Open Access Article
Open Access ArticleIn situ synthesis of MXene@Iron oxide nanoparticle composites via pulsed laser ablation in liquid applied for electrochemical H2O2 reduction
Lenka Lorencováab,
Eva Noskovičovábc,
Monika Jerigovábc,
Monika Stupavskád,
Ľubomír Orovčík e,
Lucia Kopčanováe,
Peter Kasák
e,
Lucia Kopčanováe,
Peter Kasák f,
Andrej Vikartovskýag,
Dušan Lorencch,
Khaled A. Mahmoudi and
Ján Tkáč
f,
Andrej Vikartovskýag,
Dušan Lorencch,
Khaled A. Mahmoudi and
Ján Tkáč *a
*a
aInstitute of Chemistry, Slovak Academy of Sciences, Dubravska cesta 5807/9, 845 38, Bratislava, Slovak Republic. E-mail: Jan.Tkac@savba.sk
bDepartment of Physical Chemistry and Theoretical Chemistry, Faculty of Natural Sciences, Comenius University, Mlynska dolina, 842 15, Bratislava, Slovak Republic
cInternational Laser Centre-Slovak Centre of Scientific and Technical Information, Ilkovicova 3, 841 04, Bratislava, Slovak Republic
dDepartment of Plasma Physics and Technology, CEPLANT—R&D Centre for Plasma and Nanotechnology Surface Modifications, Faculty of Science, Masaryk University, Kotlářská 267/2, 602 00 Brno, Czech Republic
eInstitute of Materials and Machine Mechanics, Dubravska cesta 9/6319, 845 13, Bratislava, Slovak Republic
fCenter for Advanced Materials, Qatar University, P.O. Box 2713, Doha, Qatar
gDepartment of Analytical Chemistry, Faculty of Natural Sciences, Comenius University, Ilkovicova 6, 842 15 Bratislava, Slovak Republic
hRCQI, Institute of Physics, Slovak Academy of Sciences, Dubravska cesta 9, 841 04, Bratislava, Slovak Republic
iQatar Environment and Energy Research Institute, Hamad Bin Khalifa University, P.O. Box 34110, Doha, Qatar
First published on 18th March 2026
Abstract
We propose to circumvent the lack of intrinsic magnetism in MXenes by in situ decorating the samples with magnetic nanoparticles during laser ablation in liquid by deposition of iron species on three types of MXenes (nanocomposites). The basic properties of such nanocomposites were characterized by a number of techniques including Scanning Electron Microscopy (SEM), Secondary Ion Mass Spectrometry (SIMS), X-ray Photoelectron Spectroscopy (XPS), electrochemical investigation and the optical Faraday effect. The results indicate significant differences in the content of Fe species deposited on MXenes (Ti3C2Tx; Nb2CTx and Nb4C3Tx) and the amount of Fe species deposited on MXenes is triggered by the density of plasmons present within MXenes. Moreover, we confimed a correlation between the capability of MXene-based Fe nanocomposites to electrochemically reduce hydrogen peroxide and electrochemical activity of Fe species present on MXenes. The highest electrochemical current density obtained for H2O2 reduction was obtained on the Ti3C2Tx MXene-based Fe nanocomposite with the value of 4399 µA cm−2. We further discuss how to improve the current density obtained for H2O2 reduction on the other MXene-based Fe nanocomposites (Nb2CTx and Nb4C3Tx) using alternative synthesis protocols.
Introduction
Over the last decade, MXenes1,2 have become prominent two-dimensional nanomaterials due to their advantageous physical and chemical properties. Their application scope3–13 has expanded considerably, driven by ease of processing and compositional versatility that yield favorable electronic,14,15 optical,16,17 mechanical,18,19 antimicrobial,20,21 electrocatalytic,22,23 thermal,24,25 and magnetic properties.26,27 Their attractiveness is further reinforced by inherent hydrophilicity, tunable surface chemistry, high flake availability, and favorable synthesis yields.28 Recent advances have focused on patterning techniques to create functional MXene-based materials with enhanced performance.29–31However, most MXenes lack spontaneous magnetism due to strong covalent M–X and M–T bonds,26 as evidenced by the limited intrinsic magnetic response of Ti3C2 and Nb2C MXenes.32,33 The former exhibited paramagnetic behavior while the latter showed superconductivity-like diamagnetism. This limitation currently restricts their use in spintronics34 and related fields.35 Two strategies have emerged to address this issue: incorporating magnetic transition metal elements such as Co, Cr, Mn, Ni, V, and Fe,36–41 or decorating MXenes with magnetic nanoparticles-an approach validated by numerous studies.
Tan et al.42 synthesized Ti3C2Tx MXene from carbon soot extracted from fossil fuel combustion waste and decorated it with Fe3O4 via hydrothermal treatment for vanadium redox flow batteries. Similarly, iron oxide nanodots were self-assembled on MXene surfaces for energy storage applications.43 Hydrothermal methods have been widely employed to fabricate Fe3O4-based MXene composites for applications including photocatalysis,44,45 electrochemical sensing,46 and pollutant adsorption.47,48 Zhang and co-workers49 introduced hierarchical pore structures in MXene/Fe3O4/polyimide aerogels through directional freezing and thermal annealing for electromagnetic interference (EMI) shielding. Various fabrication approaches-including layer-by-layer assembly,50,51 electrospinning,52 hydrothermal synthesis,53–55 and precipitation methods56,57-have been utilized to develop magnetic MXene hybrids for EMI shielding applications. The dielectric-magnetic synergistic effect for effective electromagnetic wave absorption was demonstrated in MXene/Fe3O4@Fe3C@carbon composites.55 Other notable applications include tannic acid-Fe3O4@MXene hydrogels with advanced conductivity and flexibility,58 multichamber Fe3O4/Ti3C2Tx@rGO structures for broadband microwave absorption,59 Fe3O4@Ti3C2Tx composites for antibiotic degradation,60 covalently bonded Fe3O4-MXene hybrids for microwave absorption,61 and gradient-structured cellulose/MXene/Fe3O4 films for EMI shielding.51 Magnetic N-TiO2/Fe3O4@MXene showed high photo-Fenton degradation capacity for phenol,62 while magnetically recyclable Fe3O4/Ti3C2 composites were developed for tetracycline adsorption.63 Hierarchically porous Fe3O4/SiO2/MXene/rGO64 and Fe3O4/MoS2/MXene65 aerogels were prepared for microwave absorption. Magnetically enhanced photothermal antibacterial activity was observed for Fe3O4 grown in situ on MXene nanosheets with polydopamine coating.66 Additionally, sandwich-type electrochemical biosensors based on Fe3O4–NH2 and Ti3C2 MXene were developed for exosome detection with high sensitivity.67
Most preparation methods involve at least a two-step procedure: separate synthesis of MXenes and nanoparticles, followed by their combination to obtain hybrid composites. In this work, we demonstrate that benchmark MXenes—Ti3C2, Nb4C3, and Nb2C—can be directly decorated in situ with magnetic nanoparticles fabricated via laser ablation in liquid (LAL).68 To our knowledge, only one report describes LAL-mediated Fe nanocluster formation on Ti3C2Tx for ammonia sensing.69 LAL enables production of colloidally stable, ligand-free nanoparticles with tunable surface chemistry under ambient conditions.70,71 Nanoparticles produced by LAL exhibit surface defects providing electrostatic stabilization, and the absence of surface ligands results in enhanced catalytic performance compared to chemically synthesized nanoparticles.70
Here we propose synthesis of iron oxide (FeyOz) nanoclusters on three types of MXenes (Ti3C2Tx, Nb2CTx and Nb4C3Tx). We characterized the resulting magnetic nanocomposites by applying scanning electron microscopy (SEM), secondary ion mass spectrometry (SIMS), X-ray photoelectron spectroscopy (XPS), electrochemical performance in a plain buffer and an optical Faraday effect. We also investigated performance of hybrid nanocomposites for the reduction of H2O2.
Materials and methods
Reagents and materials
The phosphate buffer (PB) components (i.e. KH2PO4 and K2HPO4, pH 7.0), dimethyl sulfoxide (DMSO) were of ≥99% purity or p.a. grade and were purchased from Sigma-Aldrich (USA). 50% aq. HF was purchased from Fisher chemicals. The PB solution was freshly prepared in 0.055 µS ultrapure deionized water (DW). DW was obtained from Milli-Q® HX 7000 SD all-in-one system.Synthesis of Ti3C2Tx, Nb4C3Tx and Nb2CTx MXenes
Ti3C2Tx MXene was prepared by selective Al etching by in situ-formed HF (reaction of LiF with HCl) using a previously described protocol.72 The multilayered (ML) Nb4C3Tx and Nb2CTx MXenes were prepared by selective Al etching from Nb4AlC3 and Nb2AlC MAX phase, respectively, by hydrofluoric acid (HF) to remove Al layer as it was previously reported.73,74 The stirred dispersions of Nb2AlC or Nb4AlC3 powders immersed in 50% HF aqueous solution were heated at temperature of 40 °C for 96 h. After cooling, the resulted dispersion was thoroughly washed by DW six times and separated by centrifugation at 3500 rpm to obtain the multilayered-MXenes (ML-MXenes) as remaining residue from the supernatants. The ML-MXenes were finally washed using ethanol, and dried at 30 °C under argon atmosphere.The subsequent delamination of ML-Nb4C3Tx was performed by intercalation and ultrasonic treatment at 60% amplitude using 750 W by Hielscher Ultrasonics instrument. The dispersion of ML-Nb4C3Tx (500 mg) in DMSO (10 mL) was stirred for 24 h. Then the residue was isolated by centrifugation followed by decantation of the supernatant. Finally, the delaminated product (DL) Nb4C3Tx was achieved by probe sonication (Hielscher Ultrasonics instrument)75 of dispersion of ML-Nb4C3Tx (20 mg mL−1) in DW at 20 °C in Ar atmosphere for 1 h. ML-Nb2C MXene was similarly processed. The resulting solution was centrifuged at 5000 rpm for 10 min and decanted followed by freeze drying to get delaminated Nb2CTx or Nb4C3Tx nanosheets (DL-Nb2CTx or DL-Nb4C3Tx).
Preparation of MXene@Iron oxide nanoparticle composites by laser ablation in liquid
An amplified Ti:Sapphire system (COHERENT Legend DUO) produced pulse trains with a repetition rate of 3 kHz at central wavelength of 800 nm and with pulse energies of 2.7 mJ at 120 fs. The beam was routed to a cuvette containing 1 mL of the dispersion (deionized water + MXene) and focused by a lens with a focal length of 175 mm onto the Fe target (purchased from Goodfellow Cambridge Ltd.) immersed 1 mm below the surface of the dispersion. The peak laser density at the focus was thus on the order of 1014 W cm−2. The Fe target was exposed to laser radiation for 30 s. Thus, the laser ablation in liquid proceeded at a rate of 3000 pulses per s. Note that we were dynamically removing water vapor from above the cuvette and thus improving the laser beam coupling onto target. The as prepared MXenes patterned by Fe oxides (FeyOz) nanoparticles (FeNPs) were carefully centrifuged and rinsed three times with deionized water to remove the unattached iron particles composites and to obtain stable nanocomposites (MXene@FeNPs).Instrumentation for characterization of MXenes and MXenes@Fe oxides nanoparticle composites
Note that at the applied MXene concentrations, scattering was severely limiting our measurements as the MXene fraction would quickly sediment to the bottom of the cuvette thus creating a concentration gradient. Hence, we have applied a following protocol. After each measurement, the sample was taken out of the solenoid core and inserted into a shaker for 1 min and subsequently reinserted into the core. As the typical measurement only takes a couple of seconds, this approach allowed us to obtain consistent results.
Results and discussion
Microscopic characterization
The SEM imaging was performed on pristine samples of Ti3C2Tx, Nb4C3Tx and Nb2CTx MXenes and subsequently on the in situ decorated samples (Fig. 1). The complex, multilayered structure of pristine Ti3C2Tx, Nb4C3Tx and Nb2CTx MXenes is shown in Fig. 1a, c and e. As confirmed by Fig. 1b, d and f all three samples had shown substantial coverage by iron oxide (FeyOz) nanoparticles (FeNPs), thereby proving our approach. Moreover, we have evaluated the NP size distribution and obtained median diameters of 65 nm, 85 nm and 115 nm for Ti3C2Tx, Nb4C3Tx, Nb2CTx, respectively (see SI, Fig. S1–S3) and a relatively broad distribution function characteristic of LAL.68 The 2D structure of MXenes was still preserved, after the nanocomposites were formed.Secondary ion mass spectrometry characterization
The characteristic ions and molecules identified in the samples of Ti3C2Tx, Nb4C3Tx and Nb2CTx MXenes and MXenes@FeNPs composites were obtained in the form of mass spectra in both positive and negative polarities. Fig. S2, S4, S6, S8, S10 and S12 show the mass spectra in a positive polarity and the identified ions and fragments with the corresponding isotopic distribution for MXenes and MXenes@FeNPs composites. Fig. S3, S5, S7, S9, S11 and S13 show the mass spectra in a negative polarity with identified ions and fragments originating from MXenes and MXenes@FeNPs composites molecules, as well as the iron oxide NPs themselves. SIMS spectra show a plethora of iron oxides, including FeO+, Fe2O+, Fe3O2+, Fe3O3+.SIMS 2D distributions for the selected fragments of Ti3C2Tx@FeNPs, Nb4C3Tx@FeNPs and Nb2CTx@FeNPs composites in both polarities are shown in Fig. 2 and 3, and additional data is shown in the supplementary information file, Fig. S14–S19. Time-of-Flight-SIMS imaging was performed to evaluate the lateral distribution of Fe species. The 2D SIMS images show a clear spatial correlation between the transition metal signal (Ti+/Nb+) and characteristic Fe oxide fragments (e.g., FeO+, Fe2O+, Fe3O3+). The images were recorded over area of 250 × 250 µm.
 | ||
| Fig. 2 SIMS 2D distribution for the selected fragments of Ti3C2Tx@FeNPs (upper row), Nb4C3Tx@FeNPs (middle row), Nb2CTx@FeNPs (lower row) in positive polarity. | ||
 | ||
| Fig. 3 SIMS 2D distribution for the selected fragments of Ti3C2Tx@FeNPs (upper row), Nb4C3Tx@FeNPs (middle row), Nb2CTx@FeNPs (lower row) in negative polarity. | ||
Fig. 2, upper row represents the 2D distribution of the selected species in positive polarity: Ti+, Fe+ and the sum of FeO+, FeOH+, Fe2O+, Fe3O2+, Fe3O3+, Fe3O3H+ and Fe3O4H+ for Ti3C2Tx MXene@FeNPs. Fig. 2, middle row represents the 2D distribution of the selected species in positive polarity: Nb+, Fe+ and the sum of Fe2O+, Fe3O3+ for Nb4C3Tx MXene@FeNPs. Fig. 2, lower row represents the 2D distribution of the selected species in positive polarity: Nb+, Fe+ and sum of FeO+, FeOH+, Fe3O+, Fe3O2+, Fe3O2+, Fe3O3+, Fe3O3H+ for Nb2CTx MXene@FeNPs.
Fig. 3, upper row represents the 2D distribution of the selected species in negative polarity: sum of TiO2−, CTi−, TiF− and OTiF−, sum of FeO−, FeOH−, FeO2−, Fe2O3−, Fe3O4− and Fe4O6− and sum of TiFeO3−, TiFeO4− and Ti3Fe3O5− for Ti3C2Tx MXene@FeNPs. Fig. 3, middle row represents the 2D distribution of the selected species in negative polarity: sum of NbO−, NbO2−, NbO2H− and NbO3−, sum of FeO−, FeOH−, FeO2− and Fe3O4− and sum of NbFe3O5H5−, Nb2Fe2O3H9− and Nb4FeO3H3− for Nb4C3Tx MXene@FeNPs. Fig. 3, lower row represents the 2D distribution of the selected species in negative polarity: sum of NbO2− and NbO3−, sum of FeO−, FeOH−, FeO2−, Fe2O3− and Fe3O4− and sum of Nb2FeO6−, Nb3FeOH4−, Nb4FeO3H3− and Nb3Fe3O6H6− for Nb2CTx MXene@FeNPs. Unlike in positive polarity, we were also able to identify the MxFeyOz cluster ions.
The values of normalized intensities for identified ions in Ti3C2Tx, Nb4C3Tx and Nb2CTx MXenes with iron oxide nanoparticles in positive and negative polarity are summarized in Table S1.
In summary, the SIMS imaging shows a clear correlation between the distribution of the respective transition metal and the characteristic iron cations. Hence, it can be concluded that the original terminations at the transition metal site of the parent MXene are now predominantly occupied by the iron oxide nanoparticles. This can be understood in terms of MXenes possessing surface terminations such as oxygen (O), hydroxyl (OH), and fluorine (F) inherited from their synthesis from MAX phases and these further act as primary binding sites.80,81 For instance, it has been shown that hydroxyl groups provide activated Ti sites that can strongly interact with metal ions (like Zn2+).82
X-ray photoelectron spectroscopy (XPS) analysis
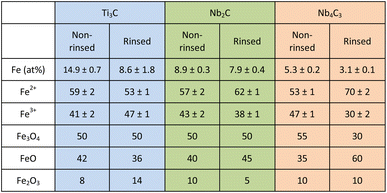
The formation of Fe nanoparticles (NPs) was further confirmed by XPS, a powerful surface-sensitive technique capable of identifying both elemental composition and oxidation states. As shown in Table 1, XPS analysis confirmed that the LAL method successfully produced MXenes enriched with Fe NPs. The highest efficiency was observed for Ti3C2, yielding a solution containing 15% Fe, of which 9% remained after water rinsing. For Nb2C and Nb4C3 MXenes, the initial Fe NP contents were 9% and 5%, respectively, decreasing to 8% and 3% after rinsing.High-resolution XPS spectra were used to determine the Fe2+/Fe3+ ratios in the metal oxides.83 Deconvolution of the Fe 2p3/2 peak revealed two main components: one at a binding energy (BE) of 710.9 eV corresponding to Fe2+, and another at 713.4 eV representing Fe3+ (Fig. 4). Relative areas of these components are summarized in Table 1. A satellite peak located 7.9 eV higher than the main Fe 2p3/2 peak was also observed, providing a clear evidence for the presence of Fe2+, since Fe3+ does not exhibit a satellite peak. The relative fractions of Fe3O4, FeO, and Fe2O3 were estimated from the deconvoluted Fe 2p3/2 spectra by quantifying Fe2+ and Fe3+ contributions (Table 1). This calculation assumed that all Fe is present exclusively in these three stoichiometric oxides. This assumption is supported by previous studies, which indicate that the LAL method frequently produces mixed FeO, Fe2O3, and Fe3O4.84–86 Stoichiometric Fe3O4 can also be written as FeO·Fe2O3; therefore, the Fe2+/Fe3+ ratio was set to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (FeO contains only Fe2+, whereas Fe2O3 contains only Fe3+). A set of linear equations was established to relate the experimentally determined Fe2+/Fe3+ ratio to the contributions from each oxide phase. Solving these equations yielded the relative phase composition, providing a straightforward means of estimating the oxide distribution directly from XPS measurements.83
2 (FeO contains only Fe2+, whereas Fe2O3 contains only Fe3+). A set of linear equations was established to relate the experimentally determined Fe2+/Fe3+ ratio to the contributions from each oxide phase. Solving these equations yielded the relative phase composition, providing a straightforward means of estimating the oxide distribution directly from XPS measurements.83
 | ||
| Fig. 4 High-resolution Fe 2p XPS spectra of Ti3C2Tx@FeNPs (a), Nb4C3Tx@FeNPs (b) and Nb2CTx@FeNPs (c). | ||
For Ti3C2 and Nb2C, Fe3O4 constitutes 50% of the total Fe oxides regardless of rinsing, indicating high stability of the mixed-valence phase. In Ti3C2, rinsing decreases FeO (from 42% to 36%) while increasing Fe2O3 (from 8% to 14%), suggesting partial oxidation of Fe2+ to Fe3+. Conversely, Nb2C exhibits a slight increase in FeO (from 40% to 45%) and a decrease in Fe2O3 upon rinsing, implying enhanced stabilization of Fe2+ species. For Nb4C3, however, a more pronounced change is observed. The Fe oxide distribution shifts from Fe3O4 = 55%, FeO = 35%, and Fe2O3 = 10% in the non-rinsed sample to Fe3O4 = 30%, FeO = 60%, and Fe2O3 = 10% after rinsing. The overall Fe content simultaneously decreases from 5 at% to 3 at%, indicating that rinsing leads primarily to the removal of weakly bound Fe3O4 nanoparticles rather than to chemical transformation of Fe species. The apparent increase in the Fe2+/Fe3+ ratio (70/30) also reflects the preferential leaching of Fe3+-containing phases.
These observations demonstrate that Fe oxide stability on MXene surfaces is composition-dependent. Ti3C2 and Nb2C retain nearly constant oxide distributions upon rinsing, confirming strong surface adhesion and chemical stability of the deposited Fe phases. In contrast, Nb4C3 shows partial loss of Fe3O4 during rinsing, suggesting that, Fe nanoparticles are more weakly anchored to this substrate and more susceptible to removal in aqueous media.
Presence of three different types of Fe oxides on MXene i.e. magnetite (Fe3O4), Fe2O3 and FeO generated from Fe target using LAL is in an excellent agreement with the literature.87
Based on SIMS analysis, the Fe-related species are distributed across the analysed surface without evidence of macroscopic segregation or localized clustering. These results indicate that Fe oxide nanoparticles are laterally distributed over the MXene surface at the micrometer scale. Although minor nanoscale variations cannot be excluded, the combined XPS and ToF-SIMS results demonstrate that the Fe loading is sufficiently homogeneous for reliable catalytic performance evaluation.
Electrochemical performance of pristine MXenes and MXene-based Fe nanocomposites
All types of MXenes exhibit a significant anodic peak, which decreases with a number of scans during CVs (Fig. 5b, d and f). While an anodic peak for Ti3C2Tx appears at a potential of 367 mV (Fig. 5b), for Nb2CTx it appears at a potential of 545 mV (Fig. 5d) and for Nb4C3Tx at a potential of 479 mV (Fig. 5f). Thus, while for oxidation of Ti3C2Tx the lowest potential is required for its oxidation such MXene material is the least electrochemically/redox stable. On the other hand, the highest potential of 545 mV was required for the oxidation of Nb2CTx; thus, Nb2CTx is the most electrochemically/redox-stable MXene among those investigated in the study, with Nb4C3Tx MXene being moderately stable. Electrochemical/redox stability in the order Nb2CTx > Nb4C3Tx > Ti3C2Tx is also confirmed by the kinetics of MXene oxidation with increasing number of scans, as shown in Fig. 6a. | ||
| Fig. 6 Stability of an anodic peak current (i.e. the highest anodic peak due to presence of surface plasmons) for Ti3C2Tx (current read at 367 mV), Nb2CTx (current read at 545 mV), and Nb4C3Tx (current read at 479 mV) MXenes during several cycles in CVs run in 0.1 M PB pH 7.0 at a sweep rate of 100 mV s−1 (a). Correlation between the content of Fe within the MXene-based nanocomposites and an anodic (plasmon) peak current observed in the 1st scan of CVs (Fig. 5b, d and e) using all types of MXenes (b). | ||
Interestingly, the Nb2CTx MXene exhibits rather wide oxidation peak, most likely due to two different redox processes at a potential of ∼310 mV and 545 mV (the main or maximal peak) (Fig. 5d), suggesting a more complex redox/electrochemical behaviour of Nb2CTx compared to Nb4C3Tx and Ti3C2Tx MXenes.
A decrease of an anodic peak with a number of scans during CVs was well described for Ti3C2Tx MXene76 and was attributed to the presence of plasmons i.e. presence of free electrons and upon oxidation of Ti3C2Tx MXene, such plasmons are channels to the electrode, resulting in oxidized Ti3C2Tx MXene with significantly altered electrochemical performance.76 Thus, Nb2CTx and Nb4C3Tx MXenes should not be exposed to conditions exceeding such an oxidation potential in order to not significantly alter electrochemical/redox performance of Nb-based MXenes, similarly to the case of Ti3C2Tx MXene. Decrease of an anodic peak for Nb2CTx and Nb4C3Tx MXenes was also described recently.88
All of the explored MXenes have metallic like bands at EF with large density of states that is due to Ti 3d bands in Ti3C2Tx and Nb 4d bands in the Nb based MXenes. Under normal conditions, they would all support collective electron oscillations and hence plasmons with the plasmon strength given by the following order: Nb4C3Tx > Nb2CTx > Ti3C2Tx. However, under strong oxygen and hydroxyl termination the order can be fully reversed. i.e. Ti3C2Tx > Nb2CTx > Nb4C3Tx. We are arguing this is indeed the case due to the following: (1) the order given by the plasmon response as identified from Fig. 5d, e and f and (2) SIMS data, specifically the images S18, S19 and Table S2.
It is worth mentioning that the electrochemical behavior of all types of MXenes is more stable in the cathodic potential window (Fig. 5a, c and e), with Nb-based MXenes being more stable compared to Ti3C2Tx MXene.
The redox/electrochemical behavior of Nb4C3Tx MXene in the cathodic potential window with a cathodic peak at ∼–700 mV and an anodic peak at a potential of ∼−530 mV (Fig. 5e) is in an excellent agreement with published data i.e. peaks at −700 mV or −490 mV, respectively, for Nb3C4Tx MXene.89 Two redox peaks at approximately the same potential were present on Nb2CTx (Fig. 5c), but more hidden in the capacitive currents.
Since the current of an anodic (plasmon) peak represents density of plasmons present in MXenes it is reasonable to assume that the density of free electrons (plasmons) can have a detrimental effect on the Fe loading capacity on MXenes during LAL process. The rationale behind is electrostatic attraction of positively charged Fe cations generated during LAL process and negatively charged MXenes. We found that the current intensity of anodic (plasmon) peak observed on pristine MXenes is increasing with an increasing the amount of Fe species deposited on MXenes during formation of MXene nanocomposites (Fig. 6b). Thus, we can conclude that the density of free electrons (plasmons) within the pristine MXenes affects effectivity of Fe deposition during LAL on pristine MXenes and thus also Fe surface coverage on the MXene nanocomposites.
Although, plasmon density expressed as anodic (plasmon) peak current is significantly lower on Nb2CTx and Nb4C3Tx MXenes, it can be effectively enhanced by the procedure of MXene synthesis from MAX phases. In the past we showed that density of plasmons expressed as anodic (plasmon) peak current was significantly lower on to Ti3C2Tx MXene when prepared using HF etching compared to LiF + HCl etching.90 Since Nb2CTx and Nb4C3Tx MXenes in this study were prepared by HF etching from their MAX phases, altered synthesis of Nb2CTx and Nb4C3Tx MXenes from their MAX phases (i.e. using LiF + HCl etchants) can enhance density of plasmons within Nb2CTx and Nb4C3Tx MXenes and thus also efficiency of Fe deposition on Nb2CTx and Nb4C3Tx MXenes using LAL.
It is noteworthly, that of an anodic peak at +550 mV on all nanocomposite electrodes (Fig. 7a, c and e), i.e. in an agreement for redox potential of Fe2+/Fe3+ redox couple (770 mV vs. NHE i.e. 570 mV vs. Ag/AgCl).91 Thus, we can attribute this peak to the redox/electrochemical activity of Fe species deposited on MXenes during LAL process. We were tempted to correlate the peak current observed at +550 mV on the nanocomposites with the electrochemical ability of nanocomposites to electrochemically reduce hydrogen peroxide by the electrochemically active Fe species present on the nanocomposites. When the peak current observed at +550 mV on nanocomposites was correlated to current observed for H2O2 reduction using nanocomposites (i.e. read at potential of −1000 mV) a good correlation was observed with R2 = 0.990 (Fig. 8). Thus, the intensity of the peak at 550 mV can be attributed to the density of electrochemically active Fe species responsible for H2O2 reduction.
 | ||
| Fig. 8 Correlation between the electrocatalytic peak current observed for electrochemical reduction of hydrogen peroxide read at −1000 mV from Fig. 7b, d and f and an anodic peak observed at +550 mV on the nanocoposites (read from Fig. 7a, c and e) with R2 = 0.990. | ||
The peak currents obtained at three different MXene-based nanocomposites can be converted to current densities using geometric surface area of 7.1 mm2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 as follows: 4399 µA cm−2 (Ti3C2Tx), 434 µA cm−2 (Nb2CTx) and 782 µA cm−2 (Nb3C4Tx). The value of current density of 4399 µA cm−2 for H2O2 reduction obtained using Ti3C2Tx MXene-based nanocomposite is significantly higher compared to the value of current density of 2020 µA cm−2 for H2O2 reduction obtained using pristine Ti3C2Tx MXene.90 Thus, LAL deposition of Fe species on MXenes is a promising way to achieve highly electrochemically active nanocomposites for H2O2 reduction using non-precious metals. We assume that synthesis of Nb2CTx and Nb4C3Tx MXenes using LiF + HCl synthetic route can enhance density of Fe species deposited on such MXenes and also increase density of electrochemically active Fe species, responsible for efficient H2O2 reduction.
90 as follows: 4399 µA cm−2 (Ti3C2Tx), 434 µA cm−2 (Nb2CTx) and 782 µA cm−2 (Nb3C4Tx). The value of current density of 4399 µA cm−2 for H2O2 reduction obtained using Ti3C2Tx MXene-based nanocomposite is significantly higher compared to the value of current density of 2020 µA cm−2 for H2O2 reduction obtained using pristine Ti3C2Tx MXene.90 Thus, LAL deposition of Fe species on MXenes is a promising way to achieve highly electrochemically active nanocomposites for H2O2 reduction using non-precious metals. We assume that synthesis of Nb2CTx and Nb4C3Tx MXenes using LiF + HCl synthetic route can enhance density of Fe species deposited on such MXenes and also increase density of electrochemically active Fe species, responsible for efficient H2O2 reduction.
Optical Faraday effect
Optical Faraday effect can be considered a benchmark technique for characterization of (optically) transparent magnetically active materials, see supplementary information, Fig. S20. Here, sample response at slowly alternating magnetic fields at a frequency of 153 Hz was observed. We started by exploring the Faraday response of LAL-prepared NP solutions in water with no MXenes added, see supplementary information, Fig. S21. They show a regular response well within the model described by Patterson et al.78 developed for characterization of superparamagnetic NPs.Next, we have switched to sample solutions containing nanocomposites. The obtained Faraday response is depicted in Fig. 9. All three MXenes under study do show magnetic properties but none of them qualitatively resembles the response of pure NPs. Most strikingly, while the NP-decorated Ti3C2Tx and Nb2CTx both show a monotonically rising Faraday response the Nb4C3Tx shows an activated response with the polarization rotation angle being negligible up until approximately 2.2–2.5 mT after which we do observe a steep rise.
 | ||
| Fig. 9 Faraday optical response of the Ti3C2Tx@FeNPs, Nb2CTx@FeNPs, and Nb4C3Tx@FeNPs nanocomposites. The solid lines are only to guide an eye. | ||
Also note that of the three MXenes under study the Ti3C2 exhibited the strongest Faraday response within the experimentally attainable range of 0–4 mT. Given the similar coverage of all three MXenes by NPs the difference cannot be simply explained by higher NP concentration for the given sample. Direct comparison between XPS data given in Table 1 and the Faraday response shows that, at low magnetic fields, up to approximately 2.5 mT, the signal magnitude for respective MXene samples follows the ordering given by either Fe or Fe3O4 content, i.e. Ti3C2Tx ≥ Nb2CTx > Nb4C3Tx. Consequently, we may speculate that at low enough mag. fields, the response is still linear and representing the actual Fe loading, while at higher fields we are entering a nonlinear regime and a direct correspondence cannot be drawn anymore.
In fact, a quantitative modeling would be needed in order to get a deeper insight into observed phenomenology. Note that our main goal here was to prove that the composite system MXene + NPs would inherit/retain magnetic properties of the NPs and not to provide a thorough quantitative analysis.
Conclusions
We have introduced a novel method for synthesis of MXene-based Fe nanocomposites using LAL allowing to prepare Fe-based nanoclusters on the surface of MXenes without any ligands, which are needed during deposition of nanoclusters on surfaces using traditional ways. The coverage of Fe on the MXene surfaces varied in the range from 5 to 15 at% of Fe depending on MXene used for deposition. Interestingly we showed that the current of plasmon peak representing density of free plasmons observed on pristine MXenes is the main driving force besides deposition of Fe species on MXenes. Furthermore, we observed that the anodic peak at +550 mV on MXene-based Fe nanocomposites represent electrochemically active Fe species with intensity of such anodic peak at +550 mV in a direct correlation with electrochemical activity of MXene-based Fe nanocomposites towards H2O2 reduction. The highest current density of electrochemical reduction of H2O2 was observed on Ti3C2Tx MXene-based Fe nanocomposite with a value of 4399 µA cm−2. We also proposed that significantly higher electrochemical reduction of H2O2 could be observed also on Nb2CTx and Nb4C3Tx MXene-based Fe nanocomposites, when using HF + HCl method for synthesis of MXenes from MAX phases. Furthermore, we have proven that the MXene-based Fe nanocomposites are magnetically active with potential to use them for some separation processes. Moreover, presence of plasmons within Nb2CTx and Nb4C3Tx MXenes could be used for spontaneous grafting of (bio)molecules using diazonium grafting-based process as was described in our earlier work on Ti3C2Tx MXene.92 Finally, it may be of great merit to test MXene-based nanocomposites using different nanoclusters deposited on MXenes using LAL for other specific purposes such as glycan enrichment and such study are underway in our laboratories.93Author contributions
Lenka Lorencová – writing – original draft, methodology, investigation, formal analysis, conceptualization. Eva Noskovičová – investigation. Monika Jerigová – investigation. Monika Stupavská – writing – original draft, investigation, formal analysis. Ľubomír Orovčík – investigation. Lucia Kopčanová – investigation. Peter Kasák – investigation. Andrej Vikartovský – investigation. Dušan Lorenc – investigation, writing – original draft. Khaled A. Mahmoudi – investigation. Ján Tkáč – writing – original draft, supervision.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI) i.e. the file “Supporting information file – “raw data.xlsx”. Supplementary information is available. See DOI: https://doi.org/10.1039/d6ra00426a.Acknowledgements
The authors would like to acknowledge the financial support received from the projects of Slovak Research and Development Agency (APVV-22-0345 and APVV-23-0083). This work was jointly supported by Qatar University and Institute of Chemistry, Slovak Academy of Sciences-IRCC-2024-429. The findings achieved herein are solely the responsibility of the authors. The work was funded by the EU NextGenerationEU through the Recovery and Resilience Plan for Slovakia under the project no. 09105-03-V02-00070. This research has been also supported by the project LM2023039 funded by the Ministry of Education, Youth and Sports of the Czech Republic.References
- M. Naguib, M. Kurtoglu, V. Presser, J. Lu, J. Niu, M. Heon, L. Hultman, Y. Gogotsi and M. Barsoum, Two-Dimensional Nanocrystals Produced by Exfoliation of Ti3AlC2, Adv. Mater., 2011, 23, 4248 CrossRef CAS PubMed.
- M. Naguib, V. Mochalin, M. Barsoum and Y. Gogotsi, 25th Anniversary Article: MXenes: A New Family of Two-Dimensional Materials, Adv. Mater., 2014, 26, 992 CrossRef CAS PubMed.
- L. Pandey, W. Liang, A. VahidMohammadi, T. Zhang, Y. Gogotsi and M. Wanunu, Hydrophilicity and Surface Charge Modulation of Ti3C2Tx MXene Based Membranes for Water Desalination, RSC Adv., 2024, 14, 21635 RSC.
- J. Simon, C. Fruhling, H. Kim, Y. Gogotsi and A. Boltasseva, MXenes for Optics and Photonics, Opt. Photonics News, 2023, 34, 44 CrossRef.
- P. Viviani, E. Gibertini, P. Fontana, F. Lissandrello, Y. Gogotsi and L. Magagnin, Flexible Inkjet-Printed Lithium-Ion Batteries with Ti3C2Tx Current Collector, J. Power Sources, 2024, 601, 234287 CrossRef CAS.
- N. Noriega, M. Shekhirev, C. Shuck, J. Salvage, A. VahidMohammadi, M. Dymond, J. Lacey, S. Sandeman, Y. Gogotsi and B. Patel, Pristine Ti3C2Tx MXene Enables Flexible and Transparent Electrochemical Sensors, ACS Appl. Mater. Interfaces, 2024, 16, 6569 CrossRef CAS PubMed.
- A. Inman, T. Parker, Y. Zhang, M. Saraf and Y. Gogotsi, Obtaining a Practical Wearable Supercapacitor Power Supply, Adv. Energy Mater., 2024, 14, 2402367 CrossRef CAS.
- F. Shahzad, M. Alhabeb, C. Hatter, B. Anasori, S. Hong, C. Koo and Y. Gogotsi, Electromagnetic Interference Shielding with 2D Transition Metal Carbides, in. MXenes: from Discovery to Applications of Two-Dimensional Metal Carbides and Nitrides, 2023, pp. 933–947 Search PubMed.
- J. Hu, N. Hu, D. Pan, Y. Zhu, X. Jin, S. Wu and Y. Lu, Smell Cancer by Machine Learning-Assisted Peptide/MXene Bioelectronic Array, Biosens. Bioelectron., 2024, 262, 116562 CrossRef CAS PubMed.
- S. Avinashi, R. Mishra, R. Singh, N. Shweta, N. Rakhi, Z. Fatima and C. Gautam, Fabrication Methods, Structural, Surface Morphology and Biomedical Applications of MXene: A Review, ACS Appl. Mater. Interfaces, 2024, 16, 47003 CrossRef CAS PubMed.
- X. Lin, D. Song, T. Shao, T. Xue, W. Hu, W. Jiang, X. Zou and N. Liu, A Multifunctional Biosensor via MXene Assisted by Conductive Metal–Organic Framework for Healthcare Monitoring, Adv. Funct. Mater., 2024, 34, 2311637 CrossRef CAS.
- L. Lorencova, P. Kasak, N. Kosutova, M. Jerigova, E. Noskovicova, A. Vikartovska, M. Barath, P. Farkas and J. Tkac, MXene-Based Electrochemical Devices Applied for Healthcare Applications, Microchim. Acta, 2024, 191, 88 CrossRef CAS PubMed.
- A. Vikartovský, N. Košútová, L. Lorencová, P. Kasák, T. Bertók and J. Tkáč, Cutting-Edge Aptasensing Approaches for Electrochemical Detection of Exosomes Utilizing MXenes, Chemelectrochem, 2025, 12, e202400713 CrossRef.
- J. Xiong, X. Zhao, Z. Liu, H. Chen, Q. Yan, H. Lian, Y. Chen, Q. Peng and X. He, Multifunctional Nacre-Like Nanocomposite Papers for Electromagnetic Interference Shielding via Heterocyclic Aramid/MXene Template-Assisted In-Situ Polypyrrole Assembly, Nano-Micro Lett., 2025, 17, 53 CrossRef CAS PubMed.
- Y. Li, Y. Wang, Z. Xu, B. Peng, N. Tran, K. Saxena, S. Vadivel and X. Liu, MXene-Based Materials for Efficient Applications in Perovskite Solar Cells: A Review, J. Mater. Sci. Technol., 2025, 215, 214 CrossRef CAS.
- D. Zhang, K. Hantanasirisakul & Y. Gogotsi, MXenes' Optical and Optoelectronic Properties and Related Applications, in. Transition Metal Carbides and Nitrides (MXenes) Handbook: Synthesis, Processing, Properties and Applications, 2024, pp. 429–452 Search PubMed.
- Y. Zhao, Z. Guan, Z. Wei, L. Fu, L. Chen, Z. Huang, M. Humphrey and C. Zhang, Enhanced Nonlinear Optical Properties of MXene (Ti3C2Tx) via Surface-Covalent Functionalization with Porphyrin, Mater. Today Phys., 2024, 48, 101577 CrossRef CAS.
- H. Zhang, C. Hao, T. Fu, D. Yu, J. Howe, K. Chen, N. Yan, H. Ren and H. Zhai, Gradient-Layered MXene/Hollow Lignin Nanospheres Architecture Design for Flexible and Stretchable Supercapacitors, Nano-Micro Lett., 2025, 17, 43 CrossRef CAS PubMed.
- X. Zhang, Y. Su, J. Xu, Y. Jin, H. Zhang, G. Ma, J. Xu, M. Zhou, X. Zhou, F. Cao, Y. Chang, Y. Wang, B. Zhao, S. Yi, J. Chen, D. Fang, X. Lv and L. Liu, Self-Adhesive ILn@MXene Multifunctional Hydrogel with Excellent Dispersibility for Human-Machine Interaction, Capacitor, Antibacterial and Detecting Various Physiological Electrical Signals in Humans and Animals, Nano Energy, 2025, 133, 110484 CrossRef CAS.
- A. Amani, A. Rahbar, E. Vafa, L. Tayebi, M. Abbasi, H. Kamyab, S. Chelliapan, S. Kasaee, A. Vaez and S. Mosleh-Shirazi, Exploring the Functionality of MXenes as Promising Versatile Antimicrobial Agents and Their Novel Applications, Mater. Today Commun., 2024, 41, 110774 CrossRef CAS.
- X. Zhao, Y. Chen, R. Niu, Y. Tang, Y. Chen, H. Su, Z. Yang, X. Jing, H. Guan, R. Gao and L. Meng, NIR Plasmonic Nanozymes: Synergistic Enhancement Mechanism and Multi-Modal Anti-Infection Applications of MXene/MOFs, Adv. Mater., 2024, 36, 2307839 CrossRef CAS PubMed.
- N. Solangi, L. Lingamdinne, R. Karri, N. Mubarak, S. Mazari and J. Koduru, Emerging 2D MXene Quantum Dots for Catalytic Conversion of CO2, Carbon, 2025, 232, 119758 CrossRef CAS.
- F. Guo, J. Ma, X. Deng and H. Gao, Interface Engineering for Improving Photoelectrocatalytic Performance of 2D/2D MXene@MoS2 for N2 Reduction Reaction: A Theoretical Insight, Fuel, 2025, 382, 133704 CrossRef CAS.
- Z. Zhao, Y. Gao and S. Zhao, Investigation of Electrothermal and Photothermal Actuation Performance in V2CTx/PE Composites, J. Alloys Compd., 2025, 1010, 177333 CrossRef CAS.
- L. Yu, L. Xu, L. Lu, Z. Alhalili and X. Zhou, Thermal Properties of MXenes and Relevant Applications, ChemPhysChem, 2022, 23, e202200203 CrossRef CAS PubMed.
- Y. Li, M. Lai, M. Hu, S. Zhao, B. Liu and J. Kai, Insights into Electronic and Magnetic Properties of MXenes: From a Fundamental Perspective, Sustain. Mater. Technol., 2022, 34, e00516 CAS.
- Y. Zhao, J. Wang, D. Yang, Z. Du, X. Zhi, R. Yu, Z. Guo and C. Tang, MXene-CNTs/Co Dielectric-Electromagnetic Synergistic Composites with Multi-Heterogeneous Interfaces for Microwave Absorption, Carbon, 2025, 232, 119825 CrossRef CAS.
- M. Downes, C. Shuck, B. McBride, J. Busa and Y. Gogotsi, Comprehensive Synthesis of Ti3C2Tx from MAX Phase to MXene, Nat. Protoc., 2024, 19, 1807 CrossRef CAS PubMed.
- Q. Zhang, J. Wang, Q. Yu, Q. Li, R. Fan, C. Li, Y. Fan, C. Zhao, W. Cheng, P. Ji, J. Sheng, C. Zhang, S. Xie, G. Henkelman and H. Li, Metal/MXene Composites via in Situ Reduction, Sustain. Mater. Technol., 2024, 4, 252 Search PubMed.
- M. Saharudin, A. Ayub, S. Hasbi, F. Muhammad-Sukki, I. Shyha and F. Inam, Recent Advances in MXene Composites Research, Applications and Opportunities, Mater. Today: Proc., 2023 DOI:10.1016/j.matpr.2023.02.435.
- P. Li, X. Zhao, Y. Ding, L. Chen, X. Wang and H. Xie, Application Studies on MXene-Based Flexible Composites, Front. Therm. Eng., 2024, 4, 1440165 CrossRef.
- Y. Yoon, T. Le, A. Tiwari, I. Kim, M. Barsoum and H. Lee, Low Temperature Solution Synthesis of Reduced Two Dimensional Ti3C2 MXenes with Paramagnetic Behaviour, Nanoscale, 2018, 10, 22429 RSC.
- Z. Babar, J. Fatheema, N. Arif, M. Anwar, S. Gul, M. Iqbal and S. Rizwan, Magnetic Phase Transition from Paramagnetic in Nb2AlC-MAX to Superconductivity-Like Diamagnetic in Nb2C-MXene: An Experimental and Computational Analysis, RSC Adv., 2020, 10, 25669 RSC.
- A. Akyildiz, I. Aysan, Y. Abdullahi, B. Hanedar, Z. Vatansever and F. Ersan, Investigation of the Electronic and Magnetic Properties of Bare and Oxygen-Terminated Ordered Double Transition-Metal MXenes for Spintronic Applications, Phys. Chem. Chem. Phys., 2024, 26, 26566 RSC.
- J. Singh, K. Singh, R. Singh and C. Adetunji, MXenes: Next-Generation 2D Materials: Fundamentals and Applications, 2024, pp. 384 Search PubMed.
- X. Jiang, A. Kuklin, A. Baev, Y. Ge, H. Ågren, H. Zhang and P. Prasad, Two-Dimensional MXenes: From Morphological to Optical, Electric, and Magnetic Properties and Applications, Phys. Rep., 2020, 848, 1 CrossRef CAS.
- S. Ullah, T. Najam, A. Rehman, S. Alarfaji, M. Ahmad, S. Riaz, B. Akkinepally, S. Shah and M. Nazir, MXene Nanomaterials: Synthesis, Properties and Applications in Energy and Environment Sector, J. Alloys Compd., 2024, 1001, 175172 CrossRef CAS.
- Q. Gao and H. Zhang, Magnetic i-MXenes: A New Class of Multifunctional Two-Dimensional Materials, Nanoscale, 2020, 12, 5995 RSC.
- H. Sarmah and S. Ghosh, Surface Passivation and Tunable Magnetic Properties of Cr-Based MXenes, J. Phys. Chem. C, 2024, 128, 16133 CrossRef CAS.
- S. Ram, N. Koshi, S. Lee and S. Bhattacharjee, Tuning the Electronic and Magnetic Properties of Double Transition Metal (MCrCT2, M = Ti, Mo) Janus MXenes for Enhanced Spintronics and Nanoelectronics, J. Phys. Chem. C, 2024, 128, 7323 CrossRef CAS.
- M. Khazaei, M. Arai, T. Sasaki, C. Chung, N. Venkataramanan, M. Estili, Y. Sakka and Y. Kawazoe, Novel Electronic and Magnetic Properties of Two-Dimensional Transition Metal Carbides and Nitrides, Adv. Funct. Mater., 2013, 23, 2185 CrossRef CAS.
- K. Tan, N. Abdullah, M. Zaed, A. Pandey, A. Zainoodin and R. Saidur, Electrochemical Performance of MXene/Fe3O4 Composite Derived from Recycled Carbon for Vanadium Redox Flow Battery, Fuel, 2024, 375, 132574 CrossRef CAS.
- P. Zhang, N. Sun, R. Soomro, S. Yue, Q. Zhu and B. Xu, Interface-Engineered Fe3O4/MXene Heterostructures for Enhanced Lithium-Ion Storage, ACS Appl. Energy Mater., 2021, 4, 11844 CrossRef CAS.
- H. Ding, M. Zhang, Y. Liu, Y. Yao, Z. Mai, H. Zheng, B. Song, B. Fan, H. Wang and H. Lu, Synthesis of CS/Fe3O4/TiO2@MXene Nanocomposite Photocatalyst with Excellent Degradation and Bacteriostatic Properties by One-Step Hydrothermal Method, Ceram. Int., 2024, 50, 46334 CrossRef CAS.
- A. Alkhudaydi, E. Danish, E. Lima, M. Gabal and M. Salam, Photocatalytic Degradation of Malachite Green Dye Using Ti3C2 MXene Nanosheets Decorated with Fe3O4 NPs under Visible Light Irradiation, Res. Chem. Intermed., 2024, 50, 5117 CrossRef CAS.
- A. Alkhudaydi, B. Samy, E. Danish, E. Lima, M. Gabal and M. Salam, Novel Sensitive Electrochemical Detection of Paracetamol Using Magnetite/MXene Electrode by Differential Pulse Voltammetry, Int. J. Electrochem. Sci., 2024, 19, 100797 CrossRef CAS.
- Z. Ma, H. Li, Y. Shen, J. Xu, X. Chen, M. Wu, L. Yang, F. Liu, R. Hao and L. Wang, Highly Selective Capture of Hg(II) Utilizing a MXene-Based Magnetic Cenosphere: Balance on Redox Sites and Magnetism, Sep. Purif. Technol., 2025, 354, 129073 CrossRef CAS.
- C. Chen, C. Chang, S. Liaw, C. Lu and J. Chen, Exfoliated MXene Nanosheet by Double Layer Forces for Preparing Magnetic Adsorbents of Toxic Metal Ions and Recycling with Hyperthermic Effect, Sep. Purif. Technol., 2024, 349, 127855 CrossRef CAS.
- P. Zhang, H. Li, H. Liang, H. Wang, X. Shan, Y. Wang, X. Fan, K. Xu, Q. Zhang and Y. Chen, Ultra-Lightweight Asymmetric Hierarchical Porous Structure for High-Efficiency Absorption-Dominated Electromagnetic Interference Shielding, Composites, Part B, 2025, 290, 111969 CrossRef CAS.
- H. Zhang, J. Cheng, K. Liu, S. Jiang, J. Zhang, Q. Wang, C. Lan, H. Jia and Z. Li, Electric-Magnetic Dual-Gradient Structure Design of Thin MXene/Fe3O4 Films for Absorption-Dominated Electromagnetic Interference Shielding, J. Colloid Interface Sci., 2025, 678, 950 CrossRef CAS PubMed.
- W. Lu, Y. Zhou, H. Xu, X. Zhang, Y. Wang and B. Zhao, Dual-Gradient MXene/AgNWs/Hollow-Fe3O4/CNF Composite Films for Thermal Management and Electromagnetic Shielding Applications, Compos. Commun., 2024, 51, 102077 CrossRef.
- M. Huang, Y. Huang, H. Yang and W. Li, Ti3C2Tx MXene/Fe3O4/Carbon Fiber Fabric/Water Polyurethane Composite Fabrics for Electromagnetic Interference Shielding and Thermal Management, ACS Appl. Nano Mater., 2024, 7, 14921 CrossRef CAS.
- M. Tao, Z. Li and H. Li, Multilayer-Structured MXene/Fe3O4 Nanosheets Composites for Electromagnetic Interference Shielding with Low Reflection, J. Mater. Sci.: Mater. Electron., 2024, 35, 1411 CrossRef CAS.
- D. Ji, Y. Wang, C. He, K. Cui, P. Han, N. Sun and Y. Li, High-Efficiency Electromagnetic Shielding Based on Multipolar Effect in RGO/MXene Aerogels Decorated with Ordered Fe3O4 Cluster, Carbon, 2024, 226, 119189 CrossRef CAS.
- J. Xu, Z. Wang, C. Ge, X. Qi, Q. Bao and C. Liu, Constructing MXene-Based Mixed-Dimensional Structure with Multiple Interfaces to Optimize Dielectric-Magnetic Synergistic Effect for Effective Electromagnetic Wave Absorption, J. Colloid Interface Sci., 2025, 677, 529 CrossRef CAS PubMed.
- M. Jia, Q. Chen, K. Chen, X. Zhang, H. Feng, C. Feng, X. Li and D. Zhang, Muscle-Inspired MXene-Based Conductive Hydrogel by Magnetic Induced for Flexible Multifunctional Sensors, Eur. Polym. J., 2024, 214, 113149 CrossRef CAS.
- B. Sun, X. Guo, D. Li, J. Zhao, X. Wang, L. Jiang, S. Chen, Y. Zhou, S. Jerrams and F. Zhou, Composites of Non-Woven Poly p-Phenylene Terephthalamide and Fe3O4-Decorated Ti3C2Tx MXene Nanosheets Coated with Ag Nanoparticles for Electromagnetic Interference Shielding, ACS Appl. Nano Mater., 2024, 7, 5610 CrossRef CAS.
- H. Sima, B. Liu, X. Shi, Z. Zhao, C. Liu and C. Zhang, Flexible and Self-Healing Tannic Acid-Fe3O4@MXene-Composited Dual Network Hydrogel Multifunctional Strain Sensor, Colloids Surf., A, 2024, 702, 135028 CrossRef CAS.
- W. Liu, K. Jia, T. Yao, L. Shen and D. Wang, Graphene-Wrapped Magnetic Multichamber Ti3C2Tx Spheres for Stable Broadband Microwave Absorption, ACS Appl. Mater. Interfaces, 2024, 16, 51118 CrossRef CAS PubMed.
- J. Huang, H. Sharif, A. Mahmood and C. Li, Unveiling the Heterojunction of Fe3O4@MXene Nanocatalyst for Ultrafast Degradation of Antibiotics via Non-Radical Pathway, Chem. Eng. J., 2024, 500, 156941 CrossRef CAS.
- J. Shi, J. Chen, X. Wang, Y. Ma, X. Yang, M. Que, H. Yuan, Y. Gao and Y. L, Covalent Assembly of Carboxyl Functionalized Ti3C2Tx with Amino-Fe3O4 for Excellent Electromagnetic Wave Absorbing Property, Ceram. Int., 2024, 50, 44793 CrossRef CAS.
- Z. Hu, W. Zhang, Y. Zhong, Q. Duan, Z. Liu, B. Wang, L. Chen, X. Wang and X. Zhang, MXene-Derived Nano N-TiO2@MXene Loaded with Fe3O4 for Photo-Fenton Synergistic Degradation of Phenol, ACS Appl. Nano Mater., 2024, 7, 20653 CrossRef CAS.
- X. Hu, G. Li, Y. Zhang, M. Lu, W. Pu, Y. Dai, M. Yang and H. Wang, A Novel Iron Oxide (Fe3O4)-Laden Titanium Carbide (Ti3C2) MXene Stacks for the Efficient Removal of Tetracycline from Aqueous Solution, Chemosphere, 2024, 364, 143210 CrossRef CAS PubMed.
- Y. Tang, S. Shao, C. Guo, K. Bi, H. Wang, T. Zhao, J. Liu and F. Wang, Multifunctional Ultralight Magnetic Fe3O4@SiO2/Ti3C2Tx/rGO Aerogel with Efficient Electromagnetic Wave Absorption and Thermal Management Properties, Carbon, 2024, 228, 119314 CrossRef CAS.
- S. Yan, S. Shao, Y. Tang, X. Zhang, C. Guo, L. Wang, J. Liu, L. Wu and F. Wang, Ultralight Hierarchical Fe3O4/MoS2/rGO/Ti3C2Tx MXene Composite Aerogels for High-Efficiency Electromagnetic Wave Absorption, ACS Appl. Mater. Interfaces, 2024, 16, 36962 CrossRef CAS PubMed.
- J. Jin, S. Wu, X. Fang, H. Li, J. Wang, S. Xuan, W. Kong, D. Wang, X. Chen, K. Leung, Q. Fang and T. Luo, MXene@Fe3O4/PDA Nanosheets with Photothermal-Magnetically Coupled Antibacterial Properties, Mater. Chem. Phys., 2024, 322, 129562 CrossRef CAS.
- L. Zhuang, Q. You, X. Su, Z. Chang, M. Ge, Q. Mei, L. Yang, W. Dong and L. Li, High-Performance Detection of Exosomes Based on Synergistic Amplification of Amino-Functionalized Fe3O4 Nanoparticles and Two-Dimensional MXene Nanosheets, Sensors, 2023, 23, 3508 CrossRef CAS PubMed.
- H. Zeng, X. Du, S. Singh, S. Kulinich, S. Yang, J. He and W. Cai, Nanomaterials via Laser Ablation/Irradiation in Liquid: A Review, Adv. Funct. Mater., 2012, 22, 1333 CrossRef CAS.
- W. Qin, X. Hu, J. Fan, Y. Liu, L. Tan, M. Zhou, C. Wu, B. Ge, S. Gao, J. Qian and W. Zhang, One-Step Laser Ablation of Fe Clusters Supported on Ti3C2Tx Nanosheets for Enhanced NH3 Sensing at Room Temperature, Ceram. Int., 2023, 49, 18353 CrossRef CAS.
- D. Zhang, B. Gokce and S. Barcikowski, Laser Synthesis and Processing of Colloids: Fundamentals and Applications, Chem. Rev., 2017, 117, 3990 CrossRef CAS PubMed.
- A. El-Khawaga, A. Zidan, A. Abd El-Mageed, M. Elsayed and M. El-Khawaga, Preparation Methods of Different Nanomaterials for Various Potential Applications: A Review, J. Mol. Struct., 2023, 1281, 135148 CrossRef CAS.
- M. Alhabeb, K. Maleski, B. Anasori, P. Lelyukh, L. Clark, S. Sin and Y. Gogotsi, Guidelines for Synthesis and Processing of Two-Dimensional Titanium Carbide (Ti3C2Tx MXene), Chem. Mater., 2017, 29, 7633 CrossRef CAS.
- P. Rasheed, R. Pandey, T. Gomez, M. Naguib and K. Mahmoud, Large Interlayer Spacing Nb4C3Tx (MXene) Promotes the Ultrasensitive Electrochemical Detection of Pb2+ on Glassy Carbon Electrodes, RSC Adv., 2020, 10, 24697 RSC.
- J. Yang, M. Naguib, M. Ghidiu, L. Pan, J. Gu, J. Nanda, J. Halim, Y. Gogotsi and M. Barsoum, Two-Dimensional Nb-Based M4C3 Solid Solutions (MXenes), J. Am. Ceram. Soc., 2016, 99, 660 CrossRef CAS.
- O. Mashtalir, M. Naguib, V. Mochalin, Y. Dall'Agnese, M. Heon, M. Barsoum and Y. Gogotsi, Intercalation and Delamination of Layered Carbides and Carbonitrides, Nat. Commun., 2013, 4, 1716 CrossRef PubMed.
- L. Lorencova, T. Bertok, E. Dosekova, A. Holazova, D. Paprckova, A. Vikartovska, V. Sasinkova, J. Filip, P. Kasak, M. Jerigova, D. Velic, K. Mahmoud and J. Tkac, Electrochemical Performance of Ti3C2Tx MXene in Aqueous Media: Towards Ultrasensitive H2O2 Sensing, Electrochim. Acta, 2017, 235, 471 CrossRef CAS PubMed.
- P. Michałowski, M. Anayee, T. Mathis, S. Kozdra, A. Wójcik, K. Hantanasirisakul, I. Jóźwik, A. Piątkowska, M. Możdżonek, A. Malinowska, R. Diduszko, E. Wierzbicka and Y. Gogotsi, Oxycarbide MXenes and MAX Phases Identification Using Monoatomic Layer-by-Layer Analysis with Ultralow-Energy Secondary-Ion Mass Spectrometry, Nat. Nanotechnol., 2022, 17, 1192 CrossRef PubMed.
- C. Patterson, M. Syed and Y. Takemura, Harmonic Decomposition of Magneto-Optical Signal from Suspensions of Superparamagnetic Nanoparticles, J. Magn. Magn. Mater., 2018, 451, 248 CrossRef CAS.
- X. Cui, F. Xiang, C. Lu, C. Liu and W. Liu, Magnetic Nanoparticles Detection Based on Nonlinear Faraday Rotation, Measurement, 2024, 227, 114309 CrossRef.
- M. Becerra and C. Toher, First-principles Investigation of Surface Functionalization and CO2 Adsorption in MXenes, NM01.07.16, MRS Spring Meeting & Exhibit, 2024 Search PubMed.
- J. Björk and J. Rosen, Functionalizing MXenes by Tailoring Surface Terminations in Different Chemical Environments, Chem. Mater., 2021, 33, 9108 CrossRef.
- C. Gu, W. Weng, C. Lu, P. Tan, Y. Jiang, Q. Zhang, X. Liu and L. Sun, Decorating MXene with tiny ZIF-8 nanoparticles: An effective approach to construct composites for water pollutant removal, Chin. J. Chem. Eng., 2022, 42, 42 CrossRef CAS.
- T. Yamashita and P. Hayes, Analysis of XPS Spectra of Fe2+ and Fe3+ Ions in Oxide Materials, Appl. Surf. Sci., 2008, 254, 2441 CrossRef CAS.
- V. Svetlichnyi, A. Shabalina, I. Lapin, D. Goncharova, D. Velikanov and A. Sokolov, Characterization and Magnetic Properties Study for Magnetite Nanoparticles Obtained by Pulsed Laser Ablation in Water, Appl. Phys. A, 2017, 123, 763 CrossRef.
- A. De Bonis, T. Lovaglio, A. Galasso, A. Santagata and R. Teghil, Iron and Iron Oxide Nanoparticles Obtained by Ultra-Short Laser Ablation in Liquid, Appl. Surf. Sci., 2015, 353, 433 CrossRef CAS.
- H. Aye, S. Choopun and T. Chairuangsri, Influence of Solvents on Characteristics of Nanoparticles Prepared by Pulsed Laser Ablation on Iron Target, Chiang Mai Univ. J. Nat. Sci., 2014, 13, 37 Search PubMed.
- V. Amendola, D. Amans, Y. Ishikawa, N. Koshizaki, S. Scirè, G. Compagnini, S. Reichenberger and S. Barcikowski, Room-Temperature Laser Synthesis in Liquid of Oxide, Metal-Oxide Core-Shells, and Doped Oxide Nanoparticles, Chem.–Eur. J., 2020, 26, 9206 CrossRef CAS PubMed.
- P. Rasheed, R. Pandey, T. Gomez, K. Jabbar, K. Prenger, M. Naguib, B. Aïssa and K. Mahmoud, Nb-Based MXenes for Efficient Electrochemical Sensing of Small Biomolecules in the Anodic Potential, Electrochem. Commun., 2020, 119, 106811 CrossRef.
- S. Zhao, X. Wang, N. Kurra, Y. Gogotsi and Y. Gao, Effect of Pinholes in Nb4C3 MXene Sheets on its Electrochemical Behavior in Aqueous Electrolytes, Electrochem. Commun., 2022, 142, 107380 CrossRef CAS.
- V. Gajdosova, L. Lorencova, M. Prochazka, M. Omastova, M. Micusik, S. Prochazkova, F. Kveton, M. Jerigova, D. Velic, P. Kasak and J. Tkac, Remarkable Differences in the Voltammetric Response Towards Hydrogen Peroxide, Oxygen and Ru(NH3)63+ of Electrode Interfaces Modified with HF or LiF-HCl Etched Ti3C2Tx MXene, Microchim. Acta, 2019, 187, 52 CrossRef PubMed.
- A. Bard, L. Faulkner and H. White, Electrochemical methods: fundamentals and applications, John Wiley & Sons, 2022, pp. 1104 Search PubMed.
- L. Lorencova, V. Gajdosova, S. Hroncekova, T. Bertok, M. Jerigova, D. Velic, P. Sobolciak, I. Krupa, P. Kasak and J. Tkac, Electrochemical Investigation of Interfacial Properties of Ti3C2Tx MXene Modified by Aryldiazonium Betaine Derivatives, Front. Chem., 2020, 8, 553 CrossRef CAS PubMed.
- J. Aguedo, Z. Pakanova, L. Lorencova, M. Nemcovic, P. Kasak, M. Barath, P. Farkas and J. Tkac, MXene as a Novel Cartridge for N-Glycan Enrichment, Anal. Chim. Acta, 2022, 1234, 340512 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |