 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
pH-driven spontaneous recovery of tyrosine via co-precipitation in the indirect aqueous carbonation of gypsum
Yujie Qua,
Yuan Gong *ab,
Baizhi Wua,
Chunlei Lia,
Lanying Wangc and
Yi Wangab
*ab,
Baizhi Wua,
Chunlei Lia,
Lanying Wangc and
Yi Wangab
aSchool of Petrochemical Technology, Lanzhou University of Technology, Lanzhou, 730050, PR China. E-mail: yuangong@lut.edu.cn
bGansu Engineering Research Center for Resource Utilization of New Energy Industry Waste and Waste-Utilized Materials, Lanzhou, 730050, PR China
cJiuquan Iron and Steel (Group) Co., Ltd., Jiayuguan, 735100, PR China
First published on 17th February 2026
Abstract
Efficient recycling of leaching additives is crucial for the economic viability of indirect aqueous carbonation of gypsum. This study presents a pH-driven spontaneous co-precipitation strategy employing tyrosine (Tyr) as a recyclable additive. The method exploits the inherent pH decrease during carbonation to spontaneously precipitate and separate Tyr from the SO42−-rich carbonation mother liquor. In a strongly alkaline medium, fully deprotonated Tyr chelates with Ca2+, significantly enhancing the solubility of CaSO4·2H2O. Response surface methodology based on a Box–Behnken design was applied to optimize the leaching conditions, which were determined as follows: Tyr/CaSO4·2H2O = 5.51 mol mol−1, KOH/Tyr = 2.00 mol mol−1, and liquid-to-solid ratio = 51.38 mL g−1. Under these conditions, the leached Ca2+ concentration reached 16.09 ± 0.30 g L−1—six times higher than the solubility of CaSO4·2H2O in pure water at 30 °C (∼2.6 g L−1). During the early stage of carbonation, the Ca2+–Tyr complexes dissociate as the pH decreases, releasing free Ca2+ for direct precipitation of homogeneous calcite, while neutral Tyr0 co-precipitates efficiently. Under the optimized carbonation conditions (30 °C, CO2 flow rate 150 mL min−1, 50 min), the carbonation efficiency and Tyr recovery efficiency exceeded 97% and 95%, respectively. The process also demonstrated excellent stability over five leaching–carbonation cycles, with average values of 12.63 ± 0.56 g L−1 for Ca2+ leaching concentration, 93.41 ± 1.85% for carbonation efficiency, and 92.73 ± 0.52% for Tyr recovery efficiency.
1. Introduction
Aqueous carbonation is widely regarded as a promising process for CO2 sequestration owing to its favorable thermodynamics, mild operating conditions, and high reactivity.1,2 Industrial waste gypsum, primarily composed of CaSO4·2H2O with a theoretical CaO content of ∼32.5%, serves as an ideal calcium source for this process.3 Its use as a substitute for natural calcium-rich minerals therefore offers a synergistic strategy to address both solid waste management and CO2 utilization/storage challenges.4–6Aqueous carbonation can be classified into direct and indirect pathways.7 The indirect two-step route involves first leaching Ca2+ from the solid feedstock, then raising the pH to an alkaline range and introducing CO2 into the leachate to precipitate CaCO3. Compared with direct carbonation, this approach effectively removes insoluble impurities, operates under milder conditions, and allows precise control over the purity, polymorph, and particle size distribution of the resulting CaCO3. These advantages make it a promising route for the high-value utilization of solid waste gypsum.8,9 The efficiency and selectivity of calcium leaching are critical, as they directly govern the subsequent carbonation efficiency and the purity of the final carbonate product. Various leaching agents have been explored to enhance leaching performance. For example, Rahmani et al.10,11 used H2SO4 to leach Ca2+ from red gypsum, while Ding et al. employed inorganic salts such as CH3COONH4, NaCl, and NH4Cl to improve the dissolution of desulfurization gypsum or phosphogypsum via the salt effect.12–15 However, despite their efficacy, the stable recycling of these leaching agents remains a major practical challenge.4 In particular, directly recycling the carbonation mother liquor containing additives in a leaching–carbonation cycle can lead to a gradual decline in Ca2+ leaching yield due to the common-ion effect from accumulating SO42−.13 Hence, developing efficient methods to separate and recycle leaching additives from SO42−-enriched carbonation mother liquor is crucial for advancing the indirect aqueous carbonation of gypsum.
In this context, organic additives such as amino acids have emerged as promising candidates due to their unique recyclability. Their distinctive molecular structure, featuring both amino and carboxyl groups, enables effective chelation of Ca2+, thereby facilitating the dissolution of calcium-based feedstocks.16 Recently, amino acids have attracted considerable interest as promoters for the indirect aqueous carbonation of fly ash.17–19 Moreover, during carbonation, the amino groups contribute to CO2 absorption by forming carbamate and protonated amino acid intermediates. These species hydrolyze upon CaCO3 precipitation, regenerating the amino acids with an efficiency surpassing conventional thermal regeneration.20 This regenerative mechanism is synergistically driven by the exothermicity of carbonation and the precipitation of CaCO3 itself, offering a promising route to reduce the energy consumption and cost associated with amino acid recovery.21,22
As is well known, amino acids exhibit minimal solubility at their isoelectric points (pI). This property suggests that selecting amino acids with a strong solubility contrast near their pI could enable their effective separation from SO42−-rich carbonation mother liquor, positioning them as candidate additives for indirect gypsum carbonation. In our previous work, using aspartic acid (Asp) as a leaching additive increased the solubility of CaSO4·2H2O to 19.50 g L−1 at 30 °C, roughly ten times higher than in pure water. Importantly, both the additive and SO42− were effectively separated from the mother liquor at the pI of Asp (2.77), enabling stable recycling with a recovery efficiency exceeding 80% over 10 cycles.16 However, the low pI of Asp necessitates excess H2SO4 for pH adjustment, which complicates the regeneration process.
This work presents a strategy for the indirect aqueous carbonation of gypsum using tyrosine (Tyr) as a leaching additive. The approach leverages the finding that Tyr co-precipitates with CaCO3 during carbonation, allowing spontaneous separation of the additive from SO42− in the mother liquor without additional acidification. The co-precipitated Tyr is then separated from CaCO3 by alkaline washing, and the resulting filtrate, containing redissolved Tyr, is recycled for subsequent Ca2+ leaching. To maximize the Ca2+ leaching yield, dissolution conditions for CaSO4·2H2O in the alkaline Tyr–KOH–H2O system were first optimized using response surface methodology (RSM). The effects of CO2 flow rate, reaction time, and temperature on carbonation efficiency and Tyr recovery were systematically investigated, leading to a proposed mechanism for the Tyr-mediated carbonation process. Finally, cyclic stability was evaluated over five consecutive leaching–carbonation cycles by monitoring Ca2+ leaching yield, carbonation efficiency, and Tyr recovery efficiency.
2. Materials and methods
2.1. Materials
Analytical-grade CaSO4·2H2O was used in all experiments to clarify the migration behavior of Ca2+ in the Tyr-mediated carbonation process. This choice avoided the complex effects of soluble impurities typically present in industrial waste gypsum, enabling a focused investigation of the underlying mechanism. Analytical-grade KOH, a strong electrolyte, was selected for the alkaline system to ensure efficient dissolution of Tyr and to provide precise stoichiometric control. Tyr and an EDTA standard solution (0.01 mol L−1), also of analytical grade, were purchased from Shanghai Macklin Biochemical Co., Ltd., China. All reagents were used as received. Ultrapure water, prepared using a purification system (Merck Chemical Technology Co., Ltd., USA), was employed throughout. CO2 gas (purity ≥ 99%) was supplied by Lanzhou Hongli Gas Co., Ltd., China.2.2. Methods
| No. | CO2 flow rate (mL min−1) | Temperature (°C) | Time (min) |
|---|---|---|---|
| 1 | 100 | 30 | 60 |
| 2 | 100 | 40 | 60 |
| 3 | 100 | 50 | 60 |
| 4 | 100 | 60 | 60 |
| 5 | 50 | 30 | 60 |
| 6 | 80 | 30 | 60 |
| 7 | 100 | 30 | 60 |
| 8 | 150 | 30 | 60 |
| 9 | 200 | 30 | 60 |
| 10 | 150 | 30 | 10 |
| 11 | 150 | 30 | 20 |
| 12 | 150 | 30 | 30 |
| 13 | 150 | 30 | 40 |
| 14 | 150 | 30 | 50 |
| 15 | 150 | 30 | 60 |
The pH was monitored in real-time throughout carbonation using a calibrated pH meter (DZS-708T, INESA Scientific Instrument Co., Ltd., China) to track the reaction progress. Upon completion, the precipitate (primarily CaCO3 and co-precipitated Tyr) was separated by vacuum filtration. It was then washed with a KOH solution (prepared at the same concentration as the initial leaching solution) to redissolve Tyr. The washed precipitate was dried at 80 °C to constant weight and characterized by scanning electron microscopy (SEM, Sigma 300, Carl Zeiss AG, Germany), thermogravimetric analysis (TGA, TGA/DSC 3+, Mettler Toledo, Switzerland), particle size distribution analysis (Mastersizer 3000, Malvern Panalytical, UK), and XRD. SEM was conducted at an accelerating voltage of 5 kV. TGA was performed under a nitrogen atmosphere at a heating rate of 10 °C min−1 from 30 to 950 °C. The alkaline wash solution containing redissolved Tyr was recycled for five consecutive leaching–carbonation cycles. Before each new cycle, lost Tyr was replenished to restore the pH of the initial alkaline leaching solution. A schematic of the overall experimental procedure is presented in Fig. 1.
Key performance indicators, including the Ca2+ leaching yield (φ, g L−1), carbonation efficiency (ω, %), and Tyr recovery efficiency (η, %), were calculated using eqn (1)–(3), respectively.
Ca2+ leaching yield:
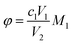 | (1) |
Carbonation efficiency:
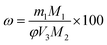 | (2) |
Tyr recovery efficiency:
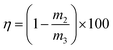 | (3) |
3. Results and discussion
3.1. Optimization of Ca2+ leaching conditions in the Tyr–KOH–H2O system
The analysis of variance (ANOVA) confirmed the statistical significance of the established model (eqn (S1)) for the Ca2+ leaching yield (φ). As detailed in Table S2, the model exhibited a high F-value of 108.96 and a highly significant p-value (<0.0001). The contour plots (Fig. 2a–c) illustrate the interaction effects of the factors on φ, revealing distinct peaks within specific operational ranges. As summarized in Table 2, the model predictions agree well with the experimental data across the leaching conditions, with a maximum deviation of 7.91% when data groups with β = 3 are excluded. The optimal leaching conditions derived from the model were α = 5.51, β = 2.00, and γ = 51.39 mL g−1, corresponding to a predicted maximum φ of 15.41 g L−1. Validation experiments performed in triplicate (Fig. 2d) showed that all results fell within the 95% prediction interval of the model, further confirming its reliability. In summary, the complexation between Tyr and Ca2+ substantially enhanced the solubility of CaSO4·2H2O, achieving a value over six times greater than in pure water (∼2.6 g L−1 at 30 °C). | ||
| Fig. 2 (a–c) Contour plots illustrating the interaction effects among the studied factors; (d) validation experiments performed under the optimized conditions. | ||
| No. | α (mol mol−1) | β (mol mol−1) | γ (mL g−1) | φ (g L−1) | Deviation (%) Abs. value | |
|---|---|---|---|---|---|---|
| Exp. value | Pred. value | |||||
| 1 | 9.00 | 2.00 | 20.00 | 6.02 | 6.25 | 3.74 |
| 2 | 9.00 | 1.00 | 50.00 | 14.51 | 14.94 | 2.98 |
| 3 | 1.00 | 3.00 | 50.00 | 8.03 | 7.50 | 6.64 |
| 4 | 9.00 | 2.00 | 80.00 | 11.67 | 11.17 | 4.33 |
| 5 | 5.00 | 3.00 | 20.00 | 0.77 | 0.96 | 24.03 |
| 6 | 5.00 | 1.00 | 80.00 | 7.80 | 7.50 | 3.88 |
| 7 | 1.00 | 2.00 | 20.00 | 9.69 | 10.02 | 3.42 |
| 8 | 1.00 | 1.00 | 50.00 | 5.10 | 5.54 | 8.69 |
| 9 | 5.00 | 3.00 | 80.00 | 2.26 | 2.94 | 29.87 |
| 10 | 5.00 | 2.00 | 50.00 | 15.16 | 15.32 | 1.04 |
| 11 | 9.00 | 3.00 | 50.00 | 0.69 | 0.15 | 78.99 |
| 12 | 1.00 | 2.00 | 80.00 | 5.62 | 5.34 | 4.96 |
| 13 | 5.00 | 2.00 | 50.00 | 15.65 | 15.32 | 2.13 |
| 14 | 5.00 | 1.00 | 20.00 | 10.03 | 9.24 | 7.91 |
| 15 | 5.00 | 2.00 | 50.00 | 15.94 | 15.32 | 3.91 |
| 16 | 5.00 | 2.00 | 50.00 | 15.14 | 15.32 | 1.17 |
| 17 | 5.00 | 2.00 | 50.00 | 14.96 | 15.32 | 2.39 |
Response surface analysis indicated that factor β (KOH/Tyr molar ratio) exerted the most significant influence on the Ca2+ leaching yield. To investigate this effect in detail, a single-factor experiment was conducted. The XRD patterns of the solid residues obtained after leaching CaSO4·2H2O at different β values are presented in Fig. 3. When β was below 2.5, the dominant crystalline phase remained CaSO4·2H2O. With a further increase in β, however, the phase converted to Ca(OH)2 due to the excess OH−. This transformation aligns with the reaction stoichiometry: the molecular structure of Tyr contains one carboxyl and one phenolic hydroxyl group, which react with KOH in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratio. Once β exceeds this stoichiometric point, the surplus OH− reacts with CaSO4·2H2O to form the less soluble Ca(OH)2.23 The significant deviation observed at β = 3 can therefore be attributed to this phase transformation under highly alkaline conditions.
2 molar ratio. Once β exceeds this stoichiometric point, the surplus OH− reacts with CaSO4·2H2O to form the less soluble Ca(OH)2.23 The significant deviation observed at β = 3 can therefore be attributed to this phase transformation under highly alkaline conditions.
 | ||
| Fig. 3 XRD patterns of the residual solid phases obtained after leaching CaSO4·2H2O at different β values. | ||
The speciation of Tyr is strongly pH-dependent. Based on its dissociation constants (Table S3),24 the distribution of its various forms as a function of pH was calculated (Fig. S1). According to acid-base proton theory, in a strongly alkaline KOH solution (pH > 11), the zwitterionic form (Tyr0) donates protons from both the α-amino (–NH3+) and phenolic hydroxyl groups, yielding the anionic species Tyr− and Tyr2−. This transformation is corroborated by the FTIR spectra shown in Fig. 4. Compared with analytical-grade Tyr, the blank sample (Tyr + KOH) exhibits the following spectral changes: the characteristic O–H stretching vibration of the phenolic hydroxyl at 3203 cm−1 and the N–H stretching vibrations of the –NH3+ group around 3000 cm−1 disappear. Concurrently, the C–O stretching vibration of the phenolic hydroxyl shifts from 1245 cm−1 to 1282 cm−1. These observations confirm that Tyr exists predominantly as the Tyr2− anion in the leachate when the KOH/Tyr molar ratio (β) equals 2. Further comparison between the blank sample and the freeze-dried solid from the filtrate (Tyr + Ca + KOH) reveals an additional peak at 1123 cm−1, assigned to the antisymmetric stretching vibration of SO42−.25 In the magnified low-frequency region (800–400 cm−1), two new absorption bands emerge at 547 cm−1 and 618 cm−1. These bands can be attributed to the stretching vibrations of Ca–O and Ca–N coordination bonds,26 indicating the formation of a Ca2+–Tyr complex intermediate.
 | ||
| Fig. 4 FTIR spectra of the freeze-dried solid from filtrate, blank sample, and analytical-grade Tyr. | ||
3.2. Characterization of the carbonation reaction process involving Tyr
The XRD patterns (Fig. 6) show that a homogeneous calcite product was obtained across the entire temperature range. This contrasts with previous reports using glycine, alanine, or aspartic acid, where vaterite typically persisted as a stable phase.26,29 In those systems, deprotonated amino acid anions adsorbed onto initially formed vaterite, inhibiting its transformation to calcite. The predominant formation of calcite in the present system is presumably due to the rapid dissociation of the Ca2+–Tyr complex during the initial carbonation stage. Driven by the sharp pH decrease, this dissociation releases free Ca2+ ions, which subsequently precipitate directly as calcite—a process with a lower activation energy than that involving chelated calcium.26 Furthermore, the crystallinity of CaCO3 decreased with increasing temperature. SEM images reveal a slight increase in particle size from 30 to 60 °C, indicating crystal growth. Distinct lamellar steps emerged on crystal surfaces at around 40 °C, which act as active nucleation sites promoting crystal growth via a spiral growth mechanism.30,31 Therefore, lower temperatures not only improve Tyr recovery efficiency but also favor the formation of well-defined CaCO3 crystals.
Fig. 7c compares the dynamic pH profiles at CO2 flow rates of 50 and 100 mL min−1. At 100 mL min−1, the pH evolution displayed three distinct stages: a rapid but decelerating drop from 11.2 to 9.8 within the first 600 s, a plateau (600–1150 s) with a further slow decline to 9.4, followed by an accelerated decrease. This progression is governed by the kinetics of CO32− formation (Reaction (R1)). In the first stage, rapid dissociation of the Ca2+–Tyr complex released abundant Tyr2−, whose fraction decreased from >90% to approximately 28% over this pH range (Fig. S1). As a strong proton acceptor, Tyr2− effectively consumed H+ generated from Reaction (R1), thereby buffering the initial pH drop and causing the observed deceleration. The plateau corresponds to a period of stable crystal growth, where the rate of CO32− formation was balanced by its consumption through CaCO3 crystallization (Reaction (R2)).32 In the final stage, the low concentration of free Ca2+ led to excess of CO32− formation. Although the dominant Tyr− species still provided buffering, its amino group likely participated in CO2 absorption, promoting HCO3− formation which exerts a stronger influence on pH and contributes to the accelerated decline.33 In contrast, at the lower CO2 flow rate of 50 mL min−1, the slower rate of CO32− formation delayed the onset of the plateau and extended its duration. XRD patterns (Fig. 7d) show that the diffraction peak intensity of CaCO3 initially increased and then decreased with rising CO2 flow rate, peaking at 150 mL min−1. Consequently, a flow rate of 150 mL min−1 was selected to ensure high Tyr recovery and well-crystallized product formation.
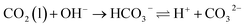 | (R1) |
| Ca2+ + CO32− → CaCO3 | (R2) |
TGA analysis (Fig. 9a) showed that extending the reaction time from 20 to 60 min significantly increased the total mass loss in the 150–550 °C range from 0.68% to 3.39%. This loss corresponds to the decomposition of Tyr (Δm1, 150–350 °C) and the dehydration of crystalline water (Δm2, 350–550 °C).29,37 During the first 20 min rapid crystallization stage, adsorption of Tyr anions on CaCO3 surfaces was insufficient to induce the formation of metastable vaterite. In the later stage, the neutral Tyr0 was predominantly incorporated into the crystals, mainly via entrapment during dissolution-reprecipitation events associated with Ostwald ripening, identified as the major cause of Tyr loss. The FTIR spectrum of the recovered Tyr (Fig. 9b) closely matches that of analytical-grade Tyr, confirming the high structural integrity of the recycled additive, which was achieved with a maximum recovery efficiency of 95.47% under the optimized reaction conditions.
 | ||
| Fig. 9 (a) TGA curves of the CaCO3 products obtained at different reaction times. (b) FTIR spectra of the recovered Tyr and analytical-grade Tyr. | ||
Based on these findings, a reaction mechanism for the Tyr-mediated indirect aqueous carbonation of CaSO4·2H2O is summarized (Fig. 10). In the strongly alkaline leaching system, Tyr deprotonates to Tyr2−, which complexes with Ca2+ to enhance CaSO4·2H2O dissolution. During subsequent carbonation, the pH drop triggers the rapid complex dissociation, releasing free Ca2+ that serves as the calcium source for precipitating calcite-type CaCO3. Concurrently, Tyr2− is progressively protonated to Tyr− and finally to the poorly soluble Tyr0, which co-precipitates with CaCO3. This spontaneous separation, driven by the intrinsic pH swing of the carbonation reaction, eliminates the need for external acid addition or energy-intensive thermal regeneration required in processes using additives such as Asp.
3.3. Cycling performance of Tyr over consecutive leaching–carbonation processes
To evaluate the recyclability of Tyr, five consecutive leaching–carbonation cycles were conducted under the optimized condition. As illustrated in Fig. 11, both the carbonation efficiency and Tyr recovery efficiency remained highly stable throughout the cycling tests, averaging 93.41 ± 1.85% and 92.73 ± 0.52%, respectively. In contrast, the Ca2+ leaching yield decreased markedly from 16.12 ± 0.30 g L−1 in the first cycle to 12.63 ± 0.56 g L−1 in the second cycle, representing a reduction of approximately 23%, and then stabilized at an average of 12.46 ± 0.43 g L−1 in the following cycles. A similar initial decline in leaching yield has been reported in glycine-mediated indirect carbonation of fly ash.38 This reduction may be attributed to the reaction of amino groups with CO2, followed by incomplete regeneration of the resulting carbamate intermediates during carbonation. Additionally, SO42− ions adsorb onto the co-precipitates of Tyr and CaCO3 during carbonation. These anions are carried into the next leaching cycle with the alkaline wash solution, where the introduced SO42− exerts a common ion effect that suppresses CaSO4·2H2O dissolution. Importantly, despite this decrease in leaching yield, the Tyr recovery efficiency remained stable throughout the cycles. The effective separation and recovery of Tyr in each cycle is key to preventing the accumulation of SO42− inhibition, which fundamentally distinguishes this process from those that directly recycle the inhibitor-containing mother liquor (e.g., NH4Cl or NaCl systems) and enables stable long-term operation.4. Conclusions
This study demonstrates a strategy for the indirect aqueous carbonation of CaSO4·2H2O using Tyr as an efficient and recyclable leaching additive. The approach is based on utilizing the inherent pH decrease during carbonation to spontaneously precipitate and separate Tyr from the SO42−-rich carbonation mother liquor, offering a potential pathway to address the challenge of additive recycling in gypsum carbonation. In a strongly alkaline medium, fully deprotonated Tyr chelates with Ca2+, increasing CaSO4·2H2O solubility by sixfold to 16.09 ± 0.30 g L−1 under optimized leaching conditions. During carbonation, the Ca2+–Tyr chelates dissociate as the pH drops, releasing free Ca2+ for the direct formation of uniform calcite, while neutral Tyr0 co-precipitates from the SO42-rich carbonation mother liquor. Under the optimized carbonation conditions, the carbonation and Tyr recovery efficiencies exceeded 97% and 95%, respectively. Furthermore, five consecutive leaching–carbonation cycles confirmed the stability of the process.Future research should focus on the influence of soluble impurities in industrial solid waste gypsum on CaCO3 polymorphism and product purity, alongside related process conditions. The underlying cause of the significant leaching yield decline during the initial cycle also requires in-depth clarification. Furthermore, integrating the process with bipolar membrane electrodialysis to recover alkaline media from the spent liquor remains a crucial direction for improving the economic feasibility of the technology.
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
All data generated or analysed during this study are included in this published article.Supplementary information (SI): Tables S1–S4 (Box–Behnken design factors, ANOVA of the calcium leaching model, dissociation constants and isoelectric point of Tyr, and lognormal distribution fit parameters for particle size distribution); Fig. S1 (speciation diagram of Tyr as a function of pH); and eqn (S1)–(S2) (polynomial regression equation and lognormal distribution function). See DOI: https://doi.org/10.1039/d5ra09358a.
Acknowledgements
This research was supported by Gansu Key Research and Development Program (Industrial Category) under Grant No. 24YFGA033.References
- M. H. Soomro, U. R. Abdullah, W. A. Wan Ab Karim Ghani and F. Mohd Kusin, J. Environ. Chem. Eng., 2025, 13, 118696 CrossRef CAS.
- X. Lin, X. Li, H. Liu, G. Boczkaj, Y. Cao and C. Wang, Sep. Purif. Technol., 2024, 338, 126558 CrossRef CAS.
- X. Lin, Y. Zhang, H. Liu, G. Boczkaj, Y. Cao and C. Wang, J. Clean. Prod., 2024, 434, 140258 CrossRef CAS.
- B. Wang, Z. Pan, H. Cheng, Z. Zhang and F. Cheng, J. Clean. Prod., 2021, 302, 126930 CrossRef CAS.
- Y. Liu, Y. Wang, T. Yin, C. Lin, H. Tan and H. Hariana, J. Cryst. Growth, 2025, 667, 128273 CrossRef CAS.
- Y. Su, B. Li, W. Zhou, W. Jie, Y. Li, H. Zhang and H. Ni, Chem. Eng. Sci., 2024, 292, 119967 CrossRef CAS.
- K. Ramasenya, B. Oladipo, V. N. Katambwe, V. R. K. Vadapalli, J. Petersen, S. Gcasamba, H. Coetzee, A. Horvat, H.-J. Ho, A. Iizuka, L. F. Petrik and T. V. Ojumu, J. Environ. Chem. Eng., 2025, 13, 117754 CrossRef CAS.
- L. Liu, M. Gan, X. Fan, Z. Sun, J. Wei, J. Li and Z. Ji, J. Environ. Chem. Eng., 2023, 11, 110655 CrossRef CAS.
- K. Ramasenya, B. Oladipo, V. N. Katambwe, V. R. K. Vadapalli, J. Petersen, S. Gcasamba, H. Coetzee, A. Horvat, H. J. Ho, A. Iizuka, L. F. Petrik and T. V. Ojumu, J. Environ. Chem. Eng., 2025, 13, 117754 CrossRef CAS.
- O. Rahmani, M. Tyrer and R. Junin, RSC Adv., 2014, 4, 45548–45557 RSC.
- O. Rahmani, J. CO2 Util., 2018, 27, 374–380 CrossRef CAS.
- Q. Chen, W. Ding, H. Sun, T. Peng and G. Ma, ACS Sustainable Chem. Eng., 2020, 8, 11649–11657 CrossRef CAS.
- W. Ding, J. Qiao, L. Zeng, H. Sun and T. Peng, Int. J. Greenhouse Gas Control, 2023, 123, 103843 CrossRef CAS.
- W. Ding, Q. Chen, H. Sun and T. Peng, J. CO2 Util., 2019, 34, 507–515 CrossRef CAS.
- Q. Chen, W. Ding, H. Sun, T. Peng and G. Ma, Energy, 2020, 206, 118148 CrossRef CAS.
- Y. Gong, X. Zhu, Z. Yang, X. Zhang and C. Li, RSC Adv., 2022, 12, 26556–26564 RSC.
- X. Zheng, J. Liu, Y. Wei, K. Li, H. Yu, X. Wang, L. Ji and S. Yan, Chem. Eng. J., 2022, 440, 135900 CrossRef CAS.
- D. Madhav, D. Hautekeete, W. Thielemans, F. Desplentere, P. Moldenaers and V. Vandeginste, Chem. Eng. J., 2025, 520, 166002 CrossRef CAS.
- C. Wang, H. Jiang, Y. Wang, T. Zhang, Z. Xiong, Y. Zhao and J. Zhang, Sep. Purif. Technol., 2025, 358, 130330 CrossRef CAS.
- Y. Li, X. Duan, W. Song, L. Ma and J. Jow, Chem. Eng. J., 2021, 405, 126938 CrossRef CAS.
- L. Ji, H. Yu, K. Li, B. Yu, M. Grigore, Q. Yang, X. Wang, Z. Chen, M. Zeng and S. Zhao, Appl. Energy, 2018, 225, 356–366 CrossRef CAS.
- M. Liu and G. Gadikota, Fuel, 2020, 275, 117887 CrossRef CAS.
- S. M. Pérez-Moreno, M. J. Gázquez and J. P. Bolívar, Chem. Eng. J., 2015, 262, 737–746 CrossRef.
- X. Yu, Handbook of MSG Industry, China Light Industry Press, 2nd edn, 2009 Search PubMed.
- Z. Chen, B. Shen, Y. Zhang, H. Guo, Z. Chen, R. Wei and J. Hu, Adv. Powder Technol., 2024, 35, 104362 CrossRef CAS.
- J. Luo, F. Kong and X. Ma, Cryst. Growth Des., 2016, 16, 728–736 CrossRef CAS.
- Y. Sun, V. Parikh and L. Zhang, J. Hazard. Mater., 2012, 209–210, 458–466 CrossRef CAS PubMed.
- Q. He, Y. Cong, M. Zheng, A. Farajtabar and H. Zhao, J. Chem. Thermodyn., 2018, 124, 123–132 CrossRef CAS.
- L. Ji, X. Zheng, Y. Ren, Y. Wang, Y. Wang and S. Yan, Sep. Purif. Technol., 2024, 329, 125171 CrossRef CAS.
- M. M. Reddy and G. H. Nancollas, J. Colloid Interface Sci., 1971, 36, 166–172 CrossRef CAS.
- J. Christoffersen and M. R. Christoffersen, J. Cryst. Growth, 1990, 100, 203–211 CrossRef CAS.
- Y. Gong, X. Zhu, Z. Yang, X. Zhang and C. Li, RSC Adv., 2022, 12, 26556–26564 RSC.
- V. Sang Sefidi and P. Luis, Ind. Eng. Chem. Res., 2019, 58, 20181–20194 CrossRef CAS.
- Y. Gong, L. Wu, J. Li and J. Zhu, J. Cryst. Growth, 2019, 522, 128–138 CrossRef CAS.
- M. M. Longuinho, V. Ramnarain, N. O. Peña, D. Ihiawakrim, R. Soria-Martínez, M. Farina, O. Ersen and A. L. Rossi, CrystEngComm, 2022, 24, 2602–2614 RSC.
- Y.-P. Ho, M.-W. Yang, L.-T. Chen and Y.-C. Yang, Rapid Commun. Mass Spectrom., 2007, 21, 1083–1089 CrossRef CAS PubMed.
- S.-Y. Pan, E.-E. Chang, H. Kim, Y.-H. Chen and P.-C. Chiang, J. Hazard. Mater., 2016, 307, 253–262 CrossRef CAS PubMed.
- X. Zheng, J. Liu, Y. Wang, Y. Wang, L. Ji and S. Yan, Chem. Eng. J., 2023, 459, 141536 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |







