Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Copper tungstate-assisted photocatalytic degradation of industrial products (dyes and pharmaceuticals) in water
Dhanalakshmi Vadivel *,
Jessica García,
Nithishkumar Kameswaran and
Daniele Dondi
*,
Jessica García,
Nithishkumar Kameswaran and
Daniele Dondi
Laboratory of Radiation and EPR Spectroscopy, Department of General Chemistry, University of Pavia, Via Torquato Taramelli 12, 27100 Pavia, Italy. E-mail: dhanalakshmi.vadivel@unipv.it
First published on 17th April 2026
Abstract
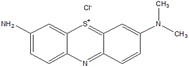
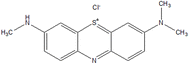
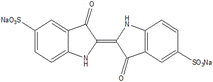
The contamination of ecosystems, specifically aquatic ecosystems, has emerged as a substantial concern in recent decades. This is mostly owing to the extensive growth of large industries that not only promote societal advancements but also impose adverse effects on the environment. Azure A (AA) and Azure B (AB) are the cationic dyes commonly employed in industrial and biomedical fields as intermediates in the production of several pharmaceuticals and as mediators for electrochemical biosensing, and indigo carmine (IC) is an anionic dye used in the textile industry for dyeing. Micropollutants such as pharmaceuticals, propranolol hydrochloride (β-blocker) (PPH) are the pollutants in the subject of discussion. In this research, the efficient degradation of AA (91%), AB (84.6%), IC (87.3%) and PPH (>80%) by the CuWO4 photocatalyst is highlighted. Both the dye and drug degradations followed pseudo-first order kinetics. CuWO4 catalyst is used to alleviate the impact of the environment on its ecosystem as a photocatalyst with ultraviolet (UV) irradiation of pollutants (AA, AB, and PPH). For the analysis of pollutant decomposition, UV-visible absorption spectroscopy and high-performance liquid chromatography (HPLC) are employed. This study highlights the potential of nanomaterial-based photocatalysis as a viable and effective method for sustainably mitigating water pollution.
1. Introduction
Over the decades, the development of industrialization has gained strength in society; nevertheless, it has also caused negative environmental impacts on ecosystems.1 As a report, about 15% of dyes are unscientifically discarded in the environment, especially in aquatic environments, which poses risks for both nature and health.2,3 Numerous dyes, including methylene blue (a phenothiazine dye)4 and indigo carmine, among others, contain aromatic rings; therefore, their biodegradation is not feasible.5,6“Emerging contaminants” are detected increasingly as low amounts in an ecosystem. The drugs,7–9 such as metoprolol, propranolol, atenolol, and pindolol belong to this group of contaminants are fall in this category. These drugs possess characteristic multi-functionalized aromatic groups, and they are soluble in water and susceptible to deionization.10–12 In European wastewater, an average concentration of 0.01 µg L−1 has been quantified up to concentrations of 0.29 µg L−1 for PPH.13 Many of the drugs are designed to be consumed orally, as they are resistant to neutral and/or basic hydrolysis, oxidation, or photolysis, which become their main routes of abiotic dissipation in natural waters. In relation to this, propranolol has a naphthalene skeleton, which indicates that it can act as a photosensitizing agent and be unstable to light.14
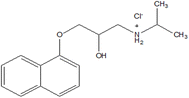
Propranolol hydrochloride (PPH) is a non-selective β-blocker drug used in clinical practice, which is administered mainly during cardiovascular therapy and for the management of primary hypertension.15–17 The assertion that the decrease in blood pressure is a consequence of diminished cardiac output caused by the inhibition of beta-receptors in the heart is subject to criticism.18 The presence of PPH in water is of concern due to its potential adverse effects on aquatic organisms and aquatic ecosystems in general.19 Although PPH is a commonly prescribed beta-blocker, it can be considered safe for humans20 only when used correctly. PPH can have adverse effects on aquatic organisms, especially at concentrations higher than the permissible limit in drinking water.
Industrial dyeing generates effluents, which comprise organic contaminants, and require treatment prior to discharge into natural water sources. Contemporary methods for treating wastewater involve the utilization of specialized oxidation techniques, including ozonation, Fenton reactions, and photocatalysis.21–24 Considerable interest has been directed towards photocatalysis due to its potential to eliminate toxic inorganic compounds, heavy metals, and organic pollutants from wastewater in a sustainable, eco-friendly, and straightforward manner.25–27
Solutions to technological and environmental challenges in the fields of solar energy conversion, catalysis, medicine, and water remediation are offered by nanomaterials.28,29 Researchers utilize metal oxide nanomaterials as a catalyst, including those of Fe3O4, TiO2, Al2O3, CuO, ZnO, and MgO, due to their versatile physical, chemical, and morphological properties that can be customized by tuning their synthesis parameters.30–33 The photodegradation of pollutants in water is a process that involve the production of reactive oxygen species.31
Conventional methods such as reverse osmosis, hydrogen peroxide treatment, dialysis, and UV photocatalysis34 are used for pollutant remediation from water. UV photocatalysis is a highly effective method owing to its easy operation, simple design, and the ability to completely degrade wastewater. The nanomaterials of metal oxides have the ability to degrade dyes present in industrial effluents. Notably, under ultraviolet light, low molecular weight metal oxide nanomaterials can engage in oxidation processes, yielding industrial CO2, H2O and aliphatic acids as final products. Researchers have shown significant interest in creating inorganic nanomaterials, such as metal oxide, metal tungstate, and metal molybdates, through synthesis.34,35
Copper tungstate (CuWO4) belongs to the tungstate family of compounds containing divalent transition metal ions that are structurally related. The 3d-orbitals of these metal ions are responsible for the observed electronic correlation effects.35 This semiconductor material with a high catalytic capacity is applied for nitric oxide gas sensing36,37 and photoelectrochemical water splitting.38–40 CuWO4 contains Cu2+ ions and has a d9 electronic configuration unlike common d0 photocatalysts such as WO3, TiO2, and BiVO4, and this distinct electronic structure results in different optical transitions, altered charge‑carrier behavior, stronger electronic correlation effects, and consequently unique photocatalytic properties.41–44 Recent research indicates that CuWO4 nanoparticles exhibit higher photocatalytic activity than P25.35 CuWO4 is formed through the reaction of Cu2+ with WO3 at high temperatures,45 and it can be produced in the form of thin films via the controlled addition of Cu2+ on the WO3 substrate.46
The present study aims to understand the degradation of PPH, AA, AB, and IC by the green photocatalyst42 CuWO4 under UVC and visible light irradiations. Analytical techniques such as UV-visible absorption spectroscopy and HPLC are used for the performance evaluation of CuWO4, and kinetic studies are also performed. The heterogeneous nature of the catalyst facilitates its subsequent removal following its utilization.47
2. Experimental methods
2.1. Chemicals
The compounds copper(II) sulphate pentahydrate CuSO4·5H2O (≥98%), AA, AB and IC were acquired from Sigma-Aldrich, USA. Sodium tungstate dihydrate, Na2WO4·2H2O (≥99%), was acquired from Sigma-Aldrich, India. DL-propranolol hydrochloride (99%) (Scheme 1) was provided by Thermo Fisher Scientific, Japan. Acetonitrile (ACN) for HPLC gradient grade (≥99.9%), phosphoric acid (H3PO4), and water for HPLC (H2O) were obtained from Sigma-Aldrich, France. For the synthesis of the catalyst, AA, and AB, we used double-distilled water. Nevertheless, to spike the IC, we used tap water from Pavia (Italy).2.2. Synthesis of CuWO4
CuWO4 was prepared via coprecipitation by mixing two aqueous solutions: 50 mL of 0.4 M solution A (CuSO4·5H2O) and 50 mL of 0.4 M solution B (Na2WO4·2H2O). Solution B was poured into solution A under stirring at a temperature of 60 °C and kept for 2 hours; subsequently, it was filtered, and the solid was dried at 100 °C for 24 hours in an oven, obtaining copper tungstate dihydrate.482.3. Instrumentation for degradation analyses
AA (Scheme 2), AB (Scheme 3) and IC dye decomposition analyses were performed by UV-visible absorption spectroscopy (Hewlett Packard 8452A diode array spectrophotometer) in 1 cm quartz cuvettes. The drug, PPH, was analyzed by HPLC. The HPLC setup is as follows: The Waters' HPLC system has an HPLC-UV-vis detector WATERS 2487 set to 291 nm. The system was equipped with a C-18 reverse phase Phenomenex column (150 × 4.6 mm, particle size 5 µL). The operating parameters for the HPLC analysis are as follows: a mobile phase consisting of a mixture of acetonitrile (ACN, 30% by volume) and water containing 0.1% of phosphoric acid (H3PO4) (70% by volume), a column temperature of 30 °C, an injection volume of 10 µL, and mobile phase flow rate set at 1 mL min−1.2.4. Sample preparation
A suspension of 50 ppm of propranolol hydrochloride, was added to 50 mg of CuWO4 in 100 mL. In the case of dye decomposition, 5 mg of CuWO4 was used in 25 mL of dye solution with a concentration of 5 × 10−5 mol dm−3. As the samples (dyes or/and drugs) were adsorbed on the CuWO4, the photocatalytic activity of the nanoparticles was tested. The dye samples were kept in darkness for 20 minutes. Then they were irradiated in quartz tubes inserted in a photoreactor equipped with 4 low pressure mercury lamps of 15 W each. All these solutions were also verified (maximum irradiation time of 2 h) by irradiating with an 18 W visible light LED lamp at 30 mW cm−2 (EvoluChem P204-18-1 6200K LED0000034) which has no appreciable reactivity. In the case of PPH this visible light is sufficient to perform the photo reaction. In the case of IC, we employed the dye in tap water to understand its decomposition efficiency as industrial waste on a real time scale (Scheme 4).3. Results and discussion
3.1. Characterization of CuWO4
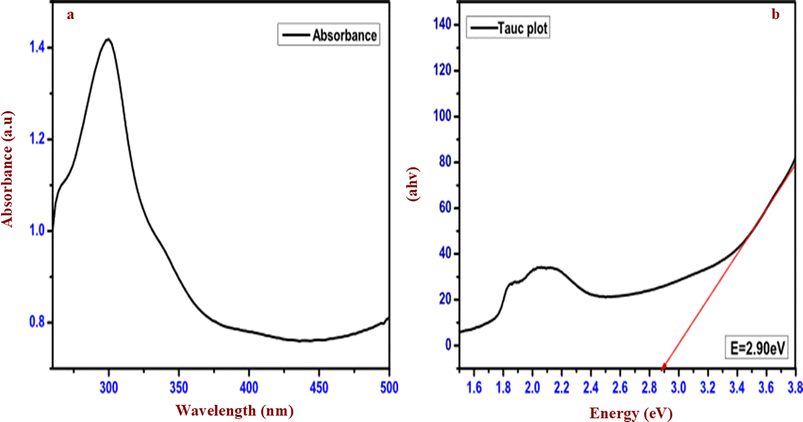
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group in the open crystallographic database 2024 (JCPDS number: 96-210-1693). The obtained diffraction patterns matched with the (101), (111), (012), (1−12), (1−22) and (3−11) planes of triclinic CuWO4.
space group in the open crystallographic database 2024 (JCPDS number: 96-210-1693). The obtained diffraction patterns matched with the (101), (111), (012), (1−12), (1−22) and (3−11) planes of triclinic CuWO4.
At lower concentrations of the dyes and the drug, the CuWO4 photocatalyst performed more efficiently under UVC light. We selected lower concentrations of dyes and drugs because the study focuses on conditions relevant to industrial effluents.
 | ||
| Fig. 3 (a and b) Scanning electron microscopic image of CuWO4. (c) Elemental distribution of CuWO4, showing the presence of (O), copper (Cu), and tungsten (W). | ||
The proportional amounts of oxygen (O), copper (Cu), and tungsten (W) obtained from the elemental analysis of CuWO4 photocatalyst as measured by SEM/EDX are shown in Table 1. Each of the values given would be acquired from the elemental analysis technique employed and indicate the proportional abundance of each element in the CuWO4 photocatalyst as measured by SEM/EDX.
| Elements | Weight percentage (%) | Atomic percentage (%) |
|---|---|---|
| Copper (Cu) | 17.24 | 11.07 |
| Tungsten (W) | 36.01 | 7.68 |
| Oxygen (O) | 28.05 | 56.78 |
Fig. 4 illustrates the process of thermal degradation of the CuWO4 sample. At temperatures below 200 °C, the TG measurements indicated a mass loss of less than 10%. This value is around 200–400 °C which depicts the necessity of the heating of CuWO4 catalyst after the synthesis above this temperature to obtain the anhydrous salt. However, since we are performing the application of the catalyst in water, the calcination step was not carried out. In addition, at temperatures above 850![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C, TG measurements indicated a mass loss of 12.86% which confirms the formation of CuWO4 which is further confirmed by the thermodynamic stability of the tungsten trioxide.55,56
°C, TG measurements indicated a mass loss of 12.86% which confirms the formation of CuWO4 which is further confirmed by the thermodynamic stability of the tungsten trioxide.55,56
3.2. Degradation of dyes using CuWO4
The utilization of photocatalysts to degrade dyes offers a viable solution for addressing water pollution issues caused by effluents containing dyes from diverse sectors such as textile, pharmaceutical, and chemical manufacturing. The dye molecules in wastewater will be bound to the photocatalyst surface by physical interactions, such as electrostatic attraction or van der Waals forces. This step increases the density of dye molecules that are in close proximity to the photocatalyst, facilitating their easy decomposition. Here, we selected the dye molecules of AA and AB (cationic dyes) and IC (an anionic dye) to be used in conjunction with the photocatalyst, CuWO4. In both cases, the photocatalyst exhibited the capability to gather and accumulate dye molecules in its close proximity.Upon absorbing photons with sufficient energy, photocatalysts produce electron–hole pairs that initiate dye degradation. Concurrently, reactive oxygen species (ROS) are generated on the catalyst surface such as hydroxyl radicals (˙OH) and superoxide ions when electron–hole pairs react with water and oxygen molecules or adsorbed substances.31,44,57–59 ROS with strong oxidative potential break chemical bonds and transform dye molecules into fragments. Photocatalysis maintains its effectiveness by converting dye molecules into non-toxic inorganic chemicals such as CO2 and H2O,60 making it a sustainable and environmentally benign method for treating wastewater.
To check the physical interaction (adsorption) of the dyes AA, AB, and IC with CuWO4, the catalyst and the solution were stirred for 20 minutes in the dark, as shown in Fig. 6, and the decrease in the absorption value of the solution was measured. It is well known that CuWO4 might be adsorbed on the dye surfaces with aromatic ring structures.61 After absorbing a photon, CuWO4 produces electron–hole pairs. These highly active charge carriers initiate the disintegration of dye molecules. The plausible mechanism reported in the literature is as follows: when the electron–hole pairs react with the oxygen molecules in water or adsorbed compounds, the surface of the photocatalyst forms reactive oxygen species (ROS), such as hydroxyl radicals (˙OH) and superoxide ions (Fig. 5).31,44,57–59 The generated ROS have a high oxidation potential, which means that they can break chemical bonds and convert dye molecules into basic chemicals, such as CO2 and H2O, which are safe for living beings. In the experiment, AA, AB, and IC were subjected to irradiation in the absence of CuWO4, as seen in Fig. 6.
The kinetics of the photoreactions were found to follow a pseudo-first-order model (Fig. 7). The kinetic constants were found to be, −2.4 × 10−2 a.u. min−1 and −2.14 × 10−2 a.u. min−1 for IC and AB, respectively. For AA, the pseudo-first order kinetic constant is −2.85 × 10−2 a.u. min−1; however, the deviation from linearity, as visible in Fig. 6, and the low R2 value (0.98) indicate that the underlying mechanism is probably more complex. The pseudo-first order rate constants were calculated using eqn (1).62,63
| ln(C/C0) = −kt | (1) |
To interpret the photoreaction dynamics, we calculated the amount of light absorbed by the catalyst with respect to dyes. Since the UVC lamp emits mainly at 254 nm, the radiation could be considered monochromatic, and hence, the calculation was performed at this wavelength. The AA, AB and IC dyes used in this experiment exhibited a measured molar absorption of 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300, 92
300, 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 and 20
600 and 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 dm3 mol−1 cm−1, respectively. In these experiments, CuWO4 is about 20 times more concentrated, thus drugs have a competitive absorption with the photocatalyst ranging from 50 to 70% of the light absorbed by dyes. Under these conditions, we expect an energy transfer from the excited state of the dye (that we demonstrated to be nonreactive) to the catalyst, giving rise to electron/hole pairs. By fast recombination of electron/hole pairs, molecules adsorbed onto the surface are mainly decomposed. This fact gives importance to the study of adsorption (a dark reaction) before irradiation.
500 dm3 mol−1 cm−1, respectively. In these experiments, CuWO4 is about 20 times more concentrated, thus drugs have a competitive absorption with the photocatalyst ranging from 50 to 70% of the light absorbed by dyes. Under these conditions, we expect an energy transfer from the excited state of the dye (that we demonstrated to be nonreactive) to the catalyst, giving rise to electron/hole pairs. By fast recombination of electron/hole pairs, molecules adsorbed onto the surface are mainly decomposed. This fact gives importance to the study of adsorption (a dark reaction) before irradiation.
3.3. Degradation of PPH by CuWO4
Fig. 8 summarizes the outcomes of the degradation of PPH under the influence of the CuWO4 catalyst. Within 140 minutes, >80% of PPH removal from the solution was achieved. Dark equilibration for 30 minutes revealed only a negligible interaction between the dye and the catalyst. On exposure to visible light, the dye concentrations reduced sharply within the first period of illumination. The kinetics of degradation were later determined using HPLC. | ||
| Fig. 8 Percentage of PPH degradation in the presence of CuWO4 under visible light and in dark conditions. | ||
The presence of CuWO4 facilitates chemical interactions, creating a conducive environment for the activation of PPH molecules. This activation process involves many transformations, such as oxidation and hydrolysis, which are particularly favored by catalysts of the metal oxide type, such as CuWO4.64
The addition of CuWO4 enhances the degradation efficiency of PPH, resulting in a higher conversion rate of the drug into breakdown products or metabolites. Thus, CuWO4 can be advantageous in wastewater treatment applications or in the elimination of medicinal compounds from the environment.64
The decomposition of PPH is directly related to the duration of irradiation, whether it is sunlight or artificial light, on the catalyst's surface because irradiation leads to the formation of electron pairs that engage in redox reactions with the adsorbed PPH. The percentage of PPH degradation is subject to variations depending on certain parameters, such as the catalyst type, PPH concentration, reaction conditions, and reaction duration, and modulating these parameters is essential to achieve optimal efficiency in the process.64–67
The kinetics of the reaction followed a pseudo-first-order model (Fig. 9). The kinetic constant was calculated to be −0.98 × 10−2 a.u. min−1 using eqn (1), but the deviation from linearity, as visible in Fig. 7, and the low R2 value (0.95) indicate that the underlying mechanism is probably more complex. This might be related to the decomposition of PPH by direct light absorption or due to the interactions between secondary products and PPH.
4. Conclusion
The effective use of CuWO4 (a green photocatalyst) eliminates pollutants such as AA dye (91%), AB dye (84.6%), and IC (87.3%) under UVC irradiation and degrades the drug PPH (>80%) under visible light irradiation. Both the dye and drug degradation reactions follow pseudo-first-order kinetics. Such research on pharmaceutical and dyeing industries will ensure that there is a sustainable and environment-friendly approach to dealing with water pollution. UV-visible absorption spectroscopy and high-performance liquid chromatography (HPLC) are employed to analyze the catalyst's efficacy. These findings indicate the advantages of using material-based photocatalysis (green photocatalyst) as an effective method to mitigate the effects of industrial and medicinal pollutants on the environment. Further exploration and advancements in this domain could potentially result in the development and wider use of enhanced methodologies for water treatment, eventually fostering the preservation of natural environments.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this paper.Data availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.Acknowledgements
This work was funded in part through the project NODES, Ministry of University and Research (M4C2 1.5 PNRR), and in part by the European Union, NextGenerationEU program under the grant agreement no. ECS00000036. D. V. and D. D. acknowledge support from the Ministry of University and Research (MUR) and the University of Pavia through the program “Dipartimenti di Eccellenza 2023–2027.”References
- A. Gaur, V. S. Chauhan and R. Vaish, Environ. Sci.:Adv., 2023, 2, 462–472 CAS.
- R. Yang, Y. Fan, R. Ye, Y. Tang, X. Cao, Z. Yin and Z. Zeng, Adv. Mater., 2021, 33, 2004862 CrossRef CAS.
- T. Wu, Q. Liang, L. Tang, J. Tang, J. Wang, B. Shao, S. Gong, Q. He, Y. Pan and Z. Liu, J. Hazard. Mater., 2023, 443, 130251 CrossRef CAS.
- T. Chellapandi, G. Madhumitha, S. M. Roopan, M. Elamathi, K. Leeladevi, E. R. Nagarajan, D. Vadivel and D. Dondi, Opt. Mater., 2023, 142, 114099 CrossRef CAS.
- F. Wang, Y. Gao, S.-S. Liu, X.-H. Yi, C.-C. Wang and H. Fu, Chem. Eng. J., 2023, 463, 142466 CrossRef CAS.
- E. Aneggi, S. Hussain, W. Baratta, D. Zuccaccia and D. Goi, Molecules, 2024, 29, 2074 CrossRef CAS PubMed.
- A. Speltini, M. Sturini, F. Maraschi, E. Mandelli, D. Vadivel, D. Dondi and A. Profumo, Microchim. Acta, 2016, 183, 2241–2249 CrossRef CAS.
- M. Hu, J. Song, X. Yi, Z. Yuan, C. Wang, R. Gao, H. Zou, H. Zhang, C. Lv, H. Cui, T. Wang, C. Zhang, Z. Wei, K. Yun and C.-E. Chen, Environ. Sci.:Adv., 2025, 4, 1782–1795 CAS.
- S. Karishma, P. R. Yaashikaa, P. S. Kumar, R. Kamalesh, A. Saravanan and G. Rangasamy, Environ. Sci.:Adv., 2023, 2, 1488–1504 CAS.
- R. Zhang, G. Zhang, Q. Zheng, J. Tang, Y. Chen, W. Xu, Y. Zou and X. Chen, Ecotoxicol. Environ. Saf., 2012, 80, 208–215 CrossRef CAS.
- S. Zou, W. Xu, R. Zhang, J. Tang, Y. Chen and G. Zhang, Environ. Pollut., 2011, 159, 2913–2920 CrossRef CAS.
- M. J. Martínez Bueno, M. D. Hernando, A. Agüera and A. R. Fernández-Alba, Talanta, 2009, 77, 1518–1527 CrossRef.
- I. Sirés, N. Oturan and M. A. Oturan, Water Res., 2010, 44, 3109–3120 CrossRef.
- R. F. Dantas, O. Rossiter, A. K. R. Teixeira, A. S. M. Simões and V. L. Da Silva, Chem. Eng. J., 2010, 158, 143–147 CrossRef CAS.
- K. Kovács, A. Tegze, A. Bezsenyi and L. Wojnárovits, J. Environ. Chem. Eng., 2023, 11, 110330 CrossRef.
- A. Piram, R. Faure, H. Chermette, C. Bordes, B. Herbreteau and A. Salvador, Int. J. Environ. Anal. Chem., 2012, 92, 96–109 CrossRef CAS.
- H. Karppanen, Naunyn-Schmiedeberg's Arch. Pharmacol., 1974, 281, 1–12 CrossRef CAS PubMed.
- E. D. Frohlich, R. C. Tarazi, H. P. Dustan and I. H. Page, Circulation, 1968, 37, 417–423 CrossRef CAS.
- D. Zhou, H. Liu, J. Wang, Y. Li, N. Wang and W. Li, Chemosphere, 2024, 357, 141985 CrossRef CAS.
- L. Luyten, A. Chalkia, A. E. Schnell, B. Özcan, L. Leng, N. Schroyens, L. Van Oudenhove, W. Vanpaemel and T. Beckers, J. Anxiety Disord., 2024, 104, 102870 CrossRef.
- S. Chen, T. Ren, Z. Zhou, K. Lu, X. Huang and X. Zhang, Chem. Eng. J., 2023, 475, 145874 CrossRef CAS.
- K.-T. Lee, K.-Y. Ho, W.-H. Chen, E. E. Kwon, K.-Y. A. Lin and S.-R. Liou, Environ. Pollut., 2023, 335, 122246 CrossRef CAS PubMed.
- V. P. Sarvothaman, V. K. Velisoju, J. Subburaj, M. S. Panithasan, S. R. Kulkarni, P. Castaño, J. Turner, P. Guida, W. L. Roberts and S. Nagarajan, Ultrason. Sonochem., 2023, 99, 106548 CrossRef CAS.
- Z. Huang, X. Huang, K. Liu, J. Fu and M. Liu, Environ. Sci.:Adv., 2026, 5, 9–42 CAS.
- S. Kumar, W. Ahlawat, G. Bhanjana, S. Heydarifard, M. M. Nazhad and N. Dilbaghi, J. Nanosci. Nanotechnol., 2014, 14, 1838–1858 CrossRef CAS.
- C. Khaokhajorn, P. Amornpitoksuk, C. Randorn, T. Rattana and S. Suwanboon, Inorg. Chem. Commun., 2023, 157, 111392 CrossRef CAS.
- M. A. Al-Nuaim, A. A. Alwasiti and Z. Y. Shnain, Chem. Pap., 2023, 77, 677–701 CrossRef CAS.
- M. A. Vallejo Pat, H. Ezekiel-Hart and C. D. Powell, Nanomaterials, 2025, 15, 1377 CrossRef CAS.
- S. Liu, G. Huang, J. Wang, J. Bao, M. Wang, Y. Wei, Y. Zhong and F. Bai, Nanomaterials, 2023, 13, 660 CrossRef CAS.
- S. Haviar, J. Čapek, Š. Batková, N. Kumar, F. Dvořák, T. Duchoň, M. Fialová and P. Zeman, Int. J. Hydrogen Energy, 2018, 43, 22756–22764 CrossRef CAS.
- B. R. Gomes, J. L. Lopes, L. Coelho, M. Ligonzo, M. Rigoletto, G. Magnacca and F. Deganello, Nanomaterials, 2023, 13, 2276 CrossRef CAS.
- N. Kumar, S. Haviar and P. Zeman, Nanomaterials, 2021, 11, 3456 CrossRef CAS.
- S. Yousafzai, M. Farid, M. Zubair, N. Naeem, W. Zafar, Z. U. Zaman Asam, S. Farid and S. Ali, Environ. Sci.:Adv., 2025, 4, 1142–1165 CAS.
- S. Narendhran, P. B. Shakila, M. Manikandan, V. Vinoth and P. Rajiv, Spectrochim. Acta, Part A, 2020, 232, 118164 CrossRef CAS.
- H. Ramezanalizadeh and F. Manteghi, J. Photochem. Photobiol., A, 2017, 338, 60–71 CrossRef CAS.
- C. M. Gonzalez, X. Du, J. L. Dunford and M. L. Post, Sens. Actuators, B, 2012, 173, 169–176 CrossRef CAS.
- W. Zhang, D. Zhang and Y. Zhang, J. Mater. Sci.:Mater. Electron., 2020, 31, 6706–6715 CrossRef CAS.
- V. Gajraj and C. R. Mariappan, J. Electroanal. Chem., 2021, 895, 115504 CrossRef CAS.
- Y. Chang, A. Braun, A. Deangelis, J. Kaneshiro and N. Gaillard, J. Phys. Chem. C, 2011, 115, 25490–25495 CrossRef CAS.
- C. M. Tian, M. Jiang, D. Tang, L. Qiao, H. Y. Xiao, F. E. Oropeza, J. P. Hofmann, E. J. M. Hensen, A. Tadich, W. Li, D. C. Qi and K. H. L. Zhang, J. Mater. Chem. A, 2019, 7, 11895–11907 RSC.
- J. E. Yourey, K. J. Pyper, J. B. Kurtz and B. M. Bartlett, J. Phys. Chem. C, 2013, 117, 8708–8718 CrossRef CAS.
- H. Ramezanalizadeh and F. Manteghi, J. Cleaner Prod., 2018, 172, 2655–2666 CrossRef CAS.
- V. Vinesh, M. Preeyanghaa, T. R. N. Kumar, M. Ashokkumar, C. L. Bianchi and B. Neppolian, Environ. Res., 2022, 207, 112112 CrossRef CAS PubMed.
- P. Raizada, S. Sharma, A. Kumar, P. Singh, A. A. Parwaz Khan and A. M. Asiri, J. Environ. Chem. Eng., 2020, 8, 104230 CrossRef CAS.
- J. C. Hill and K.-S. Choi, J. Mater. Chem. A, 2013, 1, 5006 RSC.
- D. Wang, P. Bassi, H. Qi, X. Zhao, Gurudayal, L. Wong, R. Xu, T. Sritharan and Z. Chen, Materials, 2016, 9, 348 CrossRef PubMed.
- N. A. Lima, G. C. Mendonça, G. T. S. T. Da Silva, B. S. De Lima, E. C. Paris and M. I. B. Bernardi, J. Mater. Sci.:Mater. Electron., 2021, 32, 1139–1149 CrossRef CAS.
- S. Medidi, S. Markapurapu, M. R. Kotupalli, R. K. R. Chinnam, V. M. Susarla, H. B. Gandham and P. D. Sanasi, Mod. Res. Catal., 2018, 7, 17–34 CrossRef CAS.
- N. Le Minh Tri, D. Q. Trung, D. Van Thuan, N. T. Dieu Cam, T. Al Tahtamouni, T.-D. Pham, D. S. Duc, M. H. Thanh Tung, H. Van Ha, N. H. Anh Thu and H. T. Trang, Int. J. Hydrogen Energy, 2020, 45, 18186–18194 CrossRef CAS.
- X. Hu, D. Gao, Y. Li, H. Dong, W. Zhou, L. Yang and Y. Zhang, SN Appl. Sci., 2019, 1, 119 CrossRef.
- J. E. Yourey and B. M. Bartlett, J. Mater. Chem., 2011, 21, 7651 RSC.
- M. Shekofteh-Gohari and A. Habibi-Yangjeh, J. Ind. Eng. Chem., 2016, 44, 174–184 CrossRef CAS.
- D. P. Dutta, A. Rathore, A. Ballal and A. K. Tyagi, RSC Adv., 2015, 5, 94866–94878 RSC.
- Y. Tang, N. Rong, F. Liu, M. Chu, H. Dong, Y. Zhang and P. Xiao, Appl. Surf. Sci., 2016, 361, 133–140 CrossRef CAS.
- J. Wendel, Master thesis, Lund University, 2014.
- W. Hu, F. Donat, S. A. Scott and J. S. Dennis, RSC Adv., 2016, 6, 113016–113024 RSC.
- D. Vadivel and I. Malaichamy, F1000Research, 2018, 7, 1574 Search PubMed.
- D. Vadivel, D. S. Branciforti, A. Speltini, M. Sturini, V. Bellani, I. Malaichamy and D. Dondi, Catalysts, 2020, 10, 270 CrossRef CAS.
- D. Vadivel, M. Sturini, A. Speltini and D. Dondi, Catalysts, 2022, 12, 310 CrossRef CAS.
- M. Foroughi, S. J. Peighambardoust, B. Ramavandi, R. Foroutan and N. S. Peighambardoust, Sep. Purif. Technol., 2024, 344, 127265 CrossRef CAS.
- M. F. Hanafi and N. Sapawe, Mater. Today: Proc., 2020, 31, 318–320 CAS.
- P. Kumbhakar, A. Pramanik, S. Biswas, A. K. Kole, R. Sarkar and P. Kumbhakar, J. Hazard. Mater., 2018, 360, 193–203 CrossRef CAS PubMed.
- H. Zeghioud, M. Kamagate, L. S. Coulibaly, S. Rtimi and A. A. Assadi, Chem. Eng. Res. Des., 2019, 144, 300–309 CrossRef CAS.
- E. Isarain-Chávez, R. M. Rodríguez, P. L. Cabot, F. Centellas, C. Arias, J. A. Garrido and E. Brillas, Water Res., 2011, 45, 4119–4130 CrossRef.
- J. Santiago-Morales, A. Agüera, M. D. M. Gómez, A. R. Fernández-Alba, J. Giménez, S. Esplugas and R. Rosal, Appl. Catal., B, 2013, 129, 13–29 CrossRef CAS.
- V. Romero, N. De La Cruz, R. F. Dantas, P. Marco, J. Giménez and S. Esplugas, Catal. Today, 2011, 161, 115–120 CrossRef CAS.
- A. H. Navidpour, M. B. Ahmed and J. L. Zhou, Nanomaterials, 2024, 14, 135 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |