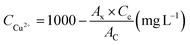Open Access Article
Open Access ArticleMultifunctional porous bio-polyurethane/biomass composites for concurrent removal of oil and heavy metal ions
Phan Huy Hoang *,
Thai Dinh Cuong and
Le Quang Dien
*,
Thai Dinh Cuong and
Le Quang Dien
Department of Chemical Engineering, School of Chemistry & Life Science, Hanoi University of Science & Technology, No 1. Dai Co Viet, Hanoi, Vietnam. E-mail: hoang.phanhuy@hust.edu.vn
First published on 5th March 2026
Abstract
The coexistence of heavy metal ions and oil in wastewater remains a major challenge for conventional treatment processes. In this study, mesoporous bio-based sorbents were fabricated by incorporating oxalic acid-treated lignocellulosic biomass (corn stover and coconut fiber) into a vegetable oil-derived polyurethane matrix. The resulting polyurethane–biomass composites exhibited dual-functionality, enabling the simultaneous adsorption of Cu2+ ions and oil from aqueous solutions. Structural characterization revealed an open-cell porous architecture and heterogeneous surface chemistry containing both polar functional groups (–OH, –COOH) and hydrophobic domains. Batch adsorption experiments demonstrated high oil uptake (up to 15.9 g g−1 for PU–CF sorbent after 180 minute adsorption) and Cu2+ adsorption capacity (up to 173.3 mg g−1 for PU–CF sorbent after 90 minute adsorption), markedly outperforming neat polyurethane and raw biomass. Adsorption equilibrium for Cu2+ was achieved within 90 min, while oil adsorption increased steadily with contact time. The enhanced performance is attributed to synergistic effects between biomass-derived metal-binding sites and the hydrophobic polyurethane network, which promotes oil absorption via capillary and van der Waals interactions. These results highlight the potential of bio-based polyurethane–biomass composites as sustainable and efficient sorbents for single-step treatment of wastewater containing mixed organic and inorganic contaminants.
Introduction
The simultaneous contamination of water bodies with heavy metal ions and hydrophobic organic pollutants (e.g., crude oil, petroleum derivatives) presents a major environmental challenge due to the different physicochemical properties and toxicological behaviors of these pollutants. Water pollution caused by the simultaneous presence of heavy metal ions and oil-based contaminants poses a significant threat to aquatic ecosystems and human health. Heavy metals such as copper (Cu2+), lead (Pb2+), and cadmium (Cd2+) are non-biodegradable and tend to bioaccumulate, posing serious risks to human health and aquatic ecosystems even at low concentrations.1–5 On the other hand, hydrophobic pollutants like oils form persistent surface films and emulsions in water, reducing oxygen transfer and threatening aquatic organisms.6–8Conventional water treatment technologies—such as membrane filtration, chemical precipitation, and traditional adsorption—are often pollutant-specific and not designed to target both hydrophilic and hydrophobic contaminants in a single step.9,10 Traditional treatment methods often address these pollutants separately, leading to increased costs and operational complexity. This limitation has motivated the development of multifunctional sorbents capable of simultaneously removing both metal ions and oil-based contaminants, thereby simplifying treatment processes and reducing costs.9
Biomass-derived sorbents have gained increasing attention due to their biodegradability, renewability, abundance, cost-effectiveness, and presence of rich surface functional groups capable of binding heavy metals.11–15 Corn stover (CS) and coconut fiber (CF), the agricultural by-product composed mainly of cellulose, hemicellulose, and lignin, provides a rich source of functional groups (e.g., –OH, –COOH, –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) suitable for metal adsorption via ion exchange or complexation.12,16–18 These two kinds of waste are abundant in Vietnam and there is no suitable method of collection and processing. Moreover, biomass itself also has ability to absorb oil due to its complex structure lignocellulose.6,19 However, biomass materials alone often exhibit limited mechanical stability and hydrophobicity, which restricts their application for simultaneous oil sorption.
O) suitable for metal adsorption via ion exchange or complexation.12,16–18 These two kinds of waste are abundant in Vietnam and there is no suitable method of collection and processing. Moreover, biomass itself also has ability to absorb oil due to its complex structure lignocellulose.6,19 However, biomass materials alone often exhibit limited mechanical stability and hydrophobicity, which restricts their application for simultaneous oil sorption.
In parallel, bio-based polyurethane (PU) synthesized from vegetable oil-derived polyols has emerged as a promising matrix for sustainable sorbent fabrication. These polyurethanes possess long aliphatic chains and urethane linkages along with porous structure that create hydrophobic domains suitable for absorbing non-polar compounds such as oils.19–22 Moreover, the use of renewable plant oils for polyol synthesis contributes to environmental sustainability and reduces dependence on fossil-derived chemicals.19
In this study, we present the fabrication of a mesoporous, bio-based sorbent composed of corn stover (CS) and coconut fiber (CF) fillers and polyurethane derived from vegetable oil-based polyol. The resulting composite exhibits dual-functionality, enabling simultaneous adsorption of heavy metal ions and hydrophobic oil contaminants from aqueous solutions. The porous architecture and chemical heterogeneity of the sorbent—combining polar and non-polar regions—facilitate effective interaction with both contaminant types. The innovation of this work lies not only in the dual-functionality of the sorbent—effectively targeting both ionic and hydrophobic contaminants in a single step—but also in its eco-friendly composition, utilizing renewable feedstocks that are often considered waste. The use of corn stover as a functional filler provides a porous structure rich in surface-active sites, while the bio-polyurethane matrix offers mechanical stability and contributes to sorption via hydrophobic interactions. To our knowledge, this is one of the first reports of a fully bio-based sorbent capable of integrated removal of such chemically distinct pollutants. This work provides new insights into the design of sustainable, multifunctional sorbents for complex wastewater treatment applications.
Materials and methods
Synthesis of bio-polyol
Vegetable oil-based bio-polyol was synthesized from sunflower oil following a previously reported method.7 Briefly, sunflower oil and formic acid (HCOOH) catalyst at a predetermined molar ratio were introduced into a conical flask and mixed thoroughly using a magnetic stirrer. Subsequently, a specified volume of hydrogen peroxide (H2O2) solution was slowly added dropwise under continuous stirring until a homogeneous reaction mixture was obtained. The flask was then sealed and heated to the desired reaction temperature using a temperature-controlled magnetic stirrer to initiate the epoxidation and ring-opening reactions leading to bio-polyol formation.Treatment of biomass
Coconut fiber (CF) was obtained from coconut shells and air-dried at room temperature for several days to remove residual moisture. The dried material was cut into smaller pieces, mechanically ground, and sieved to obtain particles with an average size of approximately 1 mm. The prepared CF samples were stored in sealed plastic bags prior to use to minimize moisture variation. Corn stover (CS) was collected from a local agricultural field in northern Vietnam after harvest. The biomass was similarly air-dried under ambient conditions for several days, followed by cutting and milling to obtain particles with a uniform size of approximately 1 mm.Both CS and CF were chemically treated using a 10 wt% oxalic acid (H2C2O4, Sigma-Aldrich) aqueous solution at 100 °C for 2 h. This treatment was employed to enhance the density of oxygen-containing functional groups, particularly carboxyl (–COOH) and hydroxyl (–OH) groups, on the biomass surface, thereby improving their affinity toward Cu2+ ions.
Fabrication of polyurethane–biomass porous biocomposite
The as-synthesized bio-polyol derived from sunflower oil was used to partially replace petrochemical polyol in the preparation of polyurethane (PU) foam. PU–biomass porous bio-composites were fabricated by incorporating treated corn stover (CS) or coconut fiber (CF) into the bio-based PU matrix following a previously reported method,23 with minor modifications.Briefly, 20 wt% of the total petrochemical polyol was replaced by bio-polyol, and the resulting polyol mixture was vigorously stirred for 3 min to ensure homogeneity. Subsequently, oxalic acid-treated CS or CF fibers (20 wt% relative to PU, average fiber length of ∼1 mm) were added to the polyol mixture and mechanically mixed for an additional 5 min to achieve uniform dispersion.
Thereafter, 4,4′-diphenylmethane diisocyanate (p-MDI, Sigma-Aldrich; average molar mass 340.6 g mol−1; NCO content ≈30 wt%) was introduced at an isocyanate-to-hydroxyl molar ratio ([NCO]/[OH]) of 1.1. The reaction mixture was rapidly stirred for 10–20 s and immediately poured into a mold for in situ foaming and curing. After completion of the curing process, the resulting porous bio-composites were demolded and cut into small specimens for subsequent characterization and adsorption experiments.
Adsorption test
80 mL of deionized water with certain concentration of ion Cu2+ was poured into a beaker, followed by the addition of 20 mL of diesel oil to form an oil–water system. A pre-weighed adsorbent sample was then placed on the oil–water system, occasionally submerged the adsorbent material deep into the water to allow the sorbent adsorb oil and Cu2+ ion for predetermined contact times. After adsorption, the sample was carefully removed and subjected to vacuum filtration for approximately 2 min to eliminate free and loosely bound water from the surface prior to weighing.Desorption and reuse procedure
The adsorbent sample after adsorption each time was simple squeezed to remove the sorbed oil and then soaked with 15 mL 0.1 mol per L HNO3 solution and shaken for hours. After shaking, the sorbent was squeezed again and was washed repeatedly with DI water in order to remove excess acid. After washing, the regenerated adsorbent was dried at 70 °C for 4 h and further used for next adsorption process with the same adsorption procedure described above. Like this, consecutive adsorption–desorption cycles were repeated eleven times by using the same sorbent.Characterizations
Fourier transform infrared (FTIR) spectra of sunflower oil, vegetable oil-based polyol, and SC–PU porous composites were recorded using an FTIR spectrometer (ALPHA, Bruker, Germany). The surface morphology of the samples was examined by scanning electron microscopy (SEM) using a JSM-7000F instrument (JEOL, Japan).The oil adsorption capacity of the PU–CS and PU–CF bio-composites were determined according to the ASTM F726 standard test method for evaluating sorbent performance for oil spill applications. The filtered adsorbent (from the adsorption process) was weighed to calculate the adsorption capacity as following equation:
The Cu2+ adsorption capacity of the PU–CS and PU–CF bio-composites were analyzed using ultraviolet-visible molecular spectroscopy (UV-VIS) absorbance measurement to determine the concentration of Cu2+ in solution after adsorption. The optical absorbance at the maximum absorption wavelength of 743 nm of the Cu–EDTA complex was recorded and then the Cu2+ concentration in each solution sample was calculated using the value of optical absorbance.
The concentration of Cu2+ adsorbed by biomass sorbent samples when was calculated according to the following formula:
The amount of Cu2+ metal ion adsorbed by the sample (adsorption capacity of biomass sorbent) was determined as follows:
| Adsorption amount = CCu2+/msample (mg Cu2+ per g material) |
Results and discussion
Characterization of the bio-based sorbent
The morphology, chemical structure, and porosity of the bio-based sorbent were thoroughly analyzed to understand its adsorption performance. Fourier-transform infrared spectroscopy (FTIR) confirmed the successful incorporation of chemical treated corn stover and coconut fiber into the polyurethane matrix. Characteristic absorption bands of lignocellulosic components such as –OH, –COOH, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups were clearly observed (Fig. 1), indicating the presence of polar functional groups that contribute to metal ion binding. In particularly, the FTIR spectra exhibited a broad absorption band in the region of 3300–3400 cm−1, corresponding to O–H stretching vibrations, which are characteristic of hydroxyl groups present in cellulose and hemicellulose matrices.24 The bands observed between 2900–2920 cm−1 were attributed to C–H stretching vibrations of aliphatic –CH2– groups, typically associated with the cellulose backbone and lignin side chains. A distinct absorption near 1730 cm−1 was ascribed to C
O groups were clearly observed (Fig. 1), indicating the presence of polar functional groups that contribute to metal ion binding. In particularly, the FTIR spectra exhibited a broad absorption band in the region of 3300–3400 cm−1, corresponding to O–H stretching vibrations, which are characteristic of hydroxyl groups present in cellulose and hemicellulose matrices.24 The bands observed between 2900–2920 cm−1 were attributed to C–H stretching vibrations of aliphatic –CH2– groups, typically associated with the cellulose backbone and lignin side chains. A distinct absorption near 1730 cm−1 was ascribed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations, likely originating from acetyl groups in hemicellulose or ester functionalities in lignin 24,25. Peaks at approximately 1600–1620 cm−1 and 1510 cm−1 were attributed to aromatic skeletal vibrations, confirming the presence of lignin. Additional peaks at around 1420 cm−1 and 1370 cm−1 corresponded to CH2 bending and C–H bending vibrations, respectively, which are characteristic of cellulose and hemicellulose structures. The absorption bands observed in the range of 1030–1050 cm−1 were ascribed to C–O stretching vibrations of alcohol and ether groups, representing glycosidic linkages and primary alcohol functionalities in cellulose.24,25 Furthermore, the peaks at 2910 cm−1 and 1708 cm−1 were associated with broad O–H stretching and C
O stretching vibrations, likely originating from acetyl groups in hemicellulose or ester functionalities in lignin 24,25. Peaks at approximately 1600–1620 cm−1 and 1510 cm−1 were attributed to aromatic skeletal vibrations, confirming the presence of lignin. Additional peaks at around 1420 cm−1 and 1370 cm−1 corresponded to CH2 bending and C–H bending vibrations, respectively, which are characteristic of cellulose and hemicellulose structures. The absorption bands observed in the range of 1030–1050 cm−1 were ascribed to C–O stretching vibrations of alcohol and ether groups, representing glycosidic linkages and primary alcohol functionalities in cellulose.24,25 Furthermore, the peaks at 2910 cm−1 and 1708 cm−1 were associated with broad O–H stretching and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations of carboxylic acid groups, respectively, which are indicative of chemical modifications in corn stover and coconut fiber following oxalic acid treatment. Additionally, characteristic absorptions corresponding to urethane linkages (–NH–COO–) derived from the vegetable oil-based polyol were observed in the 1700–1600 cm−1 region, indicating the presence of carbonyl (C
O stretching vibrations of carboxylic acid groups, respectively, which are indicative of chemical modifications in corn stover and coconut fiber following oxalic acid treatment. Additionally, characteristic absorptions corresponding to urethane linkages (–NH–COO–) derived from the vegetable oil-based polyol were observed in the 1700–1600 cm−1 region, indicating the presence of carbonyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) stretching vibrations within the urethane moiety. The peak at 1531 cm−1 was attributed to N–H bending vibrations, while the band at 1232 cm−1 corresponded to C–N stretching. Moreover, the absorption at 1105 cm−1 was assigned to C–O stretching vibrations within the urethane structure (–NHCOO–). These spectral features collectively confirm the successful formation of urethane linkages and the effective polymerization of the bio-based polyol.26,27
O) stretching vibrations within the urethane moiety. The peak at 1531 cm−1 was attributed to N–H bending vibrations, while the band at 1232 cm−1 corresponded to C–N stretching. Moreover, the absorption at 1105 cm−1 was assigned to C–O stretching vibrations within the urethane structure (–NHCOO–). These spectral features collectively confirm the successful formation of urethane linkages and the effective polymerization of the bio-based polyol.26,27
Scanning electron microscopy (SEM) images revealed a rough, mesoporous structure, with pore diameters predominantly in the 50–100 µm range. The incorporation of biomass particles generated hierarchical porosity, enhancing surface area and providing accessible adsorption sites for both aqueous ions and oil droplets.
The SEM image of the neat PU sample (Fig. 2B) reveals a relatively dense and homogeneous morphology, characterized by layered and folded polymer domains with smooth surfaces and limited open porosity. The compact microstructure suggests a continuous PU network, in which most pores are either closed or poorly interconnected. Such a morphology is beneficial for structural integrity but restricts the accessible surface area and the availability of active sites for adsorption processes.
 | ||
| Fig. 2 (A) Optical image of composite of oxalic treated-corn stover and bio-PU (PU–CS composite), (B) SEM image of neat bio-PU and (C) SEM image of PU–CS composite with 20% CS filler. | ||
In contrast, the PU–CS composite (Fig. 2C) exhibits a markedly rougher and more heterogeneous surface morphology, with the presence of abundant open pores, interconnected voids, and irregular channels. The incorporation of corn stover (CS) disrupts the dense PU matrix, leading to the formation of a highly porous architecture. The uneven surfaces and fractured features indicate good interfacial adhesion and mechanical interlocking between the lignocellulosic CS particles and the PU matrix.
The enhanced porosity and surface roughness of the PU–CS composite significantly increase the specific surface area and mass transfer pathways, facilitating the penetration of aqueous solutions into the interior of the material. Moreover, the lignocellulosic components of CS introduce additional oxygen-containing functional groups (e.g., –OH and –COOH), which serve as effective binding sites for metal ions, while the porous PU framework favors oil uptake through capillary action and hydrophobic interactions. Consequently, the PU–CS composite is expected to exhibit superior adsorption performance compared with neat PU, particularly for the simultaneous removal of heavy metal ions and oil from aqueous media.
The distinct microstructural differences observed by SEM provide a clear explanation for the adsorption behavior of the materials. The dense and less porous morphology of neat PU limits the accessibility of adsorption sites and mass transfer, whereas the highly porous and rough structure of the PU–CS composite enhances solution diffusion and exposes a larger number of active functional groups. This hierarchical porous architecture, combined with the dual hydrophilic–hydrophobic nature of the PU–CS composite, directly contributes to its improved performance in the simultaneous adsorption of Cu2+ ions and oil, as discussed in the following sections. The SEM observations confirm increased porosity and open-cell morphology by adding CS content; however, no direct structural evidence of complete pore interconnectivity is claimed in the present work.
Simultaneous adsorption of heavy metals and oil
The biocomposite sorbent composed of pre-treated biomass filler (oxalic acid treatment to improve the metal adsorption capacity) and bio-based polyurethane matrix synthesized from vegetable oil-derived bio-polyol is expected to be able to absorb heavy metals and oil in water environment at the same time. This combination provides distinct chemical domains on the sorbent surface, enabling interactions with both types of pollutants.The initial experiment aimed to evaluate the Cu2+ and oil adsorption capacities of unmodified and modified corn stover (CS) and coconut fiber (CF), and neat PU with 20% bio-polyol (20% biopolyol and 80% petro-polyol). The results was shown in Table 1.
| Material | Adsorption capacity at different contact time, g oil per g | Adsorption capacity at different contact time, mg Cu2+ per g | ||||||
|---|---|---|---|---|---|---|---|---|
| 30 min | 60 min | 120 min | 180 min | 30 min | 60 min | 90 min | 120 min | |
| CS | 1.3 ± 0.1 | 1.5 ± 0.1 | 1.8 ± 0.1 | 2.1 ± 0.1 | 32.5 ± 0.5 | 40.9 ± 0.5 | 59.4 ± 0.5 | 57.9 ± 0.5 |
| Modified CS | 1.9 ± 0.1 | 2.1 ± 0.1 | 2.7 ± 0.1 | 3.2 ± 0.1 | 60.4 ± 0.5 | 87.7 ± 0.5 | 121.1 ± 0.5 | 110.4 ± 0.5 |
| CF | 1.3 ± 0.1 | 1.8 ± 0.1 | 2.1 ± 0.1 | 2.5 ± 0.1 | 58.8 ± 0.5 | 96.4 ± 0.5 | 104.9 ± 0.5 | 101.5 ± 0.5 |
| Modified CF | 2.0 ± 0.1 | 2.6 ± 0.1 | 3.0 ± 0.1 | 3.4 ± 0.1 | 112.8 ± 0.5 | 141.2 ± 0.5 | 160.5 ± 0.5 | 136.5 ± 0.5 |
| Neat PU | 4.0 ± 0.1 | 5.1 ± 0.1 | 5.5 ± 0.1 | 5.8 ± 0.1 | 9.2 ± 0.5 | 16.6 ± 0.5 | 20.4 ± 0.5 | 17.9 ± 0.5 |
As presented in Table 1, except for the PU sample, the remaining samples all showed the ability to simultaneously adsorb oil and heavy metals in water with relatively high capacity. While PU sample only showed relatively high oil adsorption capacity and low Cu2+ adsorption capacity. In particular, both CS and CF demonstrated the ability to adsorb Cu2+ ions, although their adsorption capacities were relatively modest—approximately 59.44 mg Cu2+ per g for CS and 104.98 mg Cu2+ per g for CF. The adsorption capacity of CF was nearly twice that of CS, which may be attributed to differences in chemical composition and structural characteristics. Oxalic acid treatment significantly enhanced the Cu2+ adsorption capacity of both coconut fiber (CF) and corn stover (CS) biomass. It could be attributed to structural and chemical modifications by reaction between –COOH group with active groups in biomass that enhance both the number and accessibility of active binding sites.28,29 Notably, the highest adsorption capacities – 121.08 mg Cu2+ per g for treated CS and 160.54 mg Cu2+ per g for treated CF were achieved after oxalic acid treatment. While PU showed a low adsorption capacity of Cu2+ of 20.47 mg Cu2+ per g due to a relatively hydrophobic surface, with few polar functional groups (–OH, –NH, –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) that can interact strongly with metal ions. Moreover, all samples exhibited maximal adsorption capacities at 90 min of contact time, beyond which no further uptake of Cu2+ was observed. This plateau behavior confirms that the available active sites had become saturated and that adsorption equilibrium had been established.
O) that can interact strongly with metal ions. Moreover, all samples exhibited maximal adsorption capacities at 90 min of contact time, beyond which no further uptake of Cu2+ was observed. This plateau behavior confirms that the available active sites had become saturated and that adsorption equilibrium had been established.
For oil adsorption, both the untreated and oxalic acid-treated biomass samples exhibited comparable capacities, with CS and CF showing similar performance. Oxalic acid treatment led to a slight enhancement in oil uptake. In contrast, the PU sample demonstrated a substantially higher oil adsorption capacity, exceeding that of the biomass samples by more than twofold. Notably, the biomass samples exhibited the ability to simultaneously adsorb both oil and heavy metal ions with measurable capacities. This dual functionality arises from the intrinsic structure of lignocellulosic materials. Specifically, cellulose, hemicellulose, and lignin contain abundant hydrophilic functional groups (e.g., hydroxyl and carboxyl), which facilitate metal ion adsorption. In parallel, the capillary architecture and the presence of hydrophobic moieties contribute to effective oil uptake. However, the oil absorption capacity of natural adsorbent materials (CS, CF) is not high.
Interestingly, the new sorbent based on bio-polyol polyurethane and biomass demonstrated remarkable performance in removing both heavy metal ions and oil from aqueous solutions. By replacing 20% fossil-polyol with as synthesized bio-polyol and adding the treated biomass fiber, the porous bio-composite showed the improved oil uptake and Cu2+ adsorption compared to neat PU (without adding biomass) and only biomass fiber as seen in Table 2.
| Material | Adsorption capacity at different contact time, g oil per g | Adsorption capacity at different contact time, mg Cu2+ per g | ||||||
|---|---|---|---|---|---|---|---|---|
| 30 min | 60 min | 120 min | 180 min | 30 min | 60 min | 90 min | 120 min | |
| a PU–biomass porous bio-composites was fabricated by replacing 20% fossil-polyol with as synthesized bio-polyol and adding the 20% treated biomass fibers with 1 mm length. | ||||||||
| PU–CS | 10.0 ± 0.1 | 11.9 ± 0.1 | 13.8 ± 0.1 | 15.3 ± 0.1 | 70.5 ± 0.5 | 98.6 ± 0.5 | 129.2 ± 0.5 | 116.3 ± 0.5 |
| PU–CF | 10.7 ± 0.1 | 12.3 ± 0.1 | 14.1 ± 0.1 | 15.9 ± 0.1 | 122.6 ± 0.5 | 154.1 ± 0.5 | 173.3 ± 0.5 | 158.6 ± 0.5 |
| Neat PU | 4.0 ± 0.1 | 5.1 ± 0.1 | 5.5 ± 0.1 | 5.8 ± 0.1 | 9.25 ± 0.5 | 16.68 ± 0.5 | 20.47 ± 0.5 | 17.92 ± 0.5 |
The simultaneous adsorption experiments demonstrated that both PU–CS and PU–CF porous bio-composites reinforced exhibited significantly enhanced capacities for removing oil and Cu2+ compared to neat PU and raw biomass. Their oil adsorption capacities increased approximately 3 times relative to neat PU and nearly fivefold compared to the native lignocellulosic fillers, while Cu2+ uptake increased substantially over neat PU, and ∼10% higher than modified biomass. These results indicate a synergistic effect between the bio-PU matrix and the lignocellulosic fillers.
The enhanced oil adsorption can be primarily attributed to the increased porosity and larger interconnected pore network generated by the incorporation of natural fibers during foam formation. It can be explained that when biomass was added to the foam during polyurethane forming process, the membranes (cell walls) of PU matrix become less stable, leading to the creation of open-cell structure of the porous composite rather than closed-cells structure of original polyurethane foam.7 The open cell structure allows the oil to pass easily between the cells in the porous adsorbent instead of being impeded by the cell-walls as before. This structure facilitates rapid capillary-driven uptake and physical entrapment of hydrophobic liquids. These mechanisms are consistent with previous studies reporting strong oil sorption behavior in PU–lignocellulosic hybrid foams.19,23 Thus the oil uptake capacity of new porous bio-composite would be enhanced. Additionally, the high lignin content of coir provides intrinsically hydrophobic and oleophilic surfaces, further improving oil affinity. Hence coir fiber, treated coir fiber and PU–CF showed higher oil adsorption capacity than that of corn stalk (CS).
The improved Cu2+ adsorption performance stems from the high density of polar functional groups (–OH, –COOH, phenolic OH) present in cellulose, hemicellulose, and lignin, especially in biomass pre-treated with oxalic acid. When immobilized within the PU network, these groups become more accessible and participate in chelation, complexation, and ion-exchange interactions with Cu2+. Meanwhile, the urethane linkages (–NH–COO–) in PU also contribute to metal coordination, creating a multi-functional sorption environment. The augmented pore structure further enhances diffusion and availability of adsorption sites, leading to higher metal uptake than either component alone.
Importantly, both composites maintained high performance during simultaneous adsorption, indicating that oil and Cu2+ removal rely on distinct classes of active sites—hydrophobic domains for oil and polar/chelating sites for metal ions. This explains the limited competitive interference observed and highlights the suitability of these materials for treating wastewater containing mixed organic and inorganic pollutants.
The ability of a single sorbent to simultaneously remove both hydrophilic heavy metal ions and hydrophobic oil contaminants from aqueous environments is made possible by the presence of both polar and non-polar functional domains within the sorbent matrix. In this work, the sorbent was fabricated from biomass filler – the lignocellulosic biomass rich in surface functional groups – and bio-based polyurethane synthesized from vegetable oil-derived polyol, forming a mesoporous structure with dual adsorption functionality. The adsorption mechanism is illustrated in the following figure (Fig. 3).
Heavy metal Cu2+ ion is hydrophilic in nature and interact preferentially with polar, electron-donating functional groups. The corn stover and coir fiber components contain a high content of cellulose, hemicellulose, and lignin, which provide functional groups such as hydroxyl (–OH), carboxyl (–COOH), phenolic (–PhOH), and carbonyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) groups.2,30 These groups serve as active sites for metal ion adsorption through mechanisms such as: ion exchange, electrostatic attraction, and surface complexation or chelation. These interactions have been widely reported in lignocellulose-based biosorbents, which have demonstrated strong affinity for divalent and trivalent metal ions due to their rich surface chemistry and tunable surface charge.31
O) groups.2,30 These groups serve as active sites for metal ion adsorption through mechanisms such as: ion exchange, electrostatic attraction, and surface complexation or chelation. These interactions have been widely reported in lignocellulose-based biosorbents, which have demonstrated strong affinity for divalent and trivalent metal ions due to their rich surface chemistry and tunable surface charge.31
Oil pollutants are composed primarily of non-polar hydrocarbons and are poorly soluble in water. Their removal relies on hydrophobic interactions with non-polar surfaces. The bio-based polyurethane, synthesized from vegetable oil-based polyols, contains long aliphatic hydrocarbon chains and urethane linkages, which provide hydrophobic domains favorable for the absorption of oils and other hydrophobic organic compounds.9,20 These hydrophobic regions interact with oil molecules via van der Waals forces and partitioning effects, allowing oil droplets to adsorb onto or into the polymer matrix. Furthermore, the polymer network structure facilitates capillary absorption and swelling, enhancing oil uptake capacity.
The integration of biomass into the polyurethane matrix also generates a mesoporous structure with high surface area and interconnected pores. Moreover, this also lead to creation of open-cell structure that allows the oil to pass easily between the cells in the porous adsorbent.7 This structural feature improves mass transfer, ensures sufficient contact time between the sorbent and pollutants, and allows for the coexistence of both hydrophilic and hydrophobic adsorption sites on the same material. The resulting synergistic effect between the functionalized biomass and the hydrophobic polymer matrix enables efficient removal of both heavy metal ions and oil in a single treatment process, a capability that is still rare among biosorbents.
Overall, the combination of bio-based PU and agricultural residues yields a multifunctional sorbent with enhanced porosity and tailored surface chemistry, enabling effective removal of both hydrophobic contaminants and heavy metal ions. These findings align with literature on PU-based hybrid foams and biosorbents, confirming the feasibility of designing customizable composites for targeted water remediation.
The influence of corn stalk (CS) filler loading on the oil adsorption capacity of the PU–CS bio-composite is illustrated in Fig. 4. The results clearly demonstrate that CS content plays a crucial role in determining the oil adsorption performance of the developed sorbent. As the filler dosage increased from 5 to 20 wt%, the oil adsorption capacity increased markedly, indicating that the incorporation of CS biomass effectively enhanced the oil uptake ability of the composite.
 | ||
| Fig. 4 Oil adsorption capacity of PU–CS bio-composite with different dosage of CS filler (size of 1 mm) at different adsorption time (bio-polyol/petro-polyol ratio of 20/80 for all samples). | ||
The PU–CS sorbent containing 20 wt% CS exhibited the highest oil adsorption capacity, reaching 15.3 g g−1 after 180 min of contact time. This enhancement can be attributed to the formation of a more developed open-cell structure with higher porosity at moderate CS loadings, providing abundant pore volume and interconnected channels for oil storage and transport. The presence of open pores facilitates oil penetration into the foam, while smaller pore sizes reduce mass transfer resistance, thereby accelerating the oil absorption rate. In addition, increasing CS content improves the buoyancy of the porous composite, promoting better contact between the sorbent and the oil phase and further enhancing adsorption efficiency. However, when the CS loading was increased to 25 wt%, a slight decrease in oil adsorption capacity was observed. This decline is mainly due to the excessive filler content, which reduces the proportion of the polyurethane matrix and weakens the structural integrity of the composite. At high filler loadings, the foam structure becomes more fragile, leading to pore collapse and loss of internal channels, ultimately resulting in reduced oil adsorption efficiency.
Fig. 5 presents the effect of contact time and CS filler content on the Cu2+ adsorption capacity of PU–CS bio-composites. For all compositions, a rapid increase in adsorption capacity was observed within the initial 0–60 min, indicating the availability of accessible surface functional groups and a strong concentration gradient driving force during the early adsorption stage. This fast uptake behavior is primarily attributed to surface complexation between Cu2+ ions and oxygen-containing functional groups (–OH and –COOH) derived from the CS filler.
 | ||
| Fig. 5 Cu2+ ion adsorption capacity of PU–CS bio-composite with different dosage of CS filler (size of 1 mm) at different adsorption time (bio-polyol/petro-polyol ratio of 20/80 for all samples). | ||
As the contact time approached 90 min, the adsorption capacity gradually reached a plateau, suggesting that adsorption equilibrium was nearly achieved. A slight decrease at 120 min may be associated with partial redistribution of adsorbed ions or minor desorption effects under prolonged contact conditions. Similar behavior has been reported in biomass-based composite adsorbents, where equilibrium is attained within a finite time and extended contact does not necessarily improve metal ion uptake.
With respect to CS filler content, increasing the loading from 5 wt% to 25 wt% resulted in a consistent, though moderate, enhancement in Cu2+ adsorption capacity at all contact times. This trend can be explained by two complementary factors. First, the incorporation of CS fibers increases the overall surface porosity and promotes a more open-cell morphology within the composite matrix, facilitating improved diffusion of aqueous species into the material. Second, and more importantly, the higher CS content introduces a greater density of oxygen-containing functional groups that serve as active binding sites for Cu2+ ions.
It is important to note that, unlike oil absorption—which is predominantly governed by capillary effects and pore accessibility—the adsorption of Cu2+ ions is mainly controlled by the availability and density of surface functional groups. Therefore, the incremental increase in adsorption capacity observed with increasing CS content reflects the proportional increase in functional groups (in 5 wt% steps), rather than a structural amplification effect alone. Overall, the results indicate that CS incorporation enhances surface porosity and functional group density, both of which contribute to improved Cu2+ adsorption performance. However, the metal ion uptake behavior is predominantly functionality-driven rather than solely structure-dependent.
Desorption was conducted by simple mechanical squeezing of the PU–biomass composite and elution of metal, after which the recyclability of the sorbent was evaluated. The results showed that more than 90% of the adsorbed oil could be effectively recovered, and the collected oil could be further processed for practical reuse. The oil adsorption capacity of the PU–CS composite exhibited a slight decrease after the second cycle (≈13.3 g g−1), followed by a more pronounced decline in subsequent cycles, reaching approximately 10.2 g g−1 and 6.4 g g−1 after the third and fourth cycles, respectively. In contrast, Cu2+ adsorption remained relatively stable, with only a slight reduction observed after five cycles (≈115 mg g−1), indicating that the regeneration procedure was effective for metal removal. Overall, these results demonstrate that the PU–CS sorbent can be regenerated through oil removal and metal elution and reused for multiple cycles, confirming that the porous PU–biomass composite combines high adsorption capacity with acceptable reusability.
Conclusion
A novel, bio-based mesoporous sorbent was successfully fabricated from corn stover and vegetable oil-derived polyurethane, exhibiting excellent performance for the simultaneous adsorption of heavy metal ions and oil in aqueous media. The sorbent integrates hydrophilic functional groups from lignocellulosic biomass and hydrophobic polymer segments, enabling dual contaminant removal via distinct but synergistic mechanisms. Characterization confirmed its mesoporous structure and heterogeneous surface chemistry, while adsorption tests demonstrated high efficiency, fast kinetics, and good recyclability. This study not only introduces a sustainable solution for complex wastewater treatment, but also promotes the valorization of agricultural residues and renewable polymers for high-performance environmental materials. When compared with conventional single-function biosorbents and commercial oil adsorbents, the bio-based sorbent in this study displayed superior combined removal efficiency, material sustainability, and mechanical durability. Its entirely renewable composition – from agricultural waste and vegetable oil derivatives – offers a significant advantage for large-scale, environmentally responsible water treatment applications.Conflicts of interest
There are no conflicts to declare.Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.References
- J. Wang and C. Chen, Biosorbents for Heavy Metals Removal and Their Future, Biotechnol. Adv., 2009, 27, 195–226 Search PubMed.
- W. S. Chai, J. Y. Cheun, P. S. Kumar, M. Mubashir, Z. Majeed, F. Banat, S. H. Ho and P. L. Show, A Review on Conventional and Novel Materials towards Heavy Metal Adsorption in Wastewater Treatment Application, J. Cleaner Prod., 2021, 296, 126589 Search PubMed.
- W. D. Costa, A. M. da Silva Bento, J. A. S. de Araújo, J. M. C. Menezes, J. G. M. da Costa, F. A. B. da Cunha, H. D. M. Coutinho, F. J. de Paula Filho and R. N. Pereira Teixeira, Removal of Copper(II) Ions and Lead(II) from Aqueous Solutions Using Seeds of Azadirachta Indica A. Juss as Bioadsorvent, Environ. Res., 2020, 183, 109213 Search PubMed.
- F. G. Ávila, J. Cabrera-Sumba, S. Valdez-Pilataxi, J. Villalta-Chungata, L. Valdiviezo-Gonzales and C. Alegria-Arnedo, Removal of Heavy Metals in Industrial Wastewater Using Adsorption Technology: Efficiency and Influencing Factors, Cleaner Eng. Technol., 2025, 24, 100879 CrossRef.
- Z. Sun, Y. Liao, Y. Zhang, S. Sun, Q. Kan, Z. Wu, L. Yu, Z. Dong, Z. Wang, R. He, L. Wang, Q. Meng, H. Wang, Q. Wang, L. Mao, D. Pan, S. Wang and Z. Zhang, Recent Progress in Covalent Organic Framework-Based Membranes: Design, Synthesis, and Applications in the Fields of Energy and the Environment, ACS Macro Lett., 2025, 14, 1201–1220 Search PubMed.
- P. H. Hoang, H. T. Dat, T. D. Cuong and L. Q. Dien, Pretreatment of Coir Lignocellulose for Preparation of a Porous Coir-Polyurethane Composite with High Oil Adsorption Capacity, RSC Adv., 2022, 12, 14976–14985 RSC.
- P. H. Hoang, N. M. Dat and B. Q. Hung, One-Pot Synthesis of Bio-Polyol from Vegetable Oil for Preparation of Bio-Polyurethane Based Porous Sorbent with High Oil Adsorption Capacity, J. Appl. Polym. Sci., 2023, 1–9 Search PubMed.
- A. I. Adetunji and A. O. Olaniran, Treatment of Industrial Oily Wastewater by Advanced Technologies: A Review, Appl. Water Sci., 2021, 11, 1–19 Search PubMed.
- Q. Zhu, Q. Pan and F. Liu, Facile Removal and Collection of Oils from Water Surfaces through Superhydrophobic and Superoleophilic Sponges, J. Phys. Chem. C, 2011, 115, 17464–17470 Search PubMed.
- F. Fu and Q. Wang, Removal of Heavy Metal Ions from Wastewaters: A Review, J. Environ. Manage., 2011, 92, 407–418 CrossRef CAS PubMed.
- O. S. Bello, E. S. Owojuyigbe, M. A. Babatunde and F. E. Folaranmi, Sustainable Conversion of Agro-Wastes into Useful Adsorbents, Appl. Water Sci., 2017, 7, 3561–3571 CrossRef CAS.
- M. Petrović, T. Šoštarić, M. Stojanović, J. Milojković, M. Mihajlović, M. Stanojević and S. Stanković, Removal of Pb2+ Ions by Raw Corn Silk (Zea Mays L.) as a Novel Biosorbent, J. Taiwan Inst. Chem. Eng., 2016, 58, 407–416 CrossRef.
- H. A. Hegazi, Removal of Heavy Metals from Wastewater Using Agricultural and Industrial Wastes as Adsorbents, HBRC J., 2013, 9, 276–282 CrossRef.
- I. Abdelfattah, A. A. Ismail, F. A. Sayed, A. Almedolab and K. M. Aboelghait, Biosorption of Heavy Metals Ions in Real Industrial Wastewater Using Peanut Husk as Efficient and Cost Effective Adsorbent, Environ. Nanotechnol., Monit. Manage., 2016, 6, 176–183 Search PubMed.
- F. Wang, Y. Pan, P. Cai, T. Guo and H. Xiao, Single and Binary Adsorption of Heavy Metal Ions from Aqueous Solutions Using Sugarcane Cellulose-Based Adsorbent, Bioresour. Technol., 2017, 241, 482–490 Search PubMed.
- X. Tang, H. Wang, M. Hou, L. Song, C. Zhou, H. Zhao and L. Shi, Highly Efficient Adsorption of Cadmium(II) onto Durable Coconut Fiber Residue, Desalin. Water Treat., 2016, 57, 15098–15107 CrossRef CAS.
- K. Conrad and H. C. B. Hansen, Sorption of Zinc and Lead on Coir, Bioresour. Technol., 2007, 98, 89–97 CrossRef CAS PubMed.
- U. K. Garg, M. P. Kaur and V. K. Garg, Sud, D. Removal of Hexavalent Chromium from Aqueous Solution by Agricultural Waste Biomass, J. Hazard. Mater., 2007, 140, 60–68 Search PubMed.
- P. H. Hoang and H. T. Dat, Cascade Reaction for Bio-Polyol Synthesis from Sunflower Oil over a W/ZSM-5 Zeolite Catalyst for the Fabrication of a Bio-Polyurethane-Based Porous Biocomposite with High Oil Uptake, RSC Adv., 2024, 14, 20974–20981 Search PubMed.
- F. Olivito, V. Algieri, A. Jiritano, M. A. Tallarida, P. Costanzo, L. Maiuolo and A. De Nino, Bio-Based Polyurethane Foams for the Removal of Petroleum-Derived Pollutants: Sorption in Batch and in Continuous-Flow, Polymers, 2023, 15, 1785 Search PubMed.
- L. S. Martins, N. C. Zanini, L. S. Maia, A. G. Souza, R. F. S. Barbosa, D. S. Rosa and D. R. Mulinari, Crude Oil and S500 Diesel Removal from Seawater by Polyurethane Composites Reinforced with Palm Fiber Residues, Chemosphere, 2021, 267, 129288 Search PubMed.
- S. M. Kong, Y. Han, N. I. Won and Y. H. Na, Polyurethane Sponge with a Modified Specific Surface for Repeatable Oil-Water Separation, ACS Omega, 2021, 6, 33969–33975 Search PubMed.
- T. D. Cuong, L. Q. Dien and P. H. Hoang, Preparation of Bio-Based Porous Material with High Oil Adsorption Capacity from Bio-Polyurethane and Sugarcane Bagasse, RSC Adv., 2024, 14, 6938–6947 Search PubMed.
- O. Karnitz, L. V. A. Gurgel, J. C. P. de Melo, V. R. Botaro, T. M. S. Melo, R. P. de Freitas Gil and L. F. Gil, Adsorption of Heavy Metal Ion from Aqueous Single Metal Solution by Chemically Modified Sugarcane Bagasse, Bioresour. Technol., 2007, 98, 1291–1297 Search PubMed.
- A. Çelekli, B. Bozkuş and H. Bozkurt, Development of a New Adsorbent from Pumpkin Husk by KOH-Modification to Remove Copper Ions, Environ. Sci. Pollut. Res., 2019, 26, 11514–11523 Search PubMed.
- J. G. Vijayan, A. Chandrashekar, J. Ag and T. N. Prabhu, Polyurethane and Its Composites Derived from Bio-Sources : Synthesis , Characterization and Adsorption Studies, Polym. Polym. Compos., 2022, 30, 1–18 Search PubMed.
- Z. Fang, C. Qiu, D. Ji, Z. Yang, N. Zhu, J. Meng, X. Hu and K. Guo, Development of High-Performance Biodegradable Rigid Polyurethane Foams Using Full Modified Soy-Based Polyols, J. Agric. Food Chem., 2019, 67, 2220–2226 Search PubMed.
- S. U. Sondea, S. A. Odoemelam and F. K. Onwu, Isotherm Studies of Equilibrium Sorption of Cu2+ and Cd2+ from Aqueous Solutions by Modified and Unmodified Breadfruit Seed Hull, Orbital: Electron. J. Chem., 2015, 7(3), 215–225 Search PubMed.
- W. S. Wan Ngah and M. A. K. M. Hanafiah, Removal of Heavy Metal Ions from Wastewater by Chemically Modified Plant Wastes as Adsorbents: A Review, Bioresour. Technol., 2008, 99, 3935–3948 Search PubMed.
- K. Kaur, R. Kaur and H. Kaur, Applied Surface Science Advances A Systematic Review of Lignocellulosic Biomass for Remediation of Environmental Pollutants, Appl. Surf. Sci. Adv., 2024, 19, 100547 Search PubMed.
- P. Basnet, D. Gyawali, K. Nath and H. Paudyal, Results in Chemistry An Assessment of the Lignocellulose-Based Biosorbents in Removing Cr (VI) from Contaminated Water : A Critical Review, Results Chem., 2022, 4, 100406 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |