Open Access Article
Open Access ArticleA green and facile modification of red mud waste with MIL-100 (Fe) for multi-pollutant adsorption from wastewater: isotherms, kinetics, and thermodynamics
Adarsh Singh a,
Manne Chimyanga,
Akash Rawat
a,
Manne Chimyanga,
Akash Rawat b,
Venkatesh Uddameric and
Ashok Kumar Gupta
b,
Venkatesh Uddameric and
Ashok Kumar Gupta *a
*a
aEnvironmental Engineering Division, Department of Civil Engineering, Indian Institute of Technology Kharagpur, Kharagpur 721302, India. E-mail: agupta@civil.iitkgp.ac.in
bSchool of Environmental Science and Engineering, Indian Institute of Technology Kharagpur, Kharagpur 721302, India
cDepartment of Civil and Environmental Engineering, Lamar University, Beaumont, TX 77701, USA
First published on 7th April 2026
Abstract
The expansion of the textile and pharmaceutical industries, driven by rapid population growth and evolving lifestyles, has led to an increased release of synthetic chemicals into the environmental matrices, thereby posing risks to aquatic ecosystems and human health. A novel composite adsorbent, fabricated by integrating thermally activated red mud (ARM) with MIL-100 (Fe) (referred to as MARM) via co-precipitation, was utilized for the adsorptive removal of methylene blue (MLB), Congo red (CNR), and levofloxacin (LVX) from an aqueous solution. The assessment of textural properties showed MARM-II (containing 40% MIL-100 Fe) with a high specific surface area (SSA) of 651.741 m2 g−1. The enhanced SSA and distinctive surface charge profile of MARM-II promoted the effective adsorption of MLB, CNR, and LVX onto the MARM-II surface. At optimal conditions (contact time: 150 min, initial MLB, CNR, and LVX concentration: 10 mg L−1 each, MARM-II dose: 0.4 g L−1, solution pH: 7, and temperature: 27 ± 3 °C), MLB, CNR, and LVX removals were recorded to be 93.077 ± 0.593%, 89.739 ± 1.119%, and 96.102 ± 0.997%, respectively. The pseudo-second-order kinetic model confirmed chemisorption was the governing mechanism, while the Sips isotherm best explained the adsorptive nature of MLB, CNR, and LVX on the surface of MARM-II composite. The maximum adsorption capacity for MLB, CNR, and LVX was found to be 123.021 ± 11.926, 143.934 ± 24.248, and 97.657 ± 5.686 mg g−1, respectively. Additionally, the thermodynamic investigation indicates that the adsorption process was characterized as exothermic and spontaneous. The mechanistic insights showcased that the adsorption process was mainly driven by electrostatic interaction, hydrogen bonding, π–π interaction, and chemisorption.
1. Introduction
The decline in the quality of water resources caused by the release of organic pollutants from various industrial and domestic sources has become a significant environmental issue.1 Synthetic dyes, including methylene blue (MLB) and Congo red (CNR), are widely utilized in the textile, printing, and pharmaceutical sectors, resulting in their significant presence in the effluent from these industries.2 In addition, the wastewater discharged from domestic households and healthcare facilities often contains antibacterial drugs like levofloxacin (LVX), which also contributes to surface water contamination.3 Reportedly, the abovementioned compounds (MLB, CNR, and LVX) are recalcitrant to biodegradation owing to their xenobiotic nature.4–6 Organic dyes contribute to the coloration of water, which obstructs light penetration, consequently diminishing photosynthesis and oxygen solubility.7 Additionally, they have been documented to possess mutagenic and carcinogenic potential.8 Moreover, LVX is a fluoroquinolone-based antibiotic that acts against both Gram-positive and Gram-negative bacteria, along with pathogens.9 Thus, if released into aquatic environments, it possesses bioaccumulation potential,10 thereby threatening aquatic biota. Therefore, there is a prompt need for efficient treatment processes capable of eliminating such recalcitrant and toxic chemicals.Adsorption has emerged as a prominent method for eliminating a wide range of pollutants from water and wastewater because of its high efficiency, simplicity, cost-effectiveness, and versatility.11–15 Lately, several adsorbents have been studied for the elimination of dyes and pharmaceuticals from aqueous matrices, including metal–organic frameworks (MOFs),11 metal oxides,16 zeolites,17 activated carbon,18 polymer-based substances,19 and layered double hydroxide (LDH)-based materials.20–22 Among these materials, MOFs have attracted significant attention for adsorption applications, owing to advances in their design and synthesis.23 These porous materials consist of metal nodes and organic ligands, and showcase remarkable properties such as high porosity, pore size tunability, and a high specific surface area (SSA).24 Over the years, a wide range of MOFs, based on Al, Co, Cu, Cr, Fe, and other metals, as well as their composites, have been utilized.25–27 Unlike other MOFs, Fe-based MOFs stand out owing to the abundance of Fe on Earth,28 its relatively low toxicity, and eco-friendly nature.26
Specifically, MIL-100 (Fe) possesses high stability due to a rigid zeo-type structure consisting of octahedral Fe(III) and trimesic acid.29 However, the precisely defined structure of MOFs results in a fixed density of adsorption sites, which ultimately establishes an upper limit on their adsorption capacity.30 This limitation opens up opportunities to incorporate complementary materials that can enhance the adsorption capacity of MOF-based composites. In the context of environmental applications, the development of such composites is expected to follow principles of simplicity, versatility, economic viability, and sustainability.26 Notably, if the complementary material is derived from waste, it justifies the highest priority for use owing to both economic and environmental benefits. Given this perspective, red mud (RM) presents itself as an effective candidate for consideration, as it is a by-product produced during the refining of bauxite ore for alumina production.31 The global annual discharge of RM into the environment is approximately 120 million tons.32 Disposing of RM poses a significant environmental challenge, especially in regions with active alumina industries, due to its extreme alkalinity.33 Before 1970, alumina producing industries released RM into marine environments; however, this practice transitioned to disposal on land, notably dry stacking, in response to ecological problems.34,35 RM primarily comprises a metal-rich matrix, which includes key compounds, such as Fe2O3, Al2O3, and SiO2.36,37 Although RM has found applications in construction materials, ceramics, and surface treatment for carbon steel, its use as a low-cost adsorptive material for eliminating pollutants from aqueous matrices has garnered significant attention but has not been fully explored.38
A sustainable and cost-effective activated RM (ARM) and MIL-100 (Fe) composite (referred to as MARM hereafter) was developed in this study using a facile synthesis process at ambient temperature, anticipating synergistic interfacial interactions between the constituent materials (Fig. 1). The as-synthesized adsorptive materials were extensively characterized to investigate their crystallinity, functional groups and molecular vibrations, surface morphology and topography, elemental composition, local chemical environment, textural properties, thermal and chemical stability, and microstructural features at high resolution. Subsequently, MARM composite was employed for the adsorption of a mixture of MLB, CNR, and LVX from aqueous solution under varying operational conditions, including contact time, initial adsorbate concentration, adsorbent dosage, solution pH, and temperature. The possible impact of co-existing ions and the real wastewater matrix was evaluated to assess the applicability of the MARM-II condition under field conditions. The adsorptive potential was assessed through kinetic, isotherm, and thermodynamic analyses. A proposed sorption mechanism was subsequently proposed, along with a desorption and reusability study to assess the composite's stability and durability. Finally, an economic analysis of the MARM-II composite was done to evaluate its practical feasibility.
2. Experimental section
2.1. Chemicals and reagents
The chemicals and reagents used in this study have been numbered in Table S1. All chemicals were of reagent grade with purity >98%.2.2. Analytical and instrumentation techniques
The instruments deployed in the present study for characterizing as-prepared adsorptive materials and aliquot measurement have been enumerated in Table S2.2.3. Preparation of ARM integrated with MIL-100 (Fe) (referred to as MARM)
The pH of the raw RM sample was brought close to 7 through repeated ultrasonic-assisted dispersion, stirring, and decanting.37 Thereafter, the RM sample was thermally activated (referred to as ARM) by calcining it at 400 °C for 4 h.39 At first, 2.25 g of ferrous heptahydrate (FeSO4·7H2O) was dissolved in 125 mL of deionized (DI) water to form a homogenous solution (labeled as solution A). A predetermined amount of ARM was then ultrasonically dispersed in solution A, and continuously stirred for 3 h at 60 °C. Concurrently, 1.42 g of benzene-1,3,5-tricarboxylic acid or trimesic acid (H3BTC) and 0.80 g of sodium hydroxide (NaOH) were added to 125 mL of deionized (DI) water and subjected to ultrasonication until a clear solution was obtained (labeled as solution B). Subsequently, both solutions (A and B) were transferred to a 500 mL Erlenmeyer flask and stirred at 500 rpm for 20 h, facilitating a reaction in the presence of air. After the end of reaction period, the precipitate was filtered and rinsed three times using ethanol and DI water, alternatively. The end product, a brownish-orange colored powder (labeled as MARM-I, MARM-II, and MARM-III, corresponding to 80%, 60%, and 40% ARM added relative to FeSO4·7H2O, respectively), was obtained after overnight drying at 60 °C, followed by heating at 100 °C for 6 h. Pristine MIL-100 (Fe) was prepared following a similar route, excluding the addition of ARM, utilizing a previously reported method.402.4. Adsorption studies
The adsorption experiments were performed in batch mode using a 250 mL borosilicate Erlenmeyer flask. It contained specific dosages of MARM-II and various initial concentrations of LVX, MLB, and CNR, with the solution pH adjusted to 7 ± 0.1. The solution pH was adjusted using 0.1 M NaOH or HCl solution. The varied concentrations of the aforesaid contaminants were achieved by diluting 500 mg L−1 of LVX, MLB, and CNR stock solutions. The physicochemical properties of LVX, MLB, and CNR stock solutions are described in Table 1. The experimental sample was stirred at 200 rpm on a digital multi-position hot plate stirrer at ambient temperature (27 ± 3 °C), unless otherwise specified. At predetermined time intervals, a sample was withdrawn from the solution mixture, and the adsorbent was set apart by centrifugation at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 10 min. A UV-vis spectrophotometer was utilized to analyze the concentration of LVX, MLB, and CNR in the aliquot at wavelengths around 288 nm, 664 nm, and 497 nm, respectively. A series of dilutions was prepared from the mixed stock solution, each containing 500 mg L−1 of LVX, MLB, and CNR. Calibration curves for each analyte showcased excellent linearity with determination coefficients (R2 > 0.99). Each set of experiments was done in triplicate, and the extent of LVX, MLB, and CNR removal (η) was calculated as follows (eqn (1)):
000 rpm for 10 min. A UV-vis spectrophotometer was utilized to analyze the concentration of LVX, MLB, and CNR in the aliquot at wavelengths around 288 nm, 664 nm, and 497 nm, respectively. A series of dilutions was prepared from the mixed stock solution, each containing 500 mg L−1 of LVX, MLB, and CNR. Calibration curves for each analyte showcased excellent linearity with determination coefficients (R2 > 0.99). Each set of experiments was done in triplicate, and the extent of LVX, MLB, and CNR removal (η) was calculated as follows (eqn (1)):
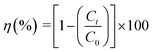 | (1) |
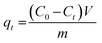 | (2) |
2.5. Adsorption kinetics
The adsorption kinetics elucidate the rate and mechanism of the adsorptive process involving various physicochemical interactions between the adsorbent and adsorbate interface. Therefore, the time-resolved adsorption data of LVX, MLB, and CNR were fitted in various kinetic models, i.e., pseudo-first-order (PFO),41 pseudo-second-order (PSO),42 Elovich,43 and intraparticle diffusion (IPD) models,44 to determine the governing mechanism of the adsorption process (eqn (3)–(6)).| qt = qe(1 − e−k1t) | (3) |
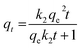 | (4) |
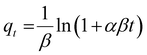 | (5) |
| qt = kit1/2 + c | (6) |
2.6. Adsorption isotherms
The interaction between adsorbate and adsorbent surface under equilibrium conditions could be interpreted through isotherm studies. The equilibrium data were fitted to various adsorption isotherm models, including Langmuir,45 Freundlich,46 Sips,47 and Redlich–Peterson isotherms,48 to analyze the adsorptive potential of LVX, MLB, and CNR molecules on MARM-II composite (eqn (7)–(10)).
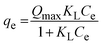 | (7) |
| qe = KFCe1/n | (8) |
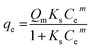 | (9) |
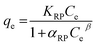 | (10) |
2.7. Adsorption thermodynamic studies
The impact of temperature on the adsorptive removal of LVX, MLB, and CNR using MARM-II composite was examined by thermodynamic assessment.49,50 The thermodynamic parameters, including changes in standard Gibbs' free energy (ΔG°) (kJ mol−1), entropy (ΔS°) (J K−1 mol−1), and enthalpy (ΔH°) (kJ mol−1), were obtained through the following equations (eqn (11) and (12)):
 | (11) |
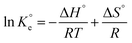 | (12) |
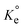 (dimensionless) is the thermodynamic equilibrium constant.
(dimensionless) is the thermodynamic equilibrium constant.
2.8. Stability and reusability studies
The thermal stability of the MARM-II composite was assessed deploying thermogravimetric analysis (TGA) and differential thermal analysis (DTA). In addition, the possible leaching of metals and organic linker from the MARM-II composite in the treated solution was quantified using inductively coupled plasma mass spectrometry (ICP-MS), and total organic carbon (TOC) analysis, respectively. Furthermore, the regenerative ability and repeated application of MARM-II were evaluated by a reusability test. Subsequent to each batch experiment, the exhausted MARM-II composite was suspended in 0.5 M HCl and agitated at 200 rpm for 90 min, followed by an analogous procedure with 0.5 M NaOH and acetone.19,51 Subsequently, the adsorbent was collected, washed, and dried as previously reported to neutralize the pH. The adsorptive removal of LVX, MLB, and CNR was quantified after each cycle, up to five successive cycles. Then, its diffractogram and FTIR spectra were acquired to examine any changes in the crystallographic properties and chemical composition of the MARM-II composite, respectively.3. Results and discussion
3.1. Characterization of the pristine and composite adsorptive materials
The crystalline nature of the as-synthesized adsorbents was investigated by X-ray diffraction spectroscopy (XRD). As displayed in Fig. 2a, MIL-100 (Fe) exhibited Bragg peaks at 2θ ∼ 10.37°, 11.03°, 12.55°, 14.27°, 20.15°, and 24.15°, aligning with the existing literature.52,53 In the diffractogram of ARM, the peaks observed at 2θ ∼ 12.34° and 24.91° can be assigned to the kaolinite phase,54 while the peak at 2θ ∼ 26.64° relates to the quartz phase.55 Moreover, the peaks determined at 2θ ∼ 24.20°, 33.18°, 35.66°, 40.88°, 49.50°, 54.11°, 62.42°, and 64.05° can be linked to the (012), (104), (110), (113), (024), (116), (214), and (300) crystal planes of the hematite phase (α-Fe2O3).56,57 The standard XRD pattern of α-Fe2O3 (reference code – 00-033-0664) and simulated XRD pattern of MIL-100 (Fe) (CIF file no. -7102029) has been presented in Fig. S1. The As anticipated, the XRD pattern of the MARM-II composite represented peaks associated with the aforementioned constituent materials, confirming its successful synthesis.Fourier transform infrared (FTIR) spectroscopy was deployed to determine the vibrational modes of the chemical bonds in the as-prepared pristine and composite materials. The FTIR spectrum of MIL-100 (Fe) exhibited two peaks at 710 cm−1 and 762 cm−1, corresponding to the Fe–OH vibrations (Fig. 2b).58 The two distinct peaks evident around 1444 cm−1 and 1375 cm−1 can be linked to the symmetric and asymmetric stretching vibrations of the O–C–O bonds in the organic ligand, respectively.59 Moreover, the peak observed at 1617 cm−1 could be linked to the phenyl and carboxylate groups of trimesic acid.60 The peak observed at 521 cm−1, common in the FTIR spectra of MIL-100 (Fe) and ARM, is indicative of the Fe–O bond.61 The peak appearing at 909 cm−1 in the latter spectrum corresponds to the bending vibration of Al–OH–Al bonds.62 The asymmetric Si–O–Al and Si–O–Si stretching bands were indicated by the intense peaks appearing at 1002 cm−1 and 1030 cm−1, respectively.63,64 Additionally, the stretching vibrations of the –OH groups were indicated by the peaks noted at 3621 cm−1 and 3693 cm−1.65 The FTIR spectrum of the MARM-II composite consisted of distinctive peaks of MIL-100 (Fe) and ARM, implying effective integration of the individual components.
The surface topographical features of the MARM-II composite were evaluated utilizing atomic force microscopy (AFM). Fig. 2c illustrates that AFM imaging uncovered a heterogeneous surface, indicating the presence of surface imperfections marked by both elevations and indentations across the MARM-II composite. According to the surface height profile, most particles exhibited sizes between roughly ∼105 nm and ∼137 nm (Fig. 2d). In addition, other height parameters are depicted in Fig. S2.
The textural properties of the as-prepared adsorptive materials were recorded through Brunauer–Emmett–Teller (BET) and Barrett–Joyner–Halenda (BJH) analysis. Fig. 2e presents the N2 adsorption–desorption isotherm of MIL-100 (Fe), ARM, and MARM-II. MIL-100 (Fe) displays a concave curve in association with the relative pressure (P/P0), characteristic of a Type I isotherm, which indicates its microporous structure and high specific surface area (SSA) (1845.522 m2 g−1). The forces from opposing pore walls overlap in micropores, causing rapid filling at low relative pressures. This phenomenon is evidenced by a sharp rise at the beginning of the adsorption isotherm, followed by a plateau at higher relative pressures.66 The N2 sorption profile of ARM had a Type IV isotherm, indicative of mesoporous material, and exhibited an H3 hysteresis loop, often associated with plate-like particle aggregates that result in slit-shaped pores.67 The SSA of ARM was found to be 21.339 m2 g−1. Furthermore, the MARM-II composite showed a combination of Type I and Type IV isotherms, and an H2 hysteresis loop, confirming the presence of both micropores and mesopores,68 with a SSA of 651.741 m2 g−1. Reportedly, the presence of micropores in adsorbents facilitates the selective adsorption of small-sized adsorbates,69,70 whereas mesopores accommodate relatively larger molecules and enhance mass transfer.70–72 The BJH pore size distribution plots for MIL-100 (Fe), ARM, and MARM-II are presented in Fig. 2f, with details of corresponding pore sizes and volumes in Table S3.
The morphological features of the as-synthesized adsorptive materials were determined by field emission gun scanning electron microscopy (FEGSEM) and high-resolution transmission electron microscopy (HR-TEM). As depicted in Fig. 3a, pristine MIL-100 (Fe) displays a morphology that slightly deviates from the typical bipyramidal octahedral shape, instead consisting of irregularly shaped crystals with non-uniform sizes. This can be attributed either to the Fe(BTC) phase or to MIL-100 (Fe) crystals that have still not developed into bipyramids.73 The ARM sample displayed a plate-like morphology (Fig. 3b), which aligns with the findings of the N2 adsorption–desorption analysis and is consistent with existing literature.74 In the context of MARM-II, the observed morphology revealed lamellar ARM particles dispersed over bipyramidal octahedral-shaped MIL-100 (Fe) crystals (Fig. 3c). Moreover, the energy-dispersive X-ray spectroscopy (EDS) spectrum (Fig. 3d), along with the corresponding elemental maps (Fig. 3e–l), confirms the co-occurrence and uniform distribution of C, O, Si, Ti, Fe, and Al within the MARM-II composite. The morphological configuration of pristine ARM and MIL-100 (Fe) within the MARM-II composite was reaffirmed by TEM imaging, as illustrated in Fig. 4a. The selected area electron diffraction (SAED) pattern, characterized by concentric rings and bright spots, indicates the polycrystalline nature of the MARM-II composite (Fig. 4b). As shown in Fig. 4c, the HR-TEM imaging of MARM-II displayed two distinct interplanar spacings of 0.265 nm and 0.318 nm, which could be associated with the (104) plane of α-Fe2O3,75 and (002) plane of sp2 hybridized graphitic carbon.76
 | ||
| Fig. 3 FEG-SEM images of (a) MIL-100 (Fe), (b) ARM, and (c) MARM-II composite. (d) EDS spectra, and (e–l) elemental mapping of MARM-II composite. | ||
 | ||
| Fig. 4 (a) TEM image, (b) SAED pattern, (c) HR-TEM image of MARM-II composite (inset selected area showcasing lattice spacings). | ||
X-ray photoelectron spectroscopy (XPS) was deployed to assess the surface chemical state and elemental composition of MARM-II. As depicted in Fig. 5a, the survey profile indicated the co-existence of various elements, including Fe, O, C, Al, Ti, and Si in MARM-II,77 with pronounced peaks for Fe, O, and C signifying their predominance in the composition. In the high-resolution spectrum of Fe 2p (Fig. 5b), the peaks observed at 722.82 eV and 709.81 eV are linked to Fe 2p1/2 and Fe 2p3/2, respectively.78 The O 1s spectrum can be deconvoluted into three peaks located at 530.87 eV, 529.96 eV, and 528.44 eV (Fig. 5c). The two major peaks observed at binding energies of 530.87 eV and 529.96 eV were ascribed to metal–oxygen bonds, associated with Al–O and Fe–O bonds, respectively.79,80 Moreover, the peak at 528.44 eV could be associated with the C–O/O–H species.81 Furthermore, the high-resolution spectrum of C 1s exhibited three peaks located at 287.06 eV, 284.37 eV, and 283.18 eV, which can be linked to C–O, C–C/C–H, and C–C/C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C species, respectively (Fig. 5d).82–84
C species, respectively (Fig. 5d).82–84
 | ||
| Fig. 5 (a) XPS survey scan of MARM-II composite, and high resolution XPS spectrum of (b) Fe 2p, (c) O 1s, and (d) C 1s. | ||
The thermal stability of the MARM-II composite was determined through thermogravimetric analysis (TGA). As illustrated in Fig. S3, the initial decrease in weight below ∼60 °C corresponds to the release of water molecules trapped in the material's pores. Thereafter, weight loss occurring between ∼60 °C and ∼300 °C relates to the loss of water molecules complexed with the iron trimer clusters present within the MIL-100 (Fe).85 The sudden weight loss of ∼35% noted in the temperature range ∼300–390 °C in the TGA profile of MARM-II is assigned mainly to the disintegration of linkers inside the MIL-100 (Fe) framework.86 Any further weight loss in the temperature range of ∼390–800 °C could likely be associated with the oxidation of the residual linkers of MIL-100 (Fe) in MARM-II. The aforementioned weight loss may also be slightly contributed to the release of CO2 resulting from the thermal decomposition of CaCO3 to CaO in the ARM portion of the MARM-II composite.87 In the differential thermal analysis (DTA) plot of MARM-II, an exothermic peak was observed at ∼386 °C.88
3.2. Adsorptive removal of organic pollutants
The effect of the initial concentrations of LVX, MLB, and CNR on their removal utilizing MARM-II was investigated by adjusting the pollutant concentration from 1 mg L−1 to 50 mg L−1 while maintaining a constant adsorbent dosage of 0.4 g L−1 and a solution pH of 7. As depicted in Fig. 6b, at first, when the initial concentrations of LVX, MLB, and CNR were raised from 1 mg L−1 to 10 mg L−1, the removal efficiencies increased by ∼17%, ∼27%, and ∼34%, respectively. It can be supported by the rationale that the initial number of pollutant molecules on the exposed surface area of the MARM-II composite is minimal at lower concentrations.90 However, when the initial concentrations of LVX, MLB, and CNR were increased from 10 mg L−1 to 50 mg L−1, their respective removal efficiencies decreased from 96.102 ± 0.997%, 93.077 ± 0.593%, and 89.739 ± 1.119% to 68.858 ± 1.648%, 79.173 ± 1.383%, and 72.426 ± 1.186±%, due to the fewer number of adsorptive sites in comparison to the number of pollutant molecules at higher concentrations.90
In contrast, MLB is a cationic phenothiazine dye that exists as a positively charged species, with its positive charge delocalized across the aromatic ring system, primarily on methylated nitrogen groups.97 Reportedly, MLB can exist in aqueous solution as either a cationic species (MLB+) or an undissociated neutral molecule (MLB0); at pH 3, MLB0 predominates (∼86%); at pH equal to its pKa (3.8), both MLB0 and MLB+ co-exist in nearly equal proportions (∼50% each); and at pH values above 6, MLB is present primarily as MLB+.98 Under acidic conditions (pH 3), the adsorption of MLB is comparatively lower than that observed at pH 5 and 7. This is ascribed to the high concentration of H+ ions in the acidic environment, which inhibits the ionization of MLB molecules to some degree, thereby diminishing the adsorption efficiency.99 At neutral pH, the MARM-II surface carries a slight negative charge, which promotes electrostatic attraction with the MLB+ species, thereby resulting in maximum removal efficiency. Furthermore, under alkaline conditions (pH 9 and 11), excess OH− ions in solution electrostatically stabilize MLB+, making it preferentially remain solvated rather than adsorbed onto the negatively charged MARM-II surface.99
In the context of LVX, the relationship between the removal efficiency and the solution pH can be elucidated based on electrostatic interactions between the LVX molecules and the positively/negatively charged surface of MARM-II. The LVX molecule exhibits two characteristic dissociation constants (pKa1 = 5.59 and pKa2 = 7.94),100 which regulate its ionization behavior, and result in the predominance of three species under different pH conditions: the protonated form (LVX+), the zwitterionic form (LVX±), and the monoanionic form (LVX−). At pH 3, the positively charged surface of MARM-II likely repels LVX+ species, resulting in a significantly low removal efficiency of 54.713 ± 1.348%. As the solution pH increased to 5, this electrostatic repulsion weakened, and the removal efficiency gradually improved, reaching around 82%. At neutral pH, LVX predominantly existed in LVX± form, where the protonated –NH+2 group interacted favorably with the slightly negatively charged MARM-II surface, and likely led to maximum LVX adsorption. Furthermore, at more alkaline pH values, increasingly negative charge of the MARM-II surface induces strong electrostatic repulsion to the LVX− species, thereby reducing removal efficiency. Overall, at neutral pH, the simultaneous removal of MLB, CNR, and LVX was maximum; therefore, it was selected as the optimum for subsequent studies.
3.3. Adsorption kinetics study
Adsorption kinetics pertains to the rate and mechanism by which pollutants transition from the solution phase to the surface of the adsorbent. Kinetic models aid in determining whether the process is regulated by surface reactions, pore diffusion, or a combination of both. The findings of the kinetics study are displayed in Fig. 7a–d, with the corresponding parameters summarized in Table S6. The kinetic study showcased that both PFO and PSO kinetic models exhibited excellent fitting (R2 > 0.99) for the adsorption of MLB, CNR, and LVX onto MARM-II composite. The fit to the PSO kinetic model showed the possibility that chemisorption, comprising valence forces through electron sharing or exchange,108 might play a primary role in the adsorptive removal of the aforementioned pollutants. However, the calculated enthalpy values for MLB (−32.47 kJ mol−1), CNR (−22.43 kJ mol−1), and LVX (−60.04 kJ mol−1) in Section 3.5, are notably lower than the chemisorption range (80–240 kJ mol−1)109 typically linked with the chemical bond formation. Therefore, the adsorption mechanism for MLB and CNR, showcasing low enthalpy values, is consistent with physisorption. On the contrary, the higher enthalpy value noted for LVX implies borderline between strong physisorption and weak chemisorption.110 The pollutant-specific kinetics indicated that LVX demonstrated rapid adsorption, characterized by the highest rate constants in both PFO (k1 = 0.1421 min−1) and PSO (k2 = 0.0140 g mg−1. min), which signifies a strong affinity and swift uptake. MLB showed slightly lower rate constants (k1 = 0.1192 min−1, k2 = 0.0113 g mg−1. min), whereas CNR demonstrated the slowest kinetics (k1 = 0.0914 min−1; k2 = 0.0065 g mg−1 min−1). The rationale for this observation is that existing adsorptive sites become increasingly shielded by previously adsorbed dye or restricted by steric hindrance, leading to a reduced adsorption rate.111The Elovich kinetic model further delved into the heterogeneous nature of the MARM-II surface. The α value was significantly high for MLB (3.26 × 105 mg g−1 min−1) and LVX (9.37 × 107 mg g−1 min−1), implying rapid coverage of adsorptive sites during the initial stage of the adsorption process.112 Contrastingly, CNR displayed a notably lower α value (497.88 mg g−1 min−1), indicating its slower uptake and weaker interaction with the active sites on the MARM-II surface. Moreover, β, which correspond to the extent of surface coverage and the change in activation energy during chemisorption,113 was lowest for CNR. This indicates a gradual increase in activation energy as adsorption proceeds, resulting in a slower decline in adsorption rate. The IPD model highlights a multi-stage adsorption mechanism marked by an initial rapid uptake (Stage I), followed by a slower intraparticle diffusion phase (Stage II), and concluding with a final equilibrium plateau (Stage III). The plots for MLB, CNR, and LVX did not exactly pass through the origin, implying that the intraparticle diffusion was not the only rate-limiting step.114 Overall, the aforementioned results establish that MLB, CNR, and LVX adsorption onto the MARM-II composite is governed mainly by physisorption with rapid initial uptake, followed by slow diffusion into pores and adsorptive site saturation.
3.4. Adsorption isotherms study
To better understand the equilibrium mechanism of MLB, CNR, and LVX adsorption onto the MARM-II composite, the experimental data were fitted to Langmuir, Freundlich, Sips, and Redlich–Peterson isotherms. The findings of the adsorption isotherms study are presented in Fig. 8a–c, and the related parameters are mentioned in Table S7. In the context of the Langmuir isotherm, adsorption occurs through chemical binding at a finite number of well-defined sites, ultimately leading to the formation of a saturated monolayer on the adsorbent surface.115 The fitted parameters showed Qmax to be 123.021 ± 11.9261 mg g−1, 143.934 ± 24.248 mg g−1, 97.657 ± 5.686 mg g−1 for MLB, CNR, and LVX, respectively, which indicated a strong affinity of the MARM-II composite towards the aforementioned pollutants. The higher Qmax value for CNR in comparison to others can be ascribed to the –SO3− and –NH2 groups, which strengthen electrostatic attraction, H-bonding, respectively, alongside its aromatic structure that facilitates π–π interactions with the adsorbent surface.92While the Langmuir isotherm for CNR showcased a satisfactory fit with R2 ranging between 0.963–0.977, the comparatively high χ2 value (∼38.622) indicated marginal deviations from ideal monolayer adsorption. Furthermore, the Freundlich isotherm (Fig. 8d–f), representative of multilayer adsorption on a heterogeneous surface,115 displayed n > 1 for all pollutants (2.454 for LVX, 1.784 for MLB, and 1.539 for CNR), confirming favorable adsorption. Notably, the Freundlich fits displayed R2 values ranging from 0.939 to 0.968, lower than those obtained from the Langmuir isotherm (0.973–0.992). This shows that the adsorption of LVX, CNR, and MLB occurs heterogeneously on the MARM-II surface, but the Freundlich isotherm cannot solely depict the equilibrium behavior of the system.
The Sips isotherm (Fig. 8g–i), comprising attributes of both Langmuir and Freundlich isotherms, displayed the best fit, with the lower χ2, compared to other models. For LVX (m = 0.941) and MLB (m = 1.185), the value of m approaches 1, suggesting that the adsorption behavior approximates the Langmuir model defined by monolayer adsorption. On the contrary, a higher m value for CNR (m = 1.589) indicates stronger heterogeneity effects, possibly attributed to steric hindrance from its bulky molecular structure, which restricts adsorptive site accessibility.116 Likewise, the Redlich–Peterson isotherm (Fig. 8j–l), which integrates aspects of both the Langmuir and the Freundlich isotherms, demonstrated satisfactory fits (R2 = 0.94–0.996 for LVX; 0.978–0.992 for MLB; 0.973–0.977 for CNR), thereby validating the aforementioned finding.
3.5. Adsorption thermodynamics study
The thermodynamics study was conducted to evaluate the spontaneity and exothermic/endothermic nature of the adsorption of MLB, CNR, and LVX onto the MARM-II composite. The findings of the thermodynamics study are depicted in Fig. S7 and the associated parameters mentioned in Table S8. The negative ΔG° values for the aforementioned pollutants, in conjunction with rising temperature, suggest that the adsorptive process is spontaneous.117 The negative ΔH° values reveal the exothermic nature of MLB, CNR, and LVX adsorption.118 Moreover, the negative ΔS° values for MLB and LVX signify increased orderliness at the adsorbent–adsorbate interface due to strong binding of the pollutants on the MARM-II surface.119,120 The slightly positive ΔS° value noted for CNR indicated an increase in randomness at the adsorbate–adsorbent interface possibly due to the partial desolvation effects during adsorption process.121 The Van't Hoff analysis's dependability is shown by the R2 values (0.913–0.981), thus explaining almost all variability in the observed data.3.6. Adsorptive mechanisms for MLB, CNR, and LVX
The mechanistic insights governing the adsorptive removal of LVX, MLB, and CNR using the MARM-II composite arise from the combined effects of their functional groups, the pH-dependent speciation of the aforementioned pollutants, and the surface chemistry of the MARM-II composite (Fig. 9). The mechanistic insights regarding MLB, CNR, and LVX adsorption onto the MARM-II surface have been elaborately covered in Section 3.2.3 and summarized in the present section. The LVX molecule primarily exists as LVX± species, depending on its pKa values, where positively charged –NH2+ group exhibits strong electrostatic affinity toward the slightly negative surface of MARM-II (pHZPC located at pH 6.35), ensuring its maximum removal. CNR was effectively adsorbed onto the MARM-II surface at multiple pH conditions, attributed to the functional groups, electrostatic attraction, and chemisorption.Under acidic conditions, the negatively charged –SO3− groups of CNR exhibit strong electrostatic attraction toward the positively charged surface of MARM-II, thereby enhancing its removal efficiency. At neutral pH, the hydrogen bonding between the –COO− groups on MIL-100 (Fe) and the –NH2 groups of CNR, alongside π–π interactions between the aromatic moieties of the adsorbent and adsorbate, overcome the mild electrostatic repulsion, promoting CNR adsorption. The chemisorption of CNR molecules onto the open Fe sites of the MIL-100 (Fe) component within the MARM-II composite contributes to the observed adsorption at alkaline pH. Furthermore, the electrostatic attraction governed the adsorption mechanism for MLB removal, as the slightly negatively charged surface of MARM-II causes interaction with the MLB+ species, leading to maximum removal at neutral pH.
As previously reported in multi-contaminant systems, adsorption capacities/removal efficiencies in binary and ternary mixtures often differ from those observed in single-adsorbate systems due to competitive interactions among co-existing contaminants.122–124 Such differences arise primarily from (i) competition for the same number of active adsorption sites, and surface coverage and pore blocking by co-adsorbates,124 (ii) antagonistic effects influenced by solution pH and resulting electrostatic interactions,123 and (iii) differences in molecular affinity and strength of non-covalent interactions (π–π interactions, H-bonding).122 In the present study, applying these established observations to our MLB-CNR-LVX system on the MARM-II surface (pHZPC ∼ 6.35), the selectivity trend (LVX > MLB > CNR) is consistent with a competitive scenario where LVX can access multiple interaction pathways (π–π interactions, H-bonding, and coordination to Fe sites), allowing it to outcompete dyes, while MLB is favored over CNR under near-neutral conditions due to stronger attraction to the slightly negative surface and faster uptake. Whereas, CNR is more easily suppressed in the mixture because it experiences less favorable electrostatic attraction and competes for the same number of active adsorptive sites, leading to reduced accessibility in the presence of higher-affinity species.
3.7. Desorption and reusability study
The regenerative potential and possible reuse of MARM-II composite for the adsorptive removal of MLB, CNR, and LVX were assessed through the reusability study. MARM-II composite showed a marginal drop in removal efficiency when tested up to five consecutive cycles. Following each cycle, 0.5 M HCl, 0.5 M NaOH, and acetone were used in a predefined order to regenerate the adsorptive sites on the MARM-II composite. After the fifth cycle, the MLB, CNR, and LVX removal was found to be ∼79%, ∼74%, and ∼83%, respectively (Fig. S8). The decrease in removal efficiencies can be linked to the saturation of adsorptive sites due to the incomplete desorption of organic pollutants,94 and the structural disintegration of the MARM-II composite19 after several washes and drying. The diffractogram and FTIR spectrum of MARM-II composite have been presented in Fig. 10a and b, depicting differences in comparison to the pristine material. Additionally, ICP-MS analysis indicated that the concentration of Fe leached in the treated solution was negligible (∼0.054 mg L−1), much lower than the acceptable limit of Fe in the drinking water (0.3 mg L−1),125 thereby confirming the chemical stability of the MARM-II composite. The leached amounts of the other two primary elements,37 i.e., Al (∼0.013 mg L−1) and Ti (∼0.071 × 10−3 mg L−1), were also insignificant. The possibility of organic linker leaching from the MARM-II composite was investigated as a control study. The optimal dosage of MARM-II was added to a beaker containing 100 mL of DI water and stirred for the specified contact time. The aliquot was then centrifuged, and the total organic carbon (TOC) of the supernatant was measured. The TOC of the supernatant was found to be in close agreement with that of the DI water used in the study, indicating negligible leaching of the H3BTC linker.3.8. Economic analysis and scalability
The cost assessment of the MARM-II adsorbent was performed, including the cost incurred for the MARM-II composite and the operational expenditures for treating a mixture of MLB, CNR, and LVX. Table S9 presents the chemical and energy costs involved in the synthesis of the MARM-II composite. The chemical costs were evaluated according to the current market value, while the energy cost was estimated based on the rate established by the West Bengal State Electricity Distribution Company Limited (WBSEDCL).126 The total cost of the MARM-II composite was calculated to be 0.56 US $ per g. Furthermore, the use of a multi-point magnetic stirrer allows scaling opportunity with altered chemical cost while the energy cost remains the same. This allows a cost reduction of about 53.6%, bringing it down to 0.27 US $ per g. In this context, the reported costs of similar MOF-based materials include MIL-100(Fe)–Fe0 (0.19 US $ per g)127 and MIL-100(Fe)-ore (0.11 US $ per g),127 MIL-101(Cr) (4.28 US $ per g),128 and UiO-66@kaolin (0.0716 US $ per g).129 Furthermore, Biswal et al. (2021) reviewed that the production cost of similar adsorbents typically ranges between 0.1 and 5 US $ per g depending on the synthesis route.130 Thus, the estimated cost of the present MARM-II adsorbent is within the reported range and remains competitive. The treatment cost (after scale-up) for a mixture of MLB, CNR, and LVX was calculated to be 0.13 US $ per L.4. Summary and conclusions
The MARM-II composite was synthesized by integrating calcined RM during the preparation of MIL-100 (Fe) via co-precipitation. The as-prepared composite (MARM-II) was utilized for the adsorptive removal of MLB, CNR, and LVX from an aqueous solution. At the optimal condition (initial MLB, CNR, and LVX concentration: 10 mg L−1 each, MARM-II dose: 0.4 g L−1, and solution pH: 7, and temperature: 27 ± 3 °C), about 93%, 96%, and 90% of MLB, LVX, and CNR were removed. The PSO kinetic model and findings from thermodynamics study suggested that physisorption is the primary mechanism governing the adsorption of the specified pollutants onto the MARM-II composite. The adsorption isotherm study indicated that the Sips isotherm, which carries characteristics of both the Langmuir and the Freundlich isotherms, effectively describes the adsorption process. The Qmax for MLB, CNR, and LVX were obtained to be 123.021 ± 11.926, 143.934 ± 24.248, and 97.657 ± 5.686 mg g−1, respectively. The remarkable adsorptive potential of MARM-II is ascribed to its enhanced SSA, which provides abundant adsorptive sites, surface functional groups (–COO−, –OH, –NH2) that facilitate H-bonding and π–π interactions. The aforementioned features act synergistically to ensure multi-pollutant adsorption onto the MARM-II surface. The thermodynamic investigation indicated that the adsorption of the aforementioned pollutants occurs as an exothermic and spontaneous process. The adsorptive process was largely affected in the presence of HCO3− ions, owing to their competition with the adsorbate molecules for the available adsorptive sites. The desorption and reusability study demonstrated that the MARM-II composite can be efficiently utilized for up to five cycles, showcasing its regenerative potential. This study advocates for the sustainable use of red mud, converting it into a valuable resource, specifically an effective adsorbent for the remediation of organic pollutants in water and wastewater, thus resulting in both water quality improvements and the reuse of waste materials.Author contributions
Adarsh Singh: writing – original draft, validation, methodology, investigation, formal analysis, data curation, conceptualization. Manne Chimyang: writing – original draft, validation, methodology, investigation, formal analysis, data curation, conceptualization. Akash Rawat: writing – original draft, validation, methodology, investigation, formal analysis, data curation, conceptualization. Venkatesh Uddameri: writing – review & editing, visualization, supervision, methodology, conceptualization. Ashok Kumar Gupta: writing – review & editing, visualization, supervision, methodology, conceptualization.Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d6ra00635c.
Acknowledgements
Adarsh Singh, Manne Chimyang, and Akash Rawat are thankful to the Indian Institute of Technology Kharagpur, India, for the financial support.References
- A. Rawat, A. Srivastava, A. Bhatnagar and A. K. Gupta, J. Cleaner Prod., 2023, 383, 135382 CrossRef CAS
.
- S. Boddu, M. Chavali, J. B. Dulla, V. N. Allugunulla, I. Mikkili, S. Malladi, S. Mannepalli and A. A. Khan, Biomass Convers. Biorefin., 2025, 15, 15399–15415 CAS
.
- N. Hanna, P. Sun, Q. Sun, X. Li, X. Yang, X. Ji, H. Zou, J. Ottoson, L. E. Nilsson, B. Berglund, O. J. Dyar, A. J. Tamhankar and C. Stålsby Lundborg, Environ. Int., 2018, 114, 131–142 CAS
.
- A. Ellafi, A. Dali, S. Mnif and S. Ben Younes, Catal. Lett., 2023, 153, 3620–3633 CrossRef CAS
.
- B. Chakraborty, L. Ray and S. Basu, Desalin. Water Treat., 2016, 57, 14077–14082 CAS
.
- A. Orzoł and A. I. Piotrowicz-Cieślak, Environ. Sci. Pollut. Res., 2017, 24, 22226–22240 Search PubMed
.
- A. Kanwal, R. Rehman, M. Imran, G. Samin, M. M. Jahangir and S. Ali, RSC Adv., 2023, 13, 26455–26474 CAS
.
- R. O. Alves de Lima, A. P. Bazo, D. M. F. Salvadori, C. M. Rech, D. de Palma Oliveira and G. de Aragão Umbuzeiro, Mutat. Res., Genet. Toxicol. Environ. Mutagen., 2007, 626, 53–60 CAS
.
- V. Bhardwaj, T. Bhardwaj, K. Sharma, A. Gupta, S. Chauhan, S. S. Cameotra, S. Sharma, R. Gupta and P. Sharma, RSC Adv., 2014, 4, 24935–24943 Search PubMed
.
- J.-Q. Xiong, M. B. Kurade and B.-H. Jeon, Chem. Eng. J., 2017, 313, 1251–1257 CrossRef CAS
.
- G. Chaturvedi, A. Kaur, A. Umar, M. A. Khan, H. Algarni and S. K. Kansal, J. Solid State Chem., 2020, 281, 121029 CrossRef CAS
.
- S. H. Mousavi, M. Yaghoobi and F. Asjadi, Sci. Rep., 2024, 14, 18505 CrossRef CAS PubMed
.
- S. Kundu and A. K. Gupta, Chem. Eng. J., 2006, 122, 93–106 CrossRef CAS
.
- S. Ayoob, A. K. Gupta, P. B. Bhakat and V. T. Bhat, Chem. Eng. J., 2008, 140, 6–14 CrossRef CAS
.
- P. S. Ghosal and A. K. Gupta, RSC Adv., 2015, 5, 105889–105900 RSC
.
- Y. Pi, C. Duan, Y. Zhou, S. Sun, Z. Yin, H. Zhang, C. Liu and Y. Zhao, J. Hazard. Mater., 2022, 424, 127577 CrossRef CAS PubMed
.
- A. Imessaoudene, S. Cheikh, A. Hadadi, N. Hamri, J.-C. Bollinger, A. Amrane, H. Tahraoui, A. Manseri and L. Mouni, Separations, 2023, 10, 57 CrossRef CAS
.
- K. Gupta and O. P. Khatri, Chem. Eng. J., 2019, 378, 122218 CrossRef CAS
.
- S. Dutta, S. K. Srivastava, B. Gupta and A. K. Gupta, ACS Appl. Mater. Interfaces, 2021, 13, 54324–54338 CrossRef CAS PubMed
.
- Z. Yu, H. Chen, J. Zhang, W. Sun, T. Li, Z. Qiu and M. Younas, Sep. Purif. Technol., 2025, 352, 128108 CrossRef CAS
.
- S. Zhu, H. Xu, M. S. Khan, M. Xia, F. Wang and Y. Chen, Water Res., 2025, 272, 122997 CrossRef CAS PubMed
.
- Y. Chen, H. Xu, M. S. Khan, S. Han and S. Zhu, Crit. Rev. Environ. Sci. Technol., 2025, 55, 1097–1123 CrossRef CAS
.
- A. Singh, S. K K, A. Bhatnagar and A. K. Gupta, Sep. Purif. Technol., 2025, 353, 128588 CrossRef CAS
.
- S.-H. Huo and X.-P. Yan, J. Mater. Chem., 2012, 22, 7449 RSC
.
- L. Oar-Arteta, T. Wezendonk, X. Sun, F. Kapteijn and J. Gascon, Mater. Chem. Front., 2017, 1, 1709–1745 Search PubMed
.
- T. Yang, Y. Teng, J. Wang, S. Jia, Y. Su, X. Li, L. Li and C. Wang, New J. Chem., 2023, 47, 3903–3909 RSC
.
- W. He, Z. Li, S. Lv, M. Niu, W. Zhou, J. Li, R. Lu, H. Gao, C. Pan and S. Zhang, Chem. Eng. J., 2021, 409, 128274 CrossRef CAS
.
- A. Rawat, S. K. Srivastava, C. S. Tiwary and A. K. Gupta, J. Mater. Chem. A, 2025, 13, 1271–1286 RSC
.
- F. Tan, M. Liu, K. Li, Y. Wang, J. Wang, X. Guo, G. Zhang and C. Song, Chem. Eng. J., 2015, 281, 360–367 CrossRef CAS
.
- R. Wang, H. Xu, X. Liu, D. Fang, S. Wei and A.-N. Yu, Surf. Interfaces, 2022, 34, 102325 CrossRef CAS
.
- Y. Liu and R. Naidu, Waste Manage., 2014, 34, 2662–2673 CrossRef CAS PubMed
.
- J. Jin, X. Liu, S. Yuan, P. Gao, Y. Li, H. Zhang and X. Meng, J. Ind. Eng. Chem., 2021, 98, 298–307 Search PubMed
.
- S. Mesgari Abbasi, A. Rashidi, A. Ghorbani and G. Khalaj, Ceram. Int., 2016, 42, 16738–16743 CrossRef CAS
.
- G. Power, M. Gräfe and C. Klauber, Hydrometallurgy, 2011, 108, 33–45 CrossRef CAS
.
- Y.-T. Xu, K.-C. Ren, Z.-M. Tao, D. K. Sam, E. Feng, X. Wang, G. Zhang, J. Wu and Y. Cao, Green Chem., 2023, 25, 589–595 Search PubMed
.
- J. Carneiro, D. M. Tobaldi, W. Hajjaji, M. N. Capela, R. M. Novais, M. P. Seabra and J. A. Labrincha, Ceram. Int., 2018, 44, 4211–4219 CrossRef CAS
.
- A. Singh and A. K. Gupta, Environ. Sci. Nano, 2026, 13, 563–581 RSC
.
- W. Fang, Y. Zhou, M. Cheng, L. Zhang, T. Zhou, Q. Cen, B. Li and Z. Liu, J. Mol. Liq., 2024, 407, 125171 CrossRef CAS
.
- Q. Wang, J. Chen, X. Li, X. Yang, Y. Wu, S. Li, Y. Ye, D. Wang, D. Wang and Z. Zheng, Fuel Process. Technol., 2022, 233, 107316 CrossRef CAS
.
- T. Steenhaut, S. Hermans and Y. Filinchuk, New J. Chem., 2020, 44, 3847–3855 RSC
.
- E. S. Dragan, D. F. Apopei Loghin and A. I. Cocarta, ACS Appl. Mater. Interfaces, 2014, 6, 16577–16592 Search PubMed
.
- Y. S. Ho and G. Mckay, Can. J. Chem. Eng., 1998, 76, 822–827 CAS
.
- S. Y. Elovich and O. G. Larionov, Bull. Acad. Sci. USSR, Div. Chem. Sci., 1962, 11, 191–197 Search PubMed
.
- W. J. Weber and J. C. Morris, J. Sanit. Eng. Div., 1963, 89, 31–59 Search PubMed
.
- I. Langmuir, J. Am. Chem. Soc., 1918, 40, 1361–1403 CAS
.
- H. Freundlich and W. Heller, J. Am. Chem. Soc., 1939, 61, 2228–2230 CAS
.
- R. Sips, J. Chem. Phys., 1948, 16, 490–495 CAS
.
- O. Redlich and D. L. Peterson, J. Phys. Chem., 1959, 63, 1024 CAS
.
- C. Chen, M. Zhang, Q. Guan and W. Li, Chem. Eng. J., 2012, 183, 60–67 CAS
.
- E. C. Lima, A. Hosseini-Bandegharaei, J. C. Moreno-Piraján and I. Anastopoulos, J. Mol. Liq., 2019, 273, 425–434 CAS
.
- J. Saleem, Z. K. B. Moghal, S. Pradhan and G. McKay, RSC Adv., 2024, 14, 33797–33808 CAS
.
- A. Dhakshinamoorthy, M. Alvaro, Y. K. Hwang, Y.-K. Seo, A. Corma and H. Garcia, Dalton Trans., 2011, 40, 10719 CAS
.
- C. Brahmi, M. Benltifa, C. Vaulot, L. Michelin, F. Dumur, E. Gkaniatsou, C. Sicard, A. Airoudj, F. Morlet-Savary, L. Bousselmi and J. Lalevée, ChemistrySelect, 2021, 6, 8120–8132 CAS
.
- M. A. El-Aal, A. E.-A. A. Said, M. H. Abdallah and M. N. Goda, Sci. Rep., 2022, 12, 9407 CAS
.
- Y. Chen, S. Gao, E. J. Jones and B. Singh, Environ. Sci. Technol., 2021, 55, 4629–4637 CAS
.
- Q. Li, G. Wei, Y. Yang, Z. Li, L. Zhang and Q. Huang, J. Hazard. Mater., 2020, 394, 122566 CrossRef CAS PubMed
.
- J. Zan, H. Song, S. Zuo, X. Chen, D. Xia and D. Li, J. Cleaner Prod., 2020, 246, 118971 CAS
.
- H. Li, F. Liu, X. Ma, Z. Wu, Y. Li, L. Zhang, S. Zhou and Y. Helian, Energy Convers. Manage., 2019, 180, 401–410 CAS
.
- Q. Li, X. Luo, J. Cai, G. Li, P. Wang, W. Han and Y. Lan, J. Catal., 2024, 436, 115625 CAS
.
- J.-G. Lee, B. N. Joshi, E. Samuel, S. An, M. T. Swihart, J. S. Lee, Y. K. Hwang, J.-S. Chang and S. S. Yoon, J. Alloys Compd., 2017, 722, 996–1001 CAS
.
- K. Vinothkumar and R. G. Balakrishna, Appl. Catal., B, 2024, 340, 123199 CrossRef CAS
.
- A. Ene, S. S. Moraru, D. I. Moraru, A. Pantelica, S. Gosav and A. M. Ceoromila, Appl.
Sci., 2024, 14, 5616 CrossRef CAS
.
- K. D. Nguyen, T. T. Thu, A. T. H. Tran, O. T. K. Le, S. Sagadevan and N. H. Mohd Kaus, ACS Omega, 2023, 8, 41258–41272 CrossRef CAS PubMed
.
- A. Čiuladienė, A. Luckutė, J. Kiuberis and A. Kareiva, Chemija, 2018, 29, 243–256 CrossRef
.
- M. A. Al-Kazragi, D. T. A. Al-Heetimi and O. S. A. Al-Khazrajy, Desalin. Water Treat., 2019, 145, 369–377 Search PubMed
.
- P. Sarawade, H. Tan, D. Anjum, D. Cha and V. Polshettiwar, ChemSusChem, 2014, 7, 529–535 CrossRef CAS PubMed
.
- Y. J. C. Martins, A. C. M. Almeida, B. M. Viegas, R. A. do Nascimento and N. F. da P. Ribeiro, Int. J. Environ. Sci. Technol., 2020, 17, 4133–4148 CrossRef CAS
.
- H. Shi, Z. Gu, M. Han, C. Chen, Z. Chen, J. Ding, Q. Wang, H. Wan and G. Guan, Colloids Surf., A, 2021, 608, 125585 CrossRef CAS
.
- F. Lian, C. Chang, Y. Du, L. Zhu, B. Xing and C. Liu, J. Environ. Sci., 2012, 24, 1549–1558 CrossRef CAS PubMed
.
- D. Liu, D. Zou, H. Zhu and J. Zhang, Small, 2018, 14, 1801454 CrossRef PubMed
.
- Hierarchically Structured Porous Materials, ed. B. Su, C. Sanchez and X. Yang, Wiley, 2011 Search PubMed
.
- H. Weng, P. Zhang, Z. Guo, G. Chen, W. Shen, J. Chen, X. Zhao and M. Lin, ACS Appl. Mater. Interfaces, 2021, 13, 8249–8262 CrossRef CAS PubMed
.
- J. J. Delgado-Marín, J. Narciso and E. V. Ramos-Fernández, Materials, 2022, 15, 6499 CrossRef PubMed
.
- V. K. Singh, A. Sett and S. Karmakar, Chem. Eng. J., 2024, 481, 148373 CrossRef CAS
.
- S. Kim, P. Anushkkaran, W.-S. Chae, S. H. Choi, M. Kumar, M. Cho, M. A. Mahadik, H. H. Lee and J. S. Jang, ACS Appl. Energy Mater., 2022, 5, 915–929 CrossRef CAS
.
- H. Wang, J. Di, Y. Sun, J. Fu, Z. Wei, H. Matsui, A. del C. Alonso and S. Zhou, Adv. Funct. Mater., 2015, 25, 5537–5547 CrossRef CAS
.
- NIST X-Ray Photoelectron Spectroscopy Database (SRD 20), Version 5.0 Search PubMed
.
- C. Dhanasekhar, S. K. Mishra, R. Rawat, A. K. Das and A. Venimadhav, J. Alloys Compd., 2017, 726, 148–153 CrossRef CAS
.
- P. Zhang, J. Wu, Y. Wang, H. Sarvari, D. Liu, Z. D. Chen and S. Li, J. Mater. Chem. A, 2017, 5, 17368–17378 RSC
.
- M. Du, J. Zhang, J. Rong, T. Peng, Y. Chen, Y. Ji and Y. Guan, MRS Commun., 2024, 14, 1371–1379 CrossRef CAS
.
- B. Zhao, X. Fu, Y. Di, L. Wei, G. Shao, H. Cui, L. Wei, N. Liu, Q. An and S. Zhai, J. Mol. Struct., 2024, 1312, 138619 CrossRef CAS
.
- R. Narukulla, U. Ojha and T. Sharma, Colloids Surf., A, 2022, 641, 128594 CrossRef CAS
.
- Q. Wu, W. Luo, Y. Du, Y. Yang, Y. Xiao, N. Cheng, B. Tang, H. Li, L. Wang, D. Wang, C. Wang, J. Guan and X. Shen, Biomacromolecules, 2023, 24, 3522–3531 CrossRef CAS PubMed
.
- A. Xie, L. Cao, T. Shen, Y. Zhou, H. Wang and S. Luo, Microchem. J., 2024, 204, 111101 CrossRef CAS
.
- N. U. Qadir, S. A. M. Said, R. B. Mansour, K. Mezghani and A. Ul-Hamid, Dalton Trans., 2016, 45, 15621–15633 RSC
.
- Y.-K. Seo, J. W. Yoon, J. S. Lee, U.-H. Lee, Y. K. Hwang, C.-H. Jun, P. Horcajada, C. Serre and J.-S. Chang, Microporous Mesoporous Mater., 2012, 157, 137–145 CrossRef CAS
.
- R. C. Sahu, R. Patel and B. C. Ray, Fuel Process. Technol., 2011, 92, 1587–1592 CrossRef CAS
.
- I. Bezverkhyy, E. Popova, N. Geoffroy, F. Herbst and J.-P. Bellat, J. Mater. Chem. A, 2016, 4, 8141–8148 RSC
.
- S. Basu, G. Ghosh and S. Saha, Process Saf. Environ. Prot., 2018, 117, 125–142 CrossRef CAS
.
- V. Basavarao and S. Rammohanrao, Chem. Eng. J., 2006, 116, 77–84 CrossRef
.
- M. H. Dehghani, M. Ghadermazi, A. Bhatnagar, P. Sadighara, G. Jahed-Khaniki, B. Heibati and G. McKay, J. Environ. Chem. Eng., 2016, 4, 2647–2655 CrossRef CAS
.
- Y. Liu, G. Qiu, Y. Liu, Y. Niu, R. Qu, C. Ji, Y. Wang, Y. Zhang and C. Sun, J. Mol. Liq., 2022, 360, 119405 CrossRef CAS
.
- L. P. Lingamdinne, G. K. R. Angaru, B. Shrestha, J. R. Koduru and R. R. Karri, Sci. Rep., 2026, 16, 7794 CrossRef CAS PubMed
.
- S. Sudarsan, G. Murugesan, T. Varadavenkatesan, R. Vinayagam and R. Selvaraj, Sci. Rep., 2025, 15, 1831 CrossRef CAS PubMed
.
- T. H. Ho, H. D. Tong and T. T. Trinh, Chem. Eng. J. Adv., 2026, 25, 101017 CrossRef
.
- S. E. Moradi, S. Dadfarnia, A. M. Haji Shabani and S. Emami, Desalin. Water Treat., 2015, 56, 709–721 CrossRef CAS
.
- G. Hähner, A. Marti, N. D. Spencer and W. R. Caseri, J. Chem. Phys., 1996, 104, 7749–7757 CrossRef
.
- J. J. Salazar-Rabago, R. Leyva-Ramos, J. Rivera-Utrilla, R. Ocampo-Perez and F. J. Cerino-Cordova, Sustainable Environ. Res., 2017, 27, 32–40 CrossRef CAS
.
- R. Song, J. Yao, M. Yang and Z. Ye, Langmuir, 2022, 38, 9400–9409 CrossRef CAS PubMed
.
- F. Hasanvandian, A. Shokri, M. Moradi, B. Kakavandi and S. Rahman Setayesh, J. Hazard. Mater., 2022, 423, 127090 CrossRef CAS PubMed
.
- W. Li, T. Zhang, L. Lv, Y. Chen, W. Tang and S. Tang, Colloids Surf., A, 2021, 624, 126791 CrossRef CAS
.
- G. M. Sawood, A. Mishra and S. K. Gupta, J. Hazard., Toxic Radioact. Waste, 2021, 25, 04020075 CrossRef CAS
.
- N. Y. Acelas, C. Hadad, A. Restrepo, C. Ibarguen and E. Flórez, Inorg. Chem., 2017, 56, 5455–5464 CrossRef CAS PubMed
.
- Q. Zheng, Y. Luo and Z. Luo, Environ. Sci. Pollut. Res., 2022, 30, 27241–27256 CrossRef PubMed
.
- R. J. Martin and K. O. Iwuco, Water Res., 1982, 16, 73–82 CrossRef CAS
.
- Y. Aldegs, M. Elbarghouthi, A. Elsheikh and G. Walker, Dyes Pigm., 2008, 77, 16–23 CrossRef CAS
.
- X. Zheng, Z. Xu, J. Liu, Y. Luo, L. Gu, D. Zhao, S. Hu and X. Pan, Carbon Res., 2022, 1, 27 CrossRef CAS
.
- W. Li, P. Zuo, D. Xu, Y. Xu, K. Wang, Y. Bai and H. Ma, Chem. Eng. Res. Des., 2017, 124, 260–270 CrossRef CAS
.
- A. Thakur, H. Assad, S. Kaya and A. Kumar, in Eco-Friendly Corrosion Inhibitors, Elsevier, 2022, pp. 283–310 Search PubMed
.
- X. Huang, N. Gao and Q. Zhang, J. Environ. Sci., 2007, 19, 1287–1292 CrossRef CAS PubMed
.
- W. A. El-Mehalmey, A. Helmy, M. A. Seleem, M. Amin, A. S. Mayhoub and M. H. Alkordi, J. Mater. Chem. A, 2025, 13, 22563–22573 RSC
.
- S. Sivalingam and A. S, Environ. Sci. Adv., 2024, 3, 732–750 CAS
.
- P. Liu, Z. Wu, X. Ge and X. Yang, RSC Adv., 2019, 9, 11696–11706 RSC
.
- S. Mohamed Nasser, M. Abbas and M. Trari, Prog. React. Kinet. Mech., 2024, 49, 14686783241226858 CrossRef
.
- É. C. Lima, M. A. Adebayo and F. M. Machado, 2015, Kinetic and Equilibrium Models of Adsorption, pp. 33–69 Search PubMed
.
- H. Lachheb, E. Puzenat, A. Houas, M. Ksibi, E. Elaloui, C. Guillard and J.-M. Herrmann, Appl. Catal., B, 2002, 39, 75–90 CrossRef CAS
.
- Z. A. Jamiu, T. A. Saleh and S. A. Ali, RSC Adv., 2015, 5, 42222–42232 RSC
.
- E. Wibowo, M. Rokhmat, Sutisna, Khairurrijal and M. Abdullah, Desalination, 2017, 409, 146–156 Search PubMed
.
- K. Wu, Q. Nan and T. Wu, Complexity, 2020, 2020, 1–11 Search PubMed
.
- T. E. Khalil, E. El-Meligy, L. A. Mohamed, A. El-dissouky and M. Amer, Int. J. Biol. Macromol., 2025, 318, 145335 CrossRef CAS PubMed
.
- F. Khalili and G. Al-Banna, J. Environ. Radioact., 2015, 146, 16–26 CrossRef CAS PubMed
.
- M. G. Oliveira, M. P. Spaolonzi, E. D. V. Duarte, H. P. S. Costa, M. G. C. da Silva and M. G. A. Vieira, Environ. Res., 2023, 233, 116503 CrossRef CAS PubMed
.
- E. Duque-Brito, D. R. Lobato-Peralta, J. A. Okolie, D. M. Arias, P. J. Sebastian and P. U. Okoye, Energy, Ecol. Environ., 2024, 9, 84–99 CrossRef CAS
.
- S. Bentahar, A. Dbik, M. El Khomri, N. El Messaoudi and A. Lacherai, J. Environ. Chem. Eng., 2017, 5, 5921–5932 CrossRef CAS
.
- BIS, IS 10500: 2012, Indian Standard Drinking Water — Specification (Second Revision), New Delhi 110002, 2012 Search PubMed
.
- WBSEDCL, Tariff Rate for the Year 2024-25, 2025 Search PubMed
.
- M. Chang, C. Tang, C.-C. Wang and C. Zhao, Prog. Nat. Sci.:Mater. Int., 2024, 34, 66–73 Search PubMed
.
- A. A. Rico-Barragán, J. R. Álvarez, S. Pioquinto-García, J. Rodríguez-Hernández, P. Rivas-García and N. E. Dávila-Guzmán, Sustainable Prod. Consumption, 2023, 40, 159–168 Search PubMed
.
- N. Kanmaz, Environ. Res. Technol., 2025, 8, 581–592 Search PubMed
.
- L. Biswal, J. E. Goodwill, C. Janiak and S. Chatterjee, Sep. Purif. Rev., 2022, 51, 408–426 CAS
.
- PubChem, PubChem Compound – NCB, accessed 21 September 2025, https://pubchem.ncbi.nlm.nih.gov/ Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |