Open Access Article
Open Access ArticleK+/Cu2+ Co-chelated diastereoselective Friedel–Crafts reaction with chiral N-sulfinyl ketimines for the facile synthesis of chiral bisindoles and their cytotoxicity
Si Yan†
ad,
Mengwei Xu†cd,
Yan Liuad,
Ziwei Duacd,
Guangshuai Zhangad,
Zishu Liua,
Jin Xiaoa,
Xiaoqian Guoa,
Gang Liaoa,
Qing Mind,
Baocheng Xie*c and
Shuanglin Qin *abd
*abd
aNational Engineering Research Center of Personalized Diagnostic and Therapeutic Technology, TCM Precision Medicine Research Department, FuRong Laboratory, Hunan University of Chinese Medicine, Changsha 410208, P.R. China. E-mail: shuanglin@tju.edu.cn
bSenior Department of Hepatology, China Military Institute of Chinese Materia, The Fifth Medical Center of PLA General Hospital, Beijing 100039, P.R. China
cDepartment of Pharmacy, The Tenth Afffliated Hospital of Southern Medical University (Dongguan People's Hospital), Dongguan 523059, P.R. China. E-mail: baochengxie@126.com
dHubei Engineering Research Center of Traditional Chinese Medicine of South Hubei Province, School of Pharmacy, Xianning Medical College, Hubei University of Science and Technology, Xianning 437100, P.R. China
First published on 4th February 2026
Abstract
A novel K+/Cu2+ co-chelated diastereoselective Friedel–Crafts reaction with chiral N-sulfinyl ketimines has been developed for the synthesis of chiral bisindoles. The method exhibits broad substrate compatibility and high stereoselectivity. The synthesized bisindoles show significant cytotoxicity against tumor cell lines such as MG63 and HepG2, with Compound 29 demonstrating selective inhibitory effects on osteosarcoma cells. This protocol provides a useful strategy for the efficient construction of chiral bisindoles. The potential of these compounds in cancer therapy was also highlighted.
Introduction
The distinguishing feature of bisindole alkaloids is the incorporation of two indole moieties in their molecular structures. These dual indole units can be interconnected indirectly via diverse structural linkages. A prime example is exemplified by vinblastine and vincristine,1,2 the antitumor agents isolated from Catharanthus roseus. Moreover, bisindole alkaloids are capable of directly undergoing polymerization to yield natural bisindole alkaloids, such as staurosporine3 derived from Streptomyces species, along with Gliocladine C and Leptosin D.4 (Fig. 1) In the contemporary context, substantial advancements have been achieved in the realm of anti-tumor therapy utilizing bisindole alkaloids. A number of small-molecule bisindole compounds have been commercialized or entered the clinical trial phase, including indirubin,5,6 vincristine,7,8 vinorelbine,9,10 midostaurin,11–13 and enzastaurin.14 Notably, the dimerization of indole monomers harbors considerable potential in augmenting biological activity, mitigating side effects, surmounting drug resistance, and optimizing pharmacokinetic, pharmacodynamic, or physicochemical characteristics.15 For instance, vinblastine and vincristine are bisindole alkaloids renowned for their potent antitumor capabilities.Fig. 1 Conversely, as monomeric entities, vindoline and catharanthine display a markedly diminished antitumor efficacy in comparison to vinblastine and vincristine.16,17 It is apparent that the dimeric indole structural motif is pivotal for the manifestation of their anti-tumor activities.Given that the majority of bisindole alkaloids are chiral compounds, the exploration and development of synthetic methodologies for the construction of chiral bisindole frameworks18,19 and bisindole alkaloids20 have emerged as a highly active and significant research frontier in the realms of organic chemistry and medicinal chemistry.21–23 With respect to the accessibility of reactants, the asymmetric Friedel–Crafts reaction involving indole and chiral tert-butanesulfinimines is recognized as the most straightforward and viable strategy for the acquisition of novel chiral compounds. Xu's team previously reported the successful implementation of the asymmetric Friedel–Crafts reaction between indole and isatin-derived N-sulfinyl ketimines, catalyzed by Bi(OTf)3. Nevertheless, favorable outcomes were exclusively attained when the nitrogen atom of isatin was substituted with a bulky dimethoxytrityl (DMTr) group. Moreover, they had previously attempted to use a Cu catalyst alone for the same reaction. However, unfortunately, the reaction exhibited extremely poor stereoselectivity.24
Chiral sulfinamides have been widely employed as versatile chiral auxiliaries in asymmetric synthesis.25 In our previous work, chiral sulfinamides were utilized as chiral directing groups. By means of chelation with alkali metals (Scheme 1a)26 and transition metals (Scheme 1b),27 as well as the concurrent chelation of alkali metals and transition metals (Scheme 1c),28 we have successfully accomplished the stereo-controlled generation of diverse chiral centers. Following our long interests in bisindoles, the chelation of chiral sulfinamide with both alkali metal K+ ions and transition metal Cu has facilitated the realization of a novel asymmetric Friedel–Crafts reaction (Scheme 1d). Leveraging this approach, a series of bisindole compounds bearing chiral amines were efficiently synthesized. Initial biological assessments of these compounds indicated promising activity against various tumor cell lines, including the osteosarcoma cell line MG63 and the hepatocellular carcinoma cell line HepG2. The selective inhibitory effect of bisindole 29 on osteosarcoma was particularly noteworthy.
 | ||
| Scheme 1 Construction of novel chiral centers via chelation-induced coordination of chiral sulfinamides with metal ions. | ||
Results and discussion
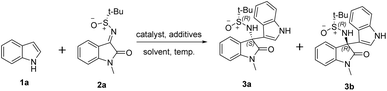
Initially, indole 1a and (R)-N-sulfinyl ketimine 2a were chosen as substrates for the screening of reaction conditions of the asymmetric Friedel–Crafts reaction. As presented in Table 1, the reaction between indole 1a and (R)-N-sulfinyl ketimine 2a was explored by screening various catalysts in THF as the solvent at room temperature. During the screening of different Cu-based catalysts (Table 1, Entries 1–7), it was observed that incorporating alkali metal ions Li+ led to the highest level of stereoselectivity in the resulting product under the catalytic action of Li2CuCl4 (Entry 6). Building upon our prior empirical insights, it was postulated that the alkali-metal ion Li+ could potentially engage in a chelation process with the reaction substrates. In light of this hypothesis, we utilized CuCl2 as the catalyst and incorporated alkali-metal ion additives into the reaction system (Entries 8–13). The subsequent substantial enhancement in the stereoselectivity of the reaction lent credence to our initial supposition. Notably, among the tested conditions, the employment of t-BuOK as the catalyst resulted in the most favorable enantioselectivity and yield (Entry 13).| Entries | Catalysts (10 mol%) | Additives (mol%) | Solvent | Temp. | Yield of 3a a (%) | drb (3a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3b) 3b) |
|---|---|---|---|---|---|---|
| a Isolated yield.b The values of dr were determined by 1H NMR.c The usage equivalent of CuCl2 is 5 mol%.d The usage equivalent of CuCl2 is 15 mol%. Temp. = temperature. NA = none available. | ||||||
| 1 | Cu(MeCN)4 | — | THF | r.t | 30 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
| (CF3SO3)2 | ||||||
| 2 | Cu(CF3SO3)2 | — | THF | r.t | 35 | 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3 |
| 3 | Cu(OAc)2 | — | THF | r.t | 18 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 4 |
| 4 | B2CuF8 | — | THF | r.t | 38 | 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3 |
| 5 | CuCl2 | — | THF | r.t | 56 | 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 6 | Li2CuCl4 | — | THF | r.t | 61 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 7 | CuBr | — | THF | r.t | NA | NA |
| 8 | CuCl2 | LiCl (100) | THF | r.t | 58 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 9 | CuCl2 | NaHMDS (100) | THF | r.t | 72 | >25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 10 | CuCl2 | LiHMDS (100) | THF | r.t | 75 | 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 11 | CuCl2 | KHMDS (100) | THF | r.t | 69 | >25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 12 | CuCl2 | t-BuONa (100) | THF | r.t | 68 | >25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 13 | CuCl2 | t-BuOK (100) | THF | r.t | 77 | >25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 14 | CuCl2 | t-BuOK (100) | PhMe | 50 | 39 | >15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 15 | CuCl2 | t-BuOK (100) | PhMe | 0 | 81 | >25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 16 | CuCl2 | t-BuOK (100) | PhMe | −20 | 92 | >25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 17 | CuCl2 | t-BuOK (100) | PhMe | −40 | 73 | >25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 18c | CuCl2 | t-BuOK (100) | PhMe | −20 | 77 | >25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 19d | CuCl2 | t-BuOK (100) | PhMe | −20 | 82 | >25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
Subsequent to these findings, we conducted a comprehensive screening of the influence of solvents on the reaction (Table S1, Entries 1–4). Protic solvents were found to exert a deleterious effect on the enantioselectivity of the reaction (Entry 1). In contrast, employing toluene as the reaction medium led to enhanced enantioselectivity and a relatively high yield (Entry 4). A systematic exploration of the impact of the reaction temperature was also made (Table 1, Entries 14–17). The results indicated that the reaction yielded 92% at a temperature of −20 °C (Entry 16). Furthermore, it was observed that any deviation from the optimal amount of the Cu catalyst, either an increase or a decrease, adversely affected the reaction yield (Entries 18–19). In conclusion, the optimal reaction conditions were identified (Entry 16).
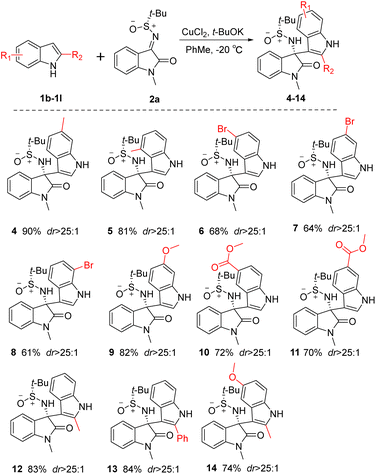
Upon establishing the optimal reaction conditions, we embarked on an investigation into the substrate scope. As depicted in Scheme 2, an initial screening of diverse indole substrates was carried out. It was observed that the desired products could be obtained with high stereoselectivity across all examined cases regardless of the nature of the substituents on the benzene ring of indole (4–11). In contrast to cases involving electron-donating groups as substituents on the benzene ring of the indole, a relatively lower yield was noted when electron-withdrawing groups were present. Remarkably, the introduction of a methyl or phenyl group at the C-2 position of the indole moiety did not hinder the reaction, resulting in high yields and excellent stereoselectivity for the products (12–14, dr > 25:1). This indicates that the reaction protocol exhibits a certain degree of tolerance towards steric hindrance at this position.
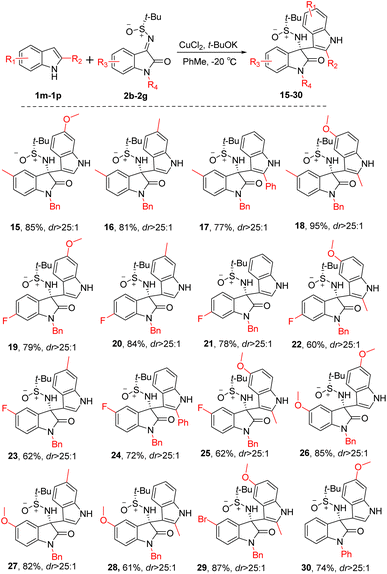
Subsequently, the present study concentrated on exploring the applicability of this novel Friedel–Crafts reaction to a diverse array of imine substrates. As illustrated in Scheme 3, reactions involving imine substrates with a benzyl group as the R4 group on the nitrogen atom consistently displayed high yields and significant stereoselectivity (15–29, dr > 25:1), irrespective of whether the R3 substituent on the benzene ring of the indole was an electron-donating or an electron-withdrawing group. Reactions involving imine substrates with a phenyl group as the R4 group on the nitrogen atom can also achieve relatively high yields and high stereoselectivity (30, dr > 25:1). This observation strongly suggests the broad substrate scope of the reaction protocol.
An X-ray crystallographic analysis of 3a confirmed the absolute stereochemistry of the newly formed compound (Scheme 4a), and the stereochemistries of the other compounds were determined based on that of 3a.
To gain deeper insights into the reaction mechanism, a series of control experiments were meticulously performed. The outcomes are detailed in Table 2. It was observed that a reduction in the t-BuOK equivalent led to a decline in the dr value. Remarkably, when Cu(MeCN)4(CF3SO3)2 was employed as the catalyst, in contrast to the condition without the addition of alkali metal ion additives (Entry 4), upon the addition of t-BuOK (Entry 5), a significant reversal in the reaction's stereoselectivity occurred (dr = 1:3 vs. dr > 25:1). This finding strongly suggests that the stoichiometric amount of potassium ions plays a pivotal role in determining the stereoselectivity of the reaction. Based on our prior studies and literature,26–30 Scheme 4b elucidates the proposed mechanism. Here, Cu2+ coordinates with the imine nitrogen and carbonyl oxygen. The N–H bond in the indole skeleton exhibits weak acidity, and t-BuOK can be used to deprotonate the N–H proton.31 This configuration promotes the transition state A, with the N-sulfinyl group adopting a synperiplanar (s-cis) conformation.29,32–34 K+ coordinates with the oxygen of the N-sulfinyl group, while indole coordinates with K+ by p-π activation of the delocalized π bond, thereby enhancing the reaction's stereoselectivity.35–37 Specifically, with (R)-N-tert-butylsulfinyl substrates, indole attacks from the less hindered Re-side of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond, avoiding steric hindrance with the bulky tert-butyl group, resulting in the formation of (S)-product C.
N bond, avoiding steric hindrance with the bulky tert-butyl group, resulting in the formation of (S)-product C.
| Entries | Catalysts (10 mol%) | Additives (mol%) | Yield of 3a a (%) | drb (3a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3b) 3b) |
|---|---|---|---|---|
| a Isolated yield.b The values of dr were determined by 1H NMR.c The standard condition. | ||||
| 1c | CuCl2 | t-BuOK (100) | 92 | >25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 2 | CuCl2 | t-BuOK (50) | 81 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 3 | CuCl2 | t-BuOK (25) | 73 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 4 | Cu(MeCN)4 | — | 30 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3 |
| (CF3SO3)2 | ||||
| 5 | Cu(MeCN)4 | t-BuOK (100) | 85 | >25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| (CF3SO3)2 | ||||
After the synthesis of several bisindoles under standard conditions, an investigation into the preliminary biological activities of these novel compounds was conducted (Table 3). Experimental findings revealed that these bisindoles exerted pronounced inhibitory effects against a diverse range of tumor cells. Notably, bisindole 29 exhibited selective inhibitory activity towards osteosarcoma cells MG63, with an IC50 value of 3.29 µM. Compound 29 exhibited significantly greater cytotoxicity towards osteosarcoma MG63 cells (IC50 = 4.03 µM) than towards normal human mesenchymal stem cells (IC50 = 18.28 µM), demonstrating its selective toxicity against cancer cells. Subsequently, we successfully achieved the gram-scale synthesis of bisindole 29, providing sufficient samples for subsequent pharmacological experiments. The diastereomeric excess (de value) was determined to be as high as 98% by HPLC analysis (Scheme 4c). Further investigations have elucidated that bisindole 29 exerts a multifaceted inhibitory action on MG63 cells. It exhibits a pronounced inhibitory effect on their proliferation, markedly curtails their colony-forming ability, induces cell-cycle arrest in the G1 phase, and suppresses their migration and invasion in a dose-dependent fashion. (See the SI for details on biological activity.)
| Compounds | IC50 (µM) | ||||||
|---|---|---|---|---|---|---|---|
| MCF-7 | H460 | A549 | SK-OV-3 | U251 | HepG2 | MG63 | |
| 3a | >100 | >100 | >100 | >100 | >100 | >100 | >100 |
| 4 | 36.78 | 81.44 | 60.69 | 53.53 | >100 | >100 | 40.72 |
| 5 | >100 | >100 | >100 | >100 | >100 | >100 | >100 |
| 6 | 26.88 | 39.89 | 41.73 | 18.50 | 29.03 | 24.98 | 16.29 |
| 7 | 16.80 | 37.78 | 38.64 | 17.78 | 29.14 | 17.95 | 15.60 |
| 8 | 32.13 | 46.89 | 67.24 | 75.12 | 36.83 | 28.57 | 29.80 |
| 9 | >100 | >100 | >100 | >100 | >100 | >100 | 66.06 |
| 10 | >100 | >100 | >100 | >100 | >100 | >100 | >100 |
| 11 | >100 | >100 | >100 | >100 | >100 | >100 | >100 |
| 12 | >100 | >100 | >100 | >100 | >100 | >100 | >100 |
| 13 | 32.17 | 30.39 | 35.37 | 55.12 | 75.23 | 42.34 | 45.08 |
| 14 | >100 | >100 | >100 | >100 | >100 | >100 | >100 |
| 15 | 42.18 | 88.2 | 22.19 | 73.90 | >100 | >100 | 61.48 |
| 16 | >100 | >100 | >100 | >100 | >100 | >100 | 75.81 |
| 17 | >100 | >100 | 60.17 | >100 | >100 | >100 | >100 |
| 18 | 41.19 | 34.89 | 45.44 | 33.14 | >100 | >100 | >100 |
| 19 | 18.84 | 35.67 | 34.54 | 23.13 | 20.70 | 26.00 | 15.95 |
| 20 | 37.09 | 39.85 | 60.51 | 43.89 | 31.28 | 36.76 | 25.11 |
| 21 | 81.78 | 83.28 | 36.34 | 83.93 | >100 | 45.24 | 51.61 |
| 22 | >100 | >100 | 68.88 | 78.02 | >100 | >100 | >100 |
| 23 | >100 | >100 | >100 | >100 | >100 | >100 | >100 |
| 24 | >100 | 74.22 | 23.64 | 63.89 | >100 | >100 | 83.72 |
| 25 | 57.64 | 19.80 | 35.23 | 40.26 | 32.10 | 56.98 | 62.84 |
| 26 | >100 | >100 | >100 | >100 | >100 | >100 | >100 |
| 27 | 47.42 | 20.43 | 33.66 | 70.29 | 43.19 | 24.58 | 20.79 |
| 28 | >100 | >100 | >100 | >100 | >100 | >100 | >100 |
| 29 | 39.10 | 25.22 | 16.87 | 28.67 | 83.36 | 17.4 | 3.29 |
| 30 | >100 | >100 | >100 | >100 | >100 | >100 | >100 |
| Doxorubicin | 6.72 | 3.13 | 20.32 | 32.17 | 16.74 | 10.13 | 4.24 |
Conclusions
In summary, we have devised a novel and expedient protocol for the synthesis of chiral bisindoles with remarkable diastereoselectivity. The developed methodology, utilizing K+/Cu2+ co-chelation catalysis, demonstrates versatility across various substituted indoles and diverse chiral imine substrates, showing its broad substrate compatibility. Initial biological investigations of the synthesized compounds have revealed that they exhibit potent inhibitory activity against the osteosarcoma cell line MG63, the hepatocellular carcinoma cell line HepG2, and other cell lines. Among all the synthesized compounds, bisindole 29 was ascertained to possess favorable anticancer activity and a pronounced selective inhibitory effect on osteosarcoma cells. Further investigations have elucidated that bisindole 29 exerts a multifaceted inhibitory action on MG63 cells. It exhibits a pronounced inhibitory effect on their proliferation, markedly curtails their colony-forming ability, induces cell-cycle arrest in the G1 phase, and suppresses their migration and invasion in a dose-dependent fashion. While we have identified the potent anti-tumor activity of bisindole 29, its underlying mechanisms require further elucidation. Our future work will be dedicated to systematically investigating the molecular mechanisms of bisindole 29 using omics technologies and novel probe technologies,38,39 with the aim of providing a robust scientific foundation for its development as a promising anti-cancer agent.Author contributions
Conceptualization, S. Q. and B. X.; methodology, S. Q. and S. Y.; validation, S. Q., S. Y. and M. X.; investigation, Y. L. and Z. D.; data curation, G. Z. and Z. L.; writing – original draft preparation, S. Y. and M. X.; writing – review and editing, S. Y., M. X., Y. L., Z. D., G. Z., Z. L., J. X., X. G., G. L. and Q. M.; supervision, S. Q. and B. X.; project administration, S. Q. and B. X.; funding acquisition, S. Q. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2423640 contains the supplementary crystallographic data for this paper.40The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ra09219a.
Acknowledgements
This research was funded by the Hunan Science Fund for Distinguished Young Scholars (No. 2025JJ20098); the National Natural Science Foundation of China (No. 82204250); the China Postdoctoral Science Foundation (No. 2021M693961); the Scientific Research Program of Fu Rong Laboratory (2023RC3071); the Natural Science Foundation of Hunan Province of China (2024JJ6487); High-level Talent Research Initiation Fund Project of Hunan University of Chinese Medicine (No. 0004010).References
- O. Allemann, M. Brutsch, J. C. Lukesh, D. M. Brody and D. L. Boger, J. Am. Chem. Soc., 2016, 138, 8376–8379 CrossRef CAS PubMed.
- J. Sugalski, F. M. Stewart, R. W. Carlson and J. N. . Compr, Canc. Netw., 2016, 14, 959–960 Search PubMed.
- M. Bonner, G. Lewis, L. Christiansen, D. Camp-bell, C. Tipper, A. Hamel, S. Shah, H. Horton, O. Negre, G. Veres and P. Gregory, Mol. Ther., 2016, 24, S86–S87 CrossRef.
- J. E. DeLorbe, D. Horne, R. Jove, S. M. Mennen, S. Nam, F. L. Zhang and L. E. Overman, J. Am. Chem. Soc., 2013, 135, 4117–4128 Search PubMed.
- L. Yang, X. Li, W. Huang, X. Rao and Y. Lai, Biomed. Pharmacother., 2022, 151, 113112 CrossRef CAS PubMed.
- Z. Xiao, Y. Hao, B. Liu and B. Qian, Leuk. Lymphoma, 2002, 43, 1763–1768 CrossRef CAS PubMed.
- A. M. Ramadan, M. A. Abdulgader, M. A. Khan, N. O. xGadalla and A. Bahieldin, PAK. J. BOT., 2020, 52, 1305–1313 CAS.
- A. B. Smitherman, M. Troy, C. Beach, A. M. Deal and S. H. Gold, JCO, 2016, 34, 10535 Search PubMed.
- M. Mano, Cancer Treat Rev., 2006, 32, 106–118 CrossRef CAS PubMed.
- D. Stuckler, J. Singhal, S. S. Singhal, S. Yadav, S. Awasthi and S. xAwasthi, Cancer Res., 2005, 65, 991–998 Search PubMed.
- E. S. Kim, Drugs, 2017, 77, 1251–1259 CrossRef CAS PubMed.
- S. Kayser, M. J. Levis and R. F. Schlenk, N. Engl. J. Med., 2017, 377, 1901–1903 CrossRef PubMed.
- W. E. Borek, W. E. Nobre, S. F. xPedicona, A. E. Camp-bell, J. A. Christopher, N. Nawaz, D. N. Perkins, P. Moreno-Cardoso, J. Kelsall, H. R. Ferguson, B. Patel, P. Gallipoli, P. Arruda, A. J. x Ambinder, A. Thompson, A. Williamson, G. Ghiaur, M. D. Minden, J. G. Gribben, D. J. Britton, P. R. Cutillas and A. D. xDokal, EBioMedicine, 2024, 108, 105316 CrossRef CAS PubMed.
- J. Zhu, Y. xHe, J. Li, N. Ding, X. Wang and Y. Song, Blood, 2018, 132, 1666 Search PubMed.
- M. W. Xu, R. Peng, Q. Min, S. W. Hui, X. Chen, G. Yang and S. L. Qin, Eur. J. Med. Chem., 2022, 243, 114748 CrossRef CAS PubMed.
- M. J. Siddiqui, Z. Ismail, A. F. A. Aisha and A. M. S. Abdul Maji, Int. J. Pharm., 2009, 6, 43–47 Search PubMed.
- H. Ishikawa, D. A. Colby, S. Seto, P. Va, A. Tam, H. Kakei, T. J. Rayl, I. Hwang and D. L. Boger, J. Am. Chem. Soc., 2009, 131, 4904–4916 CrossRef CAS PubMed.
- L. Y. Wang, J. Miao, Y. Zhao and B. M. Yang, Org. Lett., 2023, 25, 1553–1557 CrossRef CAS PubMed.
- P. Zhang, Q. Xu, X. Wang, J. Feng, C. Lu, Y. Li and R. Liu, Angew. Chem., Int. Ed., 2022, 61, e202212101 CrossRef CAS PubMed.
- W. Zhang, W. Xu, G. Y. Wang, X. Y. Gong, N. P. Li, L. Wang and W. C. Ye, Org. Lett., 2017, 19, 5194–5197 Search PubMed.
- S. Kang, Y. Wu, M. Hu, Y. Ma, X. Huang, Z. Hao, X. Li, W. Chen and H. Zhang, Org. Lett., 2023, 25, 3466–3470 Search PubMed.
- J. Wang and J. Sun, Angew. Chem., Int. Ed., 2025, 64, e202424773 CrossRef CAS PubMed.
- A. Boos, J. Most, H. Cahuzac, L. Moreira Da Silva, F. Daubeuf, S. Erb, S. Cianférani, O. Hernandez-Alba, C. Semenchenko, I. Dovgan, S. Kolodych, A. Detappe, F. Dantzer, A. Wagner, M. Zeniou and G. Chaubet, J. Med. Chem., 2025, 68, 695–705 Search PubMed.
- J. P. Chen, W. W. Chen, Y. Li and M. H. Xu, Org. Biomol. Chem., 2015, 13, 3363–3370 RSC.
- E. Wojaczyńska and J. Wojaczyński, Chem. Rev., 2020, 120, 4578–4611 CrossRef PubMed.
- S. Qin, S. Liu, Y. Cao, J. Li, C. Chong, T. Liu, Y. Luo, J. Hu, S. Jiang, H. Zhou, G. Yang and C. Yang, Org. Lett., 2018, 20, 1350–1354 CrossRef CAS PubMed.
- S. Qin, L. Yao, Y. Luo, T. Liu, J. Xu, Y. Sun, N. Wang, J. Yan, B. Tang, G. Yang and C. Yang, Org. Chem. Front., 2020, 7, 3132–3136 RSC.
- S. Qin, T. Liu, Y. Luo, S. Jiang and G. Yang, Org. Chem. Front., 2019, 6, 732–735 RSC.
- L. Wu, C. Xie, H. Mei, V. A. Soloshonok, J. Han and Y. Pan, J. Org. Chem., 2014, 79, 7677–7681 CrossRef CAS PubMed.
- C. Xie, L. Zhang, H. Mei, R. Pajkert, M. Ponoma-renko, Y. Pan, G. Röschenthaler, V. A. Soloshonok and J. Han, Chem. Eur. J., 2016, 22, 7036–7040 CrossRef CAS PubMed.
- J. Alvarez-Builla, J. Vaquero and J. Barluenga, Modern Heterocyclic Chemistry, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2011, pp. 2380–2445 Search PubMed.
- L. B. Schenkel and J. A. Ellman, Org. Lett., 2003, 5, 545–548 CrossRef CAS PubMed.
- T. D. Owens, F. J. Hollander, A. G. Oliver and J. A. Ellman, J. Am. Chem. Soc., 2001, 123, 1539–1540 CrossRef CAS.
- T. D. Owens, A. J. Souers and J. A. Ellman, J. Org. Chem., 2003, 68, 3–10 CrossRef CAS PubMed.
- S. F. Gao, X. Fu, Q. M. Zhang, R. Z. Tang, W. J. Wan and L. X. Wang, Org. Biomol. Chem., 2025, 23, 4100–4105 RSC.
- M. Hatano, T. Mochizuki, K. Nishikawa and K. Ishi-hara, ACS Catal., 2017, 8, 349–353 CrossRef.
- L. D. Quin and J. Tyrell, Fundamentals of Heterocyclic Chemistry: Importance in Nature and in the Synthesis of Pharmaceuticals, John Wiley & Sons, Inc., Hoboken, New Jersey, 2010, pp. 1–345 Search PubMed.
- S. L. Qin and X. H. Xiao, Future Med. Chem., 2025, 17, 987–989 CrossRef CAS PubMed.
- S. Yan, G. S. Zhang, W. Luo, M. W. Xu, R. Peng, Z. W. Du, Y. Liu, Z. F. Bai, X. H. Xiao and S. L. Qin, Eur. J. Med. Chem., 2024, 276, 116725 CrossRef CAS PubMed.
- CCDC 2423640: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2mbzyx.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |