 Open Access Article
Open Access ArticleUnveiling the degradation of lamotrigine in sulfate radical-mediated oxidation: kinetics, influencing factors and transformation mechanisms
Jiayue Donga,
Jing Wua,
Jialing Zhanga,
Zhuoming Denga,
Guoqiang Liu *ab and
Yonghua Liu*a
*ab and
Yonghua Liu*a
aNanjing Institute of Environmental Science, Ministry of Ecology and Environment of China, Nanjing 210042, China. E-mail: gqliushane@foxmail.com; liuyh@nies.org; Tel: +86-025-85287130
bState Key Laboratory of Pollution Control and Resource Reuse, School of the Environment, Nanjing University, Nanjing, Jiangsu Province 210023, PR China
First published on 27th March 2026
Abstract
Lamotrigine (LMG), a widely used anticonvulsant drug, has been frequently detected in aquatic environments due to its limited removal in conventional wastewater treatment. Here, we systematically investigated the degradation of LMG via heat-activated peroxydisulfate (heat/PDS) treatment. The degradation followed pseudo-first-order kinetics, with the observed rate constant (kobs) increasing at higher PDS doses or elevated temperatures. The apparent activation energy was determined to be 88.7 kJ mol−1. Radical quenching experiments using methanol or tert-butanol indicated that SO4˙− predominated in LMG degradation. The presence of halides (Br− and Cl−), natural organic matter (NOM), ammonium (NH4+), or bicarbonate (HCO3−) generally inhibited LMG degradation, primarily due to their competition for SO4˙−. Detailed transformation pathways were proposed based on quantum chemical calculations and high-resolution mass spectrometry, revealing initial attacks on electron-rich sites of the triazine ring and amino groups, leading to hydroxylated and deaminated products. Both ECOSAR predictions and luminescence bacteria assays showed reduced toxicity for most transformation products relative to LMG, suggesting that degradation of LMG by sulfate radical was a detoxification process. This study demonstrates that heat-activated PDS is an effective strategy for eliminating LMG and attenuating its ecological risk in water.
1 Introduction
Lamotrigine (LMG), 6-(2,3-dichlorophenyl)-1,2,4-triazine-3,5-diamine, is a widely prescribed anticonvulsant drug used to treat epilepsy and bipolar disorders.1 In humans, approximately 70% of the administered dose of LMG is recovered from urine with 90% as the metabolite lamotrigine-N2-glucuronide.2,3 After excretion, these metabolites can enter the environment and regenerate the parent LMG in favorable conditions. Over the past decade, LMG has been detected in several water environments. The molecular structure of LMG comprises a triazine ring moiety with two amine groups and a benzene ring moiety substituted with two chlorines, contributing to its low volatility and limited lipophilicity, which promote its persistence in aqueous environments.Ferrer et al. first reported the frequent occurrence of LMG in groundwater and surface water, with its concentration ranging from 17 to 488 ng L−1.4 In wastewater, its concentration can be up to 0.013–1.254 µg L−1.5–8 Moreover, LMG has been shown to accumulate in soils irrigated with treated wastewater, with concentrations up to 4 mg kg−1, raising concerns about potentially leaching into groundwater.9 Although studies on the ecotoxicity of LMG remain limited, emerging evidence suggests it may induce oxidative stress and neurotoxicity in aquatic organisms such as fish and has been associated with potential drug–drug interactions in humans.8,10,11 A recent study demonstrated LMG might translocate in plants, reaching leaves, and affect the expression of stress genes.12 Yang et al. found that the occurrence of LMG in surface water may pose a high risk to the environment (LD50,oral = 205 mg kg−1), potentially inducing oxidative stress and neurotoxicity in fish.13 Therefore, effective removal of LMG from water environments is imperative to mitigate ecological and public health risks.
Conventional wastewater treatment approaches (WWTPs) showed limited efficacy in LMG degradation.14,15 For instance, Bollmann et al. investigated the transformation and removal of LMG in municipal WWTPs.14 They found that LMG concentration increased from 1.1 µg L−1 in the influent to 1.6 µg L−1 in the effluent, implying the poor elimination of LMG. When LMG was subjected to simulated sunlight at pH 7.0, the degradation half-life of over 100 h, implying its high resistance to direct photodegradation as well.16–18 Karpov et al. (2021) demonstrated that redox-active minerals like bimessite (δ-MnO2) could degrade LMG via oxidation, dechlorination, and addition reactions.19 More importantly, phenolic compounds, like vanillic acid, can enhance LMG degradation while leading to the formation of numerous coupling products. These products are more stable than the parent LMG and its long-term ecological impacts remain largely unknown. These findings underscore the need for advanced treatment technologies.
Sulfate radical (SO4˙−)-based advanced oxidation processes (SR-AOPs) have emerged as a promising alternative for degrading persistent organic contaminants.20–23 SO4˙−, along with other reactive oxidants (i.e. ˙OH or O2˙−), can be generated from the activation of peroxymonosulfate (PMS) or peroxydisulfate (PDS). Several methods have been applied for the activation of persulfates, including the use of UV light, heat, ultrasound, transition metal and others.24–26 Among these, thermal activation is particularly attractive for its high efficiency, scalability, and practicality for real-world applications. To date, heat activated persulfates have been successfully implemented in wastewater treatment systems, such as hospital effluent. SO4˙− possesses a high oxidation capacity (2.5–3.1 V vs. NHE) and a long lifetime (t1/2 = 30–40 µs), enabling effective degradation of recalcitrant compounds.27–29 SR-AOPs has been successfully applied in the degradation of triazine compounds, including atrazine, simazine and propazine.30–33 Ji et al. reported that SO4˙− tends to attack alkylamino side chains of triazine ring in atrazine molecule, leading to hydroxylated products which subsequently undergo decomposition to form ketone or aldehyde.31 Given the structural similarity of LMG, which also contains a triazine core, it is plausible that SO4˙− could effectively degrade LMG, yet the underlying mechanisms and transformation pathways remain largely unexplored.
Inspired by all above, we attempt to elucidate the degradation of LMG in a heat/PDS oxidation system. Firstly, the effects of operating condition and water matrix (including halides, natural organic matter (NOM), nitrogenous ions, and bicarbonate ion (HCO3−)) on kinetics and efficiency of LMG degradation were explored. Then, solid phase extraction-liquid chromatography-mass spectroscopy (SPE-LC-MS), as a powerful tool, was applied to identify transformation products and speculate detailed degradation mechanism and pathway. Finally, the ecotoxicological impacts of LMG and its transformation products in heat/PDS system were assessed using ECOSAR and luminescent bacteria assays. Above all, this study aims to provide theoretical underpinning and technical guidance for remediation of the LMG-contaminated water using SO4˙−-based AOPs.
2 Experimental section
2.1 Materials and reagents
All chemicals and reagents used in this work were of the purest quality available. Lamotrigine (LMG), sodium peroxydisulfate (Na2S2O8), sodium chloride (NaCl), sodium bromide (NaBr), sodium carbonate (Na2CO3), sodium bicarbonate (NaHCO3), sodium nitrate (NaNO3), ammonium sulfate ((NH4)2SO4), potassium dihydrogen phosphate (KH2PO3) and sodium hydroxide (NaOH) were purchased from Aladdin (Shanghai, China). Suwannee River NOM was supplied by the International Humic Substances Society (St. Paul, MN, USA). Methanol (MEOH) and tert-butyl alcohol (TBA) were of high-performance liquid chromatography (HPLC) degrade and purchased from Thermo Fisher (MA, USA). Ultra-pure water (18.2 MΩ cm−1) was used in all prepared solution.2.2 Experiments setup
The degradation of LMG in heat/PDS was performed in the thermostatic water bath for 180 min. Appropriate volume of LMG stock solution was transferred into a series of cylindrical glass vials and specific aliquots of PDS stock solution were added to achieve a total 50 mL reaction solution with predetermined concentration. The PDS concentration varied from 0 to 2 mM and the temperature was set from 40 °C to 80 °C. The pH was maintained at 7.0 using 10 mM phosphate buffer. Parallel experiments with additional 10 mM MEOH or TBA were conducted identically to investigate the role the oxidant radicals. Effects of halides (Cl− and Br−), SRNOM, nitrogenous ions (NH4+ and NO3), and HCO3− were also investigated. At preset intervals, reaction aliquots (1 mL) were collected and immediately transferred to the vial in ice bath to stop the reaction, then stored at 4 °C before HPLC analysis.To identify the transformation products of LMG, 200 mL reaction solution containing 10 µM LMG, 1 mM PDS, 10 mM phosphate buffer was allowed to reaction for 180 min in a 60 °C water bath. At pre-determined intervals, the samples were withdrawn and immediately transferred to ice bath to quench the reaction. After that all the solutions were enriched into 1 mL MEOH with C18 solid phase extraction (SPE) and stored in a 4 °C refrigerator before HRMS analysis. Detailed procedure of SPE was displayed in Text S1. All samples were stored in a 4 °C refrigerator at most 24 h before analysis.
2.3 Analytical methods
Concentration of LMG was determined by a Shimadzu 20AD HPLC (Shimadzu, Japan) equipped with a photo-diode array detector (SPD-M20A). Separation was performed on Agilent Zorbax Eclipse Plus C18 reverse phase column (4.6 × 250 mm, 5 µm). The injection volume was 30 µL, and the flow rate was 1.0 mL min−1. The mobile phase was composed of 70% MEOH and 30% water. The retention time and detection wavelength of LMG were 5.42 min and 316 nm, respectively.LMG and its degradation products were identified by a high-resolution mass spectrometer (HRMS) comprised of a HPLC equipped with a Q-Exactive mass spectrometer (Thermo Fisher Scientific, USA). Elution was performed at a flow rate of 0.2 mL min−1 with water and MeOH, employing a linear gradient as follows: 95% A, 0–3 min; 95% to 5% A, 3–7 min; 5% A, 7–12 min; 5% to 95% A, 12–12.1 min; 95% A, 12.1–15 min. The sample injection volume was 2 µL. Mass analyses were conducted in negative mode using electrospray ionization (ESI) source. The instrument parameters used for sample analysis were as follows: ion transfer tube temperature of 350 °C; source temperature of 300 °C; sheath gas pressure of 35 arb, auxiliary gas pressure of 10 arb, spray voltage of 3500 V. Mass spectrometric data was collected at full scan mode over a mass range of m/z 60–900 to identify the degradation products.
2.4 Density functional theory calculations
Density functional theory (DFT) calculations were performed via Gaussian 16 quantum chemistry computation package. The optimization of the molecular geometry of LMG was performed at M062x/6-31g+(d) level of theory. Solvation model based on density (SMD) was used to investigate the solvent effect of water. All the computed frequencies of the optimized structure were positive, which means the energy stability of the structure. The highest occupied molecular orbital (HOMO) of LMG was obtained from Gaussview 6 software dependent on the checkpoint file from Gaussian 16. Moreover, the electrophilic Fukui function was calculated to predict reactive sites of electrophilic attack.34,35 This function was defined as follows:
 | (1) |
| Electrophilic attack: fk− = qkN − qkN−1 | (2) |
2.5 Toxicity assay
The ecotoxicity of LMG and its products to aquatic organisms were predicted using both Ecological Structure–Activity Relationship (ECOSAR, version 2.2). ECOSAR program was developed by the US EPA, OECD and EU, which has been successfully used in the estimation of chemical toxicity including the LC50 to fish (96 h) and daphnia (48 h) and chronic toxicity values (Chv) for fish and daphnia.37Additionally, the luminescent bacteria test was conducted to evaluate the acute toxicity of products during the degradation of LMG by heat/PDS treatment. Based on the previous work,38 the bacteria PPT3 was applied as the source of luminescence. The bacteria PPT3 was activated by 2% NaCl solution, diluted by 3% NaCl solution, and then exposed to sample solutions for 15 min. Then Ensight Multimode Microplate Reader (PerkinElmer, MA, USA) was adopted to record the luminescence intensity, and the higher luminescence intensity represents the lower toxicity. Detailed procedures were displayed in Text S2.
3 Results and discussion
3.1 Degradation of LMG by heat/PDS oxidation
As illustrated in Fig. 1, no removal of LMG was observed in control experiments with heating or PDS alone over 180 min, revealing that direct PDS oxidation or volatilization did not contribute to LMG degradation. In contrast, LMG was effectively removed by heat/PDS oxidation (Fig. 1a). At the PDS concentration of 0.2 mM, approximately 44.2% of LMG was decomposed at 180 min. Increasing PDS concentration to 1 mM resulted in nearly complete removal over the same period, while further increasing to 0.2 mM shorten the time for complete degradation to 90 min. The degradation of LMG can be described by a pseudo-first-order kinetic model (Fig. S1), with the pseudo-first-order rate constant determined by the linear regression of ln([LMG]0/[LMG]t) against t at condition (eqn (3)):
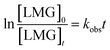 | (3) |
 | ||
| Fig. 1 Effect of PDS and temperature on the degradation of LMG by heat/PDS oxidation. Experimental conditions: LMG 10 µM, PDS 0.0–2.0 mM, pH 7.0, temperature 40–80 °C. | ||
Temperature also played a vital role in LMG degradation in the heat/PDS oxidation (Fig. 1c and S2). Experiments conducted between 40 °C and 80 °C demonstrated a strong positive correlation between temperature and removal efficiency. The kobs increased by a factor of 28.7 when the temperature was raised from 40 to 70 °C, consistent with previous findings on the degradation of atrazine in heat/PDS system.31 When the temperature further increased to 80 °C, the increase in kobs value became insignificant. This result suggested that practical application of heat/PDS for LMG contaminated water should balance efficiency with economic feasibility.
The temperature dependency of kobs can be further evaluated by Arrhenius equation (eqn (4)):
 | (4) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) kobs is decreased linearly with increasing T−1, revealing their relationship fitted well with Arrhenius equation.39 Thus, the apparent activation energy (Ea) can be measured to be 88.7 kJ mol−1, comparable to reported values, i.e. 108 kJ mol−1 for trichloroethene (TCE) and 64.8 kJ mol−1 for ciprofloxacin.31 It is noteworthy that although higher temperature facilitated the degradation of LMG in heat/PDS oxidation, evaluated scavenging reactions and faster depletion of PDS may occur simultaneously, which may increase energy consumption. Thus, identifying an optimal temperature is crucial for successful remediation. Considering economic benefits and degradation efficiency simultaneously, 60 °C was selected as the reaction temperature for subsequent experiments in this work.
kobs is decreased linearly with increasing T−1, revealing their relationship fitted well with Arrhenius equation.39 Thus, the apparent activation energy (Ea) can be measured to be 88.7 kJ mol−1, comparable to reported values, i.e. 108 kJ mol−1 for trichloroethene (TCE) and 64.8 kJ mol−1 for ciprofloxacin.31 It is noteworthy that although higher temperature facilitated the degradation of LMG in heat/PDS oxidation, evaluated scavenging reactions and faster depletion of PDS may occur simultaneously, which may increase energy consumption. Thus, identifying an optimal temperature is crucial for successful remediation. Considering economic benefits and degradation efficiency simultaneously, 60 °C was selected as the reaction temperature for subsequent experiments in this work.
3.2 Reactive radical identification
Several research reported that various reactive species can be generated in heat/PDS system. SO4˙− can be generated via activating PDS, which can react with H2O or OH− to generate OH˙. Both OH˙ and SO4˙− could potentially attack LMG and lead to degradation. In the condition of 1 mM PDS at 60 °C, nearly 100% LMG was decomposed with the kobs of 0.0255 min−1. To distinguish the roles of OH˙ and SO4˙−, MEOH and TBA were chosen as quenching agents. Generally, MEOH was known as a radical quencher for both OH˙ and SO4˙− (kMEOH,OH˙ = 9.7 × 108 M−1 s−1, kMEOH,SO4˙− = 1.1 × 107 M−1 s−1), while TBA was a selective quencher for OH˙ (kTBA,OH˙ = 3.8 × 108 M−1 s−1, kTBA,SO4˙− = 4 × 105 M−1 s−1).40 And the scavenging ratio of OH˙ and SO4˙− by MEOH or TBA can be calculated as follows:
 | (5) |
| Scavenging ratio | ||
|---|---|---|
| ˙OH | SO4˙− | |
| MEOH | 97.9% | 59.5% |
| TBA | 94.8% | 5.1% |
Consequently, introducing 10 mM MEOH into the reaction solution, 97.9% of OH˙ and 59.5% of SO4˙− was scavenged (Table 1). Under this condition, 44.4% LMG was degraded at 180 min with the kobs of 0.0034 min−1 (Fig. 2 and S3). Substitution MEOH with TBA resulted in about 94.8% of OH˙ and only 5.1% of SO4˙− being quenched, and the kobs of LMG was 0.266 min−1 which extremely close to the kobs obtained in heat/PDS system (Fig. 2 and S3). Above all, these results strongly signify the leading role of SO4˙− in LMG degradation in heat/PDS system.
 | ||
| Fig. 2 Effect of MEOH or TBA on the degradation of LMG by heat/PDS oxidation. Experimental conditions: LMG 10 µM, PDS 1.0 mM, MEOH or TBA 10 mM, pH 7.0, temperature 60 °C. | ||
3.3 Effects of nontarget natural water constituents
As can be seen in Fig. 3a, the kobs value of LMG decreased from 0.0255 min−1 to 0.0101 min−1 as Br− concentration increased from 0.00 to 0.10 mM. This inhibition is due to the competition of Br− with LMG for SO4˙−. It has been reported that Br− can scavenge SO4˙− to form Br˙ at a secondary rate constant of 3.9 × 109 M−1 s−1,45 then Br˙ reacts with Br− to form Br2˙−. Besides, the reaction of Br˙ with H2O/OH− and Br− with HO˙ can generate BrOH˙− which further reacting with Br− to form Br˙−. Finally, Br˙− is readily transformed to free bromine. Both bromide radical and free bromine can be classified as reactive bromine species (RBS). Although it has been reported that RBS can serve as secondary oxidants, their oxidation potentials (1.6–2.2 V vs. NHE) are lower than that of SO4˙−, making them less effective in oxidizing LMG. Thus, the formation of RBS does not compensate for the loss of SO4˙−, leading to net inhibition.
The effect of Cl− on LMG degradation was also explored. The kobs value was 0.0226 min−1 in the presence of 0.1 mM Cl−, similiar to the control experiment (Fig. 3b). The bimolecular rate constant of Cl− reacting with SO4˙− is 2.7 × 108 M−1 s−1,45 close to that of LMG (7.5 × 108 M−1 s−1), indicating limited competition at low Cl− level. However, in the presence of higher concentration of Cl− (10 mM), the kobs value dropped to 0.0106 min−1, suggesting the effective quenching of SO4˙− at higher Cl− concentration
| X− + SO4˙− ⇌ X˙ + SO42− | (R1) |
| X˙ + X− ⇌ X2˙− | (R2) |
| X2˙− + Br˙ ⇌ X2 + X− | (R3) |
| X˙ + X˙ ⇌ X2 | (R4) |
| X2 + H2O ⇌ HXO + H+ + X− (X = Cl− or Br−) | (R5) |
 at a bimolecular reaction rate constant of 3.5 × 105 M−1 s−1.45 Although the value is three orders of magnitude lower than that of LMG reacting with SO4˙− (7.5 × 108 M−1 s−1), the NH4+ concentration (mM level) in the solution was hundreds of times higher than LMG concentration (10 µM). This enabled NH4+ to compete for SO4˙− and thus suppressed the degradation of LMG. As to NO3−, its bimolecular reaction rate constant with SO4˙− was only 2.1 M−1 s−1,45 implying that NO3− hardly participated in the reaction. As can be seen in Fig. 3e, the effect of NO3− on LMG degradation in heat/PDS oxidation process can be neglected.
at a bimolecular reaction rate constant of 3.5 × 105 M−1 s−1.45 Although the value is three orders of magnitude lower than that of LMG reacting with SO4˙− (7.5 × 108 M−1 s−1), the NH4+ concentration (mM level) in the solution was hundreds of times higher than LMG concentration (10 µM). This enabled NH4+ to compete for SO4˙− and thus suppressed the degradation of LMG. As to NO3−, its bimolecular reaction rate constant with SO4˙− was only 2.1 M−1 s−1,45 implying that NO3− hardly participated in the reaction. As can be seen in Fig. 3e, the effect of NO3− on LMG degradation in heat/PDS oxidation process can be neglected.
 | (R6) |
 | (R7) |
3.4 Mineralization and products transformation
Synchronous mineralization was also adopted to indicate the performance of heat/PDS treatment. In presence of 2 mM PDS, complete removal of LMG required approximately 90 min, while the TOC removal stood at 20.59%. Extending the reaction time to 180 min enhanced the mineralization ratio to 51.76%. The mineralization extent reached 75.29% after 360 min reaction (Fig. S5). Overall, the LMG can be effectively mineralized by appropriately increasing reaction time, highlighting the adaptive conditions for the application of SR-AOPs.Degradation products and transformation pathways were investigated via theoretical calculation and MS analysis, (Table S1). Based on the radical quenching experiments above, SO4˙− was confirmed as the dominate oxidant for LMG degradation. It is well known that SO4˙−, as an electrophilic oxidant, preferentially attacks electron-rich sites via single-electron transfer. The highest occupied molecular orbital (HOMO) distribution shows the electron-rich sites of the molecular structure.52 In another word, HOMO indicates easy escape of electron which can be easily attacked by SO4˙−. As can be seen Fig. 4b, the HOMO mainly localized on the triazine ring and amino functional groups, revealing these sites highly prone to electrophilic attacks. This electron distribution results from the activation of triazine ring through electron-donating amino substituents, while benzene ring is deactivated by electron-withdrawing chlorine atoms.
To further evaluate the vulnerable sites of LMG, the Fukui index (f−1) was employed. A higher f−1 value means a higher reactivity.53,54 Fig. 4c shows the N16 atom of the amino group is the most reactive site on LMG with f−1 of 0.665, suggesting it is the most vulnerable for SO4˙− attack, followed by electron migration to C12, oxygen addition, and superoxide elimination to yield an imine intermediate (Fig. S4). Subsequent hydrolysis produces deaminated and hydroxylated products (Fig. 5, Pathway2, P3a). Fig. 4c shows that the f−1 value of another two sites, N13 and N15, ranked only second to N16 (0.569 and 0.542, respectively), suggesting these sites are also susceptible to SO4˙− oxidation. For N15, two degradation pathways might occur. One possibility is that the single-electron oxidation on N15 formed an N15-centered radical which underwent oxidation and hydrolysis giving rise to another de-amino and hydroxylated products (Fig. 5, Pathway2, P3b). Alternatively, the electron on N15-centered radical might undergo intramolecular transfer to the dichlorophenyl ring, facilitating the addition of N15 to C3 and leading to the formation of a tricyclic heteroaromatic product (Fig. 5c, Pathway1, P1). Similar cyclization products have been reported during the photodegradation of LMG, and such addition reactions via single-electron oxidation are known to occur on aromatic systems.18 In Pathway3, degradation was initiated on N13 by SO4˙− attack, leading to the formation of P5.
P1, P3a, P3b and P5 were identified as primary products. P3a and P3b are geometric isomers, due to the lack of authentic standards, their formation cannot be distinguished and quantified. Thus, the sum of the abundance of these two MS peaks can be applied to exhibit their generation trends. As can be seen in Fig. S6, the sum of P3a and P3b increased to a temporal maximum at 60 min, then gradually decreased. Such results implied that they were the crucial intermediates and can be further oxidized by SO4˙−. P3 (both P3a and P3b) and P4 shared the same core scaffold, differing only by the replacement of an amino group in P3 with a hydroxyl group in P4, suggesting that P3 undergoes deamination and hydroxylation to yield P4. Likewise, P1 and P5 exhibited similar formation trends (Fig. 5). Hydroxylation of P1 and P5 gave rise to P2 and P6 (both P6a and P6b), respectively. P4 and P6 underwent further oxidation contributed to P7. Additionally, P5 may undergo C–C bond cleavage to release 2-hydroxy-3,5-diamino triazine (P8).
3.5 Toxicity evaluation
The ecological safety of heat/PDS treatment was assessed by predicting the acute and chronic toxicity of LMG and its degradation products using the ECOSAR software. All the organic compounds were given in the unit of mg L−1, which can be classified as four toxicity grades, e.g., not harmful (>100.0), harmful (10.0–100.0), toxic (1.0–10.0), and very toxic (<1.0). Table 2 displays the predict acute and chronic toxicity to fish, daphnid, and green algae. The acute LC50 values of the parent LMG to Daphnid and green algae were 0.53 and 0.294 mg L−1, respectively, implying its high toxicity. The chronic toxicity of LMG to fish, daphnid, and green algae was in a very toxic degrade as well. Although P1 and P3 showed lower toxicity than LMG, their predicted acute and chronic toxicity remained between the very toxic and harmful categories (Table 2). In contrast, the polyhydroxylated products (P5–P8) displayed substantially reduced toxicity to fish, daphnid, and green algae, with most values reaching the not harmful grade. This trend indicates that progressive hydroxylation during LMG degradation generates less toxic compounds, and extended reaction times favor the further oxidation of primary intermediates.| Acute toxicity | Chronic toxicity | |||||
|---|---|---|---|---|---|---|
| Fish | Daphnid | Green algae | Fish | Daphnid | Green algae | |
| LMG | 3.64 | 0.53 | 0.294 | 0.115 | 0.053 | 0.113 |
| P1 | 4.3 | 2.91 | 4.43 | 0.516 | 0.459 | 1.71 |
| P2 | 55 | 20.9 | 68.7 | 5.29 | 2.17 | 9.49 |
| P3a | 13 | 1.08 | 2.14 | 0.122 | 0.225 | 0.362 |
| P3b | 5.52 | 0.734 | 1.07 | 0.062 | 0.145 | 0.195 |
| P4 | 6.9 | 44.2 | 3.65 | 3.52 | 16 | 0.526 |
| P5 | 21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
3220 | 1670 | 556 | 535 |
| P6a | 872 | 466 | 271 | 79.4 | 38.4 | 61.9 |
| P6b | 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 910 910![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 080 080![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
141![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
173![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 600 |
14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
| P7 | 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 890 890![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 080 080![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
140![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
172![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
| P8 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 270 270![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
473![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
62![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
6400 |
Furthermore, the toxicity during LMG degradation process via heat/PDS treatment was evaluated by luminescence bacterium test, where lower luminescence intensity corresponds to higher toxicity. Initially, LMG was the sole compound in the reaction solution, and the luminescence intensity of bacteria PPT3 was 60.98%, indicating its moderate acute toxicity. After 60 min reaction, the luminescence intensity slightly increased to 64.75%, with the toxic intermediates (P1 and P3) reaching their maximum abundances (Fig. 6). Based on ECOSAR prediction, their predicted toxicity remains relatively high, which explains the limited change in luminescence at this time point. As the reaction proceeded to 180 min, the luminescence intensity rose to 103.69%, signifying the disappearance of acute toxicity. This detoxification coincided with the substantial decline of primary products, and the subsequent products (i.e. P6–P8) classified as not harmful via ECOSAR software. The temporal correlation between the depletion of toxic intermediates strongly suggests that the detoxification is driven by the transformation of LMG and primary products into polyhydroxylated derivatives.
 | ||
| Fig. 6 The luminescence intensity of PPT3 in LMG sample following heat/PDS oxidation. Experimental conditions: LMG 10 µM, PDS 1.0 mM, pH 7.0, temperature 60 °C. | ||
Overall, the experimental data align well with ECOSAR predictions and demonstrated that prolonged treatment effectively converts toxic intermediates into innocuous products. It should be noted that toxicological performance can be affected by multiple combined factors and small changes in treatment. Additionally, the particularities of degradation products physicochemical characteristics and living organisms environmental interaction also led to different toxicological responses. Thus, it is suggested that further tests should be conducted to ensure the ecological safety of effluent disposal.
4 Conclusion
This study found that LMG can be effectively degraded by heat/PDS treatment. The degradation process follows pseudo-first-order kinetics, with the rate constants increasing with higher PDS dosage and temperature. SO4˙− were identified as the dominant reactive species responsible for the LMG oxidation. The co-existing halides, NOM, NH4+ or HCO3− served as scavengers of SO4˙− and suppressed the degradation, while NO3− exert an insignificant influence on LMG degradation. Dependent on the DFT calculations and HRMS analysis, three possible degradation pathways were proposed, including addition, hydroxylation and ring cleavage. Both ECOSAR results and luminescence bacterium test showed that most of products are less toxic than the parent LMG, thus heat/PDS treatment can be used as an effective technique to detoxify the LMG-contaminated water.Author contributions
Jiayue Dong: investigation, formal analysis, data curation, writing – original draft, conceptualization, funding acquisition. Jing Wu: writing – review & editing. Jialing Zhang: writing – review & editing. Zhuoming Deng: formal analysis, data curation. Guoqiang Liu: writing – review &editing, funding acquisition. Yonghua Liu: writing – review &editing, funding acquisition.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data that support the findings of this study are available on reasonable request from the corresponding author.Supplementary information (SI): SPE procedures, luminescent bacteria test procedures, and MS data of samples. See DOI: https://doi.org/10.1039/d5ra09514j.
Acknowledgements
This research was supported by the Special Fund of Chinese Central Government for Basic Scientific Research Operations in Commonweal (GYZX250207). The contents of the paper do not necessarily represent the views of the funding agency.References
- A. F. Cohen, G. S. Land, D. D. Breimer, W. C. Yuen, C. Winton and A. W. Peck, Lamotrigine, a new anticonvulsant: Pharmacokinetics in normal humans, Clin. Pharmacol. Ther., 1987, 42, 535–541 CrossRef CAS PubMed.
- B. Zonja, A. Delgado, J. L. Abad, S. Pérez and D. Barceló, Abiotic amidine and guanidine hydrolysis of lamotrigine-N2-glucuronide and related compounds in wastewater: The role of pH and N2-substitution on reaction kinetics, Water Res., 2016, 100, 466–475 CrossRef CAS PubMed.
- B. Zonja, S. Pérez and D. Barceló, Human Metabolite Lamotrigine-N2-glucuronide Is the Principal Source of Lamotrigine-Derived Compounds in Wastewater Treatment Plants and Surface Water, Environ. Sci. Technol., 2015, 50, 154–164 CrossRef PubMed.
- I. Ferrer and E. M. Thurman, Identification of a New Antidepressant and its Glucuronide Metabolite in Water Samples Using Liquid Chromatography/Quadrupole Time-of-Flight Mass Spectrometry, Anal. Chem., 2010, 82, 8161–8168 CrossRef CAS PubMed.
- I. Ferrer and E. M. Thurman, Analysis of 100 pharmaceuticals and their degradates in water samples by liquid chromatography/quadrupole time-of-flight mass spectrometry, J. Chromatogr. A, 2012, 1259, 148–157 CrossRef CAS PubMed.
- T. Filep, L. Szabó, A. C. Kondor, G. Jakab and Z. Szalai, Evaluation of the effect of the intrinsic chemical properties of pharmaceutically active compounds (PhACs) on sorption behaviour in soils and goethite, Ecotoxicol. Environ. Saf., 2021, 215, 112120 CrossRef CAS PubMed.
- D. Montenegro-Apraez, C. A. L. Graça, F. Machuca-Martinez and O. S. G. P. Soares, Catalytic ozonation of lamotrigine with modified activated carbon: degradation, mineralization and by-products analysis, J. Water Process Eng., 2025, 77, 108434 CrossRef.
- <LMG concentration 3 + toxicity on human being.pdf>..
- V. Baldasso, S. Sayen, C. A. R. Gomes, L. Frunzo, C. M. R. Almeida and E. Guillon, Metformin and lamotrigine sorption on a digestate amended soil in presence of trace metal contamination, J. Hazard. Mater., 2024, 466, 133635 CrossRef CAS PubMed.
- T. Malchi, Y. Maor, G. Tadmor, M. Shenker and B. Chefetz, Irrigation of Root Vegetables with Treated Wastewater: Evaluating Uptake of Pharmaceuticals and the Associated Human Health Risks, Environ. Sci. Technol., 2014, 48, 9325–9333 CrossRef CAS PubMed.
- A. Paz, G. Tadmor, T. Malchi, J. Blotevogel, T. Borch, T. Polubesova and B. Chefetz, Fate of carbamazepine, its metabolites, and lamotrigine in soils irrigated with reclaimed wastewater: Sorption, leaching and plant uptake, Chemosphere, 2016, 160, 22–29 CrossRef CAS PubMed.
- Y. Bigott, S. P. Chowdhury, S. Pérez, N. Montemurro, R. Manasfi and P. Schröder, Effect of the pharmaceuticals diclofenac and lamotrigine on stress responses and stress gene expression in lettuce (Lactuca sativa) at environmentally relevant concentrations, J. Hazard. Mater., 2021, 403, 123881 CrossRef CAS PubMed.
- H. Yang, X. Gu, H. Chen, Q. Zeng, Z. Mao and Y. Ge, Omics techniques reveal the toxicity mechanisms of three antiepileptic drugs to juvenile zebrafish (Danio rerio) brain and liver, Aquat. Toxicol., 2023, 262, 106668 CrossRef CAS PubMed.
- A. F. Bollmann, W. Seitz, C. Prasse, T. Lucke, W. Schulz and T. Ternes, Occurrence and fate of amisulpride, sulpiride, and lamotrigine in municipal wastewater treatment plants with biological treatment and ozonation, J. Hazard. Mater., 2016, 320, 204–215 CrossRef CAS PubMed.
- M. Engel and B. Chefetz, Removal of triazine-based pollutants from water by carbon nanotubes: Impact of dissolved organic matter (DOM) and solution chemistry, Water Res., 2016, 106, 146–154 CrossRef CAS PubMed.
- O. S. Keen, I. Ferrer, E. Michael Thurman and K. G. Linden, Degradation pathways of lamotrigine under advanced treatment by direct UV photolysis, hydroxyl radicals, and ozone, Chemosphere, 2014, 117, 316–323 CrossRef CAS PubMed.
- N. Klanovicz, J. M. S. de Jesus, F. M. Costa, G. C. de Assis, B. Ramos, A. F. Camargo, W. Michelon, A. Dallegrave, R. W. Becker, H. Treichel and A. C. S. C. Teixeira, A novel hybrid continuous-flow wastewater treatment for lamotrigine degradation by combining enzymatic and photo-oxidative reactions, J. Water Process Eng., 2023, 56, 104395 CrossRef.
- R. B. Young, B. Chefetz, A. Liu, Y. Desyaterik and T. Borch, Direct photodegradation of lamotrigine (an antiepileptic) in simulated sunlight – pH influenced rates and products, Environ. Sci.: Processes Impacts, 2014, 16, 848–857 RSC.
- M. Karpov, B. Seiwert, V. Mordehay, T. Reemtsma, T. Polubesova and B. Chefetz, Abiotic Transformation of Lamotrigine by Redox-Active Mineral and Phenolic Compounds, Environ. Sci. Technol., 2021, 55, 1535–1544 CrossRef CAS PubMed.
- L. Liu, H. Yan, C. Yang and G. Zhu, Dewatering of drilling sludge by ultrasound assisted Fe(ii)-activated persulfate oxidation, RSC Adv., 2018, 8, 29756–29766 RSC.
- P. V. Nidheesh, G. Divyapriya, F. Ezzahra Titchou and M. Hamdani, Treatment of textile wastewater by sulfate radical based advanced oxidation processes, Sep. Purif. Technol., 2022, 293, 121115 CrossRef CAS.
- Z. Zhang, X. Yue, Y. Duan and Z. Rao, A study on the mechanism of oxidized quinoline removal from acid solutions based on persulfate–iron systems, RSC Adv., 2020, 10, 12504–12510 RSC.
- X. Zhou, Z. Guo, X. Tang, W. Wang, M. Wu, B. Song, Y. Xiang, Y. Li, W. Xiong, D. Huang and C. Zhou, Sulfate radical-based advanced oxidation processes for simultaneous removal of antibiotic-resistant bacteria and antibiotic resistance genes and the affecting factors, Chem. Eng. J., 2024, 498, 155149 CrossRef CAS.
- Y. Gao, Y. Zhu, T. Li, Z. Chen, Q. Jiang, Z. Zhao, X. Liang and C. Hu, Unraveling the High-Activity Origin of Single-Atom Iron Catalysts for Organic Pollutant Oxidation via Peroxymonosulfate Activation, Environ. Sci. Technol., 2021, 55, 8318–8328 CrossRef CAS PubMed.
- T. Zeng, X. Tang, Z. Huang, H. Chen, S. Jin, F. Dong, J. He, S. Song and H. Zhang, Atomically Dispersed Fe–N4 Site as a Conductive Bridge Enables Efficient and Stable Activation of Peroxymonosulfate: Active Site Renewal, Anti-Oxidative Capacity, and Pathway Alternation Mechanism, Environ. Sci. Technol., 2023, 57, 20929–20940 CrossRef CAS PubMed.
- R. Zhang, P. Sun, T. H. Boyer, L. Zhao and C.-H. Huang, Degradation of Pharmaceuticals and Metabolite in Synthetic Human Urine by UV, UV/H-2O2, and UV/PDS, Environ. Sci. Technol., 2015, 49, 3056–3066 CrossRef CAS PubMed.
- S. Padmaja, Z. B. Alfassi, P. Neta and R. E. Huie, Rate constants for reactions of SO4˙− radicals in acetonitrile, Int. J. Chem. Kinet., 2004, 25, 193–198 CrossRef.
- U. Ushani, X. Lu, J. Wang, Z. Zhang, J. Dai, Y. Tan, S. Wang, W. Li, C. Niu, T. Cai, N. Wang and G. Zhen, Sulfate radicals-based advanced oxidation technology in various environmental remediation: A state-of-the–art review, Chem. Eng. J., 2020, 402, 126232 CrossRef CAS.
- C. Wang, J. Yan, J. Yuan, L. Zhang, W. Qian, G. Bian, Y. Song, X. Gao and H. Mao, Degradation of bisphenol A during heat-activated peroxodisulfate treatment: kinetics, mechanism, and transformation products, RSC Adv., 2025, 15, 28949–28958 RSC.
- X. Chen, C. Hu, F. Hong, Y. Fang, X. Yuan, H. Tian and Y. Huang, Activation of persulfate with hydrodynamic cavitation in the removal of atrazine: Regulating the concentration of ˙OH and SO4˙− and the degradation mechanism, J. Water Process Eng., 2024, 65, 105828 CrossRef.
- Y. Ji, Y. Shi, W. Dong, X. Wen, M. Jiang and J. Lu, Thermo-activated persulfate oxidation system for tetracycline antibiotics degradation in aqueous solution, Chem. Eng. J., 2016, 298, 225–233 CrossRef CAS.
- B. Liu, Y. Wang, X. Hao, X. Liang and Q. Yang, Activation of PMS degradation of chlorotriazine herbicides (atrazine and simazine) by MIL101-derived Fe/Co@C under high salinity conditions, Chem. Eng. J., 2023, 475, 146129 CrossRef CAS.
- W. Ren, X. Huang, L. Wang, X. Liu, Z. Zhou, Y. Wang, C. Lin, M. He and W. Ouyang, Degradation of simazine by heat-activated peroxydisulfate process: A coherent study on kinetics, radicals and models, Chem. Eng. J., 2021, 426, 131876 CrossRef CAS.
- W. Langenaeker, K. Demel and P. Geerlings, Quantum-chemical study of the Fukui function as a reactivity index: Part 3. Nucleophilic addition to α,β-unsaturated compounds, J. Mol. Struct.: THEOCHEM, 1992, 259, 317–330 CrossRef.
- K. Fukui, T. Yonezawa and H. Shingu, A molecular orbital theory of reactivity in aromatic hydrocarbons, J. Chem. Phys., 1952, 20, 722–725 CrossRef CAS.
- A. M. Elhorri, K. D. Belaid, M. Zouaoui-Rabah and R. Chadli, Theoretical study of the azo dyes dissociation by advanced oxidation using Fukui indices. DFT calculations, Comput. Theor. Chem., 2018, 1130, 98–106 CrossRef CAS.
- C. Chen, Y. Lu, J. Liang, L. Wang and J. Fang, Roles of nitrogen dioxide radical (˙NO2) in the transformation of aniline by sulfate radical and hydroxyl radical systems with the presence of nitrite, Chem. Eng. J., 2023, 451, 138755 CrossRef CAS.
- J. Dong, P. Yang, D. Kong, Y. Song and J. Lu, Formation of nitrated naphthalene in the sulfate radical oxidation process in the presence of nitrite, Water Res., 2024, 255, 121546 CrossRef CAS PubMed.
- A. Tsitonaki, B. Petri, M. Crimi, H. MosbÆK, R. L. Siegrist and P. L. Bjerg, In Situ Chemical Oxidation of Contaminated Soil and Groundwater Using Persulfate: A Review, Crit. Rev. Environ. Sci. Technol., 2010, 40, 55–91 CrossRef CAS.
- L. Gao, Y. Guo, J. Zhan, G. Yu and Y. Wang, Assessment of the validity of the quenching method for evaluating the role of reactive species in pollutant abatement during the persulfate-based process, Water Res., 2022, 221, 118730 CrossRef CAS PubMed.
- Y. Huang, J. Ren, Y. Wang, J. Fu, Y. Hou, B. Yang, L. Lei, A. Al-Anazi, G. Jiang and Z. Li, New insights into activation mechanisms of peroxymonosulfate by halide ions for micropollutant abatement: The generation routes and contributions of reactive radicals and hypohalous acid, Chem. Eng. Sci., 2025, 305, 121178 CrossRef CAS.
- K. Liu, J. Lu and Y. Ji, Formation of brominated disinfection by-products and bromate in cobalt catalyzed peroxymonosulfate oxidation of phenol, Water Res., 2015, 84, 1–7 CrossRef CAS PubMed.
- J. Lu, J. Wu, Y. Ji and D. Kong, Transformation of bromide in thermo activated persulfate oxidation processes, Water Res., 2015, 78, 1–8 CrossRef CAS PubMed.
- H. V. Lutze, N. Kerlin and T. C. Schmidt, Sulfate radical-based water treatment in presence of chloride: formation of chlorate, inter-conversion of sulfate radicals into hydroxyl radicals and influence of bicarbonate, Water Res., 2015, 72, 349–360 CrossRef CAS PubMed.
- P. Neta, R. E. Huie and A. B. Ross, Rate constants for reactions of inorganic radicals in aqueous solution, J. Phys. Chem. Ref. Data, 1988, 17, 1027–1284 CrossRef CAS.
- X. Lei, Y. Lei, J. Guan, P. Westerhoff and X. Yang, Kinetics and transformations of diverse dissolved organic matter fractions with sulfate radicals, Environ. Sci. Technol., 2022, 56, 4457–4466 CrossRef PubMed.
- M. Sillanpää, A. Matilainen and T. Lahtinen, in Natural Organic Matter in Water, 2015, pp. 17–53, DOI:10.1016/b978-0-12-801503-2.00002-1.
- S. Hou, L. Ling, D. D. Dionysiou, Y. Wang, J. Huang, K. Guo, X. Li and J. Fang, Chlorate formation mechanism in the presence of sulfate radical, chloride, bromide and natural organic matter, Environ. Sci. Technol., 2018, 52, 6317–6325 CrossRef CAS PubMed.
- J. Xin, Y. Liu, F. Chen, Y. Duan, G. Wei, X. Zheng and M. Li, The missing nitrogen pieces: A critical review on the distribution, transformation, and budget of nitrogen in the vadose zone-groundwater system, Water Res., 2019, 165, 114977 CrossRef CAS PubMed.
- P. Yang, Y. Ji and J. Lu, Transformation of ammonium to nitrophenolic byproducts by sulfate radical oxidation, Water Res., 2021, 202, 117432 CrossRef CAS PubMed.
- J. Liu, P. Yang, Y. Ji and J. Lu, Ammonium changes the pathway of tetrabromobisphenol S degradation by sulfate radical oxidation, Chem. Eng. J., 2023, 475, 146116 CrossRef CAS.
- L. Meng, J. Dong, J. Chen, L. Li, Q. Huang and J. Lu, Activation of peracetic acid by spinel FeCo2O4 nanoparticles for the degradation of sulfamethoxazole, Chem. Eng. J., 2023, 456, 141084 CrossRef CAS.
- Y. Feng, J. Ren, H. Li, D. Zhao, L. Sheng, Y. Wu, W. Zhao and M. Deng, Effect of thermal annealing on gas separation performance and aggregation structures of block polyimide membranes, Polymer, 2021, 219, 123538 CrossRef CAS.
- R. Pino-Rios, O. Yanez, D. Inostroza, L. Ruiz, C. Cardenas, P. Fuentealba and W. Tiznado, Proposal of a simple and effective local reactivity descriptor through a topological analysis of an orbital-weighted fukui function, J. Comput. Chem., 2017, 38, 481–488 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |



