 Open Access Article
Open Access ArticleEnhanced removal of fluoride and phosphate by nickel-doped kaolinite nano-adsorbents: mechanisms and resource recovery
Asmaa S. Morshedy *a,
Tahany Mahmoud
*a,
Tahany Mahmoud *b,
Aya M. Matlooba and
Mahmoud F. Mubarakb
*b,
Aya M. Matlooba and
Mahmoud F. Mubarakb
aRefining Department, Egyptian Petroleum Research Institute, 11727, Cairo, Egypt. E-mail: asma_2000asma@yahoo.com
bPetroleum Application Department, Egyptian Petroleum Research Institute, 11727, Cairo, Egypt. E-mail: tahany.mahmoud.a@gmail.com
First published on 5th May 2026
Abstract
The development of efficient and cost-effective catalysts for wastewater treatment is crucial for mitigating environmental pollution. In this study, NiO and Ni-metal doped kaolinite-based nanocomposites were synthesized via precipitation and calcination methods and evaluated for fluoride (F−) and phosphate (PO43−) remediation. Comprehensive characterization was performed using X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FT-IR), scanning electron microscopy-energy dispersive X-ray spectroscopy (SEM-EDX), Raman spectroscopy, Brunauer–Emmett–Teller (BET), and dynamic light scattering (DLS) analyses. The adsorption performance was systematically examined under varying pH, initial pollutant concentration, contact time, temperature, and adsorbent dosage. The Ni(OH)2@kaolinite composite exhibited the highest adsorption capacities: 52.6 mg g−1 for fluoride and 55.0 mg g−1 for phosphate, with over 90% removal efficiency under optimal conditions. Kinetic data were best described by the pseudo second order model, indicating that the adsorption rate depends on the availability of active sites and adsorption capacity; however, kinetic fitting alone was not used to assign the adsorption mechanism, which was further evaluated using diffusion and thermodynamic analyses. The composites demonstrated excellent reusability, retaining over 75% efficiency after 10 cycles. Compared to conventional adsorbents, the Ni-doped kaolinite showed superior performance and cost-effectiveness (estimated treatment cost: <$5 per 1000 L), highlighting its potential as a sustainable, scalable solution for industrial wastewater treatment and resource recovery.
1. Introduction
Water contamination due to industrial discharge remains a major environmental challenge, particularly the presence of toxic organic and inorganic pollutants. Among various remediation strategies, heterogeneous catalysis has emerged as an effective and sustainable approach for wastewater treatment. The design of cost-effective, stable, and highly active catalysts is essential for improving pollutant degradation efficiency while ensuring reusability and environmental sustainability.1–4The removal of fluoride (F−) and phosphate (PO43−) from wastewater is critical due to their harmful environmental and health impacts. Excessive fluoride causes dental/skeletal fluorosis, while phosphate contributes to eutrophication, disrupting aquatic ecosystems. Traditional methods like precipitation and ion exchange are often inefficient or costly, prompting the development of advanced adsorbents for selective and sustainable remediation.5
Clay-based composites, such as metal–oxide-functionalized clays exhibit high affinity for F−, and PO43− through ligand exchange and electrostatic-interactions. For instance, La-modified bentonite achieves >95% fluoride removal via formation of La–F complexes, while Fe-coated clays adsorb phosphate through inner-sphere complexation.5–7 Chitosan–alginate hydrogels and biochar composites further enhance adsorption capacity (50–120 mg g−1 for F− and 40–100 mg g−1 for PO43−) while offering reusability (more than 5 cycles). Innovations like ZrO2 nanoparticles or layered double hydroxides (LDHs) improve selectivity in mixed-anion systems.
Coupling these materials with membrane filtration or electrocoagulation boosts efficiency, reducing residual concentrations below World Health Organization (WHO) limits (<1.5 mg L−1 F−, <0.1 mg L−1 PO43−). Future research should optimize scalability, cost, and regeneration protocols to bridge lab-scale success with industrial adoption, ensuring compliance with stringent water-quality standards.
Nickel-based catalysts (Ni and NiO) are particularly attractive for wastewater treatment due to their strong redox properties, excellent catalytic activity, and affordability compared to noble metal catalysts.6–8 However, the interaction between the active metal species and the clay support plays a critical role in determining catalytic efficiency, necessitating a deeper investigation into their behavior.9–13
In this study, NiO@kaolinite and Ni@kaolinite catalysts were synthesized and systematically evaluated for their catalytic performance in wastewater treatment. The materials were characterized using XRD, FT-IR, SEM-EDX, and BET to understand their structural, morphological, and compositional properties. To gain insight into their catalytic behavior, a comparative kinetic study was conducted to determine the reaction order and degradation mechanisms. Furthermore, statistical modeling was applied to assess the influence of key operational parameters such as pollutant concentration, temperature, pH, and catalyst dosage on wastewater treatment efficiency. The reusability and stability of the catalysts over multiple cycles were also investigated to evaluate their practical applicability.
This study aims to provide a comprehensive understanding of NiO and Ni-modified clay-based catalysts for wastewater treatment by integrating kinetic analysis, statistical modeling, and thermodynamic evaluation. The findings will contribute to the development of efficient, cost-effective, and environmentally friendly catalysts for industrial wastewater remediation.
2. Experimental
2.1. Materials
Nickel nitrate hexahydrate (Ni(NO3)2·6H2O, ≥98% purity), ammonium hydroxide (NH3, 33%), and kaolinite (clay ≥98% purity) were purchased from Sigma-Aldrich (United Kingdom). All chemicals were used without further treatment.2.2. Chemical treatment for kaolinite support
The acid treatment was performed using 6 mol per liter sulfuric acid solution for 3 hours at 90 °C under reflux with continuous stirring.14,15 A solid-to-liquid ratio of 5 g of kaolinite per 150 mL of acid solution was maintained.16 Following the treatment, the sample was washed with distilled water until it reached a neutral pH, then dried at 100 °C for 48 hours. The dried material was then subjected to peroxide treatment.17 The sample was treated with 35% (v/v) hydrogen peroxide at 25 °C for 3 hours, using solid to liquid ratio of 5 g of kaolinite per 100 mL of peroxide solution. Afterward, it was thoroughly washed with distilled water until neutral pH was achieved, then dried at 60 °C for 24 hours. Thermal treatment was conducted in a muffle furnace for 5 hours, where the sample was gradually heated from ambient temperature (25 °C) to 800 °C at a heating rate of 10 °C min−1. After calcination, the sample was transferred to an oven at 100 °C, and then stored in a desiccator until it cooled to room temperature. Finally, the material was macerated and sieved using a 0.425 mm sieve (Tyler series), yielding sample KT.2.3. Synthesis of NiO@kaolinite and Ni@kaolinite nanocomposites
The corresponding composites were prepared following the simple precipitation method. Initially, 2 mmol of nickel nitrate hexahydrate (Ni(NO3)2·6H2O) was introduced into 1500 mL of distilled water containing 5 gram of treated kaolinite-support. The mixture stirred for 30 min, until a homogeneous solution was obtained. This mixed solution was titrated with ammonia solution until (pH = 10) and maintained at 80 °C for 1 h. The resulting faint green product was separated by filtration and washed several times by distilled water to remove any residual components. The product was dried at 60 °C to give Ni(OH)2@kaolinite.18 Finally, the powder was calcined by two different ways first at 500 °C for six hour in a muffle furnace and the second under the same conditions but under vacuum to give NiO@kaolinite and Ni@kaolinite, respectively.2.4. Characterization tools
Several apparatuses of analysis were used to determine the vital characteristics of the prepared materials such as structural, morphological, and thermal features. X-ray diffraction (XRD) patterns were determined via Cu Kα X-ray radiation (Germany, λ = 1.540 Å, PANalytical X’PERT PRO). Fourier transform infrared spectroscopy (FT-IR) was used to detect the functional groups of samples from 4000 to 400 cm−1 using a PerkinElmer spectrometer via the KBr pellet method (Spectrum One, USA). The surface area of the as-synthesized materials is recorded by the adsorption–desorption isotherms of nitrogen at −196 °C (ANOVA 3200, USA). A high-resolution scanning electron microscope (HRSEM) was formfitting (JEOL, JSM [model no: 6360]) to confirm the morphological structure of the prepared samples. Dynamic Light Scattering (DLS) at 25 °C and a laser angle of 90° was used to determine the particle size distribution (Zetasizer Nano-ZS90 apparatus, Malvern Co., UK).2.5. Parameters of application
To regenerate the spent adsorbents, various desorption agents will be tested to determine their effectiveness in removing fluoride and phosphate. Alkaline solutions such as NaOH, KOH, and Ca(OH)2 (pH 10–12) will disrupt the electrostatic interactions between the adsorbed ions and the adsorbent surface. Acidic solutions (HCl, H2SO4, pH 2–4) will also be investigated, as they can protonate functional groups and weaken the attraction between the adsorbate and adsorbent. Additionally, salt solutions such as NaCl, NaNO3, and Na2SO4 (0.1–1.0 M) will induce competitive ion exchange and promote desorption. Simple distilled water washes will be tested as a control to evaluate the weakly bound fluoride and phosphate fraction. The effect of pH on desorption will be studied to identify optimal conditions for regenerating the adsorbents. By varying the pH from 2 to 12, the influence of protonation and deprotonation on the release of contaminants will be examined. A highly efficient regeneration process should achieve maximum desorption at minimal chemical consumption, ensuring economic viability. Desorption efficiency will be calculated based on the concentration of fluoride and phosphate released into the desorbing solution, which will be measured using ion chromatography (IC) or spectrophotometric methods.25
All batch adsorption experiments were conducted in triplicate under identical conditions. The reported values represent the arithmetic mean ± standard deviation (mean ± SD). Standard deviation was calculated using:
 | (1) |
![[x with combining macron]](https://www.rsc.org/images/entities/i_char_0078_0304.gif) is the mean value and n = 3. Reported uncertainties were rounded to two significant digits, and the mean values were rounded to the same decimal place as the corresponding standard deviation, in accordance with ISO/IEC Guide 98-3 (GUM:1995) recommendations for uncertainty reporting.
is the mean value and n = 3. Reported uncertainties were rounded to two significant digits, and the mean values were rounded to the same decimal place as the corresponding standard deviation, in accordance with ISO/IEC Guide 98-3 (GUM:1995) recommendations for uncertainty reporting.
Post-desorption characterization will be performed to assess the structural integrity and stability of the regenerated adsorbents. XRD analysis will be conducted to detect any crystallinity changes while SEM will provide insights into morphological and elemental composition alterations. BET surface area analysis will determine whether repeated use affects porosity and adsorption capacity. FT-IR will also be utilized to examine changes in functional groups, helping to identify any surface modifications resulting from multiple adsorption–desorption cycles.26
The reusability of the adsorbents will be evaluated over 5–10 consecutive adsorption–desorption cycles. For each cycle, fluoride and phosphate removal efficiency will be measured, and the percentage retention of adsorption capacity will be calculated. A decline of less than 20% after multiple cycles will indicate good reusability, whereas a significant reduction in efficiency may suggest surface degradation or pore blockage. The regeneration performance of nickel-based nanoparticles will also be compared with other commonly used adsorbents such as activated alumina, biochar, hydroxyapatite, and metal oxides. This comparative analysis will consider factors such as adsorption capacity retention, desorption efficiency, material cost, and long-term stability.
3. Results and discussion
3.1. XRD analysis
Materials synthesized were analyzed using X-ray diffraction (XRD) to study their crystalline structure and phase composition, as depicted in Fig. 1. The diffraction pattern of the pristine kaolinite support (black curve) shows peaks associated with kaolinite that corroborate with JCPDS card no. 00-014-0164, therefore confirming its purity and crystalline structure.1 | ||
| Fig. 1 XRD of kaolinite-supported, Ni(OH)2@kaolinite-support, Ni@kaolinite-supported, and NiO@kaolinite-supported. | ||
Distinct changes in the diffraction profiles were observed for the Ni(OH)2@kaolinite-support sample (green curve) that exhibits distinctive reflections around 2θ = 19.2°, 33.1°, 38.5°, 52.1°, and 59.1°, which were associated with the standard diffraction peaks of brucite-type nickel hydroxide [JCPDS card no. 00-014-0117]. It could be concluded that the deposition of Ni(OH)2 onto the kaolinite-support was successful.27
The alteration results in the Ni@kaolinite-support sample were achieved by reducing the Ni(OH)2 modified support, where new reflections appeared at 2θ = 44.5°, 51.8°, and 76.4° which can be ascribed to (111), (200), and (220) planes of metallic Ni (face centered cubic, JCPDS card no. 00-004-0850), respectively.28 This change confirms the assumption of synthesizing metallic nickel nanoparticles through thermal treatment or reduction as they exhaustively showed the Ni(OH)2 peaks.
After calcination, the NiO@kaolinite-support composite (blue curve) was formed. The oxidation of metallic Ni(OH)2 to NiO is confirmed by the distinctive peaks in the XRD pattern at 2θ = 37.2°, 43.3°, and 62.9°, which correspond to the (111), (200), and (220) planes of NiO (JCPDS card no. 00-047-1049).11 These broad peaks indicate that the NiO crystallites are nanoscale in nature. The disappearance of the characteristic kaolinite peaks in the XRD patterns of NiO@kaolinite and Ni@kaolinite (compared to their presence in Ni(OH)2@kaolinite) points to a significant structural transformation of the kaolinite support during the specific synthesis or post-treatment steps used to create the NiO and Ni phases. Kaolinite is thermally unstable. When heated to 500 °C, it undergoes an irreversible dehydroxylation reaction, losing its structural hydroxyl (OH) groups. This transforms the crystalline kaolinite into an amorphous, meta-kaolin phase (Al2Si2O7) without sharp diffraction peaks. The long-range crystalline order of kaolinite is destroyed, so it's characteristic XRD peaks vanish. The remaining peaks would be solely from the newly formed crystalline NiO. So, the absence of kaolinite peaks in NiO and Ni samples is strong evidence that your preparation method involved a high-temperature step that decomposed the crystalline kaolinite into an amorphous material, which now acts as a different type of support (thermally activated aluminosilicate). In contrast, the low-temperature synthesis of the Ni(OH)2 sample successfully preserved the original clay mineral structure.
3.2. FT-IR spectroscopy
FT-IR spectroscopy is a powerful analytical technique to predict synthesized materials molecular structure and composition, providing invaluable insights into their functional groups and chemical bonds. The FT-IR spectra of the kaolinite support and nickel-modified samples are presented in Fig. 2. The spectrum of pristine kaolinite displays characteristic bands associated with the kaolinite structure. The sharp peaks at 3650 cm−1 correspond to the O–H stretching vibrations of the inner surface hydroxyl groups of kaolinite. The band near 3620 cm−1 is attributed to inner hydroxyl stretching.2 Furthermore, the band near 1110–1000 cm−1 is thought to be linked to Si–O bonds, while the peaks between 910 to 530 cm−1 suggest the presence of Al–O–H and Si–O–Al bonds, further demonstrating the structural form of the kaolinite.29 Upon modification with Ni(OH)2, dramatic changes are observed. Broadening of the O–H stretching region is indicative of increased hydrogen bonding, which shows the presence of nickel hydroxide species on the surface of kaolinite. New bands at 520–470 cm−1 are due to Ni–O vibrations, confirming the formation of Ni(OH)2 on the support surface.30 In the case of the Ni@kaolinite-support sample, a diminution of the intensity of hydroxyl stretching bands can result from partial dehydroxylation on reduction as well as free hydroxyl loss. Also, weakening or absence of features related to Ni–OH serves to indicate the reduction of Ni(OH)2 to metallic Ni.31 | ||
| Fig. 2 FT-IR spectroscopy of kaolinite-support, Ni(OH)2@kaolinite-supported, Ni@kaolinite-support, and NiO@kaolinite-support. | ||
In NiO@kaolinite-support material (blue curve), O–H stretching vibrations are weak and still evident, and additional bands at approximately 560 cm−1 appear, and can be ascribed to Ni–O stretching vibrations of NiO, indicative of successful synthesis of NiO over kaolinite.32,33 Overall, the FT-IR results complement the XRD findings, confirming the presence and transformation of nickel species and their interaction with the kaolinite support through structural and surface hydroxyl modifications.
3.3. Raman analysis
The Raman spectra of the Ni(OH)2@kaolinite, Ni@kaolinite, and NiO@kaolinite composites are presented in Fig. 3 to provide insight into the vibrational properties and structural transformations of nickel species supported on kaolinite. The pristine kaolinite spectrum shows sharp and well-defined peaks, particularly in the regions around ∼145, 207, 267, 356, 400, 466, 515, 694, and 755 cm−1, which are characteristic of kaolinite's layered silicate structure.3 These bands can be assigned to Al–O and Si–O bending and stretching vibrations, consistent with literature values.33 | ||
| Fig. 3 Raman analysis of kaolinite-support, Ni(OH)2@kaolinite-support, Ni@kaolinite-support, and NiO@kaolinite-support. | ||
For the Ni(OH)2@kaolinite sample, additional bands appear that are not present in the kaolinite spectrum. Notably, broad peaks centered around ∼465 and 525 cm−1, along with several features in the 100–400 cm−1 region, correspond to the vibrational modes of Ni–O and Ni–OH bonds, indicative of brucite-like Ni(OH)2 structure. These peaks confirm the successful deposition of nickel hydroxide onto the kaolinite support.34
In the Ni@kaolinite sample (cyan curve), the characteristic Ni(OH)2 bands are significantly suppressed or completely absent. This reduction of intensity confirms the decomposition of nickel hydroxide and the formation of metallic Ni. While metallic Ni itself is generally Raman inactive due to its centrosymmetric FCC structure, the diminished Ni–OH peaks corroborate the transformation upon reduction.35
The NiO@kaolinite composite displays a distinct and sharp for one-phonon F2g mode of cubic NiO is typically reported (TO at 400–440 cm−1 and LO at 550 cm−1 modes). Accordingly, Fig. 3 shows a broad band centered around 550 cm−1 is observed, which corresponds to the characteristic F2g mode of NiO, confirming the presence of crystalline nickel oxide.36 This confirms the oxidation of nickel species to crystalline NiO. The absence of additional peaks in the NiO@kaolinite spectrum further supports the formation of pure NiO without residual hydroxide or metallic phases. Across all modified samples, the kaolinite peaks are retained with varying intensities, confirming that the support maintains its structural integrity during the deposition and transformation of the nickel species.37 In summary, Raman spectra provide complementary evidence to XRD and FT-IR results, confirming the successful synthesis and phase transformation of nickel species (Ni(OH)2 → Ni → NiO) supported on kaolinite.
3.4. BET analysis
The Brunauer–Emmett–Teller (BET) surface area analysis of the pristine kaolinite and nickel-modified composites Ni(OH)2@clay, NiO@clay, and Ni@clay reveals key insights into the structural modifications induced by nickel incorporation and their implications for adsorption performance as shown in Table 1. The pure kaolinite clay exhibited a high surface area of 140 m2 g−1, consistent with its layered alumino-g silicate structure, which provides abundant active sites for adsorption. Upon functionalization with nickel species, a gradual decrease in surface area was observed as; Ni(OH)2@clay (135 m2 g−1) shows the slight reduction suggests that nickel hydroxide deposition partially occupies the clay's pores without significant structural disruption. NiO@clay (120 m2 g−1) shows the further decrease indicates possible nanoparticle aggregation and pore blockage during thermal conversion of Ni(OH)2 to NiO. While Ni@clay (115 m2 g−1) shows the lowest surface area, observed for the metallic Ni-loaded clay, likely results from denser Ni nanoparticle formation and poor filling during reduction.38 This trend (clay > Ni(OH)2@clay > NiO@clay > Ni@clay) highlights that while nickel modification reduces surface area, the composites retain sufficient porosity (>115 m2 g−1) for effective adsorption. The retained surface area, combined with newly introduced active nickel sites, suggests a trade-off between textural properties and enhanced chemisorption capabilities.| Sample | Surface area (m2 g−1) | Pore volume (cm3 g−1) | Average pore diameter (nm) |
|---|---|---|---|
| Clay | 140 ± 5 | 0.25 ± 0.02 | 3.5 ± 0.1 |
| Ni(OH)@clay | 135 ± 4 | 0.22 ± 0.02 | 3.7 ± 0.1 |
| NiO@clay | 120 ± 3 | 0.18 ± 0.01 | 4.2 ± 0.2 |
| Ni@clay | 115 ± 3 | 0.15 ± 0.01 | 4.5 ± 0.2 |
Despite the reduced surface area, nickel-modified clays are expected to exhibit superior fluoride (F−) and phosphate (PO43−) adsorption due to:
• Enhanced chemisorption: Ni(OH)2 and NiO provide hydroxyl groups and Lewis acid sites that strongly interact with anions.
• Electrostatic attraction: the positively charged nickel species at neutral pH improve anion uptake.
• Ligand exchange mechanisms: phosphate and fluoride may replace surface OH groups on nickel phases.
The higher fluoride/phosphate removal efficiency of Ni(OH)2@clay and NiO@clay compared to pristine clay despite their lower surface area supports the dominance of chemical interactions over pure physical adsorption. The results concluded that; while nickel doping reduces kaolinite's surface area, the introduced active sites enhance specific surface interactions and ligand-exchange affinity toward fluoride and phosphate ions, making these composites highly effective for targeted anion removal. The balance between porosity and functionalization underscores their potential as multifunctional adsorbents for water treatment and resource recovery.39
3.5. SEM mapping-EDX analysis
To analyze the surface morphology and structural characteristics of the nanocomposites, scanning electron microscopy (SEM) was performed on the samples of Ni(OH)2, NiO@kaolinite, and Ni@kaolinite. Fig. 4 shows distinct morphological differences that reflect the effect of nickel incorporation and subsequent oxidation processes on the kaolinite support. | ||
| Fig. 4 SEM mapping-EDX of (a) Ni(OH)2@kaolinite-support (b) Ni@kaolinite-support, and (c) NiO@kaolinite-support. | ||
The pristine Ni(OH)2 sample shows a flaky, layered structure which suggests low crystallinity and high surface area morphology due to its poor crystallinity as well as abundant surface area.40 On the other hand, the Ni@kaolinite composite shows the successful deposition of nickel species onto the surface of kaolinite.
Nickel particles, which appear in the form of small and semi-spherical aggregates, coat the kaolinite layers and are uniformly dispersed throughout. This disposition confirms that a strong linkage between Ni and the kaolinite support was formed, which is beneficial to the surface activity.41
For the NiO@kaolinite composite, SEM images display a more compact and granular morphology compared to Ni@kaolinite. Formation of NiO is likely to cause some sintering or cross junctioning of the particles during thermal treatment, which diminishes surface porosity but increases structural integrity of the particles.18 The structure of kaolinite retains its identity, although less prominently because its edges have been softened and lowered because of the encasement and probable infiltration of NiO species.
The SEM analysis proved that Ni and NiO (NPs) supported on kaolinite were successfully synthesized while also depicting the morphological changes due to the conversion of hydroxide to oxide. Because of the changes in surface area, porosity, and the accessibility of active sites, these structural characteristics are vital in determining the performance of the composites as catalysts or adsorbents.
The elemental distribution and composition of the prepared nanocomposites were analyzed by energy-dispersive X-ray spectroscopy (EDX) mapping. For Ni(OH)2@kaolinite-support, the maps show even distribution of nickel (Ni) signals over the kaolinite platelets separated by co-localized Si, Al and O signals, verifying that deposition of the nickel hydroxide phase does not destroy the clay sheet morphology. In contrast, the Ni@kaolinite-supported sample exhibits numerous Ni clusters with higher intensity of the metallic state that are related to high concentration appearing in the Si and Al maps (indicating a more amorphous or sintered aluminosilicate support) as a result of reduction at high temperature which has produced metallic nickel and decomposed the crystalline kaolinite. The NiO@kaolinite-support maps show a homogeneous granular distribution of Ni and O throughout the support; whereas, Si and Al signals are spread out without clear platelet morphology, further confirming that kaolinite have been converted into amorphous meta-kaolin upon calcination with formation of well-dispersed NiO. Taken together, the EDX maps support the general phase transformations proposed by XRD: retention of kaolinite-like kaocuse-structure in the low temperature Ni(OH)2 sample, loss of this (kaolinite) structure during the syntheses to produce NiO and/or Ni counterpart that exhibit significant variation in both nickel dispersion and oxidation state.
3.6. Dynamic light scattering (DLS) measurements
The particle size distribution and zeta potential of the synthesized nanomaterials were obtained using the Dynamic Light Scattering (DLS) with a laser angle 90° at 25 °C, (Zetasizer Nano-ZS90 instrument, Malvern Co., UK). The DLS is an instrument commonly used to assess the hydrodynamic diameter of nano particles based on the Brownian suspension movement of particles.42 As shown in Fig. 5 and summarized in Table 2, the measurements provide insight into the colloidal stability and surface charge of the prepared samples: kaolinite, Ni(OH)2, Ni@kaolinite, and NiO@kaolinite. The surface charge and possible stability of the suspended (NPs) are indicated by the zeta potential values. In accordance with its known anionic surface, pure kaolinite showed a negative zeta potential of −3.38 mV.43 As a sign of positively charged hydroxide surfaces, Ni(OH)2 showed a noticeably positive zeta potential of +21.3 mV.44| Sample | Zeta potential (mV) |
|---|---|
| Kaolinite | −3.38 ± 0.10 |
| Ni(OH)2 | 21.3 ± 0.5 |
| Ni@kaolinite | −3.24 ± 0.12 |
| NiO@kaolinite | −3.12 ± 0.15 |
Both Ni@kaolinite (−3.24 mV) and NiO@kaolinite (−3.12 mV) maintained somewhat negative zeta potential values when added to the kaolinite support, although not as much as pure kaolinite. This implies electrostatic interaction or partial surface coverage between the nickel-based and kaolinite phases, which could affect the stability of the dispersion and the behavior of particle aggregation.45 These interactions are further supported by the DLS size distribution patterns.
The qualitative DLS profiles most likely display differences in particle size and polydispersity, as shown in (Fig. 5). Larger hydrodynamic diameters are usually the consequence of aggregation or the creation of core–shell structures when metallic or oxide species, such as Ni and NiO, are present in a kaolinite support. The Ni@kaolinite and NiO@kaolinite samples' near-neutral zeta potential values also suggest less electrostatic repulsion, which could result in more particle agglomeration. Overall, the DLS data demonstrate that nickel incorporation into kaolinite alters the surface charge and likely the size distribution of the resulting composites.46 These changes can significantly impact their dispersion behavior and potential applications in catalysis or adsorption, where surface characteristics are critical.
3.7. Fluoride (F−) adsorption study
| Initial fluoride concentration (mg L−1) | Adsorption capacity (qe, mg g−1) – Ni | Adsorption capacity (qe, mg g−1) – NiO | Adsorption capacity (qe, mg g−1) – Ni(OH)2 | Removal efficiency (%) |
|---|---|---|---|---|
| 5 | 4.2 ± 0.2 | 4.5 ± 0.2 | 5.1 ± 0.3 | 90 ± 2 |
| 10 | 8.0 ± 0.3 | 8.6 ± 0.3 | 9.4 ± 0.4 | 85 ± 2 |
| 25 | 18.2 ± 0.5 | 19.5 ± 0.6 | 21.0 ± 0.7 | 80 ± 3 |
| 50 | 30.6 ± 0.8 | 32.1 ± 0.9 | 34.2 ± 1.0 | 75 ± 3 |
| 100 | 48.7 ± 1.2 | 50.3 ± 1.3 | 52.6 ± 1.5 | 70 ± 4 |
 | ||
| Fig. 6 Adsorption capacity (qe) of Ni, NiO, and Ni(OH)2 nanoparticles at different initial fluoride concentrations, error bars represent ±one standard deviation (n = 3). | ||
At a lower initial fluoride concentration of 5 mg L−1, the adsorption capacity was 4.2 mg g−1 for Ni, 4.5 mg g−1 for NiO, and 5.1 mg g−1 for Ni(OH)2, achieving a high removal efficiency of 90%. As the initial fluoride concentration increased to 10 mg L−1, the adsorption capacity nearly doubled, reaching 8.0, 8.6, and 9.4 mg g−1 for Ni, NiO, and Ni(OH)2, respectively, with a removal efficiency of 85%. Further an increase in fluoride concentration to 25 mg L−1 and 50 mg L−1 increased adsorption capacity to 18.2–21.0 mg g−1 and 30.6–34.2 mg g−1, respectively. However, the removal efficiency decreased, dropping from 80% at 25 mg L−1 to 75% at 50 mg L−1. This decline suggests that the available adsorption sites become saturated at higher fluoride concentrations, limiting further fluoride uptake.
At the highest concentration of 100 mg L−1, the adsorption capacities reached 48.7 mg g−1 for Ni, 50.3 mg g−1 for NiO, and 52.6 mg g−1 for Ni(OH)2, but the removal efficiency further decreased to 70%. This reduction is likely due to competition among fluoride ions for the active adsorption sites and possible repulsion between adsorbed fluoride ions and those remaining in the solution. The results confirm that Ni(OH)2 exhibits the highest adsorption capacity among the three adsorbents, followed by NiO and Ni. This can be attributed to the higher surface area and hydroxyl functional groups of Ni(OH)2, which enhance fluoride affinity.
| Contact time (min) | Adsorption capacity (qe, mg g−1) – Ni | Adsorption capacity (qe, mg g−1) – NiO | Adsorption capacity (qe, mg g−1) – Ni(OH)2 | Removal efficiency (%) |
|---|---|---|---|---|
| 5 | 5.1 ± 0.2 | 5.3 ± 0.2 | 6.0 ± 0.3 | 45 ± 2 |
| 15 | 12.4 ± 0.4 | 13.0 ± 0.5 | 14.5 ± 0.6 | 60 ± 2 |
| 30 | 20.1 ± 0.6 | 21.2 ± 0.7 | 23.5 ± 0.8 | 75 ± 3 |
| 60 | 28.7 ± 0.9 | 30.1 ± 1.0 | 32.6 ± 1.1 | 90 ± 3 |
| 120 | 30.2 ± 1.0 | 31.8 ± 1.2 | 34.3 ± 1.3 |
 | ||
| Fig. 7 Fluoride adsorption kinetics over time, error bars represent ±one standard deviation (n = 3). | ||
At an initial contact time of 5 min, the adsorption capacities were 5.1 mg g−1 for Ni, 5.3 mg g−1 for NiO, and 6.0 mg g−1 for Ni(OH)2, with a removal efficiency of 45%. This rapid initial uptake suggests that many active adsorption sites were available for fluoride binding. As the contact time increased to 15 min, the adsorption capacity nearly doubled, reaching 12.4 mg g−1 (Ni), 13.0 mg g−1 (NiO), and 14.5 mg g−1 Ni(OH)2, with an improvement in removal efficiency to 60%. This phase represents the fast adsorption stage, where fluoride ions quickly occupy the readily available surface sites. By 30 min, the adsorption capacity further increased to 20.1 mg g−1 (Ni), 21.2 mg g−1 (NiO), and 23.5 mg g−1 Ni(OH)2, with a removal efficiency of 75%, indicating continued but slightly slower adsorption as sites become occupied.
At 60 min, the adsorption process approached equilibrium, with adsorption capacities of 28.7 mg g−1 (Ni), 30.1 mg g−1 (NiO), and 32.6 mg g−1 Ni(OH)2, corresponding to a high removal efficiency of 90%. Beyond this point, the adsorption rate slowed significantly as most active sites were saturated. At the final contact time of 120 min, a slight increase in adsorption capacity was observed (30.2 mg g−1 for Ni, 31.8 mg g−1 for NiO, and 34.3 mg g−1 for Ni(OH)2), with a 95% removal efficiency. This suggests that adsorption equilibrium was reached, and no significant fluoride removal occurred beyond this time. The kinetic data were best fitted by the pseudo second order model based on correlation coefficients and error analysis. It is important to note that this model provides a mathematical description of adsorption rate behavior and does not by itself confirm a chemisorption mechanism.
Therefore, additional diffusion and thermodynamic analyses were used to interpret the adsorption mechanism. Ni(OH)2 again demonstrated the highest adsorption capacity among the three adsorbents, likely due to its higher surface area and enhanced fluoride-binding interactions.
It should be emphasized that kinetic model fitting alone cannot be used to distinguish between physisorption and chemisorption mechanisms. The pseudo second order model is widely applied because of its strong empirical fitting performance across many adsorption systems, including those controlled by surface reaction, film diffusion, or intra-particle diffusion. Therefore, mechanistic interpretation in this study was based on combined evidence from pH dependence, surface chemistry, diffusion modeling, and thermodynamic parameters rather than kinetic fitting alone.
 | ||
| Fig. 8 Variation in fluoride adsorption efficiency at different pH values, error bars represent ±one standard deviation (n = 3). | ||
As the pH increased to 4, the adsorption capacity improved significantly, reaching 15.3 mg g−1 (Ni), 16.0 mg g−1 (NiO), and 17.4 mg g−1 Ni(OH)2, with a removal efficiency of 65%. This suggests that the adsorbent surfaces were becoming more negatively charged, enhancing electrostatic attraction between fluoride anions and the positively charged active sites. At a neutral pH of 6, maximum adsorption was observed, with capacities of 25.1 mg g−1 (Ni), 26.3 mg g−1 (NiO), and 28.0 mg g−1 Ni(OH)2, achieving a removal efficiency of 80%. This can be attributed to optimal electrostatic interactions, where the adsorbent surface retains a partial positive charge, facilitating strong fluoride binding.47
The highest fluoride adsorption occurred at a slightly alkaline pH of 8, where adsorption capacities reached 30.2 mg g−1 (Ni), 31.8 mg g−1 (NiO), and 34.0 mg g−1 Ni(OH)2, with a removal efficiency of 90%. At this pH, the adsorbent surfaces were still positively charged enough to attract fluoride ions, while there was minimal competition from hydroxide ions (OH−). Beyond pH 10, a slight decline in adsorption was observed, with capacities decreasing to 28.6 mg g−1 (Ni), 30.0 mg g−1 (NiO), and 32.1 mg g−1 Ni(OH)2, and removal efficiency dropping to 85%. This reduction is likely due to increased competition between fluoride (F−) and hydroxide (OH−) ions, as both anions have similar affinities for the adsorption sites. The overall trend indicates that pH 6–8 is the optimal range for fluoride removal, where electrostatic attraction and ion exchange mechanisms are most effective. The higher adsorption capacity of Ni(OH)2 compared to Ni and NiO further confirms its superior fluoride-binding ability, possibly due to its surface hydroxyl groups facilitating stronger interactions with fluoride ions.
3.8. Phosphate (PO43−) adsorption study
 | ||
| Fig. 9 Adsorption capacity (qe) of Ni, NiO, and Ni(OH)2 nanoparticles at different initial phosphate concentrations, error bars represent ±one standard deviation (n = 3). | ||
When the initial phosphate concentration increased to 10 mg L−1, the adsorption capacity nearly doubled, reaching 9.1 mg g−1 (Ni), 9.8 mg g−1 (NiO), and 10.6 mg g−1 Ni(OH)2. However, the removal efficiency slightly decreased to 88%. This indicates that more phosphate ions were available for adsorption, but competition for binding sites started to occur. At 25 mg L−1, the adsorption capacity further increased to 20.5 mg g−1 (Ni), 21.8 mg g−1 (NiO), and 23.4 mg g−1 Ni(OH)2, but the removal efficiency dropped to 82%. This reduction is attributed to site saturation, where some phosphate ions remain in the solution after reaching equilibrium.
At 50 mg L−1, the adsorption capacities reached 33.4 mg g−1 (Ni), 35.0 mg g−1 (NiO), and 37.5 mg g−1 Ni(OH)2, with a further decline in removal efficiency to 78%. The trend suggests that although the adsorbents continue to take up phosphate, their capacity to eradicate it diminishes as surface sites become increasingly occupied. At the highest concentration of 100 mg L−1, the maximum adsorption capacities observed were 50.3 mg g−1 (Ni), 52.1 mg g−1 (NiO), and 55.0 mg g−1 Ni(OH)2, with the lowest removal efficiency of 70%. This decline confirms that competition among phosphate ions for limited adsorption sites at high phosphate concentrations leads to reduced efficiency. Among the three adsorbents, Ni(OH)2 consistently exhibited the highest adsorption capacity, followed by NiO and Ni. The superior performance of Ni(OH)2 is likely due to its enhanced surface hydroxyl groups, which facilitate stronger electrostatic interactions and ligand exchange mechanisms with phosphate ions. These findings suggest that Ni, NiO, and Ni(OH)2 nanoparticles can effectively remove phosphate from aqueous solutions, especially at moderate concentrations (5–25 mg L−1). However, adsorption site saturation limits removal efficiency at higher phosphate levels, highlighting the importance of optimizing dosage and regeneration cycles for practical applications.
 | ||
| Fig. 10 Variation in phosphate adsorption efficiency at different pH values, error bars represent ±one standard deviation (n = 3). | ||
As the pH increased to 4, a significant improvement in adsorption was observed, with adsorption capacities rising to 17.4 mg g−1 (Ni), 18.2 mg g−1 (NiO), and 19.6 mg g−1 (Ni(OH)2), achieving a removal efficiency of 68%. This increase is due to the presence of H2PO4− ions, which have a stronger affinity for the positively charged adsorption sites. The highest phosphate adsorption occurred at a pH range of 6–8, where adsorption capacities reached 30.5 mg g−1 (Ni), 32.1 mg g−1 (NiO), and 34.7 mg g−1 (Ni(OH)2), with a removal efficiency of 88–92%. At this pH range, phosphate exists primarily as H2PO4− and HPO42−, which strongly interact with the metal hydroxide surfaces through electrostatic attraction and ligand exchange mechanisms. The high adsorption efficiency in this range confirms that Ni(OH)2 exhibits the best phosphate removal performance, likely due to its abundant hydroxyl groups that facilitate effective phosphate binding.
Beyond pH 8, the adsorption capacity declined slightly, with capacities of 28.2 mg g−1 (Ni), 29.8 mg g−1 (NiO), and 32.0 mg g−1 Ni(OH)2 at pH 10, leading to a removal efficiency of 85%. This reduction is attributed to the increased competition from hydroxide ions (OH−), which can occupy active adsorption sites and reduce phosphate retention. At higher pH values, phosphate exists mainly in the form of PO43−, which experiences greater electrostatic repulsion from the negatively charged adsorbent surfaces, further limiting adsorption efficiency.
3.9. Regeneration and reusability studies
The sustainability of Ni, NiO, and Ni(OH)2 nanoparticles as adsorbents for fluoride and phosphate removal was evaluated over multiple regeneration cycles, as shown in Tables 5 (fluoride) and 6 (phosphate), with Fig. 11 and 12. The results indicate a gradual decrease in adsorption capacity with increasing cycles, although the adsorbents retained significant efficiency even after ten regeneration cycles.| Cycle no. | Ni (qe, mg g−1) | Retention (%) | NiO (qe, mg g−1) | Retention (%) | Ni(OH)2 (qe, mg g−1) | Retention (%) |
|---|---|---|---|---|---|---|
| 1 | 30.2 ± 0.8 | 100 ± 2 | 31.8 ± 0.9 | 100 ± 2 | 34.3 ± 1.0 | 100 ± 2 |
| 2 | 28.9 ± 0.7 | 96 ± 2 | 30.5 ± 0.8 | 96 ± 2 | 32.8 ± 0.9 | 96 ± 2 |
| 3 | 27.1 ± 0.7 | 90 ± 3 | 29.3 ± 0.8 | 92 ± 3 | 31.1 ± 0.9 | 91 ± 3 |
| 4 | 26.0 ± 0.6 | 86 ± 3 | 28.1 ± 0.7 | 88 ± 3 | 30.0 ± 0.8 | 87 ± 3 |
| 5 | 25.4 ± 0.6 | 84 ± 3 | 27.0 ± 0.7 | 85 ± 3 | 29.3 ± 0.8 | 85 ± 3 |
| 6 | 24.0 ± 0.6 | 79 ± 4 | 25.5 ± 0.7 | 80 ± 4 | 28.0 ± 0.8 | 82 ± 4 |
| 7 | 23.2 ± 0.5 | 77 ± 4 | 24.9 ± 0.6 | 78 ± 4 | 27.0 ± 0.7 | 79 ± 4 |
| 8 | 22.5 ± 0.5 | 75 ± 4 | 24.3 ± 0.6 | 76 ± 4 | 26.5 ± 0.7 | 77 ± 4 |
| 9 | 21.8 ± 0.5 | 72 ± 5 | 24.0 ± 0.6 | 75 ± 4 | 26.4 ± 0.7 | 77 ± 4 |
| 10 | 21.5 ± 0.5 | 71 ± 5 | 23.8 ± 0.6 | 75 ± 4 | 26.2 ± 0.7 | 76 ± 4 |
| Cycle no. | Ni (qe, mg g−1) | Retention (%) | NiO (qe, mg g−1) | Retention (%) | Ni(OH)2 (qe, mg g−1) | Retention (%) |
|---|---|---|---|---|---|---|
| 1 | 32.0 ± 0.8 | 100 ± 2 | 34.0 ± 0.9 | 100 ± 2 | 36.2 ± 1.0 | 100 ± 2 |
| 2 | 30.5 ± 0.7 | 95 ± 2 | 32.5 ± 0.8 | 96 ± 2 | 34.8 ± 0.9 | 96 ± 2 |
| 3 | 29.6 ± 0.7 | 92 ± 3 | 31.7 ± 0.8 | 93 ± 3 | 33.5 ± 0.9 | 93 ± 3 |
| 4 | 28.2 ± 0.6 | 88 ± 3 | 30.5 ± 0.7 | 90 ± 3 | 32.4 ± 0.8 | 90 ± 3 |
| 5 | 27.6 ± 0.6 | 86 ± 3 | 29.8 ± 0.7 | 88 ± 3 | 31.5 ± 0.8 | 87 ± 3 |
| 6 | 26.8 ± 0.6 | 84 ± 4 | 29.0 ± 0.7 | 85 ± 4 | 30.7 ± 0.8 | 85 ± 4 |
| 7 | 25.6 ± 0.6 | 80 ± 4 | 28.0 ± 0.7 | 82 ± 4 | 30.0 ± 0.8 | 83 ± 4 |
| 8 | 24.8 ± 0.5 | 78 ± 4 | 27.3 ± 0.6 | 80 ± 4 | 29.6 ± 0.7 | 82 ± 4 |
| 9 | 24.5 ± 0.5 | 77 ± 5 | 27.0 ± 0.6 | 79 ± 4 | 29.2 ± 0.7 | 81 ± 4 |
| 10 | 24.3 ± 0.5 | 76 ± 5 | 26.8 ± 0.6 | 79 ± 4 | 29.0 ± 0.7 | 80 ± 4 |
 | ||
| Fig. 11 Fluoride adsorption efficiency over multiple regeneration cycles, error bars represent ±one standard deviation (n = 3). | ||
 | ||
| Fig. 12 Phosphate adsorption efficiency over multiple regeneration cycles, error bars represent ±one standard deviation (n = 3). | ||
The initial adsorption capacities were 30.2 mg g−1 (Ni), 31.8 mg g−1 (NiO), and 34.3 mg g−1 Ni(OH)2 in the first cycle, with 100% adsorption retention. After the second and third regeneration cycles, adsorption capacity slightly decreased, with Ni(OH)2 maintaining the highest efficiency at 32.8 mg g−1 (cycle 2) and 31.1 mg g−1 (cycle 3), corresponding to 95% and 90% adsorption retention, respectively. By the fifth cycle, adsorption capacities further declined to 25.4 mg g−1 (Ni), 27.0 mg g−1 (NiO), and 29.3 mg g−1 Ni(OH)2, with adsorption retention of 85%, indicating a steady but controlled loss of efficiency due to possible fouling or minor structural degradation.
At the tenth cycle, the adsorption capacity was reduced to 21.5 mg g−1 (Ni), 23.8 mg g−1 (NiO), and 26.2 mg g−1 Ni(OH)2, with adsorption retention dropping to 75%. Despite this decline, the nanoparticles exhibited reasonable efficiency, confirming their suitability for multiple reuse cycles. The reduction in performance is likely attributed to residual fluoride ions blocking active sites, partial dissolution of metal oxides, or minor changes in surface chemistry.
The phosphate adsorption study followed a similar trend, with the highest initial adsorption capacities of 32.0 mg g−1 (Ni), 34.0 mg g−1 (NiO), and 36.2 mg g−1 Ni(OH)2 at 100% adsorption retention in the first cycle. By the second cycle, adsorption capacities declined slightly to 30.5 mg g−1 (Ni), 32.5 mg g−1 (NiO), and 34.8 mg g−1 (Ni(OH)2), with 96% retention, demonstrating that the adsorbents maintained high efficiency after regeneration.49
In the third and fifth cycles, adsorption capacity decreased gradually, reaching 31.5 mg g−1 for Ni(OH)2 at cycle 5, with adsorption retention of 87%, which remains highly effective. After ten cycles, the adsorption capacity further declined to 24.3 mg g−1 (Ni), 26.8 mg g−1 (NiO), and 29.0 mg g−1 Ni(OH)2, with a final retention of 78%. This trend suggests that the adsorbents remained functional even after prolonged reuse, making them viable for practical wastewater treatment applications.
3.10. Adsorption kinetics modeling
To elucidate the adsorption mechanisms and rate-controlling steps, experimental kinetic data were fitted to pseudo first order, pseudo second order, and intra-particle diffusion models. The kinetic parameters were determined from the linearized forms of these models and are presented in Tables 7–9 for fluoride and phosphate adsorption.
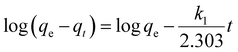 | (2) |
| Adsorbent | qe,exp. (mg g−1) | qe,calc. (mg g−1) | k1 (min−1) | R2 | χ2 |
|---|---|---|---|---|---|
| Ni@kaolinite | 30.2 ± 0.8 | 27.8 ± 1.2 | 0.031 ± 0.003 | 0.931 | 2.45 |
| NiO@kaolinite | 31.8 ± 0.7 | 29.1 ± 1.1 | 0.028 ± 0.002 | 0.918 | 3.12 |
| Ni(OH)2@kaolinite | 34.3 ± 0.9 | 31.5 ± 1.3 | 0.025 ± 0.003 | 0.902 | 3.87 |
The pseudo first order model showed moderate correlation coefficients (R2 = 0.902–0.931), with calculated qe values deviating significantly from experimental values. The higher χ2 values (2.45–3.87) indicate poor fit, suggesting that the adsorption process does not follow first-order kinetics. This deviation implies that the rate is not solely dependent on the concentration of available sites.
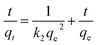 | (3) |
| Adsorbent | Ion | qe,exp. (mg g−1) | qe,calc. (mg g−1) | k2 (g mg−1 min−1) | h0 (mg g−1 min−1) | R2 | χ2 |
|---|---|---|---|---|---|---|---|
| Ni@kaolinite | F− | 30.2 ± 0.8 | 30.6 ± 0.5 | 0.0032 ± 0.0002 | 2.99 | 0.991 | 0.15 |
| Ni@kaolinite | PO43− | 32.0 ± 0.9 | 32.4 ± 0.6 | 0.0028 ± 0.0003 | 2.94 | 0.989 | 0.18 |
| NiO@kaolinite | F− | 31.8 ± 0.7 | 32.4 ± 0.4 | 0.0039 ± 0.0003 | 4.09 | 0.994 | 0.12 |
| NiO@kaolinite | PO43− | 34.0 ± 0.8 | 34.6 ± 0.5 | 0.0035 ± 0.0002 | 4.19 | 0.992 | 0.14 |
| Ni(OH)2@kaolinite | F− | 34.3 ± 0.9 | 34.9 ± 0.4 | 0.0046 ± 0.0004 | 5.60 | 0.996 | 0.08 |
| Ni(OH)2@kaolinite | PO43− | 36.2 ± 1.0 | 37.1 ± 0.5 | 0.0042 ± 0.0003 | 5.77 | 0.995 | 0.10 |
The pseudo second order model demonstrated excellent fit to the experimental data, with high correlation coefficients (R2 > 0.989) and low χ2 values (0.08–0.18). The close agreement between experimental and calculated qe values confirms that this model accurately describes the adsorption kinetics. The initial adsorption rates (h0) increased in the order: Ni@kaolinite < NiO@kaolinite < Ni(OH)2@kaolinite, indicating that Ni(OH)2@kaolinite exhibits the fastest initial uptake rate. However, it is important to note that the pseudo second order model provides a mathematical description of the rate behavior and does not by itself confirm a specific adsorption mechanism. The model's success in fitting the data indicates that the adsorption rate is proportional to the square of the number of unoccupied sites, which is consistent with various mechanisms including surface complexation, electrostatic attraction, and ligand exchange. Therefore, additional thermodynamic and diffusion analyses are necessary to fully characterize the adsorption mechanism (see Sections 3.11 and 3.12).
| qt = kipt0.5 + C | (4) |
| Adsorbent | Stage 1 | Stage 2 | Stage 3 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| kip1 | C1 | R2 | kip2 | C2 | R2 | kip3 | C3 | R2 | |
| Ni@kaolinite | 3.85 | 2.14 | 0.985 | 1.42 | 12.8 | 0.962 | 0.28 | 26.5 | 0.891 |
| NiO@kaolinite | 4.12 | 1.87 | 0.988 | 1.56 | 13.5 | 0.971 | 0.31 | 28.2 | 0.903 |
| Ni(OH)2@kaolinite | 4.58 | 1.52 | 0.992 | 1.78 | 14.2 | 0.978 | 0.35 | 30.8 | 0.912 |
The intra-particle diffusion plots exhibited three distinct linear regions, indicating a multi-stage adsorption process. Stage 1 (high kip1 values) represents rapid external surface adsorption and film diffusion. Stage 2 (moderate kip2 values) corresponds to gradual intra-particle diffusion into pores. Stage 3 (low kip3 values) represents the final equilibrium stage where adsorption rate slows due to decreased driving force. Importantly, the non-zero intercept values (C > 0) confirm that intra-particle diffusion is not the sole rate-controlling step. Instead, both surface adsorption and pore diffusion contribute to the overall adsorption process. The higher C values for Ni(OH)2@kaolinite indicate a greater contribution from boundary layer diffusion, which is consistent with its enhanced surface chemistry.
3.11. Thermodynamic analysis
Thermodynamic parameters, including Gibbs free energy change (ΔG°), enthalpy change (ΔH°), and entropy change (ΔS°), were calculated to understand the spontaneity, heat changes, and randomness associated with the adsorption process. These parameters were determined using the van 't Hoff equation:
 | (5) |
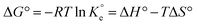 | (6) |
 is the dimensionless equilibrium constant. To ensure thermodynamic validity, the Langmuir constant (KL, in L mg−1) was converted to a dimensionless standard equilibrium constant:
is the dimensionless equilibrium constant. To ensure thermodynamic validity, the Langmuir constant (KL, in L mg−1) was converted to a dimensionless standard equilibrium constant:
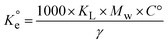 | (7) |
| Adsorbate | T (K) | KL (L mg−1) | Mw (g mol−1) | (dimensionless) |
|---|---|---|---|---|
| Fluoride | 298 | 0.082 ± 0.005 | 19.00 | 1558 |
| Fluoride | 308 | 0.094 ± 0.006 | 19.00 | 1786 |
| Fluoride | 318 | 0.109 ± 0.007 | 19.00 | 2071 |
| Fluoride | 328 | 0.128 ± 0.008 | 19.00 | 2432 |
| Phosphate | 298 | 0.065 ± 0.004 | 94.97 | 6173 |
| Phosphate | 308 | 0.072 ± 0.005 | 94.97 | 6838 |
| Phosphate | 318 | 0.081 ± 0.006 | 94.97 | 7693 |
| Phosphate | 328 | 0.093 ± 0.007 | 94.97 | 8832 |
The dimensionless equilibrium constant  increased with temperature for both fluoride and phosphate, indicating enhanced adsorption at higher temperatures. This trend is characteristic of endothermic adsorption processes. The calculated thermodynamic parameters are presented in Table 11.
increased with temperature for both fluoride and phosphate, indicating enhanced adsorption at higher temperatures. This trend is characteristic of endothermic adsorption processes. The calculated thermodynamic parameters are presented in Table 11.
The negative ΔG° values at all temperatures confirm that the adsorption of fluoride and phosphate onto Ni(OH)2@kaolinite is thermodynamically spontaneous and favorable. The magnitude of ΔG° became more negative with increasing temperature, indicating greater spontaneity at higher temperatures. The positive ΔH° values (24.6 kJ mol−1 for fluoride and 31.8 kJ mol−1 for phosphate) confirm that the adsorption process is endothermic, requiring energy input to proceed. These ΔH° values fall within the typical range of physical adsorption and surface complexation processes (<40 kJ mol−1), which distinguishes them from strong chemisorption processes that typically exhibit ΔH° > 80 kJ mol−1. Therefore, the adsorption mechanism cannot be classified as chemisorption based on thermodynamic magnitude alone. Instead, the process is attributed to a combination of electrostatic attraction, ligand exchange, and outer-sphere/inner-sphere surface complexation mechanisms. The positive ΔS° values (96 J mol−1 K−1 for fluoride and 112 J mol−1 K−1 for phosphate) indicate increased randomness at the solid–solution interface during adsorption. This entropy increase is attributed to the release of water molecules and counter-ions from the adsorbent surface upon fluoride and phosphate binding, resulting in a net increase in system disorder. The favorable entropy change further supports the spontaneous nature of the adsorption process.45
3.12. Adsorption isotherm modeling
Adsorption isotherms provide critical insights into the adsorption mechanism, surface properties, and maximum adsorption capacity. The experimental equilibrium data were analyzed using three widely applied isotherm models: Langmuir, Freundlich, and Temkin. These models were fitted to the data, and the parameters were determined from their linearized forms.
 | (8) |
| RL = 1/(1 + KLC0) | (9) |
| Adsorbent | Ion | qmax (mg g−1) | KL (L mg−1) | RL | R2 | χ2 |
|---|---|---|---|---|---|---|
| Ni@kaolinite | F− | 52.4 ± 2.1 | 0.068 ± 0.005 | 0.128–0.746 | 0.987 | 0.82 |
| Ni@kaolinite | PO43− | 54.8 ± 2.3 | 0.053 ± 0.004 | 0.159–0.790 | 0.983 | 1.05 |
| NiO@kaolinite | F− | 55.9 ± 2.0 | 0.075 ± 0.006 | 0.118–0.727 | 0.991 | 0.65 |
| NiO@kaolinite | PO43− | 58.2 ± 2.2 | 0.061 ± 0.005 | 0.141–0.766 | 0.988 | 0.78 |
| Ni(OH)2@kaolinite | F− | 58.6 ± 1.8 | 0.082 ± 0.006 | 0.109–0.709 | 0.994 | 0.45 |
| Ni(OH)2@kaolinite | PO43− | 62.1 ± 2.0 | 0.069 ± 0.005 | 0.126–0.744 | 0.992 | 0.58 |
The Langmuir model demonstrated excellent fit to the experimental data with high correlation coefficients (R2 > 0.983) and low χ2 values, indicating that monolayer adsorption is the dominant mechanism. The maximum adsorption capacities (qmax) followed the order: Ni(OH)2@kaolinite > NiO@kaolinite > Ni@kaolinite, with Ni(OH)2@kaolinite exhibiting the highest capacity (58.6 mg g−1 for F− and 62.1 mg g−1 for PO43−). The separation factor (RL) values for all adsorbents ranged between 0.109 and 0.790, confirming favorable adsorption across the entire concentration range studied. The higher KL values for Ni(OH)2@kaolinite indicate stronger adsorbent–adsorbate affinity, which is attributed to abundant surface hydroxyl groups facilitating ligand exchange and electrostatic interactions.
 | (10) |
| Adsorbent | Ion | KF ((mg g−1) (L mg−1)1/n) | n | 1/n | R2 | χ2 |
|---|---|---|---|---|---|---|
| Ni@kaolinite | F− | 8.52 ± 0.65 | 2.18 | 0.459 | 0.952 | 2.85 |
| Ni@kaolinite | PO43− | 9.14 ± 0.72 | 2.05 | 0.488 | 0.948 | 3.12 |
| NiO@kaolinite | F− | 9.87 ± 0.58 | 2.31 | 0.433 | 0.961 | 2.34 |
| NiO@kaolinite | PO43− | 10.5 ± 0.64 | 2.14 | 0.467 | 0.956 | 2.68 |
| Ni(OH)2@kaolinite | F− | 11.2 ± 0.52 | 2.45 | 0.408 | 0.968 | 1.92 |
| Ni(OH)2@kaolinite | PO43− | 12.1 ± 0.58 | 2.28 | 0.439 | 0.963 | 2.21 |
The Freundlich model showed moderate fit with R2 values ranging from 0.948 to 0.968, which are lower than those obtained for the Langmuir model. The 1/n values (0.408–0.488) all fell between 0 and 1, confirming favorable adsorption. The n values greater than 1 (2.05–2.45) indicate that the adsorbents have high adsorption affinity at low equilibrium concentrations. The higher χ2 values compared to the Langmuir model suggest that the Freundlich model is less suitable for describing the adsorption mechanism, implying that monolayer adsorption on relatively homogeneous surfaces is more representative than multilayer heterogeneous adsorption.
qe = B![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KT + B KT + B![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ce Ce
| (11) |
| Adsorbent | Ion | KT (L mg−1) | bT (kJ mol−1) | B | R2 | χ2 |
|---|---|---|---|---|---|---|
| Ni@kaolinite | F− | 1.42 ± 0.12 | 0.485 | 5.11 | 0.971 | 1.68 |
| Ni@kaolinite | PO43− | 1.28 ± 0.11 | 0.512 | 4.84 | 0.967 | 1.92 |
| NiO@kaolinite | F− | 1.67 ± 0.13 | 0.458 | 5.41 | 0.978 | 1.32 |
| NiO@kaolinite | PO43− | 1.51 ± 0.12 | 0.492 | 5.04 | 0.974 | 1.54 |
| Ni(OH)2@kaolinite | F− | 1.89 ± 0.14 | 0.432 | 5.74 | 0.983 | 1.05 |
| Ni(OH)2@kaolinite | PO43− | 1.73 ± 0.13 | 0.468 | 5.30 | 0.980 | 1.18 |
The Temkin model demonstrated good fit with R2 values ranging from 0.967 to 0.983, indicating that adsorbent–adsorbate interactions play a significant role in the adsorption process. The positive KT values confirm favorable binding. The bT values (0.432–0.512 kJ mol−1) are relatively low, suggesting weak to moderate interaction energies consistent with physical adsorption and surface complexation rather than strong chemical bonding. The B values (4.84–5.74) represent the variation in adsorption energy and confirm the heterogeneous nature of the adsorption process to some extent. Overall, Temkin model provided better fit than Freundlich but was still inferior to the Langmuir model; supporting the conclusion that monolayer adsorption on relatively uniform sites is the predominant mechanism.
| Isotherm model | Ion | R2 | χ2 | Model ranking |
|---|---|---|---|---|
| Langmuir | F− | 0.994 | 0.45 | 1st |
| Langmuir | PO43− | 0.992 | 0.58 | 1st |
| Temkin | F− | 0.983 | 1.05 | 2nd |
| Temkin | PO43− | 0.980 | 1.18 | 2nd |
| Freundlich | F− | 0.968 | 1.92 | 3rd |
| Freundlich | PO43− | 0.963 | 2.21 | 3rd |
4. Cost analysis and comparative assessment of advanced nickel-based nanostructured doped kaolinite as a multifunctional adsorbent
The increasing contamination of water bodies with fluoride (F−) and phosphate (PO43−) poses significant environmental and health risks. Conventional remediation techniques such as precipitation, ion exchange, and reverse osmosis are often costly, energy-intensive, or inefficient at low contaminant concentrations. Adsorption-based methods using nanostructured materials have emerged as promising alternatives due to their high efficiency, selectivity, and potential for resource recovery. This study evaluates the cost-effectiveness and comparative performance of nickel-doped nanostructured kaolinite as a multifunctional adsorbent for simultaneous fluoride and phosphate removal. Additionally, we assess its potential for sustainable resource recovery through desorption and regeneration. Ni–kaolinite could achieve <$5 per 1000 L treatment, making it competitive with conventional methods. Tables 16 and 17 show cost analysis.| Component | Cost estimate |
|---|---|
| Kaolinite raw material | $50–100 per ton |
| Nickel precursor | $20–50 per kg |
| Synthesis and processing | $100–200 per kg |
| Characterization | $500–1000 (initial) |
| Total estimated cost | $200–400 per kg |
| Adsorbent | Cost per kg | Adsorption capacity (mg g−1) | Regeneration cycles | Cost per 1000 L treatment |
|---|---|---|---|---|
| Ni–kaolinite | $200–400 | F−: 50–100, PO43−: 80–120 | 5–10 | $5–10 |
| Activated alumina | $300–500 | F−: 20–40, PO43−: 10–30 | 3–5 | $15–25 |
| Iron oxide NPs | $400–600 | F−: 30–60, PO43−: 40–70 | 4–7 | $20–30 |
| Biochar | $100–300 | F−: 10–30, PO43−: 20–50 | 2–4 | $10–20 |
| Ion-exchange resins | $1000–5000 | F−: 50–80, PO43−: 60–100 | 10–15 | $50–100 |
4.1. Comparative performance assessment
The use of natural kaolinite in Ni–kaolinite reduces reliance on synthetic materials, lowering environmental impacts associated with mining and processing. Nickel doping improves structural stability, preventing metal leaching and ensuring long-term adsorbent safety. Additionally, the ability to recover phosphate from treated water aligns with circular economy principles, transforming waste into a valuable resource for agricultural applications. This approach not only mitigates pollution but also promotes sustainable resource utilization, making Ni–KAO an eco-friendly solution for water remediation. Table 18 shows adsorption efficiency.| Parameter | Ni–kaolinite | Activated alumina | Iron oxide | Biochar | Ion-exchange resins |
|---|---|---|---|---|---|
| Fluoride uptake | 50–100 mg g−1 | 20–40 mg g−1 | 30–60 mg g−1 | 10–30 mg g−1 | 50–80 mg g−1 |
| Phosphate uptake | 80–120 mg g−1 | 10–30 mg g−1 | 40–70 mg g−1 | 20–50 mg g−1 | 60–100 mg g−1 |
| pH stability | 4–9 | 5–8 | 3–8 | 6–9 | 4–10 |
| Selectivity | High | Moderate | Moderate to high | Low | High |
5. Conclusions
This study successfully demonstrated the effectiveness of nickel doped kaolinite (Ni@kaolinite and NiO@kaolinite) as multifunctional adsorbents for the efficient remediation of fluoride and phosphate from aqueous systems while enabling sustainable resource recovery. The synthesized catalysts exhibited superior adsorption performance, following pseudo second order kinetics, indicating a adsorption governed by electrostatic attraction and surface complexation interactions supported by moderate enthalpy values. Advanced characterization techniques (XRD, FT-IR, SEM mapping-EDX, BET, and DLS) confirmed the enhanced dispersion of Ni species on the kaolinite support, improving catalytic activity and stability. Ni@kaolinite outperformed NiO@kaolinite, achieving higher removal efficiencies due to improved electron transfer and redox properties. Also, statistical and thermodynamic analyses revealed an endothermic, spontaneous process, optimizing parameters such as pH, temperature, and catalyst dosage. Excellent reusability over ten cycles underscores the material's long-term economic and environmental viability. Phosphate recovery potential aligns with circular economy principles, transforming waste into a reusable resource. These results highlight clay-supported Ni-based catalysts as cost-effective, sustainable, and scalable solutions for industrial wastewater treatment. Future research should focus on large-scale applications, real wastewater testing, and life-cycle assessments to further validate their industrial feasibility. This work contributes significantly to green chemistry and environmental remediation, offering a promising pathway for pollutant removal and resource recovery.Author contributions
Aya M. Matloob and Mahmoud F. Mubarak: methodology, formal analysis, data aquation, resources and wrote the original draft. Tahany Mahmoud and Asmaa S. Morshedy: resources, methodology, formal analysis, conceptualization, data curation, investigation, software, validation, visualization, writing – original draft, writing – review, and editing. All authors approved the current and final version of the manuscript for submission.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
All data is available in the manuscript and is available when requested by the corresponding authors.Acknowledgements
Authors used DeepSeek AI software during the preparation of this work to improve language, readability, and grammar of specific sections. After using this tool, authors reviewed and edited the content as needed and take full responsibility for the content of published manuscript.References
- T. E. Oladimeji, et al., Review on the impact of heavy metals from industrial wastewater effluent and removal technologies, Heliyon, 2024, 10(23), e40370 CrossRef CAS PubMed.
- H. R. Ahmed, Heterogeneous catalysts in advanced oxidation processes: A comprehensive review on antibiotic removal from wastewater, Sep. Purif. Technol., 2025, 374, 133670 CrossRef CAS.
- V. Kumar, et al., Removal of Inorganic Pollutants from Wastewater: Innovative Technologies and Toxicity Assessment, Sustainability, 2023, 15, 16376 CrossRef CAS.
- H. W. Ahmad, et al., Sustainable Wastewater Treatment Strategies in Effective Abatement of Emerging Pollutants, Water, 2024, 16, 2893 CrossRef CAS.
- E. Sanad, et al., Tailored alginate nanocomposite beads for efficient heavy metals sequestration from industrial wastewater, Int. J. Environ. Anal. Chem., 2026, 106(5), 1115–1141 CrossRef CAS.
- A. S. Morshedy, A. M. A. El-Naggar and M. T. Zaky, Sonophotocatalytic reformation: Transforming paraffin wax into hydrogen-rich fuel, Int. J. Hydrogen Energy, 2026, 223, 154271 CrossRef CAS.
- M. A. A. El-Khair, et al., Harvesting the synergistic effect of CuFe2O4@Ni-MOF nanomagnetic photocatalyst for enhanced visible light-driven green hydrogen production, Int. J. Hydrogen Energy, 2025, 101, 280–294 CrossRef CAS.
- A. S. Morshedy, et al., Ni@CaTiO3 Nanocomposites for Methane Decomposition Into Hydrogen and Multiwalled Carbon Nanotubes, ChemistrySelect, 2024, 9(32), e202401908 CrossRef CAS.
- F. Huang, et al., Catalytic performances of Ni/mesoporous SiO2 catalysts for dry reforming of methane to hydrogen, J. Energy Chem., 2016, 25(4), 709–719 CrossRef.
- Y. Cai, et al., Recent Advances in Ni-Based Catalysts for CH4-CO2 Reforming (2013–2023), Atmosphere, 2023, 14, 1323 CrossRef CAS.
- K. S. Mohammed, et al., Synthesis of Kaolin-Supported Nickel Oxide Composites for the Catalytic Oxidative Degradation of Methylene Blue Dye, ACS Omega, 2024, 9(4), 4287–4299 CrossRef CAS PubMed.
- M. A. Abo El-Khair, A. S. Morshedy and A. G. Al-Gamal, Synergistic enhancement of photocatalytic hydrogen evolution via NiSX-NiO/S-g-C3N4 heterojunctions under visible light irradiation, Fuel, 2026, 413, 138239 CrossRef CAS.
- H. M. Abd El Salam and A. S. Morshedy, TiO2@Metal organic framework Ni2(BDC)2(DABCO) as an efficient water splitting heterogeneous photocatalyst for enhanced green hydrogen generation under visible light, Int. J. Hydrogen Energy, 2025, 151, 150142 CrossRef CAS.
- O. B. Ayodele, Effect of phosphoric acid treatment on kaolinite supported ferrioxalate catalyst for the degradation of amoxicillin in batch photo-Fenton process, Appl. Clay Sci., 2013, 72, 74–83 CrossRef CAS.
- J. D. D. Melo, et al., Effects of thermal and chemical treatments on physical properties of kaolinite, Ceram. Int., 2010, 36(1), 33–38 CrossRef CAS.
- A. K. Panda, et al., Effect of sulphuric acid treatment on the physico-chemical characteristics of kaolin clay, Colloids Surf., A, 2010, 363(1), 98–104 CrossRef CAS.
- T. A. Sokolova, et al., The Effect of Treatment with Hydrogen Peroxide and the Mehra–Jackson Reagent on X-ray Diffraction Patterns of Clay Fractions, Eurasian Soil Sci., 2017, 50(12), 1386–1394 CrossRef CAS.
- K. S. Mohammed and M. Atlabachew, Synthesis of Kaolin-Supported Nickel Oxide Composites for the Catalytic Oxidative Degradation of Methylene Blue Dye, ACS Omega, 2024, 9(4), 4287–4299 CrossRef CAS PubMed.
- H. A. El-Sabban, M. F. Mubarak and M. A. Diab, PPy-NTs/C/TiO2/poly(ether sulfone) porous composite membrane: Efficient ultrafiltration of Evans blue dye from industrial wastewater, Synth. Met., 2023, 297, 117383 CrossRef CAS.
- A. El-Denglawey, M. F. Mubarak and H. Selim, Tertiary Nanocomposites of Metakaolinite/Fe3O4/SBA-15 Nanocomposite for the Heavy Metal Adsorption: Isotherm and Kinetic Study, Arabian J. Sci. Eng., 2022, 47(1), 455–476 CrossRef CAS.
- W. Plazinski, J. Dziuba and W. Rudzinski, Modeling of sorption kinetics: the pseudo-second order equation and the sorbate intraparticle diffusivity, Adsorption, 2013, 19(5), 1055–1064 CrossRef CAS.
- E. D. Revellame, et al., Adsorption kinetic modeling using pseudo-first order and pseudo-second order rate laws: A review, Cleaner Eng. Technol., 2020, 1, 100032 CrossRef.
- S. S. M. Rodrigues, et al., Immobilization of Distinctly Capped CdTe Quantum Dots onto Porous Aminated Solid Supports, ChemPhysChem, 2015, 16(9), 1880–1888 CrossRef CAS PubMed.
- M. Vigdorowitsch, et al., Freundlich Isotherm: An Adsorption Model Complete Framework, Appl. Sci., 2021, 11, 8078 CrossRef CAS.
- S. Akash, et al., Metal oxide nanobiochar materials to remediate heavy metal and dye pollution: a review, Environ. Chem. Lett., 2024, 22(4), 2091–2112 CrossRef CAS.
- W. Rahmalia, et al., Stability, reusability, and equivalent circuit of TiO2/treated metakaolinite-based dye-sensitized solar cell: effect of illumination intensity on Voc and Isc values, Mater. Renew. Sustain. Energy, 2021, 10(2), 10 CrossRef.
- I. Shakir, et al., Nickel hydroxide nanoparticles and their hybrids with carbon nanotubes for electrochemical energy storage applications, Results Phys., 2020, 17, 103117 CrossRef.
- S. Dekkar, et al., Dry Reforming of Methane over Ni–Al2O3 and Ni–SiO2 Catalysts: Role of Preparation Methods, Catal. Lett., 2020, 150(8), 2180–2199 CrossRef CAS.
- T. A. Aragaw and F. T. Angerasa, Synthesis and characterization of Ethiopian kaolin for the removal of basic yellow (BY 28) dye from aqueous solution as a potential adsorbent, Heliyon, 2020, 6(9), e04975 CrossRef CAS PubMed.
- V. Biju and M. Abdul Khadar, Fourier transform infrared spectroscopy study of nanostructured nickel oxide, Spectrochim. Acta, Part A, 2003, 59(1), 121–134 CrossRef CAS PubMed.
- C. Kavitha, et al., RETRACTED: Elimination of Lead by Biosorption on Parthenium stem powder using Box-Behnken Design, S. Afr. J. Chem. Eng., 2022, 42, 270–279 Search PubMed.
- H. Tahir, S. Atika and S. Muhammad, Synthesis of Kaolin Loaded Ag and Ni Nanocomposites and Their Applicability for the Removal of Malachite Green Oxalate Dye, Iran. J. Chem. Chem. Eng., 2018, 37(3), 11–22 Search PubMed.
- A. Moumen, et al., Removal of Malachite Green Dye from Aqueous Solution by Catalytic Wet Oxidation Technique Using Ni/Kaolin as Catalyst, Molecules, 2022, 27, 7528 CrossRef CAS PubMed.
- W. Li, et al., Sulfate assisted synthesis of α-type nickel hydroxide nanowires with 3D reticulation for energy storage in hybrid supercapacitors, Mater. Chem. Front., 2022, 6(1), 94–102 RSC.
- D. S. Hall, et al., Applications of in situ Raman spectroscopy for identifying nickel hydroxide materials and surface layers during chemical aging, ACS Appl. Mater. Interfaces, 2014, 6(5), 3141–3149 CrossRef CAS PubMed.
- N. Mironova-Ulmane, et al., Polarisation dependent Raman study of single-crystal nickel oxide, Open Phys., 2011, 9(4), 1096–1099 CrossRef CAS.
- D. Egirani, et al., Synthesis and characterization of kaolinite coated with copper oxide and its effect on the removal of aqueous Lead(II) ions, Appl. Water Sci., 2019, 9(4), 109 CrossRef.
- V. O. Shikuku and T. Mishra, Adsorption isotherm modeling for methylene blue removal onto magnetic kaolinite clay: a comparison of two-parameter isotherms, Appl. Water Sci., 2021, 11(6), 103 CrossRef CAS.
- A. A. Adeyemo, I. O. Adeoye and O. S. Bello, Adsorption of dyes using different types of clay: a review, Appl. Water Sci., 2017, 7(2), 543–568 CrossRef CAS.
- L. Mao, et al., Ultrathin Ni(OH)2 nanosheets: a new strategy for cocatalyst design on CdS surfaces for photocatalytic hydrogen generation, RSC Adv., 2019, 9(3), 1260–1269 RSC.
- K. Aimdate, et al., Natural Kaolin-Based Ni Catalysts for CO2 Methanation: On the Effect of Ce Enhancement and Microwave-Assisted Hydrothermal Synthesis, ACS Omega, 2021, 6(21), 13779–13794 CrossRef CAS PubMed.
- A. Muthuvel, et al., Biosynthesis of gold nanoparticles using Solanum nigrum leaf extract and screening their free radical scavenging and antibacterial properties, Biomed. Prev. Nutr., 2014, 4(2), 325–332 CrossRef.
- R. Greenwood, et al., The zeta potential of kaolin suspensions measured by electrophoresis and electroacoustics, Chem. Pap., 2007, 61(2), 83–92 CAS.
- M. H. Cambre, et al., Cytotoxicity of NiO and Ni(OH)2 Nanoparticles Is Mediated by Oxidative Stress-Induced Cell Death and Suppression of Cell Proliferation, Int. J. Mol. Sci., 2020, 21(7), 2355 CrossRef CAS PubMed.
- K. J. Chaudhary, et al., Enhanced hydrogen production through methane dry reforming: Evaluating the effects of promoter-induced variations in reducibility, basicity, and crystallinity on Ni/ZSM-5 catalyst performance, Energy Convers. Manage.: X, 2024, 23, 100631 CAS.
- G. Salehi and M. Bagherzadeh, Visible Light-Driven Photocatalytic Degradation of Methylene Blue Dye Using a Highly Efficient Mg-Al LDH@g-C3N4@Ag3PO4, Nanocomposite, 2024, 9(4), 4581–4593 CAS.
- E. Torrik, M. Soleimani and M. T. Ravanchi, Application of Kinetic Models for Heavy Metal Adsorption in the Single and Multicomponent Adsorption System, Int. J. Environ. Res., 2019, 13(5), 813–828 CrossRef CAS.
- J. López-Luna, et al., Linear and nonlinear kinetic and isotherm adsorption models for arsenic removal by manganese ferrite nanoparticles, SN Appl. Sci., 2019, 1(8), 950 CrossRef.
- M. F. Mubarak, et al., Meta-kaolinite metal oxide quaternary composite for layered double hydroxide applied to a new frontier in adsorption technology: Synthesis, adsorption performance and kinetics study, Inorg. Chem. Commun., 2025, 178, 114647 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |






