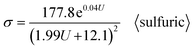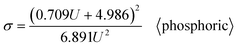DOI:
10.1039/D5RA09220E
(Review Article)
RSC Adv., 2026,
16, 7628-7647
A systematic review on competitive screening and independent variable unification focused on PAA structural parameter calculation formulas in mild anodization
Received
29th November 2025
, Accepted 11th January 2026
First published on 5th February 2026
Abstract
Porous anodized aluminium oxide (PAA) has wide and important applications in photonic crystals, energy science, nanotemplates, life science, medicine, aerospace and other scientific research and industrial manufacturing fields. The decisive factors determining its application value and specific performance are its own structural parameters. Therefore, the accurate calculation (but not destructive measurement) of each PAA structural parameter is of great significance for the design and application of PAA structures to satisfy different practical requirements. However, there is a significant problem because multiple distinct formulas proposed by different researchers are used for calculating single independent PAA structural parameters such as the pore diameter. Furthermore, these multiple distinct formulas for determining a single PAA structural parameter frequently yield different results. Compounding this issue, these single structural parameters serve as independent variables in formulas for calculating other PAA parameters. This propagation of uncertainty leads to multiple distinct results for other subsequent parameters. Consequently, in practice, for the calculation of a PAA structural parameter, it is difficult to discern which calculations are the most accurate. Regarding the aforementioned issues, this paper systematically reviews the key structural parameters of PAA and the most commonly used distinct calculation formulas of each key structural parameter. The independent variables of almost all mentioned calculation formulas are unified to the anodization voltage. Subsequently, extensive experimental data published by other researchers are substituted into all the formulas with the unified independent variable to perform an objective competitive screening for the optimal calculation formula of each PAA structural parameter. Finally, on the basis of the competitive screening and independent variable unification, an equation set of PAA structural parameter calculations is proposed for the accurate and convenient calculation of all key PAA structural parameters. The proposal of an equation set for the PAA structural parameter calculation provides a systematic, comprehensive theoretical model and mathematical tool for the design and calculation of PAA structures according to practical requirements in scientific research and engineering applications.
1 Introduction
Nanotechnology is currently one of the most researched fields of science. By some estimates, it promises to far exceed the impact of the industrial revolution. Nanotechnology provides materials like zero-dimensional nanodots,1 one-dimensional nanowires,2 two-dimensional nanoplanes of monatomic layer,3 three-dimensional nanoframes,4 periodical nanoarrays,5 and nanocomposites,6 which possess unique properties in comparison to macroscopic materials. These nanomaterials may be made from elemental carbon,7 polymers,8 metals,9 oxides,10 semiconductors,11 and superconductors,12 with potential applications in various fields of science like energy,13 electronics,14 information,15 biology,16 medicine,17 environment,18 catalysis,19 food,20 agriculture,21 military22 and many others. One of the most common nanotechnology issues is to obtain well-ordered arrays of nanostructures on a large scale with completely controllable structural parameters. Well-ordered arrays of nanostructures can be achieved by the application of various lithographic techniques such as electron beam lithography,23 X-ray lithography,24 and focused ion beam etching,25 but these techniques have two major disadvantages: a limited working area of up to a few square mm and the high cost of manufacturing.26,27 Therefore, some low-cost and controllable nanofabrication methods are more popular among researchers in nanoscience.
PAA membranes not only serve as well-ordered porous nanostructures but also as multifunctional templates for forming other nanostructures, effectively satisfying the demand for both porous nanomaterials and ordered templates. In particular, PAA membranes present many desirable properties such as tunable pore dimensions and membrane thicknesses,28 good mechanical and thermal stabilities,29 remarkable hardness,30 low-cost preparation process,31 and large area fabrication.32 In recent decades, PAA membranes have attracted increasing interest because of their potential applications in ultrafiltration and gas separation,33,34 hemodialysis,35 nanofluids,36 and Li rechargeable batteries.37 Furthermore, PAA membranes are widely used as templates for the fabrication of nanomaterials.38 Various sophisticated materials are deposited into the pores of PAA membranes by numerous techniques like electrochemical deposition,39 chemical vapor deposition,40 atomic layer deposition,41 pulse laser deposition,42 sol–gel technique,43 molecular beam epitaxy,44 and magnetron sputtering.45 The nanostructures that are fabricated by filling the pores of PAA templates have uniform and adjustable diameter and length, and they can be obtained reproducibly and economically.
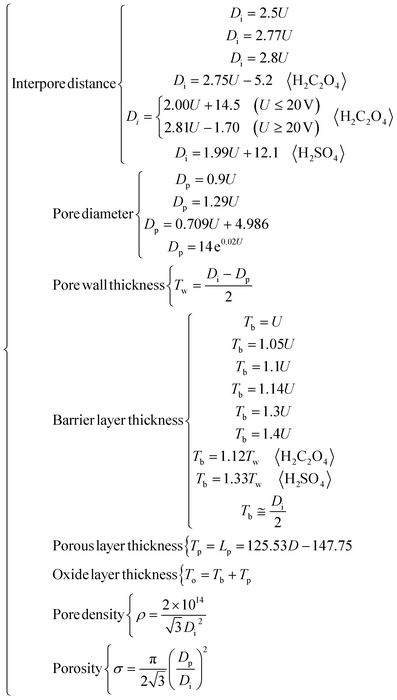
Well-ordered PAA membranes can be grown on the surface of aluminum by electrochemical oxidation,46 which is referred to as anodization in acid electrolytes. The surface of a PAA membrane is a 2-dimensional closely packed hexagonal array shaped as a honeycomb with an open cylindrical channel pore in the center of each hexagonal unit.47 The interior of a PAA membrane is characterized by parallel, non-interconnecting cylindrical channels extending from the surface down to the bottom of a PAA, and the channel bottoms are closed by a thin alumina barrier layer, as shown in Fig. 1(a). The key structural parameters of a PAA membrane include the interpore distance (Di),48 pore diameter (Dp),49 pore wall thickness (Tw),50 barrier layer thickness (Tb),51 pore channel length (Lp) which is also known as porous layer thickness (Tp),52 oxide layer thickness (To),53 pore density (ρ),54 and porosity (σ).55 Di, Dp, Tw, Tb, Lp, Tp and To are natural structural parameters, which can be measured and marked on a PAA directly, as shown in Fig. 1(a) and (c). ρ and σ are artificially defined structural parameters, which cannot be measured and marked on a PAA directly, and need to be calculated from the natural structural parameters. Every structural parameter is closely related to the capabilities of a PAA in various applications. For example, when a PAA is used for filtration, Dp will affect its filtration capability for particles with different sizes, and ρ will influence its filtration efficiency in a unit time. If a PAA is used as a template to assist the growth of various nanomaterials, Lp will directly determine the morphological characteristics of the final materials, which are nanodots, nanorods or nanowires. Thus, the accurate calculations for the PAA structural parameters are very important for the design and application of PAA structures. Depending on the accurate calculations, a PAA not only can be preliminarily designed to be the target structure according to the practical requirement before preparation but also can be directly applied after preparation without structural parameter measurements that would otherwise destroy or pollute the PAA membranes.
 |
| | Fig. 1 The schematics of a PAA structure and the marking of the PAA structural parameters: (a) perspective view, (b) top view, (c) cross-sectional view. | |
This paper summarizes various calculation formulas for each PAA structural parameter. To ensure objectivity and accuracy, this paper tests and contrasts the calculation result difference for each formula by the method of the mean square error (MSE) according to the experimental data of references, which have been published by other researchers. Based on a series of competitive screening, the optimal calculation formula of each PAA structural parameter is identified. All of the optimal calculation formulas are organized in the equation set of the PAA structural parameter calculation with a unified independent variable. The proposal of the equation set for the PAA structural parameter calculation will provide a comprehensive, systematic and accurate theoretical model and mathematical tool for predicting, designing and realizing PAA structural parameters to meet specific demands in practical applications.
2 Interpore distance (Di)
Di is the first natural structural parameter of a PAA. It is one of the two most important structural parameters, which directly or indirectly influences many other structural parameters. Di limits the maximum value of Dp and directly determines ρ, and is one of the determining factors of Tw and σ. It is well known that Di increases with increasing anodization voltage (U). Research groups have proposed different opinions to calculate Di in relation to U. Leszek et al. proposed a 2.5 nm V−1 proportional relationship between Di and U, as shown in formula (1):56
Thompson et al. proposed a 2.77 nm V−1 proportional constant for the dependence of Di on U, as shown in formula (2):57
Hélio et al. proposed a 2.8-times proportional relationship between Di and U, as shown in formula (3):58
Sun-Kyu et al. proposed a Di calculation formula that is suitable for oxalic acid anodization, as shown in eqn (4):59
| |
Di = 2.75U − 5.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉 〈oxalic〉
| (4) |
Ebihara et al. proposed another Di piecewise calculation formula suitable for oxalic acid anodization, which contains two different formulas corresponding to two different voltage intervals, as shown in formula (5):60
| |
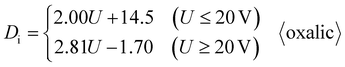 | (5) |
Furthermore, there is a Di calculation formula that is suitable for sulfuric acid anodization, as shown in eqn (6):61
| |
Di = 1.99U + 12.1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉 〈sulfuric〉
| (6) |
Eqn (1)–(6) are all formulas that can be used to calculate Di with the same independent variable U, but possess different mathematical expressions. Consequently, their calculation results differ from one another. In that case, which formula is the most accurate one? How does one choose and apply the Di calculation formulas presented above? These are questions that must be addressed in practical applications. In order to clarify the above questions, extensive experimental (U, Di) data published by other researchers (but not the authors of this paper) are substituted into formulas (1)–(6) to objectively test and compare the accuracy of formulas (1)–(6). Then, the MSE between each Di formula calculation results and the corresponding experimental measurement Di data is calculated using the MSE formula, as shown in eqn (7):
| |
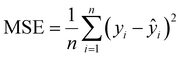 | (7) |
where MSE is the mean square error,
yi is the
i-th experimental measurement value,
ŷi is the
i-th calculation result, and
n is the number of research samples. A smaller MSE between the experimental measurement values and the calculation results indicate a more accurate formula.
Table S1 presents the experimental measurement data of Di and the corresponding U from different ref. 27, 56 and 62–72. The (U, Di) data point from the Table S1 is plotted as the black dots shown in Fig. 2(a). In this figure, the linear regressions of formulas (1)–(6) are shown as the red, orange, yellow, green, cyan and blue curve, respectively. In Fig. 2(a), visually, the linear regression of formula (1) appears to agree better for the experimental data than the other formulas when the anodization voltages are higher than 80 V. However, the differences between the experimental data and the linear regressions of formulas (1)–(6) are less clear and not easily discernible at voltages below 80 V. In order to quantitatively evaluate the errors between the experimental data and the calculation results from formulas (1)–(6), the U data from Table S1 were substituted into formulas (1)–(6), and the MSE between the results of each formula and the corresponding experimental measurement Di data were calculated.
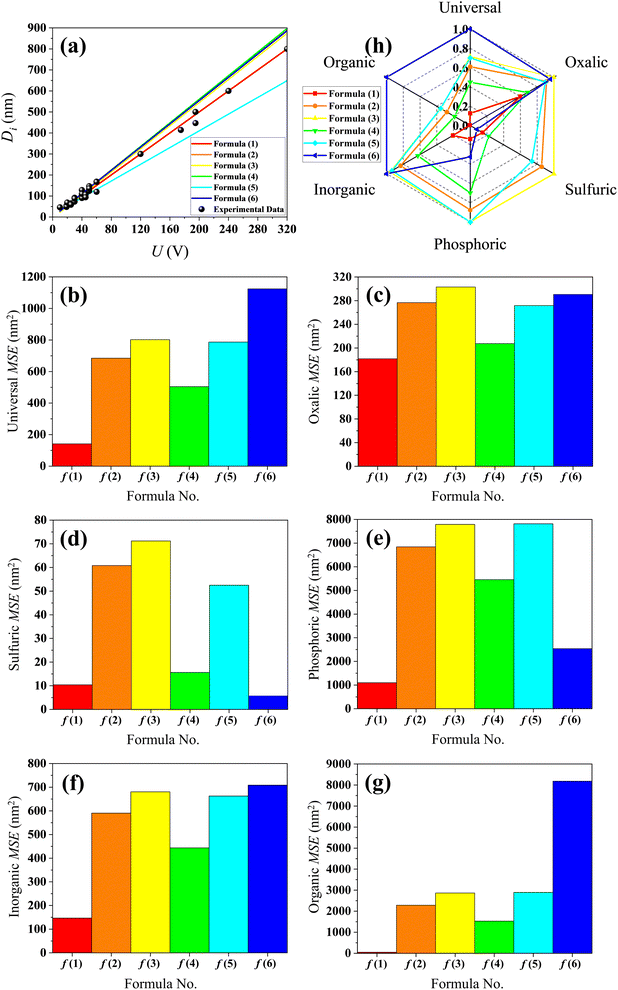 |
| | Fig. 2 Contrastive research between the calculation results of formulas (1)–(6) and the experimental data. (a) Experimental data dots of Table S1 and the linear regression of formulas (1)–(6). (b) Histogram of MSE when the electrolyte type is not specified. (c) Histogram of MSE when the electrolyte is specified as only oxalic acid. (d) Histogram of MSE when the electrolyte is specified as only sulfuric acid. (e) Histogram of MSE when the electrolyte is specified as only phosphoric acid. (f) Histogram of MSE when the electrolyte is specified as inorganic acid. (g) Histogram of MSE when the electrolyte is specified as an organic acid. (h) Normalized radar plot for MSE of formulas (1)–(6) in universal and oxalic, sulfuric, phosphoric, inorganic, organic acid anodization. | |
All the data from Table S1 were respectively substituted in formulas (1)–(6). The MSE values of the six formulas are shown in Fig. 2(b) and the ‘universal’ row of Table 1. Taken together, these results demonstrate that formula (1) is the best equation as the universal Di calculation formula because it has the smallest MSE among formulas (1)–(6), when the electrolyte type is not specially considered. When all the oxalic acid anodization data from Table S1 are substituted into formulas (1)–(6), the resulting MSE values of the six formulas shown in Fig. 2(c) and the ‘oxalic’ row of Table 1 indicate that formula (1) is the best Di calculation equation for oxalic acid anodization if the electrolyte is specified as only oxalic acid. When all the sulfuric acid anodization data of Table S1 are substituted into formulas (1)–(6), the MSE values of the six formulas shown in Fig. 2(d) and the ‘sulfuric’ row of Table 1 demonstrate that formula (6) is the best Di calculation equation for sulfuric acid anodization if the electrolyte is specified as only sulfuric acid. In the event that all the phosphoric acid anodization data of Table S1 are substituted into formulas (1)–(6), the MSE values of the six formulas shown in Fig. 2(e) and the ‘phosphoric’ row of Table 1 display that formula (1) is the best Di calculation equation for phosphoric acid anodization if the electrolyte is specified as only phosphoric acid. In the event that all the inorganic acid anodization data of Table S1 are substituted into formulas (1)–(6), the MSE values of the six formulas shown in Fig. 2(f) and the ‘inorganic’ row of Table 1 show that formula (1) is the best Di calculation equation that is suitable for inorganic acid anodization when the electrolyte is various inorganic acids. In the event that all the organic acid anodization data of Table S1 are substituted into formulas (1)–(6), the MSE values for the six formulas shown in Fig. 2(g) and the ‘organic’ row of Table 1 reveal that formula (1) is the best Di calculation equation that is suitable for organic acid anodization when the electrolyte is various organic acids. Fig. 2(h) is the normalized radar plot of the MSE values for formulas (1)–(6) in universal and oxalic, sulfuric, phosphoric, inorganic, organic acid anodization, respectively. This visualization allows for an easy comparison on the competitive rankings on MSE values from formulas (1)–(6). For example, along the inorganic axis, the normalized MSE values indicate the following accuracy ranking of inorganic acid anodization from best to worst: formula (1) (red curve) > formula (4) (green curve) > formula (2) (orange curve) > formula (5) (cyan curve) > formula (3) (yellow curve) > formula (6) (blue curve). Beyond analyzing the individual axes, the total area enclosed by each curve on the radar plot provides a measure of a formula's overall universality. Formula (1) with the smallest area (red curve) is the most universally accurate across all electrolyte types. This method is different from the above method, according to the direction and normalized MSE value to estimate, but it obtains the same conclusion.
Table 1 MSE of the calculation results from formulas (1)–(6) against the experimental measurement Di data of Table S1
| Electrolyte type |
Mean squared error |
| f (1) |
f (2) |
f (3) |
f (4) |
f (5) |
f (6) |
| Universal |
141.6 |
684.6 |
801.8 |
504.0 |
786.8 |
1124.1 |
| Oxalic |
181.7 |
276.6 |
303.1 |
207.5 |
271.9 |
290.6 |
| Sulfuric |
10.4 |
60.8 |
71.2 |
15.6 |
52.5 |
5.7 |
| Phosphoric |
1096.3 |
6841.2 |
7788.5 |
5457.3 |
7817.1 |
2536.6 |
| Inorganic |
146.8 |
590.4 |
680.5 |
443.8 |
662.8 |
708.9 |
| Organic |
52.1 |
2286.9 |
2865.3 |
1527.4 |
2894.9 |
8182.3 |
Section summary: According to a series of competitive screening based on numerous experimental data from various references, the existing optimal Di calculation formulas are verified as follows:
| |
Di = 2.5U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈universal〉 〈universal〉
| (1) |
| |
Di = 2.5U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉 〈oxalic〉
| (1) |
| |
Di = 1.99U + 12.1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉 〈sulfuric〉
| (6) |
| |
Di = 2.5U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈phosphoric〉 〈phosphoric〉
| (1) |
| |
Di = 2.5U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈inorganic〉 〈inorganic〉
| (1) |
| |
Di = 2.5U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈organic〉 〈organic〉
| (1) |
Eqn (1) is the optimal Di calculation formula in almost all cases. The only exception occurs when sulfuric acid is specified as the only electrolyte, with eqn (6) performing slightly better than eqn (1), albeit by a small margin.
3 Pore diameter (Dp)
Dp is the second natural structural parameter of a PAA, and one of the two most important PAA structural parameters. The nanoscale-size, self-organized, well-ordered nanopores of a PAA are adjustable objectively.28 This property makes the PAA membranes become one of the most popular templates for the growth of nanodots,73 nanorods,74 nanopillars,75 nanowires,76 and nanotubes,77 which exhibit special performances such as ferroelectric multi-domains,73 SERS,74 anti-biofilm,75 photocatalysis,76 and wave absorption.77
For a PAA, the maximum Dp value is limited by Di, but the born value of Dp is not directly determined by Di. Specifically, there are two kinds of Dp values for describing the size of PAA nanopores: the born value of Dp (Dp-born) and the pore-widening value of Dp (Dp-widening). Dp-born is determined by the anodization voltage during PAA fabrication. It is the initial Dp value after PAA anodization without pore-widening treatments. Dp-widening is the Dp value after pore-widening treatments on the foundation of Dp-born. It is a post-processing parameter, but not an original parameter. After anodization is complete, the Dp-widening can be easily and irreversibly adjusted to be any value from the Dp-born to the maximum (Di) by pore-widening treatments. It should be emphasized that in this paper, all of the mentioned Dp refer to Dp-born, but not Dp-widening.
It is generally considered that the anodization voltage has the most dramatic effect on Dp. Dp increases as the anodization voltage increases. Some research suggests that PAA nanopore formation is accompanied by volume expansion at the metal–oxide interface. This volume expansion is given by the Pilling–Bedworth ratio (PBR), which is expressed as eqn (8):78
| |
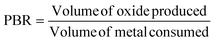 | (8) |
Due to volume expansion, the oxide is pushed in tangential and upward directions, moving the oxide walls upward, thereby increasing the height of the pore wall. A higher voltage is associated with a higher current density, which leads to a higher volume expansion, resulting in more oxide being pushed in both tangential and upward directions. Consequently, PAA pore walls will be squeezed more by the higher voltage, thereby achieving a larger Dp.
Due to the powerful influence of voltage on Dp, many research groups have proposed their own Dp calculation formulas in different mathematic expressions with U as the independent variable. Leszek et al. reported a proportionality constant of 0.9 nm V−1 between Dp and U, and the corresponding formula is shown in eqn (9):56
O'Sullivan and Wood presented a relationship between Dp and U with a correlation constant of 1.29 nm V−1, as given in formula (10):79
Palibroda et al. reported the dependence of the PAA pore diameter on the anodization voltage, as given in formula (11):80
In contrast to the above three linear formulas
(9)–(11), Alaa
et al. proposed a nonlinear
Dp calculation formula, as given in
eqn (12):
81| |
Dp = 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e0.02U e0.02U
| (12) |
Faced with the various Dp calculation formulas with different mathematical expressions, the method for confirming the optimal Dp calculation formula is the same as the competitive screening described above for the optimal Di calculation formula. Extensive experimental (U, Dp) data published by other researchers were substituted into formulas (9)–(12) to objectively test and contrast their accuracy. Then, the MSE values between each Dp formula calculation result and the corresponding experimental measurement Dp data are calculated. A smaller MSE between the experimental measurement values and calculation results indicates that the formula is more accurate.
Table S2 lists the experimental measurement data of Dp and the corresponding U from different ref. 27, 56, 62, 65–68, 70, 78 and 82–89. The (U, Dp) data points of Table S2 are shown as the black dots in Fig. 3(a), while the regression lines of formulas (9)–(12) are shown as the red, orange, green and blue curves, respectively. In Fig. 3(a), visually, the regression line of formula (11) is in better agreement with the experimental data than the other formulas when the anodization voltages are higher than 80 V. However, when the anodization voltages are lower than 80 V, the contrast between the experimental data and regression line of formulas (9)–(12) is not clear and not visually observed. In order to separately quantify the errors between the experimental data and the calculation results of formulas (9)–(12), the U data of Table S2 are substituted into formulas (9)–(12), and the MSE values between the results of each formula and the corresponding experimental measurement Dp data are then calculated.
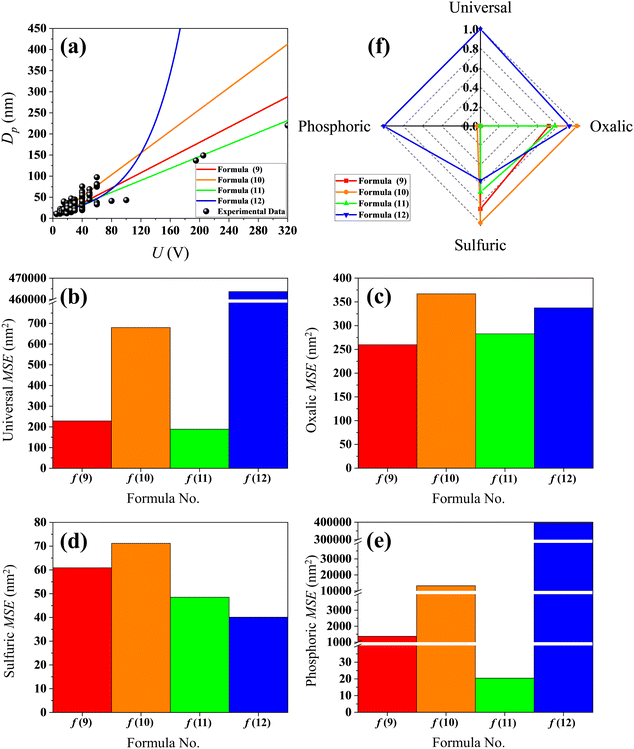 |
| | Fig. 3 Contrastive research between the calculation results of formulas (9)–(12) and the experimental data. (a) Experimental data dots of Table S2 and linear regression of formulas (9)–(12). (b) Histogram of MSE when the electrolyte type is not specified. (c) Histogram of MSE when the electrolyte is specified as only oxalic acid. (d) Histogram of MSE when the electrolyte is specified as only sulfuric acid. (e) Histogram of MSE when the electrolyte is specified as only phosphoric acid. (f) Normalized radar plot for MSE of formulas (9)–(12) in universal and oxalic, sulfuric, phosphoric acid anodization. | |
In the event that all the data of Table S2 are respectively substituted in formulas (9)–(12), the MSE values of the four formulas shown in Fig. 3(b) and the ‘universal’ row of Table 2 demonstrate that formula (11) is the best equation as the universal Dp calculation formula because of its smallest MSE among formulas (9)–(12), when the electrolyte kind is not specially considered. In the event that all the oxalic acid anodization data of Table S2 are substituted into formulas (9)–(12), the MSE values of the four formulas shown in Fig. 3(c) and the ‘oxalic’ row of Table 2 indicate that formula (9) is the best Dp calculation equation for oxalic acid anodization because of its smallest MSE, if the electrolyte is specified as only oxalic acid. In the event that all the sulfuric acid anodization data of Table S2 are substituted into formulas (9)–(12), the MSE values of the four formulas shown in Fig. 3(d) and the ‘sulfuric’ row of Table 2 indicate that formula (12) is the best Dp calculation equation for sulfuric acid anodization because of its smallest MSE if the electrolyte is specified as only sulfuric acid. In the event that all the phosphoric acid anodization data of Table S2 are substituted into formulas (9)–(12), the MSE values of the four formulas shown in Fig. 3(e) and the ‘phosphoric’ row of Table 2 show that formula (11) is the best Dp calculation equation for phosphoric acid anodization because of its smallest MSE if the electrolyte is specified as only phosphoric acid. Fig. 3(f) is the normalized radar plot for the MSE values of formulas (9)–(12) in universal and oxalic, sulfuric, phosphoric acid anodization. It exhibits the competitive ranking for the MSE values of formulas (9)–(12) in different electrolyte anodizations at a glance. The green closed curve surrounds the smallest area, which also indicates that formula (11) is the best equation for the universal calculation of Dp from a holistic view. The area method is different from the method of estimating based on the direction and MSE value of each axis, yet both approaches lead to the same conclusion.
Table 2 MSE of the calculation results from formulas (9)–(12) against the experimental measurement Dp data of Table S2
| Electrolyte type |
Mean squared error |
| f (9) |
f (10) |
f (11) |
f (12) |
| Universal |
228.8 |
679.7 |
189.02 |
463![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 669.0 669.0 |
| Oxalic |
259.7 |
367.1 |
282.8 |
337.3 |
| Sulfuric |
60.9 |
71.2 |
48.5 |
40.1 |
| Phosphoric |
1371.3 |
13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 225.2 225.2 |
20.4 |
395![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 853.4 853.4 |
It should be emphasized that the existing mainstream Dp calculation formulas are primarily expressed as single-variable functions of U, but voltage is not the sole factor influencing Dp. From the perspective of anodization conditions, besides voltage, many other factors can influence Dp to different extents, such as the anodization temperature, electrolyte concentration and electrolyte type.83 By focusing exclusively on voltage but ignoring other factors, the existing Dp formulas inevitably introduce errors, the extent of which varies across different complex anodization conditions. For example, the Dp distribution is notably broad at lower voltages in Fig. 3(a). The reason lies not in the lower voltage itself, but in the inherent limitation of the existing Dp formulas, which rely on voltage as their sole independent variable. The solution lies in systematically conducting a quantitative investigation into more influence factors on Dp, such as the anodization temperature, electrolyte concentration, electrolyte type and anodization duration, rather than focusing only on the voltage. Then, a new Dp calculation formula can be proposed with multiple independent variables that incorporate all key influencing factors, to replace all existing formulas that rely solely on voltage. The aforementioned issue exists not only in the Dp calculation formula, but more broadly in both the already-discussed Di and the various structural parameters to be discussed in subsequent chapters. This currently remains a significant research gap and a crucial research topic for future studies.
In addition, it is well known that the growth of PAA pores is a multi-mechanism process. The size of Dp is determined by the combined effect of pore growth and acid electrolyte corrosion acting simultaneously. Unfortunately, none of the existing Dp calculation formulas are derived from a separate, quantitative study of the pore growth mechanism and the acid electrolyte corrosion mechanism. Therefore, none of the existing formulas can be used to independently explain the impact of either the pore growth mechanism or the acid electrolyte corrosion effect on Dp. Any existing Dp calculation formula serves to calculate the aggregate result arising from all factors that influence Dp. Separating and quantitatively studying the individual effects of the pore growth mechanism and the acid electrolyte corrosion on Dp remains an outstanding research gap at present and a crucial research topic for future studies.
Section summary: according to a series of competitive screening based on numerous experimental data from other references, the existing optimal Dp calculation formulas are verified as follows:
| |
Dp = 0.709U + 4.986![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈universal〉 〈universal〉
| (11) |
| |
Dp = 0.9U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉 〈oxalic〉
| (9) |
| |
Dp = 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e0.02U e0.02U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉 〈sulfuric〉
| (12) |
| |
Dp = 0.709U + 4.986![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈phosphoric〉 〈phosphoric〉
| (11) |
Eqn (11) is the universal Dp calculation formula for common cases. In the case of one of the two exceptions, when oxalic acid is specified as the only electrolyte, eqn (9) is slightly better than eqn (11) with a small advantage. For the other exception, when sulfuric acid is specified as the only electrolyte, eqn (12) is slight better than eqn (11) with a small advantage.
4 Pore wall thickness (Tw)
Pore wall thickness is the third natural structural parameter of a PAA. It affects the mechanical strength of PAA membranes. A thicker pore wall thickness is indicative of a more robust and free-standing PAA membrane. It is widely recognized that in the perfect hexagonal arrangement of pores, Tw can be calculated by formula (13):90| |
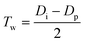 | (13) |
where Di is the interpore distance with the unit of nm, and Dp is the (born) pore diameter with the unit of nm. In formula (13), there are two independent variables, Di and Dp. According to the above discussions about Di and Dp, both Di and Dp are functions of U. Thus, Tw also can be derived to be a function of U. Therefore, if substituting the above optimal Di and Dp calculation formulas into formula (13), the optimal Tw calculation formulas as a function of U can be obtained.
After substituting the universal Di calculation formula (1) and the universal Dp calculation formula (11) into formula (13), the universal Tw calculation formula is obtained, as shown in eqn (14):
| |
Tw = 0.8955U − 2.493![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈universal〉 〈universal〉
| (14) |
After substituting oxalic Di calculation formula (1) and oxalic Dp calculation formula (9) into (13), the optimal Tw calculation formula specific to oxalic acid anodization is obtained, as shown in eqn (15):
| |
Tw = 0.8U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉 〈oxalic〉
| (15) |
After substituting sulfuric Di calculation formula (6) and sulfuric Dp calculation formula (12) into (13), the optimal Tw calculation formula specific to sulfuric acid anodization is obtained, as shown in eqn (16):
| |
Tw = 0.995U − 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e0.02U + 6.05 e0.02U + 6.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉 〈sulfuric〉
| (16) |
Based on derivations, the optimal Tw calculation formulas specific to phosphoric acid anodization are the same as the universal formula (14).
Section summary: according to a series of derivations, the existing optimal Tw calculation formula with U as the only independent variable is shown as follows:
| |
Tw = 0.8955U − 2.493![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈universal〉 〈universal〉
| (14) |
| |
Tw = 0.8U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉 〈oxalic〉
| (15) |
| |
Tw = 0.995U − 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e0.02U + 6.05 e0.02U + 6.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉 〈sulfuric〉
| (16) |
| |
Tw = 0.8955U − 2.493![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈phosphoric〉 〈phosphoric〉
| (14) |
Depending on the above optimal Tw calculation formulas for different electrolytes, the Tw of a PAA can be calculated directly from U, rather than first calculating Di and Dp separately.
5 Barrier layer thickness (Tb)
Applications of PAA structures are limited sometimes, such as when using PAA nanochannels as templates to grow nanowires by electrochemical deposition. As reported, in a system of electrochemical deposition, one of the most critical limitations is the non-conductive property of the PAA barrier layer. The high electrical resistance generated by the barrier layer isolates the metallic base from the electrochemical deposition bath. In other words, an electrochemical contact would be necessary for electrochemical deposition inside the PAA pores. For reasons similar to the above mentioned, barrier layer removal is very important for actual PAA applications in some special fields. Hence, in order to remove a barrier layer accurately with as little damage as possible to the PAA nanochannel structure, the Tb value of a PAA needs be predicted and calculated precisely in practice.
Tb is the fourth natural structural parameter of a PAA. Digby described PAA barrier layer growth using the point defect model.91 Based on this model, oxygen ion vacancies diffuse from the oxide–metal interface to solution–oxide interface. In contrast, aluminum ion vacancies diffuse from the solution–oxide interface to oxide–metal interface. The key contribution to the PAA barrier layer growth is supplied by the oxygen anion. It is demonstrated that about 60% of Tb is formed at the oxide–metal interface by migration of O2− and OH− ions inwards, and the remaining 40% of Tb is formed at the solution–oxide interface by migration of Al3+ ions outwards.92
It is well documented that Tb is proportional to U. However, different researchers have proposed different values for the proportionality coefficient between Tb and U. Lee and Park reported that Tb increases with U, as shown in formula (17):93
Thompson et al. proposed a 1.05 nm V−1 ratio relationship between Tb and U, as shown in formula (18):57
Kjyohito et al. gave a 1.1 nm V−1 proportionality coefficient between Tb and U, as shown in formula (19):94
Sachiko et al. defined a 1.14 nm V−1 ratio between Tb and U, as shown in formula (20):95
Sousa et al. presented a 1.3 nm V−1 phenomenological constant between Tb and U, as shown in formula (21):96
Dmitri et al. claimed the anodizing ratio of 1.4 nm V−1 to determine Tb by U, as shown in formula (22):97
In addition to the linear relationship between
Tb and
U,
Tw and
Di have also been reported as the independent variables that are needed to calculate
Tb. In the opinion of Ebihara
et al.,
Tb formed during oxalic or sulfuric acid anodization is different and can be calculated by
Tw, as respectively shown in formulas
(23) and
(24):
56| |
Tb = 1.12 × Tw![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉 〈oxalic〉
| (23) |
| |
Tb = 1.33 × Tw![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉 〈sulfuric〉
| (24) |
where
Tw is the pore wall thickness with the unit of nm. Nielsch
et al. suggested that
Tb is proportional to
Di, as shown in formula
(25):
98| |
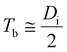 | (25) |
where
Di is the interpore distance with the unit of nm.
Apparently, the Tb calculation formulas of eqn (17)–(25) have different independent variables: U is the independent variable of formulas (17)–(22), Tw is the independent variable of formulas (23) and (24), and Di is the independent variable of formula (25). It is difficult to directly contrast the accuracies of all the formulas. However, formulas (23)–(25) can be reformulated to use U as their independent variables. This is possible because both Tw and Di are functions of U in essence, according to the previous discussions. Thus, when the oxalic Tw calculation formula (15) is substituted into formula (23), a new Tb calculation formula with U as its independent variable suitable for oxalic acid anodization is obtained, as shown in eqn (26):
| |
Tb = 0.896U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉 〈oxalic〉
| (26) |
Substituting the sulfuric Tw calculation formula (16) into (24) yields a new Tb calculation formula with U as its independent variable suitable for sulfuric acid anodization, as shown in eqn (27):
| |
Tb = 1.32335U − 9.31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e0.02U + 8.0465 e0.02U + 8.0465![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉 〈sulfuric〉
| (27) |
When the universal Di calculation formula (1) is substituted into formula (25), a new universal Tb calculation formula with U as its independent variable is obtained, as shown in eqn (28):
| |
Tb = 1.25U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈universal〉 〈universal〉
| (28) |
Substituting the sulfuric Di calculation formula (6) into (25) yields a new Tb calculation formula with U as its independent variable specific to sulfuric acid anodization, as shown in eqn (29):
| |
Tb = 0.995U + 6.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉 〈sulfuric〉
| (29) |
After all the independent variables of formulas (23)–(25) are unified by U to obtain formulas (26)–(29), all the new formulas have the same independent variable as formulas (17)–(22). Thus, the accuracy of formulas (17)–(22) and (26)–(29) can be tested and compared together. For competitively screening the optimal Tb calculation formula, numerous experimental (U, Tb) data published by other researchers are substituted into formulas (17)–(22) and (26)–(29). Then, the MSE values between each Tb formula calculation result and the corresponding experimental measurement Tb data are calculated to objectively test and contrast the accuracy of formulas (17)–(22) and (26)–(29). A smaller MSE value indicates that the formula is more accurate.
Table S3 presents experimental measurement data of Tb and the corresponding U from different ref. 56, 62, 93 and 99–104. The (U, Tb) data points of Table S3 are shown as the black dots in Fig. 4(a), and the regression lines of formulas (17)–(22) and (26)–(29) are shown as the black, pink, red, orange, yellow, green, olive, cyan, blue and violet curves, respectively. In order to separately quantify the errors between the experimental data and the calculation results of formulas (17)–(22) and (26)–(29), the U data of Table S3 are substituted into formulas (17)–(22) and (26)–(29). Then, the MSE between the result of each formula and the corresponding experimental measurement Tb data is calculated.
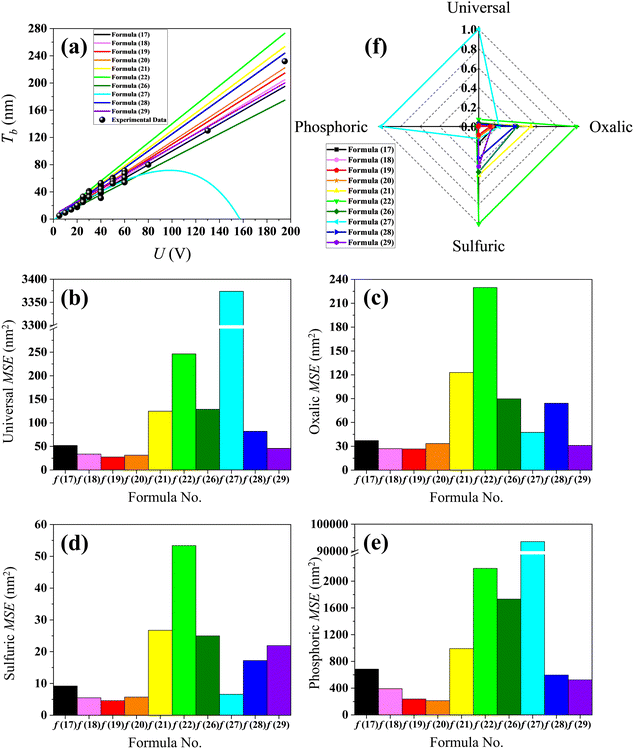 |
| | Fig. 4 Contrastive research between the calculation results of formulas (17)–(22) and (26)–(29) and the experimental data. (a) Experimental data dots of Table S3 and linear regression of formulas (17)–(22) and (26)–(29). (b) Histogram of MSE when the electrolyte type is not specified. (c) Histogram of MSE when the electrolyte is specified as only oxalic acid. (d) Histogram of MSE when the electrolyte is specified as only sulfuric acid. (e) Histogram of MSE when the electrolyte is specified as only phosphoric acid. (f) Normalized radar plot for MSE of formulas (17)–(22) and (26)–(29) in universal and oxalic, sulfuric and phosphoric acid anodization. | |
In the event that all the data of Table S3 are respectively substituted in formulas (17)–(22) and (26)–(29), the MSE values of the ten formulas shown in Fig. 4(b) and the ‘universal’ row of Table 3 demonstrate that formula (19) is the best equation as the universal Tb calculation formula due to its minimal MSE among formulas (17)–(22) and (26)–(29), when the electrolyte is not specially considered. In the event that all the oxalic acid anodization data of Table S3 are substituted into formulas (17)–(22) and (26)–(29), the MSE values of the ten formulas shown in Fig. 4(c) and the ‘oxalic’ row of Table 3 indicate that formula (19) is the best Tb calculation equation specific to oxalic acid anodization, if the electrolyte is specified as only oxalic acid. In the event that all the sulfuric acid anodization data of Table S3 are substituted into formulas (17)–(22) and (26)–(29), the MSE values of the ten formulas shown in Fig. 4(d) and the ‘sulfuric’ row of Table 3 exhibit that formula (19) is the best Tb calculation equation specific to sulfuric acid anodization, if the electrolyte is specified as only sulfuric acid. In the event that all the phosphoric acid anodization data of Table S3 are substituted into formulas (17)–(22) and (26)–(29), the MSE values of the ten formulas shown in Fig. 4(e) and the ‘phosphoric’ row of Table 3 show that formula (20) is the best Tb calculation equation specific to phosphoric acid anodization, if the electrolyte is specified as only phosphoric acid. Fig. 4(f) is the normalized radar plot for the MSE of formulas (17)–(22) and (26)–(29) in universal and oxalic, sulfuric and phosphoric acid anodization. It exhibits the competitive ranking for the MSE of (17)–(22) and (26)–(29) in different electrolyte anodization at a glance. Furthermore, the red closed curve surrounds the smallest area, which also indicates that formula (19) is the best equation for the universal calculation of Tb from a holistic view. This area-based evaluation method differs from the estimating performance based on the MSE value of each axis, yet both approaches lead to the same conclusion.
Table 3 MSE of the calculation results from formulas (17)–(22) and (26)–(29) against the experimental measurement Tb data of Table S3
| Electrolyte type |
Mean squared error |
| f (17) |
f (18) |
f (19) |
f (20) |
f (21) |
f (22) |
f (26) |
f (27) |
f (28) |
f (29) |
| Universal |
51.8 |
33.6 |
27.5 |
31.4 |
124.7 |
246.3 |
128.7 |
3373.8 |
82.1 |
45.9 |
| Oxalic |
37.2 |
27.1 |
26.7 |
33.4 |
122.9 |
229.8 |
89.7 |
47.5 |
84.2 |
31.1 |
| Sulfuric |
9.2 |
5.5 |
4.6 |
5.7 |
26.7 |
53.4 |
25.0 |
6.6 |
17.2 |
21.9 |
| Phosphoric |
684.5 |
392.4 |
237.6 |
212.7 |
991.6 |
2192 |
1731 |
93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 533 533 |
597.1 |
524.2 |
Section summary: according to a series of competitive screening based on numerous experimental data from various other references, the existing optimal Tb calculation formulas are verified as follows:
| |
Tb = 1.1U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈universal〉 〈universal〉
| (19) |
| |
Tb = 1.1U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉 〈oxalic〉
| (19) |
| |
Tb = 1.1U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉 〈sulfuric〉
| (19) |
| |
Tb = 1.14U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈phosphoric〉 〈phosphoric〉
| (20) |
Formula (19) is the optimal Tb calculation formula in almost all cases. The only exception is for phosphoric acid anodization, where formula (20) performs slightly better.
6 Pore channel length (Lp), also known as the porous layer thickness (Tp)
The pore channel length is the porous layer thickness of a PAA. Thus, Lp and Tp are the same structural parameter. They are both the fifth natural structural parameter of a PAA. Lp and Tp are closely related with practical applications of through-hole PAA. A through-hole PAA can be obtained by opening-hole process, which is typically carried out in a phosphoric acid solution.93 During the opening-hole process, the barrier layer of a PAA membrane is immersed in a phosphoric acid solution and subjected to chemical etching until removed completely.
Lp and Tp are very important parameters for through-hole PAA applications in different fields. A thick Tp is an advantageous property for separation applications, because it helps to maintain the mechanical integrity of through-hole PAA filter membranes during processing, handling and filtering. This allows a through-hole PAA to tolerate a higher pressure-difference between two sides of a through-hole PAA filter membrane during the filtration process, thereby increasing the separation rate and efficiency. However, a short Lp and thin Tp are required when a through-hole PAA is used as a mask for sputtering deposition, because a Lp that is too long and a Tp that is too thick will prevent the deposited materials from smoothly reaching the target substrate. Therefore, accurate calculations for Lp and Tp of a PAA are very important for practical applications. Hwang et al. proposed that the Lp of a PAA is proportional to the anodization duration (D). The linear relationship can be formulated as eqn (30):59
| | |
Lp = 125.53D − 147.75
| (30) |
where
Lp is the pore channel length of PAA and is equal to
Tp in units of nm, and
D is the anodization duration in units of min. They also emphasized that the linear relationship cannot be maintained when the anodization is carried out for an excessively long duration. The subsequent research demonstrates that the thickness of a PAA is not in a strictly linear relationship with the anodization duration. The appearance of a limiting thickness will be observed when an anodization duration is excessive. In fact, the thickness of a PAA membrane is usually less than 200 µm in most of the literature reports. For a PAA membrane prepared with a short or moderate duration, its pore channel length can be calculated by formula
(30). However, for a PAA membrane prepared by an excessive anodization duration, formula
(30) is not accurate. A longer duration indicates a bigger error.
Section summary: The Lp and Tp calculation formula for a short or moderate anodization duration is eqn (30):
| | |
Lp = Tp = 125.53D − 147.75
| (30) |
where
Lp is the pore channel length in units of nm,
Tp is the porous layer thickness in units of nm,
D is the anodization duration in units of min. However, formula
(30) is not accurate for a PAA fabricated with an excessive anodization duration.
7 Oxide layer thickness (To)
To is the sixth natural structural parameter of a PAA. It is the total thickness of an integral PAA membrane and is, in fact, equal to the sum of Tb and Tp. Beyond traditional applications, research in the last decade has revealed that an intact PAA with a barrier layer can be used as nanofluidic devices by taking advantage of its ionic current rectification property, such as ultrasensitive capture,105 detection,106 and controlled release of drugs107 have been demonstrated. The To calculation formula is given by eqn (31):where To is the oxide layer thickness in units of nm, Tb is the barrier layer thickness in units of nm, and Tp is the porous layer thickness in units of nm. Since Tb is the function of U, as shown as formulas (19) and (20), and Tp is the function of D, as shown as formula (30), formula (31) can be further derived to be a new To calculation formula with U and D as the independent variables. After substituting formulas (19) and (30) into (31), a new To calculation formula with anodization conditions as independent variables suitable for universal and oxalic, sulfuric acid anodization is obtained and expressed as eqn (32):| | |
To = 1.1U + 125.53D − 147.75
| (32) |
After substituting formulas (20) and (30) into (31), the new To calculation formula with anodization conditions as independent variables suitable for phosphoric anodization is obtained and expressed as eqn (33):
| | |
To = 1.14U + 125.53D − 147.75
| (33) |
Section summary: according to a series of derivations, the To calculation formulas with anodization conditions as independent variables are as follows:
| |
To = 1.1U + 125.53D − 147.75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈universal〉 〈universal〉
| (32) |
| |
To = 1.1U + 125.53D − 147.75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉 〈oxalic〉
| (32) |
| |
To = 1.1U + 125.53D − 147.75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉 〈sulfuric〉
| (32) |
| |
To = 1.14U + 125.53D − 147.75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈phosphoric〉 〈phosphoric〉
| (33) |
Eqn (32) is the optimal To calculation formula in almost all cases. The only exception is when phosphoric acid is specified as the only electrolyte; in this case, eqn (33) is slightly better than eqn (32).
8 Pore density (ρ)
ρ is the seventh PAA structural parameter, which is not a natural one but is artificially defined. For a well-ordered PAA with a standard hexagonal lattice, ρ is defined as the total number of pores on 1 cm2 of surface area.65 The formula of ρ is derived according to the definition of the pore density, given by eqn (34):65| |
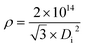 | (34) |
where ρ is the pore density in units of cm−2, and Di is the interpore distance in units of nm.
The derivation process of eqn (34) is as follows. There must be one and only one pore in the center of each hexagonal lattice cell of a PAA, as shown in the PAA surface schematic (Fig. 5). This means that the number of pores and lattice cells is equal on a PAA. Therefore, the PAA pore density is essentially the lattice cell density of a PAA. The lattice cell density equals 1 cm2 divided by the surface area of one hexagonal lattice cell, and this area can be calculated using Di. Di is the distance between two adjacent nanopore centers, as shown by the red line connecting lattice cells a and b in Fig. 5. It is equivalent to the distance between two parallel sides of a hexagonal lattice cell, as shown by the red line in lattice cell c of Fig. 5. A standard hexagonal lattice cell can be equally divided into six equilateral triangles, as shown by the six olive triangles in lattice cell d of Fig. 5. This means that the area of a lattice cell is six times the area of an equilateral triangle. As shown by the olive triangle in lattice cell e of Fig. 5, the area of an equilateral triangle is given by eqn (35):
| |
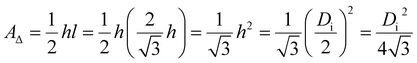 | (35) |
 |
| | Fig. 5 Schematic of PAA surface. | |
The area of a hexagonal lattice cell is given by eqn (36):
| |
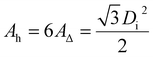 | (36) |
According to the definition of pore density, the ρ (which is the lattice cell density, in fact) calculation formula is obtained and given by eqn (37):
| |
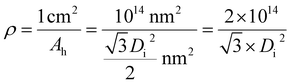 | (37) |
Going a step further, since Di is the function of U as shown in formulas (1) and (6), formula (37) can be further derived as two new ρ calculation formulas with U as the independent variable by substituting formulas (1) and (6) into it. After substituting the optimal universal Di calculation formula (1) into formula (37), the universal ρ calculation formula is obtained, as shown in eqn (38):
| |
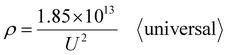 | (38) |
After substituting the Di calculation formula (6) specific to sulfuric acid anodization into formula (37), the ρ calculation formula specific to sulfuric acid anodization is obtained, as shown in eqn (39):
| |
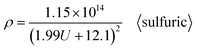 | (39) |
Based on further derivation, eqn (38) is also the optimal calculation formula for oxalic, phosphoric, inorganic, and organic acid anodization.
Section summary: According to a series of derivations, the existing optimal ρ calculation formulas with U as the independent variable are shown as follows:
| |
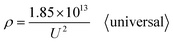 | (38) |
| |
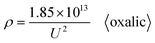 | (38) |
| |
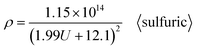 | (39) |
| |
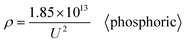 | (38) |
| |
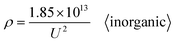 | (38) |
| |
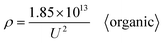 | (38) |
Eqn (38) is the optimal ρ calculation formula in almost all cases. The only exception is for sulfuric acid anodization, where formula (39) offers a slight advantage over formula (38).
9 Porosity (σ)
σ is the eighth PAA structural parameter, which is also a second artificially defined parameter. It is defined as the ratio of the total surface area occupied by all pores to the total surface area of a PAA. It is a derived structural parameter, which is calculated from Di and Dp. For a well-ordered PAA with a closely packed hexagonally arranged lattice of pores without widening hole treatment, σ can be calculated by formula (40)::27,61,64,65| |
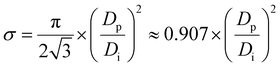 | (40) |
where σ is the porosity and a dimensionless parameter, Di is the interpore distance in units of nm, and Dp is the pore diameter in units of nm.
The derivation process of eqn (40) is as follows. There must be one and only one pore in the center of each hexagonal lattice cell of a PAA, as shown in the PAA surface schematic of Fig. 5. This means that for a perfect well-ordered PAA, σ can be calculated using the circular pore area and the hexagonal lattice area from the same single lattice cell. It equals the ratio of the circular pore area to the hexagonal lattice area. The circular pore area (Ac) can be calculated using eqn (41):
| |
 | (41) |
The hexagonal lattice area (Ah) has already been given by eqn (36) in the previous discussion. Thus, the formula (40) can be derived from Ac/Ah, as shown in the calculation process in eqn (42):
| |
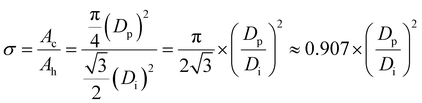 | (42) |
Going a step further, since both Di and Dp are a function of U, as shown in formulas (1), (6), (9), (11) and (12), formula (40) can be further derived into simplified σ calculation formulas with U as the only independent variable. After substituting the universal Di calculation formula (1) and universal Dp calculation formula (11) into formula (40), the universal σ calculation formula is obtained, as shown in eqn (43):
| |
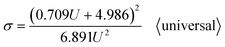 | (43) |
After substituting the oxalic Di calculation formula (1) and oxalic Dp calculation formula (9) into (40), the oxalic σ calculation formula is obtained as a constant equal to 11.75%, as shown in eqn (44):
| |
σ = 11.75%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉 〈oxalic〉
| (44) |
This result is similar to the 10% porosity rule for PAA.98 After substituting the sulfuric Di calculation formula (6) and sulfuric Dp calculation formula (12) into formula (40), the σ calculation formula specific to sulfuric acid anodization is obtained, as shown in eqn (45):
| |
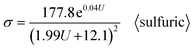 | (45) |
Moreover, based on further derivation, eqn (43) is the optimal calculation for phosphoric acid anodization.
Notably, both σ and ρ (discussed in the previous section) are artificially defined structural parameters of PAA, rather than natural ones. They cannot be directly measured experimentally, but must be derived through calculations based on other structural parameters. Despite many references concerning σ and ρ, all reported values for σ and ρ are calculated from Di and Dp, and are not measured experimentally.27,61,64,65,98 In scientific research, the accuracy of a theoretical model or formula needs to be determined through experimental verification, specifically by examining how well its calculated outcomes fit the measured data. Without experimentally measured σ and ρ data for practical validation, assessing the consistency and accuracy of their formulas is inherently meaningless, whether or not other structural parameters like Di and Dp are used for cross-reference. The key to resolving the issue of consistency and accuracy assessment for the σ and ρ formula lies in whether a new measuring technique can be developed to effectively measure the experimental data of σ and ρ in the future. Given the current lack of experimental measurement data for σ and ρ, their existing calculation formulas can only be regarded as the correct results of logical theoretical derivation. The development of an experimental measurement technique for σ and ρ remains a significant research gap at present and a meaningful research topic for future work.
Section summary: according to a series of derivations, the existing optimal σ calculation formulas with U as the only independent variable are shown as follows:
| |
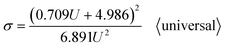 | (43) |
| |
σ = 11.75%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉 〈oxalic〉
| (44) |
| |
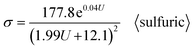 | (45) |
| |
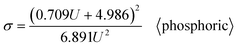 | (43) |
Eqn (43) is the universal σ calculation formula for common cases. The first exception is for oxalic acid as the electrolyte, where formula (44) is slightly better than formula (43). The second exception is for sulfuric acid as the electrolyte, where formula (45) is slightly better than formula (43).
10 Summary and outlook
There are eight key structural parameters for PAA in total: interpore distance (Di), pore diameter (Dp), pore wall thickness (Tw), barrier layer thickness (Tb), porous layer thickness (Tp) also known as pore channel length (Lp), oxide layer thickness (To), pore density (ρ) and porosity (σ), respectively. In current published research studies, the most common calculation formulas of the eight key PAA structural parameters are summarized and organized as follows:
According to a series of competitive screening for the most common calculation formulas of the eight PAA structural parameters, the optimal formulas of each structural parameter for various electrolyte systems are confirmed. After further using U to unify the independent variables of the existing optimal formulas, the universal equation set for the PAA structural parameter calculation with U as the unified independent variable is formulated as follows:
where
U is the anodization voltage with units of V, and
D is the anodization duration in units of min. The universal PAA equation set demonstrates high accuracies across most situations. It is a reliable equation set that can be widely used for all PAA structural parameter calculations.
The specialized equation set for the PAA structural parameter calculation with U as the unified independent variable is formulated as follows:
where
U is the anodization voltage in units of V, and
D is the anodization duration in units of min. The specialized equation set for the PAA structural parameter calculation will provide more accurate calculation results, if the anodization of a PAA is specified in a specific electrolyte.
The proposal of an equation set for the PAA structural parameter calculation confirms the existing optimal PAA structural parameter calculation formulas for mild anodization. It provides a systematic and accurate theoretical model and mathematical tool for designing and calculating PAA structures, according to practical requirements in scientific research and engineering applications.
It should be emphasized that this review provides a systematic summary and competitive screening of the most widely adopted existing formulas for calculating PAA structural parameters. The formulas screened here represent those regarded as the most universal and accurate up to now. However, this does not imply that they are absolutely universal or accurate in an objective sense. Significant potential remains for improving the universality and accuracy of the existing formulas, as clearly demonstrated by the MSE statistics for the Di, Dp, and Tb calculation formulas in Tables 1–3, respectively. The most critical problem of the existing mainstream PAA structural parameter calculation formulas is that these existing mainstream formulas are primarily expressed as single-variable functions of U, but that voltage is not the sole factor influencing the PAA structural parameters. Besides voltage, many other conditions such as the anodization temperature, electrolyte concentration, electrolyte type and anodization duration also influence various PAA structural parameters to different extents.59,83,108 For example, the anodization temperature is almost the second most significant factor influencing the PAA structural parameters, after voltage. The structural parameters affected by temperature include Dp, Tw, Tb, Tp, To and σ. While the impact of temperature on the PAA structural parameters has been qualitatively mentioned in numerous studies, it is unsatisfactory that systematic quantitative investigations dedicated to temperature effects, particularly those regarding local temperature, are scarcely reported. Existing calculation formulas for PAA structural parameters neither incorporate temperature as a variable nor specify their applicable temperature ranges, which constitutes a serious limitation in the existing proposed formulas. Therefore, besides the anodization voltage, the systematic and quantitative investigation of other key anodization conditions (such as the anodization temperature, electrolyte concentration, electrolyte type, and anodization duration) that influence the structural parameters of PAA remains a significant research gap at present and a meaningful research topic for future work.
The second point to emphasize is that all calculation formulas mentioned in this review are suitable for PAA prepared by mild anodization. However, it is important to note that hard anodization is another important method for fabricating PAA,108–110 and these formulas are not wholly suitable for the resulting PAA of hard anodization. A detailed discussion of the formulas applicable to hard-anodized PAA will be presented in our next forthcoming topical review.
Data availability
No primary research results, software or code have been included, and no new data were generated or analysed as part of this review.
Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ra09220e.
Conflicts of interest
The authors have no conflicts of interest to declare.
Acknowledgements
The authors acknowledge the support of the Applied Basic Research Fund of the School of Physics and Optoelectronic Engineering of Beijing University of Technology (056000513103) and the Open Project Fund of Multi-Scale Laser Forming Manufacturing Technology (056000514125528).
References
- X. Yin, H. Zhou, M. Zhang, J. Su, X. Wang, S. Li, Z. Yang, Z. Kang and R. Zhou, C3N nanodots inhibits Aβ peptides aggregation pathogenic path in Alzheimer's disease, Nat. Commun., 2023, 14, 5718 Search PubMed.
- Y. Kimura, Y. Cui, T. Suzuki, Y. Tanaka, T. Tanaka, Y. Toku and J. Yang, Growth of metal nanowire forests controlled through stress fields induced by grain gradients, Science, 2024, 385, 641–646 CrossRef CAS PubMed.
- D. Moon, W. Lee, C. Lim, J. Kim, Y. Jung, H.-Y. Choi, W. S. Choi, H. Kim, Ji-H. Baek, C. Kim, J. Joo, H.-G. Oh, H. Jang, K. Watanabe, T. Taniguchi, S. Bae, J. Son, H. Ryu, J. Kwon, H. Cheong, J. W. Han, H. Jang and G.-H. Lee, Hypotaxy of wafer-scale single-crystal transition metal dichalcogenides, Nature, 2025, 638, 957–964 CrossRef CAS PubMed.
- H. Hilal, Q. Zhao, J. Kim, S. Lee, H. MohammadNavid, S. Yoo, S. Lee, W. Park, W. Park, J. Lee, W. L. Joong, I. Jung and S. Park, Three-dimensional nanoframes with dual rims as nanoprobes for biosensing, Nat. Commun., 2022, 13, 4813 Search PubMed.
- J. Tao, R. Gao, G. Lin, C. Chu, Y. Sun, C. Yu, Y. Ma and H. Qiu, Synthesis of noble metal nanoarrays via agglomeration and metallurgy for acidic water electrolysis, Nat. Commun., 2025, 16, 4996 CrossRef CAS PubMed.
- S. Zhao, W. Peng, L. Zhou, D. Shuqi, W. Ren, E. Xu, Y. Xiao, M. Zhang, M. Huang, Y. Shen and C.-W. Nan, Metal-organic cage crosslinked nanocomposites with enhanced high-temperature capacitive energy storage performance, Nat. Commun., 2025, 16, 769 CrossRef CAS PubMed.
- A. O. Denisov, V. Reckova, S. Cances, M. J. Ruckriegel, M. Masseroni, C. Adam, C. Tong, J. D. Gerber, W. W. Huang, K. Watanabe, T. Taniguchi, T. Ihn, K. Ensslin and H. Duprez, Spin-valley protected Kramers pair in bilayer graphene, Nat. Nanotechnol., 2025, 20, 494–499 CrossRef CAS PubMed.
- H. Luo, J. Lv, P. Wen, S. Zhang, M. Wen and Z. Yang, Supramolecular polyrotaxane-based nano-theranostics enable cancer-cell stiffening for enhanced T-cell-mediated anticancer immunotherapy, Nat. Commun., 2025, 16, 2331 CrossRef CAS PubMed.
- X. Wang, Z. Yuan, L. Tingting, K. Wang, W. Dong, M. Lu, Y. Zhang, Z. Wu, A. Tang and X. Bai, Sequential addition of cations increases photoluminescence quantum yield of metal nanoclusters near unity, Nat. Commun., 2025, 16, 587 CrossRef CAS PubMed.
- S. C. Jin, S. Park, N.-U. K. Seo, Y.-C. Kang, C.-W. Lee and H. Y. Jung, Zincophilic CuO as electron sponge to facilitate dendrite-free zinc-based flow battery, Nat. Commun., 2025, 16, 844 Search PubMed.
- X. He, H. Gu, Y. Ma, Y. Cai, H. Jiang, Y. Zhang, H. Xie, M. Yang, X. Fan, L. Guo, Z. Yang and C. Hu, Light patterning semiconductor nanoparticles by modulating surface charges, Nat. Commun., 2024, 15, 9843 CrossRef CAS PubMed.
- G. Wang, T. Dvir, G. P. Mazur, C.-X. Liu, N. van Loo, S. L. D. Ten Haaf, A. Bordin, S. Gazibegovic, G. Badawy, E. P. A. M. Bakkers, M. Wimmer and L. P. Kouwenhoven, Singlet and triplet Cooper pair splitting in hybrid superconducting nanowires, Nature, 2022, 612, 448–453 CrossRef CAS PubMed.
- Y. Liu, M. Li, K. Jiang, Y. Zhang, P. Gong, S. Song, L. Dong, H. Liang, X. Huang, J. Wang, W. Li and C.-W. Nan, Radiation-hardened dendritic-like nanocomposite films with ultrahigh capacitive energy density, Nat. Commun., 2025, 16, 3882 CrossRef CAS PubMed.
- B. Lyu, J. Chen, S. Wang, S. Lou, P. Shen, J. Xie, L. Qiu, I. Mitchell, C. Li, C. Hu, X. Zhou, K. Watanabe, T. Taniguchi, X. Wang, J. Jia, L. Qi, G. Chen, T. Li, S. Wang, W. Ouyang, O. Hod, F. Ding, M. Urbakh and Z. Shi, Graphene nanoribbons grown in hBN stacks for high-performance electronics, Nature, 2024, 628, 758–764 CrossRef CAS PubMed.
- Z. Miao, W. Jiang, H. Qiao, W. Xunbin, Y.-W. Zhong, R. Hao and M. Gu, A 3D nanoscale optical disk memory with petabit capacity, Nature, 2024, 626, 772–778 CrossRef PubMed.
- S. Lee, R. D. Kibler, G. Ahn, Y. Hsia, A. J. Borst, A. Philomin, M. A. Kennedy, B. Huang, B. Stoddard and D. Baker, Four-component protein nanocages designed by programmed symmetry breaking, Nature, 2025, 638, 546–552 Search PubMed.
- W. Peng, W. Tai, B. Li, H. Wang, T. Wang, S. Guo, X. Zhang, P. Dong, C. Tian, S. Feng, L. Yang, C. Gong and B. Zheng, Inhalable nanocatalytic therapeutics for viral pneumonia, Nat. Mater., 2025, 24, 637–648 CrossRef CAS PubMed.
- Y. Song, N. Xu, G. Liu, H. Qi, W. Zhao, B. Zhu, Z. Lin and Z. Jia, High-yield solar-driven atmospheric water harvesting of metal-organic-framework-derived nanoporous carbon with fast-diffusion water channels, Nat. Nanotechnol., 2022, 17, 857–863 CrossRef CAS PubMed.
- S. Panda, T. Maity, S. Sarkar, A. K. Manna, J. Mondal and R. Haldar, Diffusion-programmed catalysis in nanoporous material, Nat. Commun., 2025, 16, 1231 Search PubMed.
- D. Wang, N. B. Saleh, A. Byro, R. Zepp, E. Sahle-Demessie, T. P. Luxton, K. T. Ho, R. M. Burgess, M. Flury, J. C. White and C. Su, Nano-enabled pesticides for sustainable agriculture and global food security, Nat. Nanotechnol., 2022, 17, 347–360 Search PubMed.
- L. Wei, J. Liu and G. Jiang, Nanoparticle-specific transformations dictate nanoparticle effects associated with plants and implications for nanotechnology use in agriculture, Nat. Commun., 2024, 15, 7389 Search PubMed.
- H. Peng, B. Cai, Y. Zhang, L. Gao, Z. Pei-Yan, L. Zhou, S. Zhang, W. Liang, Q.-F. Xuan, M. C. Koo, C.-M. Liang, L. Wen-Peng, Z.-L. Hou, T. Zhou and G.-S. Wang, Rada-terahertz-infrared compatible stealth coaxial silver nanowire@carbon nano-cable aerogel, Angew. Chem., Int. Ed., 2025, 64, e202421090 CrossRef CAS PubMed.
- V. S. Abhay Anand, M. K. Sahoo, F. Mujeeb, A. Varghese, S. Dhar, S. Lodha and A. Kumar, Novel nano-electroplating-based plasmonic platform for giant emission enhancement in monolayer semiconductors, ACS Appl. Mater. Interfaces, 2023, 15, 57783–57790 Search PubMed.
- C. Xue, J. Zhao, Y. Wu, H. Yu, S. Yang, L. Wang, W. Zhao, Q. Wu, Z. Zhu, B. Liu, X. Zhang, W. Zhou and R. Tai, Fabrication of Larger-area high-aspect-ratio periodic nanostructures on various substrates by soft X-ray interference lithography, Appl. Surf. Sci., 2017, 425, 553–557 Search PubMed.
- B. X. E. Desbiolles, E. de Coulon, A. Bertsch, S. Rohr and P. Renaud, Intracellular recording of cardiomyocyte action potentials with nanopatterned volcano-shaped microelectrode arrays, Nano Lett., 2019, 19, 6173–6181 Search PubMed.
- J. S. Wojciech and Z. Bojar, Synthesis of anodic aluminum oxide (AAO) at relatively high temperatures. Study of the influence of anodization conditions on the alumina structural features, Surf. Coat. Technol., 2011, 265–272 Search PubMed.
- I. Vida-Simiti, D. Nemes, N. Jumate, G. Thalmaier and N. Sechel, Self-ordered nanoporous alumina templates formed by anodization of aluminum in oxalic acid, JOM, 2012, 64, 1143–1147 CrossRef CAS.
- A. K. Eessaa and A. M. El-Shamy, Review on fabrication, characterization, and applications of porous anodic aluminum oxide films with tunable pore sizes for emerging technologies, Microelectron. Eng., 2023, 279, 112061 CrossRef CAS.
- B. Lee, M. Kang, K. Lee, Y. Chae, K.-J. Yoon, D.-S. Lee and I. Park, Multigas identification by temperature-modulated operation of a single anodic aluminum oxide gas sensor platform and deep learning algorithm, ACS Sens., 2025, 10, 954–964 Search PubMed.
- C.-C. Lee, C.-W. Chen, J.-S. Lin, S. H. Wang, C.-S. Lee, C.-C. Chen, Y.-H. Lin and C.-Y. Chen, Effect of anodization treatment on the thickness, hardness, and microstructural characterization of anodic aluminum oxide film on AA6061 and critical patent analysis, J. Mater. Eng. Perform., 2022, 31, 667–681 Search PubMed.
- G. R. Kramer, F. A. Bruera, P. D. Zapata and A. E. Ares, Aluminum oxide coatings as nanoadsorbents for the treatment of effluents colored with Eriochrome black T, Coating, 2025, 15, 15040488 Search PubMed.
- A. Wang, Y. Hang, J. Wang, W. Tan and N. Wu, Machine learning-assisted light management and electromagnetic field modulation of large-area plasmonic coaxial cylindrical pillar/ring nanoarray patterns, J. Phys. Chem. C, 2024, 128, 12495–12502 Search PubMed.
- S. L. Lee, J. Thomas, C.-Y. Mou, C.-L. Liu and K.-L. Tung, High-performance separation for ultra-low concentration nanoparticles with mesoporous silica thin membrane, Sep. Purif. Technol., 2024, 350, 127918 Search PubMed.
- C.-C. Fan, Y.-H. Liu and C.-J. Lu, Separation of C1 – C15 alkanes with a disk-shaped aluminum column employing mesoporous AAO as the stationary phase, Anal. Chem., 2022, 94, 15570–15577 CrossRef CAS PubMed.
- A. Anil Chandra, Z. Huang, B. Amit, W. Van Geertruyden, D. Gao and M. Wojciech, Evaluation of Nano-porous alumina membranes for hemodialysis application, ASAIO J., 2009, 55, 217–223 Search PubMed.
- M. Makela and Z. L. P. T. Lin, Surface functionalized anodic aluminum oxide membrane for opto-nanofluidic SARS-CoV-A genomic target detection, IEEE Sens. J., 2021, 21, 22645–22650 Search PubMed.
- Z. Zhang, M. Brady, B. K. Amarsingh, J.-R. Park, E. R. Jong and D. Choi, Electrochemical Characterization of LiMn2O4 nanowires fabricated by sol-gel for lithium-ion rechargeable batteries, Mater. Lett., 2020, 273, 127923 Search PubMed.
- Y. Li, L. Feng, J. Zhu, H. Zhang, S. Ha and K. Zhang, Structural and optical properties of Ga2O3 nanorod arrays sputtered using AAO templates, Mater. Lett., 2025, 392, 138569 Search PubMed.
- K. Roman, M. L. Smith, N. C. A. Seaton, M. L. Odlyzko, O. Masiuchok, R. Jeanne, P. B. Amama and B. J. H. Stadler, Sustainable manufacturing of vertical carbon nanotube (CNT) arrays inside insulating nanoporous membranes using nickel magnetic nanowires (MNWs), ACS Appl. Nano Mater., 2024, 7, 24866–24874 Search PubMed.
- G. Chen, F. Han, H. Ma, L. Pei, Z. Zhou, P. Wang, X. Li, G. Meng and B. Wei, High density 3D carbon tube nanoarray electrode boosting the capacitance of filter capacitor, Nano-Micro Lett., 2024, 16, 235 CrossRef CAS PubMed.
- Z. Lin, G.-L. Yang, W.-J. Ding, Y.-Q. Cao, W.-M. Li and Ai-D. Li, Growth behavior of Ir metal formed by atomic layer deposition in the nanopores of anodic aluminum oxide, Dalton Trans., 2022, 51, 9664 Search PubMed.
- J. Shen, Z. Hu, L. Quigley and H. Wang, Controlled growth of vertically aligned nanocomposites through an Au seeding-assisted method, ACS Omega, 2023, 8, 37140–37146 Search PubMed.
- J. Chen, Z. He and Y. Wang, AAO-ZnO nanocomposite coatings on aluminum alloy surface with high bonding strength and long-lasting antibacterial properties, Chem. Eng. J., 2024, 497, 154414 Search PubMed.
- B. A. Tran, M. T. Nguyen, T. Quang Le, T. C.-T. Pham, T.-P. T. Pham, T. L. Anh and H.-D. Nguyen, High-performance red-emitting InGaN/AlGaN nanowire light-emitting Diodes grown through porous template, Mater. Sci. Semicond. Process., 2024, 169, 107894 Search PubMed.
- D. Xu, Z. Congmian and Z. Hao, Morphology and growth mechanism of amorphous silicon carbide sputtered on anodic aluminum template by radio frequency magnetron sputtering, Ceram. Int., 2020, 46, 19629–19633 Search PubMed.
- H. Masuda and K. Fukuda, Ordered metal nanohole arrays made by a two-step replication of honeycomb structures of anodic alumina, Science, 1995, 268, 1466–1468 Search PubMed.
- I. V. Roslyakov, E. O. Sotnichuk, S. V. Sotnichuk, S. E. Kushnir and K. S. Napolskii, Kinetic and crystallographic control of self-ordering of pores in anodic aluminium oxide, J. Solid State Electrochem., 2025, 29, 1341–1373 CrossRef CAS.
- B. Tazneva, I. Vrublevshy, V. Videkow and N. Luspha, Role of electrode temperature in anodic growth of sulfuric acid alumina films, J. Solid State Electrochem., 2025, 29, 1459–1465 Search PubMed.
- Z. Song, X. Zhang, Y. Yang, P. Hou, J. Chen and S. Liang, Preparation and field emission properties of Ni nanowire arrays with different length-diameter ratios, J. Alloys Compd., 2025, 1010, 178203 Search PubMed.
- J. Dai, J. Singh and N. Yamamoto, Nonbrittle nanopore deformation of anodic aluminum oxide membranes, J. Am. Ceram. Soc., 2018, 101, 2170–2180 CrossRef CAS.
- G.-O. Ana, R. Jerome and A. Laurent, Nanocontrolled thinning of the barrier layer thickness of porous anodic films using galvanodynamic polarization of aluminum alloys, Electrochim. Acta, 2024, 479, 143894 Search PubMed.
- G. Arulkumar, P. Swaminathan and L. Neelakantan, Anodic aluminum oxide template assisted synthesis of copper nanowires using a galvanic displacement process for electrochemical denitrification, ACS Appl. Nano Mater., 2019, 2, 5981–5988 Search PubMed.
- A. Adugna Ayalew, X. Han and M. Sakairi, Effect of Substrate temperature and electrolyte composition on the fabrication of through-hole porous AAO membrane with SF-MDC, Mater. Chem. Phys., 2024, 323, 129658 Search PubMed.
- B. A. Tran, M. T. Nguyen, T. Quang Le, T. Cao-Thanh Pham, T.-P. T. Pham, T. L. Anh and H.-D. Nguyen, High-performance red-emitting InGaN/AlGaN nanowire light-emitting diodes grown through porous template, Mater. Sci. Semicond. Process., 2024, 169, 107894 CrossRef CAS.
- A. M. Abd-Elnaiem, Z. A. Mohamed, S. El. Soliman and A. Mohamed, Synthesis, characterization, and optical sensing of hydrophilic anodic alumina films, Opt. Mater., 2024, 157, 116390 Search PubMed.
- Z. Leszek, G. D. Sulka, J. Szeremeta and M. Jaskula, Porous anodic alumina formed by anodization of aluminum alloy (AAO1050) and high purity aluminum, Electrochim. Acta, 2010, 55, 4377–4386 Search PubMed.
- J. De Laet, H. Terryn and J. Vereecken, Development of an optical model for steady state porous anodic films on aluminium formed in phosphoric acid, Thin Solid Films, 1998, 320, 241–252 Search PubMed.
- H. d. L. Lira and R. Paterson, New and modified anodic alumina membranes Part III. Preparation and characterization by gas diffusion of 5 nm pore size anodic alumina membranes, J. Membr. Sci., 2002, 206, 375–387 Search PubMed.
- S.-K. Hwang, S.-H. Jeong, H.-Y. Hwang, O.-J. Lee and K.-H. Lee, Fabrication of highly ordered pore array in anodic aluminum oxide, Korean J. Chem. Eng., 2002, 19, 467473 Search PubMed.
- K. Ebihara, H. Takahashi and M. Nagayama, Structure and density of anodic oxide films formed on aluminum in oxalic acid solutions, J. Met. Finish. Soc. Jpn., 1983, 34, 548–553 Search PubMed.
- K. Ebihara, H. Takahashi and M. Nagayama, Structure and density of anodic oxide films formed on aluminum in sulfuric acid solutions, J. Met. Finish. Soc. Jpn., 1982, 33, 156–164 Search PubMed.
- P. Ramana Reddy, K. M. Ajith and N. K. Udayashankar, Morphology and photoluminescence of nano-porous anodic alumina membranes obtained in oxalic acid at different anodization potentials, Nano Express, 2020, 1, 010047 Search PubMed.
- L. Zaraska, W. J. Stepniowski, E. Ciepiela and G. D. Sulka, The effect of anodizing temperature on structural features and hexagonal arrangement of nanopores in alumina synthesized by two-step anodizing in oxalic acid, Thin Solid Films, 2013, 534, 155–161 Search PubMed.
- L. Zaraska, G. D. Sulka and M. Jaskula, Anodic alumina membranes with defined pore diameters and thicknesses obtained by adjusting the anodizing duration and pore opening/widening time, J. Solid State Electrochem., 2011, 15, 2427–2436 CrossRef CAS.
- C. V. Manzano, D. Ramos, L. Pethö, G. Bürki, J. Michler and L. Philippe, Controlling the color and effective refractive index of metal-anodic aluminum oxide (AAO)-Al nanostructures: Morphology of AAO, J. Phys. Chem. C, 2018, 122, 957–963 Search PubMed.
- C. V. Manzano, J. P. Best, J. J. Schwiedrzik, A. Cantarero, J. Michler and L. Philoppe, The influence of thickness, interpore distance and compositional structure on the optical properties of self-ordered anodic aluminum oxide films, J. Mater. Chem. C, 2016, 4, 7658 Search PubMed.
- J. Martín, C. V. Manzano, O. Caballero-Calero and M. Martín-González, High-aspect-ratio and highly ordered 15-nm porous alumina templates, ACS Appl. Mater. Interfaces, 2013, 5, 72–79 Search PubMed.
- C. V. Manzano, J. Martín and M. S. Martín-González, Ultra-narrow 12 nm pore diameter self-ordered anodic alumina templates, Microporous Mesoporous Mater., 2014, 184, 177–183 Search PubMed.
- J. Martín, C. V. Manzano and M. Martín-González, In-depth study of self-ordered porous alumina in the 140-440 nm pore diameter range, Microporous Mesoporous Mater., 2012, 151, 311–316 Search PubMed.
- A. Shunta, T. Kikuchi, S. Natsui and R. O. Suzuki, Nanostructural characterization of large-scale porous alumina fabricated via anodizing in arsenic acid solution, Appl. Surf. Sci., 2017, 403, 652–661 Search PubMed.
- S. Ono, M. Saito and H. Asoh, Self-ordering of anodic porous alumina formed in organic acid electrolytes, Electrochim. Acta, 2005, 51, 827–833 Search PubMed.
- S. Ono, M. Saito, M. Ishiguro and H. Asoh, Controlling factor of self-ordering of anodic porous alumina, J. Electrochem. Soc., 2004, 151, B473 Search PubMed.
- Y. Ahn and J. Y. Son, Formation of ferrolelctric multi-domains and electrical conduction in epitaxial BiFeO3 nanodots fabricated with AAO nanotemplates, Ceram. Int., 2023, 49, 2738–2745 Search PubMed.
- Z. Fang, J. Dong, Y. Fan, C. Li, Q. Han, C. Zhang, L. Zhu, X. Yan, J. Qi and W. Gao, Transfer of AuNRs into AAO nanoholes via self-assembly method for ultrasensitive SERS detection, ACS Omega, 2025, 10, 18764–18774 CAS.
- S. Matsumoto, S. Tanaka, T. Nagao, T. Shimizu, S. Shingubara and T. Ito, Evaluating the anti-biofilm performance of Si and resin based nanopillars, J. Photopolym. Sci. Technol., 2024, 37, 379–384 Search PubMed.
- F.-G. Zhong, X.-Y. Huang, R. Barreto and S.-H. Chen, Synthetization and photocatalytic performance of high-aspect-ratio β-Bi2O3 nanowires via the combination of vacuum die-casting and controllable oxidation processes in liquid phase, Ceram. Int., 2025, 51, 15802–15808 CrossRef CAS.
- Z. Song, Y. Yang, P. Hou, X. Zhang, S. Liang and J. Chen, Wave aborbing properties of Ni Nanoparticle/CNT composite film fabricated by AAO/CNTs electrode, Heliyon, 2024, 10, e26054 Search PubMed.
- A. Belwalkar, E. Grasing, W. Van Geertruyden, Z. Huang and W. Z. Misiolek, Effect of processing parameters on pore structure and thickness of anodic aluminum oxide (AAO) tubular membranes, J. Membr. Sci., 2008, 319, 192–198 CrossRef CAS PubMed.
- A. Ruiz-Clavijo, O. Caballero-Calero and M. Martín-González, Revisiting anodic alumina templates: from fabrication to applications, Nanoscale, 2021, 13, 2227 RSC.
- H. Akbarpour, M. Mohajeri and M. Akbarpour, Pore diameter of nanoporous anodic alumina: Experimental study and application of ANFIS and MLR, Chemom. Intell. Lab. Syst., 2016, 153, 82–91 CrossRef CAS.
- A. M. Abd-Elnaiem, A. M. Mebed, A. Gaber and M. A. Abdel-Rahim, Tailoring the porous nanostructure of porous anodic alumina membrane with the impurity control, J. Alloys Compd., 2016, 659, 270–278 CrossRef CAS.
- H. Azami and M. R. Omidkhah, Modeling and optimization of characterization of nanostructure anodized aluminium oxide membranes, J. Iran. Chem. Soc., 2019, 16, 985–997 CrossRef CAS.
- F. Alejandra Bruera, G. R. Kramer, M. L. Vera and A. E. Ares, Synthesis and morphological characterization of nanoporous aluminum oxide films by using a single anodization step, Coatings, 2019, 9, 115 CrossRef.
- W. J. Stepniowski, D. Zasada and Z. Bojar, First step of anodization influences the final nanopore arrangement in anodized alumina, Surf. Coat. Technol., 2011, 206, 1416–1422 Search PubMed.
- W. L. Hwang, J. K. Anthony, H.-D. Nguyen, M. Sun-il, K. Kim, H. Lim, J. Lee and R. Fabian, Enhanced ultrafast optical nonlinearity of porous anodized alumimum oxide nanostructures, Opt. Express, 2009, 17, 19093–19101 Search PubMed.
- B. Abad, J. Maiz and M. Martin-Gonzalez, Rules to determine thermal conductivity and density of anodic aluminum oxide (AAO) membranes, J. Phys. Chem. C, 2016, 120, 5361–5370 CrossRef CAS.
- C.-A. Ku, C.-C. Wu, C.-W. Hung and C.-K. Chung, Influence of normal-to-high anodizing voltage on AAO surface hardness from 1050 aluminum alloy in oxalic acid, Micromachines, 2024, 15, 683 Search PubMed.
- C. K. Chung, D. Dhandapani, C. J. Syu, M. W. Liao, B. Y. Chu and E. H. Kuo, Role of oxalate anions on the evolution of widened pore diameter and characteristics of room-temperature anodic aluminum oxide, J. Electrochem. Soc., 2017, 164, C121–C127 CrossRef CAS.
- V. Ali, E. Dubois, L. Michot, M. Jardat, D. Lairez, S. Durand-Vidal, C. Guibert and N. Jouault, Electrical surface properties of nanoporous alumina membranes:
influence of nanochannels' curvature, roughness and composition studied via electrokinetic experiments, R. Soc. Chem., 2023, 25, 28150 Search PubMed.
- L. González-Rovira, L. González-Souto, P. J. Astola and C. Bravo-Benítez, Francisco Javier Botana. Assessment of the corrosion resistance of self-ordered anodic aluminum oxide (AAO) obtained in tartaric-sulfuric acid (TSA), Surf. Coat. Technol., 2020, 399, 126131 Search PubMed.
- D. D. Macdonald, The point defect model for the passive state, J. Electrochem. Soc., 1992, 139, 3434 Search PubMed.
- G. Patermarakis, J. Chandrinos and K. Masavetas, Formulation of a holistic model for the kinetics of steady state growth of porous anodic alumina films, J. Solid State Electrochem., 2007, 11, 1191–1204 CrossRef CAS.
- W. Lee and S. Park, Porous anodic aluminum oxide: anodization and templated synthesis of functional nanostructures, Chem. Rev., 2014, 114, 7487–7556 Search PubMed.
- S. Kjyohito, S. Suzuki, Y. Takada, K. Abe, H. Yoshida and T. Sako, Anodization of aluminum in high-pressure carbonic acid aqueous solution, J. Chem. Eng. Jpn., 2006, 39, 90–94 CrossRef.
- I. Vrublevsky, V. Parkoun, V. Sokol, J. Schreckenbach and A. G. Werner, Dissolution behavior of anodic oxide films formed in sulfanic acid on aluminum, Microchim. Acta, 2007, 156, 173–179 Search PubMed.
- C. G. Sousa, D. C. Leitão, M. P. Proença, A. Apolinário, J. G. Correia, J. Ventura and J. P. Araújo, Tunning pore filling of anodic alumina templates by accurate control of the bottom barrier layer thickness, Nanotechnology, 2011, 22, 315602 CrossRef CAS PubMed.
- D. A. Brevnov, G. V. Rama Rao, G. P. López and P. B. Atanassov, Dynamics and temperature dependence of etching processes of porous and barrier aluminum oxide layers, Electrochim. Acta, 2004, 49, 2487–2494 CrossRef CAS.
- K. Nielsch, J. Choi, K. Schwirn, R. B. Wehrspohn and G. Ulrich, Self-ordering regimes of porous alumina: The 10% porosity rule, Nano Lett., 2002, 2, 677–680 CrossRef CAS.
- B. Benfedda, L. Hamadou, N. Benbrahim, A. Kadri, E. Chainet and F. Charlot, Electrochemical impedance investigation of anodic alumina barrier layer, J. Electrochem. Soc., 2012, 159, C372–C381 CrossRef CAS.
- S. Ono and N. Masuko, Effect of electric field strength on cell morphology and anion incorporation of anodic porous alumina, ECS Trans., 2017, 75, 23–31 Search PubMed.
- X. Zhao, S.-K. Seo, U.-J. Lee and K.-H. Lee, Controlled electrochemical dissolution of anodic aluminum oxide for preparation of open-through pore structures, J. Electrochem. Soc., 2007, 154, C553–C557 Search PubMed.
- Y. D. Kim, S. Choi, A. Kim and W. Lee, Ionic current rectification of porous anodic aluminum oxide (AAO) with a barrier oxide layer, ACS Nano, 2020, 14, 13727–13738 CrossRef CAS PubMed.
- P. L. Alexey and K. S. Napolskii, Probing barrier oxide layer of porous anodic alumina by in situ electrochemical impedance spectroscopy, J. Electrochem. Soc., 2021, 168, 071511 CrossRef.
- J. Wang, C. Suwen Law, S. Gunenthiran, H. N. Q. Tran, K. N. Tran, S. Y. Lim, A. D. Abell and A. Santos, Structural engineering of the barrier oxide layer of nanoporous anodic alumina for iontronic sensing, ACS Appl. Mater. Interfaces, 2022, 14, 21181–21197 CrossRef CAS PubMed.
- X. Li, Z. Liu, L. Yang, S. Zhou, Y. Qian, Y. wu, Z. Yan, Z. Zhang, T. Li, Q. Wang, C. Zhu, X.-Y. Kong and L. Wen, An ultrasensitive 2, 4, 6-trinitrophenol nanofluidic sensor inspired by olfactory sensory neurons in sniffer dogs, Chem. Sci., 2024, 15, 19504 RSC.
- M. Chen, C. Wang, J. Tan, J. Wang and C. Wang, Cell membrane-inspired COF/AAO hybrid nanofluidic membrane with ion current rectification properties for ultrasensitive detection of E. coli, Sens. Actuators, B, 2025, 423, 136719 CrossRef CAS.
- L. Osama, H. T. Handal, A. M. El-Sayed Sara, E. M. Elzayat and M. Mostafa, Fabrication and optimization of alumina nanoporous membranes for drug delivery applications: A comparative study, Nanomaterials, 2024, 14, 1078 CrossRef CAS PubMed.
- Y. Li, M. Zheng, L. Ma and W. Shen, Fabrication of highly ordered nanoporous alumina films by stable high-field anodization, Nanotechnology, 2006, 17, 5101–5105 CrossRef CAS.
- W. Lee, J. Ran, G. Ulrich and K. Nielsch, Fast fabrication of long-range ordered porous alumina membranes by hard anodization, Nat. Mater., 2006, 5, 741–747 Search PubMed.
- S.-Z. Chu, K. Wada, S. Inoue, M. Isogai and A. Yasumori, Fabrication of ideally ordered nanoporous alumina films and integrated alumina nanotubule arrays by high-field anodization, Adv. Mater., 2005, 17, 2115–2119 Search PubMed.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article a,
Shuang Lib,
Yan Zhaoa and
Tianrui Zhai
a,
Shuang Lib,
Yan Zhaoa and
Tianrui Zhai *a
*a

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉
〈oxalic〉
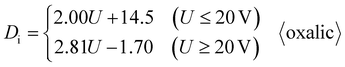
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉
〈sulfuric〉
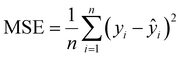

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈universal〉
〈universal〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉
〈oxalic〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉
〈sulfuric〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈phosphoric〉
〈phosphoric〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈inorganic〉
〈inorganic〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈organic〉
〈organic〉
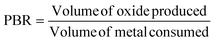
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e0.02U
e0.02U

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈universal〉
〈universal〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉
〈oxalic〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e0.02U
e0.02U![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉
〈sulfuric〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈phosphoric〉
〈phosphoric〉
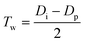
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈universal〉
〈universal〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉
〈oxalic〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e0.02U + 6.05
e0.02U + 6.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉
〈sulfuric〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈universal〉
〈universal〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉
〈oxalic〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e0.02U + 6.05
e0.02U + 6.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉
〈sulfuric〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈phosphoric〉
〈phosphoric〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉
〈oxalic〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉
〈sulfuric〉
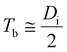
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉
〈oxalic〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e0.02U + 8.0465
e0.02U + 8.0465![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉
〈sulfuric〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈universal〉
〈universal〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉
〈sulfuric〉

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 533
533![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈universal〉
〈universal〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉
〈oxalic〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉
〈sulfuric〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈phosphoric〉
〈phosphoric〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈universal〉
〈universal〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉
〈oxalic〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈sulfuric〉
〈sulfuric〉
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈phosphoric〉
〈phosphoric〉
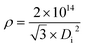
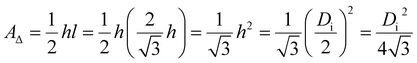
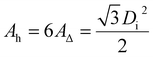
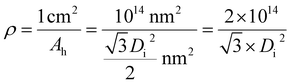
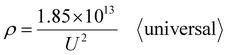
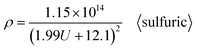
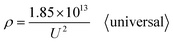
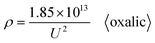
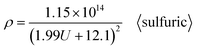
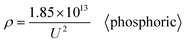
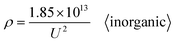
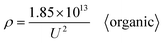
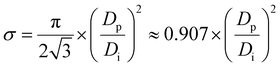

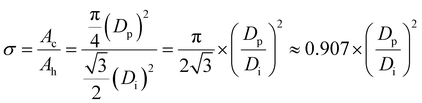
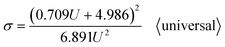
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉
〈oxalic〉
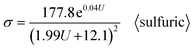
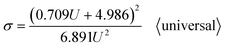
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 〈oxalic〉
〈oxalic〉