Open Access Article
Open Access ArticleSynthesis of siloxane-integrated Schiff base compounds exhibiting aggregation-induced emission properties
Yuling Li a,
Depeng Mab,
Fanzhen Kongc,
Yuanrong Wangd,
Lianfeng Wuef,
Zhihui Yangfgh and
Haifeng Lu*fgh
a,
Depeng Mab,
Fanzhen Kongc,
Yuanrong Wangd,
Lianfeng Wuef,
Zhihui Yangfgh and
Haifeng Lu*fgh
aSchool of Engineering Science, Shandong Xiehe University, Jinan, 250109, P. R. China
bSchool of Radiology, Shandong First Medical University, Shandong Academy of Medical Sciences, Taian, Shandong Province 271016, P. R. China
cSICO Performance Material (Shandong) Co., Ltd, Jining New Materials Industrial Park, Jining, Shandong Province 272200, P. R. China
dChambroad Chemical Industry Research Institute Co., Ltd, Binzhou, Shandong Province 251700, P. R. China
eMarine Chemical Research Institute Co. Ltd, Qingdao, Shandong Province 266071, P. R. China
fState Key Laboratory of Coatings for Advanced Equipment, 266071, P. R. China
gKey Laboratory of Special Functional Aggregated Materials (Shandong University), Ministry of Education, China
hSchool of Chemistry and Chemical Engineering, Shandong University, Jinan 250100, P. R. China
First published on 16th February 2026
Abstract
Schiff base compounds have garnered considerable research interest due to their promising fluorescence properties. In this work, we successfully synthesized two Schiff base compounds as aggregation-induced emission (AIE)-active luminescent agents using a simple and efficient one-step method. The molecular design of these compounds effectively combines the dual advantages of Schiff base structures and AIE effects, retaining the chemical stability and tunable optical properties of Schiff base compounds while exhibiting the unique AIE property of significantly enhanced fluorescence in the aggregated state. Owing to these AIE characteristics, the synthesized compounds show potential for application in biomedicine, including bioimaging, fluorescence sensing, and information security.
Introduction
Schiff bases are defined as compounds containing imine or methylimine groups (–RC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–), and are typically formed via the condensation of primary amines with reactive carbonyl groups.1 Due to their ease of synthesis, strong coordination ability, and favorable fluorescence properties,2–5 Schiff bases have attracted considerable research attention over recent decades. However, while most studies on Schiff bases predominantly focus on their use as fluorescence sensors,6,7 relatively few have examined their inherent fluorescence properties and structure–activity relationships.8
N–), and are typically formed via the condensation of primary amines with reactive carbonyl groups.1 Due to their ease of synthesis, strong coordination ability, and favorable fluorescence properties,2–5 Schiff bases have attracted considerable research attention over recent decades. However, while most studies on Schiff bases predominantly focus on their use as fluorescence sensors,6,7 relatively few have examined their inherent fluorescence properties and structure–activity relationships.8
Conventional fluorescent molecules often undergo aggregation-caused quenching (ACQ), a phenomenon where fluorescence diminishes at high concentrations or in the aggregated state due to π–π stacking interactions that promote the formation of quenching species, such as excimers.9 This limits their utility in applications requiring aggregated forms, such as solid films for optoelectronic devices and concentrated fluorescent probes. In contrast, aggregation-induced emission (AIE) compounds exhibit enhanced fluorescence in the aggregated state, thereby overcoming ACQ and offering promise for practical applications.10–14 AIE materials have recently attracted attention for their utility in optoelectronics and biomedical imaging, alongside their potential for use as biosensors. Their advantages—including deep tissue penetration ability,15,16 minimal autofluorescence, high photostability, and low biotoxicity17—render them suitable for analytical detection, multimodal bioimaging, light-controlled drug release, and photodynamic therapy.18–20 Therefore, synthesizing Schiff base compounds with AIE properties is critical for producing photostable probes that overcome the limitations of traditional ACQ fluorophores.
Studies on AIE have achieved success through the development of nanoplatforms that employ AIE compounds as photosensitizers.21–23 For example, Liu et al.24 prepared luminescent nanoparticles from tetraphene derivatives with strong AIE properties; by combining these with the triplet sensitizer platinum octaethylporphyrin (PtOEP), they achieved aggregation-induced photon upconversion (iPUC). Additionally, a multifunctional nanoplatform, TUT NPs, was developed using AIE-active photosensitizers, MXene Ti3C2 nanosheets, and UCNPs,25 yielding intense near-infrared fluorescence under 808 nm excitation through the combined action of the individual components. Unlike these complex methodologies, we focus here on a streamlined molecular design strategy. We report a straightforward, one-step synthesis of two novel siloxane-integrated Schiff base compounds, PBS and OBS. The deliberate incorporation of a flexible siloxane spacer aims to suppress detrimental π–π stacking and enhance AIE,26–29 while the Schiff base core offers a facile site for structural tuning and metal ion coordination.30,31
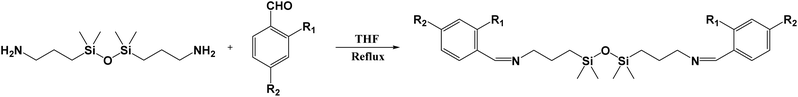
In this study, two siloxane-containing Schiff base compounds, PBS and OBS, were synthesized from MMNH2 and p-hydroxybenzaldehyde or o-hydroxybenzaldehyde, respectively (Scheme 1), in which (a) represents PBS and (b) represents OBS, and their luminescence properties were subsequently investigated. The strong solid-state emission of PBS suggests utility in developing “turn-on” fluorescent sensors for solid-phase or high-viscosity environments, such as test strips for on-site detection of analytes or probes for monitoring polymerization processes where conventional dyes quench. Furthermore, the combination of AIE-driven brightness, enhanced photostability, and the potential biocompatibility conferred by the siloxane moiety positions these compounds as promising candidates for long-term, high-fidelity bioimaging, addressing the critical limitation of photobleaching in conventional fluorescent tags 32–35. The comparative study of the para- (PBS) and ortho-substituted (OBS) analogues provides fundamental insights into the structure–property relationship, guiding the future design of AIE-active Schiff bases.
Experimental
Materials and methods
All chemicals were purchased from commercial sources and were of analytical grade unless otherwise indicated. Tetrahydrofuran (THF) was purchased from Tianjin Fuyu Chemical Co., Ltd. 1,3-Bis(aminopropyl)tetramethyldisiloxane (MMNH2) was purchased from Hangzhou Dadi Chemical Co., Ltd. p-Hydroxybenzaldehyde and o-hydroxybenzaldehyde were obtained from Shanghai Aladdin Bio-Chem Technology Co., Ltd.For the testing of the synthesized materials, 1H NMR and 13C NMR spectra were recorded on a Bruker AVANCE 300 MHz or AVANCE 400 MHz superconducting NMR spectrometer. UV-Vis absorption spectra were recorded on a Hitachi U3900H spectrophotometer, and FTIR analysis was performed synthesized materials using a Bruker TENSOR-27 FTIR spectrometer. Fluorescence properties were characterized on an F-7000 Fluorescence Spectrophotometer. All experiments were performed at room temperature (25 °C) unless otherwise specified.
Synthesis of PBS
p-Hydroxybenzaldehyde (1.22 g, 0.01 mol) was dissolved in THF (10 mL). A solution of 1,3-bis(3-aminopropyl)-1,1,3,3-tetramethyldisiloxane in THF (10 mL) was then added dropwise to the system at room temperature. The resulting mixture was continuously stirred at 50 °C for 2 h. After the evaporation of the solvent, the crude product was purified, yielding a pale-yellow solid (2.03 g; yield: 89.1%). 1H NMR (400 MHz, CD3OD, δ, ppm): 9.73 (1H, –OH), 8.14 (1H, –CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–), 7.59–7.56 (2H, Ar–H), 6.80–6.78 (2H, Ar–H), 3.52 (2H,
N–), 7.59–7.56 (2H, Ar–H), 6.80–6.78 (2H, Ar–H), 3.52 (2H, ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–CH2–), 1.74–1.66 (2H,
N–CH2–), 1.74–1.66 (2H, ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–CH2–CH2–), 0.57–0.53 (2H, –Si–CH2–CH2), 0.08 (6H, –CH3). FTIR spectrum (KBr disk, cm−1): 1051 cm−1 (Si–O–Si), 1644 cm−1 (C
N–CH2–CH2–), 0.57–0.53 (2H, –Si–CH2–CH2), 0.08 (6H, –CH3). FTIR spectrum (KBr disk, cm−1): 1051 cm−1 (Si–O–Si), 1644 cm−1 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N). 13C NMR: (100 MHz, CD3OD, δ, ppm; Fig. S1): δ 164.1, 131.8, 126.6, 117.3, 64.0, 25.8, 16.6, 0.45. ESI-HRMS (Fig. S1): m/z calculated for [C24H37N2O3Si2]+: 457.23, found 457.23 [M + H]+.
N). 13C NMR: (100 MHz, CD3OD, δ, ppm; Fig. S1): δ 164.1, 131.8, 126.6, 117.3, 64.0, 25.8, 16.6, 0.45. ESI-HRMS (Fig. S1): m/z calculated for [C24H37N2O3Si2]+: 457.23, found 457.23 [M + H]+.
Synthesis of OBS
o-Hydroxybenzaldehyde (1.22 g, 0.01 mol) was dissolved in THF (10 mL), after which a solution of 1,3-bis(3-aminopropyl)-1,1,3,3-tetramethyldisiloxane in THF (10 mL) was added dropwise to the system at room temperature. The resulting mixture was stirred continuously at 50 °C for 2 h. After rotary evaporation of the solvent, the crude product was purified, yielding a yellow-green liquid (2.16 g; yield: 94.8%). 1H NMR (400 MHz, CDCl3, δ, ppm; Fig. S3): 13.70 (1H, –OH), 8.31 (1H, –CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–), 7.32–7.23 (2H, Ar–H), 6.98–6.87 (2H, Ar–H), 3.57 (2H,
N–), 7.32–7.23 (2H, Ar–H), 6.98–6.87 (2H, Ar–H), 3.57 (2H, ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–CH2–), 1.72 (2H,
N–CH2–), 1.72 (2H, ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–CH2–CH2–), 0.58 (2H, –Si–CH2–CH2), 0.08 (6H, –CH3). FTIR spectrum (KBr disk, cm−1): 1054 cm−1 (Si–O–Si), 1633 cm−1 (C
N–CH2–CH2–), 0.58 (2H, –Si–CH2–CH2), 0.08 (6H, –CH3). FTIR spectrum (KBr disk, cm−1): 1054 cm−1 (Si–O–Si), 1633 cm−1 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N). 13C NMR: (100 MHz, CD3OD, δ, ppm; Fig. S1): δ 166.8, 165.7, 134.2, 133.1, 119.4, 118.9, 118.7. ESI-HRMS (Fig. S1): m/z calculated for [C24H37N2O3Si2]+: 457.23, found 457.23 [M + H]+.
N). 13C NMR: (100 MHz, CD3OD, δ, ppm; Fig. S1): δ 166.8, 165.7, 134.2, 133.1, 119.4, 118.9, 118.7. ESI-HRMS (Fig. S1): m/z calculated for [C24H37N2O3Si2]+: 457.23, found 457.23 [M + H]+.
Results and discussion
Structure characterization
As shown in Fig. 1A, the IR spectra of compound PBS and its reactant MMNH2, the disappearance of the primary amine bimodal peak near 3300 cm−1, and the absorption of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond at 1633 cm−1 tentatively confirmed the occurrence of the reaction. 1H NMR spectra were recorded on a Bruker 400 (400 MHz) spectrometer without tetramethylsilane as an internal reference and are shown in Fig. 1B. Resonance peaks at 0.08, 0.55, 1.70, and 3.52 ppm—corresponding to –Si–CH3, –Si–CH2–, –Si–CH2–CH2–, and –CH2–N
N bond at 1633 cm−1 tentatively confirmed the occurrence of the reaction. 1H NMR spectra were recorded on a Bruker 400 (400 MHz) spectrometer without tetramethylsilane as an internal reference and are shown in Fig. 1B. Resonance peaks at 0.08, 0.55, 1.70, and 3.52 ppm—corresponding to –Si–CH3, –Si–CH2–, –Si–CH2–CH2–, and –CH2–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) , respectively—were clearly observed for PBS. Combined, these results indicated that the Schiff base compound PBS had been successfully synthesized.
, respectively—were clearly observed for PBS. Combined, these results indicated that the Schiff base compound PBS had been successfully synthesized.
Optical properties
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N formation and coexistence of both forms. The keto-amine band at 400 nm in methanol is stabilized by intermolecular hydrogen bonding with the protic solvent, lowering its energy relative to the enol-imine form.
N formation and coexistence of both forms. The keto-amine band at 400 nm in methanol is stabilized by intermolecular hydrogen bonding with the protic solvent, lowering its energy relative to the enol-imine form.
 | ||
| Fig. 2 UV-Vis absorption spectra of PBS in (a) THF, and (b) CH3OH. Inset: enol-imine and keto-amine forms of PBS. | ||
Although precise tautomer ratios require techniques such as NMR titration or time-resolved spectroscopy, the relative intensity of the ∼400 nm band offers a clear qualitative indicator. This band is significantly stronger in PBS than in OBS, particularly in methanol, indicating a higher equilibrium population of the keto-amine tautomer in the para-substituted PBS. This trend is attributed to more effective intermolecular hydrogen-bonding stabilization in protic media and aligns with established photophysical trends in analogous Schiff-base systems.36,37
Critically, the keto-amine tautomer is recognized as the primary emitting species in such systems,38–40 governing fluorescence through mechanisms such as excited-state intramolecular proton transfer (ESIPT) and/or intramolecular charge transfer (ICT). Photoexcitation of this form promotes rapid ESIPT or strong ICT, yielding a highly conjugated, planar excited state that emits at longer wavelengths. This mechanism explains the red-shifted emission and pronounced positive solvatochromism observed in PBS, whose higher keto-amine population enhances fluorescence intensity and AIE activity. Conversely, the weaker emission of OBS correlates with its reduced keto-amine absorption, consistent with this framework. Therefore, tautomeric equilibrium not only shapes the absorption profile but also decisively dictates fluorescence outcomes—including emission wavelength, quantum yield, and AIE enhancement—highlighting the central role of tautomerism in the photophysics of these Schiff-base derivatives.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– moiety and the benzene ring extends the electron delocalization range, thereby narrowing the energy gap for electron transitions and enhancing fluorescence. The nitrogen atom of the –C
N– moiety and the benzene ring extends the electron delocalization range, thereby narrowing the energy gap for electron transitions and enhancing fluorescence. The nitrogen atom of the –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– group in the Schiff base synthesized in this work adopts sp2 hybridization; the presence of a lone pair of electrons makes it a relatively strong electron acceptor with a high propensity for protonation. Additionally, the oxygen atom contains lone-pair electrons that engage in p–π conjugation with the benzene ring. The emission peaks observed at 472 nm and 503 nm are ascribed to the intramolecular interactions between the –OH group (acting as an electron donor) on the benzene ring and the protonated imine group (acting as the acceptor).
N– group in the Schiff base synthesized in this work adopts sp2 hybridization; the presence of a lone pair of electrons makes it a relatively strong electron acceptor with a high propensity for protonation. Additionally, the oxygen atom contains lone-pair electrons that engage in p–π conjugation with the benzene ring. The emission peaks observed at 472 nm and 503 nm are ascribed to the intramolecular interactions between the –OH group (acting as an electron donor) on the benzene ring and the protonated imine group (acting as the acceptor).
To explore how this inherent emissivity translates into aggregate-state behavior, we investigated the concentration-dependent fluorescence of PBS in methanol. As shown in Fig. 3A (focusing on the intensity trend), the emission is negligible in dilute solution but increases sharply with concentration, reaching a maximum at approximately 5 × 10−3 mol L−1. This initial enhancement is characteristic of AIE, attributed to the restriction of intramolecular motion (RIM) in aggregates, which suppresses non-radiative decay. Notably, beyond this optimum concentration, a gradual decrease in intensity is observed (Fig. 3A), likely due to competitive effects such as inner-filtering or the formation of non-emissive excimers at very high concentrations.
To further investigate the influence of solubility on AIE, we altered the solvent environment. Given that the compound displayed lower solubility in hexane than in methanol, we incrementally added hexane to a methanol solution of the compound to examine its AIE response. As demonstrated in Fig. 4A, the fluorescence intensity progressively intensified with increasing hexane content, confirming the AIE characteristics of the material under solvent-induced aggregation. As shown in Fig. 4B, the fluorescence emission spectra of PBS were recorded in a series of solvents with varying polarity. The spectral behavior exhibited a clear dependence on solvent environment.
Mechanism of upconversion and AIE
Additionally, the infcorporation of Si–O–Si units disrupts the formation of a fully planar molecular structure, thereby mitigating the fluorescence quenching caused by π–π stacking interactions. This structural feature likely contributes to the compound's pronounced AIE properties.
The dipole moment of an excited state of a fluorescent molecule is larger than that of the ground state, and an increase in solvent polarity produces a greater stabilizing effect on the excited state than on the ground state. As a result, the fluorescence spectrum shifts to longer wavelengths with increasing solvent polarity. The fluorescence intensity of the compound in more polar solvents, such as THF and acetonitrile, is much greater than that in nonpolar media, such as hexane and triethylamine. This may be attributable to the fact that the energy difference (ΔE) required for the π → π* transition is reduced in polar solvents. The increased transition probability results in a redshift in both the UV absorption and fluorescence emission wavelengths, alongside an enhancement in emission intensity.
Density functional theory
To gain further insight into the influence of the molecular structure on the ICT characteristics, the frontier molecular orbitals—specifically, the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO)—were calculated. All electronic structure simulations were performed with Gaussian 16 software. The CAM-B3LYP functional was adopted for all calculations. For geometry optimization and frequency calculations, the 6-311G(d) basis set34 was used, and the optimal geometry for each compound was determined.The molecular structure, ground-state geometry optimization structure, and frontier orbital electron cloud distribution of compound PBS are shown in Fig. S7 (Fig. S6 for OBS). The geometric optimization results indicate that the torsion angles (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N and benzene ring planes) in both the ground-state and the excited state are close to 0°. This relatively planar geometric configuration is conducive to charge transfer, which provides favorable conditions for ICT.
N and benzene ring planes) in both the ground-state and the excited state are close to 0°. This relatively planar geometric configuration is conducive to charge transfer, which provides favorable conditions for ICT.
The electron clouds on the HOMO orbitals of the optimized ground-state geometry are almost predominantly distributed throughout the conjugated part (left side), while the electron clouds on the HOMO orbitals of the optimized excited-state geometry are mostly localized around C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N, which supports the presence of a strong ICT. Simultaneously, sufficient orbital overlap ensures a reasonable radiative transition rate, while the change in the electron cloud distribution distinguishes this excited state from a localized excited state.
N, which supports the presence of a strong ICT. Simultaneously, sufficient orbital overlap ensures a reasonable radiative transition rate, while the change in the electron cloud distribution distinguishes this excited state from a localized excited state.
A more rigorous explanation for the fluorescence behavior of the AIEgens in solution involves π-twisting and distortion in the excited state, which may lead to a minimum energy conical intersection and subsequent non-radiative decay. The geometrical optimization results clearly indicate that a more planar geometric configuration of the fluorescent moiety favors enhanced emission even in solution. Additionally, the distorted conformation of the Si–O–Si fragment expands the intermolecular distance, thus significantly suppressing intermolecular π–π interactions and further ensuring fluorescence emission in the aggregated state.
Conclusions
In summary, two novel siloxane-containing Schiff base compounds were synthesized, and their fluorescence properties were evaluated at different concentrations. The results showed that conjugation of the –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond with the benzene ring and the siloxane helical structure imparts unique fluorescent properties, offering new insights for enhancing the AIE characteristics of such materials. The strong AIE luminescence of compound PBS is primarily attributed to the RIM mechanism. Furthermore, the presence of ICT within the molecule was corroborated by both DFT calculations and fluorescence assays in various solvents. Such an AIE-active compound overcomes the limitation of most organic luminescent materials, which typically do not emit light in aggregated forms, such as thin films, making it highly promising for diverse applications. These include the development of organic electroluminescent devices, chemical sensing, and, in particular, advanced bioimaging platforms.
N bond with the benzene ring and the siloxane helical structure imparts unique fluorescent properties, offering new insights for enhancing the AIE characteristics of such materials. The strong AIE luminescence of compound PBS is primarily attributed to the RIM mechanism. Furthermore, the presence of ICT within the molecule was corroborated by both DFT calculations and fluorescence assays in various solvents. Such an AIE-active compound overcomes the limitation of most organic luminescent materials, which typically do not emit light in aggregated forms, such as thin films, making it highly promising for diverse applications. These include the development of organic electroluminescent devices, chemical sensing, and, in particular, advanced bioimaging platforms.
Author contributions
Yuling Li: conceptualization, resources, investigation, writing – original draft. Depeng Ma: conceptualization, data curation, validation, writing – original draft. Fanzhen Kong: data curation, validation, writing – original draft, formal analysis. Yuanrong Wang: funding acquisition, supervision, writing – review & editing. Lianfeng Wu: conceptualization, funding acquisition, supervision, validation, writing – review & editing. Zhihui Yang: conceptualization, funding acquisition, supervision, visualization, writing – review & editing. Haifeng Lu: conceptualization, funding acquisition, supervision, validation, writing – review & editing.Conflicts of interest
The authors declare no conflict of interest.Data availability
The data supporting the findings of this study are available in the supplementary information (SI), which includes characterization data (NMR, HRMS, and IR spectra), photophysical properties (UV-vis absorption, fluorescence emission in various solvents, and concentration-dependent emission), and theoretical calculations (ground- and excited-state optimized geometries and frontier molecular orbital electron cloud distributions) for PBS and OBS. See DOI: https://doi.org/10.1039/d5ra10030e.Acknowledgements
We thank the National Key R&D Program of China (2022YFF1400300) for supporting our work. This project was supported by the open project funding of the State Key Laboratory of Coatings for Advanced Equipment. The authors acknowledge the assistance of Shandong University Structural Constituent and Physical Property Research Facilities/SDU SC&PP Research Facilities/SDU SCPPRF.References
- A. L. Berhanu, I. Malik, A. K. Aulakh, J. SinghKumar and V. K. Ki-Hyun, TrAC, Trends Anal. Chem., 2019, 116, 74–91 CrossRef CAS.
- M. T. Kaczmarek, M. Zabiszak, M. Nowak and R. Jastrzab, Coord. Chem. Rev., 2018, 370, 42–54 CrossRef.
- L. Chen, T. Fu, H. B. Zhao, D. M. Guo, X. L. Wang and Y. Z. Wang, Chem. Eng. J., 2018, 336, 622–632 CrossRef.
- G. Li, Q. Lin, L. Sun, C. Feng, P. Zhang, B. Yu, Y. Chen, Y. Wen, H. Wang and L. Ji, Biomaterials, 2015, 53, 285–295 CrossRef PubMed.
- A. K. Singh and L. K. Kumawat, Sens. Actuators, B, 2014, 195, 98–108 CrossRef.
- J. Jayabharathi, V. Thanikachalam, K. Jayamoorthy and M. V. Perumal, Spectrochim. Acta, Part A, 2011, 79, 6–16 CrossRef PubMed.
- T. Jing and L. Yan, Talanta, 2017, 170, 185–192 CrossRef PubMed.
- D. Li, X. Liang, F. Zhang, J. Li, Z. Zhang, S. Wang, Z. Li, Y. Xing and K. Guo, J. Mater. Chem. C, 2022, 10, 11016–11026 Search PubMed.
- J. Mei, Y. Hong, J. W. Y. Lam, A. Qin, Y. Tang and B. Z. Tang, Adv. Mater., 2014, 26, 5429–5479 Search PubMed.
- S. Yang, Y. Yuan, X. Wang, Z. Hu and D. Guo, J. Lumin., 2022, 242, 118560 Search PubMed.
- S. Huang, J. Ding, A. Bi, K. Yu and W. Zeng, Adv. Opt. Mater., 2021, 9, 2170086 Search PubMed.
- L. Wu, N. Liang, X. Yang, W. Pan, Y. Wang and L. Zhao, J. Photochem. Photobiol., A, 2021, 420, 113506 Search PubMed.
- Y. Zhang, P. Zhuo, H. Yin, Y. Fan, J. Zhang, X. Liu and Z. Chen, ACS Appl. Mater. Interfaces, 2019, 11, 24395–24403 Search PubMed.
- C. Zhou, W. Xu, P. Zhang, M. Jiang, Y. Chen, R. T. K. Kwok, M. M. S. Lee, G. Shan, R. Qi and X. Zhou, Adv. Funct. Mater., 2019, 29, 1805986 Search PubMed.
- H. Dong, S. R. Du, X. Y. Zheng, G. M. Lyu, L. D. Sun, L. D. Li, P. Z. Zhang, C. Zhang and C. H. Yan, Chem. Rev., 2015, 115, 10725–10815 Search PubMed.
- B. Zhou, Q. Li, L. Yan and Q. Zhang, J. Rare Earths, 2020, 38, 474–482 Search PubMed.
- L. Cheng, C. Wang and Z. Liu, Nanoscale, 2012, 5, 23–37 Search PubMed.
- G. Chen, H. Qiu, P. N. Prasad and X. Chen, Chem. Rev., 2014, 114, 5161–5214 CrossRef PubMed.
- Y. Sun, W. Feng, P. Yang, C. Huang and F. Li, Chem. Soc. Rev., 2015, 44, 1299–1301 RSC.
- D. Yin, C. Wang, J. Ouyang, X. Y. Zhang and M. Wu, ACS Appl. Mater. Interfaces, 2014, 6, 18480–18488 CrossRef.
- P. Duan, N. Yanai, Y. Kurashige and N. Kimizuka, Angew. Chem., 2015, 127, 7654–7659 CrossRef.
- S. Hisamitsu, N. Yanai and N. Kimizuka, Angew. Chem., 2015, 127, 11712–11716 CrossRef.
- Y. Wang, Y. Li, Z. Zhang, L. Wang, D. Wang and B. Z. Tang, Adv. Mater., 2021, 33, 2103748 Search PubMed.
- Z. Qu, P. Duan, J. Zhou, Y. Wang and M. Liu, Nanoscale, 2018, 10, 985–991 RSC.
- Y. Wang, N. Niu, Y. Huang, S. Song, H. Tan, L. Wang, D. Wang and B. Z. Tang, Small Methods, 2022, 6, 2200393 CrossRef.
- J. H. Yang, L. Du, J. X. Guo, L. Zhang, S. G. Wang and X. H. Wang, Int. J. Biol. Macromol., 2025, 308(4), 142572, DOI:10.1016/j.ijbiomac.2025.142572.
- L. P. Zong, X. J. Li, D. R. Zhu, X. W. Guo, J. Chauvin, S. L. Zhang, G. Y. Zhang, K. Krukiewicz, S. Cosnier, X. J. Zhang and D. Shan, ACS Sens., 2025, 10(9), 7134–7143, DOI:10.1021/acssensors.5c02452.
- H. Jafari, E. Ameri, M. Hassan Vakili and A. Berisha, Electrochem. Commun., 2024, 159, 107653, DOI:10.1016/j.elecom.2023.107653.
- I. Chaturvedi, S. Vyas and R. Mishra, Dyes Pigm., 2025, 239, 112748, DOI:10.1016/j.dyepig.2025.112748.
- D. A. Chen, J. Li, Z. H. Li, T. Deng and J. S. Li, Talanta, 2025, 295, 128332, DOI:10.1016/j.talanta.2025.128332.
- R. Arabahmadi and S. Kamali, J. Water Process Eng., 2025, 76, 108230, DOI:10.1016/j.jwpe.2025.108230.
- S. Yang, J. Zhang, G. Y. Liu, R. Wang and X. Q. Zhang, Compos. Commun., 2025, 54, 102257, DOI:10.1016/j.coco.2025.102257.
- H. Liu, D. Ren, H. Geng, Y. Tian, M. Li, N. Wang, S. Yuan, J. Hao and J. Cui, Adv. Healthcare Mater., 2025, 14, 2403865, DOI:10.1002/adhm.202403865.
- L. Wang and Y. Chen, Mater. Horiz., 2025, 12, 2709–2721, 10.1039/d4mh01546k.
- V. Sharma, S. Dutta, D. Sahu, A. Pandey, D. Kumar, J. Mayee Bag and G. Kumar Patra, Inorg. Chem. Commun., 2025, 181(1), 115110, DOI:10.1016/j.inoche.2025.115110.
- N. M. D. Brown and D. C. Nonhebel, Tetrahedron, 1968, 24(16), 5655–5664, DOI:10.1016/0040-4020(68)88164-5.
- İ. Sıdır, Y. Gülseven Sıdır, H. Berber, M. L. Ramos, L. L. G. Justino and R. Fausto, Molecules, 2025, 30, 745, DOI:10.3390/molecules3003074.
- I. Sıdır, Y. G. Sıdır, H. Berber and R. Fausto, J. Mol. Struct., 2023, 1292, 136191 CrossRef.
- X. Y. Cheng, T. S. Zhang, Z. L. Li and K. Zhao, J. Phys. Chem. B, 2025, 129(24), 6061–6068 CrossRef PubMed.
- T. Jiang, J. H. Lu, C. Huang, D. M. Chen and B. X. Zhu, Dyes Pigm., 2024, 223, 111972 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |