Open Access Article
Open Access ArticleRecent advances of wearable electrochemical and optical sensors for sweat pH monitoring
Maroua Moslah a,
Chaker Tlili
a,
Chaker Tlili b and
Chérif Dridi
b and
Chérif Dridi *a
*a
aNANOMISENE Laboratory, LR16CRMN01, Centre for Research on Microelectronics and Nanotechnology of Sousse (CRMN), Technopole of Sousse B.P. 334, Sahloul, Sousse 4054, Tunisia. E-mail: cherif.dridi@crmn.rnrt.tn; Fax: +216 73 823 00; Tel: +21698940256
bResearch Center for Precision Medicine and Single-Molecule Diagnostics CIGIT, UCAS, China
First published on 9th April 2026
Abstract
Measuring the pH of human biofluids provides meaningful information about physiological balance and potential pathological changes. While blood remains the conventional medium for diagnostic evaluation, there has been an increasing focus on external biofluids, such as sweat, that enable non-invasive, continuous health assessment. Recent advances in wearable sensing technologies have resulted in the development of flexible, miniaturized and biocompatible systems that can monitor sweat pH in real time on the body. This review highlights the latest progress in wearable electrochemical and optical sensors designed for sweat pH monitoring. It first outlines the general procedures for sweat pH sensing, including sweat secretion and collection, pH sensing, signal conversion, and data interpretation. Subsequent sections focus on electrochemical approaches, particularly potentiometric sensors based on ion-selective membranes and ion-sensitive field-effect transistor (ISFET) configurations, which are recognized for their high sensitivity and rapid response times. The review further explores optical sensing technologies, such as surface-enhanced Raman scattering (SERS), fluorescence and colorimetric sensors, emphasizing their versatility for visual and spectroscopic detection. Looking ahead, the integration of artificial intelligence, microfluidic systems, and self-powered modules will facilitate the development of fully autonomous, wireless sensing platforms. The convergence of advanced materials, sensor engineering, and digital health technologies is expected to establish wearable sweat pH sensors as a cornerstone of personalized medicine, preventive care, and real-time health tracking.
1 Introduction
To facilitate the non-invasive disease diagnosis of patients, there is a growing emphasis on assessing the pH levels of external biofluids, such as urine, saliva, tears and sweat, as an alternative to traditional blood-based analyses.1,2 The pH values of these biofluids can provide valuable insights into various physiological states related to health. For example, urinary pH can provide valuable information about kidney function,3 renal lithiasis4 and tubular acidosis.5 Similarly, salivary pH has been linked to metabolic disorders such as diabetes6 and gastro-oesophageal reflux disease.7 A reduction in tear pH can act as an early marker for corneal infections before clinical symptoms manifest.8–10 Despite the fact that pH measurements in these biofluids provide diagnostic insights, they encounter several limitations. For instance, urine pH is unstable when subjected to elevated temperatures.11 The high viscosity of saliva typically requires pre-treatment processes such as filtration, thereby increasing operational complexity and cost.12 Furthermore, evaluating tear pH is technically challenging due to the small sample size, rapid evaporation rate and the need for buffer solutions to dilute the fluid for subsequent chemical analysis.13Compared to other biofluids, sweat offers the advantage of non-invasive collection, increased sample availability, reduced risk of contamination and ability to accurately reflect an individual's physiological and biochemical status. Therefore, irregular sweat pH levels have been associated with several disorders. For example, individuals with type 2 diabetes mellitus typically have lower sweat pH levels than healthy people.14 Altered sweat pH levels have also been linked to various dermatological conditions, such as irritant contact dermatitis, acne, atopic dermatitis, ichthyosis and Candida albicans infections.15 Additionally, variations in sweat pH may indicate underlying pathological states, such as tumour metastasis, microbial infections, systemic acidosis and impaired wound healing.16 Patients diagnosed with cystic fibrosis often exhibit alkaline sweat, with pH levels occasionally rising to 9 due to a deficit in bicarbonate reabsorption.17 Given these associations, reliable sweat pH monitoring shows great potential for ongoing health surveillance.
In general, pH measurements are traditionally performed using either glass electrodes or pH indicator paper. Glass electrodes, regarded as the gold standard for pH measurement, operate by measuring the potential difference across a pH-sensitive glass membrane separating two solutions of different pH. While this technique provides high accuracy and sensitivity under controlled laboratory conditions, it suffers from several inherent limitations, including the requirement for relatively large sample volumes, susceptibility to temperature fluctuations, frequent calibration, mechanical fragility, and poor portability. Consequently, glass electrodes are unsuitable for continuous, on-body, or real-time monitoring applications. Similarly, pH indicator paper offers a simple and low-cost alternative; however, it provides only semi-quantitative results, with limited sensitivity, subjective visual interpretation, and no capability for continuous or real-time measurement. These shortcomings significantly restrict its use in precise and dynamic physiological monitoring. Unlike conventional laboratory-based systems, wearable electrochemical and optical sweat pH sensors overcome these limitations by enabling real-time, continuous, and non-invasive sweat pH monitoring using flexible and skin-conformal platforms. Electrochemical sensors offer rapid response times and quantitative electrical outputs, while optical sensors enable visual or optoelectronic readout with minimal interference and enhanced user comfort. Owing to their high sensitivity within the physiological pH range, fast response, mechanical flexibility, and suitability for on-body operation, wearable pH sensors represent a promising alternative for next-generation personalized health monitoring in real-world scenarios.
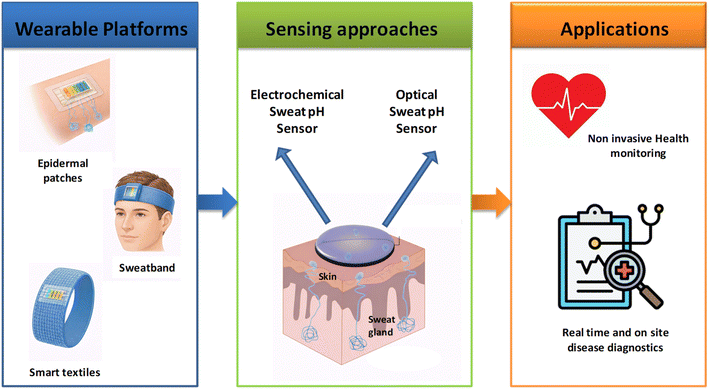
The three primary categories of wearable sweat pH sensing platforms that have recently emerged are epidermal patches (including tattoo-like sensors), smart textiles,18,19 and sweatband-type devices.20 Each format offers distinct benefits in terms of comfort, adaptability to different surfaces, and practicality for daily life and physical activity. To guarantee user safety and reliable signal acquisition, critical factors such as the flexibility, stretchability, breathability, biocompatibility and mechanical robustness of the sensing materials must be carefully evaluated. Additionally, wearable sensors can incorporate wireless communication modules to facilitate real-time transmission of physiological data to mobile devices or cloud-based systems.21–24 This connectivity converts wearable sweat pH sensors into powerful tools for continuous health monitoring, early disease detection and personalized medicine.
To the best of our knowledge, this review is the first to provide a comprehensive overview of wearable sweat sensors, with an exclusive focus on sweat pH as a critical physiological parameter for real-time, non-invasive health monitoring. The novelty of this work lies in its integrated analysis of both electrochemical and optical wearable sweat pH sensors, offering a systematic comparison of their sensing mechanisms, key performance metrics, and respective advantages and limitations. In addition, the review examines essential sensor design aspects, including flexible substrates, biocompatible materials, and electrode or optical configurations, alongside performance indicators such as sensitivity, stability, response time, and operational pH range. By critically assessing current technologies and remaining challenges, this review defines key requirements for advancing wearable sweat pH sensors toward practical healthcare applications and highlights emerging trends and future directions for their clinical translation and integration.
2 Procedure for sweat pH sensing
Fig. 1 presents a flowchart summarizing the key steps involved in sweat pH sensing using wearable devices. The workflow begins with sweat secretion being induced, either naturally through physical activity or artificially using iontophoresis or thermal methods.25 Once sweat has been produced, it is efficiently collected and transported to the sensing area via microfluidic systems designed to prevent evaporation and contamination. These systems frequently incorporate hydrophilic materials and flexible channels to ensure efficient, continuous flow of sweat. The third stage involves pH sensing, during which the hydrogen ion concentration in the collected sweat is converted into measurable signals using a specialized sensing interface. Subsequently, these signals are transmitted to the electronic processing unit, where they undergo filtering, amplification and calibration to ensure accurate readings. Finally, the processed data is transmitted wirelessly to external devices for real-time monitoring or further analysis. This integrated workflow enables the non-invasive, continuous and reliable assessment of physiological conditions through sweat pH analysis.92.1 Sweat secretion
Sweat secretion varies considerably between individuals and is influenced by factors such as physiological state, health, hydration level and physical activity. While wearable sweat sensors are commonly used during physical activity, especially by athletes, the volume of sweat produced naturally under these conditions may not always be sufficient for reliable biochemical analysis. This limitation has led to the development of controlled sweat stimulation techniques, which ensure consistent and adequate sample volumes. To this end, several sweat stimulation strategies have been explored, including thermal, physical, and chemical methods. Among these, iontophoresis has emerged as one of the most effective and widely used approaches for wearable sweat pH sensing. This technique involves applying a low-voltage electrical current through the skin to deliver a sweat-inducing agent, such as pilocarpine, into the dermis via an electrolyte gel, thereby stimulating eccrine sweat glands and inducing localised sweat secretion. The iontophoretic stimulation device can be programmed to control sweat secretion rates and collection intervals, enabling the acquisition of a consistent and sufficient sweat volume for subsequent sensor analysis. Fig. 2 illustrates a schematic representation of the iontophoresis process.26 Despite these advantages, iontophoresis presents several limitations that must be considered. Prolonged or repeated stimulation may cause skin irritation or discomfort, and sweat rates can vary significantly due to inter-subject variability related to skin properties, age, and physiological condition. In addition, the application of electrical current and chemical agents raises ethical and safety considerations, particularly for long-term or frequent use in wearable healthcare applications. Compared with exercise-induced sweating, iontophoresis offers the advantage of controlled and localised sweat generation independent of physical activity or environmental conditions, making it particularly suitable for sedentary or clinical monitoring scenarios. However, exercise-induced sweating reflects a more natural physiological state and may better represent real-world sweat composition, albeit with reduced control over sweat rate, location, and timing. A clear understanding of these trade-offs is essential for selecting appropriate sweat stimulation strategies in wearable pH sensing applications.2.2 Sweat sampling
Microfluidic technology is used to continuously collect and transport sweat from the skin to the sensing area. This enables efficient collection while minimizing the risk of evaporation and external contamination. The microfluidic system is frequently combined with a sweat-absorbent material (or pad) made from a porous, hydrophilic structure such as cellulose paper. This efficiently collects small volumes of sweat through capillary force and guides its continuous flow through channels sized in the micrometer range. Microfluidic sweat systems can be designed using flexible, biocompatible, and low-cost materials such as silicone elastomers and plastics, employing simple fabrication techniques such as molding and CO2 laser engraving.27 This platform enables autonomous sample storage, filtration, transportation and multiple detection.2.3 Sweat pH sensing
The sensing structure consists of a sensing area that effectively converts hydrogen ion concentrations into measurable signals. Key features of wearable sensors intended for pH sweat sensing include flexibility, durability, biocompatibility and light weight. To ensure optimal contact between the sensor interface and the skin, which is crucial for accurate sweat pH measurements, wearable sensors have been specifically designed for sweat analysis. These sensors are being used in an increasing number of locations on the body, including the wrist, forehead, chest, hands, feet and neck. They are presented in various formats, such as sweatbands, textile-based systems and epidermal patches (tattoos).28 Each format has its own advantages and limitations in terms of reliability, user comfort, and signal stability. Sweatbands are particularly useful for areas of the body where they can be tightened securely, such as the wrist, arm or forehead. This ensures stable skin contact and reliable signal acquisition during physical activity. However, their performance may be affected by motion artefacts and pressure variations during prolonged use. Textile-based wearable sensors are a highly comfortable and unobtrusive solution for continuous sweat monitoring as they can be seamlessly integrated into garments without restricting movement. Although textiles allow for long-term wear and good user compliance, challenges remain in ensuring consistent skin–sensor contact and stable signals under repeated deformation, washing or uneven sweat distribution.29 Recently, epidermal patch or tattoo-based sensors have emerged as a promising alternative due to their ultrathin, flexible and skin-conforming nature. This ensures intimate adhesion and reduced motion-induced noise, leading to enhanced signal stability. These sensors are usually made using inexpensive, large-scale screen-printing techniques on flexible materials such as polyethylene, polydimethylsiloxane, polyimide, polyurethane or paper. Their ease of application and removal, combined with the ability to acquire high-quality signals, makes them particularly suitable for reliable on-body sweat pH monitoring. However, long-term adhesion and skin compatibility must still be carefully considered.30Recent advances in materials science have driven the development of wearable sweat sensors with enhanced reliability and sensitivity. The incorporation of nanomaterials has been particularly influential in enhancing electrochemical and optical transduction. Their unique physicochemical properties, including high surface-to-volume ratios and superior charge-transfer capabilities, enable faster response times and lower detection limits. These advancements collectively contribute to the realization of highly efficient, flexible, and durable platforms for continuous, non-invasive health monitoring (Scheme 1).
2.4 Electronic processing, calibration, and data transmission
The electronic component is a critical part of wearable sweat pH sensing systems, as it enables signal acquisition and transmission, as well as real-time data analytics and long-term data management. Electrical signals generated in response to variations in hydrogen ion concentration are amplified and filtered first to suppress noise and motion-related artefacts, ensuring stable and accurate measurements. Embedded processing units implement adaptive calibration algorithms that compensate for environmental factors, such as temperature and humidity fluctuations, and sensor drift during prolonged operation. This maintains the reliability of the data over continuous monitoring periods. Real-time sensing is achieved by integrating flexible, miniaturised electronic circuit boards that conform to the non-planar, dynamic surface of the skin to ensure stable skin–sensor contact. Beyond on-device processing, wearable systems increasingly rely on wireless data transmission via bluetooth,20 radio-frequency identification (RFID) or near field communication (NFC) to stream data to external devices and cloud-based platforms.31–33 Cloud-based data processing enables the scalable storage of large datasets generated during long-term monitoring, as well as real-time visualisation and advanced analytics, including trend analysis, anomaly detection and user-specific baseline adaptation. Combining on-device signal conditioning with cloud-supported analytics facilitates continuous, real-time sweat pH monitoring and supports large-scale data aggregation for population-level studies and remote healthcare applications. This hybrid edge–cloud architecture enhances device portability and user comfort while enabling data-driven, personalised health monitoring.3 Wearable sensors for sweat pH monitoring
3.1 Electrochemical sensor
Recent progress in wearable sensors has led to the development of highly promising platforms for non-invasive and real-time health monitoring.34 These devices combine affordability and high analytical performance, making them suitable for continuous disease tracking. Among them, wearable electrochemical pH sensors for sweat analysis are commonly based on either potentiometric principles or ion-sensitive field-effect transistors (ISFETs).35 Compared to conventional pH sensing technologies, these systems offer superior stability, high sensitivity, rapid response, low power consumption, as well as the possibility of integration into wearable devices for continuous sweat pH monitoring. Their compact design also facilitates seamless integration into wireless and flexible wearable systems, supporting long-term and continuous physiological monitoring.A variety of potentiometric sensors have demonstrated considerable promise for non-invasive and real-time monitoring of sweat pH, along with numerous advancements in wearable technologies. For example, Yuhan Zhao et al. developed a flexible sweat pH sensor based on a two-electrode system fabricated on a polyimide (PI) substrate. The sensor integrates a three-dimensional porous polyaniline (3D PANI) working electrode onto an interdigital gold electrode (IGE), paired with a printed Ag/AgCl reference electrode. The reference electrode is formed by depositing a silver layer directly onto the same flexible substrate, then chemically chloridizing it to produce a stable Ag/AgCl interface. The polyimide substrate (PI) exhibits exceptional mechanical flexibility, positioning the sensor as a promising tool for real-time monitoring of sweat pH in medical care under-exercising (Fig. 3a). The open-circuit potential (OPC) was recorded by immersing 3D PANI sensors in various pH levels of artificial sweat (ranging from 4 to 9) under both normal and bent conditions (Fig. 3b). The proposed 3D PANI sensor exhibited exceptional sensitivity (69.33 mV pH−1), selectivity, a rapid response time (7.75 s), commendable reversibility, a low drift rate (0.10 mV h−1), minimal temperature drift (0.06 mV per pH per oC), and high repeatability (0.44% RSD) along with reproducibility (1.02% RSD). The reported sensitivity slightly exceeds the theoretical Nernstian value, which may be attributed to the redox-active nature and high surface area of the 3D PANI architecture, as well as interfacial charge storage effects. To asses the accuracy of the prepared 3D PANI sensor for detecting sweat pH, both the developed sensor and a commercial pH meter were used to measure the pH levels of five unknown pH artificial sweat samples. The findings show strong correlation in the recorded pH values between the 3D PANI sensor and the commercial pH meter (Fig. 3b).41 In a further study, Zhenying Xu et al. have developed a non-invasive wearable sweat sensing patch for simultaneous monitoring of pH and tyrosine (Tyr) levels in human sweat. This sensor consists of two parts. The first part is the electrochemical sensing system, which is based on screen printed three-electrode configuration. This setup includes an Ag/AgCl reference electrode, a carbon counter electrode, and a carbon working electrode that has been modified with a tannic acid-Ag-carbon nanotube-polyaniline (TA-Ag-CNT-PANI) composite through using the drop casting method. The second part is an iontophoretic (IP) electrode, used for direct sweat stimulation (Fig. 3c). Agarose gels were used on both cathode and anodic side, which were preloaded with a 3% pilocarpine nitrate solution. Upon energizing of the system, the positively charged pilocarpine drug is effectively driven across the skin to stimulate sweat secretion (Fig. 3d). The open circuit potential (OCP) was recorded within the pH range of 8.09 to 3.98, with a sensitivity determined to be −71.86 mV pH−1 (Fig. 3e). Such super-Nernstian behavior may result from synergistic effects between the conducting polymer and nanocomposite components, as well as non-equilibrium measurement conditions. The specificity was evaluated towards various ions present in human sweat, namely, NaCl, KCl, NH4Cl and CaCl2. The sensor response for the above ions was much lower than for H+ ions, confirming the sufficiently high specificity of the developed pH sensors. The stability of the device was assessed over a 20 days storage period, retained nearly 97.7% of its initial response. For the real sweat pH measurement, the integrated electrochemical sensors were attached to the forearms of six volunteers to accurately measure the sweat pH values. The results prove that the pH values of the sweat measured with the proposed sensor were comparable to those collected with a conventional pH meter.42 In addition, R. Liang et al. reported a Ti3C2Tx-based potentiometric pH sensor for sweat analysis. The Ti3C2Tx was synthesized using two etching methods: a mild LiF/HCl mixture and a HF solution, yielding pH-sensitive materials MILD-Ti3C2Tx and HF-Ti3C2Tx, respectively, both showing a lamellar structure (Fig. 3f). These pH-sensitive materials were used to functionalize the GCE and the electromotive force (EMF) was recorded over a pH range of 1–11. The results showed exceptional performance for HF-Ti3C2Tx in terms of sensitivity (−43.51 ± 0.53 mV pH−1), selectivity and reversibility (Fig. 3g and h). Although the sensitivity is slightly below the ideal Nernstian slope, the sensor demonstrates good linearity and stability over a wide pH range, which is advantageous for practical applications. Therefore, HF-Ti3C2Tx was selected as the ion-sensitive material for the fabrication of a flexible pH sensor on a polyethylene terephthalate (PET) substrate with an Ag/AgCl reference electrode (Fig. 3i). The developed flexible sensor was worn on the forehead of a healthy male volunteer for real-time monitoring of sweat pH (Fig. 3j and k). The recorded values were compared with those obtained using a conventional pH strip, showing comparable measurements (Fig. 3l).43 Table 1 illustrate some other example of wearable electrochemical sweat pH sensors.
 | ||
| Fig. 3 Potentiometric sweat pH sensors (a) schematic diagram of the fabrication process of flexible 3D PANI sensor; (b) response of 3D PANI sensor with increasing pH levels under mechanically normal and bending states; comparison between 3D PANI sensor with a commercial pH meter (Reproduced from ref. 41 with permission from Elseiver, Y. Zhao et al., Microchemical Journal, 2023, copyright 2023); (c) schematic illustration of the wearable sensor; (d) photograph of the sweat generated through the stimulation by the IP electrode; (e) the linear calibration curve of the OCP response versus pH (insert: OCP responses of the sensor in the pH range from 8.09 to 3.98) (Reproduced from ref. 42 with permission from Elseiver, Z. Xu et al., Biosensors and Bioelectronics, 2023, copyright 2023); (f) schematic illustration of the synthesis of Ti3C2Tx by two etching methods and SEM images of MILD-Ti3C2Tx and HF-Ti3C2Tx; (g) examination of pH reversible responses for MILD-Ti3C2Tx and HF-Ti3C2Tx; (h) pH response calibration curves; (i) schematic fabrication of flexible pH sensor; (j) photograph illustrates the on-body test of sweat pH monitoring during outdoor running; (k) on-body test pH analysis by the device; (l) comparison of sweat pH measured by HF-Ti3C2Tx-based pH sensor and pH meter (Reproduced from ref. 43 under the terms of the Creative Commons CC-BY License, published by MDPI, 2023). | ||
| pH-sensitive material | pH range | Sensitivity (mV pH−1) | Stability | Response time | Wearable format | Ref |
|---|---|---|---|---|---|---|
| PEDOT:BTB | 4–7 | 13 10−3 | — | — | Headband | 44 |
| Iridium oxide (IrOx) | 4–8 | 71.90 | — | 90 s | Patch | 45 |
| Iridium oxide | 4–8 | −47.54 | — | — | Textile | 46 |
| PANI/CNT/gold nanoshee | 4–8 | 71.44 | 10 days | — | Patch | 47 |
| Graphite-polyurethane (G-PU) | 6–9 | 4 | 2000 s | 5 s | Textile | 48 |
| Polyaniline/polyurethane | 2–7 | −60 | 50 min | 10 s | Electrode chip | 49 |
| Polyaniline/gold fibers | 4–8 | 60.60 | 8000 s | — | Textile | 50 |
| Iridium oxide | 3–8 | 79 | 2 h | 60 s | Patch | 51 |
| PANI/rGO/ITO | 2–8 | 62.30 | — | 50 s | — | 52 |
| F-Ti3C2Tx/PAN | 1–11 | −40.70 | — | — | Patch | 53 |
 | ||
| Fig. 4 ISFET based sensors for sweat pH monitoring (a) a schematic of a wearable device integrating flexible pH and temperature sensors; (b) a photograph of the fabricated device; (c) optical micrograph of the ISFET component prior to covering with polyimide; (d) photograph showing attachment of the flexible pH and temperature sensors to the test subject's neck, and (e) real-time pH and skin temperature acquired by the device. Red and blue dots represent the control experiment data for pH and skin temperature, respectively, measured using commercially available pH and IR sensors (Reproduced from ref. 54 with permission from American Chemical Society, S. Nakata et al., ACS Sensors, 2017, copyright 2017). | ||
3.2 Optical sensor
Wearable optical sensors represent a widely used technology for measuring sweat pH, and they provide exceptional rapidity and a non-invasive approach.57 Optical sensors offer several notable advantages over electrochemical sensors for detecting analytes in complex biological fluids, such as sweat. Unlike electrochemical systems, which require direct electrical contact and are susceptible to signal drift or electrode fouling, optical sensors typically rely on visible color changes in response to pH fluctuations. This allows for label-free detection, eliminating the need for electronic signal processing and making them highly compatible with low-power, wearable or disposable formats. Furthermore, recent advances in smartphone-based optical analysis have significantly improved the quantitative performance of these sensors, closing the accuracy and temporal resolution gap with electrochemical technologies. Taken together, these features make optical pH sensors promising tools for next-generation, non-invasive, continuous sweat pH monitoring applications.58In recent years, a range of optical techniques has been explored for wearable sweat pH monitoring, including surface-enhanced Raman scattering (SERS), fluorescence-based sensing, and colorimetric detection. These approaches have attracted considerable attention, owing to their non-invasive nature, rapid response, and strong compatibility with flexible and skin-conformal platforms.
Colorimetric sweat sensors are currently the most suitable for continuous and wearable pH monitoring, owing to their low cost, light weight, mechanical robustness, and minimal power requirements, as they avoid complex electronic circuits for data acquisition and transmission. These advantages make them particularly attractive for portable and commercial wearable platforms. However, their limitations include moderate sensitivity, discrete data output, and reduced temporal resolution, which may affect precise quantitative tracking.
In contrast, SERS-based sensors offer unmatched analytical performance, including label-free detection, ultra-high sensitivity, and excellent molecular specificity, even at very low analyte volumes. Despite these strengths, their reliance on external Raman spectrometers, high cost, and limited repeatability currently restrict their applicability to laboratory-based or intermittent measurements rather than long-term, autonomous wearable monitoring.
Fluorescence-based sensors provide high sensitivity, good selectivity, and rapid response, enabling the detection of small pH variations within the physiological sweat range. Nevertheless, their dependence on excitation light sources, optical filters, and signal acquisition electronics leads to higher power consumption and susceptibility to ambient light interference, which complicates long-term wearable implementation.25,59
Overall, while SERS and fluorescence-based approaches demonstrate superior sensitivity and selectivity, colorimetric sensors remain the most practical solution for continuous, low-power, and user-friendly wearable sweat pH monitoring, whereas SERS and fluorescence platforms are better suited for high-precision or validation-focused applications. Table 2 provides a comparative summary of reported optical sensors for wearable sweat pH monitoring. The following sections provide a detailed discussion of these optical sensing strategies, highlighting representative studies and recent advances in wearable sweat pH sensing technologies.
| Sensor type | Sensing mechanism | Advantages | Limitations |
|---|---|---|---|
| SERS | Raman spectral shift | Label-free detection; ultra-high sensitivity; excellent molecular specificity; minimal sample volume | Requires Raman spectrometer; high cost; limited portability; not suitable for long-term continuous monitoring |
| Fluorescence | Fluorescence intensity/wavelength shift | High sensitivity and selectivity; rapid response; capable of detecting small pH variations | Requires excitation source and optical filters; higher power consumption; susceptible to ambient light interference |
| Colorimetric | pH-responsive dyes | Low cost; lightweight; simple readout; no complex electronics; suitable for continuous and outdoor monitoring | Moderate sensitivity; discrete data; limited temporal resolution; affected by lighting conditions |
In this context, W. Wang et al. introduced a flexible SERS-based wearable sensor capable of quantitative analysis of urea and uric acid, as well as real-time pH monitoring in sweat (Fig. 5a). The hydrogel was initially prepared by photo-cross linking of sulfonated cellulose silver nanocomposites (S-CNF-Ag NPs) with acrylic acid (AA). Subsequently, the material was functionalized with 4-mercaptobenzoic acid (4-MBA), which serves as a pH-sensitive probe molecule. As a results, the hydrogel utilized as a sensor patch, demonstrated excellent skin adaptability, biocompatibility, and antimicrobial properties (Fig. 5b). Furthermore, its superabsorbent nature facilitated rapid sweat sampling, thereby addressing the challenge of difficult sweat collection. The calibration curves were generated by plotting the intensity ratios of specific Raman bands: I1382 (v(COO–))/I1075 (v12 ring), I1382/I1586 (v8a ring) and I1718(v(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O))/I1075 against pH changes in the range of 4.10–7.60 (Fig. 5c). Significantly, the S-CNF-Ag NPs/PAA/4-MBA hydrogel patch was tested on the forearms of six volunteers one hour post-running. The pH values measured by the SERS sensor were comparable with those obtained using a commercially available pH meter. This result highlights the suitability of the S-CNF-Ag NPs/PAA/4-MBA wearable sensor for sweat monitoring. Furthermore, the developed sensor demonstrated excellent reproducibility, stability, and mechanical durability throughout the detection process.64 In a similar vein, M. Chung et al. developed a mechanically flexible surface-enhanced Raman scattering (SERS)-active substrate by electrospinning thermoplastic polyurethane nanofibres, followed by gold sputtering (Au/TPU). The substrate was functionalized with two widely used pH-responsive molecules, 4-mercaptobenzoic acid (4-MBA) and 4-mercaptopyridine (4-MPy), in order to evaluate its applicability for wearable sweat pH sensing. The functionalized substrates (4-MBA/Au/TPU and 4-MPy/Au/TPU) were then incorporated into a transparent, adhesive dermal patch (see Fig. 5d). This ensured strong adhesion to the skin, protection against contaminants and inhibition of fast sweat evaporation (Fig. 5e). The SERS spectra of Au/TPU functionalized with 4-MBA and 4-MPy were recorded across a pH range of 4.25 to 7.5 using a Raman spectroscope with 785 nm laser excitation (Fig. 5f–h). The accuracy of the reported sensor for sweat pH sensing was evaluated using eccrine sweat samples collected from two individuals following 30 minutes of exercise (Fig. 5i). The proposed SERS-based pH sensor exhibited precise pH measurements with high resolution using only 1 µL of sweat, underscoring its potential for wearable sweat sensing applications. The sensor exhibits several advantageous characteristics, including mechanical stability, simple and cost-effective fabrication process, and the ability to be stored dry at room temperature while maintaining excellent accuracy, repeatability, and reversibility. However, the Au/TPU NF SERS substrate is currently limited to single-use sweat pH sensing and requires further development for continuous monitoring. The objective can be achieved through integrating microfluidic channels with an iontophoresis device to stimulate localized sweating and direct the collected sweat to the sensing surface, thereby enhancing the sensor's functionality and usability.65 C. D'Andrea et al. have developed a wearable sweat sensor chip based on surface-enhanced Raman spectroscopy (SERS) for the detection of urea and lactate, in addition to assessing of the pH level of sweat solutions. The core of the sweat pH sensor chip was fabricated by initially depositing silver nanowires onto a porous membrane comprising polytetrafluoroethylene (AgNWs@PTFE). This was followed by functionalizing the membrane with a pH-sensitive probe molecule (4-mercaptobenzoic acid (4-MBA)) which exhibits a SERS spectral signature that varies with pH levels ranging from 1 to 12 (Fig. 5j). Subsequently, the SERS chip was integrated into a biocompatible tape sandwich consisting of Tegaderm™ and Leukomed® T, which serve as protective and adhesive layers, respectively (Fig. 5k). The primary benefit of this design is that it requires only a small quantity of sweat, around tens of microliters to traverse the hole in the PTFE membrane and the Leukomed® T tape, where it is subsequently adsorbed onto the sensing area, therefore eliminating the need for microfluidic channels. Fig. 5l and m present the SERS spectra and the corresponding pH calibration curve, respectively, with the latter obtained by normalizing the area of the carboxylate bands (1400–1425 cm−1) to the benzene ring peak of 4-MBA at 1592 cm−1. The sensor was tested with three custom-simulated sweat solutions of known pH values, and the results are represented in Fig. 5m as geometric symbols (red diamond, blue square, and purple circle) corresponding to pH level of 4.8, 6.6, and 9.0, respectively, and the results fall within the 95% prediction interval. The developed SERS-active chip is a cost-effective and scalable sensing device, priced below €5, at least three times cheaper than commercially available SERS substrates, and can be produced through time-efficient processes.66
O))/I1075 against pH changes in the range of 4.10–7.60 (Fig. 5c). Significantly, the S-CNF-Ag NPs/PAA/4-MBA hydrogel patch was tested on the forearms of six volunteers one hour post-running. The pH values measured by the SERS sensor were comparable with those obtained using a commercially available pH meter. This result highlights the suitability of the S-CNF-Ag NPs/PAA/4-MBA wearable sensor for sweat monitoring. Furthermore, the developed sensor demonstrated excellent reproducibility, stability, and mechanical durability throughout the detection process.64 In a similar vein, M. Chung et al. developed a mechanically flexible surface-enhanced Raman scattering (SERS)-active substrate by electrospinning thermoplastic polyurethane nanofibres, followed by gold sputtering (Au/TPU). The substrate was functionalized with two widely used pH-responsive molecules, 4-mercaptobenzoic acid (4-MBA) and 4-mercaptopyridine (4-MPy), in order to evaluate its applicability for wearable sweat pH sensing. The functionalized substrates (4-MBA/Au/TPU and 4-MPy/Au/TPU) were then incorporated into a transparent, adhesive dermal patch (see Fig. 5d). This ensured strong adhesion to the skin, protection against contaminants and inhibition of fast sweat evaporation (Fig. 5e). The SERS spectra of Au/TPU functionalized with 4-MBA and 4-MPy were recorded across a pH range of 4.25 to 7.5 using a Raman spectroscope with 785 nm laser excitation (Fig. 5f–h). The accuracy of the reported sensor for sweat pH sensing was evaluated using eccrine sweat samples collected from two individuals following 30 minutes of exercise (Fig. 5i). The proposed SERS-based pH sensor exhibited precise pH measurements with high resolution using only 1 µL of sweat, underscoring its potential for wearable sweat sensing applications. The sensor exhibits several advantageous characteristics, including mechanical stability, simple and cost-effective fabrication process, and the ability to be stored dry at room temperature while maintaining excellent accuracy, repeatability, and reversibility. However, the Au/TPU NF SERS substrate is currently limited to single-use sweat pH sensing and requires further development for continuous monitoring. The objective can be achieved through integrating microfluidic channels with an iontophoresis device to stimulate localized sweating and direct the collected sweat to the sensing surface, thereby enhancing the sensor's functionality and usability.65 C. D'Andrea et al. have developed a wearable sweat sensor chip based on surface-enhanced Raman spectroscopy (SERS) for the detection of urea and lactate, in addition to assessing of the pH level of sweat solutions. The core of the sweat pH sensor chip was fabricated by initially depositing silver nanowires onto a porous membrane comprising polytetrafluoroethylene (AgNWs@PTFE). This was followed by functionalizing the membrane with a pH-sensitive probe molecule (4-mercaptobenzoic acid (4-MBA)) which exhibits a SERS spectral signature that varies with pH levels ranging from 1 to 12 (Fig. 5j). Subsequently, the SERS chip was integrated into a biocompatible tape sandwich consisting of Tegaderm™ and Leukomed® T, which serve as protective and adhesive layers, respectively (Fig. 5k). The primary benefit of this design is that it requires only a small quantity of sweat, around tens of microliters to traverse the hole in the PTFE membrane and the Leukomed® T tape, where it is subsequently adsorbed onto the sensing area, therefore eliminating the need for microfluidic channels. Fig. 5l and m present the SERS spectra and the corresponding pH calibration curve, respectively, with the latter obtained by normalizing the area of the carboxylate bands (1400–1425 cm−1) to the benzene ring peak of 4-MBA at 1592 cm−1. The sensor was tested with three custom-simulated sweat solutions of known pH values, and the results are represented in Fig. 5m as geometric symbols (red diamond, blue square, and purple circle) corresponding to pH level of 4.8, 6.6, and 9.0, respectively, and the results fall within the 95% prediction interval. The developed SERS-active chip is a cost-effective and scalable sensing device, priced below €5, at least three times cheaper than commercially available SERS substrates, and can be produced through time-efficient processes.66
 | ||
| Fig. 5 SERS based sensors for sweat pH quantification (a) SERS based sensor for urea and uric acid detection and pH change in sweat; (b) mechanism diagram of hydrogel adhesion to skin; (c) S-CNF-Ag NPs/PAA/4-MBA sensor's SERS spectra at different pH values (Reproduced from ref. 64 with permission from Elseiver, W. Wang et al., Chemical Engineering Journal, 2023, copyright 2023); (d) attachment of SERS-active Au/TPU electrospun fibers to a transparent adhesive dermal patch; (e) attachment of wearable pH sensor to the subject's arm; (f) Raman spectroscopy performed ; (g) SERS spectra of 4-MBA on Au/TPU nanofibers within the sweat pH range, normalized to reference peak, v(ref); (h) SERS spectra of 4-MPy on Au/TPU nanofibers within the sweat pH range, in this case normalized to reference peak, v(1095) ; (i) wearable sweat SERS pH sensing patch on human subject arm (Reproduced from ref. 65 with permission from the American Chemical Society, M. Chung et al., ACS Applied Materials & Interfaces, 2021, copyright 2021); (j) picture of a patterned AgNWs layer deposited on a hydrophobic PTFE filter membrane; (k) schematic representation of a wearable SERS-active chip; (l) SERS spectra of 4-MBA on AgNWs@PTFE at different pH values; (m) calibration curve (Reproduced from ref. 66 under the terms of the Creative Commons CC-BY License, published by the European Optical Society, 2024). | ||
 | ||
| Fig. 6 Fluorescent sweat pH sensors emission fluorescence intensity change in response to pH of (a) the anionic nanotube complexes and (b) polymer-coated anionic surfactant SWCNTs; (c) optical responses from PDADMAC-SDBS-SWCNTs in artificial sweat at various pH ranges (pH 2–4.5) and pH response curve (Reproduced from ref. 16 under the terms of the Creative Commons CC-BY License, published by the American Chemical Society, 2023); (d) schematic representation for the fabrication of the skin-mounted sweat sensor sticker (SSS); (e) (A) images of the prototype of the developed smart wearable IoT-enabled imaging module (SIMA) and its components; (f) calibration curve and the corresponding images for the fluorescence determination of pH in artificial sweat using the developed smart wearable optical sensor (SWOS); (g) human experiments conducted on athletic volunteers during exercise (Reproduced from ref. 68 with permission from American Chemical Society, A. R. Sharifi et al., Analytical Chemistry, 2023, copyright 2023); (h and i) optical photo of FHSB-GQD-PVA-PG film under 490 nm laser irradiation; (j) fluorescence spectra of FHSB-GQD-PVA-PG film at different pH stimulations, and plot of the peak fluorescence intensity vs. pH; (k) wearable device attached to a runner's arm during physical activity (Reproduced from ref. 69 with permission from Elseiver, X. Qingqing et al., Analytica Chimica Acta ,2024, copyright 2024). | ||
 | ||
| Fig. 7 Wearable colorimetric sweat pH sensor (a) photo of the optical sensing patch system developed; (b) absorption spectrum of the pH sensitive film on a glass slide with pH value of the test liquid as a variable; (c) real-time on-body monitoring of physiological parameters during cycling exercise (Reproduced from ref. 72 with permission from IEEE, G. Wang et al., IEEE Trans Biomed Eng, 2019, copyright 2019); (d) design of a thread/paper-based wearable system; (e) plots of G value against pH value of the artificial sweat. Insets show the digital images of the thread/paper-based pH sensors after reacting with artificial sweats at different pH values; (f) an arm guard integrated device after the on body measurement (Reproduced from ref. 55 under the terms of the Creative Commons CC-BY License, published by MDPI, 2010); (g) schematic diagram of the sensing procedure; (h) calibration curve of the sensor; (i) real-time results of the wearable system placed on the wristband during exercise (Reproduced from ref. 20 with permission from Elseiver, P. Escobedo et al., Sensors and Actuators B: Chemical, 2021, copyright 2021); (j) C-TPU pH sensors attached to the cloth; (k) the color of the C-TPU patch was recovered by washing in a washing machine with a neutral detergent; (l) C-TPU based colorimetric sensor attached to a baby cloth (Reproduced from ref. 74 under the terms of the Creative Commons CC-BY License, published by Elseiver, 2023). | ||
In 2021, P. Escobedo et al. proposed a wearable wristband gadget designed for continuous sweat pH monitoring, emphasizing the importance of collecting long-term health data. The design consists of several components including a sampling area using a microfluidic cloth analytical device (µCAD), for continuous sweat collection, a sensing module featuring an excitation LED and a colour sensor, positioned in front of a pH-sensitive membrane (vinyl sulfone acidochromic dye (AD-VS-1)) coated on a cotton cloth, a passive pumping path facilitating sample flows to an absorbent U-shaped material for extended operation (exceeding 1000 minutes), and a miniaturized readout module with a low-power Bluetooth interface for real time transmission of measurements to a custom-designed smartphone application. Based on their results, the Hue (H) parameter in the HSV color space has been identified as being related to the pH of sweat. The wristband gadget operates within a range of pH from 6 to 8, covering the pH range of sweat. The system was also validated in real-world conditions by being worn on the wrist during physical activity.20 J. H. Ha et al. have recently developed a wearable colorimetric sweat pH sensor based on curcumin and thermoplastic polyurethane (C-TPU) electrospun fiber, seamlessly integrated into smart textiles. This innovative technology is designed to track the health status of cystic fibrosis (CF) patients. The C-TPU fibers display a visually discernible shift in color as the sweat pH rises from a neutral control (pH 7) to pH 10, and the initial color of the C-TPU fibers can be restored through multiple washing cycles with a neutral detergent. This reusable sensor enables continuous diagnosis of CF patients while maintaining optimal performance. For real-time measurements, a clover-shaped C-TPU pH sensor was attached to baby swaddles and patient clothing to demonstrate the capabilities of wearable applications (Fig. 7l).74
4 Future perspectives and conclusions
Wearable sweat pH sensors have emerged as promising tools for non-invasive, continuous health monitoring, enabling real-time assessment of physiological and pathological states. Recent advances in materials science and nanotechnology have enhanced sensor flexibility, biocompatibility, and the integration of sensing components into skin-conformal platforms. Despite these improvements, translating these devices into practical healthcare tools requires overcoming several critical challenges.Long-term operational stability and selectivity in complex, variable sweat environments remain major concerns, particularly when sensors are exposed to mechanical stress, temperature fluctuations, or extended wear. Addressing these challenges demands the development of robust sensing materials with antifouling surfaces, mechanical resilience, and self-healing properties. Strategies such as self-calibration and energy-efficient or self-powered operation can further ensure consistent performance during prolonged use.
Seamless integration into wearable formats also requires flexible, stretchable electronics and modular architectures that maintain sensor functionality under repeated deformation. In parallel, scalable fabrication methods and standardized protocols are essential to produce reliable sensors at a commercial scale while meeting regulatory and safety requirements. Additionally, coupling sensor data with machine learning and AI algorithms can enhance personalized diagnostics, compensate for environmental and physiological variations, and enable predictive health monitoring.
By combining materials innovation, sensor engineering, electronics design, and digital health technologies, future wearable sweat pH sensors can achieve durable, reliable, and user-friendly performance. Such multidisciplinary approaches will be crucial for translating laboratory advances into practical, real-world solutions that support continuous monitoring, early disease detection, and personalized preventive medicine.
Conflicts of interest
There are no conflitcts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This research was supported by the Tunisian Ministry of Higher Education and Scientific Research, Tunisia.References
- S. Baliga, S. Muglikar and R. Kale, Salivary pH: A diagnostic biomarker, J. Indian Soc. Periodontol., 2013, 17, 461–465, DOI:10.4103/0972-124X.118317.
- N. Nakanishi, et al., Low urine pH is a predictor of chronic kidney disease, Kidney Blood Press. Res., 2012, 35, 77–81, DOI:10.1159/000330487.
- Y. He, et al., Association of acidic urine pH with impaired renal function in primary gout patients: a Chinese population-based cross-sectional study, Arthritis Res. Ther., 2022, 24, 1–12, DOI:10.1186/s13075-022-02725-w.
- F. Grases, A. Costa-Bauzá, I. Gomila, M. Ramis, A. García-Raja and R. M. Prieto, Urinary pH and renal lithiasis, Urol. Res., 2012, 40, 41–46, DOI:10.1007/s00240-011-0389-3.
- V. M. Buckalew, D. K. McCurdy, G. D. Ludwig, L. B. Chaykin and J. R. Elkinton, Incomplete renal tubular acidosis: Physiologic studies in three patients with a defect in lowering urine pH, Am. J. Med., 1968, 45, 32–42, DOI:10.1016/0002-9343(68)90005-3.
- C. Seethalakshmi, R. C. Jagat Reddy, N. Asifa and S. Prabhu, Correlation of salivary pH, incidence of dental caries and periodontal status in diabetes mellitus patients: A cross-sectional study, J. Clin. Diagn. Res., 2016, 10, ZC12–ZC14, DOI:10.7860/JCDR/2016/16310.7351.
- C. A. Eckley and H. O. Costa, Comparative study of salivary pH and volume in adults with chronic laryngopharyngitis by gastroesophageal reflux disease before and after treatment, Braz. J. Otorhinolaryngol., 2006, 72, 55–60, DOI:10.1016/s1808-8694(15)30035-5.
- S. H. Kuo, C. J. Shen, C. F. Shen and C. M. Cheng, Role of pH value in clinically relevant diagnosis, Diagnostics, 2020, 10, 107, DOI:10.3390/diagnostics10020107.
- L. Manjakkal, S. Dervin and R. Dahiya, Flexible potentiometric pH sensors for wearable systems, RSC Adv., 2020, 10, 8594–8617 RSC.
- M. Cuartero, M. Parrilla and G. A. Crespo, Wearable potentiometric sensors for medical applications, Sensors, 2019, 19, 1–24, DOI:10.3390/s19020363.
- J. D. Cook, K. A. Strauss, Y. H. Caplan, C. P. LoDico and D. M. Bush, Urine pH: The effects of time and temperature after collection, J. Anal. Toxicol., 2007, 31, 486–496, DOI:10.1093/jat/31.8.486.
- F. G. Bellagambi, et al., Saliva sampling: Methods and devices. An overview, TrAC, Trends Anal. Chem., 2020, 124, 1–48, DOI:10.1016/j.trac.2019.115781.
- J. Pieczyński, U. Szulc, J. Harazna, A. Szulc and J. Kiewisz, Tear fluid collection methods: Review of current techniques, Eur. J. Ophthalmol., 2021, 31, 2245–2251, DOI:10.1177/1120672121998922.
- H. Y. Y. Nyein, et al., A Wearable Electrochemical Platform for Noninvasive Simultaneous Monitoring of Ca2+ and pH, ACS Nano, 2016, 10, 7216–7224 CrossRef CAS PubMed.
- M. H. Schmid-Wendtner and H. C. Korting, The pH of the skin surface and its impact on the barrier function, Skin Pharmacol. Physiol., 2006, 19, 296–302, DOI:10.1159/000094670.
- N. Sultana, H. Dewey and J. Budhathoki-Uprety, Optical detection of pH changes in artificial sweat using near-infrared fluorescent nanomaterials, Sens. Diagn., 2022, 1, 1189–1197 RSC.
- W. Dang, L. Manjakkal, W. T. Navaraj and L. Lorenzelli, Stretchable Wireless System for Sweat pH Monitoring, Biosens. Bioelectron., 2018, 107, 192–202, DOI:10.1016/j.bios.2018.02.025.
- M. Caldara, C. Colleoni, E. Guido, G. Rosace, V. Re, and A. Vitali, A wearable sensor platform to monitor sweat pH and skin temperature, in. 2013 IEEE International Conference on Body Sensor Networks, 2013, pp. 1–6, DOI:10.1109/BSN.2013.6575465.
- S. Coyle, et al., Textile sensors to measure sweat pH and sweat-rate during exercise, in. 2009 3rd International Conference on Pervasive Computing Technologies for Healthcare, 2009, pp. 1–6, DOI:10.4108/ICST.PERVASIVEHEALTH2009.5957.
- P. Escobedo, et al., Wireless wearable wristband for continuous sweat pH monitoring, Sensor. Actuator. B Chem., 2021, 327, 1–16 CrossRef.
- Y. Xia, et al., Wearable electrochemical sensor based on bimetallic MOF coated CNT/PDMS film electrode via a dual-stamping method for real-time sweat glucose analysis, Anal. Chim. Acta, 2023, 1278, 341754, DOI:10.1016/J.ACA.2023.341754.
- M. G. Bruno, et al., Wearable sensor for real-time monitoring of oxidative stress in simulated exhaled breath, Biosens. Bioelectron. X, 2024, 100476 CAS.
- E. A. Choi, et al., Noninvasive wearable sensor for the continuous monitoring of human sound and movement signals in real-time, Heliyon, 2024, 10, e26307 CrossRef PubMed.
- L. Donaldson, Wearable sweat sensor for healthcare monitoring, Mater. Today, 2022, 52, 1–2 Search PubMed.
- J. Min, et al., Skin-Interfaced Wearable Sweat Sensors for Precision Medicine, Chem. Rev., 2023, 123, 5049–5138, DOI:10.1021/acs.chemrev.2c00823.Skin-Interfaced.
- A. M. V. Mohan, V. Rajendran, R. K. Mishra and M. Jayaraman, Recent advances and perspectives in sweat based wearable electrochemical sensors, TrAC, Trends Anal. Chem., 2020, 131, 116024, DOI:10.1016/j.trac.2020.116024.
- R. Vinoth, T. Nakagawa, J. Mathiyarasu and A. M. V. Mohan, Fully Printed Wearable Microfluidic Devices for High-Throughput Sweat Sampling and Multiplexed Electrochemical Analysis, ACS Sens., 2021, 6, 1174–1186, DOI:10.1021/acssensors.0c02446.
- N. Fatin, et al., A Comprehensive Review of the Recent Developments in Wearable Sweat-Sensing Devices, Sensors, 2022, 22, 7670, DOI:10.3390/s22197670.
- A. Khan, et al., A review on advanced nanocomposites materials based smart textile biosensor for healthcare monitoring from human sweat, Sens. Actuators, A Phys., 2023, 350, 114093 CrossRef CAS.
- H. Li, Y. Xiao and L. Jin, Recent Development of Wearable Electrochemical Sensors for Sweat Analysis, Int. J. Electrochem. Sci., 2020, 15, 11997–12006 CrossRef CAS.
- M. S. P Kassal, M. Sigurnjak, G. Scheipl and G. J. Mohr, Wearable sensor with RFID/NFC wireless connectivity for optical monitoring of sweat pH, in. EUROPT (R) ODE XIV Naples 2018, XIV Conference on Optical Chemical Sensors and Biosensors, 2018, pp. , pp. 156 Search PubMed.
- A. B. Barba, G. M. Bianco, L. Fiore, and C. Occhiuzzi, Design and Manufacture of Flexible Epidermal NFC Device for Electrochemical Sensing of Sweat, in. 2022 IEEE International Conference on Flexible, Printable Sensors and Systems, 2022 Search PubMed.
- M. Boada, A. Lazaro, S. Member, R. Villarino, D. Girbau and S. Member, Battery-Less NFC Sensor for pH Monitoring, IEEE Access, 2019, 7, 33226–33239, DOI:10.1109/ACCESS.2019.2904109.
- A. J. Bandodkar and J. Wang, Non-invasive wearable electrochemical sensors: a review, Trends Biotechnol., 2014, 32, 363–371, DOI:10.1016/J.TIBTECH.2014.04.005.
- M. T. Ghoneim, et al., Recent Progress in Electrochemical pH-Sensing Materials and Configurations for Biomedical Applications, Chem. Rev., 2019, 119, 5248–5297, DOI:10.1021/acs.chemrev.8b00655.
- J. Hu, A. Stein and P. Bühlmann, Rational design of all-solid-state ion-selective electrodes and reference electrodes, TrAC, Trends Anal. Chem., 2016, 76, 102–114 CrossRef CAS.
- Y. Tang, et al., Recent Advances in Wearable Potentiometric pH Sensors, Membranes, 2022, 12, 1–20 Search PubMed.
- P. Kurzweil, Metal Oxides and Ion-Exchanging Surfaces as pH Sensors in Liquids: State-of-the-Art and Outlook, Sensors, 2009, 9, 4955–4985, DOI:10.3390/s90604955.
- O. Korostynska, K. Arshak, E. Gill and A. Arshak, Review on State-of-the-art in Polymer Based pH Sensors, Sensors, 2007, 7, 3027–3042 CrossRef CAS PubMed.
- E. Bilbao, O. Garate, T. Rodr, M. Roberti and M. Mass, Electrochemical Sweat Sensors, Chemosensors, 2023, 11, 244 CrossRef CAS.
- Y. Zhao, Y. Yu, S. Zhao, R. Zhu, J. Zhao and G. Cui, Highly sensitive pH sensor based on flexible polyaniline matrix for synchronal sweat monitoring, Microchem. J., 2023, 185, 108092, DOI:10.1016/J.MICROC.2022.108092.
- Z. Xu, et al., A wearable sensor based on multifunctional conductive hydrogel for simultaneous accurate pH and tyrosine monitoring in sweat, Biosens. Bioelectron., 2023, 234, 115360 CrossRef CAS.
- R. Liang, et al., Directly Using Ti3C2Tx MXene for a Solid-Contact Potentiometric pH Sensor toward Wearable Sweat pH Monitoring, Membranes, 2023, 13, 376, DOI:10.3390/membranes13040376.
- H. Yin, et al., A Wearable Electrochemical Platform for Noninvasive Simultaneous Monitoring of Ca 2+ and pH, ACS Nano, 2016, 10, 7216–7224, DOI:10.1021/acsnano.6b04005.
- S. Anastasova, et al., A wearable multisensing patch for continuous sweat monitoring, Biosens. Bioelectron., 2017, 93, 139–145 CrossRef CAS.
- M. L. Zamora, J. M. Dominguez, R. M. Trujillo, C. B. Goy, M. A. Sánchez and R. E. Madrid, Potentiometric textile-based pH sensor, Sens. Actuators, B, 2018, 260, 601–608 CrossRef CAS.
- S. Y. Oh, S. Y. Hong, Y. R. Jeong, J. Yun, H. Park and S. W. Jin, Skin-Attachable, Stretchable Electrochemical Sweat Sensor for Glucose and pH Detection, ACS Appl. Mater. Interfaces, 2018, 10, 13729–13740, DOI:10.1021/acsami.8b03342.
- L. Manjakkal, W. Dang, N. Yogeswaran and R. Dahiya, Textile-based potentiometric electrochemical PH sensor for wearable applications, Biosensors, 2019, 9, 1–12 CrossRef PubMed.
- X. Hou, Y. Zhou, Y. Liu, L. Wang and J. Wang, Coaxial electrospun flexible PANI//PU fibers as highly sensitive pH wearable sensor, J. Mater. Sci., 2020, 55, 16033–16047 CrossRef CAS.
- R. Wang, et al., Stretchable gold fiber-based wearable electrochemical sensor toward pH monitoring, J. Mater. Chem. B, 2020, 2, 3655–3660 RSC.
- V. Mazzaracchio, L. Fiore, S. Nappi, G. Marrocco and F. Arduini, Medium-distance affordable, flexible and wireless epidermal sensor for pH monitoring in sweat, Talanta, 2021, 222, 121502 CrossRef CAS PubMed.
- F. Mazzara, et al., Pani-based wearable electrochemical sensor for ph sweat monitoring, Chemosensors, 2021, 9, 1–14 CrossRef.
- L. Chen, et al., Superhydrophobic Functionalized Ti3C2Tx MXene-Based Skin-Attachable and Wearable Electrochemical pH Sensor for Real-Time Sweat Detection, Anal. Chem., 2022, 94, 7319–7328 CrossRef CAS PubMed.
- S. Nakata, T. Arie, S. Akita and K. Takei, Wearable, Flexible, and Multifunctional Healthcare Device with an ISFET Chemical Sensor for Simultaneous Sweat pH and Skin Temperature Monitoring, ACS Sens., 2017, 2, 443–448, DOI:10.1021/acssensors.7b00047.
- C. Jimenez-jorquera, J. Orozco and A. Baldi, ISFET Based Microsensors for Environmental Monitoring, Sensors, 2010, 10, 61–83, DOI:10.3390/s100100061.
- S. Cao and S. Zhao, ISFET-Based Sensors for (Bio) Chemical Applications : A Review, pp. 1–25, 2023, DOI:10.1002/elsa.202100207.
- N. Promphet, et al., Non-invasive textile based colorimetric sensor for the simultaneous detection of sweat pH and lactate, Talanta, 2019, 192, 424–430, DOI:10.1016/J.TALANTA.2018.09.086.
- N. Promphet, S. Ummartyotin and W. Ngeontae, Non-invasive wearable chemical sensors in real-life applications, Anal. Chim. Acta, 2021, 1179, 338643, DOI:10.1016/j.aca.2021.338643.
- Z. Z. Jing Wang, Y. Luo, J. Xiao, T. X. and X. Z. Zhang, Epidermal wearable optical sensors for sweat monitoring, Commun. Mater., 2024, 5, 77, DOI:10.1038/s43246-024-00518-z.
- P. Etchegoin, et al., New limits in ultrasensitive trace detection by surface enhanced Raman scattering (SERS), Chem. Phys. Lett., 2003, 375, 84–90, DOI:10.1016/S0009-2614(03)00821-2.
- Y. Kitahama, M. Egawa, P. K. Dwivedi, W. Yang and K. Goda, An emerging tool in healthcare: wearable surface-enhanced Raman Spectroscopy, J. Phys. Photonics, 2024, 6, 021001, DOI:10.1088/2515-7647/ad38f6.
- P. Polykretis, M. Banchelli, C. D'Andrea, M. de Angelis and P. Matteini, Raman Spectroscopy Techniques for the Investigation and Diagnosis of Alzheimer's Disease, Front. Biosci., 2022, 14, 22, DOI:10.31083/j.fbs1403022.
- M. Prochazka, Surface-Enhanced Raman Spectroscopy. 2016 Search PubMed.
- W. Wang, et al., Flexible SERS wearable sensor based on nanocomposite hydrogel for detection of metabolites and pH in sweat, Chem. Eng. J., 2023, 474, 145953, DOI:10.1016/J.CEJ.2023.145953.
- M. Chung, et al., Fabrication of a Wearable Flexible Sweat pH Sensor Based on SERS-Active Au/TPU Electrospun Nanofibers, ACS Appl. Mater. Interfaces, 2021, 13, 51504–51518 CrossRef CAS PubMed.
- C. D. Andrea, et al., Development of a wearable surface enhanced Raman scattering sensor chip based on silver nanowires for rapid detection of urea , lactate and pH in sweat, J. Eur. Opt. Soc., Rapid publ, 2024, 20, 1–7 Search PubMed.
- N. Gao, G. Xu, G. Chang and Y. Wu, From Lab to Life : Self-Powered Sweat Sensors and Their Future in Personal Health Monitoring, Adv. Sci., 2025, 12, 2409178, DOI:10.1002/advs.202409178.
- A. R. Sharifi, et al., Smart Wearable Nanopaper Patch for Continuous Multiplexed Optical Monitoring of Sweat Parameters, Anal. Chem., 2023, 95, 16098–16106, DOI:10.1021/acs.analchem.3c02044.
- X. Qingqing, L. Ruiyi and L. Zaijun, Designing of multifunctional graphene quantum dot-polyvinyl alcohol-polyglycerol luminescent film for fluorescence detection of pH in sweat, Anal. Chim. Acta, 2024, 1292, 342224 CrossRef PubMed.
- A. Koh, et al., A soft, wearable microfluidic device for the capture, storage, and colorimetric sensing of sweat, Sci. Transl. Med., 2016, 8, 1–14 Search PubMed.
- F. A. A. Nedal Abu-Thabit, Y. Umar, E. Ratemi and A. Ahmad, A Flexible Optical pH Sensor Based on Polysulfone Membranes Coated with pH-Responsive Polyaniline Nanofiber, Sensors, 2016, 16, 986, DOI:10.3390/s16070986.
- G. Wang, S. Zhang, S. Dong, D. Lou, L. Ma and X. Pei, Stretchable Optical Sensing Patch System Integrated Heart Rate , Pulse Oxygen Saturation and Sweat pH Detection, IEEE Trans. Biomed. Eng., 2019, 66, 1000–1005, DOI:10.1109/TBME.2018.2866151.
- G. Xiao, et al., Facile and Low-Cost Fabrication of a Thread/Paper-Based Wearable System for Simultaneous Detection of Lactate and pH in Human Sweat, Adv. Fiber Mater., 2020, 2, 265–278 CrossRef CAS.
- J. H. Ha, et al., A wearable colorimetric sweat pH sensor-based smart textile for health state diagnosis, Mater. Horiz., 2023, 10, 4163–4171 RSC.
| This journal is © The Royal Society of Chemistry 2026 |