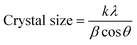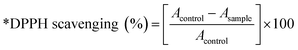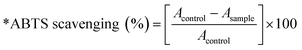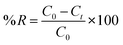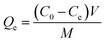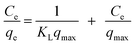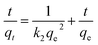Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Green synthesis of polypyrrole-SnO2 nanocomposites using Foeniculum vulgare extract for crystal violet adsorption and solvent-dependent radical scavenging
Priya Kaushika,
Ruchi Bharti *a,
Renu Sharmaa and
Annu Pandey
*a,
Renu Sharmaa and
Annu Pandey *b
*b
aDepartment of Chemistry, University Institute of Sciences, Chandigarh University, Punjab, India. E-mail: ruchi.uis@cumail.in
bDepartment of Fibre and Polymer Technology, KTH Royal Institute of Technology, Stockholm, Sweden. E-mail: annua@kth.se
First published on 3rd February 2026
Abstract
The development of multifunctional nanomaterials provides new opportunities to address both environmental and biomedical challenges. In this study, SnO2 nanoparticles were synthesized using Foeniculum vulgare seed extract and subsequently incorporated into independently synthesized polypyrrole (APS-mediated oxidative polymerization) to obtain PPy-SnO2 nanocomposites. Comprehensive structural, optical and morphological analyses, including FTIR, UV-Vis spectrophotometry, XRD, SEM-EDS, HRTEM, DLS, zeta potential, and BET, confirmed the successful formation of the nanocomposites and the uniform incorporation of SnO2 within the PPy matrix. The PPy-SnO2 nanocomposites demonstrated significant adsorption performance for crystal violet, achieving 92% removal under optimized conditions, including pH 7, a dye concentration of 10 ppm, 50 mg adsorbent, and 50 °C for 150 min. Adsorption behaviour followed a pseudo-2nd-order kinetic model, and a maximum capacity of 162.6 mg g−1 estimated from the Langmuir isotherm was achieved. The antioxidant activity assessed by DPPH and ABTS assays in methanol and hexane showed higher radical scavenging efficiency in methanol, achieving 90.8% inhibition at 800 µg mL−1. PPy-SnO2 consistently outperformed pure polypyrrole, indicating the significant role of SnO2 in enhancing electron-transfer-based scavenging. Overall, these results highlight the PPy-SnO2 NCs as an effective dual-application material that combine strong antioxidant properties with high-efficiency dye removal to provide a sustainable approach for environmental remediation.
1. Introduction
Water contamination by synthetic dyes remains a significant environmental challenge due to their high chemical stability, complex aromatic structures and resistance to biodegradation.1–3 These dyes are widely used across various industries, including textiles, pharmaceuticals, and chemicals, resulting in substantial volumes of wastewater containing these pollutants. The discharge of dye-polluted effluents into water bodies not only degrades water quality but also reduces light penetration, disrupting aquatic ecosystems and harming photosynthetic organisms. Moreover, many synthetic dyes are toxic and carcinogenic, posing serious risks to both aquatic organisms and human health.4,5 Conventional treatment methods, such as coagulation, particle aggregation, biological treatment and activated carbon adsorption, are typically insufficient for complete dye removal, highlighting the development of more efficient and environmentally friendly remediation strategies.6–8 Alongside water contamination, oxidative stress induced by reactive oxygen species (ROS) is a primary biological and industrial concern. ROS such as superoxide anions, hydroxyl radicals, and hydrogen peroxide can cause lipid peroxidation, DNA damage, protein denaturation, and accelerated material degradation.9,10 Antioxidants play a pivotal role in neutralizing ROS and preventing oxidative damage.11,12 Beyond their biological significance, antioxidants are widely employed in industrial and material applications, including food preservation, cosmetics, and protection of polymers or metals.13,14 Consequently, the development of multifunctional materials capable of both dye removal and antioxidant activity offers a promising dual-benefit approach.Conducting polymers such as polypyrrole (PPy) have drawn considerable interest due to their unique chemical, physical and electrical properties.15,16 PPy is known for its high chemical and thermal stability, good biocompatibility, large surface area and tunable conductivity, making it suitable for adsorption, catalysis and electrochemical applications.17,18 When combined with metal nanoparticles, such as tin (Sn), the resulting nanocomposites can further enhance the properties of the material.19,20 Metal nanoparticles increase surface area, enhance electron transfer, and provide additional functional sites while contributing to free radical scavenging.21 The synergistic interaction between PPy and SnO2 enables the development of dual-functional nanocomposites that can simultaneously perform pollutant removal and antioxidant activity. Previous studies have demonstrated that PPy-based nanocomposites are effective for removing cationic dyes such as crystal violet (CV). For example, PPy-chitosan and PPy-bentonite composites have shown high dye adsorption capacities under various conditions.,22,23 while PPy-metal oxide and PPy-metal nanocomposites have been examined for free radical scavenging activity.24,25 However, reports on plant-assisted Polypyrrole tin oxide nanocomposites (PPy-SnO2 NCs) with dual functionality—combining high-capacity dye adsorption and concentration-dependent antioxidant activity across multiple solvents—are limited.26 In particular, Foeniculum vulgare-assisted synthesis has not been explored in depth, despite the fact that the extract effectively facilitates green nanoparticle formation. This comparison highlights the gap in the literature that our study employs Foeniculum vulgare seed extract only for the green synthesis of SnO2 nanoparticles, while polypyrrole is synthesized by oxidative polymerization; then SnO2 nanoparticles are incorporated into the PPy matrix to synthesize PPy-SnO2 nanocomposites. Green synthesis using eco-friendly reducing agents, such as plant extracts, provides a sustainable alternative to conventional chemical methods.27,28 They minimize hazardous residues, enhance biocompatibility, and often result in nanocomposites with improved structural and functional properties.29 This study reports a low-toxicity and plant-mediated route for the synthesis of PPy-SnO2 NCs and systematically evaluates their dual-functional properties. The synthesized nanocomposites were characterized using UV-vis spectroscopy, FTIR, XRD, SEM, TEM, EDS and DLS to understand their structural, morphological and thermal features.30–34 Dye removal efficiency was investigated under varying conditions of NC dose, pH, dye concentration, contact time, and temperature. At the same time, the concentration-dependent antioxidant activity was evaluated in different solvents using standard assays.35–39 By elucidating structure–property relationships, this study highlights the potential of PPy-SnO2 NCs as sustainable, multifunctional materials for addressing both environmental pollution and oxidative stress.40,41 The findings demonstrate potential in vitro (chemical) antioxidant and ROS-scavenging activity of PPy-SnO2 nanocomposites, suggesting their suitability for future biological studies. This study aligns with the Sustainable Development Goals by facilitating efficient water decontamination (SDG 6), promoting eco-friendly synthesis (SDG 12), and exhibiting antioxidant potential (SDG 3).
2. Experimental methodology
2.1. Materials
All chemicals and solvents, including pyrrole monomer, ammonium persulfate (APS), stannous chloride (dihydrate), crystal violet dye, 0.1 M HCl, and 0.1 M NaOH, were obtained from Sigma-Aldrich, and distilled water was employed for solution preparation. Foeniculum vulgare was sourced from local store in Chandigarh. Equipment included a magnetic stirrer, centrifuge, pH meter, 1000 µL micropipette, cuvettes and a UV-vis spectrophotometer for absorbance measurements.2.2. Preparation of Foeniculum vulgare extract
Fresh Foeniculum vulgare seeds (20 g) were rigorously rinsed with distilled water to eliminate impurities and immersed in around 400 mL of distilled water. The solution was maintained at 100 °C until the colour of the solution was changed, and the volume was reduced to half. After reaching room temperature, the extract was filtered through Whatman filter paper to yield a pale-yellow solution, which act a natural reducing and capping agents for the green synthesis of tin oxide NPs, ensuring uniform and eco-friendly nanoparticle formation.2.3. Synthesis of polypyrrole
Polypyrrole was synthesized via oxidative polymerization by slowly mixing 50 ml of 0.1 M ammonium persulfate (APS, oxidant) solution to 50 ml of 0.1 M pyrrole solution, which was maintained under constant magnetic stirring for 5–6 hours at 10–20 °C. The black precipitate formed was then separated through filtration and rinsed with distilled water. The mixture was further centrifuged and then oven-dried at 70 °C for 6 h.2.4. Synthesis of tin oxide (SnO2) nanoparticles
To synthesize SnO2 nanoparticles, a solution of 0.1 M stannous chloride dihydrate (50 mL) was prepared and added dropwise to 50 ml of Foeniculum vulgare extract with constant stirring. After 30–35 minutes of stirring, a colour changes from colourless to milky yellow was observed that suggested the fabrication of SnO2 nanoparticles. The resulting product was centrifuged (3000 rpm, 10 min), rinsed with water and ethanol and dried at 70 °C for 6 h.2.5. Fabrication of polypyrrole-based tin oxide-nanocomposites
To prepare the PPy-SnO2 NCs, the SnO2 NPs solution was added gradually to the PPy solution for 40–45 minutes with stirring. A 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of PPy to SnO2 was stirred constantly at room temperature for 24 h to enable complete incorporation of SnO2 into the polypyrrole. After 24 h, nanocomposites were centrifuged at 2000 rpm for 15 minutes and washed 2–3 times with distilled water and ethanol, ensuring the removal of unbound organic phytochemicals. Nanocomposites were precipitated, and the remaining filtrate was separated. The precipitate was oven dried at 60 °C for 6 h until dryness was attained. The synthesized nanocomposites were stored in a desiccator for further characterisation and used to examine its efficiency in removing crystal dye from aqueous solution.
1 ratio of PPy to SnO2 was stirred constantly at room temperature for 24 h to enable complete incorporation of SnO2 into the polypyrrole. After 24 h, nanocomposites were centrifuged at 2000 rpm for 15 minutes and washed 2–3 times with distilled water and ethanol, ensuring the removal of unbound organic phytochemicals. Nanocomposites were precipitated, and the remaining filtrate was separated. The precipitate was oven dried at 60 °C for 6 h until dryness was attained. The synthesized nanocomposites were stored in a desiccator for further characterisation and used to examine its efficiency in removing crystal dye from aqueous solution.
3. Characterization studies
The synthesized PPy-SnO2 NCs were characterized by various analytical techniques. Fourier transform infrared (FTIR) spectra were conducted on a PerkinElmer FTIR spectrometer, and UV-vis absorption spectra were monitored using a Shimadzu UV-1900 spectrophotometer. The morphological features and elemental analysis were accessed via scanning electron microscopy with energy-dispersive spectroscopy (SEM-EDS, SU8010 series, HITACHI). Particle size and zeta potential were analyzed with a Zetasizer Nano (Malvern Panalytical, UK) provided with dynamic light scattering, and TEM-EDS was performed using a CRYO ARM™ 300 II instrument.3.1. UV-visible spectral analysis
UV-vis spectroscopy is a commonly employed technique for confirming nanoparticles synthesis and studying their optical characteristics in a liquid medium. In this study, UV-vis spectra of Foeniculum vulgare extract, SnO2 NPs, polypyrrole (PPy), and PPy-SnO2 NCs were recorded with a wavelength range from 200 to 800 nm, as shown in Fig. 1. | ||
| Fig. 1 UV-visible spectrum of Foeniculum vulgare extract, polypyrrole (PPy), SnO2 nanoparticles, and polypyrrole-SnO2 NCs. | ||
The extract exhibited a prominent absorption peak at 274 nm, attributed mainly to π → π* transitions of phytochemicals such as flavonoids and phenolics. These compounds act as natural reducing and stabilizing agents. After the synthesis of SnO2 NPs, the spectrum exhibited a new peak at approximately 360 nm, corresponding to the surface plasmon resonance of SnO2 or partially oxidized Sn species. The shift in the absorption band from 274 nm to 360 nm and the decreased intensity indicate that the phytochemicals acted as reducing and stabilizing agents during the nanoparticle formation.
PPy exhibited a characteristic absorption band at 464 nm, attributed to π–π* transitions within its conjugated polymer backbone. In the PPy-SnO2 NCs, the absorption band was broader and slightly shifted, suggesting strong electronic interactions between PPy chains and SnO2 nanoparticles. This spectral change confirms the successful incorporation of SnO2 NPs into the PPy matrix, thereby enhancing charge transfer and improving the nanocomposite's stability.42
3.2. FTIR spectral analysis
FTIR spectroscopy of Foeniculum vulgare extract, SnO2 NPs, PPy, and PPy-SnO2 NCs was carried out using the PerkinElmer Fourier-Transform Infrared Radiation spectrophotometer, with a range between 4000 cm−1 and 400 cm−1 to investigate the chemical bonds and optical performance and study the chemical bonds present in the nanoparticles and nanocomposites (Fig. 2).The FTIR spectrum of SnO2 nanoparticles (SnO2 NPs) shows a broad band at 3390–3400 cm−1 (O–H stretching) and a peak at ∼1630 cm−1 (O–H bending). The strong absorptions in the lower wavenumber region (600–500 cm−1) are attributed to Sn–O and Sn–O–Sn vibrations, confirming the presence of Sn–O bonds on the nanoparticle surface.43 The FTIR spectrum of pure polypyrrole (PPy) exhibits distinct vibrational bands characteristic of its polymeric structure. Peaks observed at around 1550 and 1460 cm−1 are attributed to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C and C–C stretching vibrations within the pyrrole ring. The band near 1300 cm−1 corresponds to C–N stretching, reflecting the polymer backbone, while the absorption around 1150 cm−1 is assigned to in-plane C–H deformation of the pyrrole units. These features confirm the successful formation of the PPy polymer network. The FTIR spectrum of PPy-SnO2 NCs exhibits the characteristic PPy peaks at 3400, 1550, 1460, 1300, and 1150 cm−1, corresponding to N–H, C
C and C–C stretching vibrations within the pyrrole ring. The band near 1300 cm−1 corresponds to C–N stretching, reflecting the polymer backbone, while the absorption around 1150 cm−1 is assigned to in-plane C–H deformation of the pyrrole units. These features confirm the successful formation of the PPy polymer network. The FTIR spectrum of PPy-SnO2 NCs exhibits the characteristic PPy peaks at 3400, 1550, 1460, 1300, and 1150 cm−1, corresponding to N–H, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, C–C, C–N, and in-plane C–H vibrations, respectively. Minor shifts and variations in peak positions are observed, indicating interactions between the PPy matrix and the SnO2 nanoparticles. Additionally, a distinct absorption peak in the 600–500 cm−1 region, assigned to Sn–O and Sn–O–Sn vibrations, confirms the successful incorporation of SnO2 and the formation of stable PPy-SnO2 NCs.44
C, C–C, C–N, and in-plane C–H vibrations, respectively. Minor shifts and variations in peak positions are observed, indicating interactions between the PPy matrix and the SnO2 nanoparticles. Additionally, a distinct absorption peak in the 600–500 cm−1 region, assigned to Sn–O and Sn–O–Sn vibrations, confirms the successful incorporation of SnO2 and the formation of stable PPy-SnO2 NCs.44
3.3. X-ray diffraction analysis
The XRD analysis was performed to examine the structural properties of PPy and PPy-SnO2 NCs, as illustrated in Fig. 3. The diffraction pattern of pure PPy demonstrated two distinct broad peaks at 2θ ≈ 23.5° and 26.1°, which is characteristic of the predominantly amorphous nature of conducting polymers. PPy exhibited a crystallinity of 42.6% with the remaining 57.4% attributed to the amorphous phase. The average crystallite size of PPy was calculated to be approximately 16.3 nm, consistent with the range reported in previous studies of PPy-based nanostructures.The average crystallite size was estimated using the Debye–Scherrer equation,
The PPy-SnO2 nanocomposites demonstrated sharp diffraction peaks over the amorphous polymer background, ensuring the presence of crystalline SnO2 NPs within the PPy matrix. Various sharp peaks observed at 2θ = 26.7°, 34.02°, 37.8°, 51.7° and 54.6° corresponds to (110), (101), (200), (211), and (220) planes of rutile SnO2, respectively (JCPDS No. 41-1445). This suggests the strong interactions among SnO2 NPs and PPy. The crystallinity of the nanocomposites improved moderately to 51.9% with less amorphous content (48.1%). Crystallite size was calculated to be 28.8 nm using the Debye–Scherrer equation, confirming the successful incorporation of SnO2 into the PPy scaffold and the development of a nanocomposite structure, consistent with earlier reports of enhanced crystallinity in PPy-based nanocomposites upon incorporation of a metal.45
3.4. Scanning-electron microscopy with energy dispersive X-ray spectroscopy (SEM-EDS)
The surface morphology and elemental analysis of PPy and PPy-SnO2 NCs were accessed by SEM-EDS on a HITACHI SU8010 series microscope. SEM enables high-resolution images of the sample through scanning with a focused electron beam, enabling detailed information about particle morphology, shape, and surface features. As shown in Fig. 4a and b, PPy particles exhibited a rough, irregular and porous surface, while SnO2 NPs mainly appeared spherical in shape with some irregularity. The PPy-SnO2 nanocomposites displayed irregularly aggregated, spherical particles with relatively smooth domains, as illustrated in Fig. 4c and d. These morphological features are consistent with those observed in prior studies of PPy-SnO2 composites.46 | ||
| Fig. 4 SEM image of (A and B) PPy and (C and D) PPy-SnO2 NCs at different magnifications (E) SEM-EDS of synthesized PPy-SnO2 nanocomposites. | ||
The average particle size of the PPy-SnO2 nanocomposites, calculated using ImageJ, was approximately 48 nm, which aligns well with previously reported values for similar polymer-metal nanocomposites. The EDS spectra, as shown in Fig. 4e, exhibited clear peaks for tin (Sn), carbon (C), nitrogen (N), and oxygen (O), validating the successful embedding of SnO2 within the PPy framework. Sn was observed as the predominant element, while C, N, and O corresponded to the characteristic composition of the PPy matrix. These results indicate that the synthesized PPy-SnO2 nanocomposites exhibit a uniform, well-defined morphology. The quantitative elemental analysis is presented in Table 1. Overall, the SEM and EDS analyses confirm the successful integration of SnO2 nanoparticles into the PPy matrix, forming a stable dual-component nanocomposite. The apparent difference between mass% and atom% is expected in EDS quantification. In particular, Sn shows a higher mass% but lower atom % because Sn has a much higher atomic weight than C, N and O; therefore, fewer Sn atoms contribute disproportionately to the total mass percentage.
| Element | Line | Mass% | Atom% |
|---|---|---|---|
| C | K | 14.1 ± 0.18 | 26.6 ± 0.34 |
| N | K | 3.8 ± 0.30 | 5.4 ± 0.41 |
| O | K | 36.3 ± 0.83 | 57.8 ± 1.17 |
| Sn | L | 45.8 ± 1.44 | 10.2. ± 0.27 |
| Total | 100.00 | 100.00 |
3.5. High-resolution transmission electron microscopy
The high-resolution transmission electron microscopy (HRTEM) images of PPy and PPy-SnO2 nanocomposites revealed distinct morphological features. The HRTEM micrographs of PPy (Fig. 5) revealed aggregation of irregularly shaped nanoparticles with an amorphous character, consistent with its broad diffraction features observed in XRD [46]. In the PPy-SnO2 NCs, as illustrated in Fig. 5, SnO2 nanoparticles are incorporated in the polymer matrix and exhibit irregular and aggregated clusters. The lighter-coloured areas show the PPy matrix, whereas the darker areas illustrate the high-electron-density SnO2 nanoparticles. | ||
| Fig. 5 HRTEM image of synthesized (A and B) PPy and (C and D) PPy-SnO2 NCs (E) HRTEM-EDS analysis of PPy-SnO2 Nanocomposites. | ||
The crystalline nature of the SnO2 nanoparticles is revealed by well-resolved lattice planes with an interplanar spacing of approximately 0.27 nm, which correlates to the (101) plane of tetragonal SnO2 (JCPDS No. 04-0673), consistent with similar PPy-metal systems.47 Furthermore, the HRTEM images of PPy-SnO2 NCs further show that SnO2 nanoparticles are predominantly spherical and uniformly distributed within the PPy matrix, with individual particle sizes (10–15 nm) and aggregate dimensions (50–150 nm) determined directly from the micrographs using ImageJ software. Energy dispersive X-ray spectroscopy (EDS) analysis confirms the presence of Sn along with N, O and C, corresponding to the composition of the PPy-SnO2 NCs as mentioned in Table 2. As shown in Table 2, the higher Sn mass% relative to atom% is consistent with EDS reporting due to higher atomic weight of Sn compared to the lighter elements (C, N and O).
| Element | Line | Mass% | Atom% |
|---|---|---|---|
| C | K | 13.5 ± 0.2 | 44.9 ± 0.3 |
| N | K | 2.5 ± 0.19 | 6.8 ± 0.2 |
| O | K | 29.5 ± 0.8 | 29.1 ± 0.9 |
| Sn | K | 54.5 ± 1.2 | 19.2 ± 0.3 |
| Total | 100 | 100 |
3.6. Dynamic light scattering analysis (DLS)
Dynamic Light Scattering (DLS) analysis was performed on the synthesized PPy-SnO2 nanocomposites in aqueous suspension to determine the hydrodynamic particle size distribution. The nanocomposites had an average hydrodynamic diameter (Z-average) of 194.1 nm, as shown in Fig. 6, which is considerably larger than the crystallite size determined by XRD (28 nm) and the particle sizes observed in SEM. This difference is attributed to the presence of solvation layers and particle aggregation in aqueous media, which are commonly observed in polymer-based nanocomposites. The corresponding polydispersity index (PDI) of 0.212 indicates a moderately narrow size distribution, reflecting good uniformity and dispersion of the nanocomposites. Such behaviour is well documented in the literature, especially for polymer-stabilized and green-synthesized nanomaterials, where biological moieties and interparticle interactions can increase the hydrodynamic size. Comparable DLS values for PPy-based nanocomposites have been reported in the range of 120–183 nm, which align with the present observation.48 The single peaked DLS distribution further confirms the formation of a relatively homogeneous population of PPy-SnO2 nanocomposites in aqueous dispersion. This hydrodynamic behaviour is particularly relevant for aqueous adsorption systems, where dispersion stability governs effective surface availability. | ||
| Fig. 6 Hydrodynamic size distribution of PPY-SnO2 nanocomposites in aqueous suspension obtained by dynamic light scattering (DLS). | ||
3.7. Zeta-potential
The zeta potential of the synthesized PPy-SnO2 NCs was assessed to evaluate their colloidal stability. The nanocomposites exhibited a mean zeta potential value of −10.2 mV (Fig. 7), indicating a moderate negative surface charge that provides sufficient electrostatic repulsion to prevent aggregation.49 | ||
| Fig. 7 Zeta potential of PPy-SnO2 nanocomposites, indicating surface charge and colloidal stability. | ||
This suggests that the synthesized nanocomposites are colloidally stable and well-dispersed in aqueous medium. The distribution peak at −4.6 mV, along with a standard deviation of 0.6 mV, indicates a relatively uniform particle dispersion in the aqueous medium.
Additional parameters, including a mean intensity of 710.1 kcounts s−1, filter optical density of 2.8738, conductivity of 0.475 mS cm−1, electrophoretic mobility of 0.7947 µm cm V−1 s−1, and transmittance of 17.7%, corroborate the colloidal stability of the PPy-SnO2 NCs. These findings confirm that the synthesized nanocomposites are well-dispersed and exhibit robust stability in aqueous suspension, which is critical for their potential applications.50
3.8. BET-analysis
The surface properties of synthesized PPy-SnO2 NCs were analyzed with nitrogen adsorption–desorption isotherms at 77 K, and the data were reported by applying the Brunauer–Emmett–Teller (BET), t-plot (DeBoer), and Barrett–Joyner–Halenda (BJH) techniques (Fig. 8a). The BET surface area of the PPy-SnO2 NCs was 24.14 m2 g−1, indicating a moderate surface area that favours possible adsorption applications. The nitrogen adsorption–desorption isotherm exhibited a type IV shape with an H3 hysteresis loop typical of mesoporous materials. | ||
| Fig. 8 (a) Nitrogen adsorption–desorption isotherm of PPy-SnO2 NCs exhibiting type IV behaviour, confirming mesoporosity, (b) exhibiting a BET surface area of 24.14 m2 g−1. | ||
BJH analysis of the desorption branch gave a total pore volume of 0.033 cm3 g−1 and an average pore diameter of 3.9 nm, which lies at the micro–mesopore boundary (Fig. 8b). The t-plot analysis indicated negligible true microporosity, suggesting that the accessible surface is dominated by narrow mesopores and external surface area, both of which are beneficial for the adsorption of dye molecules.
Overall, such porosity facilitates the diffusion and interaction of the target molecules with the nanocomposite, increasing its adsorption potential and catalytic use. These surface properties agree with the already reported mesoporous polymer-metal nanocomposites.51
4. Antioxidant studies
4.1. DPPH (2,2-diphenyl-1-picrylhydrazyl) assay
The DPPH radical scavenging method is an effective approach for determining antioxidant potential, interacting mainly through electron transfer with an additional addition via hydrogen atom transfer. In this study, antioxidants reduce the violet-coloured DPPH radical to a pale-yellow form, and a reduced absorbance at 517 nm indicates the level of radical scavenging. As shown in Fig. S9 (SI), the DPPH radical is neutralized by abstracting a hydrogen atom from the antioxidant (AH), resulting in the formation of the reduced DPPH-H and an antioxidant radical (A˙). All antioxidant measurements were assessed using PPy and PPy-SnO2 nanocomposites(NCs). Notably, Foeniculum vulgare extract was not studied for the assay medium, only washed and dried PPy and PPy-SnO2 NCs were examined to avoid any contribution from residual phytochemicals. In the current study, PPy and PPy-SnO2 nanocomposites demonstrated concentration-dependent antioxidant activity in methanol and hexane, with significantly greater effectiveness observed in methanol due to better nanomaterial dispersion and stronger interactions with radicals in polar media (Fig. 9). Ascorbic acid showed the highest activity, reaching 98.21 ± 1.14% in methanol, while PPy and PPy-SnO2 NCs achieved 62.95 ± 1.13% and 87.91 ± 1.06% inhibition, respectively, at 800 µg mL−1.Each assay was performed three times (n = 3), and values are provided as mean ± SD. The consistently higher scavenging activity of PPy-SnO2 NCs when compared with PPy can be associated with the catalytic role of SnO2 nanoparticles, which promote electron transfer. These findings, consistent with previous studies,52,53 confirm that polymer–metal nanocomposites exhibit enhanced antioxidant capacity and hold promise as effective ROS scavengers.
The Scavenging activity was evaluated by the given equation:
4.2. ABTS (2,2-azino-bis-3-ethylbenzothiazoline-6-sulphonic acid) assay
The antioxidant activity of PPy and PPy-SnO2 nanocomposites was further examined by the ABTS + cation radical approach. In this experiment, the blue-green ABTS + solution decolors upon interaction with antioxidants, as indicated by a decrease in absorbance at 734 nm (Fig. S11).Both PPy and PPy-SnO2 NCs observed an increase in radical scavenging activity with varying the concentration range over the 200–800 µg mL−1 concentration range (Fig. 10), at 800 µg mL−1, PPy-SnO2 NCs reached 90.80% inhibition in methanol compared to 57.21% for PPy, whereas in hexane the values were 60.24% and 41.51%, respectively. Ascorbic acid, used as the standard, showed the most significant inhibition in methanol at 98.21 ± 1.14%. A similar trend was observed with the DPPH method, confirming its lower activity in hexane than in methanol and highlighting the key role of solvent polarity in nanocomposite–radical interactions.52 The percentage inhibition was determined by the equation:
PPy-SnO2 NCs exhibited considerable antioxidant activity, which makes them emerging materials for various applications such as food packaging, biomedical coatings and water treatment, where the dual function of effective pollutant removal and radical scavenging is extremely favourable. However, their performance when compared with ascorbic acid, suggesting that they can support traditional antioxidants but cannot completely replace ascorbic acid.36
5. Polypyrrole-tin oxide nanocomposites for CV dye removal from aqueous solutions
The increasing contamination of the aquatic ecosystem by synthetic dyes from textile, pharmaceutical, and other industries has raised significant environmental concerns due to their toxicity, persistence, and resistance to biodegradation. Crystal violet is a commonly employed cationic triphenylmethane dye which is particularly resistant to conventional wastewater treatment. To address this concern, the adsorption performance of PPy-SnO2 nanocomposites was examined using CV, a model cationic dye, to evaluate their potential for environmental remediation. Batch studies were performed under static conditions to examine the impact of NC dose, dye concentration, time, and temperature on removal efficiency. All batch adsorption experiments were performed using washed and dried PPy-SnO2 nanocomposites as the adsorbent, without addition of Foeniculum vulgare to the adsorption medium. UV-visible spectrophotometry was employed to monitor the adsorption performance, quantified in terms of percentage removal (%R) and equilibrium adsorption capacity (Qe), evaluated by following equations:C0 represents the initial concentration (mg L−1), Ct represents the final concentration of dye at time t (mg L−1), Ce is the equilibrium concentration of dye (mg L−1), Qe is the equilibrium adsorption capacity (mg g−1), and V denotes to the volume of solution used through the experiment (L), M refers to mass of adsorbent (g).
5.1. Effect of parameters on removal of dye
 | ||
| Fig. 14 Temperature-dependent adsorption of dye was performed with fixed condition: a dye concentration of 10 ppm, 50 mg of NCs, a pH of 7 and a 90-min contact time. | ||
Where Cads denotes the adsorbed dye molar concentration on the nanocomposites at equilibrium, the free energy change (ΔG°) was evaluated from the equation ΔG° = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kc
Kc
R is the gas constant (8.314 J mol−1 K−1), T refers to absolute temperature (K), and Kc is the equilibrium constant. The positive ΔH = 38.53 kJ mol−1 indicates that adsorption is endothermic, while the positive ΔS = 121.87 J mol−1 K−1 suggests a rise in entropy at the substrate–liquid interface. Gibbs free energy (ΔG) shifts from 4.06 kJ mol−1 at 283 K to −6.00 kJ mol−1 at 363 K, indicating that adsorption becomes spontaneous at higher temperatures. The high correlation coefficient (R2 value = 0.995) from the Van't Hoff plot supports the reliability of these thermodynamic data.57
6. Kinetic and adsorption isotherms
The adsorption study of crystal violet onto PPy-SnO2 NCs was accessed through kinetic and isotherm studies. This analysis provided a detailed insight into adsorption mechanism, the dye-nanocomposite surface interaction, and the equilibrium characteristics, thereby confirming the efficiency and reliability of the nanocomposites for dye removal.6.1 Adsorption isotherm
The adsorption equilibrium of crystal violet by PPy-SnO2 NCs was analyzed to understand the adsorption mechanism and surface characteristics. The adsorption isotherm demonstrates how dye molecules are balanced among the aqueous solution and the adsorbent at equilibrium. The adsorption study was accessed employing the Langmuir and Freundlich isotherms.where qmax represents the maximum monolayer adsorption capacity (mg g−1), and KL is the Langmuir constant, which indicates the affinity between the adsorbent and dye molecules.
The results were correlated with the linearised Langmuir equation (Ce versus Qe), resulting in a high correlation coefficient (R2 ≈ 0.9698), indicating excellent agreement with the model and confirming monolayer adsorption as the dominant mechanism (Fig. 16a). This excellent agreement ensures that CV dye removal take place on the defined solid adsorption sites of the PPy-SnO2 nanocomposites rather than through homogeneous solution-phase reactions. The maximum adsorption capacity (Qmax) was determined from the slope of the Langmuir plot, yielding 162.6 mg g−1, highlighting the excellent performance of PPy-SnO2 NCs in removing crystal violet dye molecules from aqueous solutions. Furthermore, the Langmuir constant (KL) was found to be 0.48 L mg−1, suggesting a higher binding affinity of the CV molecules for the NCs surface.59 The favourability of the adsorption process was further evaluated by calculating the dimensionless separation factor (RL). The calculated RL values for all tested concentrations are within the 0–1 range, indicating that the removal of the CV dye by PPy-SnO2 NCs surface is favourable and involves significant adsorbent–adsorbate interactions.
 | ||
| Fig. 16 Langmuir and Freundlich isotherm model fitting curve for the CV dye adsorptive removal onto synthesized PPy-SnO2 NCs. | ||
The results demonstrate that CV dye molecules effectively interact with the uniform binding sites on the PPy-SnO2 NCs surface, likely by π–π interactions, electrostatic attractions, and hydrogen bonding. The excellent adsorption capacity of nanocomposites and the rapid attainment of equilibrium highlight their potential for efficient cationic dye removal in aqueous media.
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe = ln qe = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KF + (1/n) ln KF + (1/n) ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ce Ce |
KF represents the Freundlich isotherm parameters, and 1/n refers to heterogeneity factor, which indicates the adsorption intensity. The linear fitting of ln Qe against ln Ce yielded a slope value of 0.3239 (1/n = 0.3239, n = 3.09) and an intercept of 4.1101, corresponding to a KF = 60.9 mg g−1 (L g−1 (L mg−1)1/n, as shown in Fig. 16b. The value of Freundlich constants (KF and n) confirmed the favourability of adsorption, with an n value ranging from 1 to 10, signifying strong dye-NCs surface affinity. However, this model yielded slightly lower R2 values (0.8946) than the Langmuir model, suggesting heterogeneity in the PPy-SnO2 system. The high KF value further indicates the significant adsorption capacity of the synthesized nanocomposites.60 This model represents adsorption occurring on heterogeneous sites and is not limited to formation of monolayer.
6.2. Adsorption kinetics
This study was investigated to understand the efficiency of Crystal Violet (CV) dye removal and the mechanisms governing adsorption on PPy-SnO2 nanocomposites. Adsorption behaviour was evaluated by Kinetic models and determined whether the mechanism involved physisorption or chemisorption. The pseudo-1st-order and 2nd-order model are employed to study adsorption kinetics and to understand adsorption mechanisms.ln (qe − qt) = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe − k1t qe − k1t |
The pseudo-2nd-order provided a better fit to the experimental adsorption data for removal of dye by PPy-SnO2 nanocomposites. The equilibrium adsorption capacities (qe) predicted by this model closely matched the observed experimental data having a correlation coefficient (R2) of 0.998, indicating an excellent fit (Fig. 17b). These observations suggest that electron–exchange interactions among the dye molecules and reactive functional sites on the nanocomposite's surface primarily control adsorption.63 The detailed isotherm and kinetics parameters that include constants, calculated adsorption capacities and correlation coefficients are given in Table S5 (SI file).
7. Mechanism of CV dye adsorption onto PPy-SnO2 nanocomposites
The adsorptive removal of CV dye onto PPy-SnO2 NCs occurs through a synergistic combination of hydrogen bonding, electrostatic interactions and π–π stacking among the conjugated rings of PPy and the CV dye molecules. At neutral pH, the cationic Crystal Violet (CV) dye molecules are electrostatically attracted to negatively charged oxygen sites of SnO2. At the same time, –OH groups on the surface of nanocomposites participate in hydrogen bonding with dye molecules. The incorporated SnO2 NPs offer more active sites within the PPy matrix, which enhances dye adsorption and interfacial interaction. This cooperative mechanism results in efficient dye removal from aqueous solutions. A representation of the proposed adsorption mechanism is shown in Fig. 18, highlighting the key interactions responsible for the high adsorption performance of nanocomposite. | ||
| Fig. 18 Adsorption of crystal violet on PPy-SnO2 nanocomposites via electrostatic, hydrogen-bonding, and π–π interaction. | ||
8. Comparative analysis of crystal violet adsorption performance
To evaluate the efficiency of the synthesized PPy-SnO2 nanocomposites, a comparison was made with previously reported adsorbents used for crystal violet (CV) removal. The PPy-SnO2 NCs exhibited 92% removal efficiency under optimized conditions (pH 7, 10 ppm dye concentration, 50 mg adsorbent dose and 50 °C within 150 min), showing adsorption capacity (qmax) of 162.6 mg g−1 followed by the Langmuir isotherm model. Kinetic analysis confirmed that this study followed pseudo-2nd-order equation, suggesting chemisorption as the dominant mechanism. The adsorption capacities and experimental conditions of the already reported adsorbents are summarised in Table 3. The findings suggests that the qmax of synthesized PPy-SnO2 NCs is comparable to various nanomaterial-based adsorbents.| Adsorbent | Qmax (mg g−1) | Optimum pH | Temperature (°C) | Contact time (min) | Reference |
|---|---|---|---|---|---|
| PPy-SnO2 | 162.6 | 7 | 50 | 150 | [our study] |
| Ferrite-biochar composite | 325.4 | 7–8 | 30 | 120 | 64 |
| Modified carbon spheres | 134.6 | 6 | 25 | 180 | 65 |
| Carbon-sphere/Titania-nanotube composite (TNTs@Cs) | 84.7 | 5.5 | 25–30 | 180 | 66 |
| Natural clay | 203.6 | 7 | 25 | 120 | 67 |
9. Conclusions
This study successfully synthesized PPy-SnO2 nanocomposites by incorporating Foeniculum vulgare extract-derived SnO2 nanoparticles into polypyrrole synthesized via oxidative polymerization. Comprehensive characterization by UV-vis, FTIR, DLS, zeta potential, SEM-EDS, HRTEM and XRD confirmed the successful formation, stability and uniform incorporation of SnO2 within the PPy matrix. The nanocomposites showed excellent adsorption performance toward crystal violet, achieving 92% removal under optimized conditions (pH 7, 10 ppm dye, 50 mg PPy-SnO2, 50 °C, 150 min) with a maximum adsorption capacity of 162.6 mg g−1, underscoring their suitability for dye removal applications. In addition, they exhibited significant antioxidant activity in methanol at 800 µg mL−1, with 87.90% and 90.80% radical scavenging in the DPPH and ABTS assays, respectively, and higher efficiencies than in hexane. These results demonstrate that PPy-SnO2 nanocomposites are promising dual-functional materials that effectively combine high-efficiency dye removal with potent radical scavenging, thereby holding considerable potential for environmental remediation. To the best of our knowledge, this is among the first reports on Foeniculum vulgare-assisted PPy-SnO2 nanocomposites that integrate efficient crystal violet adsorption with solvent-dependent antioxidant activity. This sustainable green synthesis approach can be further tailored for use in medical, agricultural and environmental sectors. Future work may focus on optimizing the nanocomposite formulation, exploring reusability and stability under realistic conditions, and extending their application to other relevant pollutants and reactive oxygen species, thereby advancing the role of green nanotechnology in sustainable development.Author contributions
Conceptualization, methodology, software, writing – original draft preparation, P. K.; resources, project administration, data curation, R. S.; supervision, validation, formal analysis, investigation, writing – review and editing, R. B.; A. P.; all authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declare no conflicts of interest.Data availability
The data supporting this article have been included in the supplementary information (SI) and as part of the manuscript. Supplementary information: additional experimental and plant extract details, along with characterization data (Fig. S1–S19 and Tables S1–S6) supporting the results presented in this manuscript. See DOI: https://doi.org/10.1039/d5ra09693f.Acknowledgements
The authors gratefully acknowledge the Department of Chemistry, Chandigarh University, for providing laboratory facilities and resources required for this work. P. K. and R. B. also thank the Sophisticated Analytical Instrumentation Facility (SAIF), Panjab University, Chandigarh, for access to advanced characterization instruments.Notes and references
- A. Sandoval, C. Hernández-Ventura and T. E. Klimova, Fuel, 2017, 198, 22–30 CrossRef CAS.
- M. Kampa and E. Castanas, Environ. Pollut., 2008, 151, 362–367 CrossRef CAS PubMed.
- J. Huang, S. Tan, P. D. Lund and H. Zhou, Energy Environ. Sci., 2017, 10, 2284–2311 RSC.
- J. Ma, J. Shi, H. Ding, G. Zhu, K. Fu and X. Fu, Chem. Eng. J., 2017, 312, 20–29 Search PubMed.
- G. Zhu, J. Liu, J. Yin, Z. Li, B. Ren, Y. Sun, W. Peng and Y. Liu, Chem. Eng. J., 2016, 288, 390–398 CrossRef CAS.
- J. Yin, G. Zhu and B. Deng, Desalination, 2016, 379, 93–101 Search PubMed.
- G. Zhu, Y. Bian, A. S. Hursthouse, P. Wan, K. Szymanska, J. Ma, X. Wang and Z. Zhao, J. Fluoresc., 2017, 27, 1–26 Search PubMed.
- G. Zhu, Q. Wang, J. Yin, Z. Li, P. Zhang, B. Ren, G. Fan and P. Wan, Water Res., 2016, 100, 201–210 Search PubMed.
- G. Zhu, J. Liu and Y. Bian, Environ. Sci. Pollut. Res., 2018, 25, 14447–14459 CrossRef CAS PubMed.
- F. M. Christensen, Regul. Toxicol. Pharmacol., 1998, 28, 212–221 Search PubMed.
- C. A. Kinney, E. T. Furlong, S. L. Werner and J. D. Cahill, Environ. Toxicol. Chem., 2006, 25, 317–326 CrossRef CAS PubMed.
- I. Ali, H. Y. Aboul-Enein and K. Kümmerer, in Biophysico-Chemical Processes of Anthropogenic Organic Compounds in Environmental Systems, Wiley, USA, 2011, pp. 439–462 Search PubMed.
- M. J. Ahmed and B. H. Hameed, Ecotoxicol. Environ. Saf., 2018, 149, 257–266 Search PubMed.
- L. Wang, X. Zhao, J. Zhang and Z. Xiong, Environ. Sci. Pollut. Res., 2017, 24, 14198–14206 CrossRef CAS PubMed.
- M. Gutiérrez, M. Pepió and M. Crespi, Color. Technol., 2002, 118, 1–5, DOI:10.1111/j.1478-4408.2002.tb00129.x.
- V. C. Taty-Costodes, H. Fauduet, C. Porte and A. Delacroix, J. Hazard. Mater., 2003, 105, 121–142 CrossRef CAS PubMed.
- D. Mahanta, G. Madras, S. Radhakrishnan and S. Patil, J. Phys. Chem. B, 2008, 112, 10153–10157 CrossRef CAS.
- C. Giurgea, Cond. Reflex, 1973, 8, 108–115 CrossRef CAS PubMed.
- V. V. Singh, in Sustainable Nanotechnology for Environmental Remediation, Elsevier, Amsterdam, 2022, pp. 31–61 Search PubMed.
- M. Mosaviniya, T. Kikhavani, M. Tanzifi, M. T. Yaraki, P. Tajbakhsh and A. Lajevardi, Colloid Interface Sci. Commun., 2019, 33, 100211 Search PubMed.
- A. C. Burduşel, O. Gherasim, A. M. Grumezescu, L. Mogoantă, A. Ficai and E. Andronescu, Nanomaterials, 2018, 8, 681 Search PubMed.
- Z. Li, H. Shan, W. Rong, Z. Zhao, K. Ma, S. Peng and S. Wei, Toxics, 2024, 12, 687 Search PubMed.
- F. A. A. Rajathi, C. Parthiban, V. G. Kumar and P. Anantharaman, Spectrochim. Acta, Part A, 2012, 99, 166–173 Search PubMed.
- G. K. Toplak, Domace Lekovito Bilje, Mozaik Kniga, Zagreb, 2005, pp. 50–51 Search PubMed.
- C. Borek, J. Nutr., 2006, 136, 810–812 CrossRef PubMed.
- C. Borek, J. Nutr., 2001, 131, 1010–1015 CrossRef PubMed.
- M. Ates, T. Karazehir and S. Sarac, Curr. Phys. Chem., 2012, 2, 224–240 Search PubMed.
- B. Kumari, R. K. Tiwary and M. Yadav, Mater. Chem. Phys., 2022, 290, 126457 CrossRef CAS.
- P. P. Singh, in New Polymer Nanocomposites for Environmental Remediation, Elsevier, Amsterdam, 2018, pp. 223–241 Search PubMed.
- R. Prakash, R. Bharti, A. Thakur, M. Verma and R. Sharma, Sustainable Chem. Eng., 2025, 11, 11–21 Search PubMed.
- V. Saini, A. Thakur, M. Verma, R. Bharti and R. Sharma, Orbital - Electron. J. Chem., 2024, 16, 293–305 CrossRef CAS.
- A. Yadav and R. Bharti, Orbital - Electron. J. Chem., 2024, 16, 205–218 CrossRef CAS.
- A. Singh, R. Bharti, A. Thakur, M. Verma and R. Sharma, Orbital - Electron. J. Chem., 2024, 16, 271–278 CAS.
- A. Nughwal, R. Bharti, A. Thakur, M. Verma, R. Sharma and A. Pandey, RSC Adv., 2025, 15, 10287–10297 RSC.
- P. Kaushik, R. Bharti, R. Sharma, M. Verma, R. T. Olsson and A. Pandey, Eur. Polym. J., 2024, 221, 113574 CrossRef CAS.
- P. Kaushik, R. Bharti, R. Sharma and A. Pandey, Environ. Sci. Adv., 2025, 3, 45–60 Search PubMed.
- D. Kaur, M. Verma, R. Bharti, S. Naik and R. Sharma, Chem. Pap., 2025, 1–23 Search PubMed.
- S. Bansal, R. Bharti, A. Thakur, M. Verma and R. Sharma, Orbital - Electron. J. Chem., 2024, 16, 240–246 CAS.
- A. Thakur, M. Verma, R. Bharti and R. Sharma, Curr. Chin. Sci., 2023, 3, 322–348 CrossRef CAS.
- A. K. Maurya, R. Gogoi and G. Manik, Adv. Nanocompos. Mater. Environ. Energy Harvest, Springer, Cham, 2022, pp. 331–368 Search PubMed.
- S. K. Dhillon and P. P. Kundu, Chem. Eng. J., 2022, 431, 133341 CrossRef CAS.
- G. Sowmiya and G. Velraj, J. Inorg. Organomet. Polym. Mater., 2020, 30, 2494–2504 Search PubMed.
- M. Akram, A. T. Saleh, W. A. Wan Ibrahim and R. Hussain, Mater. Sci. Eng., B, 2016, 213, 1–8 Search PubMed.
- S. Sagadevan and V. Rajendran, Mater. Sci. Semicond. Process., 2021, 130, 105832 CrossRef.
- L. Zhu, Y. Zhang, X. Zhao, Y. Jiao, Z. Zhao, Y. Wang and N. Yang, Electrochem. Commun., 2021, 124, 106951 CrossRef CAS.
- M. M. Rahman Khan and M. M. H. Rumon, Polymers, 2024, 16, 3277 Search PubMed.
- B. Li, T. Zhang, S. Wei and W. Gao, Chem. Eng. J., 2022, 446, 136768 CrossRef CAS.
- D. Gil, J. González, M. Muñoz, R. Menéndez and R. Santamaría, Polymer, 2019, 160, 238–247 CrossRef.
- E. Drzymała, G. Gruzeł, A. Pajor-Świerzy, J. Depciuch, R. Socha, A. Kowal and M. Parlinska-Wojtan, J. Nanopart. Res., 2018, 20, 144 CrossRef.
- D. J. Pochapski, C. Carvalho dos Santos, G. W. Leite, S. H. Pulcinelli and C. V. Santilli, Langmuir, 2021, 37, 13379–13389 CrossRef CAS PubMed.
- B. Saha, A. Debnath and B. Saha, Mater. Today Commun., 2024, 39, 109061 CrossRef CAS.
- A. Ghazzy, R. R. Naik and A. K. Shakya, Polymers, 2023, 15, 2167 CrossRef CAS PubMed.
- S. Velpula, M. Z. Gul, S. R. Beedu and K. Rupula, Next Nanotechnol., 2025, 8, 100188 CrossRef.
- F. Mashkoor, M. Shoeb and C. Jeong, Polymers, 2023, 15, 4285 CrossRef CAS PubMed.
- E. A. Abdelrahman and M. T. Basha, Inorganics, 2025, 13, 112 Search PubMed.
- Y. Dehmani, D. S. P. Franco, J. Georgin, R. Mghaiouini, B. B. Mohammed, R. Kacimi, T. Lamhasni, E. C. Lima, N. El Messaoudi and A. Sadik, Environ. Surf. Interfaces, 2025, 3, 103–111 CrossRef.
- O. S. Omer, M. A. Hussein, B. H. Hussein and A. Mgaidi, Arab. J. Chem., 2018, 11, 615–623 CrossRef.
- A. H. Birniwa, H. N. M. E. Mahmud, S. S. A. Abdullahi, S. Habibu, A. H. Jagaba, M. N. M. Ibrahim and K. Umar, Polymers, 2022, 14, 3362 CrossRef CAS PubMed.
- G. S. Aljeddani, R. M. Alghanmi and R. A. Hamouda, Polymers, 2023, 15, 4394 Search PubMed.
- A. O. Dada, F. A. Adekola, E. O. Odebunmi, A. S. Ogunlaja and O. S. Bello, Sci. Rep., 2021, 11, 16454 CrossRef CAS PubMed.
- A. T. Ojedokun and O. S. Bello, Appl. Water Sci., 2017, 7, 1965–1977 CrossRef CAS.
- A. M. Aljeboree, A. N. Alshirifi and A. F. Alkaim, Arab. J. Chem., 2017, 10, S3381–S3393 CrossRef CAS.
- A. H. Bhat and H. T. N. Chishti, Sep. Sci. Technol., 2023, 58, 287–301 Search PubMed.
- D. Mehta, P. N. Dave and V. V. Kumar, RSC Adv., 2025, 15, 33189–33208 RSC.
- H. Li, Q. Miao, Y. Chen, M. Yin, H. Qi, M. Yang and S. Wang, Microporous Mesoporous Mater., 2020, 297, 110040 Search PubMed.
- A. M. Mohammed, A. Kotb, M. M. Sanad, M. Abdel-Hakim and A. S. Ahmed, RSC Adv., 2024, 14, 31332–31347 Search PubMed.
- A. Q. Alorabi, M. S. Hassan, M. M. Alam, S. A. Zabin, N. I. Alsenani and N. E. Baghdadi, Nanomaterials, 2021, 11, 2789 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |