DOI:
10.1039/D5RA09694D
(Paper)
RSC Adv., 2026,
16, 18612-18623
Hydrogen storage capacity, strain-improved formation enthalpy, desorption temperature, and high energy harvesting performance of SrGaH5
Received
15th December 2025
, Accepted 9th March 2026
First published on 8th April 2026
Abstract
The study of hydrogen storage (HS) characteristics in complex hydrides has become a compelling goal in recent times. Therefore, we explore the SrGaH5 hydride by focusing on its potential for HS utilization and various physical aspects via first-principles calculations under hydrostatic strain. The results indicate that strain has a positive impact on the system’s thermodynamics, which enhances its potential for practical applications as a HS material. The formation enthalpy and desorption temperature decrease to −42.93/−45.18 kJ per mol H2 and 306.67/322.74 K under −5%/+5% strain from the unstrained values of −48.03 kJ per mol H2 and 343.13 K, respectively. These values are nearly ideal, approaching the theoretical values of −40 kJ per mol H2 and 233–333 K, making it a promising candidate for HS uses. Furthermore, the volumetric hydrogen capacity is enhanced to 17% at −5% and the system exhibits a high gravimetric hydrogen capacity of 3.10%, which is good enough for practical purposes. Along with this, the system is found to be mechanically stable, where the calculated 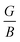 ratio and Poisson’s ratio further imply the existence of ionic bonding character in the material. Further, the non-negative phonon frequency curves validate the dynamical stability of the structure. The electronic structure calculations predicted an insulating behavior with an indirect energy gap of 3.58–2.43 eV for the considered strain range. Surprisingly, the thermoelectric performance of the system has been significantly improved under tensile strain as the figure of merit reaches a high value of 0.61 at 550 K under +5% strain. This is because the mean free path of phonons is reduced as the temperature rises due to an increase in phonon scattering driven by greater lattice vibrations, which causes a decrease in lattice thermal conductivity. Hence, the hydrogen storage capabilities and other aspects of the material indicate that it is an appropriate candidate for HS and might be crucial in various energy-generating applications.
ratio and Poisson’s ratio further imply the existence of ionic bonding character in the material. Further, the non-negative phonon frequency curves validate the dynamical stability of the structure. The electronic structure calculations predicted an insulating behavior with an indirect energy gap of 3.58–2.43 eV for the considered strain range. Surprisingly, the thermoelectric performance of the system has been significantly improved under tensile strain as the figure of merit reaches a high value of 0.61 at 550 K under +5% strain. This is because the mean free path of phonons is reduced as the temperature rises due to an increase in phonon scattering driven by greater lattice vibrations, which causes a decrease in lattice thermal conductivity. Hence, the hydrogen storage capabilities and other aspects of the material indicate that it is an appropriate candidate for HS and might be crucial in various energy-generating applications.
1 Introduction
Increasing environmental concerns, especially regarding CO2 emissions from fossil fuels, have driven worldwide investigations into sustainable alternative energy sources to create a more sustainable future. Recent work has been focused on new and renewable sources to meet the growing need for sustainable and affordable energy,1–3 where fossil fuels persist as the primary source of energy globally. However, the rapid depletion of petroleum resources and the increase in energy demand highlight the importance of sustainable energy sources.4 They could provide alternative energy sources and be environmentally friendly for countries that lack natural resources. In this sense, hydrogen is a cost-effective and non-toxic energy source that is suitable for mobile and stationary applications5,6 due to its abundance and reduces reliance on imported oil.7 However, storing and transporting hydrogen is critical for large-scale applications. Therefore, new hydrogen absorption and storage materials are being developed to ensure long-term safety.8–10 Compounds that are employed for energy storage must meet certain criteria, including large hydrogen storage (HS) capacities,11 reversibility,12 release at ambient conditions,13 and acceptable kinetics.14,15 Compared to traditional methods for HS techniques such as gas compression and liquefaction,16,17 solid-state storage is gaining popularity due to its lightweight, cost-effective, and safer materials.7,18 In advanced technology, metal and complex metal hydrides,19,20 intermetallic hydrides,21–23 complex chemical hydrides,24 nanostructured carbon materials,25 and metalorganic compounds have been used extensively.26–29
Lately, transition metal (TM)-based perovskite-type hydrides have attracted greater attention from researchers due to their potential uses in HS applications.30 Gencer et al.7 determined a hydrogen Tdes. (desorption temperature) of 446.3 K, 419.5 K, and 367.5 K for LiNiH3, NaNiH3, and KNiH3 systems, respectively. Furthermore, Xiong et al.31 reported that NaRuH3 and NaRhH3 have high gravimetric HS capacities (Cwt%) of 2.38 wt% and 2.35 wt%, respectively. However, their potassium analogs (KRuH3/KRhH3) have somewhat lower values (2.11/2.09 wt%). Interestingly, the Tdes. drops from 436.40 K (NaRuH3) to 286.37 K (KRhH3), indicating enhanced reversibility in potassium-based systems. Along with this, ARH3 shows potential for H2 storage applications due to its impressive Cwt% of 6.042 wt% for LiCaH3 and 3.678 wt% for KCaH3. Moreover, these materials satisfied the criteria for Born stability based on their mechanical parameters, which are derived from elastic constants, including Pugh’s ratio 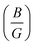 and the related moduli. The analysis of the
and the related moduli. The analysis of the 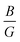 ratio and Cauchy pressure (CP), indicates that, unlike other compounds, KCaH3 exhibits a ductile nature (Poisson’s ratio (ν) = 0.29).32 Besides this, alkali metals containing ternary hydrides, such as LiBH4 (LBH), LiAlH4, NaBH4, and NaAlH4, have been intensively explored as prospective HS materials (HSMs) due to their high Cwt%.33–36 Among them, LBH is found to be a promising solid-state HSM with a volumetric capacity (ρvol.) of 121 kg H2 per m3 and the highest Cwt% of 18.3 wt% at room temperature.35,37,38 In 1997, NaAlH4 was introduced as a lightweight reversible HSM and is regarded as a promising material for solid-state hydrogen storage due to its high Cwt%, widespread availability, and low cost.33 According to Vajeeston et al.,39 the alkali boron tetrahydrides ABH4 (where A = Li, Na, K, Rb, and Cs) are insulators having an energy gap (Eg) ranging from 5.5 to 7.0 eV. Recent experimental evidence indicates that LiAlH4 and NaAlH4, processed by mechano-chemical means under ambient conditions with specific TM catalysts,40 can rapidly release 7.9 and 5.6 wt% of hydrogen, respectively. The theoretical Cwt% of LiAlH4 is as high as 10.6 wt%.41 In contrast to other chemical hydrides, which are simple and stable,42 NaBH4 is regarded as a potential candidate for HSMs due to its high hydrogen concentration of 10.6 wt%.
ratio and Cauchy pressure (CP), indicates that, unlike other compounds, KCaH3 exhibits a ductile nature (Poisson’s ratio (ν) = 0.29).32 Besides this, alkali metals containing ternary hydrides, such as LiBH4 (LBH), LiAlH4, NaBH4, and NaAlH4, have been intensively explored as prospective HS materials (HSMs) due to their high Cwt%.33–36 Among them, LBH is found to be a promising solid-state HSM with a volumetric capacity (ρvol.) of 121 kg H2 per m3 and the highest Cwt% of 18.3 wt% at room temperature.35,37,38 In 1997, NaAlH4 was introduced as a lightweight reversible HSM and is regarded as a promising material for solid-state hydrogen storage due to its high Cwt%, widespread availability, and low cost.33 According to Vajeeston et al.,39 the alkali boron tetrahydrides ABH4 (where A = Li, Na, K, Rb, and Cs) are insulators having an energy gap (Eg) ranging from 5.5 to 7.0 eV. Recent experimental evidence indicates that LiAlH4 and NaAlH4, processed by mechano-chemical means under ambient conditions with specific TM catalysts,40 can rapidly release 7.9 and 5.6 wt% of hydrogen, respectively. The theoretical Cwt% of LiAlH4 is as high as 10.6 wt%.41 In contrast to other chemical hydrides, which are simple and stable,42 NaBH4 is regarded as a potential candidate for HSMs due to its high hydrogen concentration of 10.6 wt%.
To improve the stability (formation enthalpy (ΔHf)/Tdes.) of the HSMs, numerous theoretical and experimental studies have been conducted, where size reduction,28,43 alloying,44,45 doping/functionalization,46,47 morphological control,48,49 and strain engineering50,51 are some of the most promising approaches that accomplish that goal. The calculations conducted by Zhang et al.52 indicated that under biaxial compressive and tensile strains, the ΔHf/Tdes. and hydrogen diffusion observed for magnesium hydride (MgH2) decreased simultaneously, thereby enhancing its potential for HS applications. Similarly, LBH maintains its high Cwt% (18.3 wt%), while biaxial strain improves the thermodynamic characteristics as well as hydrogenation kinetics,50 to make it a superior candidate for energy transport in mobile applications. Specifically, under compression of −6%, the ΔHf changes from −71.30 kJ per mol H2 in unstrained MgCoH3 to −37.29 kJ per mol H2, and the Tdes. decreases to 285.34 K as opposed to 545.52 K, which are both near ideal values (−40 kJ per mol H2 and 289–393 K) for practical applications.53 Likewise, the ΔHf and Tdes. are observed to decrease from −67.73 kJ per mol H2 and 521 K for unstrained ZrNiH3 to −33.73 kJ per mol H2 and 259.5 K under a maximal biaxial compressive strain of −6%, correspondingly.54 Additionally, for a maximal biaxial tensile strain of +6%, the values are −50.99 kJ per mol H2 and 392.23 K. A similar phenomenon has also been noted for uniaxial strain, wherein the ΔHf/Tdes. are reduced to −39.36 kJ per mol H2/302.78 K for a maximum uniaxial compressive strain of −12% and to −51.86 kJ per mol H2/399 K under a maximum uniaxial tensile strain of +12%.54
The aforementioned considerations clearly illustrate that strain engineering serves as an effective strategy for optimizing the HS features and overall physical attributes of materials. Various ABH5-type hydrides like CaAlH5 (a novel member of the aluminum hydride family, where Ca(AlH4)2 breaks down to generate CaAlH5),55 BAlH5 (B = Ba, Mg)56 (where the experimentally determined crystal structure of BaAlH5 is confirmed, while the MgAlH5 structure is computationally predicted), MgGaH5, CaGaH5, BaGaH5, and SrGaH5 (SGH)57 have been extensively studied via both theoretical and experimental investigations. Nevertheless, no prior research has looked at the ability to store hydrogen, despite extensive structural analyses of SGH. Therefore, we theoretically examine the effects of hydrostatic strain on the structural, HS, mechanical, dynamical, electronic, and thermoelectric (TE) performance of SGH in the context of HS applications. A detailed investigation is performed to assess its structural stabilities and HS aspects, including ΔHf, Tdes., Cwt%, and ρvol.. The findings offer important insights into the strain-dependent behavior of SGH, serving as a guide for its practical application in HS technologies. Moreover, under tensile strain, the TE performance is much improved and a particularly high ZT is achieved at high temperature.
2 Computational detail
The full-potential linearized augmented plane-wave approach as implemented in the WIEN2K code58 was used to perform density-functional-theory-based computations for the current study. The exchange–correlation functional, which is parameterized in terms of a generalized gradient approximation (GGA), is utilized.59 The plane-wave cutoff is set at Rmt × Kmax = 5 with Gmax = 20 for the wave-function expansion inside the atomic spheres, and a maximum value of 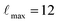 is selected. It is found that a 7 × 7 × 9 k-space grid with 128 points inside the irreducible Brillouin zone wedge gives effective convergence. Additionally, in every case, the complete relaxation of the atomic positions by reducing the atomic forces up to 2 mRy per a.u. is considered. It is expected that self-consistency occurs when the total energy (Et) convergence is smaller than 10−5 Ry.
is selected. It is found that a 7 × 7 × 9 k-space grid with 128 points inside the irreducible Brillouin zone wedge gives effective convergence. Additionally, in every case, the complete relaxation of the atomic positions by reducing the atomic forces up to 2 mRy per a.u. is considered. It is expected that self-consistency occurs when the total energy (Et) convergence is smaller than 10−5 Ry.
3 Results and discussion
3.1 Crystal structure
The SGH crystal has an orthorhombic structure with space group Pna21 (no. 33) and its primitive unit cell is composed of 28 atoms with 4/4/20 ions of Sr/Ga/H, having lattice constants a = 8.63559, b = 8.79764, and c = 4.71862.57 The Wyckoff positions of the Sr, Ga, H1, H2, H3, H4, and H5 are 4a (0.9935493, 0.8164696, 0.9540590), 4a (0.8332764, 0.4158104, 0.9539526), 4a (0.2739413, 0.1710615, 0.2299465), 4a (0.0183018, 0.4022104, 0.9541427), 4a (0.9811656, 0.0881250, 0.9541069), 4a (0.2259233, 0.6709108, 0.1778099), and 4a (0.7819938, 0.5906299, 0.9539823), respectively.57 The schematic representation of the crystal structure of SGH in various orientations is shown in Fig. 1. In any material computation science, optimizing the structural parameters is a crucial phase that yields important details about the material under study. The goal is to identify the optimum values for the structural parameters that are required for an accurate description, where the optimized unit cell volume and associated ground-state energy (E0) have been determined. Using the ground-state volume optimization curve, Birch Murnaghan’s equation of states60 was applied to get optimized lattice constants, the compressibility modulus (B), and its first derivative (B′) for the SGH hydrides as follows:| |
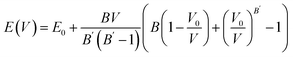 | (1) |
Fig. 2 illustrates the variance of Et as a function of volume and presents the results of the optimization process. The curve generally displays a minimum, indicative of the equilibrium volume at which the system attains the E0 configuration. The optimized lattice parameters of SGH are a = 8.63567, b = 8.79852, and c = 4.71772 Å at the lowest volume, as mentioned in Table 1 along with the calculated B/B′/E0.
 |
| | Fig. 1 Orthorhombic structure of SrGaH5 hydride (purple: GaH4 tetrahedron). | |
 |
| | Fig. 2 Computed total energy (Et) as a function of volume (V0) of the SrGaH5 hydride. | |
Table 1 Computed lattice constants (a, b, c) in Å, volume (V) in a.u3, bulk modulus (B) in GPa, B′, and ground-state energy (E0) in Ry for the SrGaH5 hydride
| SrGaH5 |
a |
b |
c |
V |
B |
B′ |
E0 |
| |
8.63567 |
8.79852 |
4.71772 |
2331.30 |
47.05 |
3.7128 |
−41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 007.124614 007.124614 |
3.2 Thermodynamic stability and desorption temperature
Next, we computed ΔHf, which serves as a crucial thermodynamic parameter for the identification and classification of HSMs. It enables the assessment of the heat generated by the overall hydration reaction, which subsequently facilitates the determination of the Tdes.. The following expression is used to evaluate the ΔHf:61| |
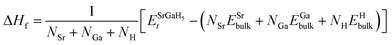 | (2) |
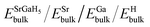 and NSr/NGa/NH represent the Et of the SGH/Sr/Ga/H in their bulk stable phases and total number of atoms of Sr/Ga/H in each case, respectively. Moreover, the hydrogen reference state used in this work is isolated H2. All energies were obtained from fully relaxed structures under each strain condition. Also, the formation enthalpy is reported in units of kJ per mol H2, which is the standard convention adopted in previous theoretical and experimental hydrogen storage studies. Using the same reference state and units allows direct comparison with previously reported hydrides in the literature.62–64 Thus, compared to the ideal value of −40 kJ per mol H2,65,66 our calculated ΔHf of the unstrained SGH system is −48.03 kJ per mol H2, which is a bit higher and may reduce the system’s functionality for the HS application. This is because, due to the substantial energy required for hydrogen absorption or desorption, a system with a reasonable ΔHf is considered the most stable.67 Here, we would like to emphasizes that the present calculations did not include contributions due to zero-point energy (ZPE) and finite-temperature contributions associated with vibrations. All of the ΔHf are reported to be the total energies of the ground states of the DFT calculations at 0 K with no phonon corrections. The ZPE may have a significant role to play in hydride systems because of the low mass of hydrogen atoms, and ZPE and vibrational terms can alter the absolute values of the ΔHf at least modestly and, as a result, the Tdes. calculated can be inaccurate. But, as our work is based on comparative trends due to applied strain, the relative stability and strain-dependent behavior should not be qualitatively affected. Along with this, the hydrogen Tdes. is also a critical factor to consider when choosing a HS system, which must be maintained between 233 and 333 K as stated by Kudiiarov et al.68 Hence, the accurate determination of Tdes. is crucial for assessing the efficiency and appropriateness of a HSM for many applications. It can be determined utilizing the conventional Gibbs equation, as illustrated in the subsequent formula:Under normal conditions of pressure and temperature, ΔG = 0, enabling us to get Tdes. as follows:
and NSr/NGa/NH represent the Et of the SGH/Sr/Ga/H in their bulk stable phases and total number of atoms of Sr/Ga/H in each case, respectively. Moreover, the hydrogen reference state used in this work is isolated H2. All energies were obtained from fully relaxed structures under each strain condition. Also, the formation enthalpy is reported in units of kJ per mol H2, which is the standard convention adopted in previous theoretical and experimental hydrogen storage studies. Using the same reference state and units allows direct comparison with previously reported hydrides in the literature.62–64 Thus, compared to the ideal value of −40 kJ per mol H2,65,66 our calculated ΔHf of the unstrained SGH system is −48.03 kJ per mol H2, which is a bit higher and may reduce the system’s functionality for the HS application. This is because, due to the substantial energy required for hydrogen absorption or desorption, a system with a reasonable ΔHf is considered the most stable.67 Here, we would like to emphasizes that the present calculations did not include contributions due to zero-point energy (ZPE) and finite-temperature contributions associated with vibrations. All of the ΔHf are reported to be the total energies of the ground states of the DFT calculations at 0 K with no phonon corrections. The ZPE may have a significant role to play in hydride systems because of the low mass of hydrogen atoms, and ZPE and vibrational terms can alter the absolute values of the ΔHf at least modestly and, as a result, the Tdes. calculated can be inaccurate. But, as our work is based on comparative trends due to applied strain, the relative stability and strain-dependent behavior should not be qualitatively affected. Along with this, the hydrogen Tdes. is also a critical factor to consider when choosing a HS system, which must be maintained between 233 and 333 K as stated by Kudiiarov et al.68 Hence, the accurate determination of Tdes. is crucial for assessing the efficiency and appropriateness of a HSM for many applications. It can be determined utilizing the conventional Gibbs equation, as illustrated in the subsequent formula:Under normal conditions of pressure and temperature, ΔG = 0, enabling us to get Tdes. as follows:| |
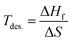 | (4) |
here ΔS represents the change in entropy and its estimated value of ΔS falls within the range of 95–140 J per mol H2 (Zhou et al.69). For the present work, ΔS = 140 J per mol H2 is used. Hence, the corresponding Tdes. of SGH is 343.13 K, which does not fall within the required range (233–333 K).68 Consequently, the objective of this investigation is to enhance the HS features of the material by diminishing its stability. To achieve this objective, we adjusted the hydrostatic strain, ranging from −5% to +5% along the abc-axes, to enhance the de-hydrogenation features (ΔHf and Tdes.) of the material. The hydrostatic strain (ε) is defined as:| |
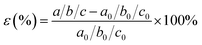 | (5) |
where a/b/c and a0/b0/c0 denote the lattice constants along the x/y/z-axis of the SGH unit cell with and without deformation, respectively. Fig. 3(a) depicts the response of the cell parameters of the SGH system under strain. For an applied compressive strain of −5%, a/b/c reduces until it attains a minimum value of 8.20381/8.35776/4.48269 Å. This signifies a decrease in the unit cell volume, as presented in Fig. 3(b)(left). Conversely, when subjected to tensile strain, a/b/c grows until it attains its maximum value of 9.06737/9.23752/4.95455 Å at +5%, which is accompanied by an augmentation of the cell volume. Fig. 3(b)(right) will be discussed later. Now, we analyze the effects of strain on ΔHf and Tdes.. It is noteworthy that by applying strain, ΔHf/Tdes. decreases and we achieve the desired range at a maximum considered strain range of ±5%, as shown in Fig. 4(a)/(b). As Fig. 4(a) illustrates, the ΔHf exhibits a decrease from −48.03 to −42.93 and −45.18 kJ per mol H2 for −5% compressive and +5% tensile deformations, respectively. The values presented here are considerably closer to the optimal ΔHf of −40 kJ per mol H2 when contrasted with the unstrained structure. These findings are consistent with those presented by Rkhis et al.,54 where the authors found that the thermodynamic stability of the ZrNiH3 perovskite type is diminished under uniaxial/biaxial tensile and compressive stress. Likewise, Benzidi et al.,50 demonstrated that deformation significantly influences the thermodynamic properties of LBH as well. They observed the application of biaxial compressive and tensile deformation results in a reduction of the material’s stability, which means hydrogen reversibility is enhanced under strain. Under −5% compressive strain, the system exhibits maximum hydrogen reversibility. Further to this, the strain-tuned Tdes. values are displayed in Fig. 4(b), which shows a decrease from 343.13 K to 306.67/322.74 K for compressive/tensile deformation of −5%/+5%. Further, compression-induced deformation diminishes the unit-cell volume and the interatomic distances, resulting in enhanced atomic interactions and reducing the material stability. On the other hand, tensile deformation enhances the volume of the unit cell and the interatomic distance, resulting in atomic dispersion and a reduction in both the ΔHf and Tdes.. Therefore, the stability of a material appears to be correlated with its volume, as suggested by Willems et al.,70 who reported a link between stability and 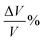 . Similarly, the ΔHf (see Fig. 5(a)) and Tdes. (see Fig. 5(b)) diminish as the
. Similarly, the ΔHf (see Fig. 5(a)) and Tdes. (see Fig. 5(b)) diminish as the 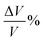 increases in the present case as well. Hence, a large
increases in the present case as well. Hence, a large 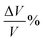 indicates a high material instability.
indicates a high material instability.
 |
| | Fig. 3 Calculated (a) optimized lattice constants (Latt. cons.) in Å, (b-left) unit-cell volume (V) in Å3, and (b-right) volumetric capacity (ρvol.) in g H2 per L as a function of ±5% hydrostatic strain for the SrGaH5 hydride. | |
 |
| | Fig. 4 Computed (a) formation enthalpy (ΔHf) in KJ per mol H2 and (b) desorption temperature (Tdes.) in K for the SrGaH5 hydride as a function of ±5% hydrostatic strain. | |
 |
| | Fig. 5 Computed (a) formation enthalpy (ΔHf) in KJ per mol H2 and (b) desorption temperature (Tdes.) in K for the SrGaH5 hydride as a function of  . . | |
The estimated ΔHf and Tdes. of SGH validate its thermodynamic stability at 0 K and moderate temperature hydrogen release. To investigate the structural changes that occur during desorption, we consider the subsequent thermal decomposition of the system proceeds via the breakdown of Ga–H and Sr–H bonds, resulting in the release of hydrogen gas and the creation of SrH2 along with metallic Ga. While subjected to heat, it undergoes thermal decomposition, as described by the reaction: 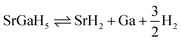 . Along with this, the reverse process, specifically rehydrogenation, takes place under appropriate hydrogen pressure and reduced temperatures as follows: SrH2 + Ga + H2 ⇌ SrGaH5. This reformation is thermodynamically viable and illustrates the reversible characteristics of SGH, highlighting its promise for practical HS applications. Now, the influence of bonding on enhancing HS features under hydrostatic strain of −5%/0%/+5% can be elucidated through the analysis of electron density distributions (EDDs; see Fig. 6(a)/(b)/(c)). Distinct responses are noted for ionic Sr–H and covalent Ga–H bonds. Table 2 illustrates that Ga–H bond lengths exhibit a consistent increase from −5% to +5% strain, leading to a decrease in covalent character (see Fig. 6) and a slight enhancement in H2 mobility. In contrast, Sr–H bonds exhibit asymmetric elongation, where −5% compressive strain leads to greater expansion compared to +5% tensile strain. This phenomenon directly destabilizes the ionic framework by lowering ΔHf (see Fig. 4) and significantly enhances the reversibility of hydrogen. This specific behavior elucidates why a −5% strain optimally enhances HS performance; the reason is that the diminished Sr–H interactions reduce the energy barrier for H2 release, while the maintained Ga–H covalent network facilitates efficient rehydrogenation. Thus, the enhanced reversibility arises from this dual mechanism; weakened ionic bonds promote desorption, whereas preserved covalent bonds ensure cyclability.
. Along with this, the reverse process, specifically rehydrogenation, takes place under appropriate hydrogen pressure and reduced temperatures as follows: SrH2 + Ga + H2 ⇌ SrGaH5. This reformation is thermodynamically viable and illustrates the reversible characteristics of SGH, highlighting its promise for practical HS applications. Now, the influence of bonding on enhancing HS features under hydrostatic strain of −5%/0%/+5% can be elucidated through the analysis of electron density distributions (EDDs; see Fig. 6(a)/(b)/(c)). Distinct responses are noted for ionic Sr–H and covalent Ga–H bonds. Table 2 illustrates that Ga–H bond lengths exhibit a consistent increase from −5% to +5% strain, leading to a decrease in covalent character (see Fig. 6) and a slight enhancement in H2 mobility. In contrast, Sr–H bonds exhibit asymmetric elongation, where −5% compressive strain leads to greater expansion compared to +5% tensile strain. This phenomenon directly destabilizes the ionic framework by lowering ΔHf (see Fig. 4) and significantly enhances the reversibility of hydrogen. This specific behavior elucidates why a −5% strain optimally enhances HS performance; the reason is that the diminished Sr–H interactions reduce the energy barrier for H2 release, while the maintained Ga–H covalent network facilitates efficient rehydrogenation. Thus, the enhanced reversibility arises from this dual mechanism; weakened ionic bonds promote desorption, whereas preserved covalent bonds ensure cyclability.
 |
| | Fig. 6 Computed electron density distribution of the SrGaH5 hydride under (a)−5%, (b) 0%, and (c) +5% hydrostatic strain. | |
Table 2 Computed nine independent elastic constants (Cij) and the interatomic distances between Sr/Ga and H (Sr–H/Ga–H) in Å for the SrGaH5 hydride under −5%/0%/+5% hydrostatic strain
| Strain |
C11 |
C12 |
C13 |
C22 |
C23 |
C33 |
C44 |
C55 |
C66 |
Sr–H |
Ga–H |
| −5% |
76.19 |
28.52 |
27.00 |
76.33 |
29.74 |
103.06 |
21.12 |
28.489 |
24.09 |
2.69 |
1.51 |
| 0% |
47.25 |
13.07 |
11.48 |
50.09 |
11.14 |
67.56 |
14.85 |
22.96 |
18.99 |
2.55 |
1.59 |
| +5% |
35.24 |
10.13 |
11.24 |
34.23 |
10.02 |
51.48 |
9.24 |
14.10 |
10.94 |
2.68 |
1.67 |
Now, the ρvol. and Cwt% (ref. 71) of SGH are calculated to predict their future use in HS as follows:72
| |
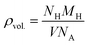 | (6) |
| |
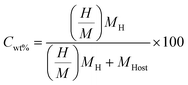 | (7) |
The amount of hydrogen that can be stored in a given volume is indicated by
ρvol. (see
eqn (6)), where
NH/
MH/
NA represents the number of hydrogen atoms/mass of hydrogen/Avogadro’s number. Hence, the computed
ρvol. of the unstrained system is 93.36787 g H
2 per L, which increases to 108.89969 g H
2 per L under −5% compressive strain and decreases to 80.65468 g H
2 per L for +5% tensile strain. The linear trend of
ρvol. against strain is illustrated in
Fig. 3(b)(right), which offers significant insights that the HS capacity of the SGH responds to strain, along with the relationship between modifications in the material’s structure and the potential for HS. The findings indicate that careful control of the strain conditions throughout the HS process is crucial for attaining maximum HS capacity. For practical hydrogen storage systems, the U.S. Department of Energy (DOE) has established system-level targets of approximately 40 g H
2 per L for
ρvol.. These standards have been commonly used to measure the performance of materials. Moreover, the
Cwt% refers to the quantity of hydrogen stored per unit mass of a substance and is calculated in
eqn (7), where
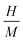
denotes the ratio of hydrogen to material atoms,
MH represents the molar mass of hydrogen, and
MHost signifies the molar mass of the host material. The determined value of
Cwt% is 3.10%, which means that the material exhibits a commendable hydrogen storage capacity, making it a suitable candidate for HS applications. Along with this, compared to associated high-energy complex hydrides, BeAlH
5 has an extremely high gravimetric capacity of 12.19 wt%, which is well beyond the DOE’s ultimate target. CaAlH
5 attains 6.9 wt%, whereas SrAlH
5 attains 4.17 wt%, which is well under the practical standard. Likewise, BeAlH
5 has a
ρvol. of 74 g H
2 L, which is much higher than the DOE 2025 objective of 40 g H
2 per L.
73 Thus, compared to these systems, SrGaH
5 has a
Cwt% of 3.10% and
ρvol. of 93.36787 g H
2 per L. It has a lower capacity than BeAlH
5 but is comparable to CaAlH
5.
3.3 Mechanical stability
The elastic constant quantifies the stiffness of a material against applied strain and is essential for evaluating the material’s mechanical behavior. In this context, nine independent elastic constants (Cij) for the orthorhombic structure of SGH are computed, utilizing conventional strain and stress relationships74,75 according to the fundamental Born criteria, expressed as: C11 > 0, C11C22 > C122, C11C22C33 + 2C12C13C23 − C11C232 − C22C132 > 0, and C44 > 0; C55 > 0; C66 > 0.76 The computed values of Cij (see Table 2) can be utilized in determining several physical parameters, including the Bulk modulus (B), Shear modulus (G), and Young’s modulus (Y). B quantifies the resistance to volumetric change under applied pressure. The highest value of B is obtained at −5%, denoting the maximum resistance to compression. Also, it has the maximum G/Y of 25.75/65.31, enabling it to endure more transverse deformation as compared to 0% and +5% (see Fig. 7).
 |
| | Fig. 7 Computed Young’s modulus (Y), Bulk modulus (B), and Shear modulus (G) of the SrGaH5 hydride under −5%/0%/+5% hydrostatic strain. | |
Furthermore, the brittleness or ductility of a material may be ascertained using the 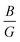 ratio. A brittle material possesses a
ratio. A brittle material possesses a 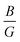 below 1.75, whereas a ductile one has a ratio over the threshold of 1.75.77 The analyzed material under 0% and +5% strain exhibits brittleness, possessing a
below 1.75, whereas a ductile one has a ratio over the threshold of 1.75.77 The analyzed material under 0% and +5% strain exhibits brittleness, possessing a  below 1.75, and is ductile under −5%, as displayed in Fig. 8(a). Similarly, the
below 1.75, and is ductile under −5%, as displayed in Fig. 8(a). Similarly, the 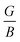 ratio (also known as Pugh’s modulus), as presented in Fig. 8(b), can be utilized to ascertain the bonding aspects of a material. If the
ratio (also known as Pugh’s modulus), as presented in Fig. 8(b), can be utilized to ascertain the bonding aspects of a material. If the  is around 0.6, ionic bonding prevails and conversely with a ratio of 1.1, covalent bonding is dominant.78 Now, ν can be utilized to ascertain the bonding features of a material. It quantifies the expansion or compression in the perpendicular axes relative to the direction of compression or stretching. If its value is 0.1, the material exhibits dominantly covalent bonding, whereas at around 0.25, it demonstrates principally ionic bonding.79 From Fig. 8(b), the analyzed compound for the strain range −5% to +5% has a ν of around 0.25, indicating a dominance of ionic bonding. Hence, for the examined material under 0% and +5% strain, the dominance of the ionic bonding is confirmed by the
is around 0.6, ionic bonding prevails and conversely with a ratio of 1.1, covalent bonding is dominant.78 Now, ν can be utilized to ascertain the bonding features of a material. It quantifies the expansion or compression in the perpendicular axes relative to the direction of compression or stretching. If its value is 0.1, the material exhibits dominantly covalent bonding, whereas at around 0.25, it demonstrates principally ionic bonding.79 From Fig. 8(b), the analyzed compound for the strain range −5% to +5% has a ν of around 0.25, indicating a dominance of ionic bonding. Hence, for the examined material under 0% and +5% strain, the dominance of the ionic bonding is confirmed by the  ratio and ν.
ratio and ν.
 |
| | Fig. 8 Computed (a) Pugh’s ratio 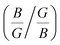 and (b) Poisson’s ratio (ν) of the SrGaH5 hydride under −5%/0%/+5% hydrostatic strain. and (b) Poisson’s ratio (ν) of the SrGaH5 hydride under −5%/0%/+5% hydrostatic strain. | |
Along with this, utilizing the ELATE tool,80,81 we have calculated the elastic anisotropy features of the material subjected to strain. Fig. 9 depicts the fluctuation of anisotropic elastic aspects, specifically Y, linear compressibility (β), G, and ν, subject to various applied strain values. It is found that Y demonstrates directional dependence (see Fig. 9(a)), exhibiting a significant anisotropy under compressive strain and displays a more uniform distribution under strain, signifying strain-induced stiffening or softening in certain crystallographic orientations. Fig. 9(b) shows the directional β, which exhibits an increasing amplitude and anisotropy with the application of tensile strain, indicating that the material becomes more compressible along certain axes under tension. The G in Fig. 9(c) displays considerable anisotropy across the strain range with marked variations in magnitude and distribution, reflecting the impact of strain on the material resistance to deformation. Further, ν in Fig. 9(d) indicates that directional variations reflect strain-induced alterations in lateral deformation behavior. Hence, Fig. 9 highlights that strain engineering not only adjusts the elastic constants but also enhances the anisotropic mechanical response of the materials (Table 3).
 |
| | Fig. 9 Directionally dependent (a) Young’s modulus (Y), (b) linear compressibility (β), (c) Shear modulus (G), and (d) Poisson’s ratio (ν) for the SrGaH5 hydride under −5% (left column)/0% (middle column)/+5% (right column) hydrostatic strain. | |
Table 3 Computed minimum/maximum values of Young’s modulus (Ymin/Ymax) in GPa, linear compressibility (βmin/βmax) in TPa−1, Shear modulus (Gmin/Gmax) in GPa, Poisson’s ratio (νmin/νmax), and their corresponding anisotropic factors (A) for the SrGaH5 hydride under −5%/0%/+5% hydrostatic strain
| Strain |
Young’s modulus |
Linear compressibility |
Shear modulus |
Poisson’s ratio |
| Ymin |
Ymax |
A |
βmin |
βmax |
A |
Gmin |
Gmax |
A |
νmin |
νmax |
A |
| −5% |
57.14 |
87.62 |
1.53 |
5.23 |
8.28 |
1.58 |
21.12 |
30.41 |
1.44 |
0.17 |
0.39 |
2.27 |
| 0 |
39.51 |
63.40 |
1.60 |
9.98 |
14.90 |
1.49 |
14.85 |
22.96 |
1.54 |
0.19 |
0.35 |
3.98 |
| +5% |
25.60 |
46.43 |
1.81 |
11.34 |
20.28 |
1.79 |
9.24 |
15.37 |
1.66 |
0.14 |
0.42 |
2.96 |
3.4 Dynamical stability
Next, to verify the dynamical stability of the system, phonon computations were conducted. Fig. 10(a)/(b)/(c) shows a graph of the computed phonon spectrum under −5%/0%/+5% hydrostatic strain. The number of phonon branches in a unit cell is exactly proportional to three times the number of atoms, with each atom normally contributing three phonon branches. It exhibits 3 acoustic and 3n − 3 optical modes that make up the 3n total branches.82 The SGH structure includes 84 vibrational modes, including 3 acoustic and 81 optical modes, and its fundamental unit cell has 28 atoms. The three calculated phonon dispersion curves for the −5%/0%/+5% strain (see Fig. 10) do not contain any negative frequencies, which confirms that they are dynamically stable as well. Furthermore, Fig. 11 displays the computed phonon lifetime of the unstrained motif, which shows a high correlation between the phonon spectra (see Fig. 10(b)) and the phonon lifetime. The linear dispersion of the acoustic modes around Γ, which has limited scattering phase-space and large group velocities, is the source of the long lifetime of low-frequency phonons (<0.3 THz). The flat optical branches, on the other hand, have low group velocities and high density of states, which encourage rapid anharmonic decay, and are consistent with the short lifetime of high-frequency phonons (>0.3 THz).
 |
| | Fig. 10 Computed phonon dispersion curves of the SrGaH5 hydride under (a) −5%, (b) 0% and (c) +5% hydrostatic strain. | |
 |
| | Fig. 11 Computed phonon lifetime of the unstrained SrGaH5 hydride. | |
3.5 Electronic structure
To qualitatively illustrate the variations in the electronic structure of the system with strain, we depicted the calculated Eg as a function of deformation in Fig. 12. When compressive strain is altered from 0% to −5%, a linear rise in Eg from 2.99 to 3.58 eV is found. In contrast, as the tensile strain is adjusted from 0% to +5%, Eg lowers from 2.99 to 2.43 eV. For a quantitative analysis, Fig. 13(a′)/(b′)/(c′) demonstrates the spin-polarized total density of states (TDOS) under varying hydrostatic strain of −5%/0%/+5%. In all three cases, a large Eg between the valence and conduction states is seen, affirming the material’s insulating aspect. The progressive reduction of the Eg with augmented tensile strain indicates improved carrier availability around the Fermi level. Along with this, Fig. 13(a)/(b)/(c) illustrates the corresponding electronic band structures for −5%/0%/+5% hydrostatic strain, which closely aligns with the TDOS measurements and further confirms the insulating behavior of the system. All the band structures indicate that Eg is consistently indirect at each strain level because the conduction band minima and valence band maxima are situated at distinct symmetry locations.
 |
| | Fig. 12 Computed energy gap (Eg) of the SrGaH5 hydride under ±5% hydrostatic strain. | |
 |
| | Fig. 13 Computed spin-polarized band structures/total density of states under (a/a′) −5%, (b/b′) 0%, and (c/c′) +5% hydrostatic strains for the SrGaH5 hydride. | |
3.6 Thermoelectric response
Finally, TE aspects including electrical conductivity per relaxation time 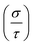 , electronic thermal conductivity per relaxation time
, electronic thermal conductivity per relaxation time 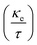 , Seebeck coefficient (S), power factor
, Seebeck coefficient (S), power factor 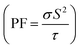 , lattice thermal conductivity per relaxation time
, lattice thermal conductivity per relaxation time 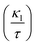 , and figure of merit
, and figure of merit 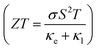 of the SGH hydride are thoroughly examined for a temperature range of 100–800 K with a consistent τ of 10−14 s using the BoltzTraP code83 and under various hydrostatic strains. Here, it is worth mentioning that we performed an extensive literature survey and found that the relaxation times for similar materials typically fall within 10−13 to 10−15 s. Therefore, selecting a constant relaxation time of 10−14 s represents a reasonable and widely accepted approximation for comparative ZT calculations, allowing us to capture the general thermoelectric trends without introducing undue uncertainty. Thus, a choice of a constant relaxation time of 10−14 s can be considered a reasonable and generally accepted approximation to comparative ZT calculations to allow us to describe the overall trends in thermoelectric behavior with a minimum amount of uncertainty. Owing to limitations in the BoltzTraP approach,
of the SGH hydride are thoroughly examined for a temperature range of 100–800 K with a consistent τ of 10−14 s using the BoltzTraP code83 and under various hydrostatic strains. Here, it is worth mentioning that we performed an extensive literature survey and found that the relaxation times for similar materials typically fall within 10−13 to 10−15 s. Therefore, selecting a constant relaxation time of 10−14 s represents a reasonable and widely accepted approximation for comparative ZT calculations, allowing us to capture the general thermoelectric trends without introducing undue uncertainty. Thus, a choice of a constant relaxation time of 10−14 s can be considered a reasonable and generally accepted approximation to comparative ZT calculations to allow us to describe the overall trends in thermoelectric behavior with a minimum amount of uncertainty. Owing to limitations in the BoltzTraP approach, 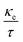 can be computed but
can be computed but 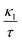 cannot. Therefore, we independently determined the
cannot. Therefore, we independently determined the 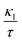 utilizing Slack’s equation as follows:84
utilizing Slack’s equation as follows:84| |
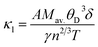 | (8) |
Mav., n, T, γ, θD, and δ stand for the average atomic mass, number of atoms in the unit cell, absolute temperature, Grüneisen parameter, Debye temperature, and cubic root of the average atomic volume, respectively. Using ν, the Grüneisen parameter (γ) is found as:| |
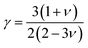 | (9) |
while A is calculated as:85| |
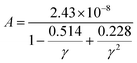 | (10) |
where θD can be established as:86| |
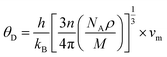 | (11) |
wherein h, n, ρ, M, NA, kB, and vm serve as Plank’s constant, the total number of atoms per unit cell, density, molecular weight, Avogadro’s number, the Boltzmann constant, and sound velocity, respectively. The average velocity of sound (vm) is calculated using:| |
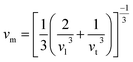 | (12) |
The longitudinal/transverse velocity is vl/vt, which can be calculated as:| |
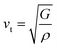 | (13) |
| |
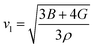 | (14) |
and the corresponding ZT as:| |
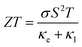 | (15) |
The material exhibits significant TE capabilities owing to its considerable Eg and degeneracy in electronic states. Fig. 14(a) illustrates the influence of temperature on  , which increases from 0.21/0.11/0.12 × 1019 1 Ωm−1 s−2 at 100 K to 0.37/0.36/0.64 × 1019 1 Ωm−1 s−2 at 750 K under −5%/0%/+5% hydrostatic strain. σ is reliant upon the quantity of free carriers. Hence, the rising temperature elevates the quantity of free carriers owing to bond dissociation, thus providing energy to free conduction electrons. Consequently,
, which increases from 0.21/0.11/0.12 × 1019 1 Ωm−1 s−2 at 100 K to 0.37/0.36/0.64 × 1019 1 Ωm−1 s−2 at 750 K under −5%/0%/+5% hydrostatic strain. σ is reliant upon the quantity of free carriers. Hence, the rising temperature elevates the quantity of free carriers owing to bond dissociation, thus providing energy to free conduction electrons. Consequently, 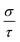 increases with increasing temperature as seen in Fig. 14(a). Among the strain conditions, tensile strain (+5%) provides the greatest
increases with increasing temperature as seen in Fig. 14(a). Among the strain conditions, tensile strain (+5%) provides the greatest 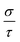 values, which signifies improved charge carrier mobility or concentration. Conversely, compressive strain (−5%) yields the lowest conductivity, probably owing to enhanced scattering or a decrease in band dispersion. To further assess the efficiency of the material, both κe and κl were examined at different strain values. The two contributions directly affect the overall thermal conductivity (κ = κe + κl) and consequently the dimensionless ZT. The κe (see Fig. 14(b)) exhibits a monotonic rise with elevated temperature throughout, at all strain levels. This tendency is predicted due to the greater mobility of the charge carriers at elevated temperature. It increases from 0.11/0.08/0.09 × 1014 W mK−1 s−1 at 100 K to 0.39/0.40/1.54 × 1014 W mK−1 s−1 under −5%/0%/+5% hydrostatic strain. In addition, the computed κl decreases with increasing temperature due to enhanced phonon–phonon scattering at elevated thermal energy (see Fig. 14(c)). It decreases from 1.76/2.01/0.9 × 1014 W mK−1 s−1 at 100 to 0.23/0.26/0.12 W mK−1 s−1 at 750 K and only reduced further with increasing temperature under −5%/0%/+5% strain. Tensile strain (+5%) results in a significant decrease of κl, probably due to enhanced lattice distortion and phonon scattering, which impede thermal conduction across the lattice, which has a positive impact on the ZT value. In contrast, compressive strain (−5%) exhibits marginally elevated values, signifying a partial restoration of lattice order and decreased phonon dispersion.
values, which signifies improved charge carrier mobility or concentration. Conversely, compressive strain (−5%) yields the lowest conductivity, probably owing to enhanced scattering or a decrease in band dispersion. To further assess the efficiency of the material, both κe and κl were examined at different strain values. The two contributions directly affect the overall thermal conductivity (κ = κe + κl) and consequently the dimensionless ZT. The κe (see Fig. 14(b)) exhibits a monotonic rise with elevated temperature throughout, at all strain levels. This tendency is predicted due to the greater mobility of the charge carriers at elevated temperature. It increases from 0.11/0.08/0.09 × 1014 W mK−1 s−1 at 100 K to 0.39/0.40/1.54 × 1014 W mK−1 s−1 under −5%/0%/+5% hydrostatic strain. In addition, the computed κl decreases with increasing temperature due to enhanced phonon–phonon scattering at elevated thermal energy (see Fig. 14(c)). It decreases from 1.76/2.01/0.9 × 1014 W mK−1 s−1 at 100 to 0.23/0.26/0.12 W mK−1 s−1 at 750 K and only reduced further with increasing temperature under −5%/0%/+5% strain. Tensile strain (+5%) results in a significant decrease of κl, probably due to enhanced lattice distortion and phonon scattering, which impede thermal conduction across the lattice, which has a positive impact on the ZT value. In contrast, compressive strain (−5%) exhibits marginally elevated values, signifying a partial restoration of lattice order and decreased phonon dispersion.
 |
| | Fig. 14 Various computed thermoelectric parameters under −5%/0%/+5% hydrostatic strain for the SrGaH5 hydride. | |
Fig. 14(d) illustrates the dependence of S on temperature, where its value decreases from 191.03/237.81/219.04 at 100 K to 104.77/97/140 at 750 K. All strained configurations have a positive S, hence confirming p-type conduction. The magnitude of S diminishes with increasing temperature, aligning with the features of degenerate semiconductors. The compressive strain condition has the greatest S, indicating a beneficial alteration in the electronic structure that improves thermopower, likely attributable to a shift in the density of states around the Fermi level. Additionally, Fig. 14(e) displays the 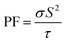 , which combines both σ and S. The value of PF at 100 K is 0.76/0.63/0.57 × 1011 W mK−2 s−1 and increases to 0.40/0.34/1.27 × 1011 W mK−2 s−1 under −5%/0%/+5% strain. Likewise, the dimensionless quantity ZT is employed to evaluate the performance of a TE material. Elevated ZT yields enhanced TE reliability, indicating that a greater amount of heat is transformed into electricity. The calculated ZT, as illustrated in Fig. 14(f), demonstrates a progressive increase with temperature from 0.04/0.03/0.05 at 100 K to 0.48/0.37/0.57 at 750 K. However, when the system is subjected to +5% tensile strain, it displays the greatest ZT of 0.61 at 550 K, whereas the unstrained and −5% compressed system demonstrated lower TE efficiency. Hence, these findings highlight the beneficial impact of tensile strain on enhancing TE performance by concurrently augmenting electrical transport and preserving an advantageous S. Finally, it is important to note that a +5% hydrostatic strain is more like a tensile pressure of roughly 3–5 GPa, assuming that there is a normal range of B for such materials. Even though +5% strain is near the upper limit of elasticity of bulk systems, +5% strain can be a useful theoretical upper limit to determine sensitivity and tunability of strain. Thus, the estimation of the improvement under the +5% strain gives a good insight into the possible practical strain-engineering approaches.
, which combines both σ and S. The value of PF at 100 K is 0.76/0.63/0.57 × 1011 W mK−2 s−1 and increases to 0.40/0.34/1.27 × 1011 W mK−2 s−1 under −5%/0%/+5% strain. Likewise, the dimensionless quantity ZT is employed to evaluate the performance of a TE material. Elevated ZT yields enhanced TE reliability, indicating that a greater amount of heat is transformed into electricity. The calculated ZT, as illustrated in Fig. 14(f), demonstrates a progressive increase with temperature from 0.04/0.03/0.05 at 100 K to 0.48/0.37/0.57 at 750 K. However, when the system is subjected to +5% tensile strain, it displays the greatest ZT of 0.61 at 550 K, whereas the unstrained and −5% compressed system demonstrated lower TE efficiency. Hence, these findings highlight the beneficial impact of tensile strain on enhancing TE performance by concurrently augmenting electrical transport and preserving an advantageous S. Finally, it is important to note that a +5% hydrostatic strain is more like a tensile pressure of roughly 3–5 GPa, assuming that there is a normal range of B for such materials. Even though +5% strain is near the upper limit of elasticity of bulk systems, +5% strain can be a useful theoretical upper limit to determine sensitivity and tunability of strain. Thus, the estimation of the improvement under the +5% strain gives a good insight into the possible practical strain-engineering approaches.
4 Conclusion
In summary, a first-principles approach utilizing density functional theory was conducted to examine the impact of hydrostatic deformation on the structural, hydrogen storage (HS), mechanical, dynamical, electronic, and thermoelectric (TE) characteristics of the complex SrGaH5 hydride. In comparison to the strain-free system, the deformed motif exhibits enhanced ΔHf (formation enthalpy) and Tdes. (desorption temperature) due to strain energy contributions. Specifically, under compressive/tensile tensile strain of −5%/+5%, the ΔHf reduces from −48.03 kJ per mol H2 to almost close to an ideal value of −42.93/−45.18 kJ per mol H2 and the corresponding Tdes. varies from 343.13 K to 306.67/322.74 K, which lies is in the desired range for HS applications. Additionally, the gravimetric hydrogen storage capacity of the motif is 3.10% and the volumetric hydrogen capacity is 108.89/93.36/80.65 g H2 per L under −5%/0%/+5% strain, further confirming the system’s potential for utilization in HS. Moreover, the electronic structure calculations confirm the insulating behavior of the system, having an indirect band gap of 3.58/2.99/2.43 eV under −5%/0%/+5% hydrostatic strain. Finally, it is found that the TE performance is significantly enhanced under tensile strain with a notable figure of merit of 0.61 achieved at 550 K under +5% strain, indicating superior heat-to-electricity conversion efficiency and improved carrier transport in the studied hydride. Hence, present findings suggest that the examined hydride is not only a promising candidate for HS but also for integration into various energy conversion systems.
Author contributions
Samia Shahzadi: writing – original draft, investigations, formal analysis, data curation A. Ibrahim: visualization, validation, resources, formal analysis. S. Nazir: writing – review and editing, validation, supervision, project administration, conceptualization.
Conflicts of interest
The authors declare no competing interests.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgements
The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through Large Research Project under grant number RGP2/713/46.
Notes and references
- A. S. Oberoi, P. Nijhawan and P. Singh, Energies, 2018, 12, 82 CrossRef.
- C. H. Tan, P. L. Show, J.-S. Chang, T. C. Ling and J. C.-W. Lan, Biotechnol. Adv., 2015, 33, 1219–1227 CrossRef CAS PubMed.
- S. Y. Lee, R. Sankaran, K. W. Chew, C. H. Tan, R. Krishnamoorthy, D.-T. Chu and P.-L. Show, BMC Energy, 2019, 1, 1–22 Search PubMed.
- R. Amirante, E. Cassone, E. Distaso and P. Tamburrano, Energy Convers. Manage., 2017, 132, 372–387 CrossRef CAS.
- I. P. Jain, Int. J. Hydrogen Energy, 2009, 34, 7368–7378 Search PubMed.
- Z. M. Wang, J. J. Li, S. Tao, J. Q. Deng, H. Zhou and Q. Yao, J. Alloys Compd., 2016, 660, 402–406 CrossRef CAS.
- A. Gencer and G. Surucu, Int. J. Hydrogen Energy, 2019, 44, 15173–15182 Search PubMed.
- S. Benlamari, S. Amara Korba, S. Lakel, H. Meradji, S. Ghemid and F. El Haj Hassan, Int. J. Hydrogen Energy, 2016, 30, 1650003 CAS.
- Y. Bouhadda, Y. Boudouma, N. Fennineche and A. Bentabet, Int. J. Hydrogen Energy, 2010, 71, 1264–1268 Search PubMed.
- J. Andersson and S. Grönkvist, Int. J. Hydrogen Energy, 2019, 44, 11901–11919 CrossRef CAS.
- P. Muthukumar, A. Kumar, M. Afzal, S. Bhogilla, P. Sharma, A. Parida, S. Jana, E. A. Kumar, R. K. Pai and I. Jain, Int. J. Hydrogen Energy, 2023, 48, 33223–33259 CrossRef CAS.
- I. Jain, C. Lal and A. Jain, Int. J. Hydrogen Energy, 2010, 35, 5133–5144 CrossRef CAS.
- X. Zhang, Y. Liu, Z. Ren, X. Zhang, J. Hu, Z. Huang, Y. Lu, M. Gao and H. Pan, Energy Environ. Sci., 2021, 14, 2302–2313 Search PubMed.
- B. Rehmat, M. Rafiq, Y. Javed, Z. Irshad, N. Ahmed and S. Mirza, Int. J. Hydrogen Energy, 2017, 42, 10038–10046 Search PubMed.
- L. Schlapbach and A. Züttel, Nature, 2001, 414, 353–358 CrossRef CAS PubMed.
- M. Al-Zareer, I. Dincer and M. A. Rosen, Energy Convers. Manage., 2018, 157, 600–618 CrossRef CAS.
- I. A. Gondal, Sustainable Energy Fuels, 2019, 3, 1468–1489 RSC.
- Y. Bouhadda, Y. Boudouma, N.-E. Fennineche and A. Bentabet, J. Phys. Chem. Solids, 2010, 71, 1264–1268 CrossRef CAS.
- K. T. Moller, D. Sheppard, D. B. Ravnsbaek, C. E. Buckley, E. Akiba, H.-W. Li and T. R. Jensen, Energies, 2017, 10, 1645 CrossRef.
- K. Yvon, Chimia, 1998, 52, 613 Search PubMed.
- V. A. Yartys and M. V. Lototskyy, J. Alloys Compd., 2022, 916, 165219 Search PubMed.
- A. R. Paul, S. Mehla and S. Bhargava, Small, 2025, 21, 2408889 CrossRef CAS PubMed.
- G. Sujan, Z. Pan, H. Li, D. Liang and N. Alam, Crit. Rev. Solid State Mater. Sci., 2020, 45, 410–427 CrossRef CAS.
- T. He, H. Cao and P. Chen, Adv. Mater., 2019, 31, 1902757 CrossRef CAS PubMed.
- Y. Yürüm, A. Taralp and T. N. Veziroglu, Int. J. Hydrogen Energy, 2009, 34, 3784–3798 CrossRef.
- P. Liu, J. Liang, R. Xue, Q. Du and M. Jiang, Int. J. Hydrogen Energy, 2019, 44, 27853–27861 CrossRef CAS.
- R. Zacharia and S. U. Rather, J. Nanomater., 2015, 2015, 914845 CrossRef.
- A. Schneemann, J. L. White, S. Kang, S. Jeong, L. F. Wan, E. S. Cho, T. W. Heo, D. Prendergast, J. J. Urban and B. C. Wood, et al., Chem. Rev., 2018, 118, 10775–10839 CrossRef CAS PubMed.
- M. He, Y. Zhang, L. Gong, Y. Zhou, X. Song, W. Zhu, M. Zhang and Z. Zhang, Int. J. Hydrogen Energy, 2019, 44, 28206–28226 Search PubMed.
- A. Siddique, A. Khalil, B. S. Almutairi, M. B. Tahir, T. Ahsan, A. Hannan, H. E. Ali, H. Alrobei and M. Alzaid, Chem. Phys., 2023, 568, 111851 CrossRef CAS.
- M. Xiong, C. Lin, Y. Xue and D. Huang, Inorg. Chem. Commun., 2025, 176, 114264 CrossRef CAS.
- Y. Song, M. K. Shahzad, S. Hussain, A. Farrukh, M. Riaz, H. Sattar, G. Khan, G. A. Ashraf, S. M. Ali and M. Alam, Int. J. Hydrogen Energy, 2024, 79, 1472–1482 CrossRef CAS.
- B. Bogdanović and M. Schwickardi, J. Alloys Compd., 1997, 253, 1–9 CrossRef.
- G. Meisner, G. Tibbetts, F. Pinkerton, C. Olk and M. Balogh, J. Alloys Compd., 2002, 337, 254–263 Search PubMed.
- A. Züttel, S. Rentsch, P. Fischer, P. Wenger, P. Sudan, P. Mauron and C. Emmenegger, J. Alloys Compd., 2003, 356, 515–520 CrossRef.
- O. Løvvik, S. M. Opalka, H. W. Brinks and B. C. Hauback, Phys. Rev. B:Condens. Matter Mater. Phys., 2004, 69, 134117 CrossRef.
- P. Chen and M. Zhu, Mater. Today, 2008, 11, 36–43 Search PubMed.
- B. B. Kitchen, N. Verdal, T. J. Udovic, J. J. Rush, M. R. Hartman and D. J. DeVries, J. Solid State Chem., 2013, 203, 51–54 CrossRef CAS.
- P. Vajeeston, P. Ravindran, A. Kjekshus and H. Fjellvåg, J. Alloys Compd., 2005, 387, 97–104 CrossRef CAS.
- V. Balema, J. Wiench, K. Dennis, M. Pruski and V. Pecharsky, J. Alloys Compd., 2001, 329, 108–114 Search PubMed.
- A. Finholt, A. Bond Jr and H. Schlesinger, J. Am. Chem. Soc., 1947, 69, 1199–1203 CrossRef CAS.
- S. Orimo, Y. Nakamori and A. Züttel, Mater. Sci. Eng., B, 2004, 108, 51–53 CrossRef.
- N. S. Norberg, T. S. Arthur, S. J. Fredrick and A. L. Prieto, J. Am. Chem. Soc., 2011, 133, 10679–10681 Search PubMed.
- F. Zhang, Y. Wang and M. Chou, J. Am. Chem. Soc., 2012, 116, 18663–18668 CAS.
- C. Mui, S. F. Bent and C. B. Musgrave, J. Phys. Chem. B, 2004, 108, 6336–6350 CrossRef CAS PubMed.
- H. Benzidi, M. Lakhal, M. Abdellaoui, M. Garara, A. Benyoussef, M. Loulidi, M. Hamedoun and O. Mounkachi, et al., Int. J. Hydrogen Energy, 2019, 44, 16793–16802 Search PubMed.
- E. German and R. Gebauer, J. Phys. Chem. C, 2016, 120, 4806–4812 CrossRef CAS.
- B. W. Chen and M. Mavrikakis, Nano Energy, 2019, 63, 103858 Search PubMed.
- F. S. Sangsefidi and M. Salavati-Niasari, ACS Appl. Energy Mater., 2018, 1, 4840–4848 CrossRef CAS.
- H. Benzidi, M. Lakhal, A. Benyoussef, M. Hamedoun, M. Loulidi and O. Mounkachi, et al., Int. J. Hydrogen Energy, 2017, 42, 19481–19486 CrossRef CAS.
- J.-J. Tang, X.-B. Yang, M. Chen, M. Zhu and Y.-J. Zhao, J. Phys. Chem. C, 2012, 116, 14943–14949 Search PubMed.
- J. Zhang, Y. Zhou, Z. Ma, L. Sun and P. Peng, Int. J. Hydrogen Energy, 2013, 38, 3661–3669 Search PubMed.
- M. Garara, H. Benzidi, M. Abdellaoui, M. Lakhal, A. Benyoussef, O. Mounkachi and M. Loulidi, et al., Mater. Chem. Phys., 2020, 254, 123417 CrossRef CAS.
- M. Rkhis, S. Laasri, S. Touhtouh, E. Hlil, M. Bououdina, R. Ahuja, K. Zaidat, S. Obbade and A. Hajjaji, Int. J. Hydrogen Energy, 2022, 47, 3022–3032 CrossRef CAS.
- C. Weidenthaler, T. J. Frankcombe and M. Felderhoff, Inorg. Chem., 2006, 45, 3849–3851 CrossRef CAS PubMed.
- A. Klaveness, P. Vajeeston, P. Ravindran, H. Fjellvåg and A. Kjekshus, Phys. Rev. B:Condens. Matter Mater. Phys., 2006, 73, 094122 CrossRef.
- A. Klaveness, O. Swang, A. Kjekshus and H. Fjellvåg, Inorg. Chem., 2006, 45, 10698–10701 CrossRef CAS PubMed.
- P. Blaha, K. Schwarz, P. Sorantin and S. Trickey, Comput. Phys. Commun., 1990, 59, 399–415 CrossRef CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865 Search PubMed.
- F. D. Murnaghan, Proc. Natl. Acad. Sci. U. S. A., 1944, 30, 244–247 Search PubMed.
- C. Yang, Y. Duan, X. Wang, M. Peng, L. Shen and H. Qi, Mater. Today Commun., 2022, 32, 103962 Search PubMed.
- A. Arharbi, H. Jebari and H. Ez-Zahraouy, J. Energy Storage, 2025, 106, 114903 Search PubMed.
- A. Arharbi and H. Ez-Zahraouy, Int. J. Hydrogen Energy, 2024, 65, 262–270 CrossRef CAS.
- H. Ez-Zahraouy, et al., Int. J. Hydrogen Energy, 2024, 87, 678–685 CrossRef.
- M. Ramzan, S. Lebegue and R. Ahuja, Int. J. Hydrogen Energy, 2010, 35, 10373–10376 CrossRef CAS.
- R. Gremaud, C. P. Broedersz, D. M. Borsa, A. Borgschulte, P. Mauron, H. Schreuders, J. H. Rector, B. Dam and R. Griessen, Adv. Mater., 2007, 19, 2813–2817 Search PubMed.
- Y. Liu, Y. Jiang, R. Zhou and J. Feng, J. Alloys Compd., 2014, 582, 500–504 CrossRef CAS.
- V. Kudiiarov, J. Lyu, O. Semyonov, A. Lider, S. Chaemchuen and F. Verpoort, Appl. Mater. Today, 2021, 25, 101208 Search PubMed.
- C. Zhou, R. C. Bowman Jr, Z. Z. Fang, J. Lu, L. Xu, P. Sun, H. Liu, H. Wu and Y. Liu, ACS Appl. Mater. Interfaces, 2019, 11, 38868–38879 CrossRef CAS PubMed.
- J. Willems, Z. Phys. Chem., 1986, 147, 231 CrossRef.
- N. Xu, Y. Chen, S. Chen, S. Li and W. Zhang, Int. J. Hydrogen Energy, 2024, 50, 114–122 CrossRef.
- M. Baaddi, R. Chami, O. Baalla, S. E. Quaoubi, A. Saadi, L. E. H. Omari and M. Chafi, Environ. Sci. Pollut. Res., 2024, 31, 62056–62064 Search PubMed.
- I. Rhrissi, A. Bouhmouche and R. Moubah, Int. J. Hydrogen Energy, 2025, 117, 146–157 CrossRef CAS.
- X. Wu, D. Vanderbilt and D. Hamann, Phys. Rev. B:Condens. Matter Mater. Phys., 2005, 72, 035105 Search PubMed.
- S. Shang, Y. Wang and Z.-K. Liu, Appl. Phys. Lett., 2007, 90, 101909 CrossRef.
- F. Mouhat and F.-X. Coudert, Phys. Rev. B:Condens. Matter Mater. Phys., 2014, 90, 224104 CrossRef.
- S. Pugh, Lond. Edinb. Dubl. Phil. Mag., 1954, 45, 823–843 CAS.
- G. Surucu, Mater. Chem. Phys., 2018, 203, 106–117 CrossRef CAS.
- A. Gencer and G. Surucu, Mater. Res. Express, 2018, 5, 076303 CrossRef.
- S. Yalameha, Z. Nourbakhsh and D. Vashaee, Comput. Phys. Commun., 2022, 271, 108195 CrossRef CAS.
- C. Ekuma and Z.-L. Liu, Comput. Phys. Commun., 2024, 300, 109161 CrossRef CAS.
- Y. Lv, X. Zhang and W. Jiang, Ceram. Int., 2018, 44, 128–135 CrossRef CAS.
- G. K. H. Madsen and D. J. Singh, Comput. Phys. Commun., 2006, 175, 67–71 CrossRef CAS.
- J. Wei, Y. Guo and G. Wang, RSC Adv., 2023, 13, 11513–11524 RSC.
- S. A. Sofi and D. C. Gupta, Int. J. Energy Res., 2021, 45, 4652–4668 CrossRef CAS.
- O. L. Anderson, J. Phys. Chem. Solids, 1963, 24, 909–917 CrossRef CAS.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article *a
*a
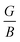 ratio and Poisson’s ratio further imply the existence of ionic bonding character in the material. Further, the non-negative phonon frequency curves validate the dynamical stability of the structure. The electronic structure calculations predicted an insulating behavior with an indirect energy gap of 3.58–2.43 eV for the considered strain range. Surprisingly, the thermoelectric performance of the system has been significantly improved under tensile strain as the figure of merit reaches a high value of 0.61 at 550 K under +5% strain. This is because the mean free path of phonons is reduced as the temperature rises due to an increase in phonon scattering driven by greater lattice vibrations, which causes a decrease in lattice thermal conductivity. Hence, the hydrogen storage capabilities and other aspects of the material indicate that it is an appropriate candidate for HS and might be crucial in various energy-generating applications.
ratio and Poisson’s ratio further imply the existence of ionic bonding character in the material. Further, the non-negative phonon frequency curves validate the dynamical stability of the structure. The electronic structure calculations predicted an insulating behavior with an indirect energy gap of 3.58–2.43 eV for the considered strain range. Surprisingly, the thermoelectric performance of the system has been significantly improved under tensile strain as the figure of merit reaches a high value of 0.61 at 550 K under +5% strain. This is because the mean free path of phonons is reduced as the temperature rises due to an increase in phonon scattering driven by greater lattice vibrations, which causes a decrease in lattice thermal conductivity. Hence, the hydrogen storage capabilities and other aspects of the material indicate that it is an appropriate candidate for HS and might be crucial in various energy-generating applications.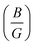 and the related moduli. The analysis of the
and the related moduli. The analysis of the 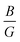 ratio and Cauchy pressure (CP), indicates that, unlike other compounds, KCaH3 exhibits a ductile nature (Poisson’s ratio (ν) = 0.29).32 Besides this, alkali metals containing ternary hydrides, such as LiBH4 (LBH), LiAlH4, NaBH4, and NaAlH4, have been intensively explored as prospective HS materials (HSMs) due to their high Cwt%.33–36 Among them, LBH is found to be a promising solid-state HSM with a volumetric capacity (ρvol.) of 121 kg H2 per m3 and the highest Cwt% of 18.3 wt% at room temperature.35,37,38 In 1997, NaAlH4 was introduced as a lightweight reversible HSM and is regarded as a promising material for solid-state hydrogen storage due to its high Cwt%, widespread availability, and low cost.33 According to Vajeeston et al.,39 the alkali boron tetrahydrides ABH4 (where A = Li, Na, K, Rb, and Cs) are insulators having an energy gap (Eg) ranging from 5.5 to 7.0 eV. Recent experimental evidence indicates that LiAlH4 and NaAlH4, processed by mechano-chemical means under ambient conditions with specific TM catalysts,40 can rapidly release 7.9 and 5.6 wt% of hydrogen, respectively. The theoretical Cwt% of LiAlH4 is as high as 10.6 wt%.41 In contrast to other chemical hydrides, which are simple and stable,42 NaBH4 is regarded as a potential candidate for HSMs due to its high hydrogen concentration of 10.6 wt%.
ratio and Cauchy pressure (CP), indicates that, unlike other compounds, KCaH3 exhibits a ductile nature (Poisson’s ratio (ν) = 0.29).32 Besides this, alkali metals containing ternary hydrides, such as LiBH4 (LBH), LiAlH4, NaBH4, and NaAlH4, have been intensively explored as prospective HS materials (HSMs) due to their high Cwt%.33–36 Among them, LBH is found to be a promising solid-state HSM with a volumetric capacity (ρvol.) of 121 kg H2 per m3 and the highest Cwt% of 18.3 wt% at room temperature.35,37,38 In 1997, NaAlH4 was introduced as a lightweight reversible HSM and is regarded as a promising material for solid-state hydrogen storage due to its high Cwt%, widespread availability, and low cost.33 According to Vajeeston et al.,39 the alkali boron tetrahydrides ABH4 (where A = Li, Na, K, Rb, and Cs) are insulators having an energy gap (Eg) ranging from 5.5 to 7.0 eV. Recent experimental evidence indicates that LiAlH4 and NaAlH4, processed by mechano-chemical means under ambient conditions with specific TM catalysts,40 can rapidly release 7.9 and 5.6 wt% of hydrogen, respectively. The theoretical Cwt% of LiAlH4 is as high as 10.6 wt%.41 In contrast to other chemical hydrides, which are simple and stable,42 NaBH4 is regarded as a potential candidate for HSMs due to its high hydrogen concentration of 10.6 wt%. is selected. It is found that a 7 × 7 × 9 k-space grid with 128 points inside the irreducible Brillouin zone wedge gives effective convergence. Additionally, in every case, the complete relaxation of the atomic positions by reducing the atomic forces up to 2 mRy per a.u. is considered. It is expected that self-consistency occurs when the total energy (Et) convergence is smaller than 10−5 Ry.
is selected. It is found that a 7 × 7 × 9 k-space grid with 128 points inside the irreducible Brillouin zone wedge gives effective convergence. Additionally, in every case, the complete relaxation of the atomic positions by reducing the atomic forces up to 2 mRy per a.u. is considered. It is expected that self-consistency occurs when the total energy (Et) convergence is smaller than 10−5 Ry.
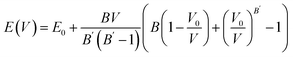
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 007.124614
007.124614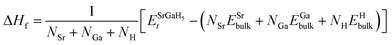
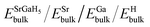 and NSr/NGa/NH represent the Et of the SGH/Sr/Ga/H in their bulk stable phases and total number of atoms of Sr/Ga/H in each case, respectively. Moreover, the hydrogen reference state used in this work is isolated H2. All energies were obtained from fully relaxed structures under each strain condition. Also, the formation enthalpy is reported in units of kJ per mol H2, which is the standard convention adopted in previous theoretical and experimental hydrogen storage studies. Using the same reference state and units allows direct comparison with previously reported hydrides in the literature.62–64 Thus, compared to the ideal value of −40 kJ per mol H2,65,66 our calculated ΔHf of the unstrained SGH system is −48.03 kJ per mol H2, which is a bit higher and may reduce the system’s functionality for the HS application. This is because, due to the substantial energy required for hydrogen absorption or desorption, a system with a reasonable ΔHf is considered the most stable.67 Here, we would like to emphasizes that the present calculations did not include contributions due to zero-point energy (ZPE) and finite-temperature contributions associated with vibrations. All of the ΔHf are reported to be the total energies of the ground states of the DFT calculations at 0 K with no phonon corrections. The ZPE may have a significant role to play in hydride systems because of the low mass of hydrogen atoms, and ZPE and vibrational terms can alter the absolute values of the ΔHf at least modestly and, as a result, the Tdes. calculated can be inaccurate. But, as our work is based on comparative trends due to applied strain, the relative stability and strain-dependent behavior should not be qualitatively affected. Along with this, the hydrogen Tdes. is also a critical factor to consider when choosing a HS system, which must be maintained between 233 and 333 K as stated by Kudiiarov et al.68 Hence, the accurate determination of Tdes. is crucial for assessing the efficiency and appropriateness of a HSM for many applications. It can be determined utilizing the conventional Gibbs equation, as illustrated in the subsequent formula:
and NSr/NGa/NH represent the Et of the SGH/Sr/Ga/H in their bulk stable phases and total number of atoms of Sr/Ga/H in each case, respectively. Moreover, the hydrogen reference state used in this work is isolated H2. All energies were obtained from fully relaxed structures under each strain condition. Also, the formation enthalpy is reported in units of kJ per mol H2, which is the standard convention adopted in previous theoretical and experimental hydrogen storage studies. Using the same reference state and units allows direct comparison with previously reported hydrides in the literature.62–64 Thus, compared to the ideal value of −40 kJ per mol H2,65,66 our calculated ΔHf of the unstrained SGH system is −48.03 kJ per mol H2, which is a bit higher and may reduce the system’s functionality for the HS application. This is because, due to the substantial energy required for hydrogen absorption or desorption, a system with a reasonable ΔHf is considered the most stable.67 Here, we would like to emphasizes that the present calculations did not include contributions due to zero-point energy (ZPE) and finite-temperature contributions associated with vibrations. All of the ΔHf are reported to be the total energies of the ground states of the DFT calculations at 0 K with no phonon corrections. The ZPE may have a significant role to play in hydride systems because of the low mass of hydrogen atoms, and ZPE and vibrational terms can alter the absolute values of the ΔHf at least modestly and, as a result, the Tdes. calculated can be inaccurate. But, as our work is based on comparative trends due to applied strain, the relative stability and strain-dependent behavior should not be qualitatively affected. Along with this, the hydrogen Tdes. is also a critical factor to consider when choosing a HS system, which must be maintained between 233 and 333 K as stated by Kudiiarov et al.68 Hence, the accurate determination of Tdes. is crucial for assessing the efficiency and appropriateness of a HSM for many applications. It can be determined utilizing the conventional Gibbs equation, as illustrated in the subsequent formula: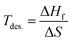
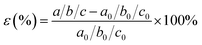
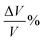 . Similarly, the ΔHf (see Fig. 5(a)) and Tdes. (see Fig. 5(b)) diminish as the
. Similarly, the ΔHf (see Fig. 5(a)) and Tdes. (see Fig. 5(b)) diminish as the 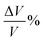 increases in the present case as well. Hence, a large
increases in the present case as well. Hence, a large 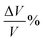 indicates a high material instability.
indicates a high material instability.


 .
.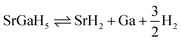 . Along with this, the reverse process, specifically rehydrogenation, takes place under appropriate hydrogen pressure and reduced temperatures as follows: SrH2 + Ga + H2 ⇌ SrGaH5. This reformation is thermodynamically viable and illustrates the reversible characteristics of SGH, highlighting its promise for practical HS applications. Now, the influence of bonding on enhancing HS features under hydrostatic strain of −5%/0%/+5% can be elucidated through the analysis of electron density distributions (EDDs; see Fig. 6(a)/(b)/(c)). Distinct responses are noted for ionic Sr–H and covalent Ga–H bonds. Table 2 illustrates that Ga–H bond lengths exhibit a consistent increase from −5% to +5% strain, leading to a decrease in covalent character (see Fig. 6) and a slight enhancement in H2 mobility. In contrast, Sr–H bonds exhibit asymmetric elongation, where −5% compressive strain leads to greater expansion compared to +5% tensile strain. This phenomenon directly destabilizes the ionic framework by lowering ΔHf (see Fig. 4) and significantly enhances the reversibility of hydrogen. This specific behavior elucidates why a −5% strain optimally enhances HS performance; the reason is that the diminished Sr–H interactions reduce the energy barrier for H2 release, while the maintained Ga–H covalent network facilitates efficient rehydrogenation. Thus, the enhanced reversibility arises from this dual mechanism; weakened ionic bonds promote desorption, whereas preserved covalent bonds ensure cyclability.
. Along with this, the reverse process, specifically rehydrogenation, takes place under appropriate hydrogen pressure and reduced temperatures as follows: SrH2 + Ga + H2 ⇌ SrGaH5. This reformation is thermodynamically viable and illustrates the reversible characteristics of SGH, highlighting its promise for practical HS applications. Now, the influence of bonding on enhancing HS features under hydrostatic strain of −5%/0%/+5% can be elucidated through the analysis of electron density distributions (EDDs; see Fig. 6(a)/(b)/(c)). Distinct responses are noted for ionic Sr–H and covalent Ga–H bonds. Table 2 illustrates that Ga–H bond lengths exhibit a consistent increase from −5% to +5% strain, leading to a decrease in covalent character (see Fig. 6) and a slight enhancement in H2 mobility. In contrast, Sr–H bonds exhibit asymmetric elongation, where −5% compressive strain leads to greater expansion compared to +5% tensile strain. This phenomenon directly destabilizes the ionic framework by lowering ΔHf (see Fig. 4) and significantly enhances the reversibility of hydrogen. This specific behavior elucidates why a −5% strain optimally enhances HS performance; the reason is that the diminished Sr–H interactions reduce the energy barrier for H2 release, while the maintained Ga–H covalent network facilitates efficient rehydrogenation. Thus, the enhanced reversibility arises from this dual mechanism; weakened ionic bonds promote desorption, whereas preserved covalent bonds ensure cyclability.
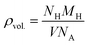
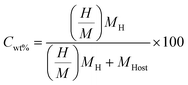
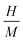 denotes the ratio of hydrogen to material atoms, MH represents the molar mass of hydrogen, and MHost signifies the molar mass of the host material. The determined value of Cwt% is 3.10%, which means that the material exhibits a commendable hydrogen storage capacity, making it a suitable candidate for HS applications. Along with this, compared to associated high-energy complex hydrides, BeAlH5 has an extremely high gravimetric capacity of 12.19 wt%, which is well beyond the DOE’s ultimate target. CaAlH5 attains 6.9 wt%, whereas SrAlH5 attains 4.17 wt%, which is well under the practical standard. Likewise, BeAlH5 has a ρvol. of 74 g H2 L, which is much higher than the DOE 2025 objective of 40 g H2 per L.73 Thus, compared to these systems, SrGaH5 has a Cwt% of 3.10% and ρvol. of 93.36787 g H2 per L. It has a lower capacity than BeAlH5 but is comparable to CaAlH5.
denotes the ratio of hydrogen to material atoms, MH represents the molar mass of hydrogen, and MHost signifies the molar mass of the host material. The determined value of Cwt% is 3.10%, which means that the material exhibits a commendable hydrogen storage capacity, making it a suitable candidate for HS applications. Along with this, compared to associated high-energy complex hydrides, BeAlH5 has an extremely high gravimetric capacity of 12.19 wt%, which is well beyond the DOE’s ultimate target. CaAlH5 attains 6.9 wt%, whereas SrAlH5 attains 4.17 wt%, which is well under the practical standard. Likewise, BeAlH5 has a ρvol. of 74 g H2 L, which is much higher than the DOE 2025 objective of 40 g H2 per L.73 Thus, compared to these systems, SrGaH5 has a Cwt% of 3.10% and ρvol. of 93.36787 g H2 per L. It has a lower capacity than BeAlH5 but is comparable to CaAlH5.

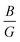 ratio. A brittle material possesses a
ratio. A brittle material possesses a 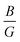 below 1.75, whereas a ductile one has a ratio over the threshold of 1.75.77 The analyzed material under 0% and +5% strain exhibits brittleness, possessing a
below 1.75, whereas a ductile one has a ratio over the threshold of 1.75.77 The analyzed material under 0% and +5% strain exhibits brittleness, possessing a  below 1.75, and is ductile under −5%, as displayed in Fig. 8(a). Similarly, the
below 1.75, and is ductile under −5%, as displayed in Fig. 8(a). Similarly, the 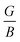 ratio (also known as Pugh’s modulus), as presented in Fig. 8(b), can be utilized to ascertain the bonding aspects of a material. If the
ratio (also known as Pugh’s modulus), as presented in Fig. 8(b), can be utilized to ascertain the bonding aspects of a material. If the  is around 0.6, ionic bonding prevails and conversely with a ratio of 1.1, covalent bonding is dominant.78 Now, ν can be utilized to ascertain the bonding features of a material. It quantifies the expansion or compression in the perpendicular axes relative to the direction of compression or stretching. If its value is 0.1, the material exhibits dominantly covalent bonding, whereas at around 0.25, it demonstrates principally ionic bonding.79 From Fig. 8(b), the analyzed compound for the strain range −5% to +5% has a ν of around 0.25, indicating a dominance of ionic bonding. Hence, for the examined material under 0% and +5% strain, the dominance of the ionic bonding is confirmed by the
is around 0.6, ionic bonding prevails and conversely with a ratio of 1.1, covalent bonding is dominant.78 Now, ν can be utilized to ascertain the bonding features of a material. It quantifies the expansion or compression in the perpendicular axes relative to the direction of compression or stretching. If its value is 0.1, the material exhibits dominantly covalent bonding, whereas at around 0.25, it demonstrates principally ionic bonding.79 From Fig. 8(b), the analyzed compound for the strain range −5% to +5% has a ν of around 0.25, indicating a dominance of ionic bonding. Hence, for the examined material under 0% and +5% strain, the dominance of the ionic bonding is confirmed by the  ratio and ν.
ratio and ν.
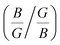 and (b) Poisson’s ratio (ν) of the SrGaH5 hydride under −5%/0%/+5% hydrostatic strain.
and (b) Poisson’s ratio (ν) of the SrGaH5 hydride under −5%/0%/+5% hydrostatic strain.

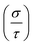 , electronic thermal conductivity per relaxation time
, electronic thermal conductivity per relaxation time 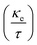 , Seebeck coefficient (S), power factor
, Seebeck coefficient (S), power factor 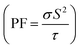 , lattice thermal conductivity per relaxation time
, lattice thermal conductivity per relaxation time 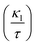 , and figure of merit
, and figure of merit 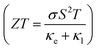 of the SGH hydride are thoroughly examined for a temperature range of 100–800 K with a consistent τ of 10−14 s using the BoltzTraP code83 and under various hydrostatic strains. Here, it is worth mentioning that we performed an extensive literature survey and found that the relaxation times for similar materials typically fall within 10−13 to 10−15 s. Therefore, selecting a constant relaxation time of 10−14 s represents a reasonable and widely accepted approximation for comparative ZT calculations, allowing us to capture the general thermoelectric trends without introducing undue uncertainty. Thus, a choice of a constant relaxation time of 10−14 s can be considered a reasonable and generally accepted approximation to comparative ZT calculations to allow us to describe the overall trends in thermoelectric behavior with a minimum amount of uncertainty. Owing to limitations in the BoltzTraP approach,
of the SGH hydride are thoroughly examined for a temperature range of 100–800 K with a consistent τ of 10−14 s using the BoltzTraP code83 and under various hydrostatic strains. Here, it is worth mentioning that we performed an extensive literature survey and found that the relaxation times for similar materials typically fall within 10−13 to 10−15 s. Therefore, selecting a constant relaxation time of 10−14 s represents a reasonable and widely accepted approximation for comparative ZT calculations, allowing us to capture the general thermoelectric trends without introducing undue uncertainty. Thus, a choice of a constant relaxation time of 10−14 s can be considered a reasonable and generally accepted approximation to comparative ZT calculations to allow us to describe the overall trends in thermoelectric behavior with a minimum amount of uncertainty. Owing to limitations in the BoltzTraP approach, 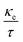 can be computed but
can be computed but 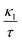 cannot. Therefore, we independently determined the
cannot. Therefore, we independently determined the 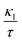 utilizing Slack’s equation as follows:84
utilizing Slack’s equation as follows:84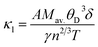
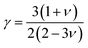
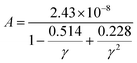
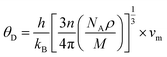
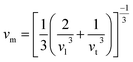
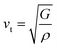
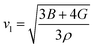
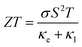
 , which increases from 0.21/0.11/0.12 × 1019 1 Ωm−1 s−2 at 100 K to 0.37/0.36/0.64 × 1019 1 Ωm−1 s−2 at 750 K under −5%/0%/+5% hydrostatic strain. σ is reliant upon the quantity of free carriers. Hence, the rising temperature elevates the quantity of free carriers owing to bond dissociation, thus providing energy to free conduction electrons. Consequently,
, which increases from 0.21/0.11/0.12 × 1019 1 Ωm−1 s−2 at 100 K to 0.37/0.36/0.64 × 1019 1 Ωm−1 s−2 at 750 K under −5%/0%/+5% hydrostatic strain. σ is reliant upon the quantity of free carriers. Hence, the rising temperature elevates the quantity of free carriers owing to bond dissociation, thus providing energy to free conduction electrons. Consequently, 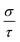 increases with increasing temperature as seen in Fig. 14(a). Among the strain conditions, tensile strain (+5%) provides the greatest
increases with increasing temperature as seen in Fig. 14(a). Among the strain conditions, tensile strain (+5%) provides the greatest 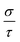 values, which signifies improved charge carrier mobility or concentration. Conversely, compressive strain (−5%) yields the lowest conductivity, probably owing to enhanced scattering or a decrease in band dispersion. To further assess the efficiency of the material, both κe and κl were examined at different strain values. The two contributions directly affect the overall thermal conductivity (κ = κe + κl) and consequently the dimensionless ZT. The κe (see Fig. 14(b)) exhibits a monotonic rise with elevated temperature throughout, at all strain levels. This tendency is predicted due to the greater mobility of the charge carriers at elevated temperature. It increases from 0.11/0.08/0.09 × 1014 W mK−1 s−1 at 100 K to 0.39/0.40/1.54 × 1014 W mK−1 s−1 under −5%/0%/+5% hydrostatic strain. In addition, the computed κl decreases with increasing temperature due to enhanced phonon–phonon scattering at elevated thermal energy (see Fig. 14(c)). It decreases from 1.76/2.01/0.9 × 1014 W mK−1 s−1 at 100 to 0.23/0.26/0.12 W mK−1 s−1 at 750 K and only reduced further with increasing temperature under −5%/0%/+5% strain. Tensile strain (+5%) results in a significant decrease of κl, probably due to enhanced lattice distortion and phonon scattering, which impede thermal conduction across the lattice, which has a positive impact on the ZT value. In contrast, compressive strain (−5%) exhibits marginally elevated values, signifying a partial restoration of lattice order and decreased phonon dispersion.
values, which signifies improved charge carrier mobility or concentration. Conversely, compressive strain (−5%) yields the lowest conductivity, probably owing to enhanced scattering or a decrease in band dispersion. To further assess the efficiency of the material, both κe and κl were examined at different strain values. The two contributions directly affect the overall thermal conductivity (κ = κe + κl) and consequently the dimensionless ZT. The κe (see Fig. 14(b)) exhibits a monotonic rise with elevated temperature throughout, at all strain levels. This tendency is predicted due to the greater mobility of the charge carriers at elevated temperature. It increases from 0.11/0.08/0.09 × 1014 W mK−1 s−1 at 100 K to 0.39/0.40/1.54 × 1014 W mK−1 s−1 under −5%/0%/+5% hydrostatic strain. In addition, the computed κl decreases with increasing temperature due to enhanced phonon–phonon scattering at elevated thermal energy (see Fig. 14(c)). It decreases from 1.76/2.01/0.9 × 1014 W mK−1 s−1 at 100 to 0.23/0.26/0.12 W mK−1 s−1 at 750 K and only reduced further with increasing temperature under −5%/0%/+5% strain. Tensile strain (+5%) results in a significant decrease of κl, probably due to enhanced lattice distortion and phonon scattering, which impede thermal conduction across the lattice, which has a positive impact on the ZT value. In contrast, compressive strain (−5%) exhibits marginally elevated values, signifying a partial restoration of lattice order and decreased phonon dispersion.

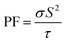 , which combines both σ and S. The value of PF at 100 K is 0.76/0.63/0.57 × 1011 W mK−2 s−1 and increases to 0.40/0.34/1.27 × 1011 W mK−2 s−1 under −5%/0%/+5% strain. Likewise, the dimensionless quantity ZT is employed to evaluate the performance of a TE material. Elevated ZT yields enhanced TE reliability, indicating that a greater amount of heat is transformed into electricity. The calculated ZT, as illustrated in Fig. 14(f), demonstrates a progressive increase with temperature from 0.04/0.03/0.05 at 100 K to 0.48/0.37/0.57 at 750 K. However, when the system is subjected to +5% tensile strain, it displays the greatest ZT of 0.61 at 550 K, whereas the unstrained and −5% compressed system demonstrated lower TE efficiency. Hence, these findings highlight the beneficial impact of tensile strain on enhancing TE performance by concurrently augmenting electrical transport and preserving an advantageous S. Finally, it is important to note that a +5% hydrostatic strain is more like a tensile pressure of roughly 3–5 GPa, assuming that there is a normal range of B for such materials. Even though +5% strain is near the upper limit of elasticity of bulk systems, +5% strain can be a useful theoretical upper limit to determine sensitivity and tunability of strain. Thus, the estimation of the improvement under the +5% strain gives a good insight into the possible practical strain-engineering approaches.
, which combines both σ and S. The value of PF at 100 K is 0.76/0.63/0.57 × 1011 W mK−2 s−1 and increases to 0.40/0.34/1.27 × 1011 W mK−2 s−1 under −5%/0%/+5% strain. Likewise, the dimensionless quantity ZT is employed to evaluate the performance of a TE material. Elevated ZT yields enhanced TE reliability, indicating that a greater amount of heat is transformed into electricity. The calculated ZT, as illustrated in Fig. 14(f), demonstrates a progressive increase with temperature from 0.04/0.03/0.05 at 100 K to 0.48/0.37/0.57 at 750 K. However, when the system is subjected to +5% tensile strain, it displays the greatest ZT of 0.61 at 550 K, whereas the unstrained and −5% compressed system demonstrated lower TE efficiency. Hence, these findings highlight the beneficial impact of tensile strain on enhancing TE performance by concurrently augmenting electrical transport and preserving an advantageous S. Finally, it is important to note that a +5% hydrostatic strain is more like a tensile pressure of roughly 3–5 GPa, assuming that there is a normal range of B for such materials. Even though +5% strain is near the upper limit of elasticity of bulk systems, +5% strain can be a useful theoretical upper limit to determine sensitivity and tunability of strain. Thus, the estimation of the improvement under the +5% strain gives a good insight into the possible practical strain-engineering approaches.





