 Open Access Article
Open Access ArticleGreen synthesis of a manganese (oxyhydr) oxide nanocomposite, utilizing Eriobotrya japonica leaf extract for adsorbing methyl blue dye from water
Honar Salah Ahmed * and
Raghad Yousif Mohammed
* and
Raghad Yousif Mohammed
Department of Physics, College of Science, University of Duhok, Duhok 42001, Kurdistan Region, Iraq. E-mail: honar.ahmed@uod.ac; raghad.mohammed@uod.ac
First published on 24th April 2026
Abstract
Manganese (oxyhydr) oxides (γ-Mn3O4 and α-MnOOH) were successfully synthesized using Eriobotrya japonica leaf extract via a green-synthesis technique. The synthesized nanomaterials exhibited a characteristic absorption peak at 302 nm with an optical bandgap of 3.71 eV. XRD analysis confirmed the formation of γ-Mn3O4 and α-MnOOH with an average crystallite size of 30.83 nm, while SEM images revealed a mixed morphology of nanoparticles and nanorods with an average particle size of 37.72 nm. EDS and elemental mapping verified the predominant presence of Mn and O. BET analysis showed a specific surface area of 2.58 m2 g−1 with mesoporous characteristics. The green-synthesized manganese (oxyhydr) oxide nanocomposite demonstrated high adsorption efficiency toward methyl blue dye, achieving a maximum removal of 91.95% at an initial dye concentration of 133.33 mg L−1. Adsorption equilibrium followed the Langmuir isotherm (R2 = 0.9846, qmax = 1138.2 mg g−1), while kinetic data were best described by the pseudo-second-order model (R2 > 0.99), confirming a chemisorption mechanism. Thermodynamic analysis indicated that the adsorption process is exothermic, spontaneous, and favorable. In addition, the adsorbent exhibited moderate reusability performance over multiple cycles, highlighting its stability and potential for sustainable wastewater treatment applications.
Introduction
Global environmental concerns have intensified in recent years due to rapid population growth, urbanization, and industrial development, which have significantly altered consumption patterns and resource utilization. These changes have resulted in the large-scale generation of municipal and industrial wastes, posing serious environmental challenges. Among them, wastewater contamination by dyes and heavy metals represents a major threat to ecosystems and human health. Improper disposal and insufficient treatment of industrial and domestic effluents introduce a wide range of organic pollutants into water, soil, and the atmosphere. Therefore, the development of effective wastewater treatment strategies has become an urgent and widely researched priority. Due to their chemically stable structures, dyes persist in aquatic systems, reducing dissolved oxygen levels and disrupting aquatic life. They also can produce toxic, carcinogenic, and mutagenic byproducts, posing serious risks to both environmental and human health.1,2 Methyl blue dye (MB), with the molecular formula C37H28N3Na2O9S3+, is a triphenylmethane and an anionic dye usually used in coloring, textiles, paper, inks, printing, cosmetics, fibers, and plastics.3,4To address this challenge, several treatment methods such as coagulation, photocatalysis, membrane filtration, chemical and biological oxidation, and adsorption have been developed. Among these, adsorption is particularly attractive due to its high efficiency, low cost, simple operation, reduced energy requirements, and minimal secondary waste generation.5 Many different adsorbents were utilized to adsorb the pollutants and dyes from the wastewater, such as active carbon, zeolites, metal and metal oxide nanostructures, fly ash, clay, and functional polymeric materials.6,7 The porous shape and functional groups of the adsorbents mainly affect the adsorption performance of organic dyes.8
Metal oxide (e.g., ZnO, TiO2, CuO, Ag2O, Mn oxides, etc.) nanoparticles are the most studied material groups. They played a significant role in various fields of applications, among them as drug delivery systems, water purification, and anti-inflammatory and antibacterial activity.9,10
The basic unit of the crystal structure of manganese oxides is a MnO6, comprising a central manganese atom surrounded by six oxygen atoms. The MnO6 octahedra connect via edge-sharing to form single chains, leading to manganese oxides with diverse structures with edge-as well as corner-sharing.11 Manganese oxides are typically classified into two different categories according to the arrangement of MnO6: the layered structure, known as phyllomanganate, and the tunnelled structure, referred to as tectomanganate.12,13 The layered structure comprises layers of edge-sharing MnO6 octahedra, with symmetry dependent on the arrangement of Mn(III) octahedra. The tunnelled structure is formed by edge-sharing MnO6 octahedra forming the walls, floor, and ceiling of the tunnels.13 Heterovalent manganese ions, as well as foreign transition-metal cations, can be present inside both structures of manganese oxides, either replacing Mn(IV) or acting as charge compensators in phyllomanganate interlayers or tectomanganate tunnels.14,15
Typical instances of tunnelled structures of manganese oxides are todorokite, pyrolusite (β-MnO2), groutite (α-MnOOH), feitknechtite (β-MnOOH), manganite (γ-MnOOH), and cryptomelane. In contrast, the layered manganese oxides have structures that include birnessite, buserite, and vernadite. The distinct manganese oxides exhibit a variety of sizes and crystal structures, leading to variations in redox reactions, adsorption, electrochemical, and photochemical processes.
Marokite (γ-Mn3O4) with an orthorhombic crystal structure, Pbcm symmetry (space group #57; Z = 4), and groutite (α-MnOOH) orthorhombic crystal structure and space group (Pnma; Z = 4) are manganese (oxyhydr) oxides that have received significant attention in the adsorption, oxidation, electrode of the batteries, and super capacitors.16–21
A variety of methods have been employed to synthesise different Mn oxide nanostructures, such as chemical, physical processes, and biological methods. Chemical methods, such as the sol–gel method,22,23 hydrothermal/solvothermal method,24 chemical vapour deposition (CVD),25 physical methods such as laser ablation,26 sputtering,27 aerosols28 and biological methods such as bacteria,29 fungus,30 yeast,31 Actinomycetes,32 algae,33 plants (leaves, roots, fruit, peel).34–36
Phytonanotechnology is an emerging approach in agricultural biotechnology that enables the green synthesis of nanoparticles using plant-derived materials such as leaves, seeds, and peels. Plant extracts contain phytochemicals and metabolites that act as reducing and stabilizing agents during nanoparticle formation. Compounds such as flavonoids, organic acids, and quinones facilitate metal ion reduction, while polyphenols and tannins contribute to nanoparticle stabilization. Owing to its environmental friendliness, low cost, simplicity, scalability, and biocompatibility, this plant-based synthesis method has gained significant attention for producing metal and metal oxide nanomaterials.37,38
Loquat (Eriobotrya japonica) is a subtropical evergreen fruit tree that originated in southeastern China. It has been cultivated in China for more than 2000 years. The plant possesses significant medicinal value, and various parts of the tree have long been utilized in traditional medicine. In Chinese folk medicine, loquat extracts have been used to treat cough, chronic bronchitis (CB), inflammation, diabetes, and cancer, as documented in ancient texts such as the Compendium of Materia Medica.39
Eriobotrya japonica is rich in diverse phytochemicals, including phenols, alkaloids, cardiac glycosides, flavonoids, mucilage, gums, and phytosterols. The leaves and fruits contain numerous bioactive compounds such as vitamins, phenolic compounds, flavonoids, and carotenoids. In particular, loquat leaves contain phenolic acids, flavonoids, tannins, sesquiterpenes, triterpenes, and megastigmane glycosides. Bioactive triterpenoids such as oleanolic acid and ursolic acid are present in both flowers and leaves, although their concentrations vary depending on cultivar type and developmental stage.40
The chemical composition of loquat leaves is complex and diverse. Khouya et al. have been reported that loquat leaves contain substantial amounts of fiber, minerals (Ca, K, Na, and Fe), and vitamins, including B2, B6, and B12.41 According to Hwang et al., the proximate composition of loquat leaves includes 8.78% moisture, 6.74% crude protein, 7.87% crude fat, 6.99% crude ash, 43.61% dietary fiber, and 26.01% carbohydrates. Their study also identified 16 amino acids and several fatty acids, such as lauric, myristic, pentadecanoic, stearic, and oleic acids.42 Furthermore, Infante-Rodríguez, D. A. et al. reported that loquat leaves contain 0.039 mg vitamin A, 0.096 mg vitamin E, and 0.575 mg vitamin C, while the mineral composition follows the order Ca > K > Mg > Na > Fe > Mn > Zn.43
Phenolic compounds represent one of the most extensively investigated classes of phytochemicals in E. japonica leaves, with phenolic acids and flavonoids being the predominant groups. Additionally, more than 164 volatile compounds have been identified in the leaves (Taniguchi et al., and Zhu et al.).44,45 Various triterpenoids—including methyl betuliate, methyl maslinate, methyl corosolate, oleanolic acid, ursolic acid, maslinic acid, corosolic acid, tormentic acid, and euscaphic acid—have also been isolated from loquat leaves (Lv et al., 2008).46 Moreover, Chen et al., identified several terpenoid compounds such as methoxy-euscophic acid, tormentic acid, methylcorosolate, corosolic acid, maslinic acid, oleanolic acid, and ursolic acid, along with phenolic–terpenoid derivatives including methoxy-3-O-p-coumaroyltormenic acid, 3-O-p-coumaroyltormenic acid, methoxy-3-O-p-coumaroylmaslinic acid, and 3-O-p-coumaroylmaslinic acid.47
The abundance of these phytochemicals, particularly phenolic compounds and flavonoids, makes loquat leaf extract a promising natural reducing and stabilizing agent for the green synthesis of nanomaterials.
In this work, marokite (γ-Mn3O4) and groutite (α-MnOOH) manganese (oxyhydr) oxides in nanoparticle and nanorod forms were synthesized via a green-synthesizing method using Eriobotrya japonica leaf extract and applied for methyl blue (MB) dye removal from water. The adsorption behavior was evaluated using Langmuir, Freundlich, Temkin, and Dubinin–Radushkevich isotherm models, while pseudo-first-order, pseudo-second-order, and intraparticle diffusion models were used to examine the adsorption kinetics. The obtained performance was compared with previously reported adsorbents, and the reusability of the green-synthesized nanocomposite was also assessed.
Materials and method
Manganese nitrate tetrahydrate Mn(NO3)2·4H2O, sodium hydroxide (NaOH) with a purity of greater than 99%, and methyl blue dye MB (C37H28N3Na2O9S3+) with maximum absorption peak (λmax = 600 nm) were acquired from Merck (Germany—Darmstadt) and used for experiments without additional purification.Collecting Eriobotrya japonica leaves
Green-synthesis of manganese (oxyhydr) oxide nanocomposite
An aqueous solution of Mn(NO3)2·4H2O (0.5 M) was prepared by dissolving 6.275 g of the salt in 50 mL of distilled water under continuous stirring for 5 min until complete dissolution was achieved. Subsequently, 25 mL of Eriobotrya japonica leaf extract (EJLE) was added gradually to the manganese nitrate solution in a dropwise manner. The pH of the resulting mixture was then adjusted using an appropriate volume of 0.1 M NaOH, followed by continuous stirring at room temperature for 30 min. The mixture was subsequently heated at 80 °C for 90 min with constant stirring. A noticeable change in the solution color from brown to dark indicated the formation of manganese (oxyhydr) oxide nanocomposite. The obtained product was then calcined at 400 °C for 2 h. Finally, the black powder was collected and stored for further characterization. The stepwise synthesis procedure of the manganese (oxyhydr) oxide nanocomposite is schematically presented in Fig. 2. | ||
| Fig. 2 Proposed step-by-step reaction mechanism for the green synthesis of Mn3O4/MnOOH composite nanomaterials using Eriobotrya japonica leaf extract as reducing, capping, and stabilizing agent. | ||
The synthesis of the green-synthesis of manganese (oxyhydr) oxide nanocomposite using Eriobotrya japonica leaf extract as an environmentally friendly reducing and capping agent proceeds through three main stages, as illustrated in Fig. 3. In the first stage, manganese ions (Mn2+) derived from manganese nitrate tetrahydrate interact with bioactive molecules in the plant extract, particularly hydroxyl (–OH) and phenolic groups. These phytochemicals function as reducing agents by donating electrons, thereby reducing Mn2+ ions to elemental manganese (Mn0) through a redox process.
 | ||
| Fig. 3 The synthesis steps of manganese (oxyhydr) oxide nanocomposite using Eriobotrya japonica leaf extract. | ||
In the second stage, the generated Mn0 atoms undergo nucleation, resulting in the formation of small clusters. These clusters subsequently aggregate as a result of energy minimization, initiating nanoparticle growth. During the growth stage, smaller clusters merge into larger ones through the Ostwald ripening mechanism, which leads to the development of larger and more stable nanoparticles. The phytochemical constituents of the Eriobotrya japonica leaf extract, including polyphenols and flavonoids, play an important role in stabilizing the growing nanoparticles. In addition, carboxylic acid groups (–COOH) present in the extract may lose hydrogen atoms to form carboxylate ions (–COO−), which attach to the nanoparticle surfaces. These carboxylate ions provide electrostatic and steric stabilization, preventing particle aggregation and helping to regulate the size and morphology of the nanoparticles.
In the final stage, the stabilized nanoparticles, which mainly consist of Mn(OH)2 intermediates, undergo thermal decomposition during the calcination process. This thermal treatment converts the hydroxide intermediates into crystalline Mn3O4/MnOOH composite nanomaterials with a well-defined morphology.
Overall, this synthesis pathway highlights the important roles of polyphenols, flavonoids, and carboxylic acids present in Eriobotrya japonica leaf extract in the reduction, growth, and stabilization of the nanoparticles. These mechanisms are consistent with those proposed by Barzinjy et al., for ZnO NPs,48 Gebreslassie & Gebretnsae for SnO2 NPs,49 Miraeez S. A. and R. Y. Mohammed for CdO NPs.50
Phytochemical screening of Eriobotrya japonica leaf extract
Qualitative phytochemical screening of the Eriobotrya japonica leaf extract (EJLE) was carried out using standard chemical assays to identify the presence of major bioactive constituents.Adsorption activity test of green-synthesized manganese (oxyhydr) oxide nanocomposite
The adsorption performance of the green-synthesized manganese (oxyhydr) oxide nanocomposite for the removal of methyl blue (MB) dye from aqueous solutions was evaluated under dark conditions to exclude any photochemical effects. A series of MB solutions with initial concentrations of 133.33, 150, 166.67, 183.33, and 200 mg L−1 were prepared in 150 mL of distilled water, and their initial absorbance values were recorded using a JENWAY 6850 UV-vis spectrophotometer. Batch adsorption experiments were then carried out according to the experimental conditions summarized in Table 1. During the adsorption process, samples were collected at 3 min intervals. The withdrawn samples were centrifuged at 4000 rpm for 15 min to separate the nanocomposite, and the absorbance of the clear supernatant was subsequently measured using a UV-vis spectrophotometer to determine the residual MB concentration. The adsorption efficiency, adsorption capacity at time (t), and at equilibrium were calculated through the following equations:61,62
 | (1) |
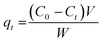 | (2) |
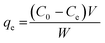 | (3) |
| Runs | pH | Initial concentration of MB dye (mg L−1) | Green-synthesized manganese (oxyhydr) oxides nanocomposite (mg/150 mL) | Temperature (K) |
|---|---|---|---|---|
| 1 | 2 | 133.33 | 20 | 283 |
| 2 | 4 | 133.33 | 20 | 283 |
| 3 | 6 | 133.33 | 20 | 283 |
| 4 | 8 | 133.33 | 20 | 283 |
| 5 | 10 | 133.33 | 20 | 283 |
| 6 | 10 | 133.33 | 7.5 | 283 |
| 7 | 10 | 133.33 | 11.25 | 283 |
| 8 | 10 | 133.33 | 15 | 283 |
| 9 | 10 | 150 | 20 | 283 |
| 10 | 10 | 166.67 | 20 | 283 |
| 11 | 10 | 183.33 | 20 | 283 |
| 12 | 10 | 200 | 20 | 283 |
| 13 | 10 | 133.33 | 20 | 298 |
| 14 | 10 | 133.33 | 20 | 308 |
Adsorption isotherms
Adsorption isotherms are widely recognized as effective tools for elucidating the interaction between methyl blue (MB) dye as the adsorbate and manganese (oxyhydr) oxide nanocomposite as the adsorbent, as well as for describing the distribution of unadsorbed dye at equilibrium.63 Different isotherm models provide insight into the distinct physical and chemical features governing the adsorption process. These models are valuable for optimizing adsorbent properties and for clarifying the underlying adsorption mechanism.64 In this study, the Langmuir, Freundlich, Temkin, and Dubinin–Radushkevich (D–R) isotherms were applied to interpret the experimental data, enabling a comprehensive evaluation of MB dye adsorption onto manganese (oxyhydr) oxide nanocomposite. The suitability of each isotherm model was determined by comparing their respective correlation coefficient (R2) values.The Langmuir isotherm assumes that adsorption occurs through the formation of a homogeneous monolayer on the surface of the adsorbent, where all active sites are equivalent and possess the same adsorption energy. In this model, each site accommodates only one adsorbate molecule, and no interactions take place between the adsorbed species.65 The linear Langmuir adsorption isotherm can be expressed as follows.66,67
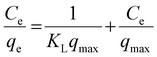 | (4) |
In this isotherm, qmax represents the maximum monolayer adsorption capacity, while Ce and qe denote the equilibrium concentration and the corresponding adsorption capacity, respectively.
The Langmuir constant KL (L mg−1), which reflects the affinity and energy of the adsorption process, can be determined along with qmax from the linear plot of Ce versus Ce/qe.
In addition, the dimensionless separation factor (RL), a key parameter for evaluating the favorability of adsorption, is calculated using the following equation.68
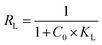 | (5) |
The dimensionless separation factor (RL) is commonly used to evaluate the nature and feasibility of an adsorption process. Depending on its value, the adsorption behavior can be classified as unfavorable (RL > 1), linear (RL = 1), irreversible (RL = 0), or favorable when RL lies between 0 and 1.
The Freundlich isotherm assumes a heterogeneous adsorbent surface characterized by active sites with different energy levels, leading to the formation of multilayer adsorption rather than a uniform monolayer. The Freundlich isotherm could be stated as.69
 | (6) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ce and log
Ce and log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe.
qe.
The Temkin isotherm describes adsorption on a heterogeneous surface by accounting for adsorbate–adsorbent interactions while neglecting the effects at extremely low and high concentrations. It assumes that the heat of adsorption decreases linearly with increasing surface coverage as temperature rises, rather than following a logarithmic dependence.70 The Temkin isotherm can be expressed as follows.68
qe = B![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A + B A + B![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ce Ce
| (7) |
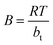 | (8) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ce.
Ce.
Dubinin–Radushkevich (D–R) is typically employed to comprehend the mechanisms of adsorption, whether chemical or physical.71 This semi-empirical adsorption isotherm is a temperature-dependent isotherm and is typically employed to represent the Gaussian energy distribution onto a heterogeneous adsorbent surface.66 The value gives the vital information about the adsorption process, such as when the energy value is less than 8 kJ mol−1, the process is physical adsorption, and when the energy value is between 8 and 16 kJ mol−1, the process is the ion exchange, and is a chemical adsorption when the energy value is above 16 kJ mol−1.71 The equations of the D–R isotherm and energy can be written as follows.68
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe = ln qe = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qm − KDRε2 qm − KDRε2
| (9) |
 | (10) |
 | (11) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe versus ε2. T and R are the temperature (K) and the universal gas constant (8.314 J mol−1 K−1), respectively.
qe versus ε2. T and R are the temperature (K) and the universal gas constant (8.314 J mol−1 K−1), respectively.
Adsorption kinetics
The adsorption kinetics were analyzed using the pseudo-first-order, pseudo-second-order, and intraparticle diffusion models, as these kinetic models provide valuable information about the adsorption equilibrium behavior.72 In particular, the pseudo-first-order and pseudo-second-order models are widely used to elucidate the nature of interactions, whether physical or chemical, between adsorbate molecules and the adsorbent surface.73 R2 values have been used to evaluate the correlation between kinetic models and experimental results. The linear equations for the three models can be represented as follows.67,74The pseudo-first-order kinetic model assumes that the rate of adsorption is directly proportional to the number of available active sites on the adsorbent surface and inversely related to the concentration of the adsorbate.75 The pseudo-first-order kinetic can be written as shown below:
ln(qe − qt) = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe − k1t qe − k1t
| (12) |
The pseudo-second-order (eqn (13)) is utilized to investigate the chemisorption of the adsorbate and adsorbent.
 | (13) |
The intraparticle diffusion kinetic model was employed to understand the diffusion mechanism of the adsorption process.70 The intraparticle diffusion model equation can be written as follows.76
| qt = kidt0.5 + C | (14) |
Thermodynamic parameters
To evaluate the thermodynamic parameters and assess the effect of temperature on the adsorption of methyl blue onto manganese (oxyhydr) oxide nanocomposite, experiments were performed at different temperatures (283, 298, and 308 K), while maintaining constant conditions of dye concentration (133.33 mg L−1), adsorbent dose (0.02 g), and pH (10). The thermodynamic quantities ΔH, ΔS, and ΔG were determined using the corresponding equations derived from the plot of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kads versus 1/T.
Kads versus 1/T.
 | (15) |
 | (16) |
The equation can express Gibbs' free energy (ΔG)79
ΔG = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kads Kads
| (17) |
Arrhenius equation used to calculate the activation energy Ea (kJ mol−1). The activation energy is the minimum energy required for a reaction to proceed.80
 | (18) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k2 versus 1/T.
k2 versus 1/T.
Characterizations
The optical properties of the green-synthesized manganese (oxyhydr) oxide nanocomposite were measured using a JENWAY 6850 UV-vis spectrophotometer. The direct optical energy bandgap of the green-synthesized manganese (oxyhydr) oxides nanocomposite was calculated using Tauc's equation.81| (αhν)2 = B(hν − Eg) | (19) |
Debye–Scherrer equation.82 It was used to calculate the crystallite size of green-synthesized manganese (oxyhydr) oxide nanocomposite.
 | (20) |
The structural properties of the green-synthesized manganese (oxyhydr) oxide nanocomposite were studied using a Philips PW1730 X-ray diffraction system in the 2θ range (10° to 80°) with CuKα = 1.54 Å.
The morphological properties of the green-synthesized manganese (oxyhydr) oxide nanocomposite were studied using the ZEISS Field Emission Scanning Electron Microscopy (FE-SEM) system, and its compositional properties were analyzed using EDX spectroscopy (FEI company, US). The N2 adsorption–desorption test of the green-synthesized manganese (oxyhydr) oxide nanocomposite was conducted using a BELSORP II mini (Japan).
Results and discussion
Phytochemical screening of Eriobotrya japonica leaf extract
The results of the phytochemical tests of EJLE are shown in Table 2 and Fig. 4. The symbols + and − denote the presence and absence of bioactive compounds in the EJLE, respectively. The indication column summarizes the outcomes of the qualitative screening tests performed for these bioactive compounds.| Bioactive compound | Chemical test | Results | Indication | Ref. |
|---|---|---|---|---|
| Flavonoids | Pb(CH3COO)2 | ++ | Yellow precipitation | 51 and 52 |
| Carbohydrates | Molisch test | + | Violet ring | 53 |
| Tannins | Braymer's test | + | Brownish-green | 54–56 |
| Alkaloids | Hager's test | − | No creamy white precipitate | 57 |
| Saponins | Foam test | + | Foam | 58 |
| Proteins and amino acid | Ninhydrin | − | No change in color | 60 |
| Phenolics | Pb(CH3COO)2 | + | White precipitation | 59 |
FT-IR results
The FTIR spectra of the green synthesized manganese (oxyhydr) oxide nanocomposite and Eriobotrya japonica leaves extract were recorded in the range 400–4000 cm−1, as shown in Fig. 5(a) and (b). From Fig. 5a, the peaks at 3398 and 3421 cm−1 corresponded to the O–H stretching and moisture in the samples.83 The peak at 2424 cm−1 may be attributed to the (–CH2–) symmetric stretching.84 The sharp peak at 1786 cm−1 corresponds to the (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) stretching.50 The peaks (1355 and 835 cm−1) can be attributed to the NO3−.85 The peaks at 466, 503, and 576 cm−1 are related to the Mn–O stretching vibration of manganese (oxyhydr) oxide nanocomposite.
O) stretching.50 The peaks (1355 and 835 cm−1) can be attributed to the NO3−.85 The peaks at 466, 503, and 576 cm−1 are related to the Mn–O stretching vibration of manganese (oxyhydr) oxide nanocomposite.
The FTIR of the Eriobotrya japonica leaf extract can be seen in Fig. 5b. The strong band at 3338 cm−1 may be attributed to the O–H and N–H stretching vibration. The peak at 2929 cm−1 corresponded to the stretching vibration of the C–H.86 The band 1610 cm−1 is attributed to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibration. The peak at 1392 cm−1 could be related to the bending of the C–H in –CH3. The band at 1070 cm−1 corresponds to the C–O or C–H stretching vibration. The band at 609 cm−1 could have corresponded to the C–H of aromatic or aliphatic carbon.87
C stretching vibration. The peak at 1392 cm−1 could be related to the bending of the C–H in –CH3. The band at 1070 cm−1 corresponds to the C–O or C–H stretching vibration. The band at 609 cm−1 could have corresponded to the C–H of aromatic or aliphatic carbon.87
Optical properties
The UV-vis absorption spectra of the Eriobotrya japonica leaf extract and the green-synthesized manganese (oxyhydr) oxide nanocomposite were recorded in the wavelength range of 250–600 nm, as illustrated in Fig. 5c and d. The plant extract exhibited two characteristic absorption bands at 278 nm and 316 nm, attributed to surface plasmon resonance, confirming the presence of phytochemical constituents, as shown in Fig. 5d. The absorption region between 250 and 320 nm is associated with n → π* transitions of functional groups such as O–H and COOH present in the extract, highlighting the role of phytochemicals as effective reducing and stabilizing agents.88,89 However, Fig. 5c presents the absorption peaks of the green synthesized manganese (oxyhydr) oxide nanocomposite. The green-synthesized manganese (oxyhydr) oxide nanocomposite exhibits an absorption peak at 302 nm. The single and sharp absorption peak of the green-synthesized manganese (oxyhydr) oxide nanocomposite indicates the uniform distribution and nanosized particles.90A plot of (αhν)2 versus hν is shown in Fig. 5e and was used to determine the energy gap of the green-synthesized manganese (oxyhydr) oxides. The linear segment of the curves is extended to the hν axis to ascertain the energy bandgap, which is 3.70 eV.
Structural properties
The XRD results of the green-synthesized manganese (oxyhydr) oxide nanocomposite are shown in Fig. 5g, and Table 3 revealed that the diffracted patterns match with the marokite (γ-Mn3O4) peaks indexed in the (JCPDS card no. 96-900-1303) with the orthorhombic crystal structure space group (Pbcm) and groutite (α-MnOOH) (JCPDS card no. 96-901-1547) with the orthorhombic crystal structure and space group (Pnma).| No. | Diffracted peaks 2θ | Planes | Phase |
|---|---|---|---|
| 1 | 16.3201 | (200) | Groutite |
| 2 | 29.6749 | (100) | Mn3O4 |
| 3 | 32.0706 | (301) | Groutite |
| 4 | 35.5736 | (210) | Groutite |
| 5 | 39.1918 | (401) | Groutite |
| 6 | 47.9899 | (140) | Mn3O4 |
| 7 | 66.8312 | (303) | Groutite |
The high intensity is an indication of good crystallinity of the (γ-Mn3O4) and (α-MnOOH) nanostructures in the nanocomposite. The crystallite size was calculated to be 30.83 nm.
N2 adsorption–desorption isotherm of green-synthesized manganese (oxyhydr) oxide nanocomposite
The porosity of green-synthesized manganese (oxyhydr) oxide nanocomposite (Fig. 5f) was acquired by measuring N2 adsorption–desorption isotherms at 77 K. The adsorbed quantity increased gradually at low pressures and sharply at high pressures, exhibiting a type-IV isotherm and a type-H3 hysteresis loop (defined by IUPAC) at a relative pressure of nearly P/Po = 0.5–1, indicating the presence of mesopores in the green-synthesized manganese (oxyhydr) oxide nanocomposite. Brunauer–Emmett–Teller (BET) isotherm was employed to analyze the porosity parameters. The results showed that the BET specific surface area of the green-synthesized manganese (oxyhydr) oxide nanocomposite is 2.5808 m2 g−1, total pore volume (P/Po = 0.990) is 0.017075 cm3 g−1, and mean pore diameter is 26.464 nm.Morphological properties
The FESEM micrographs shown in Fig. 6a reveal that the green-synthesized manganese (oxyhydr) oxide nanocomposite, composed of γ-Mn3O4 and α-MnOOH phases, exhibits two distinct nanostructures: nanoparticles and nanorods. Crystal faces with higher surface energy tend to grow more rapidly, leading to the formation of smaller facets that may eventually disappear, whereas faces with lower surface energy grow at a slower rate and are more likely to be preserved in the final morphology.91 The particle sizes calculated from the FE-SEM images were found to be 37.72 nm for the green-synthesized manganese (oxyhydr) oxide nanocomposite. The particle sized calculated from the FE-SEM image are bigger than those calculated from the XRD data this can be attributed due to their higher surface area and high surface-to-volume ratio, small particles are more likely to interact with their neighbouring surface atoms to create agglomerations of a particle because they have more surface atoms and, thus, more dangling bonds and it is essential to remember that the crystallite size (D) may not always correspond to the particle size; rather, it represents the size of a coherently diffracting domain.92 | ||
| Fig. 6 (a) FESEM images, (b) particle size distribution, (c) EDS, and (d) mapping of the green-synthesized manganese (oxyhydr) oxide nanocomposite. | ||
Compositional properties
The energy dispersive X-ray technique was utilized to study the elemental composition of the green-synthesized manganese (oxyhydr) oxide nanocomposite, as shown in Fig. 6c, which confirms the predominant presence of the peaks of the Mn and O, with the presence of unidentified peaks. The EDS results support the XRD results, indicating good crystallinity of the products. From Table 4 it can be seen that the weight and atomic ratio percentage of oxygen are dominant. The atomic ratio of green synthesized manganese (oxyhydr) oxide nanocomposite is smaller than 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4. This can be evidence that the green synthesized manganese (oxyhydr) oxide nanocomposite consists of a composite of manganese (oxyhydr) oxides (γ-Mn3O4) and (α-MnOOH).93
4. This can be evidence that the green synthesized manganese (oxyhydr) oxide nanocomposite consists of a composite of manganese (oxyhydr) oxides (γ-Mn3O4) and (α-MnOOH).93
| Weight% | Atomic% | ||
|---|---|---|---|
| Mn | O | Mn | O |
| 34.62 | 65.38 | 13.36 | 86.64 |
Adsorption activity of the green-synthesized manganese (oxyhydr) oxide nanocomposite
Fig. 7a depicts the influence of solution pH on the adsorption of methyl blue dye. The results demonstrate a clear enhancement in both adsorption capacity and removal efficiency with increasing pH. Under strongly acidic conditions, the high concentration of H+ ions leads to competitive interactions with methyl blue molecules and partial protonation of surface functional groups, which weakens the affinity between the dye and the manganese (oxyhydr) oxide nanocomposite. As the pH rises, this competitive effect diminishes, and the availability of surface functional groups such as –NH2, –OH, and –COOH increases alongside a reduction in H+ concentration and a corresponding increase in OH− ions. These conditions promote stronger interactions between the adsorbent surface and dye molecules, resulting in improved adsorption performance.95
Overall, increasing the adsorbent dosage enhances the available surface area and number of active sites, resulting in improved adsorption efficiency. However, the adsorption capacity per unit mass decreases as a consequence of the presence of unoccupied active sites at higher dosages.
Adsorption isotherms result of the green-synthesized manganese (oxyhydr) oxide nanocomposite
The equilibrium adsorption behavior of methyl blue (MB) onto the green-synthesized manganese (oxyhydr) oxide nanocomposite was evaluated using Langmuir, Freundlich, Temkin, and Dubinin–Radushkevich (D–R) isotherm models, as shown in Fig. 8 and Table 5. Among these, the Langmuir model exhibited the best fit to the experimental data, as evidenced by the high correlation coefficient (R2 = 0.9846), indicating monolayer adsorption on a homogeneous surface with finite and energetically equivalent active sites. The remarkably high maximum adsorption capacity (qmax = 1138.2 mg g−1) highlights the strong affinity of MB molecules toward the manganese (oxyhydr) oxide surface. Furthermore, the dimensionless separation factor (RL = 0.0199–0.029) lies between 0 and 1, confirming that the adsorption process is highly favorable under the studied conditions.76,100 | ||
| Fig. 8 Adsorption isotherms: (a) Langmuir isotherm, (b) Freundlich isotherm, (c) Temkin isotherm, and (d) Dubinin–Radushkevich (D–R) isotherm. | ||
| Isotherms | Parameters | Values |
|---|---|---|
| Langmuir isotherm | qmax (mg g−1) | 1138.2 |
| KL (L mg−1) | 0.2488 | |
| RL | 0.019–0.029 | |
| R2 | 0.9846 | |
| Freundlich isotherm | 1/n | 0.098 |
| n (mg g−1) | 10.16 | |
| Kf (mg g−1) | 711.49 | |
| R2 | 0.5 | |
| Temkin isotherm | AT (L g−1) | 919.18 |
| bt (J mol−1) | 24.13 | |
| B (L g−1) | 97.46 | |
| R2 | 0.5 | |
| Dubinin–Radushkevich (D–R) isotherm | qDR (mg g−1) | 0.002 |
| KDR (mol2 kJ−2) | 8.42 × 10−10 | |
| E (kJ mol−1) | 24.362 | |
| R2 | 0.484 |
In contrast, the Freundlich model showed a poor correlation (R2 = 0.5), suggesting that multilayer adsorption on a heterogeneous surface is not the dominant mechanism, despite the high Freundlich constant (Kf = 711.49 mg g−1) and the value of 1/n (0.098), which still implies favorable adsorption intensity.101,102 Similarly, the Temkin model yielded a low R2 value (0.5), indicating limited applicability; however, the Temkin constant related to adsorption heat (B = 97.46 J mol−1) suggests notable adsorbate–adsorbent interactions. The D–R isotherm also poorly described the system (R2 = 0.484). Overall, the isotherm analysis confirms that MB adsorption onto green-synthesized manganese (oxyhydr) oxide nanocomposite is a favorable, monolayer, and predominantly chemisorption process with exceptionally high adsorption capacity, underscoring its potential as an efficient adsorbent for dye-contaminated wastewater.
Table 6 represents the maximum adsorption capacity of the methyl blue dye onto the different adsorbents. This study's maximum adsorption capacity shows competitive performance with literature works, which is 1138.2 mg. This can be attributed to the presence of active sites on the green-synthesized nanoparticles.
| Adsorbents | qe mg g−1 | Ref. |
|---|---|---|
| Mespilus germanica L. fruits seed biochar (MGLfsB) | 11.148 | 103 |
| Nanomagnetic Mespilus germanica L. fruit seed biochar (nM-MGLfsB) | 13.089 | 103 |
| Amino-functionalized zeolitic imidazole framework-8 (ZIF-8/NH2 nanoparticles) | 537.4 | 104 |
| Activated carbon eggshell (ACE) | 45.23 | 105 |
| Na2Al2B2O7 | 2000 | 106 |
| Copper-based triazolate MOFs (CuTz-1-Py) | 352.3 | 107 |
| Banana leaves powder (BLP) | 84.24 | 108 |
| Activated carbon banana leaves powder (AC-BLP) | 386 | 108 |
| Crosslinked chitosan-citrate/SnO2 nanoparticles (CTN-CT/SnO2) | 511.92 | 109 |
| CMBLP | 118.24 | 110 |
| Pinus brutia biochar (PBB) | 66.667 | 111 |
| Nano-magnetic Pinus brutia biochar (nM-PBB) | 107.527 | 111 |
| Mn (oxyhyrd) oxide nanocomposite of (Mn3O4, MnOOH) | 1138.2 | This study |
Adsorption kinetics results of the green-synthesized manganese (oxyhydr) oxide nanocomposite
The pseudo-first-order and pseudo-second-order kinetics models were employed to evaluate the adsorption rate of different MB dye concentrations (133.33, 150, 166.67, 183.33, and 200 mg L−1). As illustrated and tabulated in Fig. 9, and Table 7 the kinetic parameters for methyl blue adsorption onto the green-synthesized manganese (oxyhydr) oxide nanocomposite reveal clear differences in the suitability of the applied models. The pseudo-first-order model shows relatively low correlation coefficients (R2 = 0.5957–0.9993) and calculated adsorption capacities (qe,cal) that deviate substantially from the experimental values (qe,exp), indicating that this model does not adequately describe the adsorption behavior over the studied concentration range. In contrast, the pseudo-second-order model shows excellent agreement with the experimental data, with very high correlation coefficients (R2 = 0.9841–0.99999) and qe,cal values that closely match qe,exp across all initial dye concentrations. This strong conformity suggests that the adsorption process is predominantly governed by chemisorption involving valence forces or electron sharing between the dye molecules and active sites on the nanocomposite surface.112,113 | ||
| Fig. 9 Adsorption kinetics models plot (a) pseudo-first order, (b) pseudo-second order, and (c) the intraparticle diffusion model. | ||
| Methyl blue dye concentration (mg L−1) | Pseudo-first order | Pseudo-second order | Intraparticle diffusion | qe (exp.) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| K1 (min−1) | qe (mg g−1) | R2 | K2 (g mg−1 min−1) | qe (mg g−1) | R2 | KIPD (mg g−1 min−0.5) | Cint | R2 | ||
| 133.33 | 0.5 | 44.17 | 0.9122 | 0.02 | 925.22 | 0.99999 | 9.155 | 893.56 | 0.7334 | 919.54 |
| 150 | 0.2444 | 315.26 | 0.9993 | 0.0014 | 980.39 | 0.99993 | 93.6 | 636.52 | 0.96 | 911.86 |
| 166.67 | 0.4999 | 765.48 | 0.9923 | 0.001 | 1106.71 | 0.9997 | 123.59 | 648.25 | 0.893 | 1006.88 |
| 183.33 | 0.09156 | 133.34 | 0.7663 | 0.0041 | 1122.76 | 0.9989 | 30.94 | 1003.3 | 0.7255 | 1101.32 |
| 200 | 0.1834 | 368.71 | 0.5957 | 0.0011 | 1249.33 | 0.9841 | 91.52 | 882.25 | 0.5293 | 1177.65 |
Fig. 9c and Table 7 represent the intraparticle diffusion kinetic model. Furthermore, the intraparticle diffusion model yields lower R2 values (0.5293–0.96) and nonzero intercepts (Cint), implying that intraparticle diffusion is not the sole rate-controlling step. Instead, the adsorption kinetics are influenced by multiple mechanisms, including rapid external surface adsorption followed by slower diffusion of dye molecules into the internal pores of the adsorbent.2,114
Overall, the kinetic analysis confirms that the adsorption of methyl blue onto the green-synthesized manganese (oxyhydr) oxide nanocomposite is best described by the pseudo-second-order model, with a multistep adsorption mechanism.
Thermodynamic parameters
Based on the thermodynamic parameters calculated from Fig. 10a and tabulated in Table 8. The adsorption behavior of methyl blue (MB) dye onto the green-synthesized manganese (oxyhydr) oxide nanocomposite can be interpreted as follows:| T (K) | ΔG (kJ mol−1) | ΔH (kJ mol−1) | ΔS (J mol−1 K−1) | R2 |
|---|---|---|---|---|
| 283 | −10.47 | −46.39 | −126.54 | 0.9687 |
| 298 | −8.96 | |||
| 308 | −7.22 |
The negative values of Gibbs free energy change (ΔG°) at all investigated temperatures (−10.47 to −7.22 kJ mol−1) confirm that the adsorption of MB is a spontaneous process. The gradual increase in ΔG° (becoming less negative) with increasing temperature suggests that the feasibility of adsorption slightly decreases at higher temperatures, indicating that lower temperatures are more favorable for MB uptake. The negative enthalpy change (ΔH° = −46.39 kJ mol−1) demonstrates that the adsorption process is exothermic, chemisorption, and implies strong interactions between MB molecules and the surface of the manganese (oxyhydr) oxide nanocomposite.115 Moreover, the negative entropy change (ΔS° = −126.54 J mol−1 K−1) reflects a decrease in randomness at the solid–solution interface during adsorption, which can be attributed to the orderly attachment of MB molecules onto the adsorbent surface.116–118
The high correlation coefficient (R2 = 0.9687) indicates good linearity of the thermodynamic model, confirming the reliability of the calculated parameters. Collectively, these results suggest that MB adsorption onto the manganese (oxyhydr) oxide nanocomposite is spontaneous, exothermic, and accompanied by increased structural ordering at the adsorbent–adsorbate interface.
As shown in Fig. 10b, the calculated activation energy (Ea = −45.76 kJ mol−1) indicates that the adsorption of methyl blue dye onto the green-synthesized manganese (oxyhydr) oxide nanocomposite is mainly governed by a chemisorption mechanism. In general, activation energy values within the range of 5–40 kJ mol−1 are typically attributed to physisorption, whereas higher values (approximately 40–800 kJ mol−1) are associated with chemisorption processes.119 It should be noted that, in certain adsorption systems, the adsorption rate may decrease as temperature increases. Under such conditions, the rate constant can still be described by the Arrhenius equation, which may yield a negative activation energy value.120 The negative Ea obtained in this study indicates that the adsorption process is exothermic in nature. This observation is consistent with the kinetic results, which demonstrate that the adsorption behavior follows the pseudo-second-order kinetic model, suggesting that chemical interactions play a significant role in the adsorption mechanism.
Adsorption mechanism
The FTIR spectra of the green-synthesized manganese (oxyhydr) oxide nanocomposite before and after adsorption of methyl blue (MB) Fig. 5a show noticeable changes in the positions and intensities of several characteristic peaks, indicating the participation of surface functional groups in the adsorption process.108 In particular, the –OH, –NH, C–O, C–N, and Pb–O groups were actively involved in the interaction with MB molecules.121 The broad O–H stretching vibration observed at 3398 cm−1 in the green-synthesized manganese (oxyhydr) oxide nanocomposite shifts to a lower wavenumber (3344 cm−1) after MB adsorption. This red shift suggests the formation of hydrogen bonding interactions between MB molecules and hydroxyl groups on the surface of the green-synthesized manganese (oxyhydr) oxide nanocomposite.Furthermore, a new absorption band appears at 1622 cm−1 in the MB-loaded nanocomposite, which is attributed to the deformation vibration of –NH2 groups derived from the dye molecules.110 Another new band at 1034 cm−1 corresponds to the sulfonate (–SO3−) functional group of MB, providing clear evidence for the successful adsorption of the dye onto the nanocomposite surface.122 In addition, slight shifts and reductions in the intensity of the Mn–O stretching vibration bands were observed after adsorption, indicating interactions between the dye molecules and Mn–O surface sites within the nanocomposite lattice. Collectively, these spectral changes confirm the effective adsorption of MB onto the green-synthesized manganese (oxyhydr) oxide nanocomposite.
The adsorption mechanism appears to be governed by multiple interactions, including hydrogen bonding, electrostatic attraction, n–π interactions, and pore filling, reflecting the complex nature of the adsorption behavior. Hydrogen bonding plays a significant role through interactions between hydrogen atoms on surface hydroxyl groups of green-synthesized manganese (oxyhydr) oxide nanocomposite and oxygen- and nitrogen-containing functional groups of MB dye molecules. The π bond of the benzene ring on MB can generate a specific interaction with metals. MB with rich benzene rings can generate π-interactions with manganese.123,124 The schematic of the MB adsorption on the green-synthesized manganese (oxyhydr) oxide nanocomposite can be seen in Fig. 11.
 | ||
| Fig. 11 Schematic representation of the adsorption of methyl blue dye onto the green-synthesized manganese (oxyhydr) oxide nanocomposite. | ||
In addition, the fitting of the experimental data to the Langmuir isotherm model, the pseudo-second-order (PSO) kinetic model, the magnitude of the enthalpy change, and the calculated activation energy values collectively indicate that the adsorption of MB onto green-synthesized manganese (oxyhydr) oxide nanocomposite is predominantly controlled by chemisorption. These results suggest that chemical interactions constitute the primary adsorption mechanism, although a minor contribution from physisorption cannot be excluded.
Reusability of the green-synthesized manganese (oxyhydr) oxide nanocomposite
The reusability performance of the green-synthesized manganese (oxyhydr) oxide nanocomposite was evaluated through consecutive adsorption–desorption cycles for methyl blue removal under optimized conditions (pH 10, initial dye concentration 133.33 mg L−1, adsorbent dose 20 mg/150 mL, temperature 283 K, and contact time 9 min). After each adsorption run, the used adsorbent was regenerated by thoroughly rinsing it with ethanol, followed by distilled water, to remove the dye molecules bound to its surface. As illustrated in Fig. 10c, the adsorption efficiency notably declined with increasing number of reuse cycles. The first cycle exhibited the highest removal efficiency. A moderate decrease was observed in subsequent cycles, likely due to partial blockage of adsorption sites, incomplete desorption of dye molecules, and possible structural or surface chemical changes in the nanocomposite during regeneration. Nevertheless, the adsorbent retained a considerable fraction of its initial adsorption capacity even after multiple cycles, indicating acceptable regeneration potential. These results demonstrate that the green-synthesized manganese (oxyhydr) oxide nanocomposite is a reusable and economically viable adsorbent for methyl blue removal from aqueous solutions, supporting its applicability in practical wastewater treatment processes.Conclusion
The treatment of contaminated water, particularly the removal of dyes and heavy metals, remains a critical environmental challenge, with adsorption emerging as an effective and economical solution. In this study, manganese (oxyhydr) oxide nanocomposite, namely marokite (γ-Mn3O4) and groutite (α-MnOOH), were successfully synthesized via an environmentally friendly green route using Eriobotrya japonica leaf extract. Optical, structural, and morphological characterizations confirmed the formation of pure manganese (oxyhydr) oxides composed of nanoparticles and nanorods.The green-synthesized manganese (oxyhydr) oxide nanocomposite exhibited good adsorption performance toward methyl blue dye. Adsorption equilibrium and kinetic analyses revealed that the process follows the Langmuir isotherm and pseudo-second-order kinetic model, indicating a monolayer chemisorption mechanism. Thermodynamic results demonstrated that the adsorption process is spontaneous and favorable. Moreover, the nanocomposite showed good reusability, retaining a substantial fraction of its adsorption capacity after multiple cycles, which reflects its stability and regeneration potential. The enhanced adsorption efficiency is attributed to phytochemicals in the plant extract, which play a key role in tailoring the nanomaterials' structural and surface properties. Overall, the green-synthesis technique provides an efficient and sustainable approach for producing manganese (oxyhydr) oxides with good potential for practical wastewater treatment applications.
Author contributions
Conceptualization, Raghad Yousif Mohammed; methodology, Raghad Yousif Mohammed; software, Honar Salah Ahmed; validation, Raghad Yousif Mohammed and Honar Salah Ahmed; formal analysis, Raghad Yousif Mohammed; investigation, Raghad Yousif Mohammed; resources, Raghad Yousif Mohammed; data curation, Honar Salah Ahmed; writing – original draft preparation, Honar Salah Ahmed; writing – review and editing, Raghad Yousif Mohammed.; visualization, Raghad Yousif Mohammed, and Honar Salah Ahmed; supervision, Raghad Yousif Mohammed; project administration, Raghad Yousif Mohammed all authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors have no competing interests to declare that are relevant to the content of this article.Data availability
The data were analyzed and used in this study are available upon a reasonable request from the corresponding author.Acknowledgements
The authors would like to express their gratitude to the Ministry of Higher Education and Scientific Research in the Kurdistan Region and the University of Duhok for their support.References
- F. T. Alshorifi, S. L. Ali and R. S. Salama, J. Inorg. Organomet. Polym. Mater., 2022, 32, 3765–3776 CrossRef CAS.
- N. Hasani, T. Selimi, A. Mele, V. Thaçi, J. Halili, A. Berisha and M. Sadiku, Molecules, 2022, 27, 1856 CrossRef CAS PubMed.
- B. Mu, L. Liu, W. Li and Y. Yang, J. Environ. Manage., 2019, 239, 271–278 CrossRef CAS PubMed.
- K. Azam, R. Raza, N. Shezad, M. Shabir, W. Yang, N. Ahmad, I. Shafiq, P. Akhter, A. Razzaq and M. Hussain, J. Environ. Chem. Eng., 2020, 8, 104220 CrossRef CAS.
- F. B. Loulic, R. H. S. M. Shirazi, M. Miralinaghi, H. A. Panahi and E. Moniri, Microporous Mesoporous Mater., 2023, 356, 112567 CrossRef.
- M. M. Youssif, H. G. El-Attar, V. Hessel and M. Wojnicki, Materials, 2024, 17, 5141 CrossRef CAS PubMed.
- N. Ghosh, S. Das, G. Biswas and P. K. Haldar, Water Sci. Technol., 2022, 85, 3370–3395 CrossRef CAS PubMed.
- I. Hussain, Y. Li, J. Qi, J. Li and L. Wang, J. Environ. Manage., 2018, 215, 123–131 CrossRef CAS PubMed.
- A. A. Yaqoob, H. Ahmad, T. Parveen, A. Ahmad, M. Oves, I. M. Ismail, H. A. Qari, K. Umar and M. N. Mohamad Ibrahim, Front. Chem., 2020, 8, 341 CrossRef CAS PubMed.
- S. J. Nadaf, N. R. Jadhav, H. S. Naikwadi, P. L. Savekar, I. D. Sapkal, M. M. Kambli and I. A. Desai, OpenNano, 2022, 100076 CrossRef CAS.
- S. M. Webb, B. Tebo and J. Bargar, Am. Mineral., 2005, 90, 1342–1357 CrossRef CAS.
- Z. Wu, B. Lanson, X. Feng, H. Yin, Z. Qin, X. Wang, W. Tan, Z. Chen, W. Wen and F. Liu, Geochim. Cosmochim. Acta, 2020, 271, 96–115 CrossRef CAS.
- T. E. Flores-Guia, L. F. Cano Salazar, A. Martínez-Luévanos and J. Claudio-Rizo, Handbook of Nanomaterials and Nanocomposites for Energy and Environmental Applications, 2021, pp. 2409–2428 Search PubMed.
- H. Yin, B. Lanson, S. Zhang, L. Liu, C. L. Peacock, J. E. Post, M. Zhu, W. Li, Q. Wang and J. Zhang, Geochim. Cosmochim. Acta, 2022, 333, 200–215 CrossRef CAS.
- A. G. Newton and K. D. Kwon, Geochim. Cosmochim. Acta, 2020, 291, 92–109 CrossRef CAS.
- F. Li, H. Yin, T. Zhu and W. Zhuang, Eco-Environ. & Health, 2024, 3, 89–106 Search PubMed.
- P. R. Garces Goncalves Jr, H. A. De Abreu and H. l. A. Duarte, J. Phys. Chem. C, 2018, 122, 20841–20849 CrossRef.
- M. C. Kemei, J. K. Harada, R. Seshadri and M. R. Suchomel, Phys. Rev. B:Condens. Matter Mater. Phys., 2014, 90, 064418 CrossRef CAS.
- S. V. Ovsyannikov, A. A. Aslandukova, A. Aslandukov, S. Chariton, A. A. Tsirlin, I. V. Korobeynikov, N. V. Morozova, T. Fedotenko, S. Khandarkhaeva and L. Dubrovinsky, Inorg. Chem., 2021, 60, 13440–13452 CrossRef CAS PubMed.
- T. Kohler, T. Armbruster and E. Libowitzky, J. Solid State Chem., 1997, 133, 486–500 CrossRef CAS.
- Y. O. Yesilbag, F. N. Tuzluca and M. Ertugrul, J. Mater. Sci.: Mater. Electron., 2019, 30, 8201–8209 CrossRef CAS.
- L. G. Teoh and K.-D. Li, Mater. Trans., 2012, 53, 2135–2140 CrossRef CAS.
- R. Dubey, Y. Rajesh and M. More, Mater. Today: Proc., 2015, 2, 3575–3579 CAS.
- J. Kurian and M. J. Mathew, J. Magn. Magn. Mater., 2018, 451, 121–130 CrossRef CAS.
- H. Lee, M. Y. Song, J. Jurng and Y.-K. Park, Powder Technol., 2011, 214, 64–68 CrossRef CAS.
- H. Naser, M. Alghoul, M. K. Hossain, N. Asim, M. Abdullah, M. S. Ali, F. G. Alzubi and N. Amin, J. Nanopart. Res., 2019, 21, 1–28 CrossRef.
- M. Pišlová, M. Kalbacova, L. Vrabcová, P. Slepička, Z. Kolská and V. Švorčík, Dig. J. Nanomater. Bios., 2018, 13, 1035–1044 Search PubMed.
- M. Gautam, J. O. Kim and C. S. Yong, J. Pharm. Invest., 2021, 51, 361–375 CrossRef CAS PubMed.
- X. Fang, J. Li, B. Ren, Y. Huang, D. Wang, Z. Liao, Q. Li, L. Wang and D. D. Dionysiou, J. Membr. Sci., 2019, 579, 190–198 CrossRef CAS.
- D. B. Raudabaugh, et al., Nanomater. Nanotechnol., 2013, 3, 2 CrossRef PubMed.
- M. Shu, F. He, Z. Li, X. Zhu, Y. Ma, Z. Zhou, Z. Yang, F. Gao and M. Zeng, Nanoscale Res. Lett., 2020, 15, 1–9 CrossRef PubMed.
- T. Aswani, S. Reshmi and T. Suchithra, Microbial Nanobionics: Volume 1, State-Of-The-Art, 2019, pp. 127–140 Search PubMed.
- S. Azizi, M. Mahdavi Shahri and R. Mohamad, Molecules, 2017, 22, 831 CrossRef PubMed.
- S. Ekrikaya, E. Yilmaz, C. Celik, S. Demirbuga, N. Ildiz, A. Demirbas and I. Ocsoy, J. Biotechnol., 2021, 341, 155–162 CrossRef CAS PubMed.
- R. Verma, A. Chauhan, S. Kumari, R. Jasrotia, A. Ali, C. Gopalakrishnan, R. Kumar and S. Ghotekar, Chem. Pap., 2023, 1–11 Search PubMed.
- A. Chauhan, R. Verma, S. Kumari, A. Sharma, P. Shandilya, X. Li, K. M. Batoo, A. Imran, S. Kulshrestha and R. Kumar, Sci. Rep., 2020, 10, 7881 CrossRef CAS PubMed.
- V. Mohammadzadeh, M. Barani, M. S. Amiri, M. E. T. Yazdi, M. Hassanisaadi, A. Rahdar and R. S. Varma, Sustainable Chem. Pharm., 2022, 25, 100606 CrossRef CAS.
- I. Ijaz, E. Gilani, A. Nazir and A. Bukhari, Green Chem. Lett. Rev., 2020, 13, 223–245 CrossRef CAS.
- Y. Liu, W. Zhang, C. Xu and X. Li, Int. J. Mol. Sci., 2016, 17, 1983 CrossRef PubMed.
- R. A. Syahputra, H. Helen, M. C. Gunawan, Z. D. Utari, A. Dalimunthe, E. Salim, M. Bastian, N. A. Taslim, P. Pitriani and S. E. Nugraha, J. Agric. Food Res., 2025, 22, 102091 CAS.
- T. Khouya, M. Ramchoun, H. Elbouny, A. Hmidani and C. Alem, J. Ethnopharmacol., 2022, 296, 115473 CrossRef CAS PubMed.
- Y.-G. Hwang, J.-J. Lee, A.-R. Kim and M.-Y. Lee, J. Life Sci., 2010, 20, 1625–1633 CrossRef.
- D. A. Infante-Rodríguez, M. J. Aguilar-Méndez, C. Landa-Cansigno, S. G. Vásquez-Morales, A. C. Velázquez-Narváez, J. E. Valenzuela-González, A. L. Kiel-Martínez, J. L. Monribot-Villanueva and J. A. Guerrero-Analco, Bot. Sci., 2024, 102, 1231–1250 CrossRef.
- S. Taniguchi, Y. Imayoshi, E. Kobayashi, Y. Takamatsu, H. Ito, T. Hatano, H. Sakagami, H. Tokuda, H. Nishino and D. Sugita, Phytochemistry, 2002, 59, 315–323 CrossRef CAS PubMed.
- X. Zhu, L. Wang, T. Zhao and Q. Jiang, J. Ethnopharmacol., 2022, 298, 115566 CrossRef CAS PubMed.
- H. Lv, J. Chen, W.-L. Li and H.-Q. Zhang, Zhongyaocai, 2008, 31, 1351–1354 Search PubMed.
- B. Chen, P. Long, Y. Sun, Q. Meng, X. Liu, H. Cui, Q. Lv and L. Zhang, Food Funct., 2017, 8, 687–694 RSC.
- A. A. Barzinjy, S. M. Hamad, A. F. Abdulrahman, S. J. Biro and A. A. Ghafor, Curr. Org. Synth., 2020, 17(7), 558–566, DOI:10.2174/1570179417666200628140547.
- H. G. Gebretnsae and Y. T. Gebreslassie, Nanoscale Res. Lett., 2021, 16(1), 97, DOI:10.1186/s11671-021-03555-6.
- S. A. Miraeez and R. Y. Mohammed, Int. J. Environ. Anal. Chem., 2026, 106, 378–409 CrossRef CAS.
- A. J. Enitan, L. I. Oduola and O. T. Olorunyomi, Lek. Sirovine, 2021, 41, 17–21 Search PubMed.
- A. Al Ghasham, M. Al Muzaini, K. A. Qureshi, G. O. Elhassan, R. A. Khan, S. A. Farhana, S. Hashmi, E. El-Agamy and W. E. Abdallah, Int. J. Pharm. Res. Allied Sci., 2017, 6(3), 33–46 CAS.
- K. S. Banu and L. Cathrine, Int. j. adv. res. chem. sci., 2015, 2, 25–32 Search PubMed.
- R. Verma, S. Singh, B. Priya, S. Jha and L. Rani, Res. J. Agric. Sci., 2023, 14, 1541–1546 Search PubMed.
- R. J. Ruch, S.-j. Cheng and J. E. Klaunig, Carcinogenesis, 1989, 10, 1003–1008 CrossRef CAS PubMed.
- V. Chooranam, Asian J. Pharm. Clin. Res., 2017, 10, 111–116 Search PubMed.
- J. R. Shaikh and M. Patil, Int. J. Chem. Stud., 2020, 8, 603–608 CrossRef CAS.
- S. Deshpande, J. Pharmacogn. Phytochem., 2013, 1, 23–27 Search PubMed.
- N. Raaman, Phytochemical Techniques, New India Publishing, 2006 Search PubMed.
- A. H. Al-Mossawi and Z. S. Al-Garawi, Qualitative tests of amino acids and proteins and enzyme kinetics, Mustansiriyah University, Baghdad, 2018 Search PubMed.
- A. R. Gollakota, V. S. Munagapati, V. Volli, S. Gautam, J.-C. Wen and C.-M. Shu, J. Hazard. Mater., 2021, 416, 125925 CrossRef CAS PubMed.
- Y. Wang, L. He, Y. Li, L. Jing, J. Wang and X. Li, J. Alloys Compd., 2020, 828, 154340 CrossRef CAS.
- Z. Ishak and D. Kumar, Tropical Aquatic and Soil Pollution, 2022, 2, 1–12 CrossRef.
- T. R. Das and P. K. Sharma, Mater. Sci. Semicond. Process., 2020, 105, 104721 CrossRef CAS.
- Y. He, D. B. Jiang, J. Chen and Y. X. Zhang, J. Colloid Interface Sci., 2018, 510, 207–220 CrossRef CAS PubMed.
- M. A. Al-Ghouti and M. M. Razavi, Environ. Technol. Innovation, 2020, 17, 100614 CrossRef CAS.
- F. Ahmadijokani, R. Mohammadkhani, S. Ahmadipouya, A. Shokrgozar, M. Rezakazemi, H. Molavi, T. M. Aminabhavi and M. Arjmand, Chem. Eng. J., 2020, 399, 125346 CrossRef CAS.
- M. Kasbaji, M. Mennani, N. Grimi, F. J. Barba, M. Oubenali, M. J. Simirgiotis, M. Mbarki and A. Moubarik, Process Biochem., 2022, 120, 213–226 CrossRef CAS.
- K. Naseri and A. Allahverdi, Res. Chem. Intermed., 2019, 45, 4863–4883 CrossRef CAS.
- D. Balarak, F. Kord Mostafapour, H. Azarpira and A. Joghataei, Past Name: British Journal of Pharmaceutical Research, Past ISSN: 2231-2919, 2017, pp. 1–9 Search PubMed.
- H. N. Tran, S.-J. You and H.-P. Chao, J. Environ. Chem. Eng., 2016, 4, 2671–2682 CrossRef CAS.
- M. Alizadeh, S. J. Peighambardoust, R. Foroutan, H. Azimi and B. Ramavandi, Environ. Res., 2022, 212, 113242 CrossRef CAS PubMed.
- R. Foroutan, S. J. Peighambardoust, R. Mohammadi, S. H. Peighambardoust and B. Ramavandi, Environ. Res., 2022, 211, 113020 CrossRef CAS PubMed.
- G. Yin, X. Song, L. Tao, B. Sarkar, A. K. Sarmah, W. Zhang, Q. Lin, R. Xiao, Q. Liu and H. Wang, Chem. Eng. J., 2020, 389, 124465 CrossRef CAS.
- S. I. Al-Saeedi, A. Areej, M. T. Qamar, A. Alhujaily, S. Iqbal, M. T. Alotaibi, M. Aslam, M. A. Qayyum, A. Bahadur and N. S. Awwad, Front. environ. sci., 2023, 11, 1156475 CrossRef.
- T. Chouchane, O. Khireddine and A. Boukari, J. Eng. Appl. Sci., 2021, 68, 34 CrossRef CAS.
- M. I. Khan, S. Sufian, F. Hassan, R. Shamsuddin and M. Farooq, RSC Adv., 2025, 15, 1989–2010 RSC.
- S. Salvestrini, L. Ambrosone and F.-D. Kopinke, J. Mol. Liq., 2022, 352, 118762 CrossRef CAS.
- H. Deng, Y. Wu, L. Li, P. Wang, K. Fang, J. Li, D. Hao, H. Zhu, Q. Wang and Q. Li, Sep. Purif. Technol., 2025, 354, 129455 CrossRef CAS.
- P. Patil, G. Jeppu, M. S. Vallabha and C. R. Girish, Environ. Sci. Pollut. Res., 2024, 31, 67442–67460 CrossRef CAS PubMed.
- N. M. Hosny and A. Dahshan, Mater. Chem. Phys., 2012, 137, 637–643 CrossRef CAS.
- M. Ahmad, M. Ahmad, M. H. Aziz and M. Asif, Heliyon, 2024, 10(5), 1–15, DOI:10.1016/j.heliyon.2024.e26708.
- S. Ayyappan, G. Panneerselvam, M. Antony and J. Philip, Mater. Chem. Phys., 2011, 130, 1300–1306 CrossRef CAS.
- K. V. Chandekar and K. Kant, Phys. B, 2018, 545, 536–548 CrossRef CAS.
- Z. Xu, W. Zhao, J. Liu and J. Fan, Magnetochemistry, 2023, 9, 146 CrossRef CAS.
- A. Sharma, N. Dhiman, B. P. Singh and A. K. Gathania, Mater. Res. Express, 2014, 1, 025042 CrossRef CAS.
- W. Yang, Q. Wang, K. Xu, Y. Yin, H. Bao, X. Li, L. Niu and S. Chen, Materials, 2017, 10, 956 CrossRef PubMed.
- Z. Sanaei, T. Shahrabi and B. Ramezanzadeh, Dyes Pigm., 2017, 139, 218–232 CrossRef CAS.
- S. Nikpour, M. Ramezanzadeh, G. Bahlakeh, B. Ramezanzadeh and M. Mahdavian, Constr. Build. Mater., 2019, 220, 161–176 CrossRef CAS.
- Q. Abbas, J. Nanomater. Mol. Nanotechnol., 2019, 8, 1–3 Search PubMed.
- S. Ma, L. Zheng, Y. Shi, L. Jia and X. Xu, J. Mater. Sci.: Mater. Electron., 2018, 29, 16921–16931 CrossRef CAS.
- T. Sarkar, S. Kundu, G. Ghorai, P. K. Sahoo and A. Bhattacharjee, Adv. Nat. Sci.:Nanosci. Nanotechnol., 2023, 14, 035001 CAS.
- Y. Kong, R. Jiao, S. Zeng, C. Cui, H. Li, S. Xu and L. Wang, Nanomaterials, 2020, 10, 367 CrossRef CAS PubMed.
- M. K. Goswami and A. Srivastava, J. Appl. Polym. Sci., 2025, 142, e56713 CrossRef CAS.
- C. B. Godiya, Y. Xiao and X. Lu, Int. J. Biol. Macromol., 2020, 144, 671–681 CrossRef CAS PubMed.
- E. M. Saad, M. Wagdy and A. S. Orabi, Sci. Rep., 2024, 14, 23614 CrossRef CAS PubMed.
- C. Aniagor, A. Hashem, N. Badawy and A. Aly, Hybrid Adv., 2023, 3 DOI:10.1016/j.hybadv.2023.100047.
- F. A. Adam, M. Ghoniem, M. Diawara, S. Rahali, B. Y. Abdulkhair, M. Elamin, M. A. B. Aissa and M. Seydou, RSC Adv., 2022, 12, 24786–24803 RSC.
- T. Chouchane, M. T. Abedghars, S. Chouchane and A. Boukari, Kuwait J. Sci., 2024, 51, 100210 CrossRef CAS.
- C. Toufik, K. Ouahida, C. Sana and B. Aatmane, Anal. Bioanal. Chem. Res., 2023, 10, 251–268 Search PubMed.
- C. Li, X. Li, Y. Yu, Q. Zhang, L. Li, H. Zhong and S. Wang, J. Ind. Eng. Chem., 2022, 105, 63–73 CrossRef CAS.
- T. C. Egbosiuba, T. Q. Tran, K. Arole, Y. Zhang, C. E. Enyoh, S. Mustapha, J. O. Tijani, V. K. Yadav, V. C. Anadebe and A. S. Abdulkareem, Results Eng., 2024, 22, 102073 CrossRef CAS.
- O. Bayram, U. Özkan, S. Kardeş, E. Moral, F. Göde and E. Pehlivan, Chem. Ecol., 2025, 41, 528–551 CrossRef CAS.
- D. Awang Chee, N. Kamaludin, M. Soffian, F. Abdul Halim and M. Mohamed Amin, Int. J. Environ. Sci. Technol., 2025, 22, 1591–1608 CrossRef CAS.
- S. K. D. Al Shariar Hasan, A. Bashar, P. K. Dhar and R. Haque, Extraction, 2025, 9, 10 Search PubMed.
- O. Bayram, U. Özkan, İ. Pekgözlü and F. Göde, Sep. Sci. Technol., 2025, 60, 1406–1420 CrossRef CAS.
- Y. Zhang, W. Zhang, G. Yang, Z. Chen and W. Zhang, Microporous Mesoporous Mater., 2025, 392, 113623 CrossRef CAS.
- S. Dutta, M. Jahan, N. Kaur, S. Barna, N. Sathi, R. Sultana, P. Dhar, M. Al Mamun, S. Chakrabarty and M. Amin, Int. J. Environ. Sci. Technol., 2025, 22, 1865–1890 CrossRef CAS.
- A. S. Abdulhameed, S. Abdullah, A. A. Al-Masud, M. Abualhaija and S. Algburi, Polym. Bull., 2025, 1–28 Search PubMed.
- S. D. Barna, M. N. Jahan, S. R. Sium, A. Nag, M. H. Ali and S. K. Dutta, Discov. chem., 2024, 1, 38 CrossRef.
- O. Bayram, U. Özkan, F. Göde, S. Coşkun and H. T. Şahin, J. Dispersion Sci. Technol., 2025, 46, 1737–1746 CrossRef CAS.
- T. S. Alomar, N. AlMasoud, G. Sharma, Z. A. Alothman and M. Naushad, J. Mol. Liq., 2021, 336, 116274 CrossRef CAS.
- Y. He, D. B. Jiang, J. Chen, D. Y. Jiang and Y. X. Zhang, J. Colloid Interface Sci., 2018, 510, 207–220 CrossRef CAS PubMed.
- T. Chouchane, O. Khireddine, S. Chouchane, M. T. Abedghars and H. Meradi, Turk. J. Chem., 2024, 48, 867–884 CrossRef CAS PubMed.
- F. Barracco, E. Parisi, G. Pipitone, E. Simone, S. Bensaid and D. Fino, Int. J. Environ. Sci. Technol., 2024, 21, 6513–6530 CrossRef CAS.
- K. Ezeh, I. Ogbu, K. Akpomie, N. Ojukwu and J. Ibe, Pac. J. Sci. Technol., 2017, 18, 251–264 Search PubMed.
- A. Hashem, C. O. Aniagor, S. Farag, M. Fikry, A. Aly and A. Amr, Waste Management Bulletin, 2024, 2, 172–183 CrossRef.
- S. K. M. Rozi, K. Y. Qin, H. N. A. Halim, M. M. H. Al-Rajabi and A. R. Ishak, Biomass Convers. Biorefin., 2025, 1–19 Search PubMed.
- C. R. Girish, Chem. Pap., 2025, 79, 5687–5706 CrossRef CAS.
- P. Saha and S. Chowdhury, Thermodynamics, 2011, 16, 349–364 Search PubMed.
- A. S. Abdulhameed, S. Abdullah, A. A. Al-Masud, M. Abualhaija and S. Algburi, Polym. Bull., 2025, 82, 6763–6790 CrossRef CAS.
- K. Polley and J. Bera, Int. J. Environ. Anal. Chem., 2024, 104, 5620–5637 CrossRef CAS.
- S. Mariska, J.-L. Lin, T. T. A. Tuyet, N. D. Hai and H.-P. Chao, Appl. Water Sci., 2024, 14, 95 CrossRef CAS.
- P. Arabkhani, N. Sadegh and A. Asfaram, Microchem. J., 2023, 184, 108149 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |





