 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Influence of the molecular adsorption of CO2, CO and NO on the stability of oxygen vacancies on the anatase TiO2 (101) surface
Zhi-Wen Wang *,
Jie Zhang,
Meng-Ting Yue,
Wei-Guang Chen,
Ming-Yu Zhao
*,
Jie Zhang,
Meng-Ting Yue,
Wei-Guang Chen,
Ming-Yu Zhao and
Ya-Nan Tang
and
Ya-Nan Tang *
*
College of Physics and Electronic Engineering, Zhengzhou Normal University, Zhengzhou, 450044, China. E-mail: zwwang@zznu.edu.cn; yntang2010@163.com
First published on 28th April 2026
Abstract
The impact of the molecular adsorption of CO2, CO and NO on the stability of oxygen vacancies at the anatase TiO2 (101) surface is studied through first-principles calculations. Our findings reveal that the adsorption of CO2, CO, and NO stabilizes the surface oxygen vacancy relative to the subsurface vacancy, with total energies being 0.08 eV, 0.32 eV, and 1.58 eV lower, respectively. This suggests that the adsorption of these molecules can thermodynamically reverse the relative stability between the surface and subsurface oxygen vacancies, with the surface oxygen vacancy surface becoming the most stable. Additionally, we investigate the kinetic effects of oxygen vacancy interactions with small molecules. The diffusion barriers for oxygen vacancies on surfaces with adsorbed CO2 and CO are found to be 0.68 eV and 0.36 eV, respectively—significantly lower than the diffusion barriers on the clean surface by 0.16 eV and 0.50 eV, respectively. These results suggest that CO adsorption can effectively promote the diffusion of oxygen vacancies. Overall, this study highlights the crucial role of molecular adsorption in modulating the stability and interaction of oxygen vacancies on the anatase TiO2 (101) surface, providing insights into the photocatalytic activity of this material.
Introduction
Titanium dioxide (TiO2) has been extensively studied due to its promising applications in heterogeneous catalysis, solar cells, gas sensors, photocatalysis, and environmental cleaning.1–9 Although rutile TiO2 is the thermodynamically stable phase, the (110) surface has been widely used as a model surface.10–12 However, the anatase phase exhibits greater stability relative to rutile when nanoparticle dimensions are reduced below 11 nm (ref. 13), while also demonstrating superior surface reactivity.14 Among anatase surfaces, the (101) surface is the most stable.15 The interactions between small molecules and the TiO2 (101) surface have been studied extensively in recent years.16–19Surface oxygen vacancies (VsurO) are key defects on the TiO2 (110) surface and play a crucial role in surface reactions.8,11,20–27 In contrast, studies have shown that subsurface oxygen vacancies (VsubO) are more stable than surface vacancies on the anatase TiO2 (101) surface.26,27 As a result, there has been limited research on the interaction between small molecules and defective anatase (101) surfaces.17–19 Notably, Setvin et al. reported that O2 adsorption could reverse the relative stability of VsurO and VsubO.17 Additionally, other studies have shown that water and methanol can facilitate the migration of oxygen vacancies (VOs) from the subsurface to the surface on the anatase (101) surface.18,19 We previously studied the interaction between H2S and oxygen vacancies on the anatase TiO2 (101) surface and found that H2S adsorption could reverse the stability of VsurO and VsubO.28 Upon the adsorption of small molecules such as H2O, H2S and methanol, VOs on the anatase TiO2 (101) surface undergo a stabilization transition from the subsurface to the surface layer. These VOs can now function as accessible active sites, as they are no longer obstructed by steric hindrance or electronic screening from the surface molecular adlayer. Consequently, the decomposition of adsorbed small molecules including H2O, H2S, and methanol is significantly promoted, leading to an improved surface photocatalytic performance.18,19,28
Several important reactions occur on the anatase TiO2 (101) surface, such as the photocatalytic reduction of CO2 to methanol for fuel production,16,29 low-temperature CO oxidation and CO hydrogenation reactions,30–33 and the oxidation of NO to HNO3 to reduce nitrogen oxide emissions from vehicles and industrial sources.34,35 However, studies on these reactions typically overlook the stability of oxygen vacancies in the subsurface, and the effect of adsorbed molecules on the stability of oxygen vacancies is not well understood in such works. Additionally, the role of oxygen vacancies in various photocatalytic reactions remains unclear, which limits the practical application of anatase TiO2 as a catalyst.
In this study, we systematically investigate the impact of molecular adsorption (CO2 CO, and NO) on the stability of VOs at the anatase (101) surface. Our results show that after CO2, CO, and NO adsorption, the surface with VsurO becomes more energetically favorable than the surface with VsubO. This suggests that the relative stability of the surface with either a VsubO or VsurO can be reversed, with the surface containing VsurO becoming the most stable. We also examine the kinetics of VO diffusion. We find that the diffusion barriers of VOs from the subsurface to the surface with CO2 and CO adsorption are only 0.68 eV and 0.36 eV, which are significantly lower than the clean surface by 0.16 eV and 0.50 eV, respectively. These findings indicate that CO adsorption can significantly enhance the diffusion of VOs.
Computational methods
The calculations were based on density functional theory (DFT) using the Perdew–Wang 91(PW91) generalized gradient approximation36,37 and the Vienna ab initio simulation package (VASP) code with projector-augmented wave pseudopotentials.38,39 An energy cutoff of 500 eV was used for expanding the Kohn–Sham wave functions. The anatase TiO2 (101) surface was modeled as a (1 × 4) supercell slab. The stoichiometric slab containing three O–Ti–O trilayers (Ti48O96) and a vacuum with a thickness of 20 Å, with a Γ-centered 2 × 2 mesh, has been tested to be well converged. The positions of atoms in the bottom trilayer are fixed to mimic the bulk, and the other atoms were relaxed until the forces converged to 0.01 eV Å−1. To model a reduced anatase TiO2 (101) surface, an oxygen atom was removed from the slab; the reduced slab contained 48 Ti atoms and 95 O atoms, and the VO density was 1/4, which was enough for the small molecules to get adsorbed on the surface with VOs.28 To investigate the reaction kinetics by locating the transition states, we employed the nudged elastic band (NEB) method within the VASP framework.40 For each elementary step, the reaction coordinate was represented by five intermediate images constructed through a linear interpolation between the boundary states. Structural relaxations along the reaction pathway were performed using the conjugate gradient scheme, with a force convergence criterion of 0.05 eV Å−1. The reaction barrier was defined as the energy difference between the initial state to the saddle point, identified using the image with the maximum electronic energy along the minimum energy pathway.The adsorption energy of small molecules on the TiO2 (101) was calculated using the following equation:41
| Eads = Etot − Esur − Emol − EZPE − PV + TS, | (1) |
Results and discussion
Our previous studies showed that the anatase TiO2 (101) surface system containing a VsubO is more stable than that with a VsurO as the surface containing VsurO is 0.16 eV higher in total energy than the surface containing VsubO.28 Upon introducing a VsubO into the anatase TiO2 (101) surface, we considered thirteen potential adsorption sites for small molecules, as shown in Fig. 1a. The CO2 molecule maintains a linear geometry; in the calculations for all the 13 adsorption sites, it was placed vertically above the surface, and the vertical distance between the lower oxygen atom of the molecule and the surface atom directly beneath the adsorption site was 1.92 Å in each case. After relaxation, the adsorption energy of CO2 on the anatase TiO2 (101) surface with a VsubO was calculated using eqn (1). As illustrated in Fig. 1b, the most stable adsorption site for CO2 is the B8 site, with the optimized geometry shown in Fig. 1c and e. The most stable adsorption site corresponded to the next-nearest five-coordinated titanium atom (Ti-5c) site, where the distance between the lower oxygen atom of CO2 and Ti-5c was 2.59 Å, the angle between CO2 and the surface was 63.3°, and the adsorption energy at the B8 site was −0.18 eV. Additionally, the adsorption energy of CO2 at the B3 site was −0.13 eV, where CO2 got adsorbed on the nearest Ti-5c site. The energy of this site was only 0.05 eV higher than of the B8 site. The optimized geometry for this configuration is shown in Fig. 1d and f; here, the distance between the lower oxygen atom of CO2 and Ti-5c was 2.47 Å, which was shorter than at the B8 site, and the angle between CO2 and the surface was 58.9°.Fig. 2a illustrates the thirteen possible adsorption sites for small molecules on the anatase TiO2 (101) surface with a VsurO. Similarly, for CO2 adsorption at the 13 adsorption sites on the anatase (101) surface containing a VsurO, the initial configurational setup was identical to that used for the surface models with a VsubO. When CO2 was adsorbed on a surface oxygen vacancy, the distance between the bottom oxygen atom of CO2 and the original oxygen atom at the vacancy was 1.92 Å. The adsorption energies after relaxation are shown in Fig. 2c. Results indicated that the most stable adsorption site for CO2 was the R2 site, with the optimized geometric structures shown in Fig. 2b and d. The most stable adsorption occurs at the top of the VsurO, where the angle between CO2 and the surface is 77.6°, and the adsorption energy at the R2 site is −0.37 eV.
Our previous studies showed that a VsubO is more stable than a VsurO on a clean anatase TiO2 (101) surface, with the surface containing a VsurO being 0.16 eV higher in total energy than the surface with a VsubO. As a result, after CO2 adsorption on the B3 and B8 sites, the surface with the VsurO is 0.08 eV and 0.03 eV lower in total energy than the surface with the VsubO.
After CO2 adsorption, the VO tends to migrate from the subsurface to the surface. The calculated VO diffusion pathway and optimized geometrical structures are shown in Fig. 3. Initially, the VO is located in the subsurface layer, with CO2 adsorbed on the B3 site (Fig. 3a). The oxygen atom beneath the surface (O-2c) then diffuses to the VsubO site (Fig. 3b) and forms a  complex (Fig. 3c). Subsequently, the surface O-2c atom diffuses to the
complex (Fig. 3c). Subsequently, the surface O-2c atom diffuses to the 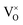 site (Fig. 3d), forming a VsurO with CO2 adsorbed at the R2 site (Fig. 3e). The energy profiles are shown in Fig. 3f. Our calculations showed that the diffusion pathway of the VO from the subsurface to the surface was similar to that of the clean surface.28 The diffusion barrier of VO on the CO2-adsorbed surface was 0.68 eV, which was 0.18 eV lower than on the clean surface, suggesting that CO2 adsorption promotes VO diffusion.
site (Fig. 3d), forming a VsurO with CO2 adsorbed at the R2 site (Fig. 3e). The energy profiles are shown in Fig. 3f. Our calculations showed that the diffusion pathway of the VO from the subsurface to the surface was similar to that of the clean surface.28 The diffusion barrier of VO on the CO2-adsorbed surface was 0.68 eV, which was 0.18 eV lower than on the clean surface, suggesting that CO2 adsorption promotes VO diffusion.
Next, we examined the adsorption energy of CO on the anatase TiO2 (101) surface with a VsubO. Thirteen possible adsorption sites and two adsorption types for CO were considered. In type 1, the CO molecule was oriented vertically with the C atom closest to the surface (Fig. 4a and c), and in type 2, the CO molecule is oriented vertically with the O atom closest to the surface (Fig. 4b). The adsorption energies of CO on the anatase TiO2 (101) surface with a VsubO are shown in Fig. 4d. Results indicated that CO preferred to adsorb via type 1 configuration, where the C atom was closest to the surface, as this configuration had a lower adsorption energy than type 2 configuration. The most stable adsorption site for CO was the B3 site (type 1), and the optimized geometric structure is shown in Fig. 4a and c. The distance between CO and Ti-5c was 2.38 Å, the angle between CO and the surface was 80.1°, and the adsorption energy was −0.25 eV. In contrast, for type 2, the distance between the O atom and Ti-5c was 2.85 Å, the angle between the CO axis and the surface was 86.5°, and the adsorption energy was −0.08 eV. Furthermore, as illustrated in Fig. 4d, the adsorption energy of type 1 is consistently lower than that of type 2 at almost all equivalent adsorption sites. This thermodynamic trend clearly indicated that the CO molecule preferentially adsorbed via type 1 mode, making it the most stable configuration for this system.
We also investigated the adsorption energy of CO on the anatase TiO2 (101) surface with a VsurO. Thirteen potential adsorption sites (Fig. 2a) and two adsorption types for CO were considered. Similar to the surface with a VsurO, in type 1, the CO molecule was oriented vertically with the C atom closest to the surface (Fig. 5a and c), and in type 2, the CO molecule was oriented vertically with the O atom closest to the surface (Fig. 5b). The adsorption energies of CO on the anatase TiO2 (101) surface with a VsurO are shown in Fig. 5d. It can be seen that, except for the R7 and R12 sites, the adsorption energies of CO in type 1 configuration were lower than those in type 2 configuration. The most stable adsorption site for CO was the R2 site in type 1 configuration, with the optimized geometry shown in Fig. 5a and c. The most stable adsorption occurred at the VsurO site, where the angle between CO and the surface was 57.8° and the adsorption energy was −0.75 eV, which was significantly larger than the energy for the same site in type 2 configuration.
Similar to CO2, the surface with a VsurO was 0.32 eV lower in total energy than the surface with a VsubO after CO adsorption. This indicates that CO adsorption can thermodynamically reverse the relative stability of the surface with either a VsubO or VsurO. After CO adsorption on the anatase TiO2 (101) surface with a VsubO, the relative stability of the configuration with the VsubO decreased, and the surface with the VsubO became the most stable.
The diffusion of the VO from the subsurface to the surface with CO adsorption is shown in Fig. 6. The diffusion pathway of the VO was similar to that observed with CO2 adsorption. However, the diffusion barrier of the VO on the surface with CO adsorption was only 0.36 eV, which was significantly lower than that on the clean surface by 0.50 eV. These results suggested that CO adsorption effectively promoted VO diffusion from the subsurface to the surface layer.
Next, we studied the adsorption energy of NO on the anatase TiO2 (101) surface with a VsubO. Similar to CO, thirteen possible adsorption sites and two adsorption types for NO were considered. In type 1, the N atom of NO was the closest to the surface (Fig. 7a and c), while in type 2, the O atom of NO was the closest to the surface (Fig. 7b). The adsorption energy of NO on the anatase TiO2 (101) surface with a VsubO is shown in Fig. 7d. Results indicated that the B3 site was the most stable adsorption site for NO, with an adsorption energy of −0.86 eV, which was much lower than the adsorption energies of CO2 and CO at the same site by 0.68 eV and 0.61 eV, respectively. The B3 site was located on the nearest Ti-5c site, where the distance between the N atom of NO and Ti-5c was 1.86 Å, and the angle between NO and the surface was 70.8°.
We also studied the adsorption energy of NO on the anatase TiO2 (101) surface with a VsurO. The adsorption sites and the two identified configurations for NO are analogous to those observed for CO. Type 1 and type 2 configurations on the R2 sites are shown in Fig. 8(a–c). The adsorption energy of NO on the anatase TiO2 (101) surface with a VsurO is shown in Fig. 8d. Results indicated that the B3 site was the most stable adsorption site for NO, with an adsorption energy of −2.60 eV, which was significantly lower than the adsorption energies of CO2 and CO by 2.23 eV and 1.85 eV, respectively. The N atom of NO was positioned on the VsurO site, and the angle between NO and the surface was 24.4°, indicating that NO is positioned almost parallel to the surface.
After comparing the total energy following NO adsorption, we found that the surface with the VsurO is 1.58 eV lower in energy than the surface with the VsubO. This suggested that the relative stability between the surfaces with the VsubO and VsurO can be reversed through NO adsorption.
To understand the adsorption characteristics of different small molecules on the anatase TiO2 (101) surface, we calculated the differential charge density for the stable adsorption configurations of CO2, CO, and NO on the anatase TiO2 (101) surface with a VsubO or VsurO, as shown in Fig. 9. When CO2 was adsorbed on the surface with a VsubO (Fig. 9a), the charge density around the oxygen and carbon atoms of CO2 decreased, while the charge density between the lowest oxygen atom of CO2 and the surface increased. However, the amount of charge transfer was very small, indicating the weak adsorption of CO2 on this surface. Conversely, when CO2 was adsorbed on the surface with a VsurO (Fig. 9d), the increase in charge density between the lowest oxygen atom of CO2 and the surface was greater than that observed on the VsubO surface, suggesting the stronger adsorption of CO2 on the surface with a VsurO. When CO was adsorbed on the surface with a VsubO (Fig. 9b), similar to the case of CO2 on this surface, less charge transfer occurred. However, when CO was adsorbed on a surface with a VsurO (Fig. 9e), the charge density around the surface atoms near the VsurO decreased, and a significant amount of charge accumulated between CO and the surface, indicating the strong bonding between CO and this surface. When NO was adsorbed on the surface with a VsubO (Fig. 9c), a high charge density from the vicinity of the surface atoms accumulated between NO and the surface. The amount of charge transfer was greater than that observed for CO2 and CO adsorption on this surface, and its adsorption energy was consequently larger. Furthermore, when NO was adsorbed on the surface with a VsurO (Fig. 9f), the angle between NO and the surface was 24.4°, indicating that NO was positioned almost parallel to the surface. A large amount of charge was accumulated not only between the N atom of NO and the surface but also between the O atom of NO and the surface, leading to a very strong bond between NO and the surface. The close agreement between the charge analysis and adsorption energy results confirmed the robustness of our findings.
Conclusions
We investigated the adsorption sites and energies of CO2, CO, and NO molecules upon their adsorption on the anatase (101) surface with a VsubO or VsurO. On the surface with a VsubO, the most stable adsorption sites and corresponding energies are B8 (−0.18 eV), B3 (−0.25 eV), and B3 (−0.86 eV) for CO2, CO, and NO, respectively. On the surface with VsurO, the most stable sites and energies are R2 (−0.37 eV), R3 (−0.75 eV), and R3 (−2.60 eV) for CO2, CO, and NO, respectively. These charge analysis results closely match the adsorption energy findings, which validates the scientific soundness of our work. Importantly, we found that the surface with a VsurO is 0.03 eV, 0.32 eV, and 1.58 eV lower in total energy than the surface with a VsubO after CO2, CO, and NO adsorption, respectively. This indicates that small molecule adsorption can thermodynamically reverse the relative stability of the surface with either a VsubO or VsurO. Additionally, we observed that the diffusion barriers of VOs on the surface with CO2 and CO adsorption are only 0.68 eV and 0.36 eV, respectively, which are much lower than those on the clean surface by 0.16 eV and 0.50 eV, respectively. These results suggest that CO adsorption effectively promotes VO diffusion.Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data supporting the findings of this study are available within the article. Additional datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.Acknowledgements
The calculations were carried out at the High-Performance Computing Center of College of Physics and Electronic Engineering, Zhengzhou Normal University. This work was supported by the National Natural Science Foundation of China (Grant No. 12204430), the Natural Science Foundation of Henan (Grant No. 222300420378), the Technologies Research and Development Program of Henan Province (Grant No. 262102231043, 262102231046), the Key Scientific Research Project of Henan College (Grant No. 26A140016), the Innovative Research Team (in Science and Technology) in University of Henan Province (Grant No. 25IRTSTHN015), the Innovation and Entrepreneurship Training Program for college students in Henan Province (Grant No. 202312949014), and the Scientific Research and Innovation Fund for college students in Zhengzhou Normal University (Grant No. 2024009).References
- H. Wang, H. Qi, H. Jia, S. Li and X. Li, High quantum efficiency of hydrogen production from methanol aqueous solution with PtCu-TiO2 photocatalysts, Nat. Mater., 2023, 22, 619 CrossRef CAS PubMed.
- O. Aldosari and U. Obaid, Unlocking the potential of TiO2-based photocatalysts for green hydrogen energy through water-splitting: Recent advances, future perspectives and techno feasibility assessment, Int. J. Hydrogen Energy, 2024, 59, 958–981 CrossRef CAS.
- W. Yuan, B. Zhu, K. Fang, X. Y. Li, J. Yang, T. S. Hansen, Y. Ou, H. Yang, J. B. Wagner, Y. Gao, Y. Wang and Z. Zhang, In situ manipulation of the active Au-TiO2 interface with atomic precision during CO oxidation, Science, 2021, 371, 517 CrossRef CAS PubMed.
- Z. Jiang, X. Xu, Y. Ma, H. S. Cho, D. Ding, C. Wang, J. Wu, P. Oleynikov, M. Jia and J. Cheng, et al., Filling Metal-Organic Framework Mesopores with TiO2 for CO2 Photoreduction, Nature, 2020, 586, 549 CrossRef CAS PubMed.
- F. Xu, K. Meng, B. Cheng, S. Wang, J. Xu and J. S. Yu, Unique, Scheme Hetero Junctions in Self-Assembled TiO2/CsPbBr3 Hybrids for CO2 Photoreduction, Nat. Commun., 2020, 11, 4613 CrossRef CAS PubMed.
- W. Li, A. Elzatahry, D. Aldhayan and D. Zhao, Core-Shell Structured Titanium Dioxide Nanomaterials for Solar Energy Utilization, Chem. Soc. Rev., 2018, 47, 8203–8237 RSC.
- E. J. W. Crossland, N. Noel, V. Sivaram, T. Leijtens, J. A. Alexander-Webber and H. J. Snaith, Mesoporous TiO2 Single Crystals Delivering Enhanced Mobility and Optoelectronic Device Performance, Nature, 2013, 495, 215–219 CrossRef CAS PubMed.
- R. Asahi, T. Morikawa, T. Ohwaki, K. Aoki and Y. Taga, Visible-Light Photocatalysis in Nitrogen-doped Titanium Oxides, Science, 2001, 293, 269–271 CrossRef CAS PubMed.
- M. Gratzel, Photoelectrochemical Cells, Nature, 2001, 414, 338–344 CrossRef CAS PubMed.
- S. Selcuk and A. Selloni, Facet-Dependent Trapping and Dynamics of Excess Electrons at Anatase TiO2 Surfaces and Aqueous Interfaces, Nat. Mater., 2016, 15, 1107–1112 CrossRef CAS PubMed.
- U. Diebold, The Surface Science of Titanium Dioxide, Surf. Sci. Rep., 2003, 48, 53–229 CrossRef CAS.
- M. A. Henderson, The Interaction of Water with Solid Surfaces: Fundamental Aspects Revisited, Surf. Sci. Rep., 2002, 46, 1–308 CrossRef CAS.
- H. Zhang and J. F. Banfield, Understanding Polymorphic Phase Transformation Behavior during Growth of Nanocrystalline Aggregates: Insights from TiO2, J. Phys. Chem. B, 2000, 104, 3481–3487 CrossRef CAS.
- L. Kavan, M. Grätzel, S. E. Gilbert, C. Klemenz and H. J. Scheel, Electrochemical and Photoelectrochemical Investigation of Single-Crystal Anatase, J. Am. Chem. Soc., 1996, 118, 6716–6723 CrossRef CAS.
- M. Lazzeri, A. Vittadini and A. Selloni, Structure and Energetics of Stoichiometric TiO2 Anatase Surfaces, Phys. Rev. B: Condens. Matter Mater. Phys., 2001, 63, 155409 CrossRef.
- J.-Y. Liu, X.-Q. Gong, R. Li, H. Shi, S. B. Cronin and A. N. Alexandrova, (Photo)Electrocatalytic CO2 Reduction at the Defective Anatase TiO2 (101) Surface, ACS Catal., 2020, 10, 4048–4058 CrossRef CAS.
- M. Setvin, U. Aschauer, P. Scheiber, Y. Li, M. Hou, W. Schmid, Y. Selloni and U. Diebold, Reaction of O2 with Subsurface Oxygen Vacancies on TiO2 Anatase (101), Science, 2013, 341, 988–991 CrossRef CAS PubMed.
- Y. Li and Y. Gao, Interplay between Water and TiO2 Anatase (101) Surface with Subsurface Oxygen Vacancy, Phys. Rev. Lett., 2014, 112, 206101 CrossRef.
- X. Lang, Y. Liang, L. Sun, S. Zhou and W.-M. Lau, Interplay between Methanol and Anatase TiO2(101) Surface: The Effect of Subsurface Oxygen Vacancy, J. Phys. Chem. C, 2017, 121, 6072–6080 CrossRef CAS.
- S. Wendt, R. Schaub, J. Matthiesen, E. Vestergaard, E. Wahlstrom, M. Rasmussen, P. Thostrup, L. Molina, E. Lagsgaard and I. Stensgaard, et al., Oxygen Vacancies on TiO2(110) and Their Interaction with H2O and O2: A Combined High-Resolution STM and DFT Study, Surf. Sci., 2005, 598, 226–245 CrossRef CAS.
- D. J. Shu, S. T. Ge, M. Wang and N. B. Ming, Interplay between External Strain and Oxygen Vacancies on A Rutile TiO2(110) Surface, Phys. Rev. Lett., 2008, 101, 116102 CrossRef PubMed.
- J. Zheng, Y. Lyu, R. Wang, C. Xie, H. Zhou, S. P. Jiang and S. Wang, Crystalline TiO2 Protective Layer with Graded Oxygen Defects for Efficient and Stable Silicon-Based, Nat. Commun., 2018, 9, 3572 CrossRef PubMed.
- S. M. Wu, X. L. Liu, X. L. Lian, G. Tian, C. Janiak, Y. X. Zhang, Y. Z. Lu, H. Z. Yu, J. Hu and H. Wei, et al., Homojunction of Oxygen and Titanium Vacancies and its Interfacial n-p Effect, Adv. Mater., 2018, 30, 1802173 CrossRef PubMed.
- Z. W. Wang and D. J. Shu, Intrinsic Interaction Between In-Plane Ferroelectric Polarization and Surface Adsorption, Phys. Chem. Chem. Phys., 2019, 21, 18680–18685 RSC.
- Z. W. Wang, W. G. Chen, D. Teng, J. Zhang, A. M. Li, Z. H. Li and Y. N. Tang, The Effect of Strain on Water Dissociation on Reduced Rutile TiO2(110) Surface, RSC Adv., 2021, 11, 8485–8490 RSC.
- Y. He, O. Dulub, H. Cheng, A. Selloni and U. Diebold, Evidence for the Predominance of Subsurface Defects on Reduced Anatase TiO2(101), Phys. Rev. Lett., 2009, 102, 106105 CrossRef PubMed.
- P. Scheiber, M. Fidler, O. Dulub, M. Schmid, U. Diebold, W. Hou, U. Aschauer and A. Selloni, (Sub)Surface Mobility of Oxygen Vacancies at the TiO2 Anatase (101) Surface, Phys. Rev. Lett., 2012, 109, 136103 CrossRef PubMed.
- Z. Wang, W. Chen, D. Teng, J. Zhang, A. Li, Z. Li and Y. Tang, Interplay between H2S and anatase TiO2(101) surface: The effect of subsurface oxygen vacancy, J. Phys. Chem. C, 2022, 126, 3939–3948 CrossRef CAS.
- J. Liu, X. Gong and A. N. Alexandrova, Mechanism of CO2 Photocatalytic Reduction to Methane and Methanol on Defected Anatase TiO2 (101): A Density Functional Theory Study, J. Phys. Chem. C, 2019, 123, 3505–3511 CrossRef CAS.
- J. Shi, F. Yang, X. Zhao, X. Ren, Y. Tang and S. Li, Spin-polarized p-block antimony/bismuth single-atom catalysts on defect-free rutile TiO2(110) substrate for highly efficient CO oxidation, Phys. Chem. Chem. Phys., 2024, 26, 16459 RSC.
- M. Setvin, M. Buchholz, W. Hou and C. Zhang, A Multitechnique Study of CO Adsorption on the TiO2 Anatase (101) Surface, J. Phys. Chem. C, 2015, 119(36), 21044–21052 CrossRef CAS.
- K. M. Bulanin, A. V. Emeline, R. V. Mikhaylov, A. Y. Mikheleva, A. V. Rudakova and V. K. Ryabchuk, Dynamics of Adsorbed CO Molecules on the TiO2 Surface under UV Irradiation, J. Phys. Chem. C, 2023, 127, 11005–11013 CrossRef CAS.
- C. Fu, H. Xu, L. Wu, Z. Zhang and W. Huang, X-ray-Induced CO2 Formation via CO Reaction with TiO2 at Cryogenic Temperature, J. Phys. Chem. Lett., 2021, 12, 9741–9747 CrossRef CAS PubMed.
- Z. Wang, G. Lv, C. Zhang and X. Sun, Catalytic Oxidation Mechanism of NO to HNO3 on TiO2 (101) and (001) Surfaces and the Influence Factors on NO Removal: A DFT Study, J. Environ. Chem. Eng., 2021, 9, 104643 CrossRef CAS.
- L. Mino, M. Cazzaniga, F. Moriggi and M. Ceotto, Elucidating NOx Surface Chemistry at the Anatase (101) Surface in TiO2 Nanoparticles, J. Phys. Chem. C, 2023, 127, 437–449 CrossRef CAS PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Generalized Gradient Approximation Made Simple, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed.
- F. Allegretti, S. O'Brien, M. Polcik, D. I. Sayago and D. P. Woodruff, Adsorption Bond Length for H2O on TiO2(110): A Key Parameter for Theoretical Understanding, Phys. Rev. Lett., 2005, 95, 226104 CrossRef CAS PubMed.
- G. Kresse and D. Joubert, From Ultrasoft Pseudopotentials to the Projector Augmented-Wave Method, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59, 1758–1775 CrossRef CAS.
- P. E. Blöchl, Projector Augmented-Wave Method, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 50, 17953–17979 CrossRef PubMed.
- G. Mills, H. Jonsson and G. K. Schenter, Reversible Work Transition State Theory: Application to Dissociative Adsorption of Hydrogen, Surf. Sci., 1995, 324, 305–337 CrossRef CAS.
- V. Wang, N. Xu, J.-C. Liu, G. Tang and W.-T. Geng, Comput. Phys. Commun., 2021, 267, 108033 CrossRef CAS.
- C. Freysoldt, B. Grabowski, T. Hickel, J. Neugebauer, G. Kresse, A. Janotti and C. G. Van de Walle, Rev. Mod. Phys., 2014, 86, 253–305 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |









