Open Access Article
Open Access ArticleA promising dual catalytic “Isothiourea + X” platform: from classic ionic to radical transformations
Bei Zhang
ab,
Yani Li
c,
Zhichao Ji
ab and
Jian Wang
 *c
*c
aSchool of Chemistry and Materials Science, Institute of Advanced Materials and Flexible Electronics (IAMFE), Nanjing University of Information Science and Technology, 219 Ningliu Road, Nanjing, 210044, China. E-mail: zhangbei@nuist.edu.cn
bChangzhou Sveck Photovoltaic New Material Co., Ltd., Changzhou, 213200, China. E-mail: Jizc@sveck.com.cn
cSchool of Pharmaceutical Sciences, Key Laboratory of Bioorganic Phosphorous Chemistry and Chemical Biology (Ministry of Education), Tsinghua University, Beijing, 100084, China. E-mail: wangjian2012@tsinghua.edu.cn
First published on 20th March 2026
Abstract
In recent decades, significant advancements have been made in isothiourea (ITU) catalysis, particularly in acyl transfer, silyl transfer, annulations, additions via C(1)-ammonium enolates and [2,3]-sigmatropic rearrangements. Despite these achievements, challenges such as a limited substrate scope of substrates and restricted reaction patterns still remain prevalent. The development of dual catalytic strategies involving secondary catalysts such as electricity, light, transition metals, and Brønsted acids has addressed these issues. This review focuses on the “Isothiourea + X” dual catalytic approach, highlighting recent breakthroughs that extend isothiourea catalysis from classic ionic reactions to radical transformations. Notable advances include the use of ITU-activated ammonium intermediates in asymmetric radical additions, as well as the light-driven generation of ketimine intermediates catalyzed by ITU. Furthermore, the integration of ITU with transition-metal catalysis has expanded its application, enabling reactions with a variety of in situ generated intermediates and promoting chiral cyclization. The review also examines the synergistic effects of ITU in combination with Brønsted acids, which enhance both reaction efficiency and stereocontrol. By summarizing these developments, the review provides valuable insights and directions for future research in ITU catalysis, particularly in the context of green, efficient, and asymmetric radical transformations across multiple fields.
1. Introduction
As is well known, aminocatalysis1,2 made significant contributions to asymmetric transformations, as recognized by the Nobel Prize in 2021. Isothiourea (ITU), an important tertiary amine organocatalyst, also deserves attention. Tracing back to its first success in the kinetic resolution (KR) of secondary alcohols in 2006,3 (S)-tetramisole and its derivative (R)-benzotetramisole made their debut. Shortly after, a nitrogen atom with Lewis basicity was successfully incorporated into a six-membered ring, creatively generating 3,4-dihydro-2H-pyrimido[2,1-b]benzothiazole (DHPB).4 DHPB demonstrated better reactivity in acyl transfer reactions compared to the classical 4-(dimethylamino)pyridine (DMAP). Two years later, the chiral (S)-homobenzotetramisole ((S)-HBTM) was reported by Birman and Li,5 exhibiting excellent efficiency and stereoselectivity in the KR process. In 2009, Birman and Smith independently developed a new generation of HyperBTM. Birman introduced a syn-methyl substituent at C(3) to synthesize (2S,3R)-HBTM-2,6 while Smith introduced a syn-isopropyl substituent at C(3) to construct (2S,3R)-HyperBTM.7 By far, most developments related to ITU have been based on making subtle modifications to these skeletons, and many asymmetric applications have also employed these structure-based catalysts.8,9Despite ITU catalysis achieving great success in acyl transfer,3,10–12 silyl transfer,13,14 annulations via α,β-unsaturated acyl ammonium intermediates,15–19 additions via C(1)-ammonium enolates,20–23 and diastereo- and enantioselective [2,3]-sigmatropic rearrangements,24,25 problems such as the narrow range of reaction substrates and monotonous reaction modes still remain. The synergistic catalytic strategy provides an effective solution to these challenges (Scheme 1). By incorporating a secondary catalytic cycle, such as photochemical,26 electrochemical,27 transition-metal-catalyzed,28 or Brønsted-acid29-catalyzed processes, a few of new catalytic modes have emerged, enabling broader utilization of common starting materials. This has significantly promoted the ITU-catalyzed chiral synthesis.
To the best of our knowledge, the known reviews on ITU mainly focus on reaction intermediates, synthetic applications or performance in stereochemistry. Therefore, the perspective on “Isothiourea + X” dual catalysis is of high value for a new understanding of ITU and its continuous development. This minireview aims to provide a specific summary of isothiourea in cooperation with other catalysts. Moreover, the economic and environmental friendliness is a common pursuit in today's scientific endeavors. The field of “Isothiourea + X” is a promising platform, potentially offering excellent applications in green and efficient asymmetric transformations via radical,30 diradical31 or ionic pathways32 for medicinal chemistry,33–36 agricultural chemistry,37 material chemistry,38–40 and beyond.41–43 Through an in-depth discussion of different interactions between catalysts and substrates, including single electron transfer, nucleophilic–electrophilic combination and acid–base non-covalent bonding, we aim to present the current achievements and the broad possibilities to readers.
2. Isothiourea/photochemical co-catalysis
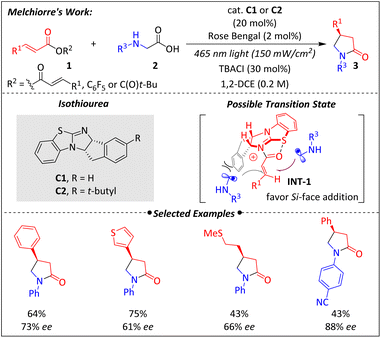
In 2022, Hartley, Melchiorre, and their colleagues reported a novel radical conjugate addition process for the synthesis of chiral pyrrolidinones44 (Scheme 2). As is well known, traditional catalytic radical conjugate addition strategies have strict limitations for the α,β-unsaturated carbonyl substrate due to the need for a suitable binding template for precise chirality control. This restriction was successfully resolved by using the unique S⋯O chalcogen interaction of isothiourea. Activating α,β-unsaturated esters via isothiourea (INT-1) made it possible to start from cheap feedstock carboxylic acids 1. Another innovation in this work was the development of radical transformations using isothiourea. Although there are many successful cases in polar nucleophilic addition, there have been no reports elucidating feasibility in enantioselective radical transformations. By employing a photocatalytic cycle, a nucleophilic α-amino radical was generated. This radical then underwent a formal Michael addition and a polar cyclization, which turned over the isothiourea catalyst and produced enantioenriched pyrrolidines.The method could be smoothly carried out with a diverse set of cinnamic acids and alkyl-substituted carboxylic acids. It is preferable to choose N-arylglycines over other N-substituted α-amino acids. Moreover, structures with pharmaceutical skeletons45–48 were also tested and showed good compatibility. The chirality control was proposed to arise from the relief of steric hindrance. The 1,5-S⋯O chalcogen interaction facilitated a favorable syn-coplanar conformation, thereby inducing selective Si-face addition (Scheme 2). Although the enantioselectivity was not excellent, this was the first case of asymmetric radical transformation utilizing isothiourea, demonstrating great potential.
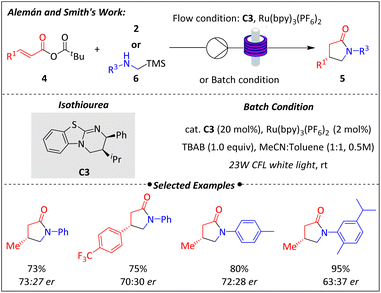
Around the same time, Alemán and Smith discovered that the (2R,3S)-HyperBTM isothiourea catalyst (C3) could mediate a radical conjugate addition involving mixed α,β-unsaturated anhydride 4 and radical precursor N-arylglycines 2, with the aid of photocatalysis49 (Scheme 3).
 | ||
| Scheme 3 Photo and isothiourea co-catalyzed asymmetric radical conjugate addition through batch or flow synthesis. | ||
Fortunately, the use of catalyst C3 enabled the synthesis of a series of enantioenriched (R)-isomers of pyrrolidinones 5, which were the enantiomers of those produced in Melchiorre's work. Although the enantiomeric excess of the products was not exceptionally high, the significance of this finding lies in demonstrating that chiral HyperBTM isothiourea has the ability to drive asymmetric radical transformations. Notably, this method was not only applicable to batch processes but also to flow synthesis. Under conditions similar to those used in batch reactions, the model reaction showed a significant improvement in yield with a slight decrease in enantioselectivity. The success was consistently replicated across many other examples, yielding high amounts of chiral pyrrolidinones with shorter reaction times. Besides N-arylglycines 2, α-silyl amines 6 were also compatible with the radical addition process. Mechanistic studies, including Stern–Volmer quenching plots, revealed no interaction between the photocatalysts and anhydride 4, weak potential for oxidation of 2 by Ru(bpy)3(PF6)2, and a clear interaction between N-arylglycines 2 and Ru(bpy)3(PF6)2.
In 2019, Song and colleagues50 discovered that visible light could trigger the formation of the ketene intermediate from α-diazoketones 8, which then generated C1-ammonium enolates (INT-3) in situ for subsequent [4 + 2] cyclizations with azadienes 7 (Scheme 4). This method enabled the efficient preparation of benzofuran-fused dihydropyridinone derivatives with an all-carbon quaternary stereocenter. The reaction required only 0.2 equivalents of a Lewis base, light irradiation, and an appropriate solvent, making the conditions straightforward. The ITU catalyst C4 demonstrated superior catalytic efficiency compared to other Lewis bases, such as NHCs and benzoylquinine. Among the precursors tested, α-diazoketones 8 were the most effective for ketene generation under light irradiation, as control experiments with carboxylic esters and carboxylic acids produced no products. The method also showed broad applicability with a range of azadienes and α-diazoketones and was easily scalable to 2.0 mmol. Mechanistic studies using 1H NMR revealed that the reaction of ketene with azadiene 7 occurred more rapidly than its formation via photoactivated Wolff rearrangement, underscoring the value of this protocol for in situ ketene generation through photoactivation.
 | ||
| Scheme 4 Sequential photoactivation and isothiourea catalysis for asymmetric synthesis of benzofuran-fused dihydropyridinone derivatives. | ||
3. Isothiourea/electrochemical co-catalysis
Most recently, Song and co-workers51 reported an elegant electricity-driven, isothiourea-catalyzed asymmetric α-enolation of carboxylic derivatives via a radical pathway (Scheme 5). Although the combination of nucleophilic ITU-activated esters with polar electrophiles to afford functionalized carbonyl compounds is well established, the coupling of nucleophilic silyl enol ethers with nucleophilic C1-ammonium enolates (INT-3) has remained challenging and rarely observed. In this work, a polarity inversion at the nucleophilic carbon of the enolate was achieved by merging electrochemical anodic single-electron oxidation with ITU-catalyzed formation of INT-3, generating an electrophilic α-carbonyl radical species (INT-4). This new type of synergistic catalysis provides an effective strategy to address the issue of mismatched polarities and the difficulty of controlling the three-dimensional structure of radical intermediates.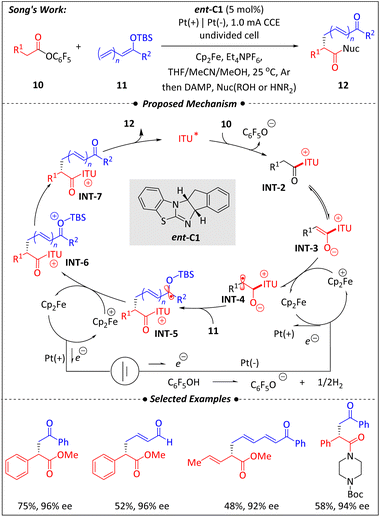 | ||
| Scheme 5 Cooperative electrochemical radical oxdiation and isothiourea catalysis for asymmetric α-enolation of esters. | ||
Control experiments revealed that even in the absence of isothiourea ent-C1, the alkylation adduct still could be obtained in 44% yield after 18 h. However, the presence of ent-C1 significantly accelerated the reaction compared to the background process. When ITU catalysis was combined with the electrocatalytic system, the oxidation potential of ester 10 was greatly reduced, thereby facilitating the anodic oxidation process, as supported by cyclic voltammetry (CV) data. Replacement of the electrode with other achiral oxidants led to a dramatic decrease in enantiomeric excess, highlighting the importance of the mutually reinforcing electro-isothiourea co-catalytic system in controlling chirality. Another crucial factor contributing to asymmetric induction may be the intramolecular 1,5-O⋯S chalcogen interaction between catalyst ent-C1 and the substrate, which restricts the conformational freedom of radical intermediate INT-4. The successful cooperation between the chiral Lewis base isothiourea and electrochemical catalysis demonstrates great promise for the development of valuable asymmetric radical transformations and underscores the potential of integrating chiral organocatalysis with electrochemical methods.
4. Isothiourea/transition metal co-catalysis
4.1. Isothiourea/Cu
In 2017, Song and Gong52 developed an effective decarboxylative cyclization of carboxylic acids (13) and 4-ethynyl dihydrobenzooxazinones (14) using synergistic catalysis by copper and isothiourea in an asymmetric manner (Scheme 6). Both catalysts were essential for this transformation. The choice of ligand for copper had little effect on the enantioselectivity but significantly impacted the diastereoselectivity. For example, changing different chiral Pybox ligands resulted in similar enantiomeric excesses (ee), and even achiral Pybox combined with C5 could yield a high ee. However, there is a significant variation in the diastereoselectivities, with ligand L1 showing the best performance. Similarly, although different substitutions and the chiral isothiourea backbones all achieved good ee values, not all could obtain satisfactory diastereomeric ratios, among which C5 was the best choice. Meanwhile, the protocol exhibited good tolerance for various functional groups.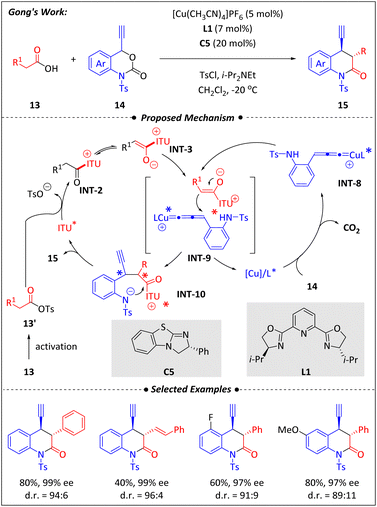 | ||
| Scheme 6 Copper and isothiourea co-catalyzed enantioselective [4 + 2] annulation of allenylidenes with C1 ammonium enolates. | ||
The plausible mechanism for the reaction involves a combination of highly compatible dual catalytic cycles. First, the carboxylic acid (13) is activated by TsCl and forms intermediate INT-2 with isothiourea C5. Tautomerization of INT-2 leads to the nucleophilic C1 ammonium enolate INT-3. Meanwhile, the copper complex combines with 4-ethynyl dihydrobenzo-oxazinones (14) to give the reactive intermediate INT-8. Subsequently, INT-3 carries out regio- and stereoselective attack on INT-8 thereby yielding the optically active intermediate INT-9.
With the release of copper and the occurrence of intramolecular lactamization, the target 3,4-dihydroquinolin-2-ones (15) with two stereocenters is ultimately formed and the isothiourea catalyst is released for next cycle.
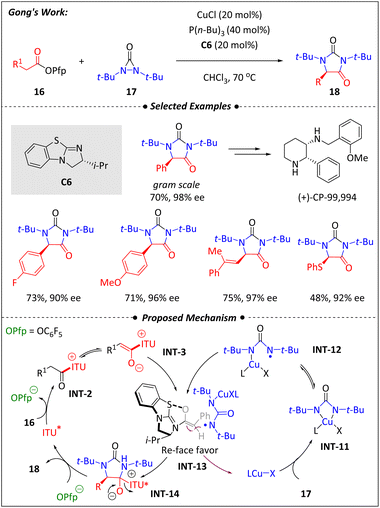
A year later, Gong and colleagues realized an asymmetric [4 + 2] annulation in α-amination of esters by employing the copper–isothiourea cooperative catalysis strategy53 (Scheme 7). Building on their discovery of an enantioselective α-propargylation of carboxylic acids, they became curious whether copper/isothiourea synergistic catalysis could be used to synthesize enantioenriched hydantoins (18). After multiple attempts, they discovered that a modified isothiourea catalyst (C6) combined with a copper chloride−tributylphosphine complex effectively catalyzed the reaction of 2-phenylacetates (16) with N,N-di-tert-butyldiaziridinone (17). Notably, the leaving group of phenylacetates (16) significantly influenced the final enantioselectivity, with pentafluorophenyl proven to be the optimal choice. Additionally, tributylphosphine played a crucial role in enhancing both the reaction efficiency and the chirality control.
Furthermore, the product served as a key intermediate in the synthesis of an NK1 antagonist.54 Theoretically, two potential pathways could lead to the target molecules. The first involves the nucleophilic attack of the C1-ammonium enolate (INT-3) on a four-membered Cu(III) species generated from the cleavage of the N–N bond of diaziridinone (17) by the Cu(I) catalyst. The second pathway involves the combination of enolate (INT-3) with a radical Cu(II) species (INT-12). The triplet signal with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 intensity ratio observed in the designed EPR experiments supports the radical pathway depicted in Scheme 7. The enantioselectivity may be attributed to the isopropyl group of C6, which shields the Si face of the C1-ammonium enolate, making the Re face more favorable for nitrogen radical addition.
1 intensity ratio observed in the designed EPR experiments supports the radical pathway depicted in Scheme 7. The enantioselectivity may be attributed to the isopropyl group of C6, which shields the Si face of the C1-ammonium enolate, making the Re face more favorable for nitrogen radical addition.
4.2. Isothiourea/Pd
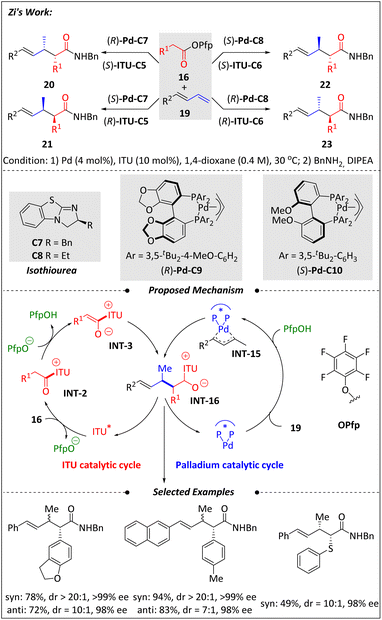
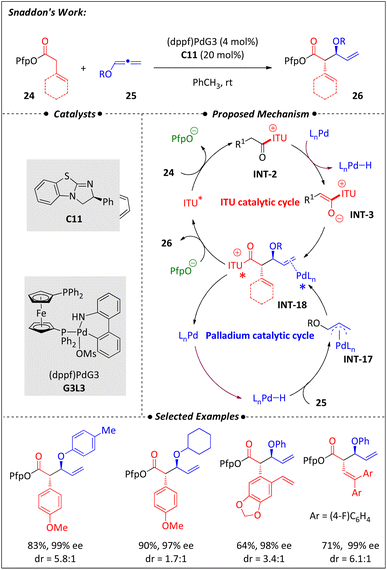
A stereodivergent coupling of pentafluorophenyl acetates (16) with 1,3-dienes (19) using cooperative palladium and isothiourea catalysis was discovered by Zi and coworkers55 (Scheme 8). Achieving stereodivergent diversity-oriented synthesis has long been a challenging issue. In response to this situation, researchers efficiently addressed these challenges by designing a dual catalytic system. By varying chiral catalysts, all four stereoisomers were synthesized with good yields and high enantioselectivities. Mechanistically, a nucleophilic C-1 ammonium enolate (INT-3) was generated via the activation of 16 by isothiourea, with the release of one equivalent of PfpOH. The acidic environment facilitated the formation of π-allyl-Pd intermediates (INT-15) from 1,3-dienes (19) and a Pd(0) complex. The coupling of the nucleophilic INT-3 with the electrophilic INT-15 resulted in the intermediate INT-16, which was then converted to the target product following a PfpO− rebound. This method exhibits a broad substrate scope and enables various product derivatizations. Notably, using this Csp3–Csp3 coupling as a key transformation step, the pain-relief drug tapentadol was synthesized in eight steps with an overall yield of 47%.In 2022, the Snaddon group56 reported a synergistic Pd–H and isothiourea catalysis method for obtaining anti-aldol fragments. This catalytic strategy enables the combination of acyclic esters (24) and oxyallenes (25) to yield various chiral β-oxy-carbonyl derivatives (26) (Scheme 9). The isothiourea skeleton has an important impact on the reaction yield, while the bisphosphine ligand influences multiple factors including reactivity, hydricity, regioselectivity, and the syn/anti ratio. The stable and efficient 3rd-generation Buchwald precatalyst G3L3 provides the best performance. This method is compatible with oxyallenes bearing both alkylated and arylated O-substituents. The compatibility of both α-aryl and α-alkenyl esters further illustrates the broad scope of this approach.57–60
 | ||
| Scheme 9 Enantioselective synthesis of anti-aldol motifs through synergistic Pd–H and isothiourea catalysis. | ||
Mechanistic studies highlight the close coupling of the two catalytic cycles through two key observations: the transformation proceeds without the need for any additional base beyond isothiourea, and the generation of the crucial LnPd–H intermediate does not require an extra Brønsted acid. The proposed mechanism begins with the formation of acylammonium ion INT-2 through the activation of achiral ester 24 via isothiourea catalysis C11. This ion was intercepted by Pd, yielding the corresponding C1-ammonium enolate INT-3 and LnPd–H. The resulting regioselective hydropalladation by LnPd–H generates O-substituted π(allyl)Pd INT-17. The nucleophilic enolate INT-3 then combines with the electrophile INT-17, producing the key intermediate INT-18 with high enantioselectivity under the influence of the dual chiral catalytic system. Finally, the rebound of PfpO− leads to the formation of the target anti-aldol β-oxy-carbonyl derivatives 26, along with the release of both catalysts.
Utilizing the similar strategy, the same group realized the asymmetric preparation of α-alkyl 2-pyrrole skeleton,61 a high valuable synthon. Employing N-methyl-2-pyrrole Pfp esters and allyl sulfonates or allyl carbonates as raw materials, isothiourea C5 and Xantphos G3P3 as the co-catalysts, a library of desired molecules could be easily obtained with good yields and enantioselectivities.
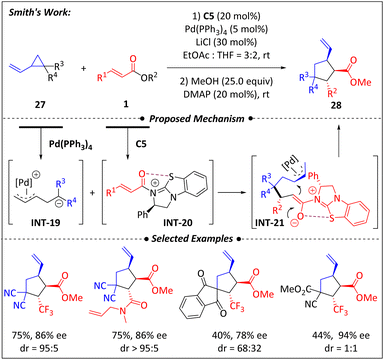
In 2022, Smith and colleagues reported an enantioselective [3 + 2] annulation of α,β-unsaturated esters 1 and vinylcyclopropanes 27 using a dual catalytic strategy involving catalyst isothiourea and palladium7 (Scheme 10). It was the first example where isothiourea/metal co-catalysis achieved high compatibility with α,β-unsaturated esters. Interestingly, the additive salts (20–30 mol%) had an unexpected influence on the stereoselectivity of the reactions. Further investigations reveals that halide ions have a positive effect on diastereocontrol, with LiCl being considered the best option. Additionally, the influence of halide ions on transition metal catalysis was explored through both experimental and computational studies. The increased π–σ–π isomerization rate within π-Pd-allyl intermediates facilitated by Cl− ions further supported the role of salt additives. The optimal solvent for this reaction was found to be a mixture of THF and EtOAc in a ratio of 2/3. The proposed mechanism involves two crucial intermediates: a zwitterionic π-allyl Pd intermediate INT-19 generated from vinylcyclopropanes 27 via a palladium-catalyzed ring-opening process, and an α,β-unsaturated acyl ammonium species INT-20 generated from the activation of 1 by isothiourea C5. These intermediates then undergo a formal [3 + 2] cycloaddition, releasing the two catalysts and forming the final products.
Gong, Han, and colleagues62 designed a cascade [1 + 1 + 4] and [1 + 1 + 2] annulation to build enantioenriched dihydropyridones (31) and β-lactams (33) using a palladium and isothiourea co-catalytic strategy (Scheme 11). The key innovation lay in the highly compatible catalysis of palladium and isothiourea, which facilitated the generation of a C1 ammonium enolate (INT-3) from commercially available halides (29) and carbon monoxide (CO). Initial trials revealed that CO pressure was a significant factor, as high pressure promoted the CO insertion process but could potentially suppress oxidative addition and ketene formation. The isothiourea scaffold (C1) played a crucial role in achieving high diastereoselectivity and enantioselectivity. Under optimized conditions, a variety of bromides (29) and N-tosylimines, including sultam-derived cyclic imines, oxindole-derived α,β-unsaturated imines, 2-naphthyl, and 3-furanyl N-tosylimines, could be utilized to obtain the desired optically pure products. These chiral products, after undergoing several common transformations, could be converted into valuable synthetic building blocks and bioactive molecules. Mechanistic kinetic isotope effect (KIE) studies suggest that the rate-limiting step likely involves C–H cleavage.
 | ||
| Scheme 11 Asymmetric three component [1 + 1 + n] (n = 2 or 4) annulations via palladium and isothiourea dual catalysis. | ||
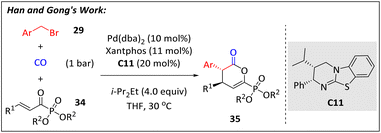
Shortly after, the same team applied this mechanism to the construction of chiral phosphorus-containing compounds63 (Scheme 12). Starting from readily accessible benzyl bromides (29), CO, and α-ketophosphonates (34), a series of phosphono dihydropyranones (35) with potential therapeutic effects against influenza viruses was smoothly prepared via isothiourea and palladium cooperative catalysis. Additionally, the same research team demonstrated that vinyl benzoxazinanones could also be utilized in this catalytic mode for the assembly of chiral quinolinones with good yields and high enantioselectivities.64
 | ||
| Scheme 12 Enantioselective three component [1 + 1 + 4] annulation for synthesis of phosphono dihydropyranones. | ||
Most recently, Sayed and Han et al. reported a three-component formal [4 + 2] cyclization using azadienes to construct a library of enantio-enriched 3,4-dihydrobenzofuro[3,2-b]pyridine derivatives via a similar dual catalytic system.65
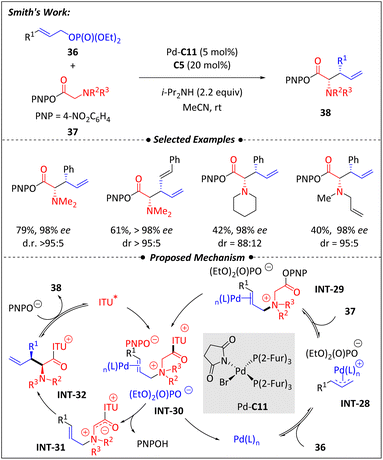
In 2017, the Smith group66 developed a palladium and isothiourea co-catalyzed tandem relay allylic amination and [2,3]-sigmatropic rearrangement for preparing enantio-riched α-amino acid fragments (Scheme 13). A series of preliminary tests were conducted for this design. Ethyl carbonates successfully underwent the reaction without the addition of extra base, achieving a yield of 75%. This result suggests that the ethyl carbonate and/or ethoxide released during the allylic substitution process has sufficient basicity to drive the [2,3]-rearrangement step. In other words, ethyl carbonates, as precursors, are not suitable for the enantio-control step.
 | ||
| Scheme 13 Asymmetric allylic amination and [2,3]-sigmatropic rearrangement for synthesis of α-amino acid. | ||
Taking into account the effect of the decreased basicity of the released counterions, cinnamyl ethyl phosphate (36) was chosen for testing. As expected, it produced the desired product only in the presence of an external base, without interfering with the [2,3]-rearrangement process. The suitable palladium is the secret to successfully achieving enantioselectivity and reaction efficiency. The succinimide-based Pd complex firstly developed by Fairlamb and co-workers showed the best performance for this reaction.67 Also the isothiourea had unignorable contribution in reaction yield as well as stereoselectivity. The variation of cinnamic aryl substituent benign to the allylic phosphate, N-substituents within the glycine ester and unsymmetrical N-allyl-N-methylglycine ester exhibited the broad utilization of this protocol. The relay catalytic process is depicted in Scheme 13. Pd coordinates with cinnamyl ethyl phosphate (36) to generate allylic ammonium intermediate (INT-28). Subsequently, the glycine ester 37 undergoes nucleophilic attack by INT-28 to form INT-29. Immediately afterward, isothiourea C5 attacks INT-29 to afford INT-31. After the [2,3]-sigmatropic rearrangement and catalyst release, the α-amino ester products 38 was finally obtained. Tests on branched cinnamyl phosphates indicate that the terminal position with minimal sterical hindrance favors the formation of the proposed Pd-π-allyl intermediate and the necessary ammonium salt required for the [2,3]-rearrangement. Testing of (Z)-cinnamylammonium salts suggests that isomerization of (Z)-36 has less possibility to occur before initial oxidative addition. A mixture of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of allylic ammonium salt (INT-29) and N-pyrrolidinylglycine ester (37) was tested, and the resuts shows that the formation of the allylic ammonium salt is partially reversible during the catalytic process.
1 ratio of allylic ammonium salt (INT-29) and N-pyrrolidinylglycine ester (37) was tested, and the resuts shows that the formation of the allylic ammonium salt is partially reversible during the catalytic process.
4.3. Isothiourea/Ir
In 2017, Hartwig and co-workers developed a convenient method for constructing all four possible stereoisomers of active esters with two adjacent chiral centers68 (Scheme 14). Starting from aryl acetic esters and allylic carbonates, the cooperation between a metallacyclic iridium complex (C12 or C13) and a chiral isothiourea (C5 or C11) leads to stereodivergent products.The desired configuration of the products can be controlled by selecting the appropriate catalyst. Aryl substituents on the phosphoramidite ligands can influence regioselectivity and diastereoselectivity to a certain degree. Additionally, the structure of the isothiourea is another crucial factor affecting reaction yields and enantioselectivity. The key intermediate INT-35 is formed from the combination of a C1 ammonium enolate INT-3, generated in the isothiourea catalytic cycle, and an allyl-Ir-complex INT-33, formed by the coordination of Ir to allylic carbonates. A rebound strategy is then employed to release the catalyst without disrupting the competitive nucleophilic attack on the electrophilic intermediate INT-33. The low concentration and low nucleophilicity of phenolate (PfpO) effectively facilitate catalyst turnover.
The resulting products can be further transformed into amides, esters, and carboxylic acids with high enantioselectivities.
In 2019, Pearson and Snaddon reported a flexible and modular protocol for preparing enantio-pure homoallylic amines in both linear and branched forms.69 By matching the appropriate transition-metal catalyst with an isothiourea catalyst, all four isomers of the targeted amines with the desired regioselectivity could be easily accessed from simple pentafluorophenyl esters and allyl electrophiles (Scheme 15). Specifically, by employing Buchwald's Xantphos-ligated 3rd generation Pd-C14 with Lewis base C5, the electrophilic π(allyl)Pd complex (INT-34) combined with the nucleophilic C1 ammonium enolate (INT-3), leading to the formation of the primary amide intermediate INT-35 after continuously bubbling NH3 gas for ten minutes. A Hofmann-type rearrangement then tends to occur, forging a C(sp3)–N bond and forming an active isocyanate INT-37, which can be easily intercepted by a nucleophilic alcohol (R3OH) to provide the final linear amines with carbamate-protected groups.
 | ||
| Scheme 15 Regio- and stereodivergent allylic alkylation and Hofmann rearrangement for synthesis homoallylic amines via Pd/Ir and isothiourea dual catalysis. | ||
When the palladium catalyst C14 or C15 is replaced by Hartwig's cyclometalated iridium(I)–phosphoramidite C12, branched homoallylic amines can be obtained through a similar pathway. This method has high value in the synthesis of medicinally important molecules. For instance, the Boc-protected benzyldimethylsilyl-substituted homoallylic amine can be easily converted into sertraline, which has therapeutic effects as a selective serotonin reuptake inhibitor,70,71 via a key Hiyama cross-coupling step. Additionally, the special branched homoallylic amine achieved via iridium catalysis can be converted into an MDM2 inhibitor.72
Song and colleagues73 developed an enantioselective [3 + 2] cyclization method for assembling chiral γ-lactam-containing derivatives using an iridium and isothiourea cooperative catalysis strategy (Scheme 16).
 | ||
| Scheme 16 Enantioselective [3 + 2] cyclization of vinyl aziridines and pentafluorophenyl esters via isothiourea and iridium co-catalysis. | ||
The resulting γ-lactam products with extended vinyl substituents can be easily transformed into various other useful chiral molecules.
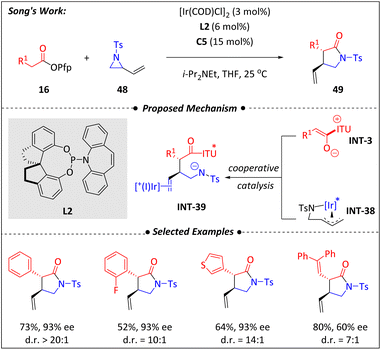
4.4. Isothiourea/Rh
The use of [Ir(COD)Cl]2, paired with the P-olefin ligand L2, facilitates the formation of (η3-allyl)iridium(III) species INT-38 and significantly influences the stereoselectivity of the subsequent reaction with the nucleophilic enolate generated from the isothiourea catalytic cycle.Recently, Song and Gong et al.74 developed a synergistic catalytic platform combining isothiourea and rhodium for the asymmetric [4 + 2] and [2 + 2] cyclization of terminal alkynes (50) with α,β-unsaturated ketimines (30) or fluorinated ketones (52) (Scheme 17). It is proposed that the terminal alkyne is first activated by rhodium to form the η1-Rh vinylidene complex INT-40. Subsequently, oxidation of INT-40 by 4-picoline N-oxide facilitates the oxygen transfer, generating the η2-Rh ketene species INT-41. The subsequent demetallation of the ketene promotes the formation of C1 ammonium enolate (INT-3), catalyzed by isothiourea C16. Annulation of INT-3 with α,β-unsaturated ketimines (30) or fluorinated ketones (52) ultimately produces optically active dihydropyridone and β-lactone frameworks. The electron-deficient P(3,5-(CF3)2C6H3)3 along with the addition of (PhO)2POOH significantly enhanced the reaction yield and enantioselectivity.
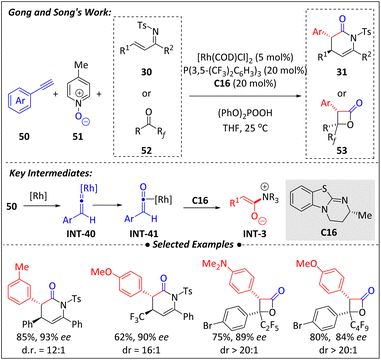 | ||
| Scheme 17 Asymmetric [4 + 2] and [2 + 2] annulations of terminal alkynes via rhodium and isothiourea synergestic catalysis. | ||
4.5. Isothiourea/Au
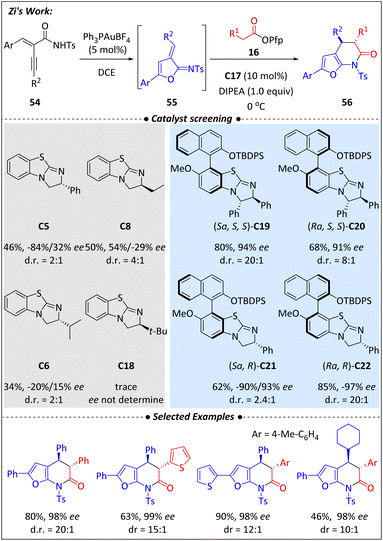
Most recently, the Zi group,75 inspired by enzyme-catalyzed reactions, designed a series newly chiral isothiourea catalysts that possess both central and axial chirality. Traditional stereo-control most rely on the adjacent bulky hindrance in the active region, while covalent activation with enzyme catalysis mainly determined by a three-dimensional (3D) pockets with long-range stereocontrol to realize an excellent stereoselectivity. Following such a mimic concept, a library of AxBTMs (C19–C22) containing two central chiral centers and one atropisomeric center was synthesized. These newly designed catalysts were first directly tested in the [2 + 2] cycloadditions between imines and α-fluoro esters, showing significant improvements in reaction yield and stereocontrol. Similar enhancements were observed in the [2 + 2] cycloaddition between α-fluoro esters and oxindoles.Based on these successful achievements, a Au/isothiourea relay catalysis for the [4 + 2] annulation of pentafluorophenyl esters and enyamides was developed (Scheme 18). However, due to the strong coordination between Au+ and the electron-riched ITU, fully merging Au/ITU cooperactive catalysis has proved to be difficult to achieve. To address this problem, a stepwise feeding strategy was employed. An in situ ortho-quinone methide intermediate (55) was first generated through the cyclizing of enyamide (54) via gold catalysis. The target products (56) were subsequently obtained through [4 + 2] cyclization in the isothiourea catalytic cycle. Notably, the newly designed ITUs (C19–C22) consistently exhibited superior yields and chiral control compared to traditional isothiourea catalysts. It was proposed that the long-range axial arm helped mitigate the Au/ITU coordination, thereby improving reaction efficiency. Additionally, the [4 + 2] cyclization exhibited varying preferences for the combinations of central and axial chiral ITUs, with the catalyst C22 proving to be the most effective.
Several control experiments were conducted to gain a deeper understanding of chirality control. It was concluded that the adjacent steric hindrance provided fundamental support for the formation of stereoselectivity, while the distal axial substituents enhanced the differentiation of isomers during the bond-forming process, resulting in higher diastereoselectivities and enantioselectivities.
5. Isothiourea/Brønsted acid co-catalysis
In 2021, the Smith group76 reported an asymmetric Mannich addition of the enolate precursor pentafluorophenyl ester (16) to the iminium precursor hemiaminal ether (57), yielding enantio-enriched α-aryl-β2-amino esters (58) via cooperative Brønsted acid and isothiourea catalysis (Scheme 19). The choice of Brønsted acid halide salts significantly affected the reaction yield, particularly the chloride salts, which contribute greatly to enantioselectivity. The scope and limitations of this method were thoroughly investigated. The variation in the aryl group on ester 16 had little influence, whereas the stronger nucleophilicity of the nitrogen in ether 57 led to the formation of amide products. The successful total syntheses of (S)-venlafaxine·HCl and (S)-N akinadine B highlight the practical effectiveness of this method. The stereo-controlling step was closely related to the Mannich addition of the C1-ammonium enolate rather than a dynamic kinetic resolution (DKR) of the α-aryl-β2-amino esters, as confirmed by control experiments. Additionally, the ee values of the products showed a linear correlation with the catalysts. The reversible addition of isothiourea to hemiaminal ether (57) also led to the formation of the desired iminium intermediate INT-42, without affecting the reaction outcome. An isolated acyl-ammonium species containing isothiourea (INT-43) can also serve as a precatalyst.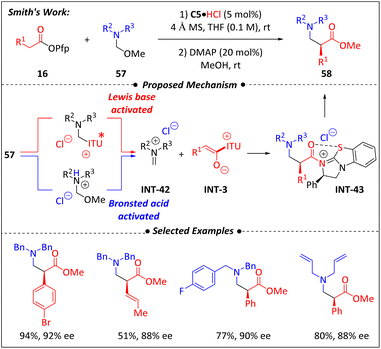 | ||
| Scheme 19 Asymmetric assembly of α-aryl-β2-amino esters via Brønsted acid and isothiourea co-catalysis. | ||
Another case of cooperative catalysis involving isothiourea and Brønsted acid was reported by Gong, Song, and colleagues.77 Through a formal [3 + 3] cyclization of unsaturated aryl esters 1 with enamines 59, a variety of 3,4-dihydropyridin-2-one derivatives 60 were efficiently prepared via this synergistic system (Scheme 20). Notably, the reaction could proceed without the additional Brønsted acid, achieving up to 72% yield and 83% ee. However, with the introduction of diphenylphosphinic acid, the yield increased significantly to 99% and the enantioselectivity improved to 97% ee. These results indicate that diphenylphosphinic acid substantially accelerates the reaction and strengthens the interaction between enamines 59 and the α,β-unsaturated acyl ammonium intermediate. A proposed transition state INT-44 involving all three species is illustrated in Scheme 20. The subsequent intermediate INT-45 undergoes an intramolecular amination and the release of the isothiourea catalyst, ultimately forming the cyclic product.
 | ||
| Scheme 20 Asymmetric [3 + 3] cyclization for synthesizing 3,4-dihydropyridin-2-ones via Brønsted acid and isothiourea dual catalysis. | ||
6. Summary and outlooks
In summary, this review highlights the remarkable efficiency of chiral isothiourea (ITU) catalysts when synergized with other catalytic systems. One of the main focuses is the radical transformation process catalyzed by ITU, which is used for the preparation of enantioenriched cyclic and acyclic compounds. A ground breaking study reported in 2022 showed that ITU-activated α,β-unsaturated ammonium intermediates can undergo asymmetric radical addition followed by amidation reaction. A recent significant advancement further confirms the tremendous potential of ITU catalysis in asymmetric radical transformations. The reaction is achieved through cooperation with electroanodic single-electron-transfer (SET) oxidation and proceeds via an inverse addition pathway. In addition, successful cases have shown that through a relay co-catalytic pathway, the photo-driven ketimine intermediates can be activated by ITU, thereby opening new avenues for photocatalytic applications. Although research in this filed is still limited, the combination of ITU and photocatalysis shows considerable application potential.Another area worth attention is the merging of ITU catalysis and transition-metal catalysis. The classical ITU-catalyzed chiral cyclization has been extended to explore in situ generated reactive intermediates such as copper–allenylidene, radical Cu(II) species, π-allyl-Pd, π-allyl-Ir, and others. Furthermore, relay catalysis involving Pd, Rh, or Au in combination with ITU has been shown to efficiently generate C1-ammonium enolates from readily available starting materials such as benzyl bromides, CO, terminal alkynes, and enynamides. Finally, synergistic systems combining ITU with Brønsted acids have also been explored. Studies have found that Lewis basic environments are highly compatible with added acids, enhancing both reaction efficiency and stereocontrol through anion counteraction or hydrogen-bonding interactions.
By summarizing these advancements, we hope to provide valuable insights and guidance for future explorations of “ITU + X” dual catalytic systems, particularly in promoting the development of radical transformations that are green, efficient, and application-oriented.78–86
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this paper.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
We thank the National Natural Science Foundation of China (22071130, 22571179), the Bayer Investigator fellow, the fellowship of Tsinghua-Peking Centre for Life Sciences (CLS), Natural Science Research Fund of Jiangsu Higher Education Institutions (25KJB150021), and the Start-up Foundation for Introducing Talent of NUIST (1513142501010).Notes and references
- B. List and J. W. Yang, Science, 2006, 313, 1584–1586 CrossRef CAS PubMed.
- D. A. Nicewicz and D. W. C. MacMillan, Science, 2008, 322, 77–80 CrossRef CAS PubMed.
- V. B. Birman and X. Li, Org. Lett., 2006, 8, 1351–1354 CrossRef CAS PubMed.
- M. Kobayashi and S. Okamoto, Tetrahedron Lett., 2006, 47, 4347–4350 CrossRef CAS.
- V. B. Birman and X. Li, Org. Lett., 2008, 10, 1115–1118 CrossRef CAS PubMed.
- Y. Zhang and V. B. Birman, Adv. Synth. Catal., 2009, 351, 2525–2529 CrossRef CAS PubMed.
- J. Bitai, A. J. Nimmo, A. M. Z. Slawin and A. D. Smith, Angew. Chem., Int. Ed., 2022, 61, e202202621 CrossRef CAS PubMed.
- J. Merad, J.-M. Pons, O. Chuzel and C. Bressy, Eur. J. Org Chem., 2016, 2016, 5589–5610 CrossRef CAS.
- A. Biswas, H. Mondal and M. S. Maji, J. Heterocycl. Chem., 2020, 57, 3818–3844 CrossRef CAS.
- M. D. Greenhalgh, S. M. Smith, D. M. Walden, J. E. Taylor, Z. Brice, E. R. T. Robinson, C. Fallan, D. B. Cordes, A. M. Z. Slawin, H. C. Richardson, M. A. Grove, P. H.-Y. Cheong and A. D. Smith, Angew. Chem., Int. Ed., 2018, 57, 3200–3206 CrossRef CAS PubMed.
- J. Merad, P. Borkar, F. Caijo, J.-M. Pons, J.-L. Parrain, O. Chuzel and C. Bressy, Angew. Chem., Int. Ed., 2017, 56, 16052–16056 CrossRef CAS PubMed.
- S. Qu, S. M. Smith, V. Laina-Martín, R. M. Neyyappadath, M. D. Greenhalgh and A. D. Smith, Angew. Chem., Int. Ed., 2020, 59, 16572–16578 CrossRef CAS PubMed.
- R. W. Clark, T. M. Deaton, Y. Zhang, M. I. Moore and S. L. Wiskur, Org. Lett., 2013, 15, 6132–6135 CrossRef CAS PubMed.
- C. I. Sheppard, J. L. Taylor and S. L. Wiskur, Org. Lett., 2011, 13, 3794–3797 CrossRef CAS PubMed.
- E. R. T. Robinson, C. Fallan, C. Simal, A. M. Z. Slawin and A. D. Smith, Chem. Sci., 2013, 4, 2193–2200 RSC.
- Y. Fukata, K. Asano and S. Matsubara, J. Am. Chem. Soc., 2015, 137, 5320–5323 CrossRef CAS PubMed.
- G. Liu, M. E. Shirley, K. N. Van, R. L. McFarlin and D. Romo, Nat. Chem., 2013, 5, 1049–1057 CrossRef CAS PubMed.
- M. E. Abbasov, B. M. Hudson, D. J. Tantillo and D. Romo, J. Am. Chem. Soc., 2014, 136, 4492–4495 CrossRef CAS PubMed.
- A. Matviitsuk, M. D. Greenhalgh, D.-J. B. Antúnez, A. M. Z. Slawin and A. D. Smith, Angew. Chem., Int. Ed., 2017, 56, 12282–12287 CrossRef CAS PubMed.
- C. McLaughlin and A. D. Smith, Chem. – Eur J., 2021, 27, 1533–1555 CrossRef CAS PubMed.
- C. A. Leverett, V. C. Purohit and D. Romo, Angew. Chem., Int. Ed., 2010, 49, 9479–9483 CrossRef CAS PubMed.
- D. Belmessieri, L. C. Morrill, C. Simal, A. M. Z. Slawin and A. D. Smith, J. Am. Chem. Soc., 2011, 133, 2714–2720 CrossRef CAS PubMed.
- J. D. Parkin, R. Chisholm, A. B. Frost, R. G. Bailey, A. D. Smith and G. Hähner, Angew. Chem., Int. Ed., 2018, 57, 9377–9381 CrossRef CAS PubMed.
- T. H. West, D. S. B. Daniels, A. M. Z. Slawin and A. D. Smith, J. Am. Chem. Soc., 2014, 136, 4476–4479 CrossRef CAS PubMed.
- T. H. West, D. M. Walden, J. E. Taylor, A. C. Brueckner, R. C. Johnston, P. H.-Y. Cheong, G. C. Lloyd-Jones and A. D. Smith, J. Am. Chem. Soc., 2017, 139, 4366–4375 CrossRef CAS PubMed.
- J. Liu, W. Zhang, X. Tao, Q. Wang, X. Wang, Y. Pan, J. Ma, L. Yan and Y. Wang, Org. Lett., 2023, 25, 3083–3088 CrossRef CAS PubMed.
- X. Xu, W. Yang, Y. Zhang, Y. Wang, Z. Mao, X. Peng, Y. Ling, Z. Zhang and H. Chen, Microchim. Acta, 2025, 192, 439 CrossRef CAS PubMed.
- H. Wang, J. Li, M. Xu, Q. Zhou, W. Xu and M. Ye, Angew. Chem., Int. Ed., 2025, 64, e202413652 CrossRef CAS PubMed.
- Q. Zhang, X. Lu, H. Wang, X. Tian, A. Wang, H. Zhou, J. Wu and Y. Tian, Chem. Commun., 2018, 54, 3771–3774 RSC.
- F. Lv, X. Guo, H. Wu, H. Li, B. Tang, C. Yu, E. Hao and L. Jiao, Chem. Commun., 2020, 56, 15577–15580 RSC.
- N. Xu, S. Liu, Q. Xu, P. Yuan, P. Zhang, S. Zhuo, C. Zhu and J. Du, Appl. Catal. Gen., 2024, 673, 119586 CrossRef CAS.
- W. Huang, G. Han, D. Wang, Y. Zhu, H. Wang, Z. Liu, K. Uvdal, J. Geng, Z. Hu, R. Zhang and Z. Zhang, J. Am. Chem. Soc., 2025, 147, 4147–4158 CrossRef CAS PubMed.
- X. Xu, J. Wang, M. Wang, X. Yuan, L. Li, C. Zhang, H. Huang, T. Jing, C. Wang, C. Tong, L. Zhou, Y. Meng, P. Xu, J. Kou, Z. Qiu, Z. Li and J. Bian, J. Med. Chem., 2021, 64, 4588–4611 CrossRef CAS PubMed.
- L. Su, X. Zhu, H. Ding, L. Hu, J. Chen, S. Qi, K. Luo, W. Ling and X. Tian, Sens. Actuators, B Chem., 2024, 412, 135792 CrossRef CAS.
- H. Wang, B. Fang, L. Kong, X. Li, Z. Feng, Y. Wu, K. Uvdal and Z. Hu, Spectrochim. Acta, Part A Mol. Biomol. Spectrosc., 2018, 198, 304–308 CrossRef CAS PubMed.
- X. Guo, W. Sheng, H. Pan, L. Guo, H. Zuo, Z. Wu, S. Ling, X. Jiang, Z. Chen, L. Jiao and E. Hao, Angew. Chem., Int. Ed., 2024, 63, e202319875 CrossRef CAS PubMed.
- X. Yang, Z. Huang, K. Ding, H. Yang, L. Wang, Q. Xia, R. Hu, G. Shi, B. Xu and A. Qin, Sci. China Chem., 2025, 68, 1426–1433 CrossRef CAS.
- Q. Gong, J. Shao, W. Li, X. Guo, S. Ling, Y. Wu, Y. Wei, X. Xu, X. Jiang, L. Jiao and E. Hao, J. Am. Chem. Soc., 2025, 147, 21041–21052 CrossRef CAS PubMed.
- Y. Wang, Y. Deng, Y. Wang, Z. Wang, J. Yang, J. Wang, Y. Wu, L. Hu and H. Wang, J. Mol. Struct., 2024, 1308, 138107 CrossRef CAS.
- W. Li, Q. Gong, Q. Zhao, Q. Wu, C. Niu, L. Jiao and E. Hao, Inorg. Chem., 2025, 64, 9114–9123 CrossRef CAS PubMed.
- D. Wang, G. Yao, J. Sun, J. Liang and H. Zhou, Anal. Chim. Acta, 2025, 1346, 343790 CrossRef CAS PubMed.
- M. Liu, F. Fan, J. Zhang, S. Fang, Y. Bai, Y. Li, Y. Zou, Y. An and X. Dai, Anal. Bioanal. Chem., 2024, 416, 3073–3083 CrossRef CAS PubMed.
- W. Cao, J. Wu, X. Zhao, Z. Li, J. Yu, T. Shao, X. Hou, L. Zhou, C. Wang, G. Wang and J. Han, Carbohydr. Polym., 2024, 325, 121565 CrossRef CAS PubMed.
- W. C. Hartley, F. Schiel, E. Ermini and P. Melchiorre, Angew. Chem., Int. Ed., 2022, 61, e202204735 CrossRef CAS PubMed.
- J. Huang, J. Su, H. Wang, J. Chen, Y. Tian, J. Zhang, T. Feng, L. Di, X. Lu, H. Sheng, Q. Zhu, X. Chen, J. Wang, X. He, Y. Yerkinkazhina, Z. Xie, Y. Shu, T. Kang, H. Tang, J. Qian and W.-G. Zhu, J. Med. Chem., 2024, 67, 17319–17349 CrossRef CAS PubMed.
- W. Wei, Q. Xu, L. Wu, G. Gong, Y. Tian, H. Huang and Z. Li, Eur. J. Med. Chem., 2024, 270, 116333 CrossRef CAS PubMed.
- L. Qin, X. Cheng, S. Wang, G. Gong, H. Su, H. Huang, T. Chen, D. Damdinjav, B. Dorjsuren, Z. Li, Z. Qiu and J. Bian, J. Med. Chem., 2024, 67, 988–1007 CrossRef CAS PubMed.
- W. Ye, C. Fan, K. Fu, X. Wang, J. Lin, S. Nian, C. Liu and W. Zhou, Eur. J. Med. Chem., 2022, 244, 114846 CrossRef CAS PubMed.
- R. del Río-Rodríguez, M. T. Westwood, M. Sicignano, M. Juhl, J. A. Fernández-Salas, J. Alemán and A. D. Smith, Chem. Commun., 2022, 58, 7277–7280 RSC.
- T. Fan, Z.-J. Zhang, Y.-C. Zhang and J. Song, Org. Lett., 2019, 21, 7897–7901 CrossRef CAS PubMed.
- N. Li, X. Ye, Y. Liu and J. Song, Nat. Catal., 2025, 8, 957–967 CrossRef CAS.
- J. Song, Z.-J. Zhang and L.-Z. Gong, Angew. Chem., Int. Ed., 2017, 56, 5212–5216 CrossRef CAS PubMed.
- J. Song, Z.-J. Zhang, S.-S. Chen, T. Fan and L.-Z. Gong, J. Am. Chem. Soc., 2018, 140, 3177–3180 CrossRef CAS PubMed.
- P. Zhu, Y. Yang, J. Qian, J. Han, D. Kong, B. Sun, J. Zhang, J. Wei, Q. Guo, S. Nian, L. Zhou, G. Wang and S. Hou, J. Mol. Struct., 2025, 1321, 140125 CrossRef CAS.
- Q. Zhang, M. Zhu and W. Zi, Chem, 2022, 8, 2784–2796 CAS.
- H.-C. Lin, G. J. Knox, C. M. Pearson, C. Yang, V. Carta and T. N. Snaddon, Angew. Chem., Int. Ed., 2022, 61, e202201753 CrossRef CAS PubMed.
- S. Feng, Y. Zhang, Y. Wang, Y. Gao and Y. Song, Chem. – Eur J., 2024, 30, e202402485 CrossRef CAS PubMed.
- L.-J. He, Y.-B. Zhu, Q.-Z. Fan, D.-D. Miao, S.-P. Zhang, X.-P. Liu and C. Zhang, Bioorg. Med. Chem. Lett., 2019, 29, 549–555 CrossRef CAS PubMed.
- D. Liu, B. Yu, X. Guan, B. Song, H. Pan, R. Wang, X. Feng, L. Pan, H. Huang, Z. Wang, H. Wu, Z. Qiu, Z. Li and J. Bian, Chem. Sci., 2023, 14, 4174–4182 RSC.
- D. Zhou, Z. Zhang, L. Pan, Y. Wang, J. Yang, Y. Gao and Y. Song, Angew. Chem., Int. Ed., 2024, 63, e202404493 CrossRef CAS PubMed.
- W. Rush Scaggs, T. D. Scaggs and T. N. Snaddon, Org. Biomol. Chem., 2019, 17, 1787–1790 RSC.
- L.-L. Li, D. Ding, J. Song, Z.-Y. Han and L.-Z. Gong, Angew. Chem., Int. Ed., 2019, 58, 7647–7651 CrossRef CAS PubMed.
- M. Sayed, Z. Shi, Z.-Y. Han and L.-Z. Gong, Org. Biomol. Chem., 2023, 21, 7305–7310 RSC.
- W.-W. Ding, Y. Zhou, Z.-Y. Han and L.-Z. Gong, J. Org. Chem., 2023, 88, 5187–5193 CrossRef CAS PubMed.
- M. Sayed, Z. Shi, T. Fan, H.-C. Shen and Z.-Y. Han, Synthesis, 2024, 56, 1381–1392 CrossRef CAS.
- S. S. M. Spoehrle, T. H. West, J. E. Taylor, A. M. Z. Slawin and A. D. Smith, J. Am. Chem. Soc., 2017, 139, 11895–11902 CrossRef CAS PubMed.
- T. O. Ronson, J. R. Carney, A. C. Whitwood, R. J. K. Taylor and I. J. S. Fairlamb, Chem. Commun., 2015, 51, 3466–3469 RSC.
- X. Jiang, J. J. Beiger and J. F. Hartwig, J. Am. Chem. Soc., 2017, 139, 87–90 CrossRef CAS PubMed.
- C. M. Pearson, J. W. B. Fyfe and T. N. Snaddon, Angew. Chem., Int. Ed., 2019, 58, 10521–10527 CrossRef CAS PubMed.
- J. Zhou, L.-Z. Yu, Y.-L. Fan, C.-H. Guo, X.-M. Lv, Z.-Y. Zhou, H.-D. Huang, D.-D. Miao, S.-P. Zhang, X.-Y. Li, P.-P. Zhao, X.-P. Liu, W.-H. Hu and C. Zhang, Eur. J. Med. Chem., 2023, 245, 114860 CrossRef CAS PubMed.
- C. Wang, R. Wang, Y. Chen, L. Wang, S. Zhou and H. Wang, Bioorg. Med. Chem. Lett., 2019, 29, 1282–1290 CrossRef CAS PubMed.
- Y. Zeng, F. Liu, J. Wang, B. Shao, T. He, Z. Xiang, Y. Wang, S. Zhu, T. Yang, S. Yu, C. Gong and L. Liu, Chin. Chem. Lett., 2025, 36, 109734 CrossRef CAS.
- Q. Wang, T. Fan and J. Song, Org. Lett., 2023, 25, 1246–1251 CrossRef CAS PubMed.
- T. Fan, Z. Shi, Q.-W. Gong, J. Song and L.-Z. Gong, Org. Lett., 2024, 26, 1421–1425 CrossRef CAS PubMed.
- Z. Lin, Y. Yu, R. Liu and W. Zi, Angew. Chem., Int. Ed., 2024, 63, e202401181 CrossRef CAS PubMed.
- F. Zhao, C. Shu, C. M. Young, C. Carpenter-Warren, A. M. Z. Slawin and A. D. Smith, Angew. Chem., Int. Ed., 2021, 60, 11892–11900 CrossRef CAS PubMed.
- Y.-C. Zhang, R.-L. Geng, J. Song and L.-Z. Gong, Org. Lett., 2020, 22, 2261–2265 CrossRef CAS PubMed.
- J. Zhao, Z. Guo, D. Feng, J. Guo, J. Wang and Y. Zhang, Microchim. Acta, 2015, 182, 2435–2442 CrossRef CAS.
- M. Yang, P. Li, W. Xu, Y. Wei, L. Li, Y. Huang, Y. Sun, X. Chen, J. Liu and X. Huang, Sens. Actuators, B, 2018, 255, 226–234 CrossRef CAS.
- J. Yang, Y. Wang, Z. Wang, J. Wang, C. Zhang, X. Gu, L. Hu and H. Wang, Anal. Chim. Acta, 2024, 1312, 342747 CrossRef CAS PubMed.
- C. Ji, H. Wu, A. Long, L. Xiao, S. Feng and S. Xu, Microchim. Acta, 2024, 191, 537 CrossRef CAS PubMed.
- L. Hu, B. Xu, H. Chen and H. Wang, Sens. Actuators, B, 2021, 340, 129950 CrossRef CAS.
- Y. Wang, S. Feng, X. Wang, C. Tao, Y. Liu, Y. Wang, Y. Gao, J. Zhao and Y. Song, Lab Chip, 2024, 24, 3521–3527 RSC.
- S. Chen, W. He, J. Li, D. Xu, R. Zhao, L. Zhu, H. Wu and F. Xu, Langmuir, 2024, 40, 5799–5808 CrossRef CAS PubMed.
- Q. Zhu, J. Song, Z. Liu, K. Wu, X. Li, Z. Chen and H. Pang, J. Colloid Interface Sci., 2022, 623, 992–1001 CrossRef CAS.
- D. Wang, R. Zhang, Q. Zhang, H. Zhou and J. Sun, Polymer, 2023, 290, 126592 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |