 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Fluorinated terpenoids and their fluorine-containing derivatives
Muhammad Badrul Hudaab,
Nurlelasaria,
Unang Supratman ac and
Yudha P. Budiman
ac and
Yudha P. Budiman *a
*a
aDepartment of Chemistry, Faculty of Mathematics and Natural Sciences, Universitas Padjadjaran, Jl. Raya Bandung-Sumedang Km 21, Sumedang 45363, West Java, Indonesia. E-mail: y.p.budiman@unpad.ac.id
bDepartment of Chemistry, Faculty of Sciences and Mathematics, Universitas Diponegoro, Jl. Prof. Soedarto No. 13 Tembalang, Semarang 50275, Central Java, Indonesia
cCentral Laboratory, Universitas Padjadjaran, Jl. Raya Bandung-Sumedang Km 21, Sumedang 45363, West Java, Indonesia
First published on 4th February 2026
Abstract
Fluorine-containing molecules have attracted increasing attention in pharmaceutical chemistry owing to their unique physicochemical properties. As there are no known examples of naturally occurring fluorinated organic molecules, fluorination of organic molecules gains significant interest in organic synthesis. Terpenoids represent one of the largest and most structurally diverse classes of natural products, exhibiting a broad range of pharmacological effects. Fluorination of terpenoid scaffolds has therefore become an active area of research aimed at enhancing or diversifying their bioactivity profiles. This review summarizes the synthetic strategies developed for the preparation of fluorinated terpenoids across different subclasses, including mono-, sesqui-, di-, tri-, and tetranor-terpenes.
1. Introduction
Terpenoids represent the most structurally diverse class of natural products, comprising more than 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 known compounds distributed across all kingdoms of life.1 Naturally occurring terpenoids exhibit a remarkably broad spectrum of biological activities, including anticancer,2 anti-inflammatory,3 antimalarial,4 cytotoxic,5 antiviral, as well as hepato- and cardioprotective properties.6 In parallel, the incorporation of fluorine into organic molecules has gained significant attention, particularly in pharmaceutical and agrochemical synthesis, owing to its profound impact on metabolic stability, bioavailability, and overall biological performance.7–14
000 known compounds distributed across all kingdoms of life.1 Naturally occurring terpenoids exhibit a remarkably broad spectrum of biological activities, including anticancer,2 anti-inflammatory,3 antimalarial,4 cytotoxic,5 antiviral, as well as hepato- and cardioprotective properties.6 In parallel, the incorporation of fluorine into organic molecules has gained significant attention, particularly in pharmaceutical and agrochemical synthesis, owing to its profound impact on metabolic stability, bioavailability, and overall biological performance.7–14
In recent years, approximately 25% of all marketed drugs have been fluoro-pharmaceuticals, representing a remarkably high proportion compared to other halogen-containing therapeutics, despite the natural abundance of organochlorine and organobromine compounds.15 Notably, several clinically relevant fluorinated pharmaceuticals belong to the terpenoid family and exhibit potent anticancer and anti-inflammatory activities. The unique properties of the fluorine atom—its high electronegativity, small atomic radius, and the low polarizability of the C–F bond—can profoundly influence the behaviour of a molecule within biological systems.16–18 More broadly, the introduction of fluorine can modulate key pharmacological processes, including receptor or enzyme binding, metabolic stability and clearance, absorption and transport, and the modulation or inhibition of enzymatic pathways.19–22
Several studies have demonstrated that fluorinated terpenoid derivatives often exhibit enhanced biological activity. For instance, omaveloxolone (1), a fluorinated oleanane derivative, has been approved for the treatment of Friedreich's ataxia (Fig. 1).23 Cytotoxicity studies have further shown that introducing a fluorine atom at the C-2 position of lupane scaffolds (e.g., compounds 2 and 3) significantly improves their antitumor potency.24 Likewise, fluorinated docetaxel (4) displays superior anticancer activity compared with the parent drug, particularly in the treatment of advanced ovarian and breast cancers.25 These examples highlight the broader trend that fluorinated terpenoids represent a valuable class of bioactive molecules with considerable therapeutic potential.
 | ||
| Fig. 1 Representative fluorinated terpenoids and their significance as pharmaceutical agents. (1) Omaveloxolone, a therapeutic agent for neurodegenerative diseases.1 (2) 2,2-Difluorodihydrobetulonic acid, an anticancer compound.24 (3) Difluoroallobetulone, an anticancer compound.2 (4) N-De-t-butoxycarbonyl-N-[2-(1,1,1-trifluoro-2-methyl)pro-pyloxycarbonyl]-30-dephenyl-30-(4-fluorophenyl)docetaxel, an antitumor agent.3 | ||
The introduction of a fluorine atom in place of a hydrogen or hydroxyl group generally does not result in substantial steric perturbation. However, intramolecular interactions involving the fluorine substituent can induce notable conformational changes within the molecule. Fluorination often slows down metabolic degradation relative to the non-fluorinated parent structure. Moreover, fluorinated compounds frequently display increased chemical stability, improved solubility, and enhanced bioavailability—attributes that are partly associated with their typically higher lipophilicity.26–29 Despite the numerous reports describing the synthesis of various fluorinated terpenoid derivatives (including mono-, di-, sesqui-, tri-, and tetranor-terpenoids), a systematic overview of the synthetic methodologies remains lacking. This review covers reports on fluorinated terpenoids and their fluorine-containing derivatives published up to the end of 2024. In this review, we aim to fill this gap by summarizing the principal strategies for the synthesis of fluorinated terpenoids, with a particular focus on the reagents and catalysts employed.
2. Synthesis of fluorinated terpenoids
2.1. Fluorinated monoterpenoids
Natural compounds, such as the monoterpenoids found in natural essential oils, are crucial sources of numerous biological activities. Research on fluorinated monoterpenoids has been conducted by numerous researchers to increase their bioactivity. Numerous of the resulting fluorinated monoterpenoids have strong biological activity; these include cytotoxic, analgesic, pesticides, and antiviral properties.30 | ||
| Scheme 1 Fluorination of hydroxyl-containing compounds using fluoroalkylation esterification.32 | ||
The key transformation—esterification of the monoterpenoid alcohol—proceeded efficiently without reported side reactions such as olefin fluorination, rearrangement, or elimination, suggesting good functional group tolerance toward isolated C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bonds and allylic systems. This is particularly noteworthy for substrates like geraniol and linalool derivatives, where competing reactions at allylic positions could be anticipated under more aggressive fluorination conditions. The mildness of the esterification protocol thus represents a practical advantage for late-stage functionalization of terpene scaffolds.32 The insecticidal activities of the fourteen fluorinated monoterpen were evaluated against the larvae of Musca domestica. It has been found that the study revealed that the fluoroacetate derivatives of cyclic monoterpenoids exhibited the highest potency and were the most effective house fly fumigants.32
C double bonds and allylic systems. This is particularly noteworthy for substrates like geraniol and linalool derivatives, where competing reactions at allylic positions could be anticipated under more aggressive fluorination conditions. The mildness of the esterification protocol thus represents a practical advantage for late-stage functionalization of terpene scaffolds.32 The insecticidal activities of the fourteen fluorinated monoterpen were evaluated against the larvae of Musca domestica. It has been found that the study revealed that the fluoroacetate derivatives of cyclic monoterpenoids exhibited the highest potency and were the most effective house fly fumigants.32
Halogenated tetrahydropyranyl rings are typically generated through Prins reactions between homoallylic alcohols and aldehydes, catalysed by suitable halogen-containing Lewis acids or ionic liquids (Scheme 2). Only a few examples describing the introduction of fluorine via halo-Prins cyclization have been reported to date.35,36 In these reactions, BF3·Et2O functions dually as the catalyst and the fluorine source.
 | ||
| Scheme 2 Fluorination via the Prins reaction.30 | ||
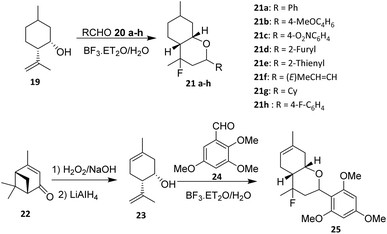
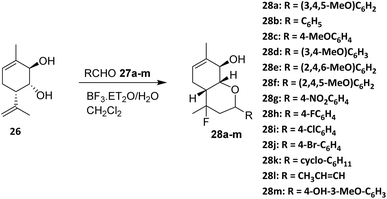
In 2016, Michalchenko et. al. reported the fluorination of para-mentha 6,8-dien-2,3diol (26) using boron trifluoride diethyl etherate (BF3·Et2O) as the fluorine source. These research was developed to study the effect of substituents at the aldehyde aromatic ring (27a–m) on the reaction yield (Scheme 3). Among the aldehyde examined, 3,4,5-trimethoxy benzaldehyde (28a) was formed with the highest yield.37 The reaction tolerates terpene-derived olefins and cyclic backbones, indicating reasonable functional group compatibility within rigid monoterpenoid scaffolds.
 | ||
| Scheme 3 Fluorination of para-mentha-6,8-dien-2,3-diol (26) via the Prins reaction.37 | ||
A key determinant of reaction efficiency is the nature of the aldehyde component. Michalchenko et al. systematically evaluated a range of aromatic aldehydes bearing different substituents and demonstrated that electron-donating groups markedly enhance reaction yields. Notably, 3,4,5-trimethoxybenzaldehyde afforded the highest yield, which is consistent with enhanced stabilization of the oxocarbenium intermediate during the cyclization step. These results underscore the dominant role of electronic effects in governing reaction outcomes, while steric congestion on the aromatic ring can impose additional constraints.
Despite its synthetic utility, the halo-Prins fluorination strategy suffers from inherent limitations. The requirement for strong Lewis acidic conditions restricts compatibility with acid-sensitive functional groups and may limit broader application in complex, multifunctional monoterpenoid systems.
 | ||
| Scheme 4 K10-clay catalyzed Prins cyclization reaction.39 | ||
Among the aldehydes examined, the reaction employing 4-(trifluoromethyl)benzaldehyde (30c) afforded the highest yield of intermolecular products. These observations highlight the role of electronic effects in steering reaction pathways and product distributions, providing insight into structure–reactivity relationships within this system. The selection of fluorine-containing substituents in the aldehydes was motivated by the need to investigate structure–activity relationships, particularly the influence of substituent electronic effects on the structures of the products formed in reactions with verbenol epoxide (29).39
 | ||
| Scheme 5 Zeolite-catalyzed Prins cyclization reaction.40 | ||
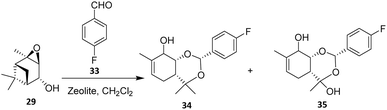
The reaction between verbenol oxide 29 and 4-fluorobenzaldehyde 33 was investigated over a temperature range of 25–70 °C using either H-Beta zeolite or Fe-modified Beta catalysts. Complete conversion of verbenol oxide was achieved over H-Beta-25 in toluene using a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 ratio of reactants, affording the isomerization products 34 and 35 as the major products. This outcome indicates that excessive acidity favors skeletal rearrangement of the terpene framework over intermolecular C–C bond formation, thereby limiting selectivity toward fluorinated products. In addition to H-Beta-25, Fe–H-Beta-150 also proved to be a promising catalyst for this transformation, yielding approximately 10% of the targeted fluorinated product under the optimized conditions.40 Overall, the study demonstrates that heterogeneous zeolite-based fluorination of monoterpenoids is feasible but highly sensitive to acid strength and catalyst composition, necessitating further optimization to expand substrate scope and improve selectivity.
5 ratio of reactants, affording the isomerization products 34 and 35 as the major products. This outcome indicates that excessive acidity favors skeletal rearrangement of the terpene framework over intermolecular C–C bond formation, thereby limiting selectivity toward fluorinated products. In addition to H-Beta-25, Fe–H-Beta-150 also proved to be a promising catalyst for this transformation, yielding approximately 10% of the targeted fluorinated product under the optimized conditions.40 Overall, the study demonstrates that heterogeneous zeolite-based fluorination of monoterpenoids is feasible but highly sensitive to acid strength and catalyst composition, necessitating further optimization to expand substrate scope and improve selectivity.
 | ||
| Scheme 6 Fluorination via Cu(I)-catalyzed click chemistry.42 | ||
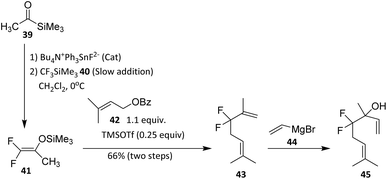 | ||
| Scheme 7 Fluoroallylation reaction to form the monoterpene 4,4-difluorolinalool.43 | ||
The key intermediate in this methodology is 1,1-difluoro-2-trimethylsilyloxypropene (41), which, upon coupling with prenyl benzoate (42), affords the difluoroheptenone derivative 43. This intermediate readily participates in coupling with prenyl benzoate to afford the difluoroheptenone derivative, indicating good compatibility with allylic esters and conjugated systems commonly found in monoterpenoids. The reaction sequence tolerates unsaturation and secondary alcohol precursors, underscoring its applicability to acyclic terpene substrates. Treatment of 43 with vinylmagnesium bromide (44) then yields the targeted gem-difluorinated monoterpenoid, 4,4-difluorolinalool (45).43 This step exploits the electrophilicity of the carbonyl group while preserving the gem-difluoro unit, highlighting the stabilizing effect of fluorine on adjacent carbocationic or anionic intermediates. The successful Grignard addition also indicates that the CF2 group does not impede nucleophilic attack, an important consideration for downstream functionalization.
In the first step of the reaction, the corresponding trimethylsilyl ethers 47 were formed. Subsequent acidic hydrolysis yielded the trifluoromethylated products—myrtenol derivatives 48—in a total isolated yield of 81% with a diastereomeric excess (de) of 17%. The diastereomeric excess was determined from the ratio of signal intensities of compounds 48 and 49 in the 1H NMR spectrum.
To avoid resinification and because the TMSCF3–TBAF·3H2O system is unstable under prolonged reaction times or elevated temperatures, the trifluoromethylation of 2-formylisopinocampheyl-3-thioacetate 50 was conducted at 30 °C with the reaction time extended to three days. Under these conditions, diastereomers 52 and 53 were obtained in a 52% combined yield with 42% de, with thioacetate 52 being the predominant component (Scheme 8). In contrast, thioacetate 54 did not react with the Ruppert–Prakash reagent (TMSCF3–TBAF·3H2O) under the same conditions, likely due to steric hindrance arising from the bulky TBAF species. Consequently, an alternative fluoride initiator, CsF, which is also employed in tandem nucleophilic CF3-addition protocols, was evaluated. Using the TMSCF3–CsF system, trifluoromethylated thioacetate 56 was formed as the sole (4S)-diastereomer in 37% yield. The reaction also produced the desulfurized product 57 in 31% yield (Scheme 8).
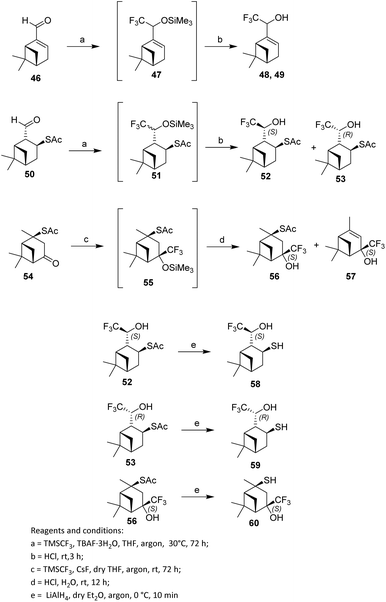 | ||
| Scheme 8 Fluorination involving the Ruppert–Prakash reagent.44 | ||
The deacylation of thioacetates 52, 53, and 56 with lithium aluminium hydride (LiAlH4) in dry Et2O under an argon atmosphere afforded the corresponding thiols 58, 59, and 60 in 73–90% yields, while retaining the stereochemical configuration at the stereocentres (Scheme 8).
The trifluoromethylation of the separated diastereomers of sulfinimines 61a,b was achieved via nucleophilic addition of the Ruppert–Prakash reagent, TMSCF3 (trifluoromethyltrimethylsilane), to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N double bond in the presence of TMAF (tetramethylammonium fluoride) as the fluoride source in THF at −65 °C. This transformation yielded the diastereomeric sulfinamides 62a,b in 42–58% yields with 87–99% de (Scheme 9). In the study by Sudarikov et al. (2018), it was shown that trifluoromethylation of (SS)-tert-butanesulfinimines using the Ruppert–Prakash system (TMSCF3–TBAT/TMAF) predominantly generates the corresponding (SSR)-trifluoromethylated derivatives, while (RS)-tert-butanesulfinimines mainly afford the (RSS)-diastereomers. These observations are consistent with single-crystal X-ray diffraction data of sulfinamide (SSR)-62b. Combined with NMR spectroscopic data—specifically, the chemical shifts and spin–spin coupling constants of the C(1′)H and NH protons—it was concluded that the other possible diastereomers, namely (SSR)-62a, (RSS)-62a,b, (SSS)-62a,b, and (RSR)-62a,b, are formed only in trace amounts.45
N double bond in the presence of TMAF (tetramethylammonium fluoride) as the fluoride source in THF at −65 °C. This transformation yielded the diastereomeric sulfinamides 62a,b in 42–58% yields with 87–99% de (Scheme 9). In the study by Sudarikov et al. (2018), it was shown that trifluoromethylation of (SS)-tert-butanesulfinimines using the Ruppert–Prakash system (TMSCF3–TBAT/TMAF) predominantly generates the corresponding (SSR)-trifluoromethylated derivatives, while (RS)-tert-butanesulfinimines mainly afford the (RSS)-diastereomers. These observations are consistent with single-crystal X-ray diffraction data of sulfinamide (SSR)-62b. Combined with NMR spectroscopic data—specifically, the chemical shifts and spin–spin coupling constants of the C(1′)H and NH protons—it was concluded that the other possible diastereomers, namely (SSR)-62a, (RSS)-62a,b, (SSS)-62a,b, and (RSR)-62a,b, are formed only in trace amounts.45
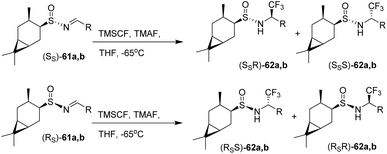 | ||
| Scheme 9 Fluorination via the Ruppert–Prakash reagent activated by TMAF.45 | ||
Petrova et al. employed ketooximes 63, 67, and 71, derived from (+)-nopinone, (−)-verbanone, and (+)-camphorquinone, respectively, to prepare the corresponding benzyl-O-oximes 64, 68, and 72. These compounds were obtained in 68%, 55%, and 45% yields, respectively. Benzyl-O-oximes 64, 68, and 72 possess two potential reactive centres—the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and C
O and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bonds—both of which, in principle, may undergo trifluoromethylation. As illustrated in Scheme 10, imines and sulfinimines typically react at the C
N bonds—both of which, in principle, may undergo trifluoromethylation. As illustrated in Scheme 10, imines and sulfinimines typically react at the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond, whereas monoterpene-derived ketooximes 64, 68, and 72 undergo trifluoromethylation exclusively at the C
N bond, whereas monoterpene-derived ketooximes 64, 68, and 72 undergo trifluoromethylation exclusively at the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond.
O bond.
 | ||
| Scheme 10 Trifluoromethylation of camphor-derived imines using the Ruppert–Prakash reagent.46 | ||
Nucleophilic addition of the Ruppert–Prakash reagent (trifluoromethyltrimethylsilane, TMSCF3) to β-keto benzyl-O-oximes 64, 68, and 72 at the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O double bond was performed in THF at 4 °C under an argon atmosphere using cesium fluoride (CsF) as the initiator. In the first step, the corresponding trimethylsilyl ethers 65, 69, and 73 were formed. Subsequent treatment with tetrabutylammonium fluoride hydrate (TBAF·3H2O) afforded the trifluoromethylated alcohols 66, 70, and 74 in 81%, 89%, and 95% yields, respectively (Scheme 10).46
O double bond was performed in THF at 4 °C under an argon atmosphere using cesium fluoride (CsF) as the initiator. In the first step, the corresponding trimethylsilyl ethers 65, 69, and 73 were formed. Subsequent treatment with tetrabutylammonium fluoride hydrate (TBAF·3H2O) afforded the trifluoromethylated alcohols 66, 70, and 74 in 81%, 89%, and 95% yields, respectively (Scheme 10).46
Application of β-fluorination conditions furnished compound 76 in 55% yield, supporting the proposed involvement of the ketone functionality in directing hydrogen-atom abstraction and subsequent fluorination.49 Notably, alternative N–F reagents—such as N-fluorobenzenesulfonimide (NFSI) and N-fluoropyridinium tetrafluoroborate—failed to produce the fluorinated product 76 (Scheme 11). Although NFSI can react with alkyl radicals, Selectfluor reacts substantially faster and is more likely to participate in ketone-mediated electron-transfer pathways crucial for this transformation.
 | ||
| Scheme 11 Ketone-directed C–H fluorination of menthone using Selectfluor under benzil-sensitized visible-light photocatalysis.49 | ||
In another study, Xia et al. (2014) demonstrated that vanadium(III) oxide efficiently catalyses the direct fluorination of C(sp3)–H bonds using Selectfluor. This protocol was applied to the monoterpene 1,4-cineole 77, affording a highly selective transformation under operationally simple conditions (Scheme 12). The heterogeneous catalyst, along with reaction by-products, can be removed readily by filtration. Fluorination occurs preferentially at the tertiary C–H site, yielding product 78 in 53% isolated yield.50
 | ||
| Scheme 12 V2O3-catalyzed direct C–H fluorination of 1,4-cineole using Selectfluor.50 | ||
Fluorination occurs preferentially at the tertiary C–H position, which is both sterically accessible and energetically favored owing to its lower bond dissociation energy relative to secondary or primary C–H bonds. This regioselectivity is consistent with a radical-type mechanism, in which hydrogen atom abstraction constitutes the rate-determining step and is governed primarily by C–H bond strength rather than by electronic directing effects.
In 2016, Ye et al. reported a palladium-catalyzed fluorination of the monoterpenoid derivative 79.52 The key to this transformation was the addition of triethyl(trifluoromethyl)silane (TESCF3), which effectively addressed the regioselectivity issues commonly encountered in Pd-catalyzed fluorination. Together with a newly designed biarylphosphine ligand, TESCF3 enabled the development of a highly regioselective and efficient fluorination protocol. Under the optimized conditions, the desired fluorinated monoterpenoid 80 was obtained in 69% yield with excellent regioselectivity (Scheme 13).
 | ||
| Scheme 13 Palladium-catalyzed fluorination of monoterpenoid derivative 79 using TESCF3 as an additive.51 | ||
In contrast, Prins-type fluorination and clay- or zeolite-catalyzed cyclizations enable skeletal reorganization and the incorporation of fluorinated aromatic units, albeit at the cost of requiring strongly Lewis acidic conditions. Consequently, their substrate scope is restricted by susceptibility to acid-promoted rearrangements, and reaction outcomes are highly sensitive to both the electronic nature of the aldehyde and the acidity of the catalyst.
Click chemistry (CuAAC) and cross-coupling methodologies provide superior regioselectivity, consistently high yields, and reliable performance, making them powerful tools for late-stage functionalization. Their principal limitation lies in the need for prefunctionalized substrates, such as alkynes or halides, which increases synthetic complexity and may limit rapid analogue generation. Fluoroallylation and gem-difluorination using silylated fluorinated reagents (e.g., acetyltrimethylsilane, TFMTMS) afford access to structurally distinctive monoterpenoid derivatives while preserving olefinic functionality; however, these multistep strategies are largely confined to acyclic systems and require careful control of reaction conditions to suppress competing side reactions.
Direct fluorination methods highlight the inherent trade-off between reactivity and selectivity. Nucleophilic trifluoromethylation with the Ruppert–Prakash reagent (TMSCF3) enables efficient CF3 incorporation with tunable diastereoselectivity, yet is limited by steric hindrance, reagent instability, and dependence on fluoride activators. Electrophilic fluorination using Selectfluor—including ketone-directed and metal-catalyzed C–H fluorination—permits late-stage, site-selective modification of rigid monoterpenoid scaffolds without prefunctionalization. Nevertheless, these transformations often suffer from modest yields, restricted positional flexibility, and strong dependence on substrate geometry and C–H bond strength. Although Pd-catalyzed fluorination can deliver enhanced regioselectivity with appropriate ligand design and additives, its broader application is curtailed by catalyst cost, narrow substrate scope, and the need for extensive optimization. Overall, no single fluorination strategy is universally optimal; rather, method selection must balance substrate architecture, the desired mode of fluorine incorporation, and tolerance to reaction conditions.
2.2. Fluorinated sesquiterpenoids
Sesquiterpenoids are a class of naturally occurring terpenoid compounds composed of 15 carbon atoms (C15), typically assembled from three isoprene units (C5H8). Unlike simple sesquiterpenes, sesquiterpenoids contain additional functional groups—such as hydroxyls, aldehydes, ketones, or epoxides—that significantly expand their chemical diversity and biological activity. Because of their structural complexity and broad pharmacological potential, incorporating fluorine atoms into sesquiterpenoid frameworks has become an important strategy for modulating their physicochemical properties, metabolic stability, and bioactivity.The conversion of α-methylene-γ-butyrolactones into α-alkylidene-γ-butyrolactones via metal-catalyzed processes has gained considerable interest due to its relevance in the efficient synthesis of biologically active natural products that contain these structural motifs.54,55
In 2009, Han et al. demonstrated the application of the Heck reaction to convert parthenolide (81), containing the α-methylene-γ-lactone substructure, into a series of α-benzylidene-γ-lactones (83a–e) with excellent selectivity for the E-olefin geometry (Scheme 14).56 This strategy provides α-benzylidene-γ-lactone derivatives in good yields and offers a versatile approach for constructing analogues of natural products bearing the α-methylene-γ-lactone motif. In this transformation, only exocyclic olefin products are obtained because the C11–C13 insertion must occur opposite the C7 proton. The required β-hydride elimination is not possible on the same face as the C7 protons, making the formation of the exocyclic olefin the exclusive pathway.56
 | ||
| Scheme 14 Palladium(II)-catalyzed Heck-type coupling of parthenolide (81) to afford α-benzylidene-γ-lactones (83a–e).56 | ||
In 2011, Woods et al. reported that PTL reacts smoothly with a variety of primary and secondary aliphatic amines to afford the corresponding amino-parthenolide analogues (85a–f) (Scheme 15). The reaction displays broad substrate applicability toward aliphatic amines with varying steric demands, indicating good functional group tolerance under mild conditions. Notably, the electrophilic α,β-unsaturated lactone moiety of PTL undergoes selective Michael addition without competitive epoxide ring opening or scaffold degradation, underscoring the high chemoselectivity of the process. Importantly, the Michael addition was found to be highly stereospecific, delivering exclusively a single stereoisomer bearing the R-configuration at C-11. The alternative diastereomer featuring the S-configuration at that stereocenter was not detected in the reaction mixture.58 This selectivity is governed by the rigid bicyclic framework of PTL, which enforces facial differentiation of the enone and directs nucleophilic attack to the less hindered face. The absence of the S-diastereomer suggests a strong conformational and steric control inherent to the natural product scaffold rather than catalyst-induced selectivity.
 | ||
| Scheme 15 Michael addition (Mannich-type) reaction of parthenolide (PTL) with aliphatic amines to afford amino-parthenolide analogues.58 | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of 87 and 88, which can be separated by recrystallization from diethyl ether to give monofluorinated analogs 87 (31% yield) and 88 (12% yield). The observed 3
1 mixture of 87 and 88, which can be separated by recrystallization from diethyl ether to give monofluorinated analogs 87 (31% yield) and 88 (12% yield). The observed 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereomeric ratio reflects competing reaction pathways governed by local steric and conformational constraints within the rigid PTL framework. Although recrystallization allows separation of the individual isomers, the modest overall yields highlight a key limitation of DAST-mediated fluorination when applied to structurally complex natural products. Furthermore, oxidation of 86 with Dess–Martin periodinane in the presence of NaHCO3 furnishes aldehyde 89, which reacts with DAST to give the difluorinated analog 90 in 43% yield.60 This result suggests that carbonyl substrates offer a more predictable and efficient route for fluorine incorporation compared to secondary alcohols.
1 diastereomeric ratio reflects competing reaction pathways governed by local steric and conformational constraints within the rigid PTL framework. Although recrystallization allows separation of the individual isomers, the modest overall yields highlight a key limitation of DAST-mediated fluorination when applied to structurally complex natural products. Furthermore, oxidation of 86 with Dess–Martin periodinane in the presence of NaHCO3 furnishes aldehyde 89, which reacts with DAST to give the difluorinated analog 90 in 43% yield.60 This result suggests that carbonyl substrates offer a more predictable and efficient route for fluorine incorporation compared to secondary alcohols.
 | ||
| Scheme 16 Fluorination with DAST.60 | ||
In 2022, Adekenov et al. carried out the fluorination of the sesquiterpene grossheimin 91 using DAST. The presence of an easily acylated secondary hydroxyl group drives grossheimin toward fluorination. The reaction of grossheimin 91 with DAST was performed in methylene dichloride and produced a complex mixture, from which two compounds, 92 and 93, were isolated in low yields (3% and 5%, respectively).
The skeletal rearrangement leading to 92 from 91 is triggered by nucleophilic attack of the 8-hydroxyl group of grossheimin on the hypervalent sulfur center of DAST. The activated hydroxyl can then be directly attacked by a fluoride anion to form 93, or alternatively rearrange to a non-classical cyclopropylmethyl cation through nucleophilic attack from the C10(14) exomethylene. Following a formal Demjanov-type ring expansion, the resulting cyclobutyl cation is ultimately trapped by a fluoride anion (Scheme 17).61
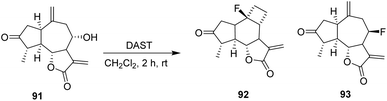 | ||
| Scheme 17 DAST-mediated deoxyfluorination of grossheimin.61 | ||
The synthesis of several MMB–1,2,3-triazole fluorinated derivatives has been reported via reaction of a C-14 azido derivative of MMB with a variety of aryl-fluorinated reagents through click-chemistry protocols. In 1982, Feraly reported melampomagnolide B (MMB) 86, an anticancer sesquiterpene lactone isolated from Magnolia grandiflora,63 a plant that produces several potent sesquiterpene-lactone derivatives active against both hematological and solid tumors.64
The MMB mesylate 94 was synthesized by reacting MMB with methanesulfonyl chloride in dichloromethane at 0 °C for 30 min. Compound 94 was then treated with sodium azide in the presence of dimethylformamide and acetonitrile at 80 °C for 1 h to afford the azido synthon 95. Intermediate 95 was subsequently reacted with a series of aryl-fluorine reagents (96a–c) in the presence of CuI/triethylamine/acetonitrile–water (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at ambient temperature to give the corresponding MMB-triazole derivatives 97a–c (Scheme 18).65 The CuAAC reaction proceeds efficiently under ambient conditions, furnishing triazole-linked products with high regioselectivity, which underscores the inherent robustness of click chemistry. Notably, aryl-fluorine substituents do not interfere with copper catalysis, demonstrating excellent functional group tolerance and enabling late-stage incorporation of fluorinated aromatic motifs.
1) at ambient temperature to give the corresponding MMB-triazole derivatives 97a–c (Scheme 18).65 The CuAAC reaction proceeds efficiently under ambient conditions, furnishing triazole-linked products with high regioselectivity, which underscores the inherent robustness of click chemistry. Notably, aryl-fluorine substituents do not interfere with copper catalysis, demonstrating excellent functional group tolerance and enabling late-stage incorporation of fluorinated aromatic motifs.
 | ||
| Scheme 18 Fluorination via click chemistry process.65 | ||
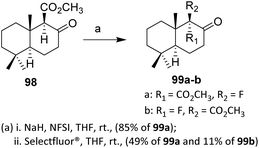
Hydrogen/fluorine interchange at the C-9 position of the decalone system was effected by electrophilic fluorination of the sodium enolate of β-keto ester 98, generated by treatment of 98 with sodium hydride in THF. The efficiency and stereoselectivity of this electrophilic fluorination depend on the nature of the fluorinating agent. The best results were achieved with N-fluorobenzenesulfonimide (NFSI), which stereoselectively afforded fluorodecalone 99 in 85% yield, likely due to its moderate electrophilicity and controlled fluorine transfer, which favor attack on the less hindered enolate face. This outcome implies the involvement of a well-organized transition state, dictated by the steric and conformational constraints of the decalone framework.
In contrast, fluorination with Selectfluor results in diminished stereocontrol, affording a mixture of the 9-epimeric fluorodecalones 99a and 99b in a combined yield of 65% after chromatographic separation (Scheme 19). The higher electrophilicity and less discriminating fluorine delivery associated with Selectfluor likely favor faster, less ordered reaction pathways, thereby reducing facial selectivity during enolate fluorination. This comparison clearly illustrates that reagent choice plays a decisive role in governing diastereoselectivity in electrophilic fluorination reactions.67,68
 | ||
| Scheme 19 Electrophilic fluorination of decalone.67,68 | ||
Vanadium(III) oxide catalyzes the direct fluorination of C(sp3)–H groups using Selectfluor. This reaction is operationally simple, and both the catalyst and reaction by-products can be easily removed by filtration. In 2014, Xia et al. reported that vanadium(III) oxide catalyzed the fluorination of the sesquiterpenoid sclareolide 100 with improved efficiency and selectivity compared to the manganese–porphyrin catalyst system. The C-2 fluoride 101 (α: β = 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was obtained in 61% yield, along with the C-3 α-fluoride in 15% yield (C-2: C-3 = 4
1) was obtained in 61% yield, along with the C-3 α-fluoride in 15% yield (C-2: C-3 = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), isolated as an inseparable mixture of isomers (Scheme 20).50 The concurrent formation of C-3-fluorinated products, albeit in minor amounts, indicates that regioselectivity is not absolute. This observation is consistent with a radical-type mechanism, in which hydrogen abstraction preferentially occurs at weaker or more accessible C–H bonds while still permitting competitive fluorination at adjacent positions. The isolation of the products as an inseparable mixture further underscores the inherent limitations in achieving precise site- and stereoselective control in direct C–H fluorination reactions.
1), isolated as an inseparable mixture of isomers (Scheme 20).50 The concurrent formation of C-3-fluorinated products, albeit in minor amounts, indicates that regioselectivity is not absolute. This observation is consistent with a radical-type mechanism, in which hydrogen abstraction preferentially occurs at weaker or more accessible C–H bonds while still permitting competitive fluorination at adjacent positions. The isolation of the products as an inseparable mixture further underscores the inherent limitations in achieving precise site- and stereoselective control in direct C–H fluorination reactions.
 | ||
| Scheme 20 Fluorination of sclareolide with Selectfluor.50 | ||
The bicyclic sesquiterpene (+)-valencene 102 is a constituent of the essential oils of various Citrus species, including sweet orange (Citrus sinensis).69 The bicyclic sesquiterpenoid (+)-valent 95 contains a ketone functional group in its structure, which can be used to direct aliphatic fluorination using Selectfluor, catalytic benzil, and visible light. Bume et al. reported a visible light-sensitized ketone-directed C–H fluorination method employing catalytic benzil (10 mol%), Selectfluor (as a putative atomic fluorine source),47 and cool white LEDs.48 Under these mild conditions, predictable β- or γ-fluorination can be achieved depending on the proximity of the C–H bond to the ketone.
It is noteworthy that other N–F reagents, such as NFSI and N-fluoropyridinium tetrafluoroborate, failed to generate the desired fluorinated product 103 (Scheme 21). Although NFSI can react with alkyl radicals, Selectfluor reacts at a significantly faster rate and is more likely to participate in electron-transfer processes. For example, compound 103 (fluorinated from the sesquiterpenoid valencene) was formed selectively, despite the presence of other tertiary carbon sites more distant from the ketone.49
 | ||
| Scheme 21 C–H fluorination of valencene using a benzil photocatalyst.49 | ||
Fluorination of sesquiterpenoid derivatives 104 can also be achieved using a palladium catalyst. As with other palladium-catalyzed fluorination reactions employing fluoride salts, control of regioselectivity remains a key challenge in developing a practical synthetic method.51 The addition of triethyl(trifluoromethyl)silane (TESCF3) was found to effectively overcome this issue, leading to dramatically improved regioselectivity in the palladium-catalyzed fluorination process.
This finding, together with the use of a newly developed biarylphosphine ligand, enabled the establishment of an efficient and highly regioselective protocol for the fluorination of monoterpenoid derivatives. Under these optimized conditions, the desired fluorinated product 105 was obtained in 41% yield (Scheme 22).52
 | ||
| Scheme 22 Fluorination of sesquiterpenoids derivatives 104 via a palladium-catalyzed reaction.52 | ||
Deoxofluorination and click chemistry provide complementary avenues for introducing fluorine into sesquiterpenoid derivatives. DAST-mediated deoxofluorination enables the direct replacement of hydroxyl or carbonyl functionalities with fluorine, granting access to mono- and difluorinated analogues. While carbonyl-containing substrates generally afford higher efficiency, reactions involving secondary alcohols often deliver only moderate yields, suffer from competing rearrangements, and display limited stereochemical control. In contrast, CuAAC click chemistry allows rapid and regioselective attachment of fluorinated aromatic moieties via triazole linkers, exhibiting excellent functional group tolerance and suitability for late-stage diversification. Nevertheless, this strategy appends fluorinated units externally rather than modifying the carbon framework of the sesquiterpenoid itself.
Direct electrophilic and catalytic fluorination methods further illustrate the delicate balance between reactivity and selectivity in structurally complex sesquiterpenoid systems. Electrophilic fluorination using reagents such as NFSI or Selectfluor enables direct C–F bond formation at enolate or activated C–H positions, with NFSI generally providing superior stereocontrol, whereas Selectfluor offers higher reactivity at the expense of selectivity. Metal- and photocatalyzed C–H fluorination strategies permit late-stage, site-selective modification without prior functionalization; however, they often generate regioisomeric mixtures and afford only modest yields. Palladium-catalyzed fluorination can improve regioselectivity through appropriate ligand and additive selection, yet its broader application is limited by catalyst cost, narrow substrate scope, and the need for extensive optimization. Collectively, these observations highlight that no single fluorination strategy is universally optimal; instead, method selection must be guided by the specific sesquiterpenoid scaffold, the desired mode of fluorine incorporation, and the tolerance of the system to the required reaction conditions.
2.3. Fluorinated diterpenoids
Nature produces an impressive assortment of more than 3300 known diterpenoids, many of which are specialized (secondary) metabolites of plant origin. Diterpenoids are widely distributed throughout the plant kingdom and have long been recognized for their broad spectrum of biological activities. For example, isopimaric acid 102 is a readily available and versatile tricyclic diterpenoid, abundantly found in the resin of conifer genera such as Pinus, Larix, and Picea.70The main synthetic interest in allenes lies in the formation of carbo- and heterocycles. Pd-catalyzed cross-coupling of allenes with aromatic iodides bearing a nucleophilic substituent at the ortho position (e.g., 2-iodoaniline and 2-iodophenol) allows cyclization via [α,β], [β,α], [β,γ], or [γ,β] attack on the allene, resulting in the formation of indole or benzofuran frameworks.
Experimental results by Gromova et al. demonstrated that the nature of the substituent in the fluorinated phenols significantly influences the outcome of the cross-coupling–cyclization reaction. Mechanistically, and according to the principles of palladium catalysis, the formation of compounds 108 and 109 can be described as follows (Scheme 23).
 | ||
| Scheme 23 Synthesis of furo[3,2-c]chromen-4-one derivatives via Pd-catalyzed coupling-cyclization.72 | ||
The pallacycle catalyst is reduced to a Pd(0) species under the reaction conditions (DMF–H2O, K2CO3, Bu4NBr, fluorine-substituted 2-iodophenol, allene). The Pd(0) species is generated via nucleophilic attack of the fluorine-substituted 2-iodophenol on the palladacycle, forming an arylated palladacycle that undergoes reductive elimination to yield the active Pd(0) catalyst.
Subsequently, a nucleophilic attack by the substituent at the α-position leads to the expected products 108. The formation of isomeric compounds 109 involves generation of an η3-allylpalladium intermediate, in equilibrium with the less stable σ-vinylpalladium species, followed by η1-rearrangement, β-elimination, and attack on the activated double bond. The substituent on 2-iodophenols 107a–c affects the stabilization of reaction intermediates, thereby influencing the selective formation of 108 or 109.72
Lu et al. reported the synthesis, cytotoxic activity, metabolic stability, and pharmacokinetics of a series of fluorinated docetaxel analogs. First, the hydroxyl groups at C-7 and C-10 of 10-deacetylbaccatin III (10-DAB) 109 were protected with 2,2,2-trichloroethyl chloroformate (TrocCl) using pyridine as the base, affording 7,10-diTroc-10-deacetylbaccatin 110. Compound 110 was then coupled with 111 in the presence of dicyclohexylcarbodiimide (DCC) and 4-dimethylaminopyridine (DMAP) to give intermediate 112 in 86% yield. The high yield obtained for intermediate 112 indicates that carbodiimide activation remains effective even in complex diterpenoid systems, provided that reactive hydroxyl groups are appropriately masked.
After removal of the 30-N-boc and acetonide protecting groups from 112 using 98% formic acid at room temperature, the amino alcohol 113 was obtained in 55% yield. Subsequent acylation of the amino group of 113 with freshly prepared fluorine-containing acyl chlorides 114a–c, followed by removal of the 7,10-troc protecting groups with zinc in acetic acid, furnished the corresponding fluorinated docetaxel derivatives 116a–c in desirable yields (Scheme 24).25 Despite its effectiveness, this approach is inherently multistep and relies heavily on protecting-group manipulations, which may limit scalability and overall synthetic efficiency. Moreover, fluorine is introduced indirectly through side-chain modification rather than by direct fluorination of the taxane core, thereby restricting structural diversification of the diterpenoid backbone.
 | ||
| Scheme 24 Fluorination via acylation of the amino group.25 | ||
In 2019, Lapuh et al. reported the synthesis of a fluorine-containing abietane derivative via a copper(0)-catalyzed process. The use of a Cu(0) catalyst enables efficient installation of a difluoroacetate fragment onto an iodoarene precursor, providing a practical entry point for the incorporation of fluorinated carbon units into aromatic terpene-derived systems. Starting from iodoarene 117, the ethyl difluoroacetate moiety was efficiently introduced through a copper(0)-mediated coupling in DMSO. The choice of DMSO as coordinating polar aprotic solvent likely facilitates copper-mediated oxidative addition and stabilizes reactive intermediates, contributing to the efficiency of the C–C bond-forming step. Subsequent selective saponification of the ethyl ester afforded carboxylic acid 118 in 81% yield. This step underscores the stability of the CF2 group under basic hydrolytic conditions and enables further functional manipulation. A three-step sequence including a Barton decarboxylation then produced the difluoromethyl derivative 119 (Scheme 25).75
 | ||
| Scheme 25 Synthesis a fluorine-containing abietane derivative via copper(0) catalysis.75 | ||
The fluorination of hydroxykauranes 120 likely begins with the formation of an intermediate hypofluorite, which acts as an electrophilic fluorinating reagent. In the case of the 15α-epimers 120a and 120b, the fluorine atom of the hypofluorite can access the equatorial H-7α, abstracting a hydrogen atom to form HF (captured by an amine) and oxidizing C-15 to generate an intermediate ketone in an enthalpy-favored process. This intermediate then reacts with a complex amine·HF (similar to commercial TREAT HF) from the less hindered side (opposite H-7α) to afford the fluoroderivatives 121a (10%) and 121b (12%) (Scheme 26).76 Nucleophilic attack occurs preferentially from the less hindered face of the molecule, opposite to H-7α, leading to the observed fluoroderivatives. This sequence illustrates that stereochemical outcome is dictated primarily by substrate topology rather than by reagent control.
 | ||
| Scheme 26 Fluorination of hydroxykauranes using Selectfluor.76 | ||
This step demonstrates that the diterpenoid framework tolerates strong oxidative conditions without degradation, thereby enabling a streamlined synthesis without intermediate purification. The target compounds 123a–e and 125a–e were obtained by treating 122 or 124 with the corresponding acid in the presence of 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDCI) and 4-dimethylaminopyridine (DMAP) in dichloromethane (DCM) at room temperature for 8–12 hours. Under these conditions, the 14-OH group was regioselectively esterified, reflecting its higher nucleophilicity and steric accessibility relative to other potential reactive sites on the oridonin skeleton. This selectivity enables predictable late-stage functionalization of a complex natural product (Scheme 27).78
 | ||
| Scheme 27 Fluorination of oridonin via an acylation method.78 | ||
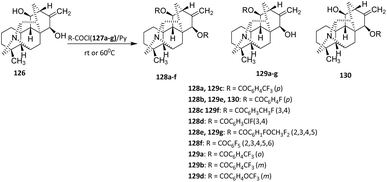
In another study, Bessonova et al. (2000) reported the fluorination of Kobusine via the acylation method. Kobusine 126 is a hetisine-type C20-diterpenoid alkaloid and a major constituent of Aconitum yesoense var. Macroyesoense.79
Kobusine 126 was reacted with various acyl chlorides (127a–g) in pyridine to afford C-11, C-15, or C-11,15-substituted acyl derivatives (128a–f, 129a–g, and 130) (Scheme 28).80,81 The formation of C-11, C-15, or C-11,15-disubstituted products reflects differences in the relative nucleophilicity and steric accessibility of the hydroxyl groups present in kobusine. Competition between these sites results in product distributions that are predominantly substrate-controlled rather than catalyst-controlled. While the ability to access multiple substitution patterns indicates a degree of substrate flexibility, it also underscores the limited inherent regioselectivity of the transformation under the selected conditions. This transformation proceeds via nucleophilic attack of the hydroxyl group on the carbonyl carbon of the acid chloride. Pyridine acts both as a base to neutralize the released HCl and as a catalyst to facilitate ester bond formation.
 | ||
| Scheme 28 Fluorination of Kobusine via acylation method.80,81 | ||
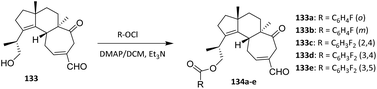
Cao et al. also performed fluorination of the diterpenoid sarcodonin G 133 using the acylation method. Sarcodonin G 133 was obtained quantitatively from the fruit bodies of S. scabrosus. A series of 19-O-benzoyl derivatives was prepared as shown in Scheme 29. Briefly, 133 was treated with dry Ac2O or the respective benzoyl chloride derivatives in the presence of 4-dimethylaminopyridine (DMAP) at room temperature. Use of DMAP as a nucleophilic acyl transfer catalyst facilitates efficient ester bond formation under mild conditions, minimizing side reactions and preserving the integrity of the polycyclic framework. Workup of the resulting residue provided new compounds (134a–e) as yellowish oils with purity >99.0% by HPLC and in good to excellent yields (Scheme 29).82 The acylation reaction proceeds with high regioselectivity at the C-19 hydroxyl group, reflecting its enhanced nucleophilicity and steric accessibility relative to other potential reactive sites on the diterpenoid scaffold.
 | ||
| Scheme 29 Fluorination of sarcodonin G 133.82 | ||
 | ||
| Scheme 30 Deoxofluorination of indaconitine 131.84 | ||
Reactivity and selectivity in DAST-mediated deoxofluorination are governed by a combination of steric and electronic factors. More accessible hydroxyl groups and those capable of forming stable alkoxysulfur intermediates are preferentially fluorinated, while sterically hindered or intramolecularly hydrogen-bonded OH groups are less reactive. In indaconitine, the conformational exposure of the 3-OH in the A-ring likely facilitates its activation by DAST, whereas other hydroxyl groups remain unreacted under the same conditions.
 | ||
| Scheme 31 Fluorination of diterpenoid (135) via amide reaction.85 | ||
Diterpenoid 135 was reacted in one-pot with 2-fluoroaniline 136a, 3-fluoroaniline 136b, and 4-fluoroaniline 136c in absolute ethanol under catalyst-free conditions at room temperature to yield three new fluorinated diterpenoids 137a–c (Scheme 31). The reaction proceeds without external activation, underscoring the intrinsic reactivity of the diterpenoid scaffold and its suitability for mild, late-stage functionalization.
From a selectivity and substrate-scope perspective, the method tolerates different positional isomers of fluoroaniline, indicating that the electronic effects of the fluorine substituent exert only a limited influence on the overall reaction outcome. However, the longer reaction times observed with fluoroanilines compared to non-fluorinated anilines suggest that the electron-withdrawing fluorine atom reduces aniline nucleophilicity, thereby slowing the transformation.
Reactivity is governed primarily by intramolecular cyclization: in situ generation of a reactive intermediate enables nucleophilic attack by the hydroxyl group of the acid moiety, leading to γ-lactone formation. This intramolecular step confers high regioselectivity, as lactonization is dictated by spatial proximity and conformational rigidity of the diterpenoid framework rather than by external catalysts or additives.85
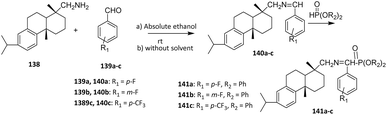
In another study, Rao et al. (2008) reported the fluorination of diterpenoid dehydroabietylamine 138 via two-step reactions through imine intermediates 140a–c to yield novel α-aminophosphonates 141a–c. Despite the high steric hindrance of the tricyclic structure, dehydroabietylamine 138 and substituted benzaldehydes 139a–c afforded imines in good yields without catalysts. The subsequent addition of phosphites to the imines was challenging due to steric hindrance; benzyl phosphonates gave relatively low yields of 20–30%, whereas ethyl phosphonates provided higher yields owing to less steric congestion (Scheme 32).86
 | ||
| Scheme 32 Fluorination of diterpenoid dehydroabietylamine 138.86 | ||
Acylation strategies, including the acylation of amino or hydroxyl functionalities, represent some of the most reliable and widely applicable methods for introducing fluorinated moieties into diterpenoids such as docetaxel, oridonin, kobusine, and sarcodonin G. These transformations generally proceed under mild conditions with high regioselectivity dictated by the intrinsic nucleophilicity and steric accessibility of specific functional groups. Their advantages include broad functional group tolerance, reproducibility, and suitability for late-stage modification of bioactive compounds. In these cases, however, fluorine is incorporated indirectly via fluorinated acyl substituents rather than through direct C–F bond formation on the diterpenoid core. In multistep systems such as taxanes, extensive protecting-group manipulations further reduce synthetic efficiency and constrain the rapid generation of analogues.
Direct fluorination approaches, including electrophilic fluorination and DAST-mediated deoxofluorination, enable the direct installation of C–F bonds, thereby exerting a more pronounced influence on electronic properties, metabolic stability, and biological activity. Electrophilic fluorination of hydroxykauranes illustrates that stereochemical outcomes are largely governed by substrate control, dictated by molecular topology rather than reagent identity, although yields are often modest. DAST-mediated deoxofluorination constitutes a valuable late-stage strategy for highly functionalized diterpenoids, allowing selective fluorination of accessible hydroxyl groups. Nevertheless, such reactions may suffer from limited regioselectivity, competing side reactions, and sensitivity to steric and conformational constraints. Complementary amide-based transformations further expand structural diversity but are often limited by the reduced nucleophilicity of fluoroanilines. Overall, no single fluorination strategy is universally optimal; instead, the choice of method must balance structural complexity, desired fluorine placement, reaction efficiency, and compatibility with the diterpenoid framework.
2.4. Fluorinated triterpenoids
Triterpenoids are one of the largest classes of natural products, with over 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 known members. They are biosynthesized in plants via cyclization of squalene. Over the last decades, pentacyclic triterpenoids have shown a unique range of pharmacological activities, including chemopreventive and antineoplastic potential.87–91
000 known members. They are biosynthesized in plants via cyclization of squalene. Over the last decades, pentacyclic triterpenoids have shown a unique range of pharmacological activities, including chemopreventive and antineoplastic potential.87–91
Asiatic acid (AA, 142), a member of the ursane family extracted mainly from Centella asiatica, exhibits hepatoprotective, neuroprotective, antidiabetic, antihyperlipidemic, anti-inflammatory, antioxidant, and anti-Alzheimer activities. Goncalves et al. (2016) used Selectfluor to introduce a fluorine atom into AA to increase its anticancer activity. Treatment of 142 with Selectfluor in a mixture of nitromethane and dioxane at 80 °C afforded the 12β-fluorolactone derivative 143 in 61% yield (Scheme 33). This transformation highlights the high regio- and stereoselectivity of Selectfluor, as fluorination occurs exclusively at the activated C-12 position to give the 12β-fluorolactone, reflecting strong substrate control imposed by the lactone framework and neighboring functional groups. While the method exhibits good efficiency for this specific diterpenoid scaffold, its substrate scope appears limited, as elevated temperatures (80 °C) are required and overoxidation or competing electrophilic reactions may occur in more densely functionalized or sensitive analogues.
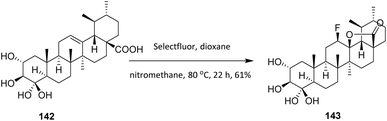 | ||
| Scheme 33 Fluorination of asiatic acid (142) using Selectfluor.92 | ||
In a different study, Leal et al. (2018) developed fluorinated ursolic acid 144 derivatives in an effort to further improve their antitumor properties activity. Fluorine was introduced into the C ring of ursolic acid 145. Ursolic acid 144 is a pentacyclic triterpenoid present in plants, vegetables and fruits, which has been found to have many biological properties, such as anti-inflammatory, antidiabetic, cardioprotective, hepactoprotective, antitumoral and chemopreventive activities.93 The fluorination of ursane-type triterpenoids, when performed in a reaction solution without a nucleophilic donor, allows the synthesis of a fluorolactone. The free acid behaves like the nucleophilic donor, losing the proton and promoting cyclization to the carbon C13 in the ursane backbone. This reaction is not possible in ursane-type compounds where the acid is protected, however, as the free acid alone does not react with Selectfluor. Compound 144 was reacted with Selectfluor at 80 °C in a mixture nitromethane and dioxane, produced 145 with the insertion of β-fluorine at C12 with a yield above 88% (Scheme 34).94
 | ||
| Scheme 34 Fluorination of ursolic acid (144) using Selectfluor.94 | ||
In a separate study, compound 142, a pentacyclic triterpenoid 18-β-glycyrrhizinate with an enone on the C-ring, was fluorinated by Pitts et al. (2017). Fluorination at the C1 position afforded the product 143 in good yield (up to 72%).95,96 This selectivity is governed by the intrinsic polarization of the α,β-unsaturated carbonyl system, which directs electrophilic fluorine toward the more electron-rich and sterically accessible site. In addition to enones targeting the γ position, Bume et al. (2018) performed enone fluorination at the β position. Compound 144, containing an enone in ring D, was fluorinated to give 145 in 70% yield (Scheme 35), highlighting that the reaction outcome is sensitive to both the enone position and the ring topology within the pentacyclic scaffold.97,98 The reactivity and selectivity of these transformations are governed by conjugation effects within the enone system, steric shielding imposed by the rigid triterpenoid framework, and the strong electrophilicity of Selectfluor. Collectively, these factors favor controlled monofluorination while suppressing over-fluorination and competing side reactions, even in highly functionalized substrates.
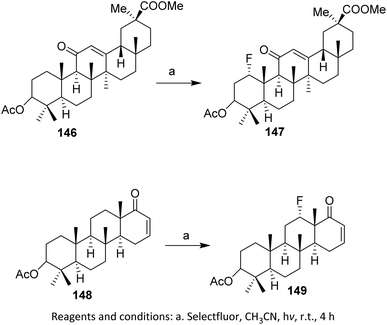 | ||
| Scheme 35 Photoinduced C–H fluorination with Selectfluor.97,98 | ||
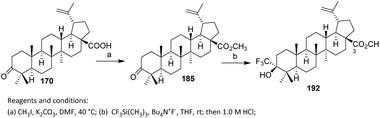
Li et al. introduced fluorine atoms directly at the C-2 side chain of 185 as shown in Scheme 36. Methyl ester 185 was prepared from betulonic acid 170 following reported methods. Fluorination was achieved by treating 185 with lithium diisopropylamide (LDA) and N-fluorobenzenesulfonimide (NFSI) at −78 °C, resulting in the addition of one fluorine atom at C-2 to afford 186 in 43% yield via an electrophilic substitution reaction.99,100 The selection of NFSI as a mild and selective electrophilic fluorine source is critical, as it promotes single fluorine transfer while suppressing over-fluorination and oxidative side reactions. The use of low temperature further stabilizes the enolate and minimizes enolate equilibration, thereby contributing to the observed site selectivity. With respect to substrate scope, this strategy is best suited to triterpenoids bearing acidic α-protons adjacent to carbonyl functionalities, such as esters or ketones, and thus suggests potential applicability to other betulonic acid derivatives. However, substrates lacking enolizable positions or containing base-sensitive functional groups may be incompatible.
 | ||
| Scheme 36 Electrophilic fluorination of betulinic acid derivatives.99,100 | ||
Trifluoromethyl hypofluorite (CF3OF) was applied to methyl olean-2,12(13)-dien-ll-on-30-oat-3β-yl acetate 187. Under sufficiently mild conditions, only the enol acetate system reacts, while the double bond at the 12(13) position remains unaffected. This selectivity reflects the strong peference of CF3OF for activated enol systems adjacent to carbonyl groups, where electrophilic fluorine transfer is kinetically favored. Scheme 37 shown an electrophilic fluorination using trifluoromethyl hypofluorite (CF3OF) as the fluorinating agent. In this process, CF3OF functions as a highly potent electrophilic fluorinating agent, delivering fluorine to the α-position of the carbonyl via an enol or enol acetate intermediate. Subsequent base treatment is essential, as it promotes elimination and rearrangement to afford the thermodynamically more stable methyl 2α-fluoroolean-12-ene-3,11-dion-30-oate 188, thereby completing the functional group interconversion in a controlled manner.101 The limitations of this methodology include the reliance on CF3OF, a highly reactive and potentially hazardous reagent that necessitates careful handling and strictly controlled reaction conditions.
 | ||
| Scheme 37 Fluorination of steroidal compound using CF3OF reagent.101 | ||
Nitrosyl fluoride (NOF) generally adds to olefinic bonds to give fluoro-nitrimino compounds, which decompose on alumina to fluoroketones.102 However, the reaction of NOF with dienes has not been widely reported. An excess of NOF with methyl 3-O-acetyl-9,11-dehydro-11-deoxo-glycyrrhetate 189 in methylene chloride (10 days at 5 °C) gave a mixture, which upon separation by thin-layer chromatography (TLC) afforded 190 (10%) and 191 (25%) (Scheme 38).103 Unlike its more common reactions with isolated olefins, NOF engages the diene only under prolonged, low-temperature conditions, indicating low intrinsic reactivity that necessitates the use of excess reagent and extended reaction times. From a selectivity and mechanistic perspective, the formation of two separable products (190 and 191) points to competing regio- and/or stereochemical pathways during electrophilic addition across the conjugated system. The modest yields further suggest that multiple addition modes or parallel decomposition pathways operate concurrently, thereby diminishing overall efficiency and reaction control.
 | ||
| Scheme 38 Simultaneous nitration–fluorination of a steroidal derivative.103 | ||
 | ||
| Scheme 39 Fluorinateion of triterpenoid via deoxofluorination reaction.24,104 | ||
The substantial drop in yield in the absence of pyridine (40%) highlights the importance of additive control in optimizing reaction efficiency. From a reactivity standpoint, pyridine plays a crucial role by scavenging HF generated in situ and stabilizing reactive sulfur intermediates, thereby suppressing side reactions such as elimination or rearrangement. This additive effect is particularly important in rigid terpenoid frameworks, where neighboring functional groups and steric congestion might otherwise divert the reaction pathway. The same procedure was applied to convert hydroxyketone 152 to 2,2-difluoroallobetulone 153.24
In 2002, Slavikova et al. reacted hydroxy ketones 154 with DAST to form a single difluoroketone product 155. Optimization of the reaction conditions afforded 38% yield. Enol ketone 156 and alcohol 158 were also used as substrates, providing the desired fluoro derivatives 157 and 159 in 77–80% yield.105
Utilization of DAST for converting ketones and aldehydes into geminal difluoro derivatives requires more forcing conditions than alcohol substitution, often resulting in lower yields. For example, aldehyde 160 gave difluoro derivative 161 in only 22% yield after 48 hours, while heptanorketone 162 yielded difluoro derivative 163 in 30%.
DAST is also effective for the synthesis of acyl fluorides under mild conditions. Acetylpreculinic acid 164 was fluorinated to acyl fluoride 165. Biederman et al. successfully applied the same procedure to saturated derivatives 166 and 21-oxo acid 168, obtaining the respective acyl fluorides 167 and 169 (Scheme 40).104
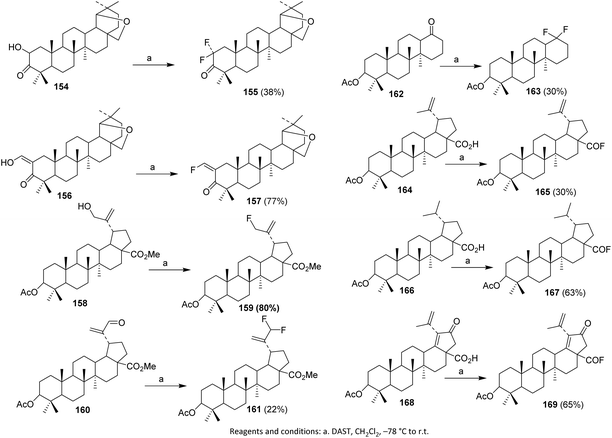 | ||
| Scheme 40 Fluorination of triterpenoids via deoxofluorination reaction.104,105 | ||
 | ||
| Scheme 41 Synthesis of fluorinated triterpenoids via amine acylation reaction.106 | ||
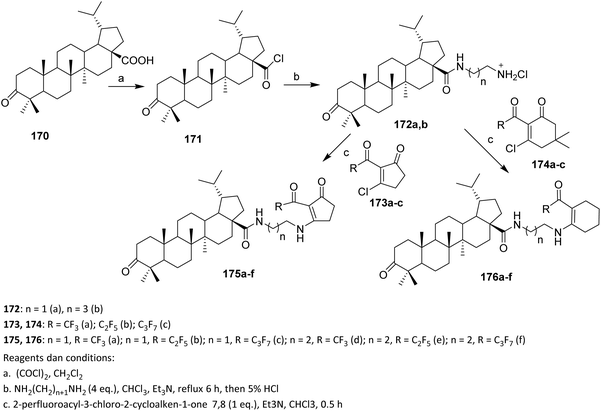
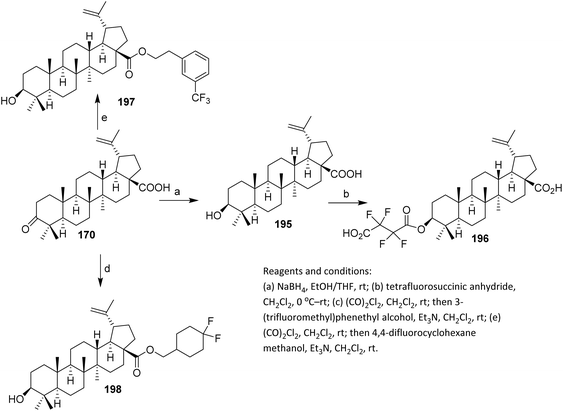
The efforts to obtain new anti-inflammatory agents were focused on structural modifications at C-3 and C-28 positions of betulonic acid 170. Scheme 42 shows the methodology used to produce the investigated fluorine-containing amides 179a–d and hybrids 183a–e. To synthesize the fluorinated amide derivatives of betulonic acid, an acylation of the corresponding fluoroaromatic amines with betulonic acid chloride 177, obtained as per literature procedure, was carried out. Compound 177 was allowed to react with the fluorine-containing aromatic amines in dry benzene in the presence of pyridine to produce betulonic acid amides 179a–d in 66–72% yields.
 | ||
| Scheme 42 Synthesis of fluorinated amide derivatives of betulinic acid.107 | ||
For obtaining the hybrids 183a–e, a reaction of a vinylogous substitution of cyclic β-triketone enol derivatives by N-nucleophiles was used. Betulonic acid 170 was methylated with diazomethane to give the methyl ester 180. Then oxime 180 was prepared by treatment of this intermediate with hydroxylamine hydrochloride in pyridine. Obtained oxime 180 was reduced under an action of NaBH4 in the presence of NiCl2 in dry ethanol to give methyl 3-amino-3-deoxybetulinate 181 as a mixture of 3β- and 3α-isomers (65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 35 according to 1H NMR data). A vinylogous substitution of 2-acyl-3-chlorocyclopent-2-en-1-ones, obtained from corresponding cyclic β-triketones, with amine 182 was carried out in chloroform at room temperature to give hybrids 183a–e.107
35 according to 1H NMR data). A vinylogous substitution of 2-acyl-3-chlorocyclopent-2-en-1-ones, obtained from corresponding cyclic β-triketones, with amine 182 was carried out in chloroform at room temperature to give hybrids 183a–e.107
The betulonic acid 170 was transformed into the corresponding chloride 177 according to the previously reported procedure and immediately coupled to the selected oxime. With both key components in hand, a one-pot procedure led to the target conjugates 184a–b. In a typical experiment, a mixture consisting of a selected 6,7-dihydro-1H-indazol-4(5H)-one oxime, betulonic acid chloride 177, and pyridine in CHCl3 was refluxed for 5 h. The acylated products 184a–b were obtained in moderate to good yields (Scheme 43).108 From a reactivity and mechanism perspective, the success of the one-pot protocol relies on the high electrophilicity of the acid chloride and the sufficient nucleophilicity of the oxime oxygen. Pyridine plays a dual role as an HCl scavenger and as a mild base, thereby suppressing acid-promoted decomposition of the oxime and minimizing side reactions such as hydrolysis or rearrangement under reflux conditions.
 | ||
| Scheme 43 Synthesis of fluorinated heterocyclic derivatives of betulinic acid.108 | ||
 | ||
| Scheme 44 Trifluoromethylation of a betulinic acid derivative using (CF3)3Si and TBAF.99,100 | ||
In step (b), 185 is treated with CF3Si(CH3)3 (Ruppert–Prakash reagent) and Bu4N+F− in THF at room temperature, followed by acidification with HCl, to introduce a trifluoromethyl group at the C-3 position. This nucleophilic trifluoromethylation produces the final compound 192, functionalized with both a methyl ester at C-28 and a CF3 substituent at C-3, enhancing its chemical and biological properties. The mild reaction conditions and controlled acidic quench favor clean trifluoromethyl addition over competing reduction or elimination pathways, underscoring effective chemoselectivity in a densely functionalized triterpenoid scaffold.
In terms of substrate scope, this methodology is best suited to ketone-containing triterpenoids analogous to betulonic acid, particularly those featuring an accessible and relatively unhindered carbonyl group. The approach therefore suggests broader applicability to other lupane- or oleanane-type scaffolds, provided that the carbonyl functionality is sufficiently reactive toward nucleophilic trifluoromethylation.99,100
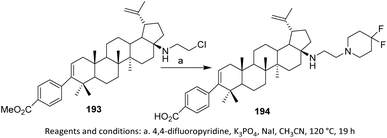
Piperidine derivative 193 was obtained by replacement of the terminal chloride with an amine, in which the amine acts as a strong nucleophile to displace the chloride under controlled conditions. This transformation demonstrates high chemoselectivity, as substitution occurs preferentially at the activated terminal position without affecting other functional groups within the molecule.
Subsequent deprotection of the benzoate ester furnishes compound 194, restoring the free hydroxyl group and increasing the polarity of the final product. The stepwise sequence allows independent control over C–N bond formation and protecting-group removal, which is advantageous when working with multifunctional terpenoid or alkyl halide frameworks (Scheme 45).109
 | ||
| Scheme 45 Formation of a 4,4-difluoropiperidine-linked steroidal derivative.109 | ||
 | ||
| Scheme 46 Synthesis of fluorinated ester and ether derivatives of betulinic acid.99,100 | ||
 | ||
| Scheme 47 Fluorination of a betulinic acid derivative via Sonogashira coupling reaction.3 | ||
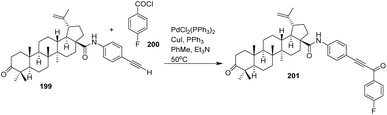
From a reactivity and catalytic standpoint, the PdCl2(PPh3)2/CuI system efficiently mediates oxidative addition of the aryl chloride 200, transmetalation with the copper acetylide, and subsequent reductive elimination to form the Csp–Csp2 bond. Triethylamine serves both as a base and as a scavenger for HX, while toluene at moderate temperature (50 °C) balances catalytic activity with suppression of alkyne homocoupling or decomposition. The catalytic system enables the formation of a new Csp–Csp2 bond between the alkyne and aryl chloride, leading to the integration of a fluorinated aromatic group onto the BA structure and producing 201.
In terms of substrate scope, this approach is well suited for BA derivatives bearing terminal alkynes and for aryl halides containing fluorinated substituents, allowing modular introduction of diverse fluorinated aromatic motifs. The strategy is potentially extensible to other triterpenoid scaffolds, provided that the alkyne functionality is sterically accessible and compatible with palladium catalysis.3
 | ||
| Scheme 48 Fluorination of a triterpenoid via an oxidation–reduction reaction.2 | ||
From a reactivity control perspective, the use of sodium hydride (NaH) in tetrahydrofuran (THF) promotes efficient enolate formation at C-3, enabling controlled C–C bond formation with a range of aromatic aldehydes. The subsequent sodium borohydride (NaBH4) reduction selectively reduces the C-3 carbonyl in 204a–c to regenerate the hydroxyl group in 205a–c, while preserving the newly installed benzylidene moiety, demonstrating good functional group tolerance and predictable chemoselectivity.2
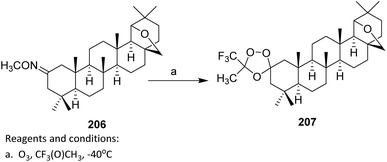 | ||
| Scheme 49 Oxidative ozonolysis of a steroidal alkene to a trifluoroacetate derivative.110,111 | ||
From a reactivity and mechanistic perspective, the use of fluorinated ketones such as CF3C(O)CH3 is crucial. The strongly electron-withdrawing CF3 group markedly enhances the dipolarophilicity of the carbonyl moiety, thereby promoting rapid and selective [3 + 2] cycloaddition with the carbonyl oxide intermediate. This electronic activation offsets steric constraints imposed by the bulky terpenoid framework and directs efficient spirocyclization.
With respect to substrate scope, the successful conversion of isomeric mixtures of 205 indicates a notable tolerance toward olefin geometry and substitution patterns. Consequently, this strategy is particularly attractive for late-stage diversification of complex terpenoid scaffolds, especially when fluorinated dipolarophiles are employed to drive cycloaddition efficiency.111
Deoxofluorination and nucleophilic fluorination approaches provide complementary routes to mono- and gem-difluorinated triterpenoids, enabling substantial electronic perturbation of the core scaffold. DAST-mediated deoxofluorination allows late-stage substitution of hydroxyl or carbonyl functionalities, even in structurally complex systems; however, yields are strongly dependent on steric effects, reaction conditions, and the nature of the functional group (alcohols versus carbonyls). Nucleophilic trifluoromethylation using the Ruppert–Prakash reagent enables site-selective installation of CF3 groups at carbonyl positions under relatively mild conditions, offering high chemoselectivity and expanded physicochemical diversity. Despite these advantages, such methods are largely confined to substrates bearing reactive carbonyl groups and may suffer from reduced efficiency or competing side reactions in more elaborate molecular settings.
Indirect fluorination strategies—including esterification, amide formation, cross-coupling reactions (e.g., Sonogashira coupling), oxidation–reduction sequences, and ozonolysis—constitute some of the most versatile and reliable approaches for generating libraries of fluorinated triterpenoids. These methods generally exhibit excellent functional group tolerance, predictable regioselectivity, and compatibility with late-stage diversification, enabling the modular incorporation of fluorinated aromatic, heterocyclic, or aliphatic fragments. Their principal limitation lies in the fact that fluorine is introduced as a peripheral substituent rather than being directly embedded within the triterpenoid carbon framework, thereby exerting a more modest influence on the intrinsic electronic properties of the core. Consequently, no single fluorination strategy is universally optimal for triterpenoids; instead, the selection of an appropriate method depends on the targeted position, the desired fluorine motif (F, CF2, or CF3), scaffold sensitivity, and the balance between synthetic efficiency and structural impact.
2.5. Fluorinated tetranorterpenoids (limonoids)
Limonoids, a group of highly oxygenated triterpenoids, mainly exist in the Rutaceae and Meliaceae plant families. Tetranortriterpenoids is an alternative name for limonoids because, during oxidative changes of triterpenoids, the side chain is eventually oxidized to an α-substituted furyl ring by the loss of four carbon atoms. Limonoids exhibit a wide spectrum of biological properties, including cytotoxic, antioxidant, anti-inflammatory, neuroprotective, antiviral, antimicrobial, antiprotozoal, antimalarial, insect antifeedant, and insecticidal activities.112 | ||
| Scheme 50 Fluorination of limonoid using DAST.114 | ||
From a reactivity standpoint, the requirement for a large excess of DAST (10 equiv.) and an extended reaction time (24 h) reflects both steric hindrance at the secondary alcohol and the presence of competing reaction pathways. The formation of dehydrated by-product 210 in 10% yield indicates partial elimination, likely promoted by in situ – generated HF, underscoring the delicate balance between substitution and elimination in DAST-based transformations.114
 | ||
| Scheme 51 Fluorination of limonoid via esterification using DCC/DMAP.116 | ||
The sequence shown in Scheme 52 illustrates a regioselective, stepwise functionalization strategy for modifying the fraxinellone scaffold. The initial chromium trioxide–mediated allylic oxidation under microwave irradiation selectively targets the allylic position of 213 to afford fraxinellone 214, demonstrating effective site selectivity despite the presence of multiple oxidizable sites within the limonoid framework. From a reactivity control perspective, microwave irradiation accelerates the oxidation process and enhances regioselectivity by promoting rapid and uniform heating. Subsequent NaBH4 reduction cleanly converts the carbonyl group of 214 into alcohol 215 without affecting other sensitive functionalities. This reduction step reintroduces a reactive hydroxyl handle, thereby enabling further derivatization.
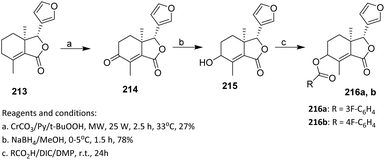 | ||
| Scheme 52 Fluorination of limonoid via esterification using DIC/DMAP.117 | ||
The substrate scope of the final esterification step is governed by the accessibility of the hydroxyl group in 215 and the nature of the carboxylic acids employed. The use of DIC/DMAP enables efficient coupling with different acids to afford esters 216a and 216b, indicating reasonable tolerance toward varied acyl substituents and suitability for structure–activity relationship (SAR)-driven diversification.
However, this approach is limited by the relatively low yield of the initial oxidation step (27%) and the reliance on chromium-based oxidants, which raise concerns regarding environmental impact and functional group compatibility.117
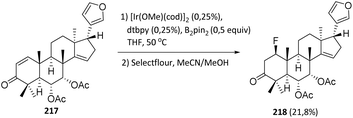 | ||
| Scheme 53 One-pot, two-step borylation/fluorination reaction of dysobinin.118 | ||
3. Conclusion
Fluorinated terpenoids exhibit diverse and significant bioactivities, underscoring their growing importance in medicinal and synthetic chemistry. In the past, the fluorination of terpenoids often resulted in product mixtures with low conversions and poor overall yields. Moreover, the use of fluorinating reagents required extensive precautions during synthesis, and some reagents such as perchloryl fluoride are considered highly hazardous. These factors have limited the practical utilization of fluoro-substituted terpenoids.However, the development and commercialization of new electrophilic and nucleophilic fluorinating agents, along with the emergence of metal-catalyzed fluorination reactions, have eliminated the need for direct handling of elemental fluorine in pharmaceutical laboratories. These advances have opened efficient pathways for the selective introduction of fluorine atoms into terpenoid frameworks with high yields. Among the most widely used reagents are DAST, trifluoromethyl hypofluorite, Selectfluor, and NFSI, which are readily available for electrophilic fluorination. Meanwhile, nucleophilic fluorination agents are tetrabutylammonium fluoride hydrate (TBAF·3H2O), triethyl(trifluoromethyl)silane (TESCF3), and Ruppert—Prakash reagent (TMSCF3—TBAT/TMAF). In addition, fluorination can also be achieved through the incorporation of aryl groups containing fluorine atoms. This approach encompasses various reaction types, including esterification, alcohol reduction, michael addition, oxidation–reduction, C–C cross-coupling (e.g., Heck coupling and Sonogashira coupling), click chemistry, ozonolysis of an alkene and N-acylation reactions (Table 1).
| Strategy type | Monoterpenoids | Sesquiterpenoids | Diterpenoids | Triterpenoids | Tetranortriterpenoids |
|---|---|---|---|---|---|
| Functional group transformation–driven approaches | Esterification; Prins-type reactions; acid-catalyzed transformations | Michael addition; acylation reactions; functional group interconversions | Esterification; oxidation–reduction; ozonolysis of alkenes | Esterification and multistep functional modification | Reduction of hydroxy limonoids; functional group manipulation |
| Nucleophilic fluorination/deoxofluorination | Alcohol-based deoxofluorination; site-selective C–F installation | Limited application due to skeletal complexity | Deoxofluorination reactions; nucleophilic trifluoromethylation | Applicable but requires pre-functionalized substrates | Deoxofluorination of hydroxylated limonoids; late-stage modification |
| Electrophilic fluorination | Applicable but limited regioselectivity | Electrophilic fluorination of activated sites | Direct electrophilic fluorination with good functional-group tolerance | Sterically hindered; limited direct application | Electrophilic fluorination following borylation |
| Metal-catalyzed & cross-coupling strategies | Pd- and Cu-catalyzed transformations; click reactions | Pd-catalyzed cross-coupling reactions | Pd/Cu-catalyzed cross-coupling; alkynylation | Limited due to steric congestion | Borylation–fluorination sequences enabling scaffold diversification |
| Overall synthetic features | Simple skeletons favor acid-catalyzed and functional-group-based strategies | Moderate complexity allows multiple fluorination pathways | Larger frameworks tolerate diverse and selective fluorination methods | Structural rigidity limits direct fluorination | Highly functionalized scaffolds favor controlled, multistep strategies |
Despite these advances, several challenges remain and will likely define future research directions in this field. Achieving site-selective and stereoselective fluorination on complex terpenoid scaffolds continues to be difficult, particularly for late-stage functionalization. Furthermore, many reported transformations rely on stoichiometric reagents or multistep sequences, highlighting the need for more sustainable, catalytic, and environmentally benign fluorination strategies. From a biological perspective, systematic structure–activity relationship studies are still limited, and deeper insight into how fluorine incorporation influences target selectivity, metabolic stability, and toxicity is required. Addressing these challenges will be essential for translating fluorinated terpenoids from synthetic curiosities into practical candidates for medicinal, agrochemical, and materials applications.
Conflicts of interest
There are no conflicts to declare.Data availability
No new primary research data, software, or code were generated or analysed in the preparation of this review.Acknowledgements
Y.P.B. acknowledges financial support from LPDP through the Equity–WCU Kemdiktisaintek Program (Grant No. 3871/UN6.3.1/PT.00/2025). This publication charge is funded by Unpad through the Indonesian Endowment Fund for Education (LPDP) on behalf of the Indonesian Ministry of Higher Education, Science and Technology and managed under the EQUITY Program (Contract No. 4303/B3/DT.03.08/2025 and 3927/UN6.RKT/HK.07.00/2025).References
- D. W. Christianson, Chem. Rev., 2017, 117, 11570–11648 CrossRef CAS PubMed.
- L. Ma, X. Wang, W. Li, D. Miao, Y. Li, J. Lu and Y. Zhao, Eur. J. Med. Chem., 2020, 187, 111964 Search PubMed.
- M. D. Semenova, S. A. Popov, I. V. Sorokina, Y. V. Meshkova, D. S. Baev, T. G. Tolstikova and E. E. Shults, Steroids, 2022, 184, 109042 CrossRef CAS PubMed.
- A. Abouabdellah, J. P. Bégué, D. Bonnet-Delpon, J. C. Gantier, T. T. T. Nga and T. D. Thac, Bioorg. Med. Chem. Lett., 1996, 6, 2717–2720 CrossRef CAS.
- L. Dumitrescu, D. T. M. Huong, N. V. Hung, B. Crousse and D. Bonnet-Delpon, Eur. J. Med. Chem., 2010, 45, 3213–3218 Search PubMed.
- H. Sheng and H. Sun, Nat. Prod. Rep., 2011, 28, 543–593 RSC.
- Y. Zhou, J. Wang, Z. Gu, S. Wang, W. Zhu, J. L. Aceña, V. A. Soloshonok, K. Izawa and H. Liu, Chem. Rev., 2016, 116, 422–518 CrossRef CAS PubMed.
- P. Jeschke, Pest Manag. Sci., 2017, 73, 1053–1066 CrossRef CAS PubMed.
- J. Wang, M. Sanchez-Rosello, J. L. Acena, C. Del Pozo, A. E. Sorochinsky, S. Fustero, V. A. Soloshonok and H. Liu, Chem. Rev., 2014, 114, 2432–2506 CrossRef CAS PubMed.
- H. J. Bohm, D. Banner, S. Bendels, M. Kansy, B. Kuhn, K. Müller, U. Obst-Sander and M. Stahl, ChemBioChem, 2004, 5, 637–643 CrossRef PubMed.
- C. Isanbor and D. O'Hagan, J. Fluorine Chem., 2006, 127, 303–319 CrossRef CAS.
- J. P. Bégué and D. Bonnet-Delpon, J. Fluorine Chem., 2006, 127, 992–1012 CrossRef.
- K. L. Kirk, J. Fluorine Chem., 2006, 127, 1013–1029 CrossRef CAS.
- P. Fischer, M. Morris, H. Müller-Bunz and P. Evans, Eur. J. Org Chem., 2020, 2020(9), 1165–1176 CrossRef CAS.
- Y. P. Budiman, S. A. Westcott, U. Radius and T. B. Marder, Adv. Synth. Catal., 2021, 363, 2224–2255 CrossRef CAS.
- Y. P. Budiman, R. N. Perutz, P. G. Steel, U. Radius and T. B. Marder, Chem. Rev., 2024, 124, 4822–4862 CrossRef CAS PubMed.
- Y. P. Budiman, P. A. Tasripin, M. R. Ramadhan, R. Hannifah, Y. Deawati, T. Mayanti, Solihudin, M. H. Putra, U. Radius and T. B. Marder, Chem.–Asian J., 2025, 20(10), e202500073 CrossRef CAS PubMed.
- Y. P. Budiman, M. H. Putra, M. R. Ramadhan, R. Hannifah, C. Luz, I. Z. Ghafara, R. Rustaman, E. E. Ernawati, T. Mayanti, A. Groß, U. Radius and T. B. Marder, Chem.–Asian J., 2024, 19(8), e202400094 CrossRef CAS PubMed.
- N. A. Meanwell, J. Med. Chem., 2018, 61, 5822–5880 CrossRef CAS PubMed.
- Q. Wang, H. Song and Q. Wang, Chin. Chem. Lett., 2022, 33(2), 626–642 Search PubMed.
- W. K. Hagmann, J. Med. Chem., 2008, 51, 4359–4369 CrossRef CAS PubMed.
- D. O'Hagan, J. Fluorine Chem., 2010, 131, 1071–1081 CrossRef.
- S. Dayalan Naidu and A. T. Dinkova-Kostova, Trends Pharmacol. Sci., 2023, 44, 394–395 Search PubMed.
- L. Borkova, L. Jasikova, J. Rehulka, K. Frisonsova, M. Urban, I. Frydrych, I. Popa, M. Hajduch, N. J. Dickinson, M. Vlk, P. Dzubak and J. Sarek, Eur. J. Med. Chem., 2015, 96, 482–490 Search PubMed.
- H. F. Lu, C. Xie, J. Chang, G. Q. Lin and X. Sun, Eur. J. Med. Chem., 2011, 46, 1743–1748 Search PubMed.
- H. J. Böhm, D. Banner, S. Bendels, M. Kansy, B. Kuhn, K. Müller, U. Obst-Sander and M. Stahl, ChemBioChem, 2004, 5, 637–643 CrossRef PubMed.
- K. Müller, C. Faeh and F. Diederich, Science, 2007, 317(5846), 1881–1886 CrossRef PubMed.
- D. O'Hagan, J. Fluorine Chem., 2010, 131, 1071–1081 CrossRef.
- W. K. Hagmann, J. Med. Chem., 2008, 51, 4359–4369 Search PubMed.
- I. V. Il’ina, O. S. Patrusheva, D. V. Korchagina, K. P. Volcho and N. F. Salakhutdinov, Chem. Heterocycl. Compd., 2020, 56, 867–874 Search PubMed.
- J. R. Coats, L. L. Karr and C. D. Drewes, ACS Symp. Ser., 1991, 449, 305–316 CrossRef CAS.
- P. J. Rice and J. R. Coats, Pestic. Sci., 1994, 41, 195–202 CrossRef CAS.
- I. V. Ilyina, O. S. Patrusheva, V. V. Zarubaev, M. A. Misiurina, A. V. Slita, I. L. Esaulkova, D. V. Korchagina, Y. V. Gatilov, S. S. Borisevich, K. P. Volcho and N. F. Salakhutdinov, Bioorg. Med. Chem. Lett., 2021, 31, 127677 Search PubMed.
- N. S. Li-Zhulanov, I. V. Il’ina, A. Chicca, P. chenker, O. S. Patrusheva, E. V. Nazimova, D. V. Korchagina, M. Krasavin, K. P. Volcho and N. F. Salakhutdinov, Med. Chem. Res., 2019, 28, 450–464 CrossRef CAS.
- Y. Kishi, S. Inagi and T. Fuchigami, Eur. J. Org Chem., 2009, 2009(1), 103–109 CrossRef.
- A. J. Cresswell, S. G. Davies, P. M. Roberts and J. E. Thomson, Chem. Rev., 2015, 115, 566–611 CrossRef CAS PubMed.
- O. S. Mikhalchenko, D. V. Korchagina, K. P. Volcho and N. F. Salakhutdinov, Beilstein J. Org. Chem., 2016, 12, 648–653 Search PubMed.
- I. V. Il'Ina, D. V. Korchagina, K. P. Volcho and N. F. Salakhutdinov, Russ. J. Org. Chem., 2010, 46, 998–1001 Search PubMed.
- S. Kurbakova, I. Il'Ina, A. Pavlova, D. Korchagina, O. Yarovaya, T. T. Y. Tolstikova, K. Volcho and N. Salakhutdinov, Med. Chem. Res., 2014, 23, 1709–1717 CrossRef CAS.
- A. Torozova, P. Mäki-Arvela, A. Aho, N. Kumar, A. Smeds, M. Peurla, R. Sjöholm, I. Heinmaa, D. V. Korchagina, K. P. Volcho, N. F. Salakhutdinov and D. Y. Murzin, J. Mol. Catal. A Chem., 2015, 397, 48–55 CrossRef CAS.
- X. Zhang, H. Li, L. You, Y. Tang and R. P. Hsung, Adv. Synth. Catal., 2006, 348, 2437–2442 Search PubMed.
- J. K. Addo, E. Owusu-Ansah, N. T. K. D. Dayie, X. Cheseto and B. Torto, Heliyon, 2022, 8, e10836 CrossRef CAS PubMed.
- O. Lefebvre, T. Brigaud and C. Portella, J. Org. Chem., 2001, 66, 1941–1946 CrossRef CAS PubMed.
- N. O. Ilchenko, D. V. Sudarikov, P. A. Slepukhin, S. A. Rubtsova and A. V. Kutchin, ChemistrySelect, 2021, 6, 1710–1714 Search PubMed.
- D. V. Sudarikov, Y. V. Krymskaya, N. O. Il’chenko, P. A. Slepukhin, S. A. Rubtsova and A. V. Kutchin, Russ. Chem. Bull., Int. Ed., 2018, 67, 731–742 CrossRef CAS.
- P. A. Petrova, D. V. Sudarikov, L. L. Frolova, R. V. Rumyantcev, S. A. Rubtsova and A. V. Kutchin, Molecules, 2022, 27, 7068 Search PubMed.
- M. Rueda-Becerril, C. Chatalova Sazepin, J. C. T. Leung, T. Okbinoglu, P. Kennepohl, J. F. Paquin and G. M. Sammis, J. Am. Chem. Soc., 2012, 134, 4026–4029 CrossRef CAS PubMed.
- N. A. Romero and D. A. Nicewicz, Chem. Rev., 2016, 116, 10075–10166 CrossRef CAS PubMed.
- D. D. Bume, C. R. Pitts, F. Ghorbani, S. A. Harry, J. N. Capilato, M. A. Siegler and T. Lectka, Chem. Sci., 2017, 8, 6918–6923 Search PubMed.
- J. B. Xia, Y. Ma and C. Chen, Org. Chem. Front., 2014, 1, 468–472 RSC.
- A. C. Sather, H. G. Lee, V. Y. De La Rosa, Y. Yang, P. Müller and S. L. Buchwald, J. Am. Chem. Soc., 2015, 137, 13433–13438 Search PubMed.
- Y. Ye, T. Takada and S. L. Buchwald, Angew. Chem., 2016, 128, 15788–15792 Search PubMed.
- S. Heptinstall, D. V. C. Awang, B. A. Dawson, D. Kindack, D. W. Knight and J. May, J. Pharm. Pharmacol., 1992, 44, 391–395 Search PubMed.
- A. Arcadi, M. Chiarini, F. Marinelli, Z. Berente and L. Kollàr, Org. Lett., 2000, 2, 69–72 CrossRef CAS PubMed.
- J. Moïse, S. Arseniyadis and J. Cossy, Org. Lett., 2007, 9, 1695–1698 CrossRef.
- C. Han, F. J. Barrios, M. V. Riofski and D. A. Colby, J. Org. Chem., 2009, 74, 7176–7179 Search PubMed.
- S. Neelakantan, S. Nasim, M. L. Guzman, C. T. Jordan and P. A. Crooks, Bioorg. Med. Chem. Lett., 2009, 19, 4346–4349 Search PubMed.
- J. R. Woods, H. Mo, A. A. Bieberich, T. Alavanja and D. A. Colby, J. Med. Chem., 2011, 54, 7934–7941 CrossRef CAS PubMed.
- A. Ghantous, A. Sinjab, Z. Herceg and N. Darwiche, Drug Discov. Today, 2013, 18, 894–905 CrossRef CAS PubMed.
- Z.-J. Yang, W.-Z. Ge, Q.-Y. Li, Y. Lu, J.-M. Gong, B.-J. Kuang, X. Xi, H. Wu, Q. Zhang and Y. Chen, J. Med. Chem., 2015, 58(17), 7007–7020 Search PubMed.
- S. Adekenov, Z. R. Shaimerdenova, K. S. Adekenova and A. S. Kishkentayeva, Fitoterapia, 2022, 158, 105154 CrossRef CAS PubMed.
- P. Thirumurugan, D. Matosiuk and K. Jozwiak, Chem. Rev., 2013, 113, 4905–4979 CrossRef CAS PubMed.
- F. S. El-Feraly, Melampolides from Magnolia Grandiflora, 1984, p. 23 Search PubMed.
- V. Janganati, N. R. Penthala, N. R. Madadi, Z. Chen and P. A. Crooks, Bioorg. Med. Chem. Lett., 2014, 24, 3499–3502 Search PubMed.
- V. Janganati, J. Ponder, M. Balasubramaniam, P. Bhat-Nakshatri, E. E. Bar, H. Nakshatri, C. T. Jordan and P. A. Crooks, Eur. J. Med. Chem., 2018, 5(157), 562–581 CrossRef PubMed.
- C. L. Lee, L. C. Chiang, L. H. Cheng, C. C. Liaw, M. H. Abd El-Razek, F. R. Chang and Y. C. Wu, J. Nat. Prod., 2009, 72, 1568–1572 Search PubMed.
- A. Abad, C. Agulló, A. C. Cuñ and D. Pardo, Synthesis of Fluorinated Drimanes, Preparation of 9aF-drimenin, 2003, p. 44 Search PubMed.
- A. Abad, C. Agulló, A. C. Cuñat, A. González-Coloma and D. Pardo, Eur. J. Org Chem., 2010, 2010(11), 2182–2198 Search PubMed.
- J. Frohwitter, S. A. E. Heider, P. Peters-Wendisch, J. Beekwilder and V. F. Wendisch, J. Biotechnol., 2014, 191, 205–213 Search PubMed.
- C. I. Keeling and J. Bohlmann, Phytochemistry, 2006, 67, 2415–2423 CrossRef CAS PubMed.
- M. A. Gromova, Y. V. Kharitonov, I. Y. Bagryanskaya and E. E. Shults, ChemistryOpen, 2018, 7, 890–901 CrossRef CAS PubMed.
- M. A. Gromova, Y. V. Kharitonov, L. V. Politanskaya, E. V. Tretyakov and E. E. Shults, J. Fluorine Chem., 2020, 236, 109554 CrossRef CAS.
- I. Ojima, J. Chen, L. Sun, C. P. Borella, T. Wang, M. L. Miller, S. Lin, X. Geng, L. Kuznetsova, C. Qu, D. Gallager, X. Zhao, I. Zanardi, S. Xia, S. B. Horwitz, J. M. Mallen-St Clair, J. L. Guerriero, D. Bar-Sagi, J. M. Veith, P. Pera and R. J. Bernacki, J. Med. Chem., 2008, 51, 3203–3221 CrossRef CAS PubMed.
- M. A. González, Nat. Prod. Rep., 2015, 32, 684–704 RSC.
- M. I. Lapuh, A. Dana, P. H. Di Chenna, B. Darses, F. J. Durán and P. Dauban, Org. Biomol. Chem., 2019, 17, 4736–4746 RSC.
- J. Anaya, C. Grande, M. Grande, A.-I. Patino and P. Torres, Synlett, 1999, 9, 1429–1431 Search PubMed.
- L. Wang, D. Li, S. Xu, H. Cai, H. Yao, Y. Zhang, J. Jiang and J. Xu, Eur. J. Med. Chem., 2012, 52, 242–250 CrossRef CAS PubMed.
- D. Li, T. Han, S. Xu, T. Zhou, K. Tian, X. Hu, K. Cheng, Z. Li, H. Hua and J. Xu, Molecules, 2016, 21, 575 Search PubMed.
- I. A. Bessonova and S. A. Saidkhodzhaeva, Chem. Nat. Compd., 2000, 36, 419–477 CrossRef CAS.
- K. Wada, M. Goto, H. Tanaka, M. Mizukami, Y. Suzuki, K. H. Lee and H. Yamashita, ACS Omega, 2022, 7, 28173–28181 CrossRef CAS PubMed.
- K. Wada, M. Hazawa, K. Takahashi, T. Mori, N. Kawahara and I. Kashiwakura, J. Nat. Med., 2011, 65, 43–49 CrossRef CAS PubMed.
- C. Y. Cao, C. C. Zhang, X. W. Shi, D. Li, W. Cao, X. Yin and J. M. Gao, ACS Chem. Neurosci., 2018, 9, 1607–1615 CrossRef CAS PubMed.
- K. Wada, E. Ohkoshi, Y. Zhao, M. Goto, S. L. Morris-Natschke and K. H. Lee, Bioorg. Med. Chem. Lett., 2015, 25, 1525–1531 Search PubMed.
- L. Zhang, Y. Xie, X. Liang, L. Yin, C. He, Z. Yin, G. Yue, Y. Zou, L. Li, X. Song and H. Tang, Bioorg. Chem., 2023, 135, 106501 Search PubMed.
- A. K. Khan, A. Ahmed, M. Hussain, I. A. Khan, S. A. Ali, A. D. Farooq and S. Faizi, Eur. J. Med. Chem., 2017, 138, 480–490 Search PubMed.
- X. Rao, Z. Song and L. He, Heteroatom Chem., 2008, 19, 512–516 Search PubMed.
- U. Supratman, W. Naibaho, S. Salam, R. Maharani, A. T. Hidayat, D. Harneti, Nurlelasari and Y. Shiono, Phytochem. Lett., 2019, 30, 81–87 CrossRef CAS.
- J. Sianturi, D. Harneti, Darwati, T. Mayanti, U. Supratman and K. Awang, Nat. Prod. Res., 2016, 30, 2204–2208 CrossRef CAS PubMed.
- A. A. Naini, T. Mayanti and U. Supratman, Arch. Pharm. Res., 2022, 45, 63–89 CrossRef CAS PubMed.
- E. Hilmayanti, Nurlelasari, U. Supratman, K. Kabayama, A. Shimoyama and K. Fukase, Phytochemistry, 2022, 203, 113469 CrossRef PubMed.
- S. A. Riyadi, A. A. Naini and U. Supratman, Molecules, 2023, 28, 4874 CrossRef CAS PubMed.
- B. M. F. Gonçalves, J. A. R. Salvador, S. Marín and M. Cascante, Eur. J. Med. Chem., 2016, 114, 101–117 CrossRef PubMed.
- D. Y. Seo, S. R. Lee, J. W. Heo, M. H. No, B. D. Rhee, K. S. Ko, H. B. Kwak and J. Han, Korean J. Physiol. Pharmacol., 2018, 22, 235–248 Search PubMed.
- A. S. Leal, R. Wang, J. A. R. Salvador and Y. Jing, ChemMedChem, 2012, 7, 1635–1646 CrossRef CAS PubMed.
- C. R. Pitts, D. D. Bume, S. A. Harry, M. A. Siegler and T. Lectka, J. Am. Chem. Soc., 2017, 139, 2208–2211 CrossRef CAS PubMed.
- C. R. Pitts, M. A. Siegler and T. Lectka, J. Org. Chem., 2017, 82, 3996–4000 Search PubMed.
- D. D. Bume, S. A. Harry, T. Lectka and C. R. Pitts, J. Org. Chem., 2018, 83, 8803–8814 CrossRef CAS PubMed.
- D. D. Bume, S. A. Harry, C. R. Pitts and T. Lectka, J. Org. Chem., 2018, 83, 1565–1575 Search PubMed.
- J. Li, M. Goto, X. Yang, S. L. Morris-Natschke, L. Huang, C. H. Chen and K. H. Lee, Bioorg. Med. Chem. Lett., 2016, 26, 68–71 Search PubMed.
- J. Li, L. C. Chang, K. Y. Hsieh, P. L. Hsu, S. J. Capuzzi, Y. C. Zhang, K. P. Li, S. L. Morris-Natschke, M. Goto and K. H. Lee, Bioorg. Med. Chem., 2019, 27, 2871–2882 Search PubMed.
- S. Rozen, I. Shahak and E. D. Bergmann, J. Org. Chem., 1975, 40(20), 2966–2969 CrossRef CAS.
- W. S. Johnson, W. A. Vredenburgh and J. E. Pike, J. Am. Chem. Soc., 1960, 82, 3409–3415 CrossRef CAS.
- S. Rozen and E. D. Bergmann, Isr. J. Chem., 1973, 2, 825–833 CrossRef.
- D. Biedermann, J. Sarek, J. Klinot, M. Hajduch and P. Dzubak, Synthesis, 2005, 2005(7), 1157–1163 Search PubMed.
- B. Slavíková, A. Kasal, H. Chodounská and Z. Krištofíková, Collect. Czech. Chem. Commun., 2002, 67, 30–46 CrossRef.
- T. S. Khlebnikova, Y. A. Piven and F. A. Lakhvich, Chem. Nat. Compd., 2017, 53, 486–491 CrossRef CAS.
- T. S. Khlebnicova, Y. A. Piven, A. V. Baranovsky, F. A. Lakhvich, I. V. Sorokina and T. G. Tolstikova, Steroids, 2019, 147, 62–69 CrossRef CAS PubMed.
- D. Zicāne, Z. Tetere, I. Rāviņa, V. Kumpiņš, I. Rijkure, I. Mieriņa, U. Peipiņš and M. Turks, Steroids, 2017, 117, 77–89 CrossRef PubMed.
- A. Regueiro-Ren, J. J. Swidorski, Z. Liu, Y. Chen, N. Sin, S. Y. Sit, J. Chen, B. L. Venables, J. Zhu, B. Nowicka-Sans, B. Protack, Z. Lin, B. Terry, H. Samanta, S. Zhang, Z. Li, J. Easter, B. R. Beno, V. Arora, X. S. Huang, S. Rahematpura, D. D. Parker, R. Haskell, K. S. Santone, M. I. Cockett, M. Krystal, N. A. Meanwell, S. Jenkins, U. Hanumegowda and I. B. Dicker, J. Med. Chem., 2018, 61, 7289–7313 CrossRef CAS PubMed.
- O. B. Kazakova, E. F. Khusnutdinova, A. V. Petrova, E. Y. Yamansarov, A. N. Lobov, A. A. Fedorova and K. Y. Suponitsky, J. Nat. Prod., 2019, 82, 2550–2558 CrossRef CAS PubMed.
- S. H. Lau, A. Galván, R. R. Merchant, C. Battilocchio, J. A. Souto, M. B. Berry and S. V. Ley, Org. Lett., 2015, 17, 3218–3221 CrossRef CAS PubMed.
- Y. Zhang and H. Xu, RSC Adv., 2017, 7, 35191–35220 RSC.
- K. Mitsui, H. Saito, R. Yamamura, H. Fukaya, Y. Hitotsuyanagi and K. Takeya, J. Nat. Prod., 2006, 69, 1310–1314 CrossRef CAS PubMed.
- Y. Hitotsuyanagi, K. Mitsui, H. Fukaya and K. Takeya, Phytochem. Lett., 2012, 5, 486–489 CrossRef CAS.
- L. C. Tavares, T. S. Fernandes, V. Ilha, A. T. Neto, E. W. dos Santos, R. A. Burrow, F. A. Duarte, E. M. M. Flores, U. F. Silva, M. A. Mostardeiro and A. F. Morel, J. Braz. Chem. Soc., 2016, 27, 161–178 CAS.
- X. Huang, T. Li, X. Shan, R. Lu, M. Hao, M. Lv, Z. Sun and H. Xu, J. Agric. Food Chem., 2020, 68, 12241–12251 CrossRef CAS PubMed.
- Y. Guo, Y. Yan, X. Yu, Y. Wang, X. Y. Zhi, Y. Hu and H. Xu, J. Agric. Food Chem., 2012, 60, 7016–7021 Search PubMed.
- M. B. Huda, Nurlelasari, W. Safriansyah, M. Fajar, I. I. Widiyowati, U. Supratman, Y. Permana and Y. P. Budiman, Commun. Sci. Technol., 2024, 9, 366–371 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |
