 Open Access Article
Open Access ArticleTransforming plastic waste into liquid fuels: novel upgrading of polypropylene pyrolysis oil and its utilization in a diesel engine
Abhishek Kumar Yadav†
a,
Suparna Maity†a,
Sandeep Kumar Yadav a,
Vasudeva Madav
a,
Vasudeva Madav b,
G. N. Kumarb and
Saikat Dutta
b,
G. N. Kumarb and
Saikat Dutta *a
*a
aDepartment of Chemistry, National Institute of Technology Karnataka, Surathkal, Mangalore–575025, Karnataka, India. E-mail: sdutta@nitk.edu.in
bDepartment of Mechanical Engineering, National Institute of Technology Karnataka, Surathkal, Mangalore–575025, Karnataka, India
First published on 24th April 2026
Abstract
Pyrolysis of end-of-life polypropylene (EOL-PP) items into polypropylene pyrolysis oil (PPO) is a scalable, economically feasible, and environmentally acceptable waste-to-energy pathway. The poor stability (e.g., storage and oxidative) and inferior physicochemical properties (e.g., low flash point) of crude PPO (CPPO) mandate an inexpensive, scalable, and eco-friendly upgrading method to enable its use as engine fuel. This work reports a novel upgrading of CPPO into upgraded PPO (UPPO) by sequentially removing the excessively volatile and chemically unstable components in a two-step process. The chemical composition of the volatile fraction (b.p. <150 °C) resembled that of gasoline and the heavy naphtha fraction, which promises to diversify the applications and markets of PPO. The physicochemical properties and spectral characteristics of CPPO and UPPO were analyzed to quantify the improvements in fuel properties. UPPO showed noticeably better physicochemical properties than CPPO. The UPPO (10–25 vol%)-diesel blends were used in a diesel engine to study the engine performance and emission characteristics, which afforded comparable engine performance (e.g., brake thermal efficiency) and reduced emissions (i.e., NOx, CO, and unburnt hydrocarbons) compared to unblended diesel.
1. Introduction
The applications of polypropylene (PP) have rapidly expanded over the past two decades across various industries due to its pliable properties, including chemical and moisture resistance, light weight, high impact strength, and good moldability.1 However, end-of-life polypropylene (EOL-PP) items have created serious environmental concerns due to their slow environmental degradation and accumulation in landfill sites.2 Indiscriminate disposal of EOL-PP leads to pollution (air, water, and soil), posing health-, fire-, and ecological hazards.2 A scalable, economical, and eco-friendly solution for the waste management of EOL-PP is paramount from social, economic, and environmental perspectives.3 In this regard, pyrolysis of EOL-PP into crude polypropylene pyrolysis oil (CPPO) has shown promise due to the process simplicity, scalability, and marketability of CPPO as a hydrocarbon-based liquid fuel.4 The hydrocarbon backbone of EOL-PP can be cleaved by heat in the absence (thermal) or presence of a catalyst (catalytic) to form a mixture of liquid hydrocarbons of varying carbon chain lengths, mainly in the diesel fuel range.5–7 A considerable portion of a barrel of petroleum is transformed into synthetic thermoplastics (e.g., PP); therefore, the production of CPPO from EOL-PP is critical in a resource-constrained world helping to reclaim the resources while minimizing waste accumulation.8,9 The pyrolysis of EOL-PP produces CPPO, combustible gases, and carbonized char, and their relative proportions depend on the feedstock and the process parameters.10 Even though all the components obtained during the pyrolysis process can be valorized individually, CPPO is the most important fraction due to its diverse applications and huge market potential.11,12 Significant research has been devoted to fine-tuning the pyrolysis of EOL-PP to maximize CPPO yield and improve its quality as a liquid fuel.13 CPPO can be used as a substitute for diesel in furnaces and diesel engines.14 However, the inconsistent quality of CPPO produced during thermal pyrolysis of EOL-PP and its degradation during storage poses a major challenge for using it directly as furnace and engine fuel. Several process parameters, including the quality of the feedstock (i.e., the extent of degradation and ageing of EOL-PP and contamination from other plastics), reactor type, pyrolysis temperature, and residence time, determine the quality of CPPO. The oxygen content in EOL-PP increases over time since the PP backbone gets partially oxidized by photooxidation, leading to oxygenated compounds in the CPPO.15 The inferior thermal, oxidative, and storage stability of CPPO primarily originates from these polar components. Moreover, the unstable compounds produced during the pyrolysis of EOL-PP, such as terminal olefins, can undergo oxidation and oligomerization, forming a waxy residue during the storage and transportation of CPPO.16,17 Therefore, attempts have been made to improve the quality of CPPO by suitable upgrading. The upgraded PPO (UPPO) can be produced using two primary strategies. The catalytic pyrolysis of EOL-PP can produce UPPO, as the catalyst promotes selective bond cleavage at lower temperatures and minimizes the formation of unstable, undesirable compounds.18 However, the catalysts are often expensive, and recovering them from the carbonized char is very challenging.19 Catalyst has also been used to upgrade the CPPO from thermal pyrolysis in a separate step.20 However, this strategy often requires expensive catalysts (e.g., zeolite), additional energy (i.e., heat), and a catalyst recycling setup. An alternative strategy for obtaining UPPO is to upgrade CPPO downstream via processes like fractional distillation. However, the process is energy-intensive, low-yielding, and produces significant waste from repolymerization and carbonization reactions.21,22 Attempts have been made to remove the acidic and oxygenated polar compounds by preferential solubility and selective adsorption over a suitable adsorbent.23 However, the cost of solvents and adsorbents increases the operational expenditures.24 Attempts have been made on radical-mediated catalytic deconstruction of plastics into valuable chemicals and monomers using a uranyl-based photocatalytic system. Uranyl-based photocatalytic systems involve radioactive materials, raising concerns towards process scalability and environmental and health safety during plastic upgradation at the commercial scale.25 We envisioned that the excessively volatile fraction (b.p. <150 °C) could be distilled from CPPO under atmospheric pressure to improve the flash point. The acid-promoted degumming process can remove the relatively unstable and waxy compounds from the remaining liquid.26 A suitable organic or mineral acid insoluble in CPPO can react with oxygenates and unstable hydrocarbons (e.g., terminal olefins) and precipitate them from the CPPO.Typically, a multi-stage treatment and purification process is involved in acid degumming of vegetable oil.27 For example, the oil sample must be treated with aqueous acid at high temperature or under sonication. Then the oil sample must be phase-separated, pH-adjusted with a base, and dried to remove any trace water. However, such elaborate operational steps would compromise the process's scalability and increase its cost. We reasoned that the hydrocarbon-rich CPPO is radically different from triglyceride-containing vegetable oil. Therefore, the acid-promoted degumming of CPPO can be simplified by selecting an appropriate acid catalyst.
This work shows how CPPO can be upgraded into UPPO by a two-stage process. In the first step, the excessively volatile fraction (b.p. <150 °C) from CPPO was separated by fraction distillation under atmospheric pressure. The heavier fraction was then degummed with acids to easily separate unstable compounds in their oligomerized or polymerized forms. The colorless volatile fraction containing light hydrocarbons can be used as the naphtha fraction or as aviation fuel and can be conveniently integrated into the petrorefinery infrastructure. This high-value fraction can improve the economic feasibility of producing PPO from EOL-PP. The physicochemical characteristics, thermal properties, and chemical composition of CPPO and UPPO (prepared over two steps) were analyzed. The UPPO was then blended with diesel in 10–20 vol%, and the blended fuels were then used as fuels in a diesel engine (Fig. 1). The engine performance and emission characteristics of the UPPO-diesel blends were studied.
 | ||
| Fig. 1 Overall scheme and objectives of this work on using UPPO as an alternative liquid fuel in the diesel engine. | ||
2. Experimental methodology
2.1 Materials
Crude polypropylene pyrolysis oil (CPPO) was obtained from a local PPO manufacturer. The end-of-life polypropylene chairs were used as feedstock for producing CPPO. The powdered EOL-PP was fed into a 500 kg batch reactor. The thermal pyrolysis was performed at 450 °C, which took a total of 4 h to complete. The vaporized CPPO emanating from the reactor was cooled in a water-cooled condenser and stored in HDPE drums. Diesel was purchased from a commercial outlet of the Indian Oil Corporation Limited (IOCL) in Mangalore, Karnataka. Orthophosphoric acid (85%) and polyphosphoric acid (98%) were procured from Loba Chemie Pvt. Ltd.2.2 Instrumental analysis
The molecular composition of CPPO and UPPO was analyzed by Fourier transform infrared (FTIR), 1H-NMR, and GC-MS. The FTIR spectra were recorded in a Bruker Alpha-II instrument using the attenuated total reflectance (ATR) method. A few drops of the fuel samples were diluted in dichloromethane, and a thin film was formed on the ATR counter top by evaporating a drop of the solution at room temperature. The recorded FTIR spectrum for each sample was an average of 24 scans collected at a scan rate of 4 scans per second. The selected spectral range was 600 cm−1 to 4000 cm−1 to analyze the full width of the functional group and fingerprint regions. The 1H-NMR data of the fuel samples were used to study the chemical composition, H/C ratio, and iso-paraffin index of pyrolysis oil. The 1H-NMR spectra were recorded on a Bruker NanoBay® NMR instrument operating at 400 MHz. Deuterated chloroform (CDCl3) was chosen as the solvent for diluting the fuel samples and recording the NMR data. The gas chromatography (GC) analyses of CPPO and UPPO were performed in a LECO Pegasus BT 4D spectrometer. Given the hydrocarbon-rich composition of CPPO and UPPO, a non-polar column (Rtx-5 ms, 30 m × 0.25 mm × 0.25 µm) and a polar column (Rxi-17) were employed to help separate the compounds. High-purity helium gas was employed as the carrier gas at a constant flow rate of 1.40 mL min−1. The injector temperature was fixed at 250 °C with a 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 split ratio. The oven temperature was initially set to 60 °C for 2 min, then ramped at 10 °C min−1 to 300 °C. LECO Chroma software was used to process the acquired chromatograms further. The compounds were characterized by mass spectrometry and matched against the library integrated into the software to determine the percentage similarity.
1 split ratio. The oven temperature was initially set to 60 °C for 2 min, then ramped at 10 °C min−1 to 300 °C. LECO Chroma software was used to process the acquired chromatograms further. The compounds were characterized by mass spectrometry and matched against the library integrated into the software to determine the percentage similarity.
2.3 Upgradation of CPPO
A straightforward and scalable two-step upgradation procedure was employed to convert CPPO into UPPO. In the first step, the excessively volatile compounds in CPPO (b.p. < 150 °C), which are responsible for a very low flash point, were removed by fractional distillation under atmospheric pressure. CPPO was taken in a round-bottomed flask and placed in an electrically heated furnace. The flask was connected to a water-cooled condenser through a glass adapter. The other end of the condenser was connected to a receiver flask (a distillation setup) through another glass adapter with a vacuum joint exposed to the atmosphere. The reaction flask containing CPPO (50 mL) was heated to 150 °C until the volatile compounds distilled out and condensed in the collecting flask as a transparent liquid (Scheme 1). The heavier fraction remaining in the distillation flask was evaluated as a diesel substitute because its molecular composition matched that of diesel fuel. | ||
| Scheme 1 Schematic representation of the two-step upgrading of CPPO into UPPO, containing process details and mass balance. | ||
In the second step of the upgrading process, the residual polypropylene pyrolysis oil (DPPO) post-distillation was treated with 3% (v/v) of orthophosphoric acid (85%) for 2 h at room temperature to remove the gum-forming and polar components (e.g., oxygenates, heterocycles). The gum-forming compounds and various polar compounds (e.g., oxygenates) separate. The supernatant oil was decanted and stored in sealed glass containers. It was observed that a slightly elevated temperature (50 °C) accelerated the degumming process, and the contact time was reduced significantly (<1 h). The loss during this upgrading step was found to be 5–8 vol%, depending on the quality of different batches of CPPO. The upgraded polypropylene pyrolysis oil (UPPO) was blended with commercial diesel at different volumetric ratios to assess their combustion properties and emission characteristics in a compression-ignition internal combustion engine.
2.4 Characterization of CPPO and UPPO
The chemical composition of the oil samples was determined as the volume percentages of paraffins, olefins, and aromatics by integrating the peaks at various regions of the 1H-NMR spectrum.28 Similarly, the isoparaffin index and H/C ratio were estimated from the integral areas of peaks in different chemical shift regions, as shown in Table 1.29| Proton type | Chemical shift | |
|---|---|---|
| A | Aromatic | 8.0–6.6 |
| B | Olefin | 6.0–4.5 |
| C | Methylene/methane (attached to aromatics/allyl) | 3.0–2.0 |
| D | Methine (paraffins) | 2.0–1.5 |
| E | Methylene (paraffins) | 1.5–1.0 |
| F | Methyl (paraffins) | 1.0–0.6 |
The following eqn (1)–(5) have been used to calculate the percentages of various types of protons in the molecular components of the PPO samples.28,29
 | (1) |
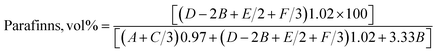 | (2) |
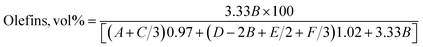 | (3) |
 | (4) |
 | (5) |
2.5 Preparation of fuel blends
UPPO was blended (10–25 vol%) with petrodiesel to study the combustion performance and emission characteristics of the UPPO-diesel blends in a compression-ignition single-cylinder diesel engine. The required volume of UPPO was measured in a calibrated measuring cylinder (100 mL) and introduced into a beaker (2 L). The required quantity of diesel was added using the same measuring cylinder, and the total volume was made up to 1 L. For example, the B10 fuel blend was prepared by mixing 100 mL of UPPO with 900 mL of diesel. A Teflon-coated magnetic stirring rod was introduced in the beaker, and the mixture was stirred at 500 rpm for 1 h. Similarly, 150 mL, 200 mL, and 250 mL UPPO were used to make B15, B20, and B25 UPPO-diesel blends of 1 L volume, respectively. The fuel mixtures were stored in air-tight glass bottles.2.6 Engine testing setup and instrumentation
The UPPO-diesel blends were homogenized by magnetic stirring and then filtered through a Whatman filter paper (grade 4) before studying their performance in a single-cylinder compression-ignition engine. The engine setup consisted of a single-cylinder, naturally aspirated, direct-injection, water-cooled diesel engine (make: Kirloskar, model: TV1) coupled to an eddy-current dynamometer as a load. The engine was designed for a power output of 3.5 kW at 1500 rpm. Complete specifications of the single-cylinder diesel engine are shown in the SI file (Table S1, SI).Single-cylinder diesel engines are widely used as the primary power source for irrigation and decentralized energy production across the Indian subcontinent, particularly in rural and semi-urban areas. Due to their simple design, robustness, and adaptability to a wide range of fuels, these engines are routinely used to test alternative fuels at most research institutions. Fuel consumption was measured using a conventional gravimetric method with a calibrated burette and a stopwatch, enabling accurate determination of the fuel flow rate under varying engine load. A piezoelectric Kistler pressure sensor was mounted on the cylinder to record the in-cylinder combustion characteristics. The pressure signals were digitized and recorded by a high-speed data-acquisition system synchronized with the crank angle encoder. The recorded pressure data were subsequently analyzed using dedicated software for combustion analysis (Engine Soft, Version 9.0). Thermal measurements were obtained using a K-type thermocouple made of Cr–Al alloy interfaced with a temperature sensor. This setup was used to record the temperature of lubrication oil and exhaust gas. The emission of various gases from the exhaust was studied using the INDUS 5 Gas Analyzer (Model: PEA 205N). The fuel consumption was determined volumetrically using a calibrated burette and stopwatch, while the airflow rate was measured manually for each engine load. Initially, the engine was run for 30 minutes at 1500 rpm at a fixed load to reach the steady-state conditions before data recording was initiated. Technical specifications of the exhaust gas analyzer used in the present work are listed in Table S2 of the SI. The photographic images of the engine setup are shown in the SI (Fig. S4–S7).
3. Results and discussion
The following subsections present detailed characterizations of CPPO and UPPO, the preparation of UPPO-diesel blends, their use as fuels in a diesel engine, and a discussion of their combustion properties and emission characteristics.3.1 Physicochemical and thermal analyses of CPPO and UPPO
The upgradation of PPO by distillation followed by degumming significantly improved the physicochemical properties of UPPO, with most values conforming to regulatory guidelines and being comparable to those of diesel fuel (Table 2). The calorific value of UPPO improved to 45.99 MJ kg−1 from 44.88 MJ kg−1 in CPPO, suggesting the elimination of oxygenates during the two-step upgrading process. The second-step degumming process removed oxygenates and acidic compounds (e.g., carboxylic acids) from CPPO, resulting in a significant decrease in the acid value. The acid value of CPPO was 5.82 mg KOH per g, whereas that of UPPO was only 0.54 mg KOH per g. A decrease in the sulfur content in UPPO (compared to CPPO) was also observed, but it was higher than the acceptable value in petrodiesel. Therefore, a desulfurization step may be added to improve the values. In this study, UPPO was blended (i.e., 10–25 vol%) with commercial diesel to minimize any complications. For example, for 20 vol% UPPO blended with diesel (i.e., B20), the sulfur content will be around 12 ppm.
| Physicochemical properties | ASTM methods | Unit | Diesel | CPPO | UPPO |
|---|---|---|---|---|---|
| Density (15 °C) | D4052-22 | g cm−3 | 0.831 | 0.799 | 0.812 |
| Kinematic viscosity (40 °C) | D7042-21a | cSt | 2.781 | 2.126 | 3.385 |
| Flash point | D93-20 | °C | 47 | <20 | 47.5 |
| Pour point | D5950-14 | °C | −6 | −24 | −9 |
| Acid value | D974-22 | mg KOH per g | 0.06 | 5.82 | 0.54 |
| Sulfur content | D7220-22 | ppm | 6 | 46.5 | 38 |
| Calorific value | D240-19 | MJ kg−1 | 45.50 | 44.88 | 45.99 |
3.2 Compositional and spectral analyses
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching frequency of olefins. The peak at 1458 cm−1 is due to the bending (scissoring) vibration of methyl and methylene groups. The peak at 1377 cm−1 is due to the bending vibration (umbrella mode) of the methyl groups. Peaks in the fingerprint region 968 cm−1, 887 cm−1, 845 cm−1, 811 cm−1, and 738 cm−1 are also of C–H bending vibration but of the aromatic ring. The C–O stretching appeared around 1100 cm−1 in CPPO, suggesting the presence of oxygenates, which reduced significantly in UPPO.
C stretching frequency of olefins. The peak at 1458 cm−1 is due to the bending (scissoring) vibration of methyl and methylene groups. The peak at 1377 cm−1 is due to the bending vibration (umbrella mode) of the methyl groups. Peaks in the fingerprint region 968 cm−1, 887 cm−1, 845 cm−1, 811 cm−1, and 738 cm−1 are also of C–H bending vibration but of the aromatic ring. The C–O stretching appeared around 1100 cm−1 in CPPO, suggesting the presence of oxygenates, which reduced significantly in UPPO.
| Proton type | Chemical shift | Integral area of CPPO | Integral area of UPPO | |
|---|---|---|---|---|
| A | Ring aromatic | 8.0–6.6 | 0.31 | 0.15 |
| B | Olefin | 6.0–4.5 | 1 | 1 |
| C | α-Methyl | 3.0–2.0 | 0.65 | 0.93 |
| D | Methine (paraffins) | 2.0–1.5 | 3.45 | 4.31 |
| E | Methylene (paraffins) | 1.5–1.0 | 5.22 | 5.53 |
| F | Methyl (paraffins) | 1.0–0.6 | 9.60 | 9.70 |
| Aromatics, vol% | 0.8 | 0.79 | ||
| Paraffins, vol% | 65 | 69.2 | ||
| Olefins, vol% | 29 | 26.7 | ||
| H/C | 1.8 | 1.73 | ||
| Isoparaffin index | 1.23 | 1.24 |
3.3 Fuel properties analysis of various blends
Density of the UPPO-diesel fuel blends (B10, B15, and B20) was comparable to unblended diesel. More specifically, the density of unblended diesel and the B25 blend were 0.831 g cm−3 and 0.829 g cm−3, respectively, at 15 °C. Kinematic viscosity (at 40 °C) of the UPPO-diesel blends did not show any specific trend and were within acceptable limits. The pour point of the unblended diesel was −6 °C, whereas the blends showed a marginal increase to −3 °C. The pour point of the fuel is a crucial property for assessing cold–flow properties, and all the UPPO-diesel blends performed satisfactorily. The flash point, acid value, and calorific value of the fuel mixtures studied in this work were within the acceptable limits of diesel fuel. Table 4 lists some major physicochemical properties and their experimentally determined values for unblended diesel and UPPO-diesel blends.| Properties | Unit | Diesel | B10 | B15 | B20 | B25 |
|---|---|---|---|---|---|---|
| Density at 15 °C | g cc−1 | 0.831 | 0.832 | 0.827 | 0.829 | 0.826 |
| Kinematic viscosity (40 °C) | cSt | 2.781 | 2.782 | 2.757 | 2.802 | 2.825 |
| Acid value | mg KOH per g | 0.06 | 0.14 | 0.15 | 0.18 | 0.16 |
| Flash point | °C | 47 | 45 | 44.5 | 44 | 44.5 |
| Pour point | °C | −6 | −3 | −3 | −3 | −6 |
| Sulfur content | ppm | 6 | 10 | 11.5 | 14.5 | 13.5 |
| Calorific value | MJ kg−1 | 45.5 | 45.8 | 45.7 | 45.9 | 46.2 |
3.4 Utilization of UPPO-diesel fuel blends in the diesel engine
The brake-specific energy consumption (BSEC) is an important performance parameter that evaluates the energy efficiency of internal combustion engines, and it is used to determine the efficiency of converting heat energy into mechanical power. BSEC expresses the total energy input required to produce one kilowatt-hour of brake power. A lower BSEC indicates that the engine converts the fuel's chemical energy into useful mechanical energy more efficiently. Fig. 7 shows that the BSEC decreases with increasing load, indicating that the various blends perform more efficiently at higher loads than at lower ones. It was observed that the BSEC decreases from pure diesel to various fuel blends and remains almost constant with increasing blend percentage. The higher volatility of UPPO improves fuel atomization and homogenizes the air–fuel mixture, leading to more efficient combustion and reduced energy waste, thereby increasing the BTE. As a result, the engine requires less fuel energy input per unit of brake power output for the UPPO-diesel blends than for unblended diesel fuel.
Uncertainty analysis was performed to assess the accuracy of the instrumental data. The uncertainty in BTE was calculated using the root-sum-square approach. The uncertainty analysis is included in the SI (Page S2, SI) based on the published literature.30,31
MGT is a critical parameter that provides insight into heat release and use during the engine's compression, combustion, and expansion strokes. MGT refers to the average temperature of the gases inside the combustion chamber over a complete engine cycle. It signifies the thermal state of combustion characteristics, heat transfer, and overall engine performance, providing insights about the heat release inside the cylinder during each engine cycle. The average temperature of 100 MGT cycles was determined for each engine load. A higher MGT generally indicates more efficient combustion and greater heat release, thereby improving the fuel's thermal efficiency. As load increases, MGT generally rises due to increased fuel consumption. More fuel is injected at higher loads, leading to a higher heat release rate and elevating the MGT. This trend indicates improved combustion with increasing engine load. The variation of MGT across diesel and various UPPO-diesel fuel blends is shown in Fig. 9.
At lower engine loads (0–50%), the MGT of B10–B20 fuel blends were comparable with diesel, whereas the B25 fuel blend had noticeably higher values. At higher engine loads (75–100%), all the fuel blends had higher MGT than diesel, especially B25. The higher aromatic content in the UPPO may be responsible for this behavior.
Carbon monoxide (CO) forms from the incomplete combustion of carbon-based fuels in the compression-ignition internal combustion engine. In this study, CO emissions from UPPO-diesel blends were measured at varying engine loads. At 100% engine load, CO emissions decreased for UPPO-diesel blends compared to unblended diesel. The percentage CO emissions of diesel and UPPO-diesel blends at various engine loads are depicted in Fig. 11. With increasing engine load, CO emissions increased slowly across all fuel samples. The combustion process became more vigorous as the engine load reached 100%. The available time for the further oxidation of CO to CO2 was reduced, leading to a significant increase in CO emissions. The UPPO-diesel blends also showed a similar pattern, and CO emissions were comparable to those of diesel, with some variations.
Carbon dioxide emissions indicate the availability of sufficient oxygen in the combustion chamber for complete combustion of the fuels under study. Density, viscosity, oxygenates, molecular composition of the fuels, and the air-fuel ratio influence carbon dioxide emissions. It was noted that diesel had higher CO2 emissions, whereas CO2 emissions decreased slightly with increasing UPPO percentage in the UPPO-diesel blends. Since the molecular composition of UPPO was similar to diesel, the impact on CO2 emissions was less pronounced. Higher engine loads increase CP-max and MGT, thereby enhancing oxidation reactions and elevating CO2 emissions (Fig. 12).
The emission of unburnt hydrocarbons (UHC) results from the insufficient or incomplete combustion inside the cylinder in internal combustion engines. Despite some variations, UHC was lowest in the B10 blend and increased as the engine load increased from 0% to 100%. A decrease in UHC is typically observed with increasing engine load, due to improved combustion efficiency at higher in-cylinder temperatures. However, as engine load increases, the required fuel-to-air ratio increases to maintain a safe operating temperature and avoid overheating the cylinder. The published literature reported both increasing and decreasing trends for UHC with increasing engine load. At higher engine loads (i.e., 75% & 100%), UHC decreased noticeably for the UPPO-diesel blends compared to unblended diesel. The presence of oxygenated compounds in UPPO facilitates the complete combustion of the fuel mixture and reduces UHC emissions. The UHC of diesel and UPPO-diesel blends under varying engine loads is shown in Fig. 13.
4. Conclusion
This study demonstrates an effective method for upgrading CPPO to UPPO by selectively removing volatiles and unstable components. The straightforward, scalable upgrade significantly improved the physicochemical and thermal properties, as well as the storage stability, of UPPO, making it a suitable diesel substitute. The UPPO-diesel blends (10–25 vol% of UPPO) showed promising engine performance. For example, the BTE improved in the UPPO-diesel blends across all engine loads compared to unblended diesel, resulting in a decrease in BSEC and indicating improved energy utilization. The maximum cylinder pressure dropped in the UPPO-diesel blends, leading to smoother engine functioning. Compared to unblended diesel, the UPPO-diesel blends resulted in reduced emissions of NOx, CO, and UHC. This result can be explained by the presence of various oxygenates in UPPO, which reduces the total oxygen requirement for combusting the fuel mixtures and lowers the temperature produced during ignition. The mean gas temperature was comparable in UPPO-diesel blends to that in diesel alone. Therefore, UHC-diesel blends can be used as cleaner fuels in diesel engines while maintaining engine performance.Author contributions
Abhishek Kumar Yadav and Suparna Maity performed the experiments and analyzed the data. Sandeep Kumar Yadav helped collect data on the physicochemical characteristics of the fuel blends. Kumar G N and Vasudeva Madav assisted in engine performance, emission analysis, and edited the manuscript. Saikat Dutta conceptualized the work, supervised, and wrote the original manuscript.Conflicts of interest
The author declares no competing interests.Data availability
Additional supporting experimental data, photographs of the experimental setup, and the appearance of the fuel blends are available in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ra09026a.Acknowledgements
SD thanks the Science and Engineering Research Board (SERB), India, for research funding under the Core Research Grant (CRG) scheme (file no. CRG/2022/009346). The authors thank the central library of NITK, Surathkal, for providing access to academic journals and databases. The authors thank the Internal Combustion Laboratory at the Department of Mechanical Engineering, NITK, Surathkal.References
- M. T. Hossain, M. A. Shahid, N. Mahmud, A. Habib, M. M. Rana, S. A. Khan and M. D. Hossain, Discover Nano, 2024, 19, 2 CrossRef PubMed.
- V. Mannheim and Z. Simenfalvi, Polymers, 2020, 12, 1901 CrossRef CAS PubMed.
- J.-P. Lange, ACS Sustainable Chem. Eng., 2021, 9, 15722–15738 CrossRef CAS.
- J. Uebe, Z. Kryzevicius, R. Majauskiene, M. Dulevicius, L. Kosychova and A. Zukauskaite, Waste Manage. Res., 2022, 40, 1220–1230 CrossRef CAS PubMed.
- C. Park and J. Lee, Int. J. Energy Res., 2021, 45, 13088–13097 CrossRef CAS.
- A. I. Eldahshory, K. Emara, M. S. Abd-Elhady and M. A. Ismail, Sci. Rep., 2023, 13, 11766 CrossRef CAS PubMed.
- P. Rex, M. K. Rahiman, P. Barmavatu, S. B. Aryasomayajula Venkata Satya Lakshmi and N. Meenakshisundaram, Environ. Qual. Manag., 2024, 33, 501–511 CrossRef.
- R. Geyer, J. R. Jambeck and K. L. Law, Sci. Adv., 2017, 3, e1700782 CrossRef PubMed.
- L. Zou, R. Xu, H. Wang, Z. Wang, Y. Sun and M. Li, Natl. Sci. Rev., 2023, 10, nwad207 CrossRef CAS PubMed.
- M. M. Harussani, S. M. Sapuan, U. Rashid, A. Khalina and R. A. Ilyas, Sci. Total Environ., 2022, 803, 149911 CrossRef CAS PubMed.
- M. J. B. Kabeyi and O. A. Olanrewaju, J. Energy, 2023, 2023, 1–25 Search PubMed.
- M. Laghezza, S. Fiore and F. Berruti, J. Anal. Appl. Pyrolysis, 2024, 179, 106479 CrossRef CAS.
- Y. Cui, Y. Zhang, L. Cui, Y. Liu, B. Li and W. Liu, J. Cleaner Prod., 2023, 411, 137303 CrossRef CAS.
- A. Mohan, S. Dutta, S. Balusamy and V. Madav, RSC Adv., 2021, 11, 9807–9826 RSC.
- P. Gijsman and R. Fiorio, Polym. Degrad. Stab., 2023, 208, 110260 CrossRef CAS.
- M. S. Klippel and M. F. Martins, Polym. Degrad. Stab., 2022, 204, 110090 CrossRef CAS.
- M. I. Jahirul, F. Faisal, M. G. Rasul, D. Schaller, M. M. K. Khan and R. B. Dexter, Energy Rep., 2022, 8, 730–735 CrossRef.
- P. Palmay, C. Medina, C. Donoso, D. Barzallo and J. C. Bruno, Clean Technol. Environ. Policy, 2023, 25, 1539–1549 CrossRef CAS.
- H. Wang, M. van Akker, J. G. M. Winkelman, A. Heeres and H. J. Heeres, Energy Fuels, 2025, 39, 3564–3574 CrossRef CAS PubMed.
- L. Briones, A. Cordero, M. Alonso-Doncel, D. P. Serrano and J. M. Escola, Appl. Catal., B, 2024, 341, 123359 CrossRef CAS.
- M. Sogancioglu Kalem, Sep. Purif. Technol., 2022, 300, 121859 CrossRef CAS.
- W. Zeb, T. De Somer, M. Roosen, P. Knockaert, M. S. Abbas-Abadi, U. Kresovic, J. Hogie, K. M. Van Geem and S. De Meester, Fuel, 2025, 379, 133055 CrossRef CAS.
- T. G. Kailas, A. A R, S. Dutta and V. Madav, Energy Convers. Manage.:X, 2025, 25, 100824 CAS.
- A. Mohan, S. Dutta and V. Madav, Fuel, 2019, 250, 339–351 CrossRef CAS.
- G. Wei, Z. Chen, X. Tai, Z. Sun and X. Wang, Trans. Tianjin Univ., 2025, 31, 390–402 CrossRef CAS.
- C. Allegretti, A. Bono, P. D'Arrigo, F. G. Gatti, S. Marzorati, L. A. M. Rossato, S. Serra, A. Strini and D. Tessaro, ChemistrySelect, 2021, 6, 9157–9163 CrossRef CAS.
- A. Demydova and T. Berezka, Innovative Biosyst. Bioeng., 2021, 5, 105–116 CrossRef CAS.
- M. E. Myers, J. Stollsteimer and A. M. Wims, Anal. Chem., 1975, 47, 2010–2015 CrossRef CAS.
- P. Das and P. Tiwari, Resour., Conserv. Recycl., 2018, 128, 69–77 CrossRef.
- A. K. Yadav, S. K. Yadav, G. N. Kumar, V. Madav and S. Dutta, RSC Adv., 2025, 15, 27933–27940 RSC.
- J. Manu, T. G. Kailas and V. Madav, J. Cleaner Prod., 2023, 419, 137991 CrossRef CAS.
Footnote |
| † Equal contribution. |
| This journal is © The Royal Society of Chemistry 2026 |












