Open Access Article
Open Access Article2-Pyridone-assisted palladium-catalyzed regioselective arylation of naphthamides
Jayprakash Prajapati and
Sourav Chatterjee *
*
Department of Chemistry, Ashoka University, Sonipat, Haryana-131029, India. E-mail: sourav.chatterjee@ashoka.edu.in
First published on 3rd March 2026
Abstract
The 2-pyridone scaffold receives attention in medicinal chemistry due to its bioisostere properties, which significantly contributing to lipophilicity, solubility, metabolic stability, and drug off-target effects. In the present article, we report the 2-pyridone-assisted regioselective arylation of naphthamides. Using diversified naphthamides and aryl iodides, a wide variety of 2-pyridone-containing molecular scaffolds were synthesized in good yields, which could be potentially useful in medicinal chemistry.
Introduction
The development of a new potential drug molecule has always been associated with many challenges. Design of a molecular architecture and its compelling synthetic approach for a specific biological function always remain a challenge. The 2-pyridone scaffold functions both as a hydrogen-bond acceptor and donor; serves as a bioisostere for amides, phenyls, pyridines, and other N- or O-containing heterocycles; and modulates lipophilicity, aqueous solubility, and metabolic stability in drug molecules. Compared to phenyls, pyridines, or phenols, pyridones are more polar (higher c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P, log
P, log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) D, and PSA values), often resulting in improved drug-like properties. Unlike basic nitrogen heterocycles, such as pyridine, the neutral, less lipophilic pyridone ring system helps reduce off-target activities. Its embedded amide functionality makes 2-pyridone a valid peptide-bond isostere and improves its physicochemical properties, such as metabolic clearance. Fig. 1 highlights a few representative examples of how the systematic incorporation of 2-pyridone onto a hit molecule through structure–activity relationship (SAR) studies could afford an optimized lead molecule with improved potency.1–5 Similarly, Kohji Hanasaki and co-workers developed a human cannabinoid receptor type II (CB2R) inhibitor bearing the 2-pyridone backbone that regulates analgesic and anti-inflammatory responses without central adverse effects.6 Stephen T. Worland and co-workers report 2-pyridone-containing HRV-2 3CP inhibitors as anti-rhinoviral agents.7 Another report from Alan S. Rosenthal and co-workers highlights HLE inhibitors bearing the 2-pyridone moiety, used for the treatment of emphysema and cystic fibrosis.8
D, and PSA values), often resulting in improved drug-like properties. Unlike basic nitrogen heterocycles, such as pyridine, the neutral, less lipophilic pyridone ring system helps reduce off-target activities. Its embedded amide functionality makes 2-pyridone a valid peptide-bond isostere and improves its physicochemical properties, such as metabolic clearance. Fig. 1 highlights a few representative examples of how the systematic incorporation of 2-pyridone onto a hit molecule through structure–activity relationship (SAR) studies could afford an optimized lead molecule with improved potency.1–5 Similarly, Kohji Hanasaki and co-workers developed a human cannabinoid receptor type II (CB2R) inhibitor bearing the 2-pyridone backbone that regulates analgesic and anti-inflammatory responses without central adverse effects.6 Stephen T. Worland and co-workers report 2-pyridone-containing HRV-2 3CP inhibitors as anti-rhinoviral agents.7 Another report from Alan S. Rosenthal and co-workers highlights HLE inhibitors bearing the 2-pyridone moiety, used for the treatment of emphysema and cystic fibrosis.8
Owing to the growing application of 2-pyridone as a fundamental heterocyclic building block in medicinal chemistry, we planned to explore its use as a directing group for facile access to important molecular architectures, which potentially could be used in medicinal chemistry. As a proof of concept, initially, we chose the naphthamide system for 2-pyridone-mediated regioselective sp2 C–H activation.
Regioselective C–H activation in naphthamide is important because it allows late-stage functionalization of complex naphthalene-containing molecules, useful in drug molecules, fungicides, organic electronics, dyes and pigments. In 2018, N. Lv et al. showcased Co and Mn-dual catalysis cross-coupling as a strategy for chelation-assisted C–H activation from readily available benzamides and oximes (Scheme 1a). The resulting difunctional biaryl synthesis also responded well for 1-naphthamide (Scheme 1a).9 This dual-chelation-assisted strategy provides access to diverse difunctional biaryls, which offer promising biological activities considering their substrate scope and controlled regioselectivity. B. M. Bhanage and coworkers in 2016 implemented a deaminative C–C bond coupling using N-methoxybenzamide/naphthamide in combination with aromatic amines and Pd(OAc)2 as a catalyst to generate diverse phenanthridinones (Scheme 1b).10 Along this line, a report by P. Li et al. in 2018 disclosed the nickel-catalyzed regioselective arylation of aromatic amides with aryl iodides, where pyridine-N-oxide served as a directing group (Scheme 1c).11
Owing to the importance of 2-pyridone in medicinal chemistry and its growing functional utility as a directing group,12 here, we report 2-pyridone as a bidentate directing group for the regioselective sp2 C–H arylation of 1-naphthamide. To the best of our knowledge, this is the first study to adopt 2-pyridone as a directing group for the regioselective C2 arylation of 1-naphthamide. This 2-pyridone-assisted C2–H arylation proceeds via Pd catalysis (with Ag2O employed for catalyst regeneration) in acetic acid as a solvent, affording N-(1-alkyl-2-pyridone-3-yl)-2-aryl-1-naphthamides in good-to-excellent yields (Scheme 1d).
Results and discussion
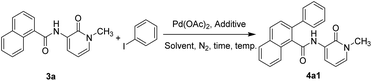
To optimize the 2-pyridone-directed regioselective C2–H arylation of 1-naphthamide, we first considered N-(1-methyl-2-pyridone)-2-phenyl-1-naphthamide (4a1) as a representative example to start with. The precursor N-(1-methyl-2-pyridone)-1-naphthamide (3a) was synthesized from the N-methylation of 3-nitro-2-pyridone (1a), followed by the reduction of its nitro group to amine (2a) and, further, its conjugation with 1-naphthoic acid via acid-amine coupling to form the corresponding N-(1-methyl-2-pyridone-3-yl)-1-naphthamide (3a) (Scheme 2). We tested our hypothesis with 10 mol% Pd(OAc)2 and 0.36 mmol (2 eqv.) Ag2O under inert conditions, at 80 °C for 10 h (Table 1, entry 1). Further isolation and characterization confirmed the formation of 4a1 as the desired product in 25% isolated yield (Table 1, entry 1). To optimize the reaction condition, we planned to determine the yield of the crude reaction mixture by HPLC analysis. A growth curve was generated using HPLC for 4a1, with an increase in the 4a1 product concentration. The reaction temperature was further increased to 100 °C, 120 °C and then to 150 °C. An increase in the yield was observed when the reaction temperature was raised to 100 °C (73%) and 120 °C (96%) (Table 1, entry 2–3). However, further increase in the reaction temperature to 150 °C reduced the yield to 71% (Table 1, entry 4), which could be because trace impurity formation was observed at this temperature. Under this condition, reducing the reaction time to 6 h or increasing the reaction time to 15 h had a very little influence in terms of product yield (Table 1, entries 5–6). We further extended our observation to determine the right solvent for this reaction. While H2O and HFIP offered negligible and moderate reaction yields (Table 1, entries 7 and 8), respectively, using an identical acetic-acid composition (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) with these solvents did not greatly enhance the reaction outcome (Table 1, entries 9–10). The use of DMF also failed to establish it as a solvent of choice (Table 1, entry 11). Our findings highlight acetic acid as the optimized solvent tested. We were curious to understand the effect of other additives compared to Ag2O. Ag2CO3 (2.0 eqv., 0.36 mmol) and a mixture of Ag(OAc) (3.0 eqv., 0.54 mmol) and sodium salt of TFA (2.0 eqv., 0.36 mmol) were tested both in acetic acid (Table 1, entries 12 and 18) and dioxane (Table 1, entry 13) as a solvent. To our dissatisfaction, none of these was as good as Ag2O (Table 1, entries 12–13 vs. 3). Reducing the silver oxide stoichiometry (1.5 eqv., 0.27 mmol and 1.0 eqv., 0.18 mmol) was not shown to decrease the product yield significantly; however, 2.0 eqv. (0.36 mmol) of Ag2O was considered to be the optimized additive loading. The catalyst loading was also monitored. Reducing Pd(OAc)2 from 10 mol% (Table 1, entry 3) to 5 mol% (Table 1, entry 16) and further to 2 mol% (Table 1, entry 17) dramatically compromised the reaction yield. Finally, we found that the use of 10 mol% of Pd(OAc)2 as a catalyst, in combination with 2.0 eqv. (0.36 mmol) of Ag2O and 2.0 eqv. of iodobenzene, in the reaction of amide 3a in acetic acid (as the solvent), at 120 °C for 10 h produced the corresponding C2-arylated product 4a1 in 86% isolated yield, with a high selectivity. Under the optimized condition, to explore the substrate scope, 1-naphthamide 3a was primarily subjected to selective coupling with various aryl/heteroaryl/cycloalkyl halides. The investigation encompassed aryl iodides, aryl bromides, 2-iodo-pyridine, 4-iodo-pyridine, 5-iodo-1H-indole and 5-iodo-1H-imidazole, cyclopentyl bromide, and cyclohexyl bromide. The results demonstrated that only aryl iodides successfully afforded the desired products, while cycloalkyl bromides remained unreactive. With different heterocyclic iodides, we observed that the starting material 3a majorly remained unconsumed, with a very trace non-isolable new product formation observed, as determined by the HPLC analysis of the crude reaction mixture after 10 h (SI, Table S1). The poor product conversion observed with nitrogen-containing heterocyclic iodides is presumably because of the ring nitrogen coordination with palladium, which might restrict further reaction. To gain further insight into the aryl iodide versatility, we examined various aryl iodides bearing electron-donating groups (EDG) and electron-withdrawing groups (EWG) at different positions for the regioselective C2–H arylation of 1-naphthamide. Both electron-deficient and electron-rich aryl iodides proved compatible with the reaction conditions, delivering the corresponding regioselective C2-arylated products 4a1–4d5 in good yields (Scheme 3). Different N-protected 2-pyridones served equally well (Scheme 3), offering a broader scope for structural modality. To identify the reasons for the few low yields, such as for products 4b5 (16%), 3b6 (21%) and 4c3 (23%), we performed HPLC analysis on the crude reaction mixture after 10 h and compared the HPLC retention time with that for the isolated standard sample. In all three reactions, we observed the complete consumption of both the reactants and the formation of undesired by-products, which lowered the yield of these products. The chromatography results are summarized in SI (Table S2). We further investigated whether our optimized reaction protocol was also successful in the 2-pyridone-directed 2-naphthamide system. N-(1-methyl-2-pyridone)-2-naphthamide (3e) was subjected to C–H arylation with iodobenzene under an identical reaction condition (Scheme 4). To our satisfaction, the method confirmed the formation of a C3-selective arylated product N-(1-methyl-2-pyridone)-3-phenyl-2-naphthamide (4 × 101) in 84% isolated yield (Scheme 5). To validate that the catalytic pathway proceeds through five-membered palladacycles, we protected the amide nitrogen of N-(1-methyl-2-pyridone)-1-naphthamide (3a) with a methyl group to prepare N-methyl-(1-methyl-2-pyridone)-1-naphthamide (3f). When 3f was chosen as a substrate, as expected, no arylated product was observed even after 20 h, under an identical reaction condition (Scheme 5). This finding confirms the essential role of the free amide nitrogen in facilitating the transformation through coordination-assisted metalation.
1) with these solvents did not greatly enhance the reaction outcome (Table 1, entries 9–10). The use of DMF also failed to establish it as a solvent of choice (Table 1, entry 11). Our findings highlight acetic acid as the optimized solvent tested. We were curious to understand the effect of other additives compared to Ag2O. Ag2CO3 (2.0 eqv., 0.36 mmol) and a mixture of Ag(OAc) (3.0 eqv., 0.54 mmol) and sodium salt of TFA (2.0 eqv., 0.36 mmol) were tested both in acetic acid (Table 1, entries 12 and 18) and dioxane (Table 1, entry 13) as a solvent. To our dissatisfaction, none of these was as good as Ag2O (Table 1, entries 12–13 vs. 3). Reducing the silver oxide stoichiometry (1.5 eqv., 0.27 mmol and 1.0 eqv., 0.18 mmol) was not shown to decrease the product yield significantly; however, 2.0 eqv. (0.36 mmol) of Ag2O was considered to be the optimized additive loading. The catalyst loading was also monitored. Reducing Pd(OAc)2 from 10 mol% (Table 1, entry 3) to 5 mol% (Table 1, entry 16) and further to 2 mol% (Table 1, entry 17) dramatically compromised the reaction yield. Finally, we found that the use of 10 mol% of Pd(OAc)2 as a catalyst, in combination with 2.0 eqv. (0.36 mmol) of Ag2O and 2.0 eqv. of iodobenzene, in the reaction of amide 3a in acetic acid (as the solvent), at 120 °C for 10 h produced the corresponding C2-arylated product 4a1 in 86% isolated yield, with a high selectivity. Under the optimized condition, to explore the substrate scope, 1-naphthamide 3a was primarily subjected to selective coupling with various aryl/heteroaryl/cycloalkyl halides. The investigation encompassed aryl iodides, aryl bromides, 2-iodo-pyridine, 4-iodo-pyridine, 5-iodo-1H-indole and 5-iodo-1H-imidazole, cyclopentyl bromide, and cyclohexyl bromide. The results demonstrated that only aryl iodides successfully afforded the desired products, while cycloalkyl bromides remained unreactive. With different heterocyclic iodides, we observed that the starting material 3a majorly remained unconsumed, with a very trace non-isolable new product formation observed, as determined by the HPLC analysis of the crude reaction mixture after 10 h (SI, Table S1). The poor product conversion observed with nitrogen-containing heterocyclic iodides is presumably because of the ring nitrogen coordination with palladium, which might restrict further reaction. To gain further insight into the aryl iodide versatility, we examined various aryl iodides bearing electron-donating groups (EDG) and electron-withdrawing groups (EWG) at different positions for the regioselective C2–H arylation of 1-naphthamide. Both electron-deficient and electron-rich aryl iodides proved compatible with the reaction conditions, delivering the corresponding regioselective C2-arylated products 4a1–4d5 in good yields (Scheme 3). Different N-protected 2-pyridones served equally well (Scheme 3), offering a broader scope for structural modality. To identify the reasons for the few low yields, such as for products 4b5 (16%), 3b6 (21%) and 4c3 (23%), we performed HPLC analysis on the crude reaction mixture after 10 h and compared the HPLC retention time with that for the isolated standard sample. In all three reactions, we observed the complete consumption of both the reactants and the formation of undesired by-products, which lowered the yield of these products. The chromatography results are summarized in SI (Table S2). We further investigated whether our optimized reaction protocol was also successful in the 2-pyridone-directed 2-naphthamide system. N-(1-methyl-2-pyridone)-2-naphthamide (3e) was subjected to C–H arylation with iodobenzene under an identical reaction condition (Scheme 4). To our satisfaction, the method confirmed the formation of a C3-selective arylated product N-(1-methyl-2-pyridone)-3-phenyl-2-naphthamide (4 × 101) in 84% isolated yield (Scheme 5). To validate that the catalytic pathway proceeds through five-membered palladacycles, we protected the amide nitrogen of N-(1-methyl-2-pyridone)-1-naphthamide (3a) with a methyl group to prepare N-methyl-(1-methyl-2-pyridone)-1-naphthamide (3f). When 3f was chosen as a substrate, as expected, no arylated product was observed even after 20 h, under an identical reaction condition (Scheme 5). This finding confirms the essential role of the free amide nitrogen in facilitating the transformation through coordination-assisted metalation.
| Entry | Additive/Temp. (°C)/Time (h) | Solvent | Yieldg (%) |
|---|---|---|---|
| a Reaction conditions: 3a (0.18 mmol), Ph-I (0.36 mmol), Pd(OAc)2 (10 mol%), and Ag2O (0.36 mmol), heated at a specified temperature for the mentioned time.b 0.54 mmol of Ag(OAc) and 0.36 mmol of CF3COONa were used.c Ag2O (0.27 mmol).d Ag2O (0.18 mmol).e Pd(Oac)2 (5 mol%), and.f Pd(OAc)2 (2 mol%).g Yields are reported as the HPLC yield from the reaction mixture.h Ag2CO3 (0.36 mmol).i Isolated yield by column chromatography. | |||
| 1 | Ag2O/80/10 | AcOH | 32 (25i) |
| 2 | Ag2O/100/10 | AcOH | 73 |
| 3 | Ag2O/120/10 | AcOH | 96 (86i) |
| 4 | Ag2O/150/10 | AcOH | 71 |
| 5 | Ag2O/120/6 | AcOH | 87 |
| 6 | Ag2O/120/15 | AcOH | 89 |
| 7 | Ag2O/120/10 | H2O | 6 |
| 8 | Ag2O/120/10 | HFIP | 65 |
| 9 | Ag2O/120/10 | H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) AcOH (1 AcOH (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
24 |
| 10 | Ag2O/120/10 | HFIP![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) AcOH (1 AcOH (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
69 |
| 11 | Ag2O/120/10 | DMF | Trace |
| 12b | Ag(OAc)/CF3COONa/120/10 | AcOH | 45 |
| 13b | Ag(OAc)/CF3COONa/120/10 | Dioxane | 67 |
| 14c | Ag2O/120/10 | AcOH | 91 |
| 15d | Ag2O/120/10 | AcOH | 88 |
| 16e | Ag2O/120/10 | AcOH | 48 |
| 17f | Ag2O/120/10 | AcOH | 41 |
| 18h | Ag2CO3/120/10 | AcOH | 91i |
The presence of Ag2O is important, as it regenerates the Pd catalyst, generating by-products like AgI, H2O, and Ag in the cycle. Also, the directing group, 2-pyridone, helps in providing stability by forming a bidentate complex with the Pd(II) intermediate. On the basis of the known Pd-catalyzed cycle13 and DFT calculation (Fig. 2), we proposed a plausible Pd(OAc)2-catalyzed regioselective reaction mechanism for the reaction of 3a with iodobenzene to give 4a1 as the product. The first step involved the coordination of 3a with Pd(II), followed by the formation of the active catalyst complex of Pd(II) INT-1 via TS-1, with the elimination of AcOH (Scheme 6).
 | ||
| Fig. 2 Predicted mechanistic pathway and free-energy variation (in kcal mol−1). Calculation was carried out at the B3LYP def2-TZVP, SMD(CH3COOH) level of theory. | ||
Further, INT-1 undergoes C–H activation at the C2 position in 1-naphthamide through TS-2 to yield INT-2, accompanied by the elimination of AcOH. In the next step, INT-2 undergoes the oxidative addition of iodobenzene through TS-3 to generate INT-3, where the Pd acquires an oxidation state of +4. The addition of AcOH afforded INT-4 via TS-4, with the highest energy barrier of 14.14 kcal mol−1. This step is the rate-determining step of the reaction. During the reductive elimination, the Pd(II)-metal complex gives the C2-arylated 1-naphthamide (INT-5), followed by proto-demetallation through TS-5 to afford the arylated product 4a1. In situ Pd(II) undergoes catalyst regeneration for the next catalytic cycle using Ag2O as the oxidant and AcOH as the solvent, with the formation of AgI, H2O, and Ag as side products (Scheme 6).
To validate the role of 2-pyridone in the catalytic cycle, we extended our study to understand the stability of palladium intermediate complex at deferent dihedral angles (Pd–N–C–CO) of 0°, 90°, 180°, and 270°. We conducted computational calculation using Orca at the RHF def2-SVP, SMD(CH3COOH) level of theory. With a constraint dihedral angle, all geometry were optimized at the same level of theory. The relative energy suggests that the intermediate with a dihedral angle of 0° has the lowest Gibbs free energy. With increasing the dihedral angle, the energy value increases; for example, 90° and 180° dihedral angles give 15.55 kcal mol−1 and 24.44 kcal mol−1, respectively. To our expectation, when Pd comes in proximity to the carbonyl group of 2-pyridone, the energy goes down to 15.55 kcal mol−1 (Fig. 3). The results highlight that 2-pyridone assists in the catalytic cycle by stabilizing the palladium center via a five-membered palladacycle intermediate.
Conclusions
In conclusion, we demonstrated that 2-pyridone, a fundamental building block in medicinal chemistry, could be used as a bidentate directing group for the palladium-catalyzed regioselective C2(sp2)–H arylation of 1-naphthamides. Our wide structural diversification also extended to showcase that 2-naphthamides can equally offer regioselective C3(sp2)–H arylated products. The advantageous use of the ligand-free reaction condition and the formation of non-toxic byproducts, such as H2O and Ag, highlight more possibilities for structural diversification in medicinal chemistry.Author contributions
All authors have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
Crystallographic data for 4a1, 4d2 has been deposited at the CCDC under number: 2472814 and 2475895.14a,bThe data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6ra00358c.
Acknowledgements
SC and JP acknowledge the financial aid provided by Axis Bank to support this research work. SC and JP also acknowledge Ashoka University for providing research infrastructure. JP thanks Ashoka University for funding support in terms of a Research Scholarship. The authors gratefully acknowledge the high-performance computing facility provided by Ashoka University.References
- S. Lin, C. Liu, X. Zhao, X. Han, X. Li, Y. Ye and Z. Li, Front. Chem., 2022, 10, 869860 Search PubMed.
- M. A. Ahmed, S. I. Alqasoumi, A. E. Ashour, V. Masand, N. A. Al-Jaber, B. H. Taibi and M. A. Mohamed, Eur. J. Med. Chem., 2012, 53, 133–140 Search PubMed.
- K. S. Kim, L. Zhang, R. Schmidt, Z.-W. Cai, D. Wei, D. K. Williams, L. J. Lombardo, G. L. Trainor, D. Xie, Y. Zhang, A. Yongmi, J. S. Sack, J. S. Tokarski, C. Darienzo, A. Kamath, P. Marathe, Y. Zhang, J. Lippy, J. Sr. Robert, W. Barri, H. Benjamin, J. Gullo-Brown, V. Manne, J. T. Hunt, J. Fargnoli and R. M. Borzilleri, J. Med. Chem., 2008, 51(17), 5330–5341 CrossRef CAS PubMed.
- Z. Liu, Y. Yuan, M. Kogiso, B. Zheng, L. Deng, J. J. Qiu, S. Dong, H. Lv, J. M. Gallo, X.-N. Li and Y. Song, J. Med. Chem., 2014, 57(20), 8307–8318 CrossRef CAS.
- F. Wu, H. Jiang, B. Zheng, M. Kogiso, Y. Yuan, C. Zhou, X.-N. Li and Y. Song, J. Med. Chem., 2015, 58(17), 6899–6908 Search PubMed.
- K. Kusakabe, Y. Tada, Y. Iso, M. Sakagami, Y. Morioka, N. Chomei, S. Shinonome, K. Kawamoto, H. Takenaka, K. Yasui, H. Hamana and K. Hanasaki, Bioorg. Med. Chem., 2013, 21(7), 2045–2055 Search PubMed.
- P. S. Dragovich, T. J. Prins, Ru Zhou, E. L. Brown, F. C. Maldonado, S. A. Fuhrman, L. S. Zalman, T. Tuntland, C. A. Lee, A. K. Patick, D. A. Matthews, T. F. Hendrickson, M. B. Kosa, Bo Liu, M. R. Batugo, J.-P. R. Gleeson, S. K. Sakata, L. Chen, M. C. Guzman, J. W. Meador, A. F. Rose and S. T. Worland, J. Med. Chem., 2002, 45(8), 1607–1623 CrossRef CAS PubMed.
- J. W. Skiles, R. Sorcek, S. Jacober, C. Miao, P. W. Mui, D. McNeil and A. S. Rosenthal, Bioorg. Med. Chem. Lett., 1993, 3(4), 773–778 CrossRef CAS.
- L. Ningning, C. Zhengkai, L. Yue, L. Zhanxiang and Z. Yuhong, Org. Lett., 2018, 20(18), 5845–5848 CrossRef.
- S. L. Yedage and B. M. Bhanage, J. Org. Chem., 2016, 81(10), 4103–4111 Search PubMed.
- P. Li, G.-W. Wang, H. Chen and L. Wang, Org. Biomol. Chem., 2018, 16, 8783–8790 Search PubMed.
- (a) T. K. Pati, S. Debnath, M. Kundu, U. Khamrai and D. K. Maiti, Org. Lett., 2018, 20(13), 4062–4066 Search PubMed; (b) T. K. Pati, S. Ali Molla, N. N. Ghosh, M. Kundu, S. Ajarul, P. Maity, U. Khamrai and D. K. Maiti, J. Org. Chem., 2024, 89(10), 6798–6812 CrossRef CAS PubMed.
- V. G. Zaitsev, D. Shabashov and O. Daugulis, J. Am. Chem. Soc., 2005, 127(38), 13154–13155 CrossRef CAS.
- (a) CCDC 2472814: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p0563; (b) CCDC 2475895: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p3cls.
| This journal is © The Royal Society of Chemistry 2026 |