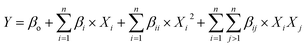Open Access Article
Open Access ArticleValorization of tannery fleshing waste into high energy biofuel through pyrolysis: process optimization and fuel properties analysis
Mohammad Al Shahriar Khan a,
Manjushree Chowdhurya,
Masud Hassan
a,
Manjushree Chowdhurya,
Masud Hassan b and
Amal Kanti Deb
b and
Amal Kanti Deb *a
*a
aInstitute of Leather Engineering and Technology, University of Dhaka, Dhaka-1000, Bangladesh. E-mail: debak.ilet@du.ac.bd
bCollege of Resources and Environmental Engineering, Guizhou University, Guiyang, Guizhou 550025, China
First published on 27th April 2026
Abstract
Fleshing waste (FW) is an unavoidable solid waste generated during the fleshing operation of the leather tanning process. FW is hazardous because it contains significant amounts of sulfides and harms the environment by polluting soil and water, and emits greenhouse gases. This study examined the pyrolysis process for producing biofuel from FW and optimized the process parameters using response surface methodology. FW contains 3.77% moisture, 15.68% ash, 71.24% volatile matter, and 9.31% fixed carbon based on dry mass, according to proximate analysis. Ultimate analysis determined 50.41% C, 9.79% H, 4.02% N, 0.24% S, and 19.85% O in FW by dry mass, yielding a higher heating value of 27.62 MJ kg−1 and a lower heating value of 25.48 MJ kg−1. Under optimized conditions (555 °C, 60 minutes, and 1 L h−1 N2 flow rate), the pyrolysis process yielded 55% biofuel from FW. FTIR analysis of biofuel identified absorption bands at 3350, 2920, 2855, 1660, and 1455 cm−1 corresponding to –OH and –NH stretching, –CH stretching, –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching, and –CH bending groups. The biofuel exhibits enhanced energy density and ease of handling, with a calorific value of 40.24 MJ kg−1, a kinematic viscosity of 2.12 cSt, and a density of 860 kg m−3. Chemical stability of the biofuel is ensured by an acid value of 16 mg KOH per g. Fuel safety is ensured by a flash point of 60 °C and a fire point of 78 °C. GC-MS analysis of biofuel detected significant compounds like decahydro-8a-ethyl-1,1,4a,6-tetramethylnaphthalene and 1-methyl-2-propylpyrazolium bromide along with other active components. Therefore, the produced biofuel offers practical application potential, promotes sustainable waste management in the leather industry, and creates opportunities for a renewable energy source.
O stretching, and –CH bending groups. The biofuel exhibits enhanced energy density and ease of handling, with a calorific value of 40.24 MJ kg−1, a kinematic viscosity of 2.12 cSt, and a density of 860 kg m−3. Chemical stability of the biofuel is ensured by an acid value of 16 mg KOH per g. Fuel safety is ensured by a flash point of 60 °C and a fire point of 78 °C. GC-MS analysis of biofuel detected significant compounds like decahydro-8a-ethyl-1,1,4a,6-tetramethylnaphthalene and 1-methyl-2-propylpyrazolium bromide along with other active components. Therefore, the produced biofuel offers practical application potential, promotes sustainable waste management in the leather industry, and creates opportunities for a renewable energy source.
1. Introduction
The leather tanning sector has a long history and is essential to the demands of society and the world economy. Animal hides and skins are tanned to make leather, but this process generates significant waste. When one ton of hide is processed, only 200 kg of leather is produced, along with 600 kg of tanned and non-tanned solid waste, and 200 kg of dissolved residue appears in wastewater.1 Every year, the leather processing sector produces over 6 million tons of solid waste globally, of which 50–60% is fleshing waste (FW).2 FW is generated during the fleshing operation of hides and skins (pelts) after the liming-unhairing process, and usually contains 4–18% fat, 5–7% protein, 2–6% lime, and 2–4% sulfide on a wet-mass basis.3 Additionally, FW contains sodium sulfide, a caustic and toxic substance that poses environmental risk.4 The high concentration of toxic water from FW pollutes the soil, surface water, and groundwater substantially. When FW comes into contact with acidic substances and the pH drops to 8.5 or lower, a high concentration of H2S gas is produced, causing severe health hazards and air pollution.5 In addition, when the protein and fatty substances in FW decompose, it produces greenhouse gases (GHGs), primarily methane. At present, FW is generally not recycled and instead ends up in landfills or is dumped in open areas, which can lead to soil contamination, increase alkalinity, and leave toxic residues. Rainwater transports hazardous constituents from fleshing waste into nearby water bodies, posing serious risks to aquatic ecosystems and humans.Several approaches to valorize FW have been reported in the literature to overcome ecological and economic pitfalls associated with it. These include anaerobic digestion for biogas production,6 bacterial composting,7 biodiesel production via transesterification,8 and hydrolysate production.9 Although these methods have advantages, there are also shortcomings, such as low efficiency, the generation of toxic by-products, secondary pollution, high operating costs, and complex procedures. To address these challenges, the pyrolysis process is proposed in this study as an alternative to conventional recovery methods to reduce the environmental impact and financial costs associated with leather solid waste.
Pyrolysis involves heating carbonaceous materials within an inert atmosphere, transforming them into gases, oils, and carbonaceous residue (biochar) at high temperatures.10 Pyrolysis is superior to combustion because it produces fewer greenhouse gas emissions that contribute to global warming. The results of pyrolysis depend on various parameters, including biomass composition, temperature, residence time, carrier gas flow rate, heating rate, and particle size.11 Various biomasses have been employed for pyrolysis over the years. Shadangi et al., 2014 pyrolyzed Karanja seeds in a semi-batch reactor at 550 °C, and the highest biofuel obtained was 55.2%.12 The pyrolysis of Mediterranean seaweed (Posidonia oceanica) generated biofuel yield of 52.4% with 32% biochar at 400 °C, and white pine produced 47.4% biofuel at 500 °C and 26.4% biochar at 400 °C.13 A microwave-based pyrolysis of lignocellulosic feedstock such as bamboo leaves, rice husks, corn stalks, sugarcane bagasse, sugarcane peel trash, coffee ash, and paddy straw was carried out with biochar yields between 18–22%, biofuel yields between 40–48%, and syngas of 30–40%.14 Tallow (animal fat) was thermally decomposed by pyrolysis in a fixed-bed reactor at laboratory scale, with a highest 12.47% biofuel yield obtained in optimum conditions of 350 °C and 140 minutes.15 The pyrolysis of chrome shaving dust from tannery waste yielded 49% biofuel, 21% syngas, and 30% biochar, whereas leather finished trimmings produced 52% biofuel, 21% syngas, and 27% biochar in a fixed-bed pyrolysis unit.16 However, the heavy metals, including chromium, lead, and cadmium, in the solid residue from the pyrolysis of tanned and finished solid leather wastes limit their practical application. On the other hand, research on non-tanned leather solid waste, such as FW, is scarce, and it contains no heavy metals; the char after pyrolysis can be safely utilized. Therefore, the current study attempts to fill this gap by considering the pyrolysis of tannery FW to produce high-energy content biofuel.
At present, fossil fuels have extremely high global demand owing to their dominant use in both the industrial and transportation sectors.17 The burning of fossil fuels results in pollutants, including unburned hydrocarbons, nitrogen oxides, carbon dioxide, and volatile organic compounds. The GHGs exacerbate climate change, the implications of which (warmer temperature) is a serious threat to humanity around the world.18 As a result, researchers, development practitioners, and industrialists are working hard to develop sustainable and viable solutions in the field of biofuels.19 There is a global consensus on the advantages of using biofuel, such as green sourcing, low or no pollution, and affordability. In addition, biofuels reduce GHG emissions that cause global warming, reduce environmental pollution, and enhance the local economy.20 FW pyrolysis to biofuel may have the potential to be an alternative to fossil fuels, as it may produce heat, electricity, and other value added products.
Although biofuel combustion results in fewer CO2 emissions, it does not completely eliminate GHG emissions. The main objective of this study is to demonstrate a waste-to-energy valorization pathway for tannery fleshing waste. This approach can reduce environmental impacts by avoiding methane (CH4) emissions from conventional FW disposal methods, such as landfilling. According to the Intergovernmental Panel on Climate Change (IPCC) Fifth Assessment Report (AR5), CH4 has 28 times the 100-year global warming potential (GWP) of CO2.21 Additionally, the biofuel produced has the potential to replace fossil fuels partially, thereby reducing net carbon emissions.
The overarching goal of this work is to provide a sustainable solution for tannery FW through resource recovery, i.e., biofuel production. The primary objectives are to pyrolyze FW to biofuel, determine the ideal operational conditions for maximum biofuel yield, examine the influence of key parameters including temperature, holding time, and N2 flow rate on biofuel production, and conduct a comprehensive characterization of the synthesized biofuel.
2. Materials and methods
2.1 Raw materials
Fleshing waste (FW) was collected from Arafat Tannery, located at the tannery industrial estate in Savar, Dhaka (TIED), Bangladesh. The samples were taken during the fleshing operation of cow limed pelts after the liming-unhairing process. After washing several times, FW was delimed using ammonium chloride and ammonium sulfate to remove lime and sulfides. Afterward, the samples were allowed to air-dry for 7 days to reduce moisture content to less than 5% and were subsequently stored in glassware for future analytical assessment and experiments.2.2 Raw material characterization
For elemental analysis, the pre-dried FW was analyzed using ASTM standard methods E777, E775, and E778 to determine carbon, hydrogen, nitrogen, and sulfur content with a CHNS analyzer (Vario MICRO Cube, Germany).25 The oxygen (O) level was determined by subtracting the combined percentages of hydrogen, nitrogen, sulfur, carbon, and ash from 100%. About 3 mg of the sample was transferred to a tin capsule and then introduced into the automatic elemental analyzer. During the analysis, the combustion and reduction temperatures were 1150 °C and 850 °C, respectively, while the gases used for conveying were helium and oxygen. The higher and lower heating values (HHV and LHV) of the sample were determined using the Dulong equations, as depicted in SI eqn (S1) and (S2) (SI text I).26,27
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 by mass) into transparent pellets. After preparation, each specimen was placed into the spectrometer's sample holder, and absorption data were collected over the 400–4000 cm−1 wavenumber range. The observed spectra were analyzed to identify the functional groups present in the sample.28
100 by mass) into transparent pellets. After preparation, each specimen was placed into the spectrometer's sample holder, and absorption data were collected over the 400–4000 cm−1 wavenumber range. The observed spectra were analyzed to identify the functional groups present in the sample.282.3 Pyrolysis experiment
The pyrolysis experiments were conducted in a small-scale fixed-bed reactor with an interior diameter of 15 cm and a height of 30 cm. The reactor was heated using an electric furnace. A photoionization detector was used to control the temperature accurately, and the K-type thermocouple in the middle was used to monitor it. The reactor was connected to a 200 mm-long steel condenser via a stainless-steel pipe, where the liquid condensate was collected. Water circulation was used to keep the condenser at a steady temperature of 25 °C. The experiment was performed under nitrogen gas flow at atmospheric pressure. Approximately 200 g of pre-dried FW was loaded onto the sample holder in the pyrolysis reactor, raised to a target temperature at various heating rates, and held at that temperature for specific durations.2.4 Experimental design
Response surface methodology (RSM) with central composite design (CCD) was employed to optimize the pyrolysis process. CCD was chosen because it can model interaction and curvature with fewer experimental runs.30 Temperature, holding time, and N2 flow rate were chosen as key parameters. The total number of trials required was determined using the following equation.| N = 2n + 2n + Cp = 23 + 2.3 + 3 = 17 |
However, three additional experiments were added to the original design to enhance the model's robustness and accuracy. The relationship between biofuel yield and influencing factors (temperature, time, and N2 flow rate) can be expressed with the following second-order polynomial equation.
Analysis of variance (ANOVA) was performed to assess how well the model fits the data. To better understand the influence of parameters, 3D response surface plots and 2D contour plots were generated to visualize the interactions among the variables. Optimization analysis, model validation, and graphical representation were all carried out using Design-Expert v25.0 software of Stat-Ease Inc.
2.5 Biofuel characterization
Biofuel's calorific value was assessed using the ASTM D240-19 standard test on a bomb calorimeter.31 The biofuel sample was placed in a sealed combustion chamber (bomb) and filled with oxygen to ensure complete combustion. The bomb was submerged in a water bath with a known heat capacity, and the biofuel was ignited electrically. The heat produced during combustion increases the temperature of the surrounding water, and the energy content is derived from this temperature variation. Kinematic and dynamic viscosities were measured using a Saybolt Seconds Universal (SSU) viscometer in accordance with ASTM D445-24.32 The SSU viscometer measured kinematic viscosity by measuring the flow time of 60 mL of fluid through a calibrated orifice under controlled temperature in an oil bath. The recorded SSU time was then converted to centistokes (Cst). The dynamic viscosity was subsequently calculated as the product of kinematic viscosity and liquid density.
The acid value of biofuel was determined by titration following the standard method of ASTM D664.33 In that process, 2 g of the biofuel sample was refluxed with neutral ethanol and titrated with a standard potassium hydroxide (KOH) solution. Phenolphthalein was used as an indicator to determine the endpoint upon a pale pink color change. Acid value was determined by the equation as illustrated in SI eqn (S5) (SI text I).
A closed-cup standard method, as per ASTM D93, was employed to assess the flash and fire points of the biofuel.34 This experiment used the Pensky-Martens Closed Cup Tester (Germany), in which the biofuel sample was enclosed in a sealed chamber and heated at a controlled rate. A small test flame was introduced at regular intervals to determine the minimum temperature at which vapors ignite (flash point). The temperature at which the biofuel sustained combustion for at least 5 seconds was determined as the fire point. The biofuel's moisture and ash contents were measured under the same conditions as Section 2.2.1. A digital pH meter with a glass electrode was used to measure the biofuel's pH in accordance with ASTM E70-19.35 The pH meter was standardized with buffer solutions before analyzing the pH of the biofuel sample.
3. Results and discussion
3.1 Feed material characteristics
| Characteristics | Fleshing waste (wt%) |
|---|---|
| Proximate analysis | |
| Moisture content | 3.77% |
| Ash content | 15.68% |
| Volatile matter | 71.24% |
| Fixed carbon | 9.31% |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|
| Ultimate analysis | |
| C | 50.41% |
| H | 9.79% |
| N | 4.02% |
| S | 0.24% |
| O | 19.85% |
| H/C | 2.33 |
| O/C | 0.30 |
| HHV | 27.62 MJ kg−1 |
| LHV | 25.48 MJ kg−1 |
The ultimate analysis of FW shows high carbon and hydrogen contents of 50.41 wt% and 9.79 wt%, respectively. FW contains very low sulfur content (0.24 wt%), enabling cleaner energy conversion than other wastes with higher sulfur content, including chrome-tanned leather waste (1.09 wt%) and finished leather waste (3.43 wt%).38,40 Since the FW were pre-treated, delimed, and washed several times, most of the unreacted inorganic sulfides were removed from the FW. The HHV of 27.62 MJ kg−1 and LHV of 25.48 MJ kg−1 highlight the energy potential of the tannery FW, as these values are higher than those of chrome-tanned leather waste (12–14 MJ kg−1) and other tannery wastes.37–41
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations that are generally assigned to the carbonyl groups.44 Another peak at 1640 cm−1 is attributed to the amide I vibration of collagen. The sharp peak at 1539 cm−1 is attributed to amide II vibrations, confirming that the waste material was proteinaceous.45 The absorption band at 1459 cm−1 corresponds to –CH bending vibration of alkanes, and signals at around 1175 and 1104 cm−1 can be ascribed to –CO stretching, indicating the presence of esters or ethers.46 The peak observed at 720 cm−1 in the low-wavenumber region is due to the rocking vibration of the methylene group (–CH2–) and is characteristic of long-chain hydrocarbons.47
O stretching vibrations that are generally assigned to the carbonyl groups.44 Another peak at 1640 cm−1 is attributed to the amide I vibration of collagen. The sharp peak at 1539 cm−1 is attributed to amide II vibrations, confirming that the waste material was proteinaceous.45 The absorption band at 1459 cm−1 corresponds to –CH bending vibration of alkanes, and signals at around 1175 and 1104 cm−1 can be ascribed to –CO stretching, indicating the presence of esters or ethers.46 The peak observed at 720 cm−1 in the low-wavenumber region is due to the rocking vibration of the methylene group (–CH2–) and is characteristic of long-chain hydrocarbons.47
 | ||
| Fig. 1 (A) FTIR spectra of FW showing the different functional groups, (B) TGA and DTG curves exhibiting various thermal phase decomposition of FW. | ||
The thermogram analysis of pre-dried FW, obtained from thermogravimetric analysis (TGA) and derivative thermogravimetric (DTG) curves, is illustrated in Fig. 1(B), highlighting four distinct decomposition phases. The first weight-loss stage occured at approximately 100 °C and was attributed to the gradual volatilization of moisture present in the sample. This was followed by an exothermic peak at 325 °C, corresponding to the decomposition of fatty substances. This stage was characterized by the maximum decomposition rate of −0.47% °C−1 and a total mass loss of 39.11%. The third stage, with a peak at 460 °C, indicateed the degradation of proteinaceous material, with a peak decomposition rate of −0.24% °C−1 and an additional weight loss of 18.79%. The last decomposition stage occured at approximately 700 °C, indicating the complete breakdown of FW, and contributed 7.0% mass loss at a maximum rate of −0.12% °C−1. Overall, the TGA and DTG analyses provide insights into the active pyrolysis zone, which falls between 325 °C and 700 °C.
3.2 Statistical modeling and process optimization of biofuel yield
| Biofuel = 46.91 + 0.39A + 0.65B − 6.23C + 0.823AB − 2.74AC + 4.05BC − 14.70A2 − 4.89B2 + 2.00C2 |
| Run | A: Temperature (°C) | B: Time (min) | C: N2 flow rate (L h−1) | Response biofuel (%) |
|---|---|---|---|---|
| 1 | 400 | 90 | 5 | 30 |
| 2 | 550 | 60 | 3 | 47 |
| 3 | 400 | 60 | 3 | 33 |
| 4 | 700 | 30 | 5 | 15 |
| 5 | 550 | 60 | 5 | 45 |
| 6 | 600 | 60 | 1 | 54.5 |
| 7 | 550 | 60 | 3 | 47 |
| 8 | 550 | 90 | 3 | 42 |
| 9 | 400 | 90 | 1 | 28 |
| 10 | 700 | 30 | 1 | 40 |
| 11 | 550 | 60 | 3 | 45 |
| 12 | 400 | 30 | 5 | 20 |
| 13 | 400 | 30 | 1 | 38 |
| 14 | 600 | 90 | 1 | 48 |
| 15 | 700 | 60 | 3 | 35 |
| 16 | 700 | 90 | 5 | 25 |
| 17 | 550 | 30 | 3 | 43 |
| 18 | 400 | 60 | 1 | 36 |
| 19 | 550 | 60 | 3 | 46 |
| 20 | 550 | 60 | 3 | 45 |
The evaluation of various regression models such as Linear, 2FI (Two-Factor Interaction), Quadratic, and Cubic to identify the most suitable match for this experimental dataset is presented in Table S1. Among these, the Quadratic model is the most appropriate, with a highly significant sequential p-value (<0.0001), an adequate p-value (0.0647), a high adjusted R2 (0.9664), and a predicted R2 (0.8077). These values imply that the model can interpret and predict the data's variance. The Quadratic model's strong fit is also supported by its statistical measures; a low standard deviation (1.89) and high R2 (0.9823) indicate minimal variation and excellent correlation with the data. The adequate precision value of 29.4692 is considerably larger than the minimum threshold of 4, indicating an excellent signal-to-noise ratio.
Moreover, the Quadratic model is also validated by the diagnostic plots in Fig. 2. The Fig. 2(A) shows that externally studentized residuals align closely with the expected normal distribution, which confirms the normality of the residuals. The predicted vs. actual plot in Fig. 2(B) shows that the linear relationship is strong, implying high predictive accuracy. The residuals vs. predicted plot in Fig. 2(C) reveals random scatter within the standard control limits, supporting homoscedasticity and model stability. Collectively, these diagnostics confirm that the Quadratic model has good statistical integrity and reliability in representing the data structure in this study.
 | ||
| Fig. 2 Diagnostic plots for the Quadratic model validation: (A) normal probability plot of residuals, (B) predicted versus actual response values, and (C) residuals versus predicted values. | ||
Table 3 also demonstrates that the overall Quadratic model is statistically significant (F-value = 61.64, and p-value < 0.0001). Among the individual factors, nitrogen flow rate has a significant impact (p-value < 0.0001), whereas temperature and time alone are not significant (p-values of 0.5506 and 0.3137, respectively). Interaction terms, such as AC (temperature × N2 flow rate) and BC (time × N2 flow rate), as well as quadratic terms like A2, B2, and C2, are also significant. This means there is a non-linear association and interaction between the variables. The insignificant lack-of-fit (p-value = 0.0647), low residual variance (35.66), and significant contribution of key terms demonstrate that this model is robust and offers a valuable contribution to the study of the pyrolysis process.
| Source | Sum of squares | Degree of freedom | Mean square | F-Value | p-Value | Remarks |
|---|---|---|---|---|---|---|
| Model | 1978.28 | 9 | 219.81 | 61.64 | <0.0001 | Significant |
| A-Temperature | 1.36 | 1 | 1.36 | 0.3816 | 0.5506 | |
| B-Time | 4.01 | 1 | 4.01 | 1.13 | 0.3137 | |
| C-N2 flow rate | 392.69 | 1 | 392.69 | 110.13 | <0.0001 | Significant |
| AB | 4.30 | 1 | 4.30 | 1.21 | 0.2979 | |
| AC | 52.62 | 1 | 52.62 | 14.76 | 0.0033 | Significant |
| BC | 120.45 | 1 | 120.45 | 33.78 | 0.0002 | Significant |
| A2 | 579.07 | 1 | 579.07 | 162.39 | <0.0001 | Significant |
| B2 | 83.82 | 1 | 83.82 | 23.51 | 0.0007 | Significant |
| C2 | 11.87 | 1 | 11.87 | 3.33 | 0.0980 | Significant |
| Residual | 35.66 | 10 | 3.57 | |||
| Lack of fit | 31.66 | 6 | 5.28 | 5.28 | 0.0647 | Not significant |
| Pure error | 4.00 | 4 | 1.00 | |||
| Cor. total | 2013.94 | 19 |
 | ||
| Fig. 3 (A) 3D surface plot, and (B) 2D contour plot depicting the combined influence of temperature and time on biofuel yield. | ||
 | ||
| Fig. 4 (A) 3D surface plot, and (B) 2D contour plot illustrating the combined influence of temperature and N2 flow rate on biofuel yield. | ||
 | ||
| Fig. 5 (A) 3D surface plot, and (B) 2D contour plot showing the combined effect of time and N2 flow rate on biofuel yield. | ||
The effect of temperature and time on biofuel yield was examined at a constant N2 flow rate of 3 L h−1, as shown in Fig. 3. The analysis revealed that, as temperature increased, biofuel yield initially rose to a maximum before declining as temperature rose further. This is probably due to increased formation of volatiles (H2, CH4, and CO) at higher temperatures.48 Also, beyond critical temperature, exposure to higher temperature speeds up crosslinking and repolymerization, resulting in enhanced biochar and syngas production rather than biofuel.49 These results are consistent with the ANOVA analysis, which indicated that the quadratic term for temperature (temperature2) was significant, whereas the linear term for temperature was not. This nonlinear effect is statistically significant (F-statistic = 162.39 and p-value < 0.0001 for the quadratic term). Time also showed a similar nonlinear trend in its effect on biofuel yield, although it was smaller than that observed for temperature (F-value of 23.51 and p-value of 0.0007 for the quadratic term). However, the interaction of time and temperature (AB) was not statistically significant with a p-value of 0.2979.
Subsequently, a further evaluation was performed to examine the interaction of temperature and N2 flow rate with the reaction time fixed at 60 minutes (Fig. 4). The findings showed that biofuel yield increased up to the optimum temperature, and further temperature increases induced secondary cracking of volatiles, thereby reducing yield (consistent with previous findings). In comparison, the impact of N2 flow rate on biofuel yield was inversely linear: higher N2 flow rates reduced biofuel production. The decrease was due to the reduced residence time of the volatile compounds in the pyrolysis reactor, which prevented them from condensing and producing biofuel.50 ANOVA analysis demonstrated that N2 flow rate had a highly significant effect on biofuel yield (F = 110.13, p < 0.0001). In addition, the interaction term between temperature and N2 flow rate (AC) is significant (F = 14.76, p = 0.0033).
Finally, the impact of time and N2 flow rate on biofuel yield was explored by keeping the temperature constant at 550 °C (Fig. 5). It was found that time had a parabolic effect on biofuel yield, and N2 flow rate had an inverse effect (consistent with previous findings). Statistical analysis confirmed the significance of the quadratic term for time and the linear term for N2 flow rate. The interaction of time and N2 flow rate (BC) was also statistically significant with an F-value of 33.78 and a p-value of 0.0002.
Three experimental validation tests were performed using these optimum conditions, and the average biofuel yield was obtained to be 55.10%, which is consistent with the predicted value (Table 4). This yield is comparable to that of biofuel produced from other feedstocks. However, the quality of fuel produced in this case appears superior, as evaluated in Section 3.3 of the biofuel property section. This enhancement is due to the composition of fleshing waste, which is especially high in protein and fat.
| Temperature (°C) | Time (min) | N2 flow rate (L h−1) | Biofuel yield (%) | |
|---|---|---|---|---|
| Predicted | 558 | 63 | 1 | 54.66 |
| Experimental | 555 | 60 | 1 | 55.10 |
3.3 Biofuel properties
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations, which are typically associated with conjugated carbonyl groups.53,54 Furthermore, the presence of an absorption band at 1455 cm−1 indicates –CH bending vibrations in alkanes and highlights the biofuel's hydrocarbon structure.55 At lower wavelengths, peaks at 1034 and 721 cm−1 that correspond to –C–O stretching and methylene (–CH2–) rocking vibrations were observed.46,47,56 These signals indicate a complex interaction between oxygenated and hydrocarbon components in the biofuel.
O stretching vibrations, which are typically associated with conjugated carbonyl groups.53,54 Furthermore, the presence of an absorption band at 1455 cm−1 indicates –CH bending vibrations in alkanes and highlights the biofuel's hydrocarbon structure.55 At lower wavelengths, peaks at 1034 and 721 cm−1 that correspond to –C–O stretching and methylene (–CH2–) rocking vibrations were observed.46,47,56 These signals indicate a complex interaction between oxygenated and hydrocarbon components in the biofuel.
| Fuel properties | Studied biofuel | Switch grass58 | Wood59 | Palm shell60 | Animal fat (poultry)57 | ASTM burner fuel standard61 |
|---|---|---|---|---|---|---|
| Calorific value (MJ kg−1) | 40.24 | 18.00 | 23.10 | 26.93 | 40.32 | Min 15 |
| Dynamic viscosity (cP) | 1.82 | — | 45.00 | — | — | — |
| Kinematic viscosity at 40 °C (cSt) | 2.12 | 19.30 | 134.00 | 1.75 | 5.32 | Max 125 |
| Specific gravity | 0.86 | — | 1.18 | — | — | — |
| Density (kg m−3) | 860.00 | 1150.00 | — | 1110 | 886.20 | — |
| Acid value (mg KOH per g) | 16.00 | — | — | — | 124.34 | — |
| Flash point (°C) | 60.00 | — | 50.00 | 99.00 | 46.00 | Min 45 |
| Fire point (°C) | 78.00 | — | — | — | — | — |
| Moisture content (%) | 30.00 | 32.70 | 22.00 | 48.78 | 0.70 | Max 30 |
| Ash content (%) | 0.01 | 0.51 | >0.01% | 0.07 | — | Max 0.25 |
| pH | 8.50 | 3.24 | 2.50 | 2.98 | 2.90 | — |
The biofuel also has a very low ash content (0.01%) and a moderate alkaline pH of 8.5, which makes it less corrosive and harmful to the environment. Other pyrolysis biofuels are much more acidic, with a pH of 3.24 or lower.57–60 Overall, the clean characteristics of the derived biofuel suggest it as a more eco-friendly and sustainable fuel alternative and further emphasize its effectiveness for use in industrial burners, engines, and power generation systems (Fig. S1 and S2). Besides, the cost of biofuel production from FW through the proposed method is significantly lower than that of conventional methods. The lab-scale production cost is only 0.33 USD L−1, and this cost will be significantly lower at large-scale production (Table S2). In addition to biofuel, this system produces around 40% solid biochar, which is free of toxic chemicals, and suitable for environmental remediation, including soil and water treatment and carbon sequestration (Fig. S3). The remaining 5% is syngas, which can also be utilized as an energy source.
Among the identified compounds, decahydro-8a-ethyl-1,1,4a,6-tetramethylnaphthalene (MW: 222, area: 11.57%) stands out for its significant contribution to the biofuel's energy density and combustion efficiency. Long-chain hydrocarbons, such as z-23-dotriaconten-2-one (MW: 462, area: 3.06%), underscore the biofuel's potential as a high-energy fuel source. 1-methyl-2-propyl-pyrazolium bromide (MW: 204, area: 10.40%) is another important component that affects fuel stability and ignition properties. Moreover, chemical compounds such as 2-pentadecanone (MW: 226, area: 3.43%) and n-[4-cyclooctylaminobutyl] aziridine (MW: 224, area: 3.58%) with the existing oxygen and nitrogen functional groups influence fuel stability, ignition performance, and emission remediation. The biofuel also contained trace levels of sulfur and halogen compounds, indicating that further purification or detoxification is necessary to meet strict environmental standards. Finally, the presence of low concentration silane derivatives and urea-derived compounds suggests potential for selective material recovery or niche chemical applications.
Based on the tanning process, sulfur in FW is expected to originate mainly from inorganic sulfides (e.g., sodium sulfide/hydrosulfide used during unhairing) and sulfur-containing amino acids (such as cysteine and methionine) present in collagen of lime fleshing.64 During pyrolysis, these sulfur sources might transform into various organosulfur compounds. Supporting this, GC-MS analysis of the produced biofuel identified trace sulfur-containing compounds, including thiane derivatives (e.g., trans-2-methyl-4-n-pentylthiane S,S-dioxide) and sulfur pentafluoride-related species, confirming the presence and transformation of sulfur species (SI Table S3). Importantly, the ultra-low sulfur content (Table 1) suggests minimal environmental impact and reduced potential for SOX emissions.
4. Conclusion
This study confirms the feasibility of producing biofuel from fleshing waste (FW) from the leather industry via pyrolysis, with optimization using response surface methodology. The pyrolysis process produced 55% biofuel at 555 °C for 60 minutes with a nitrogen flow rate of 1 L h−1. The Quadratic model provided a good fit for identifying the relationships between biofuel yield and the parameters (temperature, time, and N2 flow rate). This is confirmed by a high R2 of 0.9823 and an adequate precision of 29.4692. The effects of temperature and time were parabolic for biofuel yield, while the N2 flow rate showed a linear inverse correlation. Characterization of the fuel properties showed that the synthesized biofuel is suitable for direct energy use, with a calorific value of 40.24 MJ kg−1, a dynamic viscosity of 1.82 cP, a kinematic viscosity of 2.12 cSt, and a density of 860 kg m−3. Its better stability is reflected in an acid value of 16 mg KOH per g of fuel, and safety is indicated by flash and fire points of 60 °C and 78 °C, respectively. FTIR analysis of biofuel showed absorption bands at around 3350, 2920, 2855, 1660, and 1455 cm−1 associated with –OH and –NH stretching, –CH stretching, –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching, and –CH bending groups. The result is also well correlated with the detection of a range of hydrocarbons, oxygenates, and nitrogen-containing compounds by GC-MS.
O stretching, and –CH bending groups. The result is also well correlated with the detection of a range of hydrocarbons, oxygenates, and nitrogen-containing compounds by GC-MS.
The research concentrates on the advantages of mitigating waste issues in Bangladesh's leather industry for developing a sustainable waste management system aligning with SDG 7 (Affordable and clean energy), SDG 8 (Decent work and economic growth), SDG 12 (Responsible consumption and production), and SDG 13 (Climate action). In addition to biofuel, the pyrolysis of FW also generates about 40% biochar and 5% syngas under optimized conditions. Although external energy input is required for pyrolysis, the process can be optimized by utilizing pyrolysis by-products (e.g., syngas or char) as internal energy sources, thereby reducing reliance on fossil-based electricity. The char has also potential for value-added applications, such as carbon sequestration, soil amendment, and environmental remediation. A comprehensive life cycle assessment (LCA) would be necessary to fully quantify the net GHG emissions from the biofuel produced. Hence, future investigations should explore LCA, characterization, and utilization of by-products to enhance overall sustainability and circularity with zero waste discharge.
Author contributions
Mohammad Al Shahriar Khan: investigation, writing – original draft, visualization, validation, formal analysis, software, Manjushree Chowdhury: writing – review & editing, validation, Masud Hassan: writing – review & editing, validation, Amal Kanti Deb: conceptualization, methodology, data curation, visualization, validation, writing – review and editing, supervision, resources.Conflicts of interest
The authors declare that they have no affiliations with any organization or entity that could have influenced the content of this manuscript.Data availability
On behalf of all authors, the corresponding author would like to confirm that most of the data generated in this research is presented in the article. The additional data supporting this article has been included as part of the supplementary information (SI). Supplementary information: formulas eqn (S1)–(S5) (SI text I), statistical comparison of regression models (Table S1), cost analysis of biofuel (Table S2), compounds identified in GC-MS analysis (Table S3), purified biofuel fraction based on boiling point at 40–60 °C (Fig. S1), purified biofuel fraction based on boiling point at 60–80 °C (Fig. S2), biochar synthesized at 550 °C (Fig. S3). See DOI: https://doi.org/10.1039/d5ra10133f.Acknowledgements
Mohammad Al Shahriar Khan extends his gratitude to the Ministry of Science and Technology, Bangladesh, for providing him with the National Science and Technology (NST) fellowship. The authors express their gratitude to the Centre for Advanced Research in Sciences (CARS) at the University of Dhaka and ILET Advanced Lab for their assistance with experimental works and instrumental analyses. The authors are especially grateful to the managment of Arafat Tannery for providing the fleshing waste samples.References
- D. Masilamani, B. Madhan, G. Shanmugam, S. Palanivel and B. Narayan, J. Clean. Prod., 2016, 113, 338–344 CrossRef CAS.
- K. V. Sandhya, S. Abinandan, N. Vedaraman and K. C. Velappan, Waste Manage., 2016, 48, 638–643 CrossRef CAS PubMed.
- M. Hashem, M. N. A. Tomal and B. Mondal, Bangladesh J. Sci. Ind. Res., 2015, 50, 227–232 CrossRef CAS.
- P. Puhazhselvan, A. Pandi, P. B. Sujiritha, G. S. Antony, S. N. Jaisankar, N. Ayyadurai, P. Saravanan and N. R. Kamini, Process Saf. Environ. Prot., 2022, 157, 59–67 CrossRef CAS.
- N. Uddin, T. R. Tamanna, M. Fateh, A. Khan Panni and M. I. Hossain, Eur. J. Eng. Technol. Res., 2023, 8, 31–36 CrossRef.
- C. B. Agustini, F. Spier, M. da Costa and M. Gutterres, Resour. Conserv. Recycl., 2018, 130, 51–59 CrossRef.
- B. Ravindran and G. Sekaran, Waste Manage., 2010, 30, 2622–2630 CrossRef CAS PubMed.
- E. Alptekin, M. Canakci and H. Sanli, Fuel, 2012, 95, 214–220 CrossRef CAS.
- S. Chatterjee, A. Das, D. Paul, S. Chakraborty and P. Choudhury, J. Environ. Manage., 2023, 343, 118141 CrossRef CAS PubMed.
- S. Yaman, Energy Convers. Manag., 2004, 45, 651–671 CrossRef CAS.
- A. Al-Rumaihi, M. Shahbaz, G. Mckay, H. Mackey and T. Al-Ansari, Renewable Sustainable Energy Rev., 2022, 167, 112715 CrossRef CAS.
- K. P. Shadangi and K. Mohanty, Fuel, 2014, 115, 434–442 CrossRef CAS.
- V. Chiodo, G. Zafarana, S. Maisano, S. Freni and F. Urbani, Fuel, 2016, 164, 220–227 CrossRef CAS.
- S. L. Lo, Y. F. Huang, P. Te Chiueh and W. H. Kuan, in Energy Procedia, Elsevier Ltd, 2017, 105, 41–46 Search PubMed.
- K. A. Roni, Z. Mufrodi, B. Putri Irani and L. Legiso, Int. J. Res. Innovat. Appl. Sci., 2025, 10, 625–631 CrossRef.
- M. Velusamy, B. Chakali, S. Ganesan, F. Tinwala and S. Shanmugham Venkatachalam, Environ. Sci. Pollut. Res., 2020, 27, 29778–29790 CrossRef CAS.
- S. A. Afolalu, O. O. Yusuf, A. A. Abioye, M. E. Emetere, S. O. Ongbali and O. D. Samuel, IOP Conf. Ser. Earth Environ. Sci., 2021, 665, 012040 CrossRef.
- M. S. Karimi, S. Arif and B. Noori, Int. J. Biol. Phys. Chem. Stud., 2025, 7, 06–13 CrossRef.
- S. Chakraborty, V. Aggarwal, D. Mukherjee and K. Andras, Asia-Pac. J. Chem. Eng., 2012, 7, 254–262 Search PubMed.
- X. Ji and X. Long, Renewable Sustainable Energy Rev., 2016, 61, 41–52 CrossRef.
- T. F. Stocker, D. Qin, G.-K. Plattner, M. M. B. Tignor, S. K. Allen, J. Boschung, A. Nauels, X. Yu, V. Bex and P. M. Midgley, Climate Change 2013: the Physical Science Basis: Summary for Policymakers, Technical Summary and Frequently Asked Questions, Intergovernmental Panel on Climate Change, 2013, vol. 1, pp. 31–115 Search PubMed.
- H. Dagne, R. Karthikeyan and S. Feleke, J. Energy, 2019, 2019, 7329269 Search PubMed.
- A. M. Yahya, A. A. Adeleke, P. Nzerem, P. P. Ikubanni, S. Ayuba, H. A. Rasheed, A. Gimba, I. Okafor, J. A. Okolie and P. Paramasivam, ACS Omega, 2023, 8, 43771–43791 CrossRef CAS PubMed.
- C. C. Seah, S. H. Habib, R. S. R. M. Hafriz, A. H. Shamsuddin, N. M. Razali and A. Salmiaton, Results Eng., 2024, 22, 102301 CrossRef CAS.
- K. Akubo, M. A. Nahil and P. T. Williams, J. Energy Inst., 2019, 92, 1987–1996 CrossRef CAS.
- M. E. A. Dahou, S. Dehmani, C. Dehmani and D. Zerrouki, Pol. J. Environ. Stud., 2025, 34, 5067–5075 CrossRef.
- J. A. Oyebanji, P. O. Okekunle, O. A. Lasode and S. O. Oyedepo, Biofuels, 2018, 9, 479–487 CrossRef CAS.
- A. Koochzakzaei, M. S. Bidgoli and S. Safapour, J. Am. Leather Chem. Assoc., 2022, 117, 515–519 CrossRef.
- G. Griyanitasari, I. F. Pahlawan and E. Kasmudjiastuti, IOP Conf. Ser. Mater. Sci. Eng., 2018, 432, 012040 CrossRef.
- A. R. Akhi, A. K. Deb, M. Hassan and S. K. Paul, ACS Omega, 2025, 10, 48349–48369 CrossRef CAS PubMed.
- D. Glushkov, D. Klepikov, A. Nigay, K. Paushkina and A. Pleshko, Appl. Sci., 2023, 13, 3501 CrossRef CAS.
- A. Wolak, G. Zajac and T. Słowik, Sensors, 2021, 21, 2530 CrossRef CAS.
- P. A. P. Decote, L. Negris, A. P. Vidoto, L. A. N. Mendes, E. M. M. Flores, M. A. Vicente and M. F. P. Santos, Fuel, 2022, 313, 122642 CrossRef CAS.
- J. M. Hughes, R. H. Dennis and E. J. Beal, Energy Fuels, 1996, 10, 1276–1277 CrossRef CAS.
- D. J. Graham, B. Jaselskis and C. E. Moore, J. Chem. Educ., 2013, 90, 345–351 CrossRef CAS.
- L. R. Cavonius, N. G. Carlsson and I. Undeland, Anal. Bioanal. Chem., 2014, 406, 7313–7322 CrossRef CAS.
- A. Ganesh Kumar, S. Swarnalatha, B. Sairam and G. Sekaran, Bioresour. Technol., 2008, 99, 1939–1944 CrossRef CAS.
- M. González-Lucas, M. Peinado, J. J. Vaquero, L. Nozal, J. L. Aguirre and S. González-Egido, Energies, 2022, 15, 1273 CrossRef.
- A. Bahillo, L. Armesto, A. Cabanillas and J. Otero, Trans. ASME, J. Energy Resour. Technol., 2006, 128, 99–103 CrossRef CAS.
- J. Kluska, M. Ochnio, D. Kardaś and Ł. Heda, Waste Manage., 2019, 88, 248–256 CrossRef CAS PubMed.
- R. Chagtmi, A. Ben Hassen Trabelsi, A. Ben Abdallah, A. Maaoui, G. Lopez, M. Cortazar, H. Khedira, C. Chaden and M. Olazar, Sustain. Chem. Pharm., 2023, 33, 101130 CrossRef CAS.
- C. Branca, G. D'Angelo, C. Crupi, K. Khouzami, S. Rifici, G. Ruello and U. Wanderlingh, Polymer, 2016, 99, 614–622 CrossRef CAS.
- M. Asemani and A. R. Rabbani, J. Pet. Sci. Eng., 2020, 185, 106618 CrossRef CAS.
- S. Ulpathakumbura, N. Marikkar, L. Jayasinghe and A. History, Int. J. Plant Biol. Based Pharmaceut., 2023, 3, 104–113 CrossRef.
- T. Riaz, R. Zeeshan, F. Zarif, K. Ilyas, N. Muhammad, S. Z. Safi, A. Rahim, S. A. A. Rizvi and I. U. Rehman, Appl. Spectrosc. Rev., 2018, 53, 703–746 CrossRef CAS.
- E. Apaydın Varol and Ü. Mutlu, Energies, 2023, 16, 3674 CrossRef.
- M. Abdulla-Al-Mamun, N. Hossain, M. I. Hossain and R. Sultana, Text. Leather Rev., 2023, 6, 37–56 CrossRef CAS.
- O. Farobie, A. Amrullah, N. Syaftika, A. Bayu, E. Hartulistiyoso, W. Fatriasari and A. B. Dani Nandiyanto, ACS Omega, 2024, 9, 16665–16675 CrossRef CAS.
- N. Rambhatla, T. F. Panicker, R. K. Mishra, S. K. Manjeshwar and A. Sharma, Results Eng., 2025, 25, 103679 CrossRef CAS.
- I. Y. Mohammed, Y. A. Abakr, F. K. Kazi, S. Yusuf, I. Alshareef and S. A. Chin, Bioresources, 2015, 10, 6457–6478 CAS.
- N. H. Elsayed, G. M. Taha and O. A. Mohamed, J. Biomed. Res. Environ. Sci., 2021, 2, 1035–1043 CrossRef.
- G. C. Basak, L. Goswami, B. Chattopadhyay and A. Bandyopadhyay, Polym. Polym. Compos., 2012, 20, 279–288 CAS.
- M. F. Ali, M. S. Hossain, S. Ahmed and A. M. Sarwaruddin Chowdhury, Heliyon, 2021, 7, e06954 CrossRef.
- M. F. Ali, M. A. Ahmed, M. S. Hossain, S. Ahmed and A. M. Sarwaruddin Chowdhury, Compos., Part C: Open Access, 2022, 9, 100320 CAS.
- C. Invernizzi, T. Rovetta, M. Licchelli and M. Malagodi, Int. J. Anal. Chem., 2018, 2018, 7823248 Search PubMed.
- A. Elena, I. Gozescu, A. Dabici, P. Sfirloaga and Z. Szabadai, Macro to Nano Spectroscopy, IntechOpen, London, 2012 Search PubMed.
- A. Ben Hassen-Trabelsi, T. Kraiem, S. Naoui and H. Belayouni, Waste Manage., 2014, 34, 210–218 CrossRef CAS.
- R. He, X. P. Ye, B. C. English and J. A. Satrio, Bioresour. Technol., 2009, 100, 5305–5311 CrossRef CAS PubMed.
- D. Vamvuka, Int. J. Energy Res., 2011, 35, 835–862 CrossRef CAS.
- P. Weerachanchai, C. Tangsathitkulchai and M. Tangsathitkulchai, SAE Technical Papers, 2007, DOI:10.4271/2007-01-2024.
- A. Oasmaa, A. Källi, C. Lindfors, D. C. Elliott, D. Springer, C. Peacocke and D. Chiaramonti, Energy Fuels, 2012, 26, 3864–3873 CrossRef CAS.
- D. Pradhan, R. K. Singh, H. Bendu and R. Mund, Energy Convers. Manag., 2016, 108, 529–538 CrossRef CAS.
- S. Nanda, P. Mohanty, J. A. Kozinski and A. K. Dalai, Energy Environ. Res., 2014, 4, 21–32 Search PubMed.
- A. D. Covington and W. R. Wise, in Tanning Chemistry: the Science of Leather, The Royal Society of Chemistry, Cambridge, 2nd edn, 2019, vol. 1, pp. 05–26 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |