freundlich model also demonstrates a good fit, further confirming the heterogeneity of the adsorbent surface, which aligns with the disordered structure of the PDA layer and the uneven energy distribution of active sites. Its parameter n > 1 indicates a strong affinity between phosphate and the adsorbent, making the adsorption process favorable. Based on the D–R model, the average adsorption free energy (E) was calculated to be 34.32 kJ mol−1. This value falls within the range of 20–40 kJ mol−1,39 indicating that the adsorption process is primarily governed by chemisorption, consistent with the potential formation of inner-sphere complexes between La3+ and PO43−.
To further investigate the proposed chemisorption mechanism, X-ray photoelectron spectroscopy (XPS) was performed on the material before and after phosphate adsorption. As shown in Table S5, the La 3d5/2 binding energy shifted from 835.1 eV to 835.6 eV after phosphate adsorption, accompanied by an increase in the satellite-to-main peak intensity ratio (from 0.44 to 0.51). These spectral changes are characteristic of the formation of a La–O–P inner-sphere complex, as previously reported for LaPO4 and related La-based adsorbents. The Fe 2p binding energy remained unchanged after adsorption, ruling out direct Fe participation in phosphate binding. The P 2p signal approaches the detection limit, but a weak peak is still observed at approximately 133.6 eV, which is higher than that of free phosphate (≈133.2 eV) and is consistent with La–O–P coordination.40 Collectively, these XPS results (shift in La 3d5/2 binding energy and increased satellite intensity, together with a weak P 2p signal at ∼133.6 eV) are consistent with the formation of inner-sphere La–O–P complexes. Although the P 2p signal intensity is low, the observed spectral changes align with literature reports on La-phosphate interactions and support the chemisorption mechanism inferred from the D–R model (E = 34.32 kJ mol−1).
The adsorption performance of 2.5%La-Fe3O4@PDA was compared with previously reported phosphate adsorbents (Table S6). Although the equilibrium time of 180 min is not the shortest among the compared materials, it is comparable to or even shorter than that of many conventional adsorbents, which often require equilibrium times of 600–1440 min.41,42 The adsorption capacity of 123.44 mg P per g is among the higher values reported for similar materials, indicating that the present adsorbent exhibits competitive phosphate uptake capability. In addition to adsorption performance, the material offers practical advantages such as magnetic recoverability, environmental safety, and satisfactory reusability, and low cost of regeneration (using NaCl as the eluent), making it a promising candidate for phosphate removal applications.
To further evaluate the stability of the material during the regeneration process, the potential leaching of La was investigated under different conditions. The 2.5%La-Fe3O4@PDA was immersed in three different media (20 mg P per L phosphate solution, deionized water, and lake water), and La concentrations in the supernatant were measured by ICP-MS at 0 h, 3 h, and 24 h. As shown in Fig. 4c, La was below the detection limit in deionized water at all time points. In phosphate solution and lake water, La remained below the detection limit at 0 h and 3 h, while a concentration of only 0.01 µg L−1 was detected at 24 h, a value well below the detection limit and considered negligible. These results confirm that La is stably and homogeneously incorporated throughout the Fe3O4 particles and does not leach into the aqueous phase during the adsorption and regeneration processes, further supporting the excellent stability and reusability of the material.
The influence of ionic strength on phosphate adsorption was examined using NaCl as the background electrolyte at concentrations ranging from 0 to 1.0 mol L−1. As shown in Fig. 4d, the phosphate removal efficiency gradually decreased with increasing ionic strength. In the absence of NaCl (control), the removal efficiency was 91.24%. At NaCl concentrations of 0.01 and 0.05 mol L−1, the efficiencies slightly decreased to 89.68% and 89.45%, respectively. Further increases to 0.1, 0.5, and 1.0 mol L−1 resulted in removal efficiencies of 87.13%, 87.07%, and 86.24%, respectively. The moderate decrease in adsorption efficiency with increasing ionic strength suggests that electrostatic interactions contribute to phosphate adsorption, but the material retains a high removal capacity (>86%) even under high ionic strength. This behavior is consistent with an inner-sphere complexation mechanism, wherein specific chemical bonding between La3+ and phosphate is the dominant driving force.
The selectivity of 2.5%La-Fe3O4@PDA toward phosphate was assessed in the presence of common competing anions, SO42− and CO32−, at concentrations of 0.2 and 2.0 mmol L−1. A mixed solution containing both anions (each at 1.0 mmol L−1) was also tested. The initial phosphate concentration was fixed at 20 mg P per L. As shown in Fig. 4e, the control (without competing anions) achieved a removal efficiency of 92.53%. In the presence of 0.2 mmol L−1 SO42− or CO32−, the efficiencies were 91.91% and 91.94%, respectively. At the higher concentration of 2.0 mmol L−1, the removal efficiencies remained above 90%. The mixed anion system gave a removal efficiency of 91.73%. These results demonstrate that coexisting anions do not significantly interfere with phosphate adsorption, confirming the excellent selectivity of the material. This high selectivity is also attributed to the strong affinity of La3+ sites for phosphate via inner-sphere complexation.
To further evaluate the practical applicability of 2.5%La-Fe3O4@PDA, the standard addition method was used to test tap water and lake water (collected from Shihezi City). Both water samples were filtered through 0.22 µm membrane filters to remove suspended solids. The initial phosphate concentrations in the raw samples were negligible. Therefore, phosphate standard solution was spiked into each sample to achieve an initial phosphate concentration of 20 mg P per L. Subsequently, 10 mg of 2.5%La-Fe3O4@PDA was added to 50 mL of each spiked water sample. As shown in Fig. 4f, the phosphate removal efficiencies in tap water and lake water were 91.76% and 91.69%, respectively, which are comparable to those obtained in deionized water (92.53%). These results demonstrate that the material remains highly effective under near-neutral pH conditions (the natural pH of the tested waters), even though the maximum adsorption capacity is observed at pH 3.0 in controlled laboratory experiments. Thus, the presence of natural organic matter and co-existing ions does not significantly impair performance, confirming the feasibility of 2.5%La-Fe3O4@PDA for practical phosphate removal applications.
Taken together, the material exhibits stable and efficient phosphate adsorption across a wide range of water matrix conditions, including high ionic strength, the presence of competing anions, and real natural water samples. These findings further support that phosphate uptake proceeds primarily through an inner-sphere complexation mechanism, and underscore the potential of 2.5%La-Fe3O4@PDA as a robust adsorbent for real-world wastewater treatment.
Notably, at higher concentrations (100 and 200 mg L−1), the final OD680 values were comparable to, or even slightly higher than, those of the control group (0.8225 and 0.8725 vs. 0.7875). This observation suggests that the material does not exert acute toxicity toward Chlorella sp. within the tested concentration range. The slightly lower OD values observed at low concentrations (20 and 40 mg L−1) may be attributed to physical effects such as light scattering or shading caused by well-dispersed particles in the culture medium, rather than chemical toxicity. At higher concentrations (100 and 200 mg L−1), the particles tend to aggregate and settle, reducing light interference and allowing more efficient algal growth. This interpretation is strongly supported by the negligible La leaching (<0.01 µg L−1) confirmed by ICP-MS analysis, which rules out the possibility of toxic metal ion release. Furthermore, scanning electron microscopy (SEM) images of Chlorella sp. after 30 days of exposure (Fig. S4) showed intact cell morphology with no visible surface damage, cracking, or lysis across all tested concentrations, further confirming the absence of acute toxic effects.
Cost and scalability considerations
To assess the practical potential of 2.5%La-Fe3O4@PDA, we estimated the raw material cost based on current market prices (Table S7). The composite was synthesized via a simple one-pot hydrothermal method (200 °C, 10 h) followed by room-temperature PDA coating. The estimated raw material cost is approximately 0.04–0.05 USD per g when excluding the recoverable solvent (ethylene glycol). Magnetic separation eliminates the need for energy-intensive centrifugation or filtration, significantly reducing operational costs compared with non-magnetic nano-adsorbents. The use of 1 M NaCl as a low-cost regenerant further enhances economic viability.From a scalability perspective, hydrothermal synthesis is industrially mature. Future optimization may include continuous-flow hydrothermal reactors and more cost-effective dopamine analogs (e.g., tannic acid) to further lower costs. Although the raw material cost (≈40 USD per kg) is higher than that of some commercial adsorbents (e.g., activated alumina or lanthanum-modified resins, typically $5–20 per kg),25 the high reusability (>85% capacity retention after five cycles) reduces the effective cost per cycle to approximately 8 USD per kg, making the material economically competitive on a life-cycle basis. Overall, these attributes position 2.5%La-Fe3O4@PDA as a cost-effective and scalable option for large-scale wastewater treatment.
Conclusions
In this study, a La-doped Fe3O4@PDA core–shell composite was fabricated and comprehensively evaluated as a high-performance phosphate adsorbent, with emphasis on mechanistic validation of uniform La incorporation into the Fe3O4 matrix and inner-sphere La–P complexation. XPS depth profiling confirmed that La is uniformly distributed throughout the Fe3O4 particles rather than merely present on the surface, with a constant La/Fe ratio from the surface to 60 nm depth. The optimized material, 2.5%La-Fe3O4@PDA, exhibited excellent phosphate adsorption capacity (123.44 mg P per g) and rapid equilibrium (180 min), following pseudo-second-order kinetics and the Sips isotherm model. XPS analysis revealed shifts in the La 3d spectra consistent with the formation of inner-sphere La–P complexes, further supporting the chemisorption mechanism. The adsorbent retained over 85% of its initial capacity after five consecutive adsorption–desorption cycles using 1 M NaCl as the eluent, demonstrating excellent reusability. Although the maximum adsorption capacity was observed at pH 3.0 in controlled laboratory experiments, the material maintained high removal efficiency (>91%) in spiked real water samples at their natural near-neutral pH, indicating its potential practical applicability without extensive pH adjustment. Moreover, the material showed negligible La leaching (<0.01 µg L−1) and low aquatic toxicity toward Chlorella sp., supporting its environmental safety. This study provides a high-performance, magnetically separable, and reusable material solution for phosphate removal from wastewater.Author contributions
All authors have accepted responsibility for the entire content of this manuscript and approved its submission.Conflicts of interest
There are no conflicts to declare.Data availability
All data that support the findings of this study are included within the article.Supplementary information (SI): Tables S1–S7 and Fig. S1–S4, covering XPS depth profiling, magnetic properties, adsorption kinetics and isotherms, binding energy changes, adsorbent comparison, material cost, elemental mapping, FT-IR, and SEM images of Chlorella sp. See DOI: https://doi.org/10.1039/d6ra00725b.
Acknowledgements
This work was supported by the Tianshan Elite Youth Talent Program of Xinjiang Uygur Autonomous Region (grant no. 2024TSYCJC0076) and the National Natural Science Foundation of China (grant no. 32460237).Notes and references
- T. L. Martinon, RSC Sustainability, 2023, 1, 1594–1598 RSC.
- M. M. Mekonnen and A. Y. Hoekstra, Water Resour. Res., 2018, 54, 345–358 CrossRef CAS.
- M. N. Nadagouda, G. Varshney, V. Varshney and C. A. Hejase, ACS Environ. Au, 2024, 4, 271–291 CrossRef CAS PubMed.
- E. Zran, B. Yao, A. Trokourey, A. Yobouet and P. Drogui, Int. J. Environ. Sci. Technol., 2015, 12, 3117–3124 CrossRef CAS.
- D. Mulkerrins, A. Dobson and E. Colleran, Environ. Int., 2004, 30, 249–259 CrossRef CAS PubMed.
- A. Anwar, C. M. Rosnelly, I. Ramli, N. Arahman and A. Fahrina, Elkawnie, 2023, 9, 260–274 CrossRef.
- H. Bacelo, A. M. Pintor, S. C. Santos, R. A. Boaventura and C. M. Botelho, Chem. Eng. J., 2020, 381, 122566 CrossRef CAS.
- Y. Huang, X. Lee, M. Grattieri, M. Yuan, R. Cai, F. C. Macazo and S. D. Minteer, Chem. Eng. J., 2020, 380, 122375 CrossRef CAS.
- L.-g. Yan, K. Yang, R.-r. Shan, T. Yan, J. Wei, S.-j. Yu, H.-q. Yu and B. Du, J. Colloid Interface Sci., 2015, 448, 508–516 CrossRef CAS PubMed.
- M. Belaye, A. M. Taddesse, E. Teju, M. Sanchez-Sanchez and J. M. Yassin, ACS Omega, 2023, 8, 23860–23869 CrossRef CAS PubMed.
- Ł. Wujcicki, T. Mańdok, W. Budzińska-Lipka, K. Pawlusińska, N. Szozda, G. Dudek, K. Piotrowski, R. Turczyn, M. Krzywiecki and A. Kazek-Kęsik, Sci. Rep., 2023, 13, 13049 CrossRef PubMed.
- A. E. Angkawijaya, Y. N. Tran-Chuong, Q. N. Ha, P. L. Tran-Nguyen, S. P. Santoso, V. Bundjaja, A. W. Go, H.-Y. Hsu and Y.-H. Ju, J. Taiwan Inst. Chem. Eng., 2022, 131, 104162 CrossRef CAS.
- D. Pattappan, S. Kapoor, S. S. Islam and Y.-T. Lai, ACS Omega, 2023, 8, 24727–24749 CrossRef CAS PubMed.
- C. Su, J. Hazard Mater., 2017, 322, 48–84 CrossRef CAS PubMed.
- H. Ardiyanti, E. Suharyadi, T. Kato and S. Iwata, AIP Conf. Proc., 2016, 1725, 020007 CrossRef.
- S. Duan, J. Liu, Y. Pang, F. Lin, X. Meng, K. Tang and J. Li, Molecules, 2022, 27, 6812 CrossRef CAS PubMed.
- S. Laurent, D. Forge, M. Port, A. Roch, C. Robic, L. Vander Elst and R. N. Muller, Chem. Rev., 2008, 108, 2064–2110 CrossRef CAS PubMed.
- Y. Li, Q. Xie, Q. Hu, C. Li, Z. Huang, X. Yang and H. Guo, Sci. Rep., 2016, 6, 30651 CrossRef CAS PubMed.
- L. Paltrinieri, M. Wang, S. Sachdeva, N. A. Besseling, E. J. Sudhölter and L. C. De Smet, J. Mater. Chem. A, 2017, 5, 18476–18485 RSC.
- H. Shen, Z. Wang, A. Zhou, J. Chen, M. Hu, X. Dong and Q. Xia, RSC Adv., 2015, 5, 22080–22090 RSC.
- K.-W. Jung, S. Lee and Y. J. Lee, Bioresour. Technol., 2017, 245, 751–759 CrossRef CAS PubMed.
- A. I. Salim, N. A. Abdelgawad, E. Rozaik and N. G. Mostafa, Sci. Rep., 2025, 15, 24038 CrossRef CAS PubMed.
- K. Kim, D. Kim, T. Kim, B.-G. Kim, D. Ko, J. Lee, Y. Han, J. C. Jung and H. B. Na, RSC Adv., 2019, 9, 15257–15264 RSC.
- J. Mo, Q. Li, X. Sun, H. Zhang, M. Xing, B. Dong and H. Zhu, Water, 2024, 16, 418 CrossRef CAS.
- H. Wang, H. Luo, R. Han, W. Wu, J. Yang, T. Qin, L. Chen, M. Liu, B.-J. He and Z. Jing, Sci. Rep., 2025, 15, 3145 CrossRef CAS PubMed.
- Y. Zhu, X. Yue and F. Xie, Adsorpt. Sci. Technol., 2020, 38, 254–270 CrossRef CAS.
- Y. Lu, X. Jin, X. Li, M. Liu, B. Liu, X. Zeng, J. Chen, Z. Liu, S. Yu and Y. Xu, Polymers, 2023, 15, 248 CrossRef CAS PubMed.
- F. Liu, R. Jamal, T. Abdiryim and X. Liu, Cellulose, 2022, 29, 8025–8064 CrossRef CAS.
- H. Hemmatpour, O. De Luca, D. Crestani, M. C. Stuart, A. Lasorsa, P. C. Van Der Wel, K. Loos, T. Giousis, V. Haddadi-Asl and P. Rudolf, Nat. Commun., 2023, 14, 664 CrossRef CAS PubMed.
- G. Siciliano, A. G. Monteduro, A. Turco, E. Primiceri, S. Rizzato, N. Depalo, M. L. Curri and G. Maruccio, Nanomaterials, 2022, 12, 1145 CrossRef CAS PubMed.
- M. Saraf, P. Prateek, R. Ranjan, B. Balasubramaniam, V. K. Thakur and R. K. Gupta, Adv. Mater. Interfaces, 2024, 11, 2300670 CrossRef CAS.
- S. I. A. Shah, W. Ahmad, M. Anwar, R. Shah, J. A. Khan, N. S. Shah, A. Al-Anazi and C. Han, Appl. Catal. O: Open, 2025, 203, 207049 CAS.
- F. da Silva Bruckmann, C. E. Schnorr, T. da Rosa Salles, F. B. Nunes, L. Baumann, E. I. Müller, L. F. Silva, G. L. Dotto and C. R. Bohn Rhoden, Polymers, 2022, 14, 4854 CrossRef CAS PubMed.
- S. G. Divakara and B. Mahesh, Results Eng., 2024, 21, 101702 CrossRef.
- E. F. Aboelfetoh, Sci. Rep., 2025, 15, 27342 CrossRef CAS PubMed.
- W. Wang, L. Zheng, F. Lu, R. Hong, M. Z. Chen and L. Zhuang, AIP Adv., 2017, 7, 056317 CrossRef.
- M. Zeng, K. Thummavichai, W. Chen, G. Liu, Z. Li, X. Chen, C. Feng, Y. Li, N. Wang and Y. Zhu, RSC Adv., 2021, 11, 37246–37253 RSC.
- C. T. Yavuz, J. Mayo, W. W. Yu, A. Prakash, J. C. Falkner, S. Yean, L. Cong, H. J. Shipley, A. Kan and M. Tomson, Science, 2006, 314, 964–967 CrossRef PubMed.
- S. A. Odoemelam, F. K. Onwu, S. C. Uchechukwu and M. A. Chinedu, Am. Chem. Sci. J., 2015, 5, 253–269 CrossRef.
- Y. He, X. Qi, J. Li, W. Wang, J. Zhang, L. Yang, M. Xue and K. Lan, ACS Omega, 2024, 9, 30826–30833 CrossRef CAS PubMed.
- W. Xiong, J. Tong, Z. Yang, G. Zeng, Y. Zhou, D. Wang, P. Song, R. Xu, C. Zhang and M. Cheng, J. Colloid Interface Sci., 2017, 493, 17–23 CrossRef CAS PubMed.
- Q. Yin, H. Ren, R. Wang and Z. Zhao, Sci. Total Environ., 2018, 631, 895–903 CrossRef PubMed.
Footnote |
| † Equal contribution. |
| This journal is © The Royal Society of Chemistry 2026 |





