Open Access Article
Open Access ArticleTheoretical study on the geometry, aromaticity and electronic properties of porphyrin analogues
Xiahe Chena,
Dawei Lia,
Chengwei Zhanga,
Yuanbin She *ab and
Yun-Fang Yang
*ab and
Yun-Fang Yang *ac
*ac
aCollege of Chemical Engineering, Zhejiang University of Technology, Hangzhou, Zhejiang 310014, China. E-mail: sheyb@zjut.edu.cn; yangyf@zjut.edu.cn
bState Key Laboratory of Green Chemical Synthesis and Conversion, Hangzhou 310014, China
cZhejiang Key Laboratory of Surface and Interface Science and Engineering for Catalysts, Hangzhou 310014, China
First published on 13th April 2026
Abstract
Density functional theory calculations were employed to investigate a family of porphyrin analogues differing in macrocyclic size and bridging motif. The geometrical features of these macrocycles exhibit pronounced variations in bond lengths, bond angles, and overall conformations as the framework size and linkage patterns are altered. Nucleus independent chemical shift (NICS) values and anisotropy of the induced current density (AICD) analyses were calculated to evaluate the aromaticity of the porphyrin analogues. The porphyrin analogues exhibit pronounced aromatic character. The electronic properties were further examined by frontier molecular orbital analysis, revealing systematic changes in orbital energies with increasing π-conjugation length and with nitrogen substitution at the meso positions. We expect that these studies will provide new insights and will aid the further development of diverse novel porphyrin analogues.
Introduction
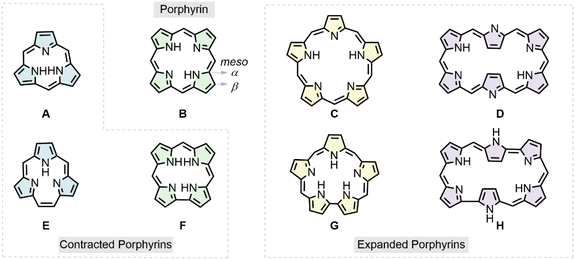
Macrocyclic complexes have attracted considerable attention in bioinorganic chemistry, with porphyrins representing one of the most extensively studied classes. Porphyrins consist of four pyrrolic subunits, in which the pyrrole rings are interconnected through methine carbons at the meso positions.1 In nature, porphyrin frameworks participate in numerous essential biological processes, including photosynthesis in chlorophyll, oxygen transport in hemoglobin, and oxygen storage in myoglobin, earning them the designation “pigments of life”.2 Beyond their biological significance, synthetic porphyrins have also been widely investigated because of their broad applications in diverse fields.3 In addition, recent studies have employed machine-learning approaches to predict properties based on existing porphyrin databases.4Porphyrin macrocycles exhibit a wide variety of structural variants, which can be generated by altering the number of pyrrole rings, substituting pyrrolic nitrogen atoms with other heteroatoms, replacing meso-carbon atoms with heteroatoms, or adding or removing meso-carbons from the macrocycle, among other modifications.5 Representative examples include contracted porphyrins,6 expanded porphyrins,7 and heteroporphyrins8 (Scheme 1). Such structural modifications profoundly influence the intrinsic properties of the macrocycle, including its cavity size, degree of planarity, π-electron configuration, and aromatic character.9 For example, expanded porphyrins possess enlarged ring frameworks that extend the π-conjugated system, resulting in bathochromic shifts in their absorption spectra. The nonlinear optical (NLO) properties of these compounds have been extensively investigated, revealing that their NLO responses and excited-state dynamics are closely related to structural variations and to their aromatic or antiaromatic character, which is influenced by the total number of π-electrons.10 In heteroporphyrin systems, our previous theoretical studies have shown that regular porphyrins and O-substituted porphyrins follow a 22π-electron aromaticity model, whereas heteroporphyrins substituted with PH, AsH, S, Se, Te, CH2, or SiH2 conform to an 18π-[18]annulene aromaticity model.11 The aromaticity of porphyrin analogues has been investigated,12,13 and recent reviews have summarized the methodologies used to assess the aromatic character of expanded porphyrins.14
Although porphyrins themselves have been extensively investigated, the structure–property relationships of their analogues remain insufficiently understood and have not yet been systematically characterized. This makes it difficult to elucidate functional regulatory mechanisms at the molecular level and to further expand their practical applications across diverse fields. To address these issues, the present study provides a detailed theoretical investigation of porphyrin analogues, with the aim of establishing a rational basis for their future molecular design and functional development. The porphyrin analogues considered in this work are illustrated in Scheme 1 and classified according to the number of pyrrole rings and methine carbon atoms. In groups A–D, the number of pyrrole rings increases stepwise from three to six, with adjacent units connected through meso-atom bridges. Groups E–H also contain three to six pyrrole rings but involve the insertion or removal of one or two atoms at the meso positions. In addition, porphyrazine analogues are included in each group, in which the meso-carbons atoms are replaced by nitrogen atoms.
Computational methods
All calculations were performed using the Gaussian 16 package.15 Geometry optimizations and energy calculations were carried out with B3LYP-D3,16 which has been widely applied in computational studies of porphyrin systems.17 The def2-SVP basis set18 was used for all atoms. Frequency analysis was conducted at the same level of theory to verify that the stationary points are minima or saddle points. Computed structures were visualized using CYLview.19 The molecular planarity parameter (MPP)20 was calculated using Multiwfn.21Frontier molecular orbitals (FMOs) and their energies were calculated at the HF/def2-TZVP22 level using the gas-phase optimized geometries. Both nucleus independent chemical shifts (NICS)23 values and anisotropy of the induced current density (AICD)24 analyses were computed at the B3LYP-D3/def2-TZVP level on the same geometries. For NICS calculations of both planar and nonplanar structures, the reference plane was defined by least-squares fitting to the heavy atoms of the macrocycle, and the ring center was determined accordingly. NICS(0) values were obtained at the geometrical center of the heavy atoms, while NICS(1) values correspond to the average of values calculated 1 Å above and below the ring center. Aromatic species exhibit negative NICS values and clockwise induced current densities, whereas antiaromatic species show positive NICS values and counterclockwise currents.
Results and discussion
Geometry of porphyrin analogues
The optimized structures of the corresponding porphyrin analogues A1–D1 are shown in Fig. 1. Across this series, the average bond lengths of the α-Cpyrrole–Cmeso (meso-methine bridges) remain approximately 1.40 Å, whereas the α-Cpyrrole–Cmeso–α-Cpyrrole angles vary significantly. The method for calculating the average bond lengths and bond angles of the porphyrin analogues is illustrated in Fig. S1. The average angle increases from 122° in A1 to 127° in B1 and 135° in C1, indicating a progressive widening of this angle with an increasing number of pyrrole units. However, the average angle in D1 decreases to 129°, deviating from this trend. This difference may arise from variations in the overall molecular conformation, with C1 adopting an approximately circular arrangement and D1 appearing closer to a rectangular geometry. Notably, the geometry of D1 appears more similar to that of B1, which may account for their similar bond angles. An alternative conformation of D1, designated D1-1, was analyzed to assess the effect of molecular geometry. This structure adopts a circular geometry similar to that of C1 and shows an average α-Cpyrrole–Cmeso–α-Cpyrrole angle of 140° (Fig. S2). Computational results indicate that D1 is 26.3 kcal mol−1 lower in energy than D1-1, suggesting that D1 represents the more stable conformation, consistent with experimental observations.25The planarity of these molecules was also evaluated. The molecular planarity parameters (MPP) for A1–D1 are 0.39, 0.00, 0.00, and 0.22 Å, respectively. The ideal bond angle of a sp2-hybridized atom is approximately 120°.26 The average α-Cpyrrole–Cmeso–α-Cpyrrole angle in A1 is close to the ideal value, at 122°. However, steric repulsion from the internal NH protons in its small central cavity forces one pyrrole ring to protrude outside the macrocyclic center, reducing the overall planarity. In B1 and C1, the α-Cpyrrole–Cmeso–α-Cpyrrole angles are slightly larger than 120° and the structures remain relatively stable, possibly due to electronic delocalization. In contrast, two imine-type pyrrole rings in D1 are flipped, with their nitrogen atoms oriented away from the porphyrin core, introducing additional steric strain and further reducing planarity.
For E1–H1 (Fig. 2), the average bond lengths and angles of the α-Cpyrrole–Cmeso–α-Cpyrrole units are comparable to those in A1–D1. In addition, the planarity of the structures shows little variation, except for the tri-pyrrole system. Compared with A1, E1 contains an additional carbon atom inserted between two pyrrole units, which enlarges the central cavity of the macrocycle. At the same time, the number of protonated pyrrolic nitrogen decreases to compensate for the additional meso-carbons, resulting in a higher degree of planarity for E1 (MPP = 0.00 Å) compared with A1 (MPP = 0.39 Å).
The structural effects of different bridging motifs, including meso-methine bridges and aza-N bridges, were further examined in porphyrin analogues. The optimized geometries of the porphyrazine analogues A2–H2, in which nitrogen atoms occupy the meso positions, are shown in Fig. 3. In A2–H2, the average α-Cpyrrole–Nmeso bond length is approximately 1.33 Å, which is shorter than the corresponding α-Cpyrrole–Cmeso bond length in A1–H1 (about 1.40 Å, Fig. 1 and 2). This difference is consistent with the slightly smaller covalent radius of nitrogen compared to carbon (0.71 Å vs. 0.75 Å).27 Similarly, the average α-Cpyrrole–Nmeso–α-Cpyrrole angles in A2–H2 are smaller than the corresponding α-Cpyrrole–Cmeso–α-Cpyrrole angles in A1–H1. These geometric differences affect the overall molecular planarity. Compared to A1–H1, the A2–H2 structures generally show larger MPP values, indicating increased structural distortion.
Among these systems, the structural change is particularly pronounced for C1 and C2: C1 maintains a planar conformation with a MPP value of 0.00 Å (Fig. 1), whereas C2 exhibits the largest MPP of 0.70 Å (Fig. 3). This difference is likely associated with increased ring strain arising from a key structural feature: an aza-N bridge linking the two imine-type pyrrole moieties. Notably, the bond length of the aza-N bridge is shorter than the corresponding meso-methine bridge, leading to steric repulsion between the nitrogen atoms of two imine-type pyrrole rings. To relieve this unfavourable interaction, the fragment undergoes dihedral rotation, causing the two pyrrole rings to deviate from coplanarity and resulting in an overall twisted conformation for C2. To examine this hypothesis, geometry optimizations were carried out on two representative fragments. The structure in which an aza-N bridge connects two imine-type pyrrole rings was defined as fragment 1, whereas the structure in which an meso-methine bridge connects two imine-type pyrrole rings was defined as fragment 2 (Fig. S3). The optimized structures show that the most stable conformation of fragment 1 adopts a dihedral angle N–C–N–C of −69°, whereas fragment 2 remains nearly planar with a corresponding dihedral angle of −1° (Fig. S3). Fragment 1 adopts a −69° dihedral angle, likely in an effort to relieve unfavourable interaction between the nitrogen atoms of two imine-type pyrrole rings. These results indicate that incorporation of the fragment 1 induces pronounced nonplanar distortion, providing an explanation for the markedly reduced planarity observed for C2.
Aromaticity of porphyrin analogues
Studies of aromaticity and antiaromaticity are essential for understanding fundamental molecular properties and reactivity. To evaluate the aromatic character of the porphyrin analogues, NICS values and AICD analyses were calculated. The NICS(1) values at the center of the macrocycle for A1–H1 are shown in Fig. 4 and 5, and the NICS values for individual pyrrole rings are summarized in Table S1 and Table S2. The calculated NICS(1) values for A1, B1, C1, and D1 are −13.11, −13.84, −14.86, and −15.08 ppm, respectively. The gradual increase in the magnitude of the NICS(1) values from A1 to D1 is primarily attributed to the increasing number of π-electrons. Both NICS scans28 and harmonic oscillator model of aromaticity (HOMA)29 values exhibit trends consistent with the NICS(1) results (Fig. 4 and S4). A similar trend is observed for E1–H1, with the NICS(1) values following the order H1 < G1 < F1 < E1. The dominant current pathways, determined from AICD, line integral convolution (LIC),30 and bond current strength (BCS)31 analyses (Fig. S5–S6) and indicated by red lines, are shown in Fig. 4 and 5. AICD and LIC plots show that all porphyrin analogues from A1 to H1 exhibit a clockwise ring current. This indicates aromatic character, in good agreement with the NICS results.A correlation between structural planarity and aromaticity is observed in most systems, with more planar structures generally exhibiting stronger aromatic character (Fig. S7). For example, in the tetra-pyrrole systems, B1 and F1 display MPP values of 0.00 and 0.15 Å, respectively, accompanied by NICS(1) values of −13.84 and −12.49 ppm (Fig. 1, 2, 4 and 5).
Although E1 is more planar than A1, with MPP values of 0.00 and 0.39 Å, respectively, its aromaticity is slightly weaker, as indicated by NICS(1) values of −12.21 ppm for E1 and −13.11 ppm for A1. This behavior may be partly explained by the bowl-shaped conformation of A1. As reported by Casabianca, molecular curvature can influence aromaticity.32 In A1, the NICS values on the concave and convex sides are −16.34 and −9.88 ppm, respectively (Fig. S8). Compared with the planar structure E1, the enhancement of the concave side NICS value in A1 is more pronounced, ultimately leading to a more negative overall NICS(1) value. In the hexa-pyrrole systems, D1 and H1 exhibit significant NICS(1) values of −15.08 and −16.86 ppm, with corresponding MPP values of 0.22 and 0.36 Å, respectively. This result may be related to the placement of the NICS probe (bq), with NICS values calculated at the ring centers defined as the nonweighted mean of the heavy atom coordinates. In the NICS calculations for D1 and H1, the NICS probe is positioned close to the Cβ atoms of the two flipped pyrrole rings. This placement may have a considerable impact on the calculated NICS values. NICS scans show that when the probe is placed more than 1.4 Å from the ring center, the NICS value of D1 becomes more negative than that of H1, indicating relatively stronger aromaticity for D1. (Fig. S7).
The porphyrazine analogues with nitrogen at the meso positions exhibit NICS(1) values in the range of −17.00 to −10.00 ppm (Table S1), comparable to those of the A1–H1 series of porphyrin analogues, suggesting that their overall aromatic character remains largely unchanged.
HOMO–LUMO gap of porphyrin analogues
To better understand the influence of structural variations on the electronic properties of these porphyrin analogues, the HOMO–LUMO energy gaps were analyzed, which serve as important indicators of molecular conjugation, optical properties, and electronic stability.5d The spatial distributions of the frontier molecular orbitals for porphyrin analogues A1–D1 are depicted in Fig. 6. For A1–D1, the HOMOs are predominantly localized on the pyrrole carbon atoms, corresponding to a1u-type orbitals whereas the LUMOs are mainly distributed over the whole π-conjugated backbone, corresponding to eg-type orbitals.33 The calculated HOMO–LUMO energy gaps for A1–D1 are 7.42, 6.25, 5.32, and 4.59 eV, respectively. As the π-conjugation pathways become longer from A1 to D1, the HOMO–LUMO energy gap decreases. Extension of the π-conjugated system affects both the HOMO and LUMO energy levels, resulting in an increase in the HOMO energy and a decrease in the LUMO energy, with the effect being more pronounced for the LUMO. A clear decrease in the HOMO–LUMO energy gap is also observed from E1 to H1. HOMO–LUMO energy gap calculations performed with other functionals exhibit the same trend (Table S3). | ||
| Fig. 6 Molecular orbital distributions and diagrams of the frontier molecular orbitals for A1–H1. The energies are shown in eV. | ||
Compared with porphyrin analogues with carbon at the meso positions, porphyrazine analogues, in which the meso-carbons are replaced by nitrogen, generally exhibit a smaller HOMO–LUMO gap (Fig. 7 and Table S4). In most cases, the narrowing of the HOMO–LUMO gap is primarily associated with stabilization of the LUMO. For instance, the HOMO–LUMO gap decreases from 6.25 eV in B1 to 5.45 eV in B2 (Table S4). This reduction is primarily due to a 0.81 eV lowering of the LUMO energy upon substitution of meso-carbon atoms by nitrogen, which is likely related to the higher electronegativity of nitrogen (3.04) compared with carbon (2.55).5b In contrast, the HOMO energy remains largely unchanged, as the contribution of the meso positions to the HOMO orbital is negligible.
Conclusions
In this study, the structural, aromatic, and electronic properties of a series of porphyrin analogues were systematically investigated. Structural analyses reveal that variations in the number of pyrrole units within the macrocyclic framework, as well as differences in bridging motifs, have a pronounced influence on molecular planarity. In particular, incorporation of aza-N bridges can introduce local steric strain, leading to significant nonplanar distortion in certain analogues. Aromaticity analyses based on NICS and AICD calculations indicate that more planar structures generally exhibit stronger aromatic character, whereas deviations from planarity can noticeably affect aromaticity. Increasing π-conjugation in porphyrin analogues generally raises the HOMO energy and lowers the LUMO energy, resulting in a smaller HOMO–LUMO gap. In addition, substitution of meso-carbon atoms by nitrogen typically leads to smaller HOMO–LUMO gaps compared with porphyrin analogues containing meso-carbon atoms. We hope that these findings offer guidance for the rational design of functional porphyrin-like systems.Author contributions
Xiahe Chen: calculation, data curation, and writing – original draft. Dawei Li: calculation, data curation, and writing – original draft. Chengwei Zhang: calculation. Yuanbin She: conceptualization, and project administration. Yun-Fang Yang: conceptualization, supervision, writing – review & editing, and funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementry information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6ra00486e.Acknowledgements
This work was also supported by the National Natural Science Foundation of China (22371256 and 22138011), the Zhejiang Provincial Natural Science Foundation of China (LR25B020002), the Fundamental Research Funds for the Provincial Universities of Zhejiang (RF-C2022006).Notes and references
- (a) S. Hiroto, Y. Miyake and H. Shinokubo, Chem. Rev., 2017, 117, 2910–3043 CrossRef CAS; (b) B. Sekaran and R. Misra, Coord. Chem. Rev., 2022, 453, 214312 CrossRef CAS.
- (a) A. R. Battersby, C. J. R. Fookes, G. W. J. Matcham and E. McDonald, Nature, 1980, 285, 17–21 CrossRef CAS; (b) M. Taniguchi and J. S. Lindsey, Chem. Rev., 2017, 117, 344–535 CrossRef CAS PubMed.
- (a) C.-M. Che, V. K.-Y. Lo, C.-Y. Zhou and J.-S. Huang, Chem. Soc. Rev., 2011, 40, 1950–1975 RSC; (b) R. Paolesse, S. Nardis, D. Monti, M. Stefanelli and C. Di Natale, Chem. Rev., 2017, 117, 2517–2583 CrossRef CAS; (c) F. Luo, L. Liu, H. Wu, L. Xu, Y. Rao, M. Zhou, A. Osuka and J. Song, Nat. Commun., 2023, 14, 5028 CrossRef CAS PubMed; (d) X. Chen, Y.-F. Yang and Y. She, Org. Chem. Front., 2024, 11, 1039–1049 RSC.
- (a) A. Su, C. Zhang, Y.-B. She and Y.-F. Yang, Catalysts, 2022, 12, 1485 CrossRef CAS; (b) A. Su, X. Zhang, C. Zhang, D. Ding, Y.-F. Yang, K. Wang and Y.-B. She, Phys. Chem. Chem. Phys., 2023, 25, 10536–10549 RSC; (c) S. Fite and Z. Gross, J. Chem. Inf. Model., 2025, 65, 4403–4411 CrossRef CAS.
- (a) B. Szyszko and L. Latos-Grażyński, Chem. Soc. Rev., 2015, 44, 3588–3616 RSC; (b) Y. Matano, Chem. Rev., 2017, 117, 3138–3191 CrossRef CAS PubMed; (c) T. Sarma and P. K. Panda, Chem. Rev., 2017, 117, 2785–2838 CrossRef CAS; (d) J. Mack, Chem. Rev., 2017, 117, 3444–3478 CrossRef CAS.
- (a) C. G. Claessens, D. González-Rodríguez, M. S. Rodríguez-Morgade, A. Medina and T. Torres, Chem. Rev., 2014, 114, 2192–2277 CrossRef CAS; (b) S. Shimizu and N. Kobayashi, Chem. Commun., 2014, 50, 6949–6966 RSC; (c) S. Shimizu, Chem. Rev., 2017, 117, 2730–2784 CrossRef CAS; (d) G. Lavarda, J. Labella, M. V. Martínez-Díaz, M. S. Rodríguez-Morgade, A. Osuka and T. Torres, Chem. Soc. Rev., 2022, 51, 9482–9619 RSC.
- (a) B. Szyszko, M. J. Białek, E. Pacholska-Dudziak and L. Latos-Grażyński, Chem. Rev., 2017, 117, 2839–2909 CrossRef CAS; (b) T. Tanaka and A. Osuka, Chem. Rev., 2017, 117, 2584–2640 CrossRef CAS PubMed; (c) T. Chatterjee, A. Srinivasan, M. Ravikanth and T. K. Chandrashekar, Chem. Rev., 2017, 117, 3329–3376 CrossRef CAS PubMed; (d) Y. Rao, J. Lee, J. Chen, L. Xu, M. Zhou, B. Yin, J. Kim, A. Osuka and J. Song, Angew. Chem., Int. Ed., 2024, 63, e202409655 CrossRef CAS.
- (a) T. Chatterjee, V. S. Shetti, R. Sharma and M. Ravikanth, Chem. Rev., 2017, 117, 3254–3328 CrossRef CAS; (b) R. Sengupta, M. Ravikanth and T. K. Chandrashekar, Chem. Soc. Rev., 2021, 50, 13268–13320 RSC.
- (a) M. Stępień, N. Sprutta and L. Latos-Grażyński, Angew. Chem., Int. Ed., 2011, 50, 4288–4340 CrossRef PubMed; (b) M. Pawlicki and L. Latos-Grażyński, Chem.–Asian J., 2015, 10, 1438–1451 CrossRef CAS PubMed; (c) D. W. Thuita and C. Brückner, Chem. Rev., 2022, 122, 7990–805 CrossRef CAS PubMed; (d) K. Watanabe, N. N. Pati and Y. Inokuma, Chem. Sci., 2024, 15, 6994–7009 RSC.
- (a) M. Yahya, Y. Nural and Z. Seferoğlu, Dyes Pigm., 2022, 198, 109960 CrossRef CAS; (b) E. Desmedt, D. Smets, T. Woller, M. Alonso and F. De Vleeschouwer, Phys. Chem. Chem. Phys., 2023, 25, 17128–17142 RSC; (c) E. Desmedt, M. Jacobs, M. Alonso and F. De Vleeschouwer, Phys. Chem. Chem. Phys., 2025, 27, 1256–1273 RSC.
- X. Fang, X. Chen, Q. Wang, Y.-F. Yang and Y.-B. She, Org. Biomol. Chem., 2020, 18, 4415–4422 RSC.
- (a) J.-i. Aihara, J. Phys. Chem. A, 2008, 112, 5305–5311 CrossRef CAS PubMed; (b) H. Fliegl, D. Sundholm, S. Taubert and F. Pichierri, J. Phys. Chem. A, 2010, 114, 7153–7161 CrossRef CAS PubMed; (c) M. Bröring, Angew. Chem., Int. Ed., 2011, 50, 2436–2438 CrossRef.
- (a) R. R. Valiev, I. Benkyi, Y. V. Konyshev, H. Fliegl and D. Sundholm, J. Phys. Chem. A, 2018, 122, 4756–4767 CrossRef CAS PubMed; (b) R. Pino-Rios, G. Cárdenas-Jirón and W. Tiznado, Phys. Chem. Chem. Phys., 2020, 22, 21267–21274 RSC; (c) I. Casademont-Reig, T. Woller, V. García, J. Contreras-García, W. Tiznado, M. Torrent-Sucarrat, E. Matito and M. Alonso, Chem. Eur. J., 2023, 29, e202202264 CrossRef CAS PubMed.
- F. De Vleeschouwer, E. Desmedt and M. Alonso, Chem. Methods, 2025, 5, e202500064 CrossRef CAS.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian 16, Revision C.01, Gaussian, Inc., Wallingford CT, 2019 Search PubMed.
- (a) S. Grimme, J. Antony, S. Ehrlich and H. Krieg, J. Chem. Phys., 2010, 132, 154104 CrossRef; (b) S. Grimme, S. Ehrlich and L. Goerigk, J. Comput. Chem., 2011, 32, 1456–1465 CrossRef CAS; (c) J. Witte, N. Mardirossian, J. B. Neaton and M. Head Gordon, J. Chem. Theory Comput., 2017, 13, 2043–2052 CrossRef CAS.
- (a) D. A. Sharon, D. Mallick, B. Wang and S. Shaik, J. Am. Chem. Soc., 2016, 138, 9597–9610 CrossRef CAS PubMed; (b) D. Mallick and S. Shaik, J. Am. Chem. Soc., 2017, 139, 11451–11459 CrossRef CAS PubMed.
- F. Weigend and R. Ahlrichs, Phys. Chem. Chem. Phys., 2005, 7, 3297–3305 RSC.
- C. Y. Legault, CYLview, 1.0b, Université de Sherbrooke, Canada, 2009, https://www.cylview.org Search PubMed.
- T. Lu, J. Mol. Model., 2021, 27, 263 CrossRef CAS.
- T. Lu and F. Chen, J. Comput. Chem., 2012, 33, 580–592 CrossRef CAS PubMed.
- F. Weigend, Phys. Chem. Chem. Phys., 2006, 8, 1057–1065 RSC.
- (a) P. v. R. Schleyer, C. Maerker, A. Dransfeld, H. Jiao and N. J. R. van Eikema Hommes, J. Am. Chem. Soc., 1996, 118, 6317–6318 CrossRef CAS; (b) Z. Chen, C. S. Wannere, C. Corminboeuf, R. Puchta and P. v. R. Schleyer, Chem. Rev., 2005, 105, 3842–3888 CrossRef CAS; (c) T. Stuyver, M. Perrin, P. Geerlings, F. De Proft and M. Alonso, J. Am. Chem. Soc., 2018, 140, 1313–1326 CrossRef CAS PubMed.
- (a) R. Herges and D. Geuenich, J. Phys. Chem. A, 2001, 105, 3214–3220 CrossRef CAS; (b) D. Geuenich, K. Hess, F. Köhler and R. Herges, Chem. Rev., 2005, 105, 3758–3772 CrossRef CAS.
- (a) L. Simkhovich, I. Goldberg and Z. Gross, Org. Lett., 2003, 5, 1241–1244 CrossRef CAS PubMed; (b) M. Suzuki and A. Osuka, Org. Lett., 2003, 5, 3943–3946 CrossRef CAS PubMed.
- M. Diociaiuti, S. Casciardi and R. Sisto, Micron, 2016, 90, 97–107 CrossRef CAS PubMed.
- P. Pyykkö and M. Atsumi, Chem. Eur. J., 2009, 15, 186–197 CrossRef.
- (a) A. Stanger, J. Org. Chem., 2006, 71, 883–893 CrossRef CAS PubMed; (b) A. Espinosa Ferao, Inorg. Chem., 2025, 64, 11832–11844 CrossRef CAS.
- (a) J. Kruszewski and T. M. Krygowski, Tetrahedron Lett., 1972, 13, 3839–3842 CrossRef; (b) T. M. Krygowski, J. Chem. Inf. Comput. Sci., 1993, 33, 70–78 CrossRef CAS.
- (a) J. Jusélius, D. Sundholm and J. Gauss, J. Chem. Phys., 2004, 121, 3952–3963 CrossRef; (b) H. Fliegl, S. Taubert, O. Lehtonen and D. Sundholm, Phys. Chem. Chem. Phys., 2011, 13, 20500–20518 RSC.
- G. Monaco, F. F. Summa and R. Zanasi, J. Chem. Inf. Model., 2021, 61, 270–283 CrossRef CAS PubMed.
- L. B. Casabianca, J. Phys. Chem. A, 2016, 120, 7011–7019 CrossRef CAS.
- (a) S. I. Yang, J. Seth, T. Balasubramanian, D. Kim, J. S. Lindsey, D. Holten and D. F. Bocian, J. Am. Chem. Soc., 1999, 121, 4008–4018 CrossRef CAS; (b) R.-J. Cheng, P.-Y. Chen, T. Lovell, T. Liu, L. Noodleman and D. A. Case, J. Am. Chem. Soc., 2003, 125, 6774–6783 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |