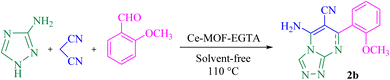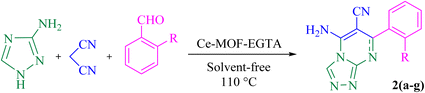Open Access Article
Open Access ArticleA novel flexible egtazic acid/cerium-based metal organic framework as a potent catalyst for the synthesis of benzochromenoquinolinones and triazolopyrimidines
Ghazale Almasi,
Davood Habibi *,
Hosein Khodakarami and
Masoumeh Beiranvand
*,
Hosein Khodakarami and
Masoumeh Beiranvand
Department of Organic Chemistry, Faculty of Chemistry and Petroleum Sciences, Bu-Ali Sina University, Hamedan, Iran. E-mail: davood.habibi@gmail.com; dhabibi@basu.ac.ir; Fax: +98 81 31408025; Tel: +98 81 31406070
First published on 20th February 2026
Abstract
The sponge-like structures of metal–organic frameworks (MOFs) are ideal platforms for various applications due to their structural diversity and adjustability, which allow for targeted design and synthesis. In recent years, substantial advances in the field of MOFs have been achieved, especially those containing aromatic ligands. Although there are several publications related to aliphatic ligands, the aliphatic MOFs developed are rather limited. Herein, a novel cerium-based flexible MOF (Ce-MOF-EGTA) constructed from an aliphatic tetratopic EGTA linker is reported. The framework exhibits high crystallinity, permanent porosity, and thermal stability up to 360 °C, as confirmed by FT-IR, EDX, elemental mapping, FESEM, XRD, TGA/DTA, and BET/BJH analyses. The presented flexible MOF is highly crystalline in nature and is stable up to 360 °C. Owing to the combination of intrinsic framework flexibility and accessible Ce Lewis acid sites, Ce-MOF-EGTA functions as an efficient heterogeneous catalyst for the synthesis of 6H-chromeno [4,3-b]quinolin-6-ones and 1,2,4-triazolo[4,3-a]pyrimidines, providing high yields under mild conditions with short reaction times. The catalyst demonstrates excellent stability and reusability over five consecutive cycles, with negligible cerium leaching and preservation of the original framework structure.
Introduction
MOFs are self-assembled materials formed by covalent bonds between metal ions or clusters and organic linkers. MOFs have attracted much attention from researchers over the past few decades due to their properties, such as various synthesis methods, high porosity, high surface area, spatially separated catalytic active sites, adjustable pore size and void volume, ordered crystalline shapes, structural, high thermal and chemical stabilities, and so on.1–6MOFs have wide applications in various fields such as catalysts, photocatalysis, photo-luminescence, sorption and separation, water remediation, drug storage and delivery, imaging, and sensors.7–12 The catalytic behavior of MOFs arises from both their metal centers and organic linkers. Broadly, three different types of active sites in MOFs act as reaction chambers:
(i) coordinatively unsaturated metal centers that coordinate with the substrate and catalyze the reaction,
(ii) the second type of site involves encapsulation or incorporation of active catalytic species inside the pores of MOF for synergistically enhanced catalysis, and
(iii) Functionalized ligands present in the framework to initiate the reaction.13
In general, most MOFs are based on aromatic carboxylates14,15 and N-donor ligands,16 which possess their structural rigidity and, therefore, allow a more straightforward design and predictable functionalization of the polymeric lattice, but an important advantage of MOFs is the variety of metal ions and organic ligands used in them.
According to the theories of Susumu Kitagawa and the type of ligand used in the synthesis of the MOFs based on the intactness of the framework upon interaction with the guest molecule, there are four types of MOFs (Fig. 1):
(i) in which the framework collapses upon desorption of the guest molecule, (ii) the framework remains intact during the removal of a guest molecule, (iii) a flexible type framework adjusts its shape by changing pore volume on guest uptake and restores its size upon guest removal, and (iv) structures that are associated with post-synthetic modifications.17,18
Many MOFs have been built based on aromatic or rigid ligands, such as 2-amino-terephthalic acid, benzene dicarboxylic acid, 1,3,5-benzene-tricarboxylic acid, 2-methylimidazole, biphenyl-4,4′-dicarboxylic acid, 2-(5-pyridin-4-yl-2H-[1,2,4]triazol-3-yl)-pyrimidine and so on.19–24
The synthesis of MOFs using aliphatic ligands is an emerging field that has attracted the attention of researchers due to its features, such as increased structural freedom, non-polar core, low light absorption, high hydrophobicity, controllable dynamics, and application in medicine. It has many applications in gas absorption, heterogeneous catalysis, and proton conduction.25–28 The presence of a saturated hydrocarbon core is a promising trend for the synthesis of biocompatible and biostable MOFs compared to MOFs with aromatic ligands.
The use of aliphatic ligands results in flexible frameworks, which are capable of responding to external stimuli such as temperature, pressure, light, electric field, inclusion of chemicals, etc., but they are also crystalline and can reversibly change their channels while maintaining their topology. These FL-MOFs are often associated with reversible transformations between two or more states related to expansion and contraction. This phenomenon is often known as the breathing effect,29,30 sponge-like effect,31–33 or accordion effect.34
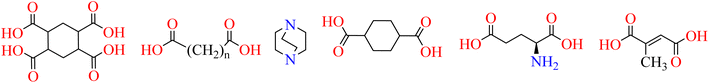
So far, various MOFs have been constructed using aliphatic ligands such as cyclohexane-1,2,4,5-tetracarboxylic acid, alkane dioic acids, glutamic acid, mesaconic acid, 1,4-cyclohexane dicarboxylic acid, and 1,4-diazabicyclo[2,2,2]octane (DABCO) (Fig. 2).35–41
Chromeno[4,3-b]quinolines consist of two fused chromene and quinoline rings, which have wide applications in various fields such as biology, medicine, physics, and electronics.42–46
Triazolopyrimidines are also an attractive class of N-heterocycles because these compounds are widely used in the pharmaceutical and agricultural industries and have significant potential in a wide range of biological activities, such as antibacterial, antifungal, antiviral, anticancer, anti-inflammatory, analgesic, and central nervous system activators.47–53
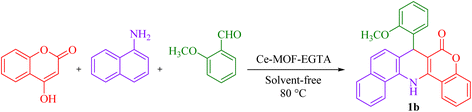
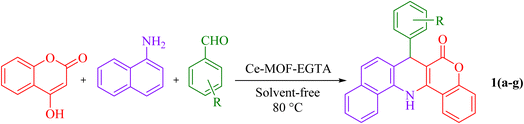
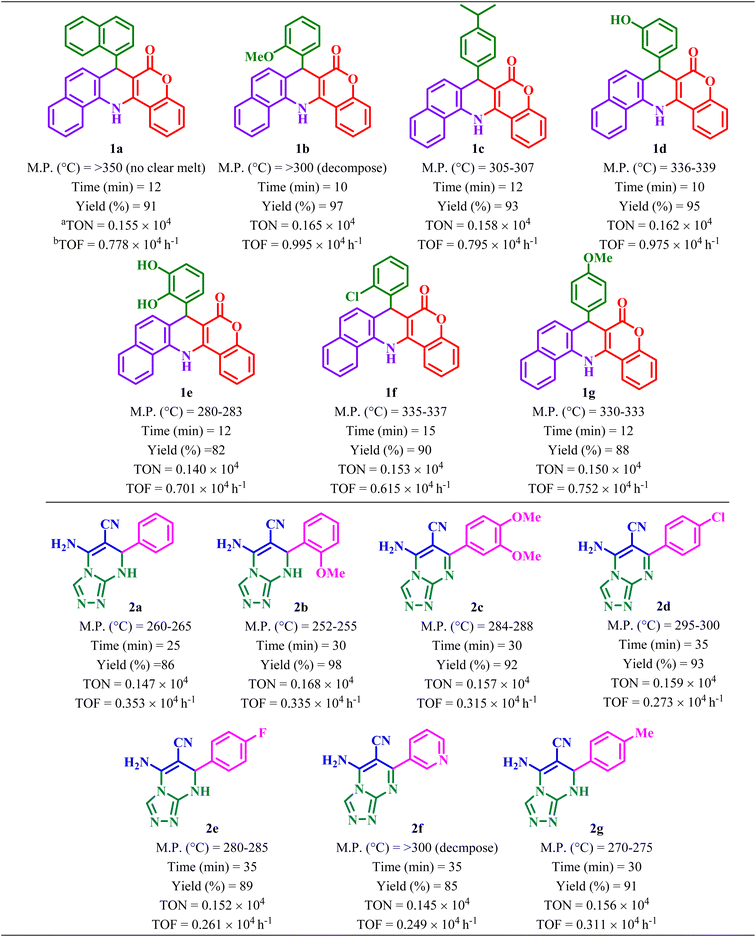
In this work, a new FL-MOF (Ce-MOF-EGTA) was synthesized from the reaction of Ce(IV) ammonium nitrate {CAN = (NH4)2[Ce(NO3)6]} with an aliphatic ligand, namely ethylene glycol-bis(β-aminoethyl ether)-N,N,N′,N′-tetraacetic acid (EGTA), also known as egtazic acid. EGTA is an aminopolycarboxylic tetratopic acid ligand, which can bind to metal ions as a chelating agent. Then, the catalytic potential of Ce-MOF-EGTA was explored as a heterogeneous catalyst for the synthesis of benzochromenoquinolines 1(a–g) and triazolopyrimidines 2(a–g). The Ce-MOF-EGTA was synthesized in a simple, one-step method and showed high catalyst activity. Compared to other catalysts used in the synthesis of benzochromenoquinolines and triazolopyrimidines, Ce-MOF-EGTA produced products with higher yields and a more convenient purification method.
Experimental
General
All the commercial reagents were provided by the foreign chemical companies and used as received. The reaction progression and purity of the prepared compounds were monitored by thin-layer chromatography (TLC) performed with silica gel 60 F 254 plates. Fourier transform infrared (FT-IR) spectra were recorded on a PerkinElmer Spectrum Version 10.02.00 employing KBr pellets. The 1H NMR (250 and 400 MHz) and 13C NMR (62.5 and 125 MHz) spectra were taken on a Bruker spectrometer (δ in ppm) using DMSO-d6 as a solvent with chemical shifts measured relative to TMS as the internal standard. Melting points were taken with a BUCHI 510 melting point apparatus. Elemental analysis was performed using a MIRA II analyzer. The field-emission scanning electron microscopy (FESEM) images were recorded using a MIRA III analyzer. The X-ray diffraction (XRD) measurements were carried out with an XRD Philips PW1730. Thermo-gravimetric differential thermal analysis (TGA-DTA) was done using an SDT-Q600 device.General strategy for the synthesis of Ce-MOF-EGTA
Ce-MOF-EGTA was synthesized according to the previously established solvothermal procedure by Wang and Li.54 Briefly, CAN (0.0437 mmol) and EGTA (0.0368 mmol) were stirred in a mixed solvent (DMF/HCOOH, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 7.5 mL) for 10 min at room temperature. The mixture was then placed in a Teflon reactor and placed in an oven at 120 °C for 72 h. The mixture was then slowly cooled to room temperature, centrifuged, washed with DMF and dichloromethane, and dried in vacuo at 100 °C for 24 h. The obtained white Ce-MOF-EGTA was then characterized by FT-IR, EDX, Elemental mapping, XRD, FESEM, TGA/DTA, and BET/BJH analyses (Fig. 3).
2, 7.5 mL) for 10 min at room temperature. The mixture was then placed in a Teflon reactor and placed in an oven at 120 °C for 72 h. The mixture was then slowly cooled to room temperature, centrifuged, washed with DMF and dichloromethane, and dried in vacuo at 100 °C for 24 h. The obtained white Ce-MOF-EGTA was then characterized by FT-IR, EDX, Elemental mapping, XRD, FESEM, TGA/DTA, and BET/BJH analyses (Fig. 3).
General procedure for the synthesis of 1(a–g) and 2(a–g) by Ce-MOF-EGTA
For the synthesis of 1(a–g), 4-hydroxycumarine (0.1621 g, 1.0 mmol), 1-naphthylamine (0.1431 g, 1.0 mmol), aromatic aldehydes (1 mmol), and Ce-MOF-EGTA (20 mg) were mixed and stirred at 80 °C under solvent-free conditions. The progress of the reaction was pursued using TLC (n-hexane/EtOAc, 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3). At the end, the resultant solid was dissolved in DMF (10 mL) and centrifuged to separate the catalyst. Then, the crude product was then purified using ethanol and acetone and characterized by FT-IR, NMR, and melting point analysis. Also, for the synthesis of 2(a–g), 3-amino-1,2,4-triazole (84 mg, 1.0 mmol), malononitrile (66 mg, 1.0 mmol), aromatic aldehydes (1.0 mmol), and Ce-MOF-EGTA (20 mg) were mixed at 110 °C under solvent-free conditions for appropriate times, and the reaction progress was monitored by TLC (n-hexane/EtOAc, 5
3). At the end, the resultant solid was dissolved in DMF (10 mL) and centrifuged to separate the catalyst. Then, the crude product was then purified using ethanol and acetone and characterized by FT-IR, NMR, and melting point analysis. Also, for the synthesis of 2(a–g), 3-amino-1,2,4-triazole (84 mg, 1.0 mmol), malononitrile (66 mg, 1.0 mmol), aromatic aldehydes (1.0 mmol), and Ce-MOF-EGTA (20 mg) were mixed at 110 °C under solvent-free conditions for appropriate times, and the reaction progress was monitored by TLC (n-hexane/EtOAc, 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). After completion of the reaction, the solid was dissolved in DMF (10 mL) and centrifuged to separate the catalyst. The product was purified using ethanol and ethyl acetate, and characterized by FT-IR, NMR, and melting point analyses.
5). After completion of the reaction, the solid was dissolved in DMF (10 mL) and centrifuged to separate the catalyst. The product was purified using ethanol and ethyl acetate, and characterized by FT-IR, NMR, and melting point analyses.
Results and discussion
Characterization of Ce-MOF-EGTA
The novel MOF was characterized by several techniques, including FT-IR, XRD, EDX, elemental mapping, FESEM, TGA/DTA, and BET/BJH.Characterization by FT-IR spectroscopy
The FT-IR spectra of EGTA and Ce-MOF-EGTA are demonstrated in Fig. 4. The FT-IR spectrum of the EGTA ligand shows the peaks at 3436, 1739, 1134, and 1100 cm−1, which are related to the O–H, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, C–N, and C–O. The FT-IR spectrum of Ce-MOF-EGTA shows distinct absorption bands at 2912, 1601, 1574, 1428, 1404, 1357 and 778 cm−1 for C–H, C
O, C–N, and C–O. The FT-IR spectrum of Ce-MOF-EGTA shows distinct absorption bands at 2912, 1601, 1574, 1428, 1404, 1357 and 778 cm−1 for C–H, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, CH2, C–N, C–O, C–C and Ce–O, respectively, as well as a sharp decrease in O–H absorption, carbonyl peak shift and the presence of a new peak at 778 cm−1 corresponding to Ce–O, confirms the successful synthesis of the catalyst.
O, CH2, C–N, C–O, C–C and Ce–O, respectively, as well as a sharp decrease in O–H absorption, carbonyl peak shift and the presence of a new peak at 778 cm−1 corresponding to Ce–O, confirms the successful synthesis of the catalyst.
Characterization by EDX and elemental mapping analysis
The existence of Ce, O, C, and N elements in the chemical composition of Ce-MOF-EGTA was confirmed by EDX analysis. In addition, EDX mappings represent the homogeneous distribution of all elements in the framework (Fig. 5).Characterization by the FESEM analysis
The morphology of the synthesized framework was examined using field emission scanning electron microscopy (FESEM). The particle size distribution histogram was also examined. FESEM images showed rod-like particles with a uniform size and homogeneous particle distribution for Ce-MOF-EGTA. As can be observed, the size of the rod-like particles is approximately 0.345–0.770 µm (Fig. 6).Characterization by XRD analysis
The XRD analysis confirms the highly crystalline nature of the newly synthesized framework and EGTA ligand (Fig. 7) with the characteristic peaks observed at 2θ = 18.7, 25.8, 31.2, 35.8, 43.5, 47.1, 50, 53.6, 56.5, 59.7, 62.1 and 65.1. For comparison, the free EGTA ligand shows peaks at 2θ = 13.7, 15.5, 16.4, 17.1, 20.4, 21.1, 23.5, 24.8, 27.1, 27.8, 28.6, 3., 31.4, 32.1, 34.7 and 41.8, confirming its incorporation into the MOF framework.Characterization by BET/BJH
The specific surface area of Ce-MOF-EGTA was characterized using the N2 adsorption–desorption analysis. The results are presented in Fig. 8, and the corresponding detailed parameters are listed in Table 1.| Entry | Parameter | Ce-MOF-EGTA |
|---|---|---|
| 1 | as (cm3 g−1) | 5.15 |
| 2 | Vm (cm3 g−1) | 1.18 |
| 3 | Total pore volume (cm3 g−1) | 0.014 |
| 4 | Mean pore diameter (nm) | 11.27 |
As observed from the BET analysis results, the surface area, pore volume (Vm), and total pore volume (cm3 g−1) of Ce-MOF-EGTA are 5.1 m2 g−1, 1.18 cm3 g−1, and 0.014 cm3 g−1, respectively (Fig. 8a). Additionally, the obtained BJH adsorption indicated that the pore size of Ce-MOF-EGTA is approximately 1.22 nm (Fig. 8b).
The BET surface areas and gas uptake capacities of most FL-MOFs are relatively low compared with the estimations by computational studies. This phenomenon can be reasonably explained by channel collapse (or partial collapse) upon solvent removal or channel blockage on account of partial solvent retention. On the one hand, FL-MOFs have been found particularly susceptible to incomplete activation or loss of porosity owing to the flexibility of the bridging organic linkers, which can hardly sustain the frameworks.25 In general, as the length of the linking chain increases, the surface area and pore volume of the framework decrease.27 Despite the low BET values, the catalytic activity of Ce-MOF-EGTA can be affected by external surfaces and partially open pores. In addition, the measured mean pore diameter in the mesoporous range suggests that the framework still contains open channels sufficient for substrate diffusion.
Characterization by TGA-DTA
Thermogravimetric analysis (TGA-DTA) was conducted under an air flow and a temperature increase rate of 10 °C min−1 to investigate the thermal behavior of the Ce-MOF-EGTA. The TGA-DTA curves of Ce-MOF-EGTA exhibit a two-step weight loss upon heating in the temperature range of 25 to 800 °C under airflow conditions (Fig. 9).The first weight loss between 50 and 110 °C is attributed to the loss of solvent molecules from the pores and also to molecules coordinated within the cage of the MOF. The second weight loss, occurring at 320 to 360 °C, is probably due to the collapse of the MOF structure and the loss of the organic linkers. Moreover, approximately 60% of the initial mass remains at 800 °C.
Catalytic studies
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio of starting materials with 20 mg of Ce-MOF-EGTA at 80 °C in solvent-free conditions (entry 2, Table 2).
1 molar ratio of starting materials with 20 mg of Ce-MOF-EGTA at 80 °C in solvent-free conditions (entry 2, Table 2).
| Entry | Condition | Catal. amount (mg) | Time (min) | Yield (%) |
| 1 | Solvent-free, 70 °C | 20 | 10 | 86 |
| 2 | Solvent-free, 80°C | 20 | 10 | 97 |
| 3 | Solvent-free, 100 °C | 20 | 10 | 83 |
| 4 | Solvent-free, 110 °C | 20 | 10 | 75 |
| 5 | Solvent-free, 80 °C | 10 | 10 | 80 |
| 6 | Solvent-free, 80 °C | 30 | 10 | 78 |
| 7 | Solvent-free, 80 °C | 20 | 20 | 70 |
| 8 | Solvent-free, 80 °C | 20 | 30 | 65 |
| 9 | Solvent-free, 80 °C | — | 30 | 12 |
| 10 | DMF, 80 °C | 20 | 10 | 63 |
| 11 | EtOH, reflux | 20 | 10 | 75 |
| 12 | CH3CN, reflux | 20 | 10 | 68 |
| 13 | THF, reflux | 20 | 10 | 45 |
| 14 | Toluene, reflux | 20 | 10 | 23 |
| 15 | n-Hexane, reflux | 20 | 10 | 15 |
| 16 | H2O, reflux | 20 | 10 | 20 |
With increasing the reaction time and catalyst amount, lower yields were observed, which is likely due to the increase the side reactions. In general, the polar solvents showed more efficacy than non-polar solvents; however, they gave lower yields compared to the solvent-free conditions.
In addition, to confirm the role of Ce-MOF-EGTA in the synthesis of 1(a–g), three separate similar reactions were carried out in the presence of CAN, EGTA, and a mixture of CAN + EGTA as catalysts, and it was observed that the corresponding yields of 1b decreased to 58%, 47%, and 62%, respectively.
For the synthesis of 2(a–g), synthesis of 2b (5-amino-7-(2-methoxyphenyl)-7,8-dihydro-[1,2,4]triazolo[4,3-a] pyrimidine-6-carbonitrile) by Ce-MOF-EGTA from the reaction of 3-amino-1,2,4-triazole, malononitrile, and 2-methoxy benzaldehyde was chosen as a model reaction. The best condition was found to be the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio of starting materials with 20 mg of Ce-MOF-EGTA at 110 °C in solvent-free conditions (entry 2, Table 3).
1 molar ratio of starting materials with 20 mg of Ce-MOF-EGTA at 110 °C in solvent-free conditions (entry 2, Table 3).
| Entry | Condition | Catal. amount (mg) | Time (min) | Yield (%) |
| 1 | Solvent-free, 90 °C | 20 | 30 | 79 |
| 2 | Solvent-free, 110°C | 20 | 30 | 98 |
| 3 | Solvent-free, 130 °C | 20 | 30 | 85 |
| 4 | Solvent-free, 110 °C | 10 | 30 | 75 |
| 5 | Solvent-free, 110 °C | 30 | 30 | 68 |
| 6 | Solvent-free, 110 °C | 20 | 20 | 82 |
| 7 | Solvent-free, 110 °C | 20 | 40 | 77 |
| 8 | Solvent-free, 110 °C | 20 | 60 | 65 |
| 9 | Solvent-free, 110 °C | — | 60 | Trace |
| 10 | DMF, 110 °C | 20 | 30 | 63 |
| 11 | EtOH, reflux | 20 | 30 | 70 |
| 12 | CH3CN, reflux | 20 | 30 | 55 |
| 13 | THF, reflux | 20 | 30 | 60 |
| 14 | Toluene, reflux | 20 | 30 | 57 |
| 15 | n-Hexane, reflux | 20 | 30 | 50 |
| 16 | H2O, reflux | 20 | 30 | 70 |
With increasing the reaction time and catalyst amount, lower yields were observed, which is likely due to the increase in the side reactions. In general, the polar solvents showed more efficacy than non-polar solvents; however, they gave lower yields compared to the solvent-free conditions.
In addition, to confirm the role of Ce-MOF-EGTA in the synthesis of 2(a–g), three separate similar reactions were carried out in the presence of CAN, EGTA, and a mixture of CAN + EGTA as catalysts, and it was observed that the corresponding yields of 2b decreased to 61%, 53%, and 75%, respectively.
Based on the results obtained from the model reaction (2b), diverse 2(a–g) were synthesized.
7-(Naphthalen-1-yl)-7,14-dihydro-6H-benzo[h]chromeno[4,3-b]quinolin-6-one (1a). Cream solid, M.P. >300 °C, IR (KBr, cm−1): 3369 (NH), 3040 (C–H aromatic), 1663 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1621 (NH bend), 1473 (C
O), 1621 (NH bend), 1473 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic) and 1267 (C–O). 1H NMR (250 MHz, DMSO-d6) δ = 9.46 (s, 1H, NH), 8.78 (d, J = 8.6 Hz, 1H, H aromatic), 8.61 (t, J = 10.1 Hz, 2H, H aromatic), 7.87 (d, J = 8.0 Hz, 1H, H aromatic), 7.76 (d, J = 7.8 Hz, 1H, H aromatic), 7.61 (q, J = 7.5 Hz, 4H, H aromatic), 7.51 (d, J = 7.4 Hz, 3H, H aromatic), 7.38 (d, J = 8.6 Hz, 4H, H aromatic), 7.15 (d, J = 8.3 Hz, 1H, H aromatic), 6.25 (s, 1H, H aliphatic). 13C NMR (62.5 MHz, DMSO-d6) δ = 152.7 (C
C aromatic) and 1267 (C–O). 1H NMR (250 MHz, DMSO-d6) δ = 9.46 (s, 1H, NH), 8.78 (d, J = 8.6 Hz, 1H, H aromatic), 8.61 (t, J = 10.1 Hz, 2H, H aromatic), 7.87 (d, J = 8.0 Hz, 1H, H aromatic), 7.76 (d, J = 7.8 Hz, 1H, H aromatic), 7.61 (q, J = 7.5 Hz, 4H, H aromatic), 7.51 (d, J = 7.4 Hz, 3H, H aromatic), 7.38 (d, J = 8.6 Hz, 4H, H aromatic), 7.15 (d, J = 8.3 Hz, 1H, H aromatic), 6.25 (s, 1H, H aliphatic). 13C NMR (62.5 MHz, DMSO-d6) δ = 152.7 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 145.5, 144.5, 133.8, 133.0, 132.3, 130.8, 129.8, 128.6, 126.8, 126.4, 124.5, 124.1, 123.3, 122.4, 121.8, 117.2, 114.0, 100.1 (C aromatic), 37.0 (C aliphatic).
O), 145.5, 144.5, 133.8, 133.0, 132.3, 130.8, 129.8, 128.6, 126.8, 126.4, 124.5, 124.1, 123.3, 122.4, 121.8, 117.2, 114.0, 100.1 (C aromatic), 37.0 (C aliphatic).
7-(2-Methoxyphenyl)-7,14-dihydro-6H-benzo[h]chromeno[4,3-b]quinolin-6-one (1b). Yellow solid, M.P. >300 °C, IR (KBr, cm−1): 3343 (NH), 3060 (C–H aromatic), 1665(C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1621(NH bend), 1471(C
O), 1621(NH bend), 1471(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic) and 1241(C–O). 1H NMR (250 MHz, DMSO-d6) δ = 9.37 (s, 1H, NH), 8.57 (t, J = 7.7 Hz,2H, H aromatic), 7.80 (d, J = 8.0 Hz, 1H, H aromatic), 7.60 (dd, J = 13.8, 7.7 Hz, 2H, H aromatic), 7.49 (dt, J = 8.4, 4.3 Hz, 3H, H aromatic), 7.37 (d, J = 8.4 Hz, 2H, H aromatic), 7.08 (t, J = 8.7 Hz, 2H, H aromatic), 6.94 (d, J = 8.0 Hz, 1H, H aromatic), 6.74 (t, J = 7.3 Hz, 1H, H aromatic), 5.76 (d, J = 2.6 Hz, 1H, H aliphatic) 3.83 (s, 3H, OCH3). 13C NMR (62.5 MHz, DMSO-d6) δ = 160.7 (C
C aromatic) and 1241(C–O). 1H NMR (250 MHz, DMSO-d6) δ = 9.37 (s, 1H, NH), 8.57 (t, J = 7.7 Hz,2H, H aromatic), 7.80 (d, J = 8.0 Hz, 1H, H aromatic), 7.60 (dd, J = 13.8, 7.7 Hz, 2H, H aromatic), 7.49 (dt, J = 8.4, 4.3 Hz, 3H, H aromatic), 7.37 (d, J = 8.4 Hz, 2H, H aromatic), 7.08 (t, J = 8.7 Hz, 2H, H aromatic), 6.94 (d, J = 8.0 Hz, 1H, H aromatic), 6.74 (t, J = 7.3 Hz, 1H, H aromatic), 5.76 (d, J = 2.6 Hz, 1H, H aliphatic) 3.83 (s, 3H, OCH3). 13C NMR (62.5 MHz, DMSO-d6) δ = 160.7 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 156.1, 152.8, 145.2, 136.0, 132.9, 132.2, 130.2, 129.0, 128.5, 128.2, 127.3, 126.3, 124.1, 123.2, 122.2, 121.4, 121.2, 117.1, 114.1, 112.1, 98.5 (C aromatic), 56.2 (OCH3), 35.6 (C aliphatic).
O), 156.1, 152.8, 145.2, 136.0, 132.9, 132.2, 130.2, 129.0, 128.5, 128.2, 127.3, 126.3, 124.1, 123.2, 122.2, 121.4, 121.2, 117.1, 114.1, 112.1, 98.5 (C aromatic), 56.2 (OCH3), 35.6 (C aliphatic).
7-(4-Isopropylphenyl)-7,14-dihydro-6H-benzo[h]chromeno[4,3-b]quinolin-6-one (1c). Yellow solid, M.P. 305–307 °C, IR (KBr, cm−1): 3463 (NH), 3044 (C–H aromatic), 2954 (C–H aliphatic), 1702 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1639 (NH bend), 1473 (C
O), 1639 (NH bend), 1473 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic) and 1267 (C–O). 1H NMR (250 MHz, DMSO-d6) δ = 9.49 (s, 1H, NH), 8.59 (dd, J = 16.6, 8.2 Hz, 2H, H aromatic), 7.85 (d, J = 8.0 Hz, 2H, H aromatic), 7.57 (ddt, J = 25.4, 18.9, 8.3 Hz, 6H, H aromatic), 7.36 (t, J = 7.9 Hz, 2H, H aromatic), 7.18 (d, J = 7.8 Hz, 2H, H aromatic), 7.05 (d, J = 7.6 Hz, 2H, H aromatic), 5.32 (s, 1H, H aliphatic), 2.73 (s, 1H, –CH2–), 1.08 (d, J = 6.9 Hz, 6H, isopropyl). 13C NMR (62.5 MHz, DMSO-d6) δ = 160.9 (C
C aromatic) and 1267 (C–O). 1H NMR (250 MHz, DMSO-d6) δ = 9.49 (s, 1H, NH), 8.59 (dd, J = 16.6, 8.2 Hz, 2H, H aromatic), 7.85 (d, J = 8.0 Hz, 2H, H aromatic), 7.57 (ddt, J = 25.4, 18.9, 8.3 Hz, 6H, H aromatic), 7.36 (t, J = 7.9 Hz, 2H, H aromatic), 7.18 (d, J = 7.8 Hz, 2H, H aromatic), 7.05 (d, J = 7.6 Hz, 2H, H aromatic), 5.32 (s, 1H, H aliphatic), 2.73 (s, 1H, –CH2–), 1.08 (d, J = 6.9 Hz, 6H, isopropyl). 13C NMR (62.5 MHz, DMSO-d6) δ = 160.9 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 156.7, 154.1, 152.7, 147.6, 147.0, 144.9, 139.4, 134.9, 132.3, 130.5, 128.6, 127.6, 126.8, 126.6, 124.4, 123.9, 121.7, 117.2 (C aromatic), 33.4 (C aliphatic), 24.2 (isopropyl).
O), 156.7, 154.1, 152.7, 147.6, 147.0, 144.9, 139.4, 134.9, 132.3, 130.5, 128.6, 127.6, 126.8, 126.6, 124.4, 123.9, 121.7, 117.2 (C aromatic), 33.4 (C aliphatic), 24.2 (isopropyl).
7-(3-Hydroxyphenyl)-7,14-dihydro-6H-benzo[h]chromeno[4,3-b]quinolin-6-one (1d). Yellow solid, M.P. 336–339 °C, IR (KBr, cm−1): 3442 (NH), 3275 (OH), 3066 (C–H aromatic), 2903 (C–H aliphatic), 1661 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1472 (C
O), 1472 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic) and 1268 (C–O). 1H NMR (250 MHz, DMSO-d6) δ = 9.45 (s, 1H, NH), 9.21 (s, 1H, OH), 8.61 (d, J = 8.4 Hz,1H, H aromatic), 8.56 (d, J = 8.0 Hz, 1H, H aromatic), 7.85 (d, J = 8.0 Hz, 1H, H aromatic), 7.68–7.56 (m, 2H, H aromatic), 7.53 (d, J = 7.3 Hz, 2H, H aromatic), 7.37 (q, J = 11.0 Hz, 3H, H aromatic), 6.98 (t, J = 7.8 Hz, 1H, H aromatic), 6.74 (d, J = 7.6 Hz, 1H, H aromatic), 6.66 (s,1H, H aromatic), 6.49 (d, J = 5.7 Hz, 1H, H aromatic), 5.26 (s, 1H, H aliphatic). 13C NMR (62.5 MHz, DMSO-d6) δ = 161.0 (C
C aromatic) and 1268 (C–O). 1H NMR (250 MHz, DMSO-d6) δ = 9.45 (s, 1H, NH), 9.21 (s, 1H, OH), 8.61 (d, J = 8.4 Hz,1H, H aromatic), 8.56 (d, J = 8.0 Hz, 1H, H aromatic), 7.85 (d, J = 8.0 Hz, 1H, H aromatic), 7.68–7.56 (m, 2H, H aromatic), 7.53 (d, J = 7.3 Hz, 2H, H aromatic), 7.37 (q, J = 11.0 Hz, 3H, H aromatic), 6.98 (t, J = 7.8 Hz, 1H, H aromatic), 6.74 (d, J = 7.6 Hz, 1H, H aromatic), 6.66 (s,1H, H aromatic), 6.49 (d, J = 5.7 Hz, 1H, H aromatic), 5.26 (s, 1H, H aliphatic). 13C NMR (62.5 MHz, DMSO-d6) δ = 161.0 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 157.8, 152.7, 148.6, 144.3, 133.1, 132.3, 130.5, 129.7, 128.6, 127.8, 126.5, 126.4, 124.4, 124.2, 123.9, 123.2, 122.3, 121.0, 118.5, 117.2, 114.7, 114.1, 113.9, 99.1 (C aromatic), 41.9 (C aliphatic).
O), 157.8, 152.7, 148.6, 144.3, 133.1, 132.3, 130.5, 129.7, 128.6, 127.8, 126.5, 126.4, 124.4, 124.2, 123.9, 123.2, 122.3, 121.0, 118.5, 117.2, 114.7, 114.1, 113.9, 99.1 (C aromatic), 41.9 (C aliphatic).
7-(2,3-Dihydroxyphenyl)-7,14-dihydro-6H-benzo[h]chromeno[4,3-b]quinolin-6-one (1e). Cream solid, M.P. 280–283 °C, IR (KBr, cm−1): 3417 (NH), 2924 (C–H aromatic), 1647 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1472 (C
O), 1472 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic) and 1266 (C–O). 1H NMR (250 MHz, DMSO-d6) δ = 9.28 (d, J = 13.8 Hz, 2H, OH), 8.64–8.47 (m, 3H, H aromatic), 7.80 (d, J = 7.8 Hz, 1H, NH), 7.70–7.33 (m, 7H, H aromatic), 6.59–6.32 (m, 3H, H aromatic), 5.78 (s,1H, H aliphatic).
C aromatic) and 1266 (C–O). 1H NMR (250 MHz, DMSO-d6) δ = 9.28 (d, J = 13.8 Hz, 2H, OH), 8.64–8.47 (m, 3H, H aromatic), 7.80 (d, J = 7.8 Hz, 1H, NH), 7.70–7.33 (m, 7H, H aromatic), 6.59–6.32 (m, 3H, H aromatic), 5.78 (s,1H, H aliphatic).
7-(2-Chlorophenyl)-7,14-dihydro-6H-benzo[h]chromeno[4,3-b]quinolin-6-one (1f). Cream solid, M.P. 335–337 °C, IR (KBr, cm−1): 3327 (NH), 3065 (C–H aromatic), 1663 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1619 (NH bend), 1471 (C
O), 1619 (NH bend), 1471 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic) and 1272 (C–O). 1H NMR (250 MHz, DMSO-d6) δ = 9.38 (s, 1H, NH), 8.57 (dd, J = 13.2, 8.3 Hz, 2H, H aromatic), 7.81 (d, J = 8.1 Hz, 1H, H aromatic), 7.65 (d, J = 8.6 Hz, 2H, H aromatic), 7.51 (dd, J = 11.7, 7.8 Hz, 4H, H aromatic), 7.37 (d, J = 8.1 Hz, 2H, H aromatic), 7.29 (d, J = 8.6 Hz,1H, H aromatic), 7.16–7.10 (m, 2H, H aromatic), 5.88 (s, 1H, H aliphatic).
C aromatic) and 1272 (C–O). 1H NMR (250 MHz, DMSO-d6) δ = 9.38 (s, 1H, NH), 8.57 (dd, J = 13.2, 8.3 Hz, 2H, H aromatic), 7.81 (d, J = 8.1 Hz, 1H, H aromatic), 7.65 (d, J = 8.6 Hz, 2H, H aromatic), 7.51 (dd, J = 11.7, 7.8 Hz, 4H, H aromatic), 7.37 (d, J = 8.1 Hz, 2H, H aromatic), 7.29 (d, J = 8.6 Hz,1H, H aromatic), 7.16–7.10 (m, 2H, H aromatic), 5.88 (s, 1H, H aliphatic).
7-(4-Methoxyphenyl)-7,14-dihydro-6H-benzo[h]chromeno[4,3-b]quinolin-6-one (1g). Yellow solid, M.P. 330–333 °C, IR (KBr, cm−1): 3295 (NH), 2992 (C–H aromatic), 1665 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1622 (NH bend), 1400 (C
O), 1622 (NH bend), 1400 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic) and 1246 (C–O). 1H NMR (250 MHz, DMSO-d6) δ = 9.44 (s, 1H, NH), 8.58 (dd, J = 18.4, 8.2 Hz,2H, H aromatic), 7.84 (d, J = 7.9 Hz, 1H, H aromatic), 7.69–7.28 (m, 7H, H aromatic), 7.17 (d, J = 8.2 Hz, 2H, H aromatic), 6.74 (d, J = 8.2 Hz, 2H, H aromatic), 5.29 (s,1H, H aliphatic), 3.61 (s, 3H, OCH3).
C aromatic) and 1246 (C–O). 1H NMR (250 MHz, DMSO-d6) δ = 9.44 (s, 1H, NH), 8.58 (dd, J = 18.4, 8.2 Hz,2H, H aromatic), 7.84 (d, J = 7.9 Hz, 1H, H aromatic), 7.69–7.28 (m, 7H, H aromatic), 7.17 (d, J = 8.2 Hz, 2H, H aromatic), 6.74 (d, J = 8.2 Hz, 2H, H aromatic), 5.29 (s,1H, H aliphatic), 3.61 (s, 3H, OCH3).
5-Amino-7-phenyl-7,8-dihydro-[1,2,4]triazolo[4,3-a]pyrimidine-6-carbonitrile (2a). Yellow solid, M.P. 259–262 °C, IR (KBr, cm−1): 3357 (NH2), 3267 (NH), 3183 (C–H aromatic), 2190 (C
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N), 1531 (C
N), 1531 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) and 1367 (C
N) and 1367 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic). 1H NMR (300 MHz, DMSO-d6) δ = 8.80 (s, 1H, NH), 7.74 (s, 1H, H aromatic), 7.48–7.35 (m, 2H, NH2), 7.32 (ddd, J = 8.2, 6.6, 1.6 Hz, 3H, H aromatic), 7.24 (s, 2H, H aromatic), 5.37 (d, J = 2.0 Hz, 1H, H aliphatic). 13C NMR (75 MHz, DMSO-d6) δ = 160.5, 158.3, 157.6, 156.4, 154.8, 154.5, 152.4, 147.5, 143.7, 135.7, 131.0, 130.2, 129.2, 128.5, 128.3, 126.5 (C aromatic), 119.5 (CN), 56.5 (C aliphatic).
C aromatic). 1H NMR (300 MHz, DMSO-d6) δ = 8.80 (s, 1H, NH), 7.74 (s, 1H, H aromatic), 7.48–7.35 (m, 2H, NH2), 7.32 (ddd, J = 8.2, 6.6, 1.6 Hz, 3H, H aromatic), 7.24 (s, 2H, H aromatic), 5.37 (d, J = 2.0 Hz, 1H, H aliphatic). 13C NMR (75 MHz, DMSO-d6) δ = 160.5, 158.3, 157.6, 156.4, 154.8, 154.5, 152.4, 147.5, 143.7, 135.7, 131.0, 130.2, 129.2, 128.5, 128.3, 126.5 (C aromatic), 119.5 (CN), 56.5 (C aliphatic).
5-Amino-7-(2-methoxyphenyl)-7,8-dihydro-[1,2,4]triazolo[4,3-a]pyrimidine-6-carbonitrile (2b). Yellow solid, M.P. 252–255 °C, IR (KBr, cm−1): 3456 (NH2), 3360 (NH), 3265 (C–H aromatic), 3008 (C–H aliphatic), 2189 (C
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N), 1630 (C
N), 1630 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) and 1485 (C
N) and 1485 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic), 1236 (C–O). 1H NMR (500 MHz,DMSO-d6) δ = 8.41 (d, J = 2.5 Hz, 1H, NH), 7.66 (s, 1H, H aromatic), 7.30–7.26 (m, 1H, H aromatic), 7.10 (dd, J = 7.5, 1.7 Hz, 1H, H aromatic), 7.06 (s, 2H, NH2), 7.02 (d, J = 8.2 Hz, 1H, H aromatic), 6.91 (t, J = 7.4 Hz, 1H, H aromatic), 5.45 (d, J = 2.5 Hz, 1H, H aliphatic), 3.68 (s, 3H, OCH3). 13C NMR (125 MHz, DMSO-d6) δ = 157.1, 154.7, 152.0, 147.8, 131.0, 129.8, 127.5, 120.8, 119.4 (C aromatic), 112.0 (CN), 55.9 (C aliphatic), 55.5 (OCH3).
C aromatic), 1236 (C–O). 1H NMR (500 MHz,DMSO-d6) δ = 8.41 (d, J = 2.5 Hz, 1H, NH), 7.66 (s, 1H, H aromatic), 7.30–7.26 (m, 1H, H aromatic), 7.10 (dd, J = 7.5, 1.7 Hz, 1H, H aromatic), 7.06 (s, 2H, NH2), 7.02 (d, J = 8.2 Hz, 1H, H aromatic), 6.91 (t, J = 7.4 Hz, 1H, H aromatic), 5.45 (d, J = 2.5 Hz, 1H, H aliphatic), 3.68 (s, 3H, OCH3). 13C NMR (125 MHz, DMSO-d6) δ = 157.1, 154.7, 152.0, 147.8, 131.0, 129.8, 127.5, 120.8, 119.4 (C aromatic), 112.0 (CN), 55.9 (C aliphatic), 55.5 (OCH3).
5-Amino-7-(3,4-dimethoxyphenyl)-7,8-dihydro-[1,2,4]triazolo[4,3-a]pyrimidine-6-carbonitrile (2c). Yellow solid, M.P. 284–288 °C, IR (KBr, cm−1): 3323 (NH2), 3196 (C–H aromatic), 2963 (C–H aliphatic), 2219 (C
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N), 1682 (C
N), 1682 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) and 1466 (C
N) and 1466 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic). 1H NMR (500 MHz, DMSO-d6) δ = 8.96 (s, 2H, NH2), 8.36–8.33 (m, 1H, H aromatic), 7.24 (d, J = 15.8 Hz, 2H, H aromatic), 6.89 (s, 1H, H aromatic), 3.61 (s, 3H, OCH3), 3.10 (s, 3H, OCH3). 13C NMR (125 MHz, DMSO-d6) δ = 164.1, 156.4, 155.5, 152.1, 151.4, 148.7, 129.5, 122.5, 116.4, 112.6, 111.6 (C aromatic),75.5 (CN), 56.1 (OCH3).
C aromatic). 1H NMR (500 MHz, DMSO-d6) δ = 8.96 (s, 2H, NH2), 8.36–8.33 (m, 1H, H aromatic), 7.24 (d, J = 15.8 Hz, 2H, H aromatic), 6.89 (s, 1H, H aromatic), 3.61 (s, 3H, OCH3), 3.10 (s, 3H, OCH3). 13C NMR (125 MHz, DMSO-d6) δ = 164.1, 156.4, 155.5, 152.1, 151.4, 148.7, 129.5, 122.5, 116.4, 112.6, 111.6 (C aromatic),75.5 (CN), 56.1 (OCH3).
5-Amino-7-(4-chlorophenyl)-[1,2,4]triazolo[4,3-a]pyrimidine-6-carbonitrile (2d). White solid, M.P. 295–300 °C, IR (KBr, cm−1): 3351 (NH2), 3056 (C–H aromatic), 2197 (C
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N), 1661 (C
N), 1661 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N), 1593 (NH2 in plan bend), 1485 (C
N), 1593 (NH2 in plan bend), 1485 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic) and 1157 (C–N). 1H NMR (250 MHz, DMSO-d6) δ = 9.30 (s, 2H, NH2), 8.61 (s, 1H, H aromatic), 7.85 (d, J = 8.2 Hz, 2H, H aromatic), 7.64 (d, J = 8.4 Hz, 2H, H aromatic). 13C NMR (62.5 MHz, DMSO-d6) δ = 156.4, 151.9, 136.2, 131.0, 128.9 (C aromatic), 115.9 (CN).
C aromatic) and 1157 (C–N). 1H NMR (250 MHz, DMSO-d6) δ = 9.30 (s, 2H, NH2), 8.61 (s, 1H, H aromatic), 7.85 (d, J = 8.2 Hz, 2H, H aromatic), 7.64 (d, J = 8.4 Hz, 2H, H aromatic). 13C NMR (62.5 MHz, DMSO-d6) δ = 156.4, 151.9, 136.2, 131.0, 128.9 (C aromatic), 115.9 (CN).
5-Amino-7-(4-fluorophenyl)-7,8-dihydro-[1,2,4]triazolo[4,3-a]pyrimidine-6-carbonitrile (2e). White solid, M.P. 280–285 °C, IR (KBr, cm−1): 3351 (NH2), 3238 (NH), 3114 (C–H aromatic), 2195 (C
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N), 1661 (C
N), 1661 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) and 1485 (C
N) and 1485 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic). 1H NMR (300 MHz, DMSO-d6) δ = 8.97 (s, 1H, NH), 8.30–8.27 (m, 2H, NH2), 7.77 (s, 1H, H aromatic), 7.60 (d, J = 8.8 Hz, 2H, H aromatic), 7.38 (s, 2H, H aromatic), 5.60 (s, 1H, H aliphatic).
C aromatic). 1H NMR (300 MHz, DMSO-d6) δ = 8.97 (s, 1H, NH), 8.30–8.27 (m, 2H, NH2), 7.77 (s, 1H, H aromatic), 7.60 (d, J = 8.8 Hz, 2H, H aromatic), 7.38 (s, 2H, H aromatic), 5.60 (s, 1H, H aliphatic).
5-Amino-7-(pyridin-3-yl)-[1,2,4]triazolo[4,3-a]pyrimidine-6-carbonitrile (2f). Light brown solid, M.P. > 300 °C, IR (KBr, cm−1): 3434 (NH2), 2928 (C–H aromatic), 2190 (C
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N), 1684 (C
N), 1684 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N), 1537 (C
N), 1537 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic) and 1580 (NH2 in plane bend). 1H NMR (250 MHz, DMSO-d6) δ = 9.37 (s, 2H, NH2), 8.97 (s, 1H, H aromatic), 8.67 (d, J = 27.8 Hz, 2H, H aromatic), 8.21 (d, J = 7.8 Hz, 1H, H aromatic), 7.60 (s, 1H, H aromatic).
C aromatic) and 1580 (NH2 in plane bend). 1H NMR (250 MHz, DMSO-d6) δ = 9.37 (s, 2H, NH2), 8.97 (s, 1H, H aromatic), 8.67 (d, J = 27.8 Hz, 2H, H aromatic), 8.21 (d, J = 7.8 Hz, 1H, H aromatic), 7.60 (s, 1H, H aromatic).
5-Amino-7-(p-tolyl)-7,8-dihydro-[1,2,4]triazolo[4,3-a]pyrimidine-6-carbonitrile (2g). White solid, M.P. 270–275 °C, IR (KBr, cm−1): 3349 (NH2), 3263 (NH), 3187 (C–H aromatic), 3118 (C–H aliphatic), 2194 (C
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N), 1661 (C
N), 1661 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) and 1530 (C
N) and 1530 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic). 1H NMR (250 MHz, DMSO-d6) δ = 9.21 (s, 1H, NH), 8.63 (d, J = 32.0 Hz, 2H, NH2), 7.75–7.64 (m, 1H, H aromatic), 7.15 (s, 5H, H aromatic), 5.25 (s, 1H, H aromatic), 2.26 (d, J = 30.9 Hz, 3H, CH3).
C aromatic). 1H NMR (250 MHz, DMSO-d6) δ = 9.21 (s, 1H, NH), 8.63 (d, J = 32.0 Hz, 2H, NH2), 7.75–7.64 (m, 1H, H aromatic), 7.15 (s, 5H, H aromatic), 5.25 (s, 1H, H aromatic), 2.26 (d, J = 30.9 Hz, 3H, CH3).
The proposed mechanism for the synthesis of 2(a–g) is shown in Fig. 11. The aldehyde is activated by the Ce Lewis acid sites of Ce-MOF-EGTA, which increases its electrophilicity and facilitates the initial condensation with malononitrile to form the benzylidene intermediate A. Intermediate A then undergoes a Michael addition with 3-amino-1,2,4-triazole, generating intermediate B. Subsequent intramolecular cyclization of intermediate B affords the expected cyclic products. Anomeric-based oxidation or air-oxidation leads to hydride transfer, controlling the ring aromaticity. In this reaction as well, the structural arrangement of the ligand can assist in aligning the reactants within the framework.
 | ||
| Fig. 11 Plausible mechanism for the synthesis of 2(a–g) by Ce-MOF-EGTA. Generally, the high catalytic efficiency observed for Ce-MOF-EGTA highlights the functional importance of its flexible framework. In this MOF, the aliphatic tetratopic EGTA ligand coordinates to Ce centers through its four binding sites, forming a stable yet flexible network. This coordination arrangement allows the ligand to adopt different conformations, creating an adaptive pore environment that facilitates substrate diffusion and proper alignment of reactants near the Lewis acidic Ce sites. The flexible nature of the ligand framework enables the MOF to accommodate substrates of varying sizes and shapes, consistent with a breathing or conformational flexibility observed in other flexible MOFs.55,56 | ||
Moreover, the used catalyst was evaluated after the fifth catalytic cycle by the FT-IR, XRD, and FESEM techniques. As shown in Fig. 13, the IR index peaks of the synthesized framework were preserved in the recycled catalyst, which indicates the stability of the recycled catalyst.
On the other hand, the XRD spectrum of the used Ce-MOF-EGTA catalyst shows that the crystalline phase is preserved (Fig. 14).
In addition, the SEM images reveal that the catalyst's structure remains intact (Fig. 15).
According to the structural analysis (FT-IR, XRD and SEM), the recovered catalyst retains its original structure, indicating negligible Ce leaching. Moreover, a hot filtration test was performed: the catalyst was removed from the reaction mixture midway, and the reaction continued without noticeable change in rate or yield, suggesting minimal Ce leaching. We have not performed ICP or EDX analysis to quantitatively measure leaching; however, based on the hot filtration test, any leaching is expected to be very low.
Conclusion
A novel cerium-based flexible MOF (Ce-MOF-EGTA) constructed from an aliphatic tetratopic ligand was developed as an efficient and reusable heterogeneous catalyst. EGTA introduces intrinsic conformational flexibility due to its aliphatic backbone and chelating ether groups, leading to an adaptive framework. Moreover, Ce provides accessible Lewis acid sites, which are rarely explored in flexible MOFs for catalytic applications. Its intrinsic framework flexibility combined with accessible Ce Lewis acid sites enables high-yield synthesis of biologically relevant heterocycles under mild and green conditions. The catalyst retains its structure and activity over multiple cycles, demonstrating excellent stability and recyclability. This study highlights the potential of aliphatic-ligand-based flexible MOFs for sustainable and adaptive catalytic applications, opening avenues for future design of functional MOFs.Conflicts of interest
There is no conflicts to declare.Data availability
All data generated or analyzed during this study are included in the Main manuscript text.References
- V. Mishra and A. Mishra, Introduction of metal-organic frameworks (MOFs), in Metal–Organic Frameworks as Forensic Detectors, Springer Nature Singapore, Singapore, 2025, pp. 1–20 Search PubMed
.
- V. F. Yusuf, N. I. Malek and S. K. Kailasa, Review on metal–organic framework classification, synthetic approaches, and influencing factors: applications in energy, drug delivery, and wastewater treatment, ACS Omega, 2022, 7, 44507–44531 CrossRef CAS PubMed
.
- M. Rubio-Martinez, C. Avci-Camur, A. W. Thornton, I. Imaz, D. Maspoch and M. R. Hill, New synthetic routes towards MOF production at scale, Chem. Soc. Rev., 2017, 46, 3453–3480 RSC
.
- Z. Han, Y. Yang, J. Rushlow, J. Huo, Z. Liu, Y. C. Hsu, R. Yin, M. Wang, R. Liang, K. Y. Wang and H. C. Zhou, Development of the design and synthesis of metal–organic frameworks (MOFs)–from large scale attempts, functional oriented modifications, to artificial intelligence (AI) predictions, Chem. Soc. Rev., 2025, 54, 367–395 RSC
.
- H. Laeim, V. Molahalli, P. Prajongthat, A. Pattanaporkratana, G. Pathak, B. Phettong, N. Hongkarnjanakul and N. Chattham, Porosity tunable metal-organic framework (MOF)-based composites for energy storage applications: recent progress, Polymers, 2025, 17, 130 CrossRef CAS PubMed
.
- C. Liu, C. Tian, J. Guo, X. Zhang, L. Wu, L. Zhu and B. Du, Research progress of metal organic frameworks as drug delivery systems, ACS Appl. Mater. Interfaces, 2024, 16, 43156–43170 CrossRef CAS PubMed
.
- A. Ali, J. M. Moradian, A. Naveed, S. Zhang, M. H. Tahir, K. Shehzad and M. Sillanpää, Progress in cathode materials for rechargeable zinc-ion batteries: from inorganic and organic systems to hybrid frameworks and biomass-derived innovations, Prog. Mater. Sci., 2025, 101543 Search PubMed
.
- S. Essalmi, S. Lotfi, A. BaQais, M. Saadi, M. Arab and H. A. Ahsaine, Design, and application of metal organic frameworks for heavy metals adsorption in water: a review, RSC Adv., 2024, 14, 9365–9390 RSC
.
- G. R. Xu, Z. H. An, K. Xu, Q. Liu, R. Das and H. L. Zhao, Metal organic framework (MOF)-based micro/nanoscaled materials for heavy metal ions removal: the cutting-edge study on designs, synthesis, and applications, Coord. Chem. Rev., 2021, 427, 213554 CrossRef CAS
.
- D. Wang, H. Yao, J. Ye, Y. Gao, H. Cong and B. Yu, Metal organic frameworks (MOFs): classification, synthesis, modification, and biomedical applications, Small, 2024, 20, 2404350 CrossRef CAS PubMed
.
- S. Kumar, S. Jain, M. Nehra, N. Dilbaghi, G. Marrazza and K. H. Kim, Green synthesis of metal organic frameworks: a state-of-the-art review of potential environmental and medical applications, Coord. Chem. Rev., 2020, 420, 213407 CrossRef CAS
.
- P. S. Sharanyakanth and M. Radhakrishnan, Synthesis of metal-organic frameworks (MOFs) and its application in food packaging: a critical review, Trends Food Sci. Technol., 2020, 104, 102–116 CrossRef CAS
.
- S. Yadav, R. Dixit, S. Sharma, S. Dutta, K. Solanki and R. K. Sharma, Magnetic metal organic framework composites: structurally advanced catalytic materials for organic trans-formations, Mater. Adv., 2021, 2, 2153–2187 RSC
.
- H. Ghasempour, K. Y. Wang, J. A. Powell, F. Zare Karizi, X. L. Lv, A. Morsali and H. C. Zhou, Metal–organic frameworks based on multicarboxylate linkers, Coord. Chem. Rev., 2021, 426, 213542 CrossRef CAS
.
- D. Yang, Y. Chen, Z. Su, X. Zhang, W. Zhang and K. Srinivas, Organic carboxylate-based MOFs and derivatives for electrocatalytic water oxidation, Coord. Chem. Rev., 2021, 428, 213619 CrossRef CAS
.
- A. V. Desai, S. Sharma, S. Let and S. K. Ghosh, N-donor linker-based metal-organic frame-works (MOFs): advancement and prospects as functional materials, Coord. Chem. Rev., 2019, 395, 146–192 CrossRef CAS
.
- J. H. Lee, S. Jeoung, Y. G. Chung and H. R. Moon, Elucidation of flexible metal-organic frameworks: research progresses and recent developments, Coord. Chem. Rev., 2019, 389, 161–188 CrossRef CAS
.
- J. Kaur and G. Kaur, Review on flexible metal-organic frameworks, Mater. Adv., 2021, 6, 8227–8243 CAS
.
- J. Chen, Q. Zhang, J. Dong, F. Xu and S. Li, Amino-functionalized Cu metal organic framework nanosheets as fluorescent probes for detecting TNP, Anal. Methods, 2021, 13, 5328–5334 RSC
.
- M. A. Farajzadeh, S. Rahimzadeh, M. R. A. Mogaddam and M. B. Aghdam, A fast and simple procedure for the synthesis of a zinc and 1,4-benzene dicarboxylic acid metal–organic framework and its evaluation as a sorbent for dispersive micro solid phase extraction of pesticide residues, RSC Adv., 2024, 14, 28035–28043 RSC
.
- R. Darabi and H. Karimi-Maleh, Hierarchical copper-1,3,5-benzene-tricarboxylic acid-MOF-derived with nitrogen-doped graphene nanoribbons as a novel assembly nanocomposite for asymmetric supercapacitors, Adv. Compos. Hybrid Mater., 2023, 6, 114 CrossRef CAS
.
- N. Kumar Sundarraj, J. Q. S. Pandiyan, A. Al Souwaileh, J. J. Wu and S. Anandan, Sono-chemical synthesis of Ni-MOF using 2-methylimidazole as an organic linker: pushing the boundaries of energy storage, Electrochim. Acta, 2025, 523, 145975 CrossRef
.
- L. Biswal, J. E. Goodwill, C. Janiak and S. Chatterjee, Versatility, cost analysis, and scale-up in fluoride and arsenic removal using metal-organic framework-based adsorbents, Sep. Purif. Rev., 2022, 51, 408–426 CrossRef CAS
.
- H. Li, Q. Li, X. He, N. Zhang, Z. Xu and Y. Wang, The magnetic hybrid Cu(I)-MOF@Fe3O4 with hierarchically engineered micropores for highly efficient removal of Cr(VI) from aqueous solution, Cryst. Growth Des., 2018, 18, 6248–6256 CrossRef CAS
.
- P. A. Demakov, Properties of aliphatic ligand-based metal organic frameworks, Polymers, 2023, 15, 2891 CrossRef CAS PubMed
.
- Z. J. Lin, J. Lü, M. Hong and R. Cao, Metal organic frameworks based on flexible ligands (FL-MOFs): structures and applications, Chem. Soc. Rev., 2014, 43, 5867–5895 RSC
.
- M. Chang, Y. Zhao, D. Liu, J. Yang, J. Li and C. Zhong, Methane-trapping metal organic frameworks with an aliphatic ligand for efficient CH4/N2 separation, Sustain. Energy Fuels, 2020, 4, 138–142 RSC
.
- D. H. Hong and M. P. Suh, Enhancing CO2 separation ability of a metal organic framework by post-synthetic ligand exchange with flexible aliphatic carboxylates, Chem. Eur. J., 2014, 20, 426–434 CrossRef CAS PubMed
.
- G. Férey and C. Serre, Large breathing effects in three-dimensional porous hybrid matter: facts, analyses, rules and consequences, Chem. Soc. Rev., 2009, 38, 1380–1399 RSC
.
- P. A. Demakov, A. S. Poryvaev, K. A. Kovalenko, D. G. Samsonenko, M. V. Fedin, V. P. Fedin and D. N. Dybtsev, Structural dynamics and adsorption properties of the breathing microporous aliphatic metal organic framework, Inorg. Chem., 2020, 59, 15724–15732 CrossRef CAS PubMed
.
- G. Férey, A selective magnetic sponge, Nat. Mater., 2003, 2, 136–137 CrossRef PubMed
.
- D. Maspoch, D. Ruiz-Molina, K. Wurst, N. Domingo, M. Cavallini, F. Biscarini, J. Tejada, C. Rovira and J. Veciana, A nanoporous molecular magnet with reversible solvent-induced mechanical and magnetic properties, Nat. Mater., 2003, 2, 190–195 CrossRef CAS PubMed
.
- N. Zigon, V. Duplan, N. Wada and M. Fujita, Crystalline sponge method: X-ray structure analysis of small molecules by post-orientation within porous crystals-principle and proof-of-concept studies, Angew.
Chem., Int. Ed., 2021, 60, 25204–25222 CrossRef CAS PubMed
.
- G. Alberti, S. Murcia-Mascaros and R. Vivani, Pillared derivatives of γ-zirconium phosphate containing nonrigid alkyl chain pillars, J. Am. Chem. Soc., 1998, 120, 9291–9295 CrossRef CAS
.
- T. K. Kim, K. J. Lee, M. Choi, N. Park, D. Moon and H. R. Moon, Metal–organic frameworks constructed from flexible ditopic ligands: conformational diversity of an aliphatic ligand, New J. Chem., 2013, 37, 4130–4139 RSC
.
- H. Reinsch, R. S. Pillai, R. Siegel, J. Senker, A. Lieb, G. Maurin and N. Stock, Structure and properties of Al-MIL-53-ADP, a breathing MOF based on the aliphatic linker molecule adipic acid, Dalton Trans., 2016, 45, 4179–4186 RSC
.
- H. Reinsch, T. Homburg, N. Heidenreich, D. Fröhlich, S. Hennninger, M. Wark and N. Stock, Green synthesis of a new Al-MOF based on the aliphatic linker mesaconic acid: structure, properties and in situ crystallization studies of Al-MIL-68-Mes, Chem. Eur. J., 2018, 24, 2173–2181 CrossRef CAS PubMed
.
- V. D. Slyusarchuk, P. E. Kruger and C. S. Hawes, Cyclic aliphatic hydrocarbons as linkers in metal-organic frameworks: new frontiers for ligand design, ChemPlusChem, 2020, 85, 845–854 CrossRef CAS PubMed
.
- K. D. Abasheeva, P. A. Demakov, E. V. Polyakova, A. N. Lavrov, V. P. Fedin and D. N. Dybtsev, Synthesis, structural versatility, magnetic properties, and (I) adsorption in a series of cobalt(II) metal–organic frameworks with a charge-neutral aliphatic (O, O)-donor bridge, Nanomater., 2023, 13, 2773 CrossRef CAS PubMed
.
- P. A. Demakov, A. S. Poryvaev, K. A Kovalenko, D. G. Samsonenko, M. V. Fedin, V. P. Fedin and D. N. Dybtsev, Structural dynamics and adsorption properties of the breathing microporous aliphatic metal–organic framework, Inorg. Chem., 2020, 59, 15724–15732 CrossRef CAS PubMed
.
- J. Wang, Y. C. Ou, Y. Shen, L. Yun, J. D. Leng, Z. Lin and M. L Tong, Coordination chemistry of cyclohexane-1,2,4,5-tetracarboxylate (H4L). Synthesis, structure, and magnetic properties of metal-organic frameworks with conformation-flexible H4L ligand, Cryst. Growth Des., 2009, 9, 2442–2450 CrossRef CAS
.
- M. Beiranvand, D. Habibi and H. Khodakarami, Novel UiO-NH2-like Zr-based MOF (Basu-DPU) as an excellent catalyst for preparation of new 6H-chromeno[4,3-b]quinolin-6-ones, ACS Omega, 2023, 8, 25924–25937 CrossRef CAS PubMed
.
- P. Sanati-Tirgan and H. Eshghi, Synthesis, characterization, and application of MOF/COF hybrid composite as a highly active and recyclable catalyst for multicomponent synthesis of chromeno[4,3-b]quinoline-6-ones, Appl. Organomet. Chem., 2025, 39, e70039 CrossRef CAS
.
- G. Rahimzadeh, S. Bahadorikhalili, E. Kianmehr and M. Mahdavi, Ionic liquid-functionalized magnetic nanostructures as an efficient catalyst for the synthesis of 6H-chromeno[4,3-b] quinolin-6-ones, Mol. Divers., 2017, 21, 597–609 CrossRef CAS PubMed
.
- R. Singha, A. Islam and P. Ghosh, One-pot three-component tandem annulation of 4-hydroxy-coumarine with aldehyde and aromatic amines using graphene oxide as an efficient catalyst, Sci. Rep., 2021, 11, 19891 CrossRef CAS PubMed
.
- Z. Chen, J. Bi and W. Su, Synthesis, and antitumor activity of novel coumarin derivatives via a three-component reaction in water, Chin. J. Chem., 2013, 31, 507–514 CrossRef CAS
.
- A. Benrashid, D. Habibi and M. Beiranvand, Deep eutectic solvent-immobilized magnetic nanoparticles as a capable catalyst for the green synthesis of triazolopyrimidines, J. Mol. Liq., 2024, 409, 125504 CrossRef CAS
.
- M. Lashkari, M. Ghashang and A. Abedi-Madiseh, Soluble glass, an efficient promoter for the cascade addition-cyclization reaction of 4-hydroxycoumarins to chalcone derivatives, Org. Prep. Proced. Int., 2020, 53, 52–58 CrossRef
.
- A. Dandia, P. Sarawgi, K. Arya and S. Khaturia, Mild and ecofriendly tandem synthesis of 1, 2,4-triazolo[4,3-a]pyrimidines in aqueous medium, Arkivoc, 2006, 16, 83–92 Search PubMed
.
- N. Jamasbi, M. Irankhah-Khanghah, F. Shirini, H. Tajik and M. S. N. Langarudi, DABCO-based ionic liquids: introduction of two metal-free catalysts for one-pot synthesis of 1,2,4-triazolo[4,3-a]pyrimidines and pyrido[2,3-d]pyrimidines, New J. Chem., 2018, 42, 9016–9027 RSC
.
- S. S. Pandit, S. K. Bhalerao, U. S. Aher, G. L. Adhav and V. U. Pandit, Amberlyst A-15: reusable catalyst for the synthesis of 2,4,5-trisubstituted and 1,2,4,5-tetrasubstituted-1H-imidazoles under MW irradiation, J. Chem. Sci., 2011, 123, 421–426 CrossRef CAS
.
- P. Chhattise, S. Saleh, V. Pandit, S. Arbuj and V. Chabukswar, ZnO nanostructures: a heterogeneous catalyst for the synthesis of benzoxanthene and pyranopyrazole scaffolds via a multi-component reaction strategy, Mater. Adv., 2020, 1, 2339–2345 RSC
.
- S. Pandit, R. Shaikh and V. Pandit, Synthesis of 5-unsubstituted-3,4-dihydropyridine-2-(1H)-ones using nbs as a catalyst under solvent free conditions, Rasayan J. Chem., 2009, 2, 907–911 CAS
.
- X. Wang, Y. Zhang, Z. Shi, T. Lu, Q. Wang and B. Li, Multifunctional Zr-MOF based on bisimidazole tetracarboxylic acid for pH sensing and photoreduction of Cr(VI), ACS Appl. Mater. Interfaces, 2021, 13, 54 Search PubMed
.
- A. Dhakshinamoorthy and H. Garcia, Metal–organic frameworks as solid catalysts for the synthesis of nitrogen-containing heterocycles, Chem. Soc. Rev., 2014, 43, 5750–5765 RSC
.
- P. A. Demakov, Properties of aliphatic ligand-based metal–organic frameworks, Polymers, 2023, 15, 2891 CrossRef CAS PubMed
.
| This journal is © The Royal Society of Chemistry 2026 |