 Open Access Article
Open Access ArticleMagnetic nanocrystalline cellulose-poly(methacrylic acid-co-acrylonitrile) composite: a versatile material for analytical and biological applications
Ali Khanab,
Hamayun Khan*a,
Nasrullah Shah *c,
Mahmoud Elodemi
*c,
Mahmoud Elodemi d,
S. Tasqeeruddine,
Ajmal Khan*fg and
Ahmed Al-Harrasi
d,
S. Tasqeeruddine,
Ajmal Khan*fg and
Ahmed Al-Harrasi *f
*f
aDepartment of Chemistry, Islamia College University, Peshawar 25120, Pakistan. E-mail: hamayun84@yahoo.com
bDepartment of Chemical Engineering, Bay Campus, Swansea University, Swansea, SA1 8EN, UK
cDepartment of Chemistry, Abdul Wali Khan University, Mardan 23200, Pakistan. E-mail: nasrullah@awkum.edu.pk
dDepartment of Pharmacology, Faculty of Medicine, University of Tabuk, Tabuk 71491, Saudi Arabia
eDepartment of Pharmacognosy, College of Pharmacy, King Khalid University, Asir 61441, Saudi Arabia
fNatural and Medical Sciences Research Center, University of Nizwa, PO Box 33, 616 Birkat Al Mauz, Nizwa, Oman. E-mail: ajmalchemist@yahoo.com; aharrasi@unizwa.edu.om
gDepartment of Chemical and Biological Engineering, College of Engineering, Korea University, Seoul 02841, Republic of Korea
First published on 23rd April 2026
Abstract
Developing new materials for advanced analytical and biological applications is always a key target in the field of advanced materials. In the present study, a poly(methacrylic acid-co-acrylonitrile)-grafted-magnetic nanocrystalline cellulose composite [P(MAA-co-AN)-g-MNCC] was synthesized through a modified free-radical grafting co-polymerization method using cellulose recovered from waste paper. The synthesized composite was characterized via FTIR spectroscopy, SEM, EDX, and TGA, and its Eg, PZC, and surface area were determined. The as-synthesized P(MAA-co-AN)-g-MNCC was studied as an adsorbent for the removal of gallic acid (GA) from aqueous environments and as an antimicrobial and antioxidant agent. The influence of various experimental factors, including contact time, pH, temperature, and GA concentration, was investigated to optimize the adsorption process. The adsorption process follows pseudo-second-order kinetics and the Freundlich model with a spontaneous and exothermic nature. Similarly, the synthesized adsorbent maintained its excellent adsorption capacity when it was recycled and reused for four successive cycles in real samples. The synthesized P(MAA-co-AN)-g-MNCC also displayed good antioxidant and antibacterial activities against the tested pathogenic bacteria. The results indicated that the as-synthesized P(MAA-co-AN)-g-MNCC has the potential to be used as an effective advanced material for wastewater treatment and biomedical applications.
1. Introduction
In the field of material science, chemistry, medicine, and biology, magnetic nanoparticles (MNPs) have significant importance due to their notable properties such as biocompatibility, non-toxicity, and cost-effective synthesis.1,2 MNPs (magnetite Fe3O4 and maghemite γ-Fe2O3) show superparamagnetic properties and a high surface area-to-volume ratio, which endow them with the ability to carry, bind, and absorb various compounds like small drug molecules, proteins, DNA, RNA, and probes with remarkable efficiency.3–11 Therefore, MNPs have attracted global attention, especially in the field of biomedical applications, such as cancer treatment, drug delivery, magnetic hyperthermia, and MRI.12–17 However, their hydrophobic nature and the presence of van der Waals/electrostatic forces cause them to agglomerate, which restricts their applications.18,19 Therefore, to avoid agglomeration, MNPs are coated with suitable inorganic/organic materials such as natural polysaccharides. To improve the chemical and thermal stability, biocompatibility, and biodegradability of synthesized MNPs, various polysaccharides, including chitosan, agar, carboxymethyl cellulose sodium, and carboxymethyl chitosan, are employed as stabilizers during their synthesis.2,5,16,20Polysaccharide-based materials, especially the cellulose-based magnetic composites, are suitable for biomedical and environmental applications. However, the poor mechanical stability, high sensitivity to microbial attack, low sorption capacity, and uncontrolled water uptake of cellulose limit its application as a substrate for highly specific adsorbents.21–23 In this case, its amorphous part can be removed to overcome these restrictions. For this purpose, nanocrystalline cellulose (NC) is produced using concentrated H2SO4, which has a highly crystalline nature and excellent mechanical properties and offers maximum stability in solutions by preventing agglomeration. Graft copolymerization using vinyl monomers can further improve the dispersibility of NC in various solvents; furthermore, the addition of ionizable and hydrophobic functional groups to composite materials makes them pH-sensitive.24
Fe3O4 nanoparticles are incorporated into composite materials to impart magnetic responsiveness, enabling the rapid magnetic separation and efficient recovery of the adsorbent after analyte adsorption, thereby enhancing its reusability and practical applicability.25,26 Cellulose acts as a hydroxyl-rich support that improves the grafting efficiency and structural stability while contributing to adsorption via hydrogen bonding interactions with gallic acid (GA).27,28 Alternative hydroxyl-containing substrates may provide similar active sites; however, variations in functional group density and physicochemical properties could influence their adsorption performance and recyclability.
GA is a phenolic bioactive compound with a molecular mass of 170.12 mg L−1 and low water solubility.29,30 GA has a wide range of applications, including anti-metastatic, antioxidant, anti-allergic, and anti-mutagenic agents. It is also utilized as a raw material in the manufacturing of imaging agents, dyes, inks, paints, foods, household chemicals, cosmetics, and pharmaceutical drugs.31–36 A significant amount of GA is released into wastewater during the manufacturing process, which can have serious environmental consequences. The ecological impact of GA is concentration dependent. While it is generally considered safe and biodegradable at low concentrations, studies indicate that levels exceeding 200 mg L−1, and particularly 500 mg L−1, exhibit inhibitory effects on microbial populations and aquatic organisms due to its pro-oxidant potential and high chemical oxygen demand.37,38
For the isolation and degradation of GA from aqueous solution, different methods have been used, such as electrochemical oxidation, adsorption, ozonation treatment, UV/H2O2 treatment, biological degradation, and extraction.29,36–40 However, these methods have benefits and drawbacks. Among them, the adsorption approach is the most popular and widely used for removing GA from aqueous environments due to its low cost and high efficiency. Various adsorbents, including biomass, resin, activated carbon, and magnetite nanoparticles, have been used to remove GA from the aqueous environment. Herein, we illustrate the synthesis of novel water-insoluble P(MAA-co-AN)-g-MNCC as a magnetically functionalized crosslinked polymer network. The pH sensitivity of the synthesized adsorbent was enhanced due to the presence of highly ionizable functionalities such as –COOH, and our goal was to utilize these functionalities for the removal of GA from an aqueous environment using the newly synthesized adsorbent under optimized conditions and determine its potential for biological applications. We previously reported the synthesis of chitosan-grafted terpolymer hydrogels for controlled drug delivery applications.41 The synthetic procedure in this study differs fundamentally from our previous report. The current approach emphasizes a sustainable waste-to-material strategy by using waste-paper-derived nanocrystalline cellulose as the primary backbone. Furthermore, the integration of acrylonitrile (AN) provides a unique nitrogenous functionality, shifting the application from general drug delivery toward targeted removal and enhanced antibacterial activity.
2. Experimental
2.1. Materials
Analytical grade chemicals and reagents were used. Methacrylic acid (MAA), acrylonitrile (AN), potassium disulfate (K2S2O7), and ethylene glycol dimethacrylate (EDGMA) were purchased from Sigma-Aldrich, USA. Iron(III) chloride hexahydrate (FeCl3·6H2O), iron(II) chloride tetrahydrate (FeCl2·4H2O), H2SO4 (98%), NaOH, and gallic acid (GA) were supplied by Merck KGaA, Darmstadt, Germany. Furthermore, Luria–Bertani (LB) agar was obtained from Basingstoke, Hampshire, England. Petri dishes were obtained from Sigma-Aldrich, and pathogenic strains, including Escherichia coli (E. coli) and Staphylococcus aureus (S. aureus), and the positive control ceftriaxone were sourced from the Microbiology Department, Abdul Wali Khan University, Mardan (AWKUM), Pakistan. In the experiment, waste copy paper (A4 Printing Paper, Trade International, Pakistan) was used to extract cellulose, and deionized double-distilled water (DDDW) was utilized.2.2. Synthesis of P(MAA-co-AN)-g-MNCC
P(MAA-co-AN)-g-MNCC was prepared via the four steps depicted in Fig. 1. Briefly, Fe2+/Fe3+ salts were co-precipitated under alkaline conditions in the presence of cellulose, leading to the in situ formation and deposition of Fe3O4 nanoparticles on the cellulose surface. Then, the polymerization proceeds via a free-radical polymerization mechanism, which is initiated by the initiator system. The vinyl group (–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–) of methacrylic acid participates in radical polymerization, forming the polymer backbone. The –COOH groups do not react directly with the –OH groups of cellulose under these conditions because no esterification catalyst or activation step was used. Instead, cellulose primarily serves as a substrate for grafting through radical initiation sites and physical/chemical interactions.
C–) of methacrylic acid participates in radical polymerization, forming the polymer backbone. The –COOH groups do not react directly with the –OH groups of cellulose under these conditions because no esterification catalyst or activation step was used. Instead, cellulose primarily serves as a substrate for grafting through radical initiation sites and physical/chemical interactions.
2.3. Characterization of P(MAA-co-AN)-g-MNCC
The synthesized P(MAA-co-AN)-g-MNCC was studied using various analytical techniques, including TGA, FTIR, SEM, and EDX, and its Eg, PZC, and surface area were determined (S1).2.4. Adsorption studies
The adsorption studies were carried out in a batch setup (250 mL conical flask), where the synthesized adsorbent (0.05 g) was mixed with 100 mL GA solution (1–25 mg L−1) at different pH (3–11). The mixture was shaken at 200 rpm for different intervals (2.5–35 min) at a temperature from 298.15 to 328.15 K and then centrifuged for 10 min at 9000 rpm at room temperature. After centrifugation, the aliquot was analyzed for its GA content using a UV-visible spectrophotometer (Lambda 900, PerkinElmer UV Win Lab, USA) at 259 nm. The amount of GA adsorbed (qe) and % adsorption were determined using the following equations:43–45
 | (1) |
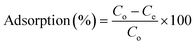 | (2) |
2.5. Kinetic, equilibrium, and thermodynamic studies
To comprehend the adsorption process, different kinetic models [pseudo-first-order, pseudo-second-order, intraparticle diffusion, and Elovich (Table S1)], equilibrium models [Freundlich, Langmuir, Temkin, and Dubinin–Radushkevich (Table S2)], and thermodynamic models [Arrhenius and van't Hoff (Table S3)] were applied.46–482.6. Desorption and reusability studies
For this purpose, the first step involved adsorbing GA on the surface of the synthesized adsorbent under optimized conditions to determine qe and % adsorption. For the desorption study, an aqueous HCl (0.1 M) solution containing EtOH (45 vol%) was used as the desorbing agent (100 mL) at pH 2 and the mixture was shaken at 200 rpm and room temperature for 30 min. Afterwards, the mixture was centrifuged for 10 min at 9000 rpm, and the supernatant was analyzed for GA content at 259 nm using a UV-visible spectrophotometer. The amount of GA desorbed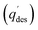 and the desorption percentage were calculated using the following equations:49,50
and the desorption percentage were calculated using the following equations:49,50
 | (3) |
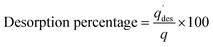 | (4) |
 (mg g−1) represents the quantity of GA desorbed, CL (mg L−1) denotes the GA concentration loaded on the adsorbent during adsorption, Cd (mg L−1) indicates the GA concentration desorbed, W (g) is the weight of the adsorbent, and V′ (L) represents the volume of the solution.
(mg g−1) represents the quantity of GA desorbed, CL (mg L−1) denotes the GA concentration loaded on the adsorbent during adsorption, Cd (mg L−1) indicates the GA concentration desorbed, W (g) is the weight of the adsorbent, and V′ (L) represents the volume of the solution.
After desorption, the same adsorbent [P(MAA-co-AN)-g-MNCC] was used for four consecutive adsorption–desorption cycles.
2.7. Application to real samples
The adsorptive potential of the synthesized P(MAA-co-AN)-g-MNCC towards GA was investigated in real samples. The selected real samples include tap water, river water, industrial wastewater, and commercially available green tea. The real water samples were obtained from Mardan district, and an industrial wastewater sample from the effluent from Rashakai Special Economic Zone, Mardan, Pakistan. These samples were filtered for the removal of suspended particles and spiked with GA (10.0 mg L−1). Similarly, the green tea samples (100 mg) were boiled in DDDW (100 mL) for 10 min and then filtered. These real samples were then used for the adsorptive removal of GA under the optimal conditions.2.8. Biological applications of P(MAA-co-AN)-g-MNCC
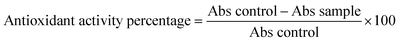 | (5) |
Three separate bores were created in each agar plate. For the treatment groups, 30 µL of the composite suspension at various concentrations (25–100 mg L−1) was introduced into the bores. Ceftriaxone (10 mg L−1) was utilized as a positive control in separate bores. The plates were incubated in a biochemical oxygen demand (BOD) incubator at 37 °C for 24 h. Following incubation, the antibacterial efficacy was quantified by measuring the diameter of the zones of inhibition (ZOI) in millimeters.
3. Results and discussion
3.1. Synthesis of P(MAA-co-AN)-g-MNCC
P(MAA-co-AN)-g-MNCC was synthesized through the mini-emulsion polymerization method. MNC was produced via chemical precipitation and subsequently integrated into NC. Graft co-polymerization of AN and MAA as functional monomers was carried out using K2S2O8 as the radical initiator and cross-linker monomer (EDGMA) under an N2 environment. Initially, upon heating, the persulfate initiator decomposes to produce the free radical . This radical removes hydrogen from the hydroxyl group of MNC, forming an alkoxy radical. The vinyl monomer functions firstly as a radical acceptor, resulting in chain growth. After that, it functions as a radical donor for other monomers. In chain propagation, the end vinyl groups of EGDMA react with the polymeric chain, forming a three-dimensional network structure with –COOH and –C
. This radical removes hydrogen from the hydroxyl group of MNC, forming an alkoxy radical. The vinyl monomer functions firstly as a radical acceptor, resulting in chain growth. After that, it functions as a radical donor for other monomers. In chain propagation, the end vinyl groups of EGDMA react with the polymeric chain, forming a three-dimensional network structure with –COOH and –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N functionalities. Compared to AN, MAA, a vinyl monomer with higher hydrophilicity, interacts more rapidly with MNC due to its hydrophilic hydroxyl group.
N functionalities. Compared to AN, MAA, a vinyl monomer with higher hydrophilicity, interacts more rapidly with MNC due to its hydrophilic hydroxyl group.
The yield of the P(MAA-co-AN)-g-MNCC synthesis was determined to be 84.6%. This was calculated by comparing the weight of the final dried and purified composite against the total mass of the initial MNC and monomers used in the feed.
The degree of swelling was evaluated gravimetrically in distilled water. The composite exhibited an equilibrium swelling capacity of 12.4 g g−1. The presence of hydrophobic acrylonitrile units and the EGDMA crosslinking network endows this material with structural integrity while maintaining sufficient porosity for adsorption.
The degree of crosslinking was determined based on the molar ratio of the EGDMA crosslinker to the total monomer concentration in the feed. The crosslinking density was calculated to be 0.078 mol%. This level of crosslinking ensures a robust three-dimensional network that prevents the dissolution of the copolymer during application in aqueous media.
3.2. Characterization of P(MAA-co-AN)-g-MNCC
Fig. 2a shows the FTIR spectrum of P(MAA-co-AN)-g-MNCC, displaying characteristic peaks at 538 cm−1, 592 cm−1, 589 cm−1, and 632 cm−1 attributed to the vibration of iron oxide in Fe3O4 nanoparticles. The peak at 2922 cm−1 is associated with the stretching vibration of the –CH group. Additionally, the peaks at 1114 cm−1 and 1636 cm−1 indicate the presence of a saccharide structure and glycosidic linkage in cellulose.53–55 The peak at 1551 cm−1 is attributed to the stretching vibration of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C group.54,56 In the spectrum, a broad O–H stretching band appears at 3441–3256 cm−1.24 The peaks at 1217 cm−1 and 1700 cm−1 indicate the presence of –C–O and –C
C group.54,56 In the spectrum, a broad O–H stretching band appears at 3441–3256 cm−1.24 The peaks at 1217 cm−1 and 1700 cm−1 indicate the presence of –C–O and –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups, confirming the existence of hydroxyl and carboxylic acid groups in the synthesized composite, respectively.57 Additionally, some new peaks appear compared to the spectra of the monomers and the Fe3O4/cellulose precursor.
O groups, confirming the existence of hydroxyl and carboxylic acid groups in the synthesized composite, respectively.57 Additionally, some new peaks appear compared to the spectra of the monomers and the Fe3O4/cellulose precursor.
SEM was used to analyze the morphology and structure of P(MAA-co-AN)-g-MNCC, as depicted in Fig. 2c. The SEM image of P(MAA-co-AN)-g-MNCC appears undulant and coarse. The increase in the amount of Fe3O4 inside the cellulose network resulted in an evident increase in surface roughness, which greatly contributed to the increased surface area. SEM analysis was explicitly performed for studying the surface topology, porosity, and morphology of P(MAA-co-AN)-g-MNCC.
The elemental composition of P(MAA-co-AN)-g-MNCC was analyzed using EDX analysis (Fig. 2d). The EDX spectrum of the synthesized composite shows the presence of C, Fe, O, and N, indicating the efficient synthesis of P(MAA-co-AN)-g-MNCC. EDX confirms the elemental incorporation and homogeneous distribution, while successful network formation is supported collectively by the FTIR analysis, swelling behavior, and adsorption performance rather than elemental composition alone.
The thermal stability of P(MAA-co-AN)-g-MNCC was analyzed by TGA, as can be seen in Fig. 2e. It is evident from the curve that the decomposition of P(MAA-co-AN)-g-MNCC occurs at various stages. The first weight loss (below ∼120 °C) corresponds to moisture removal. The second stage (≈220–350 °C) is attributed mainly to the degradation of its cellulose backbone.
The subsequent weight loss at higher temperatures corresponds to the degradation of the grafted P(MAA-co-AN) network.58,59
The band gap energy for P(MAA-co-AN)-g-MNCC was determined by the Tauc method (Fig. 2f), which was found to be 2.30 eV. This band gap energy value provides support for the adsorptive nature of P(MAA-co-AN)-g-MNCC.
The PZC was determined to investigate the influence of pH on the surface properties of P(MAA-co-AN)-g-MNCC and its adsorption behavior. By plotting ΔpH vs. pHi, the PZC of the synthesized material was calculated to be about 8.9 (Fig. 2g). This result showed that the synthesized material carries a positive charge at pH below 8.9 and a negative charge at pH above 8.9, and vice versa. GA has four pKa values: 4.16, 8.55, 11.40, and 12.8, indicating that it has a negative charge at pH greater than 4.16.37 The results confirmed the presence of an electrostatic contact force between GA and P(MAA-co-AN)-g-MNCC at pH 7.
The surface properties of P(MAA-co-AN)-g-MNCC were analyzed by recording its N2 gas adsorption–desorption isotherms at 77.35 K using a surface area analyzer. The Brunauer–Emmett–Teller (BET) equation was employed to calculate the surface area of the composite. The de Boer method was used to calculate the pore radius and volume of the composite. According to the results, the synthesized composite possesses a surface area of about 46.034 m2 g−1, a pore radius of 16.231 Å, and a pore volume of 0.001 cm3 g−1. The surface area of the synthesized composite was reduced because of the presence of multiple functional groups. Fig. 2h illustrates the adsorption–desorption isotherm of the synthesized material, which is a type IV isotherm.
3.3. Adsorption of GA using P(MAA-co-AN)-g-MNCC
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C to 55
°C to 55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C (298 K to 328 K). Variations in temperature had a negligible influence within this studied range. The adsorptive removal percentage of GA decreased slightly as the temperature increased, indicating the exothermic nature of the adsorption process. At 25
°C (298 K to 328 K). Variations in temperature had a negligible influence within this studied range. The adsorptive removal percentage of GA decreased slightly as the temperature increased, indicating the exothermic nature of the adsorption process. At 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C (298.15 K), the adsorption capacity was 16.01 mg g−1 (77.68% removal) for an initial GA concentration of 10.27 mg L−1. Consequently, 25
°C (298.15 K), the adsorption capacity was 16.01 mg g−1 (77.68% removal) for an initial GA concentration of 10.27 mg L−1. Consequently, 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C was selected as the optimal temperature for further experiments.
°C was selected as the optimal temperature for further experiments.3.4. Kinetic, equilibrium, and thermodynamic studies
The results indicate that the data were well-suited to the pseudo-second-order kinetic model, as evidenced by the high linear regression coefficient (R2) value shown in Table 1. The details of the various equations used for the determination of the kinetic and thermodynamic parameters are given in the SI (Tables S1–S3), while the data obtained are shown in Fig. S1–S11 in the SI. Fig. 4 and 5 present the pseudo-second-order kinetics and the Freundlich adsorption isotherm studies, respectively. The results indicated that the adsorption rate increased in direct proportion to the square of the number of vacant sites, suggesting that ionic interactions or ion-exchange mechanisms control the adsorption process. The pseudo-second-order adsorption mechanism primarily comprises two steps. The first step is external diffusion, where GA molecules move from the solution bulk to the external surface of P(MAA-co-AN)-g-MNCC. This is followed by the adsorption of the adsorbate molecules on the adsorbent surface, likely due to ionic interactions.| Model | Parameter | Unit | Value |
|---|---|---|---|
| a The experimental qe value is 15.886 mg g−1. | |||
| Kinetic models | |||
| Pseudo-first-order kinetic | qe (calculated) | mg g−1 | 15.890 |
| K1 | min−1 | 0.285 | |
| R2 | — | 0.869 | |
| Pseudo-first-order kinetic | qe (calculated) | mg g−1 | 14.010 |
| K2 | g mg−1 min−1 | 0.049 | |
| H | mg g−1 min−1 | 9.170 | |
| R2 | — | 0.999 | |
| Intraparticle diffusion | Kint | mg g−1 min−1/2 | 0.879 |
| C | — | 8.776 | |
| R2 | — | 0.894 | |
| Elovich | A | mg g−1 min−1 | 2.161 |
| B | g mg−1 | 0.655 | |
| R2 | — | 0.981 | |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||
| Equilibrium | |||
| Freundlich | KF (calculated) | mg g−1 | 7.761 |
| n | L g−1 | 1.668 | |
| 1/n | g L−1 | 0.600 | |
| R2 | — | 0.982 | |
| Langmuir | Qo (calculated) | mg g−1 | 52.460 |
| KL | L g−1 | 10.171 | |
| aL | L g−1 | 0.194 | |
| RL | g L−1 | 0.0023–0.089 | |
| R2 | — | 0.9300 | |
| Temkin | BT (calculated) | mg g−1 | 7.691 |
| AT | L g−1 | 5.849 | |
| bT | J mol−1 | 322.15 | |
| R2 | — | 0.840 | |
| Dubinin–Radushkevich (D–R) | Qm (calculated) | mg g−1 | 21.913 |
| K | mol2 kJ−2 | 7.81 × 10−8 | |
| E | kJ mol−1 | 12.903 | |
| R2 | — | 0.750 | |
| Thermodynamic | |||
|---|---|---|---|
| Temperature (K) | ΔG° (kJ mol−1) | ΔH° (kJ mol−1) | ΔS° (kJ mol−1) |
| 298.15 | −4.810 | −2.113 | 0.009 |
| 308.15 | −4.877 | ||
| 318.15 | −4.992 | ||
| 328.15 | −5.072 | ||
The data fit the Freundlich adsorption model well, as indicated by the high linear regression coefficient (R2) value in Table 1. This suggests a heterogeneous surface with a non-uniform distribution of the heat of adsorption, making this model appropriate for such a highly heterogeneous surface. The Freundlich equation implies that as the adsorption sites on an adsorbent become saturated, the adsorption energy decreases exponentially. KL and aL are associated with the maximum adsorption capacity (L g−1) and bonding strength (L g−1), respectively. The RL value determines whether the isotherm is unfavorable (RL > 1), linear (RL = 1), favorable (0 < RL < 1), or irreversible (RL = 0). RL values ranging from 0 to 1 indicate favorable adsorption. In this study, the RL values are greater than zero and less than one, confirming the favorable nature of GA adsorption on P(MAA-co-AN)-g-MNCC. The constant K, which is related to the adsorption energy, was calculated to be 0.003 mol2 kJ−2, resulting in a computed E value of 12.903 kJ mol−1, using P(MAA-co-AN)-g-MNCC. It has been reported that when the mean adsorption energy (E) is less than 8 kJ mol−1, it indicates physical adsorption. In comparison, values between 8 and 16 kJ mol−1 suggest chemical ion-exchange, and values above 40 kJ mol−1 indicate chemical adsorption. In this study, the lower E values indicate that the removal of GA from the aqueous environment mainly occurs through chemical adsorption and ion-exchange processes.
Based on the data obtained, the values of ΔG°, ΔH°, and ΔS° were calculated and listed in Table 1. The negative ΔG° value indicates that the adsorption process is spontaneous and exothermic, involving an ion-exchange mechanism. Furthermore, the decrease in ΔG° as the temperature increases suggests a higher adsorption capacity at lower temperatures. An increase in temperature causes GA molecules to move more rapidly, causing them to move from the solid medium to the liquid medium. As a result, the adsorption of GA decreases. This trend is also reflected in the KD values. The negative ΔH° values indicate that the adsorption process is exothermic, suggesting that adsorption may be more favorable at lower temperatures and confirming the occurrence of physisorption. The positive value of ΔS° (0.009 kJ K−1 mol−1) indicates an increase in randomness at the solid–liquid interface during the sorption of GA on P(MAA-co-AN)-g-MNCC. Additionally, the activation energy (Ea) and sticking probability (S*) parameters are used to determine whether the adsorption process is primarily chemical or physical in nature. The findings are shown in Fig. 4, with the Ea and S* values determined from the slope and intercept, respectively. The calculated Ea value is 1.263 kJ mol−1, indicating that physicochemical and ion exchange mechanisms might be dominant. Similarly, in the case of a favorable system, the S* value should be greater than zero but less than one (0 < S* < 1). The S* value in this study was determined to be 0.144, indicating that the adsorbate (GA) adhered favorably to the synthesized adsorbent [P(MAA-co-AN)-g-MNCC] (Table 1).
3.5. Desorption and reusability studies
A desorption batch experiment was designed to study the reusable potential of the synthesized P(MAA-co-AN)-g-MNCC. It was found that during the desorption process, >98% of the adsorbed GA was desorbed, and the same adsorbent was reused in four consecutive adsorption–desorption cycles, maintaining excellent adsorption capacity (Table 2). From the first to the fourth batch of adsorption–desorption process, the overall decrease in the % adsorption was <4%. This confirms the effective recyclable and reusable nature of the synthesized P(MAA-co-AN)-g-MNCC.| Reusability | Real sample applications | ||
|---|---|---|---|
| Batch | % adsorption | Real sample | % adsorption |
| 1st | 77.1 ± 2.51 | Tap water | 78.3 ± 2.56 |
| 2nd | 76.3 ± 2.43 | River water | 75.2 ± 2.14 |
| 3rd | 75.1 ± 2.51 | Industrial wastewater | 70.3 ± 1.99 |
| 4th | 73.5 ± 2.16 | Green tea | 84.9 ± 3.01 |
3.6. Application to real samples
The removal of GA from real samples was investigated. For this purpose, two real samples (river water and tap water), one from industrial effluent from the Rashakai Special Economic Zone, and one Lipton green tea sample, were utilized. The amount of GA adsorbed, qe, and the adsorption percentage were determined, as shown in Table 2. The result shows the efficient removal of GA from the real samples. This confirms the applicability of P(MAA-co-AN)-g-MNCC for the treatment of real samples and industrial waste.3.7. Biological applications of P(MAA-co-AN)-g-MNCC
| P(MAA-co-AN)-g-MNCC concentration (mg L−1) | Antioxidant activity (% DPPH scavenging) | Antibacterial activity (zone of inhibition in mm) | |
|---|---|---|---|
| Escherichia coli | Staphylococcus aureus | ||
| 25 | 58 ± 1.2 | 3.9 ± 0.12 | 4.2 ± 0.11 |
| 50 | 61 ± 1.5 | 4.3 ± 0.20 | 5.1 ± 0.13 |
| 75 | 68 ± 1.8 | 6.2 ± 0.18 | 6.4 ± 0.22 |
| 100 | 77 ± 2.2 | 7.1 ± 0.22 | 7.5 ± 0.19 |
4. Conclusions
In this study, a novel P(MAA-co-AN)-g-MNCC composite was successfully engineered using a robust stepwise and free-radical graft co-polymerization strategy. Comprehensive characterization confirmed the structural integrity and high density of active functional groups within the composite, which directly contributed to its superior performance. This material demonstrated exceptional multi-functionality, exhibiting high efficiency in the removal of gallic acid (GA) from complex water and wastewater matrices, alongside potent antioxidant and antibacterial activities. Notably, the high recyclability and biological stability of P(MAA-co-AN)-g-MNCC suggest that it is a sustainable alternative to conventional sorbents. These findings highlight the potential of this composite not only for advanced wastewater treatment but also as a versatile platform for controlled drug release, solid-phase extraction, and biomedical applications.Author contributions
Ali Khan: methodology, formal analysis, writing – original draft, Hamayun Khan: conceptualization, supervision, project administration, writing – review & editing. Nasrullah Shah: conceptualization, supervision, project administration, writing – review & editing. Mahmoud Elodemi: formal analysis, software, validation, data curation. S. Tasqeeruddin: validation, software, formal analysis and writing – review & editing, funding acquisition, resources, Ajmal Khan: writing – review & editing, supervision & project administration, Ahmed Al-Harrasi: writing – review & editing, supervision & project administration.Conflicts of interest
The authors declare no conflict of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: (a) procedure and details of instruments used in characterization techniques. (b) data (tables and plots) obtained by applying kinetic, equilibrium and thermodynamic models. See DOI: https://doi.org/10.1039/d6ra00474a.Acknowledgements
The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University, KSA, for funding this work through Small Research Group Project under grant number (RGP.1/73/46).References
- A. Dias, A. Hussain, A. S. Marcos and A. C. A. Roque, A biotechnological perspective on the application of iron oxide magnetic colloids modified with polysaccharides, Biotechnol. Adv., 2011, 29, 142–155 CrossRef CAS PubMed.
- P. R. Chang, J. Yu, X. Ma and D. P. Anderson, Polysaccharides as stabilizers for the synthesis of magnetic nanoparticles, Carbohydr. Polym., 2011, 83, 640–644 CrossRef CAS.
- H. Hu, H. Liu, D. Zhang, J. Wang and X. Zhang, pH and electromagnetic dual-remoted drug delivery based on bimodal super paramagnetic Fe3O4@porous silica nanoparticles, Eng. Sci., 2018, 2, 43–48 Search PubMed.
- M. A. C. M. Haniffa, Y. C. Ching, H. A. Illias, K. Munawar, S. Ibrahim, D. H. Nguyen and C. H. Chuah, Cellulose supported promising magnetic sorbents for magnetic solid-phase extraction: A review, Carbohydr. Polym., 2021, 253, 117245 CrossRef PubMed.
- H. Khan, A. K. Khalil, A. Khan, K. Saeed and N. Ali, Photocatalytic degradation of bromophenol blue in aqueous medium using chitosan conjugated magnetic nanoparticles, Korean J. Chem. Eng., 2016, 33, 2802–2807 CrossRef CAS.
- A. Aseri, S. K. Garg, A. Nayak and J. Ahsan, Magnetic nanoparticles: magnetic nano-technology using biomedical applications and future prospects, Int. J. Pharm. Sci. Rev. Res., 2015, 31, 119–131 CAS.
- B. G. Maciel, R. J. da Silva, A. E. Chávez-Guajardo, J. C. Medina-Llamas, J. J. Alcaraz-Espinoza and C. P. de Melo, Magnetic extraction and purification of DNA from whole human blood using a γ-Fe2O3@chitosan@polyaniline hybrid nanocomposite, Carbohydr. Polym., 2018, 197, 100–108 CrossRef CAS PubMed.
- I. A. Perales, L. M. P. Pineda, L. M. Lozano-Sánchez, J. G. Puente-Cordova and A. Elías-Zúñiga, Enhancement of a magnetorheological PDMS elastomer with carbonyl iron particles, Polym. Test., 2017, 57, 78–86 CrossRef.
- S. J. Kiprono, M. W. Ullah and G. Yang, Encapsulation of E. coli in biomimetic and Fe3O4-doped hydrogel: structural and viability analyses, Appl. Microbiol. Biotechnol., 2018, 102, 933–944 CrossRef CAS PubMed.
- M. W. Ullah, Z. Shi, X. Shi, D. Zeng, S. Li and G. Yang, Microbes as structural templates in biofabrication: Study of surface chemistry and applications, ACS Sustain. Chem. Eng., 2017, 5, 11163–11175 CrossRef CAS.
- A. S. Thakor and S. S. Gambhir, Nanooncology: the future of cancer diagnosis and therapy, Ca-Cancer J. Clin., 2013, 63(6), 395–418 CrossRef PubMed.
- C. W. Leung, K. Y. Chan, W. K. Chan, W. Zhong and P. W. T. Pong, Effect of synthesis conditions on the properties of citric-acid coated iron oxide nanoparticles, Microelectron. Eng., 2013, 110, 329–334 CrossRef.
- G. Kandasamy, A. Sudame, P. Bhati, A. Chakrabarty, S. Kale and D. Maity, Systematic magnetic fluid hyperthermia studies of carboxyl functionalized hydrophilic superparamagnetic iron oxide nanoparticles based ferrofluids, J. Colloid Interface Sci., 2018, 514, 534–543 CrossRef CAS PubMed.
- Z. Shaterabadi, G. Nabiyouni and M. Soleymani, High impact of in-situ dextran coating on biocompatibility, stability and magnetic properties of iron oxide nanoparticles, Mater. Sci. Eng., 2017, 75, 947–956 CrossRef CAS PubMed.
- E. Rauwel, S. Al-Arag, H. Salehi, C. O. Amorim, F. Cuisinier, M. Guha and P. Rauwel, Assessing cobalt metal nanoparticles uptake by cancer cells using live Raman spectroscopy, Int. J. Nanomed., 2020, 15, 7051 CrossRef CAS PubMed.
- H. Khan, A. K. Khalil and A. Khan, Photocatalytic degradation of alizarin yellow in aqueous medium and real samples using chitosan conjugated tin magnetic nanocomposites, J. Mater. Sci.: Mater. Electron., 2019, 30, 21332–21342 CrossRef CAS.
- G. Salmanian, S. A. Hassanzadeh-Tabrizi and N. Koupaei, Magnetic chitosan nanocomposites for simultaneous hyperthermia and drug delivery applications: A review, Int. J. Biol. Macromol., 2021, 184, 618–635 CrossRef CAS PubMed.
- X. Sun, S. Wang, Y. Wang and K. Sun, Synthesis and characterization of hydrophobic Fe3O4 magnetic nanoparticles with high saturation magnetization, J. Supercond. Novel Magn., 2019, 32, 2903–2911 CrossRef CAS.
- S. Khan, Z. Shah, S. Riaz, N. Ahmad, S. Islam, M. A. Raza and S. Naseem, Antimicrobial activity of citric acid functionalized iron oxide nanoparticles-superparamagnetic effect, Ceram. Int., 2020, 46, 10942–10951 CrossRef CAS.
- S. M. Fotukian, A. Barati, M. Soleymani and A. M. Alizadeh, Solvothermal synthesis of CuFe2O4 and Fe3O4 nanoparticles with high heating efficiency for magnetic hyperthermia application, J. Alloys Compd., 2020, 816, 152548 CrossRef CAS.
- A. Barra, Z. Alves, N. M. Ferreira, M. A. Martins, H. Oliveira, L. P. Ferreira and P. Ferreira, Biocompatible chitosan-based composites with properties suitable for hyperthermia therapy, J. Mater. Chem., 2020, 8, 1256–1265 CAS.
- M. A. Dheyab, A. A. Aziz, M. S. Jameel, O. A. Noqta and B. Mehrdel, Synthesis and coating methods of biocompatible iron oxide/gold nanoparticle and nanocomposite for biomedical applications, Chin. J. Phys., 2020, 64, 305–325 CrossRef CAS.
- X. Huang, J. Zhuang, D. Chen, H. Liu, F. Tang, X. Yan and J. Ren, General strategy for designing functionalized magnetic microspheres for different bioapplications, Langmuir, 2009, 25, 11657–11663 CrossRef CAS PubMed.
- T. S. Anirudhan and S. R. R. J. Binusree, Adsorptive separation of myoglobin from aqueous solutions using iron oxide magnetic nanoparticles modified with functionalized nanocrystalline cellulose, J. Chem. Eng. Data, 2013, 58, 1329–1339 CrossRef CAS.
- J. Hu, G. Chen and I. M. Lo, Removal and recovery of Cr (VI) from wastewater by maghemite nanoparticles, Water Res., 2005, 39(18), 4528–4536 CrossRef CAS PubMed.
- M. M. Woldeamanuel, S. Mohapatra, S. Senapati, T. K. Bastia, A. K. Panda and P. Rath, Role of magnetic nanomaterials in environmental remediation, in Iron Oxide-Based Nanocomposites and Nanoenzymes: Fundamentals and Applications, Springer International Publishing, Cham, 2024, pp. 185–208 Search PubMed.
- D. Klemm, B. Heublein, H. P. Fink and A. Bohn, Cellulose: fascinating biopolymer and sustainable raw material, Angew. Chem., Int. Ed., 2005, 44(22), 3358–3393 CrossRef CAS PubMed.
- Y. Habibi, L. A. Lucia and O. J. Rojas, Cellulose nanocrystals: chemistry, self-assembly, and applications, Chem. Rev., 2010, 110(6), 3479–3500 CrossRef CAS PubMed.
- L. Ding, C. Guo, Y. Zhu, L. Ma, Y. Kong, M. Zhong and H. Zhang, Adsorptive removal of gallic acid from aqueous solution onto magnetic ion exchange resin, Water Sci. Technol., 2020, 81, 1479–1493 CrossRef PubMed.
- K. Kerdsomboon, W. Chumsawat and C. Auesukaree, Effects of Moringa oleifera leaf extracts and its bioactive compound gallic acid on reducing toxicities of heavy metals and metalloid in Saccharomyces cerevisiae, Chemosphere, 2021, 270, 128659 CrossRef CAS PubMed.
- S. Verma, A. Singh and A. Mishra, Gallic acid molecular rival of cancer, Environ. Toxicol. Pharmacol., 2013, 35, 473–485 CrossRef CAS.
- Y. Hao, R. Gao, D. Liu, Y. Tang and Z. Guo, Selective extraction of gallic acid in pomegranate rind using surface imprinting polymers over magnetic carbon nanotubes, Anal. Bioanal. Chem., 2015, 407, 7681–7690 CrossRef CAS PubMed.
- X. Tang, G. I. Urujeni, X. Ni, Z. Lu, D. Wang, J. Gao and P. Dramou, Polyethyleneimine in designed nanocomposite based magnetic halloysite nanotubes for extraction and determination of gallic acid in green tea, Int. J. Biol. Macromol., 2024, 265, 130914 CrossRef CAS PubMed.
- F. Ferk, A. Chakraborty, W. Jäger, M. Kundi, J. Bichler, M. Mišík and C. Hoelzl, Potent protection of gallic acid against DNA oxidation: Results of human and animal experiments, Mutat. Res.-Fund. Mol. M., 2011, 715, 61–71 CAS.
- M. Kundi, J. Bichler, M. Mišík and C. Hoelzl, Potent protection of gallic acid against DNA oxidation: Results of human and animal experiments, Mutat. Res. Fundam. Mol. Mech. Mutagen., 2011, 715, 61–71 CrossRef PubMed.
- M. Arabi, M. Ghaedi and A. Ostovan, Synthesis and application of in-situ molecularly imprinted silica monolithic in pipette-tip solid-phase micro extraction for the separation and determination of gallic acid in orange juice samples, J. Chromatogr. B, 2017, 1048, 102–110 CrossRef CAS PubMed.
- F. J. Benitez, F. J. Real, J. L. Acero, A. I. Leal and C. Garcia, Gallic acid degradation in aqueous solutions by UV/H2O2 treatment, Fenton's reagent and the photo-Fenton system, J. Hazard Mater., 2005, 126(1–3), 31–39 CrossRef CAS PubMed.
- X. Guan, S. Yan, Z. Xu and H. Fan, Gallic acid-conjugated iron oxide nanocomposite: An efficient, separable, and reusable adsorbent for remediation of Al (III)-contaminated tannery wastewater, J. Environ. Chem. Eng., 2017, 5(1), 479–487 CrossRef CAS.
- G. G. Celestino, R. R. Henriques, A. L. Shiguihara, V. R. Constantino and J. A. Júnior, Adsorption of gallic acid on nanoclay modified with poly(diallyldimethylammonium chloride), Environ. Sci. Pollut. Res., 2019, 26, 28444–28454 CrossRef CAS PubMed.
- Y. Zhang, P. He, L. Jia, T. Zhang, H. Liu, S. Wang and S. Zhou, Dimensionally stable Ti/SnO2-RuO2 composite electrode based highly efficient electrocatalytic degradation of industrial gallic acid effluent, Chemosphere, 2019, 224, 707–715 CrossRef CAS PubMed.
- N. Majeed, N. Shah, A. Hameed, M. Shah, H. Gul, N. Ullah, T. Rehan, M. Elodemi, H. A. Ogaly, A. Khan and A. Al-Harrasi, Synthesis and characterization of multifunctional chitosan-grafted hydrogel for controlled drug delivery and electrochemical applications, J. Drug Delivery Sci. Technol., 2025, 107, 106770 CrossRef CAS.
- D. R. Mulinari and M. L. C. Silva, Adsorption of sulphate ions by modification of sugarcane bagasse cellulose, Carbohydr. Polym., 2008, 74, 617–620 CrossRef CAS.
- J. Shah, M. R. Jan, M. Khan and S. Amir, Removal and recovery of cadmium from aqueous solutions using magnetic nanoparticle-modified sawdust: kinetics and adsorption isotherm studies, Desalination Water Treat., 2016, 57, 9736–9744 CrossRef CAS.
- S. Hasan, A. Krishnaiah, T. K. Ghosh, D. S. Viswanath, V. M. Boddu and E. D. Smith, Adsorption of divalent cadmium Cd(II) from aqueous solutions onto chitosan-coated perlite beads, Ind. Eng. Chem. Res., 2006, 45, 5066–5077 CrossRef CAS.
- H. Khan, Iram, K. Gul, B. Ara, A. Khan, N. Ali, N. Ali and M. Bilal, Adsorptive removal of acrylic acid from the aqueous environment using raw and chemically modified alumina: Batch adsorption, kinetic, equilibrium and thermodynamic studies, J. Environ. Chem. Eng., 2020, 8, 103927 CrossRef CAS.
- J. Golveia, M. F. Santiago, L. B. Silva, L. C. Campos and F. Schimidt, Utilization of the corncob agro industrial residue as a potential adsorbent in the biosorption of bisphenol-A, J. Braz. Chem. Soc., 2021, 32, 1396–1404 CAS.
- N. Ayawei, A. N. Ebelegi and D. Wankasi, Modelling and interpretation of adsorption isotherms, J. Chem., 2017, 11, 2017 Search PubMed.
- A. K. Agarwal, Kinetic behavior for the adsorption of different metal ions using fly ash, J. Chem. Technol. Metall., 2021, 56, 775–781 CAS.
- T. Ullah, K. Gul, H. Khan, B. Ara and T. U. H. Zia, Efficient removal of selected fluoroquinolones from the aqueous environment using reduced magnetic graphene oxide/polyaniline composite, Chemosphere, 2022, 293, 133452 CrossRef CAS PubMed.
- H. Khan, W. Ahmad, I. Hussain, M. Imran, M. S. Afridi and S. Ullah, Phytochemical composition, antioxidant and antimicrobial activities of leaves of Olea europaea wild variety, J. Food Meas. Char., 2020, 14, 640–648 CrossRef.
- S. Alahyaribeik, S. D. Sharifi, F. Tabandeh, S. Honarbakhsh and S. Ghazanfari, Stability and cytotoxicity of DPPH inhibitory peptides derived from biodegradation of chicken feather, Protein Expression Purif., 2021, 177, 105748 CrossRef CAS PubMed.
- D. Yin, Y. Guo, M. Li, W. Wu, J. Tang, Y. Liu and F. Hu, Performance of VITEK 2, E-test, Kirby-Bauer disk diffusion and modified Kirby-Bauer disk diffusion compared to reference broth microdilution for testing tigecycline susceptibility of carbapenem-resistant K. pneumoniae and A. baumannii in a multicenter study in China, Eur. J. Clin. Microbiol. Infect. Dis., 2021, 40, 1149–1154 CrossRef CAS PubMed.
- R. Atchudan, T. N. J. I. Edison, S. Perumal, R. Vinodh, N. Muthuchamy and Y. R. Lee, One-pot synthesis of Fe3O4@graphite sheets as electrocatalyst for water electrolysis, Fuel, 2020, 277, 118235 CrossRef CAS.
- X. Huang, J. Zhuang, D. Chen, H. Liu, F. Tang, X. Yan and J. Ren, General strategy for designing functionalized magnetic microspheres for different bioapplications, Langmuir, 2009, 25, 11657–11663 CrossRef CAS PubMed.
- A. M. El-Nahas, T. A. Salaheldin, T. Zaki, H. H. El Maghrabi, A. M. Marie, S. M. Morsy and N. K. Allam, Functionalized cellulose-magnetite nanocomposite catalysts for efficient biodiesel production, Chem. Eng. J., 2017, 322, 167–180 CrossRef CAS.
- D. Shao, G. Hou, F. Chi, X. Lu and X. Ren, Transformation details of poly(acrylonitrile) to poly(amidoxime) during the amidoximation process, RSC Adv., 2021, 11, 1909–1915 RSC.
- N. Priyadarshni, P. Nath and N. Chanda, Sustainable removal of arsenate, arsenite and bacterial contamination from water using biochar stabilized iron and copper oxide nanoparticles and associated mechanism of the remediation process, J. Water Proc. Eng., 2020, 37, 101495 CrossRef.
- A. Azizi, Green synthesis of Fe3O4 nanoparticles and its application in preparation of Fe3O4/cellulose magnetic nanocomposites: A suitable proposal for drug delivery systems, J. Inorg. Organomet. Polym. Mater., 2020, 30, 3552–3561 CrossRef CAS.
- F. Osanlou, F. Nemati and S. Sabaqian, An eco-friendly and magnetized biopolymer cellulose-based heterogeneous acid catalyst for facile synthesis of functionalized pyrimido[4,5-b]quinolines and indeno fused pyrido[2,3-d]pyrimidines in water, Res. Chem. Intermed., 2017, 43, 2159–2174 CrossRef CAS.
- S. Chowdhury, R. Mishra, P. Saha and P. Kushwaha, Adsorption thermodynamics, kinetics and isosteric heat of adsorption of malachite green onto chemically modified rice husk, Desalination, 2011, 265, 159–168 CrossRef CAS.
- N. Baylan, I. Ilalan and I. Inci, Copper oxide nanoparticles as a novel adsorbent for separation of acrylic acid from aqueous solution: Synthesis, characterization and application, Water, Air, Soil Pollut., 2020, 231, 465 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |






