 Open Access Article
Open Access ArticleModelling simple and complex metal-oxide and -hydroxide surface structures using their point of zero charge
Miriam Leffler *a and
Steven L. Suib
*a and
Steven L. Suib b
b
aDepartment of Chemistry, University of Connecticut, USA. E-mail: Miriamleffler1066@gmail.com
bDepartment of Chemistry, Director of IMS Board of Trustees Distinguished Professor University of Connecticut, USA. E-mail: Steven.Suib@uconn.edu
First published on 15th May 2026
Abstract
Multiple methods are available to measure organic and inorganic structures in an aqueous environment. These techniques though are mainly used for measuring and modelling the global structures of species in solution, molecules, polymers, biological compounds, and individual structure factors, not surfaces. Since surface structure and composition are directly responsible for a materials point of zero charge it is the ideal property for determining its global structure and composition. Multiple metal-oxides and -hydroxides in an aqueous environment, under varying conditions are also subject to changes in their surface structure and composition. Therefore, this work focuses solely on modelling the global surface structure of simple and complex metal-oxides and –hydroxides in an aqueous environment at their solid/liquid interface, as there are no other methods currently available for this task. The model developed for this work was obtained by augmenting a model originally used for predicting the point of zero charge for both simple and complex metal-oxides and –hydroxides using only their structure factors. To adapt the original model, it was necessary to determine not only each materials set of structure factors but also its surface properties (i.e. bond ionic content). The method then developed required a determination of each of the possible stable surface crystal structures at its point of zero charge (pH). Each of these structures was then placed in the model to determine which global structure converged to its experimental point of zero charge.
1 Introduction
1.1 The uses of point of zero charge
The importance of a material's point of zero charge (PZC) cannot be overstated as the property is used to determine the attraction between materials. Areas such as flotation1 use it to assure the adsorption of surfactants to allow the extraction of the components within the ore matrix for collection. In catalysis,2 it allows the adsorption of a metal catalyst onto its support material. Contact angles and surface energy3,4 are also affected by this property. PZC is also used in allowing the best conditions for dyeing of clothing.5 Charge storage methods using activated Carbons in Aqueous Electrolytes6 also employs this property. While this is only small sample size of this property uses, these examples make it clear its importance.1.2 Techniques for identification of structures in solution
Determining the structure of both crystalline and amorphous materials in an aqueous solution has been accomplished using multiple techniques. These range from X-ray diffraction and small-angle neutron scattering,7 Fast Fourier Infrared Spectroscopy,8 Raman and X-ray Absorption Fine Structure (XAFS) Spectroscopy,9 to Extended X-ray Absorption Fine Spectroscopy (EXAFS),10 and Nuclear Magnetic Spectroscopy (NMR).11 These techniques though are geared toward the identification of species in solution and individual and global structure factors for molecules, polymers, biological compounds, and inorganic particles in an aqueous environment.It is important to understand the importance of a materials surface structure and composition's effect on its PZC value. Work by Yoon et al.,12 Leffler et al.,13 Lutzenkirchen14 and Auer et al.15 demonstrated that the surface structure is one of the critical factors responsible for PZC values. Work by Di Paola et al.16 found that changing the surface composition of TiO2 using different surface dopant atoms, shifted the material's PZC values significantly, while not affecting the global surface structure of the catalyst support (i.e. TiO2). Work by Suchanek et al.17 also demonstrated the same effect of changing the surface composition of 3% yttrium doped zirconia (YSZ) by doping its surface with neodymium (Nd) and gadolinium (Gd). X-ray diffraction patterns demonstrated that the bulk structure remained constant while PZC values of the doped surfaces shifted upwards of 1 pH unit away from the PZC of the undoped YSZ surface. Taken together, these works demonstrate that the surface composition and structure at the solid/aqueous interface are the critical factors determining a material's PZC value.
At present none of the available methods for determining the global surface structure and composition of a material can be used in an aqueous environment. It has been demonstrated though that PZC is determined solely by the material's global surface structure and composition at its solid/liquid interface.12–14,16,17 Therefore, knowledge of a material's PZC value was determined to be ideal for modelling its global surface structure and composition. This was the impetus for developing the augmented model12 and the new method presented in this work to model the material's global surface structure, composition and properties (i.e. bond ionic content) at the solid/aqueous interface using its PZC.
1.3 Structure and composition of the Stern Layer, outer helmholtz layer and diffuse region (general solution) around a metal-oxide surface
When constructing a model, it is critical to understand the physical system under consideration. An examination of the literature on metal-oxide/hydroxide surfaces in an aqueous environment reveals a complex interaction between its surface structure, composition and the makeup of the solution. Therefore, to draw the correct assumptions for a model describing a given system it was imperative to obtain the full understanding of how the entire system functions.Beginning with Livey and Murray's18 work on the geometry of solid oxides and carbide surfaces, they determined that oxygen atoms at the surface screen the cations to achieve the lowest possible surface energy. It is accomplished by the oxygen pulling up and over the metal cations. This results in the surface metal cations being situated below the level at which the surface oxygens are located, having retracted downward toward the interior bulk structure.
Work by Leffler et al.'s13 findings support Livey and Murray's18 surface model. They determined that above a diameter of approximately 29 nm for anatase titania (Region I), the surface structure, PZC, surface charge, and bond ionic content remain constant. In Region I, the surface has the highest measurable PZC value (i.e. pH ≅ 7.17) of this material. They also determined that electrons transferred to the surrounding oxygen atoms appear to bow back towards the metal cations, acting to partially shield them, in the manner described by Livey and Murray.18
Declining particle size below a diameter of approximately 29 nm (Region II), pHPZC values decreased almost linearly with increasing surface bond lengths, indicating that the global percentage of the of the surface which is positive increased significantly. This demonstrated that the metal cations below the oxygen atoms were moving upward toward the surface with decreasing particle size. At a given particle size the metal cations would most likely be situated at approximately the same elevation as the surface oxygen.
Recent work by Brown et al.,19 using a micro-jet, coupled with X-ray Photoelectron Spectroscopy (XPS) determined the structure and composition of the Stern Layer, Outer Helmholtz Plane (OHP) and Diffuse Region (general solution) above silica (SiO2) metal-oxide particles [Fig. 1]. Their work focused on how the cations of four different electrolyte solutions, LiCl, NaCl, KCl, and CsCl were located with respect to the Stern layer, OHP and Diffuse Layer (general solution). Lastly, they determined the composition of the diffuse region (general solution).
 | ||
| Fig. 1 The structure and composition of the Stern layer, outer Helmholtz plane and diffuse region (general solution) at and above the surface of a silicon dioxide (SiO2) particle. [After Livey and Murray,18 and Brown et al.19]. The positioning of the oxygen to silicon atoms at the surface are in accord with Livey and Murray.18 | ||
They determined that the Stern layer was a single layer of water molecules in various orientations with only the hydrogen atoms on the water molecules being adsorbed onto the surface (i.e. in contract with). This single layer of water molecules is bounded by the Outer Helmholtz Plane (OHP). At the line defining the OHP layer they identified only hydrated cations from each electrolyte, which would most likely be neutralizing the charge on the oxygen of the adsorbed water molecules. Beyond the OHP, in the diffuse region (general solution) they identified water, hydrated cations and hydrated anions. In addition to Brown et al.'s findings,19 a small amount of water is also known to dissociate into hydroxyls (O–H−) and hydronium ions (H+)20 ergo they would also be present in the diffuse region (general solution).
The findings of Brown et al.19 indicated that the water molecules and hydrated cations of the dissociated electrolyte molecules along the OHP, neutralized the negatively charged fraction of the surface (i.e. oxygen). Therefore, the remaining portion of the un-neutralized surface charge would be due to the surface cations. Since the surface cations sit below a surface dominated by oxygen atoms and are surrounded by oxygens in their primary coordination sphere this indicates that each of the positive regions created by these cations at the surface are isolated from each other. This suggests that the surface cations effectively function as independent point charges at the solid/aqueous interface.
Since Brown et al.'s19 work demonstrated that hydrated anions are found only in the diffuse region (general solution) this indicates that they come from the dissociated electrolyte (LiCl, NaCl, KCl, and CsCl) and titrating acid or base solutions (i.e. H+–Cl−, Na+–OH−, etc.). The dissociated water molecules (H+–OH−) would most likely also provide another source of negatively charged ions. All these together would then be responsible for neutralizing the surface cations charge. Therefore, each of these ions would be electrostatically adsorbed above the hydrated cations at the OHP, in the diffuse region (general solution).
This suggests that the concentration of H+ ions from the dissociated water molecules (i.e. H+ and OH−) and acid titrating solutions (i.e. H+–Cl−) are what is measured to obtain the materials pHPZC below a pH = 7. Above a pH = 7, the titrant used would be a base, such as NaOH, which dissociates into Na+ and OH−. The anions from the electrolyte (LiCl, NaCl, KCl, and CsCl), base (i.e. NaOH) and dissociated water would then be adsorbed above the OHP to neutralize surface cations. This suggests that the H+ ions which constitute the measured pHPZC value are only from the dissociated water molecules. Therefore, the smaller source of H+ ions most likely explains why a pH ≥ 7.0 is measured.
1.4 Adsorption models predicting point of zero charge
The mathematics for each model developed to predict PZC values are based on the concept of the change in Free Energy of the surface [eqn (1)]12 as protons (H+) and hydroxyls (O–H−) adsorb onto the surface out of solution.| ΔG0 = ΔGel + ΔGch | (1) |
Eqn (1) can be rewritten in the manner of eqn (2) or (3) (ref. 12)
 | (2) |
| ν = ZM/M |
| L = L + r |
ΔG0 = −2.3RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K K
| (3) |
Multiple models were considered for use in this work. One important model considered was the Multisite Complexation (MUSIC) model developed by Heimstra et al.21 Many of the other models available are essentially iterations of Heimstra et al.'s21 basic concepts. The basis of these models is that all the adsorption sites at the surface behave as a single system. Therefor the equilibrium constant(s) (K) for the surface represents the value for the entire surface. In addition, the MUSIC model21 incorporates different K values for metal-oxide and -hydroxide surfaces.
These models though do not fit the physical system determined by Brown et al.19 They demonstrated that (1) the oxygen atoms are fully neutralized by water molecules and that the hydrated electrolyte cations, appear to neutralize the adsorbed water molecule's oxygen charge at the surface of the Stern layer. The remaining source of unresolved charge is from the surface cations which behave as separate point charge sources due to their position below the oxygen atoms. In addition, the multisite adsorption complex models cannot model complex metal-oxide and -hydroxide surface structures or phases in Region II, where the pHPZC decreases below Region I. Therefore, they could not be used for the modelling objectives in this work.
The model which does treat each cation adsorption site as an individual point source was developed by Yoon et al.12,21 They expanded the initial model developed by Parks,16 incorporating the material's entire crystal structure and composition into their model. In addition, their model allows the calculation of a predicted point of zero charge (PPZC) for both simple and complex metal-oxide and -hydroxide values. Finally, their model most closely resembles the physical system determined by Livey and Murray,18 Brown et al.19 and Leffler et al.13
To model the surface structure of a simple or complex metal-oxide/–hydroxide, it is necessary for each of the variables in the model to be a quantifiable parameter. These include physical structure factors and/or surface properties. In addition, the know structures of the material need to be identified. Multiple metal-oxides/hydroxides have multiple structures, such as mercuric oxide (HgO) which possess coordination numbers (C.N.) of both 2 and 6 (ref. 12 and 22–24) Therefore, the objectives of this work were to: (1) eliminate any non-structure factors used in Yoon et al.'s model eqn (2),12 (2) identify, confirm and incorporate any unidentified physical parameters of the material (i.e. surface properties), (3) identify all the possible crystal structures and their structure factors at equilibrium, and (4) develop a method for applying the model to determine which is the most likely global surface structure present at its solid/aqueous interface.
2 Augmentation of the model used to determine surface structure and composition from its PZC
The crystal structure factors included in Yoon et al.'s12 model are metal–oxygen bond length(s) (L), coordination number(s) (M), bond valence(s) (ν), metal oxidation state(s) (ZM) and the fraction of Dangling surface bonds per metal cation (fi) being modelled. The variable which is not a crystal structure factor is L = L + 1.01 Å. It is the average metal–oxygen bond length (L) (i.e. M2+–O2−) for each of the cations in the structure, with the addition of a hydroxyl (i.e. O–H1−) bond length. This hydroxyl is formed when hydrogen ions (H+) in solution adsorbs and/or bonds to oxygens bonded to the central cation. The reason O–H−1 (r = 1.01 Å) is not considered part of the bond length L is because it is not part of the primary coordination sphere surrounding the metal cation which is defined by its coordination number (M). Rather the adsorbed H+ is considered part of the secondary coordination sphere which surrounds the primary coordination sphere. These consist of ligands that are attached to the ligands in the primary coordination sphere.25 Since the H+ is part of the secondary coordination sphere the O–H− is a separate bond from the M2+–O2− bond and therefore their lengths cannot be added and treated as a single bond length.To illustrate the effect of adding the hydroxyl's bond length to the average metal–oxygen bond length (L) in their model12 it was found to shift the predicted pHPPZC to a more basic value. An example of the use of this variable in their model with (L) and without (i.e. only L) comes from Table I in Yoon et al.'s12 work. The experimental PZC value for SiO2 is with a pHPZC = 1.80. Using L and all the other structure factors in Table I12 for SiO2 in the model, a pHPPZC = −1.85 is obtained. Replacing L with the structures average bond length L, results in a pHPPZC = −14.56. Therefore, it appears that by using the extended bond length L in the model compensated for the lower pHPPZC values predicted without it.
This variable (L) also poses a problem with regards to the findings of Leffler et al.13 They determined that pHPZC values shift toward significantly lower pH values as the surface M2+–O2− surface bonds expand at the anatase titania solid/aqueous interface in Region II.13 This indicates that there is a physical parameter absent from Yoon et al.'s original model12 that would perform the same function as the L variable and account for changes in surface bond lengths. Therefore, it was decided to remove the variable L from the model, replace it with the structures average bond length (L) and then determine the physical property not accounted for in the original model.
Leffler et al.13 identified the property that might replace the L variable and account for the effect of changes in surface bond lengths effect on the materials PZC value. It is based on the knowledge that ionic/covalent bonds form an electric dipole which is defined by its dipole moment µ (Coulombmeters). As the ionic/covalent bond length expands/contracts its dipole moment increases/decreases, resulting in a shift in the ionic character of the bond. The ratio of an ionic/covalent bond's observed dipole moment (µo) with its ideal dipole moment (µI = 100%) provides the bond's percent ionic character (I% = µo/µI).26
Leffler et al.13 identified the surface ionic/covalent bond(s) in Region I as having observed dipole moment(s) (µo) as they are smaller than the bulk structure bond(s) due to their shorter length.27 Bulk structure bonds possess the maximum possible length(s) within the structure, giving them the greatest possible dipole moment(s). This identifies them as possessing the ideal dipole moment (µI = 100%). Therefore, in Region II, where the surface bonds expand toward those in the bulk structure,27 this shifts the observed dipole moment(s) toward the ideal dipole moment. The result is that the surface bond's ionic character increases toward an ionic character (I%) = 100%.
Use of the bonds ionic content in the augmented model12 is supported by work of Bickmore et al.28 They presented the mathematical justification for a method incorporating a bonds ionic content into the modelling of the pKa (acidity of a substance) value for solution monomers. Their work was then use by Mitchel29 to augment the MUSIC model to predict PZC values for gibbsite (Al(OH)3).
To determine if the ionic content of surface bonds can replace L in the model, the L variable was removed from Yoon et al.'s model12 and replaced with L. The original equation [eqn (2)] was then rearranged to form eqn (4), where X represents the value of the possible variable that might replace L. Structure factors and PZC values for titania were used in eqn (4). These were L = 1.969 Å, oxidation state (ZM) = +4, and the coordination number (M) = 6, which came from the example for TiO2 in Table I in Yoon et al.'s work.12 The PZC value used was determined by Leffler et al.13 in Region I, where pHPZC ≅ 7.17, as it remains constant above an average diameter ≅29 nm. The value calculated from eqn (4) gave an X = 0.6214.
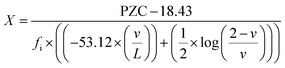 | (4) |
To determine if X = 0.6214 is equivalent to Pauling's percent ionic content (I%), eqn (5) and values from Pauling's Electronegative Scale30 were used to determine the ionic character for the Ti4+–O2− bond.
 | (5) |
| XOx = 3.5 |
| XTi = 1.5 |
The value determined from eqn (5) was I% = 0.6321. The difference between the calculated value X [eqn (4)] and Pauling's I% is 0.0107 (1.69%), which indicates that the ionic character of the bond is one of the variables responsible for determining the surface's PZC value. Therefore, X was replaced with the variable for the ionic ch of the surface bond (fI% = I%) in eqn (4). Rearranging eqn (4) gave eqn (6). Eqn (5) is used to determine the ionic character of the bonds (fI%) for each atom in the structure in Region I. Eqn (6) constitutes the augmented Yoon et al. model12 used in this work.
 | (6) |
| fI% = I% |
The calculated value for fI% = 0.6321 for anatase titania was then used to model the pHPPZC for anatase titania using eqn (6), along with the structure factors in Table I.12 This resulted in a pHPPZC = 6.97. This pHPPZC value is within the error range (pH = ± 0.2) of the Hanna pH meter used by Leffler et al.13 to obtain their experimental PZC values. These results demonstrate that the ionic content of the bond is the correct physical property to replace L in the original Yoon et al. model.12
3 Development of the modelling method
3.1 Determination of surface structure in an aqueous environment
The method developed for this work is modelled after the approach used for the Rietveld Refinement method31 used for determining the structure of a material's X-ray diffraction (XRD) pattern. The initial XRD pattern is obtained for a known phase. The composition and atomic positions are placed in a control file from which an XRD pattern is calculated. The program then calculates the differences between the experimental and modelled pattern. Each variable in the control file is then altered within the given parameters of each value, to calculate a pattern closest to the experimental pattern that is being modelled. The goodness of fit (R2) determines how close the experimental and modelled patterns are. This same approach alters the modelled PZC values within their allowed physical parameters (i.e. bond lengths, composition, bond ionic content, and coordination number) until the difference between the two values is equal to or less than 0.5 pH units. When the model cannot converge, it indicates that the structure in the control file does not match the experimental XRD pattern. This is equivalent to the modelled pHPPZC being greater than 0.5 pH units than the experimental pHPZC.The structure information used by Parks32 and Yoon et al.12 in their modelling work is for material not in an aqueous environment. When a metal-oxide or -hydroxide is placed in an aqueous environment, the global surface crystal structure and composition may not be the thermodynamically preferred phase present in the dominant species phase in the solution at its measured pHPZC value. This might result in the surface structure and/or composition transforming to that of the dominant species phase in the solution at the pHPZC.
To test this possibility, each of the phases presented in Yoon et al.'s work (Tables I–III)12 were compared to the dominant species at their pHPZC values, using their Pourbaix Diagrams.33–37 In Table I12 six of the phases were found not to be the dominant species phase at their pHPZC. In Table II12 two samples varied from the dominant species phase at the material's pHPZC values. In some cases, both metal-oxides and their -hydroxide counterparts are stable in the same pH range. When this occurred, secondary information was consulted to identify the most likely surface structure at the material's pHPZC. Once the dominant species phase was identified it replaced the phase modelled in Tables I–III.12 Examples from Tables I–III12 are presented in Sections 3.1.1–3.1.3. The remaining phases from Tables I and II not presented Tables 1 and 2 are included in the SI Section A. Explanations for how and why certain phases were replaced with the dominant species phase are presented after each table and for those tables contained in the SI Section A.
| Sample | Dominant species | Experimental point of zero charge (pH) |
|---|---|---|
| MgO | Mg(OH)2 | 12.4 |
| La2O3 | La(OH)3 | 10.4 |
| ZrO2 | Zr(OH)4 | 10.0–11.0 |
| α-AlO(OH) | Al(OH)3 | 7.7 |
| γ- AlO(OH) | Al(OH)3 | 7.5 |
| Y2O3 | Y(OH)3 | 9.0 |
| Sample | Dominant species | Experimental point of zero charge (pH) |
|---|---|---|
| Fe(OH)2 | Fe(OH)3 | 12.0 ± 0.5 |
| Ni(OH)2 | NiO | 11.1 |
| Cu(OH)2 | CuO | 7.7 |
3.1.1.1 Magnesium hydroxide (Mg(OH)2). Magnesium hydroxide is more thermodynamically stable than magnesium oxide (MgO)35,38 in an aqueous environment. The Pourbaix diagrams present a pH stability range for Mg(OH)2 from ≅9–16.33–35 In the Pourbaix diagrams for magnesium, only Mg(OH)2 is present in all the Eh-pH fields examined. Based on this information, it was determined that the surface of magnesium oxide most likely transforms to its hydroxide phase in an aqueous environment at its pHPZC value.
3.1.1.2 Lanthanum hydroxide (La(OH)3). When exposed to moisture at ambient temperature and pressure La2O3 rapidly transforms to La(OH)3 making it the thermodynamically preferred phase39. This explains why La(OH)3 is the dominant species in the Pourbaix diagrams33–35 examined. Therefore, the experimental PZC value given for La2O3 in Table I12 is the most likely surface structure of lanthanum hydroxide in an aqueous environment.
3.1.1.3 Zirconium hydroxide (Zr(OH)4). According to multiple Pourbaix diagrams,33–35 there are two stable dominant species present at a pHPZC = 10–11,12 these are ZrO2 and Zr(OH)4. While ZrO2 is stable in an aqueous environment, the undoped meta-stable tetragonal phase (C.N. = 8) was used to model the pHPPZC in Table 1.12 Meta-stable tetragonal ZrO2 phase though, rapidly transforms to its most stable form at ambient temperature and pressure, which is the monoclinic structure with a C.N. = 7.40–42 The lower coordination number would shift the predicted value toward a lower pHppzc in this model [eqn (6)]. This is supported by the literature value for monoclinic zirconia of pHPZC = 8.5,43 which comes from a sample with a particle size of 0.1 µm, placing it within the material's Region I.13,44
The second dominant species in the Pourbaix diagrams examined was for zirconium hydroxide [Zr(OH)4, (ZrO2·2H2O)].33–35 This phase has a C.N. = 8.45 As the coordination number in the model [eqn (6)] increases, the predicted PZC value shifts towards a more basic pHPPZC. Other PZC values measured for zirconium hydroxide are also in the range of pH = 10–11.45,46 Therefore, the structure modelled in Table I12 in the pHPZC = 10–11 range is most likely zirconium hydroxide.
3.1.1.4 Aluminium hydroxide (γ – Al(OH)3/α – Al(OH)3: gibbsite). There are four phases of aluminium hydroxide, gibbsite, diaspore, boehmite, and bayerite.47 The phase with the most stable free energy of these four aluminium hydroxides is gibbsite (hydrargillite).35 The Eh–pH diagram for aluminium hydroxide also shows only the gibbsite structure in the pH range from pH = 4–11.48 Therefore, the examples of diaspore (α-AlO(OH)) and Boehmite (γ-AlO(OH)) in Table I were not modelled as their surface structure had most likely transformed to the gibbsite phase.
3.1.1.5 Yttrium hydroxide (Y(OH)3). Yttrium oxide (Y2O3), when placed in an aqueous environment, rapidly hydrates and transforms to yttrium hydroxide (Y(OH)3).49 This makes it the thermodynamically preferred structure in the presence of moisture. In addition, only yttrium hydroxide (Y(OH)3) is present in the Pourbaix diagrams33–35 examined and are present in a pH range of 6.5 to 16. Therefore, yttrium hydroxide was most likely the surface structure present in the aqueous environment at its pHPZC = 9.0.
3.1.2.1 Iron hydroxide (Fe(OH)3). The Pourbaix diagrams33–35 consulted indicate that Fe(OH)3 is the dominant species at the pHPZC value presented in Table II.12 This is supported by the thermodynamic data, for the transformation equation: 4Fe(OH)2 + O2 + 2H2O => 4Fe(OH)3 that gives a Gibbs free energy (ΔG) of −91.53 kJ mol−1.50 Therefore, it indicates that in an aqueous environment Fe(OH)2 spontaneously transforms to Fe(OH)3.
3.1.2.2 Nickel oxide (NiO). Until recently, the pHPZC value for NiO had been identified as Ni(OH)2 being the dominant species.33–35 Recent work though has redefined the dominant species as NiO.51,52 While the phase boundaries in these Pourbaix diagrams vary, they overlap from pH = 9.0 to 12.5. This range includes the most reliable basic value for NiO at a pHPZC = 11.1.53
3.1.2.3 Copper oxide (CuO). Copper hydroxide (Cu(OH)2) is a meta-stable phase which transforms to the stable CuO at ambient temperature and pressure in a basic aqueous solution (pH > 7).54 Pourbaix diagrams for copper,33–35 under these conditions, indicate that copper oxide (CuO) is the dominant stable species up to a pH ≅ 12.8. The most basic PZC value found in the literature is for a pHPZC = 10.0,55 which lies within this range of values. Therefore, the surface of the Cu(OH)2 would most likely have transformed to CuO at a pHPZC > 7.
While SiO2 remains stable in this pH range [0–12.5],33 aluminium oxide hydrates to its stable form Al(OH)3.57 An examination of the difference between the ionic radii for OH1− and O2− at coordination numbers of 4 and 6, determined that they are effectively equal.22 Therefore, based on this information, the surface and bulk bond lengths for the Al–O sites, in these three complex oxides, are the same as the hydroxide (i.e. Al–OH) in an aqueous environment at their pHPZC values. Therefore, the surface Al3+ was modelled with an oxide bond (i.e. Al3+–O2−).
3.2 Modelling results
Where the pHPZC and pHPPZC values were greater than pH = ±0.5, literature values for each phase's structure factors such as bond length and coordination number were examined. After identifying the structure factor(s) which varied from the information in Table I,12 these different structure factor(s) for the material were replaced in the model. The final structure values, pHPZC and pHPPZC values presented in Table 1 are given in Table 4. Explanations for each phase where a structure factor and/or pHPZC value was replaced are presented after Table 4. The phases in Table I, not included in Table 4 and their explanations are given in SI Material B.
3.2.1.1 Lanthanum hydroxide (La(OH)3). While La2O3 has a C.N. = 6,12 La(OH)3 has a C.N. = 9 and an average bond length of L = 2.566 Å.22 The most basic experimental PZC value found for La(OH)3 is a pHPZC = 12.40.1 When the structure factors were placed in the model, it [eqn (6)] predicted a pHPPZC = 12.90, with a difference of 0.5 pH units.
3.2.1.2 Yttrium hydroxide (Y(OH)3). The structure of Y(OH)3 has a C. N. = 9 (ref. 62) and an average bond length L = 2.43 Å.22 The most basic pHPZC available in the literature is for a pH = 12.8.63 The pHPPZC = 12.83, a difference of only 0.03 pH units.
3.2.2.1 Iron(III) hydroxide (Fe(OH)3). The dominant species found on the Pourbaix diagram for iron is Fe(OH)3. The common coordination number for iron is six coordinated,12 with an average surface bond length of L = 2.09 Å in an aqueous environment. This is longer than the bond length of 1.99 Å measured using XRD for the dry powder of Fe(OH)3.64 When these structure factors, which included an oxidation state (ZM) = +3 were placed in the model [eqn (6)] a pHPPZC = 11.65, a difference of 0.35 pH units from pHPZC = 12.0 ± 0.5 of the experimental value was obtained.
3.2.2.2 Nickel oxide (NiO). Using the structure factors provided by Yoon et al.12 resulted in a pHPPZC = 13.68, more than two pH units above the experimental value of pHPZC = 11.10. Based on the most current Pourbaix diagrams,51,52 NiO was identified as the dominant species, not Ni(OH)2, which ruled out changing the oxidation state structure factor. This indicates another structure factor might vary from the bulk structure.
This was found to be the case. It was determined that while the bulk crystal structure of NiO has a coordination number of 6, work by Che and Bonneviot65 demonstrated that at the [1 0 0] crystal face of NiO the surface atom, Ni2+, has a square pyramidal symmetry (C.N. = 4). Since NiO has a simple rock salt cubic structure,69 each crystal lattice face is identical, and all the Ni2+ atoms are four coordinated.70 They also found that corners and edges present lower coordination numbers of 3 and 4. Therefore, replacing C.N. = 6 with a C.N. = 4 and calculating the average bond length for this value (L = 1.91 Å)22 resulted in a pHPPZC = 11.04, only 0.06 pH units different than the experimental value.
3.2.2.3 Copper oxide (CuO). Copper oxide is the thermodynamically stable structure with its most basic pHPZC = 10.0.66 Therefore, its oxidation state is +2. Using the bulk structure factors from Yoon et al.12 (C. N. = 4) resulted in a pHPPZC = 11.91. Reducing the C.N. value to 2 results in a pHPPZC = 5.87, 4.03 pH units below the materials PZC value. Modelling work by Maimaiti et al.67 using Density Functional Theory (DFT) predicted a C.N. = 3 for its surface structure with an average L = 2.024 Å.68 Using these values in the model resulted in a pHPPZC = 10.01, a difference of 0.01 pH units.
Each of the pHPPZC values calculated using eqn (6), are presented in Table 7. The pHPPZC values all fall within the range of experimental values presented in Yoon et al.'s work12,58,59 and within ±0.6 pH units of the average of these experimental values. Therefore, no changes to the structure factors values were considered necessary for these three aluminosilicate samples. These results also confirm that the surface structure and composition are not affected by an aqueous environment.38,56
A comparison of the two experimental pHPZC values58,59 though suggests that surface composition and structure may have varied from the bulk structure. These variations in the surface may have been due to processing work (i.e. grinding) exposing different cleavages with slightly different compositions or contamination.71 Based on the model's sensitivity to slight changes in each of the structure factors used to determine a pHPPZC, one or all these factors might explain the larger variation than pH = ± 0.5 from some of the materials average pHPZC in Table 7.
The measured pHPZC values were for the surface of a 10 × 20 mm2 hematite crystal electrode. The initial pHPZC value was measured right after its surface had been freshly fractured. Following each measurement, the electrode was then cleaned with ethanol and milliQ-water. The sample was then aged in water for 30-minutes intervals, eight times (240 min. total) at 600 °C. Dividing up the total period from 0 minutes to 240 minutes, gave intervals of 80 minutes between each PZC measurement. What they found was a significant shift in the PZC values of the hematite electrode's surface, from the initial pHPZC = 3.85 to their steady state value of pHPZC = 8.80.
The most basic pHPZC found in the literature is from work done by Klein Wolterink et al.72 Their hematite powder was aged for 480 minutes (8 h) at 120 °C under 1.2 atmospheres. The extended aging time resulted in a pHPZC = 9.80. This value is most likely closest to the final equilibrium PZC for the hematite surface in Region I.13 The equilibrium structure for hematite in Region I comes from the work by Jones et al.73 They determined that the most stable surface structure on the most stable crystal face [0 1 1] of hematite exhibited a five coordinated Fe3+ atom. When placed into eqn (6), the model converged to a pHPPZC = 9.92, only 0.12 pH units above Klein Wolterink et al.'s72 measured PZC value. While the final value measured by Lutzenkirchen et al.14 was more than 1.1 pH units below Klein Wolterink et al.'s.72 Therefore, the value obtained by Klein Wolterink72 was placed in Fig. 2, as it appears to be the equilibrium PZC value for hematite.
 | ||
| Fig. 2 The point of zero charge for the hematite surface after aging against the time interval at which it was measured.14 The data point at 480 min. is for the equilibrium PZC value in Region I.72 | ||
The first step in modelling the hematite electrode's surface was to determine the initial coordination value for the F3+ atoms on the freshly fractured hematite electrode. This was obtained from work by Schottner et al.74 They used adsorbed carbon monoxide (CO) to determine the oxygen coordination for the surface Fe3+ atoms of a freshly fractured pristine hematite surface. Their results determined that the Fe3+ atoms had a coordination number of three oxygen atoms surrounding them. Work by Jones et al.73 identified a bond length for the three coordinated Fe3+ of L = 1.88 Å. Placing these structure factors into the model [eqn (6)] gave a pHPPZC = 3.89, only 0.04 pH units different from the experimental value presented in Fig. 2 and `s C1 and C2 [SI Section C]. The model [eqn (6)] also confirmed the experimental findings by Schottner et al.74 that the surface for a pristine, newly fractured surface has an Fe3+ atom with a C.N. = 3 (ref. 12, 73 and 74) in an aqueous environment.
The critical information explaining how the surface structure changed also comes from Lutzenkirchen et al.'s14 work. Their examination of the freshly fractured electrode revealed that the surface had two doubly coordinated hydroxyl groups present. This suggests that the oxygen of these doubly coordinated hydroxyls might have been sitting in oxygen vacancies adjacent to but not bonded to the Fe+3 atoms. As the PZC values shifted, they found that the number of surface hydroxyls decreased from two to one. This suggests that an oxygen atom from each of the doubly coordinated hydroxyls bonded with Fe3+ atoms. It is supported by the increased roughness of the surface, which suggests it was due to a newly formed Fe3+–O2− bonds extending upward from the surface.
To determine the most reasonable bonding sequence responsible for the change in PZC values [Fig. 2] two different models were executed. Since hematite is known to exhibit 4 coordinated sites,75 the first model was for a bonding sequence of Fe3+ sites shifting in a sequence of C.N. = 3 => C.N. = 4, then 4 => C.N. = 5. These results are presented in Fig. 3 and Table C1 [SI Section C]. The second bonding sequence modelled is for the C.N. = 3 going directly to a C.N. = 5. These results are presented in Fig. 4 and Table C2 [SI Section C].
Fig. 3 and 4 were compared with Fig. 2. The results in Fig. 2 and 4 demonstrate a parallel shift in their curves over time. Therefore, these results suggest the sequence responsible for the shift in PZC values is presented in Fig. 4 and Table C2 [SI Section C] where the C.N. = 3 => C.N. = 5.
The argument for choosing the Fig. 4 sequence is that the two double hydroxyls identified in the surface structure14 were probably sitting in the oxygen vacancies next to each Fe3+ atom. Therefore, the simplest and most expedient path available to drive surface energy to its lowest value [18] would be for the double hydroxyl oxygens to bond directly to the Fe3+ atoms. Whereas the C.N. = 3 => C.N. = 4 => C.N. = 5 would most likely have been reflected in a different set of shifts in PZC values over time and might result in a higher surface energy during this transition.
3.3 Determination of the anatase titania bond ionic character in regions I and II
In Section 2 the bond ionic content in Region I for anatase titania was calculated using eqn (4), the structure inputs from Table I12 and the pHPZC from work by Leffler et al.13,76 Their work also demonstrated that in Region II, surface bond lengths (i.e. Band gaps) expand with decreasing particle size. Therefore, it was necessary to obtain the average surface bond length as the average primary particle size decreased. This would allow use of the correct surface bond length (L) be used in eqn (4) as well as the samples pHPZC.This was accomplished by obtaining the average bond length within the bulk structure for anatase titania. Work by Pantaleone et al.77 provided both the average bond length for anatase titania at the surface [L ≅ 1.8 Å–1.9 Å] and within the bulk structure [L ≅ 2.1 Å]. Assigning the bond length [L = 1.986] from Yoon et al.'s12 Table I for anatase titania to the surface in Region I at d = 29 nm, and L ≅ 2.1 Å at d = 3.30 nm76 the simple equation for a line was fitted to these two points [eqn (7)].
| y = −0.0051x + 2.1168 | (7) |
The bond lengths calculated using eqn (7) are presented in Table C1 [SI Materials C], along with the bond ionic content [eqn (4)] pHPZC, and average primary particle diameter13,76 for each sample.
Surface bond ionic content was then plotted against their average primary particle diameter and is presented in Fig. 5. What is most interesting is that the bond ionic content of particle (d = 3.30 nm) where superconductivity occurred76 has an I% = 99.5%, indicating surface bonds were essentially purely ionic. The calculated bond length [eqn (4)] where I% = 100% for the particle where superconductivity occurred is at L = 2.115 Å, a difference of only 0.71%. An interesting note, while the surface bond lengths were calculated using the equation for a line, the curve for the percent ionic content presents the same form as the pHPZC values in Region II,13 a polynomial.
 | ||
| Fig. 5 Modelled bond ionic character of anatase titania against their average primary particle diameters from literature values.13,76 | ||
Lastly, the bulk bond length can also be used to determine the lowest pHPZC where the surface bonds are fully ionic. This is accomplished by using all the structure factors in Region I, save the average surface bond length (L) and the bond ionic content determined from Pauling's electronegative table and eqn (4). To obtain the pHPZC value where surface bonds are fully ionic, f%I = 1.0 and L = Average bulk structure bond length (2.115 Å).
4 Discussion
Prior to this work, most of the models predicting PZC values used the central premise, that all the adsorption sites at the surface interacted with each other. This required determining global equilibrium constant(s) for different surfaces using various methods. Work by Livey and Murray,18 Brown et al.19 and Leffler et al.13 though demonstrated that surface oxygens dominate the surface and are fully neutralized by water molecules in the Stern layer. Then the hydrated cations from the electrolyte at the outer Helmholtz plane neutralize the charge on the water's oxygen adsorbed on to the surface.The anions from the dissociated electrolyte, titrant and water molecules are most likely responsible for neutralizing the cations at the surface, adsorbing above the OHP over the surface cations in the Diffuse Region (general solution). The PZC value below a pHPZC ≤ 7 is determined by measuring the H+ ions from the dissociated acid titrant and water. In the basic region (i.e. pHPZC ≥ 7) the O–H− from the dissociated base and water molecules are what most likely neutralize the surface cations. Only the H+ ion concentration from dissociated water molecules is what is measured to determine the material's PZC value.
Prior to this work, modelling PZC values for simple and complex metal-oxides and –hydroxides were carried out under the assumption that their bulk and surface structures were identical.12,32 Based on Pourbaix diagrams of each simple and complex metal-oxides or -hydroxides suggests they may possess a structure which, at times, is independent of their bulk phase when exposed to an aqueous environment at different pH values. Therefore, from the literature and modelling results presented in Tables 1–7, and SI Tables A1–D1, it has been demonstrated that in an aqueous environment the surface structure being modelled can vary significantly from the bulk structure.
| Sample | Dominant species | Experimental point of zero charge (pH) | Experimental point of zero charge (pH) |
|---|---|---|---|
| Kyanite | Kyanite | 7.9 | 6.9 |
| Andalusite | Andalusite | 7.2 | 5.2 |
| Sillimanite | Sillimanite | 6.8 | 6.0 |
| Dominant species | C.N. | ZM | L (Å) | fI% (%) | fi | =fi× I% | Exp. pHPZC | Cal. pHPPZC | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Mg(OH)2 | 6 | +2 | 2.06 | 0.7335 | 1 | 0.7335 | 12.0 | 11.87 | 12 and 60 |
| La(OH)3 | 9 | +3 | 2.566 | 0.7631 | 1 | 0.7631 | 12.40 | 12.90 | 12, 1, 22 and 61 |
| Zr(OH)4 | 8 | +4 | 2.26 | 0.6680 | 1 | 0.6680 | 10–11 | 10.422 | 12, 45 and 46 |
| Y(OH)3 | 9 | +3 | 2.43 | 0.7335 | 1 | 0.7335 | 12.80 | 12.83 | 12, 62 and 63 |
| Sample | I% | =fi× I% Si–O(IV)a | =fi× I% Al–O(IV)a | =fi× I% Al–O(VI)a | =fi× I% Al–O(VI)a | Exp. pHPZC | Exp. pHPZC | Avg. Exp. pHPZC | Calc. pH PPZC |
|---|---|---|---|---|---|---|---|---|---|
| a Coordination number. | |||||||||
| Kyanite | Si = 0.5145 Al = 0.6321 | 0.128 | 0.4742 | 6.9 | 7.9 | 7.4 | 7.58 | ||
| Andalusite | Si = 0.5145 Al = 0.6321 | 0.137 | 0.211 | 0.253 | 5.2 | 7.2 | 6.2 | 6.79 | |
| Sillimanite | Si = 0.5145 Al = 0.6321 | 0.135 | 0.181 | 0.270 | 5.6 | 6.8 | 6.2 | 5.74 | |
There are several modelling methods that look at material systems surfaces. The first is the Molecular Dynamic (MD)78 modelling method which focuses on the motion of molecules and atoms within a system. Work by Brown et al.'s19 and Leffler et al.'s13 on the interface structure though shows they possess a stable structure at the solid/aqueous interface. This is because surface molecules and ions are electrostatically adsorbed (i.e. attached). In addition, no literature using this method was ever located that provided the needed surface structure and composition at the solid/water interface. Therefor this method found not to be applicable in determining the stable surface structure and composition at its solid/aqueous interface.
A second modelling method of a surface's structure and composition is the Density Functional Theory (DFT). This method is a quantum mechanical method for modelling stable multi-body systems, making it ideal for determining possible surface structures in an aqueous environment.79 Were the solid/aqueous structures not stable, measurements such as reproducible zeta potential curves that converge at a zeta potential of 0 mV where the particle move through an electric field at differing pH values and electrolyte concentration would not be possible. In addition, the literature searches for surface structures all produced models using DFT. These were for copper oxide (CuO),68,80 and hematite (α-Fe2O3)81 and were used as possible surface structures which varied from the materials bulk structure at their PZC in the model [eqn (6)].
Work by Leffler et al.13,76 found that the surface bonds in Region I all have the essentially the same bulk indirect band gap value (∼3.2 eV) for anatase titania. This indicates that they are all essentially the same length above a particle diameter of ≅29 nm. In addition, the material's bond ionic content (fI%) determined using eqn (4) (ref. 30) and presented in Fig. 5 are constant in Region I. Further, these values (fI%) were determined to be essentially equal to what is obtained using Pauling's eqn (5) and his table of electronegative values30 in Region I for TiO2. Therefore, determination of a material's surface structure needs to be done using the pHPZC value in Region I, as all the structure factors needed are available.
Once the surface structure in Region I is determined, eqn (4) can be employed to determine the ionic content of the surface bonds (fI%) in Region II, using the method described in Section 3.3. This makes it possible to model the bond ionic content for the surface bonds for each average primary particle in Region II. At present, though the limitation for determining the ionic character of the surface bonds in Region II using this method can only be applied to simple metal-oxides and -hydroxides.
Based on the findings in this work it has been demonstrated that the augmented model developed in this paper is applicable to both simple and complex metal-oxides and -hydroxides, but only in Region I. At present the modelling of surface bond ionic content in Region II is restricted to simple metal-oxides and -hydroxides, as demonstrated in Fig. 5 and Table D1 in SI Section D. The reason for this is that at present it is not known if all the metal oxide bonds for different cations in complex metal-oxides and hydroxides expand linearly. Future work will focus on how each of the bonds for complex metal-oxides and hydroxides change in Region II.
Based on the methodology developed for this work, it is evident that it is not a stand-alone method. To obtain reliable results the user must be able draw from multiple areas such as thermodynamics, crystallography, X-ray diffraction, electrochemistry, Density Functional Theory modelling, etc. In addition, Leffler et al.13 developed a protocol for correctly measuring PZC values with a minimum of error. This method should be incorporated into the measurement scheme for the sample. Therefore, it is critical that when using this method all these factors be considered for the surface structure under test.
5 Conclusions
This work demonstrates a completely new method for modelling the global surface structure, and composition at the solid/aqueous interface for both simple and complex metal-oxides and -hydroxides in Region I using the material's PZC. In Region II it has been shown that as the materials PZC decreases with its average primary particle diameter, the surface bond ionic content for simple metal-oxides and – hydroxides can be measured directly. To accomplish this the model [eqn (2)] developed by Yoon et al.'s12 was augmented [eqn (6)] to determine both the structure factors for the primary coordination sphere of each cation and their bond ionic content using Pauling's electronegative table and eqn (5).30A method was then developed to apply Yoon et al.'s12 augmented model [eqn (6)] to obtain the global surface structure and composition at the solid/aqueous interface of simple and complex metal-oxides and -hydroxides in Region I.
This method requires knowledge of:
1. The correct dominant species at the material's pHPZC.
2. All the possible phases formed by this dominant species.
3. The most basic pHPZC value for the surface structure available either experimentally or in the literature.
4. The most thermodynamically stable phase present in an aqueous environment where multiple phases might be present in the same aqueous environment, and
5. All the possible bulk and surface structure information of the dominant species phase at the material's pHPZC from both experimental and/or modelling methods such as DFT.
Once this information has been acquired, each of the possible structures at the materials PZC values are entered into model until it converges to its measured experimental PZC value.
What makes this model and method so useful, is that it can be applied in multiple fields such as catalysis, flotation, electrochemistry for battery and capacitor development, remediation of contaminated regions, confirming surface models (i.e. DFT) in an aqueous environment and the treatment of clothing (i.e. dying). It can also be used to predict a materials PZC value when the average primary particle size is in its Region I for complex metal-oxides and -hydroxides over a series of compositions. This is also the only experimental means at present of determining metal-oxides and hydroxides global surface structure, composition and bond ionic content of each cation in an aqueous environment. Lastly, the PZC measurement is economical and SI and multiple analytical tests on the material to model the surface are readily available in the literature.
Future work will focus on developing this modelling method to determine the structure, composition and changes in the bond ionic content of each cation–oxygen bond in complex metal-oxides and -hydroxides in Region II. This will allow its application to complex nanoscale metal-oxides and -hydroxides with an average primary particle diameter that are under test in Region II. Lastly, an examination of whether this modelling method can be applied to phases under test using cyclic voltammetry. The reason for investigating this possibility is because the shift in voltage values translates to changing pH values at the surface of the material across this range.
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
All data used in this work will be available on request to any interested party.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d6ra00388e.
Acknowledgements
SLS thank NEUCORSE for partial support of this research.References
- S. R. Rao and J. Leja, Surface Chemistry of Froth Flotation, Second Edition, Volume 1, Fundamentals, Springer Science + Business Media, LLC, New York. 2004 Search PubMed.
- W. Wu and E. V. Shevchenko, The surface science of nanoparticles for catalysis: electronic and steric effects of organic ligands, J. Nanopart. Res., 2018, 20, 1–14 Search PubMed.
- A. M. Munshi, R. N. Singh, M. Kumar and J. P. Singh, Effect of nanoparticle size on sessile droplet contact angle, J. Appl. Phys., 2008, 103, 0843315 CrossRef.
- H. Zhang, R. L. Penn, R. J. Hamers and J. F. Banfield, Enhanced Adsorption of Molecules on Surfaces of Nanocrystalline Particles, J. Phys. Chem. B, 1999, 103(22), 4656–4662 CrossRef CAS.
- S. N. Guilhen, T. Watanabe, T. T. Silva, S. Rovani, J. T. Marumo, J. A. S. Tenorio, O. Masek and L. Goulart de Araujo, Role of Point of Zero Charge in the Adsorption of Cationic Textile Dye on Standard Biochars from Aqueous Solutions: Selection Criteria and Performance Assessment, Recent Progress in Materials, 2022, 4(2), 1–20 CrossRef.
- S. Slesinska, P. Galek, J. Menzel, S. W. Donne, K. Fic and A. Ptatek-Mielczarek, Fundamentals and Implication of Point of Zero Charge (PZC) Determination for Activated Carbons in Aqueous Electrolytes, Adv. Sci., 2024, 11, 2409162–2409172 CrossRef CAS PubMed.
- T. Megyes, S. Balint, T. Grosz, T. Radnai and I. Bako, The Structure of aqueous sodium hydroxide solutions: A combined solution x-ray diffraction and simulation study, J. Chem. Phys., 2008, 128(4), 044501–044512 CrossRef PubMed.
- H. Jiang, Z. Song, M. Ling and S. Du Z. Yang, FTIR studies of recombinant human graulocyte-macrophage colony-stimulating factor in aqueous solutions: secondary structure, disulfide reduction and thermal behavior, Biochim. Biophys. Acta, 1996, 1294(2), 121–128 CrossRef PubMed.
- A. M. Noval, D. Nishio, T. Kuruma and S. Hayakawa, Coordination and structure of Ca(II)-acetate complexes in aqueous solution studied by combination of Raman and XAFS spectroscopies, J. Mol. Struct., 2018, 1161, 512–518 CrossRef.
- L. Guo, R. H. Colby, M. Y. Lin and G. P. Dado, Micellar structure changes in aqueous mixtures of nonionic surfactants, J. Rheol., 2001, 45(5), 1223–1243 CrossRef CAS.
- H. Yang and Y. Lim, Solution Structure Determination of Four Diploptera punctata Allatostatins by NMR Spectroscopy and Molecular Modeling, Bull. Korean Chem. Soc., 2005, 26(5), 845–848 CrossRef CAS.
- R. H. Yoon, T. Salman and G. Donnay, Predicting Points of Zero Charge of Oxides and Hydroxides, J. Colloid Interface Sci., 1979, 70(3), 483–493 CrossRef CAS.
- M. Leffler, A. Mirich, J. Fee, S. March and S. L. Suib, Part I: determination of a structure/property transformation mechanism responsible for changes in the point of zero charge of anatase titania with decreasing particle size, RSC Adv., 2024, 14, 30543–30565 RSC.
- J. Lutzenkirchen, F. Heberling, F. Suplijika, T. Preocanin, N. Kallay, F. Johann and E. P. J. Weisser, Structure – charge relationship – the case of hematite (0 l 0), Faraday Discuss., 2015, 180, 55–79 RSC.
- A. Auer, X. Ding, A. S. Bandarenka and J. Kunze-Liebhauser, The Potential of Zero Charge and Electrochemical Interface Structure of Cu(111) in Alkaline Solutions, J. Phys. Chem. C, 2021, 125, 5020–5028 CrossRef CAS PubMed.
- P. A. Di, S. Ikeda, G. Marci, B. Ohtani and L. Palmisano, Transition metal doped TiO2: physical properties and photocatalytic behavior, Int. J. Photoenergy, 2001, 3, 171–176 CrossRef.
- M. Suchanek, E. Niewiara, K. Wilkosz and W. W. Kubiak, Nanopowders of Yttria-Stabilized Zirconia Doped with Rare Earth Elements as Adsorbents of Humic Acids, Materials, 2019, 12, 3915–3930 CrossRef CAS PubMed.
- D. T. Livey and P. Murray, Surface Energies of Solid Oxides and Carbides, J. Am. Ceram. Soc., 1956, 39(11), 363–372 CrossRef.
- M. A. Brown, Z. Abbas, A. Kleibert, R. G. Green, A. Goel, S. May and T. M. Squires, Determination of Surface Potential and Electrical Double-Layer Structure at the Aqueous Electrolyte-Nanoparticle Interface, Phys. Rev. X, 2016, 6, 011007 Search PubMed.
- R. Chang, Chemistry, Random House, New York, 1st edn, 1981 Search PubMed.
- T. Heimstra, W. H. Van Riemsdijk and G. H. Bolt, Multisite Proton Adsorption Modeling at the Solid/Solution Interface of (hydr)oxides: A New Approach: Model description and evaluation of intrinsic reaction constants, J. Colloid Interface Sci., 1989, 133(1), 91–104 CrossRef.
- R. D. Shannon, Revised effective ionic radii and systematic studies of inter-atomic distances in halides and chalcogenides, Acta Crystallogr., 1976, A32, 751–767 CrossRef CAS.
- L. R. Merte, M. S. Jorgensen, K. Pussi, J. Gustafson, M. Shipilin, A. Schaefer, C. Zang, J. Rawle, C. Nicklin, G. Thornton, R. Linday, B. Hammer and E. Lundgren, Structure of the SnO2(110) - (4 × 1) Surface, Phys. Rev. Lett., 2017, 119(9), 1–6 CrossRef PubMed.
- O. W. L. Carter, Y. Xu and P. J. Sadler, Minerals in biology and medicine, RSC Adv., 2021, 11(4), 1939–1951 RSC.
- L. V. A. Hale and N. K. Szymczak, Hydrogen Transfer Catalysis beyond the Primary Coordination Sphere, ACS Catal., 2018, 8, 6446–6461 CrossRef CAS.
- M. Tuckerman, E. Vitz, J. W. Moore, J. Shorb, X. Prat-Resina, T. Wendorff, A. Hahn and R. J. Lancashire, Contributors, Electronegativity and Dipole Moment, https://chem.libretexts.org/@go/page/41350.2020.
- C. Q. Sun, L. Li, B. K. Tay and H. Huang, An extended ‘quantum confinement’ theory: Surface-coordination imperfection modifies the entire band structure of a nanosolid, J. Phys. D: Appl. Phys., 2001, 34(24), 3470–3479 CrossRef CAS.
- B. R. Bickmore, C. J. Tadanier, K. M. Rosso, W. D. Monn and D. L. Eggett, Bond-valence methods for pKa prediction: critical reanalysis and a new approach, Geochem. Cosmochim. Acta, 2004, 68(9), 2025–2042 CrossRef CAS.
- S. C. Mitchell, An Improved MUSIC Model for Gibbsite, Brigham Young University, Masters, 2005 Search PubMed.
- L. Pauling, The Nature of the Chemical Bond, Cornell University Press, Ithaca New York, 3rd edn, 1960 Search PubMed.
- R. A. Young and D. B. Wiles, National Bureau of Standards Special Publication 567, National Bureau of Standards, Washington D. C., 1980, 567, 143 – 162 Search PubMed.
- G. A. Parks, The Isoelectric Points of Solid Oxides, Solid Hydroxides, and Aqueous Hydroxo Complex Systems, Chem. Rev., 1965, 65(2), 177–198 CrossRef CAS.
- D. G. Brookins, Eh-pH Diagrams for Geochemistry, Springer – Verlag Berlin Heidelberg, 1st edn, 1988 Search PubMed.
- N. Takeno, Atlas of Eh – pH Diagrams, Geological Survey of Japan Open File Report No. 419, National Institute of Advanced Industrial Science and Technology Research Center for Deep Geological Environments, 2005 Search PubMed.
- M. Pourbaix, Atlas of Electrochemical Equilibria in Aqueous Solutions, J. A. Translator Franklin, National Association of Corrosion Engineers, Houston TX, 1974 Search PubMed.
- F. Basharat, U. A. Rana, M. Shahid and M. Serwar, Heat treatment of electrodeposited NiO films for improved catalytic water oxidation, RSC Adv., 2015, 5, 86713–86722 RSC.
- L.-F. Huang, M. J. Hutchison, Jr. R. J. Santucci, J. R. Scully and J. M. Rondinelli, Improved Electrochemical Phase Diagrams from Theory and Experiment: The Ni – Water System and Its Complex Compounds, J. Phys. Chem. C, 2017, 121(18), 9782–9789 CrossRef CAS.
- R. C. Weast, D. R. Lide, M. J. Astle and W. H. Beyer, Handbook of Chemistry and Physics, CRC Press, Boca Ratan, Florida, 70th edn, 1990 Search PubMed.
- P. Fleming, R. A. Farrell, J. D. Holmes and M. A. Morris, The Rapid Formation of La(OH)3 from La2O3 Powders on Exposure to Water Vapor, J. Am. Ceram. Soc., 2010, 93(4), 1187–1194 CrossRef CAS.
- S. Xie, E. Iglesta and A. T. Bell, Water-Assisted Tetragonal-to-Monoclinic Phase Transformation of ZrO2 at Low Temperature, Chem. Mater., 2000, 12(8), 2442–2447 CrossRef CAS.
- F. Zhang, P. J. Chupas, S. L. A. Liu, J. C. Hanson, W. A. Caliebe, P. L. Lee and S. W. Chan, In situ Study of the Crystallization from Amorphous to Cubic Zirconium Oxide: Rietveld and Reverse Monte Carlo Analyses, Chem. Mater., 2007, 19(13), 3118–3126 CrossRef CAS.
- P. Southon, Structural Evolution during the Preparation and Heating of Nanophase Zirconia Gels, Ph. D. Thesis, University of Technology, Sydney Australia, 2000 Search PubMed.
- G. A. El, Measurement of the diffuse double-layer between zirconia and alumina, J. Appl. Phys., 1999, 86(10), 5894–5897 CrossRef.
- M. Leffler, Chemistry Dept., University of Connecticut, Unpublished results, 2021 Search PubMed.
- D. Petrescu, L. Velciu and C. Bucur, Antimony Removal from Aqueous Solutions Using Zirconium Hydroxide, International Atomic Energy Agency, Nuclear, 2016 Search PubMed.
- A. S. C. Chen, G. M. Lewis and L. Wang, Arsenic Removal from Drinking Water by Adsorptive Media EPA Demonstration Project at Golden Hills Community Services District in Tehachapi, CA Final Performance Evaluation Report, National Risk Management Research Laboratory, Office of Research and Development, U. S. Environmental Protection Agency, Cincinnati, Ohio 45268, 2010 Search PubMed.
- G. A. Parks, Free Energies of Formation and Aqueous Solubilities of Aluminium Hydroxides and Oxide Hydroxides at 25oC, Am. Mineral., 1972, 57, 1163–1189 CAS.
- V. G. Krivovichev, M. V. Charykova and A. V. Vishnevsky, The Thermodynamics of Selenium Minerals in Near-Surface Environments, Minerals, 2017, 7(10), 188 CrossRef.
- P. Lindqvist-Reis, Hydration of the Yttrium (III) Ion in Aqueous Solution. An X-ray Diffraction and XAFS Structural Study, J. Phys. Chem. B, 2000, 104(2), 402–408 CrossRef CAS.
- R. C. Weast, D. R. Lide, M. J. Astle and W. H. Beyer, Handbook of Chemistry and Physics, CRC Press, Boca Ratan, Florida, 70th edn, 1990 Search PubMed.
- F. Basharat, U. A. Rana, M. Shahid and M. Serwar, Heat treatment of electrodeposited NiO films for improved catalytic water oxidation, RSC Adv., 2015, 5, 86713–86722 RSC.
- L.-F. Huang, M. J. Hutchison, Jr. R. J. Santucci, J. R. Scully and J. M. Rondinelli, Improved Electrochemical Phase Diagrams from Theory and Experiment: The Ni – Water System and Its Complex Compounds, J. Phys. Chem. C, 2017, 121(18), 9782–9789 CrossRef CAS.
- G. A. Parks, The Isoelectric Points of Solid Oxides, Solid Hydroxides, and Aqueous Hydroxo Complex Systems, Chem. Rev., 1965, 65(2), 177–198 CrossRef CAS.
- Y. Cudennec and A. Lecerf, The transformation of Cu(OH)2 into CuO, revisited, Solid State Sci., 2003, 5(11–12), 1471–1474 CrossRef CAS.
- H. Chang, C. S. Jwo, C. J. Lo, T. T. Tsung, M. J. Kao and H. M. Lin, Rheology of CuO Nanoparticle Suspension Prepared by ASNSS, Rev. Adv. Mater. Sci., 2005, 10, 128–132 CAS.
- Y. Zhang, D. J. Rimstidt, Y. Huang and C. Zhu, Kyanite far from equilibrium dissolution rate at 0-22oC and pH of 3.5 – 7.5, Acta Geochim., 2019, 38, 472–480 CrossRef CAS.
- T. Shirai, h. Watanabe, M. Fuji and M. Takahashi, Structural Properties and Surface Characteristics on Aluminum Oxide Powders, Nagoya Institute of Technology Ceramic Research Annual Report, 2009, vol. 9, pp. 23–31 Search PubMed.
- J. J. Smolik, E. Harman and D. W. Fuerstenau, Surface characteristics and flotation of aluminosilicate, Trans. AIME, 1967, 235, 367–375 Search PubMed.
- H. S. Choi, and J. H. Oh, Surface properties and floatability of kyanite and andalusite, Jour. Min, Met. Inst., Japan, 1965, 81(927), pp. 614–620 Search PubMed.
- J. X. Lin and L. Wang, Adsorption of dyes using magnesium hydroxide-modified diatomite, Desalination Water Treat., 2009, 8(1–3), 263–271 CrossRef CAS.
- J. M. Trillo, M. D. Alba, M. A. Castro, A. Munoz, J. Poyato and M. M. Tobias, Local Environment of Lanthanum Ions in Montmorillonite Upon Heating, Clay Miner., 1992, 27(4), 423–3 CrossRef CAS.
- T. Guner, A. Kus, M. Ozcan, A. Genc, H. Sahin and M. M. Demir, Green fabrication of lanthanide –doped hydroxide-based phosphors: Y(OH)3:Eu3+ nanoparticles for white light generation, Beilstein J. Nanotechnol., 2019, 10, 1200–1210 CrossRef CAS PubMed.
- V. A. Hackley and S. G. Malghan, The surface chemistry of silicon nitride powder in the presence of dissolved ions, J. Mater. Sci., 2004, 29, 4420–4430 CrossRef.
- K. Shinoda, E. Matsubara, A. Muramatsu and Y. Waseda, Local Structure of Ferric Hydroxide Fe(OH)3 in Aqueous Solution by the Anomalous X-ray Scattering and EXAFS Method, Mater. Trans., 1994, 35(6), 394–398 CrossRef CAS.
- M. Che and L. Bonneviot, Role of oxide surface in coordination chemistry of transition metal ions in catalytic systems, Pure Appl. Chem., 1988, 60(8), 1369–1378 CrossRef CAS.
- H. Chang, C. S. Jwo, C. J. Lo, T. T. Tsung, M. J. Kao and H. M. Lin, Rheology of CuO Nanoparticle Suspension Prepared by ASNSS, Rev. Adv. Mater. Sci., 2005, 10, 128–132 CAS.
- Y. Maimaiti, M. Nolan and S. D. Elliott, Reduction mechanisms of the CuO (1 1 1) surface through surface oxygen vacancy formation and hydrogen adsorption, Phys. Chem. Chem. Phys., 2014, 16(7), 3036–3046 RSC.
- A. K. Mishra, A. Roldan and N. H. de Leeuw, CuO Surfaces and Co 2 Activation: A Dispersion-Corrected DFT+U Study, 2016 Dispersion-Corrected DFT+U Study, J. Phys. Chem. C, 2016, 120(4), 2198–2214 CrossRef CAS.
- C.-C. Diao, C.-Y. Huang, C.-F. Yang and C.-C. Wu, Morphological, Optical, and Electrical Properties of p-type Nickel Oxide Thin Films by Nonvacuum Deposition, Nanomaterials, 2020, 10(4), 636–641 CrossRef CAS PubMed.
- R. W. Cairns and E. Ott, X-Ray Studies of the System Nickel – Oxygen – Water. I. Nickelous Oxide and Hydroxide, J. Am. Chem. Soc., 1933, 55(2), 527–533 CrossRef CAS.
- C. M. Koretsky, D. A. Sverjensky and N. Sahai, A model of surface site types on oxide and silicate minerals based on crystal chemistry: Implications for surface site types and densities, multi-site adsorption, surface infrared spectroscopy, and dissolution kinetics, Am. J. Sci., 1998, 298, 349–438 CrossRef CAS.
- W. J. K. Klein, L. K. Koopal, M. A. Cohen Stuart and W. H. Van Riemskijk, Surface charge regulation upon polyelectrolyte adsorption, hematite, polystyrene sulfonate, surface charge regulation Theoretical calculations and hematite-poly(styrene sulfonate) system, Colloids Surf., A, 2006, 291, 13–23 CrossRef.
- F. Jones, A. L. Rohl, J. B. Farrow and W. van Bronswijk, Molecular modeling of water adsorption on hematite, Phys. Chem. Chem. Phys., 2000, 14, 3209–3216 RSC.
- L. Schottner, A. Nefedov, C. Yang, S. Heissier, Y. Wang and C. Woll, Structural Evolution of α-Fe2O3 Surfaces Under Reduction Conditions Monitored by Infrared Spectroscopy, Front. Chem., 2019, 7(451), 1–12 Search PubMed.
- E. Wasserman, J. R. Rustad, A. R. Felmy, B. P. Hay and J. W. Halley, Ewald methods for polarizable surfaces with application to hydroxylation and hydrogen bonding on the (012) and (001) surfaces of α-Fe2O3, Surf. Sci., 1997, 385(2 – 3), 217–239 CrossRef CAS.
- M. Leffler, J. Fee, S. March, Y. Wu and S. L. Suib, Part II: Superconductivity Observed in Magnetically Separated Nanoscale Anatase Titania at Ambient Temperature and Pressure in an Aqueous Environment at its Point of Zero Charge, RSC Adv., 2024, 14, 30317–30335 RSC.
- S. Pantaleone, F. Pellegrino, V. Maurino, M. Corno, P. Ugliengo and L. Mino, Disclosing the true atomic structure of {0 0 1} facets in shape-engineered TiO2 anatase titania nanoparticles, J. Mater. Chem. A, 2024, 12, 4325–4332 RSC.
- Molecular Dynamics, Wikipedia, https://en.wikipedia.org/wiki/Molecular_dynamics Search PubMed.
- Density Functional Theory, Wikipedia, https://en.wikipedia.org/wiki/Density_functional_theory Search PubMed.
- Y. Maimaiti, M. Nolan and S. D. Elliott, Reduction mechanisms of the CuO (1 1 1) surface through surface oxygen vacancy formation and hydrogen adsorption, Phys. Chem. Chem. Phys., 2014, 16(7), 3036–3046 RSC.
- N. Naveas, R. Pulido, C. Marini, J. Hernandez-Montelongo and M. M. Silvan, First-principles calculations of hematite (α-Fe2O3) by self-consistent DFT+U+V, iScience, 2023, 26(2), 1–17 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |


