 Open Access Article
Open Access ArticleSunlight-driven fast photo-degradation of Eriochrome Black T dye using highly efficient La-doped Ag3PO4 decorated with ZnS QDs
Shabana Bibia,
Amna Bashir b,
Noshabah Tabassuma,
It Ee Lee*cd,
Hafsa Yasmeenb,
Syed Mustansar Abbasf,
Qamar Walicd,
Muhammad Aamir
b,
Noshabah Tabassuma,
It Ee Lee*cd,
Hafsa Yasmeenb,
Syed Mustansar Abbasf,
Qamar Walicd,
Muhammad Aamir e,
Chingmai Kog and
Imtiaz Ahmad
e,
Chingmai Kog and
Imtiaz Ahmad *g
*g
aDepartment of Environment Sciences, Fatima Jinnah Women University, The Mall, Rawalpindi, 46000, Pakistan
bDepartment of Chemistry, Fatima Jinnah Women University, The Mall, Rawalpindi, 46000, Pakistan. E-mail: amnabashir79@yahoo.com
cFaculty of Artificial Intelligence and Engineering, Multimedia University, 63100 Cyberjaya, Malaysia. E-mail: ielee@mmu.edu.my
dCentre for Smart Systems and Automation, COE for Robotics and Sensing Technologies, Multimedia University, 63100 Cyberjaya, Selangor, Malaysia
eDepartment of Chemistry, Mirpur University of Science and Technology (MUST), Mirpur-10250 (AJK), Pakistan
fDepartment of Chemistry, College of Science, Imam Abdulrahman Bin Faisal University, P.O. Box 1982, Dammam, Saudi Arabia
gSchool of Electronics and Communication Engineering, Quanzhou University of Information Engineering, Quanzhou, 362000, China
First published on 5th May 2026
Abstract
The untreated discharge of dye-contaminated effluents into aquatic environments poses serious risks to both environmental integrity and public health. Therefore, the development of efficient dye removal strategies is essential for pollution control and ecosystem protection. This study aims to investigate and optimize the photocatalytic degradation of Eriochrome Black T (EBT) in aqueous solutions using novel Ag3PO4-based composites, including lanthanum-doped Ag3PO4, and ZnS quantum dots under sunlight irradiation. Pure Ag3PO4, 2% and 6% La-doped Ag3PO4, and ZnS quantum dots were synthesized via a co-precipitation method, and their composite photocatalysts were fabricated using a hybrid mixing approach. The characterizations of the materials were carried out using X-ray diffraction, BET surface area analysis, UV-visible diffuse reflectance spectroscopy (UV-vis DRS), Fourier transform infrared (FTIR) spectroscopy, Raman spectroscopy, X-ray photoelectron spectroscopy (XPS), thermogravimetric analysis (TGA), and scanning electron microscopy with energy-dispersive spectroscopy (SEM–EDS) to evaluate their structural, optical, and morphological properties. The synthesized photocatalysts, including pure Ag3PO4, 2% and 6% La-doped Ag3PO4, ZnS quantum dots, and their composite systems, exhibited well-defined crystalline structures, as confirmed by X-ray diffraction (XRD) analysis. UV-vis DRS analysis showed that pure Ag3PO4 had a band gap of 2.41 eV, which decreased to 2.39 eV for 2% La-doped Ag3PO4 and 2.36 eV for 6% La-doped Ag3PO4, while ZnS quantum dots exhibited a band gap of 3.6 eV, and the 6% La-doped Ag3PO4/ZnS QD composite showed a significantly reduced band gap of 1.5 eV. Upon the incorporation of ZnS QDs into the 6% La-Ag3PO4 particles, the surface area of the 6% La-Ag3PO4 heterojunction composite increased from 133.446 to 141.120 m2 g−1. The photocatalytic activity of the synthesized Ag3PO4-based materials was evaluated through the degradation of Eriochrome Black T (EBT) under light irradiation. The influence of key operational parameters, including the solution pH (3–10), dye concentration (10–20 ppm), photocatalyst dosage (0.05–0.15 g), and irradiation time (5–120 min), was systematically investigated to assess their effect on degradation efficiency. Among the examined photocatalysts, 6% La-doped Ag3PO4 exhibited the highest photocatalytic performance, demonstrating the beneficial role of lanthanum incorporation. The degradation efficiencies of 97.84% and 84.88% were achieved using 6% La-doped Ag3PO4 and the 6% La-doped Ag3PO4/ZnS QD composite, respectively, under the optimized conditions of pH 6 and an irradiation time of 120 min. Overall, these results indicate that La-modified Ag3PO4-based photocatalysts are promising and sustainable materials for the effective treatment of dye-contaminated wastewater, offering significant potential for environmental remediation applications.
1 Introduction
Rapid population growth and ongoing industrial development are major contributors to water pollution, creating serious environmental, economic, and public health challenges by contaminating vital water resources.1–4 Organic substances like dyes and hazardous pollutants like heavy metals are the main sources of water contamination.5,6 Azo dyes, commonly applied in textile industries, are a significant source of water pollution. These dyes are extremely resistant to degradation, even under sunlight, with half-lives exceeding two thousand hours.7 More than 50% of the dye produced worldwide is Eriochrome Black T (EBT).8–10 Many industries, including pharmaceutical, paper printing, leather, paint, and cosmetics, utilize it, which contaminates the water.11 Its intricate chemical structure, resistance to photodegradation, and light stability make it a significant target for removal.12–14Indiscriminate effluent discharge from industries is largely responsible for the rise in water pollution, causing rapid deterioration in water quality and posing serious environmental threats. Pollutants like EBT accumulate in different parts of plants or plant products, which are then consumed by humans and animals, entering the food chain and having negative effects. Additionally, groundwater may become contaminated by EBT-containing industrial effluents, rendering it unsafe for human consumption.15–18 Numerous health problems, such as pulmonary toxicity, genetic lesions, genetic mutation, congenital malformations, and oncogenesis, can result from long-term exposure to high concentrations of EBT dye.19,20 For this reason, wastewater must be properly treated before being released into the environment. It is also vital to develop affordable, efficient, and eco-friendly technology for removing EBT and azo dyes from both water and wastewater sources.
All dyes, especially reactive dyes, cannot always be completely removed using conventional dye removal techniques, which include physical and biological processes. Photocatalysis is one of the most promising processes (AOPs) which helps in the oxidative elimination of organic contaminants. The breakdown of organic pollutants is accelerated by this process, which uses catalysts and light sources. Studies reveal that the pigments in industrial effluents can be removed by photocatalysis at a rate of 70–80%. In an effort to achieve efficient dye degradation, numerous scientists are studying heterogeneous photocatalysts, including WO3, ZnO, and TiO2.21
Ag3PO4 has been synthesized in various shapes as reported in literature such as tetrapod, tetrahedral, cubic, spherical, and trisoctahedral.22–25 When exposed to visible light, the produced Ag3PO4 showed 100% photodegradation of the MB dye. Concave trioctahedral Ag3PO4 particles were created. using the same process but with a different phosphate precursor. Furthermore, when employed as a photocatalyst for the degradation of RhB dye, they similarly achieved 100% degradation under visible-light illumination.23 Many studies have shown that silver phosphate displays excellent photocatalytic performance. However, it also has a high photogenerated charge-carrier recombination rate. Therefore, doping has been incorporated into the host Ag3PO4's lattice site to increase its photocatalytic potential. Dopants such as nickel, bismuth, lanthanum, fluorine, sulfate, carbonate, and rare-earth elements (dysprosium, erbium, and gadolinium) have been identified in several studies. These elements have the potential to enhance the chemical characteristics and photocatalytic efficiency of Ag3PO4.26–32 It has also been reported that lanthanum, as a dopant, alters the surface structure of the host lattice.33,34 Amirulsyafiee et al. used the coprecipitation method to synthesize La-doped Ag3PO4 and studied its photocatalytic performance under visible light. The 1% La-doped Ag3PO4 demonstrated superior photocatalytic activity, degrading 81% of methylene blue in 60 minutes and 94% of methyl orange in 30 minutes. The enhanced photocatalytic efficiency was attributed to reduced electron–hole recombination and morphological changes resulting from lanthanum incorporation.32,35 Ag3PO4 suffers from photocorrosion due to Ag+ reduction under visible light. Therefore, La3+ doping confers Ag3PO4 antiphotocorrosion properties by altering internal electron migration pathways, introducing new electronic states, and improving electron migration, thereby enhancing charge separation, facilitating internal electron transfer, and increasing photocatalytic stability.36
Zinc sulfide (ZnS) is one of the most attractive metal chalcogenides due to its intrinsic properties, including high electronic mobility, water insolubility, low cost, and non-toxicity.37,38 ZnS QDs have been modified for increased photocatalytic effectiveness using techniques such as heterojunction creation and dopant inclusion.39 Recent studies have revealed that tuning the electronic structure and coordination environment of catalysts is an effective strategy to enhance charge separation and improve photocatalytic efficiency.40 Furthermore, the construction of heterojunction systems has been widely explored to promote interfacial charge transfer and extend light absorption. In addition, the local structural distortion of catalytic sites can regulate electron density and facilitate charge transfer, which plays a crucial role in determining catalytic performance.41 The photocatalytic activity of a material is also strongly influenced by its band gap. An effective photocatalyst typically has a band gap in the range of 2–3 eV, allowing efficient visible-light absorption while providing sufficient redox potential for photocatalytic reactions, whereas materials with too low a band gap (<1.23 eV) are generally not suitable, as they cannot facilitate the necessary redox processes effectively.42
In the current work, Ag3PO4, La-doped Ag3PO4, and ZnS QDs were synthesized via a co-precipitation method. The composites were formed by mixing. The photocatalytic performance of individual components and the composites was studied for the degradation of Eriochrome Black T (EBT) under sunlight irradiation. Comprehensive characterizations of the synthesized materials were carried out using X-ray diffraction, BET surface area analysis, UV-visible diffuse reflectance spectroscopy (UV-vis DRS), Fourier transform infrared (FTIR) spectroscopy, Raman spectroscopy, X-ray photoelectron spectroscopy (XPS), thermogravimetric analysis (TGA), and scanning electron microscopy with energy-dispersive spectroscopy (SEM–EDS) to evaluate their structural, optical, and morphological properties. The influence of key factors, including the dye concentration, catalyst dose, pH, and temperature, was thoroughly investigated. Interestingly, the individual components exhibited higher degradation efficiency for EBT dye than the composites under the conditions studied. No studies are available on undoped Ag3PO4 and doped Ag3PO4 for EBT degradation. Furthermore, the ZnS quantum dots synthesized via the presented co-precipitation approach have not been explored for EBT degradation. However, direct comparison with previously reported studies is limited, as, to the best of our knowledge, Ag3PO4-based photocatalysts have not been explored for EBT degradation. Therefore, this study not only introduces a new photocatalytic system but also extends the application of Ag3PO4-based materials to a previously unexplored EBT azo dye, providing new insights into their photocatalytic behaviour.
2 Materials and methods
The chemicals used for synthesis in this research were silver nitrate (AgNO3; Sigma-Aldrich), potassium dihydrogen phosphate (KH2PO4; Scharlau), lanthanum nitrate hexahydrate (La(NO3)3·6H2O; Sigma-Aldrich), zinc acetate dihydrate (Zn(CH3COO)2·2H2O; Scharlau), sodium sulfide (Na2S; Sigma-Aldrich), sodium dodecyl sulfate (Sigma-Aldrich), deionized water, distilled water, and Eriochrome Black T (Sigma-Aldrich).2.1 Synthesis of Ag3PO4 and La-doped Ag3PO4
Silver phosphate (Ag3PO4) and lanthanum-doped silver phosphate (La-Ag3PO4) were synthesized via a modified co-precipitation method.35 Briefly, 0.1 M AgNO3 and 0.3 M KH2PO4 solutions were prepared separately in 20 mL of deionized water. The KH2PO4 solution was added dropwise to the AgNO3 solution under continuous stirring, resulting in the formation of yellow precipitates (confirming the formation of Ag3PO4). The suspension was left undisturbed for 2 hours to allow the precipitates to settle, followed by vacuum filtration and repeated washing with deionized water. The obtained product was dried in an oven for 2 hours and stored.For La-doped Ag3PO4 synthesis, the same procedure was followed with the addition of an appropriate amount of La(NO3)3·6H2O to the AgNO3 solution to achieve 2% and 6% La doping. The schematic representation of the synthesis route of Ag3PO4 and La-doped Ag3PO4 is shown in Fig. 1(a).
 | ||
| Fig. 1 Schematic of the synthesis routes to (a) doped and undoped Ag3PO4, (b) ZnS QDs, and (c) Ag3PO4/ZnS QD and La-doped Ag3PO4/ZnS QD composites. | ||
2.2 Synthesis of ZnS quantum dots
The co-precipitation method was used to synthesize ZnS quantum dots with slight modifications.43 In the standard synthesis procedure, a 0.5 M zinc acetate dihydrate solution and a 0.5 M sodium sulfide solution were prepared in 25 mL of distilled water. Then, the zinc acetate solution was added to the sodium sulfide solution dropwise under constant stirring at neutral pH. Additionally, 25 mL of a 0.5 M SDS solution was added dropwise to the solution while being constantly stirred. The above solution was stirred for 2 hours until white precipitates were formed. The solution was filtered and washed with a mixture of water and ethanol several times. The precipitates were dried in an oven at 60 °C for 5 hours. In Fig. 1(b), a schematic of the synthesis of ZnS quantum dots is presented.2.3 Synthesis of composites
The Ag3PO4/ZnS composite was prepared by combining Ag3PO4 and ZnS quantum dots in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 weight ratio. In the typical process, 0.5 g of the synthesized Ag3PO4 was dispersed in 25 mL of deionized water and ultrasonicated at 30 °C for 15 minutes. Subsequently, 0.5 g of ZnS quantum dots was added to the mixture, followed by ultrasonication for 15 min to achieve uniform dispersion. The mixture was then stirred for 2 hours, during which dark-green precipitates formed, indicating the formation of the Ag3PO4/ZnS composite. The product was allowed to settle, filtered, washed several times with deionized water, and dried in an oven for 2 hours at 50–60 °C. Fig. 1(c) presents the schematic illustration of the synthesis method of Ag3PO4/ZnS QDs and La-doped Ag3PO4/ZnS QDs. The synthesis of La-Ag3PO4/ZnS composites followed the same procedure, using La-doped Ag3PO4 (2% and 6%) instead of pure Ag3PO4.
1 weight ratio. In the typical process, 0.5 g of the synthesized Ag3PO4 was dispersed in 25 mL of deionized water and ultrasonicated at 30 °C for 15 minutes. Subsequently, 0.5 g of ZnS quantum dots was added to the mixture, followed by ultrasonication for 15 min to achieve uniform dispersion. The mixture was then stirred for 2 hours, during which dark-green precipitates formed, indicating the formation of the Ag3PO4/ZnS composite. The product was allowed to settle, filtered, washed several times with deionized water, and dried in an oven for 2 hours at 50–60 °C. Fig. 1(c) presents the schematic illustration of the synthesis method of Ag3PO4/ZnS QDs and La-doped Ag3PO4/ZnS QDs. The synthesis of La-Ag3PO4/ZnS composites followed the same procedure, using La-doped Ag3PO4 (2% and 6%) instead of pure Ag3PO4.
The crystal structure and crystalline size of the synthesized materials were analysed using XRD. FTIR spectroscopy was utilized to examine the functional groups of the synthesized materials. An FTIR spectrophotometer (SHIMADZU-8400) was used to record FTIR spectra in the range from 400 to 4000 cm−1. The surface morphology of the synthesized materials was analysed by scanning electron microscopy (SEM). The elemental composition of the synthesized materials was determined using energy-dispersive X-ray spectroscopy (EDS). The thermal behaviour of the synthesized materials was analysed by thermogravimetric analysis (TGA) in an inert gas atmosphere. Raman spectroscopy was used to investigate the structural and vibrational properties of the synthesized materials.
2.4 Adsorption experiments
The adsorption studies were conducted using a method already reported in the literature, with slight modifications.44 Briefly, an adsorption experiment was conducted using a 15 ppm dye solution at 25 °C. An appropriate quantity of the catalyst (0.1 g) and 50 mL of the dye solution were combined, and the mixture was stirred for 1 hour in the dark. After 1 hour, an aliquot of the solution was taken out, followed by the separation of the catalyst by centrifugation. The absorbance was measured at 550 nm using a UV-visible spectrophotometer. Fig. S1(a) presents the schematic representation of the adsorption experiment.2.5 Photocatalytic degradation experiments
The photocatalytic degradation experiment was conducted in direct sunlight using a previously reported method.45,46 Briefly, a known quantity of the catalyst (0.1 g) was added to 50 mL of a dye solution. The dye solution was exposed to direct sunlight for 2 hours under continuous stirring. An adequate volume of the solution was removed after 5, 15, 30, 45, 60, and 120 min. The catalyst was removed from the sample via centrifugation, and the absorbance was measured at 550 nm. The schematic representation of the photocatalytic experiment is shown in Fig. S1(b). The equation used to determine the percentage of photocatalytic degradation is given below.47
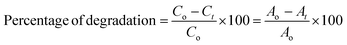 | (1) |
3 Results and discussion
3.1 Characterization of the synthesized photocatalysts
Fig. 2(a) displays the XRD patterns of pure and doped silver phosphate (Ag3PO4).26,32 In the XRD patterns of Ag3PO4 and La-doped Ag3PO4, the characteristic peaks observed at different 2θ values correspond to the (110), (200), (210), (211), (220), (310), (222), (320), (321), (400), and (411) planes, respectively.26,32 All these peaks are well-matched with the JCPDS card no. 06-0505.26,32 Compared to pure Ag3PO4, no change in the crystal structures and no impurity peaks are observed in the XRD patterns of 2% and 6% La-doped Ag3PO4, which indicates that the La(III) ions are successfully integrated and uniformly distributed in the host lattice. The XRD pattern of ZnS quantum dots (ZnS QDs) is also shown in Fig. 2(a).48,49 In the XRD pattern of ZnS QDs, the major peaks observed at 2θ values of 29.20°, 48.53°, and 57.52° correspond to the (111), (220), and (311) planes, respectively. All these peaks are in accordance with JCPDS card no. 05-0566. The observed XRD pattern closely resembles the typical pattern of the cubic phase of the zinc blende structure.49 The XRD patterns of the composites are displayed in Fig. 2(a). In the XRD patterns (Fig. 2(a)) of the composites, the diffraction peaks corresponding to Ag3PO4 and ZnS QDs are observed, confirming the coexistence of both phases in the composites. The crystal structure of Ag3PO4 is shown in Fig. 2(b).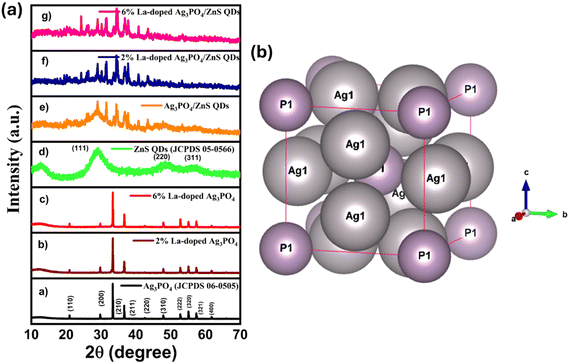 | ||
| Fig. 2 (a) XRD patterns of the synthesized nanomaterials. (b) Crystal structure of silver phosphate. | ||
The crystalline size of the synthesized materials was calculated using the Debye–Scherrer equation, as shown in eqn (2).50
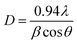 | (2) |
The dislocation density (δ) was calculated using the following formula:51
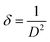 | (3) |
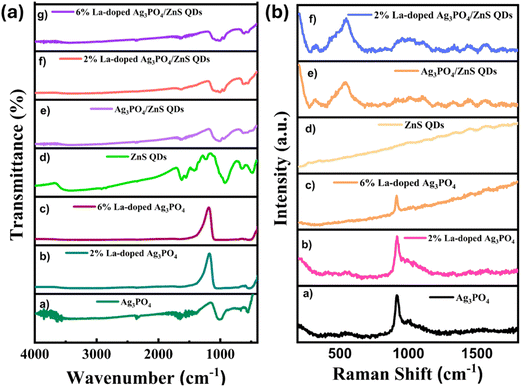
The crystal microstain (ε) was calculated using the following formula:52
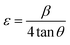 | (4) |
All calculated parameters are given in Table S1 in the SI. The average crystalline size of pure Ag3PO4 is found to be 158.13 nm. A clear reduction in the crystallite size is observed after La incorporation. The average crystallite sizes of 2% La-doped Ag3PO4 and 6% La-doped Ag3PO4 samples are 73.07 nm and 118 nm, respectively. This decrease can be attributed to the introduction of lattice strain and structural defects due to the addition of La, which restricts crystal growth, which is consistent with a previous report.53 The calculated average crystallite size of the ZnS quantum dots is 2.26 nm, closely matching the previously reported value of 2.09 nm for ZnS quantum dots.43,54 The Ag3PO4/ZnS composite further exhibits a reduced crystallite size of 10.33 nm. After the incorporation of La into this composite, the crystallite sizes are 37.80 and 21.54 nm for the 2% and 6% La-doped Ag3PO4, respectively.
FT-IR analysis was utilized to examine the functional groups of the synthesized materials. The FT-IR spectra of pure and La-doped Ag3PO4 are presented in Fig. 3(a). The main characteristic peaks are observed at 507, 550, 558, 670, 1007, and 2350 cm−1, respectively. The peak observed at 507 cm−1 is due to the stretching vibrations of the Ag–O band, while the peak at 550 cm−1 corresponds to the O–P–O bending vibrations.35 The stretching and bending vibrations of the P–O bond in the PO43− group appear at 1007 and 558 cm−1, respectively.55–57 The peak at 2350 cm−1 is due to atmospheric carbon dioxide.48 In the case of La-doped Ag3PO4, the main peak is observed at 610 cm−1, corresponding to the La–O bond vibrations.35
 | ||
| Fig. 3 (a) FTIR spectra of the synthesized nanomaterials. (b) Raman spectra of the synthesized nanomaterials. | ||
In the FTIR spectrum of ZnS quantum dots (Fig. 3(a)), the main characteristic peaks appear at 651 cm−1 and 484 cm−1, corresponding to the stretching vibrations of the Zn–S bond.49,58 The absorption peaks at 1384,59,60 1419,61 156362 and 1620 cm−1 (ref. 61) correspond to the symmetric stretching vibrations of the –COO− bond. The broad peak observed at 3400 cm−1 is assigned to the stretching vibrations of the O–H bond, which arises from the adsorption of water molecules on the surface of ZnS.63 The absorption peaks observed at 2852 cm−1 and 2921 cm−1 are attributed to the C–H stretching,64 while the peaks at 1458 cm−1 and 1466 cm−1 correspond to the CH3 bending vibrations.65 The peak observed at 928 cm−1 is assigned to the S–C bond stretching vibrations.66
The Raman spectra of pure and doped Ag3PO4 are shown in Fig. 3(b). The Raman spectrum of pure Ag3PO4 displays distinct vibrational bands characteristic of the phosphate tetrahedron, which are closely related to those reported in a previous study.67 The high-intensity band at 912 cm−1 is assigned to the stretching vibration of the terminal oxygen of the PO43− groups. A weak band is observed at 555 cm−1, which corresponds to the asymmetrical bending vibration of the P–O–P bond. In the case of La-doped Ag3PO4, no new band is identified in the range of 200–1800 cm−1. However, the band at 912 cm−1 becomes progressively weaker and narrower with an increase in the concentration of La. Thus, the incorporation of La3+ alters only the intrinsic phosphate vibrations.
In the Raman spectrum of ZnS quantum dots (Fig. 3(b)), the characteristic bands appear at 270 cm−1 and 347 cm−1, which correspond to the transverse optical (TO) and first-order longitudinal optical (1LO) phonon modes of cubic ZnS, respectively.68–70 Additional bands appearing between ∼997 and 1562 cm−1 are linked to multiphonon processes and SDS vibrational modes.71 These spectral characteristics not only confirm the structural crystallinity of ZnS QDs but also reveal the phonon confinement effects in nanocrystals. The Raman spectra of the composites display the vibrational modes of both components with slight band broadening, confirming that the two crystalline phases coexist in the composites.
The thermal stability of the synthesized photocatalysts was evaluated using thermogravimetric analysis (TGA), and the results are shown in Fig. 4(a). A slight weight loss is observed in the case of pure and doped Ag3PO4 up to 750 °C, demonstrating high thermal stability. The minor weight loss in these TGA curves is correlated with water loss. Similarly, the TGA curve of ZnS quantum dots, shown in Fig. 4(a), displays approximately 21% weight loss. The 7% weight loss at 150 °C is due to the removal of adsorbed water from the quantum dots' surface. However, the loss of sulfuric acid groups and degradation occur up to 400 °C. At 600 °C, the weight loss is due to the oxidation of ZnS to ZnO. Furthermore, in the case of Ag3PO4/ZnS quantum dot composites, three weight loss regions are observed up to 600 °C. The first weight loss at 150 °C corresponds to the loss of adsorbed water molecules, while the second one at 400 °C involves the evaporation of organic compounds. The final weight loss at 600 °C relates to the breakdown of the residual inorganic content, the oxidation of metal species, or internal structural modification.
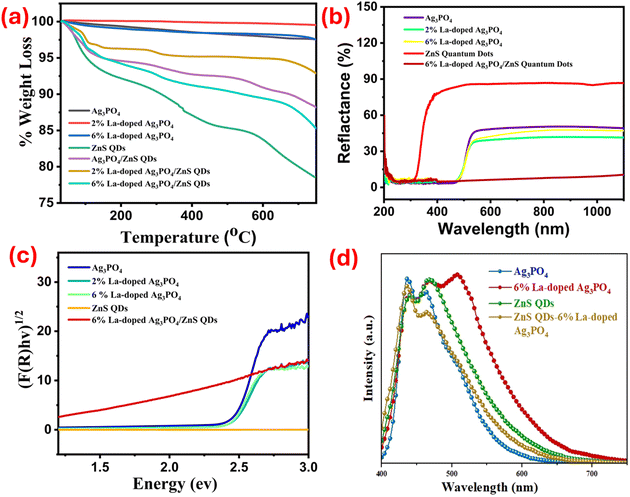 | ||
Fig. 4 (a) TGA analysis of the synthesized nanomaterials. (b) Relactance vs. wavelength spectra. (c) Tauc plots showing 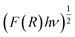 vs. hv for band gap estimation. (d) PL spectra of selected samples. vs. hv for band gap estimation. (d) PL spectra of selected samples. | ||
The composite exhibits a 12% weight loss, indicating superior thermal stability compared to pure ZnS. In the doped composites, overall, 14% and 7.15% weight loss are observed for 6% La-doped Ag3PO4/ZnS and 2% La-doped Ag3PO4, respectively.
The optical characteristics of the synthesized catalysts were analysed by UV-visible diffuse reflectance spectroscopy (UV-DRS). The data were collected in terms of the reflectance (R) versus wavelength (λ), and the spectra are presented in Fig. 4(b). The band gap energies (Eg) were estimated using the Kubelka–Munk function,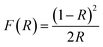 ,72 and Tauc plots were created by plotting (F(R)·hv)1/2 against the photon energy (hν) (Fig. 4(c)). The band gap values were determined by extrapolating the linear portion of each Tauc plot to the energy axis. The calculated band gap energies are shown in Table 1.
,72 and Tauc plots were created by plotting (F(R)·hv)1/2 against the photon energy (hν) (Fig. 4(c)). The band gap values were determined by extrapolating the linear portion of each Tauc plot to the energy axis. The calculated band gap energies are shown in Table 1.
| S. no. | Catalyst | Band gap (eV) |
|---|---|---|
| 1 | Pure Ag3PO4 | 2.41 |
| 2 | 2% La-doped Ag3PO4 | 2.39 |
| 3 | 6% La-doped Ag3PO4 | 2.36 |
| 4 | ZnS quantum dots (QDs) | 3.60 |
| 5 | 6% La-doped Ag3PO4/ZnS QD composite | 1.50 |
Pure Ag3PO4 exhibits a band gap of 2.41 eV. Upon the addition of 2% and 6% La to the lattice, the band gap decreases slightly to 2.39 eV and 2.36 eV, respectively. The decrease in the band gap is possibly due to the creation of oxygen vacancies within the lattice upon La doping.35 In contrast, ZnS QDs have a wider band gap of 3.60 eV and mainly absorb in the UV region. The 6% La-doped Ag3PO4/ZnS QD composite shows a markedly reduced band gap of 1.50 eV.
Photoluminescence (PL) spectroscopy was employed to identify defect sites and oxygen vacancies within the synthesized nanoparticles. As shown in Fig. 4(d), the results clearly indicate that all samples exhibit two distinct emission peaks. Pure Ag3PO4 exhibits two peaks at 436 and 463 nm.73 6% La-doped Ag3PO4 shows three peaks at 439, 468, and 507 nm. ZnS quantum dots (QDs) show two peaks at 439 and 467 nm. The 6% La-doped Ag3PO4/ZnS QD composite shows two peaks at 436 and 464 nm. It is observed that 6% La-doped Ag3PO4 shows an additional peak. The emission band observed within the 490–550 nm range is attributed to the radiative recombination of photogenerated charge carriers at surface-active oxygen vacancies.74,75
The surface area of the synthesized samples was investigated using BET analysis (Fig. 5(a)). The BET specific surface areas are determined to be 130.109, 89.934, 133.446, 116.537, and 141.120 m2 g−1 for pure Ag3PO4, 2% La-Ag3PO4, 6% La-Ag3PO4, ZnS QDs, and 6% La-Ag3PO4/ZnS QDs, respectively. Upon the incorporation of ZnS QDs into the 6% La-Ag3PO4 particles, the surface area of the 6% La-Ag3PO4 heterojunction composite increases from 133.446 to 141.120 m2 g−1.
The pore size distribution of the samples was analysed by the BJH adsorption method (Fig. 5(b)). The pore radii of Ag3PO4, 2% La-Ag3PO4, 6% La-Ag3PO4, ZnS QDs, and 6% La-Ag3PO4/ZnS nanoparticles are 15.578, 15.564, 15.581, 15.610, and 14.761 Å, respectively. The pore sizes of Ag3PO4, 2% La-Ag3PO4, 6% La-Ag3PO4, ZnS QDs, and 6% La-Ag3PO4/ZnS nanoparticles are 3.116 nm, 3.113 nm, 3.116 nm, 3.122 nm, and 2.952 nm, respectively. This indicates a mesoporous structure according to the IUPAC classification (pores between 2 and 50 nm).76
Additionally, the pore volumes of Ag3PO4, 2% La-Ag3PO4, 6% La-Ag3PO4, ZnS QDs, and 6% La-Ag3PO4/ZnS are 0.036, 0.031, 0.038, 0.040, and 0.053 cm3 g−1, respectively. This suggests that the BET surface area and pore size of the material are not the primary factors influencing its photocatalytic activity.77 The comparison between different catalysts is presented in Fig. 5(c).
The surface morphology of the catalysts was studied using SEM images at various magnifications, as shown in Fig. 6(a–l). The pure Ag3PO4 particles have irregular spherical shapes with a porous, rough surface, and some are aggregated78 (Fig. 6(a–d)). The SEM images of La-doped Ag3PO4 reveal that the morphology of silver phosphate changes from spherical to tetrapod, with a rough surface consisting of some pores (Fig. 6(e–h)). In the case of ZnS QDs, a regular morphology with slight agglomeration is observed, as shown in Fig. 6(i–l).79
 | ||
| Fig. 6 SEM images of (a–d) Ag3PO4, (e–h) 6% La-doped Ag3PO4, and (i–l) ZnS quantum dots at different magnifications. | ||
The elemental composition of the materials was determined using energy-dispersive X-ray spectroscopy (EDS). The presence of Ag, P, and O elements in the EDS spectrum (Fig. 7(a)) of Ag3PO4 confirms the formation of Ag3PO4. In the case of 6% La-doped Ag3PO4, the EDS spectrum (Fig. 7(b)) displays the peaks of Ag, P, La, and O. The existence of the peaks of La in the EDS spectrum of 6% La-doped Ag3PO4 confirms that lanthanum (La) is successfully introduced into the lattice. The existence of carbon in the EDS spectra of pure and doped Ag3PO4 is probably due to the latex material of the SEM sample holder.80 The presence of Zn and S peaks in the EDS spectrum (Fig. 7(c)) confirms the synthesis of the ZnS QDs. The presence of the small peaks of copper (Cu) and silicon (Si) in the EDS spectrum is due to the sample holder and EDS detector, respectively.
Fig. 8(a–f) presents the TEM micrographs of pure and 6% La-doped Ag3O4 at varying magnifications, revealing the nanostructured morphology of the synthesized photocatalysts. The TEM micrograph of the nanoparticles shows that the synthesized nanoparticles are nearly spherical, tend to form soft agglomerates due to magnetic interactions, which range in size from 0.5 µm to less than 100 nm, and exhibit a smooth surface texture due to proper dispersion. The TEM images of ZnS are presented in Fig. 8(g–h).
 | ||
| Fig. 8 TEM images of (a–c) pure Ag3PO4, (d–f) 6% La-Ag3PO4, and (g–h) ZnS QDs at different magnifications. | ||
The XPS survey spectra of La-Ag3PO4/ZnS nanocomposites reveal the presence of peaks corresponding to silver (Ag), oxygen (O), zinc (Zn), sulfur (S), phosphorus (P), lanthanum (La), and carbon (C), as illustrated in Fig. 9(a–f) and S2. The two characteristic peaks observed at 366.4 eV and 370.2 eV correspond to Ag 3d5/2 and Ag 3d3/2, respectively, indicating the presence of Ag+ in Ag3PO4.81 The O 1s peak at 530.2 eV corresponds to lattice oxygen in Ag3PO4.82 As shown in Fig. 9(d), the Zn 2p spectrum displays two characteristic peaks at 1020.2 eV and 1044.5 eV, assigned to Zn 2p3/2 and Zn 2p1/2, respectively, confirming the presence of ZnS.83 In the S 2p spectrum shown in Fig. 9(e), three distinct peaks are observed. The two peaks at binding energies of 162 and 163.1 eV are attributed to the S 2p3/2 and S 2p1/2 orbitals, respectively, confirming the presence of metal–sulfur bonds, such as those found in ZnS. The additional peak appearing at 169.1 eV is assigned to sulfur species in a higher oxidation state, which likely results from surface oxidation effects.83
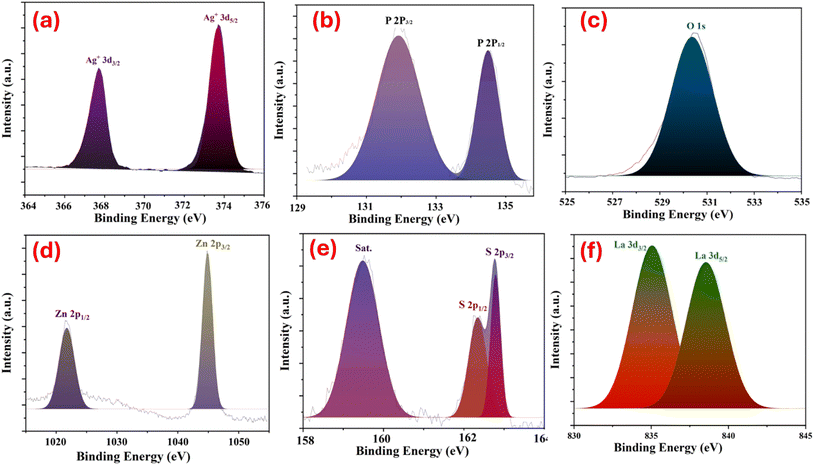 | ||
| Fig. 9 XPS spectra of the La-doped Ag3PO4 decorated with ZnS quantum dots: (a) Ag 3d, (b) P 2p, (c) O 1s, (d) Zn 2p, (e) S 2p, and (f) La 3d. | ||
The P 2p XPS spectrum shown in Fig. 9(b) exhibits a characteristic peak at a binding energy of 133.72 eV, which is attributed to phosphorus in the phosphate species.84 The lanthanum 3d XPS spectrum is illustrated in Fig. 9(f). For lanthanum, the closed-shell La3+ ion shows La 3d5/2 and La 3d3/2 peaks. As shown in the spectrum, the La 3d peaks are deconvoluted into distinct subpeaks, with binding energies of 832.5 eV and 837.1 eV, corresponding to the La 3d5/2 and La 3d3/2 levels, respectively, confirming the presence of La in the trivalent oxidation state.85 The deconvoluted C 1s XPS spectrum of Ag3PO4 nanoparticles, shown in Fig. S2(a), displays two distinct peaks at 284.2 eV and 286.9 eV. The C1 peak at 284.2 eV is assigned to sp2-bonded carbon C–C, while the higher-binding-energy peak, C2, at 286.9 eV corresponds to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O species. These results are consistent with the literature.86 The complete XPS survey is shown in Fig. S2(b).
O species. These results are consistent with the literature.86 The complete XPS survey is shown in Fig. S2(b).
3.2 Application of the synthesized photocatalysts
The results are presented in Fig. 11(a–f) and S4. The extent of dye degradation was examined by monitoring the change in the absorbance of the dye at regular intervals. The continuous decrease in absorbance with increasing irradiation time clearly indicates the effective photodegradation of EBT molecules. The photocatalytic activities of pure Ag3PO4, 2% La-doped Ag3PO4, 6% La-doped Ag3PO4, and ZnS quantum dots are 94.71%, 95.63%, 97.84% and 97.59%, respectively. Furthermore, the photocatalytic degradation efficiencies of the composite materials, i.e., Ag3PO4/ZnS QDs, 2% La-doped Ag3PO4/ZnS QDs, and 6% La-doped Ag3PO4/ZnS QDs, is 83.33%, 79.54%, and 84.88%, respectively.
La3+ doping improves photocatalytic activity primarily through lattice distortion and defect formation. As shown in Table S2, the crystallite size decreases with increasing La content, while the microstrain and dislocation density increase, indicating the introduction of structural defects. These defects increase the number of active sites and slightly reduce the band gap, enhancing light absorption and photocatalytic performance.
Although heterojunctions generally enhance photocatalytic activity, the lower degradation efficiency of the 6% La-Ag3PO4/ZnS composite than that of 6% La-Ag3PO4 can be explained by surface coverage effects. A similar behaviour has been reported for Ag3PO4/g-C3N4 composites, where the excessive loading of one component reduces photocatalytic activity due to the shielding of active sites.88 In this work, the presence of ZnS may reduce the accessibility of Ag3PO4 active sites, leading to lower efficiency.
Although heterojunctions usually improve photocatalytic activity by reducing electron–hole recombination,89 the 6% La-Ag3PO4/ZnS composite showed lower degradation efficiency than 6% La-Ag3PO4, likely because its low band gap (1.5 eV) can lead to faster charge recombination.
The slightly lower photocatalytic activity of the composite materials can be attributed to several factors. First, the combination of different components may have led to poor interfacial contact or inefficient charge transfer between the constituents, which hinders the separation and migration of photogenerated charge carriers. Second, there may have been an unfavourable band alignment between materials, preventing efficient electron–hole pair separation. Additionally, synthesis conditions or improper doping ratios might have resulted in surface defects, recombination centres, or agglomeration, all of which can negatively affect photocatalytic efficiency. These limitations collectively reduced the overall degradation efficiency of composites compared to individual photocatalysts.
3.3 Factors affecting the photocatalytic degradation of EBT
The impact of pH on the photocatalytic degradation performance of the catalyst is fundamentally explained by variations in the electric double layer at the solid-electrolyte interface. This variation in the electric double layer influences the adsorption–desorption process, generating electron–hole pairs on the photocatalyst surface. At low pH values (e.g., pH = 4), the stronger adsorption of dye molecules reduces the number of active sites on the photocatalyst surface, thereby decreasing light absorption and photodegradation. Moreover, strong adsorption leads to the accumulation of multiple layers of dye molecules on the catalyst, hindering direct contact between dye molecules and the catalyst surface. Therefore, they do not participate in photodegradation.94–96
In case of 6% La-doped Ag3PO4/ZnS QD composite, the highest photodegradation efficiency of 88% was observed at pH 4. The higher photocatalytic activity is due to the effective electron–hole pair separation, higher dye adsorption capacity, and suitable band alignment between the 6% La-doped Ag3PO4 and ZnS QDs.
Although degradation curves showed slight overlap for various factors (dye concentration, catalyst dose, pH, and temperature), it can be attributed to minor experimental variations in the sunlight intensity,98 catalyst dispersion,99 and light attenuation by dye molecules and other components in the solution.98 Overall, the trends still clearly demonstrated how each factor affects EBT degradation.
3.4 Kinetic study of the photodegradation process
The kinetic study of photodegradation was conducted using three kinetic models: zero-order, pseudo-first-order, and pseudo-second-order models.In the case of pure Ag3PO4, 2–6% La-doped Ag3PO4, and the Ag3PO4/ZnS quantum dot composite, regression coefficient (R2) values from the pseudo-second-order kinetic model were higher than the regression coefficient values from the zero-order kinetic model and pseudo-first-order kinetic model. The result showed that the degradation of EBT followed second-order kinetics. By contrast, in the case of ZnS quantum dots, the regression coefficient value (R2 = 0.94308) from the pseudo-first-order kinetic model indicated that the degradation of EBT followed the first-order kinetics. In Fig. S8(a–c), the kinetic study of the photodegradation process of EBT for all catalysts is displayed.
3.5 Catalyst reusability
For practical applications, photocatalysts must exhibit not only high catalytic performance but also good stability and reusability. The reusability of the 6% La-doped Ag3PO4 photocatalyst was examined through repeated photocatalytic cycles. After each cycle, the catalyst was recovered, washed, and reused under the same reaction conditions. The results are shown in Fig. 14(a and b). The photocatalytic degradation efficiency of 6% La-silver phosphate is 97%, 91%, 85%, 77%, and 65% in cycles 1, 2, 3, 4, and 5, respectively. The decrease in degradation efficiency may be due to the loss of the photocatalyst during washing between cycles. The SEM images after 5 cycles are shown in Fig. 14(c and d). | ||
| Fig. 14 (a and b) Catalyst reusability for up to 5 cycles using 6% La-Ag3PO4 in a 15 ppm dye solution. (c and d) SEM images after 5 cycles. | ||
Table S3 presents the recently reported efficiencies of various photocatalysts for the degradation of Eriochrome Black T (EBT). 6% La-doped Ag3PO4 and ZnS QDs exhibit superior performance under natural sunlight, achieving 97.84% and 97.59% degradation of EBT, respectively, within 120 minutes. Compared modified efficiency and operational simplicity.
3.6 Mechanism of photocatalytic degradation
Based on the UV-vis diffuse reflectance spectroscopy (DRS) results and photocatalytic degradation performance, a plausible photocatalytic mechanism is proposed. Under sunlight, Ag3PO4 (2.41 eV), 2% La-doped Ag3PO4 (2.39 eV), 6% La-doped Ag3PO4 (2.36 eV), ZnS QDs (3.6 eV), and 6% La-doped Ag3PO4/ZnS QDs (1.5 eV) generate electron–hole (e−/h+) pairs. The reduced band gap of the composite enhances sunlight absorption; however, an excessively low band gap may decrease the redox potential required for efficient photocatalytic reactions.42 The photogenerated electrons are expected to react with dissolved oxygen to produce superoxide radicals (˙O2−), while holes oxidize water to generate hydroxyl radicals (˙OH), which are responsible for the degradation of the dye. These reactive species attack the dye adsorbed on the catalyst surface, breaking it down into simpler, less-toxic molecules.37,100 Nevertheless, interfacial interactions in the composites may promote charge recombination, resulting in lower photocatalytic efficiency compared to the individual components. The chemical reactions involved in the degradation of EBT are presented in Fig. 15(a). The schematic diagram for EBT degradation via photocatalysts is shown in Fig. 15(b). | ||
| Fig. 15 (a) Photocatalytic degradation equations. (b) Schematic of EBT degradation using the proposed photocatalysts. | ||
4 Conclusion
In summary, pure Ag3PO4, La-doped Ag3PO4 (2% and 6%), and ZnS quantum dots were successfully synthesized via a co-precipitation method, while the composites were synthesized through a hybrid mixing method. Different characterization techniques were used to analyse the structural, morphological, and optical properties of the synthesized materials. The XRD results confirmed the successful formation of crystalline Ag3PO4, La-doped Ag3PO4, ZnS quantum dots, and composites. The morphological study of the synthesized materials was performed using SEM micrographs. The elemental composition of the synthesized materials was confirmed by EDS analysis. TGA analysis indicated that all synthesized materials exhibited good thermal stability. UV-DRS results further verified that La incorporation into the Ag3PO4 lattice altered its band gap energy.The photocatalytic capability of the synthesized materials was examined by observing the breakdown of Eriochrome Black T (EBT), an azo dye, under solar illumination. Notably, all synthesized individual materials showed higher degradation efficiency than the composites. This may be due to limitations such as inefficient charge separation and the limited availability of surface active sites on the composites. Additionally, particle clustering or weak interactions between the two components may have reduced the photocatalytic efficiency. Under the optimized reaction conditions, EBT photodegradation efficiencies of 97.84% and 84.88% were achieved using 6% La-doped Ag3PO4 and the 6% La-doped Ag3PO4/ZnS QD composite, respectively, at pH 6, with an initial dye concentration of 15 ppm, a photocatalyst dosage of 0.1 g, and an irradiation time of 120 min. Among the tested materials, 6% La-doped Ag3PO4 showed the highest photocatalytic performance, which can be attributed to enhanced charge carrier utilization and favourable electronic interactions resulting from lanthanum doping. These results indicate that La-modified Ag3PO4-based photocatalysts are promising and sustainable materials for the effective treatment of dye-contaminated wastewater, offering significant potential for environmental remediation applications.
Author contributions
Shabana Bibi: experimental work, writing original draft; Dr Amna Bashir, Dr Noshabah Tabassum: supervision, data analysis, writing, proofreading. The rest of the authors helped in the characterization of materials.Conflicts of interest
There is no conflict of interest.Data availability
Data would be made available on request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d6ra00544f.
Acknowledgements
This research work was supported by the Ministry of Higher Education (MOHE) under the 2023 Translational Research Program for the Energy Sustainability Focus Area (Project ID: MMUE/240001), the 2024 ASEAN IVO (Project ID: 2024-02), and Multimedia University, Malaysia.References
- F. Arias Arias, M. Guevara, T. Tene, P. Angamarca, R. Molina, A. Valarezo, O. Salguero, C. Vacacela Gomez, M. Arias and L. S. Caputi, The adsorption of methylene blue on eco-friendly reduced graphene oxide, Nanomaterials, 2020, 10(4), 681 CrossRef PubMed.
- S. Jayapandi, P. Backialakshmi, P. Soundarrajan, M. Senthil Pandian, P. Ramasamy, S. Suresh Kumar and C. Gopinathan, Construction of p–n junction type Ag2O/SnO2 heterostructure photocatalyst for enhanced organic dye degradation under direct sunlight irradiation: Experimental and theoretical investigations, J. Mater. Res., 2023, 38(3), 753–766 CrossRef CAS.
- M. Khatun, P. Mitra and S. Mukherjee, Effect of band gap and particle size on photocatalytic degradation of NiSnO3 nanopowder for some conventional organic dyes, Hybrid Adv., 2023, 4, 100079 CrossRef.
- T. Naseem and T. Durrani, The role of some important metal oxide nanoparticles for wastewater and antibacterial applications: A review, Environ. Chem. Ecotoxicol., 2021, 3, 59–75 CrossRef CAS.
- M. Al-Ghouti, M. Khraisheh, S. Allen and M. Ahmad, The removal of dyes from textile wastewater: a study of the physical characteristics and adsorption mechanisms of diatomaceous earth, J. Environ. Manag., 2003, 69(3), 229–238 CrossRef CAS PubMed.
- A. Alinsafi, M. Khemis, M.-N. Pons, J.-P. Leclerc, A. Yaacoubi, A. Benhammou and A. Nejmeddine, Electro-coagulation of reactive textile dyes and textile wastewater, Chem. Eng. Process. Process Intensif., 2005, 44(4), 461–470 CrossRef CAS.
- A. Bashir, F. Bashir, Z. Mehmood, M. S. Satti and Z. Akhter, Synthesis, characterisation and investigation of enhanced photocatalytic activity of Sm+ 3, Ni+ 2 co-doped TiO2 nanoparticles on the degradation of azo dyes in visible region, Int. J. Nanoparticles, 2019, 11(1), 37–61 CrossRef CAS.
- R. Rashidi, Y. Omidi Khaniabadi and M. Ghaderpoori, Adsorption of Eriochrome black-T from aqueous environment by raw Montmorillonite, Int. J. Environ. Anal. Chem., 2023, 103(11), 2601–2615 CrossRef CAS.
- N. Barka, M. Abdennouri and M. E. Makhfouk, Removal of Methylene Blue and Eriochrome Black T from aqueous solutions by biosorption on Scolymus hispanicus L.: Kinetics, equilibrium and thermodynamics, J. Taiwan Inst. Chem. Eng., 2011, 42(2), 320–326 CrossRef CAS.
- M. Alamzeb, M. Tullah, S. Ali, Ihsanullah, B. Khan, W. N. Setzer, N. Al-Zaqri and M. N. M. Ibrahim, Kinetic, thermodynamic and adsorption isotherm studies of detoxification of Eriochrome Black T dye from wastewater by native and washed garlic peel, Water, 2022, 14(22), 3713 CrossRef CAS.
- M. T. Yagub, T. K. Sen, S. Afroze and H. M. Ang, Dye and its removal from aqueous solution by adsorption: a review, Adv. Colloid Interface Sci., 2014, 209, 172–184 CrossRef CAS PubMed.
- F. Moeinpour, A. Alimoradi and M. Kazemi, Efficient removal of Eriochrome black-T from aqueous solution using NiFe2O4 magnetic nanoparticles, J. Environ. Health Sci. Eng., 2014, 12(1), 112 CrossRef PubMed.
- A. V. Padalkar and R. Kumar, Common effluent treatment plant (CETP): Reliability analysis and performance evaluation, Water Sci. Eng., 2018, 11(3), 205–213 CrossRef.
- J. Rathore, Studies on pollution load induced by dyeing and printing units in River Bandi at Pali, Rajasthan, India, Int. J. Environ. Sci., 2012, 3(1), 735–742 CAS.
- S. Garcia-Segura, J. D. Ocon and M. N. Chong, Electrochemical oxidation remediation of real wastewater effluents—A review, Process Saf. Environ. Prot., 2018, 113, 48–67 CrossRef CAS.
- E. S. Priya and P. S. Selvan, Water hyacinth (Eichhornia crassipes)–An efficient and economic adsorbent for textile effluent treatment–A review, Arab. J. Chem., 2017, 10, S3548–S3558 CrossRef.
- K. K. Wilson, Assessment of the impact of effluents discharge from Murang'a town and its environs on the water quality of river Mathioya, Murang'a County, Kenya, COPAS, JKUAT, 2017, pp. 1–84 Search PubMed.
- A. Srivastava, N. Verma, A. Mistri, B. Ranjan, A. K. Nigam, U. Kumari, S. Mittal and A. K. Mittal, Alterations in the skin of Labeo rohita exposed to an azo dye, Eriochrome black T: a histopathological and enzyme biochemical investigation, Environ. Sci. Pollut. Res., 2017, 24(9), 8671–8681 CrossRef CAS PubMed.
- M. Al Kausor and D. Chakrabortty, Graphene oxide based semiconductor photocatalysts for degradation of organic dye in waste water: A review on fabrication, performance enhancement and challenges, Inorg. Chem. Commun., 2021, 129, 108630 CrossRef CAS.
- C. M. Simonescu, A. Tătăruş, D. C. Culiţă, N. Stănică, I. A. Ionescu, B. Butoi and A.-M. Banici, Comparative study of CoFe2O4 nanoparticles and CoFe2O4-chitosan composite for Congo red and methyl orange removal by adsorption, Nanomaterials, 2021, 11(3), 711 CrossRef CAS PubMed.
- F. Harrelkas, A. Paulo, M. Alves, L. El Khadir, O. Zahraa, M.-N. Pons and F. Van Der Zee, Photocatalytic and combined anaerobic–photocatalytic treatment of textile dyes, Chemosphere, 2008, 72(11), 1816–1822 CrossRef CAS PubMed.
- Y. Bi, H. Hu, S. Ouyang, G. Lu, J. Cao and J. Ye, Photocatalytic and photoelectric properties of cubic Ag 3 PO 4 sub-microcrystals with sharp corners and edges, Chem. Commun., 2012, 48(31), 3748–3750 RSC.
- L. Dong, P. Wang, S. Wang, P. Lei and Y. Wang, A simple way for Ag3PO4 tetrahedron and tetrapod microcrystals with high visible-light-responsive activity, Mater. Lett., 2014, 134, 158–161 CrossRef CAS.
- J. Cruz-Filho, T. Costa, M. Lima, L. Silva, R. Santos, L. Cavalcante, E. Longo and G. Luz Jr, Effect of different synthesis methods on the morphology, optical behavior, and superior photocatalytic performances of Ag3PO4 sub-microcrystals using white-light-emitting diodes, J. Photochem. Photobiol., A, 2019, 377, 14–25 CrossRef CAS.
- A. Khan, M. Qamar and M. Muneer, Synthesis of highly active visible-light-driven colloidal silver orthophosphate, Chem. Phys. Lett., 2012, 519, 54–58 CrossRef.
- E. Ghazalian, N. Ghasemi and A. Amani-Ghadim, Enhanced visible light photocatalytic performance of Ag3PO4 through doping by different trivalent Lanthanide cations, Mater. Res. Bull., 2017, 88, 23–32 CrossRef CAS.
- M. S. Hussien and I. Yahia, Fabrication progress of selective and durable Ni2+-doped Ag3PO4 for visible-light degradation of various textile dyes, J. Photochem. Photobiol., A, 2019, 368, 210–218 CrossRef CAS.
- S. Zhang, S. Zhang and L. Song, Super-high activity of Bi3+ doped Ag3PO4 and enhanced photocatalytic mechanism, Appl. Catal. B Environ., 2014, 152, 129–139 CrossRef.
- W. Cao, Z. Gui, L. Chen, X. Zhu and Z. Qi, Facile synthesis of sulfate-doped Ag3PO4 with enhanced visible light photocatalystic activity, Appl. Catal. B Environ., 2017, 200, 681–689 CrossRef CAS.
- J. Luo, Y. Luo, Q. Li, J. Yao, G. Duan and X. Liu, Synthesis of doughnut-like carbonate-doped Ag3PO4 with enhanced visible light photocatalytic activity, Colloids Surf., A, 2017, 535, 89–95 CrossRef CAS.
- S. Li, S. Shi, G. Huang, Y. Xiong and S. Liu, Synergetic tuning charge dynamics and potentials of Ag3PO4 photocatalysts with boosting activity and stability by facile in-situ fluorination, Appl. Surf. Sci., 2018, 455, 1137–1149 CrossRef CAS.
- Y. P. Xie and G. S. Wang, Visible light responsive porous Lanthanum-doped Ag3PO4 photocatalyst with high photocatalytic water oxidation activity, J. Colloid Interface Sci., 2014, 430, 1–5 CrossRef CAS PubMed.
- S. Anandan, A. Vinu, K. S. Lovely, N. Gokulakrishnan, P. Srinivasu, T. Mori, V. Murugesan, V. Sivamurugan and K. Ariga, Photocatalytic activity of La-doped ZnO for the degradation of monocrotophos in aqueous suspension, J. Mol. Catal. A: Chem., 2007, 266(1–2), 149–157 CrossRef CAS.
- H. Kato, K. Asakura and A. Kudo, Highly efficient water splitting into H2 and O2 over lanthanum-doped NaTaO3 photocatalysts with high crystallinity and surface nanostructure, J. Am. Chem. Soc., 2003, 125(10), 3082–3089 CrossRef CAS PubMed.
- A. Amirulsyafiee, M. M. Khan, M. Y. Khan, A. Khan and M. H. Harunsani, Visible light active La-doped Ag3PO4 for photocatalytic degradation of dyes and reduction of Cr (VI), Solid State Sci., 2022, 131, 106950 CrossRef CAS.
- B. Wang, Y. Liu, H. Hao, Y. Zhao, Z. He, W. Song, E. Liu, Z. Li and Z. Miao, Alteration of internal electron migration pathways in La-doped Ag 3 PO 4 for improved photocatalytic stability, Mater. Chem. Front., 2025, 9(7), 1189–1204 RSC.
- D. Chen, F. Huang, G. Ren, D. Li, M. Zheng, Y. Wang and Z. Lin, ZnS nano-architectures: photocatalysis, deactivation and regeneration, Nanoscale, 2010, 2(10), 2062–2064 RSC.
- C. Venkata Reddy, N. Bandaru, J. Shim and S. P. Vattikuti, Synthesis of CdO/ZnS heterojunction for photodegradation of organic dye molecules, Appl. Phys. A, 2017, 123(6), 396 CrossRef.
- K. Sharma, P. Raizada, V. Hasija, P. Singh, A. Bajpai, V.-H. Nguyen, S. Rangabhashiyam, P. Kumar, A. K. Nadda and S. Y. Kim, ZnS-based quantum dots as photocatalysts for water purification, J. Water Proc. Eng., 2021, 43, 102217 CrossRef.
- Y. Zhao, X. Wu, H. Wang, M. Ma, J. Tian and X. Wang, Phosphorus regulates coordination number and electronegativity of cobalt atomic sites triggering efficient photocatalytic water splitting, Nano Lett., 2024, 24(50), 16175–16183 CrossRef CAS PubMed.
- C. Xing, J. Ren, L. Fan, J. Zhang, M. Ma, S. Wu, Z. Liu and J. Tian, π-d conjugated copper chloranilate with distorted Cu-O4 site for efficient electrocatalytic ammonia production, Adv. Funct. Mater., 2024, 34(49), 2409064 CrossRef CAS.
- S. M. Yahaya, N. Abdu, I. A. Aliyu and B. Mukhtar, Revolutionizing nitrogen fixation: communicating the potentials of nanostructured photocatalysts for sustainable ammonia/nitrate synthesis, Circ. Agric. Syst., 2024, 4(1), 1–13 Search PubMed.
- S. Kaur, S. Sharma, A. Umar, S. Singh, S. Mehta and S. K. Kansal, Solar light driven enhanced photocatalytic degradation of brilliant green dye based on ZnS quantum dots, Superlattices Microstruct., 2017, 103, 365–375 CrossRef CAS.
- A. Bashir, F. Bashir, M. Sultan, M. Mubeen, A. Iqbal and Z. Akhter, Influence of nickel and lanthanum ions co-doping on photocatalytic properties of TiO2 for effective degradation of reactive yellow 145 in the visible region, J. Sol-Gel Sci. Technol., 2020, 93(2), 438–451 CrossRef CAS.
- R. A. Shathy, S. A. Fahim, M. Sarker, M. S. Quddus, M. Moniruzzaman, S. M. Masum and M. A. I. Molla, Natural sunlight driven photocatalytic removal of toxic textile dyes in water using B-doped ZnO/TiO2 nanocomposites, Catalysts, 2022, 12(3), 308 CrossRef CAS.
- K. Bano, S. K. Mittal, P. P. Singh and S. Kaushal, Sunlight driven photocatalytic degradation of organic pollutants using a MnV 2 O 6/BiVO 4 heterojunction: Mechanistic perception and degradation pathways, Nanoscale Adv., 2021, 3(22), 6446–6458 RSC.
- J. Goyal, S. Sharma and S. Basu, Solar light-induced photocatalytic response of BiOCl/PANI composite towards the degradation of tetracycline, Catalysts, 2023, 13(5), 795 CrossRef CAS.
- H. Yu and M. Obrovac, Quantitative determination of carbon dioxide content in organic electrolytes by infrared spectroscopy, J. Electrochem. Soc., 2019, 166(12), A2467 CrossRef CAS.
- A. B. Alwany, G. Youssef, O. Samir, M. A. Algradee, N. A. A. Yahya, M. A. Swillam, S. Humaidi and R. Abd-Shukor, Annealing temperature effects on the size and band gap of ZnS quantum dots fabricated by co-precipitation technique without capping agent, Sci. Rep., 2023, 13(1), 10314 CrossRef CAS PubMed.
- A. Patterson, The Scherrer formula for X-ray particle size determination, Phys. Rev., 1939, 56(10), 978 CrossRef CAS.
- M. Srivathsa and B. V. Rajendra, Effect of lanthanum doping on the structural, morphological, and optical properties of spray-coated ZnO thin films, Eng. Proc., 2023, 59(1), 32 Search PubMed.
- S.-Y. Ting, P.-J. Chen, H.-C. Wang, C.-H. Liao, W.-M. Chang, Y.-P. Hsieh and C. Yang, Crystallinity improvement of ZnO thin film on different buffer layers grown by MBE, J. Nanomater., 2012, 2012(1), 929278 CrossRef.
- A. A. Khan, A. T. Partho, M. H. Arnab, M. A. Khyam, N. Kumar and M. Tahir, Recent advances in Lanthanum-based photocatalysts with engineering aspects for photocatalytic hydrogen production: A critical review, Mater. Sci. Semicond. Process., 2024, 184, 108809 CrossRef CAS.
- L. Saravanan, S. Diwakar, R. Mohankumar, A. Pandurangan and R. Jayavel, Synthesis, structural and optical properties of PVP encapsulated CdS nanoparticles, Nanomater. Nanotechnol., 2011, 1, 17 CrossRef PubMed.
- Y. Yue, L. Han, B. Ding, Y. Yang, X. Yue, S. Wang, Q. Song and C. Du, Straw-derived activated carbon decorated with Ag3PO4 for organic pollutant removal by a circular degradation mechanism: adsorption and photocatalysis, ACS Omega, 2024, 9(22), 23584–23596 CrossRef CAS PubMed.
- Y. Wang, M. Ding, Z. Li and M. Li, Visible light photocatalytic degradation of dyes by Ag3PO4/g-C3N4/CQDs composite, Surf. Interfaces, 2024, 44, 103585 CrossRef CAS.
- Y. Wang, J. Niu, X. Gao and Y. Zhang, Synergetic tuning of photocatalytic activity and photostability of Ag3PO4 via yttrium doping, carbon quantum dots and BiVO4 for atenolol degradation, Appl. Surf. Sci., 2020, 533, 147458 CrossRef.
- A. Dhupar, S. Kumar, H. S. Tuli, A. K. Sharma, V. Sharma and J. K. Sharma, In-doped ZnS nanoparticles: structural, morphological, optical and antibacterial properties, Appl. Phys. A, 2021, 127(4), 263 CrossRef CAS.
- A. Sudha, I. Manimehan and P. Sakthivel, Structural, morphological, electrochemical and optical properties ZnS quantum dots: Influence of La3+ ions, Results Opt., 2024, 16, 100727 CrossRef.
- P. Sakthivel, A. Krishnamoorthy and I. Devadoss, Influence of Fe3+ ions on the crystallographic, optical and electrochemical performance of ZnS quantum dots, Indian J. Phys., 2024, 98(3), 937–946 CrossRef CAS.
- A. Jain, S. Panwar, T. Kang, H. Jeon, S. Kumar and R. Choubey, Effect of zinc oxide concentration in fluorescent ZnS: Mn/ZnO core–shell nanostructures, J. Mater. Sci.: Mater. Electron., 2014, 25(4), 1716–1723 CrossRef CAS.
- A. B. Alwany, G. Youssef, E. E. Saleh, M. A. Algradee, A. Alnakhlani and B. Hassan, Effect of lead doping on the structural, optical, and radiation shielding parameters of chemically synthesized ZnS nanoparticles, J. Mater. Sci.: Mater. Electron., 2023, 34(3), 233 CrossRef CAS.
- P. Sakthivel and S. Muthukumaran, Crystallographic, structural and band gap tailoring of Zn0. 98Mn0. 02S quantum dots co-doped with Cu by co-precipitation method, J. Inorg. Organomet. Polym. Mater., 2016, 26(3), 563–571 CrossRef CAS.
- T. Watanabe, C. Wada, Y. Iso, T. Isobe and H. Sasaki, Preparation of photostable fluorescent InP/ZnS quantum dots embedded in TMAS-derived silica, ECS J. Solid State Sci. Technol., 2017, 6(7), R75 CrossRef CAS.
- H. Qu, L. Cao, G. Su, W. Liu, R. Gao, C. Xia and J. Qin, Silica-coated ZnS quantum dots as fluorescent probes for the sensitive detection of Pb2+ ions, J. Nanoparticle Res., 2014, 16(12), 2762 CrossRef.
- M. Gordel-Wójcik, M. Malik, A. Siomra, M. Samoć and M. Nyk, Third-order nonlinear optical properties of aqueous silver sulfide quantum dots, J. Phys. Chem. Lett., 2023, 14(49), 11117–11124 CrossRef PubMed.
- H. Agbe, N. Raza, D. Dodoo-Arhin, A. Chauhan and R. V. Kumar, H2O2 rejuvenation-mediated synthesis of stable mixed-morphology Ag3PO4 photocatalysts, Heliyon, 2018, 4(4), 1–25 CrossRef PubMed.
- J. Trajić, R. Kostić, N. Romčević, M. Romčević, M. Mitrić, V. Lazović, P. Balaž and D. Stojanović, Raman spectroscopy of ZnS quantum dots, J. Alloys Compd., 2015, 637, 401–406 CrossRef.
- S. Dhara, A. Arora, J. Ghatak, K. Chen, C. Liu, L. Chen, Y. Tzeng, B. Raj, Deformation potential dominated phonons in ZnS quantum dots, arXiv, preprint arXiv: 0709.1773, 2007 Search PubMed.
- A. Milekhin, N. Yeryukov, L. Sveshnikova, T. Duda, C. Himcinschi, E. Zenkevich and D. Zahn, Resonant Raman scattering of ZnS, ZnO, and ZnS/ZnO core/shell quantum dots, Appl. Phys. A, 2012, 107(2), 275–278 CrossRef CAS.
- S. Mandal, S. I. Ali, S. Pramanik, A. C. Mandal, Impact of capping agent on structural and optical properties of ZnS nanoparticles, arXiv, preprint arXiv: 2304.13420, 2023 Search PubMed.
- B. Choudhury, M. Dey and A. Choudhury, Defect generation, d-d transition, and band gap reduction in Cu-doped TiO2 nanoparticles, Int. Nano Lett., 2013, 3(1), 25 CrossRef.
- G. Botelho, J. Andres, L. Gracia, L. S. Matos and E. Longo, Photoluminescence and photocatalytic properties of Ag3PO4 microcrystals: an experimental and theoretical investigation, ChemPlusChem, 2016, 81(2), 202–212 CrossRef CAS PubMed.
- X. Ma, B. Lu, D. Li, R. Shi, C. Pan and Y. Zhu, Origin of photocatalytic activation of silver orthophosphate from first-principles, J. Phys. Chem. C, 2011, 115(11), 4680–4687 CrossRef CAS.
- S. S. Patil, D. R. Patil, S. K. Apte, M. V. Kulkarni, J. D. Ambekar, C.-J. Park, S. W. Gosavi, S. S. Kolekar and B. B. Kale, Confinement of Ag3PO4 nanoparticles supported by surface plasmon resonance of Ag in glass: efficient nanoscale photocatalyst for solar H2 production from waste H2S, Appl. Catal. B Environ., 2016, 190, 75–84 CrossRef CAS.
- I. Kilic, Y. Bayrak, H. Topallar and B. Karagoz, Preparation of Rice Hull-Based Activated Carbon with Ammonium Nitrate, Asian J. Chem., 2013, 25(9), 4911 CrossRef CAS.
- B. Ju, F. Yang, K. Huang and Y. Wang, Fabrication, characterization and photocatalytic mechanism of a novel Z-scheme BiOBr/Ag3PO4@ rGO composite for enhanced visible light photocatalytic degradation, J. Alloys Compd., 2020, 815, 151886 CrossRef CAS.
- H. Khiar, N. Barka and A. Puga, Metal phosphates for the design of advanced heterogeneous photocatalysts, Coord. Chem. Rev., 2024, 510, 215814 CrossRef CAS.
- S. C. Tudu, T. Sarkar, S. Kundu, G. Ghorai, P. K. Sahoo and A. Bhattacharjee, Synthesis and structural characterization of ZnS quantum dots (< 2 nm) vis-à-vis studies on their spectroscopic and dielectric properties, J. Mater. Sci.: Mater. Electron., 2024, 35(9), 619 CrossRef CAS.
- L. Ju, Z. Chen, L. Fang, W. Dong, F. Zheng and M. Shen, Sol–gel synthesis and photo-Fenton-like catalytic activity of EuFeO3 nanoparticles, J. Am. Ceram. Soc., 2011, 94(10), 3418–3424 CrossRef CAS.
- S.-A. Chang, P.-Y. Wen, T. Wu and Y.-W. Lin, Microwave-assisted synthesis of chalcopyrite/silver phosphate composites with enhanced degradation of Rhodamine B under photo-Fenton process, Nanomaterials, 2020, 10(11), 2300 CrossRef CAS PubMed.
- X. Chen, C. Yu, R. Zhu, N. Li, J. Chen, S. Li, W. Xia, S. Xu, H. Wang and X. Chen, Ag3PO4 deposited on CuBi2O4 to construct Z-scheme photocatalyst with excellent visible-light catalytic performance toward the degradation of diclofenac sodium, Nanomaterials, 2019, 9(7), 959 CrossRef CAS PubMed.
- X. Li, L. Liu, C. Tu, Q. Zhang, X. Yang, D. I. Kolokolov, H. Maltanava, N. Belko, S. Poznyak and M. Samtsov, Zn-BTC MOF as self-template to hierarchical ZnS/NiS2 heterostructure with improved electrochemical performance for hybrid supercapacitor, Nanomaterials, 2023, 14(1), 22 CrossRef CAS PubMed.
- S. Tong, Y. Lin, S. Zhang, J. Nie, C. Luo, Y. Zhang, S. Wu and C. Yang, Performance and mechanisms of photocatalysis of silver phosphate modified by carbon nanosheets doped with both nitrogen and boron for removal of norfloxacin, J. Environ. Chem. Eng., 2023, 11(5), 110595 CrossRef CAS.
- Y. Lee, C. H. Lee, T. Nam, S. Lee, I.-K. Oh, J. Y. Yang, D. W. Choi, C. Yoo, H.-j. Kim and W.-H. Kim, Hydrogen barrier performance of sputtered La2O3 films for InGaZnO thin-film transistor, J. Mater. Sci., 2019, 54(16), 11145–11156 CrossRef CAS.
- P. Nagajyothi, T. Sreekanth, R. Ramaraghavulu, K. Devarayapalli, K. Yoo, S. P. Vattikuti and J. Shim, Photocatalytic dye degradation and hydrogen production activity of Ag3PO4/g-C3N4 nanocatalyst, J. Mater. Sci.: Mater. Electron., 2019, 30(16), 14890–14901 CrossRef CAS.
- X.-F. Tan, S.-S. Zhu, R.-P. Wang, Y.-D. Chen, P.-L. Show, F.-F. Zhang and S.-H. Ho, Role of biochar surface characteristics in the adsorption of aromatic compounds: Pore structure and functional groups, Chin. Chem. Lett., 2021, 32(10), 2939–2946 CrossRef CAS.
- W. Zhang, L. Zhou, J. Shi and H. Deng, Synthesis of Ag3PO4/G-C3N4 composite with enhanced photocatalytic performance for the photodegradation of diclofenac under visible light irradiation, Catalysts, 2018, 8(2), 45 CrossRef.
- K. Kaur, R. Badru, P. P. Singh and S. Kaushal, Photodegradation of organic pollutants using heterojunctions: A review, J. Environ. Chem. Eng., 2020, 8(2), 103666 CrossRef.
- C.-C. Wang, C.-K. Lee, M.-D. Lyu and L.-C. Juang, Photocatalytic degradation of CI Basic Violet 10 using TiO2 catalysts supported by Y zeolite: An investigation of the effects of operational parameters, Dyes Pigm., 2008, 76(3), 817–824 CrossRef CAS.
- Y. A. Shaban, Enhanced Photocatalytic Removal of Methylene Blue From Seawater Under Natural Sunlight Using Carbon-Modified n-TiO^ sub 2^ Nanoparticles, Environ. Pollut., 2014, 3(1), 41 CAS.
- Z. Vasiljevic, M. Dojcinovic, J. Vujancevic, I. Jankovic-Castvan, M. Ognjanovic, N. Tadic, S. Stojadinovic, G. Brankovic and M. Nikolic, Photocatalytic degradation of methylene blue under natural sunlight using iron titanate nanoparticles prepared by a modified sol–gel method, R. Soc. Open Sci., 2020, 7(9), 200708 CrossRef CAS PubMed.
- M. Abdellah, S. Nosier, A. El-Shazly and A. Mubarak, Photocatalytic decolorization of methylene blue using TiO2/UV system enhanced by air sparging, Alex. Eng. J., 2018, 57(4), 3727–3735 CrossRef.
- I. Poulios, A. Avranas, E. Rekliti and A. Zouboulis, Photocatalytic oxidation of Auramine O in the presence of semiconducting oxides, J. Chem. Technol. Biotechnol. Int. Res. Process Environ. Clean Technol., 2000, 75(3), 205–212 CAS.
- A. Bashir, U. Rafique, R. Bashir, S. Jamil, F. Bashir, M. Sultan, M. Mubeen, Z. Mehmood, A. Iqbal and Z. Akhter, Synthesis and comparative evaluation of optical and electrochemical properties of Ni+ 2 and Pr+ 3 ions co-doped mesoporous TiO2 nanoparticles with undoped Titania, Appl. Nanosci., 2021, 11(9), 2397–2413 CrossRef CAS.
- I. Poulios and I. Tsachpinis, Photodegradation of the textile dye Reactive Black 5 in the presence of semiconducting oxides, J. Chem. Technol. Biotechnol. Int. Res. Process Environ. Clean Technol., 1999, 74(4), 349–357 CAS.
- L. Karimi, S. Zohoori and M. E. Yazdanshenas, Photocatalytic degradation of azo dyes in aqueous solutions under UV irradiation using nano-strontium titanate as the nanophotocatalyst, J. Saudi Chem. Soc., 2014, 18(5), 581–588 CrossRef.
- H. D. Tran, D. Q. Nguyen, P. T. Do and U. N. Tran, Kinetics of photocatalytic degradation of organic compounds: a mini-review and new approach, RSC Adv., 2023, 13(25), 16915–16925 RSC.
- L. Y. Kunz, J. Hong, A. R. Riscoe, A. Majumdar and M. Cargnello, Reducing instability in dispersed powder photocatalysis derived from variable dispersion, metallic co-catalyst morphology, and light fluctuations, J. Photochem. Photobiol., 2020, 2, 100004 CrossRef.
- A. Amirulsyafiee, M. M. Khan, M. Y. Khan, A. Khan and M. H. Harunsani, La, Zr co-doped Ag3PO4 for enhanced visible-light photocatalytic degradation of dyes and Cr (VI) photoreduction, Chem. Phys. Impact, 2022, 5, 100102 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |






