Open Access Article
Open Access ArticleSulfenylnitrene-mediated aminative cyclizations for the diastereoselective synthesis of fused bicyclic indolines
Deacon Herndon ,
Prakash Kafle†
,
Prakash Kafle†
 ,
Rishav Mukherjee† and
Indrajeet Sharma
,
Rishav Mukherjee† and
Indrajeet Sharma *
*
Department of Chemistry and Biochemistry, University of Oklahoma, 101 Stephenson Parkway, Norman, Oklahoma 73019, USA. E-mail: isharma@ou.edu; Web: https://indrajeetsharma.com/
First published on 24th March 2026
Abstract
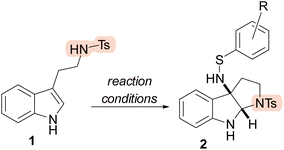
We disclose the sulfenylnitrene-mediated synthesis of aminated, fused bicyclic indolines from tryptamine and tryptophol derivatives. This method provides rapid, metal-free access to valuable bioactive indoline scaffolds under operationally simple conditions. The approach accommodates a variety of nucleophiles including unprotected indoles, works in a variety of solvents, and provides access to 5, 6, and 7-membered bicycles. We have also demonstrated the scalability of our methodology through a gram scale synthesis.
Introduction
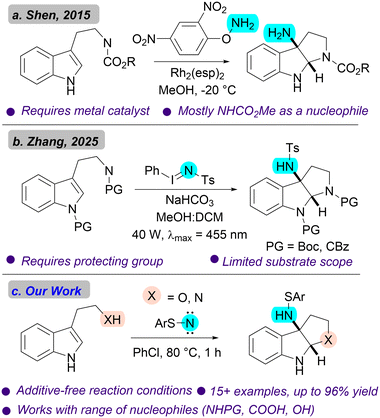
Fused bicyclic indolines are a key structural motif in a number of biologically active natural products and drug molecules.1,2 Indolines themselves are saturated derivatives of indoles and serve as an important class of aromatic heterocycles (Scheme 1a).3,4 A popular strategy for the synthesis of indolines has been dearomative difunctionalization of the 2,3-position of indole through cyclization pathways that simultaneously capture an electrophile, enabling functionalization of the resulting indoline.5–9 (Scheme 1b).From a total synthesis standpoint, amino-pyrroloindoline is a key intermediate leveraged in the synthesis of a number of indole alkaloid natural products.10–12 As early as the 1970s, 3-azido pyrroloindolines were being synthesized from tryptamine and iodine azide, a technique which has been improved upon in recent years.13–16 These azido compounds can then be reduced to amines in a separate step. Reports from Movassaghi and coworkers showcased that amino-pyrroloindolines could be synthesized from pyrroloindolines through either substitution of bromine or direct C–H activation.17,18 Furthermore, a number of reports have utilized transition metals, including copper and rhodium, for aminocyclization of tryptamines.19–21 Notably, organocatalytic synthesis of enantioenriched cyclotryptamines was reported by Antilla and coworkers in 2015, but this was relatively limited in scope.22 Importantly, there has been progress in this area in the form of aminative cyclization of tryptamines via the capture of nitrenes. A seminal report of this strategy comes from Shen and coworkers in 2015 using rhodium oxynitrenes (Scheme 2a).23 This work is further noteworthy as it provided access to naked amino-pyrroloindolines in a single step. However, it is still limited by its use of rhodium, a precious metal catalysis. In general, these reports have been scattered and limited in scope. Critically, there has been very little progress made in the area of metal-free aminative cyclizations for the synthesis of amino-pyrroloindoline and its analogues.
 | ||
| Scheme 2 (a) Metal-mediated aminative synthesis of cyclotryptamines. (b) Light-mediated aminative synthesis of cyclotryptamines. (c) Our approach. | ||
Despite the power of organometallic chemistry in modern synthesis, there has been an increasing awareness of the sustainability issues associated with metal-mediated transformations.24,25 Indeed, mining 1 kilogram of rhodium produces 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 kilograms of CO2.26,27 Furthermore, metal-free conditions lower drug costs and avoid strict manufacturing as many pharmaceuticals have a ≤10 ppm limit for precious metals.28
100 kilograms of CO2.26,27 Furthermore, metal-free conditions lower drug costs and avoid strict manufacturing as many pharmaceuticals have a ≤10 ppm limit for precious metals.28
Zhang and coworkers reported an important, metal-free synthesis of amino-pyrroloindolines using photogenerated iodonitrenes.29 Notably, they expanded the scope of this reaction to include the cyclotryptophol scaffold as well as its 6- membered analogue (Scheme 2b). Despite the advances that these approaches represent, existing methods are limited in substrate scope, rely on costly transition metal catalysts, and often have a need for indole –NH protecting groups.
These methods have not been shown to be tolerant of amide or carboxylic acid nucleophiles which would provide access to valuable lactam and lactone scaffolds, respectively. To overcome these challenges, we disclose sulfenylnitrene as a general purpose aminocyclization reagent capable of producing fused bicyclic indole ethers, lactams, and lactones under mild, metal- and additive-free conditions (Scheme 2c).
Our group has recently shown that sulfenylnitrenes can be generated under purely thermal or photochemical conditions, making them a logical choice to serve as a nitrene transfer reagent.30,31 Sulfenylnitrenes are electron-deficient, singlet organic nitrenes bearing a thiol moiety attached to the nitrenic center (Fig. 1a).32 Our group has developed a series of benchtop stable precursors, which release sulfenylnitrenes under a wide of range of thermal and photochemical conditions (Fig. 1b). These precursors have been utilized in a variety of organic transformations including the single atom editing of nitrogen heterocycles.31,33,34 Density functional theory (DFT) calculations indicate that sulfenylnitrenes exist as a ground state singlet and likely exhibit a considerable ylide-like resonance. Depending on the electronics of the system in question, they can participate in aziridination reactions with olefins in either a concerted-asynchronous or stepwise process.31,33–35 Our group reported the serendipitous discovery of such a transformation during a mechanistic study in our 2025 paper using sulfenylnitrenes for skeletal editing of N- heterocycles (Fig. 1c).31
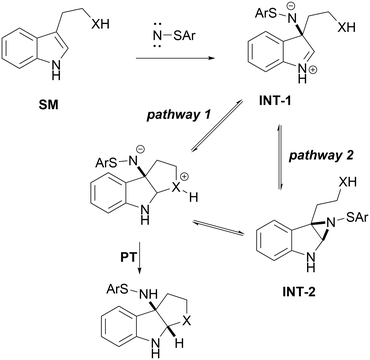
After observing that SNP-1a was capable of accomplishing the aminative cyclization of -Ts protected tryptamine at 120 °C in chlorobenzene (PhCl), we hypothesized that this process involved either capture of an indole C2-carbocation or proximity driven ring-opening of an aziridine intermediate by a pendant nucleophile. This hypothesis is supported by calculations that have showed that the aziridination of olefins by sulfenylnitrene is highly asynchronous and sometimes even stepwise.31,35 Indeed, the mechanisms of both cycloadditions and rearrangements of carbocations generally exist on a spectrum between concerted synchronous/asynchronous and stepwise.36–38 With this reactivity spectrum in mind we sought to leverage a transient indole C2-carbocation to enable an efficient, general cyclization of pendant nucleophiles.
Results and discussion
Encouraged by these findings, we moved to optimize this reaction by screening a variety of sulfenylnitrene precursors (Table 1). We sought to improve the initial yield from SNP-1a at 120 °C by changing the electronics of the aryl ring. However, using SNP-1b at 100 °C offered a lower yield. This led us to try increased heating (180 °C, SNP-2a), but here the yield dramatically decreased. Seeking the right balance between temperature and class of precursor, we then utilized SNP-2b/c and observed formation of the product in excellent yield.| Entry | SNP | Solvent | T (°C) | Yieldb (%) |
|---|---|---|---|---|
| a Reaction conditions: 1 and SNPs were mixed in solvent (0.1 M) at the specified temperature for 1 hour.b Determined by 1H NMR using 1,3,5-trimethoxybenzene as internal standard.c Reaction was exposed to 390 nm blue light for 12 hours at room temperature. | ||||
| 1 | SNP-la | PhCl | 120 °C | 82% |
| 2 | SNP-1b | PhCl | 100 °C | 53% |
| 3 | SNP-2a | PhCl | 180 °C | 18% |
| 4 | SNP-2b | PhCl | 80 °C | 95% |
| 5 | SNP-2c | PhCI | 80°C | 96% |
| 6 | SNP-2c | MeCN | 80 °C | 91% |
| 7 | SNP-2c | THE | 80 °C | 77% |
| 8 | SNP-2c | DHE | 80 °C | 73% |
| 9 | SNP-2c | DCM | r.t hνc | 25% |
 |
||||
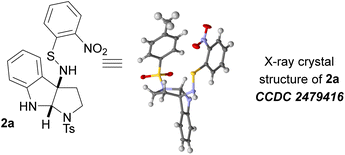
We additionally screened the photochemical generation of sulfenylnitrene with little success. It is likely that light alone does not provide sufficient energy to overcome the entropic cost associated with cyclization. We chose SNP-2c as our optimized precursor and went on to show that the reaction was compatible with a variety of solvents with a broad range of polarities. Chlorobenzene was chosen as the ideal solvent for its high yield and clean reaction. We were additionally able to obtain a crystal structure of compound 2a (Table 1), which unambiguously confirmed the relative stereochemistry of the product. This provides direct evidence that the reaction proceeds in a diastereoselective manner, consistent with the proposed mechanistic hypothesis.
With these optimized conditions in hand, we moved forward towards expanding the substrate scope of this transformation (Fig. 2). First, we blocked the indole –NH with a methyl protecting group, which afforded the product (2b) in good yield (Fig. 2a). We next screened a handful of tryptamine –NH protecting groups (2c, 2d) as well as indole substitution (2e, 2f), both of which were well-tolerated. The 6-membered tryptamine analogue also afforded the product in moderate yield (2g).
 | ||
| Fig. 2 (a) Substrate scope. (b) Application to synthesis of a melatonin analogue. (c) Limitations of our methodology. | ||
Critically, benzyl protected amide and free carboxylic acid cleanly afforded the corresponding lactam (2h) and lactone (2i), respectively. To our delight, our conditions were capable of converting the sleep hormone melatonin directly into its aminated cyclic analogue (2j) in high yield (Fig. 2b). Notably, previous literature has showed that sodium borohydride is capable of reducing thioaryl functionalities to access naked amines.31 Finally, subjecting an open chain alkene-amide (1k) to our conditions did not afford the corresponding lactam (2k), and the starting material was recovered (Fig. 2c). We postulate that this is because of the decreased electrophilicity of sulfenylnitrenes compared other reagents used for electrophilic nitrogen triggered 1,2-difunctionalization of alkenes.39,40 Because of this, aziridination of the relatively unactivated styrene alkene is inefficient. A combination of factors including steric hindrance, electronic effects, and ring size may also account for the comparatively lower yields observed with sulfenylnitrene in certain indole substrates.
Ring-size effect study and mechanistic discussion
We were interested in the potential power of this C2-carbocation capture strategy to make a variety of ring-sizes, including classically challenging medium-sized, 7-membered rings (2n). As such, we developed a scope of O-nucleophiles of various ring-sizes (Fig. 3). While our method was capable of accessing 5-, 6-, and 7-membered rings, we observed a clear trend as larger ring-sizes resulted in lower reaction yields. This is presumably because as the ring-closure step becomes slower, more side-products are formed from the reactive C2-carbocation intermediate, resulting in lower yields.41 In the case of 5-membered rings, our methodology also tolerated a variety of different substitutions. Specifically, this substrate scope showcases a 6-substituted indole (2o), as well as weakly (2p) and strongly (2q) electron-withdrawing functional groups.Based on our experimental observations and previous work with concerted-asynchronous and stepwise aziridinations using sulfenylnitrenes, we propose a plausible mechanism for this transformation (Scheme 3).31,35 We postulate that the starting indole (SM) reacts with sulfenylnitrene to produce a zwitterionic structure (INT-1). This structure may be classified as a true intermediate, or some other transient species depending on its lifetime.42 Regardless, INT-1 is capable of undergoing cyclization either directly (pathway 1) or in an aziridine (INT-2) dependent fashion (pathway 2) to form the protonated bicyclic indoline which can tautomerize to form the observed product.
Gram-scale synthesis and deprotection
To demonstrate the feasibility and scalability of our approach, we next endeavoured to perform a gram–scale reaction. For this reaction, we utilized nosyl-protected tryptamine (1r) due to the lability of this protecting group, which would enable us to derivatize the product later (Scheme 4). Subjecting this molecule to our standard conditions cleanly resulted in the cyclized indoline product (2r) in 68% yield. The nosyl-group was then removed using K2CO3 to afford the deprotected indoline (3) in good yield.Conclusions
In conclusion, we have demonstrated that sulfenylnitrenes may be utilized as a general-purpose reagent for the synthesis of aminated bicyclo-indolines capable of providing access to 5-, 6-, and 7-membered rings from a wide variety of nucleophiles. This approach is operationally simple and provides a high yield without the need of precious metal catalysts or specialized experimental setup. Additionally, the high diastereoselectivity of this transformation was unambiguously determined through X-ray crystallography and the method was applied to the functionalization of the drug molecule melatonin as well as a gram scale synthesis of cyclotryptamine.Experimental
General procedure for aminative cyclization
To a solution of indole (1 equiv.) in PhCl (0.1 M), SNP-2c (1 equiv.) was added and heated to 80 °C for 1 hour. The reaction was then cooled to room temperature. The crude reaction mixture was concentrated under reduced pressure (700 mmHg, bath temperature: 50 °C). The products were purified by silica-gel flash column chromatography.Spectral data for parent substrate
Author contributions
This manuscript was prepared by D. H. and I. S. All authors participated in research discussions and approved the final manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2479416 (for 2a) contains the supplementary crystallographic data for this paper.43The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: detailed experimental protocols and NMR/HRMS spectra. See DOI: https://doi.org/10.1039/d5ra09687a.
Acknowledgements
We thank Dr Novruz Akhmedov, Dr Steven Foster, and Dr Nobuyuki Yamamoto of the Research Support Services (University of Oklahoma) for assistance with NMR, mass spectra, and X-ray crystallography respectively. Support for the acquisition of the Bruker D8 Quest diffractometer from the NSF MRI program (CHE-1726630) is gratefully acknowledged. Helpful discussions with Prof. Yihan Shao are also greatly appreciated. Furthermore, we would like to acknowledge the Dodge Family College of Arts and Sciences for their financial support of DH and RM though the Dissertation Research Fellowship. We also thank the Graduate College (OU) for supporting PK through the Nancy L. Mergler Dissertation Completion Fellowship. This work was supported by the Research Council of the University of Oklahoma Norman Campus, and the American Chemical Society Petroleum Research Fund (ACS-PRF) New Directions grant (PRF no. 69051-ND1).References
- H. Wei, B. Li, N. Wang, Y. Ma, J. Yu, X. Wang, J. Su and D. Liu, ChemistryOpen, 2023, 12, e202200235 CrossRef CAS PubMed.
- C. Bowman, M. Denis and S. Canesi, Chem. Commun., 2025, 61, 5563–5576 RSC.
- Q. Li, T. Xia, L. Yao, H. Deng and X. Liao, Chem. Sci., 2015, 6, 3599–3605 RSC.
- Y. Liu, Y. Zhang, F. Zhang, M. Wang and J. Xu, Tetrahedron, 2025, 134672 CrossRef CAS.
- P. Ruiz-Sanchis, S. A. Savina, F. Albericio and M. Alvarez, Chem. Eur J., 2011, 17, 1388–1408 CrossRef CAS PubMed.
- L. M. Repka and S. E. Reisman, J. Org. Chem., 2013, 78, 12314–12320 CrossRef CAS PubMed.
- R. Dalpozzo, G. Bartoli and G. Bencivenni, Chem. Soc. Rev., 2012, 41, 7247–7290 RSC.
- Y. Lee, Y. S. Nam, S. Y. Kim, J. E. Ki and H. G. Lee, Chem. Sci., 2023, 14, 7688–7698 RSC.
- A. R. Liandi, A. H. Cahyana, D. N. Alfariza, R. Nuraini, R. W. Sari and T. P. Wendari, Green Synth. Catal., 2024, 5, 1–13 CAS.
- T. Newhouse and P. S. Baran, J. Am. Chem. Soc., 2008, 130, 10886–10887 CrossRef CAS PubMed.
- Y. Matsuda, M. Kitajima and H. Takayama, Org. Lett., 2008, 10, 125–128 CrossRef CAS PubMed.
- T. Newhouse, C. A. Lewis, K. J. Eastman and P. S. Baran, J. Am. Chem. Soc., 2010, 132, 7119–7137 CrossRef CAS PubMed.
- H. M. Nelson, S. H. Reisberg, H. P. Shunatona, J. S. Patel and F. D. Toste, Angew. Chem., Int. Ed., 2014, 53, 5600–5603 CrossRef CAS PubMed.
- M. Ikeda, F. Tabusa, Y. Nishimura, S. Kwon and Y. Tamura, Tetrahedron Lett., 1976, 17, 2347–2350 CrossRef.
- C.-Z. Lin, L.-F. Jiang, G.-Y. Zhang, F.-S. Zhou, S.-H. Wu, J. Cao and Q.-H. Deng, Chem. Commun., 2023, 59, 7831–7834 RSC.
- J. Guo, S. Chen, J. Liu, J. Guo, W. Chen, Q. Cai, P. Liu and P. Sun, Eur. J. Org Chem., 2017, 2017, 4773–4777 CrossRef CAS.
- S. P. Lathrop and M. Movassaghi, Chem. Sci., 2014, 5, 333–340 RSC.
- M. Movassaghi, O. K. Ahmad and S. P. Lathrop, J. Am. Chem. Soc., 2011, 133, 13002–13005 CrossRef CAS PubMed.
- T. Benkovics, I. A. Guzei and T. P. Yoon, Angew Chem. Int. Ed. Engl., 2010, 49, 9153 CrossRef CAS PubMed.
- C. Liu, J. C. Yi, Z. B. Zheng, Y. Tang, L. X. Dai and S. L. You, Angew. Chem., 2016, 128, 761–764 Search PubMed.
- S. Beaumont, V. Pons, P. Retailleau, R. H. Dodd and P. Dauban, Angew. Chem., 2010, 122, 1678–1681 CrossRef.
- Z. Zhang and J. C. Antilla, Angew. Chem., 2012, 124, 11948–11952 Search PubMed.
- Z. Shen, Z. Xia, H. Zhao, J. Hu, X. Wan, Y. Lai, C. Zhu and W. Xie, Org. Biomol. Chem., 2015, 13, 5381–5384 RSC.
- L.-C. Campeau and D. E. Fogg, Organometallics, 2019, 38, 1–2 Search PubMed.
- P. Kafle, B. Ghosh, A. C. Hunter, R. Mukherjee, K. M. Nicholas and I. Sharma, ACS Catal., 2024, 14, 1292–1299 CrossRef CAS.
- P. Nuss and M. J. Eckelman, PLoS One, 2014, 9, e101298 CrossRef PubMed.
- R. M. Bullock, J. G. Chen, L. Gagliardi, P. J. Chirik, O. K. Farha, C. H. Hendon, C. W. Jones, J. A. Keith, J. Klosin and S. D. Minteer, Science, 2020, 369, eabc3183 CrossRef CAS PubMed.
- E. B. Bauer, in Iron Catalysis II, Springer, 2015, pp. 1–18 Search PubMed.
- L.-H. Zhang, Y. Xie and J. Xuan, Chem. Commun., 2025, 61, 8528–8531 RSC.
- P. Kafle, P. Kharel, D. Nilson, D. Herndon, S. Yasuda and I. Sharma, Chem, 2026, 102753 Search PubMed.
- B. Ghosh, P. Kafle, R. Mukherjee, R. Welles, D. Herndon, K. M. Nicholas, Y. Shao and I. Sharma, Science, 2025, 387, 102–107 Search PubMed.
- P. Kafle, D. Herndon and I. Sharma, Chem. Soc. Rev., 2025, 54, 10344–10362 Search PubMed.
- R. S. Atkinson, M. Lee and J. R. Malpass, Chem. Commun., 1984, 919–920 RSC.
- T. Fujii, T. Kousaka and T. Yoshimura, Tetrahedron Lett., 2002, 43, 5841–5843 Search PubMed.
- P. Kafle and I. Sharma, 9‐((2,4‐Dinitrophenyl)thio)‐1,4‐Dihydro‐1,4‐Epiminonaphthalene, Encyclopedia of Reagents for Organic Synthesis, 2026, pp. 1–2, DOI:10.1002/047084289X.rn02658.
- D. J. Tantillo, J. Phys. Org. Chem., 2008, 21, 561–570 Search PubMed.
- D. J. Tantillo, Chem. Soc. Rev., 2010, 39, 2847–2854 RSC.
- K. N. Houk, F. Liu, Z. Yang and J. I. Seeman, Angew. Chem., Int. Ed., 2021, 60, 12660–12681 CrossRef CAS PubMed.
- M. J. Smith, W. Tu, C. M. Robertson and J. F. Bower, Angew. Chem., 2023, 135, e202312797 CrossRef.
- A. M. Rodriguez Trevino, Y.-D. Kwon and L. Kurti, Org. Lett., 2025, 27, 9430–9435 CrossRef CAS PubMed.
- G. Illuminati and L. Mandolini, Acc. Chem. Res., 1981, 14, 95–102 CrossRef CAS.
- International Union of Pure and Applied Chemistry (IUPAC), 5.00 edn, 2025, DOI: DOI:10.1351/goldbook.I03096.
- CCDC 2479416: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p7155.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |