Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A divergent synthetic approach to tricyclic Furo[3,2-e]indolizines via base-mediated tandem annulations of diethyl malonate with aromatic bromomethyls
Sandya Tambi Dorai and
Sandeep Chandrashekharappa
and
Sandeep Chandrashekharappa *
*
Department of Medicinal Chemistry, National Institute of Pharmaceutical Education and Research-Raebareli, Transit Campus, Bijnor-Sisendi Road, Sarojini Nagar, Near CRPF Base Camp, Lucknow, UP-226002, India. E-mail: c.sandeep@niperraebareli.edu.in; c.sandeep@niperrbl.ac.in; Fax: +91-522-2975587; Tel: +91-522-2497903
First published on 19th February 2026
Abstract
A base-promoted, transition-metal-free one-pot method for the divergent synthesis of tricyclic furo[3,2-e]indolizines is described. The reaction of 1-(2-oxo-2-phenylethyl)-1H-pyrrole-2-carbaldehyde, diethyl malonate, and aromatic bromomethyl derivatives proceeds through a tandem Knoevenagel–aldol annulation, O-alkylation, and intramolecular cyclization to afford the corresponding furoindolizine products in good to excellent yields. The protocol employs readily accessible starting materials, operates under simple and transition-metal-free conditions, and enables the rapid construction of structurally diverse furo[3,2-e]indolizine frameworks. These features highlight the synthetic efficiency and practical applicability of the approach, making it a valuable platform for heterocyclic synthesis and potential applications in medicinal chemistry.
Introduction
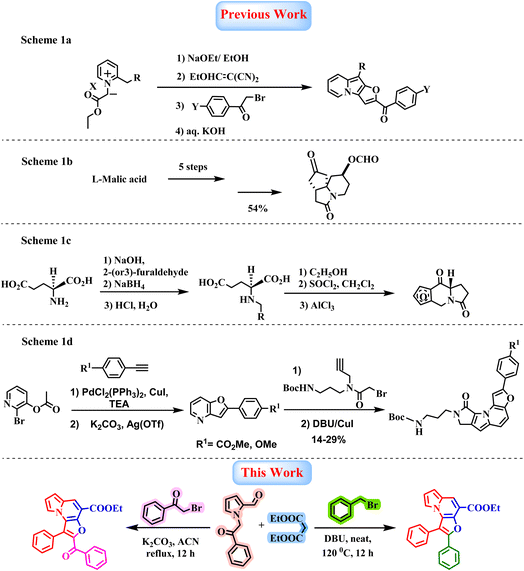
The indolizine scaffold is recognized as a privileged structural motif and it serves as a bioisostere of indole, a well-established pharmacophore which is found in natural products, pharmaceuticals, and clinically approved drugs.1,2 Owing to its structural resemblance to indoles, indolizine has emerged as a promising framework in medicinal chemistry, offering valuable alternatives for drug discovery and synthetic applications.3–7 Indolizine derivatives have attracted significant attention due to their diverse therapeutic potential against cancer, inflammation conditions,8,9 tuberculosis,10,11 and other infectious diseases. Additionally, these compounds exhibit diverse biological and pharmacological properties such as antimicrobial,12 antioxidant,13 antifungal and larvicidal activities,14 and show use as NmeNANAS inhibitors,15 fluorescent biotaggers and probes,16 as represented in Fig. 1. Notably, the indolizine scaffold displays intrinsic fluorescence properties, commonly referred to as Seoul-Fluor.17 Structural modification of substituents and their positions enables fine-tuning of photophysical properties, including emission wavelength and quantum yield. Such variations in structure can influence π-conjugation and intramolecular charge transfer, leading to environment-sensitive fluorescence across the visible spectrum. Its modularity supports bioorthogonal labelling applications, with indolizine-based probes demonstrating strong fluorescence turn-on behaviour in live-cell imaging.16–18Indolizine-fused heterocycles have recently garnered significant attention across diverse research areas, particularly those incorporating a furan moiety due to its inherent fluorescence properties.16–18 The presence of multiple reactive sites within these frameworks renders them highly versatile, enabling extensive structural diversification. As a result, substantial efforts have been directed towards developing efficient synthetic methodologies for their construction. Among these, a notable strategy involves the Michael addition of various vinylating agents to 2(3H)-indolizinones, generated in situ from the corresponding 1-(ethoxycarbonylmethyl)-2-picolinium salts, followed by an alkaline treatment (Scheme 1a).19 While this strategy provides access to indolizine-based architecture, its applicability is limited by the narrow substrate scope of suitable vinylating agents. In parallel, several chiral furoindolizine derivatives have been reported. For instance, a multistep asymmetric synthesis of the furoindolizine framework starting from L-malic acid relies on a key N-acyliminium ion cyclization step (Scheme 1b).20 Similarly, condensation of (S)-glutamic acid with 2-(or 3)-furaldehyde to form Schiff base, followed by sodium borohydride reduction, affords (S)-n-(furylmethyl)-glutamic acid, which undergoes cyclization in ethanol to yield oxoproline. Subsequently, conversion to the corresponding acid chlorides using thionyl chloride and Friedel–Crafts cyclization using aluminium trichloride furnishes the desired furoindolizine product (Scheme 1c).21 Additionally, Lee and co-workers reported a metal-catalyzed coupling strategy for constructing the furan scaffold, which, upon further reaction with tert-butyl (3-(2-bromo-N-(prop-2-yn-1-yl)acetamido)propyl)carbamate, delivers the furo[3,2-e]indolizine core (Scheme 1d).17 Although several approaches for the construction of furoindolizine scaffolds have been reported, many suffer from limitations such as multistep procedures, pre-functionalized substrates, transition-metal catalysts, harsh reaction conditions, limited substrate scope, and lower yields, which restrict their synthetic utility. A key challenge in this area is developing a simple, high-yielding, base catalyzed, transition-metal-free one-pot methodology22–24 that enables the divergent synthesis of tricyclic heterocyclic frameworks is highly desirable. Herein, we report a base-promoted tandem, Knoevenagel–Aldol annulation involving 1-(2-oxo-2-phenylethyl)-1H-pyrrole-2-carbaldehyde and diethyl malonate, followed by O-alkylation of bromomethyl substrates and subsequent intramolecular cyclization. This sequence efficiently furnishes structurally diverse furo[3,2-e]indolizine derivatives under mild conditions, providing a versatile and divergent synthetic methodology (Scheme 1).
Result and discussion
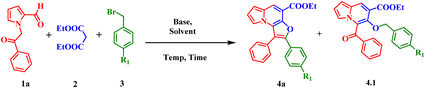
Initially, to identify the optimal reaction conditions, 1-(2-oxo-2-phenylethyl)-1H-pyrrole-2-carbaldehyde (1a), diethyl malonate (2), and 1-bromo-4-(bromomethyl)benzene (3a) were selected as model substrates (Table 1). Preliminary attempts employing the weak organic base triethylamine under neat conditions (110 °C) failed to yield the desired product (Table 1, Entry 1). Substitution with DIPEA likewise did not promote the reaction (Table 1, Entry 2 and Table S1, Entry 4). To enhance reactivity, the base strength was increased by using piperidine. However, conducting the reaction neat at room temperature for 24 h did not yield the target compound; instead, ethyl 5-benzoyl-6-hydroxyindolizine-7-carboxylate25 was obtained (Table S1, Entry 5). Elevating the temperature to 100 °C did not enhance product formation, and most of the starting materials, particularly the benzyl bromide, remained unreacted, with only trace amounts of compound 4.1 detected. Similarly, refluxing the reaction mixture in ethanol proved ineffective (Table S1, Entries 6 and 7). These observations suggested that moderate base strength was insufficient to drive the desired transformation.| Entry | Base (equiv.) | Solvent | Time (h) | Temp. (°C) | R1 | Yieldb % (4.1) | Yieldb % (4a) |
|---|---|---|---|---|---|---|---|
| a Reaction condition: 1a (0.9 mmol), 2 (1.8 mmol), 3a (0.9 mmol), DBU (2.8 mmol), 120 °C, 12 h.b Isolated yields. | |||||||
| 1 | Et3N (3.0) | — | 12 | 110 | Br | — | — |
| 2 | DIPEA (2.0) | — | 24 | 100 | Br | — | — |
| 3 | Piperidine (Cat.) | — | 24 | 100 | Br | Trace | — |
| 4 | K2CO3 (1.0) | CH3CN | 48 | rt | Br | 20 | — |
| 5 | K2CO3 (1.5) | DMF | 12 | 120 | Br | 72 | — |
| 6 | Cs2CO3 (2.0) | CH3CN | 12 | 85 | Br | 78 | — |
| 7 | t-BuOK (2.0) | DMF | 12 | 120 | Br | 67 | — |
| 8 | NaH (2.5) | CH3CN | 12 | 85 | Br | 61 | |
| 9 | DBU (3.0) | CH3CN | 24 | 85 | Br | 68 | — |
| 10 | t-BuOK (4.0) | DMSO | 12 | 120 | NO2 | 52 | Trace |
| 11 | DBU (2.0) | DMF | 24 | 120 | NO2 | — | 20 |
| 12 | DBU (2.5) | DMF | 24 | 120 | NO2 | — | 58 |
| 13 | DBU (3.0) | — | 12 | 120 | NO2 | — | 80 |
| 14 | DBU (3.0) | CH3CN | 24 | 85 | NO2 | — | 72 |
Subsequently, stronger inorganic bases were investigated. Use of potassium carbonate (K2CO3) in a polar aprotic solvent such as acetonitrile resulted in the formation of compound 4.1 in 20% yield after 48 h at room temperature, with no detectable formation of compound 4a. (Table 1, Entry 4). Increasing the base equivalents and screening solvents, including acetonitrile and dimethylformamide, led to improved yields of 4.1, with no observation of 4a (Table S1, Entries 9–11). Further screening of bases such as cesium carbonate, potassium tert-butoxide, sodium hydride, and DBU were carried out in various solvents. However, these conditions did not enhance the formation of compound 4a (Table 1, Entries 6–9 and Table S1, Entries 12–19). Reactions employing benzyl bromides bearing electron-donating substituents and other halogen groups consistently resulted in the formation of O-alkylated indolizine intermediate 4.1.
To promote cyclization, the effect of electron-withdrawing substituents on the benzyl bromide was examined. Benzyl bromides containing nitro and cyano groups were subjected to the reaction under different conditions. Notably, in the presence of potassium tert-butoxide and polar aprotic solvents such as DMF and DMSO, trace amount of the desired cyclized product 4a was detected (Table 1, Entry 10 and Table S1, Entries 20–22), indicating that electron-withdrawing groups promote the cyclization process.
To improve the yield of the cyclized product, stronger bases and reaction parameters were systematically evaluated. Optimal results were obtained using 3.0 equivalents of DBU under neat condition at 120 °C, affording compound 4a in 80% yield (Table 1, Entries 11–14). These results highlight the importance of base strength and electronic effects in facilitating cyclization, and the optimized reaction condition (Table 1, Entry 13) was subsequently taken for further studies.
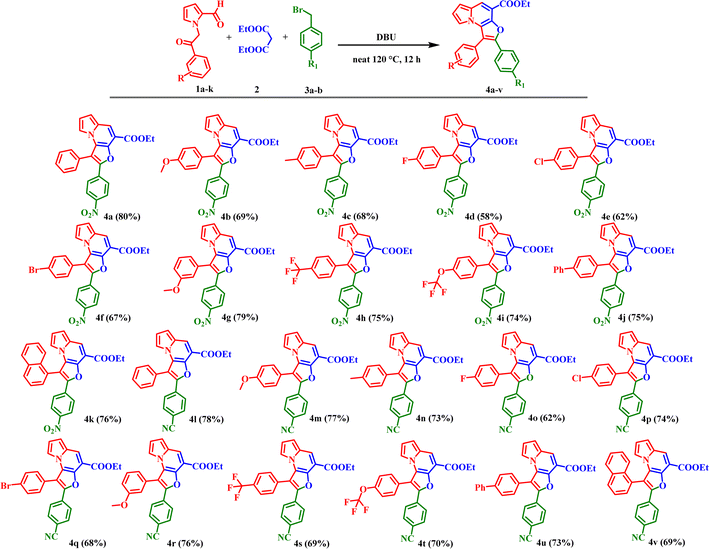
As illustrated in Scheme 2, the substrate scope was explored with respect to the benzoyl group substitutions at the meta-and para-positions of 1-(2-oxo-2-phenylethyl)-1H-pyrrole-2-carbaldehyde (1a–k) in reaction with aromatic bromomethyls (3a–b), affording the corresponding furo[3,2-e]indolizines (4a–4v) in moderate to good yields. Pyrrole substrates containing electron-donating groups (EDGs) on the benzoyl moiety, such as OCH3, CH3, phenyl, and naphthyl substituents, provided the desired products (4b–c, 4j–n, 4u–v) in good yields ranging from 68–77%. Notably, electron-withdrawing groups (EWGs), including Br, Cl, and F, were also well tolerated under the optimized conditions, delivering the corresponding products (4d–f, 4o–q) in moderate yields (58–74%). These results demonstrate the broad functional-group compatibility of the protocol, although EDG-substituted substrates generally afforded slightly higher yields than EWG-substituted substrates.
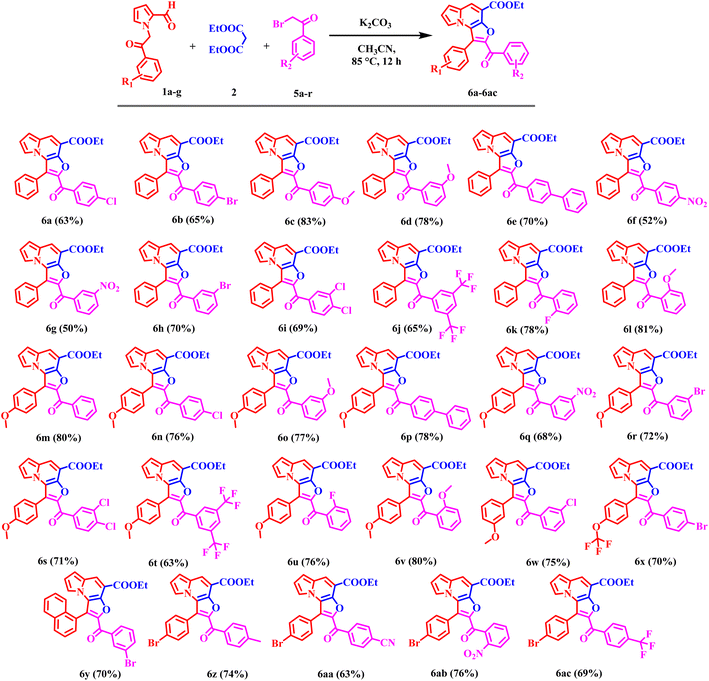
Encouraged by these results, the scope of the tandem Knoevenagel–Aldol annulation was further extended to phenacyl bromide derivatives (5a–r) and pyrrole substituted intermediate (7a–b) as depicted in Schemes 3 and 4. Unlike benzyl bromide substrates, initial reactions using piperidine under neat or ethanol conditions were ineffective, yielding no or only trace amount of product (Table S2, Entries 1–3). This outcome is likely due to insufficient activation of the phenacyl bromide towards C–C bond formation under mild basic conditions. In contrast, the use of K2CO3 in acetonitrile significantly improved the reaction outcome, yielding 20% at room temperature and increasing to 80% at 85 °C (Table S2, Entries 4–6). Other bases, including Cs2CO3, NaH, potassium tert-butoxide, and DBU, yielded only moderate results, likely due to competing side reactions under strongly basic conditions (Table S2, Entries 7–11).
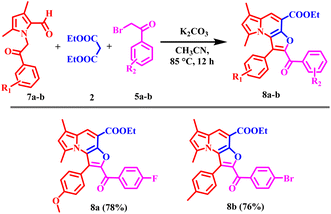 | ||
| Scheme 4 Substrate scope of 6,8-dimethyl furo[3,2-e]indolizine derivatives (8a–b); reaction condition: 7 (0.7 mmol), 2 (1.5 mmol), 5 (0.7 mmol), K2CO3 (1.1 mmol), 85 °C, 12 h, isolated yields. | ||
Overall, K2CO3 in acetonitrile was identified as the optimal condition for phenacyl bromide substrate. Under optimized conditions, phenacyl bromide derivatives bearing substituents at the para-, meta-, and ortho-positions of the phenyl ring yielded the desired products (6a–6ac) in good to excellent yields. Para-substituted electron-donating groups, such as CH3 and OCH3, phenyl provided compounds 6c, 6e, 6p, and 6z in high yield (70–83%). Corresponding meta-substituted derivatives 6d and 6o were obtained in 78% and 77% yields, respectively, while ortho-substituted analogues 6l and 6v furnished the products in similarly high yields (81% and 80%) respectively. In contrast, electron-withdrawing groups including F, Cl, Br, CF3, NO2, and CN at the para-position resulted in comparatively lower yields, ranging from 52–76%. Meta-substituted substrates afforded the corresponding products (6g, 6h, 6q, 6r, 6w, and 6y) in moderate yields (50–70%). While the ortho-substituted derivative 6u was obtained in 76% yield. Disubstituted phenacyl bromides were also compatible under the optimized conditions; 3,5-di-CF3 substituted substrate furnished products 6j and 6t in 65% and 63% yields, respectively, whereas 3,4-dichloro analogue provided products 6i and 6s in 69% and 71% yields. Overall, these results indicate that the electron-donating substituents, particularly at the para position, favor higher reaction efficiency. In contrast, the electron-withdrawing and disubstituted substrates moderately reduce yields, consistent with electronic modulation of the annulation process.
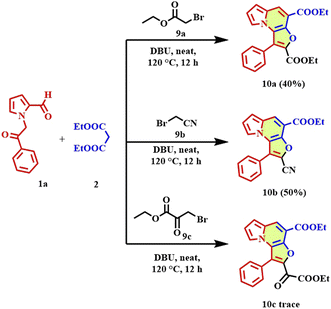
To further assess the scope and applicability of the developed protocol, the reaction was extended to aliphatic bromomethyl derivatives, including ethyl bromoacetate, bromoacetonitrile, and ethyl 3-bromopyruvate. Under the optimized neat conditions in the presence of DBU (3.0 equiv) at 120 °C for 12 h, these substrates successfully underwent the tandem Knoevenagel-O-alkylation-cyclization sequence to afford the corresponding furo[3,2-e]indolizine derivatives (Scheme 5). The reaction with ethyl bromoacetate, bromoacetonitrile, and ethyl 3-bromopyruvate afforded products 10a–b in 40%, 50% respectively, and compound 10c was found in trace amount. The successful incorporation of ester, nitrile functionalities demonstrates the tolerance of the protocol towards diverse aliphatic electrophiles. Importantly, this expanded substrate scope highlights the versatility of the base-mediated, transition-metal-free one-pot strategy.
Plausible mechanism
Based on the preliminary results and the previous literature,25 a plausible mechanism is proposed in Scheme 6. The reaction proceeds through a series of steps, beginning with a base-mediated Knoevenagel condensation reaction between compounds 1 and 2, to form intermediate (A). This intermediate then undergoes cyclization to produce ethyl 5-benzoyl-6-hydroxyindolizine-7-carboxylate (C). Next, the β-carbon, which bears a hydroxyl group and is situated between the ester and ketone functional groups, undergoes deprotonation to generate intermediate (D), which acts as a nucleophile and attacks the alkyl halide, forming the O-alkylated intermediate (E). After alkylation, the base deprotonates the active methylene proton, which then acts on the carbonyl carbon of the benzoyl group, forming intermediate (G). Finally, this intermediate undergoes dehydrogenation to yield compound 4.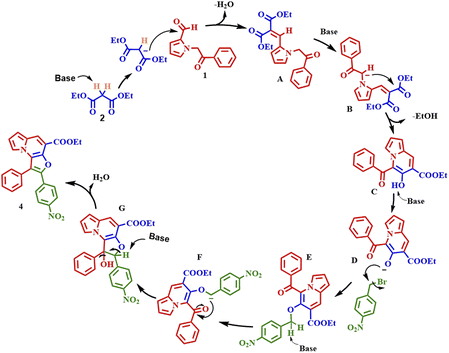 | ||
| Scheme 6 Plausible reaction pathway for the base-mediated formation of furo[3,2-e]indolizines from 1-(2-oxo-2-phenylethyl)-1H-pyrrole-2-carbaldehyde and 4-nitro benzyl bromide. | ||
Conclusion
In summary, an efficient and transition metal-free strategy for the synthesis of furo[3,2-e]indolizine derivatives has been developed. This method employs readily accessible starting materials and proceeds under mild, base-mediated conditions through a tandem Knoevenagel condensation, O-alkylation, and cyclization sequence. A broad range of para-, meta-, and ortho-substituted 1-(2-oxo-2-phenylethyl)-1H-pyrrole-2-carbaldehyde were successfully converted to the corresponding furoindolizine in good to excellent yields. The operational simplicity, metal-free nature, and broad substrate scope highlights the utility of this method for rapid access to structurally diverse furoindolizine frameworks.Conflicts of interest
The authors declare no competing financial interest.Data availability
The authors declare that all the required spectral data available in supporting information (SI). Supplementary information: the synthetic procedure, characterisation details, and spectral information. See DOI: https://doi.org/10.1039/d6ra00540c.Acknowledgements
The authors are thankful to Dr Shubhini A Saraf, Director, NIPER Raebareli, and Dr USN Murty. Former Director, NIPER Raebareli, for their support and encouragement for this research work. The authors are thankful to the Department of Pharmaceuticals, Ministry of Chemicals and fertilizers, and Govt of India for providing fellowship. The authors are also thankful to CIF, and NIPER Raebareli for recording the spectra of synthesized compounds. NIPER-R/Communication/844.References
- A. Gomtsyan, Heterocycles in drugs and drug discovery, Chem. Heterocycl. Compd., 2012, 48, 7–10 CrossRef CAS.
- J. Jampilek, Heterocycles in medicinal chemistry, MDPI, 2019, vol. 24, p 3839 Search PubMed.
- K. M. Dawood and A. A. Abbas, Inhibitory activities of indolizine derivatives: a patent review, Expert Opin. Ther. Pat., 2020, 30, 695–714 CrossRef CAS.
- I. O. Ghinea and R. M. Dinica, Breakthroughs in indole and indolizine chemistry–new synthetic pathways, new applications, in Scope of Selective Heterocycles from Organic and Pharmaceutical Perspective, IntechOpen, 2016 Search PubMed.
- C. Sandeep, K. N. Venugopala, A. Mohammed, A. Mahesh, P. Basavaraj, S. Rashmi, V. Rashmi and B. Odhav, Review on chemistry of natural and synthetic indolizines with their chemical and pharmacological properties, J. Basic Clin. Pharm, 2016, 8, 49–61 Search PubMed.
- K. N. Venugopala, S. Chandrashekharappa, S. Bhandary, D. Chopra, M. A. Khedr, B. E. Aldhubiab, M. Attimarad and B. Odhav, Efficient synthesis and characterization of novel substituted 3-benzoylindolizine analogues via the cyclization of aromatic cycloimmoniumylides with electron-deficient alkenes, Curr. Org. Synth., 2018, 15, 388–395 CrossRef CAS.
- K. Lakshmikanth, S. M. Saini, S. T. Dorai and S. Chandrashekharappa, Tandem-Michael-cyclization cascade to make pyridines: Use of electron-deficient acetylenes for the synthesis of indolizines in aqueous media, Tetrahedron, 2023, 142, 133516 CrossRef CAS.
- K. N. Venugopala, S. Chandrashekharappa, C. Tratrat, P. K. Deb, R. D. Nagdeve, S. K. Nayak, M. A. Morsy, P. Borah, F. M. Mahomoodally and R. P. Mailavaram, Crystallography, molecular modeling, and COX-2 inhibition studies on indolizine derivatives, Molecules, 2021, 26, 3550 CrossRef CAS PubMed.
- H. R. Singh, P. Kushwaha, R. Tandon, N. Srivastava and S. Chandrashekharappa, Synthesis, characterization, and anti-inflammatory properties of novel ethyl 3-benzoyl-7-(trifluoromethyl) indolizine-1-carboxylate derivatives: In silico and in vitro analysis, Chem. Biol. Drug Des., 2024, 103, e14514 CrossRef CAS.
- P. Mundhe, S. Kidwai, S. M. Saini, H. R. Singh, R. Singh and S. Chandrashekharappa, Design, synthesis, characterization, and anti-tubercular activity of novel ethyl-3-benzoyl-6, 8-difluoroindolizine-1-carboxylate analogues: Molecular target identification and molecular docking studies, J. Mol. Struct., 2023, 1284, 135359 CrossRef CAS.
- P. Tiwari, G. S. Mangubhai, S. Kidwai, R. Singh and S. Chandrashekharappa, Design, synthesis and characterization of ethyl 3-benzoyl-7-morpholinoindolizine-1-carboxylate as anti-tubercular agents: In silico screening for possible target identification, Chem. Biol. Drug Des., 2024, 103, e14512 CrossRef CAS PubMed.
- C. Sandeep, P. Basavaraj and S. Rashmi, Synthesis of isomeric subtituted 6-acetyl-3-benzoylindolizine-1-carboxylate and 8-acetyl-3-benzoylindolizine-1-carboxylate from subtituteded 3-acetyl pyridinium bromides and their antimicrobial activity, J. Appl. Chem, 2013, 2, 1049 CAS.
- P. Tiwari, S. Tripathi, R. Ningegowda, G. M. Dhanush, H. Vivek and S. Chandrashekharappa, One-pot synthesis and biological evaluation of substituted 7-chloroindolizines as antimicrobial, antioxidant, and anti-inflammatory agents, Mol. Diversity, 2026, 1–22 Search PubMed.
- S. U. Shende, N. A. Al-Shar’i, S. M. Saini, V. Mohanlall, R. M. Gleiser, P. K. Deb, M. A. Morsy, K. N. Venugopala and S. Chandrashekharappa, Synthesis, characterization and larvicidal studies of ethyl 3-benzoyl-7-(piperidin-1-yl) indolizine-1-carboxylate analogues against Anopheles arabiensis and cheminformatics approaches, J. Biomol. Struct. Dyn., 2024, 1–13 Search PubMed.
- O. I. Alwassil, S. Chandrashekharappa, S. K. Nayak and K. N. Venugopala, Design, synthesis, and structural elucidation of novel NmeNANAS inhibitors for the treatment of meningococcal infection, PLoS One, 2019, 14, e0223413 CrossRef CAS.
- Y. Lee, D. Kim and S. B. Park, Systematic Exploration of Furoindolizine-Based Molecular Frameworks towards a Versatile Fluorescent Platform, Chem. - Eur. J., 2022, 28, e202200533 CrossRef CAS PubMed.
- Y. Lee, A. Jo and S. B. Park, Rational Improvement of Molar Absorptivity Guided by Oscillator Strength: A Case Study with Furoindolizine-Based Core Skeleton, Angew. Chem., Int. Ed., 2015, 54, 15689–15693 Search PubMed.
- E. J. Choi, E. Kim, Y. Lee, A. Jo and S. B. Park, Rational Perturbation of the Fluorescence Quantum Yield in Emission-Tunable and Predictable Fluorophores (Seoul-Fluors) by a Facile Synthetic Method Involving C–H Activation, Angew. Chem., Int. Ed., 2014, 53, 1346–1350 Search PubMed.
- A. Kakehi, S. Ito, T. Ohizumi and M. Ito, Preparation of new nitrogen-bridged heterocycles. 6. First synthesis of 2-aroylfuro (2, 3-b) indolizine derivatives, Bull. Chem. Soc. Jpn., 1983, 56, 1219–1222 Search PubMed.
- Y. S. Lee, J. Y. Lee, D. W. Kim and H. Park, Asymmetric synthesis of furo [3, 2-i] indolizines from L-malic acid, Tetrahedron, 1999, 55, 4631–4636 Search PubMed.
- F. Szemes, Š. Marchalín, N. Bar and B. Decroix, Synthesis of indolizidinediones annelated to a furan ring, J. Heterocycl. Chem., 1998, 35, 1371–1375 CrossRef CAS.
- S. M. Saini, H. R. Singh, R. Katiyar and S. Chandrashekharappa, DDQ-promoted synthesis of 2-benzoylbenzofurans and benzofuro [2, 3-c] pyridines using 2′-hydroxy ethyl cinnamate and phenacyl bromides, RSC Adv., 2026, 16, 3129–3137 RSC.
- S. M. Saini and S. Chandrashekharappa, Metal-free synthesis of tricyclic benzofuro [2, 3-c] pyridin-3-ol derivatives, characterization, and photoluminescence properties, RSC Adv., 2025, 15, 31840–31852 RSC.
- G. Singh, S. Kumar, A. Chowdhury and R. Vijaya Anand, Base-mediated one-pot synthesis of oxygen-based heterocycles from 2-hydroxyphenyl-substituted para-quinone methides, J. Org. Chem., 2019, 84, 15978–15989 CrossRef CAS.
- S. T. Dorai and S. Chandrashekharappa, Tandem Knoevenagel–Aldol Cyclization Towards Di and Trisubstituted Pyridines: Use of Malonaldehyde and Diethyl Malonate for the Synthesis of Indolizine, Eur. J. Org Chem., 2024, 27, e202400906 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |