Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Mechanistic insights into carbon steel corrosion inhibition by benzodiazepine derivatives in hydrochloric acid
N. Timoudan *a,
L. Chahira,
A. Marzaqa,
R. Saddikb,
S. Tighadouinib,
I. Waradc,
B. Dikicid,
Ahmed A. Farag
*a,
L. Chahira,
A. Marzaqa,
R. Saddikb,
S. Tighadouinib,
I. Waradc,
B. Dikicid,
Ahmed A. Farag e,
F. Bentissf and
A. Zarrouk
e,
F. Bentissf and
A. Zarrouk *a
*a
aLaboratory of Molecular Spectroscopy Modelling, Materials, Nanomaterials, Water and Environment, CERNE2D, Faculty of Sciences, Mohammed V University, Av. Ibn Battuta. P. O. Box 1014, Rabat, Morocco. E-mail: nadia.timoudan1995@gmail.com; a.zarrouk@um5r.ac.ma; azarrouk@gmail.com; Tel: +212673923069 Tel: +212665201397
bLaboratory of Organic Chemistry, Materials, Electrochemistry and Environment, Faculty of Sciences Ain Chock, Hassan II University, BP 5366, Casablanca, Morocco
cDepartment of Chemistry, AN-Najah National University, P. O. Box 7, Nablus, Palestine
dAtaturk University, Department of Mechanical Engineering, 25240, Erzurum, Turkey
eEgyptian Petroleum Research Institute (EPRI), Cairo, 11727, Egypt
fLaboratory of Catalysis and Corrosion of Materials, Faculty of Sciences, Chouaib Doukkali University, P. O. Box 20 M-24000, El Jadida, Morocco
First published on 21st April 2026
Abstract
The protection of carbon steel from aggressive acidic environments remains a critical challenge in industrial and defence-related applications. In this study, two newly synthesised benzodiazepine derivatives, namely 9-ethyl-2,3,4,9-tetrahydrobenzo[b]cyclopenta[e][1,4]diazepin-10(1H)-one (SR3) and 1-ethyl-4-phenyl-1H-benzo[b][1,4]diazepin-2(3H)-one (SR4), were investigated as corrosion inhibitors for carbon steel in 1 M HCl solution. Electrochemical measurements, including potentiodynamic polarisation and electrochemical impedance spectroscopy, were complemented by comprehensive surface and spectroscopic analyses (SEM, EDS, AFM, XRD, contact angle, and UV-Vis). Both compounds exhibited high inhibition efficiencies, reaching 85.1% for SR3 and 92.8% for SR4, with adsorption behaviour following the Langmuir isotherm model. Polarisation results indicated a mixed-type inhibition mechanism, while surface analyses confirmed the formation of a compact and adherent protective film on the steel surface. Density functional theory and molecular dynamics simulations provided molecular-level insights into the adsorption configurations and interaction strength of the inhibitors on the Fe(110) surface, in good agreement with the experimental observations. Overall, these findings demonstrate the effectiveness of benzodiazepine-based frameworks as promising molecular inhibitors for carbon steel corrosion in acidic media.
1. Introduction
Every year, corrosion of metals and alloys causes major economic losses, especially in industry. Owing to its mechanical characteristics, carbon steel is frequently employed as a base material for various types of construction. However, corrosion problems can unexpectedly affect the profitability of these projects, as the decision to use this material is based on the initial cost. Given its importance, researchers, engineers, theoreticians, and electrochemists are analyzing its resilience to corrosion in harsh conditions.On the other hand, many chemical processes and industrial procedures, including pickling, cleaning, localised-deposit removal, and acidification of oil wells, rely heavily on acid solutions, particularly hydrochloric acid. Today, corrosion prevention is both a scientific & technical research field and a financial requirement. The search for new, environmentally friendly inhibitors is justified by banning harmful modifiers such as chromates and nitrites in anti-corrosion treatments.
The presence of heteroatoms (N, O, S, and P), aromatic rings, functional/groups, a large number of π-electron-containing bonds, and electron density are all crucial factors that affect how efficiently these organic molecules inhibit.1–4 Amine derivatives are especially valued because they can create very effective chemicals as corrosion inhibitors and add new functional groups to their structures. Both physical and chemical adsorption depend on the molecules' electrostatic interaction with the metal surface and the d-orbital charge sharing between the heteroatoms and aromatic ring of organic compounds, which makes it easier for the molecules to adhere to the metal surface.
Recent studies on stainless steel have highlighted the importance of alloy composition and surface interactions in corrosion resistance.5,6 While these studies focus on carbon steel, the insights into microstructural stability and surface interactions are also relevant for understanding corrosion mechanisms in carbon steel. Such knowledge supports the evaluation of organic inhibitors, including benzodiazepine derivatives, by emphasizing the importance of adsorption and protective film formation in acidic media.
It seems that benzodiazepine molecules are the most effective at preventing corrosion. In fact, a benzodiazepine, which is sometimes shortened to “BZD” or simply “benzo,” is a psychotropic medication whose chemical structure is based on the combination of a diazepine and a benzene ring. Leo Sternbach discovered the first benzodiazepine, chlordiazepoxide (Librium), in 1955. Hoffmann-La Roche put it on the market in 1960 and introduced diazepam (Valium) in 1963.7 Furthermore, there is a dearth of information regarding the application of benzodiazepine derivatives in acidic settings.8–11
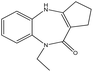
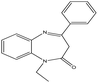
This study evaluates the ability of two synthetic benzodiazepine compounds, specifically 9-ethyl-2,3,4,9-tetrahydrobenzo[b]cyclopenta[e][1,4]diazepin-10(1H)-one (SR3) & 1-ethyl-4-phenyl-1H-benzo[b][1,4]diazepin-2(3H)-one (SR4), to prevent corrosion of C.steel in a solution of 1 M HCl. Electrochemical impedance spectroscopy, contact angle, SEM/EDS, AFM, XRD, isothermal computations, potentiodynamic polarization curves, and UV-visible examination were among the methods used at different phases to evaluate the efficacy of these substances. DFT computations and molecular-dynamics/simulations (MD) were used to ascertain the connection between the molecular structures of the studied derivatives and their capacity to inhibit corrosion. Along with determining the adsorption & thermodynamic-parameters, the adsorption mechanism was also suggested and discussed. The molecular structures of the SR3 & SR4 compounds examined are shown in Table 1.
2. Experimental
2.1. Materials
Corrosion tests were conducted using C.stl plates that had the composition below (in weight percentage): S-0.01600%, Cr-0.07700%, Ti-0.011000%, Ni-0.059000%, Co-0.009000%, Cu-0.16000%, C-0.37000%, Si-0.230000%, Mn-0.680000%, and iron as the predominant element. Acetone was used to clean the C.stl plates for electrochemical investigations after they were mechanically abraded using SiC sandpaper grades 180–1200. The plates were then allowed to dry at room temperature. The test solution was diluted an analytical-grade 37% HCl solution with double-distilled water to create a 1 M HCl environment.2.2. Electrochemical assessments
A typical three-electrode cell is formed by immersing three electrodes in the electrolyte in all electrochemical test experiments. This arrangement consists of a C.steel working electrode (WE) with a surface area of 1 cm2 exposed to the electrolyte, a saturated calomel electrode (SCE) as the reference electrode, and a platinum electrode (Pt) as the counter-electrode. The computer-connected PGZ100 potentiostat was used for the tests, and the ‘VoltaMaster 4.0’ electrochemical analysis program was used to gather electrochemical data. The C.stl's open circuit potential (OCP) was stabilized after 30 minutes of immersion in hydrochloric acid 1 M. The potentiodynamic-polarization analysis was set between −800 and −100 mV per SCE.Electrochemical-impedance tests were conducted in the 100 kHz to 10.0 mHz range with a signal amplitude of 10 mV at a 0.5 mV s−1 scan rate. Volta-master transforms the data from electrochemical studies into semicircular, which are fitted using ZView software.
2.3. Surface investigations
• Using JEOL JSM-IT 100 equipment, SEM was employed to characterize the C.steel surface before and after a 24 hours immersion in a 1 M HCl solution, with or without adding 1 × 10−3 M benzodiazepines. Since our team has already published a study with comparable conditions, we have used the blank's results in both the presence and absence of HCl without inhibitor.12• Atomic Force Microscopy (AFM) by a Hitachi 5100N was used to observe the deposited film shape at T = 303 K. Using an imaging methods system, the AFM experiments are conducted by immersing the electrode in 1 M HCl for twenty-four hours, in the presence and absence of 1 × 10−3 M of the two compounds being studied.
• Contact angle analysis was performed using the Biolin Scientific Attension Theta device. In contrast, an X-ray diffraction (XRD) assessment was conducted using a Shimadzu 6100 diffractometer (JEOL/Model JSM-IT) operating at a 20 kV voltage.
The electrochemical and surface examination findings for the non-inhibited solution used in this study are identical to those in our previous publication. This is significant because the same equipment and setup were used for the trials.13
2.4. UV-visible analysis
The effectiveness of corrosion inhibitors SR3 and SR4 for C.stl has been investigated using UV-visible spectroscopy, a technique based on light absorption characteristics at particular wavelengths. To analyze the interactions between these organic molecules & the surface/C.steel in a 1 M HCl medium, absorption tests of the solution were conducted both with and without a specimen of C.stl at a concentration of 1 × 10−3 M for SR3 and SR4 molecules. The lengths of the onde were measured using a UV-visible spectrophotometer (JASCO series V-730) that covered the 190–600 nm range.2.5. DFT computations
Gaussian 09 software was used to perform theoretical calculations in both gas phase and solution, utilizing density-functional/theory (DFT) with the hybrid function B3LYP,14–16 Water's effect as a solvent was considered using the polarizable continuum model (PCM), as electrochemical corrosion usually takes place in an aquatic environment. For optimization, the bases 6-31G(d,p) & 6-311+G(d,p) were employed for all species examined.This article presents quantum chemical studies within the framework of Kohn–Sham density-functional/theory. The equations of this theory have been employed to estimate various global quantum chemical descriptors, such as ionization potential (I), electronic affinity (A), energy gap (ΔE), electronegativity (χ), hardness (η), and softness (σ), among others.17–20
The following equations are used to calculate the overall energy change of back-donation (ΔEb–d) and the number (fraction) of electrons transferred (ΔN) from the inhibitor the inhibitor to the metal/surface.
 | (1) |
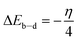 | (2) |
2.6. MD computational studies
The Forcite module, which was integrated into the Materials Studio 8.0 program created by BIOVIA Inc., carried out molecular dynamics simulation.21,22 A 40-high vacuum slab and periodic boundary conditions were used to model the interaction between the Fe (110) surface and the macromolecular matrix of benzodiazepine derivatives in a box measuring 32.27 × 32.27 × 30.13 Å3. The simulated species, such as 500H2O, 5Cl−, 5H3O+, and benzodiazepine-derivatives (SR3, SR4), interact with the Fe (110) surface via the COMPASS force environment. Using the canonical ensemble (NVT) with a step count of 1 fs and a simulation length of 1000 ps, the Andersen thermostat controls the temperature at 303 K during this simulation.233. Results & discussion
3.1. Polarization curves
Fig. 1 shows the polarization curve of C.steel that was submerged in a corrosion solution for half an hour at 303 K. Both the cathodic & anodic branches are reduced using SR3 and SR4. The benzodiazepine derivatives appear to function as mixed-type inhibitors, as evidenced by the maximum measured shift of 51.5 mV, which is below the threshold of 85 mV.24 All PDP curves showed parallel cathodic branches, suggesting that the cathodic reaction mechanism was unaffected by the existence of benzodiazepine derivatives.25 Furthermore, Fig. 1 shows that incorporating benzodiazepine compounds into 1 M HCl blocked both the anodic & cathodic branches. As the concentration rose, the influence of these derivatives on current densities became more noticeable. C.steel dissolving and hydrogen reduction at the corresponding reaction sites is significantly reduced due to the suppression of both anodic & cathodic/reactions on the C.steel/surface.26According to the extrapolated/electrochemical values presented in Table 2, the incorporation of the benzodiazepine derivatives notably decreases icorr when compared to the 1 M hydrochloric acid environment. At 10−3 M, the inhibition efficiency (η%) rises with the derivatives' concentration, peaking at 87.9% for SR3 and 94.2% for SR4. According to the extrapolated electrochemical values presented in Table 2, the incorporation of benzodiazepine derivatives significantly decreases icorr compared to the 1 M HCl environment. At 10−3 M, the inhibition efficiency (η%) increases with the derivatives' concentration, reaching 87.9% for SR3 and 94.2% for SR4. While the cathodic branches of the polarization curves appear roughly parallel, the observed changes in the cathodic Tafel slopes (βc, Table 2) indicate that the cathodic reaction or the rate-determining step may be affected by the presence of the inhibitors. The maximum measured shift corresponds to the offset of the corrosion potential (ΔEcorr), which remains below the 85 mV threshold, confirming that the inhibitors act as mixed-type, with a significant influence on the hydrogen evolution process.
| Environment | Conc. | −Ecor (SCE) (mV) | icorr (µA cm−2) | βa (mV dec−1) | −βc (mV dec−1) | ηPPD (%) |
|---|---|---|---|---|---|---|
| HCl | 1 M | 456.3 | 1104.1 | 112.8 | 155.4 | — |
| SR3 | 10−3 | 443.1 | 133.3 | 68.5 | 79.7 | 87.9 |
| 10−4 | 429.8 | 216.3 | 70.7 | 89.8 | 80.4 | |
| 10−5 | 405.2 | 428.1 | 75.5 | 165.1 | 61.2 | |
| 10−6 | 442.7 | 723.7 | 95.2 | 109.3 | 34.4 | |
| SR4 | 10−3 | 435.6 | 63.5 | 56.1 | 105.1 | 94.2 |
| 10−4 | 427.4 | 118.5 | 68.8 | 76.1 | 89.2 | |
| 10−5 | 429.1 | 136.1 | 84.9 | 82.5 | 87.6 | |
| 10−6 | 422.2 | 176.8 | 88.5 | 69.9 | 83.9 |
The addition of benzodiazepine derivatives also leads to a decrease in the anodic Tafel slopes (βa), suggesting an effect on the anodic dissolution reaction.27 This behavior implies that benzodiazepine molecules adsorb onto the C.stl/surface, creating an insulating layer & lowering the number of available active sites. Moreover, the heteroatoms of the benzodiazepine molecules (oxygen and nitrogen) may encourage the development of a shield that blocks the dissolution of C.stl. Consequently, when fewer active sites occur where the corrosion process occurs, the rate of anodic dissolution and hydrogen release is slowed.28,29 The detailed adsorption behavior and the formation of a protective film on the steel surface are discussed in the following section, based on Langmuir isotherm analysis and surface characterization techniques.
3.2. EIS investigation
Fig. 2 displays the Nyquist & Bode graphs of the EIS curves for C.stl in a hydrochloric acid environment. A charge transfer mechanism is shown by the spectrum's capacitive loop when no additives are present.30,31 Furthermore, as the concentration of SR3 and SR4 rises, the size of the semicircles in the inhibited electrolytes increases significantly. This enlargement indicates the formation of a protective film on the steel surface due to the adsorption of these derivatives. In addition, the phase diagrams in Fig. 2 show a single peak, suggesting that the SR3 and SR4 corrosion processes occur in a single step, controlled by charge transfer.32Modeling the EIS data using the equivalent circuit model allowed for a more thorough examination of the effects of SR3 and SR4 on C.stl, as seen in Fig. 3. Rp (polarization-resistance), a constant-phase-element (CPE) that represents the double-layer/capacitance (Cdl) on a heterogeneous/surface, & Rs (solution-resistance) are among the electrochemical parameters that were determined. When SR3 or SR4 is present or absent, eqn (3) (ref. 33) describes the impedance.
The matching electrochemical values are shown in Table 3. The findings show that the presence of benzodiazepine derivatives lowers double-layer/capacitance (Cdl) by increasing polarization-resistance (Rp). The widening of the semicircle in the Nyquist diagram, which indicates an elevation of Rp values in inhibited systems, verifies that SR3 and SR4 molecules prevent corrosion. The drop in Cdl parameters, which are usually associated with the thickness of the double-layer, could be due to the adsorption of benzodiazepine derivatives on the C.stl/acid interface. These derivatives have a lower dielectric constant than adsorbed water, which protects against metal corrosion.34,35 When compared to the hydrochloric acid environment, Table 3 shows a significant decrease in Rp with the addition of the benzodiazepine derivatives, while the inhibition efficiency (ηSIE%) improves as the derivative concentration increases. At 10−3 M, SR4 showed the highest corrosion inhibition (ηSIE% = 92.8%), whereas SR3 showed the highest corrosion inhibition (ηSIE% = 85.1%), indicating that benzodiazepine derivatives act as excellent corrosion inhibitors for steel in acidic environments.
| Environment | Conc. (M) | Rs (Ω cm2) | Rp (Ω cm2) | Q (µF sn−1 cm−2) | n | Cdl (µF cm−2) | χ2 | ηSIE (%) |
|---|---|---|---|---|---|---|---|---|
| HCl | 1 M | 0.83 | 21.57 | 293.9 | 0.845 | 116.2 | 0.002 | — |
| SR3 | 10−3 | 1.46 | 144.4 | 135.2 | 0.868 | 74.3 | 0.006 | 85.1 |
| 10−4 | 1.55 | 102.6 | 155.8 | 0.857 | 78.1 | 0.008 | 79.0 | |
| 10−5 | 1.70 | 54.9 | 182.1 | 0.851 | 81.3 | 0.007 | 60.8 | |
| 10−6 | 1.93 | 33.1 | 203.5 | 0.847 | 82.5 | 0.008 | 35.0 | |
| SR4 | 10−3 | 2.17 | 300.7 | 67.01 | 0.875 | 38.4 | 0.008 | 92.8 |
| 10−4 | 3.47 | 184.2 | 80.4 | 0.866 | 41.9 | 0.007 | 88.3 | |
| 10−5 | 1.46 | 166.0 | 101.5 | 0.858 | 51.6 | 0.008 | 87.0 | |
| 10−6 | 1.10 | 146.3 | 123.8 | 0.856 | 63.0 | 0.007 | 85.3 |
An excellent method for evaluating a solid surface's inhibitory behavior is to use adsorption isotherms. Based on formula (3), the Langmuir-isotherm with a linear regression coefficient (R2) and a slope of around one was found to be the best model for predicting the EIS results.
 | (3) |
ΔGads = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(55.5 × K) ln(55.5 × K)
| (4) |
Fig. 4 illustrates the Langmuir isotherm, showing the adsorption of benzodiazepine derivatives onto the surface of carbon steel in a 1 M HCl solution.
The range of ΔGads is −40.7 kJ mol−1 to −43.1 kJ mol−1, as seen in Table 4. Therefore, when both benzodiazepine equivalents are adsorbed onto the C.stl/surface, chemical adsorption is predominant. Furthermore, the value for SR4 is substantially greater and more damaging than SR3's, indicating that SR4 provides superior adsorption, in agreement with the higher inhibition efficiency observed in the PDP results. This demonstrates that the mixed-type inhibition behavior observed in the polarization measurements arises from the adsorption of the inhibitor molecules and the formation of a stable protective layer on the steel surface.
| R2 | Slope | K (L mol−1) | ΔGads (kJ mol−1) | |
|---|---|---|---|---|
| SR3 | 0.9999 | 1.170 | 1.93 × 105 | −40.7 |
| SR4 | 0.9999 | 1.075 | 4.879 × 105 | −43.1 |
3.3. Effect of temperature
Temperature can cause several changes at the electrolyte/steel interface, including increased surface attack, inhibitor molecule desorption, and even inhibitor breakdown.36 In light of this, the development of inhibitor corrosion rates was examined during a 30 minutes immersion in a corrosive solution of hydrochloric-acid at various T (303 to 333 K), both with & without the SR3 and SR4 inhibitors (at a concentration of 10−3 M).The icorr current density rises with increasing temperature, as demonstrated by the potentiodynamic polarization curves in Fig. 5. Additionally, Table 5 shows that the percentage inhibition efficiency (IE) for compounds SR3 and SR4 drops somewhat with temperature, from 87.9% to 75.6% and 94.2% to 84.5%, respectively. This highlights the reduced adsorption of SR3 & SR4 compounds, especially at elevated temperatures.
 | ||
| Fig. 5 PDP plots in the non-existence and existence of 10−3 M SR3 & SR4 molecules at various temperatures. | ||
| Medium | Temp (K) | −Ecorr (SCE) (mV) | icorr (µA cm−2) | βa (mV dec−1) | −βc (mV dec−1) | ηpp (%) |
|---|---|---|---|---|---|---|
| 1 M HCl | 303 | 456.3 | 1104.1 | 112.8 | 155.4 | — |
| 313 | 423.5 | 1477.4 | 91.3 | 131.3 | — | |
| 323 | 436.3 | 2254.0 | 91.4 | 117.8 | — | |
| 333 | 433.3 | 3944.9 | 103.9 | 134.6 | — | |
| SR3 | 303 | 443.1 | 133.3 | 68.5 | 79.7 | 87.9 |
| 313 | 448.2 | 255.1 | 66.7 | 64.6 | 82.7 | |
| 323 | 441.3 | 465.1 | 91.3 | 165.7 | 79.3 | |
| 333 | 459.8 | 962.1 | 89.6 | 109.7 | 75.6 | |
| SR4 | 303 | 435.6 | 63.5 | 84.9 | 105.1 | 94.2 |
| 313 | 447.3 | 154.1 | 60.5 | 65.6 | 89.5 | |
| 323 | 442.1 | 287.2 | 87.0 | 103.6 | 87.2 | |
| 333 | 458.05 | 610.6 | 79.6 | 104.1 | 84.5 |
Although a decrease in inhibition efficiency was observed with increasing temperature, SR4 retained a relatively high performance at 10−3 M. However, it should be noted that temperature-dependent measurements were carried out only at this concentration. Therefore, broader conclusions regarding adsorption behavior at lower concentrations require further investigation.
An analysis of the literature demonstrates how important temperature is to metal corrosion. Because hydrogen's in acidic environments, the reduction potential typically diminishes, hydrogen depolarization is preferred, and corrosion rates are accelerated. Activation energy (Ea), activation entropy (ΔSa), and activation enthalpy (ΔHa) are among the qualities that can be better understood through studies conducted at different temperatures. The Arrhenius equation37 often demonstrates a strong correlation between temperature and corrosion rate:
 | (5) |
 | (6) |
In this case, T represents the absolute temperature, R the gas constant, and A the Arrhenius pre-exponential factor. Fig. 6 shows that when ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) icorr is plotted against 1/T, straight lines are produced.
icorr is plotted against 1/T, straight lines are produced.
Fig. 6 presents an Arrhenius plot that exhibits a linear relationship with a slope of −Ea/R. The calculated activation energy (Ea) for systems incorporating SR3 & SR4 ({C.stl/SR3 or SR4/1 M HCl}) is higher compared to the reference medium. An increase in Ea parameters suggests a reduction in the rate of the corrosion reaction.38 In other words, SR3 and SR4 slow down the breakdown of C.stl in a hydrochloric-acid/environment by decreasing the corrosive-reactivity agents or preventing them from reaching the metal/surface. The positive ΔHa values in Table 6 suggest that the dissolution process in C.stl is endothermic. The increase in ΔSa when switching from the electrolyte solution to the electrolyte solution in the presence of both inhibitors implies that an increase in disorder occurs when the reactants switch to the activated complex.39
| Medium | R2 | Ea (kJ mol−1) | ΔHa (kJ mol−1) | ΔSa (J mol−1 K−1) |
|---|---|---|---|---|
| Reference | 0.9710 | 35.40 | 32.70 | −79.21 |
| SR3 | 0.994 | 54.60 | 52.0 | −32.80 |
| SR4 | 0.995 | 62.10 | 59.50 | −13.71 |
3.4. SEM/EDS analysis
The surface topography of samples can be effectively analyzed using the scanning electron microscope (SEM). HCl containing benzodiazepine derivatives at 10−3 M or pure HCl was applied to C.stl samples for 24 hours. Uninhibited and inhibited samples' SEM images are displayed in Fig. 7a–c, respectively. Fig. 7a shows C.stl not exposed to HCl, whose surface appears smooth and homogeneous, with only minor imperfections and no obvious signs of corrosion. This is characteristic of a metal not subjected to a corrosive environment. In contrast, Fig. 7b shows the steel after exposure to 1 M HCl without an inhibitor, where the surface is heavily damaged, with deep cavities, pitting, and a rough structure. These alterations are the result of severe corrosion due to the dissolution of iron under the effect of the acid, accompanied by the formation of corrosion products (iron oxides and chlorides). The surface shows a significant loss of material, confirming a rapid and aggressive attack in the absence of an inhibitor. Fig. 7c, showing the steel in the presence of 1 M HCl + 10−3 M SR3, shows a less damaged surface than in Fig. 7b, although irregularities and porosities are still visible. The SR3 inhibitor appears to have limited acid attack, but signs of corrosion remain, suggesting partial protection. A protective film formed by the adsorption of SR3 could be present in the form of deposits on the metal surface. Finally, Fig. 7d shows the steel in the presence of 1 M HCl + 10−3 M SR4, where the surface is much more protected compared to the previous conditions. Very few cavities or pits are visible, indicating that SR4 has formed a more effective protective layer, reducing acid attack. The homogeneous structure observed suggests stronger adsorption of SR4, which significantly limits metal degradation. These observations confirm that SR4 offers better protection than SR3 against corrosion of carbon steel in an acidic environment. | ||
| Fig. 7 SEM/EDS images of C.stl alone (a), with hydrochloric acid (b), and with 10−3 M SR3 (c) & SR4 (d) after 24 hours of submersion. | ||
In the case of carbon steel alone, not exposed to HCl (Fig. 7a), the dominant signal corresponds to iron (Fe), the main constituent of steel. Faint traces of oxygen (O) are detected, probably due to a thin layer of natural oxidation on the surface. The absence of chlorine (Cl) and other elements indicates that the steel has not undergone any corrosion or adsorption of inhibitors. When exposed to 1 M HCl without an inhibitor (Fig. 7b), the EDS reveals a significant decrease in the Fe signal, reflecting partial dissolution of the metal by the acid. Simultaneously, an increase in the Cl signal is observed, confirming the formation of corrosion products, notably FeCl3, by reaction between the iron and the acid. In addition, the increased presence of oxygen (O) indicates significant oxidation, forming iron oxides and hydroxides. These observations confirm severe and unprotected corrosion of the steel in this acidic environment. With the addition of 10−3 M SR3 in 1 M HCl medium (Fig. 7c), we observed an increase in the Fe signal compared with the condition without inhibitor, indicating a reduction in metal dissolution due to the protective effect of SR3. In addition, a decrease in the Cl signal suggests that SR3 limited the formation of corrosion products. The appearance of new elements, such as C and N, confirms the adsorption of SR3 to the metal surface. However, the residual presence of Cl and O indicates that inhibition is not complete, and that traces of corrosion remain. Finally, in the presence of 10−3 M SR4 (Fig. 7d), inhibition is even more marked. The Fe signal is more intense than in the case of SR3, indicating better protection against dissolution. The Cl signal is strongly reduced, showing that SR4 limits FeCl3 formation more than SR3. In addition, the marked appearance of C and N suggests more efficient adsorption of SR4, reinforcing the protective layer formed on the steel surface. The decrease in the O signal also indicates better control of oxidation, reducing the formation of iron oxides.
3.5. AFM
The AFM images (Fig. 8) reveal that the surface/C.stl becomes highly rough Following contact with hydrochloric acid. In contrast, the existence of benzodiazepine derivatives suggests a protective effect through forming an adsorbed layer. Samples containing these derivatives exhibited reduced surface roughness, with SR3 showing an average roughness of 70.7 nm and SR4 at 21.51 nm, compared to the untreated steel, which had a roughness (Ra) of 120.7 nm. This roughness reduction highlights the derivatives' effectiveness, with SR4 demonstrating superior performance due to better adsorption. These AFM results are consistent with findings from other surface analysis techniques.3.6. Contact angle (CA)
The results of CA measurement suggests that the angle is higher when SR3 & SR4 are present than when they are absent (Fig. 9). CA analysis shows a decrease in the metal/surface's contact with water, indicating that these compounds make the surface more hydrophobic by creating a protective organic layer. With SR4 outperforming SR3, contact angles rise from 78.17° for the untreated surface to 99.78° and 102.08° for SR3 & SR4, respectively. The efficacy of the protective barrier produced by benzodiazepine derivatives is confirmed by a contact angle larger than 90°, which denotes a hydrophobic surface. | ||
| Fig. 9 Contact angle measurements of C.steel submerged in hydrochloric acid for 6 hours, both in the non-existence and existence of 1 × 10−3 M of SR3 & SR4. | ||
3.7. XRD examination
Diffractograms of C.stl (Fig. 10) show significant differences depending on the existence and non-existence of SR3 and SR4 after six hours of contact with hydrochloric acid. In the non-existence of these inhibitors, iron & iron-oxide peaks are observed, whereas in their presence, oxide creation is delayed, and the intensity of the iron peak decreases. This illustrates the ability of SR3 and SR4 to prevent corrosion by encouraging the creation of a protective layer. The XRD method is crucial for analyzing the composition of corrosion products in steel before and after inhibitor treatment.3.8. UV-visible
As seen in Fig. 11, UV-visible/absorption spectra were taken for a medium including 1 × 10−3 M of SR3 & SR4, both before and after C.stl was soaked for 72 hours at 303 K to confirm the likelihood of complex creation between SR3, SR4, and Fe. According to earlier research, a wavelength shifts and/or a change in absorbance are signs of complex formation between C.stl and inhibitor molecules. The n–π* electronic transition caused by the ring is represented by a single band at around 231.1 nm for SR4 and two bands at about 225.7 nm and 312.5 nm for SR3 in the UV-visible spectra of SR3 and SR4 dissolved in an acidic medium before C.stl immersion. Following a 72 hours soak, these bands see a redshift and an increase in absorbance. These modifications imply that interactions between Fe2+ and benzodiazepine molecules have occurred in the acidic media. | ||
| Fig. 11 UV-visible spectra of SR3 and SR4 in the acid medium before (black) and after (red) 72 hours of C.stl quenching. | ||
3.9. Comparison of SR3 and SR4
According to the data summarized in Table 7, it is possible to confirm that SR4 has a better corrosion inhibition performance compared to SR3. SR4 has a lower corrosion current density, high inhibition efficiency, and polarization resistance, suggesting the formation of a more effective protective coating on the surface of carbon steel. Lower roughness and increased contact angle of SR4 are also observed through surface analysis, which indicates better surface protection. Moreover, the theory of more negative ΔGads of SR4 (−43.1 kJ mol−1) than that of SR3 (−40.7 kJ mol−1) indicates that it is adsorbed more on the steel surface. This could be because of the existence of the phenyl group in SR4, which increases the interaction of the molecule with the Fe surface.| Parameter/method | SR3 (10−3 M) | SR4 (10−3 M) |
|---|---|---|
| Nomenclature | 9-Ethyl-2,3,4,9-tetrahydrobenzo[b]cyclopenta[e][1,4]diazepin-10(1H)-one | 1-Ethyl-4-phenyl-1H-benzo[b][1,4]diazepin-2(3H)-one |
| icorr (µA cm−2) | 133.3 | 63.5 |
| ηPPD (%) | 87.9 | 94.2 |
| RP (Ω cm2) (EIS) | 144.4 | 300.7 |
| ηEIS (%) | 85.1 | 92.8 |
| Surface roughness (nm) | 70.7 | 21.5 |
| Contact angle | 99.8 | 102.1 |
| ΔGads | −40.7 | −43.1 |
3.10. Theoretical analysis
The reactivity of SR3 and SR4 molecules can be measured by measuring the energies of the border molecular orbitals (HOMO & LUMO). Table 8 displays the overall reactivity indices for SR3 & SR4's neutral & protonated forms. It should be borne in mind that a molecule's propensity to give up electrons during interactions with metal orbitals increases with its EHOMO value.43–45 An essential feature affecting the reactivity of a compound is its energy-gap (ΔE = ELUMO – EHOMO). HOMO & LUMO interactions become stronger as this gap is reduced, increasing the reactivity of the compound, & conversely. According to our results, the neutral, protonated form of SR4 shows a reduced energy gap, indicating that it is more reactive than SR3. This could also mean that SR4 has a higher inhibitory efficiency. These observations offer convincing confirmation and are align with our experimental findings.
| Descriptor | EHOMO (eV) | ELUMO (eV) | ΔE (eV) | χ (eV) | η (eV) | σ (eV−1) | ΔN110 |
|---|---|---|---|---|---|---|---|
| SR3 neutral | −5.645 | −1.145 | 4.500 | 3.395 | 2.250 | 0.444 | 0.316 |
| SR3 protonated | −7.117 | −1.953 | 5.164 | 4.535 | 2.582 | 0.387 | 0.055 |
| SR4 neutral | −6.310 | −2.016 | 4.293 | 4.163 | 2.147 | 0.465 | 0.153 |
| SR4 protonated | −7.109 | −3.343 | 3.765 | 5.226 | 1.883 | 0.531 | −0.107 |
The values of ΔN110, 0.316 for SR3, 0.153 for SR4, 0.055 for SR3-H, and −0.1078 for SR4-H respectively, confirm this observation. Positive ΔN110 values suggest that neutral forms tend to give up electrons, while negative values indicate that charged compounds do not share electrons. ELUMO values are also used to assess the attraction effect: a lesser value of this descriptor demonstrates a larger propensity of species to accept electrons. According to our findings, electrons are transferring from the HOMO of benzodiazepines to the metal/surface, as indicated by ΔN110 values below 3.6, which is consistent with the inferences made from the energy gap.46
Furthermore, other properties of the molecule being studied, such as absolute-electronegativity (χ) & hardness (η), influence the reactivity of this species. The interchange of electrons between the inhibitor compound & the metal/surface is also impacted by variations in electronegativity. Electrons tend to move from the protective compound, which is less electronegative, to the metal support, which is more electronegative, to balance chemical potentials.47 According to our findings, the protonated molecules SR3-H and SR4-H have larger χ values than the neutral forms, indicating that the charged/forms interact more with the treated surface (see Table 8). Additionally, the η parameters, which are also displayed in Table 8, indicate that the protonated/compounds are more reactive because they have lower values than their neutral counterparts.
In conclusion, quantum chemical calculations are proving very helpful. They significantly support the experimental findings and demonstrate that SR4 has a greater inhibitory capacity than SR3.
 | ||
| Fig. 13 Mulliken atomic charges representations for the SR3 and SR4 molecules obtained by the DFT analysis in their neutral & protonated forms. | ||
| Einteraction = Etotal − (Esurface+solution) + Esolution | (7) |
| Ebinding = −Einteration | (8) |
3.11. Adsorption mechanism
The results of the study indicate that organic compounds act as corrosion inhibitors by attaching themselves to the metal surface. The adsorption of molecules onto the metal can occur through various mechanisms. Retrodonation involves the interaction of the molecule's π electrons with the metal surface, while chemisorption results from the interaction between the non-bonding electron doublets of the molecules and the metal. Physisorption, on the other hand, is governed by electrostatic interactions between charged species present in the medium.In an acidic environment, nitrogen-containing compounds can exist in neutral or protonated form. Furthermore, oxidation of the metal surface leads to a loss of electrons, giving it a positive charge. This situation then promotes the adsorption of negative ions, such as chloride ions, onto the metal surface.65
The adsorption mechanism of SR3 and SR4 on the carbon steel surface in 1 M HCl is illustrated in Fig. 16. In an acidic medium, the inhibitor molecules become protonated, forming SR3H+ and SR4H+ species. Chloride ions (Cl−) in the solution are first adsorbed onto the positively charged steel surface, thereby creating negatively charged sites. The adsorption of SR3H+ and SR4H+ occurs through a mixed mechanism involving both physisorption and chemisorption. Physisorption results from electrostatic interactions between the protonated inhibitor molecules and the negatively charged surface. Chemisorption occurs through donor–acceptor interactions between the lone pair electrons of nitrogen and oxygen atoms, as well as the π-electrons of aromatic rings, and the vacant d-orbitals of Fe atoms. In addition, retrodonation from the metal surface to the inhibitor molecules may further strengthen the adsorption and stabilize the protective film formed on the steel surface.
 | ||
| Fig. 16 Schematic illustration of the adsorption mechanism of SR3 and SR4 on the C.stl surface in 1 M HCl. | ||
4. Conclusion
The effectiveness and inhibitory mechanisms of benzodiazepine derivatives in halting the corrosion of C.stl in a hydrochloric Acid environment are assessed in this study using a variety of methodologies. The compounds' exceptional efficacy as inhibitors was demonstrated by their highest performance of 87.9% for SR3 and 94.2% for SR4 at the ideal concentration. The Langmuir isotherm governs the chemical adsorption of SR3 & SR4/C.stl, where they function as a mixed inhibitor. While UV-visible analysis of the environment demonstrated the production of complexes, surface investigation using SEM-EDS, AFM, contact angle, and XDR, verified the development of a protective layer on the steel/surface. Molecular-dynamics (MD) simulations display that SR3 & SR4 adsorb in parallel to the Fe(110) iron surface, boosting corrosion-protection, whereas the findings of DFT predictions correlate significantly with experimental performance.However, several limitations should be considered. The inhibition efficiency of SR4 decreases with increasing temperature, suggesting limited thermal stability under elevated temperatures. SR3 provided incomplete protection, as residual corrosion was observed in SEM analyses. Additionally, all experiments were conducted over short-term immersion periods (up to 72 hours), and long-term stability under industrially relevant conditions has not been assessed. These limitations highlight the need for further studies to fully validate the practical applicability of these inhibitors.
Conflicts of interest
There are no conflicts to declare.Data availability
All supporting data are contained within the manuscript.References
- F. Tezcan, G. Yerlikaya, A. Mahmood and G. Kardaş, J. Mol. Liq., 2018, 269, 398–406 CrossRef CAS.
- H. Zarrok, A. Zarrouk, R. Salghi, Y. Ramli, B. Hammouti, M. Assouag, E. M. Essassi, H. Oudda and M. Taleb, J. Chem. Pharmaceut. Res., 2012, 4, 5048–5055 CAS.
- H. Zarrok, S. S. Al-Deyab, A. Zarrouk, R. Salghi, B. Hammouti, H. Oudda, M. Bouachrine and F. Bentiss, Int. J. Electrochem. Sci., 2012, 7, 4047–4063 CrossRef CAS.
- M. El Faydy, B. Lakhrissi, A. Guenbour, S. Kaya, F. Bentiss, I. Warad and A. Zarrouk, J. Mol. Liq., 2019, 280, 341–359 CrossRef CAS.
- Y. Zhao, H. L. Liu, L. L. Wei and L. Q. Chen, Tungsten, 2023, 5(4), 467–480 CrossRef.
- Y. Han, Z. H. Liu, C. B. Wu, Y. Zhao, G. Q. Zu, W. W. Zhu and X. Ran, Tungsten, 2023, 5(4), 419–439 CrossRef.
- E. Shorter, A Historical Dictionary of Psychiatry, Oxford University Press, Oxford, 2005 Search PubMed.
- N. Benzbiria, A. Thoume, S. Echihi, M. E. Belghiti, A. Elmakssoudi, A. Zarrouk, M. Azzi and M. Zertoubi, J. Mol. Struct., 2023, 1281, 135139 CrossRef CAS.
- T. Laabaissi, M. Rbaa, F. Benhiba, Z. Rouifi, U. P. Kumar, F. Bentiss, H. Oudda, B. Lakhrissi, I. Warad and A. Zarrouk, Colloids Surf., A, 2021, 629, 127428 CrossRef CAS.
- W. Niouri, B. Zerga, M. Sfaira, M. Taleb, M. E. Touhami, B. Hammouti, M. Mcharfi, S. S. Al-Deyab, H. Benzeid and E. M. Essassi, Int. J. Electrochem. Sci., 2014, 9, 8283–8298 CrossRef.
- Y. El Aoufir, J. Sebhaoui, H. Lgaz, Y. El Bakri, A. Zarrouk, F. Bentiss and H. Oudda, J. Mater. Environ. Sci., 2017, 8, 2161–2173 CAS.
- N. Timoudan, A. Titi, M. El Faydy, F. Benhiba, R. Touzani, I. Warad, A. Bellaouchou, A. Alsulmi, B. Dikici, F. Bentiss and A. Zarrouk, Colloids Surf., A, 2024, 682, 132771 CrossRef CAS.
- A. Barrahi, M. E. M. Mekhzoum, A. Thakur, A. E. K. Qaiss, G. Kaichouh, M. El Faydy, F. Benhiba, B. Dikici, R. Bouhfid, H. A. Abuelizz, I. Warad and A. Zarrouk, Int. J. Electrochem. Sci., 2025, 20, 100917 CrossRef CAS.
- A. D. Becke, J. Chem. Phys., 1993, 98, 5648–5652 CrossRef CAS.
- C. Lee, W. Yang and R. G. Parr, Phys. Rev. B, 1988, 37, 785–789 CrossRef CAS PubMed.
- R. Hsissou, O. Dagdag, S. Abbout, F. Benhiba, M. Berradi, M. El Bouchti, A. Berisha, N. Hajjaji and A. El Harfi, J. Mol. Liq., 2019, 284, 182–192 CrossRef CAS.
- O. Dagdag, Z. Safi, R. Hsissou, H. Erramli, M. El Bouchti, N. Wazzan, L. Guo, C. Verma, E. E. Ebenso and A. El Harfi, Sci. Rep., 2019, 9, 11715 CrossRef PubMed.
- L. Guo, Z. S. Safi, S. Kaya, W. Shi, B. Tüzün, N. Altunay and C. Kaya, Front. Chem., 2018, 6, 155 CrossRef PubMed.
- R. Hsissou, S. Abbout, Z. Safi, F. Benhiba, N. Wazzan, L. Guo, K. Nouneh, S. Briche, H. Erramli and M. E. Touhami, Constr. Build. Mater., 2021, 270, 121454 CrossRef CAS.
- Ş. Erdoğan, Z. S. Safi, S. Kaya, D. Ö. Işın, L. Guo and C. Kaya, J. Mol. Struct., 2017, 1134, 751–761 CrossRef.
- F. Benhiba, H. Serrar, R. Hsissou, A. Guenbour, A. Bellaouchou, M. Tabyaoui, S. Boukhris, H. Oudda, I. Warad and A. Zarrouk, Chem. Phys. Lett., 2020, 743, 137181 CrossRef.
- D. S. BIOVIA, BIOVIA Materials Studio, Dassault Systèmes, San Diego, 2016 Search PubMed.
- H. C. Andersen, J. Chem. Phys., 1980, 72, 2384–2393 CrossRef CAS.
- R. Nabah, F. Benhiba, Y. Ramli, M. Ouakki, M. Cherkaoui, H. Oudda, R. Touir, I. Warad and A. Zarrouk, Anal. Bioanal. Electrochem., 2018, 10, 1375–1398 CAS.
- V. Saraswat and M. Yadav, J. Mol. Liq., 2020, 297, 111883 CrossRef CAS.
- L. O. Olasunkanmi and E. E. Ebenso, J. Colloid Interface Sci., 2020, 561, 104–116 CrossRef CAS PubMed.
- M. El Belghiti, Y. Karzazi, A. Dafali, B. Hammouti, F. Bentiss, I. B. Obot, I. Bahadur and E. E. Ebenso, J. Mol. Liq., 2016, 218, 281–293 CrossRef CAS.
- S. Echihi, N. Benzbiria, M. Beraich, M. Elfal, M. Elbelghiti and M. Boudalia, Chem. Data Collect., 2023, 46, 101049 CrossRef CAS.
- A. Thoume, F. Benhiba, A. Elmakssoudi, D. B. Left, N. Benzbiria, I. Warad, M. Dakir, M. Azzi, M. Zertoubi and A. Zarrouk, J. Appl. Electrochem., 2021, 51, 1755–1770 CrossRef CAS.
- W. Zhang, H.-J. Li, M. Wang, L.-J. Wang, Q. Pan, X. Ji, Y. Qin and Y.-C. Wu, J. Mol. Liq., 2019, 293, 111478 CrossRef CAS.
- A. El-Yaktini, A. Lachiri, M. El Faydy, F. Benhiba, H. Zarrok, M. El-Azzouzi, M. Zertoubi, M. Azzi, B. Lakhrissi and A. Zarrouk, Orient. J. Chem., 2018, 34, 3016–3024 CAS.
- S. Yesudass, L. O. Olasunkanmi, I. Bahadur, M. M. Kabanda, I. B. Obot and E. E. Ebenso, J. Taiwan Inst. Chem. Eng., 2016, 64, 252–268 CrossRef CAS.
- H. Zarrok, S. Daoui, N. Benzbiria, A. Barrahi, F. Benhiba, M. Galai, M. E. Touhami, I. Warad, K. Karrouchi and N. Benchat, Chem. Data Collect., 2024, 51, 101140 CrossRef CAS.
- L. Chahir, F. Benhiba, N. Abad, H. Zarrok, I. Warad, M. Al-Noaimi, D. B. Left, M. Zertoubi, M. Allali and A. Bellaouchou, J. Electrochem. Sci. Eng., 2024, 14, 275–296 CAS.
- L. Chahir, A. Marzaq, N. Timoudan, M. El Faydy, F. Benhiba, D. B. Left, M. Zertoubi, R. Saddik, S. Tighadouini, I. Warad, B. Dikici, M. Allali and A. Zarrouk, Colloids Surf., A, 2025, 712, 136431 CrossRef CAS.
- B. Xu, W. Yang, Y. Liu, X. Yin, W. Gong and Y. Chen, Corros. Sci., 2014, 78, 260–268 CrossRef CAS.
- M. El Faydy, F. Benhiba, N. Timoudan, B. Lakhrissi, I. Warad, S. Saoiabi, A. Guenbour, F. Bentiss and A. Zarrouk, J. Mol. Liq., 2022, 354, 118900 CrossRef CAS.
- S. M. Shaban, I. Aiad, M. M. El-Sukkary, E. A. Soliman and M. Y. El-Awady, J. Mol. Liq., 2015, 203, 20–28 CrossRef CAS.
- A. Zarrouk, B. Hammouti, H. Zarrok, R. Salghi, A. Dafali, Lh. Bazzi, L. Bammou and S. S. Al-Deyab, Der Pharma Chem, 2012, 4, 337–346 Search PubMed.
- Z. Rouifi, M. Rbaa, A. S. Abousalem, F. Benhiba, T. Laabaissi, H. Oudda, B. Lakhrissi, A. Guenbour, I. Warad and A. Zarrouk, Surf. Interfaces, 2020, 18, 100442 CrossRef CAS.
- I. B. Obot and N. O. Obi-Egbedi, Corros. Sci., 2010, 52, 657–660 CrossRef CAS.
- V. S. Sastri and J. R. Perumareddi, Corrosion, 1997, 53, 617–622 CrossRef CAS.
- B. El Mehdi, B. Mernari, M. Traisnel, F. Bentiss and M. Lagrenée, Mater. Chem. Phys., 2003, 77, 489–496 CrossRef CAS.
- K. Fukui, Science, 1982, 218, 747–754 CrossRef CAS PubMed.
- L. Kadiri, A. Ouass, R. Hsissou, Z. Safi, N. Wazzan, Y. Essaadaoui, I. Lebkiri, O. El Khattabi and A. Lebkiri, J. Mol. Liq., 2021, 343, 116971 CrossRef CAS.
- I. Lukovits, K. Pálfi, I. Bako and E. Kálmán, Corrosion, 1997, 53, 915–919 CrossRef CAS.
- S. Alaoui Mrani, E. Ech-Chihbi, N. Arrousse, Z. Rais, F. El Hajjaji, C. El Abiad, S. Radi, J. Mabrouki, M. Taleb and S. Jodeh, Arabian J. Sci. Eng., 2021, 46, 5691–5707 CrossRef CAS.
- A. Djermoune, R. Maizia, M. Zahzouh and Y. Khelfaoui, Colloids Surf., A, 2025, 709, 136068 CrossRef CAS.
- A. Hmada, R. Sayed, F. Hamouche, N. Errahmany, H. Anahmadi, M. Galai, S. Boukhris, M. Harcharras and N. Dkhireche, Inorg. Chem. Commun., 2025, 172, 113640 CrossRef CAS.
- A. Toghan, A. Fawzy, A. I. Alakhras, M. M. S. Sanad, M. Khairy and A. A. Farag, Metals, 2023, 13, 1–15 Search PubMed.
- A. Barrahi, M. E. M. Mekhzoum, A. Thakur, A. E. K. Qaiss, G. Kaichouh, M. El Faydy, F. Benhiba, B. Dikici, R. Bouhfid, H. A. Abuelizz, I. Warad and A. Zarrouk, Int. J. Electrochem. Sci., 2025, 20, 100917 CrossRef CAS.
- A. G. Al-Gamal, A. A. Farag, E. M. Elnaggar and K. I. Kabel, Compos. Interfaces, 2018, 25, 959–980 CrossRef CAS.
- A. A. Farag, S. M. Al-Shomar and N. S. Abdelshafi, Int. J. Biol. Macromol., 2024, 245, 135408 CrossRef PubMed.
- J. J. Martinez-Gonzalez, I. Tello-Salgado, A. K. Larios-Galvez, R. Lopez-Sesenes, Z. Zarhri, A. M. Ramirez-Arteaga and J. G. Gonzalez-Rodriguez, J. Mol. Struct., 2025, 1321, 140014 CrossRef CAS.
- A. Toghan, H. Alhussain, A. Fawzy, M. M. S. Sanad, S. A. Al-Hussain, E. M. Masoud, H. Jiang and A. A. Farag, J. Mol. Struct., 2024, 1318, 139315 CrossRef CAS.
- M. A. El-Monem, A. A. Farag, M. M. H. Khalil and M. A. Migahed, J. Mol. Struct., 2024, 1314, 138702 CrossRef CAS.
- N. S. Abdelshafi, A. A. Farag, F. E.-T. Heakal, A.-S. Badran, K. M. Abdel-Azim, A.-R. Manar El Sayed and M. A. Ibrahim, J. Mol. Struct., 2024, 1304, 137638 CrossRef CAS.
- B. H. A. Mohamed, A. A. Farag and B. M. Elshafey, Eurasian J. Chem., 2008, 10, 67–77 Search PubMed.
- A. A. Farag, S. M. Tawfik, A. A. Abd-Elaal and N. S. Abdelshafi, J. Ind. Eng. Chem., 2024, 138, 237–255 CrossRef CAS.
- A. A. Farag, A. Toghan, M. S. Mostafa, C. Lan and G. Ge, Catalysts, 2022, 12, 1–18 CrossRef.
- A. Toghan, A. Fawzy, A. I. Alakhras and A. A. Farag, Int. J. Electrochem. Sci., 2022, 17, 2212108 CrossRef CAS.
- A. Toghan, M. Khairy, M. Huang and A. A. Farag, Int. J. Electrochem. Sci., 2023, 18, 100072 CrossRef CAS.
- A. Fawzy, A. Toghan, N. Alqarni, M. Morad, M. E. A. Zaki, M. M. S. Sanad, A. I. Alakhras and A. A. Farag, Polymers, 2023, 15, 1–22 CrossRef.
- A. A. Farag, Corros. Rev., 2018, 36, 575–588 CrossRef CAS.
- N. Timoudan, A. S. Al-Gorair, L. El Foujji, I. Warad, Z. Safi, B. Dikici and A. Zarrouk, RSC Adv., 2024, 14, 30295–30316 RSC.
| This journal is © The Royal Society of Chemistry 2026 |