Open Access Article
Open Access ArticleProduction of phenols from a cyclic sugar alcohol in high-temperature water over a charcoal-supported platinum catalyst
Kiyoyuki Yamazaki *,
Rumiko Samata,
Norihito Hiyoshi
*,
Rumiko Samata,
Norihito Hiyoshi ,
Osamu Sato and
Aritomo Yamaguchi
,
Osamu Sato and
Aritomo Yamaguchi
Research Institute for Chemical Process Technology, National Institute of Advanced Industrial Science and Technology (AIST), 4-2-1 Nigatake, Miyagino, Sendai 983-8551, Japan. E-mail: k-yamazaki@aist.go.jp
First published on 19th February 2026
Abstract
Biomass is a useful carbon-neutral feedstock for the production of various chemicals. The conversion of cyclic sugar alcohols to aromatic compounds can be expected to promote greater use of biomass resources. Here, we examined the conversion of myo-inositol, a cyclic sugar alcohol present in high amounts in rice bran, to aromatic compounds over supported metal catalysts in high-temperature water. We confirmed that myo-inositol could be converted into phenolic compounds such as phenol and catechol over several charcoal-supported metal catalysts in high-temperature water at 250 °C; the yield of phenolic compounds decreased in the order of Pt/C (42%) > Rh/C (24%) > Ru/C (12%) > Pd/C (0.8%) > no catalyst (0%). To understand more about the reaction pathway, the conversion was repeated using various other cyclic alcohols and phenolic compounds. The yields of phenolic compounds decreased progressively with decreasing number of hydroxy groups in the cyclic alcohols; however, phenolic compounds were also produced from cyclohexanetriol and cyclohexanediol, indicating that the formation of aromatic rings from these compounds cannot be explained only by dehydration or dehydroxylation reactions. Therefore, the aromatic ring formation potentially involves dehydrogenation of the cyclic alcohols. Recycling tests confirmed that the Pt/C catalyst retained its catalytic activity after being reused twice.
1. Introduction
As we move toward a more sustainable society to address the issues of resource depletion and greenhouse gas emissions, greater utilization of certain underused raw materials is needed.1 Biomass is a truly renewable carbon resource because it can be replenished on the human timescale and it comes from plants that absorb CO2 from the atmosphere, which offsets emissions when converted to chemical stocks. Thus, greater use of biomass for the production of raw materials would be environmentally beneficial. Currently, biomass is used for the production of monomers such as ethylene and propylene for the subsequent production of polymers such as polyolefins.2–5 However, methods for the production of aromatic monomers from biomass for use in the production of aromatic plastics such as polystyrene, phenolic resin, and epoxy resin are yet to be developed.Woody biomass consists mainly of lignocellulose, which itself is composed of cellulose, hemicellulose, and lignin. Cellulose is a polymer of glucose, and hemicellulose is a polymer of several kinds of monosaccharides. Lignin is an aromatic polymer and its depolymerization is known to produce aromatic monomers.6–9 However, depolymerization of lignin to aromatic monomers requires severe reaction conditions because of lignin's robust three-dimensional structure. Previously, we reported that organosolv lignin10 and biomass of Japanese cedar11 can be converted to aromatic compounds by using charcoal-supported metal catalysts(Pt, Pd, Rh, or Ru/C) in supercritical water (385–415 °C). The yields of aromatic products, which were calculated based on the number of aromatic rings in the reactant, were 6.4% for organosolv lignin10 and 17.4% for Japanese cedar11 over Pt/C for 1 h at 400 °C. Thus, under identical reaction conditions, the direct conversion of biomass produced a higher yield of aromatic compounds compared with the conversion of lignin alone. In addition, the aromatic compounds obtained from the conversion of biomass were derived not only from the lignin but also from the cellulose and/or hemicellulose. Based on these findings, we subsequently investigated the conversion of cellulose and hemicellulose xylan, under the same reaction conditions and found that both reactants were converted into aromatic compounds even at a temperature of 250 °C, which is lower than that required for lignin depolymerization, despite the fact that cellulose and hemicellulose do not themselves contain aromatic rings.12 To gain insights into the reaction pathways, we then carried out the conversion of various monosaccharides and their derivatives (e.g., glucose [C6] and xylose [C5]) into aromatic compounds.13 The total yields of aromatic products were comparable (about 4%) for the conversion of polysaccharides and monosaccharides regardless of the carbon number of the reactant. This result indicates that aromatic compounds were obtained even from monosaccharides that contain fewer carbon atoms than a benzene ring. Thus, the reaction conditions permitted the C–C bond formation and subsequent cyclization that were necessary for producing aromatic compounds from monosaccharides. However, the conversion of saccharides to aromatic compounds involved multiple reaction steps, resulting in low yields of aromatic compounds. Thus, we hypothesized that alcohols with a six-membered ring (cyclohexane ring) should be convertable to aromatic products at high yield because initial C–C bond formation and cyclization would not be required for aromatic ring formation. Additionally, we focus on the reactions following cyclization in the pathway from monosaccharides to aromatic compounds, with particular attention to aromatic ring formation and dehydroxylation processes. By elucidating this conversion pathway, we aim to the effective utilization of biomass resources for the production of aromatic compounds.
Inositol is a cyclic sugar alcohol particularly abundant in rice bran but often discarded as waste.14 Previously, other groups have reported the production of catechol from 2-deoxy-scyllo-inosose, a cyclic alcohol derived from cyanobacterium, by using hydroiodic acid and acetic acid, citing a yield of 58%.15,16 However, this approach requires the use of large quantities of hydroiodic acid, which is expensive and needs special corrosion-resistant equipment. Dehydrogenation of cyclohexanediol over supported Ni nanoparticles has also been reported for the production of catechol.17,18 What has not been reported, however, is the direct conversion of cyclic sugar alcohols into aromatic compounds in high-temperature water over supported metal catalysts. In the present study, we therefore examined the conversion of myo-inositol to aromatic compounds in high-temperature water using supported metal catalysts.
Once a catalyst with high activity was identified, we optimized the reaction conditions with respect to the total yield of aromatic compounds. To investigate the reaction pathways underlying the conversion of the cyclic alcohols to aromatic compounds, we also examined the conversion of several other cyclic alcohols under the optimized conditions. The recyclability of the selected catalyst was also examined.
2. Experimental
2.1. Materials
Charcoal-supported metal catalysts with 5 wt% metal loading (i.e., Pt/C, Rh/C, Ru/C, and Pd/C) were purchased from Fujifilm Wako Pure Chemical Corporation. myo-Inositol, cyclohexanol, resorcinol, hydroquinone, tetrahydrofuran, 1-propanol, and 1-butanol were also obtained from Fujifilm Wako Pure Chemical Corporation. Catechol, 1,2,3-trihydroxybenzene (pyrogallol), 1,2,4-trihydroxybenzene, 1,3,5-trihydroxybenzene (phloroglucinol), cis-1,2-cyclohexanediol, and trans-1,2-cyclohexanediol were obtained from Tokyo Chemical Industry Co., Ltd. chiro-Inositol was purchased from Merck & Co., Inc.2.2. Reaction procedure
The conversion of cyclic alcohols was performed in a batch reactor made from a 316 stainless steel tube (volume: 6.0 cm3). All reagents were used without any pretreatment. The reactor was charged with catalyst (0.15 g), reactant (0.2 g), and water (0.5–3.0 g). The reactor was purged with argon to remove air, then heated for 1 h at 200–300 °C using an air-fluidized sand bath (ACRAFT). After the reaction, the reactor was rapidly cooled in a water bath. Gaseous products were collected using a glass syringe and analyzed with a gas chromatograph (Shimadzu GC-8A) equipped with a Shincarbon ST column and a thermal conductivity detector. The reaction slurry was recovered by rinsing the reactor with tetrahydrofuran, and solid residue was separated by filtering the slurry. Liquid products in the filtrate were quantitatively analyzed by using a second gas chromatograph (Agilent HP-6890) equipped with a DB-WAX capillary column and a flame ionization detector using 1-propanol or 1-butanol as the internal standard. Detailed operating conditions of gas chromatographs were shown in SI. Additionally, GC-MS (Agilent 5977A MSD) analysis was performed to comprehensively identify the byproducts. The yields of aromatic products and gaseous products, gas composition, and solid yield were calculated using eqn (1)–(4):| Yield of aromatic products (%) = (moles of carbon atoms in aromatic products)/(moles of carbon atoms in initial reactant) × 100 | (1) |
| Yield of gaseous products (%) = (moles of carbon atoms in gaseous products)/(moles of carbon atoms in initial reactant) × 100 | (2) |
| Gas composition (%) = (moles of gaseous products)/(sum of moles of total gaseous products) × 100 | (3) |
| Solid yield (wt%) = {(weight of solid residue) − (weight of supported metal catalyst)}/(weight of initial reactants) × 100 | (4) |
3. Results and discussion
3.1. Confirmation that myo-inositol is converted into aromatic compounds over charcoal-supported metal catalysts
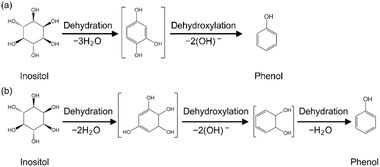
The conversion of myo-inositol to phenol requires the dehydration of three water molecules and the elimination of two OH groups from the reactant (Scheme 1). Here, the conversion of myo-inositol was performed over Pt/C in water for 1 h at 250 °C. The total yield of phenolic compounds was 42% (Fig. 1). To the best of our knowledge, this is the first report of direct conversion of cyclic sugar alcohols into aromatic compounds over supported metal catalysts without any acids. In addition to phenol, the other major products observed were catechol, resorcinol, and hydroquinone. Several minor products at yields below 1% were also observed and included cresol, ethylphenol, propylphenol, benzene, toluene, o-xylene, ethylbenzene, propylbenzene, naphthalene, 2-methylnaphthalene, biphenyl, cyclohexane, cyclohexanone, 1,2-cyclohexanediol, 1,2,3-cyclohexanetriol, and 1,3,5cyclohexanetriol. These products are labeled as “Other aromatic products” in the figure legend. The total carbon yield of the gaseous products was 31%, with the individual products being H2, CH4, CO2, and C2H6 obtained at a gas composition of 1.7%, 3.5%, 89%, and 5.9%, respectively (Table 1). The solid yield was 2.6 wt%. Then, all byproducts identified by GC-MS analysis are summarized in Table S1 in SI. In addition, the myo-inositol conversion experiments were repeated three times under the conditions (Pt/C in water at 250 °C for 1 h) to ensure the reliability of experimental results. The standard errors of the yields of aromatic compounds, gaseous products, and solid residues are also presented in Table S1 as representative values.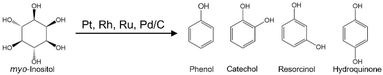 | ||
| Scheme 1 Conversion of myo-inositol into phenolic compounds in high-temperature water over charcoal-supported metal catalysts. | ||
| Catalyst | Gas yield (C%) | Gas composition (%) | Solid yield (wt%) | |||
|---|---|---|---|---|---|---|
| H2 | CH4 | CO2 | C2H6 | |||
| Pt/C | 31 | 1.7 | 3.5 | 89 | 5.9 | 2.6 |
| Rh/C | 26 | 1.7 | 6.1 | 86 | 6.2 | 12 |
| Ru/C | 15 | 0.7 | 0.4 | 99 | 0.0 | 25 |
| Pd/C | 0.0 | — | — | — | — | 63 |
| None | 0.0 | — | — | — | — | 86 |
To identify the most active catalyst, we repeated the conversion of myo-inositol over three additional charcoal-supported metal catalysts (i.e., Rh/C, Ru/C, Pd/C) (Fig. 1). The total yields of phenolic compounds decreased in the order of Pt/C (42%) > Rh/C (24%) > Ru/C (12%) > Pd/C (0.8%) > no catalyst (0%). Notably, the Rh/C catalyst afforded resorcinol at a yield of 6.3%, whereas Pt/C and Ru/C afforded yields of only around 1.0%. Furthermore, the Ru/C catalyst provided mainly catechol with only a small yield of phenol, which was quite different from the products afforded by the Pt/C and Rh/C catalysts, where phenol was the main product. Supported platinum catalysts are known to be stable in high-temperature water and to progress dehydroxylation of o-cresol.19 Consistent with this, Pt/C also facilitated the dehydroxylation of myo-inositol, showing high activity in its conversion to phenols in this study.
The yields of gaseous products followed the same order as the total yields of phenolic compounds: Pt/C (31%) > Rh/C (26%) > Ru/C (15%) > Pd/C (0%) > no catalyst (0%) (Table 1). Solid products were observed in all reactions, with the amounts inversely correlated to the yields of phenolic compounds. The formation of solid products decreased in the order of no catalyst (86%) > Pd/C (63%) > Ru/C (25%) > Rh/C (12%) > Pt/C (2.6%). It is possible that these solid products are humins, which are commonly formed during the aqueous-phase conversion of carbohydrate-based reactants.20 If that was the case, the implications would be that myo-inositol under the present reaction conditions but without catalyst is polymerized to humins at 250 °C, and that the Pt/C, Rh/C, and Ru/C catalysts avoid the formation of humins. The yields of aromatic compounds and gaseous products were calculated on a carbon basis, while the yield of solid products was calculated on a weight basis. For example, in the conversion of myo-inositol over Pt/C, the carbon yield of aromatic compounds was 44%, and that of gaseous products was 31%. The solid product yield was 2.6 wt%, which likely corresponds to a somewhat higher carbon yield, considering that the solid residue is primarily composed of carbonaceous humins. Thus, approximately 75–80% of the carbon input was accounted for. The remaining carbon is presumed to be present as unidentified organic compounds dissolved in the aqueous phase. However, total organic carbon analysis could not be performed to quantify these dissolved components because the reaction mixture was recovered using tetrahydrofuran as the solvent.
3.2. Optimization of reaction conditions over Pt/C
The most active catalyst, Pt/C, was characterized previously using XRD and adsorption measurements.21 The surface area of Pt/C was 1120 m2 g−1, and the Pt dispersion was 24.2%, as determined by gas adsorption analysis. In addition, the XRD pattern of Pt/C showed no diffraction peak attributable to platinum, indicating that the platinum particles were highly dispersed.Using the Pt/C catalyst, we optimized the reaction conditions for the conversion of myo-inositol into phenolic compounds. The optimal reaction temperature with respect to yield of phenolic compounds was 250 °C (Fig. 2). At 200 °C, the yields of both phenolic compounds and gaseous products were reduced, while the total amount of solid products was increased (Table S2). Also, the percentage contributions of catechol and resorcinol to the total yield of phenolic compounds at 200 °C were higher than those obtained at 250 °C. This implied that the formation of aromatic compounds and humins progressed competitively at the lower temperature. Meanwhile, the total yield of phenolic compounds at 300 °C decreased compared with that at 250 °C. Phenol was the predominant product at 300 °C and only trace amounts of other phenolic compounds were observed. This implies that increasing the reaction temperature from 250 to 300 °C enhanced phenol production by increasing the catalytic activity of the Pt/C catalyst for dehydroxylation.
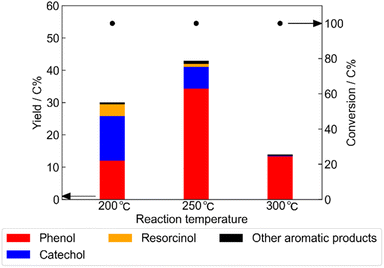 | ||
| Fig. 2 Yields of phenolic compounds obtained from the conversion of myo-inositol over Pt/C catalyst at various reaction temperatures (Pt/C 0.15 g; myo-inositol 0.20 g; water 1.0 g; reaction time 1 h). Data at 250 °C were taken from Fig. 1. | ||
The optimal amount of water with respect to yield of phenolic compounds was 1.0 g (Fig. 3). The yield of phenolic compounds decreased progressively with increasing amount of water (2.0 or 3.0 g). When the amount of water was 0.5 g, the yield of phenolic compounds decreased and the amount of solid products increased (Table S3). This implied that increasing the amount of water to more than 1.0 g suppressed the catalytic activity due to dilution of the myo-inositol. In contrast, decreasing the amount of water to below 1.0 g enhanced the formation of humins, likely due to the higher concentration of myo-inositol, resulting in a decrease in the yield of phenolic compounds.
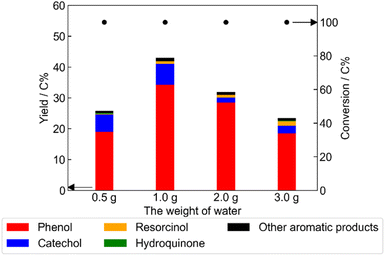 | ||
| Fig. 3 Yields of phenolic compounds obtained from myo-inositol over Pt/C catalyst with various weights of water (Pt/C 0.15 g; myo-inositol 0.20 g; reaction temperature 250 °C; reaction time 1 h). Data for 1.0 g of water were taken from Fig. 1. | ||
The optimal reaction time with respect to yield of phenolic compounds was 1 h (Fig. 4). Phenol and catechol yields increased with reaction time up to 1 h, but subsequently decreased, accompanied by an increase in gaseous product formation (Table S4). This implied that extending the reaction time beyond 1 h led to the increased production of phenol and catechol.
 | ||
| Fig. 4 Yields of phenolic compounds obtained from the conversion of myo-inositol over Pt/C as a function of reaction time from 0.5 to 2.0 h (Pt/C 0.15 g; myo-inositol 0.20 g; water 1.0 g; reaction temperature 250 °C). Data for the conversion at 60 min were taken from Fig. 1. | ||
Taking these findings together, we concluded that the optimized reaction conditions for the conversion of myo-inositol to phenolic compounds over Pt/C catalyst were Pt/C 0.15 g, myo-inositol 0.20 g, water 1.0 g, reaction temperature 250 °C, and reaction time 1 h. These optimized reaction conditions afforded a total phenolic compound yield of 42%, with the products primarily being phenol and catechol.
3.3. Reaction pathway from cyclic alcohol to aromatic compounds
In the context of converting cyclohexane rings into aromatic compounds, three key types of chemical reactions are often discussed: (1) dehydration, (2) dehydroxylation, and (3) dehydrogenation. The differences will be clearly illustrated using a cyclohexane ring as a model system. (1) Dehydration refers to the elimination of a water molecule (H2O) from the substrate. In a cyclohexane ring having hydroxy groups involves the removal of a hydroxy group (–OH) and a hydrogen atom (–H) from adjacent carbon atoms, resulting in the formation of a carbon–carbon double bond. For example, dehydration of cyclohexanol can lead to the formation of a cyclohexene intermediate. In the case of cyclohexanetriol, sequential dehydration of three water molecules can theoretically lead to the formation of a fully conjugated system, ultimately yielding a benzene ring. (2) Dehydroxylation involves the direct removal of a hydroxy group (–OH) from the substrate, typically using hydrogen to form water, without necessarily forming a double bond at that step. This process requires cleavage of the C–O bond and is usually facilitated by the surface of a solid catalyst, such as Pt/C. In the case of polyhydroxylated cyclohexane derivatives like myo-inositol, dehydroxylation steps are required to reduce the oxygen content of the products, enabling the formation of phenol. (3) Dehydrogenation refers to the removal of hydrogen atoms, typically as molecular hydrogen H2, from the substrate. In petrochemical processes, for example, cyclohexane can be converted to benzene via successive dehydrogenation steps over metal catalysts such as Pt or Cr-based systems at high temperatures. This process increases the degree of unsaturation and ultimately leads to aromatic ring formation. However, in aqueous phase biomass conversion systems especially under conditions where hydrogen evolution is small, dehydrogenation is less likely to be the dominant pathway. Instead, oxygen-removal processes such as dehydration and dehydroxylation are more relevant under these conditions.To examine the reaction pathway, we repeated the conversion over the Pt/C catalyst using different cyclic alcohols (Fig. 5 and Table 2). These additional cyclic alcohols were chiro-inositol, an isomer of myo-inositol, and cyclohexanetriol, cyclohexanediol, and cyclohexanol, which are cyclic alcohols with different numbers of hydroxy groups.
 | ||
| Fig. 5 Yields of products from the conversion of cyclic alcohols (Pt/C 0.15 g; cyclic alcohols 0.20 g; water 1.0 g; reaction temperature 250 °C; reaction time 1 h). Data for the myo-inositol conversion were taken from Fig. 1. | ||
| Cyclic alcohol | Gas yield (C%) | Gas composition (%) | Solid yield (wt%) | |||
|---|---|---|---|---|---|---|
| H2 | CH4 | CO2 | C2H6 | |||
| myo-Inositol | 31 | 1.7 | 3.5 | 89 | 5.9 | 2.6 |
| chiro-Inositol | 38 | 1.7 | 2.9 | 89 | 6.2 | 0.3 |
| 1,3,5-Cyclohexanetriol | 12 | 20 | 4.7 | 75 | 0.7 | 0.0 |
| 1,2,3-Cyclohexanetriol | 5.0 | 30 | 0.0 | 70 | 0.0 | 0.0 |
| cis-1,2-Cyclohexanediol | 5.4 | 51 | 0.0 | 49 | 0.0 | 0.0 |
| trans-1,2-Cyclohexanediol | 6.7 | 57 | 0.0 | 43 | 0.0 | 0.0 |
| Cyclohexanol | 0.4 | 90 | 0.0 | 9.6 | 0.0 | 0.0 |
The yields of phenolic compounds obtained from chiro-inositol were comparable with those from myo-inositol, implying that the stereochemistry of the hydroxy groups had little effect on the product yields. It is notable that the total yields of phenolic compounds decreased progressively with decreasing number of hydroxy groups in the cyclic alcohols (inositol > cyclohexanetriol > cyclohexanediol > cyclohexanol) while the yield of non-aromatic products progressively increased in the same order. This implied that the hydroxy groups in the cyclic alcohols were essential for the formation of aromatic rings. However, given that aromatic compounds were also produced from cyclohexanetriol and cyclohexanediol, the formation of aromatic rings from these compounds cannot be explained only by dehydration or dehydroxylation involving hydroxy groups.
The hydrogen yield increased with decreasing number of hydroxy groups in the cyclic alcohols (Table 2), suggesting that dehydrogenation of cyclic alcohols is also likely involved in the aromatic ring formation. If dehydrogenation is slower than dehydration in forming aromatic rings from the cyclic alcohols, the formation of aromatic rings from cyclic alcohols with fewer hydroxy groups becomes slower because cyclohexanetriol and cyclohexanediol require the slow dehydrogenation step. This would explain why the total yield of phenolic compounds decreased with decreasing number of hydroxy groups.
As shown in Table 2, the hydrogen yield from myo-inositol conversion was indeed very low, which strongly suggests that aromatic ring formation proceeds primarily through dehydration and dehydroxylation, rather than dehydrogenation. This is consistent with the highly oxygenated nature of myo-inositol, where the removal of hydroxy groups is a prerequisite for aromatization. Moreover, this result remains reasonable when considering that the hydrogen formed by gasification is consumed during the dehydroxylation. In contrast, for substrates such as cyclohexanediol and cyclohexanetriol, which possess fewer hydroxy groups, both dehydration and dehydrogenation appear to contribute to aromatic ring formation (Scheme S1 in SI). However, our data indicate that dehydrogenation competes with dehydroxylation, leading to the formation of non-phenolic byproducts and a decrease in phenol yield. Given the low hydrogen yield and the complexity of possible reaction pathways, the evidence for dehydrogenation remains circumstantial. While some contribution from dehydrogenation cannot be ruled out, the dominant pathway for myo-inositol conversion appears to be oxygen-removal-driven aromatization. In the conversion of myo-inositol to phenol, several plausible reaction pathways can be envisioned based on the molecular structure and the observed product distribution. myo-Inositol contains six hydroxy groups on a cyclohexane ring, and its transformation into phenol requires both the removal of oxygen-containing functional groups and the formation of an aromatic ring. One possible pathway involves the sequential dehydration of three water molecules (each involving the elimination of an –OH and an adjacent –H) to produce the benzene ring, followed by two dehydroxylation steps to remove additional hydroxy groups (Scheme 2(a)). This sequence could lead to the formation of phenol. However, we do not consider that the dehydroxylation of trihydroxybenzene shown in Scheme 2(a) is the main route to phenol formation, as discussed later. Alternatively, another plausible route involves two dehydration steps to introduce partial unsaturation, followed by two dehydroxylation steps and an additional dehydration to complete the aromatization process (Scheme 2(b)). These variations reflect the multiple ways in which oxygen can be removed from the highly hydroxylated inositol molecule. Considering the complexity of the system and the potential for parallel pathways, further mechanistic studies will be necessary to definitively determine the dominant route to phenol. Nonetheless, the current data suggest that both dehydration and dehydroxylation play critical roles in the oxygen-removal-driven aromatization of myo-inositol under high-temperature aqueous conditions over Pt/C catalysts. Griffin et al. investigated the deoxygenation of m-cresol over Pt/C and Pt/TiO2 using both experimental and computational approaches.22 Their DFT calculations revealed that the hydrogen-adsorbed Pt(111) surface facilitates ring hydrogenation and subsequent hydrodeoxygenation, with relatively low activation barriers. This surface state promotes C–O bond cleavage via hydrodeoxygenation, which is a key step in the conversion of myo-inositol to phenols.
High-temperature water exhibits unique properties that make it a suitable solvent for such transformations. At elevated temperatures (e.g., 250 °C), water has a significantly higher ionic product (e.g., [H+][OH−] ≈ 10−11) compared to room temperature (10−14), resulting in increased proton availability. This facilitates acid-catalyzed dehydration and dehydroxylation reactions, even in the absence of added acids. Indeed, we previously demonstrated that sorbitol or other sugar alcohols can be selectively dehydrated in high-temperature water without any acid additives, highlighting the catalytic role of water itself.23,24
To understand more about the reactions that occurred after the aromatic ring formation, we repeated the conversions using several dihydroxybenzenes (i.e., catechol, resorcinol, and hydroquinone) and three trihydroxybenzenes (Fig. 6 and Table S5). The conversions of the dihydroxybenzenes were all below 20%. Phenol was the only observed aromatic product, which was presumably obtained from dehydroxylation of the dihydroxybenzenes. The yields of phenol decreased in the order of hydroquinone > resorcinol > catechol, which was the opposite of the yields obtained from myo-inositol. In contrast, when the trihydroxybenzenes were used, the conversion was almost 100% and the obtained aromatic products were phenol and dihydroxybenzenes. In addition, the amount of solid products from the trihydroxybenzenes was greater than that from the dihydroxybenzenes, suggesting that the higher number of hydroxy groups promoted humin formation via polymerization of the reactants. Overall, the yields of phenolic compounds from the conversion of trihydroxybenzenes were 25% at most, which is lower than that from myo-inositol (42%). This result suggests that the dehydroxylation pathway of trihydroxybenzene shown in Scheme 2(a) is not the main route to phenol formation, as previously stated. The stability of the dihydroxybenzenes toward further conversion also indicated that phenol and dihydroxybenzenes were obtained as direct products of the conversion of inositol.
 | ||
| Fig. 6 Yields of aromatic products from the conversion of phenolic compounds over Pt/C catalyst (Pt/C 0.15 g; phenolic compounds 0.20 g; water 1.0 g; reaction temperature 250 °C; reaction time 1 h). | ||
3.4. Reusability of the Pt/C catalyst
We conducted recycling tests with the Pt/C catalyst (Fig. 7), subjecting the catalyst to a total of three use cycles. The total yield of phenolic compounds increased from 42% to 52% on the second use of the catalyst. However, when the catalyst was used for the third time, the total yield of phenolic compounds decreased slightly to 49%, but still remained higher than that obtained on the first use. Overall, the Pt/C catalyst remained highly active over the three runs; however, the phenol yield did progressively decrease with each reuse of the catalyst, while the yields of the dihydroxybenzenes (i.e., catechol, resorcinol, and hydroquinone) increased. This suggested that the Pt/C catalyst was gradually deactivated with respect to the dehydroxylation of myo-inositol. | ||
| Fig. 7 Recycling test using the Pt/C catalyst for the conversion of myo-inositol (Pt/C 0.15 g; myo-inositol 0.20 g; water 1.0 g; reaction temperature 250 °C; reaction time 1 h). Data for the conversion with fresh Pt/C were taken from Fig. 1. | ||
4. Conclusions
Here, we investigated the conversion of the cyclic sugar alcohol myo-inositol to aromatic compounds using charcoal-supported metal catalysts in high-temperature water. The highest total yield of phenolic compounds, which included phenol and catechol, was 42% when reacted over Pt/C catalyst. Notably, the yield of aromatic products obtained from myo-inositol was about ten times higher than that from glucose over Pt/C.13 The optimal conditions for the conversion of myo-inositol to aromatic compounds over Pt/C catalyst were Pt/C 0.15 g, myo-inositol 0.20 g, water 1.0 g, reaction temperature 250 °C, and reaction time 1 h. This is the first report of the direct conversion of a cyclic sugar alcohol into aromatic compounds over supported metal catalysts alone in water.To understand more about the formation of aromatic rings from cyclic sugar alcohols, we also carried out conversions using various other cyclic alcohols. The total yield of phenolic compounds decreased with decreasing number of hydroxy groups in the cyclic alcohols: inositol > cyclohexanetriol > cyclohexanediol > cyclohexanol, implying that the hydroxy groups in cyclic alcohols are important for the formation of aromatic rings. However, phenolic compounds were also produced from cyclohexanetriol and cyclohexanediol, indicating that the formation of aromatic rings from these compounds cannot be explained only by dehydration or dehydroxylation; thus, dehydrogenation of cyclic alcohols is also likely involved in the aromatic ring formation. The yields of phenolic compounds from trihydroxybenzenes were 25% at most, which is lower than those from myo-inositol (42%). This implies that dehydration of inositol to trihydroxybenzene is not the main route for the production of phenol. The stability of dihydroxybenzenes against further conversion indicated that phenol and dihydroxybenzenes were obtained directly as the main products of inositol conversion.
Recycling tests revealed that the Pt/C catalyst retained its catalytic activity for producing phenolic compounds from myo-inositol over three use cycles.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: tables S1, S2, S3, S4, and S5. See DOI: https://doi.org/10.1039/d5ra09573e.Acknowledgements
This work was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI (grant number JP22H00578).Notes and references
- BP Energy Outlook – 2025 edition; 2025, https://www.bp.com/en/global/corporate/energy-economics/energy-outlook.html, accessed 26 November 2025 Search PubMed.
- A. Busic, N. Mardetko, S. Kundas, G. Morzak, H. Belskaya, M. I. Santek, D. Komes, S. Novak and B. Santek, Food Technol. Biotechnol., 2018, 56, 289–311 Search PubMed.
- H. Z. Chen and X. G. Fu, Renewable Sustainable Energy Rev., 2016, 57, 468–478 CrossRef.
- L. Ouayloul, I. Agirrezabal-Telleria, P. Sebastien and M. El Doukkali, ACS Catal., 2024, 14, 17360–17397 CrossRef.
- X. Li, A. Kant, Y. He, H. Thakkar, M. Atanga, F. Rezaei, D. Ludlow and A. Rownaghi, Catal. Today, 2016, 276, 62–77 CrossRef.
- J. G. Zhang, J. Teo, X. Chen, H. Asakura, T. Tanaka, K. Teramura and N. Yan, ACS Catal., 2014, 4, 1574–1583 CrossRef.
- C. Z. Li, X. C. Zhao, A. Q. Wang, G. W. Huber and T. Zhang, Chem. Rev., 2015, 115, 11559–11624 CrossRef PubMed.
- K. Barta, G. R. Warner, E. S. Beach and P. T. Anastas, Green Chem., 2014, 16, 191–196 RSC.
- Y. X. Zhai, C. Li, G. Y. Xu, Y. F. Ma, X. H. Liu and Y. Zhang, Green Chem., 2017, 19, 1895–1903 RSC.
- A. Yamaguchi, N. Mimura, A. Segawa, H. Mazaki and O. Sato, J. Jpn. Pet. Inst., 2020, 63, 221–227 CrossRef.
- A. Yamaguchi, T. Watanabe, K. Saito, S. Kuwano, Y. Murakami, N. Mimura and O. Sato, Mol. Catal., 2019, 477, 6 Search PubMed.
- K. Yamazaki, A. Segawa, H. Mazaki, N. Hiyoshi, N. Mimura, O. Sato and A. Yamaguchi, RSC Adv., 2023, 13, 13472–13476 RSC.
- K. Yamazaki, N. Hiyoshi, O. Sato and A. Yamaguchi, J. Jpn. Pet. Inst., 2024, 67, 224–229 CrossRef.
- H. Choi, Water Sci. Technol., 2020, 82, 1120–1130 CrossRef PubMed.
- S. Watanabe, H. Ozawa, H. Kato, K. Nimura-Matsune, T. Hirayama, F. Kudo, T. Eguchi, K. Kakinuma and H. Yoshikawa, Biosci., Biotechnol., Biochem., 2018, 82, 161–165 CrossRef PubMed.
- K. Kakinuma, E. Nango, F. Kudo, Y. Matsushima and T. Eguchi, Tetrahedron Lett., 2000, 41, 1935–1938 CrossRef.
- B. Chen, W. Liu, A. Li, Y. Liu and Z. Chao, Dalton Trans., 2015, 44, 1023–1038 RSC.
- D. Yan, J. Feng, S. Li, Y. Yang, X. Yang, X. Shi, M. Li and Y. Zhang, J. Catal., 2023, 421, 1–11 CrossRef CAS.
- J. Dickinson and P. Savage, J. Mol. Catal. A:Chem., 2014, 388, 56–65 CrossRef.
- M. Schlaf, Z. Zhang, Reaction Pathways and Mechanisms in Thermocatalytic Biomass Conversion II: Homogeneously Catalyzed Transformations, Acrylics from Biomass, Theoretical Aspects, Lignin Valorization and Pyrolysis Pathways, Springer-Verlag Singapore Pte Ltd., Singapore, 2016 Search PubMed.
- K. Yamazaki, R. Sasaki, T. Watanabe, S. Kuwano, Y. Murakami, N. Mimura, O. Sato and A. Yamaguchi, Waste Biomass Valorization, 2021, 12, 6081–6089 CrossRef CAS.
- M. B. Griffin, G. A. Ferguson, D. A. Ruddy, M. J. Biddy, G. T. Beckham and J. A. Schaidle, ACS Catal., 2016, 6, 2715–2727 CrossRef CAS.
- A. Yamaguchi, N. Hiyoshi, O. Sato and M. Shirai, Green Chem., 2011, 13, 873–881 RSC.
- A. Yamaguchi, N. Muramatsu, N. Mimura, M. Shirai and O. Sato, Phys. Chem. Chem. Phys., 2017, 19, 2714–2722 RSC.
| This journal is © The Royal Society of Chemistry 2026 |