Open Access Article
Open Access ArticleRongalite-mediated sequential homologative fluorination of oxindoles en route to 3-(fluoromethyl)-3-methylindolin-2-ones
Nagaraju Naddi ,
Hari Prasad Kokatla
,
Hari Prasad Kokatla *,
Neetika Singh
*,
Neetika Singh and
Rambabu Kandukuri
and
Rambabu Kandukuri
Department of Chemistry, National Institute of Technology Warangal, Warangal, Telangana-506004, India. E-mail: harikokatla@nitw.ac.in
First published on 20th January 2026
Abstract
A rongalite-mediated, transition-metal-free reductive homologative-fluorination strategy has been developed for the synthesis of 3-(fluoromethyl)-3-methylindolin-2-ones. This methodology involves the monofluoromethylation of indolin-2-one derivatives, employing rongalite as a C1-homologating reagent, and diethylaminosulfur trifluoride (DAST) as the fluorine source. The reaction proceeds under mild conditions, offering excellent yields, high selectivity, and broad functional group tolerance on oxindoles. Overall, this approach provides a practical and sustainable route for CH2F incorporation, thereby expanding the scope of fluoroalkylation chemistry and opening new avenues for the synthesis of fluorinated bioactive molecules.
Introduction
Fluorinated organic compounds have increasingly permeated diverse scientific disciplines, especially pharmaceuticals, agrochemicals, and materials science.1 Significantly, approximately 20% of marketed pharmaceuticals are fluorine-bearing, and in the agrochemical domain, the contribution of organofluorine compounds has exceeded 50% over recent decades.2 The distinctive physicochemical characteristics of fluorine-particularly its high electronegativity and the robustness of the carbon–fluorine (C–F) bond exert a dominant influence on the behaviour of fluorine-bearing molecules.3 These traits are strategically exploited in the design of molecular scaffolds of value, thereby attracting broad interest across pharmacology,4 materials development,5 and agricultural chemistry6 due to their improved physicochemical properties relative to non-fluorinated analogues.Incorporation of a fluorine atom often enhances the biological activity,7 lipophilicity,8 and binding selectivity9 of lead molecules (Fig. 1). From a synthetic chemistry perspective, the construction of fluorinated molecular frameworks remains a significant challenge,10 highlighting the importance of organofluorine chemistry as a vital, and interdisciplinary area of research.
The development of novel methods for incorporating fluorine atoms has attracted extensive interest. In particular, difluoromethyl- and trifluoromethylarenes have been widely explored by synthetic chemists,11 resulting in a multitude of available synthetic approaches.
Within this context, monofluoromethyl (–CH2F) is of particular value by mimicking methyl (CH3) or hydroxymethyl (CH2OH) groups, which are frequently present in biologically active compounds.12 These units enable retention of structural features while imparting the “fluorine effect”. Their application spans pharmaceuticals,13 agrochemicals,14 and material science,15 where they can contribute to enhanced molecular stability, bioactivity, and selectivity.
In contrast, despite the proven significance of the monofluoromethyl (CH2F) group, synthetic methodologies for its incorporation remain limited. Most reported approaches rely on: (i) transition-metal-catalyzed reactions employing fluoro(halo)methanes (e.g., ICH2F or BrCH2F) as monofluoromethyl sources (Scheme 1a);16 (ii) carbenoid-mediated nucleophilic fluoromethylation under strongly basic conditions (Scheme 1b);17 and, (iii) fluorobis(phenylsulfonyl)methane (FBSM)-based monofluoromethylation strategies (Scheme 1c).18
Most established monofluoromethylation protocols utilize reagents such as fluoromethyllithium (LiCH2F) or fluoroiodomethyllithium (LiCHIF), typically in the presence of metals or strong bases.
These transformations generally require prior preparation of fluoromethyl precursors, and involve multi-step homologation sequences. Although effective, they are often limited by harsh conditions, costly reagents, and operational complexity. Notably, a one-pot C1 homologation/fluorination protocol for the direct introduction of a monofluoromethyl group has not yet been reported.
On the other hand, oxindole skeleton represents a privileged heterocyclic framework that is widely distributed in natural products, pharmaceuticals, and bioactive compounds, attracting sustained interest in both synthetic, and medicinal chemistry.19 Among these, 3,3-disubstituted oxindoles are of particular significance owing to their rigid structures, extended π-systems, and nitrogen-rich environments.20 These features render them suitable pharmacophores exhibiting diverse biological activities such as spermicidal,21 antimicrobial,22 enzyme inhibitory,23 and anticancer properties.24 Further, 3-hydroxymethyl-3-methyl-2-oxindoles serve as key intermediates in the total synthesis of (–)-physostigmine and (–)-esermethole,25 and their biological activity can be enhanced by fluorine incorporation.
Continuing our studies on the synthetic versatility of rongalite,26 a multifaceted reagent functioning as a reducing agent,27 a single-electron donor,28 and a dual C1-unit donor,29 we now report a straightforward rongalite-mediated monofluoromethylation of indolin-2-one. In this transformation, rongalite serves as the C1-unit donor and, in the presence of an external fluorine source, provides an efficient route to fluorinated oxindole derivatives (Scheme 1d).
Results and discussion
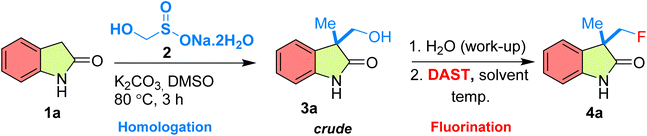
In our preliminary investigations, we aimed to develop a streamlined and efficient monofluoromethylation strategy for the synthesis of 3-(fluoromethyl)-3-methylindolin-2-one 4a from indolin-2-one 1a, Rongalite 2, and DAST as model substrates. The study commenced with the C1-homologation of 1a (1.0 mmol) using Rongalite 2 (3.0 mmol), and K2CO3 (2.5 mmol) in DMSO (2 mL) at 80 °C, affording the hydroxymethyl crude 3a. After completing the C1 homologation, the reaction mixture 3a was washed with water and extracted with Et2O, evaporated and the crude 3a was directly subjected to fluorination in CH2Cl2 without purification, providing the desired product 4a in 22% overall yield (Table 1, entry 1). Encouraged by this initial result, we proceeded to optimize the DAST stoichiometry and reaction temperature. Lowering the temperature to 0 °C improved the yield to 30% after 45 minutes (Table 1, entry 2). Further increasing the amount of DAST from 1.0 mmol to 2.0 mmol enhanced the yield to 46% in 30 minutes (Table 1, entry 3), while 3.0 mmol of DAST afforded a significantly higher yield of 60% within 20 minutes (Table 1, entry 4). Remarkably, using 4.0 mmol of DAST under the same conditions delivered 4a in 85% yield within only 5 minutes (Table 1, entry 5), demonstrating that an excess of DAST effectively drives the fluorination to completion. To further understand the effect of temperature, reactions using 4.0 mmol DAST were carried out under varied conditions. At room temperature, 4a was obtained in 60% yield in 15 minutes (Table 1 entry 6), whereas at −10 °C, the yield slightly decreased to 80% in 5 minutes (Table 1, entry 7). These observations revealed that 0 °C offered the optimal balance between reactivity and selectivity, providing the highest yield in the shortest time. Next, the solvent effect was examined (Table 1, entries 8–11). When CH2Cl2 was replaced by THF, CHCl3, CH3CN, and 1,2-dichloroethane (DCE), the yields decreased to 65%, 55%, 48%, and 64%, respectively. Notably, performing the fluorination in DMSO resulted in no product formation, even after 24 hours (Table 1, entry 12). It is worth mentioning that the other fluorinating agents such as PyFluor and AlkylFluor are failed to give desired product 4a (see SI, Table S1). In summary, the optimization studies established that treating 3a (1.0 mmol) with DAST (4.0 mmol) in CH2Cl2 at 0 °C constitutes the most effective set of conditions, affording 3-(fluoromethyl)-3-methylindolin-2-one 4a in 85% yield within 5 minutes (Table 1, entry 5). These mild, and efficient reaction parameters were subsequently adopted for the substrate-scope investigations.| Entry | DAST (mmol) | Solvent | Temp. (°C) | Time (min) | Yieldb (%) |
|---|---|---|---|---|---|
| a Reaction conditions: indolin-2-one 1a (1 mmol), rongalite 2 (3 mmol) and K2CO3 (2.5 mmol) in 2 mL of DMSO at 80 °C, after completing the C1 homologation, the reaction mixture 3a was washed with water and extracted with Et2O, evaporated, re-dissolved in CH2Cl2 (2 mL), and added DAST.b Isolated yield from homologation-fluorination sequence.c n.r. = no reaction, rt = room temperature. | |||||
| 1 | 1.0 | CH2Cl2 | rt | 1 h | 22 |
| 2 | 1.0 | CH2Cl2 | 0 | 45 | 30 |
| 3 | 2.0 | CH2Cl2 | 0 | 30 | 46 |
| 4 | 3.0 | CH2Cl2 | 0 | 20 | 60 |
| 5 | 4.0 | CH2Cl2 | 0 | 5 | 85 |
| 6 | 4.0 | CH2Cl2 | rt | 15 | 60 |
| 7 | 4.0 | CH2Cl2 | −10 | 5 | 80 |
| 8 | 4.0 | THF | 0 | 30 | 65 |
| 9 | 4.0 | CHCl3 | 0 | 20 | 55 |
| 10 | 4.0 | CH3CN | 0 | 30 | 48 |
| 11 | 4.0 | C2H4Cl2 | 0 | 15 | 64 |
| 12 | 4.0 | DMSO | 0 | 24 h | n.rc |
The fluorination behaviour of various 3-(hydroxymethyl)-3-methylindolin-2-one derivatives using DAST was systematically investigated (Table 2). The method displayed broad substrate tolerance: electron-donating substituents (methyl, dimethyl, methoxy) on the aromatic ring afforded fluoromethyl derivatives 4b–4d in good yields (72–75%). These substituents likely enhance electron density on the indolinone ring, stabilizing the transition state in fluorination. Similarly, substrates bearing electron-withdrawing halogens (F, Cl, Br, I) at various positions tolerated the conditions well, giving products 4e–4l in yields of 75–84%. Moreover, an –OCF3 substituent was well accommodated, affording 4m in 80% yield. These results demonstrate a versatile and efficient fluorination protocol for the preparation of fluoromethyl-substituted indolin-2-ones under mild conditions. Notably, N-alkylated, and N-aryl-substituted derivatives demonstrated remarkably enhanced reactivity compared to their N–H counterparts. Substrates with N-methyl, N-ethyl, N-butyl, and N-4-ethylphenyl groups afforded products 4n–4q in excellent yields 91–93% (Table 2) within a very short reaction time. Likewise, N-substituted cyclopentyl, allyl, propargyl, and benzyl afforded the desired fluorinated products 4r–4u in good to excellent yields 76–89% (Table 2).
| a Reaction conditions: indolin-2-one 1 (1 mmol), rongalite 2 (3 mmol) and K2CO3 (2.5 mmol) in 2 mL of DMSO at 80 °C, after completing the C1 homologation, the reaction mixture 3a was washed with water, and extracted with Et2O, evaporated, re-dissolved in CH2Cl2 (2 mL), and added DAST (4 mmol).b Isolated yield from homologation-fluorination sequence. |
|---|
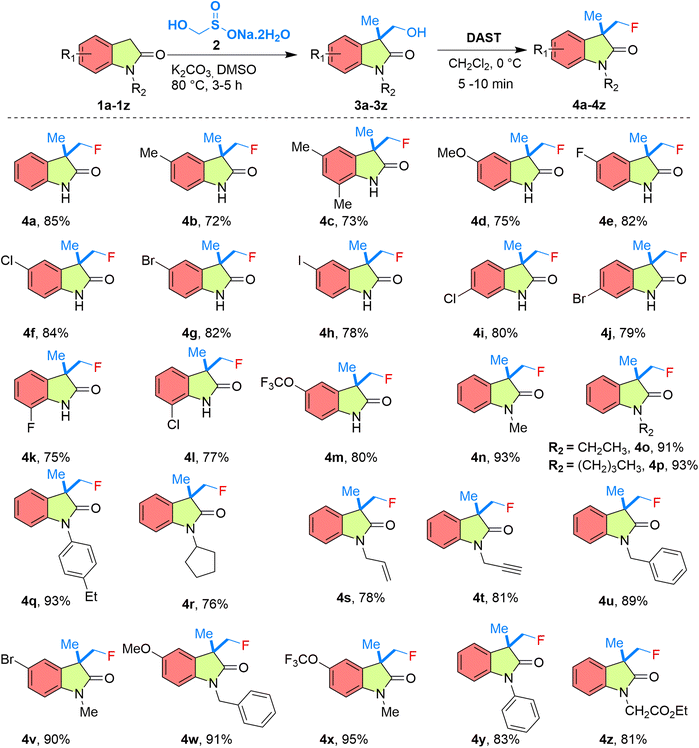 |
Further, the substitutions on the benzene ring, and the nitrogen atom enhanced the formation of products 4v–4x in excellent yields 90–95% (Table 2), confirming the high efficiency of the reaction. Further, N-phenyl 3-(hydroxymethyl)-3-methylindolin-2-one also actively participated in the reaction giving the product 4y in excellent yield 83% (Table 2). The N-substituted ethyl acetate also participated in the reaction forming the product 4z in good yield 81% (Table 2). Overall, both electron-donating, and electron-withdrawing groups on the indolinone ring were well tolerated, but N-alkylation, and N-arylation significantly improved the reaction efficiency, leading to excellent yields within a short time frame.
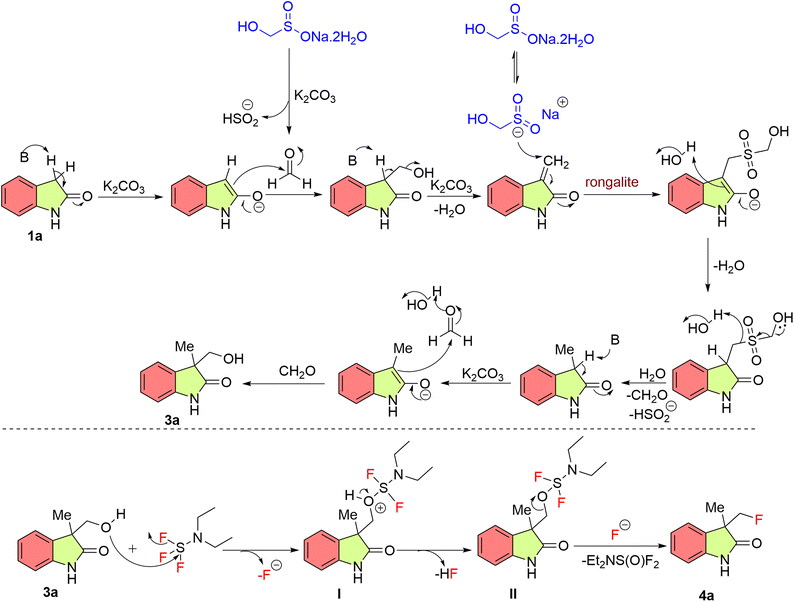
Based on the literature reports27,30 a plausible mechanism is outlined in Scheme 2. Initially, indolin-2-one 1a reacts with in situ generated formaldehyde from rongalite 2 to form 3-(hydroxymethyl)-3-methylindolin-2-one 3a via aldol condensation reaction followed by a reductive aldol reaction.27 Which further undergoes nucleophilic attack of oxygen atom from alcohol 3a to the electrophilic sulfur atom of diethylaminosulfur trifluoride would form an alkoxy-sulfur intermediate I, subsequent deprotonation facilitates the generation of a neutral intermediate II, which then undergoes an intramolecular displacement by the liberated fluoride ion on intermediate, yielding 3-(fluoromethyl)-3-methylindolin-2-one 4a with loss of the sulfur-containing leaving group.
Conclusions
We have developed an efficient monofluoromethylation by sequential hydroxymethylation, and monofluorination strategy, which involves a classical aldol condensation reaction followed by a reductive aldol reaction using rongalite. In this method, rongalite is an industrial product with low cost (1 g, 0.03$), which plays a vital role as a hydride-free reducing agent, and double C1 unit donor. This transition metal, and hydride-free reductive aldol protocol allows rapid access to 3-(hydroxymethyl)-3-methylindolin-2-ones, and DAST (diethylaminosulfur trifluoride) is commercially available, easy to handle, and highly selective for hydroxyl groups, making it ideal for deoxyfluorination of alcohols. It can also work under mild conditions, preserving sensitive functional groups in the molecule. 3-(fluoromethyl)-3-methylindolin-2-one is an organofluorine compound that belongs to the class of fluorinated indolinones, a group of molecules with significant importance in medicinal, and pharmaceutical chemistry.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental procedures, characterization data, and copies of the 1H, 13C{1H}, 19F NMR and HRMS spectra of all compounds are included. See DOI: https://doi.org/10.1039/d5ra08987e.Acknowledgements
The author H.P.K. is grateful to the Anusandhan National Research Foundation (SCP/2022/000273 & EEQ/2022/000511) and to the DST-FIST grant (SR/FST/CSII/2018/65) awarded to the Department of Chemistry, NIT Warangal, for financial support of this work. The authors also thank the CRIF Centre, NIT Warangal, for NMR and HRMS analysis and NIT Warangal for providing infrastructure support.Notes and references
- (a) A. M. Thayer, Chem. Eng. News, 2006, 84, 15–24 Search PubMed; (b) R. Britton, V. Gouverneur, J.-H. Lin, M. Meanwell, C. Ni, G. Pupo, J.-C. Xiao and J. Hu, Nat. Rev. Methods Primers, 2021, 1, 47 CrossRef CAS.
- D. A. M. Alexandrino, C. M. R. Almeida, A. P. Mucha and M. F. Carvalho, Environ. Pollut., 2022, 292, 118315 CrossRef CAS PubMed.
- (a) K. Shinoda, M. Hato and T. Hayashi, J. Phys. Chem., 1972, 76, 909–914 CrossRef CAS; (b) M. A. Kaiser, D. P. Cobranchi, C.-P. C. Kao, P. J. Krusic, A. A. Marchione and R. C. Buck, J. Chem. Eng. Data, 2004, 49, 912–916 CrossRef CAS; (c) T. Hasegawa, Chem. Rec., 2017, 17, 903–917 CrossRef CAS PubMed; (d) C. M. Burba, K. Feightner, M. Liu and A. Hawari, ChemPhysChem, 2022, 23, e202100548 CrossRef CAS PubMed.
- G. Shabir, A. Saeed, W. Zahid, F. Naseer, Z. Riaz, N. Khalil, Muneeba and F. Albericio, Pharmaceuticals, 2023, 16, 1162 CrossRef CAS PubMed.
- (a) M. J. A. Dinglasan Panlilio and S. A. Mabury, Environ. Sci. Technol., 2006, 40, 1447–1453 CrossRef CAS; (b) S. Guzman Puyol, npj Sci. Food, 2024, 8, 82 CrossRef PubMed; (c) G. Chandra, B. Mahto, V. R. Singh, G. K. Mahato and U. Rani, J. Photochem. Photobiol. C: Photochem. Rev., 2024, 60–61, 100677 CrossRef CAS; (d) F. Babudri, G. M. Farinola, F. Naso and R. Ragni, Chem. Commun., 2007, 1003–1022 RSC.
- (a) C. Gan, Z. Gan, S. Cui, R. Fan, Y. Fu, M. Peng and J. Yang, Sci. Total Environ., 2021, 771, 144809 CrossRef CAS PubMed; (b) R. Jagani, H. Patel, J. Chovatiya and S. S. Andra, J. Agric. Food Chem., 2025, 73, 2217–2220 CrossRef CAS PubMed; (c) P. Jeschke, Pest Manag. Sci., 2024, 80, 3065–3087 CrossRef CAS PubMed.
- (a) T. A. Unzner and T. Magauer, Tetrahedron Lett., 2015, 56, 877–883 CrossRef CAS; (b) K. Fan, X. Chen, X. Wang, X. Liu, Y. Liu, W. Lai and X. Liu, ACS Appl. Mater. Interfaces, 2018, 10, 28828–28838 CrossRef CAS PubMed.
- (a) Z. Wang, H. R. Felstead, R. I. Troup, B. Linclau and P. T. F. Williamson, Angew. Chem., Int. Ed., 2023, 62, e202301077 CrossRef CAS PubMed; (b) B. Jeffries, Z. Wang, H. R. Felstead, J.-Y. Le Questel, J. S. Scott, E. Chiarparin, J. Graton and B. Linclau, J. Med. Chem., 2020, 63, 1002–1031 CrossRef CAS PubMed; (c) K. P. Melnykov, O. S. Liashuk, O. Smyrnov, D. Lesyk, Y. Holota, P. Borysko, V. Yakubovskyi and O. O. Grygorenko, J. Fluorine Chem., 2025, 281, 110384 CrossRef CAS; (d) I. Columbus, L. Ghindes-Azaria, I. M. Herzog, E. Blum, G. Parvari, Y. Eichen, Y. Cohen, E. Gershonov, E. Drug, S. Saphier, S. Elias, B. Smolkin and Y. Zafrani, Commun. Chem., 2023, 6, 197 CrossRef CAS PubMed; (e) T. D. Le, R. A. Arlauskas and J. G. Weers, J. Fluorine Chem., 1996, 78, 155–163 CrossRef CAS.
- (a) T. Fuchigami and S. Inagi, Acc. Chem. Res., 2020, 53, 322–334 CrossRef CAS PubMed; (b) S. Rozen, Chem. Rev., 1996, 96, 1717–1736 CrossRef CAS PubMed; (c) T. Fuchigami, J. Fluorine Chem., 2007, 128, 311–316 CrossRef CAS.
- (a) A. Joseph, R. Rajan, J. Paul, K. E. Cherian, N. Kapoor, F. Jebasingh, H. S. Asha, N. Thomas and T. V. Paul, J. Clin. Transl. Endocrinol. Case Rep., 2022, 24, 100114 Search PubMed; (b) J. J. Petkowski, S. Seager and W. Bains, Sci. Rep., 2024, 14, 15575 CrossRef CAS PubMed; (c) Y. Pan, ACS Med. Chem. Lett., 2019, 10, 1016–1019 CrossRef CAS PubMed.
- (a) H. Sun, Y. Jiang, Y.-S. Yang, Y.-Y. Li, L. Li, W.-X. Wang, T. Feng, Z. Li and J.-K. Liu, Org. Biomol. Chem., 2019, 17, 6629–6638 RSC; (b) Y. Guo, R. Wang, H. Song, Y. Liu and Q. Wang, Chem. Commun., 2021, 57, 8284–8287 RSC.
- (a) B.-J. Li, Y.-L. Ruan, L. Zhu, J. Zhou and J.-S. Yu, Chem. Commun., 2024, 60, 12302–12314 RSC; (b) J. B. I. Sap, C. F. Meyer, N. J. W. Straathof, N. Iwumene, C. W. am Ende, A. A. Trabanco and V. Gouverneur, Chem. Soc. Rev., 2021, 50, 8214–8247 RSC; (c) M. Y. Moskalik, Int. J. Mol. Sci., 2023, 24, 17593 CrossRef CAS PubMed; (d) A. Knieb, V. Krishnamurti, Z. Zhu and G. K. Surya Prakash, J. Fluorine Chem., 2023, 267, 110095 CrossRef CAS.
- (a) A. Citarella and N. Micale, Molecules, 2020, 25, 4031 CrossRef CAS PubMed; (b) C. M. Lloyd, N. Colgin and S. L. Cobb, Molecules, 2020, 25, 5601 CrossRef CAS PubMed; (c) Z. Wang, Y.-F. Zeng, C. Zeng and Y. Wang, Asian J. Org. Chem., 2025, 14, 257–276 Search PubMed; (d) B. M. Johnson, Y.-Z. Shu, X. Zhuo and N. A. Meanwell, J. Med. Chem., 2020, 63, 6315–6386 CrossRef CAS PubMed.
- T. Fujiwara and D. O'Hagan, J. Fluorine Chem., 2014, 167, 16–29 CrossRef CAS.
- Z.-K. Zhang, W.-Y. Xu, T.-J. Gong and Y. Fu, ChemSusChem, 2024, 17, e202301072 CrossRef CAS PubMed.
- (a) Q. Lan, X. Yu, M. Qiao and X. Zhang, Angew. Chem., Int. Ed., 2015, 54, 9079–9083 CrossRef PubMed; (b) H. Yin, J. Sheng, K.-F. Zhang, Z.-Q. Zhang, K.-J. Bian and X.-S. Wang, Chem. Commun., 2019, 55, 7635–7638 RSC; (c) J. Hu, B. Gao, L. Li, C. Ni and J. Hu, Org. Lett., 2015, 17, 3086–3089 CrossRef CAS PubMed.
- G. Parisi, M. Colella, S. Monticelli, G. Romanazzi, W. Holzer, T. Langer, L. Degennaro, V. Pace and R. Luisi, J. Am. Chem. Soc., 2017, 139, 13648–13651 CrossRef CAS PubMed.
- A. Knieb, V. Krishnamurti, Z. Zhu and G. K. Surya Prakash, J. Fluorine Chem., 2023, 267, 110095 CrossRef CAS.
- (a) M. Kaur, M. Singh, N. Chadha and O. Silakari, Eur. J. Med. Chem., 2016, 123, 858–894 CrossRef CAS PubMed; (b) L.-M. Zhou, R.-Y. Qu and G.-F. Yang, Expert Opin. Drug Discov., 2020, 15, 603–625 CrossRef CAS PubMed; (c) B. Yu, D.-Q. Yu and H.-M. Liu, Eur. J. Med. Chem., 2015, 97, 673–698 CrossRef CAS PubMed; (d) C. V. Galliford and K. A. Scheidt, Angew. Chem., Int. Ed., 2007, 46, 8748–8758 CrossRef CAS PubMed; (e) A. Millemaggi and R. J. K. Taylor, Eur. J. Org Chem., 2010, 2010, 4527–4547 CrossRef; (f) K. Huber, J. Schemies, U. Uciechowska, J. M. Wagner, T. Rumpf, F. Lewrick, R. Süss, W. Sippl, M. Jung and F. Bracher, J. Med. Chem., 2010, 53, 1383–1396 CrossRef CAS PubMed; (g) B. Volk, J. Barkóczy, E. Hegedus, S. Udvari, I. Gacsalyi, T. Mezei, K. Pallagi, H. Kompagne, G. Levay, A. Egyed, L. G. Harsing, M. Spedding and G. Simig, J. Med. Chem., 2008, 51, 2522–2532 CrossRef CAS PubMed.
- S. Sharma, Y. Monga, A. Gupta and S. Singh, RSC Adv., 2023, 13, 14249–14267 RSC.
- P. Paira, A. Hazra, S. Kumar, R. Paira, K. B. Sahu, S. Naskar, P. Saha, S. Mondal, A. Maity, S. Banerjee and N. B. Mondal, Bioorg. Med. Chem. Lett., 2009, 19, 4786–4789 CrossRef CAS PubMed.
- N. G. Kandile, H. T. Zaky, M. I. Mohamed, H. M. Ismaeel, N. A. Ahmed and J. Enzyme, Inhib. Med. Chem., 2012, 27, 599–608 CrossRef CAS PubMed.
- P. Czeleń, M. Wierzchowski, R. Kluge, E. Kedzierska and J. Płonka, Biology, 2021, 10, 332 CrossRef PubMed.
- M. S. Christodoulou, V. Kollia, G. Papageorgiou, P. Nikolaou, T. Tselios and V. Psycharis, Eur. J. Med. Chem., 2020, 192, 112168 Search PubMed.
- (a) K. Asakawa, N. Noguchi and M. Nakada, Heterocycles, 2008, 76, 183–200 CrossRef CAS PubMed; (b) K. Asakawa, N. Noguchi, S. Takashima and M. Nakada, Tetrahedron: Asymmetry, 2008, 19, 2304–2312 CrossRef CAS.
- (a) X. Zhang, Y. Li, J. Wang, Z. Chen and H. Liu, Chin. J. Chem., 2023, 41, 3388–3400 CrossRef; (b) S. Kotha and P. Khedkar, Chem. Rev., 2012, 112, 1650–1680 CrossRef CAS PubMed.
- (a) S. Golla, S. Jalagam, S. Poshala and H. P. Kokatla, Org. Biomol. Chem., 2022, 20, 4926–4932 RSC; (b) S. Golla, N. Anugu, S. Jalagam and H. P. Kokatla, Org. Biomol. Chem., 2022, 20, 808–816 RSC; (c) Y. Jain, M. Kumari, H. Laddha and R. Gupta, ChemistrySelect, 2019, 4, 7015–7026 CrossRef CAS.
- (a) S. Golla and H. P. Kokatla, J. Org. Chem., 2022, 87, 9915–9925 CrossRef CAS PubMed; (b) S. Golla, S. Poshala, R. Pawar and H. P. Kokatla, Tetrahedron Lett., 2020, 61, 151539 Search PubMed; (c) S. Poshala, S. Thunga, S. Manchala and H. P. Kokatla, ChemistrySelect, 2018, 3, 13759–13764 CrossRef CAS.
- (a) S. Thunga, M. Inapanuri, N. Singh and H. P. Kokatla, J. Org. Chem., 2024, 89, 18313–18321 CrossRef CAS PubMed; (b) S. Thunga, N. Singh, N. Anugu and H. P. Kokatla, J. Org. Chem., 2024, 89, 18649–18653 Search PubMed; (c) S. Thunga, N. Singh, M. Inapanuri and H. P. Kokatla, Org. Biomol. Chem., 2024, 22, 8787–8792 RSC.
- M. Kaźmierczak and M. Bilska-Markowska, Eur. J. Org Chem., 2021, 2021, 5585–5604 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |