Open Access Article
Open Access ArticleEffective extraction and determination of 9 β-lactams in water and milk samples by a novel magnetic covalent organic framework combined with UPLC-MS/MS
Lu Liab,
Na Lia,
Wendan Niuab,
Aijing Guoab,
Ling Ma*ab,
Wei Zhao*a,
Yawei Zhang*a and
Ke Wang *ab
*ab
aShijiazhuang Center for Disease Control and Prevention, Shijiazhuang 050011, China. E-mail: mamalin001@163.com; 13933887009@139.com; 2211291167@qq.com
bShijiazhuang Technology Innovation Center for Chemical Poison Detection and Risk Early Warning, Shijiazhuang 050011, China. E-mail: wkecdc@163.com
First published on 8th April 2026
Abstract
β-Lactams (β-LAs) are a class of widely used antibiotics. However, their residues in food may pose a threat to human health. Therefore, it is of great necessity to develop a method that can sensitively and effectively detect β-LAs at trace levels in complex matrices. In this study, a novel functionalized magnetic covalent organic framework was synthesized based on biphenyl-3,3′,5,5′-tetracarbaldehyde and p-phenylenediamine. This material was used as an adsorbent for the effective extraction of 9 β-LAs in water and milk samples, which were analyzed by ultra-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS). The experimental parameters were optimized, then the sensitive and accurate detection method was developed with excellent linearity (R2 ≥ 0.9990), low limits of detection (0.001–0.347 µg L−1) and limits of quantitation (0.002–1.051 µg L−1) for β-LAs. Meanwhile, good recoveries (71.7–118.8%) of 9 β-LAs in water and milk samples were obtained with the relative standard deviations (RSD) <10% (n = 5). In conclusion, Fe3O4@SiO2@Ap-COF can be regarded as an effective magnetic adsorbent, and the established method holds significant promise for rapid detection of 9 β-LAs in water and milk samples.
1 Introduction
β-Lactam antibiotics (β-LAs) constitute a class of important antibacterial drugs containing β-lactam rings, mainly including penicillins and cephalosporins.1 In recent years, β-LAs have found widespread use for the prevention or treatment of bacterial infections, accounting for roughly two-thirds of commonly administered antibiotics owing to their broad-spectrum activity, low toxicity, and cost-effectiveness.2However, when animals ingest β-lactam antibiotics, these drugs and their metabolites accumulate in animal organs and tissues, this retention could pose risks to public health.3,4 On the one hand, β-LAs can lead to water pollution through the excretion of animal feces and the discharge of wastewater from livestock farms and pharmaceutical factories.5 On the other hand, unmetabolized antibiotics in the animal's body can migrate into animal derived foods, such as milk, eggs, fish and meat, posing health risks to consumers. Milk is one of the most consumed foods owing to its rich content of essential nutrients. However, cephalosporins are widely used as feed additives in veterinary medicine, which may compromise the quality of milk.6 Therefore, regulatory authorities and institutions including the European Commission, China, and the United States have established maximum residue limits (MRLs) for β-LAs in foods of animal origin to safeguard consumer safety. In view of this, it is crucial to develop sensitive, reliable and simple analytical methods for the monitor of β-LAs in water and milk samples to ensure public health security.
Current methods for β-LAs analysis are based on capillary electrophoresis (CE),7 enzyme-linked immunosorbent assay,8 liquid chromatography (LC),9 chemiluminescence methods,10 and liquid chromatography-tandem mass spectrometry (LC-MS/MS).11 Among these methods, LC-MS/MS has been well adopted owing to its high sensitivity, high specificity, high-throughout qualitative and quantitative analysis. Due to the complex matrix in the actual sample, which contains many potential interference components, simple and effective sample pretreatment is critical before analysis and detection. Commonly used sample pretreatment methods include liquid–liquid extraction (LLE),12 molecular imprinting technology,13 pressurized liquid extraction,14 matrix solid-phase dispersion (MSPD),1 solid phase extraction (SPE),15,16 and QuECHERS.17,18
Different from the traditional sample pretreatment methods, magnetic solid-phase extraction (MSPE) is an advanced technique based on dispersed solid-phase extraction, which uses a magnetic adsorbent for the separation and enrichment of analytes via an applied magnetic field. The magnetic adsorbent is a magnetic nanocomposite with a core–shell structure, which is formed by combining a magnetic nanoparticle (MNPs) core with a non-magnetic adsorptive material. Since MSPE has the characteristics of an easy, green, environmental-friendly and effective extraction approach, it has received great attention for complex sample preparation in recent years.19 Among them, Fe3O4 is the most common MNPs because of advantages of high magnetic moments, good biocompatibility, simple preparation methods, and low production costs.20 Various materials including carbon nanotubes (CNTs), graphene,21,22 molecularly imprinted polymers (MIPs),23 and metal–organic frameworks (MOFs),24,25 have been explored as MSPE adsorbent materials. Covalent organic frameworks (COFs) are a new type of porous polymers, which have attracted extensive attention due to their excellent properties of large specific surface area, unique topological structure, high thermochemical stability and functional modification.26–28 COFs exhibit a high affinity for target compounds and can extract a wide range of pollutants, including pesticides, polycyclic aromatic hydrocarbons (PAHs), antibiotics, heavy metal ions, illegal additives, hormones, and biotoxins.29–33 Compared with MIPs, COFs possess a higher specific surface area, porosity, mass transfer limitations, and adsorption capacity.34,35 Besides, compared with MOFs, COFs have been proved to be more stable in water and acidic media.36,37 However, since the sizes of COFs are typically in the micro- and nano-scale, it is difficult to precipitate COFs with low density from complex matrix.38
Fascinatingly, the combination of COFs and Fe3O4 effectively makes up for this limitation, not only possess high adsorption capacity, but also facilitate separation.33,39
In recent years, a large number of literature have reported the application of magnetic materials in the separation of macromolecular substances,40 organic compounds41 and metal ion enrichment.42 Some studies have shown that magnetic materials have great potential in extracting antibiotics from water samples, such as the detection of tetracycline drugs,43 the extraction of sulfonamides,44 but there are few applications in the detection of β-LAs. Most reported MSPE methods employ molecularly imprinted polymers (MIPs) as the magnetic adsorbent,13,45,46 In contrast, the use of magnetic covalent organic frameworks (COFs) is rare. Furthermore, these methods are often limited to a single sample matrix and cover only a small number of target analytes.
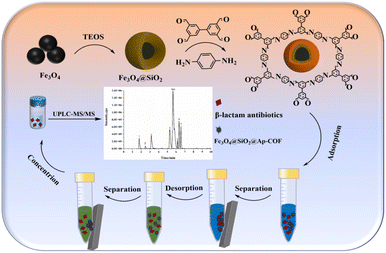
In the present study, a novel adsorption material was synthesized by using silanized Fe3O4 as the core of the magnetic material and COFs formed by the combination of 3,3′,5,5′-tetraldehyde-biphenyl and p-phenylenediamine as the shell of the magnetic material. The obtained Fe3O4@SiO2@Ap-COF was used as an MSPE adsorbent and combined with UPLC-MS/MS for the enrichment of 9 kinds of β-LAs, demonstrating excellent adsorption performance. Subsequently, the main parameters influencing the MSPE procedure were optimized and the developed method was validated.
2 Experimental
2.1 Reagents and materials
Ferric chloride hexahydrate (FeCl3·6H2O), ethylene glycol (EG), sodium acetate anhydrous (NaAc), 1,4-dioxane, aodium chloride (NaCl) and anhydrous ethanol were obtained from Tianjin Yong da Chemical Reagent Ltd (Tianjin, China). Biphenyl-3,3′,5,5′-tetracarbaldehyde and p-phenylenediamine were purchased from Shanghai Macklin Biochemical Ltd (Shanghai, China). Tetraethoxysilane (TEOS) was sourced from Aladdin Chemistry Ltd (Shanghai, China). Polyethylene glycol (PEG-4000) was supplied by Xiya Chemical Technology Ltd (Shandong, China). 37% (w/w) hydrochloric acid were acquired from Tianjin Kernel Chemical Reagent Ltd (Tianjin, China). Acetonitrile (ACN) was purchased from Sigma-Aldrich (St. Louis, MO, USA), methanol (MA), hexane and ethyl acetate were procured from Thermo Fisher Ltd (Waltham, USA). Formic acid (FA) was purchased from Dikma (Lake Forest, USA). Superior pure dichloro-methane was purchased from Tianjin Kemiou Chemical Reagent (China). Formic acid, acetonitrile and methanol were HPLC grade, whereas the remaining reagents were analytical grade. The ultrapure water was manufactured using a Milli-Q purification system (Millipore, Bedford, USA).Cefpiramide acid (CRA), cefoperazone (CFP), ceftiofur (EFT), cefpodoxime proxetil (CPD), cefetamet pivoxil hydrochloride (CETP), dicloxacillin sodium (DIC), flucloxacillin (FLU), cloxacillin (CLO), and oxacillin sodium (OXA) were obtained from Alta Scientific Co., Ltd (Tianjin, China). The properties of β-LAs of all standards were shown in Table S1.
2.2 Instrumental and analytical conditions
The UPLC-MS/MS instrument (Exion TRILPLE QUAD 5500, AB SCIEX, USA) was used to determine analytes. Chromatographic separation was performed on an Agilent ZORBAX SB-Aq column (2.1 × 150 mm, 3.5 µm). The injection volume was 5 µL and a constant flow rate of 0.5 mL min−1 was maintained. Mobile phase A was 0.1% formic acid aqueous solution, while mobile phase B was ACN. The gradient elution program was set as follows: (1) 0–1.0 min, 30% B (v/v); (2) 1.1–4.0 min, 40% B (v/v); (3) 4.1–6.0 min 70% B (v/v); (4) 6.1–7.0 min 85% B (v/v); (5) 7.1–10.0 min 30%, B (v/v). The column temperature was set at 40 °C. Mass spectrometric analysis was conducted in multiple reaction monitoring (MRM) mode using an electrospray ionization source (ESI) in the positive ion mode. The mass spectrometry parameters have been configured, including spray voltage (5500 V), vaporizer temperature (550 °C), nebulizing gas pressure (55 psi), auxiliary gas pressure (55 psi), and curtain gas pressure (40 psi). In addition, the other parameters including retention times, precursor ion and product ion mass, declustering potential (DP) and collision energy (CE) were listed in Table S2. Besides, the chromatograms of 9 β-LAs were listed in Fig. S1.Fourier infrared transform (FT-IR) spectras were obtained via a Nicole iS5 spectrometer (Thermo Nicolet Co. Ltd, USA). The Brunauer–Emmett–Teller (BET) surface areas were measured by Quadrasorb EVO (Quantachrome Instruments, USA). The X-ray diffraction (XRD) graphs were acquired using a German Bruker D8 (Bruker, Germany). The magnetizations of the materials were determined by a SQUID XL-7 vibrating sample magnetometer (Quantum Design, USA). Morphology of the materials were observed using a Regulus 810 scanning electron microscopy (SEM, Hitachi, Japan) and a FEI Tecnai G2 F20 transmission electron microscope (TEM, FEI, USA).
2.3 Preparation of magnetic adsorbents
2.4 Collection and sample preparation
20 water samples were collected from different rivers during autumn in Shijiazhuang City, China, which were filtered by 0.45 µm filter membrane and stored 4 °C. Before MSPE, the sample solution pH was adjusted to 3.0 with dilute hydrochloric acid.15 milk samples (10 whole milk samples and 5 skimmed milk samples) were purchased from local supermarkets during autumn in Chang'an District, Shijiazhuang City, China. Before MSPE, 1.0 g of the homogenized sample was transferred to a 50 mL centrifuge tube, then 12 mL acetonitrile–water (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) was added and vortexed for 3 min to extract β-LAs. After centrifuged for 5 min at 4 °C under 10
1, v/v) was added and vortexed for 3 min to extract β-LAs. After centrifuged for 5 min at 4 °C under 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, 6 mL aliquot supernatant was added into a 15 mL centrifuge tube, and dried under nitrogen at 40 °C. Subsequently, the dried residue was dissolved in 10 mL of ultra-pure water, and the pH was adjusted to 3.0 with dilute hydrochloric acid before MSPE.
000 rpm, 6 mL aliquot supernatant was added into a 15 mL centrifuge tube, and dried under nitrogen at 40 °C. Subsequently, the dried residue was dissolved in 10 mL of ultra-pure water, and the pH was adjusted to 3.0 with dilute hydrochloric acid before MSPE.
2.5 Procedure of MSPE
The extraction procedure is depicted in Scheme 1. Specifically, 10 mL of sample solution and 8 mg of Fe3O4@SiO2@Ap-COF were added to a 50 mL centrifuge tube. The mixture was subjected to 15 min of intense vortexing for adsorption, followed by separating the material from the solution using a magnet. 8 mL 0.3% (v/v) acidified methanol was added to desorb the β-LAs from Fe3O4@SiO2@Ap-COF by vortex for 7 min, and the eluent obtained was filtered through a 0.20 µm filter for HPLC-MS/MS analysis. In addition, the magnetic material Fe3O4@SiO2@Ap-COF was vibrately washed several times with 0.3% acidified methanol and recycled after vacuum drying at 60 °C.3 Results and discussions
3.1 Characterization of the Fe3O4@SiO2@Ap-COF
The SEM and TEM images revealed the structural characteristics of Fe3O4 nanoparticles and Fe3O4@SiO2@Ap-COF nanoparticles. As shown in Fig. 1A and B, it can be seen that Fe3O4 had a smooth and well-dispersed surface, while Fe3O4@SiO2@Ap-COF had a rough surface. The typical core–shell structure, comprising a Fe3O4 nanoparticle core and a gray COF layer, can be clearly observed in the TEM images presented in Fig. 1C and D. Besides, based on SEM, the average particle diameter of Fe3O4 was approximately 55 nm, while that of Fe3O4@SiO2@Ap-COF was about 66 nm, which showed the formation of a coating shell with thickness of approximately 11 nm, further approving the successful coating of COF.To further prove the successful synthesis of Fe3O4@SiO2@Ap-COF, the crystalline structures and characteristic spectrum of Fe3O4, Fe3O4@SiO2 and Fe3O4@SiO2@Ap-COF were determined through XRD and FT-IR.
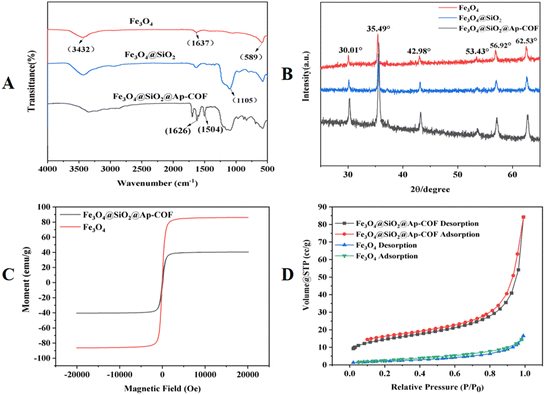
The scanning FTIR images of the Fe3O4@SiO2@Ap-COF revealed that synthesis process of Fe3O4@SiO2@Ap-COF. As depicted in Fig. 2A, the typical band at 587 cm−1 was assigned to the Fe–O–Fe vibration. The broad bands of –OH at 3432 cm−1 and 1637 cm−1 could be classified as the stretching and bending vibrations of water molecules on the surface of Fe3O4. The peaks around 1105 cm−1 were induced by the Si–O bending vibration, indicating that the silica shells were successfully coated on the surface of Fe3O4. Furthermore, the emerging stretching at 1504 cm−1 and 1626 cm−1 was caused by the benzene skeleton of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching and the C
C stretching and the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching vibrations,51 which show the successful coating of COFs on the surface of Fe3O4@SiO2 by Schiff-base condensation reaction. The FT-IR results confirmed the successful fabrication of Fe3O4@ SiO2@Ap-COF.
N stretching vibrations,51 which show the successful coating of COFs on the surface of Fe3O4@SiO2 by Schiff-base condensation reaction. The FT-IR results confirmed the successful fabrication of Fe3O4@ SiO2@Ap-COF.
As seen in Fig. 2B, all the synthesized MNPs had the same peaks at wide angles (2θ = 30.01°, 35.49°, 42.98°, 53.43°, 56.92°, and 62.53°), Corresponding to (220), (311), (400), (422), (511) and (440) in the standard magnetite XRD pattern (JCPDS 19-0629).39 The results show that Fe3O4@SiO2@Ap-COF has good crystallization and high crystallinity after coating, and the crystalline phase of Fe3O4 was well maintained during the synthesis of the Fe3O4@SiO2 and Fe3O4@SiO2@Ap-COF. The VSM was used to further validate the magnetic behaviors of Fe3O4 and Fe3O4@SiO2@Ap-COF. The magnetization of the material was the key factor in the rapid separation of the adsorbent from the sample solution. As depicted in Fig. 2C, the saturation magnetization values of the Fe3O4 and Fe3O4@SiO2@Ap-COF were 79.12 and 40.43 emu g−1, respectively. Compared with Fe3O4, the Fe3O4@SiO2@Ap-COF showed a significant decrease in saturation magnetization, which could be due to the shielding effect of the coated layer on the magnetization of the Fe3O4, further confirming the successful coating of the COF material. However, under an external magnetic field, the magnetization of Fe3O4@SiO2@Ap-COF can still ensure the rapid separation of it from liquid samples within 1 min.
The porosity and surface area was investigated via N2 adsorption–desorption analysis at 77 K. As presented in Fig. 2D, Fe3O4 is a type II adsorption isotherm,52 and Fe3O4@SiO2@Ap-COF shows a type IV adsorption isotherm. After calculation, the BET surface area of Fe3O4@SiO2@Ap-COF was 52.32 m2 g−1, which was higher than that of the Fe3O4 with a surface area of 7.89 m2 g−1. A high specific surface area facilitated contact between the material and analyte, thereby providing more available adsorption sites and consequently enhancing adsorption performance of material toward β-LAs. Compared with Fe3O4, Fe3O4@SiO2@Ap-COF possesses a higher specific surface area, which gives it an advantage in adsorbing β-LAs. All these results demonstrated that the Fe3O4@SiO2@Ap-COF was suitable for acting as an adsorbent for β-LAs with high adsorption efficiency.
3.2 Optimization of MSPE condition
To achieve high extraction efficiency, some influencing factors such as adsorbent dosage, sample solution pH, salt concentration, extraction time, type and volume of the desorption solvent, and desorption time were optimized in detail. Such optimization experiments were performed using single factor approach.The eluent is an important factor to obtain good desorption efficiency. Therefore, the elution effect of MA and ACN was first investigated. As seen in Fig. 4A, the elution effect of MA is significantly better than that of ACN. Additionally, the addition of an appropriate amount of acidic solution facilitates the separation of analytes. Therefore, the effect of methanol acidification was further investigated. When the concentration of formic acid was 0.3% (v/v), the highest overall elution efficiency for the nine analytes was achieved, with recoveries ranging from 70.3% to 95.8%. However, the elution efficiency gradually decreased as the formic acid concentration in the methanol increased. We speculate that this decline is because that the chemical structure of β-LAs may easily destroyed under acidic environment.57 Therefore, 0.3% (v/v) acidified MA was selected for elution.
Notably, the volume of the eluent influenced the recoveries of the analytes. In this study, different eluent volumes (4 mL, 6 mL, 8 mL, 10 mL, 12 mL) were investigated. As presented in Fig. 4B, when eluent volume was 8 mL, the recoveries of 9 β-LAs were optimal, in the range of 87.9–110.8%.
Additionally, when the elution volume was fixed, the effects of different elution time on β-LAs were further explored from 3 to 9 min. Fig. 4C showed that when the vortex time was 7 min, the recoveries of 9 β-LAs reached the best level (79.2–109.8%), and the recoveries did not significantly alter with the increase of the vortex time. Therefore, the elution time of 7 min was selected in this study.
3.3 Adsorption mechanism
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N group and benzene ring of the Fe3O4@SiO2@Ap-COF could form the large π-conjugation system, which was prone to induce π–π interaction with β-LAs containing carbonyl group and benzene ring.58
N group and benzene ring of the Fe3O4@SiO2@Ap-COF could form the large π-conjugation system, which was prone to induce π–π interaction with β-LAs containing carbonyl group and benzene ring.58
Furthermore, Fe3O4@SiO2@Ap-COF exhibited optimal adsorption performance for β-LAs at pH 3. This could be attributed to the fact that pKa values of most β-LAs were close to 3, then β-LAs predominantly existed in original state at pH 3. Correspondingly, the abundant N atoms of the Fe3O4@SiO2@Ap-COF could interact with carboxyl group, hydroxyl group or amino group of β-LAs via hydrogen bond, thereby achieving efficient adsorption.
Hydrophilicity and hydrophobicity are related to the log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P value59,60 (the higher log
P value59,60 (the higher log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P value, the better hydrophobicity). The log
P value, the better hydrophobicity). The log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P values of most β-LAs were above 1, indicating their good hydrophobicity. Meanwhile, the Fe3O4@SiO2@Ap-COF contained abundant benzene rings, increasing the hydrophobicity. Therefore, hydrophobic interaction was also an important factor for the effective adsorption of β-LAs by Fe3O4@SiO2@Ap-COF. In conclusion, the dominant adsorption mechanism of Fe3O4@SiO2@Ap-COF with β-LAs involved π–π interaction, hydrogen bonding and hydrophobic interactions.
P values of most β-LAs were above 1, indicating their good hydrophobicity. Meanwhile, the Fe3O4@SiO2@Ap-COF contained abundant benzene rings, increasing the hydrophobicity. Therefore, hydrophobic interaction was also an important factor for the effective adsorption of β-LAs by Fe3O4@SiO2@Ap-COF. In conclusion, the dominant adsorption mechanism of Fe3O4@SiO2@Ap-COF with β-LAs involved π–π interaction, hydrogen bonding and hydrophobic interactions.
To further investigate the adsorption selectivity of Fe3O4@SiO2@Ap-COF for β-LAs, the other two categories of antibiotics including nitroimidazoles (NMZs) and tetracyclines (TCs) were also tested. As seen in Table S4, Fe3O4@SiO2@Ap-COF had low adsorption efficiency (<30%) for NMZs and TCs, exhibiting good adsorption selectivity towards β-LAs.
3.4 Reusability of the adsorbent
For MSPE adsorbents, it is very important to evaluate their reusability. Through reusing multiple times, the good adsorption performance is a key indicator of evaluating material's reusability.18 In this experiment, Fe3O4@SiO2@Ap-COF containing β-LAs was washed with 8 mL, 0.3% acidified methanol for several times, dried in vacuum at 60 °C for 4 h, and then several continuous adsorption–desorption tests were carried out to investigate the reusability of the adsorbent. As seen in Fig. 5, Fe3O4@SiO2@Ap-COF could be reused for 4 times, and the recoveries of 9 β-LAs were all above 65%. The decline in adsorption efficiency after repeated use can be due to fouling from the analyzed sample. The results showed that Fe3O4@SiO2@Ap-COF has good reusability.3.5 Matrix effect
Matrix effect (ME) refers to influence of other co-extracted components in the sample matrix on the measurement of the target analyte, which can cause signal suppression or enhancement during UPLC-MS/MS analysis.61 In this study, ME were assessed by the ratio of slopes between the matrix-matched calibration curves and the solvent calibration curves. ME could be ignored if the ratio was closer to 80–120%. The formula of ME is listed below:The blank matrix solutions of surface water and milk blank matrix solutions without the target substances were obtained according to the sample pretreatment and MSPE steps, and the ME of 9 β-LAs were assessed at different concentrations. According to the calculation results (Table 1), the range of the ME of water and milk were respectively 85.3–118.2% and 98.2–114.9%, demonstrating that the tested analytes showed no obvious matrix effect. Therefore, to the sensitive and accurate determination of water and milk samples, we chose an external standard method using solvent standard curve for quantitative analysis in this experiment.
| Analytes | ME (%) | |
|---|---|---|
| Water | Milk | |
| CRA | 85.3 | 98.2 |
| CFP | 94.5 | 110.7 |
| EFT | 100.5 | 114.7 |
| CPD | 94.7 | 109.0 |
| CETP | 99.9 | 105.8 |
| DIC | 118.2 | 114.9 |
| CLO | 113.8 | 112.4 |
| OXA | 115.5 | 114.8 |
| FLU | 115.8 | 110.7 |
3.6 Method validation
| Analytes | Linear range (µg L−1) | Calibration curve | R2 | LOD | LOQ | ||
|---|---|---|---|---|---|---|---|
| Water (µg L−1) | Milk (µg kg−1) | Water (µg L−1) | Milk (µg kg−1) | ||||
| CRA | 0.2–200 | Y = 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 007.8X + 2205.6 007.8X + 2205.6 |
0.9998 | 0.001 | 0.005 | 0.004 | 0.015 |
| CFP | 0.2–200 | Y = 3005.7X + 858.1 | 0.9997 | 0.013 | 0.018 | 0.041 | 0.056 |
| EFT | 0.2–200 | Y = 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 390.2X + 790.1 390.2X + 790.1 |
0.9998 | 0.003 | 0.004 | 0.009 | 0.011 |
| CPD | 0.2–200 | Y = 918![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 54X + 17 54X + 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 470.8 470.8 |
0.9997 | 0.001 | 0.002 | 0.002 | 0.006 |
| CETP | 0.2–200 | Y = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 432 432![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 26X + 36 26X + 36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 026.3 026.3 |
0.9995 | 0.001 | 0.003 | 0.003 | 0.008 |
| DIC | 0.2–200 | Y = 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 412.7X + 7253.3 412.7X + 7253.3 |
0.9994 | 0.002 | 0.006 | 0.007 | 0.018 |
| FLU | 0.2–200 | Y = 28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 092.4X + 1298.7 092.4X + 1298.7 |
0.9996 | 0.007 | 0.006 | 0.020 | 0.019 |
| CLO | 0.2–200 | Y = 378.5X + 32.4 | 0.9999 | 0.102 | 0.347 | 0.310 | 1.051 |
| OXA | 0.2–100 | Y = 27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 891.2X + 687.9 891.2X + 687.9 |
0.9993 | 0.001 | 0.005 | 0.003 | 0.016 |
3.7 Method application in real samples
The method was applied to analyze 15 milk samples and 20 water samples. As a result, none of the target 9 β-LAs were detected.4 Conclusions
In summary, a new core–shell magnetic material Fe3O4@SiO2@Ap-COF was synthesized, which exhibited good adsorption efficiency and selectivity for 9 β-LAs. Using this material as an MSPE adsorbent, a new method for determining 9 β-LAs in water and milk were developed. The method showed that the LODs were 0.001 µg L−1–0.102 µg L−1 (water) and 0.002 µg kg−1–0.347 µg kg−1 (milk), and the LOQs were 0.002 µg L−1–0.310 µg L−1 (water) and 0.006 µg kg−1–1.051 µg kg−1 (milk), respectively, with RSDs less than 9.46%, indicating high sensitivity, accuracy and precision, thus enabling the rapid, high-throughput determination of 9 β-LAs in water and milk.Author contributions
Lu Li: writing – original draft, conceptualization, formal analysis, methodology. Na Li: data curation, formal analysis, methodology, writing – original draft. Wendan Niu: data curation, validation, investigation. Aijing Guo: methodology, validation. Ling Ma: conceptualization, supervision, funding acquisition. Wei Zhao: conceptualization, supervision. Yawei Zhang: conceptualization, supervision. Ke Wang: conceptualization, supervision, writing – review & editing, project administration, funding acquisition, resources. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting the findings of this study are available within the article and its supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ra09307d.Acknowledgements
This work was supported by the National Natural Science Foundation of China (81903322), S&T Program of Hebei (223777116D) and the funding for the Fourth Batch of Discipline Leading Talents in Shijiazhuang City.References
- Z. Huang, X.-D. Pan, B. Huang, J.-J. Xu, M.-L. Wang and Y.-P. Ren, Food Control, 2016, 66, 145–150 CrossRef CAS.
- V. G. Alekseev, Pharm. Chem. J., 2010, 44, 14–24 CrossRef CAS.
- M. Cycoń, A. Mrozik and Z. Piotrowska-Seget, Front. Microbiol., 2019, 10, 338 CrossRef PubMed.
- Y. M. Mesfin, B. A. Mitiku and H. Tamrat Admasu, Vet. Med. Sci., 2024, 10, e70049 CrossRef PubMed.
- Y. Yang, Y. S. Ok, K.-H. Kim, E. E. Kwon and Y. F. Tsang, Sci. Total Environ., 2017, 596–597, 303–320 CrossRef CAS PubMed.
- V. Samanidou and S. Nisyriou, J. Sep. Sci., 2008, 31, 2068–2090 CrossRef CAS PubMed.
- T. A. H. Nguyen, T. N. M. Pham, T. B. Le, D. C. Le, T. T. P. Tran, T. Q. H. Nguyen, T. K. T. Nguyen, P. C. Hauser and T. D. Mai, J. Chromatogr. A, 2019, 1605, 360356 CrossRef CAS PubMed.
- S. Ahmed, J. Ning, G. Cheng, M. K. Maan, T. Chen, I. Ahmad, S. A. Algharib and Z. Yuan, Microchem. J., 2020, 152, 104354 CrossRef CAS.
- M. Cámara, A. Gallego-Picó, R. M. Garcinuño, P. Fernández-Hernando, J. S. Durand-Alegría and P. J. Sánchez, Food Chem., 2013, 141, 829–834 CrossRef PubMed.
- M. Iranifam and M. Khabbaz Kharameh, Luminescence, 2015, 30, 625–630 CrossRef CAS PubMed.
- Q. Yang, Y. Liu, Z. Jiang, M. Xu, S. Yao, C. Li, G. Chen, M. Xu, W. Liu, L. Yin and Z. Hu, Anal. Biochem., 2021, 631, 114299 CrossRef CAS PubMed.
- J. Li, X. Ren, Y. Diao, Y. Chen, Q. Wang, W. Jin, P. Zhou, Q. Fan, Y. Zhang and H. Liu, Food Chem., 2018, 257, 259–264 CrossRef CAS PubMed.
- Y. Wang, X. Ma, Y. Peng, Y. Liu and H. Zhang, J. Hazard. Mater., 2021, 416, 126098 CrossRef CAS PubMed.
- R. B. Hoff, L. Molognoni, C. T. P. Deolindo, M. O. Vargas, C. R. Kleemann and H. Daguer, J. Chromatogr. B, 2020, 1152, 122232 CrossRef CAS PubMed.
- S. Zhi, J. Zhou, H. Liu, H. Wu, Z. Zhang, Y. Ding and K. Zhang, J. Chromatogr. B, 2020, 1154, 122286 CrossRef CAS PubMed.
- Y. Zhang, X. Xue, S. Su, Z. Guo, J. Wang, L. Ding, Y. Liu and J. Zhu, Food Anal. Methods, 2018, 11, 2865–2884 CrossRef.
- Q. Chen, X.-D. Pan, B.-F. Huang and J.-L. Han, J. Pharm. Biomed. Anal., 2017, 145, 525–530 CrossRef CAS PubMed.
- L. Li, Y. Yin, G. Zheng, S. Liu, C. Zhao, L. Ma, Q. Shan, X. Dai, L. Wei, J. Lin and W. Xie, Arab. J. Chem., 2022, 15, 103912 CrossRef CAS.
- S. A. Khatibi, S. Hamidi and M. R. Siahi-Shadbad, Crit. Rev. Food Sci. Nutr., 2021, 61, 3361–3382 CrossRef CAS PubMed.
- L. Xia, L. Liu, X. Lv, F. Qu, G. Li and J. You, J. Chromatogr. A, 2017, 1500, 24–31 CrossRef CAS PubMed.
- L. Fu, H. Zhou, E. Miao, S. Lu, S. Jing, Y. Hu, L. Wei, J. Zhan and M. Wu, Food Chem., 2019, 289, 701–707 CrossRef CAS PubMed.
- X. He, G. N. Wang, K. Yang, H. Z. Liu, X. J. Wu and J. P. Wang, Food Chem., 2017, 221, 1226–1231 CrossRef CAS PubMed.
- M. Hernández-Mesa, C. Cruces-Blanco and A. M. García-Campaña, Talanta, 2017, 163, 111–120 CrossRef PubMed.
- H. Duo, X. Lu, X. Nie, L. Wang, S. Wang, X. Liang and Y. Guo, J. Chromatogr. A, 2020, 1626, 461328 CrossRef CAS PubMed.
- T. Liu, C. Sang, Y. Liu, B. Shao and G. He, Food Chem., 2025, 492, 145624 CrossRef CAS PubMed.
- A. Azzouz, L. Hejji, A. Assafi and K.-H. Kim, TrAC, Trends Anal. Chem., 2025, 193, 118468 CrossRef CAS.
- N. Yuan, C. Zhang, X. Zhang and R. Zhang, J. Cleaner Prod., 2024, 434, 140259 CrossRef CAS.
- Z. Shen, X. Xu, X. Wang, Z. Du and F. Zhang, Food Chem., 2025, 496, 146712 CrossRef CAS PubMed.
- J. Ge, J. Xiao, L. Liu, L. Qiu and X. Jiang, J. Porous Mater., 2016, 23, 791–800 CrossRef CAS.
- K. Hu, Y. Shi, W. Zhu, J. Cai, W. Zhao, H. Zeng, Z. Zhang and S. Zhang, Food Chem., 2021, 339, 128079 CrossRef CAS PubMed.
- C. Zhang, G. Li and Z. Zhang, J. Chromatogr. A, 2015, 1419, 1–9 CrossRef CAS PubMed.
- L. Wen, L. Liu, X. Wang, M.-L. Wang, J.-M. Lin and R.-S. Zhao, J. Chromatogr. A, 2020, 1625, 461275 CrossRef CAS PubMed.
- N. Li, D. Wu, N. Hu, G. Fan, X. Li, J. Sun, X. Chen, Y. Suo, G. Li and Y. Wu, J. Agric. Food Chem., 2018, 66, 3572–3580 CrossRef CAS PubMed.
- P. N. Nomngongo, S. K. Selahle, A. Mpupa, A. Nqombolo, T. S. Munonde and L. M. Madikizela, TrAC, Trends Anal. Chem., 2024, 179, 117906 CrossRef CAS.
- J. Peng, D. Liu, T. Shi, H. Tian, X. Hui and H. He, Anal. Bioanal. Chem., 2017, 409, 4157–4166 CrossRef CAS PubMed.
- X. Yang, X. Zhou, Q. Wang, Q. Yang, J. Wang and L. Chen, J. Chromatogr. A, 2025, 1762, 466341 CrossRef CAS PubMed.
- G. Wang, T. Zhou and Y. Lei, RSC Adv., 2020, 10, 11557–11564 RSC.
- Q. Li, S. Zhu, F. Wu, F. Chen and C. Guo, Microchim. Acta, 2023, 190, 369 CrossRef CAS PubMed.
- J. Xin, X. Wang, N. Li, L. Liu, Y. Lian, M. Wang and R.-S. Zhao, Food Chem., 2020, 330, 127255 CrossRef CAS PubMed.
- J. Li, M. Chen, Z. Gao, J. Du, W. Yang and M. Yin, Colloids Surf. B Biointerfaces, 2016, 146, 468–474 CrossRef CAS PubMed.
- S. Lin, N. Gan, L. Qiao, J. Zhang, Y. Cao and Y. Chen, Talanta, 2015, 144, 1139–1145 CrossRef CAS PubMed.
- M. A. Habila, Z. A. ALOthman, A. M. El-Toni, J. P. Labis and M. Soylak, Talanta, 2016, 154, 539–547 CrossRef CAS PubMed.
- L. Lian, J. Lv, X. Wang and D. Lou, J. Chromatogr. A, 2018, 1534, 1–9 CrossRef CAS PubMed.
- P. Shi and N. Ye, Talanta, 2015, 143, 219–225 CrossRef CAS PubMed.
- Y. Hao, M. Yang, X. Chen, F. Zhang, N. Li, M. He and M. Xu, J. Agric. Food Chem., 2023, 71, 8656–8664 CrossRef CAS PubMed.
- X. Zhang, L. Chen, Y. Xu, H. Wang, Q. Zeng, Q. Zhao, N. Ren and L. Ding, J. Chromatogr. B, 2010, 878, 3421–3426 CrossRef CAS PubMed.
- Y. Li, H. Zhang, Y. Chen, L. Huang, Z. Lin and Z. Cai, ACS Appl. Mater. Interfaces, 2019, 11, 22492–22500 CrossRef CAS PubMed.
- N. Li, M. Liang, H. Zhang, Z. Hua, L. Ma, Y. Qi and K. Wang, RSC Adv., 2024, 14, 8303–8312 RSC.
- D. Li, M. He, B. Chen and B. Hu, J. Chromatogr. A, 2019, 1601, 1–8 CrossRef CAS PubMed.
- Y. Chen and Z. Chen, Talanta, 2017, 165, 188–193 CrossRef CAS PubMed.
- D. Wei, A. Pan, C. Zhang, M. Guo, C. Lou, J. Zhang, H. Wu and X. Wang, Food Chem., 2023, 404, 134464 CrossRef CAS PubMed.
- M. Zhang, J. Li, C. Zhang, Z. Wu, Y. Yang, J. Li, F. Fu and Z. Lin, J. Chromatogr. A, 2020, 1615, 460773 CrossRef CAS PubMed.
- M. Shirani, B. Akbari-adergani, H. Rashidi Nodeh and S. Shahabuddin, Microchim. Acta, 2020, 187, 634 CrossRef CAS PubMed.
- R. Mirzaei, M. Yunesian, S. Nasseri, M. Gholami, E. Jalilzadeh, S. Shoeibi, H. S. Bidshahi and A. Mesdaghinia, J. Environ. Health Sci. Eng., 2017, 15, 21 CrossRef PubMed.
- H. Su, Y. Lin, Z. Wang, Y.-L. E. Wong, X. Chen and T.-W. D. Chan, J. Chromatogr. A, 2016, 1466, 21–28 CrossRef CAS PubMed.
- D. Li, J. Zhu, M. Wang, W. Bi, X. Huang and D. D. Y. Chen, J. Chromatogr. A, 2017, 1491, 27–35 CrossRef CAS PubMed.
- F. J. Lara, M. Del Olmo-Iruela, C. Cruces-Blanco, C. Quesada-Molina and A. M. García-Campaña, TrAC, Trends Anal. Chem., 2012, 38, 52–66 CrossRef CAS.
- R. Li, Y. Zhu, X. Zhang, S. Li, D. Wang, Z. Liu, X. Wang, Y. Hou and S. Li, Polymer, 2025, 320, 127973 CrossRef CAS.
- N. Wang, H. Li, D. Kong, X. Feng, C. Li, X. Cui and T. Wang, Dyes Pigm., 2025, 240, 112841 CrossRef CAS.
- M. S. Islam and S. Mitra, Hybrid Adv., 2023, 3, 100074 CrossRef.
- J. Wu, E. Guo, M. Wang, K. Wang, L. Ma and K. Lian, J. Food Compos. Anal., 2023, 122, 105437 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |